- 1Department of Neurosurgery & Brain and Nerve Research Laboratory, The First Affiliated Hospital of Soochow University, Suzhou, China
- 2Department of Neurosurgery, First People’s Hospital of Kunshan, Suzhou, China
Objective: To retrospective analyze the clinical data of 162 patients with small cerebellopontine angle meningiomas. To compare with the nature of tumors, symptoms pre- and post-treatments, neurological deficit, and prognosis in literatures. To explore the surgical outcomes of small cerebellopontine angle meningiomas and summarize the surgical experience.
Methods: All of 162 patients with small cerebellopontine angle meningiomas underwent surgery between January 2010 and December 2019 in the neurosurgery department of the First Affiliated Hospital of Soochow University. This cohort of eight literatures reported about stereotactic radiotherapy of small cerebellopontine angle meningiomas between January 2010 and December 2019. All clinical data were obtained for analysis.
Results: Compared with stereotactic radiotherapy, surgical treatment for small cerebellopontine angle meningiomas lead to the better results in relieving symptoms and inhibiting tumor progression. Surgical treatment can obtain the exact pathological examination results to guide the further treatment.
Conclusions: Surgical treatment should be the first choice for small cerebellopontine angle meningiomas.
Introduction
Cerebellopontine angle meningiomas account for 6–15% of the tumors in the cerebellopontine angle region (1). They are characterized by the deep tumor location, narrow surgical field, and proximity to the brainstem, multiple pairs of (V–XI) cranial nerves (2). At present, surgical treatment is the first choice for large cerebellopontine angle meningiomas, while small cerebellopontine angle meningiomas are always treated by stereotactic radiotherapy, pharmacotherapy and experimental therapy (3). In recent years, many studies have found that stereotactic radiotherapy had the limitations of low tumor control rate, post-treatment brain edema, and tissue adhesion, which hindered the further treatments (4). Therefore, more and more small cerebellopontine angle meningioma patients are turning to surgical treatment.
Materials and Methods
Study Design
A retrospective analysis of small cerebellopontine angle meningioma patients was performed. These patients were operated on in the neurosurgery department of the First Affiliated Hospital of Soochow University between January 2010 and December 2019. These patients were classified as the surgery group. We also reviewed the literature on stereotactic radiotherapy of small cerebellopontine angle meningiomas from the past 10 years. The clinical data of patients in the literature were collected and analyzed, and these patients were classified as the radiotherapy group. We analyzed the differences between the two groups, including the nature of tumors, symptoms pre- and post-treatments, neurological deficits, and prognosis. Finally, we explored the surgical outcomes of small cerebellopontine angle meningiomas and summarized the surgical experience.
Inclusion Criteria
Diagnosis
To distinguish meningiomas from other cerebellopontine angle tumor, such as acoustic neuromas and gliomas, all the patients were diagnosed by both radiological and histopathological examination, including MRI, CT, PET, and SSTR2 ligands. Both the dural tail sign on MRI and no expansion of internal auditory canal on CT are the main differential points between cerebellopontine angle meningiomas and acoustic neuromas. To differentiate cerebellopontine angle meningiomas from gliomas and metastases, patients were conventionally tested by MR spectroscopy (5). In terms of histological aspects, immunohistochemical analysis was also conventionally tested, such as HE staining, Vimentin staining, EMA staining, Ki-67 and CD56.
Surgery Group
(1) The patients with small cerebellopontine angle meningioma were operated between January 2010 and December 2019, (2) tumor volume ≤ 8 cm3, (3) no related treatment before surgery, (4) no other nervous system diseases, and (5) kept in touch during follow-up.
Radiotherapy Group
(1) The patients with small cerebellopontine angle meningioma were collected from the literature about stereotactic radiotherapy, which published between January 2010 and December 2019, (2) the included patients had complete pre- and post-treatment data, (3) no other nervous system diseases, and (4) kept in touch during follow-up.
Follow-up consisted of routinely visiting the patients and performing MRI or CT tests every 3–6 months for the first 3 years after treatment, and then visiting and testing every year.
Classification of Tumors
Based on the central site of dural attachment, cerebellopontine angle meningiomas were classified into three types (6): anterior tumors were those that originated from the tentorium cerebelli or the petrous bone dura anterior the internal auditory canal; middle tumors were those that originated from the dura mater in the internal auditory canal; posterior tumors were those that originated from the sigmoid and transverse sinuses or the petrous bone dura posterior the internal auditory canal (Figure 1).
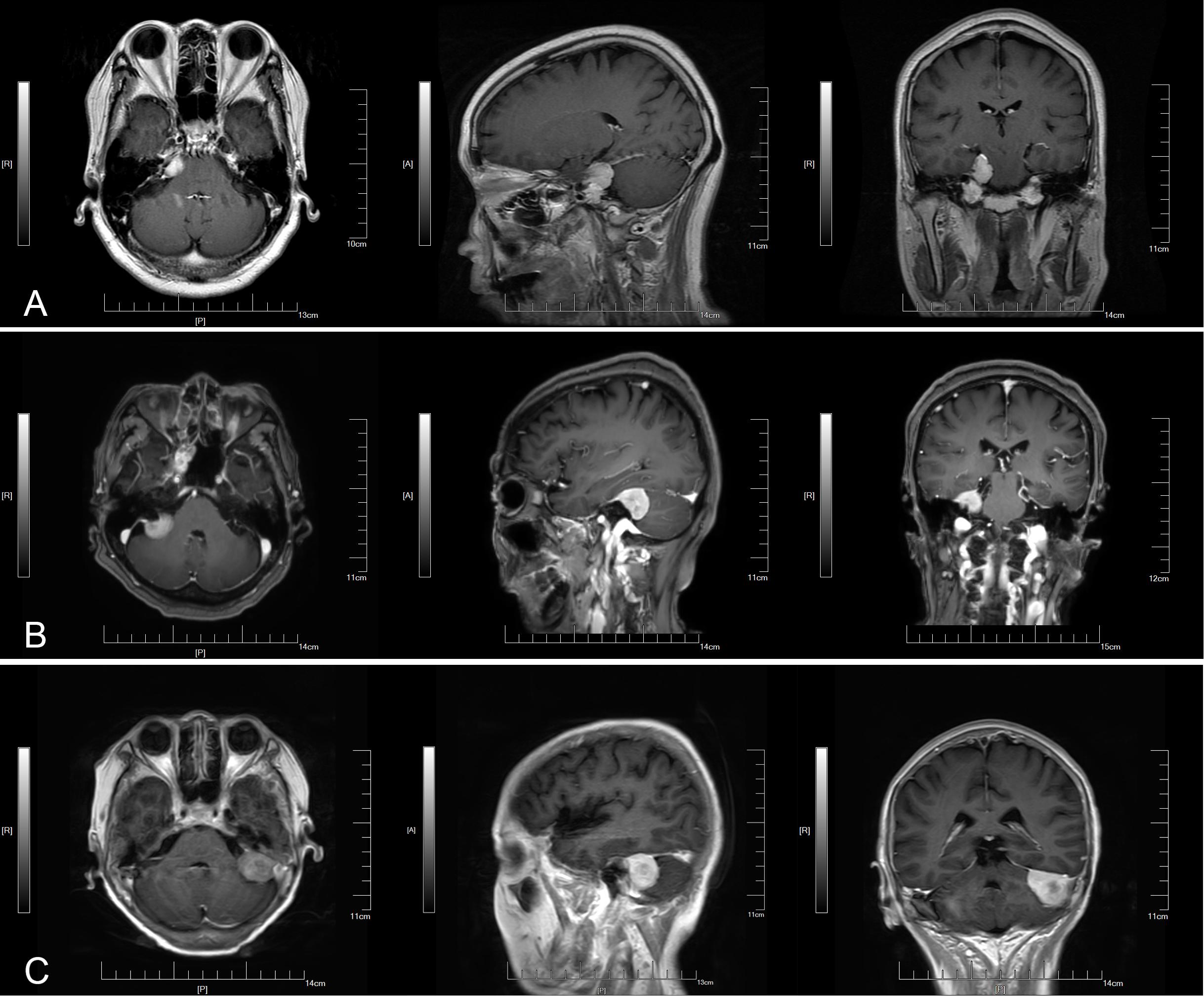
Figure 1. Three types of small cerebellopontine angle meningiomas. (A) Anterior. (B) Middle. (C) Posterior.
Based on tumor pathology, cerebellopontine angle meningiomas were classified into three grades: WHO grade I (low recurrence and low invasive growth), WHO grade II (high recurrence and high invasive growth), and WHO grade III (strong recurrence and metastasize systemically).
Based on the grade of tumor resection, patients with cerebellopontine angle meningiomas were divided into five grades: Simpson grade I: total resection of the meningioma, dural attachment, and skull; Simpson grade II: total resection of the meningioma and electrocoagulation or laser treatment with dural attachment; Simpson grade III: total resection of meningiomas and no treatment with the dural attachment and skull; Simpson grade IV: partial resection of meningiomas; Simpson grade V: decompression and tumor biopsy.
Tumor progression and regression were defined as volume changes of more than 15% on radiological examination.
Surgical Program
All of the patients were using the suboccipital retrosigmoid approach. A suboccipital retrosigmoid straight incision of about 8–10 cm was made on the affected side. The long diameter of the oval bone window was 5 cm, and the short diameter was 3.5 cm. The bone window up to the transverse sinus, lateral to the mastoid root, exposed the angle between the sigmoid and transverse sinus. Cut the dura, stretched the cerebellum, opened up the cisterna magna, and released the cerebrospinal fluid. Finally, the cerebellopontine angle meningioma was completely resected, taking care to protect the petrous vein, the trigeminal nerve, and the abducens nerve. During the operation, electrophysiological monitoring was used to monitor the facial and acoustic nerves.
Statistical Analysis
All of the statistical analyses were performed with SPSS version 22.0. The parametric continuous variables were reported as mean ± standard deviation. The non-parametric variables were reported as the median with the range. Clinical outcomes and signs and symptoms were reported as three-category data (improvement or enlargement, no change, deterioration or diminution). The independent samples t-test was performed for two categories of data, and ANOVA was performed for three-category data. The chi-square test was performed to compare nominally distributed categorical variables. Logistic regression analysis was performed for multivariate analyses. P-values less than 0.05 were considered to be statistically significant.
Results
Surgery Group
Participants
A total of 162 patients with small cerebellopontine angle meningioma were included, including 53 males and 109 females, with an average age of 54.85 years (21–89 years). The preoperative symptoms were headache and dizziness (96), hearing loss and tinnitus (48), facial sensation and paralysis (29), walking instability and ataxia (28), hoarseness, poor cough reflex (16). There were 77 patients with two or more symptoms and 64 patients without symptoms before diagnosis.
The Nature of the Tumors
The volume of tumors ranged from 1.042 to 8.161 cm3, with an average of 4.710 cm3; 72 patients had anterior tumors, 41 patients had middle tumors, and 49 patients had posterior tumors; 148 patients had WHO grade I tumors, and 14 patients had WHO grade II tumors.
Surgical Outcomes
There were 152 cases of Simpson grade I and 10 cases of Simpson grade II, 41 cases of post-operative symptom relief, 110 cases of no significant change, 11 cases of aggravation or new symptoms.
Radiotherapy Group
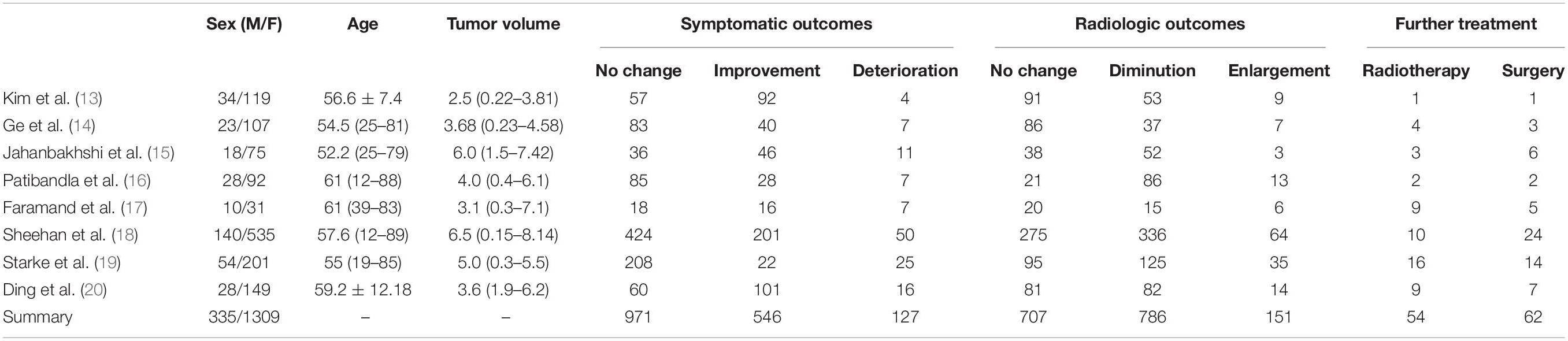
A total of 1644 patients with small cerebellopontine angle meningioma were included, including 335 males and 1309 females (Table 1).
Of the radiotherapy group, 971 patients (59.1%) had no significant change in symptoms after treatment, 546 patients (33.2%) showed an improvement in symptoms, and 127 patients (7.7%) showed a worsening of symptoms or developed new neurological symptoms.
There were 151 patients (9.2%) in the radiotherapy group with tumor recurrence during follow-up.
There were 54 patients (3.3%) received second stereotactic radiotherapy, and 62 patients (3.8%) underwent surgery during follow-up.
Symptomatic Outcomes
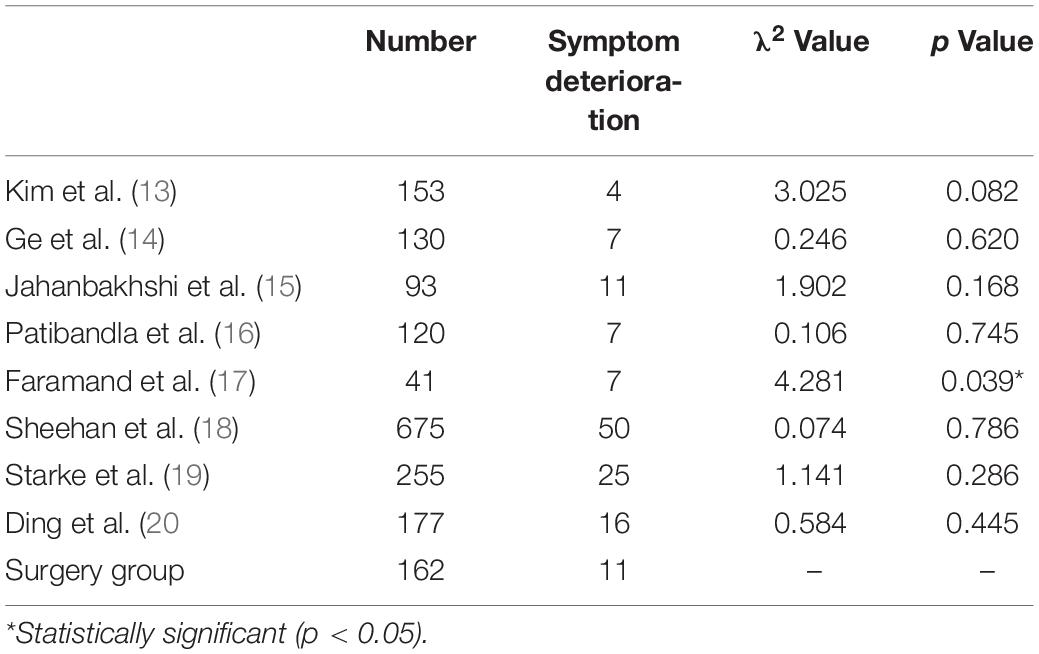
Of the surgery group, 102 patients (63.0%) had no significant change in symptoms after surgery, 49 patients (30.2%) showed an improvement in symptoms, and 11 patients (6.8%) showed a worsening of symptoms or developed new neurological symptoms. There was no significant difference in the symptom deterioration rate between the surgery group and the radiotherapy group, with the exception of Andrew et al.’s study (Table 2). The symptom deterioration rate of Andrew et al. was significantly higher than that of the surgery group.
Radiologic Outcomes
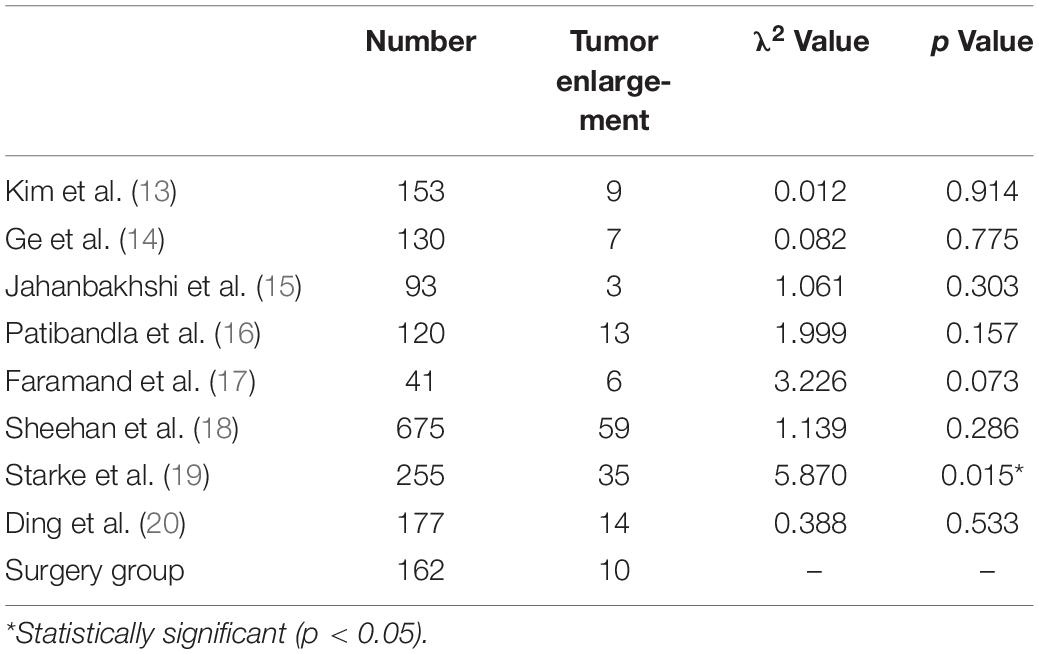
There were 10 patients (6.2%) in the surgery group with tumor recurrence during follow-up. We found no significant difference in tumor enlargement rate between the surgery group and the radiotherapy group, with the exception of Robert et al. (Table 3). The tumor enlargement rate of Robert et al. was significantly higher than that of the surgery group.
Further Treatment
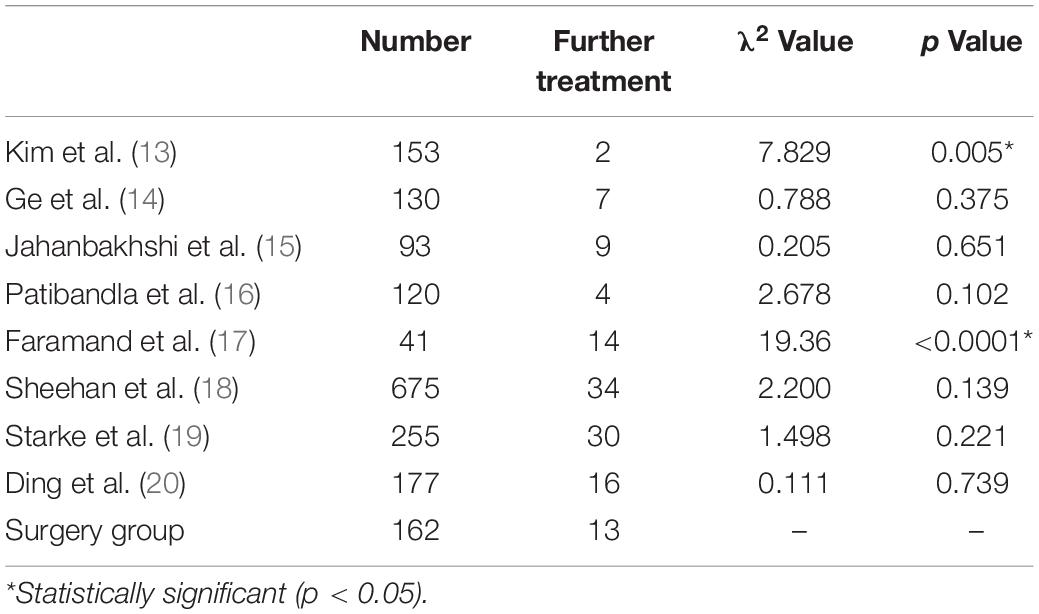
In the surgery group, there was one patient (0.6%) who underwent a second resection, and 12 patients (7.4%) received stereotactic radiotherapy during follow-up. We found no significant difference in the further treatment rate between the surgery group and the radiotherapy group, except for Kyung et al. and Andrew et al. (Table 4). The further treatment rate of Kyung et al. was significantly lower than that of the surgery group, while the further treatment rate of Andrew et al. was significantly higher than that of the surgery group.
In the surgery group, WHO grade II and Simpson grade II were risk factors of further treatment (Table 5).
Discussion
The choice of surgery or stereotactic radiotherapy depends on the general situation of patients and the nature of tumors (7). Most of cerebellopontine angle meningiomas belong to benign tumors. The surgical effect of cerebellopontine angle meningiomas is always satisfied, and the rates of both post-operative symptom deterioration and tumor enlargement are lower than other nervous system tumors. However, the compression of the brain stem and cerebellum is a frequent occurrence of cerebellopontine angle meningiomas, especially in patients with large cerebellopontine angle meningiomas, the high intracranial pressure can lead to herniation and acute hydrocephalus. Either complete or partial resection can significantly reduce the risk of complications. Hence, surgery is the best choice for patients with large cerebellopontine angle meningiomas. For small cerebellopontine angle meningiomas, stereotactic radiotherapy, including Gamma Knife, cyber knife and other types of linear accelerator (8), is universally acknowledged as the first choice. With the development of medical treatment and the popularization of MRI, the early diagnosis of cerebellopontine angle meningioma in the small-volume or asymptomatic stage turns to possible. Early diagnosis and treatment greatly improve the prognosis of cerebellopontine angle meningioma, as well as bringing a confusion of choice between surgery and stereotactic radiotherapy for small cerebellopontine angle meningioma. In consideration of the edema and adhesion of brain tissue after stereotactic radiotherapy, which hindered the further surgery, more and more studies have supported early surgical treatments.
In terms of relieving pre-operative symptoms, the symptom deterioration rate in surgery group was similar to or even lower than the rate in radiotherapy group. Compared with stereotactic radiotherapy, surgical treatment can sometimes lead to better results in relieving pre-operative symptoms. Edema of peripheral brain tissue is a common side effect of stereotactic radiotherapy. Swelling of brain tissue will aggravate the tension and compression of nerves, which is the reason why the symptoms become worse after stereotactic radiotherapy. In order to reduce nerve injury and relieve symptom deterioration, operators need to carefully protect brain tissue, nerves, and blood vessels during the surgery. Compared with regular cerebellopontine angle meningiomas, surgical operation on small cerebellopontine angle meningiomas requires more protection for nerves and blood vessels, and neuroelectrophysiological monitoring during the whole surgery process is deemed essential, which contributes to the lower symptom deterioration rate in the surgery group. The most common clinical manifestation of small cerebellopontine angle meningiomas is functional defects of the facial and auditory nerves. Hence, protecting the facial and auditory nerves is a key point of surgery. Different types of cerebellopontine angle meningiomas will push the facial and auditory nerves to different positions (9). Thus, the first step of small cerebellopontine angle meningiomas surgery is locating the facial and auditory nerves. Anterior tumors generally push the facial and auditory nerves to the lateral or lateral inferior side. Posterior tumors generally push the facial and auditory nerves to the medial or medial inferior side. Middle tumors generally push the facial and auditory nerves vertically (Figure 2). Due to the compression of the tumor, the facial and auditory nerves are often elongated and become thin and discolored.
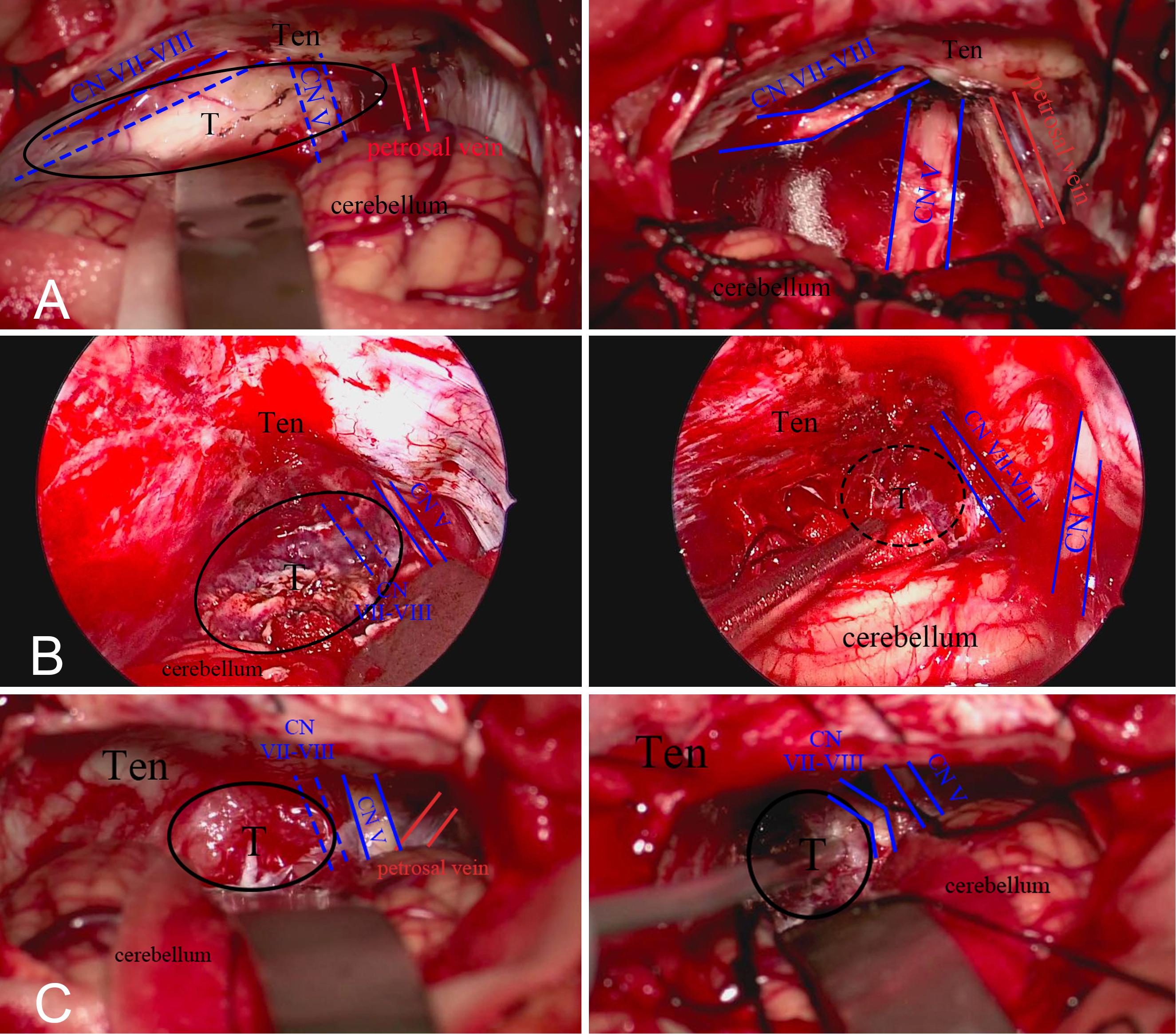
Figure 2. (A) Anterior tumors push the facial and auditory nerve to the lateral side. (B) Middle tumors push nerve to the ventral aspect. (C) Posterior tumors push nerve to the medial inferior side.
During the surgery, the operator needs to insist on sharp separation when separating the tumor from the facial and auditory nerves. The traction of the facial and auditory nerves and cerebellum should be minimized. The nutrient vessels around the facial and auditory nerves should be preserved as much as possible. Although the facial and auditory nerves are anatomically preserved after surgery, the loss of nerve function still exists in many patients, which may be result from the invasive growth of tumors, intraoperative traction, and heat conduction injury caused by electrocoagulation (10). Effective intraoperative neuroelectrophysiological monitoring can lessen the neuron injury by enabling the timely location of the facial and auditory nerves, thus increasing the rate of nerve function preservation.
In terms of inhibiting tumor progression, the tumor recurrence rate in the surgery group is similar to or even lower than the tumor enlargement rate in the radiotherapy group. Compared to stereotactic radiotherapy, surgical treatment can lead to the better results in inhibiting tumor progression. Stereotactic radiotherapy, mainly referring to Gamma Knife and cyber knife, suppresses tumor progression by killing tumor cells. The target of stereotactic radiotherapy is generally located in the center of tumors, and its dose decreases with the distance away from the center of tumors. Although peripheral dose enhancement technology has emerged in recent years, the problem of incomplete tumor boundary inactivation still exists, which also leads to the increase of the tumor enlargement rate in the radiotherapy group (11). For the surgical treatment of small cerebellopontine angle meningiomas, total resection of the tumor, dural attachment, and skull are key to preventing tumor recurrence. In the surgical principle, the operator needs to resect the dural attachment and skull after the tumor resection to achieve Simpson grade I. When the dural attachment and skull were resected incompletely, electrocoagulation is essential to reduce the possibility of tumor recurrence and achieve Simpson grade II. With the development of radiofrequency laser scalpels and other microinstruments, the surgical treatment of dural attachment and skull resection is becoming more and more standardized. The overall tumor resection rate of small cerebellopontine angle meningiomas is increasing, and the tumor recurrence rate is decreasing year by year.
Many radiotherapy studies have reported that the recurrence of small cerebellopontine angle meningiomas is connected with the pathology of the tumors. WHO grade II meningiomas are more likely to recur than WHO grade I meningiomas, which is related to the characteristic of high invasive growth. However, stereotactic radiotherapy cannot obtain the tumor tissue to examine the pathology of the tumor directly. Therefore, the grade of cerebellopontine angle meningiomas treated with radiotherapy is mostly inferred from imaging examination, which is uncertain. Contrary to stereotactic radiotherapy, surgery can directly obtain tumor tissue for pathological examination and guide further treatment through the exact pathological examination results. In the present study, WHO grade II and Simpson grade II were the risk factors of tumor recurrence after surgery. Therefore, patients with WHO grade II or Simpson grade II who undergo surgery need to receive further treatments at the early stage instead of waiting for the recurrence of tumors. This is also the reason why the recurrence rate in the surgery group was slightly lower than the tumor enlargement rate in the radiotherapy group.
In terms of further treatments, the further treatment rate in the surgery group was similar to or even lower than that reported in the radiotherapy group. Hence, compared with stereotactic radiotherapy, patients with surgical treatment might have a lower likelihood of further treatments. In Kyung et al.’s study, the subjects were asymptomatic patients with small cerebellopontine angle meningiomas. A lack of symptoms or relatively mild symptoms could significantly reduce the subjective desire of patients for treatment, which may be why the further treatment rate in Kyung et al. was lower than that reported in the surgery group. In the radiotherapy group, there was no significant difference between the number of patients who chose further radiotherapy and the number of patients who turned to surgery. In the surgery group, there was 1 patient (0.6%) who underwent a second resection, and 12 patients (7.4%) received stereotactic radiotherapy during follow-up. The reason for further radiotherapy for patients in the surgery group was the high invasiveness of the tumor and the incomplete treatment of the dural attachment and skull (12). Similar to other meningiomas, stereotactic radiotherapy of small cerebellopontine angle meningiomas is more likely to be an auxiliary treatment for inhibiting tumor progression after surgery.
The surgical complications mainly included dysfunction of the facial and auditory nerves, trigeminal nerve, and posterior cranial nerves as well as hydrocephalus. Facial paralysis, facial numbness, and hearing loss can seriously affect the quality of life of patients after surgery. The symptoms of posterior cranial nerve damage, such as hoarseness, dysarthria, and weakened cough reflex, significantly affect the prognosis of patients after surgery. Because of the small size of the tumor, complications, such as hydrocephalus and intracerebral hemorrhage, are rare in patients with small cerebellopontine angle meningiomas.
Conclusion
Compared with stereotactic radiotherapy, surgical treatment for small cerebellopontine angle meningiomas can sometimes lead to better results in relieving pre-operative symptoms and inhibiting tumor progression. In terms of further treatments, compared with the uncertainty of stereotactic radiotherapy, surgical treatment can obtain exact pathological examination results to guide the further treatment. Similar to large tumors, surgical treatment should be the first choice for small cerebellopontine angle meningiomas, while stereotactic radiotherapy, pharmacotherapy and experimental therapy are more suitable as supplement to surgical treatment.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Ethics Statement
The studies involving human participants were reviewed and approved by the Institutional Review Board and Ethics Committee of the First Affiliated Hospital of Soochow University. The patients/participants provided their written informed consent to participate in this study.
Author Contributions
JW and GC designed the study. JB, PP, HY, WG, YL, ZY, and ZW performed the experiments and analyzed the data. JB wrote the manuscript. All the authors read and approved the manuscript. JB, PJ, and HY have contributed equally to this work. All authors contributed to the article and approved the submitted version.
Funding
This work was supported by the Jiangsu Provincial Medical Key Talent grant (ZDRCA2016040) and grant (SYS2019045) from Suzhou Government.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
1. Kankane VK, Warade AC, Misra BK. Nonvestibular schwannoma tumors in the cerebellopontine angle: a single-surgeon experience. Asian J Neurosurg. (2019) 14:154–61. doi: 10.4103/ajns.ajns_335_17
2. Faramand A, Kano H, Niranjan A, Park K-J, Flickinger JC, Lunsford LD. Tumor control and cranial nerve outcomes after adjuvant radiosurgery for low grade skull-base meningiomas. World Neurosurg. (2019) 127:e221–9. doi: 10.1016/j.wneu.2019.03.052
3. Spena G, Sorrentino T, Altieri R, Zinis LR, Stefini R, Panciani PP, et al. Early-career surgical practice for cerebellopontine angle tumors in the era of radiosurgery. J Neurol Surg B Skull Base. (2018) 79:269–81. doi: 10.1055/s-0037-1606826
4. Magill ST, Lee DS, Yen AJ, Lucas CG, Raleigh DR, Aghi MK, et al. Surgical outcomes after reoperation for recurrent skull base meningiomas. J Neurosurg. (2018) 130:876–83. doi: 10.3171/2017.11.jns172278
5. Nowosielski M, Galldiks N, Iglseder S, Kickingereder P, Deimling A, Bendszus M, et al. Diagnostic challenges in meningioma. J Neuro Oncol. (2017) 19:1588–98.
6. Magill ST, Rick JW, Chen WC, Haase DA, Ralelgh DR, Aghl MK, et al. Petrous face meningiomas: classification, clinical syndromes, and surgical outcomes. World Neurosurg. (2018) 114:e1266–74. doi: 10.1016/j.wneu.2018.03.194
7. Tomasello F, Angileri FF, Conti A, Scibilia A, Cardali S, Torre DL, et al. Petrosal meningiomas: factors affecting outcome and the role of intraoperative multimodal assistance to microsurgery. Neurosurgery. (2019) 84:1313–24. doi: 10.1093/neuros/nyy188
8. Goldbrunner R, Minniti G, Preusser M, Jenkinson MD, Sallabanda K, Houdart E, et al. EANO guidelines for the diagnosis and treatment of meningiomas. Lancet Oncol. (2016) 17:e383–91. doi: 10.1016/S1470-2045(16)30321-7
9. Ehresman JS, Garzon-Muvdi T, Rogers D, Lim M, Gallia GL, Weingart J, et al. Risk of developing postoperative deficits based on tumor location after surgical resection of an intracranial meningioma. J Neurol Surg B Skull Base. (2019) 80:59–66. doi: 10.1055/s-0038-1667066
10. D’Amico RS, Banu MA, Petridis P, Bercow AS, Malone H, Praver M, et al. Efficacy and outcomes of facial nerve-sparing treatment approach to cerebellopontine angle meningiomas. J Neurosurg. (2017) 127:1231–41. doi: 10.3171/2016.10.JNS161982
11. Iwai Y, Yamanaka K, Shimohonji W, Ishibashi K. Staged gamma knife radiosurgery for large skull base meningiomas. Cureus. (2019) 11:e6001. doi: 10.7759/cureus.6001
12. Magill ST, Dalle Ore CL, Diaz MA, Jalili DD, Raleigh DR, Aghi MK, et al. Surgical outcomes after reoperation for recurrent non-skull base meningiomas. J Neurosurg. (2018) 1:1–9. doi: 10.3171/2018.6JNS18118
13. Kim KH, Kang SJ, Choi JW, Kong DS, Seol HJ, Nam DH, et al. Clinical and radiological outcomes of proactive Gamma Knife surgery for asymptomatic meningiomas compared with the natural course without intervention. J Neurosurg. (2018) 1:1–10. doi: 10.3171/2017.12JNS171943
14. Ge Y, Liu D, Zhang Z, Li Y, Li Y, Wang G, et al. Gamma Knife radiosurgery for intracranial benign meningiomas: follow-up outcome in 130 patients. Neurosurg Focus. (2019) 46:E7. doi: 10.3171/2019.3.FOCUS1956
15. Jahanbakhshi A, Azar M, Kazemi F, Jalessi M, Chanideh I, Amini E. Gamma Knife stereotactic radiosurgery for cerebellopontine angle meningioma. Clin Neurol Neurosurg. (2019) 187:105557. doi: 10.1016/j.clineuro.2019.105557
16. Patibandla MR, Lee CC, Tata A, Addagada GC, Sheehan JP. Stereotactic radiosurgery for WHO grade I posterior fossa meningiomas: long-term outcomes with volumetric evaluation. J Neurosurg. (2018) 129:1249–59. doi: 10.3171/2017.6JNS17993
17. Faramand A, Kano H, Niranjan A, Johnson SA, Hassib M, Park KJ, et al. Cranial nerve outcomes after primary stereotactic radiosurgery for symptomatic skull base meningiomas. J Neurooncol. (2018) 139:341–8. doi: 10.1007/s11060-018-2866-9
18. Sheehan JP, Starke RM, Kano H, Barnett GH, Mathieu D, Chiang V, et al. Gamma knife radiosurgery for posterior fossa meningiomas: a multicenter study. J Neurosurg. (2015) 122:1479–89. doi: 10.3171/2014.10JNS14139
19. Starke RM, Williams BJ, Hiles C, Nguyen JH, Elsharkawy MY, Sheehan JP. Gamma Knife surgery for skull base meningiomas. J Neurosurg. (2012) 116:588–97.
Keywords: small cerebellopontine angle meningioma, stereotactic radiotherapy, surgical treatment, symptom deterioration, tumor enlargement, further treatments
Citation: Bu J, Pan P, Yao H, Gong W, Liu Y, Yu Z, Wang Z, Wu J and Chen G (2020) Small Cerebellopontine Angle Meningioma—Surgical Experience of 162 Patients and Literature Review. Front. Oncol. 10:558548. doi: 10.3389/fonc.2020.558548
Received: 03 May 2020; Accepted: 04 September 2020;
Published: 09 October 2020.
Edited by:
Hailiang Tang, Huashan Hospital Affiliated to Fudan University, ChinaCopyright © 2020 Bu, Pan, Yao, Gong, Liu, Yu, Wang, Wu and Chen. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Gang Chen, bmp1X25ldXJvc3VyZ2VyeUAxNjMuY29t; Jiang Wu, c3pqaWFuZ3d1QDE2My5jb20=
†These authors have contributed equally to this work
 Jiyuan Bu
Jiyuan Bu Pengjie Pan
Pengjie Pan Hui Yao1†
Hui Yao1† Yuan Liu
Yuan Liu Jiang Wu
Jiang Wu Gang Chen
Gang Chen