- Pharmacology Laboratory, Pharmacy Faculty, Nancy University, Villers, France
This review considers whether a case can be made for a protective effect of inhibitors and blockers of the renin–angiotensin–aldosterone system (RAAS) on the cerebral circulation. It first looks at whether there exists a preferential effect on the cerebral circulation during a drug-induced lowering of high arterial blood pressure and cardiovascular morbi-mortality. It then goes on to consider background studies on the relationship between inhibition of the RAAS and stroke. This is followed by exploration of possible new directions in the inhibition of the RAAS and its effect on stroke.
Drug-Induced Lowering of High Arterial Blood Pressure and Cardiovascular Morbi-Mortality – Existence of a Preferential Effect on the Cerebral Circulation?
Once antihypertensive drugs that were reasonably well tolerated became available in the 1950s and 1960s, it became possible to evaluate whether chronically lowering high blood pressure [at first from moderately to severely elevated diastolic blood pressure (DBP) levels of 90–114 mmHg], would lower cardiovascular morbi-mortality (Freis, 1995). Thus the Veterans’ Administration (VA) cooperative study, one of the first randomized, placebo-controlled, double blind, multi-institutional clinical trials of antihypertensive treatment demonstrated that target organ damage was lowered by 50% in treated groups and furthermore that the beneficial effect on stroke was greater than that on heart attacks (Veterans’ Administration Cooperative Study Group on Antihypertensive Agents, 1970). This “preferential” effect of antihypertensive agents on the cerebral circulation was also seen in the Australian Therapeutic Trial in Mild Hypertension (The Management Committee, 1980) in which there were fewer cerebrovascular events in the treated group – but little overall difference in ischemic heart disease.
A similar result was obtained in the MRC trial (The Medical Research Council Working Party, 1985) on the treatment of mild hypertension (DBP 90–109 mmHg). In this trial stroke was lower on active treatment but there was no difference in overall rates of coronary events. Thus from an early date it appeared that antihypertensive treatment could have a beneficial effect on the cerebral circulation – and that this effect was (perhaps) greater than that on other circulations such as the coronary.
The question then arose as to whether there was any difference amongst the different antihypertensive drugs.
The MRC trial was one of the few trials to compare the effects of two different drugs – benzofluazide, a diuretic, and propranolol, a beta-blocker. They reported that “…neither of the two drug regimens had any clear overall advantage over the other…” and yet “…the diuretic was perhaps better than the beta-blocker in preventing stroke”. The latter observation was not confirmed however, in a comparison of 14 unconfounded randomized trials of antihypertensive drugs – with mainly diuretics and beta-blockers (Collins et al., 1990). They concluded that “…no consistent differences in stroke…were demonstrated in trials….”
The fact that lowering blood pressure with a wide range of drugs with very different mechanisms of action has a beneficial effect on stroke occurrence, would argue that the important factor is lowering blood pressure rather than the pharmacological way in which this is done (Figure 1). This is backed up by epidemiological data showing that stroke occurrence, survival and recurrence are all affected by high blood pressure (Sacco et al., 1982).

Figure 1. Possible pressure-dependent (A) and pressure-independent (B) effects of antihypertensive drugs on the cerebral circulation and stroke.
Inhibition of the RAAS and Stroke – Background Studies
Although drugs such as diuretics and beta-blockers do impact on the renin–angiotensin–aldosterone system (RAAS), their effect on blood pressure is not primarily related to their effects on the RAAS.
Drugs that lower blood pressure by blocking the RAAS became available in the 1980s.
When the first inhibitor of the RAAS that was reasonably well tolerated – captopril an inhibitor of the angiotensin I converting enzyme (ACEI) – began to be more and more widely used in the early 1980s, several questions were asked. Were ACEIs safe drugs compared to the well-known conventional treatment available at the time (with diuretics and beta-blockers)? If ACEIs were safe to use then was their effect on cardiovascular morbi-mortality as large as (or greater than) that obtained with conventional therapy?
The Nordic Captopril Prevention Project (CAPP; Hansson et al., 1999) was one of the few trials comparing “newer” to “older” treatment, viz captopril with conventional antihypertensive therapy with diuretics and beta-blockers. The two regimens did not differ in preventing cardiovascular morbi-mortality excepting that stroke was apparently more common with captopril. The authors suggested that the difference in stroke risk “…is probably due to the lower levels of blood pressure obtained initially in previously treated people randomized to conventional therapy.” Again in this trial blood pressure appeared to be the predominant factor.
Albeit in other trials the results on the relative risk for strike were in favor of the ACEI. Thus in the HOPE trial (The Heart Outcomes Prevention Evaluation (HOPE) Study Investigators, 2000b) using the ACEI ramipril, the relative risk for stroke (0.68, P < 0.01) was in favor of the ACEI. The authors argued that the beneficial effect of ramipril was linked to inhibition of the RAAS or to some other effect of ramipril unrelated to lowering of blood pressure. In a related study – the microalbuminuria, cardiovascular and renal outcomes (MICRO) HOPE sub-study (The Heart Outcomes Prevention Evaluation (HOPE) Study Investigators, 2000a), the risk reduction of cardiovascular events was again greater than that to be expected from the difference in blood pressure, suggesting that ramipril, and perhaps ACEIs in general, have a “protective” effect on the vascular wall.
This could be brought about by inhibition of the RAAS and/or an increase in bradykinin (Unger, 2002). Regarding the latter however, it should be pointed out that stimulation of B2 receptors may produce brain damage (Schöller et al., 2011) and so offset any beneficial effect obtained with bradykinin-induced cerebral vasodilatation.
Following the establishment of ACEIs as effective and safe antihypertensive drugs with potential protective effects on the vasculature, angiotensin II type 1 receptor antagonists (ARB) were introduced. Losartan was the first ARB available clinically and in the LIFE trial (Dahlöf et al., 2002) its cardiovascular effects were compared to those of the beta-blocker, atenolol. The conclusion was that “…losartan prevents more cardiovascular morbidity…than atenolol for a similar reduction in blood pressure”. Once again a trial that showed a beneficial effect of RAAS blockade.
Debate continues over whether a certain mechanism of action (in this case blockade of the RAAS) confers additional beneficial impact on cardiovascular morbi-mortality over and above the reduction in blood pressure (Figure 1). Potential mechanisms involved – especially for ARBs – have been discussed in several reviews (Chrysant, 2005; Thöne-Reineke et al., 2006). These include the possibility that blockade of the angiotensin II type 1 receptor (vasoconstrictor) by ARB treatment, “uncovers” angiotensin II stimulation of the vasodilator angiotensin II type 2 receptor. Our group has shown that in the presence of the ARB, telmisartan, angiotensin II produces dilatation of cerebral arterioles in an animal model of hypertension, the spontaneously hypertensive rats (SHR; Vincent et al., 2005; Figure 2).
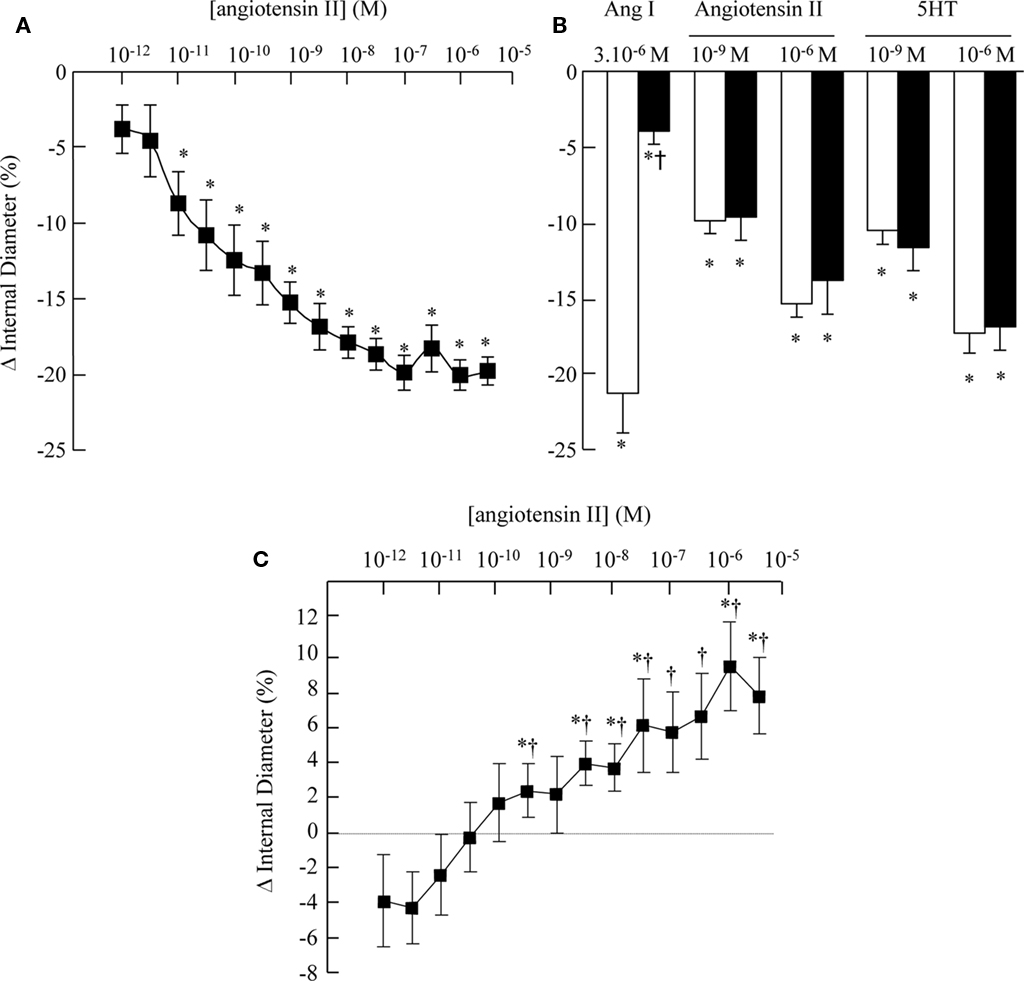
Figure 2. Effects of angiotensins on cerebral arteriolar diameter (from Vincent et al., 2005). Cumulative concentration–response curve for arteriolar vasoconstriction evoked by angiotensin II [(A), n = 7], and effects of the ACEI, captopril (10−5 M, solid bar), on vasoconstriction induced by angiotensin I, angiotensin II, and 5HT [(B), n = 4]. Results are expressed as % fall in diameter. Values are mean ± SEM. *P ≤ 0.05 vs. baseline; †P ≤ 0.05 vs. responses induced by angiotensin I. (C) Cumulative concentration–response curve for the arteriolar vasomotor response to angiotensin II in presence of the angiotensin AT1 receptor antagonist, telmisartan (10−5 M). Results are expressed as % change in diameter. Values are mean ± SEM; n = 5. *P ≤ 0.05 vs. baseline; †P ≤ 0.05 vs. responses induced by angiotensin II alone.
Inhibition of the RAAS and Stroke – New Directions
Use of Combinations of Existing Drugs
Bi- and tri-therapy regimens have been used in the treatment of high arterial blood pressure for several decades. Regimens generally use drugs that act on different pharmacological pathways. Thus ACEIs or ARBs that block the RAAS are combined with diuretics that lower blood pressure by causing the body to rid itself of excess fluids and sodium (whilst at the same time activating the RAAS).
An original approach using combinations of drugs that act on the same pharmacological system has been proposed (Unger, 2002). Treatment with an ACEI may not completely block the RAAS as angiotensin I levels may rise due to interruption of the negative feedback control of renin release by angiotensin II, dissociation of the ACEI from the active sites of the enzyme and/or production of angiotensin II by other enzymatic pathways such as those involving chymase (Menard et al., 1997). When a combination of low doses of enalapril (ACEI) and losartan (ARB) was given to SHR, a synergistic effect on blood pressure and left ventricular hypertrophy was observed and this was attributed to more effective inhibition of the RAAS (Menard et al., 1997).
In the ONTARGET clinical trial (The ONTARGET Investigators, 2008) telmisartan (ARB) was combined with ramipril (ACEI) again on the basis that an ACEI does not completely block the RAAS so addition of an ARB may be beneficial. However no significant benefit was seen in the primary outcome and side effects such as the risk of hypotension and hyperkalemia were more marked. In the ONTARGET trial standard clinical doses of telmisartan and ramipril were used. This may have given an overall antihypertensive dose that was too high, thus producing side effects and masking the potentially beneficial synergism. Thus we carried out a study in the SHR similar to that of Menard et al. (1997) using a combination of suboptimal doses, i.e., doses that did not normalize blood pressure when given separately (Dupuis et al., 2010). The 0.8 mg/kg/day telmisartan plus 0.1 mg/kg/day ramipril combination (TEL8/RAM1) normalized cerebral arteriolar diameter (62 ± 18 μm, P < 0.05 vs. untreated SHR 42 ± 16 and normotensive WKY rats 59 ± 16, n = 18 or 19 per group) and the lower limit of cerebral blood flow autoregulation (50 ± 10 mmHg vs. 77 ± 28 for SHR and 53 ± 17 for WKY). TEL8/RAM1 reversed the effect of angiotensin II on cerebral arterioles to vasodilatation. Although the translation of such beneficial structural and functional effects in an animal model into a beneficial effect on stroke in man is debatable, on the basis of these results and those of Menard et al. (1997), quoted above, there may be a case for studying the effects of combinations of low doses of different inhibitors of the RAAS (Budzyn et al., 2010; see later).
New Drugs
Renin inhibitors
The work of Goldblatt and others (Katz et al., 1939) in the early 1930s suggested that the link between renal ischemia provoked, for instance, by renal artery stenosis was renin. Building on this, the group of Page and Helmer at Lilly, USA provided evidence of the existence of an endogenous inhibitor for renin (Page and Helmer, 1940) and this stimulated research on the use of a renin inhibitor for the treatment of high blood pressure. Gross and others showed that renin inhibitors such as the Streptomyces pentapeptide, pepstatin, would lower blood pressure in animal models (Lazar et al., 1972).
At this time work was being carried out simultaneously on the first ACEI (teprotide, the peptide from the venom of the snake, Bothrops jararaca) and the first ARB (the peptide analog of angiotensin II, saralasin). All three – pepstatin, teprotide, and saralasin – are peptides presenting pharmacokinetic difficulties when used for daily oral treatment, as is the case for hypertension. In the pharmaco-chemical race to find therapeutically usable analogs, ACEI research came in first with captopril, and from then on, interest waned in other forms of peptide research in the RAAS such as the research on renin inhibitors.
Albeit, research on renin inhibitors did not cease and in the 1990s Novartis, Switzerland developed an orally active renin inhibitor, aliskiren. The rational behind this was the same as that of the research of the 1960s and 1970s: a “more complete” inhibition of the RAAS can be obtained by blocking the first rate-limiting enzyme (renin) at the summit of the metabolic cascade of production of active peptides (angiotensins; Paulis and Unger 2010; Riccioni, 2011). Recent clinical trials suggest that aliskiren may have beneficial cardiovascular and renal effects especially in diabetes (Parving et al., 2008); other trials are looking at the effect of aliskiren on stroke (Parving et al., 2009).
It is interesting to note that in the above trials on renin inhibition, dual RAAS blockade discussed previously is in fact being used: the renin inhibitor is added onto existing treatment with an ACEI or an ARB. Albeit, it has recently been concluded that for the time being: “…the use of dual RAAS blockade should be avoided unless ironclad data emerge to the contrary” (Messerli et al., 2010).
Specific aldosterone antagonists
In parallel to blocking the RAAS cascade at the summit (with a renin inhibitor) there may be a case for blocking the system at the lowest level of the cascade of active products – aldosterone. The RAAS presents evolutionary duality with the appearance firstly of a blood pressure regulatory system (using angiotensins) followed by the evolution of a regulatory system for sodium metabolism (using aldosterone; Withers, 1992). Thus although the RAAS is absent in cyclostomes and elasmobranchs, juxtaglomerular cells are present in other chondrichthyean fish and all other vertebrates. The macula densa, however, appears later in some amphibia and higher tetrapods, suggesting that it evolved at a more advanced stage. Thus the RAAS may have evolved as a blood pressure regulatory system with sodium regulation developing later.
On the basis of the argument it has been postulated that aldosterone acts strictly as a mineralocorticoid hormone on renal and other epithelia, to maintain electrolytic homeostasis (Delyani et al., 2001). However, already in the 1970s it was suggested that aldosterone may play a negative role in vascular diseases such as stroke (Brunner et al., 1972). More recently evidence has revealed that aldosterone acts also on non-epithelial vascular sites; other results have shown that blocking the effects of aldosterone with antagonists such as spironolactone diminishes cerebrovascular lesions in the rat model of stroke (the SHR-SP; Rocha et al., 1998). Recently our group has detected an inverse linear relationship between plasma aldosterone levels and the diameter of the cerebral arteriole in SHR (Figure 3). Arguments are emerging therefore, that suggest the mineralocorticoid receptor as a potential therapeutic target for stroke prevention (Osmond et al., 2008).
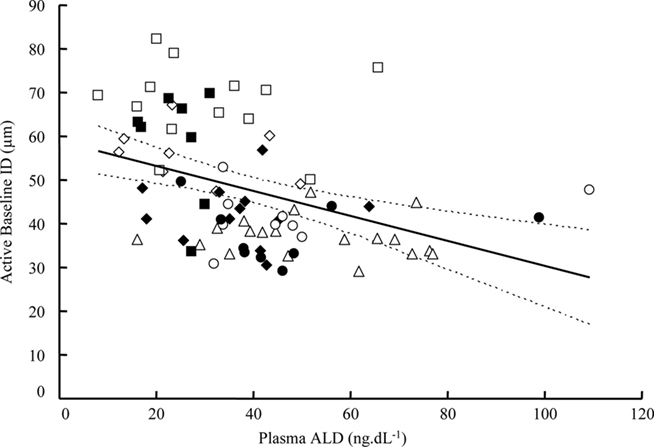
Figure 3. Relationship between cerebral arteriolar diameter and aldosterone level. Relationship between aldosterone (ALD) plasma level and cerebral arteriolar internal diameter in WKY-solvent (open square), WKY-ALD (full square), SHR-solvent (open triangle), SHR-ARB (open circle), SHR-ARB + ALD (full circle), SHR-ACEI (open lozenge), and SHR-ACEI + ALD (full lozenge). Pooled n = 78. Linear regression equation: Y = −0.29X + 59; r2 = 0.161; degrees of freedom = 76 (P < 0.05).
New Therapeutic Indications and Pathways
RAAS, neurological performance and cognition
In clinical trials on stroke, parameters such as occurrence, survival, and recurrence are measured. This contrasts with preclinical studies. Except in some rare species such as dogs, spontaneous stroke rarely occurs. In most laboratory animal models cerebral ischemia is induced by various means such as surgical stenosis. Thus frequency of occurrence of stroke is by definition 100% and other parameters such as neurological performance are measured. Inhibitors of the RAAS are active in such models. Thus our group has shown that the ACEI captopril will improve neurological performance following induced ischemia in renovascular hypertensive rats (Capdeville et al., 1990). Again the translation of preclinical results into clinical reality is not assured. Recent clinical investigation into a possible beneficial effect of RAAS inhibition on neurological performance and cognition in the TRANSCEND trial (Anderson et al., 2011) came to the conclusion that “…blocking of the renin-angiotensin system had no clear effects of cognitive performance”. This question remains open.
RAAS inhibition in non-hypertensive patients
If it is hypothesized that inhibition of the RAAS can have beneficial effects that are not necessarily related to blood pressure then a case could be made for treating patients presenting cardiovascular risk, e.g., previous stroke or transient ischemic attacks, that are not necessarily hypertensive. This was investigated in the PROGRESS trial (MacMahon et al., 2001). In this trial treatment with an ACEI, perindopril, plus a diuretic, indapamide, reduced stroke risk in the non-hypertensive group.
Such observations raise another question regarding the risks and benefits of lowering blood pressure below classical normotensive goals. In the APRIL study, normotensive rats were treated from the age of 6 through 30 months with a hypotensive dose of the ACEI, perindopril, that lowered DBP from 90 to 80 mmHg (Lartaud et al. 1994). Cerebrovascular dysfunction in old rats as evaluated by an increase in the lower limit of autoregulation of cerebral blood flow, was corrected by ACEI treatment. Thus in this animal study, life-long ACEI treatment and/or systemic arterial hypotension improved cerebrovascular function.
There have been few clinical trials looking at the beneficial effects of arterial “hypotension.” In the HOT study with the calcium entry blocker, felodipine, lowering DBP below 90 mmHg had a beneficial effect on myocardial infarction but not on stroke incidence (Hansson et al., 1998). The question remains open.
RAAS and large artery stiffness
With aging and cardiovascular disease such as hypertension, the elastic properties of large central arteries change leading to stiffening of the wall. This leads to a decrease in compliance and an increase in pulse pressure. This can be important in vascular beds with low flow resistance and torrential flow such as that of the brain (O’Rourke and Safar, 2005). Exposure of small vessels to highly pulsatile pressure and flow can lead to structural modification and microvascular damage. This was shown for instance in the SHR rat by the group of Baumbach (1996) who found that an increase in pulse pressure – even in the absence of an increase in mean pressure – is enough to produce arteriolar wall hypertrophy.
Angiotensin I converting enzyme inhibitors may be useful in this context as they appear to protect the elastic fiber network of the arterial wall and reduced the age-related increase in wall stiffness (Figure 4).
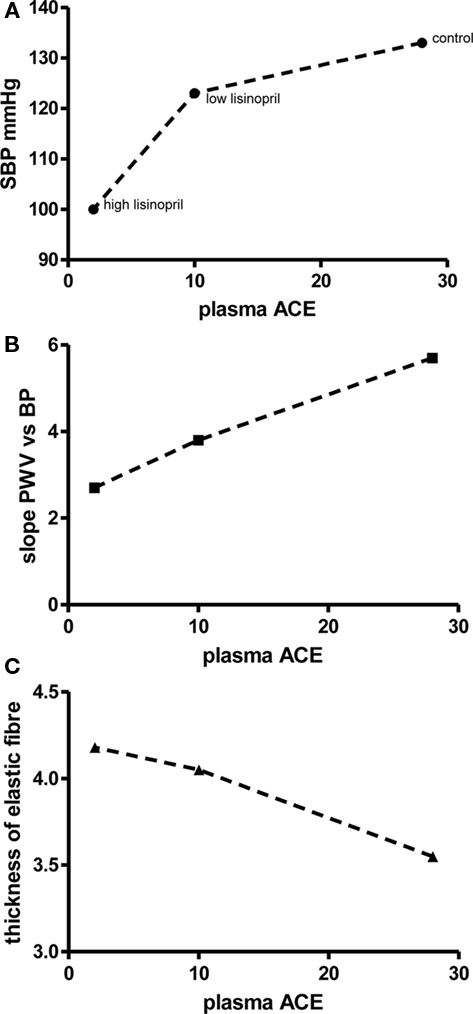
Figure 4. Increases in arterial distensibility produced by the ACEI, lisinopril (recalculated and redrawn from Makki et al., 1994). Four-month-old normotensive rats were treated orally for 9 months with a low dose of lisinopril (0.9 mg/kg/day) or a high dose (9 mg/kg/day). ACE was inhibited in a dose-related fashion but the low dose treatment did not significantly modify systolic blood pressure (A). Arterial stiffness (judged from the relationship between pulse wave velocity and mean blood pressure) was reduced in a dose-dependent fashion (B) and this was probably due in part to a reduction in the loss of thickness of arterial elastic fibers with age (C).
RAAS genomics and stroke
Analysis of RAAS gene polymorphisms may provide information for a targeted pharmaco-genomic approach to stroke prevention. The MOBILIZE Boston study (Hajjar et al., 2010) revealed that the angiotensinogen gene may be involved in cerebrovascular reactivity independently of blood pressure. Previous studies have also shown links between the RAAS gene polymorphisms and other forms of cerebrovascular dysfunction such as lacunar infarction (Takami et al., 2000) or cerebral white matter pathology (van Rijn et al., 2007) although the link to clinical stroke seems more tenuous (van Rijn et al., 2007). Again the question remains open as to whether future research will reveal RAAS gene polymorphism linked to stroke that could form the basis of pharmaco-genomic treatment with inhibitors and antagonists of the RAAS.
Concluding Remarks
The important issue in antihypertensive therapy is whether the latter prevents target organ damage in the brain, kidney, and other vascular beds. This review looks at the preclinical and clinical evidence suggesting that drugs acting via inhibition of the RAAS have a cerebrovascular protective effect. Whilst a case can be made for this hypothesis, the latter should be strengthened by further evidence from clinical trials. Furthermore a deeper investigation into the predictability of animal models of hypertension when used to test drugs and elaborate mechanisms is required.
Conflict of Interest Statement
The author declares that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
Anderson, C., Teo, K., Gao, P., Arima, H., Dans, A., Unger, T., Commerford, P., Dyal, L., Schumacher, H., Pogue, J., Paolasso, E., Holwerda, N., Chazova, I., Binbrek, A., Young, J., and Yusuf, S. for the ONTARGET and TRANSCEND Investigators. (2011). Renin–angiotensin system blockade and cognitive function in patients at high risk of cardiovascular disease: analysis of data from the ONTARGET and TRANSCEND studies. Lancet Neurol. 10, 43–53.
Baumbach, G. L. (1996). Effects of increased pulse pressure on cerebral arterioles. Hypertension 27, 159–167.
Brunner, H., Laragh, J. H., Baer, L., Newton, M. A., Goodwin, F. T., Krakoff, L. R., Bard, R. H., and Bühler, F. (1972). Essential hypertension: renin and aldosterone, heart attack and stroke. N. Engl. J. Med. 286, 441–449.
Budzyn, K., Sobey, C. G., and Drummond, G. R. (2010). Reduced cardiovascular remodelling and functional impairment in spontaneously hypertensive rats following combined treatment with suboptimal doses of telmisartan and ramipril: is less really more? J. Hypertension 28, 1384–1389.
Capdeville, C., el Bouhtoury, F., Parache, R. M., Boulu, R. G., Guillou, J., and Atkinson, J. (1990). The angiotensin I converting enzyme inhibitors captopril and Wy-44,655 attenuate the consequences of cerebral ischemia in renovascular hypertensive rats. Life Sci. 47, 539–549.
Chrysant, S. G. (2005). Possible pathophysiologic mechanisms supporting the superior stroke protection of angiotensin receptor blockers compared to angiotensin-converting enzyme inhibitors: clinical and experimental evidence. J. Hum. Hypertens. 19, 923–931.
Collins, R., Peto, R., MacMahon, S., Hebert, P., Fiebach, N. H., Eberlein, K. A., Godwin, J., Qizilbash, N., Taylor, J. O., and Hennekens, C. H. (1990). Blood pressure, stroke, and coronary heart disease. Part 2. Short-term reductions in blood pressure: overview of randomised drug trials in their epidemiological context. Lancet 335, 827–838.
Dahlöf, B., Devereux, R. B., Kjeldsen, S. E., Julius, S., Beevers, G., de Faire, U., Fyhrquist, F., Ibsen, H., Kristiansson, K., Lederballe-Pedersen, O., Lindholm, L. H., Nieminen, M., Omvik, P., Oparil, S., and Wedel, H., for the LIFE Investigators. (2002). Cardiovascular morbidity and mortality in the losartan intervention for endpoint reduction in hypertension study (LIFE): a randomised trial against atenolol. Lancet 359, 995–1002.
Delyani, J. A., Rocha, R., Cook, C. S., Tolbert, D. S., Levin, S., Roniken, B., Workman, D. L., Sing, Y.-L., and Whelhan, B. (2001). Eplerenone: a selective aldosterone receptor antagonist (SARA). Cardiovasc. Drug Rev. 19, 185–200.
Dupuis, F., Vincent, J.-M., Liminana, P., Chillon, J.-M., Capdeville-Atkinson, C., and Atkinson, J. (2010). Effects of suboptimal doses of the AT1 receptor blocker, telmisartan, with the angiotensin-converting enzyme inhibitor, ramipril, on cerebral arterioles in spontaneously hypertensive rat. J. Hypertens. 28, 1566–1573.
Freis, E. (1995). “Historical development of antihypertensive treatment,” in Hypertension: Patho-physiology, Diagnosis and Management, 2nd Edn., eds J. H. Laragh, and B. M. Brenner (New York: Raven Press), 2741–2751.
Hajjar, I., Sorond, F., Hsu, Y.-H., Galica, A., Cupples, L. A., and Lipsitz, L. A. (2010). Renin angiotensin system gene polymorphisms and cerebral blood flow regulation. The MOBILIZE Boston Study. Stroke 41, 635–640.
Hansson, L., Lindholm, L. H., Niskanen, L., Lanke, J., Hedner, T., Niklason, A., Luomanmaki, K., Dahlöf, B., de Faire, U., Mörlin, C., Karlberg, B. E., Wester, P. O., and Björck, J.-E. for the Captopril Prevention Project (CAPPP) study group. (1999). Effect of angiotensin-converting enzyme inhibition combined with conventional therapy on cardiovascular morbidity and mortality in hypertension: the Captopril Prevention Project (CAPPP) randomised trial. Lancet 353, 611–616.
Hansson, L., Zanchetti, A., Carruthers, S. G., Dahlöf, B., Elmfeldt, D., Julius, S., Menard, J., Rahn, K. H., Wedel, H., and Westerling, S. for the HOT study group. (1998). Effects of intensive blood-pressure lowering and low-dose aspirin in patients with hypertension: principal results of the Hypertension Optimal Treatment (HOT) randomised trial. Lancet 351, 1755–1762.
Katz, L. N., Friedman, M., Rodbard, S., and Weinstein, W. (1939). Observations on the genesis of renal hypertension. American Heart Journal 17, 334–356.
Lartaud, I., Makki, T., Bray-des-Boscs, L., Niederhoffer, N., Atkinson, J., Corman, B., and Capdeville-Atkinson, C. (1994). Effect of chronic ANG I-converting enzyme inhibition on aging processes. IV. Cerebral blood flow regulation. Am. J. Physiol. 267, R687–R694.
Lazar, J., Orth, H., Möhring, J., and Gross, F. (1972). Effects of the renin inhibitor pepstatin on blood pressure in intact and nephrectomised rats. N. S. Arch. Pharmacol. 275, 114–118.
MacMahon, S., Neal, B., and Tzourio, C., the PROGRESS study group. (2001). Randomised trial of a perindopril-based blood-pressure-lowering regimen among 6105 individuals with previous stroke or transient ischemic attack. Lancet 358, 1033–1041.
Makki, T., Tatchum-Talom, R., Niederhoffer, N., Amin, F., Tankosic, P., Mertes, P.-M., and Atkinson, J. (1994). Increased arterial distensibility induced by the angiotensin-converting enzyme inhibitor lisinopril, in normotensive rats. Br. J. Pharmacol. 111, 555–560.
Menard, J., Campbell, D. J., Azizi, M., and Gonzales, M.-F. (1997). Synergistic effects of ACE inhibition and Ang II antagonism on blood pressure, cardiac weight, and renin in spontaneously hypertensive rats. Circulation 96, 3072–3078.
Messerli, F. H., Staessen, J. A., and Zannad, F. (2010). Of fads, fashion, surrogate endpoints and dual RAS blockade. Eur. Heart J. 31, 2205–2209.
O’Rourke, M. F., and Safar, M. E. (2005). Relationship between aortic stiffening and microvascular disease in brain and kidney. Cause and logic of therapy. Hypertension 46, 200–204.
Osmond, J. M., Rigsby, C. S., and Dorrance, A. M. (2008). Is the mineralocorticoid receptor a potential target for stroke prevention? Clin. Sci. 114, 37–47.
Page, I. H., and Helmer, G. M. (1940). Angiotonin-activator, renin- and angiotonin-inhibitor, and the mechanism of angiotonin tachyphylaxis in normal, hypertensive, and nephrectomised animals. J. Exp. Med. 71, 495–519.
Parving, H.-H., Brenner, B. M., McMurray, J. J. V., de Zeeuw, D., Haffner, S. M., Solomon, S. D., Chaturvedi, N., Ghadanfar, M., Weissbach, N., Xiang, Z., Armbrecht, J., and Pfeffer, M. A. (2009). Aliskiren trial in type 2 diabetes using cardio-renal endpoints (ALTITUDE): rationale and study design. Nephrol. Dial. Transplant. 24, 1663–1671.
Parving, H.-H., Persson, F., Lewis, J. B., Lewis, E. J., and Hollenberg, N. K. (2008). Aliskiren combined with losartan in type 2 diabetes and nephropathy. N. Engl. J. Med. 358, 2433–2446.
Paulis, L., and Unger, T. (2010). Novel therapeutic targets in hypertension. Nat. Rev. Cardiol. 7, 431–441.
Riccioni, G. (2011). Aliskiren in the treatment of hypertension and organ damage. Cardiovasc. Ther. 29, 77–87.
Rocha, R., Chander, P. N., Khanna, K., Zuckerman, A., and Stier, C. T. (1998). Mineralocorticoid blockade reduces vascular injury in stroke-prone hypertensive rats. Hypertension 31, 451–458.
Sacco, R. L., Wolf, P. A., Kannel, W. B., and McNamara, P. M. (1982). Survival and recurrence following stroke. The Framingham study. Stroke 13, 290–286.
Schöller, K., Feiler, S., Anetsberger, S., Kim, S.-W., and Plesnila, N. (2011). Contribution of bradykinin receptors to the development of secondary brain damage after experimental subarachnoid haemorrhage. Neurosurgery 68, 1118–1123.
Takami, S., Imai, Y., and Katsuya, T. (2000). Gene polymorphism of the renin–angiotensin system associates with risk for lacunar infarction. Am. J. Hypertens. 13, 121–127.
The Heart Outcomes Prevention Evaluation (HOPE) Study Investigators. (2000a). Effects of ramipril on cardiovascular and microvascular outcomes in people with diabetes mellitus: results of the HOPE study and MICRO-HOPE substudy. Lancet 355, 253–258.
The Heart Outcomes Prevention Evaluation (HOPE) Study Investigators. (2000b). Effects of an angiotensin-converting enzyme inhibitor, ramipril, on cardiovascular events in high risk patients. N. Engl. J. Med. 342, 145–152.
The Management Committee. (1980). The Australian therapeutic trial in mild hypertension (ATTMH). Report by the Management Committee. Lancet 1, 1261–1267.
The Medical Research Council Working Party. (1985). MRC trial on the treatment of mild hypertension: principal results. Br. Med. J. 291, 97–104.
The ONTARGET Investigators. (2008). Telmisartan, ramipril, or both in patients at high risk for vascular events. N. Engl. J. Med. 358, 1547–1559.
Thöne-Reineke, C., Steckelings, U. M., and Unger, T. (2006). Angiotensin receptor blockers and cerebral protection in stroke. J. Hypertens. 24(Suppl. 1), S115–S121.
Unger, T. (2002). The role of the renin–angiotensin system in the development of cardiovascular disease. Am. J. Cardiol. 89(Suppl.), 3A–10A.
van Rijn, M. J. E., Bos, M. J., Isaacs, A., Yazdanpanah, M., Arias-Vasquez, A., Strikcer, B. H. C., Klungel, O. H., Oostra, B. A., Koudstaal, P. J., Witteman, J. C., Hofman, A., Breteler, M. M. B., and van Duijn, C. M. (2007). Polymorphisms of the renin–angiotensin system are associated with blood pressure, atherosclerosis and cerebral white matter pathology. J. Neurol. Neurosurg. Psychiatry 78, 1083–1087.
Veterans’ Administration Cooperative Study Group on Antihypertensive Agents. (1970). Effects of treatment on morbidity in hypertension: II. Results in patients with diastolic blood pressure averaging 90 through 114 mmHg. J. Am. Med. Assoc. 213, 143–152.
Keywords: blood pressure, cerebral circulation, rat model, stroke, angiotensin I converting enzyme inhibitors, angiotensin AT1 receptor blockers
Citation: Atkinson J (2011) Stroke, high blood pressure and the renin–angiotensin–aldosterone system – new developments. Front. Pharmacol. 2:22. doi: 10.3389/fphar.2011.00022
Received: 08 March 2011; Paper pending published: 18 March 2011;
Accepted: 06 April 2011; Published online: 27 April 2011.
Edited by:
Y. W. Kwan, Chinese University of Hong Kong, Hong KongReviewed by:
Michel Félétou, Institut De Recherches Servier, FranceXiao Qiang Yao, Chinese University of Hong Kong, China
Copyright: © 2011 Atkinson. This is an open-access article subject to a non-exclusive license between the authors and Frontiers Media SA, which permits use, distribution and reproduction in other forums, provided the original authors and source are credited and other Frontiers conditions are complied with.
*Correspondence: Jeffrey Atkinson, Pharmacology Laboratory, Pharmacy faculty, Nancy University, 12 rue de Versigny, F-54600 Villers, France. e-mail:amVmZnJleS5ha2luc29uQHBoYXJtYS51aHAtbmFuY3kuZnI=
