- 1Program in Anemia Signaling Research, Division of Nephrology, Program in Membrane Biology, Center for Systems Biology, Massachusetts General Hospital, Harvard Medical School, Boston, MA, USA
- 2INSERM, U1043, CNRS, U5282, Université Paul Sabatier, Centre de Physiopathologie de Toulouse Purpan, Toulouse, France
Matriptase-2, encoded by the TMPRSS6 gene, is a member of the type II transmembrane serine protease family. Matriptase-2 has structural and enzymatic similarities to matriptase-1, which has been implicated in cancer progression. Matriptase-2 was later established to be essential in iron homeostasis based on the phenotypes of iron-refractory iron deficiency anemia identified in mouse models as well as in human patients with TMPRSS6 mutations. TMPRSS6 is expressed mainly in the liver and negatively regulates the production of hepcidin, the systemic iron regulatory hormone. This review focuses on the current understanding of matriptase-2 biochemistry, and its role in iron metabolism and cancer progression. In light of recent investigations, the function of matriptase-2 in hepcidin regulation, how it is being regulated, as well as the therapeutic potential of matriptase-2 are also discussed.
Biochemistry of Matriptase-2
Type II transmembrane serine protease matriptase-2, encoded by the TMPRSS6 gene, belongs to the family of type II transmembrane serine proteases (TTSP). Matriptase-2 is comprised of a transmembrane domain, followed by a sea urchin sperm protein, enteropeptidase and agrin (SEA) domain, a stem region containing two complement factor C1r/C1s, urchin embryonic growth factor and bone morphogenetic protein (CUB) domains and three low-density lipoprotein receptor (LDLR) class A repeats, and a C-terminal trypsin-like serine protease domain (Velasco et al., 2002; Ramsay et al., 2008). Matriptase-2 is synthesized as a single chain inactive proenzyme, which auto-activates itself by a cleavage at an arginine residue at the RIVGG consensus site between the prodomain and the catalytic domain (Ramsay et al., 2009b; Altamura et al., 2010). After the auto-activation, it remains membrane-bound through a single disulphide bond linking the pro- and catalytic domains (Ramsay et al., 2009a). Once the catalytic domain is released, it migrates as a single or dimeric species (Silvestri et al., 2008). Matriptase-2 shares high structural and enzymatic similarities with matriptase-1, which contains four LDLR repeats instead of three (Sanders et al., 2010), is expressed in epithelial cells, and has been implicated in the progression of cancers, such as breast, prostate, and colorectal cancer (Oberst et al., 2001; Velasco et al., 2002; Kang et al., 2003; Riddick et al., 2005).
The structural features of matriptase-2 are highly conserved across mammalian species, including human, macaque monkey, dog, cow, mouse and rat, with human protein sharing >80% identity to matriptase-2 from other species (Ramsay et al., 2008). The expression pattern of TMPRSS6 determined from mRNA expression studies and analysis of GenBank Unigene database indicates that matriptase-2 is predominantly expressed in the liver (Velasco et al., 2002; Finberg et al., 2008) but also to a lower extent in the kidney, spleen, brain, lung, mammary gland, testis, and uterus (Ramsay et al., 2008). In addition, aberrant expression of TMPRSS6 is observed in different human cancers such as breast and prostate cancer (Parr et al., 2007; Sanders et al., 2008).
Matriptase-1, a close relative of matriptase-2, is known to be associated with two endogenous inhibitors: hepatocyte growth factor activator inhibitor (HAI)-1 and HAI-2, which inhibit matriptase-1 dependent activation of its physiological substrates, likely through an interaction with the second CUB domain (Szabo et al., 2008; Inouye et al., 2010). With 35% identity and structural similarities with matriptase-1 (Velasco et al., 2002), it is possible that matriptase-2 is also associated with an endogenous inhibitor. Indeed, Maurer et al. (2013) recently demonstrated that HAI-2 is a cognate inhibitor of matriptase-2 that inhibits its proteolytic activity, and thus increases hepcidin expression in vitro. However, the physiological role of HAI-2 in the regulation of hepcidin and iron metabolism remains to be investigated.
Following the identification and characterization of matriptase-2, Velasco et al. (2002) also examined the enzymatic activity of the catalytic serine protease domain against extracellular matrix components. It was found that matriptase-2 has the capacity to degrade fibronectin, fibrinogen, and type I collagen. Recently, membrane bound hemojuvelin has also been identified as a substrate for matriptase-2 in vitro (Silvestri et al., 2008), providing a straightforward mechanism for the effects of TMPRSS6 mutations on hepcidin and iron regulations. However, as will be discussed below, evidence exists in vivo that is not consistent with this hypothesis.
Role of Matriptase-2 in Iron Metabolism
Matriptase-2 is produced mainly by the liver and negatively regulates the production of hepcidin, the systemic iron regulatory hormone encoded by the HAMP gene (Du et al., 2008; Finberg et al., 2008). Hepcidin is a peptide secreted by the liver that plays a central role in adjusting iron absorption to meet iron needs of the body (Nicolas et al., 2001). Hepcidin negatively regulates cellular iron export by promoting the degradation of ferroportin (Nemeth et al., 2004), the only known iron exporter present on the surface of duodenal enterocytes, macrophages, and hepatocytes and thus limits iron absorption and iron release. It is now well established that Hamp expression is regulated by the bone morphogenetic protein (BMP)/sons of mothers against decapentaplegic (SMAD) signaling pathway (Babitt et al., 2006, 2007).
At the molecular level, BMP6, the endogenous ligand of BMP/SMAD signaling, activates BMP-receptor complex by binding to type I and type II BMP receptors that induces phosphorylation (Andriopoulos et al., 2009; Meynard et al., 2009). The activated complex, in turn, phosphorylates Smad1,5,8/Smad4 complex, which then translocates to nucleus to modulate gene transcription (Wang et al., 2005; Babitt et al., 2006; Kautz et al., 2008). Hemojuvelin (HJV) acts as a coreceptor and is required to fully activate the BMP signaling ability (Babitt et al., 2006). The expression of BMP6 is proportional to hepatic iron concentrations and consistent with Hamp mRNA expression (Kautz et al., 2008).
TMPRSS6 Mutations in Mice and Human
Matriptase-2 regulates Hamp expression through the BMP/SMAD pathway (Finberg et al., 2010; Lenoir et al., 2011) in an as yet unfully characterized manner. Mice without functional matriptase-2 (both mask mice with truncated Tmprss6 lacking the protease domain and Tmprss6 knockout mice) showed a hypochromic microcytic anemia and an alopecia (Du et al., 2008; Folgueras et al., 2008). These phenotypes resulted from inappropriately high levels of Hamp mRNA expression (Du et al., 2008; Folgueras et al., 2008; Finberg et al., 2010).
Mutations in TMPRSS6 in humans led to iron-refractory iron deficiency anemia (IRIDA) that is unresponsive to oral iron treatment and only partially responsive to parental iron therapy (Finberg et al., 2008). IRIDA is also characterized by congenital hypochromic, microcytic anemia, low mean corpuscular erythrocyte volume, low transferrin saturation, and defects in iron absorption and utilization (Finberg et al., 2008; Guillem et al., 2008; Melis et al., 2008). Currently, there are 42 different TMPRSS6 mutations reported in humans, scattered throughout all the different extracellular domains (Figure 1).
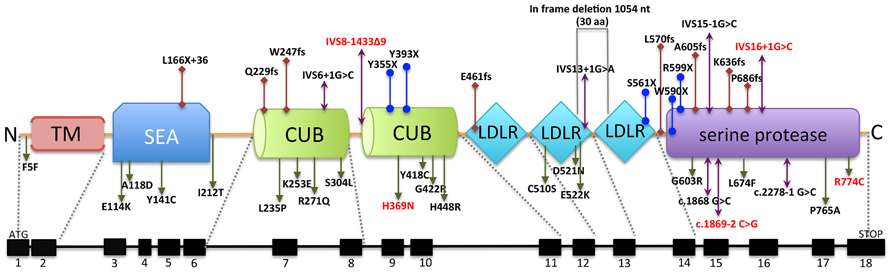
FIGURE 1. TMPRSS6 gene structure and schematic representation of corresponding matriptase-2 mutations reported in IRIDA patients. The genomic organization and the corresponding structural domains of matriptase-2 with currently identified mutations are shown. The missense, nonsense, frameshift and splice junction mutations are shown in green, blue, red and purple arrows, respectively. One in-frame deletion is boxed in gray. The mutations highlighted in red represent those appear to have haploinsufficiency.
Interestingly, in contrast to current understanding of autosomal recessive disorder, haploinsufficiency is observed in some TMPRSS6 mutations (Figure 1; Finberg et al., 2008; Pellegrino et al., 2012; Jaspers et al., 2013). Haploinsufficiency is also observed in animal models. Nai et al. (2010) reported that Tmprss6 heterozygous knockout mice are more susceptible to iron deficiency compared to their wild-type littermates. Finberg et al. (2011) also demonstrated that, compared to mice deficient for Hfe alone, heterozygous loss of Tmprss6 in Hfe knockout mice had higher hepcidin levels at 4 weeks of age, which presumably resulted in decreased hepatic iron concentrations at 8 weeks of age.
Human genome wide association studies (GWAS) highlighted the significance of matriptase-2 in control of iron homeostasis by identifying common TMPRSS6 variants associated with abnormal hematological parameters, including hemoglobin, transferrin saturation, erythrocyte mean cell volume (MCV) and serum iron concentrations (Benyamin et al., 2009; Chambers et al., 2009; Tanaka et al., 2010). Following GWAS, population-based cohort studies were investigated in China and Italy to study the association between serum iron parameters, iron-related diseases and specific TMPRSS6 single nucleotide polymorphisms (SNPs): rs855791 (V736A) and rs4820268 (D521D). It was found that TMPRSS6 SNPs was associated with lowered serum iron, hemoglobin, and plasma ferritin levels, consistent with lowered risk of iron overload and increased risk of iron deficiency anemia in Chinese population (An et al., 2012; Gan et al., 2012). A retrospective cohort study in northern Italy also suggested that TMPRSS6 V736A polymorphism is likely to be a gene modifier in hemochromatosis patients, influencing the susceptibility of cirrhosis (Valenti et al., 2012). Nai et al. (2011) demonstrated that TMPRSS6 V736A directly modulates HAMP expression in vitro and that healthy individuals with the homozygous substitution had lower levels of serum hepcidin, higher serum iron and higher transferrin saturation. Taken together, these studies clearly establish TMPRSS6/matriptase-2 as an important regulator of iron homeostasis in humans. A recent review focused more on the anemia induced by matriptase-2 mutations is complementary to the current review (De Falco et al., 2013).
Function of Matriptase-2 in Hepcidin Regulation
Matriptase-2 inhibition of hepcidin activation by cleaving membrane hemojuvelin has been established in vitro (Silvestri et al., 2008). When overexpressed in HeLa cells, matriptase-2 interacts and induces the cleavage of membrane hemojuvelin at the cell surface, resulting in the generation of soluble hemojuvelin that is released into the cell medium (Silvestri et al., 2008). However, in both mask and Tmprss6 knockout mice, hepatic hemojuvelin levels at the membrane were found unexpectedly to be decreased, compared to wild-type animals (Krijt et al., 2011; Frydlova et al., 2013). In addition, the levels of serum soluble hemojuvelin, which one would expect to be decreased in Tmprss6 knockout, did not differ from wild-type mice (Chen et al., 2013). Although the possibility that soluble hemojuvelin and fragments are rapidly degraded in vivo cannot be excluded, these data suggested that hemojuvelin may not be the endogenous substrate of matriptase-2 and that matriptase-2 functions in a more complicated way in vivo than by merely cleaving hemojuvelin to regulate hepcidin and iron.
Several studies have been conducted to study the role of matriptase-2, by crossing Tmprss6 knockout mice with several iron overload mouse models, including the generations of Hjv/Tmprss6, Bmp6/Tmprss6, Hfe/Tmprss6, and Tfr2/Tmprss6 double mutant mice (Truksa et al., 2009; Finberg et al., 2011; Lenoir et al., 2011; Lee et al., 2012). In mice lacking both Hjv and Tmprss6, Id1, a target gene of BMP6 signaling, and Hamp mRNA levels were low, whereas serum iron, transferrin saturation, and liver iron concentration were high, similar to phenotypes of mice deficient for Hjv alone (Truksa et al., 2009; Finberg et al., 2010). These results indicate that if the substrate of matriptase-2 is downstream of hemojuvelin, it is likely to be along the SMAD signaling pathway. It is known that inflammatory cytokines, such as LPS and IL6, can induce Hamp expression in the absence of Hjv (Niederkofler et al., 2005), presumably via the Stat3 and Stat5 pathways (Verga Falzacappa et al., 2007; Meynard et al., 2013). However, it was surprising to find that the lack of both Hjv and Tmprss6 in mice did not impair the responsiveness of hepcidin to BMP2 and IL6, but did fail to respond to iron challenge (Truksa et al., 2009). In mice deficient for both Bmp6 and Tmprss6, the levels of Hamp and Id1 mRNAs did not differ from mice deficient for Bmp6 alone; however, their plasma iron levels and hepatic iron stores were significantly lower, suggesting the loss of matriptase-2 ameliorates iron overload conditions in Bmp6 knockout mice (Lenoir et al., 2011). It is unclear why Bmp6/Tmprss6 mice had less iron loading compared to mice deficient for Bmp6 alone, but Hamp mRNA levels did not differ between Bmp6/Tmprss6 and Bmp6 knockout mice. Whether matriptase-2 has a significant role besides effects on BMP/SMAD signaling in iron metabolism, remain to be investigated.
Mice deficient for Hfe or Tfr2 alone also develop iron overload phenotypes with inappropriately low Hamp mRNA expression and high serum iron parameters, compared to wild-type animals (Ahmad et al., 2002; Wallace et al., 2005). It is suggested that Hfe competes with transferrin for binding to transferrin receptor-1 and thus inhibits Hamp expression (Giannetti and Bjorkman, 2004; Schmidt et al., 2008). Others also showed that Hfe knockout mice had high Bmp6 mRNA expression but inappropriately low Smad1/5/8 phosphorylation, suggesting Hfe facilitates signal transduction initiated by BMP6 (Corradini et al., 2009; Kautz et al., 2009). However, the underlying mechanisms of how Hfe and Tfr2 contribute in BMP/SMAD signaling pathway is unclear. Mice deficient for both Hfe or Tfr2 and Tmprss6, had high Hamp mRNA expression and exhibited iron deficiency microcytic anemia mimicking the phenotypes of mice lacking functional matriptase-2 alone (Finberg et al., 2011; Lee et al., 2012). This suggests that Hfe and Tfr2, if involved in BMP/SMAD pathway, are likely to be upstream of matriptase-2 signaling.
Regulation of Matriptase-2
Studies have shown that matriptase-2 expression can be modulated by iron status (Meynard et al., 2011; Zhang et al., 2011). In rats under acute iron deprivation, hepatic matriptase-2 protein levels are upregulated to repress hepcidin production (Zhang et al., 2011). Interestingly, matriptase-2 levels are also increased in response to chronic iron treatment and BMP6 administration in mice, possibly to prevent excessive hepcidin production, suggesting a dual role of matriptase-2 in the maintenance of tight systemic iron balance in response to iron (Meynard et al., 2011). In addition, studies also suggest that TMPRSS6 mRNA expression is suppressed by conditions of inflammation (Meynard et al., 2013) and is upregulated in hypoxia (Lakhal et al., 2011; Maurer et al., 2012) and by erythropoietin (Peng et al., 2010). Human hepatoma Hep3B cells treated with interleukin-6 and mice injected with lipopolysaccharide demonstrated a downregulation of TMPRSS6 via a decrease in Stat5 phosphorylation, independent of BMP/SMAD pathway (Meynard et al., 2013). Studies using Hep3B cells revealed that TMPRSS6 is upregulated by HIF-1α and HIF-2α. This upregulation resulted in a decrease in membrane hemojuvelin and thus reducing hepcidin production (Lakhal et al., 2011). In mice, Tmprss6 mRNA expression is induced by erythropoietin (Peng et al., 2010), which is also shown to be a negative regulator of hepcidin expression (Sasaki et al., 2012). Whether the downregulation of hepcidin by erythropoietin is dependent on Tmprss6 or through other unidentified mechanisms remains to be investigated.
Matriptase-2 as a Therapeutic Target
Genetic studies of mice deficient for both Tmprss6 and Hfe or Tfr2 or Hbbth3/+, the mouse model of β-thalassemia intermedia, have shown that iron overload can be prevented by targeting Tmprss6 (Finberg et al., 2011; Lee et al., 2012; Nai et al., 2012). It is believed that the therapeutic effect did not come from silencing Tmprss6 directly but from increased hepcidin production, resulting in lowered circulating iron burden (Camaschella, 2013). Studies targeting Tmprss6 in Hbbth3/+ and Hfe knockout mice by injecting silencing RNA (Schmidt et al., 2013) and anti-sense oligonucleotides (Guo et al., 2013) have successfully suppressed Tmprss6 mRNA expression, leading to elevated hepcidin levels, improved iron overload in Hfe knockout and anemia and β-thalassemic mice. It is unclear how the ineffective erythropoiesis is improved by dampening Tmprss6 expression in Hbbth3/+ mice. However, higher hepcidin level inhibiting iron delivery to the erythroid precursors seems to play a role as evident by the similar effects achieved by overexpression of Hamp, iron restriction, and the injection of transferrin to Hbbth3/+ mice (Gardenghi et al., 2010; Li et al., 2010; Finberg, 2013).
One limitation of using this method is that, unlike traditional phlebotomy and chelation therapies, iron is not removed or excreted from the body, and therefore, may not be an ideal treatment for patients with severe iron overload and transfusion-dependent thalassemia (Camaschella, 2013). It could, however, improve therapeutic efficacy when used in combination with other traditional therapies by preventing intestinal iron absorption. A key issue for the use of RNA interference for clinical applications is the delivery method. There are safety concerns with viral vectors and non-viral delivery methods, which are still in their early development stage. Concerns have also been raised regarding the potential for off-target effects of siRNAs and their possible induction of interferon-stimulated genes. Other novel inhibitors of TMPRSS6, such as small molecule inhibitors, once identified, may eventually become useful therapeutic agents as well.
Role of Matriptase-2 in Cancer
Numerous members of the type II transmembrane serine protease family have been associated with a variety of different human cancers due to the differential expression patterns observed in these proteases between normal and cancerous tissues and cells (Webb et al., 2011). However, there are only a limited number of studies examining the involvement of matriptase-2 in human cancer, including breast cancer (Hartikainen et al., 2006; Parr et al., 2007; Tuhkanen et al., 2013) and prostate cancer (Sanders et al., 2008; Webb et al., 2012).
The association between matriptase-2 and breast cancer was established by a case control study in eastern Finnish population where they found a SNP (rs733655) in TMPRSS6 gene associated with increased breast cancer risk (Hartikainen et al., 2006). It was later shown that TMPRSS6 mRNA expression inhibits breast tumor development and thus correlates with favorable prognostic outcome in patients (Parr et al., 2007). Recently, Tuhkanen et al. (2013) also demonstrated the association of several TMPRSS6 variants with breast cancer risk and survival. It was highlighted that matriptase-2 protein levels decrease with tumor progression, and lower gene expression is seen in poor-prognosis-related triple-negative breast cancers (Tuhkanen et al., 2013). Mastriptase-2 is also implicated in tumor invasion and metastasis in prostate cancer in vitro (Sanders et al., 2008; Webb et al., 2012). These results indicate the involvement of matriptase-2 in tumor development. However, it is not clear whether the role of TMPRSS6 in cancer progression is due to its ability to cleave extracellular matrix component such as fibronectin or due to a modification of iron parameters in cancer cells.
TMPRSS6 expression is predominantly found in low invasive breast cancer cell lines such as MCF-7 and is absent in more invasive breast cancer cell lines such as MDA-MB-231 (Parr et al., 2007). Overexpression of matriptase-2 in MDA-MB-231 leads to a reduction of invasiveness and motility of the transfected cells and suppresses their tumorigenesis when xenografted in athymic nude mice suggesting that matriptase-2 could be involved in cancer progression through its capacity to cleave extracellular matrix components (Parr et al., 2007). However, variations of the iron status and iron regulatory genes expression were not addressed in the transfected cells in this study.
Many cancers exhibit an increased requirement for iron, presumably because of the need for iron as a cofactor in proteins essential to sustain growth and proliferation. The iron exporter ferroportin is expressed in breast cancer cells. Pinnix et al. (2010) showed that cells with high hepcidin and low ferroportin levels tended to be more aggressive. They concluded that having a breast cancer with low hepcidin and high ferroportin levels is an independent predictor of prognosis for a >90% 10-year survival rate (Pinnix et al., 2010), however, the mechanism is still to be investigated. Further studies are required to clarify the role of matriptase-2 in cancer progression.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
This study was supported by National Institutes of Health, National Institute of Diabetes and Digestive, and Kidney Diseases grants R01 DK069533 and R01 DK071837 (Herbert Y. Lin).
References
Ahmad, K. A., Ahmann, J. R., Migas, M. C., Waheed, A., Britton, R. S., Bacon, B. R., et al. (2002). Decreased liver hepcidin expression in the Hfe knockout mouse. Blood Cells Mol. Dis. 29, 361–366. doi: 10.1006/bcmd.2002.0575
Altamura, S., D’Alessio, F., Selle, B., and Muckenthaler, M. U. (2010). A novel TMPRSS6 mutation that prevents protease auto-activation causes IRIDA. Biochem. J. 431, 363–371. doi: 10.1042/BJ20100668
An, P., Wu, Q., Wang, H., Guan, Y., Mu, M., Liao, Y., et al. (2012). TMPRSS6, but not TF, TFR2 or BMP2 variants are associated with increased risk of iron-deficiency anemia. Hum. Mol. Genet. 21, 2124–2131. doi: 10.1093/hmg/dds028
Andriopoulos, B. Jr., Corradini, E., Xia, Y., Faasse, S. A., Chen, S., Grgurevic, L., et al. (2009). BMP6 is a key endogenous regulator of hepcidin expression and iron metabolism. Nat. Genet. 41, 482–487. doi: 10.1038/ng.335
Babitt, J. L., Huang, F. W., Wrighting, D. M., Xia, Y., Sidis, Y., Samad, T. A., et al. (2006). Bone morphogenetic protein signaling by hemojuvelin regulates hepcidin expression. Nat. Genet. 38, 531–539. doi: 10.1038/ng1777
Babitt, J. L., Huang, F. W., Xia, Y., Sidis, Y., Andrews, N. C., and Lin, H. Y. (2007). Modulation of bone morphogenetic protein signaling in vivo regulates systemic iron balance. J. Clin. Invest. 117, 1933–1939. doi: 10.1172/JCI31342
Benyamin, B., Ferreira, M. A., Willemsen, G., Gordon, S., Middelberg, R. P., Mcevoy, B. P., et al. (2009). Common variants in TMPRSS6 are associated with iron status and erythrocyte volume. Nat. Genet. 41, 1173–1175. doi: 10.1038/ng.456
Camaschella, C. (2013). Treating iron overload. N. Engl. J. Med. 368, 2325–2327. doi: 10.1056/NEJMcibr1304338
Chambers, J. C., Zhang, W., Li, Y., Sehmi, J., Wass, M. N., Zabaneh, D., et al. (2009). Genome-wide association study identifies variants in TMPRSS6 associated with hemoglobin levels. Nat. Genet. 41, 1170–1172. doi: 10.1038/ng.462
Chen, W., Sun, C. C., Chen, S., Meynard, D., Babitt, J. L., and Lin, H. Y. (2013). A novel validated enzyme-linked immunosorbent assay to quantify soluble hemojuvelin in mouse serum. Haematologica 98, 296–304. doi: 10.3324/haematol.2012.070136
Corradini, E., Garuti, C., Montosi, G., Ventura, P., Andriopoulos, B. Jr., Lin, H. Y., et al. (2009). Bone morphogenetic protein signaling is impaired in an HFE knockout mouse model of hemochromatosis. Gastroenterology 137, 1489–1497. doi: 10.1053/j.gastro.2009.06.057
De Falco, L., Sanchez, M., Silvestri, L., Kannengiesser, C., Muckenthaler, M. U., Iolascon, A., et al. (2013). Iron refractory iron deficiency anemia. Haematologica 98, 845–853. doi: 10.3324/haematol.2012.075515
Du, X., She, E., Gelbart, T., Truksa, J., Lee, P., Xia, Y., et al. (2008). The serine protease TMPRSS6 is required to sense iron deficiency. Science 320, 1088–1092. doi: 10.1126/science.1157121
Finberg, K. E. (2013). Striking the target in iron overload disorders. J. Clin. Invest. 123, 1424–1427. doi: 10.1172/JCI68889
Finberg, K. E., Heeney, M. M., Campagna, D. R., Aydinok, Y., Pearson, H. A., Hartman, K. R., et al. (2008). Mutations in TMPRSS6 cause iron-refractory iron deficiency anemia (IRIDA). Nat. Genet. 40, 569–571. doi: 10.1038/ng.130
Finberg, K. E., Whittlesey, R. L., and Andrews, N. C. (2011). Tmprss6 is a genetic modifier of the Hfe-hemochromatosis phenotype in mice. Blood 117, 4590–4599. doi: 10.1182/blood-2010-10.315507
Finberg, K. E., Whittlesey, R. L., Fleming, M. D., and Andrews, N. C. (2010). Down-regulation of Bmp/Smad signaling by Tmprss6 is required for maintenance of systemic iron homeostasis. Blood 115, 3817–3826. doi: 10.1182/blood-2009-05-224808
Folgueras, A. R., De Lara, F. M., Pendas, A. M., Garabaya, C., Rodriguez, F., Astudillo, A., et al. (2008). Membrane-bound serine protease matriptase-2 (Tmprss6) is an essential regulator of iron homeostasis. Blood 112, 2539–2545. doi: 10.1182/blood-2008-04-149773
Frydlova, J., Fujikura, Y., Vokurka, M., Necas, E., and Krijt, J. (2013). Decreased hemojuvelin protein levels in mask mice lacking matriptase-2-dependent proteolytic activity. Physiol. Res. 62, 405–411.
Gan, W., Guan, Y., Wu, Q., An, P., Zhu, J., Lu, L., et al. (2012). Association of TMPRSS6 polymorphisms with ferritin, hemoglobin, and type 2 diabetes risk in a Chinese Han population. Am. J. Clin. Nutr. 95, 626–632. doi: 10.3945/ajcn.111.025684
Gardenghi, S., Ramos, P., Marongiu, M. F., Melchiori, L., Breda, L., Guy, E., et al. (2010). Hepcidin as a therapeutic tool to limit iron overload and improve anemia in beta-thalassemic mice. J. Clin. Invest. 120, 4466–4477. doi: 10.1172/JCI41717
Giannetti, A. M., and Bjorkman, P. J. (2004). HFE and transferrin directly compete for transferrin receptor in solution and at the cell surface. J. Biol. Chem. 279, 25866–25875. doi: 10.1074/jbc.M401467200
Guillem, F., Lawson, S., Kannengiesser, C., Westerman, M., Beaumont, C., and Grandchamp, B. (2008). Two nonsense mutations in the TMPRSS6 gene in a patient with microcytic anemia and iron deficiency. Blood 112, 2089–2091. doi: 10.1182/blood-2008-05-154740
Guo, S., Casu, C., Gardenghi, S., Booten, S., Aghajan, M., Peralta, R., et al. (2013). Reducing TMPRSS6 ameliorates hemochromatosis and beta-thalassemia in mice. J. Clin. Invest. 123, 1531–1541. doi: 10.1172/JCI66969
Hartikainen, J. M., Tuhkanen, H., Kataja, V., Eskelinen, M., Uusitupa, M., Kosma, V. M., et al. (2006). Refinement of the 22q12-q13 breast cancer - associated region: evidence of TMPRSS6 as a candidate gene in an eastern Finnish population. Clin. Cancer Res. 12, 1454–1462. doi: 10.1158/1078-0432.CCR-05-1417
Inouye, K., Tsuzuki, S., Yasumoto, M., Kojima, K., Mochida, S., and Fushiki, T. (2010). Identification of the matriptase second CUB domain as the secondary site for interaction with hepatocyte growth factor activator inhibitor type-1. J. Biol. Chem. 285, 33394–33403. doi: 10.1074/jbc.M110.115816
Jaspers, A., Caers, J., Le Gac, G., Ferec, C., Beguin, Y., and Fillet, G. (2013). A novel mutation in the CUB sequence of matriptase-2 (TMPRSS6) is implicated in iron-resistant iron deficiency anaemia (IRIDA). Br. J. Haematol. 160, 564–565. doi: 10.1111/bjh.12147
Kang, J. Y., Dolled-Filhart, M., Ocal, I. T., Singh, B., Lin, C. Y., Dickson, R. B., et al. (2003). Tissue microarray analysis of hepatocyte growth factor/Met pathway components reveals a role for Met, matriptase, and hepatocyte growth factor activator inhibitor 1 in the progression of node-negative breast cancer. Cancer Res. 63, 1101–1105.
Kautz, L., Meynard, D., Besson-Fournier, C., Darnaud, V., Al Saati, T., Coppin, H., et al. (2009). BMP/Smad signaling is not enhanced in Hfe-deficient mice despite increased Bmp6 expression. Blood 114, 2515–2520. doi: 10.1182/blood-2009-02–206771
Kautz, L., Meynard, D., Monnier, A., Darnaud, V., Bouvet, R., Wang, R. H., et al. (2008). Iron regulates phosphorylation of Smad1/5/8 and gene expression of Bmp6, Smad7, Id1, and Atoh8 in the mouse liver. Blood 112, 1503–1509. doi: 10.1182/blood-2008-03-143354
Krijt, J., Fujikura, Y., Ramsay, A. J., Velasco, G., and Necas, E. (2011). Liver hemojuvelin protein levels in mice deficient in matriptase-2 (Tmprss6). Blood Cells Mol. Dis. 47, 133–137. doi: 10.1016/j.bcmd.2011.04.009
Lakhal, S., Schodel, J., Townsend, A. R., Pugh, C. W., Ratcliffe, P. J., and Mole, D. R. (2011). Regulation of type II transmembrane serine proteinase TMPRSS6 by hypoxia-inducible factors: new link between hypoxia signaling and iron homeostasis. J. Biol. Chem. 286, 4090–4097. doi: 10.1074/jbc.M110.173096
Lee, P., Hsu, M. H., Welser-Alves, J., and Peng, H. (2012). Severe microcytic anemia but increased erythropoiesis in mice lacking Hfe or Tfr2 and Tmprss6. Blood Cells Mol. Dis. 48, 173–178. doi: 10.1016/j.bcmd.2011.12.005
Lenoir, A., Deschemin, J. C., Kautz, L., Ramsay, A. J., Roth, M. P., Lopez-Otin, C., et al. (2011). Iron-deficiency anemia from matriptase-2 inactivation is dependent on the presence of functional Bmp6. Blood 117, 647–650. doi: 10.1182/blood-2010-07-295147
Li, H., Rybicki, A. C., Suzuka, S. M., Von Bonsdorff, L., Breuer, W., Hall, C. B., et al. (2010). Transferrin therapy ameliorates disease in beta-thalassemic mice. Nat. Med. 16, 177–182. doi: 10.1038/nm.2073
Maurer, E., Gutschow, M., and Stirnberg, M. (2012). Matriptase-2 (TMPRSS6) is directly up-regulated by hypoxia inducible factor-1: identification of a hypoxia-responsive element in the TMPRSS6 promoter region. Biol. Chem. 393, 535–540. doi: 10.1515/hsz-2011-0221
Maurer, E., Gutschow, M., and Stirnberg, M. (2013). Hepatocyte growth factor activator inhibitor type 2 (HAI-2) modulates hepcidin expression by inhibiting the cell surface protease matriptase-2. Biochem. J. 450, 583–593. doi: 10.1042/BJ20121518
Melis, M. A., Cau, M., Congiu, R., Sole, G., Barella, S., Cao, A., et al. (2008). A mutation in the TMPRSS6 gene, encoding a transmembrane serine protease that suppresses hepcidin production, in familial iron deficiency anemia refractory to oral iron. Haematologica 93, 1473–1479. doi: 10.3324/haematol.13342
Meynard, D., Kautz, L., Darnaud, V., Canonne-Hergaux, F., Coppin, H., and Roth, M. P. (2009). Lack of the bone morphogenetic protein BMP6 induces massive iron overload. Nat. Genet. 41, 478–481. doi: 10.1038/ng.320
Meynard, D., Sun, C. C., Wu, Q., Chen, W., Chen, S., Nelson, C. N., et al. (2013). Inflammation Regulates TMPRSS6 Expression via STAT5. PLoS ONE 8:e82127. doi: 10.1371/journal.pone.0082127
Meynard, D., Vaja, V., Sun, C. C., Corradini, E., Chen, S., Lopez-Otin, C., et al. (2011). Regulation of TMPRSS6 by BMP6 and iron in human cells and mice. Blood 118, 747–756. doi: 10.1182/blood-2011-04-348698
Nai, A., Pagani, A., Mandelli, G., Lidonnici, M. R., Silvestri, L., Ferrari, G., et al. (2012). Deletion of TMPRSS6 attenuates the phenotype in a mouse model of beta-thalassemia. Blood 119, 5021–5029. doi: 10.1182/blood-2012-01-401885
Nai, A., Pagani, A., Silvestri, L., and Camaschella, C. (2010). Increased susceptibility to iron deficiency of Tmprss6-haploinsufficient mice. Blood 116, 851–852. doi: 10.1182/blood-2010-04-278655
Nai, A., Pagani, A., Silvestri, L., Campostrini, N., Corbella, M., Girelli, D., et al. (2011). TMPRSS6 rs855791 modulates hepcidin transcription in vitro and serum hepcidin levels in normal individuals. Blood 118, 4459–4462. doi: 10.1182/blood-2011-06-364034
Nemeth, E., Tuttle, M. S., Powelson, J., Vaughn, M. B., Donovan, A., Ward, D. M., et al. (2004). Hepcidin regulates cellular iron efflux by binding to ferroportin and inducing its internalization. Science 306, 2090–2093. doi: 10.1126/science.1104742
Nicolas, G., Bennoun, M., Devaux, I., Beaumont, C., Grandchamp, B., Kahn, A., et al. (2001). Lack of hepcidin gene expression and severe tissue iron overload in upstream stimulatory factor 2 (USF2) knockout mice. Proc. Natl. Acad. Sci. U.S.A. 98, 8780–8785. doi: 10.1073/pnas.151179498151179498
Niederkofler, V., Salie, R., and Arber, S. (2005). Hemojuvelin is essential for dietary iron sensing, and its mutation leads to severe iron overload. J. Clin. Invest. 115, 2180–2186. doi: 10.1172/JCI25683
Oberst, M., Anders, J., Xie, B., Singh, B., Ossandon, M., Johnson, M., et al. (2001). Matriptase and HAI-1 are expressed by normal and malignant epithelial cells in vitro and in vivo. Am. J. Pathol. 158, 1301–1311. doi: 10.1016/S0002-9440(10)64081-3
Parr, C., Sanders, A. J., Davies, G., Martin, T., Lane, J., Mason, M. D., et al. (2007). Matriptase-2 inhibits breast tumor growth and invasion and correlates with favorable prognosis for breast cancer patients. Clin. Cancer Res. 13, 3568–3576. doi: 10.1158/1078-0432.CCR-06-2357
Pellegrino, R. M., Coutinho, M., D’Ascola, D., Lopes, A. M., Palmieri, A., Carnuccio, F., et al. (2012). Two novel mutations in the tmprss6 gene associated with iron-refractory iron-deficiency anaemia (irida) and partial expression in the heterozygous form. Br. J. Haematol. 158, 668–672. doi: 10.1111/j.1365-2141.2012.09198.x
Peng, H., Truksa, J., and Lee, P. (2010). EPO-mediated reduction in Hamp expression in vivo corrects iron deficiency anaemia in TMPRSS6 deficiency. Br. J. Haematol. 151, 106–109. doi: 10.1111/j.1365-2141.2010.08306.x
Pinnix, Z. K., Miller, L. D., Wang, W., D’Agostino, R. Jr., Kute, T., Willingham, M. C., et al. (2010). Ferroportin and iron regulation in breast cancer progression and prognosis. Sci. Transl. Med. 2:43ra56. doi: 10.1126/scisignal.3001127
Ramsay, A. J., Hooper, J. D., Folgueras, A. R., Velasco, G., and Lopez-Otin, C. (2009a). Matriptase-2 (TMPRSS6): a proteolytic regulator of iron homeostasis. Haematologica 94, 840–849. doi: 10.3324/haematol.2008.001867
Ramsay, A. J., Quesada, V., Sanchez, M., Garabaya, C., Sarda, M. P., Baiget, M., et al. (2009b). Matriptase-2 mutations in iron-refractory iron deficiency anemia patients provide new insights into protease activation mechanisms. Hum. Mol. Genet. 18, 3673–3683. doi: 10.1093/hmg/ddp315
Ramsay, A. J., Reid, J. C., Velasco, G., Quigley, J. P., and Hooper, J. D. (2008). The type II transmembrane serine protease matriptase-2–identification, structural features, enzymology, expression pattern and potential roles. Front. Biosci. 13:569–579. doi: 10.2741/2702
Riddick, A. C., Shukla, C. J., Pennington, C. J., Bass, R., Nuttall, R. K., Hogan, A., et al. (2005). Identification of degradome components associated with prostate cancer progression by expression analysis of human prostatic tissues. Br. J. Cancer 92, 2171–2180. doi: 10.1038/sj.bjc.6602630
Sanders, A. J., Parr, C., Martin, T. A., Lane, J., Mason, M. D., and Jiang, W. G. (2008). Genetic upregulation of matriptase-2 reduces the aggressiveness of prostate cancer cells in vitro and in vivo and affects FAK and paxillin localisation. J. Cell. Physiol. 216, 780–789. doi: 10.1002/jcp.21460
Sanders, A. J., Webb, S. L., Parr, C., Mason, M. D., and Jiang, W. G. (2010). The type II transmembrane serine protease, matriptase-2: possible links to cancer? Anticancer Agents Med. Chem. 10, 64–69. doi: 10.2174/1871520611009010064
Sasaki, Y., Noguchi-Sasaki, M., Yasuno, H., Yorozu, K., and Shimonaka, Y. (2012). Erythropoietin stimulation decreases hepcidin expression through hematopoietic activity on bone marrow cells in mice. Int. J. Hematol. 96, 692–700. doi: 10.1007/s12185-012-1217-4
Schmidt, P. J., Toran, P. T., Giannetti, A. M., Bjorkman, P. J., and Andrews, N. C. (2008). The transferrin receptor modulates Hfe-dependent regulation of hepcidin expression. Cell Metab. 7, 205–214. doi: 10.1016/j.cmet.2007.11.016
Schmidt, P. J., Toudjarska, I., Sendamarai, A. K., Racie, T., Milstein, S., Bettencourt, B. R., et al. (2013). An RNAi therapeutic targeting Tmprss6 decreases iron overload in Hfe(-/-) mice and ameliorates anemia and iron overload in murine beta-thalassemia intermedia. Blood 121, 1200–1208. doi: 10.1182/blood-2012-09-453977
Silvestri, L., Pagani, A., Nai, A., De Domenico, I., Kaplan, J., and Camaschella, C. (2008). The serine protease matriptase-2 (TMPRSS6) inhibits hepcidin activation by cleaving membrane hemojuvelin. Cell Metab. 8, 502–511. doi: 10.1016/j.cmet.2008.09.012
Szabo, R., Hobson, J. P., List, K., Molinolo, A., Lin, C. Y., and Bugge, T. H. (2008). Potent inhibition and global co-localization implicate the transmembrane Kunitz-type serine protease inhibitor hepatocyte growth factor activator inhibitor-2 in the regulation of epithelial matriptase activity. J. Biol. Chem. 283, 29495–29504. doi: 10.1074/jbc.M801970200
Tanaka, T., Roy, C. N., Yao, W., Matteini, A., Semba, R. D., Arking, D., et al. (2010). A genome-wide association analysis of serum iron concentrations. Blood 115, 94–96. doi: 10.1182/blood-2009-07-232496
Truksa, J., Gelbart, T., Peng, H., Beutler, E., Beutler, B., and Lee, P. (2009). Suppression of the hepcidin-encoding gene Hamp permits iron overload in mice lacking both hemojuvelin and matriptase-2/TMPRSS6. Br. J. Haematol. 147, 571–581. doi: 10.1111/j.1365-2141.2009.07873.x
Tuhkanen, H., Hartikainen, J. M., Soini, Y., Velasco, G., Sironen, R., Nykopp, T. K., et al. (2013). Matriptase-2 gene (TMPRSS6) variants associate with breast cancer survival, and reduced expression is related to triple-negative breast cancer. Int. J. Cancer 133, 2334–2340. doi: 10.1002/ijc.28254
Valenti, L., Fracanzani, A. L., Rametta, R., Fraquelli, M., Soverini, G., Pelusi, S., et al. (2012). Effect of the A736V TMPRSS6 polymorphism on the penetrance and clinical expression of hereditary hemochromatosis. J. Hepatol. 57, 1319–1325. doi: 10.1016/j.jhep.2012.07.041
Velasco, G., Cal, S., Quesada, V., Sanchez, L. M., and Lopez-Otin, C. (2002). Matriptase-2, a membrane-bound mosaic serine proteinase predominantly expressed in human liver and showing degrading activity against extracellular matrix proteins. J. Biol. Chem. 277, 37637–37646. doi: 10.1074/jbc.M203007200
Verga Falzacappa, M. V., Vujic Spasic, M., Kessler, R., Stolte, J., Hentze, M. W., and Muckenthaler, M. U. (2007). STAT3 mediates hepatic hepcidin expression and its inflammatory stimulation. Blood 109, 353–358. doi: 10.1182/blood-2006-07-033969
Wallace, D. F., Summerville, L., Lusby, P. E., and Subramaniam, V. N. (2005). First phenotypic description of transferrin receptor 2 knockout mouse, and the role of hepcidin. Gut 54, 980–986. doi: 10.1136/gut.2004.062018
Wang, R. H., Li, C., Xu, X., Zheng, Y., Xiao, C., Zerfas, P., et al. (2005). A role of SMAD4 in iron metabolism through the positive regulation of hepcidin expression. Cell Metab. 2, 399–409. doi: 10.1016/j.cmet.2005.10.010
Webb, S. L., Sanders, A. J., Mason, M. D., and Jiang, W. G. (2011). Type II transmembrane serine protease (TTSP) deregulation in cancer. Front. Biosci. (Landmark Ed.) 16:539–552.
Webb, S. L., Sanders, A. J., Mason, M. D., and Jiang, W. G. (2012). The influence of matriptase-2 on prostate cancer in vitro: a possible role for beta-catenin. Oncol. Rep. 28, 1491–1497. doi: 10.3892/or.2012.1945
Keywords: iron, TMPRSS6, matriptase-2, iron overload, IRIDA
Citation: Wang C-Y, Meynard D and Lin HY (2014) The role of TMPRSS6/matriptase-2 in iron regulation and anemia. Front. Pharmacol. 5:114. doi: 10.3389/fphar.2014.00114
Received: 17 March 2014; Paper pending published: 07 April 2014;
Accepted: 29 April 2014; Published online: 19 May 2014.
Edited by:
Raffaella Gozzelino, Instituto Gulbenkian de Ciência, PortugalReviewed by:
Clara Camaschella, Vita Salute San Raffaele University and San Raffaele Scientific Institute, ItalyCarole Beaumont, Institut National de la Santé et de la Recherche Médicale, France
Copyright © 2014 Wang, Meynard and Lin. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Herbert Y. Lin, Program in Anemia Signaling Research, Division of Nephrology, Program in Membrane Biology, Center for Systems Biology, Massachusetts General Hospital, Harvard Medical School, 185 Cambridge Street, CPZN-8216, Boston, MA 02114, USA e-mail:bGluLmhlcmJlcnRAbWdoLmhhcnZhcmQuZWR1
 Chia-Yu Wang
Chia-Yu Wang Delphine Meynard2
Delphine Meynard2 Herbert Y. Lin
Herbert Y. Lin