- 1Department of Pharmacy, Stamford University Bangladesh, Dhaka, Bangladesh
- 2Department of Pharmacy, Primeasia University, Dhaka, Bangladesh
Fruits of Phoenix sylvestris Roxb. (Arecaceae) are used to treat back pain, toothache, headache, arthritis, nervous debility and as sedative. The aim of this study was to evaluate the antinociceptive and neuropharmacological activities of methanol extract of P. sylvestris fruit pulp (MEPS). The antinociceptive activity of MEPS was evaluated by heat-induced (hot plate, tail immersion test) and chemical-induced pain models (acetic acid-induced writhing, formalin-induced nociception, glutamate-induced nociception and paw edema test). The effect of MEPS on central nervous system (CNS) was studied using hole cross test, open field test, sodium thiopental-induced sleeping time and elevated plus maze test. MEPS showed strong, significant and dose-dependent antinociceptive activity in all heat-induced and chemical-induced pain models at all experimental doses. Involvement of opioid receptor mediated analgesia was evident from the reversal of analgesic effect by naloxone. MEPS also showed reduced locomotor activity in both hole cross and open field tests. The increase in sleeping time in sodium thiopental-induced sleeping test and anxiolytic activity in elevated plus maze test were also significant. So, it is evident that MEPS possesses strong central and peripheral antinociceptive activity as well as CNS depressant, sedative and anxiolytic activity. The results justify the ethnomedicinal use of P. sylvestris fruit in different painful conditions and CNS disorders.
Introduction
Investigation of pharmacological basis of the therapeutic uses of plants by traditional practitioners is an integral part of ethnopharmacology (Leonti and Casu, 2013). Ethnopharmacological knowledge continues to contribute to the discovery of new antinociceptive agents from plants (Calixto et al., 2000). Phytotherapy based on this knowledge is also being used as a guide for the development of CNS depressant, sedative, anxiolytic drugs (Gurib-Fakim, 2006).
Phoenix sylvestris Roxb. (Arecaceae), locally known as Khejur, is a palm tree cultivated for its syrupy juice and edible fruit in Bangladesh. Fruits of the plant are used to treat back pain, stomachache, toothache, headache, arthritis, pain of buttocks, fever, piles, nervous debility, and as nervine tonic, restorative, sedative in ethnomedicine (Ghani, 2003; Acharya and Pokhrel, 2006; Harney, 2013; Manikandan, 2013). Besides, fruits are used in cough, diarrhea, dysentery, opthalmia, opacity of cornea, and toothworm (Ghani, 2003; Ahmad et al., 2008; Rajkumari et al., 2013; Murthy and Madhav, 2015). Fruits are also employed as aphrodisiac, cooling and gastric stimulant in traditional medicine (Goswami et al., 2012; Sambandan and Dhatchanamoorthy, 2012). Amino acids (mainly alanine), vitamins A, B, and D, sugars, tannins, mucilage and ascorbic acid have been found in fruits (Ghani, 2003). Proanthocyanidin, a polyphenolic substance belonging to the flavonoid group, has been isolated from the fruits of P. sylvestris (Rao et al., 1980). The antioxidant activity of fruits has been reported (Mukherjee et al., 2014). Aqueous extract of the fruits has been found to inhib the angiotensin I-converting enzyme (ACE), α-glucosidase and α-amylase (Das et al., 2012; Das and De, 2013). The erythropoietic activity of the fruits has also been reported (Lohar et al., 2009).
Ethnomedicinal reports on P. sylvestris fruits demonstrate its extensive application in different painful conditions and neurological disorders. However, there is lack of scientific study regarding antinociceptive as well as neuropharmacological activities of P. sylvestris fruits. Therefore, we aimed to investigate the central and peripheral antinociceptive activities of P. sylvestris fruit pulp using heat-induced (hot plate and tail immersion test) and chemical-induced pain models (acetic acid-induced writhing, formalin induced-licking and glutamate-induced paw licking test). For the evaluation of neuropharmacological activities we studied locomotor activity by open field and hole cross method, sedative activity by sodium thiopental-induced sleeping time test and anxiolytic activity by elevated plus maze test. In addition, we have also reported the bioactive compounds by gas chromatography-mass spectroscopy (GC-MS) analysis of P. sylvestris fruit pulp.
Materials and Methods
Collection, Identification and Extraction of Plant Material
Mature but unripe fruits of P. sylvestris were collected from Akabpur, Mainamati, Comilla, Bangladesh in July 23, 2013. Fruits were identified by Sarder Nasir Uddin, Senior Scientific Officer, Bangladesh National Herbarium, Mirpur, Dhaka, Bangladesh. A voucher specimen (DACB: 38499) has been deposited in the herbarium for further reference. The pulp of the fruits was separated from pit, dried and grounded. The powdered pulp (230 g) was macerated using 1000 mL of methanol with occasional stirring at 25 ± 2°C for 7 days. The extract was then filtered using sterilized cotton filter and Buchner funnel. A rotary evaporator was used to remove the solvent at 40° C and 50 r.p.m. under reduced pressure. After removal of solvent, 30.19 g (yield 13.13 %) dried extract was obtained and this crude extract was used for the experiments.
Ethical Statements
Ethical Principles and Guidelines of Scientific Experiments on Animals (1995) formulated by The Swiss Academy of Medical sciences and the Swiss Academy of Sciences were followed for the care and treatment of the animals. All the procedures and protocols were approved by the Ethics Committee of Stamford University Bangladesh (SUB/IAEC/13.02). The animals were euthanized using pentobarbital in accordance with AVMA guidelines for the Euthanasia of Animals: 2013 edition and sufficient measures were taken to minimize suffering prior to, during and after the experiments.
Animals
Swiss albino mice were collected from Animal Resources Branch of International Centre for Diarrhoeal Disease Research, Bangladesh (icddr,b). The animals were housed in 120 × 30 × 30 cm cages and flake wood shavings were provided for bedding. The health status of mice was checked by daily monitoring during acclimatization period and before and after experiments. The animals were provided with standard diet and tap water ad libitum. For the period of 14 days animals were acclimatized in the laboratory environment before experiments. The animals were fasted overnight only before experiments. The standard laboratory conditions (relative humidity 55–60%; room temperature 25 ± 2°C; 12 h light/dark cycle with lights on at 7:00 a.m. and off at 7:00 p.m.) were maintained. Animals weighing between 25–30 g and 8–10 weeks aged were used for the experiments. All the experiments were performed between 9:00 a.m. and 5.00 p.m.
Drugs and Chemicals Used in the Experiments
The drugs and chemicals used in this study are: diclofenac sodium (Novartis Bangladesh Ltd.), morphine sulfate, sodium thiopental (Gonoshasthaya Pharmaceuticals Ltd.), diazepam (Square Pharmaceuticals Ltd.), naloxone hydrochloride (Samarth Life Sciences Pvt. Ltd., India), methanol, acetic acid, formalin, L-glutamic acid (Merck, Germany).
Standard Drugs and Treatments
Physiological saline (0.9% sodium chloride) was used for the preparation of all the standard drugs and MEPS doses. It was used as vehicle at the dose of 10 mL/kg for the control groups. The doses (50, 150, 300, and 450 mg/kg body weight) of MEPS were selected on the basis of the effects of the trial dose and previously reported effective analgesic doses of the methanol extract of P. sylvestris (Howlader et al., 2006). The vehicle and MEPS were administered per oral (p.o) 30 min before the experiments. Morphine sulfate and diclofenac sodium were used as standard drug at the dose of 5 mg/kg and 10 mg/kg respectively in antinociceptive tests. Both of them were administered intraperitoneally (i.p.) 15 min before the experiments. In hot plate and tail immersion test naloxone was employed (i.p.) 15 min before the administration of morphine sulfate or MEPS, at the dose of 2 mg/kg to evaluate opioid mediated antinociceptive activity. Diazepam was used as standard drug in open field, hole cross, sodium thiopental-induced sleeping time and elevated plus maze test at the dose of 1 mg/kg (i.p.). In sodium thiopental-induced sleeping time and elevated plus maze test diazepam was administered 30 min prior to starting experiments. Sodium thiopental was administered (i.p.) at the dose of 40 mg/kg.
Preliminary Phytochemical Screening
The crude extract of P. sylvestris fruits was qualitatively tested for phythochemicals using following reagents and chemicals: Carbohydrates with Molisch's and Fehling's reagents, reducing sugars with Benedict's reagent, glycosides with aqueous NaOH, glucosides with Fehling's reagent and H2SO4, resins with acetic anhydride and H2SO4, tannins with Ferric chloride and Lead acetate, proteins with NaOH and Cu2SO4, saponins with stable foam producing ability, flavonoids with the use of Zn and HCl, alkaloids with Mayer's, Hager's, Wagner's and Dragendroff's reagents and steroids with Libermann-Burchard's reagent. The change of color in respective tests was observed according to the standard procedures to detect the chemical constituents (Ghani, 2005).
GC-MS (Gas Chromatography-mass Spectroscopy) Analysis
The GC-MS analysis was performed using Agilent Technologies 7890A capillary gas chromatograph, directly coupled to a mass spectrometer system (Model: 5975C inert XL EI/CI MSD with triple axis detector). A fused silica capillary column of 5% phenyl, 95% dimethyl-poly-silloxane (HP-5MSI; length: 90 m, diameter: 0.250 mm and film: 0.25 μm) was used. The GC parameter was set as follows: the inlet temperature was set at 250°C and oven temperature was programmed as 90°C for 0 min, then 3°C/min to 200°C for 2 min and then 15°C/min to 280°C for 2 min. Total run time was 46 min and column flow rate was 1.1 mL/min Helium gas. The auxiliary (GC to MS interface) temperature was set to 280°C. The MS parameter was set as the MS was in scan mode. The ionization mode was EI (electron ionization) type. The mass range was set in the range of 50–550 m/z. MS quad temperature and source temperature was set at 150°C and 230°C respectively. Each component were searched and identified by using “NIST-MS Library 2009.” Peak area of the total ionic chromatogram (TIC) was used to determine the relative percentage amounts of separated compounds and calculation were done automatically.
Acute Toxicity Test
The acute toxicity of MEPS was observed in seven experimental groups. Each group contained five animals (n = 5). The experimental groups received MEPS at the dose of 500, 1000, 2000, 3000, 4000, 5000, and 6000 mg/kg body weight (p.o). Each group of animals were placed in separate cages and allowed for free access of water ad libitum and food. The animals were observed for the next 72 h to find any mortality, adverse reactions like skin rashes, itching, swelling and behavioral changes (Walker et al., 2008).
Experimental Procedures for Antinociceptive Test
Hot Plate Test
Hot plate test was performed using Eddy's hot plate (Kshitij Innovations, Haryana, India) for determining the central analgesic activity of MEPS. The temperature of hot plate was maintained at 55 ± 1°C. A pre-treatment latency was recorded as baseline for the study. Acute responses like jumping, forepaw licking and withdrawal of paw(s) were considered as nociception. After the treatment of mice with vehicle, MEPS (p.o) or standard drug morphine (i.p.) the latency periods were recorded at 30, 45, 60, 90, and 120 min (Eddy and Leimbach, 1953). The mice were placed in hot plate not more than for 20 s to avoid any tissue damage. The procedure was repeated by employing naloxone (i.p.) 15 min before morphine sulfate or MEPS administration in mice for the evaluation of opioid receptor involvement and the data were pooled. To determine the percentage of maximal possible effect (% MPE) following formula was used (Coelho et al., 2005):
Tail Immersion Test
The procedure was carried out as previously reported (Janssen et al., 1963). The temperature of the water bath was set constant at 52 ± 1°C. Mice that exerted deflection of tail from warm water within 1.5–3.5 s were selected for the study. Mice were gently immobilized for 25–30 s using “Chux” and 1–2 cm of their tail was submersed into the warm water. The violent flick response of mice was taken as end point of nociception. A cut off period of 20 s was maintained to avoid injury to mice. The latency time at 30, 45, 60, 90, and 120 min following administration of vehicle, morphine (i.p.) or MEPS were recorded. A pre-treatment latency was obtained before administration. The same test was carried out with the prior administration of naloxone (i.p.) as described in hot plate test and % MPE was calculated from the latency periods.
Acetic Acid-induced Writhing Test
The effect of MEPS against chemical induced central and peripheral nociception was evaluated by performing acetic acid-induced writhing test. To induce writhing, 0.7% acetic acid (10 mL/kg) was injected (i.p.) 30 min after the administration of vehicle or MEPS and 15 min of diclofenac sodium treatment. Mice were placed in box, left for 5 min and then the number of writhing was counted for the next 10 min (Vogel, 2007). The writhing was characterized by abdominal contraction, stretching or bending of the body, trunk and/or pelvis ending with limbs extension.
Formalin-induced Licking Test
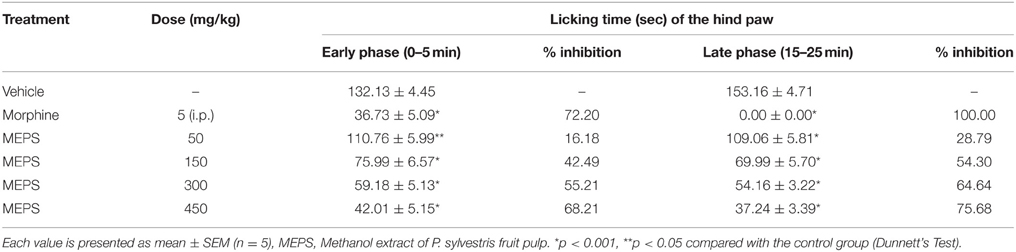
Formalin was injected in the sub-plantar region of the right hind paw of mice to induce pain. Each mouse was injected with a volume of 20 μl of 1.35% formalin solution (0.5% formaldehyde) made up in saline. Formalin was injected 30 min after the administration of vehicle or MEPS. The standard drug morphine was injected 15 min before the formalin injection. Right hind paw licking of mice was measured from 0 to 5 min as early phase response (neurogenic phase) and from 15 to 25 min as late phase response (inflammatory phase). The licking was considered as indicative of nociception (Coelho et al., 2005).
Glutamate-induced Paw Licking and Edema Test
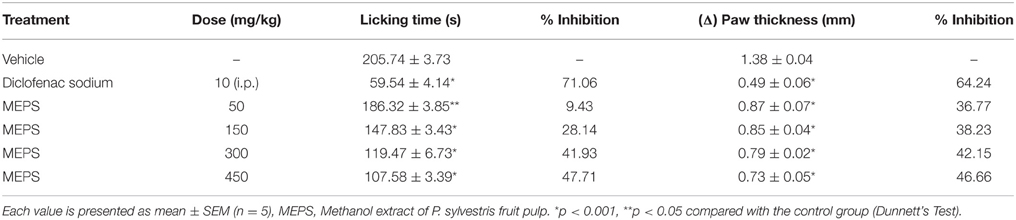
The mice were treated with vehicle, MEPS or diclofenac sodium first. The thickness of right hind paw of each mouse was measured at this stage using digital slide calipers. Glutamate was administered to the sub-plantar region of the right hind paw of each mouse (20 μL/20 μmol per paw) 30 min after the administration of vehicle or MEPS and 15 min after standard drug treatment. To avoid irritation, pH of the preparation was adjusted at 7.4. After glutamate challenge the mice were placed in observation chambers. The licking of injected paw was counted for 15 min and paw thickness was measured again for each mouse. The measure of paw thickness before glutamate injection was subtracted from paw thickness after glutamate challenge to determine the degree of edema (Δ) in mm (Rodrigues et al., 2012).
Experimental Procedures for Neruopharmacological Test
Open Field Test
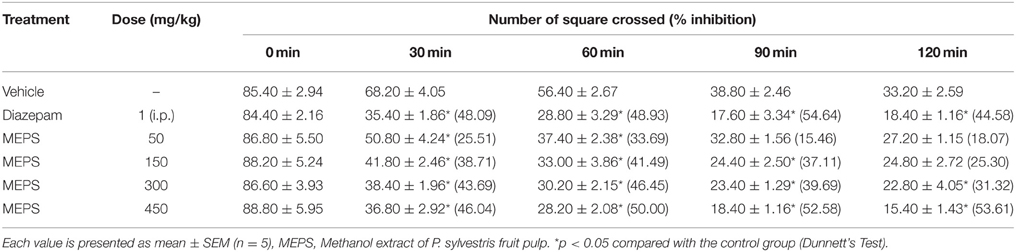
Open field test was performed to evaluate effect of MEPS on locomotion of mice. The experiment was performed in an isolated, silent and dimly lit area. The dimension of apparatus of open field was 50 × 50 × 50 cm3 in size. The floor of the open field was divided into series of squares and each square were alternatively colored black and white. Each mouse was placed at the center of the field after the treatment with vehicle, diazepam or MEPS. Then the number of squares traveled was counted for 3 min at 0, 30, 60, 90, and 120 min (Gupta et al., 1971). The % inhibition was calculated by using the formula, where “MST” means mean squares traveled:
Hole Cross Test
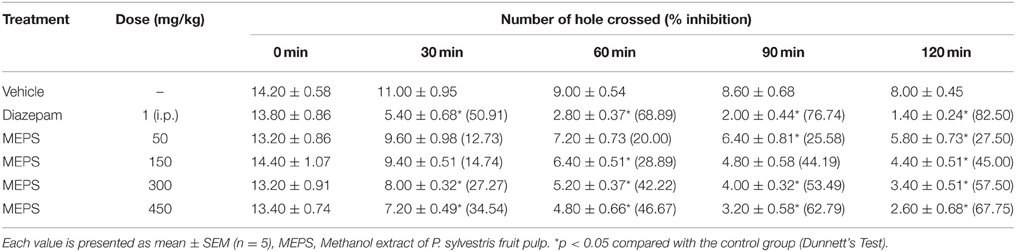
The hole cross test was performed as previously describe by Takagi et al. (1971). A 30 × 20 × 14 cm3 size hole cross box was used to perform the experiment. A fixed partition into the middle of the box divided it into two compartments. There was a 3 cm hole to make easy passage for mice in the partition. Mice were treated with vehicle, diazepam or MEPS and immediately after treatment mice were placed beside the wall of one compartment facing to the hole. Then the number of movement from one compartment to other through the hole was counted for a period of 3 min at 0, 30, 60, 90, and 120 min. The % inhibition was calculated from the number of movements.
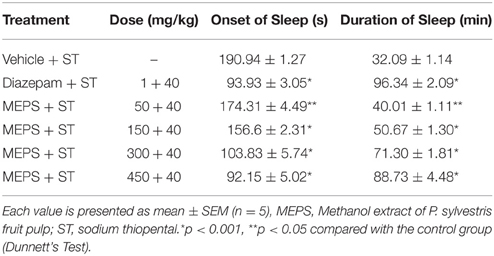
Sodium Thiopental-induced Sleeping Time Test
Mice were treated with vehicle, diazepam or MEPS. To induce sleep, sodium thiopental (40 mg/kg) was administered to each mouse 30 min after the treatments and placed in an observation chamber. After administration of sodium thiopental, time was counted till loss of righting reflex and recorded as the latent period. The duration of sleep was recorded from loss of righting reflex to recovery of righting reflex (Ferrini et al., 1974).
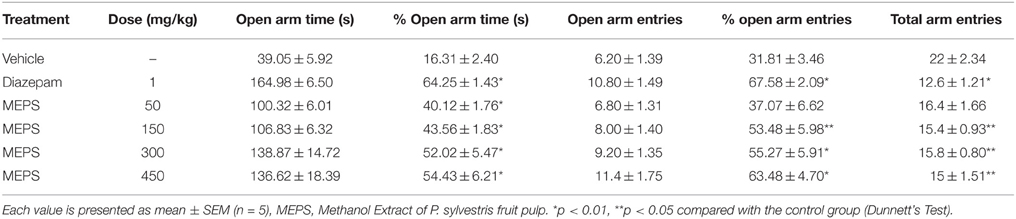
Elevated Plus Maze Test
Elevated plus maze test was performed to evaluate the anxiolytic effect of MEPS. The maze consisted of two open arms (50 × 10 cm) and two enclosed arms (50 × 10 × 40 cm). The arms were arranged such a way that each type was opposite to each other and was elevated to 50 cm height above the floor. The experiment was performed in an isolated and silent area. Vehicle, MEPS or diazepam were administered 30 min before the experiment. Each mouse was placed individually in the center of the maze facing a closed arm. Entry of four paws in an arm was defined as arm entries (Pellow et al., 1985). Then the following parameter was observed and counted: (a) number of entries, (b) time spent into close and open arms during 5 min observation period. The percentage of time spent on open arm by each animal was calculated as follows:
Statistical Analysis
The results were expressed as mean ± SEM (n = 5). To perform statistical analysis of the results, One-Way analysis of variance (ANOVA) followed by Dunnett's post-hoc or Bonferroni test was used as appropriate. All statistical analysis was performed by SPSS 19 software. Differences between groups were considered significant at the level of p < 0.05.
Results
Preliminary Phytochemical Screening
The presence of carbohydrates, reducing sugars, resins, tannins, proteins, saponins, falvonoids, alkaloids, and steroids in MEPS was found in the preliminary phytochemical screening.
GC-MS Analysis
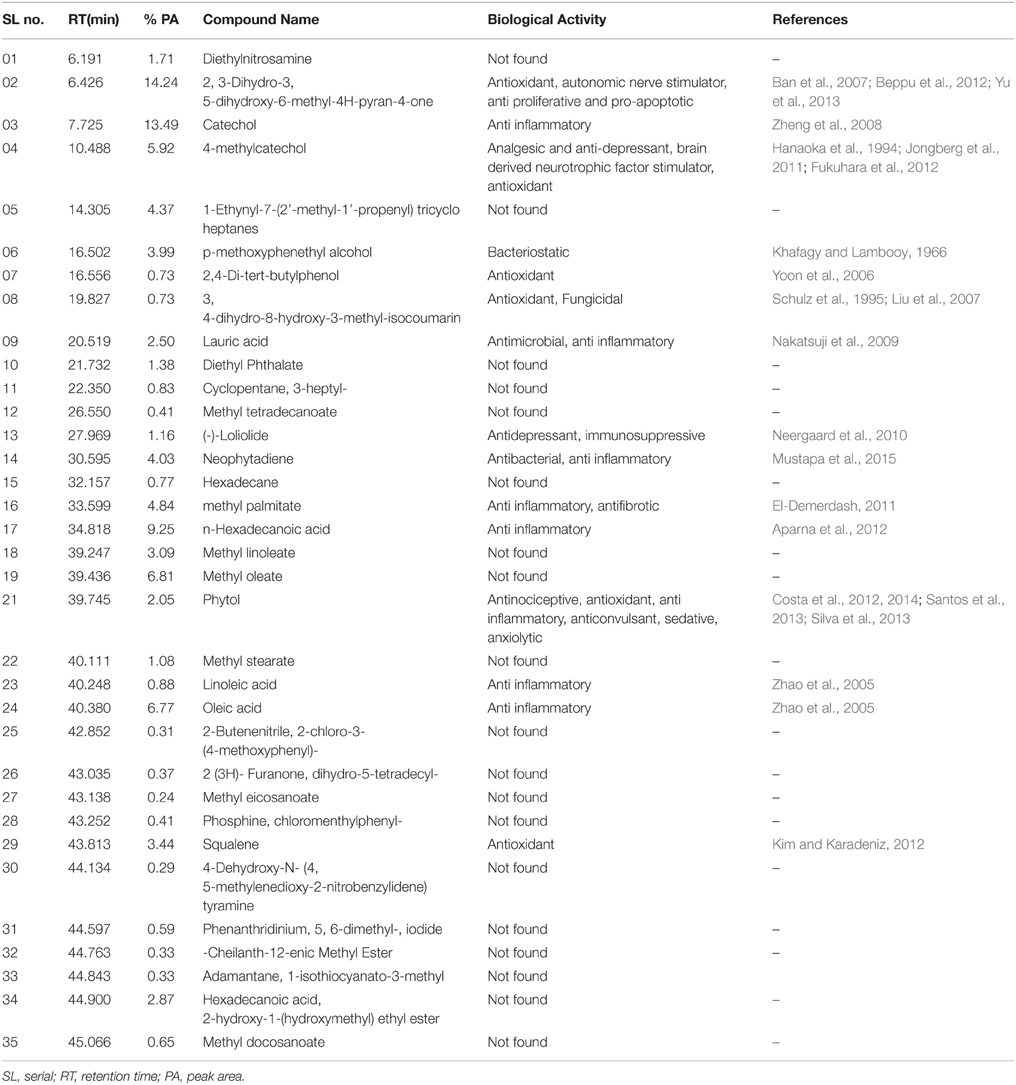
The compounds identified by GC-MS analysis in MEPS are listed in Table 1 and the chromatogram is shown in Figure 1. Among 35, the major constituents were identified by comparing the relative percentage amount and they are: 2,3-Dihydro-3,5-dihydroxy-6-methyl-4H-pyran-4-one (14.24%), Catechol (13.49%), n-Hexadecanoic acid (9.25%), Methyl oleate (6.81%), Oleic acid (6.77%), 4-methylcatechol (5.92%), methyl palmitate (4.84%), 1-Ethynyl-7-(2'-methyl-1'-propenyl) tricycloheptanes (4.37%), Neophytadiene (4.03%), p-methoxyphenethyl alcohol (3.93%), Squalene (3.44%), Linoleate (3.09%), Hexadecanoic acid, 2-hydroxy-1-(hydroxymethyl) ethyl ester (2.87%), Lauric acid (2.50%), Phytol (2.05%), Ethanamine, N-ethyl-N-nitroso (1.17%), (-)-Loliolide (1.16%).
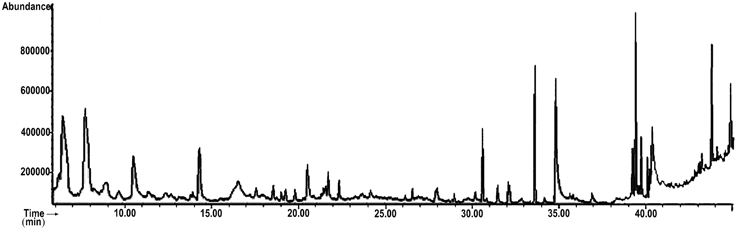
Figure 1. Total ionic chromatogram (TIC) of methanol extract of P. sylvestris fruit (MEPS). TIC of MEPS obtained by GC-MS with energy of ionization of 70 eV.
Acute Toxicity
Oral administration of MEPS did not produce any toxicity at doses of 500–6000 mg/kg. During the 72 h observation period no behavioral changes, allergic reaction or mortality were observed. This result demonstrates that MEPS have lower toxicity profile.
Hot Plate Test
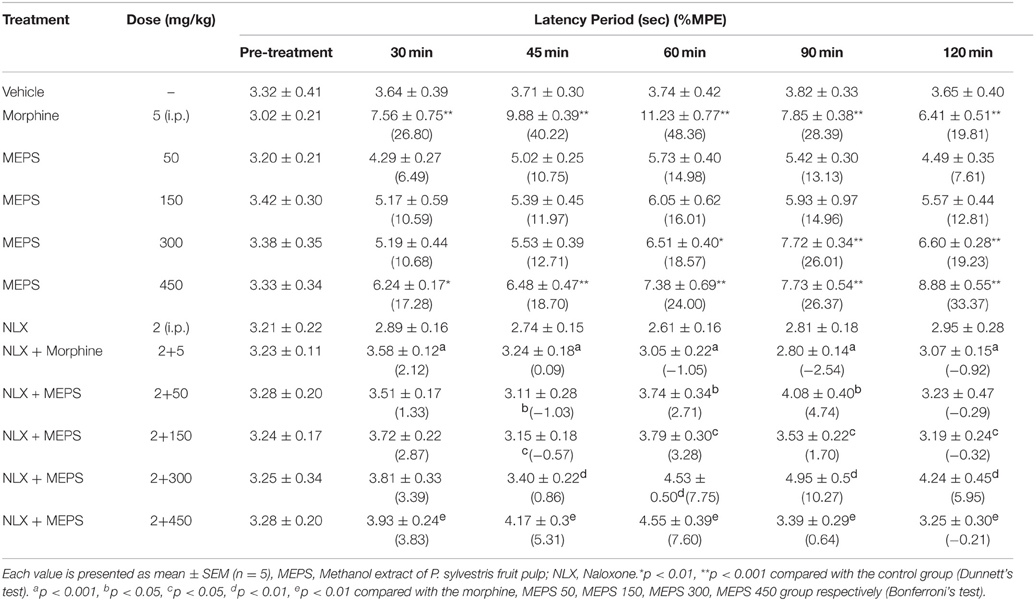
Oral administration of MEPS demonstrated significant increase in latency period to thermal stimulus at the doses of 300 and 450 mg/kg in hot plate test (p < 0.01) as shown in Table 2. The protection against heat-induced pain response of MEPS was dose-dependent and stronger at 450 mg/kg dose than other doses. The reaction time for standard drug morphine was more than any dose of MEPS at all observation period. MEPS also showed significant % MPE at all experimental doses (p < 0.05). The antinoceptive activity of MEPS at all four doses and of morphine was significantly antagonized by naloxone (p < 0.05); (Table 2).
Tail Immersion Test
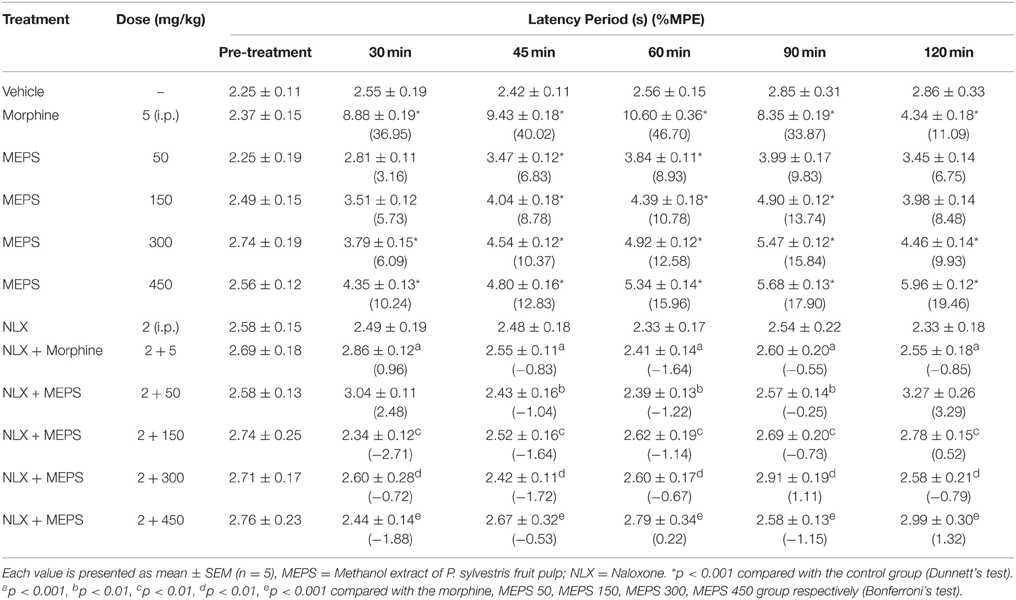
MEPS significantly reduced the hot-water induced nociception at all experimental doses in tail immersion test (p < 0.001). The antinociceptive effect of MEPS and morphine are shown in Table 3. Both MEPS and morphine significantly increased the latency time. The % MPE of the extract was also significant at all experimental doses (p < 0.05). The % MPE value for morphine was higher than MEPS at all observation periods. The activity of MEPS against thermal nociception was dose-dependent. Naloxone significantly reversed the antinoceptive effect of morphine and of MEPS at all experimental doses (P < 0.01).0.1.
Acetic Acid-induced Writhing
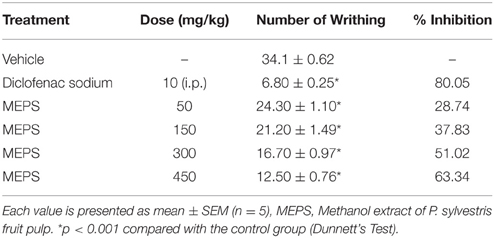
As shown in Table 4, acetic acid-induced writhing response was significantly decreased by standard drug (diclofenac sodium) and MEPS at all doses (p < 0.001). The % inhibition of writhing by MEPS was dose-dependent and highest at the dose of 450 mg/kg (63.34%). The standard drug (diclofenac sodium) showed maximum % inhibition (80.05%).
Formalin-induced Licking
MEPS at the doses of 150, 300, and 450 mg/kg and the centrally acting analgesic, morphine, produced significant inhibition of formalin-induced licking (p < 0.001) in both early and late phase of formalin-induced licking test (Table 5). The inhibition of licking by MEPS was dose-dependent and percent of inhibition was highest at 450 mg/kg in both early and late phases (68.21% and 75.68% respectively). Morphine completely inhibited the nociceptive effect at late phase.
Glutamate-induced Paw Licking and Edema
MEPS significantly reduced the glutamate-induced paw licking at the doses of 150, 300, and 450 mg/kg (p < 0.001). The inhibitory effect of MEPS was dose-dependent and highest at the dose of 450 mg/kg (47.71%). However, the standard drug (diclofenac sodium) showed maximum inhibition (71.06%). The extract also showed significant reduction of paw edema (p < 0.001) at all the test doses (Table 6).
Open Field Test
MEPS significantly inhibited the locomotor activity at the doses of 50 and 150 mg/kg for first 60 and 90 min respectively. However, at the doses of 300 and 450 mg/kg, MEPS significantly reduced the locomotor activity from 30 to 120 min like the reference drug diazepam (p < 0.05). The percent inhibition of the dose 450 mg/kg was close to that of diazepam at 30 and 60 min (Table 7).
Hole Cross Test
MEPS demonstrated significant inhibition of movement at the doses of 300 and 450 mg/kg throughout the observation periods in hole cross test (p < 0.05). On the other hand the dose 50 and 150 mg/kg showed significant effect at later observation periods. The standard drug diazepam showed significant (p < 0.05) reduction of movement and highest percent inhibition of locomotion at all the study periods. The reduction of locomotor activity by MEPS was dose-dependent (Table 8).
Sodium Thiopental-induced Sleeping Time Test
The doses 150, 300, and 450 mg/kg of MEPS showed significant (p < 0.001) reduction in onset of sleep and increased sleep duration. The dose 50 mg/kg also increased the sleeping time and decreased onset of sleep (p < 0.05). The dose-dependent effect of MEPS was clear from the observations of 50–450 mg/kg doses. The effect of diazepam was found most significant considering the onset of sleeping and highest duration of sleeping (p < 0.001) (Table 9).
Elevated Plus Maze Test
Table 10 clearly shows that the time spent in open arm was increased and the percent of time spent was significant (p < 0.05) at all the doses of MEPS in elevated plus maze test. The standard drug diazepam also significantly increased the percent time spent in open arm (p < 0.05). The percent open arm entries of MEPS at the doses of 150, 300, and 450 mg/kg and of diazepam were significant (p < 0.05). In addition, total number of entries was also reduced significantly by the extract and diazepam compared to the control group.
Discussion
The results of the present study indicate peripheral and central antinociceptive, CNS depressant, sedative and anxiolytic effect of MEPS at different doses in mice models. In addition, study of acute toxicity at the doses of 500–6000 mg/kg suggests that MEPS possess low toxicity profile.
The significant (p < 0.01) inhibition of the thermally induced nociception and increase of latency time in hot plate test demonstrates the central antinociceptive activity of MEPS (Table 2). The effect was also observed (p < 0.001) in the tail immersion test (Table 3). The tail withdrawal response is selective only for centrally acting analgesics (Srinivasan et al., 2003). Therefore, the outcome of tail immersion test supports the effect of MEPS in hot plate test. The hot plate test induces nociception via supraspinal reflex whereas tail immersion test induce nociceptive pain by spinal reflexes (Chapman et al., 1985). Opioid agents exhibit their analgesic effect via both supraspinal (μ1, κ3, δ1, σ2) and spinal (μ2, κ1, δ2) receptors (Hosseinzadeh et al., 2002; Jinsmaa et al., 2004, 2005). Therefore the antinociceptive activity of MEPS is due to action on spinal and supraspinal receptors. Naloxone significantly antagonized (p < 0.05) the antinociceptive effect of morphine and MEPS at different doses level in both thermal pain models. This confirms the opioid receptors mediated antinociceptive activity of MEPS.
Acetic acid-induced writhing test is a commonly used method to evaluate the antinociceptive activity of peripheral and central analgesic agents. The action of acetic acid causes the release of endogenous histamine, prostaglandins (PGs), serotonin, bradykinin, cycloxoygenase (COX), lipoxygenase (LOX), and cytokines (TNF-α, IL-1β, and IL-8). These visceral inflammatory mediators enter the dorsal horn of central nervous system and stimulate primary afferent nociceptors (Ikeda et al., 2001) and results in induction of pain expressing abdominal constriction (Bley et al., 1998). Oral administration of MEPS (p < 0.001) significantly reduced the number of abdominal constriction induced by acetic acid in mice (Table 4). The result clearly indicates that the antinociception produced by MEPS is due to its inhibition of COX, LOX and other endogenous inflammatory mediators as well as of primary afferent nociceptors signal transduction. The identified compound phytol has been reported to give antinociceptive activity via central and peripheral mechanisms (Santos et al., 2013). Besides histamine, serotonin, bradykinin, PGE2 and cytokines inhibitory activity of phytol (Silva et al., 2013) provide justification of the result.
The formalin-induced nociception is mediated via direct stimulation of sensory afferent fibers by the chemical nociceptors especially C-fibers in early phase and peripheral inflammatory mediators such as histamine, prostaglandins (PGs), serotonin and bradykinin in late phase (Le Bars et al., 2001; Parada et al., 2001). In formalin-induced licking test peripherally acting analgesics inhibit early phase nociception where the centrally acting analgesics inhibit the nociception of both phases (Hunskaar and Hole, 1987; Tjølsen et al., 1992). The results showed that MEPS significantly (p < 0.001) reduced paw licking at both early and late phases in a dose-dependent manner. The effect was stronger in late phase (Table 5). Therefore the inhibition of nociception by MEPS in both phases gives further evidence of its central antinociceptive effect. Moreover, the late phase inhibition suggests the anti-inflammatory potential of MEPS.
MEPS produced significant dose-dependent inhibition (p < 0.001) in both glutamate-induced paw licking and edema (Table 6). The glutamate induced nociceptive response and edema formation involves (N-methyl-D-aspartate) NMDA and non-NMDA (AMPA, Kainate) receptors respectively in peripheral, spinal and supraspinal sites of action. Glutamate causes neuropeptides (substance P) and pro-inflammatory cytokines (TNF-α, IL-1β) release to transmit nociceptive signals from the peripheral nervous system to the dorsal horn of the spinal cord. Moreover, the pro-inflammatory signals are mediated by, ROS (Reactive oxygen species) and NOS (Nitric oxide synthase) participated stimulation of TNF-α, IL-1, and IL-6 genes through the activation of the redox-sensitive transcription factor NF-κB (Beauparlant and Hiscott, 1996; Beirith et al., 2002; Ribas et al., 2008). Therefore, from the results it can be suggested that MEPS inhibited the release of neuropeptides and activation of NMDA and non-NMDA receptors as well as interrupted the ROS and NOS mediated pro-inflammatory signals. The compounds such as 2,3-Dihydro-3,5-dihydroxy-6-methyl-4H-pyran-4-one, catechol, oleic acid and linoleic acids found in MEPS has been reported to inhibit the NF-κB, NOS, TNF-α, and TNF-α gene expression respectively (Zhao et al., 2005; Ban et al., 2007; Zheng et al., 2008) which strongly suggests the pro-inflammatory signal inhibitory activity of MEPS.
We have detected the presence of carbohydrates, steroids, alkaloids, saponins, and tannins by preliminary phytochemical screening and considerable amount of phenolic content (128.6 ± 2.7 mgGAE/g) has been found in P. sylvestris fruit (Prakash et al., 2013). Scientific investigations reported that plant materials containing flavonoids, phenols and tannins are responsible for analgesic and CNS activity (Takahashi et al., 1986; Starec et al., 1988; Mills and Bone, 2000; Morteza-Semnani et al., 2006).
Beside antinociceptive activity, MEPS has also been found to have neurobehavioral effects. Both in open field and hole cross test MEPS depressed the central nervous system. The locomotor activity of mice gradually decreased during the period of observation (Tables 7, 8). In addition, MEPS significantly decreased latency time and increased the sleeping time in sodium thiopental-induced sleeping time test (Table 9) suggesting its possible sedative effect. CNS depressant and sedative drugs potentiate the gamma aminobutyric acid (GABA) mediated post synaptic inhibition via allosteric modification of GABA receptors as well as decrease the sleep latency and increase sleeping time (Sieghart and Sperk, 2002; Aladeokin and Umukoro, 2011; O'Donnell and Shelton, 2011). Therefore the significant decrease of locomotion and sleep enhancement activity of MEPS reveals its potential action on GABA receptors. The GABAA receptor mediated sedative activity of identified compound phytol (Costa et al., 2014) provides further evidence of GABAergic mechanism of action of MEPS.
Another potentiality of MEPS was revealed in elevated plus maze test. This test is frequently used to evaluate the anxiolytic or anxiogenic properties of drug. The fact is rodents are extremely aversive to an open area and the major index of anxiety is the frequency and time spent in the open arms (Pellow and File, 1986). The benzodiazepine drugs exert their anxiolytic activity via their action on GABA/benzodiazepine/chloride (GBC) complex mechanism and increase the percent of spent time, frequency of entries in open arm and also reduce total number of entries (Pellow et al., 1985; Stephens et al., 1986). The results showed that diazepam and MEPS significantly (p < 0.05) increased percentage of spent time, frequency of entries in open arm and reduced total arm entries in elevated plus maze (Table 10). The findings of probable GABAergic-like mechanism of MEPS and action like reference anxiolytic drug diazepam may be a potential indicator of its anxiolytic-like activity. In addition the anxiolytic effect of phytol (Costa et al., 2014) found in MEPS support our results. Therefore the outcomes of our investigation rationalize the traditional implication of P. sylvestris fruit as nervine tonic in the treatment of CNS disorder like nervous debility.
From the results of different pain models it may be suggested that crude methanol extract of P. sylvestris fruit possesses potent and long-lasting antinociceptive activity. Inhibition of MEPS-induced antinociception by selective opioid antagonist, naloxone, suggests the involvement of opioid receptor mediated analgesia. The results also provide the evidence of inhibition of peripheral inflammatory mediators such as COX, LOX, PGs in pain inhibition. In addition the neuropharmacological evaluation also suggests the sleep enhancing and anxiolytic potentiality of P. sylvestris fruit which is possibly mediated via GABAergic pathway. These outcomes rationalize the use of P. sylvestris fruit in traditional medicine in painful conditions such as toothache, headache, arthritis backache or pain in buttocks, and also to induce sedation, as nervine tonic and in the treatment of nervous debility. The GC-MS analysis shows there are potential biologically active compounds present in P. sylvestris fruit. Taking the findings into account, it seems quite possible that P. sylvestris fruit may lead to the development of new natural products having analgesic and neuropharmacological effect.
Author Contributions
MZI, TA, and MS conceived and designed the study. MS performed the experiments and collected data. Data analysis was performed by MZI and MS. MS, SA, and MZI drafted the manuscript and all authors revised and approved the content of manuscript.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
The authors are grateful to Mirola Afroze, Scientific Officer, Designated Reference Institute for Chemical Measurements (DRICM), Bangladesh Council of Scientific and Industrial Research (BCSIR) for her cooperation in managing the GC-MS analysis from BCSIR.
Abbreviations
MEPS, Methanol extract of Phoenix sylvestris fruit pulp; CNS, central nervous system; GC-MS, Gas chromatography-mass spectroscopy; ACE, Angiotensin I-converting enzyme; r.p.m, Rotation per minute; AVMA, American Veterinary Medical Association; icddr,b, International Center for Diarrhoeal Disease Research, Bangladesh; p.o, per oral, i.p. intraperitoneal; NIST-MS, National Institute of Standards and Technology- mass spectroscopy; EI, electron ionization; TIC, total ionic chromatogram; % MPE, percentage of maximal possible effect; MST, mean square traveled by animals; ANOVA, analysis of variance; LD50, median lethal dose; COX, cycloxoygenase, LOX, lipoxygenase, TNF-α, Tumor necrosis factor alpha; IL-1β, Interleukin-1 beta; IL-8, Interleukin 8; PGs, Prostaglandins; NMDA, N-methyl-D-aspartate; AMPA, Alpha-amino-3-hydroxy-5-methylisoxazole-4-propionic acid; NF-κB, nuclear factor kappa-light-chain-enhancer of activated B cells; ROS, Reactive oxygen species; NOS, Nitric oxide synthase; GAE, Gallic acid equivalence method; GABA, gamma amino butyric acid; GBC, GABA/benzodiazepine/chloride complex; NLX, naloxone; ST, sodium thiopental; n, number of mice; RT, retention time; PA, peak area; eV, electron volt.
References
Acharya, E., and Pokhrel, B. (2006). Ethno-medicinal plants used by Bantar of Bhaudaha, Morang, Nepal. Our Nat. 4, 96–103. doi: 10.3126/on.v4i1.508
Ahmad, M., Khan, M. A., and Zafar, M. (2008). Traditional herbal cosmetics used by local women communities in district Attock of northern Pakistan. Indian J. Tradit. Knowl. 7, 421–424.
Aladeokin, A. C., and Umukoro, S. (2011). Psychopharmacological properties of an aqueous extract of Tetracarpidium conophorum Hutch. & Dalziel in mice. J. Nat. Med. 65, 411–416. doi: 10.1007/s11418-011-0506-1
Aparna, V., Dileep, K. V., Mandal, P. K., Karthe, P., Sadasivan, C., and Haridas, M. (2012). Anti-inflammatory property of n-hexadecanoic acid: structural evidence and kinetic assessment. Chem. Biol. Drug Des. 80, 434–439. doi: 10.1111/j.1747-0285.2012.01418.x
Ban, J. O., Hwang, I. G., Kim, T. M., Hwang, B. Y., Lee, U. S., Jeong, H. -S., et al. (2007). Anti-proliferate and pro-apoptotic effects of 2, 3-Dihydro-3, 5-dihydroxy-6-methyl-4H-pyranone through inactivation of NF-κB in human colon cancer cells. Arch. Pharm. Res. 30, 1455–1463. doi: 10.1007/BF02977371
Beauparlant, P., and Hiscott, J. (1996). Biological and biochemical inhibitors of the NF-kappa B/ Rel proteins and cytokine synthesis. Cytokine Growth Factor Rev. 7, 175–190. doi: 10.1016/1359-6101(96)00020-2
Beirith, A., Santos, A. R. S., and Calixto, J. B. (2002). Mechanisms underlying the nociception and paw oedema caused by injection of glutamate into the mouse paw. Brain Res. 924, 219–228. doi: 10.1016/S0006-8993(01)03240-1
Beppu, Y., Komura, H., Izumo, T., Horii, Y., Shen, J., Tanida, M., et al. (2012). Identificaton of 2,3-Dihydro-3,5-dihydroxy-6-methyl-4H-pyran-4-one isolated from Lactobacillus pentosus strain S-PT84 culture supernatants as a compound that stimulates autonomic nerve activities in rats. J. Agric. Food. Chem. 60, 11044–11049. doi: 10.1021/jf302355e
Bley, K. R., Hunter, J. C., Eglen, R. M., and Smith, J. A. (1998). The role of IP prostanoid receptors in inflammatory pain. Trends Pharmacol. Sci. 19, 141–147. doi: 10.1016/S0165-6147(98)01185-7
Calixto, J. B., Beirith, A., Ferreira, J., Santos, A. R., Filho, V. C., and Yunes, R. A. (2000). Naturally occurring antinociceptive substances from plants. Phytother. Res. 14, 401–418. doi: 10.1002/1099-1573(200009)14:6<401::AID-PTR762>3.0.CO;2-H
Chapman, C. R., Casey, K. L., Dubner, R., Foley, K. M., Gracely, R. H., and Reading, A. E. (1985). Pain measurement: an overview. Pain 22, 1–31. doi: 10.1016/0304-3959(85)90145-9
Coelho, L. P., Reis, P. A., de Castro, F. L., Gayer, C. R., da Silva Lopes, C., da Costa e Silva, M. C., et al. (2005). Antinociceptive properties of ethanolic extract and fractions of Pterodon pubescens Benth. seeds. J. Ethnopharmacol. 98, 109–116. doi: 10.1016/j.jep.2005.01.014
Costa, J. P., de Oliveira, G. A., de Almeida, A. A., Islam, M. T., de Sousa, D. P., and de Freitas, R. M. (2014). Anxiolytic-like effects of phytol: possible involvement of GABAergic transmission. Brain Res. 1547, 34–42. doi: 10.1016/j.brainres.2013.12.003
Costa, J. P., Ferreira, P. B., de Sousa, D. P., Jordan, J., and Freitas, R. M. (2012). Anticonvulsant effect of phytol in a pilocarpine model in mice. Neurosci. Lett. 523, 115–118. doi: 10.1016/j.neulet.2012.06.055
Das, S., Das, S., and De, B. (2012). In vitro inhibition of key enzymes related to diabetes by the aqueous extracts of some fruits of West Bengal, India. Curr. Nutr. Food Sci. 8, 19–24. doi: 10.2174/157340112800269614
Das, S., and De, B. (2013). Evaluation of Angiotensin I-Converting Enzyme (ACE) inhibitory potential of some underutilized indigenous fruits of West Bengal using an in vitro model. Fruits 68, 499–506. doi: 10.1051/fruits/2013092
Eddy, N. B., and Leimbach, D. (1953). Synthetic analgesics. II. Dithienylbutenyl-and dithienylbutylamines. J. Pharmacol. Exp. Ther. 10, 385–393.
El-Demerdash, E. (2011). Anti-inflammatory and antifibrotic effects of methyl palmitate. Toxicol. Appl. Pharmacol. 254, 238–244. doi: 10.1016/j.taap.2011.04.016
Ferrini, R., Miragoli, G., and Taccardi, B. (1974). Neuropharmacological studies on SB 5833, a new psychotherapeutic agent of the benzodiazepine class. Arzneimittelforschurch 24, 2029–2032.
Fukuhara, K., Ishikawa, K., Yasuda, S., Kishishita, Y., Kim, H. K., Kakeda, T., et al. (2012). Intracerebroventricular 4-Methylcatechol (4-MC) ameliorates chronic pain associated with depression-like behavior via induction of brain-derived neurotrophic factor (BDNF). Cell. Mol. Neurobiol. 32, 971–977. doi: 10.1007/s10571-011-9782-2
Ghani, A. (2003). Medicinal Plants of Bangladesh: Chemical Constituents and Uses. Dhaka: Asiatic Society of Bangladesh.
Goswami, S. K., Kumar, P. M., Jamwal, R., Dethe, S., Agarwal, A., and Naseeruddin, I. M. (2012). Screening for Rho-kinase 2 inhibitory potential of Indian medicinal plants used in management of erectile dysfunction. J. Ethnopharmacol. 144, 483–489. doi: 10.1016/j.jep.2012.07.045
Gupta, B. D., Dandiya, P. C., and Gupta, M. L. (1971). A psychopharmacological analysis of behavior in rat. Jpn. J. Pharmacol. 21, 293–298. doi: 10.1254/jjp.21.293
Gurib-Fakim, A. (2006). Medicinal plants: traditions of yesterday and drugs of tomorrow. Mol. Aspects Med. 27, 1–93. doi: 10.1016/j.mam.2005.07.008
Hanaoka, Y., Ohi, T., Furukawa, S., Furukawa, Y., Hayashi, K., and Matsukura, S. (1994). The therapeutic effects of 4-methylcatechol, a stimulator of endogenous nerve growth factor synthesis, on experimental diabetic neuropathy in rats. J. Neurol. Sci. 122, 28–32. doi: 10.1016/0022-510X(94)90048-5
Harney, N. V. (2013). Ethnomedicinal plants diversity of Bhadrawati Tahsil of Chandrapur district, Maharashtra, India. Int. J. Sci. Res. Pub. 3:8.
Hosseinzadeh, H., Ramezani, M., Fadishei, M., and Mahmoudi, M. (2002). Antinociceptive, anti-inflammatory and acute toxicity effects of Zhumeria majdae extracts in mice and rats. Phytomed 9, 135–141. doi: 10.1078/0944-7113-00097
Howlader, M. A., Bachar, S. C., Begum, F., and Rouf, A. S. (2006). Diuretic and analgesic effects of the methanol extract of Phoenix sylvestris root. Pak. J. Pharm. Sci. 19, 330–332.
Hunskaar, S., and Hole, K. (1987). The formalin test in mice: dissociation between inflammatory and non-inflammatory pain. Pain 30, 103–114. doi: 10.1016/0304-3959(87)90088-1
Ikeda, Y., Ueno, A., Naraba, H., and Oh-ishi, S. (2001). Involvement of vanilloid receptor VR1 and prostanoids in the acid-induced writhing responses of mice. Life Sci. 69, 2911–2919. doi: 10.1016/S0024-3205(01)01374-1
Janssen, P. A., Niemegeers, C. J., and Dony, J. G. (1963). The inhibitory effect of fentanyl and other morphine-like analgesics on the warm water induced tail withdrawal reflex in rats. Arzneimittelforschung 13, 502–507.
Jinsmaa, Y., Fujita, Y., Shiotani, K., Miyazaki, A., Li, T., Tsuda, Y., et al. (2005). Differentiation of opioid receptor preference by [Dmt1] endomorphin-2-mediated antinociception in the mouse. Eur. J. Phamrcol. 509, 37–42. doi: 10.1016/j.ejphar.2004.12.015
Jinsmaa, Y., Okada, Y., Tsuda, Y., Shiotani, K., Sasaki, Y., Ambo, A., et al. (2004). Novel 2′, 6′dimethyl- L-tyrosine-containing pyrazinone opioid mimetic μ-agonists with potent antinociceptive activity in mice. J. Pharmacol. Exp. Ther. 309, 432–438. doi: 10.1124/jpet.103.060061
Jongberg, S., Lund, M. N., Waterhouse, A. L., and Skibsted, L. H. (2011). 4-Methylcatechol inhibits protein oxidation in meat but not disulfide formation. J. Agric. Food Chem. 59, 10329–10335. doi: 10.1021/jf202268q
Khafagy, E. Z., and Lambooy, J. P. (1966). Inhibition of protein, RNA and DNA synthesis in Escherichia coli by p-methoxyphenethyl alcohol. Biochim. Biophys. Acta. 123, 646–648. doi: 10.1016/0005-2787(66)90240-1
Kim, S. K., and Karadeniz, F. (2012). Biological importance and applications of squalene and squalane. Adv. Food Nutr. Res. 65, 223–233. doi: 10.1016/B978-0-12-416003-3.00014-7
Le Bars, D., Gozariu, M., and Cadden, S. W. (2001). Animal models of nociception. Pharmacol. Rev. 53, 597–652.
Leonti, M., and Casu, L. (2013). Traditional medicines and globalization: current and future perspectives in ethnopharmacology. Front. Pharmacol. 4:92. doi: 10.3389/fphar.2013.00092
Liu, X., Dong, M., Chen, X., Jiang, M., Lv, X., and Yan, G. (2007). Antioxidant activity and phenolics of an endophytic Xylaria sp. from Ginkgo biloba. Food Chem. 105, 548–554. doi: 10.1016/j.foodchem.2007.04.008
Lohar, P. S., Lohar, M. S., and Roychoudhury, S. (2009). Erythropoitic effects of some medicinal plants of India on experimental rat model. Slovak. J. Anim. Sci. 42, 95–98.
Manikandan, S. (2013). Ethnomedicinal flora of Ivanur Panchayat in Cuddalore district, Tamil Nadu, India. Int. Res. J. Plant Sci. 3, 39–46.
Mills, S., and Bone, K. (2000). Principles and Practice of Phytotherapy: Modern Herbal Medicine. Edinburgh: Churchill Livingstone.
Morteza-Semnani, K., Mahmoudi, M., and Heidar, M. R. (2006). Analgesic activity of the methanol extract and total alkaloids of Glaucium paucilobum. Methods Find. Exp. Clin. Pharmacol. 28, 151–155. doi: 10.1358/mf.2006.28.3.985229
Mukherjee, K., Paul, P., and Banerjee, E. R. (2014). Free radical scavenging activities of Date Palm (Phoenix sylvestris) fruit extracts. Nat. Prod. Chem. Res. 2:151. doi: 10.4172/2329-6836.1000151
Murthy, E. N., and Madhav, N. V. (2015). Enumeration of medicinal plants of Ramagiri-Khilla forests of Karimnagar district, Telangana, India. Int. J. Pharm. Life Sci. 6, 4405–4416.
Mustapa, A. N., Martin, Á., Mato, R. B., and Cocero, M. J. (2015). Extraction of phytocompounds from the medicinal plant Clinacanthus nutans Lindau by microwave-assisted extraction and supercritical carbon dioxide extraction. Ind. Crops. Prod. 74, 83–94. doi: 10.1016/j.indcrop.2015.04.035
Nakatsuji, T., Kao, M. C., Fang, J. Y., Zouboulis, C. C., Zhang, L., Gallo, R. L., et al. (2009). Antimicrobial property of lauric acid against Propionibacterium acnes: its therapeutic potential for inflammatory Acne vulgaris. J. Invest. Dermatol. 129, 2480–2488. doi: 10.1038/jid.2009.93
Neergaard, J. S., Rasmussen, H. B., Stafford, G. I., Staden, J. V., and Jäger, A. K. (2010). Serotonin transporter affinity of (-)-loliolide, a monoterpene lactone from Mondia whitei. S. Afr. J. Bot. 76, 593–596. doi: 10.1016/j.sajb.2010.02.091
O'Donnell, J. M., and Shelton, R. C. (2011). “Drug therapy of depression and anxiety disorders,” in Goodman and Gilman's The Pharmacological Basis of Therapeutics, ed L. L. Brunton (New York, NY: McGraw-Hill Medical), 413.
Parada, C. A., Tambeli, C. H., Cunha, F. Q., and Ferreira, S. H. (2001). The major role of peripheral release of histamine and 5-hydroxy tryptamine in formalin-induced nociception. Neuroscience 102, 937–944. doi: 10.1016/S0306-4522(00)00523-6
Pellow, S., Chopin, P., File, S. E., and Briley, M. (1985). Validation of open: closed arm entries in an elevated plus maze as a measure of anxiety in the rat. J. Neurosci. Methods 14, 149–167. doi: 10.1016/0165-0270(85)90031-7
Pellow, S., and File, S. E. (1986). Anxiolytic and anxiogenic drug effects on exploratory activity in an elevated plus maze: a novel test of anxiety in the rat. Pharmacol. Biochem. Behav. 24, 525–529. doi: 10.1016/0091-3057(86)90552-6
Prakash, D., Upadhyay, G., and Gupta, C. (2013). Total phenol and antioxidant activity of some fruits and their under-utilized parts. Int. Food Res. J. 20, 1717–1724.
Rajkumari, R., Singh, P. K., Das, A. K., and Dutta, B. K. (2013). Ethnobotanical investigation of wild edible and medicinal plants used by the Chiru Tribe of Manipur, India. Pleione 7, 167–174.
Rao, K. V. N., Rao, S. S. R., Rao, K. N., and Srimannarayana, G. (1980). Effect of some proanthocyanidins and catechins on the growth of Lemna paucicostata Hegelm. Proc. Plant Sci. 89, 73–77.
Ribas, C. M., Meotti, F. C., Nascimento, F. P., Jacques, A. V., Dafre, A. L., Rodrigues, A. L., et al. (2008). Antinociceptive effect of the Polygala sabulosa hydroalcoholic extract in mice: evidence for the involvement of glutamatergic receptors and cytokine pathways. Basic Clin. Pharmacol. Toxicol. 103, 43–47. doi: 10.1111/j.1742-7843.2008.00245.x
Rodrigues, M. R., Kanazawa, L. K., das Neves, T. L., da Silva, C. F., Horst, H., Pizzolatti, M. G., et al. (2012). Antinociceptive and anti-inflammatory potential of extract and isolated compounds from the leaves of Salvia officinalis in mice. J. Ethnopharmacol. 139, 519–526. doi: 10.1016/j.jep.2011.11.042
Sambandan, K., and Dhatchanamoorthy, N. (2012). Studies on the phytodiversity of a sacred grove and its traditional uses in Karaikal district, UT Puducherry. J. Phytology 4, 16–21.
Santos, C. C., Salvadori, M. S., Mota, V. G., Costa, L. M., de Almeida, A. A., de Oliveira, G. A., et al. (2013). Antinociceptive and antioxidant activities of phytol in vivo and in vitro models. Neurosci. J. 2013:949452. doi: 10.1155/2013/949452
Schulz, B., Sucker, J., Aust, H. J., Krohn, K., Ludewig, K., Jones, P. G., et al. (1995). Biologically active secondary metabolites of endophytic Pezicula species. Mycol. Res. 99, 1007–1015. doi: 10.1016/S0953-7562(09)80766-1
Sieghart, W., and Sperk, G. (2002). Subunit composition, distribution and function of GABA-A receptor subtypes. Curr. Top. Med. Chem. 2, 795–816. doi: 10.2174/1568026023393507
Silva, R. O., Sousa, F. B., Damasceno, S. R., Carvalho, N. S., Silva, V. G., Oliveira, F. R., et al. (2013). Phytol, a diterpene alcohol, inhibits the inflammatory response by reducing cytokine production and oxidative stress. Fundam. Clin. Pharmacol. 28, 455–464. doi: 10.1111/fcp.12049
Srinivasan, K., Muruganandan, S., Lal, J., Chandra, S., Tandan, S. K., Raviprakash, V., et al. (2003). Anti-nociceptive and antipyretic activities of Pongamia pinnata leaves. Phytother. Res. 17, 259–264. doi: 10.1002/ptr.1126
Starec, M., Waitzova, D., and Elis, J. (1988). Evaluation of the analgesic effect of RG-tannin using the “hot plate”and“tailflick”method in mice. Cesk. Farm. 37, 319–321.
Stephens, D. N., Meldrum, B. S., Weidmann, R., Schneider, C., and Grützner, M. (1986). Does the excitatory amino acid receptor antagonist 2-APH exhibit anxiolytic activity? Psychopharmacology 90, 166–169. doi: 10.1007/BF00181234
Takagi, K., Watanabe, M., and Saito, H. (1971). Studies on the spontaneous movement of animals by the hole cross test: effect of 2-dimethylaminoethane, its acyl esters on the central nervous system. Jpn. J. Pharmacol. 21, 797–810. doi: 10.1254/jjp.21.797
Takahashi, R. N., de Lima, T. C., and Morato, G. S. (1986). Pharmacological actions of tannic acid; II. Evaluation of CNS activity in animals. Planta Med. 52, 272–275. doi: 10.1055/s-2007-969150
Tjølsen, A., Berge, O. G., Hunskaar, S., Rosland, J. H., and Hole, K. (1992). The formalin test: an evaluation of the method. Pain 51, 5–17. doi: 10.1016/0304-3959(92)90003-T
Vogel, H. (2007). Drug Discovery and Evaluation: Pharmacological Assays. Berlin: Springer Science and Business Media.
Walker, C. I., Trevisan, G., Rossato, M. F., Franciscato, C., Pereira, M. E., Ferreira, J., et al. (2008). Antinociceptive activity of Mirabilis jalapa in mice. J. Ethnopharmacol. 120, 169–175. doi: 10.1016/j.jep.2008.08.002
Yoon, M. A., Jeong, T. S., Park, D. S., Xu, M. Z., Oh, H. W., Song, K. B., et al. (2006). Antioxidant effects of quinoline alkaloids and 2,4-Di-tert-butylphenol isolated from Scolopendra subspinipes. Biol. Pharm. Bull. 29, 735–739. doi: 10.1248/bpb.29.735
Yu, X., Zhao, M., Liu, F., Zeng, S., and Hu, J. (2013). Identification of 2, 3-dihydro-3, 5-dihydroxy-6-methyl-4H-pyran-4-one as a strong antioxidant in glucose–histidine Maillard reaction products. Food Res. Int. 51, 397–403. doi: 10.1016/j.foodres.2012.12.044
Zhao, G., Etherton, T. D., Martin, K. R., Vanden Heuvel, J. P., Gillies, P. J., West, S. G., et al. (2005). Anti-inflammatory effects of polyunsaturated fatty acids in THP-1 cells. Biochem. Biophy. Res. Commun. 336, 909–917. doi: 10.1016/j.bbrc.2005.08.204
Keywords: Phoenix sylvestris Roxb., Arecaceae, ethnomedicine, antinociceptive, CNS depressant, sedative, anxiolytic
Citation: Shajib MS, Akter S, Ahmed T and Imam MZ (2015) Antinociceptive and neuropharmacological activities of methanol extract of Phoenix sylvestris fruit pulp. Front. Pharmacol. 6:212. doi: 10.3389/fphar.2015.00212
Received: 21 July 2015; Accepted: 11 September 2015;
Published: 02 October 2015.
Edited by:
Adolfo Andrade-Cetto, Universidad Nacional Autónoma de México, MexicoReviewed by:
Keliang Xie, General Hospital of Tianjin Medical University, ChinaJelena Cvejic, University of Novi Sad, Serbia
Copyright © 2015 Shajib, Akter, Ahmed and Imam. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Mohammad Zafar Imam, Department of Pharmacy, Stamford University Bangladesh, 51 Siddeswari Road, Dhaka 1217, Bangladesh,emFmYXJpbWFtQGdtYWlsLmNvbQ==
 Md. Shafiullah Shajib
Md. Shafiullah Shajib Saleha Akter
Saleha Akter Tajnin Ahmed1
Tajnin Ahmed1 Mohammad Zafar Imam
Mohammad Zafar Imam