- 1Department of Cardiology, Hippokration Hospital, Athens, Greece
- 2School of Medicine, University of Patras, Patras, Greece
- 3Department of Cardiology, Asklepeion General Hospital, Athens, Greece
- 4Faculty of Medicine, School of Health Sciences, University of Ioannina, Ioannina, Greece
- 5Department of Cardiology, Elena Venizelou Hospital, Athens, Greece
- 6First University Department of Cardiology, Hippokration Hospital, University of Athens, Athens, Greece
Preeclampsia has been linked to high morbidity and mortality during pregnancy. However, no efficient pharmacological options for the prevention of this condition are currently available. Preeclampsia is thought to share several pathophysiologic mechanisms with cardiovascular disease, which has led to investigations for the potential role of statins (HMG CoA reductase inhibitors) in its prevention and early management. Pravastatin seems to have a safer pharmacokinetic profile compared to other statins, however, the existing preclinical evidence for its effectiveness in preeclampsia treatment has been mostly restricted to animal models. This review aims to summarize the current data and delineate the potential future role of statins in the prevention and management of preeclampsia.
Introduction
Preeclampsia is a pregnancy-related disorder with multi-organ involvement, mainly including the emergence of high blood pressure (BP) in the second trimester of pregnancy. Affecting about 1 in 12 pregnant women in the USA, preeclampsia is the most common hypertensive condition complicating pregnancy, as well as one of the major causes of maternal and fetal complications and death (Frishman et al., 2006; American College of Obstetricians and Gynecologists and Task Force on Hypertension in Pregnancy, 2013; Lo et al., 2013; Say et al., 2014). Although many therapeutic interventions have been proposed, mortality and morbidity rates still remain considerable. The aim of this review is to assess the possible role of statins in the therapeutic management of preeclampsia through exploration of the current preclinical and clinical data.
Clinical Presentation
Preeclampsia is characterized by the development of new onset hypertension (HTN) and the establishment of proteinuria. Other signs and symptoms that accompany the disease include: headache, visual disturbances, epigastric or abdominal pain, weakness, altered mental status, HELLP syndrome (Aloizos et al., 2013) dyspnea and edema (American College of Obstetricians and Gynecologists and Task Force on Hypertension in Pregnancy, 2013). Many factors have been found to increase the risk of preeclampsia and their association with the disease has been adequately elucidated. Previous preeclamptic pregnancy, family history of preeclampsia, late age of maternity (>40 years), multiple gestation, obesity, diabetes mellitus and history of thrombophilia have been identified as predisposing risk factors (American College of Obstetricians and Gynecologists and Task Force on Hypertension in Pregnancy, 2013). In particular, the presence of HTN and chronic renal impairment before gestation has been strongly correlated to the development of preeclampsia later during pregnancy (Foo et al., 2015).
Preeclampsia can result in a great number of severe and, in some cases, fatal short- and long-term consequences affecting both the mother and the fetus. Maternal complications include cardiometabolic disorders (diabetes, ischemic heart disease, metabolic syndrome), cerebrovascular disease (stroke, intracranial bleeding), neurologic abnormalities (eclamptic seizures) and renal impairment (Ramsay et al., 2003; Wilson et al., 2003; Haukkamaa et al., 2004; Funai et al., 2005). Fetal outcomes include intrauterine growth restriction (IUGR), prematurity and higher risk of developing HTN, obesity, metabolic syndrome, dyslipidemias, and cardiovascular disease (Lo et al., 2013; Nice guidelines, 2016).
Delivery of the fetus is the only efficient therapy (Everett et al., 2012; Gangooly et al., 2014; Nice guidelines, 2016). If the gestational age is less than 34 weeks and the BP can be sufficiently controlled with the absence of other symptoms, pregnancy can be prolonged in order to avoid prematurity complications for the fetus. The main therapeutic goal in preeclampsia is the management of HTN, aiming for SBP of 140–150 mmHg and DBP of 80–100 mmHg (Nice guidelines, 2016). Oral antihypertensive therapy including a-methyldopa, calcium channel blockers, b-blockers and labetalol, coupled with anti-platelet agents and magnesium sulfate are considered as a therapy in hypertensive disorders in order to limit maternal and fetal complications (Sandrim et al., 2008; Nice guidelines, 2016).
Pathophysiologic Mechanisms
Even though the precise etiology of preeclampsia remains obscure, the main pathophysiological mechanisms include maternal endothelial dysfunction (Maynard et al., 2003; Levine et al., 2004) and placental vascular impairment (Redman and Sargent, 2005; Burton et al., 2009; Brosens et al., 2011). Endothelial dysfunction has been correlated with abnormalities in pregnant lipid profile, with high levels of triglycerides. LDL oxidation, oxidative stress and increase of reactive oxygen species (ROS) play a key role in placental vascular dysfunction (Costantine and Cleary, 2013). In early onset disease, an adverse remodeling of spiral arteries, in which endothelial cells are partially replaced by cytotrophoblast cells, may cause malperfusion of the placenta (Burton et al., 2009; Brosens et al., 2011). As a result, ischemic injury and consequent inflammatory response will be generated.
The integrity of endothelial cells is crucial for the maintenance of angiogenic balance. Any endothelial damage such as deficient spiral artery remodeling mentioned above, can lead to placental oxidative stress and imbalance in the production of vasoconstrictive and vasodilative factors. Anti-angiogenic factors, such as soluble Fms-like tyrosine kinase (sFlt-1) and soluble endoglin (sEng), have been shown to increase in preeclamptic gestations (Hertig et al., 2004; Levine et al., 2004; Wang et al., 2009). Specifically, sFlt-1 blocks the binding of vascular endothelial growth factor (VEGF) and placental growth factor (PIGF) to their receptors, resulting in reduction of nitric oxide (NO) synthesis through inactivation of endothelial NO synthase (eNOs) (Shen et al., 1999; Noris et al., 2005). The low levels of NO invert the beneficial impact of vasodilatation in preventing placental ischemia (Sladek et al., 1997; Venkatesha et al., 2006; Ramma and Ahmed, 2014). Inhibition of NO production can be caused through an asymmetric dimethylarginine (ADMA)-mediated eNOS inactivation (Ehsanipoor et al., 2013). Another pathophysiologic mechanism in preeclampsia is the hemeoxygenase/carbon monoxide (HO-1/CO) pathway (Ozen et al., 2015; Zenclussen et al., 2015). Hemeoxygenase 1 has anti-inflammatory and vasoprotective properties and its down regulation seems to lead to sFlt-1 and sEng overexpression. The result of this angiogenic imbalance is the endothelial dysfunction and the appearance of the maternal clinical syndrome.
The Potential Role of Statins
Statins inhibit HMG-CoA reductase leading to increase of LDL receptors (LDLr) and thus, to the reduction of plasma cholesterol levels. The most serious adverse effects include rhabdomyolysis and transaminase elevation but their incidence is about 1%. Statin administration has been shown to be safe in pregnancy (Kazmin et al., 2007; Ofori et al., 2007; Taguchi et al., 2008) presenting a protective role in endothelial function (Laufs et al., 1997). Beyond lowering cholesterol levels, they also have other pleiotropic actions such as antioxidant, anti-inflammatory and anti-thrombogenic effects. They have been shown to increase eNOs activity in vitro and in vivo, resulting into the overexpression of vasodilating factor NO (Laufs et al., 1997, 1998; Endres et al., 1998; Fox et al., 2011). Moreover, statins induce the activity of HO-1/CO pathway and reduce platelet aggregation (Grosser et al., 2004; Lee et al., 2004) (Figure 1).
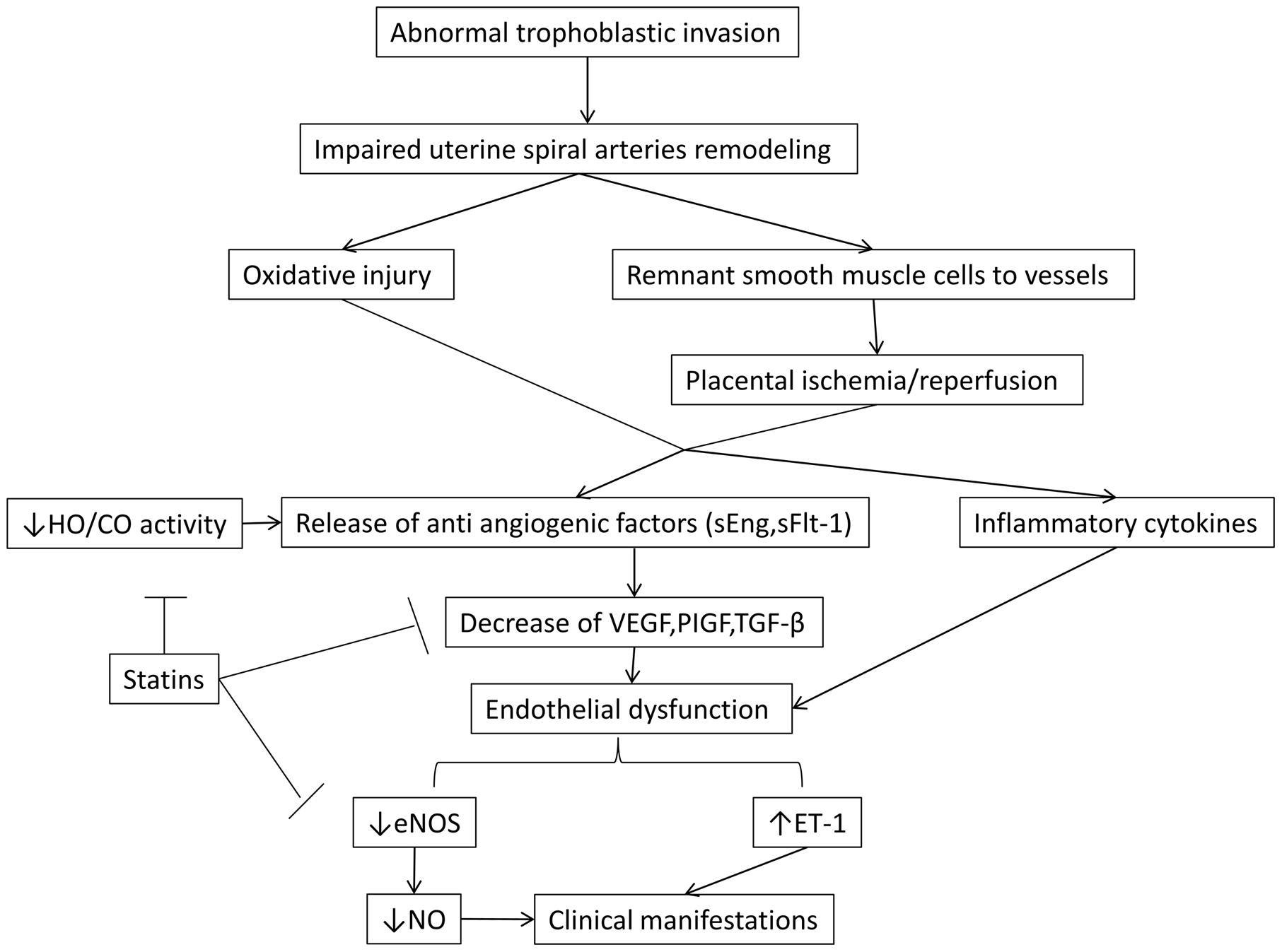
FIGURE 1. The malfunction in the invasion of trophoblastic cells is thought to be fundamental in the cascade of preeclampsia. Also, the role of NO and HO/CO activity in the release of antiangiogenic factors sFlt-1 and sEng is crucial in the pathogenesis of the disease. By blocking these regulatory points, statins beneficially allow the synthesis of NO by eNOS, leading to vasodilation and decrease of the secretion of antiangiogenic factors. The latter effect results in the increase of VEGF and PIGF which are key components in placental and endothelial integrity. eNOS, endothelial nitric oxidase synthase; ET -1, endothelin 1; HO-1/CO, hemeoxygenase -1/ carbon monoxide; NO, nitric oxide; PIGF, placental growth factor; sEng, soluble endoglin; sFlt-1, soluble Fms- like tyrosine kinase; TGF – β, transforming growth factor β; VEGF, vascular endothelial growth factor.
The preclinical evidence supporting the use of pravastatin in the treatment of preeclampsia is currently only limited to animal models and it has focused on simulation of endothelial pathophysiologic changes in preeclamptic mice. In these studies pregnant mice were injected with adenovirus carrying sFlt-1, a factor responsible for many clinical manifestations of preeclampsia. Subsequently, a statin was administered and preeclampsia-related markers, such as sFlt-1 and sEng were monitored. Pravastatin was the most commonly used agent in the majority of preclinical animal models due to its biochemical profile, namely hydrophilic and hepatoselective properties.
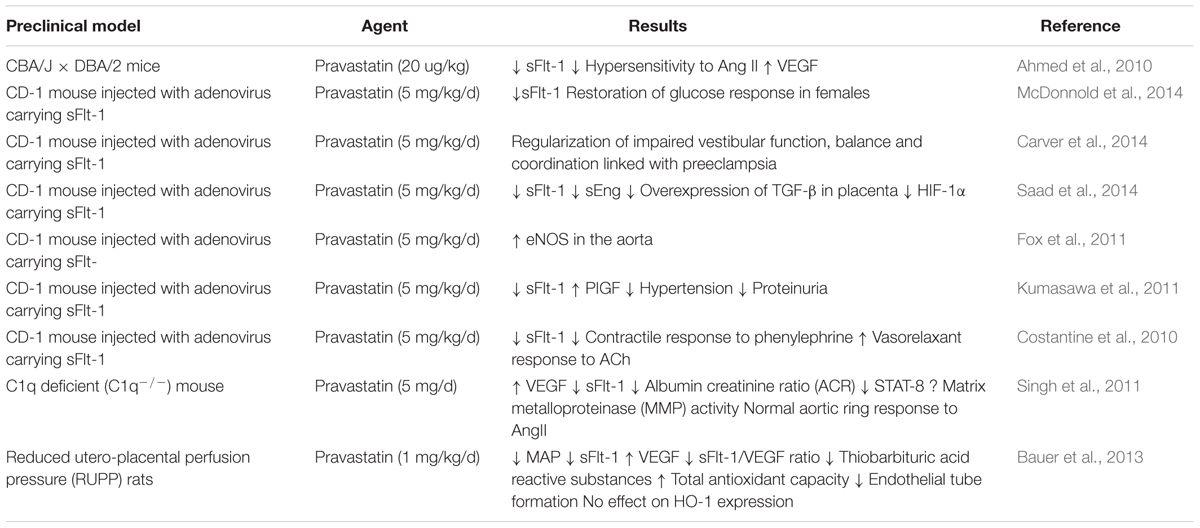
Ahmed et al. (2010) focused on depicting the features of human preeclampsia at rodents, such as albuminuria and endotheliosis, by presenting their prototype CBA/J × DBA/2 mice. When treated with pravastatin CBA/J × DBA/2 demonstrated decreased levels of sFlt-1, elevated levels of VEGF and decreased hypersensitivity to Angiotensin II. Saad et al. (2014) showed that pravastatin diminished the increase of sFlt-1 and sEng in their mice model and observed a down regulation in hypoxia inducible factor 1a (HIF- 1a) and in placental TGF-β. Fox et al. (2011) concentrated on the impact of pravastatin in vascular function by measuring the levels of eNOS protein. They reached to the conclusion that pravastatin increased the levels of eNOS in the aorta and reduced the levels of sFlt-1. The animal studies of Kumasawa et al. (2011) were of paramount importance in depicting the role of pravastatin in preeclampsia. They focused on the augmentation of PIGF levels, which seemed to play an important role in improving glomerular function, reducing BP and sFlt-1 levels. Costantine et al. (2010) affirms that pravastatin administration improved endothelial function by reducing sFlt-1 and amplifying NOS expression. They also suggested that pravastatin plays an important role in prevention of IUGR in offspring’s. Except from the CBA/J × DBA/2 model, Singh et al. (2011) presented a C1q-deficient mouse model that mimics human PE. When treated with pravastatin, the pregnant C1q-deficient mice restored their placental blood flow and the angiogenic balance. Bauer et al. (2013) with their experiments in rat models with placental ischemia- induced hypertension, diverged themselves from the other investigators, not only for his mouse model but also for noticing some defects of pravastatin. They reported elevated levels of VEGF and decreased levels of sFlt-1, complying with the results of other investigators. Although they observed a restoration of antioxidant capacity, there wasn’t a restoration in angiogenic potential of serum as estimated by an endothelial tube formation assay, which is an essential disadvantage. Table 1 summarizes the results of the animal studies reported above.
Studies in mice focused also on the neonatal/offspring benefit. McDonnold et al. (2014), apart from reduced sFlt-1 levels with pravastatin treatment, underlined the role of maternal treatment with pravastatin in restoration of glucose response and post-partum growth. They suggested that the effect of pravastatin in metabolic changes took place only in female offspring’s. Carver et al. (2014) supported that pravastatin administration during pregnancy in murine models ameliorated the neuromotor dysfunction by optimizing vestibular function and balance. They also showed that statins promoted the expression of genes pertained to myelination and oxidative stress, especially in male offspring’s.
Moreover, a few case reports in humans have been recently published. A case of a 30-year-old woman with known antiphospholipid syndrome, thrombosis and preeclampsia at the 23rd week of gestation has been described (Lefkou et al., 2014). The patient was treated with combined medication including pravastatin (20 mg), enoxaparin (0.4 BD) and aspirin (100 mg OD). BP and proteinuria improved and normalization of previous pathological Doppler findings of the uterine arteries were noticed. In another study (Brownfoot et al., 2015) of four patients, presented with hypertension, proteinuria, preeclampsia, and growth restricted fetuses before the 30th week of gestation, the daily administration of pravastatin (40 mg), resulted in reduced levels of sFlt-1 and stabilization of impaired glomerular function and hypertension.
A double blind, randomized placebo-controlled, multicenter trial of pravastatin to ameliorate early onset pre-eclampsia (StAmP) is currently recruiting and investigators focus on the impact of pravastatin in the reduction of anti-angiogenic markers in women with early preeclampsia (Clinical trials register.eu, 2009). Additionally, in the Pravastatin for Prevention of Preeclampsia trial, a phase I interventional trial under progress, preliminary data suggest safe pharmacokinetic profile of pravastatin with no serious adverse effects for the fetus or the mother in high-risk pregnancy (Clinical Trials.gov, 2016; Costantine et al., 2016).
Conclusion
Preeclampsia remains a pregnancy-related health issue with severe and life-threatening complications and clinicians should be alerted for early recognition, prompt diagnosis, prevention and effective treatment. Preliminary data suggest that statins can be a promising therapeutic alternative for the prevention and treatment of preeclampsia, however, larger clinical studies are still required.
Author Contributions
All authors listed, have made substantial, direct and intellectual contribution to the work, and approved it for publication.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
Ahmed, A., Singh, J., Khan, Y., Seshan, S. V., and Girardi, G. (2010). A new mouse model to explore therapies for preeclampsia. PLoS ONE 5:e13663. doi: 10.1371/journal.pone.0013663
Aloizos, S., Seretis, C., Liakos, N., Aravosita, P., Mystakelli, C., Kanna, E., et al. (2013). HELLP syndrome: understanding and management of a pregnancy-specific disease. J Obstet. Gynaecol. 33, 331–337. doi: 10.3109/01443615.2013.775231
American College of Obstetricians and Gynecologists and Task Force on Hypertension in Pregnancy (2013). Hypertension in pregnancy. Report of the American College of Obstetricians and Gynecologists’ Task Force on Hypertension in Pregnancy. Obstet. Gynecol. 122, 1122–1131.
Bauer, A. J., Banek, C. T., Needham, K., Gillham, H., Capoccia, S., Regal, J. F., et al. (2013). Pravastatin attenuates hypertension, oxidative stress, and angiogenic imbalance in rat model of placental ischemia-induced hypertensionNovelty and significance. Hypertension 61, 1103–1110. doi: 10.1161/HYPERTENSIONAHA.111.00226
Brosens, I., Pijnenborg, R., Vercruysse, L., and Romero, R. (2011). The “Great Obstetrical Syndromes” are associated with disorders of deep placentation. Am. J. Obstet. Gynecol. 204, 193–201. doi: 10.1016/j.ajog.2010.08.009
Brownfoot, F. C., Tong, S., Hannan, N. J., Binder, N. K., Walker, S. P., Cannon, P., et al. (2015). Effects of pravastatin on human placenta, endothelium, and women with severe preeclampsia. Hypertension 66, 687–697. doi: 10.1161/HYPERTENSIONAHA.115.05445
Burton, G. J., Woods, A. W., Jauniaux, E., and Kingdom, J. C. P. (2009). Rheological and physiological consequences of conversion of the maternal spiral arteries for uteroplacental blood flow during human pregnancy. Placenta 30, 473–482. doi: 10.1016/j.placenta.2009.02.009
Carver, A. R., Andrikopoulou, M., Lei, J., Tamayo, E., Gamble, P., Hou, Z., et al. (2014). Maternal pravastatin prevents altered fetal brain development in a preeclamptic CD-1 mouse model. PLoS ONE 9:e100873. doi: 10.1371/journal.pone.0100873
Clinical Trials. gov (2016). Pravastatin for the Prevention of Preeclampsia in High-Risk Women: A Phase I Pilot Study [Pravastatin for Prevention of Preeclampsia]. Available at: https://clinicaltrials.gov/ct2/show/NCT01717586?term$=$NCT01717586&rank$=$1
Clinicaltrialsregister.eu (2009). A Proof of Principle, Double-Blind, Randomised Placebo-Controlled, Multi-Centre Trial of pravaStatin to Ameliorate Early Onset Pre-eclampsia [StAmP]. Available at: https://www.clinicaltrialsregister.eu/ctr-search/trial/2009-012968-13/GB
Costantine, M. M., and Cleary, K. (2013). Pravastatin for the prevention of preeclampsia in high-risk pregnant women. Obstet. Gynecol. 121, 349–353. doi: 10.1016/j.ajog.2015.12.038
Costantine, M. M., Cleary, K., Hebert, M. F., Ahmed, M. S., Brown, L. M., Ren, Z., et al. (2016). for the Eunice Kennedy Shriver National Institute of Child Health and Human Development Obstetric-Fetal Pharmacology Research Units Network (ORPU), Safety and Pharmacokinetics of Pravastatin used for the prevention of preeclampsia in high-risk pregnant women: a pilot randomized controlled trial. Am. J. Obstet. Gynecol. 214, 720.e1–720. doi: 10.1016/j.ajog.2015.12.038
Costantine, M. M., Tamayo, E., Lu, F., Bytautiene, E., Longo, M., Hankins, G. D., et al. (2010). Using pravastatin to improve the vascular reactivity in a mouse model of soluble fms-like tyrosine kinase-1-induced preeclampsia. Obstet. Gynecol. 116, 114–120. doi: 10.1097/AOG.0b013e3181e10ebd
Ehsanipoor, R. M., Fortson, W., Fitzmaurice, L. E., Liao, W. X., Wing, D. A., Chen, D. B., et al. (2013). Nitric oxide and carbon monoxide production and metabolism in preeclampsia. Reprod. Sci. 20, 542–548. doi: 10.1177/1933719112459231
Endres, M., Laufs, U., Huang, Z., Nakamura, T., Huang, P., Moskowitz, M. A., et al. (1998). Stroke protection by 3-hydroxy-3-methylglutaryl (HMG)-CoA reductase inhibitors mediated by endothelial nitric oxide synthase. Proc. Natl. Acad. Sci. U.S.A. 95, 8880–8885. doi: 10.1073/pnas.95.15.8880
Everett, T. R., Wilkinson, I. B., and Lees, C. C. (2012). Drug development in preeclampsia: a ‘no go’ area? J. Matern. Fetal Neonatal Med. 25, 50–52. doi: 10.3109/14767058.2011.557791
Foo, L., Tay, J., Lees, C. C., McEniery, C. M., and Wilkinson, I. B. (2015). Hypertension in pregnancy: natural history and treatment options. Curr. Hypertens. Rep. 17, 36. doi: 10.1007/s11906-015-0545-1
Fox, K. A., Longo, M., Tamayo, E., Kechichian, T., Bytautiene, E., Hankins, G. D., et al. (2011). Effects of pravastatin on mediators of vascular function in a mouse model of soluble Fms-like tyrosine kinase-1-induced preeclampsia. Am. J. Obstet. Gynecol. 205, 366.e1–5. doi: 10.1016/j.ajog.2011.06.083
Frishman, W. H., Veresh, M., Schlocker, S. J., and Tejani, N. (2006). Pathophysiology and medical management of systemic hypertension in preeclampsia. Curr. Hypertens. Rep. 8, 502–511. doi: 10.1007/s11906-006-0030-y
Funai, E. F., Friedlander, Y., Paltiel, O., Tiram, E., Xue, X., Deutsch, L., et al. (2005). Long-term mortality after preeclampsia. Epidemiology 16, 206–215. doi: 10.1097/01.ede.0000152912.02042.cd
Gangooly, S., Muttukrishna, S., and Jauniaux, E. (2014). In-vitro study of the effect of anti-hypertensive drugs on placental hormones and angiogenic proteins synthesis in pre-eclampsia. PLoS ONE. 9:e107644. doi: 10.1371/journal.pone.0107644
Grosser, N., Hemmerle, A., Berndt, G., Erdmann, K., Hinkelmann, U., Schürger, S., et al. (2004). The antioxidant defense protein heme oxygenase 1 is a novel target for statins in endothelial cells. Free Radic. Biol. Med. 37, 2064–2071. doi: 10.1016/j.freeradbiomed.2004.09.009
Haukkamaa, L., Salminen, M., Laivuori, H., Leinonen, H., Hiilesmaa, V., and Kaaja, R. (2004). Risk for subsequent coronary artery disease after preeclampsia. Am. J. Cardiol. 93, 805–808. doi: 10.1016/j.amjcard.2003.11.065
Hertig, A., Berkane, N., Lefevre, G., Toumi, K., Marti, H. P., Capeau, J., et al. (2004). Maternal serum sFlt1 concentration is an early and reliable predictive marker of preeclampsia. Clin. Chem. 2004, 1702–1703. doi: 10.1373/clinchem.2004.036715
Kazmin, A., Garcia-Bournissen, F., and Koren, G. (2007). Risks of statin use during pregnancy: a systematic review. J. Obstet. Gynaecol. Can. 2007, 906–908. doi: 10.1016/S1701-2163(16)32656-1
Kumasawa, K., Ikawa, M., Kidoya, H., Hasuwa, H., Saito-Fujita, T., Morioka, Y., et al. (2011). Pravastatin induces placental growth factor (PGF) and ameliorates preeclampsia in a mouse model. Proc. Natl. Acad. Sci. U.S.A. 108, 1451–1455. doi: 10.1073/pnas.1011293108
Laufs, U., Fata, V. L., and Liao, J. K. (1997). Inhibition of 3-hydroxy-3-methylglutaryl (HMG)-CoA reductase blocks hypoxia-mediated down-regulation of endothelial nitric oxide synthase. J. Biol. Chem. 272, 31725–31729. doi: 10.1074/jbc.272.50.31725
Laufs, U., La Fata, V., Plutzky, J., and Liao, J. K. (1998). Upregulation of endothelial nitric oxide synthase by HMG CoA reductase inhibitors. Circulation 97, 1129–1135. doi: 10.1161/01.CIR.97.12.1129
Lee, T. S., Chang, C. C., Zhu, Y., and Shyy, J. Y. (2004). Simvastatin induces heme oxygenase-1: a novel mechanism of vessel protection. Circulation 2004, 1296–1302. doi: 10.1161/01.CIR.0000140694.67251.9C
Lefkou, E., Mamopoulos, A., Fragakis, N., Dagklis, T., Vosnakis, C., Nounopoulos, E., et al. (2014). Clinical improvement and successful pregnancy in a preeclamptic patient with antiphospholipid syndrome treated with pravastatin. Hypertension 63, e118–9. doi: 10.1161/HYPERTENSIONAHA.114.03115
Levine, R. J., Maynard, S. E., Qian, C., Lim, K. H., England, L. J., Yu, K. F., et al. (2004). Circulating angiogenic factors and the risk of preeclampsia. N. Engl. J. Med. 350, 672–683. doi: 10.1056/NEJMoa031884
Lo, J. O., Mission, J. F., and Caughey, A. B. (2013). Hypertensive disease of pregnancy and maternal mortality. Curr. Opin. Obstet. Gynecol. 25, 124–132. doi: 10.1097/GCO.0b013e32835e0ef5
Maynard, S. E., Min, J. Y., Merchan, J., Lim, K. H., Li, J., Mondal, S., et al. (2003). Excess placental soluble fms-like tyrosine kinase 1 (sFlt1) may contribute to endothelial dysfunction, hypertension, and proteinuria in preeclampsia. J. Clin. Invest. 111, 649–658. doi: 10.1172/JCI17189
McDonnold, M., Tamayo, E., Kechichian, T., Gamble, P., Longo, M., Hankins, G. D., et al. (2014). The effect of prenatal pravastatin treatment on altered fetal programming of postnatal growth and metabolic function in a preeclampsia-like murine model. Am. J. Obstet. Gynecol. 210, 542.e1–7. doi: 10.1016/j.ajog.2014.01.010
Nice guidelines (2016). Hypertension in Pregnancy: Diagnosis and Management | 1-Guidance | Guidance and guidelines | NICE. [cited 2016 May 27]. Available at: https://www.nice.org.uk/guidance/cg107/chapter/1-guidance
Noris, M., Perico, N., and Remuzzi, G. (2005). Mechanisms of disease: pre-eclampsia. Nat. Clin. Pract. Nephrol. 2005, 98–114. doi: 10.1038/ncpneph0035
Ofori, B., Rey, E., and Berard, A. (2007). Risk of congenital anomalies in pregnant users of statin drugs. Br. J. Clin. Pharmacol. 2007, 496–509. doi: 10.1111/j.1365-2125.2007.02905.x
Ozen, M., Zhao, H., Lewis, D. B., Wong, R. J., and Stevenson, D. K. (2015). Heme oxygenase and the immune system in normal and pathological pregnancies. Front. Pharmacol. 6:84. doi: 10.3389/fphar.2015.00084
Ramma, W., and Ahmed, A. (2014). Therapeutic potential of statins and the induction of heme oxygenase-1 in preeclampsia. J. Reprod. Immunol. 101–102, 153–160. doi: 10.1016/j.jri.2013.12.120
Ramsay, J. E., Stewart, F., Green, I. A., and Sattar, N. (2003). Microvascular dysfunction: a link between pre-eclampsia and maternal coronary heart disease. BJOG 2003, 1029–1031. doi: 10.1111/j.1471-0528.2003.02069.x
Redman, C. W., and Sargent, I. L. (2005). Latest advances in understanding preeclampsia. Science 308, 1592–1597. doi: 10.1126/science.1111726
Saad, A. F., Kechichian, T., Yin, H., Sbrana, E., Longo, M., Wen, M., et al. (2014). Effects of pravastatin on angiogenic and placental hypoxic imbalance in a mouse model of preeclampsia. Reprod. Sci. 21, 138–145. doi: 10.1177/1933719113492207
Sandrim, V. C., Palei, A. C., Metzger, I. F., Gomes, V. A., Cavalli, R. C., and Tanus-Santos, J. E. (2008). Nitric oxide formation is inversely related to serum levels of antiangiogenic factors soluble fms-like tyrosine kinase-1 and soluble endogline in preeclampsia. Hypertension 52, 402–407. doi: 10.1161/HYPERTENSIONAHA.108.115006
Say, L., Chou, D., Gemmill, A., Tunçalp,Ö, Moller, A. B., Daniels, J., et al. (2014). Global causes of maternal death: a WHO systematic analysis. Lancet Global Health 2, e323–e333. doi: 10.1016/S2214-109X(14)70227-X
Shen, B. Q., Lee, D. Y., and Zioncheck, T. F. (1999). Vascular endothelial growth factor governs endothelial nitric-oxide synthase expression via a KDR/Flk-1 Receptor and a protein kinase C signaling pathway. J. Biol. Chem. 1999, 33057–33063. doi: 10.1074/jbc.274.46.33057
Singh, J., Ahmed, A., and Girardi, G. (2011). Role of complement component C1q in the onset of preeclampsia in mice. Hypertension 58, 716–724. doi: 10.1161/HYPERTENSIONAHA.111.175919
Sladek, S. M., Magness, R. R., and Conrad, K. P. (1997). Nitric oxide and pregnancy. Am. J. Physiol. 1997, R441–R463.
Taguchi, N., Rubin, E. T., and Hosokawa, A. (2008). Prenatal exposure to HMGCoA reductase inhibitor: effects on fetal and neonatal outcomes. Reprod. Toxicol. 2008, 175–177. doi: 10.1016/j.reprotox.2008.06.009
Venkatesha, S., Toporsian, M., Lam, C., Hanai, J., Mammoto, T., Kim, Y. M., et al. (2006). Soluble endoglin contributes to the pathogenesis of preeclampsia. Nat. Med. 12, 642–649. doi: 10.1038/nm1429
Wang, A., Rana, S., and Karumanchi, S. A. (2009). Preeclampsia: the role of angiogenic factors in its pathogenesis. Physiology (Bethesda) 2009, 147–158. doi: 10.1152/physiol.00043.2008
Wilson, B. J., Watson, M. S., Prescott, G. J., Sunderland, S., Campbell, D. M., Hannaford, P., et al. (2003). Hypertensive diseases of pregnancy and risk of hypertension and stroke in later life: results from cohort study. BMJ 326, 845. doi: 10.1136/bmj.326.7394.845
Keywords: preeclampsia, hypertension, statins, pravastatin, prevention
Citation: Katsi V, Georgountzos G, Kallistratos MS, Zerdes I, Makris T, Manolis AJ, Nihoyannopoulos P and Tousoulis D (2017) The Role of Statins in Prevention of Preeclampsia: A Promise for the Future? Front. Pharmacol. 8:247. doi: 10.3389/fphar.2017.00247
Received: 04 September 2016; Accepted: 19 April 2017;
Published: 05 May 2017.
Edited by:
Jean-Paul Deslypere, Proclin Therapeutic Research Pte Ltd, SingaporeReviewed by:
Sunita Nair, Capita India Pvt Ltd, IndiaEric George, University of Mississippi Medical Center School of Dentistry, USA
Copyright © 2017 Katsi, Georgountzos, Kallistratos, Zerdes, Makris, Manolis, Nihoyannopoulos and Tousoulis. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Manolis S. Kallistratos, a2FsbGlzdHJhdDE5NzJAZ21haWwuY29t
 Vasiliki Katsi
Vasiliki Katsi Georgios Georgountzos
Georgios Georgountzos Manolis S. Kallistratos
Manolis S. Kallistratos Ioannis Zerdes
Ioannis Zerdes Thomas Makris5
Thomas Makris5