- 1Department of Endocrinology, Guang’anmen Hospital, China Academy of Chinese Medical Sciences, Beijing, China
- 2Graduate School, Beijing University of Chinese Medicine, Beijing, China
Insulin resistance is a condition in which insulin sensitivity is reduced and the insulin signaling pathway is impaired. Although often expressed as an increase in insulin concentration, the disease is characterized by a decrease in insulin action. This increased workload of the pancreas and the consequent decompensation are not only the main mechanisms for the development of type 2 diabetes (T2D), but also exacerbate the damage of metabolic diseases, including obesity, nonalcoholic fatty liver disease, polycystic ovary syndrome, metabolic syndrome, and others. Many clinical trials have suggested the potential role of herbs in the treatment of insulin resistance, although most of the clinical trials included in this review have certain flaws and bias risks in their methodological design, including the generation of randomization, the concealment of allocation, blinding, and inadequate reporting of sample size estimates. These studies involve not only the single-flavored herbs, but also herbal formulas, extracts, and active ingredients. Numerous of in vitro and in vivo studies have pointed out that the role of herbal medicine in improving insulin resistance is related to interventions in various aspects of the insulin signaling pathway. The targets involved in these studies include insulin receptor substrate, phosphatidylinositol 3-kinase, glucose transporter, AMP-activated protein kinase, glycogen synthase kinase 3, mitogen-activated protein kinases, c-Jun-N-terminal kinase, nuclear factor-kappaB, protein tyrosine phosphatase 1B, nuclear factor-E2-related factor 2, and peroxisome proliferator-activated receptors. Improved insulin sensitivity upon treatment with herbal medicine provides considerable prospects for treating insulin resistance. This article reviews studies of the target mechanisms of herbal treatments for insulin resistance.
Introduction
Insulin resistance (IR) is a pathological condition in which target tissues (primarily skeletal muscle, liver, and adipose tissue) have an impaired biological response to insulin stimulation. During IR, the body’s compensatory release of excess insulin to maintain blood sugar stability causes hyperinsulinemia that can progress to type 2 diabetes mellitus (T2D). Prospective studies have highlighted the importance of IR in the pathogenesis of T2D and suggest that IR is the best predictor of future T2D diagnosis (Lillioja et al., 1993). IR and obesity are connected with chronic inflammation in metabolic tissues such as adipose tissue and the liver (Winer et al., 2016). Some studies have pointed out that body mass index is positively associated with IR (Li W. et al., 2014) and inflammation in visceral adipose tissue is a main driver of IR (Lumeng et al., 2007). Closely linked to the epidemic of obesity (Ng et al., 2014), the number of adults with diabetes increased from 108 million in 1980 to 422 million in 2014 (Zhou et al., 2016), and this figure is projected to rise to 642 million people by 2040. There are many vascular and nerve-related complications in diabetes such as diabetes-induced dysregulation of cardiac function, instability of microvasculature of the heart, and increased risk for heart failure (Riehle and Abel, 2016; Levelt et al., 2016; Hinkel et al., 2017). The risk of dementia, Alzheimer’s disease, and cognitive decline are elevated in people with IR (Biessels et al., 2006; Willette et al., 2015; Kullmann et al., 2016) and T2D; the global prevalence of diabetic foot pathologies is 6.3%, and 12.9 to 49.0 million people worldwide have a history of foot ulceration (Armstrong et al., 2017; Zhang P. et al., 2017). These complications bring a tremendous medical and socioeconomic burden. IR is associated with increased risk for other associated disorders, including polycystic ovary syndrome (PCOS), hepatitis C virus, nonalcoholic fatty liver disease (NAFLD), and metabolic syndrome (Diamanti-Kandarakis and Dunaif, 2012; Meex and Watt, 2017; Aytug et al., 2003). Improving IR may provide a therapeutic strategy for controlling T2D, obesity, and many other diseases. Current interventions for IR include intensive lifestyle interventions, thiazolidinedione, DPP-4 inhibitors, and metformin. However, IR is not well controlled and poses a threat to modern society (Kahn et al., 2006). Some herbal medicines such as Coptis chinensis Franch (Zhen et al., 2011), Ganoderma lucidum, and Panax ginseng C. A. Mey result in enhanced insulin sensitivity through modulation of diverse physiological and cellular pathways (Chang et al., 2015; Martel et al., 2017; Bai et al., 2018). For centuries, natural herbs and herbal formulae derived from systemic traditional Chinese medicine theory and practice have been used to treat many kinds of ailments in China. At present, Chinese medicine has received strong support from the World Health Organization and will be included in Chapter 26 of the 11th edition of the Global Medical Program. Chinese medicine also provides treatments for obesity and T2D (D, 2018). In the third century BC, Huang Di Nei Jing, the most classic book of Chinese medicine, recorded similar diseases related to diabetes and obesity and provided treatment principles. Now, traditional Chinese medicine is widely used to clinically treat IR. In this review, we explored whether herbs and their formulations or monomers can improve IR and the mechanisms of herbal compounds that increase insulin sensitivity.
Methodology
According to the Pharmacopoeia of the People’s Republic of China that was revised by the China Food and Drug Administration in 2015, herbal medicine is defined as therapy using herbs and materials derived from botanical herbal products and mineral and animal sources. Interventions that were used in this study include single-flavored herbs and their extracts, active ingredients, and herbal formulas. There were no geographical restrictions on the herbs included.
We reviewed literature (from PubMed) published between July 8, 2013 and July 6, 2018 on IR that had been treated with herbal medicine. The following combination of terms were used as search keywords: “herbal,” “phytochemical,” “phytomedicine,” “natural product,” and “insulin resistance” or “IR.” The search did not exclude articles based on language or status of the publication.
The specified exclusion criteria include: a) case reports, case series, editorials, reviews; b) interventions containing ingredients other than herbs; and c) relevant indicators of IR, such as homeostatic model assessment of IR (HOMA-IR), and IR-index, not involved in the primary and secondary outcomes of clinical trials.
Results
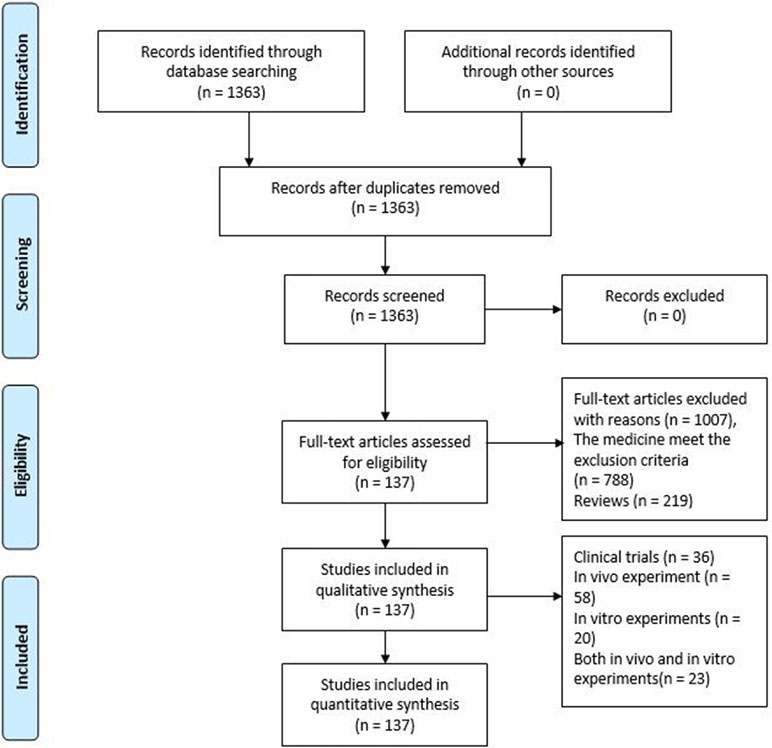
The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) flow chart (Figure 1) of article processing shows that our search yielded 1,363 articles, and 1,007 articles were excluded based on the exclusion criteria. After excluding these 1,007 articles, we included 137 articles, including 36 clinical trials, 58 in vivo experiments, 20 in vitro experiments, and 23 that were a combination of both in vitro and in vivo experiments. Based on further reading of this literature, we divided 101 in vivo and in vitro experiments into three parts based on the interventional drug used: active ingredients (31 articles), natural products (38 articles), and herbal formulas (32 articles). The results suggest that most clinical trials (30 articles) indicate that herbal active ingredients, natural products, and herbal formulas, such as JTTZ formula, Jinlida, and Curcumin, have a therapeutic effects on IR. There were a few clinical trials (six articles) that did not support the above results, such as those that used marjoram tea, hydroalcoholic extract of Juglans regia (walnut) leaves, Fraxinus excelsior L. seeds/fruit extract, garlic extract, bee propolis, red wine polyphenols. On the other hand, we concluded that the effects of herbal medicine on IR may be related to 11 important target molecules that affect insulin signaling, such as insulin receptor substrate, phosphatidylinositol 3-kinase, and glucose transporter.
Clinical Trials to Improve Insulin Resistance
In clinical trials, participants in the treatment group should be restricted to using herbs alone. If medication other than herbs are being used, then the medication must be administered to both the treatment group and the control group. Participants in the control group should receive hypoglycemic agents, placebo, or life interventions. There is no limit to the number of predetermined herbs, recipes, sample sizes, or duration of treatment.
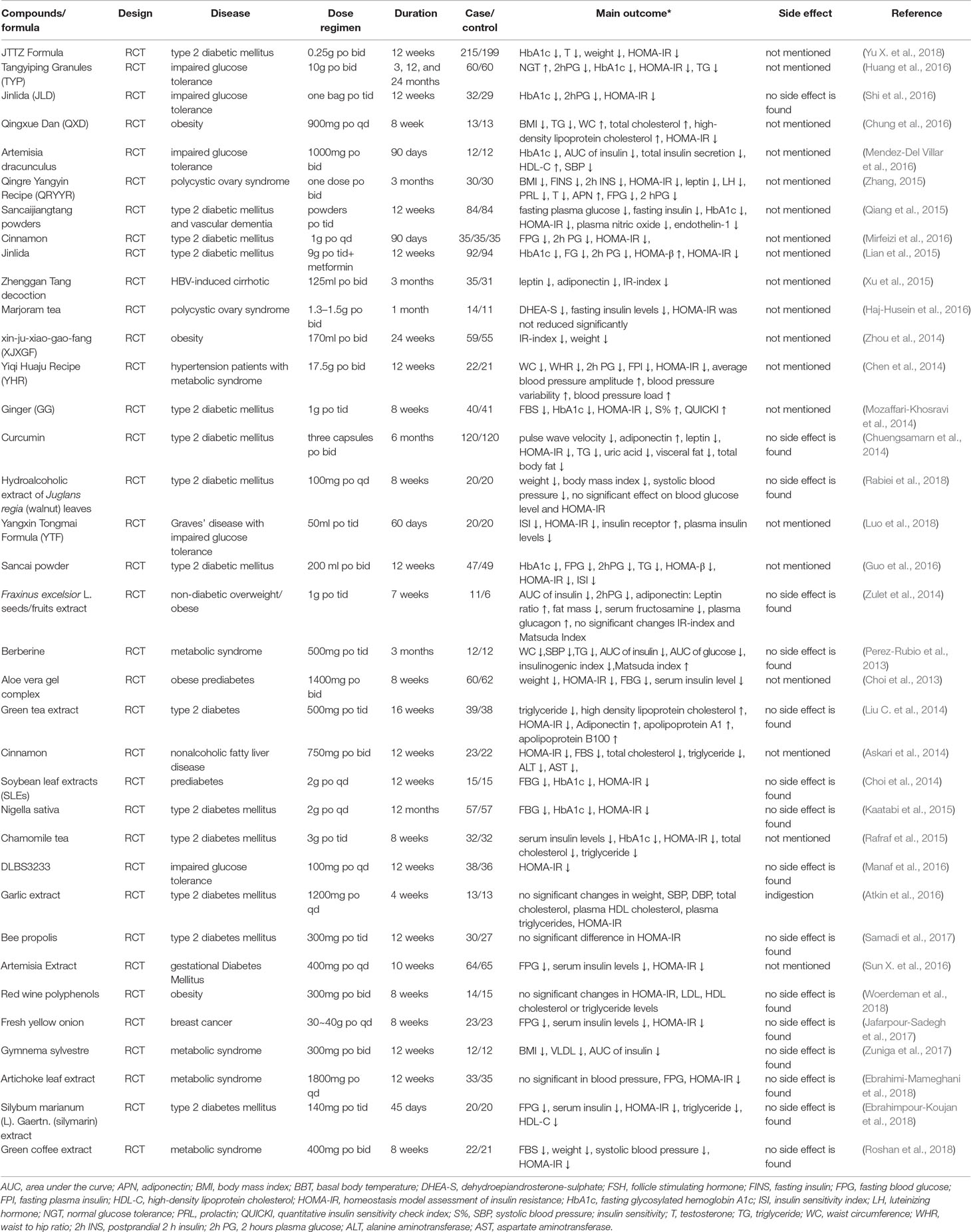
IR can be accurately assessed by clinical examination. It is generally believed that hyperinsulinemic-euglycemic clamps are the “gold standard” for determining IR, but the complex nature and inherent potential hazards of this technique limit its routine use (Park et al., 2015). Commonly used alternatives are primarily HOMA-IR and quantitative insulin sensitivity check index (QUICKI), which use fasting insulin and glucose concentrations to assess IR and correlate with the results of the clamp study (Matthews et al., 1985). In the 36 clinical trials (Table 1) in the above table, most of the drugs examined were herbal formulas, including herbal extracts and active ingredients, and the diseases of concern included T2D, metabolic syndrome, obesity, impaired glucose tolerance, PCOS, and cirrhosis. Most studies have shown that herbal medicine can not only reduce IR but also improve blood sugar, blood lipids, glycosylated hemoglobin, and other biochemical indicators. Indicators of IR in these studies include HOMA-IR, QUICKI, area under the curve of insulin, and IR index. Among them, HOMA-IR was the indicator and was used in 29 studies (29/36, 80%). In order to objectively observe the therapeutic effect of herbs on IR, the changes in IR evaluation indicators before and after treatment in 36 clinical trials are listed in Table 2.
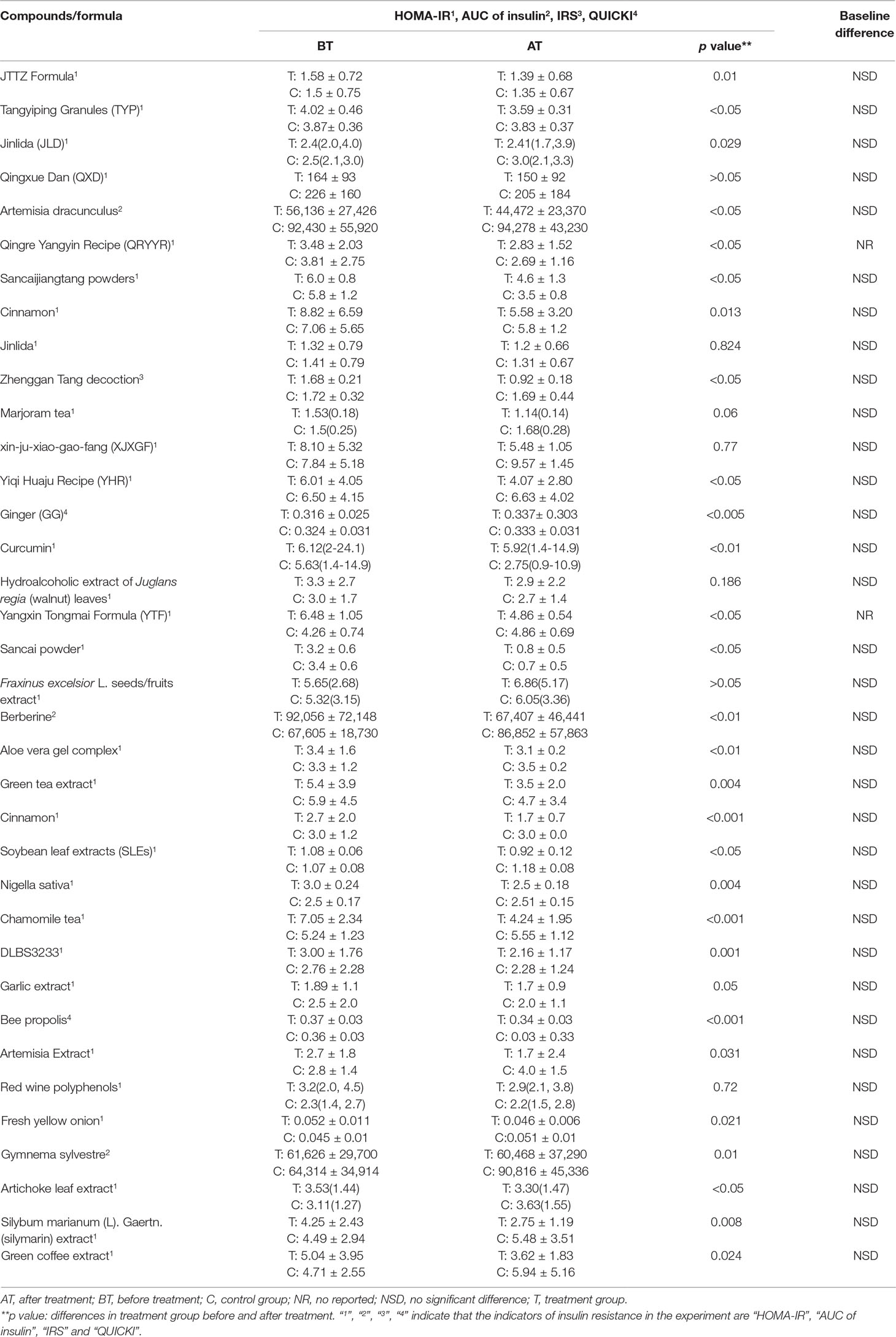
Table 2 Changes in insulin resistance evaluation indicators before and after treatment in clinical trials.
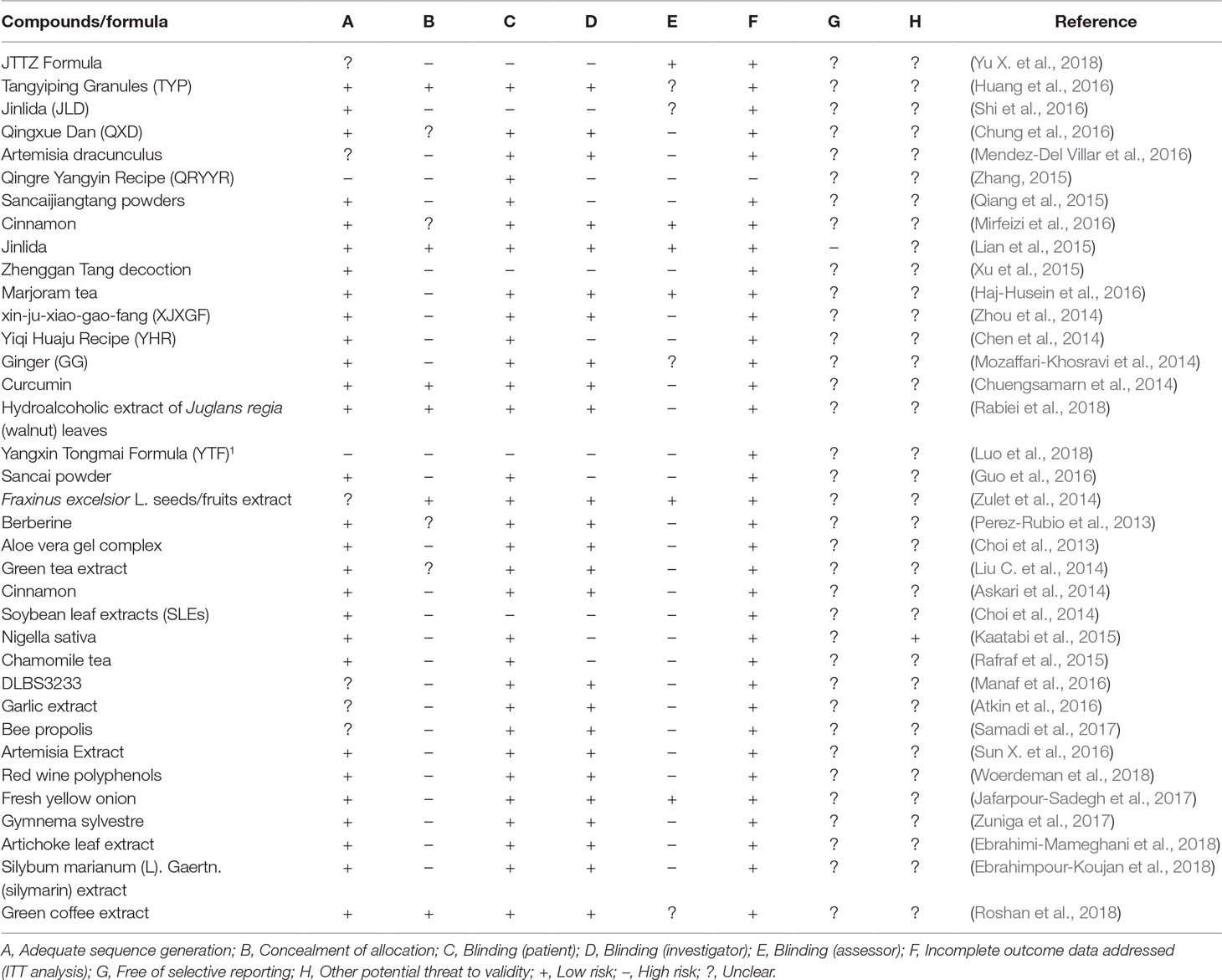
Two evaluators independently assessed the risk of bias in each study and provided the methodological quality of inclusion in clinical trials according to predetermined criteria in the Cochrane Handbook (Table 3). In general, the methodological quality was assessed to be poor. There are few reports of randomized sequence generation and allocation concealment. Fifteen studies (30/36, 83%) detailed how patients were randomized. Only six trials (6/36, 17%) in this study adequately reported the allocation of hidden methods. Twenty-five trials (25/36, 69%) used blinding on their subjects and investigators. Five trials (5/36, 14%) used blinding on their subjects, investigators, and outcome evaluators. The following restrictions should be considered before accepting the conclusion. First, most of the clinical trials included in this review have certain flaws and bias risks in their methodological design, including the generation of randomization, the concealment of allocation, blinding, and inadequate reporting of sample size estimates. Secondly, the duration of 21 trials (21/36, 58%) was greater than or equal to 12 weeks, and 3 trials (3/36, 8%) lasted longer than 6 months. The impact of duration on the results of the study cannot be ignored. Finally, outcome measures for evaluation of major adverse clinical events, such long-term follow-up, were not considered in this review. Therefore, this review indicates that the evidence for both the benefits and harms of herbal treatment for IR is not strong, and it is necessary to rigorously design further trials with high methodological quality to confirm the conclusion.
It must be mentioned that six clinical trials (Juglans regia leaves, marjoram tea, Fraxinus excelsior L. seeds/fruit extract, garlic extract, bee propolis, and red wine polyphenols) have not confirmed the therapeutic effect of herbs on IR. Possible reasons may include that, firstly, all herbal medicines are not necessarily effective for treating IR while also improving the metabolic index and secondly, these two herbs were not studied using conventional methods of their consuming (they were obtained from extraction using ethanol and soaking in boiling water, respectively). This may have altered the concentration and composition of the drug solution. Despite this, the potential therapeutic effects of herbs on IR are worthy of attention.
Insulin Signal Transduction
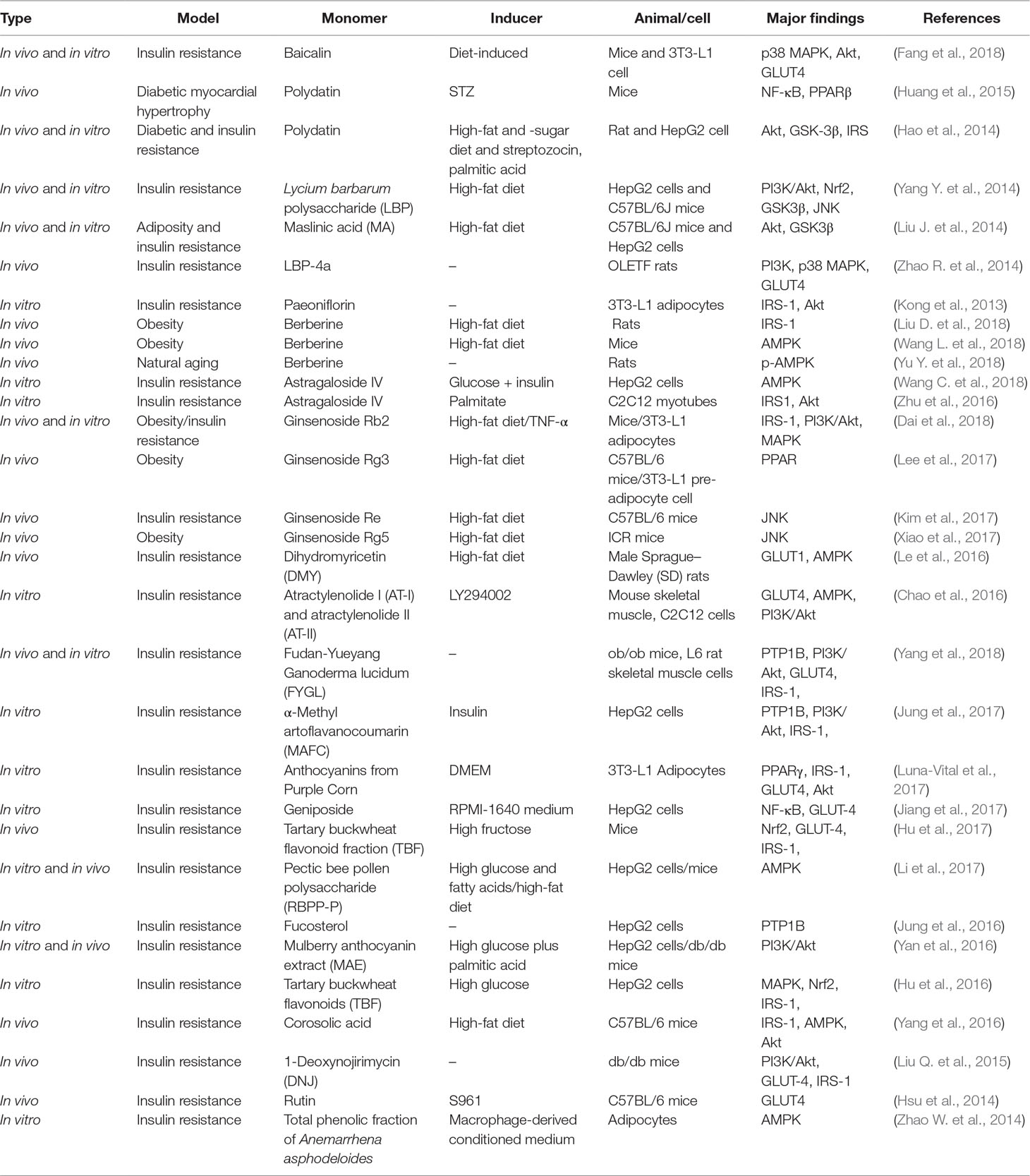
The physiology of insulin involves a complex network of signaling pathways that is activated by the insulin receptor (Samuel and Shulman, 2016). Insulin binding to an insulin receptor on a cell triggers autophosphorylation followed by phosphorylation of intracellular receptor substrates 1 and 2 (IRS-1/IRS-2) (Cheng et al., 2013). Several upstream and downstream key signaling molecules in the insulin signaling pathway have been identified, including the phosphoinositide 3-kinase (PI3K)/Akt pathway that is known to be involved in the translocation of glucose transporter 4 (GLUT-4) from intracellular vesicles to cells and promote glucose uptake to adipose tissue and skeletal muscle, eventually decreasing blood glucose levels. Other related pathways include the mitogen-activated protein kinase (MAPK), adenosine monophosphate-activated protein kinase (AMPK), and stress-activated c-Jun-N-terminal kinase (JNK) pathways (Belwal et al., 2017), among others, and these key players in signal transduction processes are potential targets for drug interventions in IR. IR is characterized by multiple defects, with decreases in receptor concentration and kinase activity, PI3K activity (Anitha et al., 2006), the concentration and phosphorylation of IRS-1 and IRS-2 (Hoehn et al., 2008), and glucose transporter translocation (Bogan, 2012). Previous studies have found that abnormalities in insulin signaling pathways caused by lipid metabolism disorders, inflammatory responses, oxidative stress, endoplasmic reticulum stress, and mitochondrial dysfunction lead to IR (Guilherme et al., 2008; Szendroedi et al., 2011; Gurzov et al., 2014; Park E. et al., 2014; Siwicki et al., 2016). Metabolic disorder and inflammation cause IR and promote leukocytes to secrete proinflammatory cytokines, including IL-6 and tumor necrosis factor-α (TNF-α) (Feve and Bastard, 2009; Wen et al., 2011; Wensveen et al., 2015), which provides a framework to understand how physiological stress, obesity, and diet promote IR. We have placed the experimental research on the treatment of IR by herbal medicine into three categories: active ingredients (Table 4), natural product (Table 5), and herbal formula (Table 6). The active ingredient is a relatively single component, and research has proven to play a major role in the therapeutic effects of herbal medicine. The active ingredients listed in Table 4 are berberine, ginsenoside, astragaloside, polydatin, baicalin, maslinic acid, paeoniflorin, Lycium barbarum polysaccharide, dihydromyricetin, atractylenolide, etc. Natural products are also extracted from herbs, but the ingredients are relatively complex. The herbs involved in the natural products in the table include mulberry leaves, Coptis chinensis, litchi seed, red ginseng, and Gastrodia elata Blume. There are many herbs involved in herbal formulas. Some of these herbs have been used frequently, such as C. chinensis, mulberry leaves, Pueraria montana lobata, Salvia miltiorrhiza, and Astragalus membranaceus. Therapeutic targets for these herbs include: insulin receptor substrate, phosphatidylinositol 3-kinase, glucose transporter, AMP-activated protein kinase (AMPK), glycogen synthase kinase 3, MAPKs, JNK, nuclear factor-kappaB (NF-κB), protein tyrosine phosphatase 1B, nuclear factor-E2-related factor 2, and peroxisome proliferator-activated receptors. The results suggest that herbal interventions for IR are mostly multi-targeted, sometimes interfering with the same target through different pathways. Insulin receptor substrate signals transduction.
Insulin Receptor Substrate Signal Transduction
The insulin receptor belongs to the subfamily of receptor tyrosine kinases, including insulin-like growth factor 1 receptors and insulin receptor-related receptors (White, 2003). Most insulin signals promote or regulate phosphorylation of IRS-1 or its homolog IRS-2 via tyrosine (Haeusler and Accili, 2008), and IRS-1 is the major substrate of the insulin receptor. IRS mediates insulin action differently in different tissues, with IRS-1 playing a prominent role in skeletal muscle and IRS-2 in the liver (Kido et al., 2000). Studies have shown that liver IRS-1 and IRS-2 have complementary effects in controlling liver metabolism; IRS-1 is more closely related to glucose homeostasis (Bouzakri et al., 2006), and IRS-2 is more closely related to lipid metabolism (Taniguchi et al., 2005). Insufficient expression of IRS-1 and IRS-2 can lead to IR (Tamemoto et al., 1994; Shimomura et al., 2000). Jinlida particles can raise insulin sensitivity in skeletal muscle in fat-induced insulin-resistant ApoE-/- mice by increasing the expression of IRS-1 mRNA and protein (Jin et al., 2015). Treating high insulin-induced HepG2 cells with FTZ in vitro upregulated the expression of IRS-1 protein while attenuating in vitro glucose levels (Hu et al., 2014). Abnormal phosphorylation of IRS is also an important mechanism of IR.
IRS1 and IRS2 appear to lack intrinsic catalytic activity but contain many serine and tyrosine phosphorylation sites (White, 2003). Serine/threonine phosphorylation of IRS-1 at the phosphorylation site Ser307 may inhibit insulin signaling (Rui et al., 2001) and attenuate tyrosine phosphorylation levels (Saad et al., 1992). Salvia-Nelumbinis naturalis (SNN) improves hepatic insulin sensitivity in rats and increases IRS phosphorylation (Zhang et al., 2014). Tumor necrosis factor-α (TNF-α) reduces insulin receptor substrate tyrosine phosphorylation and is an important mediator of IR in obesity and diabetes (Hotamisligil et al., 1996). Erchen decoction and Linguizhugan decoction reduce the level of TNF-α in diet-induced insulin-resistant rats to improve IR (Zhang H. et al., 2017), similar to the pharmacological action of thiazolidinedione (Peraldi et al., 1997). In addition, degenerative neuropathies such as Alzheimer’s disease (Talbot and Wang, 2014) and multiple system atrophy (Bassil et al., 2017) are also closely related to brain IR caused by blocked IRS signaling. Defects in IRS-1 may cause vascular damage and accelerate the progression of atherosclerosis (Abe et al., 1998), while IRS-2 delays neointimal formation under IR (Kubota et al., 2003). Gal-geun-dang-gwi-tang attenuates endothelial dysfunction by promoting nitric oxide (NO)-cyclic guanosine monophosphate (cGMP) signaling and improves insulin sensitivity in individuals with diabetic atherosclerosis. Gal-geun-dang-gwi-tang was also shown to be associated with restored expression of IRS-1 in the thoracic aorta and skeletal muscle (Lee et al., 2014).
PI3K/Akt Signaling Pathway Signal Transduction
Glucose is mainly metabolized in insulin-sensitive tissues by two pathways: the classical phosphatidylinositol 3-kinase (PI3K) pathway and the 5’-AMP activating kinase (AMPK) signal transduction pathway (Jeong et al., 2017). Tyrosine phosphorylation of the insulin receptor substrate activates PI3K, and activated PI3K catalyzes 4,5-2 phosphatidylinositol (PIP2) and produces PIP3, which acts as a second messenger that activates Akt (White, 2003). Activated Akt promotes downstream molecules that regulate metabolism. Liuwei Dihaung decoction can be used to treat IR by regulating the PI3K/Akt signaling pathway in the liver of rats with T2D, accompanied by phosphorylation and upregulation of PI3K/Akt pathway-associated proteins (Dai et al., 2016). PI3K is composed of a regulatory subunit p85 and a catalytic subunit p110 (Geering et al., 2007) and has dual activities of phosphatidylinositol kinase and serine/threonine (Ser/Thr) protein kinase. Heterozygous mutations in the PI3Kp85 regulatory subunit gene often result in metabolic disorders such as IR associated with decreased ability to activate PI3K in muscle and adipose tissue (Winnay et al., 2016). Significant damage to PI3K signaling in muscle often results in muscle IR and systemic glucose intolerance (Luo et al., 2006). Central glucagon-like peptide 2 can enhance hepatic insulin sensitivity by activating G3-2R-p85a interactions in PI3K signaling in proopiomelanocortin neurons (Shi et al., 2013). Different proportions of Jiaotai Pill enhance PI3K pathway insulin signaling by upregulating the expression of the PI3K p85 subunit in skeletal muscle, attenuating the development of diabetes in a rat model of T2D (Dong et al., 2013). Akt has three isoforms, of which Akt1 and Akt2 are highly expressed in skeletal muscle and have unique and overlapping functions (Matheny et al., 2018). Overexpression of Akt1 is associated with increased beta cell size and total islet mass (Tuttle et al., 2001), and Akt2 is more important for insulin-stimulated glucose metabolism (Bouzakri et al., 2006). Defects in signaling pathways caused by mutations in the protein kinase Akt2/PKB gene often impair the ability of insulin to lower blood glucose in the liver and skeletal muscle (George et al., 2004). Paeoniflorin improves TNF-α-induced IR in adipocytes and is associated with insulin-stimulated Akt phosphorylation recovery in adipocytes (Kong et al., 2013). Akt2 is involved in glucose uptake by insulin-regulated muscle and adipocytes by promoting the transport of GLUT-4 to the cell surface (Ng et al., 2008), as is metformin, which also increases liver Akt phosphorylation and promotes GLUT-4 translocation (Garabadu and Krishnamurthy, 2017). Jiangzhi capsule improved fructose-induced IR and repaired the damaged muscle fiber membrane GLUT-4 cycle by regulating the ratio of phosphorylated Akt to total Akt in the gastrocnemius muscle (Jiang L. et al., 2016). Akt2 is required for hepatic lipid accumulation in obese and insulin-resistant states induced by leptin deficiency or high-fat diet (HFD) (Leavens et al., 2009). Modified lingguizhugan decoction improves liver fat accumulation and IR in rats with metabolic syndrome by inhibiting abnormal increases in leptin and PKB in the liver (Yao et al., 2017).
Glucose Transporters Signal Transduction
There are currently 13 known sugar transporter proteins (GLUT1-12 and HMIT) encoded in the human genome (Joost and Thorens, 2001). Based on sequence similarity and characteristic elements, the extended GLUT family can be divided into three subfamilies, of which class I contains the glucose transporters GLUT1-4, which all show tissue/cell specific expression (Wood and Trayhurn, 2003). GLUT-4 is highly expressed in adipose tissue and skeletal muscle (Bogan, 2012). The main cellular mechanism for handling exogenous glucose load is insulin-stimulated glucose transport into skeletal muscle, and the primary glucose transporter that mediates this uptake is GLUT-4 (Huang and Czech, 2007). Oxidative stress caused by overnutrition often induces GLUT-4 inactivation by carbonylation and oxidation (Boden et al., 2015), and related gene variants (Stenbit et al., 1997) often lead to GLUT-4 deficiency and downregulation in skeletal muscle and adipose tissue (Zisman et al., 2000; Abel et al., 2001). Exercise and caloric restriction can upregulate GLUT-4 gene expression and increase insulin-induced GLUT-4 transport to the plasma membrane (Richter and Hargreaves 2013; Zanquetta et al., 2003). A novel botanical formula containing standardized extracts of mulberry leaf, fenugreek seed, and American ginseng can attenuate the decrease in GL UT-4 expression induced by an HFD and alloxan (Kan et al., 2017). A key step in the physiological role of GLUT-4 is translocation (Bai et al., 2007), in which GLUT-4 is redistributed from the intracellular pool to the plasma membrane under the regulation of the Akt substrate AS160 rab GTPaseactivating protein (Sano et al., 2007). Studies have shown that cold exposure therapy can significantly increase GLUT-4 translocation in basal skeletal muscle and may be a potential treatment for diabetes (Hanssen et al., 2015). Oroxylum indicum stem bark extract significantly enhanced insulin sensitivity in mature 3T3-L1 adipocytes, as evidenced by increased skeletal muscle GLUT-4 translocation (Singh and Kakkar, 2013). GLUT-2 is mainly found in the liver, intestine, kidney, and pancreatic beta cells (Wood and Trayhurn, 2003), and it plays an important role in glucose transport and energy metabolism because it promotes glucose uptake or liver efflux (Thorens et al., 1992). Sang-Tong-Jian, a new formulation of flavonoids and alkaloids from mulberry leaves, improves IR in KKAy mice, which is associated with upregulation of GLUT-2 (liver) gene, and protein expression (Kuai et al., 2016). GLUT-1 is expressed at the highest level in the endothelial tissues of barrier tissues such as blood vessels and blood-brain barriers, and moderate levels of expression are also observed in adipose tissue, muscle, and liver, which play an important role in supplying glucose to organs such as the brain (Deng et al., 2014; Tang et al., 2017). Chinese herbal formula TZQ-F treatment upregulates the expression of related proteins such as GLUT-1, which regulates the potency of insulin action and is beneficial for reducing hyperinsulinemia (Nan Xia et al., 2013).
AMP-Activated Protein Kinase Signal Transduction
AMPK is a conserved, ubiquitously expressed heterotrimeric serine/threonine protein kinase (Kahn et al., 2005) that plays a key role in regulating cellular energy metabolism (Lage et al., 2008). AMPK integrates nutrient and hormonal signals in peripheral tissues and the hypothalamus and is regulated by multiple hormones such as leptin, adiponectin, ciliary neurotrophic factor, and ghrelin (Minokoshi et al., 2002; Watt et al., 2006; Zhao L. et al., 2015). AMPK plays an important role in regulating food intake, body weight, glucose, and lipid metabolism (Minokoshi et al., 2004). In addition, AMPK can also be activated in response to cellular stress, exercise (Cantó et al., 2010), and drugs (Sasaki et al., 2009). Based on traditional Chinese medicine, Dai-Zong-Fang improves insulin sensitivity in db/db diabetic mice by inhibiting liver lipids and enhancing energy metabolism in skeletal muscle by inhibiting AMPK activation (Zhu et al., 2018). Under physiological conditions, AMPK is mainly present in an inactive form complexed with Mg-ATP, which is more abundant than AMP (Xiao et al., 2011). An increase in AMP concentration activates AMPK, and by phosphorylating the activation loop within the kinase domain (Li et al., 2011), AMPK transitions from an inactive form to a catalytic form: binding of AMP to the c-regulatory domain promotes phosphorylation of the upstream kinase and causes allosteric activation, and inhibition of the dephosphorylation of Thr172 in the kinase domain activation loop regulates AMPK phosphorylation levels (Xiao et al., 2007). Goka fruit supplements improve IR and liver lipid accumulation in mice with HFD-induced obesity by increasing AMPK phosphorylation in the liver. AMPK in adipocytes is critical for maintaining mitochondrial integrity, responding to pharmacological agents and heat stress, and protecting against nutrient overload-induced NAFLD and IR (Kishton et al., 2016; Mottillo et al., 2016). Modified Si-Miao-San positively regulates AMPK phosphorylation to promote basal glucose uptake by 3T3-L1 adipocytes and beneficially improves insulin signaling by inhibiting inflammation in adipocytes (Yang J. et al., 2014). Adiponectin is a major insulin-sensitized adipokine (Kadowaki and Yamauchi, 2011). It has been shown that adiponectin and its receptors AdipoR1 and AdipoR2 enhance glucose and fatty acid metabolism by activating AMPK in peripheral tissues (Kubota et al., 2007). The herbal composition GGEx18 can treat visceral obesity and visceral obesity-related IR by upregulating visceral fat expression of fatty acid oxidation genes. The results show that the expression of fatty acid oxidation genes, including genes encoding adiponectin, AMPK, and others, is significantly increased in mesenteric adipose tissue of 3T3-L1 adipocytes and obese mice (Oh et al., 2015).
Glycogen Synthase Kinase 3 Signal Transduction
GSK-3 is a ubiquitous cytosolic serine/threonine protein kinase expressed in mammalian tissues as two closely related isoforms: GSK-3α and GSK-3β (Dajani et al., 2001). GSK-3 is constitutively active under resting conditions and regulates human metabolism through phosphorylation of glycogen synthase and other substrates (Kaidanovich and Eldar-Finkelman, 2002). Inhibition of GSK-3 is required for insulin-stimulated glycogen and protein synthesis, and its inhibition is critical for the normal functioning of insulin-activated signaling pathways (Ali et al., 2001). The skeletal muscle GSK-3 activity and its expression level were significantly increased in patients with T2D (Saltiel and Kahn, 2001), and abnormally excessively elevated GSK-3 resulted in further inhibition of glycogen synthase activity. A large body of evidence indicates that GSK-3 inhibitors have therapeutic uses in neurodegenerative diseases, cancer, and T2D (Patel and Woodgett, 2008; Hur and Zhou, 2010; Martinez et al., 2002). Jiangtangxiaoke granules are an effective drug for T2D, since this treatment can regulate the expression of glycogen synthase kinase 3β (GSK3β) by regulating the PI3K/Akt signaling pathway in skeletal muscle of mice with T2D (Yu et al., 2017). Insulin-induced GSK-3 inhibition is mediated through its downstream target protein kinase B (PKB), which phosphorylates and inactivates GSK-3 at Ser9/21 (Cross et al., 1995; Frame et al., 2001). ZiBu PiYin recipe treatment alters insulin signaling in T2DM rats in association with inhibited GSK3β overexpression resulting from increased p-GSK3β levels in the pre-frontal cortex and hippocampus (Sun Z. et al., 2016). Abnormal overactivity of GSK-3 may also limit IR-mediated signaling through phosphorylation of IRS-1. Polydatin significantly increased phosphorylated GSK-3β and increased protein levels of phosphorylated IRS in liver and insulin-resistant HepG2 cells of diabetic rats (Hao et al., 2014). Therefore, GSK-3 inhibitors can be a promising new drug for diabetic IR.
P38 Mitogen-Activated Protein Kinase Signal Transduction
MAPKs and their downstream targets are important signaling modules for cellular responses to changes in the physical and chemical properties of the environment (Cuenda et al., 2017). It is known that MAPK has at least four subfamilies in mammalian cells: p38 kinase (p38α, β, γ, and δ), extracellular signal-regulated kinase (ERK1/2), ERK5, and Jun amino terminal kinase (JNK1-3) (Gehart et al., 2010). Insulin activates the PI3K/Akt pathway, which is responsible for glucose uptake, and the MAPK pathway, which is critical for IR (Saltiel and Kahn, 2001). MAPK is involved in a variety of processes that control hepatic metabolism (Lawan and Bennett, 2017). Obesity and inflammation-related stress responses in insulin-responsive tissues activate liver MAPKs, which are thought to impair insulin action and lipid metabolism (Hotamisligil and Davis, 2016). MAPK phosphatases (MKPs) can dephosphorylate MAPK to catalyze the inactivation of MAPK (Gehart et al., 2010). Baicalin plays an important role in reversing HFD-induced glucose intolerance and IR in diet-induced obese mice, and its mechanism is associated with downregulation of p-p38 MAPK levels (Fang et al., 2018). Reactive oxygen species (ROS)-mediated activation of p38MAPK stress response signaling has been recognized as one of the causes of insulin signaling damage and hepatic IR (Al-Lahham et al., 2016). Jinlida, a compound preparation based on traditional Chinese medicine, can attenuate oxidative stress and reduce phosphorylation of p38MAPK and JNK in high-fat fed rats, showing antioxidant effects and upregulation of insulin signaling (Liu Y. et al., 2015).
C-Jun-N-Terminal Kinase Signal Transduction
The JNK is a member of the MAPK family, and three JNK isoforms exist in mammals: JNK1, JNK2, and JNK3 (Lawan and Bennett, 2017). JNK1 and JNK2 are expressed in almost all celRls, including liver parenchymal cells, while JNK3 is mainly expressed in the brain, heart, and testis (Seki et al., 2012). Cytokines, ROS, endoplasmic reticulum stress, and free fatty acids activate JNK (Ozcan et al., 2004; Holzer et al., 2011; Zhao H. et al., 2015) and play a key role in metabolic disorders such as obesity, IR, and T2D (Vallerie and Hotamisligil, 2010). Lycium barbarum polysaccharide treatment effectively inhibits phospho-JNK levels in HFD-fed mice and reduces ROS levels via the PI3K/AKT/Nrf2 axis, acting as a novel anti-hyperlipide-induced IR oxidizer (Yang Y. et al., 2014). Stress factors such as non-esterified fatty acids are generally thought to induce inhibitory serine phosphorylation of IRS-1 through the JNK pathway and impair insulin signaling (Hirosumi et al., 2002; Gao et al., 2018). Studies have also confirmed that hepatocyte-specific deletion of JNK1 enhances IR (Sabio et al., 2009). Therefore, JNK may positively regulate hepatic insulin signaling, and in other insulin-sensitive organs, JNK negatively regulates insulin action, especially under stress conditions. This is the potential protective effect of mulberry leaf extract (MLE) and a formula consisting of MLE, fenugreek seed extract, and cinnamon cassia extract (MLEF) on hyperglycemia induced by high-energy diet and toxic chemicals in rats and recovery of insulin sensitivity, the most likely mechanism is the upregulation of phosphorylation of JNK and other related proteins in the liver to promote IRS-1 phosphorylation (Liu et al., 2016).
Nuclear Factor-kappaB Signal Transduction
NF-κB is a sequence-specific transcription factor that is a major regulator of inflammatory responses, including responses to inflammation and oxidative stress (Chiang et al., 2009). In the quiescent state, NF-κB binds to the inhibitor subunit IκB in an inactive form in the cytoplasm. IKK-β is required for activation during acute inflammation. Phosphorylation-activated IKKβ induces phosphorylation of IκB kinase α (Arkan et al., 2005) and IκBα phosphorylates IκB and leads to proteolysis of IκB, which exposes nuclear recognition sites for NF-κB. NF-κB is translocated into the nucleus, resulting in the expression of related target genes such as inflammatory cytokines (Cai et al., 2005). Thus, blocking NF-κB signaling improves IR and prevents the development of diabetes (Wang et al., 2014). Jiao-Tai-Wan, composed of Rhizome Coptidis and Cortex Cinnamomi, reversibly increases markers of systemic inflammation and IR caused by sleep loss in Sprague-Dawley rats, and these changes are related to downregulation of NF-κB mRNA expression in peripheral blood mononuclear cells (Zou et al., 2017). NF-κB may represent an attractive therapeutic target for obesity, IR, diabetes, and other complications associated with these diseases.
Protein Tyrosine Phosphatase 1B Signal Transduction
Protein tyrosine phosphatase 1B belongs to the protein tyrosine phosphatase (PTP) family (Tiganis, 2013) and has a catalytic domain characterized by an 11 amino acid sequence motif containing cysteine (Cys215) and arginine (Arg221) (Haque et al., 2011). These residues are critical to the catalytic activity of the enzyme. As a negative regulator of the insulin signaling cascade, PTP1B overexpression inhibits tyrosine phosphorylation of IR and IRS-1, enhances serine phosphorylation, thereby terminating insulin signaling (Johnson et al., 2002). Oligonol, a low molecular weight polyphenol mixture derived from lychee fruit, can significantly reduce PTP1B expression and reduce serine phosphorylation of IRS-1, improving insulin sensitivity in insulin-resistant HepG2 cells (Bhakta et al., 2017). In general, PTP1B inhibitors are a promising class of insulin sensitizers.
Nuclear Factor-E2-Related Factor 2 Signal Transduction
Nuclear factor erythrocyte 2-related factor 2 (Nrf2) is a key regulator of antioxidant signaling and plays a crucial role in maintaining redox homeostasis (Seo and Lee, 2013). Under physiological conditions, Nrf2 remains in the cytoplasm by binding to the endogenous inhibitor Keap1, which mediates rapid activation of the proteasome and subsequent degradation of Nrf2 (Zhang et al., 2015). Under exogenous and endogenous oxidative stress, Nrf2 becomes stable and released from the Keap1/Nrf2 complex, Nrf2 degradation is inhibited, and Nrf2 accumulates in the nucleus, synergistically enhancing the expression of various genes encoding antioxidant enzymes (Bhakkiyalakshmi et al., 2015). Many studies have shown that increased Nrf2 signaling can inhibit oxidative stress and improve insulin and leptin resistance (Yagishita et al., 2017). L. barbarum polysaccharide, an antioxidant from wolfberry, increases Nrf2 phosphorylation in livers of HFD-fed mice and HepG2 cells by inducing PI3K/AKT signaling and induces Nrf2/ARE signaling to reduce oxidative stress and maintain peripheral insulin sensitivity (Yang Y. et al., 2014).
Peroxisome Proliferator-Activated Receptor Signal Transduction
Peroxisome proliferator-activated receptors (PPARs) are nuclear receptors involved in the transcriptional control of genes encoding proteins involved in adipocyte differentiation, lipid and carbohydrate metabolism, and adipokine synthesis, including three isoforms encoded by different genes (Eldor et al., 2013). PPARα is highly expressed in liver, kidney, and skeletal muscle. PPARγ is also highly expressed in various cell types and organs, including fat cells, muscle cells, liver, and kidneys, and is considered to be a major regulator of glucose homeostasis (Haluzík and Haluzík, 2006). Studies have suggested that the production of future PPARα and γ double agonists will simultaneously bring about favorable changes in PPARα lipid mass spectrometry and blood glucose benefits of PPARγ agonists (Massaro et al., 2016). The existing insulin-sensitizing drug thiazolidinedione is a potent agonist of nuclear PPAR-γ (Hevener et al., 2003). Based on the traditional anti-diabetic formula, TZQ-F can improve IR in KKA(y) mice through its efficacy in regulating adipocyte differentiation and insulin action, and the results indicate that its therapeutic effect is related to the upregulation of PPARγ expression in liver tissue (Nan Xia et al., 2013) and PPARγ coactivator 1α (PGC1α), which is a PPAR-mediated transcriptional coactivator of fatty acid oxidation (Koo et al., 2004). Tang-Nai-Kang is a mixture of five herbal plant extracts that has been shown to improve glucose metabolism abnormalities in patients with pre-diabetes. Tang-Nai-Kang treatment can deacetylate PGC1α to activate it and synergize with PPAR expression to enhance fatty acid oxidation and improve insulin levels in rats (Li et al., 2015).
Conclusions and Perspectives
IR is a pathological condition common to many metabolic diseases; the most well known of which is T2D. Some surveys show that China is one of the countries with the highest incidence of T2D in the world, and the number of people with diabetes in the adult population have exceeded 113.9 million in 2010 (Zheng et al., 2018). The global trend of other metabolic diseases associated with IR is also not optimistic. From 2003 to 2012, the overall prevalence of metabolic syndrome in the United States was 33% (Aguilar et al., 2015). It is estimated that nearly 100 million people in the United States have NAFLD (Rinella, 2015). Global Burden of Disease studies have shown that the prevalence of obesity has doubled in 73 countries between 1980 and 2015 (Inoue et al., 2018). In addition, based on the 1990 National Institutes of Health standard, PCOS affects 6–10% of women worldwide (Goodarzi et al., 2011), and studies have indicated that 56.3% of Han women in China with PCOS have IR (Li et al., 2018). Therefore, in the face of such a large potential patient population, the harm caused by IR cannot be ignored. Herbs have been used in China for more than 2,000 years and are still considered effective drugs to prevent and treat various diseases. The discovery and application of artemisinin, a compound derived from Artemisia annua, is a good example. In recent years, research on Chinese medicine has gradually been standardized and systematized. China’s latest guidelines for preventing and treating T2D are also the first to include Chinese medicine treatment.
The information presented in this review shows that herbal formulas, active ingredients, and natural products can be effective to improve IR. Targets of herbal compounds that affect insulin signaling include insulin receptor substrate, phosphatidylinositol 3-kinase, glucose transporter, AMPK, glycogen synthase kinase 3, MAPKs, JNK, NF-κB, protein tyrosine phosphatase 1B, nuclear factor-E2-related factor 2, and peroxisome proliferator-activated receptors.
In Table 7, we have listed the composition of the above herbal formula in detail. In addition, we have found that certain herbs have a good application prospect in the treatment of IR. Such single botanicals include Coptis, Pueraria, Mulberry, Salvia, and others. Effective extracts of herbal medicines include berberine and mulberry leaves. There are also effective active ingredients such as berberine, ginsenoside, astragaloside, and resveratrol. The effects of these drugs are not only limited to enhancing insulin sensitivity, but also can be beneficial for improving systemic metabolism, such as reducing fasting blood glucose and postprandial blood glucose, improving blood lipid metabolism, reducing body weight, lowering blood pressure, and regulating female hormone secretion. Therefore, the role of herbal medicine in the treatment of IR is not only beneficial for treating T2D, but also provides new ideas for treating obesity, metabolic syndrome, PCOS, and NAFLD.
However, there were some shortcomings in our research. First, most of the interventions studied were herbal formulas and extracts. The diversity of ingredients in the herbal formulas and extracts results in complex potential therapeutic mechanisms. Not only does it make understanding the role of drugs difficult, but it also makes in-depth research impossible. However, the existing research also has the same shortcomings as our research. In addition, there are studies questioning the role of herbal medicine in the treatment of IR. The beneficial effects of ginsenosides on IR are listed in Table 4, but clinical trials have shown that oral ginseng or ginsenosides do not improve insulin sensitivity in glucose-tolerant or obese/overweight subjects who are newly diagnosed with diabetes (Reeds et al., 2011). However, the clinical trial lasted only 8 weeks, and in most in vitro and in vivo tests, ginsenosides were administered by intraperitoneal injection. In clinical trials, the systemic utilization and metabolic processes have to be considered only after oral administration. Studies have indicated that ginsenoside is a precursor. The pharmacological action of ginsenoside is to activate intestinal deglycosylation and fatty acid esterification (Hasegawa, 2004). Therefore, the in vitro and in vivo levels of ginsenosides should not be the only focus of the trial.
In response to these shortcomings, we can make some improvements in future studies. For example, regarding the bioavailability of herbal medicines, we can use targeted drug delivery systems (TDDSs) to improve the way in which the active ingredients of herbal medicines are administered. Research on targeted preparations related to TDDSs has become a popular topic, especially for applications in the field of anti-cancer research. Targeted administration can be divided into liposomes, granules, nanoparticles, emulsions, and other similar preparations (Li et al., 2009). The combination of drugs and specific target carriers can play a synergistic and attenuating role. At present, research on herbal drug delivery systems is still in the exploration stage, and the design, synthesis, and quality evaluation of TDDS are more suitable for single herbal ingredients. Therefore, separating the active ingredients of herbs is particularly important, including those known or unknown. We can select safe and effective single herbs from the many herbal formulas obtained from the literature and experimental research and separate the active ingredients, such as monomers, by pharmacological methods. Then, based on the active ingredients of the herbal medicine, the effective target mechanism of the drug can be studied. Finally, the active ingredient, target, and TDDS are combined. This not only makes the research on herbal medicine more targeted and efficient, but also provides a good prospect for the development and application of anti-insulin drugs.
Author Contributions
JL and LB designed the work of review. JL, LB, and FW reviewed the literature available on this topic and wrote the paper. JZ, DW, YX, and WY contributed in the scientific writing of the manuscript. JL, LB, and JW revised the manuscript. All authors approved the paper for publication. JL, LB, FW, JZ, DW, YX, and WY contributed equally to this work. JL and LB contributed equally to this study and share first authorship.
Funding
This paper was supported by Central Health Research Project W2017BJ43.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
Abe, H., Yamada, N., Kamata, K., Kuwaki, T., Shimada, M., Osuga, J., et al. (1998). Hypertension, hypertriglyceridemia, and impaired endothelium-dependent vascular relaxation in mice lacking insulin receptor substrate-1. J. Clin. Invest. 101 (8), 1784–1788. doi: 10.1172/JCI1594
Abel, E., Peroni, O., Kim, J., Kim, Y., Boss, O., Hadro, E., et al. (2001). Adipose-selective targeting of the GLUT4 gene impairs insulin action in muscle and liver. Nature 409 (6821), 729–733. doi: 10.1038/35055575
Aguilar, M., Bhuket, T., Torres, S., Liu, B., Wong, R. J. (2015). Prevalence of the metabolic syndrome in the United States, 2003–2012. JAMA 313 (19), 1973–1974. doi: 10.1001/jama.2015.4260
Ali, A., Hoeflich, K., Woodgett, J. (2001). Glycogen synthase kinase-3: properties, functions, and regulation. Chem. Rev. 101 (8), 2527–2540. doi: 10.1021/cr000110o
Al-Lahham, R., Deford, J. H., Papaconstantinou, J. (2016). Mitochondrial-generated ROS down regulates insulin signaling via activation of the p38MAPK stress response pathway. Mol. Cell Endocrinol. 419, 1–11. doi: 10.1016/j.mce.2015.09.013
Anitha, M., Gondha, C., Sutliff, R., Parsadanian, A., Mwangi, S., Sitaraman, S. V., et al. (2006). GDNF rescues hyperglycemia-induced diabetic enteric neuropathy through activation of the PI3K/Akt pathway. J. Clin. Invest. 116 (2), 344–356. doi: 10.1172/JCI26295
Araujo, T. G., de Oliveira, A. G., Vecina, J. F., Marin, R. M., Franco, E. S., Abdalla Saad, M. J., et al. (2016). Parkinsonia aculeata (Caesalpineaceae) improves high-fat diet-induced insulin resistance in mice through the enhancement of insulin signaling and mitochondrial biogenesis. J. Ethnopharmacol. 183, 95–102. doi: 10.1016/j.jep.2016.02.048
Arkan, M., Hevener, A., Greten, F., Maeda, S., Li, Z., Long, J., et al. (2005). IKK-beta links inflammation to obesity-induced insulin resistance. Nat. Med. 11 (2), 191–198. doi: 10.1038/nm1185
Armstrong, D. G., Boulton, A. J. M., Bus, S. A. (2017). Diabetic foot ulcers and their recurrence. N. Engl. J. Med. 376 (24), 2367–2375. doi: 10.1056/NEJMra1615439
Askari, F., Rashidkhani, B., Hekmatdoost, A. (2014). Cinnamon may have therapeutic benefits on lipid profile, liver enzymes, insulin resistance, and high-sensitivity C-reactive protein in nonalcoholic fatty liver disease patients. Nutr. Res. 34 (2), 143–148. doi: 10.1016/j.nutres.2013.11.005
Atkin, M., Laight, D., Cummings, M. H. (2016). The effects of garlic extract upon endothelial function, vascular inflammation, oxidative stress and insulin resistance in adults with type 2 diabetes at high cardiovascular risk. J. Diabetes Complications. 30 (4), 723–727. doi: 10.1016/j.jdiacomp.2016.01.003
Aytug, S., Reich, D., Sapiro, L. E., Bernstein, D., Begum, N. (2003). Impaired IRS-1/PI3-kinase signaling in patients with HCV: a mechanism for increased prevalence of type 2 diabetes. Hepatology 38 (6), 1384–1392. doi: 10.1016/j.hep.2003.09.012
Bae, U. J., Park, J., Park, I. W., Chae, B. M., Oh, M. R., Jung, S. J., et al. (2018). Epigallocatechin-3-gallate-rich green tea extract ameliorates fatty liver and weight gain in mice fed a high fat diet by activating the sirtuin 1 and AMP activating protein kinase pathway. Am. J. Chin. Med. 46 (3), 617–632. doi: 10.1142/S0192415X18500325
Bai, L., Gao, J., Wei, F., Zhao, J., Wang, D., Wei, J. (2018). Therapeutic potential of ginsenosides as an adjuvant treatment for diabetes. Front. Pharmacol. 9, 423. doi: 10.3389/fphar.2018.00423
Bai, L., Wang, Y., Fan, J., Chen, Y., Ji, W., Qu, A., et al. (2007). Dissecting multiple steps of GLUT4 trafficking and identifying the sites of insulin action. Cell Metab. 5 (1), 47–57. doi: 10.1016/j.cmet.2006.11.013
Banin, R. M., Hirata, B. K., Andrade, I. S., Zemdegs, J. C., Clemente, A. P., Dornellas, A. P., et al. (2014). Beneficial effects of Ginkgo biloba extract on insulin signaling cascade, dyslipidemia, and body adiposity of diet-induced obese rats. Braz. J. Med. Biol. Res. 47 (9), 780–788. doi: 10.1590/1414-431X20142983
Bassil, F., Canron, M. H., Vital, A., Bezard, E., Li, Y., Greig, N. H., et al. (2017). Insulin resistance and exendin-4 treatment for multiple system atrophy. Brain 140 (5), 1420–1436. doi: 10.1093/brain/awx044
Belwal, T., Nabavi, S. F., Nabavi, S. M., Habtemariam, S. (2017). Dietary anthocyanins and insulin resistance: when food becomes a medicine. Nutrients 9 (10), 1–22. doi: 10.3390/nu9101111
Bhakkiyalakshmi, E., Sireesh, D., Rajaguru, P., Paulmurugan, R., Ramkumar, K. M. (2015). The emerging role of redox-sensitive Nrf2-Keap1 pathway in diabetes. Pharmacol. Res. 91, 104–114. doi: 10.1016/j.phrs.2014.10.004
Bhakta, H. K., Paudel, P., Fujii, H., Sato, A., Park, C. H., Yokozawa, T., et al. (2017). Oligonol promotes glucose uptake by modulating the insulin signaling pathway in insulin-resistant HepG2 cells via inhibiting protein tyrosine phosphatase 1B. Arch. Pharm. Res. 40 (11), 1314–1327. doi: 10.1007/s12272-017-0970-6
Biessels, G. J., Staekenborg, S., Brunner, E., Brayne, C., Scheltens, P. (2006). Risk of dementia in diabetes mellitus: a systematic review. Lancet Neurol. 5 (1), 64–74. doi: 10.1016/S1474-4422(05)70284-2
Boden, G., Homko, C., Barrero, C., Stein, T., Chen, X., Cheung, P., et al. (2015). Excessive caloric intake acutely causes oxidative stress, GLUT4 carbonylation, and insulin resistance in healthy men. Sci. Transl. Med. 7 (304), 304re307. doi: 10.1126/scitranslmed.aac4765
Bogan, J. S. (2012). Regulation of glucose transporter translocation in health and diabetes. Annu. Rev. Biochem. 81, 507–532. doi: 10.1146/annurev-biochem-060109-094246
Bouzakri, K., Zachrisson, A., Al-Khalili, L., Zhang, B. B., Koistinen, H. A., Krook, A., et al. (2006). siRNA-based gene silencing reveals specialized roles of IRS-1/Akt2 and IRS-2/Akt1 in glucose and lipid metabolism in human skeletal muscle. Cell Metab. 4 (1), 89–96. doi: 10.1016/j.cmet.2006.04.008
Cai, D., Yuan, M., Frantz, D., Melendez, P., Hansen, L., Lee, J., et al. (2005). Local and systemic insulin resistance resulting from hepatic activation of IKK-beta and NF-kappaB. Nat. Med. 11 (2), 183–190. doi: 10.1038/nm1166
Cantó, C., Jiang, L., Deshmukh, A., Mataki, C., Coste, A., Lagouge, M., et al. (2010). Interdependence of AMPK and SIRT1 for metabolic adaptation to fasting and exercise in skeletal muscle. Cell Metab. 11 (3), 213–219. doi: 10.1016/j.cmet.2010.02.006
Chang, C. J., Lin, C. S., Lu, C. C., Martel, J., Ko, Y. F., Ojcius, D. M., et al. (2015). Ganoderma lucidum reduces obesity in mice by modulating the composition of the gut microbiota. Nat. Commun. 6, 7489. doi: 10.1038/ncomms8489
Chao, C. L., Huang, H. C., Lin, H. C., Chang, T. C., Chang, W. L. (2016). Sesquiterpenes from Baizhu stimulate glucose uptake by activating AMPK and PI3K. Am. J. Chin. Med. 44 (5), 963–979. doi: 10.1142/S0192415X16500531
Chen, G., Yang, X., Yang, X., Li, L., Luo, J., Dong, H., et al. (2017). Jia-Wei-Jiao-Tai-Wan ameliorates type 2 diabetes by improving beta cell function and reducing insulin resistance in diabetic rats. BMC Complement. Altern. Med. 17 (1), 507. doi: 10.1186/s12906-017-2016-5
Chen, Y., Fu, D. Y., Fu, X. D., He, Y. M., Wang, W. J. (2014). Effect of yiqi huaju recipe combined with routine therapy in treating hypertension patients with metabolic syndrome: a clinical study. Zhongguo Zhong Xi Yi Jie He Za Zhi. 34 (6), 680–687.
Cheng, K. C., Asakawa, A., Li, Y. X., Liu, I. M., Amitani, H., Cheng, J. T., et al. (2013). Opioid mu-receptors as new target for insulin resistance. Pharmacol. Ther. 139 (3), 334–340. doi: 10.1016/j.pharmthera.2013.05.002
Chiang, S. H., Bazuine, M., Lumeng, C. N., Geletka, L. M., Mowers, J., White, N. M., et al. (2009). The protein kinase IKKepsilon regulates energy balance in obese mice. Cell 138 (5), 961–975. doi: 10.1016/j.cell.2009.06.046
Choi, H. C., Kim, S. J., Son, K. Y., Oh, B. J., Cho, B. L. (2013). Metabolic effects of aloe vera gel complex in obese prediabetes and early non-treated diabetic patients: randomized controlled trial. Nutrition 29 (9), 1110–1114. doi: 10.1016/j.nut.2013.02.015
Choi, M. S., Ryu, R., Seo, Y. R., Jeong, T. S., Shin, D. H., Park, Y. B., et al. (2014). The beneficial effect of soybean (Glycine max (L). Food Funct. 5 (7), 1621–1630. doi: 10.1039/c4fo00199k
Chuengsamarn, S., Rattanamongkolgul, S., Phonrat, B., Tungtrongchitr, R., Jirawatnotai, S. (2014). Reduction of atherogenic risk in patients with type 2 diabetes by curcuminoid extract: a randomized controlled trial. J. Nutr. Biochem. 25 (2), 144–150. doi: 10.1016/j.jnutbio.2013.09.013
Chung, W., Ryu, J., Chung, S., Kim, S. (2016). [Effect of Qingxue Dan on obesity and metabolic biomarker: a double-blind randomized-controlled pilot study]. J. Tradit. Chin. Med. 36 (3), 291–298. doi: 10.1016/S0254-6272(16)30040-1
Cross, D., Alessi, D., Cohen, P., Andjelkovich, M., Hemmings, B. (1995). Inhibition of glycogen synthase kinase-3 by insulin mediated by protein kinase B. Nature 378 (6559), 785–789. doi: 10.1038/378785a0
Cuenda, A., Lizcano, J. M., Lozano, J. (2017). Editorial: mitogen activated protein kinases. Front Cell Dev. Biol. 5, 80. doi: 10.3389/fcell.2017.00080
D, C. (2018). Why Chinese medicine is heading for clinics around the world. Nature 561, 448–450. doi: 10.1038/d41586-018-06782-7
Dai, B., Wu, Q., Zeng, C., Zhang, J., Cao, L., Xiao, Z., et al. (2016). The effect of Liuwei Dihuang decoction on PI3K/Akt signaling pathway in liver of type 2 diabetes mellitus (T2DM) rats with insulin resistance. J. Ethnopharmacol. 192, 382–389. doi: 10.1016/j.jep.2016.07.024
Dai, S., Hong, Y., Xu, J., Lin, Y., Si, Q., Gu, X. (2018). Ginsenoside Rb2 promotes glucose metabolism and attenuates fat accumulation via AKT-dependent mechanisms. Biomed. Pharmacother. 100, 93–100. doi: 10.1016/j.biopha.2018.01.111
Dajani, R., Fraser, E., Roe, S., Young, N., Good, V., Dale, T., et al. (2001). Crystal structure of glycogen synthase kinase 3 beta: structural basis for phosphate-primed substrate specificity and autoinhibition. Cell 105 (6), 721–732. doi: 10.1016/S0092-8674(01)00374-9
Deng, D., Xu, C., Sun, P., Wu, J., Yan, C., Hu, M., et al. (2014). Crystal structure of the human glucose transporter GLUT1. Nature 510 (7503), 121–125. doi: 10.1038/nature13306
Diamanti-Kandarakis, E., Dunaif, A. (2012). Insulin resistance and the polycystic ovary syndrome revisited: an update on mechanisms and implications. Endocr. Rev. 33 (6), 981–1030. doi: 10.1210/er.2011-1034
Dong, H., Wang, J. H., Lu, F. E., Xu, L. J., Gong, Y. L., Zou, X. (2013). Jiaotai Pill enhances insulin signaling through phosphatidylinositol 3-kinase pathway in skeletal muscle of diabetic rats. Chin. J. Integr. Med. 19 (9), 668–674. doi: 10.1007/s11655-013-1560-1
Ebrahimi-Mameghani, M., Asghari-Jafarabadi, M., Rezazadeh, K. (2018). TCF7L2-rs7903146 polymorphism modulates the effect of artichoke leaf extract supplementation on insulin resistance in metabolic syndrome: a randomized, double-blind, placebo-controlled trial. J. Integr. Med. 16 (5), 329–334. doi: 10.1016/j.joim.2018.05.006
Ebrahimpour-Koujan, S., Gargari, B. P., Mobasseri, M., Valizadeh, H., Asghari-Jafarabadi, M. (2018). Lower glycemic indices and lipid profile among type 2 diabetes mellitus patients who received novel dose of Silybum marianum (L). Phytomedicine 44, 39–44. doi: 10.1016/j.phymed.2018.03.050
Eldor, R., DeFronzo, R. A., Abdul-Ghani, M. (2013). In vivo actions of peroxisome proliferator-activated receptors: glycemic control, insulin sensitivity, and insulin secretion. Diabetes Care 36 Suppl 2, S162–174. doi: 10.2337/dcS13-2003
Fang, P., Yu, M., Min, W., Han, S., Shi, M., Zhang, Z., et al. (2018). Beneficial effect of baicalin on insulin sensitivity in adipocytes of diet-induced obese mice. Diabetes Res. Clin. Pract. 139, 262–271. doi: 10.1016/j.diabres.2018.03.007
Feve, B., Bastard, J. P. (2009). The role of interleukins in insulin resistance and type 2 diabetes mellitus. Nat. Rev. Endocrinol. 5 (6), 305–311. doi: 10.1038/nrendo.2009.62
Frame, S., Cohen, P., Biondi, R. (2001). A common phosphate binding site explains the unique substrate specificity of GSK3 and its inactivation by phosphorylation. Mol. Cell 7 (6), 1321–1327. doi: 10.1016/S1097-2765(01)00253-2
Gao, L. H., Liu, Q., Liu, S. N., Chen, Z. Y., Li, C. N., Lei, L., et al. (2014). A refined-JinQi-JiangTang tablet ameliorates prediabetes by reducing insulin resistance and improving beta cell function in mice. J. Ethnopharmacol. 151 (1), 675–685. doi: 10.1016/j.jep.2013.11.024
Gao, S., Guo, Q., Qin, C., Shang, R., Zhang, Z. (2017). Sea buckthorn fruit oil extract alleviates insulin resistance through the PI3K/Akt signaling pathway in type 2 diabetes mellitus cells and rats. J. Agric. Food Chem. 65 (7), 1328–1336. doi: 10.1021/acs.jafc.6b04682
Gao, W., Du, X., Lei, L., Wang, H., Zhang, M., Wang, Z., et al. (2018). NEFA-induced ROS impaired insulin signalling through the JNK and p38MAPK pathways in non-alcoholic steatohepatitis. J. Cell Mol. Med. 22 (7), 3408–3422. doi: 10.1111/jcmm.13617
Garabadu, D., Krishnamurthy, S. (2017). Metformin attenuates hepatic insulin resistance in type-2 diabetic rats through PI3K/Akt/GLUT-4 signalling independent to bicuculline-sensitive GABAA receptor stimulation. Pharm. Biol. 55 (1), 722–728. doi: 10.1080/13880209.2016.1268635
Geering, B., Cutillas, P., Nock, G., Gharbi, S., Vanhaesebroeck, B. (2007). Class IA phosphoinositide 3-kinases are obligate p85-p110 heterodimers. Proc. Natl. Acad. Sci. U.S.A. 104 (19), 7809–7814. doi: 10.1073/pnas.0700373104
Gehart, H., Kumpf, S., Ittner, A., Ricci, R. (2010). MAPK signalling in cellular metabolism: stress or wellness? EMBO Rep. 11 (11), 834–840. doi: 10.1038/embor.2010.160
George, S., Rochford, J., Wolfrum, C., Gray, S., Schinner, S., Wilson, J., et al. (2004). A family with severe insulin resistance and diabetes due to a mutation in AKT2. Science 304 (5675), 1325–1328. doi: 10.1126/science.1096706
Goodarzi, M. O., Dumesic, D. A., Chazenbalk, G., Azziz, R. (2011). Polycystic ovary syndrome: etiology, pathogenesis and diagnosis. Nat. Rev. Endocrinol. 7 (4), 219–231. doi: 10.1038/nrendo.2010.217
Guilherme, A., Virbasius, J. V., Puri, V., Czech, M. P. (2008). Adipocyte dysfunctions linking obesity to insulin resistance and type 2 diabetes. Nat. Rev. Mol. Cell Biol. 9 (5), 367–377. doi: 10.1038/nrm2391
Guo, Q., Zhang, H., Li, M., Zhao, Z., Luo, Y., Luo, Y., et al. (2016). Efficacy and safety of Sancai powder in patients with type 2 diabetes mellitus: a randomized controlled trial. J. Tradit. Chin. Med. 36 (5), 640–648. doi: 10.1016/S0254-6272(16)30084-X
Gurzov, E. N., Tran, M., Fernandez-Rojo, M. A., Merry, T. L., Zhang, X., Xu, Y., et al. (2014). Hepatic oxidative stress promotes insulin-STAT-5 signaling and obesity by inactivating protein tyrosine phosphatase N2. Cell Metab. 20 (1), 85–102. doi: 10.1016/j.cmet.2014.05.011
Haeusler, R. A., Accili, D. (2008). The double life of Irs. Cell Metab. 8 (1), 7–9. doi: 10.1016/j.cmet.2008.06.010
Haj-Husein, I., Tukan, S., Alkazaleh, F. (2016). The effect of marjoram (Origanum majorana) tea on the hormonal profile of women with polycystic ovary syndrome: a randomised controlled pilot study. J. Hum. Nutr. Diet 29 (1), 105–111. doi: 10.1111/jhn.12290
Haluzík, M., Haluzík, M. (2006). Peroxisome proliferator activated receptors (PPAR) and insulin sensitivity: experimental studies. Cesk Fysiol. 55 (4), 163–168.
Han, J., Yang, N., Zhang, F., Zhang, C., Liang, F., Xie, W., et al. (2015). Rhizoma Anemarrhenae extract ameliorates hyperglycemia and insulin resistance via activation of AMP-activated protein kinase in diabetic rodents. J. Ethnopharmacol. 172, 368–376. doi: 10.1016/j.jep.2015.05.016
Hanssen, M. J., Hoeks, J., Brans, B., van der Lans, A. A., Schaart, G., van den Driessche, J. J., et al. (2015). Short-term cold acclimation improves insulin sensitivity in patients with type 2 diabetes mellitus. Nat. Med. 21 (8), 863–865. doi: 10.1038/nm.3891
Hao, J., Chen, C., Huang, K., Huang, J., Li, J., Liu, P., et al. (2014). Polydatin improves glucose and lipid metabolism in experimental diabetes through activating the Akt signaling pathway. Eur. J. Pharmacol. 745, 152–165. doi: 10.1016/j.ejphar.2014.09.047
Haque, A., Andersen, J. N., Salmeen, A., Barford, D., Tonks, N. K. (2011). Conformation-sensing antibodies stabilize the oxidized form of PTP1B and inhibit its phosphatase activity. Cell 147 (1), 185–198. doi: 10.1016/j.cell.2011.08.036
Hasegawa, H. (2004). Proof of the mysterious efficacy of ginseng: basic and clinical trials: metabolic activation of ginsenoside: deglycosylation by intestinal bacteria and esterification with fatty acid. J. Pharmacol. Sci. 95 (2), 153–157. doi: 10.1254/jphs.FMJ04001X4
Hevener, A., He, W., Barak, Y., Le, J., Bandyopadhyay, G., Olson, P., et al. (2003). Muscle-specific Pparg deletion causes insulin resistance. Nat. Med. 9 (12), 1491–1497. doi: 10.1038/nm956
Hinkel, R., Howe, A., Renner, S., Ng, J., Lee, S., Klett, K., et al. (2017). Diabetes Mellitus-Induced Microvascular Destabilization in the Myocardium. J. Am. Coll. Cardiol. 69 (2), 131–143. doi: 10.1016/j.jacc.2016.10.058
Hirosumi, J., Tuncman, G., Chang, L., Gorgun, C. Z., Uysal, K. T., Maeda, K., et al. (2002). A central role for JNK in obesity and insulin resistance. Nature 420 (6913), 333–336. doi: 10.1038/nature01137
Hoehn, K. L., Hohnen-Behrens, C., Cederberg, A., Wu, L. E., Turner, N., Yuasa, T., et al. (2008). IRS1-independent defects define major nodes of insulin resistance. Cell Metab. 7 (5), 421–433. doi: 10.1016/j.cmet.2008.04.005
Holzer, R. G., Park, E. J., Li, N., Tran, H., Chen, M., Choi, C., et al. (2011). Saturated fatty acids induce c-Src clustering within membrane subdomains, leading to JNK activation. Cell 147 (1), 173–184. doi: 10.1016/j.cell.2011.08.034
Hong, Y. L., Wu, F. (2014). Effect of Bushen Huatan Recipe on the Akt signal pathway in polycystic ovarian syndrome model rats with insulin resistance: an experimental research. Zhongguo Zhong Xi Yi Jie He Za Zhi. 34 (2), 230–234.
Hotamisligil, G. S., Davis, R. J. (2016). Cell signaling and stress responses. Cold Spring Harb. Perspect. Biol. 8 (10), 1–20. doi: 10.1101/cshperspect.a006072
Hotamisligil, G., Peraldi, P., Budavari, A., Ellis, R., White, M., Spiegelman, B. (1996). IRS-1-mediated inhibition of insulin receptor tyrosine kinase activity in TNF-alpha- and obesity-induced insulin resistance. Science 271 (5249), 665–668. doi: 10.1126/science.271.5249.665
Hsu, C. Y., Shih, H. Y., Chia, Y. C., Lee, C. H., Ashida, H., Lai, Y. K., et al. (2014). Rutin potentiates insulin receptor kinase to enhance insulin-dependent glucose transporter 4 translocation. Mol. Nutr. Food Res. 58 (6), 1168–1176. doi: 10.1002/mnfr.201300691
Hu, X., Wang, M., Bei, W., Han, Z., Guo, J. (2014). The Chinese herbal medicine FTZ attenuates insulin resistance via IRS1 and PI3K in vitro and in rats with metabolic syndrome. J. Transl. Med. 12 (1479-5876 (Electronic)), 47–56. doi: 10.1186/1479-5876-12-47
Hu, Y., Hou, Z., Liu, D., Yang, X. (2016). Tartary buckwheat flavonoids protect hepatic cells against high glucose-induced oxidative stress and insulin resistance via MAPK signaling pathways. Food Funct. 7 (3), 1523–1536. doi: 10.1039/C5FO01467K
Hu, Y., Hou, Z., Yi, R., Wang, Z., Sun, P., Li, G., et al. (2017). Tartary buckwheat flavonoids ameliorate high fructose-induced insulin resistance and oxidative stress associated with the insulin signaling and Nrf2/HO-1 pathways in mice. Food Funct. 8 (8), 2803–2816. doi: 10.1039/C7FO00359E
Huang, B., Xue, L., Wu, Y., Jiang, Q. S. (2015). Effect and mechanism of polydatin on diabetic myocardial hypertrophy in mice. Zhongguo Zhong Yao Za Zhi. 40 (21), 4256–4261.
Huang, S., Czech, M. P. (2007). The GLUT4 glucose transporter. Cell Metab. 5 (4), 237–252. doi: 10.1016/j.cmet.2007.03.006
Huang, Y. Q., Yang, Q. F., Wang, H., Xu, Y. S., Peng, W., Jiang, Y. H. (2016). Long-term clinical effect of Tangyiping Granules () on patients with impaired glucose tolerance. Chin. J. Integr. Med. 22 (9), 653–659. doi: 10.1007/s11655-016-2512-3
Hur, E. M., Zhou, F. Q. (2010). GSK3 signalling in neural development. Nat. Rev. Neurosci. 11 (8), 539–551. doi: 10.1038/nrn2870
Inoue, Y., Qin, B., Poti, J., Sokol, R., Gordon-Larsen, P. (2018). Epidemiology of obesity in adults: latest trends. Curr. Obes. Rep. 7 (4), 276–288. doi: 10.1007/s13679-018-0317-8
Jafarpour-Sadegh, F., Montazeri, V., Adili, A., Esfehani, A., Rashidi, M. R., Pirouzpanah, S. (2017). Consumption of fresh yellow onion ameliorates hyperglycemia and insulin resistance in breast cancer patients during doxorubicin-based chemotherapy: a randomized controlled clinical trial. Integr. Cancer Ther. 16 (3), 276–289. doi: 10.1177/1534735416656915
Jeong, M., Park, J., Youn, D., Jung, Y., Kang, J., Lim, S., et al. (2017). Albiflorin ameliorates obesity by inducing thermogenic genes via AMPK and PI3K/AKT in vivo and in vitro. Metab. Clin. Exp. 73, 85–99. doi: 10.1016/j.metabol.2017.05.009
Jiang, B., Le, L., Zhai, W., Wan, W., Hu, K., Yong, P., et al. (2016). Protective effects of marein on high glucose-induced glucose metabolic disorder in HepG2 cells. Phytomedicine 23 (9), 891–900. doi: 10.1016/j.phymed.2016.05.004
Jiang, C., Yao, N., Wang, Q., Zhang, J., Sun, Y., Xiao, N., et al. (2014). Cyclocarya paliurus extract modulates adipokine expression and improves insulin sensitivity by inhibition of inflammation in mice. J. Ethnopharmacol. 153 (2), 344–351. doi: 10.1016/j.jep.2014.02.003
Jiang, H., Ma, Y., Yan, J., Liu, J., Li, L. (2017). Geniposide promotes autophagy to inhibit insulin resistance in HepG2 cells via P62/NFkappaB/GLUT4. Mol. Med. Rep. 16 (5), 7237–7244. doi: 10.3892/mmr.2017.7503
Jiang, L., Yao, L., Yang, Y., Ke, D., Batey, R., Wang, J., et al. (2016). Jiangzhi capsule improves fructose-induced insulin resistance in rats: association with repair of the impaired sarcolemmal glucose transporter-4 recycling. J. Ethnopharmacol. 194, 288–298. doi: 10.1016/j.jep.2016.09.009
Jin, X., Zhang, H. X., Cui, W. W. (2015). Effect of Jinlida on DGAT1 in skeletal muscle in fat-induced insulin resistance ApoE -/- mice. Zhong Yao Cai. 38 (6), 1237–1241.
Johnson, T. O., Ermolieff, J., Jirousek, M. R. (2002). Protein tyrosine phosphatase 1B inhibitors for diabetes. Nat. Rev. Drug Discov. 1 (9), 696–709. doi: 10.1038/nrd895
Joost, H., Thorens, B. (2001). The extended GLUT-family of sugar/polyol transport facilitators: nomenclature, sequence characteristics, and potential function of its novel members (review). Mol. Membr. Biol. 18 (4), 247–256. doi: 10.1080/09687680110090456
Jung, H. A., Bhakta, H. K., Min, B. S., Choi, J. S. (2016). Fucosterol activates the insulin signaling pathway in insulin resistant HepG2 cells via inhibiting PTP1B. Arch. Pharm. Res. 39 (10), 1454–1464. doi: 10.1007/s12272-016-0819-4
Jung, H. J., Seong, S. H., Ali, M. Y., Min, B. S., Jung, H. A., Choi, J. S. (2017). Alpha-Methyl artoflavanocoumarin from Juniperus chinensis exerts anti-diabetic effects by inhibiting PTP1B and activating the PI3K/Akt signaling pathway in insulin-resistant HepG2 cells. Arch. Pharm. Res. 40 (12), 1403–1413. doi: 10.1007/s12272-017-0992-0
Kaatabi, H., Bamosa, A. O., Badar, A., Al-Elq, A., Abou-Hozaifa, B., Lebda, F., et al. (2015). Nigella sativa improves glycemic control and ameliorates oxidative stress in patients with type 2 diabetes mellitus: placebo controlled participant blinded clinical trial. PLoS One 10 (2), e0113486. doi: 10.1371/journal.pone.0113486
Kadowaki, T., Yamauchi, T. (2011). Adiponectin receptor signaling: a new layer to the current model. Cell Metab. 13 (2), 123–124. doi: 10.1016/j.cmet.2011.01.012
Kahn, B. B., Alquier, T., Carling, D., Hardie, D. G. (2005). AMP-activated protein kinase: ancient energy gauge provides clues to modern understanding of metabolism. Cell Metab. 1 (1), 15–25. doi: 10.1016/j.cmet.2004.12.003
Kahn, S. E., Hull, R. L., Utzschneider, K. M. (2006). Mechanisms linking obesity to insulin resistance and type 2 diabetes. Nature 444 (7121), 840–846. doi: 10.1038/nature05482
Kaidanovich, O., Eldar-Finkelman, H. (2002). The role of glycogen synthase kinase-3 in insulin resistance and type 2 diabetes. Expert Opin. Ther.Targets. 6 (5), 555–561. doi: 10.1517/14728222.6.5.555
Kan, J., Velliquette, R. A., Grann, K., Burns, C. R., Scholten, J., Tian, F., et al. (2017). A novel botanical formula prevents diabetes by improving insulin resistance. BMC Complement. Altern. Med. 17 (1), 352. doi: 10.1186/s12906-017-1848-3
Kho, M. C., Lee, Y. J., Cha, J. D., Choi, K. M., Kang, D. G., Lee, H. S. (2014). Gastrodia elata ameliorates high-fructose diet-induced lipid metabolism and endothelial dysfunction. Evid. Based Complement. Alternat. Med. 2014, 101624. doi: 10.1155/2014/101624
Kho, M. C., Lee, Y. J., Park, J. H., Kim, H. Y., Yoon, J. J., Ahn, Y. M., et al. (2016). Fermented red ginseng potentiates improvement of metabolic dysfunction in metabolic syndrome rat models. Nutrients 8 (6), 1–15. doi: 10.3390/nu8060369
Kido, Y., Burks, D., Withers, D., Bruning, J., Kahn, C., White, M., et al. (2000). Tissue-specific insulin resistance in mice with mutations in the insulin receptor, IRS-1, and IRS-2. J. Clin. Invest. 105 (2), 199–205. doi: 10.1172/JCI7917
Kim, J. M., Park, C. H., Park, S. K., Seung, T. W., Kang, J. Y., Ha, J. S., et al. (2017). Ginsenoside Re ameliorates brain insulin resistance and cognitive dysfunction in high fat diet-induced C57BL/6 mice. J. Agric. Food Chem. 65 (13), 2719–2729. doi: 10.1021/acs.jafc.7b00297
Kim, Y. J., Choi, J. Y., Ryu, R., Lee, J., Cho, S. J., Kwon, E. Y., et al. (2016). Platycodon grandiflorus root extract attenuates body fat mass, hepatic steatosis and insulin resistance through the interplay between the liver and adipose tissue. Nutrients 8 (9), 1–10. doi: 10.3390/nu8090532
Kishton, R., Barnes, C., Nichols, A., Cohen, S., Gerriets, V., Siska, P., et al. (2016). AMPK is essential to balance glycolysis and mitochondrial metabolism to control T-ALL cell stress and survival. Cell Metab. 23 (4), 649–662. doi: 10.1016/j.cmet.2016.03.008
Kong, P., Chi, R., Zhang, L., Wang, N., Lu, Y. (2013). Effects of paeoniflorin on tumor necrosis factor-alpha-induced insulin resistance and changes of adipokines in 3T3-L1 adipocytes. Fitoterapia. 91, 44–50. doi: 10.1016/j.fitote.2013.08.010
Koo, S. H., Satoh, H., Herzig, S., Lee, C. H., Hedrick, S., Kulkarni, R., et al. (2004). PGC-1 promotes insulin resistance in liver through PPAR-alpha-dependent induction of TRB-3. Nat. Med. 10 (5), 530–534. doi: 10.1038/nm1044
Kuai, M., Li, Y., Sun, X., Ma, Z., Lin, C., Jing, Y., et al. (2016). A novel formula Sang-Tong-Jian improves glycometabolism and ameliorates insulin resistance by activating PI3K/AKT pathway in type 2 diabetic KKAy mice. Biomed. Pharmacother. 84, 1585–1594. doi: 10.1016/j.biopha.2016.10.101
Kubota, N., Yano, W., Kubota, T., Yamauchi, T., Itoh, S., Kumagai, H., et al. (2007). Adiponectin stimulates AMP-activated protein kinase in the hypothalamus and increases food intake. Cell Metab. 6 (1), 55–68. doi: 10.1016/j.cmet.2007.06.003
Kubota, T., Kubota, N., Moroi, M., Terauchi, Y., Kobayashi, T., Kamata, K., et al. (2003). Lack of insulin receptor substrate-2 causes progressive neointima formation in response to vessel injury. Circulation 107 (24), 3073–3080. doi: 10.1161/01.CIR.0000070937.52035.25
Kullmann, S., Heni, M., Hallschmid, M., Fritsche, A., Preissl, H., Haring, H. U. (2016). Brain insulin resistance at the crossroads of metabolic and cognitive disorders in humans. Physiol. Rev. 96 (4), 1169–1209. doi: 10.1152/physrev.00032.2015
Kuo, Y. H., Lin, C. H., Shih, C. C. (2015). Ergostatrien-3beta-ol from Antrodia camphorata inhibits diabetes and hyperlipidemia in high-fat-diet treated mice via regulation of hepatic related genes, glucose transporter 4, and AMP-activated protein kinase phosphorylation. J. Agric. Food Chem. 63 (9), 2479–2489. doi: 10.1021/acs.jafc.5b00073
Lage, R., Dieguez, C., Vidal-Puig, A., Lopez, M. (2008). AMPK: a metabolic gauge regulating whole-body energy homeostasis. Trends Mol. Med. 14 (12), 539–549. doi: 10.1016/j.molmed.2008.09.007
Lawan, A., Bennett, A. M. (2017). Mitogen-activated protein kinase regulation in hepatic metabolism. Trends Endocrinol. Metab. 28 (12), 868–878. doi: 10.1016/j.tem.2017.10.007
Le, L., Jiang, B., Wan, W., Zhai, W., Xu, L., Hu, K., et al. (2016). Metabolomics reveals the protective of Dihydromyricetin on glucose homeostasis by enhancing insulin sensitivity. Sci. Rep. 6, 36184. doi: 10.1038/srep36184
Leavens, K. F., Easton, R. M., Shulman, G. I., Previs, S. F., Birnbaum, M. J. (2009). Akt2 is required for hepatic lipid accumulation in models of insulin resistance. Cell Metab. 10 (5), 405–418. doi: 10.1016/j.cmet.2009.10.004
Lee, J. B., Yoon, S. J., Lee, S. H., Lee, M. S., Jung, H., Kim, T. D., et al. (2017). Ginsenoside Rg3 ameliorated HFD-induced hepatic steatosis through downregulation of STAT5-PPARgamma. J. Endocrinol. 235 (3), 223–235. doi: 10.1530/JOE-17-0233
Lee, S. M., Lee, Y. J., Choi, J. H., Kho, M. C., Yoon, J. J., Shin, S. H., et al. (2014). Gal-geun-dang-gwi-tang improves diabetic vascular complication in apolipoprotein E KO mice fed a western diet. BMC Complement. Altern. Med. 14, 453. doi: 10.1186/1472-6882-14-453
Leem, K. H., Kim, M. G., Hahm, Y. T., Kim, H. K. (2016). Hypoglycemic effect of Opuntia ficus-indica var. Nutrients 8 (12), 1–15. doi: 10.3390/nu8120800
Levelt, E., Rodgers, C. T., Clarke, W. T., Mahmod, M., Ariga, R., Francis, J. M., et al. (2016). Cardiac energetics, oxygenation, and perfusion during increased workload in patients with type 2 diabetes mellitus. Eur. Heart J. 37 (46), 3461–3469. doi: 10.1093/eurheartj/ehv442
Li, D. C., Zhong, X. K., Zeng, Z. P., Jiang, J. G., Li, L., Zhao, M. M., et al. (2009). Application of targeted drug delivery system in Chinese medicine. J. Control Release 138 (2), 103–112. doi: 10.1016/j.jconrel.2009.05.008
Li, J. B., Xu, L. J., Dong, H., Huang, Z. Y., Zhao, Y., Chen, G., et al. (2013). Effects of Chinese Fructus Mume formula and its separated prescription extract on insulin resistance in type 2 diabetic rats. J. Huazhong Univ. Sci. Technol. Med. Sci. 33 (6), 877–885. doi: 10.1007/s11596-013-1215-7
Li, L., Yoshitomi, H., Wei, Y., Qin, L., Zhou, J., Xu, T., et al. (2015). Tang-Nai-Kang alleviates pre-diabetes and metabolic disorders and induces a gene expression switch toward fatty acid oxidation in SHR.Cg-Leprcp/NDmcr rats. PLoS One 10 (4), e0122024. doi: 10.1371/journal.pone.0122024
Li, W., Chen, Q., Xie, Y., Hu, J., Yang, S., Lin, M. (2018). Prevalence and degree of insulin resistance in Chinese Han women with PCOS: Results from euglycemic-hyperinsulinemic clamps. Clin. Endocrinol. (Oxf). 90 (1), 138–144. doi: 10.1111/cen.13860
Li, X., Gong, H., Yang, S., Yang, L., Fan, Y., Zhou, Y. (2017). Pectic bee pollen polysaccharide from Rosa rugosa alleviates diet-induced hepatic steatosis and insulin resistance via induction of AMPK/mTOR-mediated autophagy. Molecules 22 (5), 1–13. doi: 10.3390/molecules22050699
Li, Y., Tran, V. H., Kota, B. P., Nammi, S., Duke, C. C., Roufogalis, B. D. (2014). Preventative effect of Zingiber officinale on insulin resistance in a high-fat high-carbohydrate diet-fed rat model and its mechanism of action. Basic Clin. Pharmacol. Toxicol. 115 (2), 209–215. doi: 10.1111/bcpt.12196
Li, Y., Xu, S., Mihaylova, M. M., Zheng, B., Hou, X., Jiang, B., et al. (2011). AMPK phosphorylates and inhibits SREBP activity to attenuate hepatic steatosis and atherosclerosis in diet-induced insulin-resistant mice. Cell Metab. 13 (4), 376–388. doi: 10.1016/j.cmet.2011.03.009
Li, W., Zhang, S., Liu, H., Wang, L., Zhang, C., Leng, J., et al. (2014). Different associations of diabetes with β-cell dysfunction and insulin resistance among obese and nonobese chinese women with prior gestational diabetes mellitus. Diabetes Care 37 (9), 2533–2539. doi: 10.2337/dc14-0573
Lian, F., Tian, J., Chen, X., Li, Z., Piao, C., Guo, J., et al. (2015). The efficacy and safety of Chinese herbal medicine Jinlida as add-on medication in type 2 diabetes patients ineffectively managed by metformin monotherapy: a double-blind, randomized, placebo-controlled, multicenter trial. PLoS One 10 (6), e0130550. doi: 10.1371/journal.pone.0130550
Lillioja, S., Mott, D. M., Spraul, M., Ferraro, R., Foley, J. E., Ravussin, E., et al. (1993). Insulin resistance and insulin secretory dysfunction as precursors of non-insulin-dependent diabetes mellitus. N. Engl. J. Med. 329 (27), 1988–1992. doi: 10.1056/NEJM199312303292703
Liu, C. Y., Huang, C. J., Huang, L. H., Chen, I. J., Chiu, J. P., Hsu, C. H. (2014). Effects of green tea extract on insulin resistance and glucagon-like peptide 1 in patients with type 2 diabetes and lipid abnormalities: a randomized, double-blinded, and placebo-controlled trial. PLoS One 9 (3), e91163. doi: 10.1371/journal.pone.0091163
Liu, D., Zhang, Y., Liu, Y., Hou, L., Li, S., Tian, H., et al. (2018). Berberine modulates gut microbiota and reduces insulin resistance via the TLR4 signaling pathway. Exp. Clin. Endocrinol. Diabetes 126 (8), 513–520. doi: 10.1055/s-0043-125066
Liu, H. W., Huang, W. C., Yu, W. J., Chang, S. J. (2015). Toona Sinensis ameliorates insulin resistance via AMPK and PPARgamma pathways. Food Funct. 6 (6), 1855–1864. doi: 10.3390/molecules201219794
Liu, J., Wang, X., Chen, Y.-P., Mao, L.-F., Shang, J., Sun, H.-B., et al. (2014). Maslinic acid modulates glycogen metabolism by enhancing the insulin signaling pathway and inhibiting glycogen phosphorylase. Chin. J. Nat. Med. 12 (4), 259–265. doi: 10.1016/S1875-5364(14)60052-2
Liu, Q., Li, X., Li, C., Zheng, Y., Peng, G. (2015). 1-Deoxynojirimycin alleviates insulin resistance via activation of insulin signaling PI3K/AKT pathway in skeletal muscle of db/db mice. Molecules 20 (12), 21700–21714. doi: 10.1039/C5FO00056D
Liu, Q., Liu, S., Gao, L., Sun, S., Huan, Y., Li, C., et al. (2017). Anti-diabetic effects and mechanisms of action of a Chinese herbal medicine preparation JQ-R in vitro and in diabetic KK(Ay) mice. Acta. Pharm. Sin B. 7 (4), 461–469. doi: 10.1016/j.apsb.2017.04.010
Liu, Y., Li, X., Xie, C., Luo, X., Bao, Y., Wu, B., et al. (2016). Prevention effects and possible molecular mechanism of mulberry leaf extract and its formulation on rats with insulin-insensitivity. PLoS One 11 (4), e0152728. doi: 10.1371/journal.pone.0152728
Liu, Y., Song, A., Zang, S., Wang, C., Song, G., Li, X., et al. (2015). Jinlida reduces insulin resistance and ameliorates liver oxidative stress in high-fat fed rats. J. Ethnopharmacol. 162, 244–252. doi: 10.1016/j.jep.2014.12.040
Liu, Z. Q., Liu, T., Chen, C., Li, M. Y., Wang, Z. Y., Chen, R. S., et al. (2015). Fumosorinone, a novel PTP1B inhibitor, activates insulin signaling in insulin-resistance HepG2 cells and shows anti-diabetic effect in diabetic KKAy mice. Toxicol. Appl. Pharmacol. 285 (1), 61–70. doi: 10.1016/j.taap.2015.03.011
Liu, Z., Kuang, W., Xu, X., Li, D., Zhu, W., Lan, Z., et al. (2018). Putative identification of components in Zengye decoction and their effects on glucose consumption and lipogenesis in insulin-induced insulin-resistant HepG2 cells. J. Chromatogr. B Analyt. Technol. Biomed. Life Sci. 1073, 145–153. doi: 10.1016/j.jchromb.2017.12.019
Lumeng, C. N., Bodzin, J. L., Saltiel, A. R. (2007). Obesity induces a phenotypic switch in adipose tissue macrophage polarization. J. Clin. Invest. 117 (1), 175–184. doi: 10.1172/JCI29881
Luna-Vital, D., Weiss, M., Gonzalez de Mejia, E. (2017). Anthocyanins from purple corn ameliorated tumor necrosis factor-alpha-induced inflammation and insulin resistance in 3T3-L1 adipocytes via activation of insulin signaling and enhanced GLUT4 translocation. Mol. Nutr. Food Res. 61 (12), 1–41. doi: 10.1002/mnfr.201700362
Luo, J., Sobkiw, C. L., Hirshman, M. F., Logsdon, M. N., Li, T. Q., Goodyear, L. J., et al. (2006). Loss of class IA PI3K signaling in muscle leads to impaired muscle growth, insulin response, and hyperlipidemia. Cell Metab. 3 (5), 355–366. doi: 10.1016/j.cmet.2006.04.003
Luo, Y. H., Zhu, M., Wang, D. G., Yang, Y. S., Tan, T., Zhu, H., et al. (2018). Yangxin Tongmai formula ameliorates impaired glucose tolerance in children with Graves’ disease through upregulation of the insulin receptor levels. Acta. Pharmacol. Sin. 39 (6), 923–929. doi: 10.1038/aps.2017.94
Ma, C., Yu, H., Xiao, Y., Wang, H. (2017). Momordica charantia extracts ameliorate insulin resistance by regulating the expression of SOCS-3 and JNK in type 2 diabetes mellitus rats. Pharm. Biol. 55 (1), 2170–2177. doi: 10.1080/13880209.2017.1396350
Manaf, A., Tjandrawinata, R. R., Malinda, D. (2016). Insulin sensitizer in prediabetes: a clinical study with DLBS3233, a combined bioactive fraction of Cinnamomum burmanii and Lagerstroemia speciosa. Drug Des. Devel. Ther. 10, 1279–1289. doi: 10.2147/DDDT.S97568
Martel, J., Ojcius, D. M., Chang, C. J., Lin, C. S., Lu, C. C., Ko, Y. F., et al. (2017). Anti-obesogenic and antidiabetic effects of plants and mushrooms. Nat. Rev. Endocrinol. 13 (3), 149–160. doi: 10.1038/nrendo.2016.142
Martinez, A., Castro, A., Dorronsoro, I., Alonso, M. (2002). Glycogen synthase kinase 3 (GSK-3) inhibitors as new promising drugs for diabetes, neurodegeneration, cancer, and inflammation. Med. Res. Rev. 22 (4), 373–384. doi: 10.1002/med.10011
Massaro, M., Scoditti, E., Pellegrino, M., Carluccio, M. A., Calabriso, N., Wabitsch, M., et al. (2016). Therapeutic potential of the dual peroxisome proliferator activated receptor (PPAR)alpha/gamma agonist aleglitazar in attenuating TNF-alpha-mediated inflammation and insulin resistance in human adipocytes. Pharmacol. Res. 107, 125–136. doi: 10.1016/j.phrs.2016.02.027
Matheny, R. W., Jr., Geddis, A. V., Abdalla, M. N., Leandry, L. A., Ford, M., McClung, H. L., et al. (2018). AKT2 is the predominant AKT isoform expressed in human skeletal muscle. Physiol. Rep. 6 (6), e13652. doi: 10.14814/phy2.13652
Matthews, D. R., Hosker, J. P., Rudenski, A. S., Naylor, B. A., Treacher, D. F., Turner, R. C. (1985). Homeostasis model assessment: insulin resistance and beta-cell function from fasting plasma glucose and insulin concentrations in man. Diabetologia 28 (7), 412–419. doi: 10.1007/BF00280883
Meex, R. C. R., Watt, M. J. (2017). Hepatokines: linking nonalcoholic fatty liver disease and insulin resistance. Nat. Rev. Endocrinol. 13 (9), 509–520. doi: 10.1038/nrendo.2017.56
Mendez-Del Villar, M., Puebla-Perez, A. M., Sanchez-Pena, M. J., Gonzalez-Ortiz, L. J., Martinez-Abundis, E., Gonzalez-Ortiz, M. (2016). Effect of Artemisia dracunculus administration on glycemic control, insulin sensitivity, and insulin secretion in patients with impaired glucose tolerance. J. Med. Food. 19 (5), 481–485. doi: 10.1089/jmf.2016.0005
Miki, S., Inokuma, K. I., Takashima, M., Nishida, M., Sasaki, Y., Ushijima, M., et al. (2017). Aged garlic extract suppresses the increase of plasma glycated albumin level and enhances the AMP-activated protein kinase in adipose tissue in TSOD mice. Mol. Nutr. Food Res. 61 (5), 1–22. doi: 10.1002/mnfr.201600797
Minokoshi, Y., Alquier, T., Furukawa, N., Kim, Y., Lee, A., Xue, B., et al. (2004). AMP-kinase regulates food intake by responding to hormonal and nutrient signals in the hypothalamus. Nature 428 (6982), 569–574. doi: 10.1038/nature02440
Minokoshi, Y., Kim, Y., Peroni, O., Fryer, L., Müller, C., Carling, D., et al. (2002). Leptin stimulates fatty-acid oxidation by activating AMP-activated protein kinase. Nature 415 (6869), 339–343. doi: 10.1038/415339a
Mirfeizi, M., Mehdizadeh Tourzani, Z., Mirfeizi, S. Z., Asghari Jafarabadi, M., Rezvani, H. R., Afzali, M. (2016). Controlling type 2 diabetes mellitus with herbal medicines: a triple-blind randomized clinical trial of efficacy and safety. J. Diabetes 8 (5), 647–656. doi: 10.1111/1753-0407.12342
Mottillo, E. P., Desjardins, E. M., Crane, J. D., Smith, B. K., Green, A. E., Ducommun, S., et al. (2016). Lack of adipocyte AMPK exacerbates insulin resistance and hepatic steatosis through brown and beige adipose tissue function. Cell Metab. 24 (1), 118–129. doi: 10.1016/j.cmet.2016.06.006
Mozaffari-Khosravi, H., Talaei, B., Jalali, B. A., Najarzadeh, A., Mozayan, M. R. (2014). The effect of ginger powder supplementation on insulin resistance and glycemic indices in patients with type 2 diabetes: a randomized, double-blind, placebo-controlled trial. Complement. Ther. Med. 22 (1), 9–16. doi: 10.1016/j.ctim.2013.12.017
Na, H. Y., Seol, M. H., Kim, M., Lee, B. C. (2017). Effect of seyoeum on obesity, insulin resistance, and nonalcoholic fatty liver disease of high-fat diet-fed C57BL/6 Mice. Evid. Based Complement. Alternat. Med. 2017, 4658543. doi: 10.1155/2017/4658543
Nan Xia, J., Qin Zhang, D., Du, J., Wen, J. (2013). Regulation effects of TZQ-F on adipocyte differentiation and insulin action. J. Ethnopharmacol. 150 (2), 692–699. doi: 10.1016/j.jep.2013.09.038
Ng, M., Fleming, T., Robinson, M., Thomson, B., Graetz, N., Margono, C., et al. (2014). Global, regional, and national prevalence of overweight and obesity in children and adults during 1980-2013: a systematic analysis for the Global Burden of Disease Study 2013. Lancet 384 (9945), 766–781. doi: 10.1016/S0140-6736(14)60460-8
Ng, Y., Ramm, G., Lopez, J. A., James, D. E. (2008). Rapid activation of Akt2 is sufficient to stimulate GLUT4 translocation in 3T3-L1 adipocytes. Cell Metab. 7 (4), 348–356. doi: 10.1016/j.cmet.2008.02.008
Nie, J., Chang, Y. (2017). Caffeic acid phenethyl ester (propolis extract) ameliorates insulin resistance by inhibiting JNK and NF-kappaB inflammatory pathways in diabetic mice and HepG2 cell models. J. Agric. Food Chem. 65 (41), 9041–9053. doi: 10.1021/acs.jafc.7b02880
Oh, J., Lee, H., Lim, H., Woo, S., Shin, S. S., Yoon, M. (2015). The herbal composition GGEx18 from Laminaria japonica, Rheum palmatum, and Ephedra sinica inhibits visceral obesity and insulin resistance by upregulating visceral adipose genes involved in fatty acid oxidation. Pharm. Biol. 53 (2), 301–312. doi: 10.3109/13880209.2014.917328
Ooi, J., Adamu, H. A., Imam, M. U., Ithnin, H., Ismail, M. (2018). Polyphenol-rich ethyl acetate fraction isolated from Molineria latifolia ameliorates insulin resistance in experimental diabetic rats via IRS1/AKT activation. Biomed. Pharmacother. 98, 125–133. doi: 10.1016/j.biopha.2017.12.002
Ozcan, U., Cao, Q., Yilmaz, E., Lee, A., Iwakoshi, N., Ozdelen, E., et al. (2004). Endoplasmic reticulum stress links obesity, insulin action, and type 2 diabetes. Science 306 (5695), 457–461. doi: 10.1126/science.1103160
Pandey, J., Maurya, R., Raykhera, R., Srivastava, M. N., Yadav, P. P., Tamrakar, A. K. (2014). Murraya koenigii (L). J. Sci. Food Agric. 94 (11), 2282–2288. doi: 10.1002/jsfa.6555
Parimala, M., Debjani, M., Vasanthi, H. R., Shoba, F. G. (2015). Nymphaea nouchali Burm. J. Adv. Pharm. Technol. Res. 6 (4), 183–189. doi: 10.4103/2231-4040.165013
Park, C. H., Kim, D. H., Park, M. H., Kim, M. K., Kim, N. D., Kim, C. M., et al. (2014). Chinese prescription Kangen-karyu and Salviae Miltiorrhizae Radix improve age-related oxidative stress and inflammatory response through the PI3K/Akt or MAPK pathways. Am. J. Chin. Med. 42 (4), 987–1005. doi: 10.1142/S0192415X14500621
Park, E. C., Kim, S. I., Hong, Y., Hwang, J. W., Cho, G. S., Cha, H. N., et al. (2014). Inhibition of CYP4A reduces hepatic endoplasmic reticulum stress and features of diabetes in mice. Gastroenterology 147 (4), 860–869. doi: 10.1053/j.gastro.2014.06.039
Park, S. E., Park, C. Y., Sweeney, G. (2015). Biomarkers of insulin sensitivity and insulin resistance: Past, present and future. Crit. Rev. Clin. Lab. Sci: 52 (4), 180–190. doi: 10.3109/10408363.2015.1023429
Patel, S., Woodgett, J. (2008). Glycogen synthase kinase-3 and cancer: good cop, bad cop? Cancer Cell. 14 (5), 351–353. doi: 10.1016/j.ccr.2008.10.013
Peraldi, P., Xu, M., Spiegelman, B. (1997). Thiazolidinediones block tumor necrosis factor-alpha-induced inhibition of insulin signaling. J. Clin. Invest. 100 (7), 1863–1869. doi: 10.1172/JCI119715
Perez-Rubio, K. G., Gonzalez-Ortiz, M., Martinez-Abundis, E., Robles-Cervantes, J. A., Espinel-Bermudez, M. C. (2013). Effect of berberine administration on metabolic syndrome, insulin sensitivity, and insulin secretion. Metab. Syndr. Relat. Disord. 11 (5), 366–369. doi: 10.1089/met.2012.0183
Qiang, G., Wenzhai, C., Huan, Z., Yuxia, Z., Dongdong, Y., Sen, Z., et al. (2015). Effect of Sancaijiangtang on plasma nitric oxide and endothelin-1 levels in patients with type 2 diabetes mellitus and vascular dementia: a single-blind randomized controlled trial. J. Tradit. Chin. Med. 35 (4), 375–380. doi: 10.1016/S0254-6272(15)30112-6
Qin, J. H., Ma, J. Z., Yang, X. W., Hu, Y. J., Zhou, J., Fu, L. C., et al. (2015). A triterpenoid inhibited hormone-induced adipocyte differentiation and alleviated dexamethasone-induced insulin resistance in 3T3-L1 adipocytes. Nat. Prod. Bioprospect. 5 (3), 159–166. doi: 10.1007/s13659-015-0063-5
Rabiei, K., Ebrahimzadeh, M. A., Saeedi, M., Bahar, A., Akha, O., Kashi, Z. (2018). Effects of a hydroalcoholic extract of Juglans regia (walnut) leaves on blood glucose and major cardiovascular risk factors in type 2 diabetic patients: a double-blind, placebo-controlled clinical trial. BMC Complement. Altern. Med. 18 (1), 206. doi: 10.1186/s12906-018-2268-8
Rafraf, M., Zemestani, M., Asghari-Jafarabadi, M. (2015). Effectiveness of chamomile tea on glycemic control and serum lipid profile in patients with type 2 diabetes. J. Endocrinol. Invest. 38 (2), 163–170. doi: 10.1007/s40618-014-0170-x
Reeds, D. N., Patterson, B. W., Okunade, A., Holloszy, J. O., Polonsky, K. S., Klein, S. (2011). Ginseng and ginsenoside Re do not improve beta-cell functionor insulin sensitivity in overweight and obese subjects with impaired glucosetolerance or diabetes. Diabetes Care 34, 1071–1076. doi: 10.2337/dc10-2299
Richard, A. J., Burris, T. P., Sanchez-Infantes, D., Wang, Y., Ribnicky, D. M., Stephens, J. M. (2014). Artemisia extracts activate PPARgamma, promote adipogenesis, and enhance insulin sensitivity in adipose tissue of obese mice. Nutrition 30 (7–8 Suppl), S31–36. doi: 10.1016/j.nut.2014.02.013
Richter, E., Hargreaves, M. (2013). Exercise, GLUT4, and skeletal muscle glucose uptake. Physiol. Rev. 93 (3), 993–1017. doi: 10.1152/physrev.00038.2012
Riehle, C., Abel, E. D. (2016). Insulin signaling and heart failure. Circ. Res. 118 (7), 1151–1169. doi: 10.1161/CIRCRESAHA.116.306206
Rinella, M. E. (2015). Nonalcoholic fatty liver disease: a systematic review. JAMA 313 (22), 2263–2273. doi: 10.1001/jama.2015.5370
Roshan, H., Nikpayam, O., Sedaghat, M., Sohrab, G. (2018). Effects of green coffee extract supplementation on anthropometric indices, glycaemic control, blood pressure, lipid profile, insulin resistance and appetite in patients with the metabolic syndrome: a randomised clinical trial. Br. J. Nutr. 119 (3), 250–258. doi: 10.1017/S0007114517003439
Rui, L., Aguirre, V., Kim, J., Shulman, G., Lee, A., Corbould, A., et al. (2001). Insulin/IGF-1 and TNF-alpha stimulate phosphorylation of IRS-1 at inhibitory Ser307 via distinct pathways. J. Clin. Invest. 107 (2), 181–189. doi: 10.1172/JCI10934
Saad, M., Araki, E., Miralpeix, M., Rothenberg, P., White, M., Kahn, C. (1992). Regulation of insulin receptor substrate-1 in liver and muscle of animal models of insulin resistance. J. Clin. Invest. 90 (5), 1839–1849. doi: 10.1172/JCI116060
Sabio, G., Cavanagh-Kyros, J., Ko, H., Jung, D., Gray, S., Jun, J., et al. (2009). Prevention of steatosis by hepatic JNK1. Cell Metab. 10 (6), 491–498. doi: 10.1016/j.cmet.2009.09.007
Saito, T., Nishida, M., Saito, M., Tanabe, A., Eitsuka, T., Yuan, S. H., et al. (2016). The fruit of Acanthopanax senticosus (Rupr. Nutr. Res. 36 (10), 1090–1097. doi: 10.1016/j.nutres.2016.09.004
Saltiel, A., Kahn, C. (2001). Insulin signalling and the regulation of glucose and lipid metabolism. Nature 414 (6865), 799–806. doi: 10.1038/414799a
Samadi, N., Mozaffari-Khosravi, H., Rahmanian, M., Askarishahi, M. (2017). Effects of bee propolis supplementation on glycemic control, lipid profile and insulin resistance indices in patients with type 2 diabetes: a randomized, double-blind clinical trial. J. Integr. Med. 15 (2), 124–134. doi: 10.1016/S2095-4964(17)60315-7
Samuel, V. T., Shulman, G. I. (2016). The pathogenesis of insulin resistance: integrating signaling pathways and substrate flux. J. Clin. Invest. 126 (1), 12–22. doi: 10.1172/JCI77812
Sano, H., Eguez, L., Teruel, M. N., Fukuda, M., Chuang, T. D., Chavez, J. A., et al. (2007). Rab10, a target of the AS160 Rab GAP, is required for insulin-stimulated translocation of GLUT4 to the adipocyte plasma membrane. Cell Metab. 5 (4), 293–303. doi: 10.1016/j.cmet.2007.03.001
Sasaki, H., Asanuma, H., Fujita, M., Takahama, H., Wakeno, M., Ito, S., et al. (2009). Metformin prevents progression of heart failure in dogs: role of AMP-activated protein kinase. Circulation 119 (19), 2568–2577. doi: 10.1161/CIRCULATIONAHA.108.798561
Sathiyabama, R. G., Rajiv Gandhi, G., Denadai, M., Sridharan, G., Jothi, G., Sasikumar, P., et al. (2018). Evidence of insulin-dependent signalling mechanisms produced by Citrus sinensis (L). Food Chem. Toxicol. 116 (Pt B), 86–99. doi: 10.1016/j.fct.2018.03.050
Seki, E., Brenner, D. A., Karin, M. (2012). A liver full of JNK: signaling in regulation of cell function and disease pathogenesis, and clinical approaches. Gastroenterology 143 (2), 307–320. doi: 10.1053/j.gastro.2012.06.004
Seo, E., Kim, S., Lee, S. J., Oh, B. C., Jun, H. S. (2015). Ginseng berry extract supplementation improves age-related decline of insulin signaling in mice. Nutrients 7 (4), 3038–3053. doi: 10.3390/nu7043038
Seo, H. A., Lee, I. K. (2013). The role of Nrf2: adipocyte differentiation, obesity, and insulin resistance. Oxid. Med. Cell Longev. 2013, 184598. doi: 10.1155/2013/184598
Shen, Y., Honma, N., Kobayashi, K., Jia, L. N., Hosono, T., Shindo, K., et al. (2014). Cinnamon extract enhances glucose uptake in 3T3-L1 adipocytes and C2C12 myocytes by inducing LKB1-AMP-activated protein kinase signaling. PLoS One 9 (2), e87894. doi: 10.1371/journal.pone.0087894
Shi, X., Zhou, F., Li, X., Chang, B., Li, D., Wang, Y., et al. (2013). Central GLP-2 enhances hepatic insulin sensitivity via activating PI3K signaling in POMC neurons. Cell Metab. 18 (1), 86–98. doi: 10.1016/j.cmet.2013.06.014
Shi, Y. L., Liu, W. J., Zhang, X. F., Su, W. J., Chen, N. N., Lu, S. H., et al. (2016). Effect of Chinese herbal medicine Jinlida granule in treatment of patients with impaired glucose tolerance. Chin. Med. J. (Engl) 129 (19), 2281–2286. doi: 10.4103/0366-6999.190676
Shimomura, I., Matsuda, M., Hammer, R., Bashmakov, Y., Brown, M., Goldstein, J. (2000). Decreased IRS-2 and increased SREBP-1c lead to mixed insulin resistance and sensitivity in livers of lipodystrophic and ob/ob mice. Mol. Cell 6 (1), 77–86. doi: 10.1016/S1097-2765(05)00010-9
Singh, J., Kakkar, P. (2013). Modulation of liver function, antioxidant responses, insulin resistance and glucose transport by Oroxylum indicum stem bark in STZ induced diabetic rats. Food Chem. Toxicol. 62, 722–731. doi: 10.1016/j.fct.2013.09.035
Siwicki, M., Engblom, C., Pittet, M. J. (2016). Gal3 links inflammation and insulin resistance. Cell Metab. 24 (5), 655–656. doi: 10.1016/j.cmet.2016.10.014
Stenbit, A., Tsao, T., Li, J., Burcelin, R., Geenen, D., Factor, S., et al. (1997). GLUT4 heterozygous knockout mice develop muscle insulin resistance and diabetes. Nat. Med. 3 (10), 1096–1101. doi: 10.1038/nm1097-1096
Sun, X., Sun, H., Zhang, J., Ji, X. (2016). Artemisia extract improves insulin sensitivity in women with gestational diabetes mellitus by up-regulating adiponectin. J. Clin. Pharmacol. 56 (12), 1550–1554. doi: 10.1002/jcph.755
Sun, Z., Zhan, L., Liang, L., Sui, H., Zheng, L., Sun, X., et al. (2016). ZiBu PiYin recipe prevents diabetes-associated cognitive decline in rats: possible involvement of ameliorating mitochondrial dysfunction, insulin resistance pathway and histopathological changes. BMC Complement. Altern. Med. 16, 200. doi: 10.1186/s12906-016-1177-y
Szendroedi, J., Phielix, E., Roden, M. (2011). The role of mitochondria in insulin resistance and type 2 diabetes mellitus. Nat. Rev. Endocrinol. 8 (2), 92–103. doi: 10.1038/nrendo.2011.138
Talbot, K., Wang, H. Y. (2014). The nature, significance, and glucagon-like peptide-1 analog treatment of brain insulin resistance in Alzheimer’s disease. Alzheimers Dement. 10 (1 Suppl), S12–25. doi: 10.1016/j.jalz.2013.12.007
Tamemoto, H., Kadowaki, T., Tobe, K., Yagi, T., Sakura, H., Hayakawa, T., et al. (1994). Insulin resistance and growth retardation in mice lacking insulin receptor substrate-1. Nature 372 (6502), 182–186. doi: 10.1038/372182a0
Tang, M., Gao, G., Rueda, C. B., Yu, H., Thibodeaux, D. N., Awano, T., et al. (2017). Brain microvasculature defects and Glut1 deficiency syndrome averted by early repletion of the glucose transporter-1 protein. Nat. Commun. 8, 14152. doi: 10.1038/ncomms14152
Taniguchi, C., Ueki, K., Kahn, R. (2005). Complementary roles of IRS-1 and IRS-2 in the hepatic regulation of metabolism. J. Clin. Invest. 115 (3), 718–727. doi: 10.1172/JCI23187
Thorens, B., Wu, Y., Leahy, J., Weir, G. (1992). The loss of GLUT2 expression by glucose-unresponsive beta cells of db/db mice is reversible and is induced by the diabetic environment. J. Clin. Invest. 90 (1), 77–85. doi: 10.1172/JCI115858
Tiganis, T. (2013). PTP1B and TCPTP – nonredundant phosphatases in insulin signaling and glucose homeostasis. FEBS J. 280 (2), 445–458. doi: 10.1111/j.1742-4658.2012.08563.x
Tuttle, R., Gill, N., Pugh, W., Lee, J., Koeberlein, B., Furth, E., et al. (2001). Regulation of pancreatic beta-cell growth and survival by the serine/threonine protein kinase Akt1/PKBalpha. Nat. Med. 7 (10), 1133–1137. doi: 10.1038/nm1001-1133
Vallerie, S., Hotamisligil, G. (2010). The role of JNK proteins in metabolism. Sci. Transl. Med. 2 (60), 60rv65. doi: 10.1126/scitranslmed.3001007
Wang, C., Li, Y., Hao, M., Li, W. (2018). Astragaloside IV inhibits triglyceride accumulation in insulin-resistant HepG2 cells via AMPK-induced SREBP-1c phosphorylation. Front. Pharmacol. 9, 345. doi: 10.3389/fphar.2018.00345
Wang, L. H., Wang, X., Yu, X. Z., Xu, W. T. (2016). Potent therapeutic effects of shouwu jiangqi decoction on polycystic ovary syndrome with insulin resistance in rats. Chin. J. Integr. Med. 22 (2), 116–123. doi: 10.1007/s11655-015-2147-9
Wang, L., Ye, X., Hua, Y., Song, Y. (2018). Berberine alleviates adipose tissue fibrosis by inducing AMP-activated kinase signaling in high-fat diet-induced obese mice. Biomed. Pharmacother. 105, 121–129. doi: 10.1016/j.biopha.2018.05.110
Wang, X. A., Zhang, R., She, Z. G., Zhang, X. F., Jiang, D. S., Wang, T., et al. (2014). Interferon regulatory factor 3 constrains IKKbeta/NF-kappaB signaling to alleviate hepatic steatosis and insulin resistance. Hepatology 59 (3), 870–885. doi: 10.1002/hep.26751
Wang, Z. Q., Zhang, X. H., Yu, Y., Tipton, R. C., Raskin, I., Ribnicky, D., et al. (2013). Artemisia scoparia extract attenuates non-alcoholic fatty liver disease in diet-induced obesity mice by enhancing hepatic insulin and AMPK signaling independently of FGF21 pathway. Metabolism 62 (9), 1239–1249. doi: 10.1016/j.metabol.2013.03.004
Watt, M. J., Dzamko, N., Thomas, W. G., Rose-John, S., Ernst, M., Carling, D., et al. (2006). CNTF reverses obesity-induced insulin resistance by activating skeletal muscle AMPK. Nat. Med. 12 (5), 541–548. doi: 10.1038/nm1383
Wen, H., Gris, D., Lei, Y., Jha, S., Zhang, L., Huang, M. T., et al. (2011). Fatty acid-induced NLRP3-ASC inflammasome activation interferes with insulin signaling. Nat. Immunol. 12 (5), 408–415. doi: 10.1038/ni.2022
Wensveen, F. M., Jelencic, V., Valentic, S., Sestan, M., Wensveen, T. T., Theurich, S., et al. (2015). NK cells link obesity-induced adipose stress to inflammation and insulin resistance. Nat. Immunol. 16 (4), 376–385. doi: 10.1038/ni.3120
White, M. (2003). Insulin signaling in health and disease. Science 302 (5651), 1710–1711. doi: 10.1126/science.1092952
Willette, A. A., Johnson, S. C., Birdsill, A. C., Sager, M. A., Christian, B., Baker, L. D., et al. (2015). Insulin resistance predicts brain amyloid deposition in late middle-aged adults. Alzheimers Dement. 11 (5), 504–510 e501. doi: 10.1016/j.jalz.2014.03.011
Winer, D. A., Luck, H., Tsai, S., Winer, S. (2016). The intestinal immune system in obesity and insulin resistance. Cell Metab. 23 (3), 413–426. doi: 10.1016/j.cmet.2016.01.003
Winnay, J., Solheim, M., Dirice, E., Sakaguchi, M., Noh, H., Kang, H., et al. (2016). PI3-kinase mutation linked to insulin and growth factor resistance in vivo. J. Clin. Invest. 126 (4), 1401–1412. doi: 10.1172/JCI84005
Woerdeman, J., Del Rio, D., Calani, L., Eringa, E. C., Smulders, Y. M., Serne, E. H. (2018). Red wine polyphenols do not improve obesity-associated insulin resistance: a randomized controlled trial. Diabetes Obes. Metab. 20 (1), 206–210. doi: 10.1111/dom.13044
Wood, I. S., Trayhurn, P. (2003). Glucose transporters (GLUT and SGLT): expanded families of sugar transport proteins. Br. J. Nutr. 89 (1), 3–9. doi: 10.1079/BJN2002763
Xiao, B., Heath, R., Saiu, P., Leiper, F. C., Leone, P., Jing, C., et al. (2007). Structural basis for AMP binding to mammalian AMP-activated protein kinase. Nature 449 (7161), 496–500. doi: 10.1038/nature06161
Xiao, B., Sanders, M. J., Underwood, E., Heath, R., Mayer, F. V., Carmena, D., et al. (2011). Structure of mammalian AMPK and its regulation by ADP. Nature 472 (7342), 230–233. doi: 10.1038/nature09932
Xiao, N., Yang, L. L., Yang, Y. L., Liu, L. W., Li, J., Liu, B., et al. (2017). Ginsenoside Rg5 inhibits succinate-associated lipolysis in adipose tissue and prevents muscle insulin resistance. Front. Pharmacol. 8, 43. doi: 10.3389/fphar.2017.00043
Xing, Q., Chen, Y. (2018). Antidiabetic effects of a Chinese herbal medicinal compound Sangguayin preparation via PI3K/Akt signaling pathway in db/db mice. Evid. Based Complement. Alternat. Med. 2018, 2010423. doi: 10.1155/2018/2010423
Xu, J., Pan, F., Xu, H. (2015). [Effect of Zhenggan Tang decoction on the serum levels of leptin, adiponectin and insulin resistance on HBV-induced cirrhotic patients]. Zhonghua Liu Xing Bing Xue Za Zhi. 36 (4), 399–401.
Yagishita, Y., Uruno, A., Fukutomi, T., Saito, R., Saigusa, D., Pi, J., et al. (2017). Nrf2 improves leptin and insulin resistance provoked by hypothalamic oxidative stress. Cell Rep. 18 (8), 2030–2044. doi: 10.1016/j.celrep.2017.01.064
Yan, F., Dai, G., Zheng, X. (2016). Mulberry anthocyanin extract ameliorates insulin resistance by regulating PI3K/AKT pathway in HepG2 cells and db/db mice. J. Nutr. Biochem. 36, 68–80. doi: 10.1016/j.jnutbio.2016.07.004
Yan, J., Wang, C., Jin, Y., Meng, Q., Liu, Q., Liu, Z., et al. (2018). Catalpol ameliorates hepatic insulin resistance in type 2 diabetes through acting on AMPK/NOX4/PI3K/AKT pathway. Pharmacol. Res. 130, 466–480. doi: 10.1016/j.phrs.2017.12.026
Yang, J., Leng, J., Li, J. J., Tang, J. F., Li, Y., Liu, B. L., et al. (2016). Corosolic acid inhibits adipose tissue inflammation and ameliorates insulin resistance via AMPK activation in high-fat fed mice. Phytomedicine 23 (2), 181–190. doi: 10.1016/j.phymed.2015.12.018
Yang, J.-L., Wang, J.-L., Huang, F., Liu, K., Liu, B.-L. (2014). Modified Si-Miao-San inhibits inflammation and promotes glucose disposal in adipocytes through regulation of AMP-kinase. Chin. J. Nat. Med. 12 (12), 911–919. doi: 10.1016/S1875-5364(14)60134-5
Yang, S. J., Choi, J. M., Park, S. E., Rhee, E. J., Lee, W. Y., Oh, K. W., et al. (2015). Preventive effects of bitter melon (Momordica charantia) against insulin resistance and diabetes are associated with the inhibition of NF-kappaB and JNK pathways in high-fat-fed OLETF rats. J. Nutr. Biochem. 26 (3), 234–240. doi: 10.1016/j.jnutbio.2014.10.010
Yang, X., Li, L., Fang, K., Dong, R., Li, J., Zhao, Y., et al. (2017). Wu-Mei-Wan reduces insulin resistance via inhibition of NLRP3 inflammasome activation in HepG2 cells. Evid. Based Complement. Alternat. Med. 2017, 7283241. doi: 10.1155/2017/7283241
Yang, Y., Li, W., Li, Y., Wang, Q., Gao, L., Zhao, J. (2014). Dietary Lycium barbarum polysaccharide induces Nrf2/ARE pathway and ameliorates insulin resistance induced by high-fat via activation of PI3K/AKT signaling. Oxid. Med. Cell Longev. 2014, 145641. doi: 10.1155/2014/145641
Yang, Z., Wu, F., He, Y., Zhang, Q., Zhang, Y., Zhou, G., et al. (2018). A novel PTP1B inhibitor extracted from Ganoderma lucidum ameliorates insulin resistance by regulating IRS1-GLUT4 cascades in the insulin signaling pathway. Food Funct. 9 (1), 397–406. doi: 10.1039/C7FO01489A
Yao, L., Wei, J., Shi, S., Guo, K., Wang, X., Wang, Q., et al. (2017). Modified lingguizhugan decoction incorporated with dietary restriction and exercise ameliorates hyperglycemia, hyperlipidemia and hypertension in a rat model of the metabolic syndrome. BMC Complement. Altern. Med. 17 (1), 132. doi: 10.1186/s12906-017-1557-y
You, H. N., Park, M. H., Hwang, S. Y., Han, J. S. (2018). Nardostachys jatamansi DC extract alleviates insulin resistance and regulates glucose metabolism in C57BL/KsJ-db/db mice through the AMP-activated protein kinase signaling pathway. J. Med. Food. 21 (4), 324–331. doi: 10.1089/jmf.2017.4015
Yu, N., Fang, X., Zhao, D., Mu, Q., Zuo, J., Ma, Y., et al. (2017). Anti-diabetic effects of Jiang Tang Xiao Ke granule via PI3K/Akt signalling pathway in type 2 diabetes KKAy mice. PLoS One 12 (1), e0168980. doi: 10.1371/journal.pone.0168980
Yu, S. H., Chen, S. Y., Li, W. S., Dubey, N. K., Chen, W. H., Chuu, J. J., et al. (2015). Hypoglycemic activity through a novel combination of fruiting body and mycelia of Cordyceps militaris in high-fat diet-induced type 2 diabetes mellitus mice. J. Diabetes Res. 2015, 723190. doi: 10.1155/2015/723190
Yu, X., Xu, L., Zhou, Q., Wu, S., Tian, J., Piao, C., et al. (2018). The efficacy and safety of the Chinese herbal formula, JTTZ, for the treatment of type 2 diabetes with obesity and hyperlipidemia: a multicenter randomized, positive-controlled, open-label clinical trial. Int. J. Endocrinol. 2018, 9519231. doi: 10.1155/2018/9519231
Yu, Y., Zhao, Y., Teng, F., Li, J., Guan, Y., Xu, J., et al. (2018). Berberine improves cognitive deficiency and muscular dysfunction via activation of the AMPK/SIRT1/PGC-1a pathway in skeletal muscle from naturally aging rats. J. Nutr. Health Aging 22 (6), 710–717. doi: 10.1007/s12603-018-1015-7
Yuan, Y., Wang, X., Lu, X., Marunaka, Y., Wang, X. (2014). Effect of Coptidis Rhizoma extracts in a water-based solution on insulin resistance in 3T3-L1 adipocytes. Biomed. Res. 35 (5), 321–327. doi: 10.2220/biomedres.35.321
Zanquetta, M., Seraphim, P., Sumida, D., Cipolla-Neto, J., Machado, U. (2003). Calorie restriction reduces pinealectomy-induced insulin resistance by improving GLUT4 gene expression and its translocation to the plasma membrane. J. Pineal. Res. 35 (3), 141–148. doi: 10.1034/j.1600-079X.2003.00067.x
Zhang, L., Xu, J., Song, H., Yao, Z., Ji, G. (2014). Extracts from Salvia-Nelumbinis naturalis alleviate hepatosteatosis via improving hepatic insulin sensitivity. J. Transl. Med. 12 (1479-5876 (Electronic)), 236–247. doi: 10.1186/s12967-014-0236-8
Zhang, H., Ta, N., Chen, P., Wang, H. (2017). Erchen decoction and Linguizhugan decoction ameliorate hepatic insulin resistance by inhibiting IRS-1Ser307 phosphorylation in vivo and in vitro. Evid. Based Complement. Alternat. Med. 2017, 1589871. doi: 10.1155/2017/1589871
Zhang, P., Lu, J., Jing, Y., Tang, S., Zhu, D., Bi, Y. (2017). Global epidemiology of diabetic foot ulceration: a systematic review and meta-analysis (dagger). Ann. Med. 49 (2), 106–116. doi: 10.1080/07853890.2016.1231932
Zhang, T. (2015). Effect of Qingre Yangyin recipe on endocrine and metabolism of polycystic ovary syndrome patients. Zhongguo Zhong Xi Yi Jie He Za Zhi. 35 (10), 1175–1180.
Zhang, Z., Zhou, S., Jiang, X., Wang, Y. H., Li, F., Wang, Y. G., et al. (2015). The role of the Nrf2/Keap1 pathway in obesity and metabolic syndrome. Rev. Endocr. Metab. Disord. 16 (1), 35–45. doi: 10.1007/s11154-014-9305-9
Zhao, H., Huang, X., Jiao, J., Zhang, H., Liu, J., Qin, W., et al. (2015). Protein phosphatase 4 (PP4) functions as a critical regulator in tumor necrosis factor (TNF)-alpha-induced hepatic insulin resistance. Sci. Rep. 5, 18093. doi: 10.1038/srep18093
Zhao, H., Zhou, D., Chen, Y., Liu, D., Chu, S., Zhang, S. (2017). Beneficial effects of Heqi san on rat model of polycystic ovary syndrome through the PI3K/AKT pathway. Daru. 25 (1), 21. doi: 10.1186/s40199-017-0188-7
Zhao, L., Fu, Z., Wu, J., Aylor, K. W., Barrett, E. J., Cao, W., et al. (2015). Globular adiponectin ameliorates metabolic insulin resistance via AMPK-mediated restoration of microvascular insulin responses. J. Physiol. 593 (17), 4067–4079. doi: 10.1113/JP270371
Zhao, R., Qiu, B., Li, Q., Zhang, T., Zhao, H., Chen, Z., et al. (2014). LBP-4a improves insulin resistance via translocation and activation of GLUT4 in OLETF rats. Food Funct. 5 (4), 811–820. doi: 10.1039/C3FO60602C
Zhao, W., Wang, M., Shao, L., Liao, M., Liu, K., Huang, F., et al. (2014). The total phenolic fraction of Anemarrhena asphodeloides inhibits inflammation and reduces insulin resistance in adipocytes via regulation of AMP-kinase activity. Planta Med. 80 (2–3), 146–152. doi: 10.1055/s-0033-1360197
Zhen, Z., Chang, B., Li, M., Lian, F. M., Chen, L., Dong, L., et al. (2011). Anti-diabetic effects of a Coptis chinensis containing new traditional Chinese medicine formula in type 2 diabetic rats. Am. J. Chin. Med. 39 (1), 53–63. doi: 10.1142/S0192415X11008646
Zheng, Y., Ley, S. H., Hu, F. B. (2018). Global aetiology and epidemiology of type 2 diabetes mellitus and its complications. Nat. Rev. Endocrinol. 14 (2), 88–98. doi: 10.1038/nrendo.2017.151
Zhou, B., Lu, Y., Hajifathalian, K., Bentham, J., Di Cesare, M., Danaei, G., et al. (2016). Worldwide trends in diabetes since 1980: a pooled analysis of 751 population-based studies with 4•4 million participants. Lancet 387 (10027), 1513–1530. doi: 10.1016/S0140-6736(16)00618-8
Zhou, Q., Chang, B., Chen, X. Y., Zhou, S. P., Zhen, Z., Zhang, L. L., et al. (2014). Chinese herbal medicine for obesity: a randomized, double-blinded, multicenter, prospective trial. Am. J. Chin. Med. 42 (6), 1345–1356. doi: 10.1142/S0192415X14500840
Zhu, L., Zhu, X., Sun, G., Meng, X., Wang, M., Cui, H., et al. (2018). Dai-Zong-Fang, a traditional chinese herbal formula, ameliorates insulin resistance in db/db Mice. Front. Physiol. 9, 224. doi: 10.3389/fphys.2018.00224
Zhu, R., Zheng, J., Chen, L., Gu, B., Huang, S. (2016). Astragaloside IV facilitates glucose transport in C2C12 myotubes through the IRS1/AKT pathway and suppresses the palmitate-induced activation of the IKK/IkappaBalpha pathway. Int. J. Mol. Med. 37 (6), 1697–1705. doi: 10.3892/ijmm.2016.2555
Zisman, A., Peroni, O., Abel, E., Michael, M., Mauvais-Jarvis, F., Lowell, B., et al. (2000). Targeted disruption of the glucose transporter 4 selectively in muscle causes insulin resistance and glucose intolerance. Nat. Med. 6 (8), 924–928. doi: 10.1038/78693
Zou, X., Huang, W., Lu, F., Fang, K., Wang, D., Zhao, S., et al. (2017). The effects of Jiao-Tai-Wan on sleep, inflammation and insulin resistance in obesity-resistant rats with chronic partial sleep deprivation. BMC Complement. Altern. Med. 17 (1), 165. doi: 10.1186/s12906-017-1648-9
Zulet, M. A., Navas-Carretero, S., Lara y Sanchez, D., Abete, I., Flanagan, J., Issaly, N., et al. (2014). A Fraxinus excelsior L. Phytomedicine 21 (10), 1162–1169. doi: 10.1016/j.phymed.2014.04.027
Keywords: herbal medicine, insulin resistance, signal transduction, pathway, mechanism
Citation: Li J, Bai L, Wei F, Zhao J, Wang D, Xiao Y, Yan W and Wei J (2019) Therapeutic Mechanisms of Herbal Medicines Against Insulin Resistance: A Review. Front. Pharmacol. 10:661. doi: 10.3389/fphar.2019.00661
Received: 17 December 2018; Accepted: 23 May 2019;
Published: 14 June 2019.
Edited by:
Alejandro Urzua, Universidad de Santiago de Chile, ChileReviewed by:
Susan Semple, University of South Australia, AustraliaYun K. Tam, Sinoveda Canada Inc., Canada
Copyright © 2019 Li, Bai, Wei, Zhao, Wang, Xiao, Yan and Wei. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Junping Wei, d2VpanVucGluZ0AxMjYuY29t
 Jun Li
Jun Li Litao Bai
Litao Bai Fan Wei1
Fan Wei1