- C&PNS and General Pharmacology, Research and Development, Sigma-Tau Industrie Farmaceutiche Riunite S.p.A., Pomezia, Rome, Italy
Adenosine A2A receptors seem to exist in typical (more in striatum) and atypical (more in hippocampus and cortex) subtypes. In the present study, we investigated the affinity of two adenosine A2A receptor antagonists, ST1535 [2 butyl -9-methyl-8-(2H-1,2,3-triazol 2-yl)-9H-purin-6-xylamine] and KW6002 [(E)-1,3-diethyl-8-(3,4-dimethoxystyryl)-7-methyl-3,7-dihydro-1H-purine-2,6,dione] to the “typical” and “atypical” A2A binding sites. Affinity was determined by radioligand competition experiments in membranes from rat striatum and hippocampus. Displacement of the adenosine analog [3H]CGS21680 [2-p-(2-carboxyethyl)phenethyl-amino-5’-N-ethylcarbox-amidoadenosine] was evaluated in the absence or in the presence of either CSC [8-(3-chlorostyryl)-caffeine], an adenosine A2A antagonist that pharmacologically isolates atypical binding sites, or DPCPX (8-cyclopentyl-1,3-dipropylxanthine), an adenosine A1 receptor antagonist that pharmacologically isolates typical binding site. ZM241385 [84-(2-[7-amino-2-(2-furyl) [1,2,4]-triazol[2,3-a][1,3,5]triazin-5-yl amino]ethyl) phenol)] and SCH58261 [(5-amino-7-(β-phenylethyl)-2-(8-furyl)pyrazolo(4,3-e)-1,2,4-triazolo(1,5-c) pyrimidine], two other adenosine A2A receptor antagonists, which were reported to differently bind to atypical and typical A2A receptors, were used as reference compounds. ST1535, KW6002, ZM241385 and SCH58261 displaced [3H]CGS21680 with higher affinity in striatum than in hippocampus. In hippocampus, no typical adenosine A2A binding was detected, and ST1535 was the only compound that occupied atypical A2A adenosine receptors. Present data are explained in terms of heteromeric association among adenosine A2A, A2B and A1 receptors, rather than with the presence of atypical A2A receptor subtype.
Introduction
Adenosine represents an endogenous inhibitory modulator widely distributed in the central nervous system, exerting its physiological actions through activation of four structurally distinct surface receptors (A1, A2A, A2B and A3), all of which represent attractive targets for several human diseases (Fredholm et al., 2001; Sebastião and Ribeiro, 2009). Some A2A antagonists are currently studied for their role in controlling motor function and as potential neuroprotective agents in Parkinson’s disease (Ikeda et al., 2002; Yu et al., 2008; Morelli et al., 2009; Pinna, 2009). A role for adenosine A2A receptors was also suggested in drug addiction (Ferré et al., 2007) and sensitization (Bastia et al., 2005), narcolepsy and pain (Ferré et al., 2007), and respiration in immature animals (Mayer et al., 2005).
Characterization of adenosine receptor subtypes are traditionally made by the means of specific pharmacological tools (Fredholm et al., 2001). Among these, CGS21680 (Wan et al., 1990) is widely used to study the A2A receptor subtype. It has Ki values for adenosine A2A and A1 receptors of about 22 and 3,100 nM, respectively (Hutchison et al., 1989). [3H]CGS21680 was also used to characterize two high-affinity binding sites in cortical, hippocampal and striatal membranes. The first high-affinity binding site predominates in the striatum and has binding characteristics compatible with A2A receptors. The second high-affinity binding site predominates in the cortex and hippocampus, and has binding characteristics intermediary between those of A1 and A2A receptors (Wan et al., 1990; Johansson et al., 1993; Kirk and Richardson, 1995; Cunha et al., 1996). These pharmacologically distinct binding sites were referred as “typical”, i.e., striatal-like, also present with low abundance in the cortex and hippocampus, and “atypical”, predominant in the cortex and hippocampus. The greatest pharmacological differences between the [3H]CGS21680 binding to “typical” and “atypical” binding sites were observed with the selective A2A antagonists KF17837 (Nonaka et al., 1994) and CSC (Jacobson et al., 1993) and the selective A1 antagonist DPCPX (Lohse et al., 1987). KF17837, which is 62-fold A2A/A1 selective, and CSC which is 520-fold A2A/A1 selective, inhibited most of the [3H]CGS21680 binding to striatal membranes, but they were 10-fold less potent in inhibiting most of the [3H]CGS21680 binding in the hippocampus and cortex. On the contrary, in the hippocampus and cortex, but not in the striatum, most of the [3H]CGS21680 binding was displaced by DPCPX at low nanomolar concentrations (Cunha et al., 1996, 1997). This distinct pharmacological affinity profile, even though the high selectivity of [3H]CGS21680 for A2A receptors, has raised reasonable doubts as whether these atypical binding sites might represent A2A receptors.
ST1535 and KW6002 are two known A2A receptor antagonists, proposed for the treatment of Parkinson’s disease (Kase et al., 2003; Pinna, 2009), which differentiate for their in vitro selectivity profile on adenosine receptors. ST1535 has a preferential antagonistic activity on A2A receptors, but it is also an antagonist on A1 receptors (Stasi et al., 2006). On the other hand, KW6002 also binds to adenosine A2B receptors (Stasi et al., 2006).
Present work was aimed at determining ST1535 and KW6002 affinities toward the adenosine A2A receptor subtypes in two rat brain regions, striatum and hippocampus.
ST1535 and KW6002-mediated displacement of [3H]CGS21680 binding was evaluated in the absence or in the presence of either CSC, which pharmacologically isolates atypical CGS21680 binding sites, or DPCPX, which pharmacologically isolates typical CGS21680 binding site (Cunha et al., 1996). ZM241385, an adenosine A2A receptor antagonist with equal potency to display [3H]CGS21680 binding in striatal and limbic regions (Cunha et al., 1997), and SCH21680, an adenosine A2A receptor antagonist that clearly discriminates between the two different binding sites in different brain regions (Lindström et al., 1996), were used as reference compounds.
Materials and Methods
Sources of Compounds
ST1535, KW6002 and SCH58261 were supplied by sigma-tau Chemical Department. CSC [8-(3-chlorostyryl) caffeine] and DPCPX [8-cyclopentyl-1,3-dipropylxantine] were purchased from Sigma-Aldrich (Sigma-Aldrich, St. Louis, MO, USA), while ZM241385 was obtained from Tocris (Tocris, Ellisville, MI, USA).
Animal Husbandry
Six male Wistar rats, 11- to 12-week old (Harlan, S. Pietro al Natisone, Udine, Italy) were used. Rats were housed in makrolon cages (Tecniplast Gazzada 42.5 cm × 26.6 cm × 18 cm height) (three rats/cage) with stainless steel covered feed racks and sterilized, dust-free bedding cobs. Animals were housed under a light-dark cycle, with constant temperature and humidity. Parameters of the animal rooms were as follows: temperature 22 ± 2°C, relative humidity 55 ± 10%, about 15–20 filtered air exchanges/hour and a 12-h circadian cycle of artificial light (7 a.m.–7 p.m.). A diet (in pellets) coded GLP 4RF 21, produced by Mucedola S.r.l. of Settimo Milanese (a licensee of Charles River – Italia S.p.A.), was used. All parts of the study concerning animal care were performed under the control of sigma-tau veterinarians, in compliance with Italian regulatory system (D.L.vo 116 of 27 January 1992, art. 6). Animals were sacrificed by beheading and striatum and hippocampus were collected and stored at −80°C.
Membrane Preparation and Competition Binding
The collected tissues were homogenized in about 25 volumes of incubation buffer and centrifuged at 40,000 g for 10 min at 4°C. The pellet was resuspended in the same fresh buffer, homogenized, centrifuged once again and the final pellet stored at −80°C until use. The methods used for binding studies were as those described by Cunha et al. (1996). Prior to the competition binding assay, the pellet was resuspended in appropriate buffer at the wanted protein concentration and membrane suspension incubated with 5 U/ml adenosine deaminase (ADA, Sigma-Aldrich) for 30 min at 37°C to remove endogenous adenosine. The protein concentration of membrane suspension was determined using the Bradford method (Pierce, Rockford, IL, USA) with bovine albumin as standard. Competition binding experiments with [3H]CGS21680 were performed by incubating membranes (50–100 μg of protein/sample) with a single concentrations of [3H]CGS21680 (30 nM) for 2 h at room temperature in a solution containing 50 mM Tris-HCl, pH 7.4, 10 mM MgCl2 and 5 U/ml ADA and various concentrations (ranging from 10−5 to 10−12 M) of test compounds. Non-specific binding was determined in the presence of an excess of cold 2-chloroadenosine (Sigma-Aldrich). The binding reactions were stopped by vacuum filtration through GF/B filters, followed by washing of the filters with ice-cold washing buffer (same composition as the binding buffer) and filter-bound radioactivity measured by scintillation spectrometry. Competition studies were performed with the various compounds alone or in the presence of an excess of either CSC or DPCPX (both from Sigma-Aldrich). The compounds were dissolved in 100% DMSO (Sigma-Aldrich) at a concentration of 10 mM and stored at −20°C. An intermediate 100 μM working solution was prepared in water and subsequent scalar working solutions in incubation buffer.
Data Evaluation
Concentration-response binding curves obtained for every test compound (n = 3–4) were analyzed together using non-linear regression with GraphPad PRISM commercial software and expressed as IC50 (concentration of compounds that inhibits 50% of specific radioligand binding) and 95% confidence intervals. Inhibitory binding constant (Ki) values were calculated from IC50 values according to the Cheng and Prusoff equation Ki = IC50/(1 + [C]/Kd), where [C] is the concentration of the radioligand and Kd its dissociation constant. Kd of 62 nM and 12 nM were used for hippocampus and striatum, respectively, as reported by Cunha et al. (1996).
Results
Binding experiments were performed using in both brain tissues a radioligand concentration of 30 nM, which is intermediate between its Kd values in the striatum and hippocampus, respectively (Cunha et al., 1996). In addition, to exclude the role of different endogenous adenosine concentrations in the tissues, experiments were performed after a 30 min treatment with 5 U/ml ADA.
In such conditions, [3H]CGS21680 specific binding in the striatum represented about 80% of the total binding. All compounds bound to adenosine A2A receptors in the striatum both in the absence and in the presence of either CSC or DPCPX, which were supposed to isolate “atypical” and “typical” receptor subtypes, respectively (Cunha et al., 1996) (Table 1, Figures 1, 3 and 4).
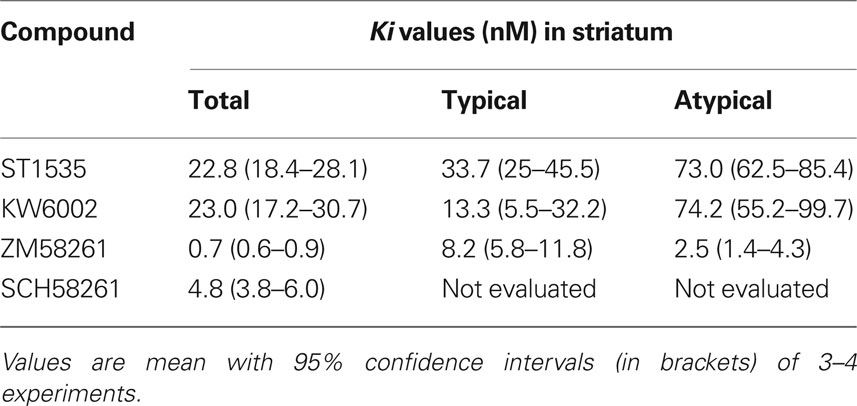
Table 1. Displacement of [3H]CGS 21680 binding from total, typical and atypical adenosine A2A receptors in rat striatal membranes.
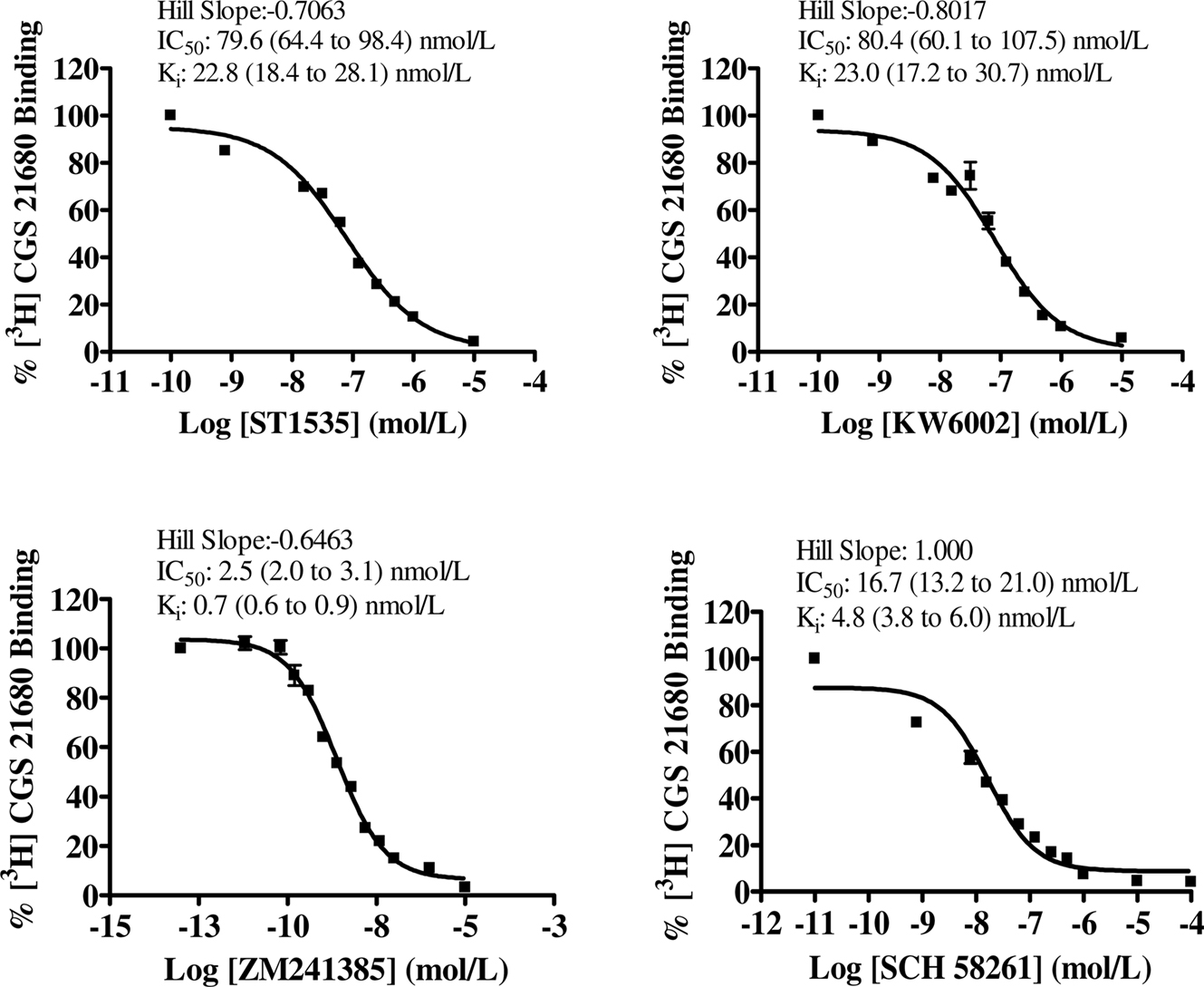
Figure 1. Dose–response curves of compound-mediated displacement of [3H]CGS21680 binding to rat striatum. The ordinates represent the specific binding of 30 nmol/L [3H]CGS21680 in the presence of different concentrations of compounds. Non-specific binding was determined with 100 μM 2-chloroadenosine. Curves were obtained from 3 to 4 independent experiments performed in duplicate. Values in brackets represent 95% confidence intervals.
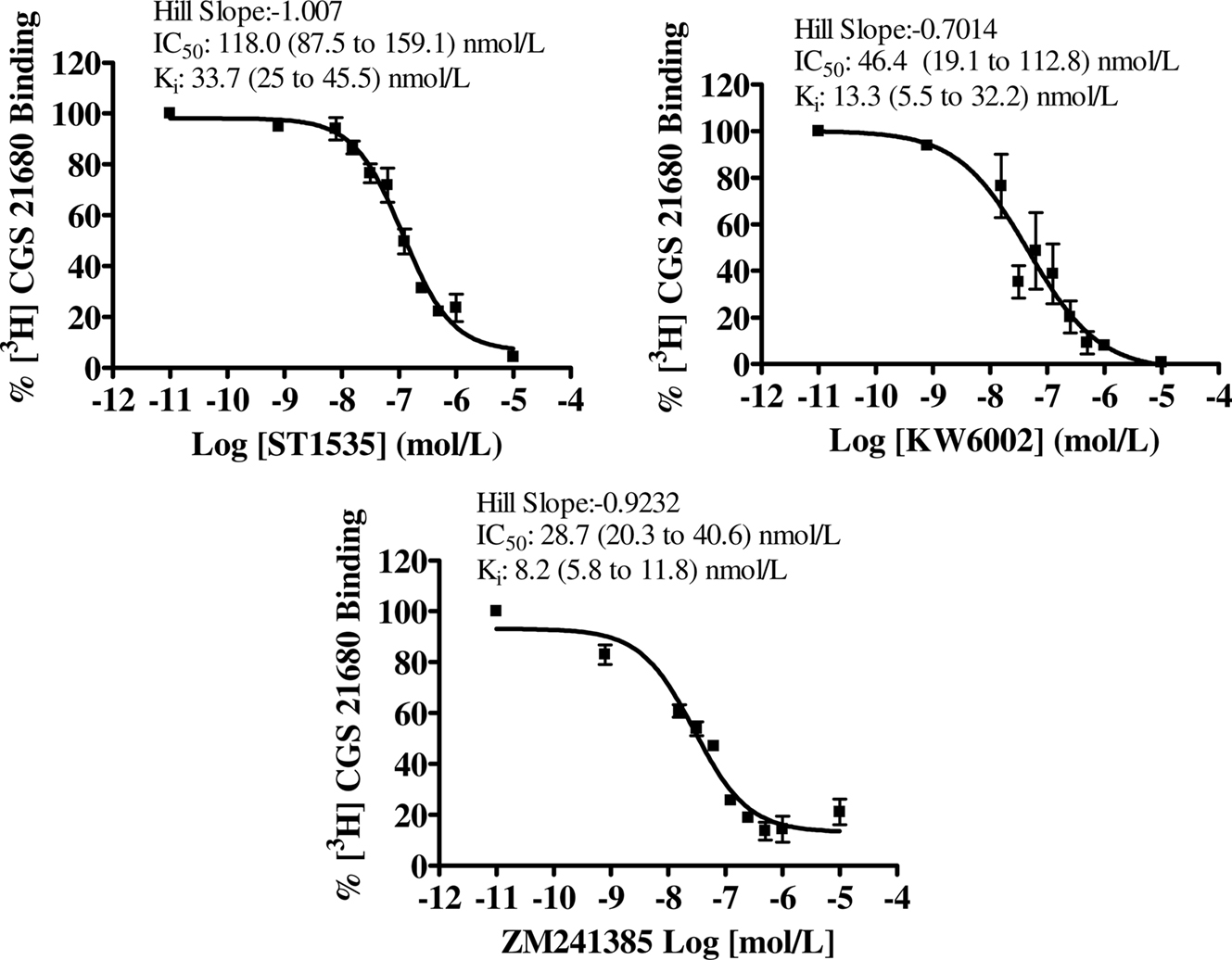
Figure 2. Dose–response curves of compound-mediated displacement of [3H]CGS21680 binding to rat striatum in the presence of 100 nmol/L DPCPX, to isolate typical adenosine A2A receptors. The ordinates represent the specific binding of 30 nmol/L [3H]CGS21680 in the presence of different concentrations of compounds and a fixed saturating concentration of DPCPX. Non-specific binding was determined with 100 μM 2-chloroadenosine. Curves were obtained from three independent experiments performed in duplicate. Values in brackets represent 95% confidence intervals.
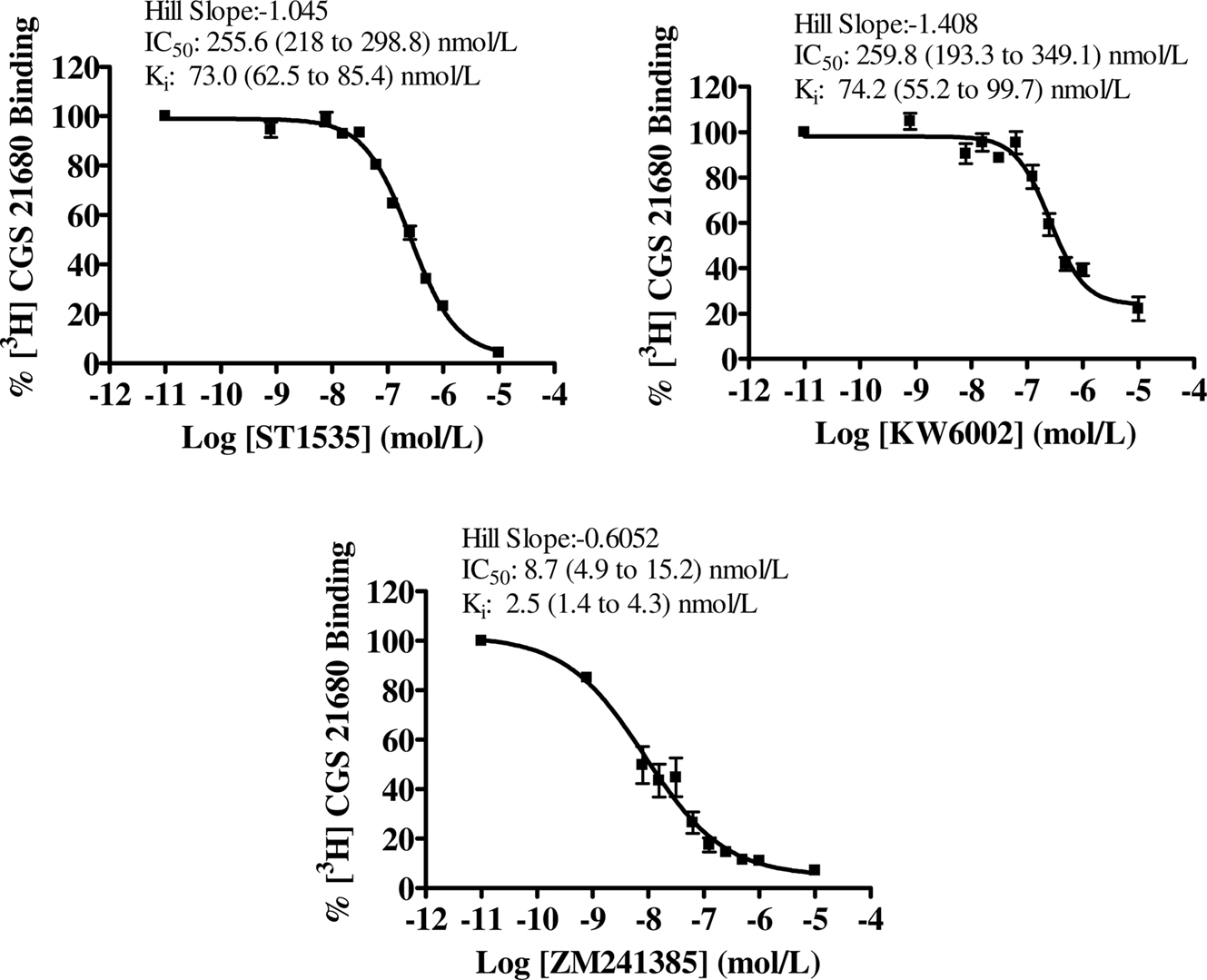
Figure 3. Dose–response curve of compound-mediated displacement of [3H]CGS21680 binding to rat striatum in the presence of 200 nmol/L CSC, to isolate atypical adenosine A2A receptors. The ordinates represent the specific binding of 30 nmol/L [3H]CGS21680 in the presence of different concentrations of compounds and a fixed saturating concentration of CSC. Non-specific binding was determined with 100 μM 2-chloroadenosine. Curves were obtained from three independent experiments performed in duplicate. Values in brackets represent 95% confidence intervals.
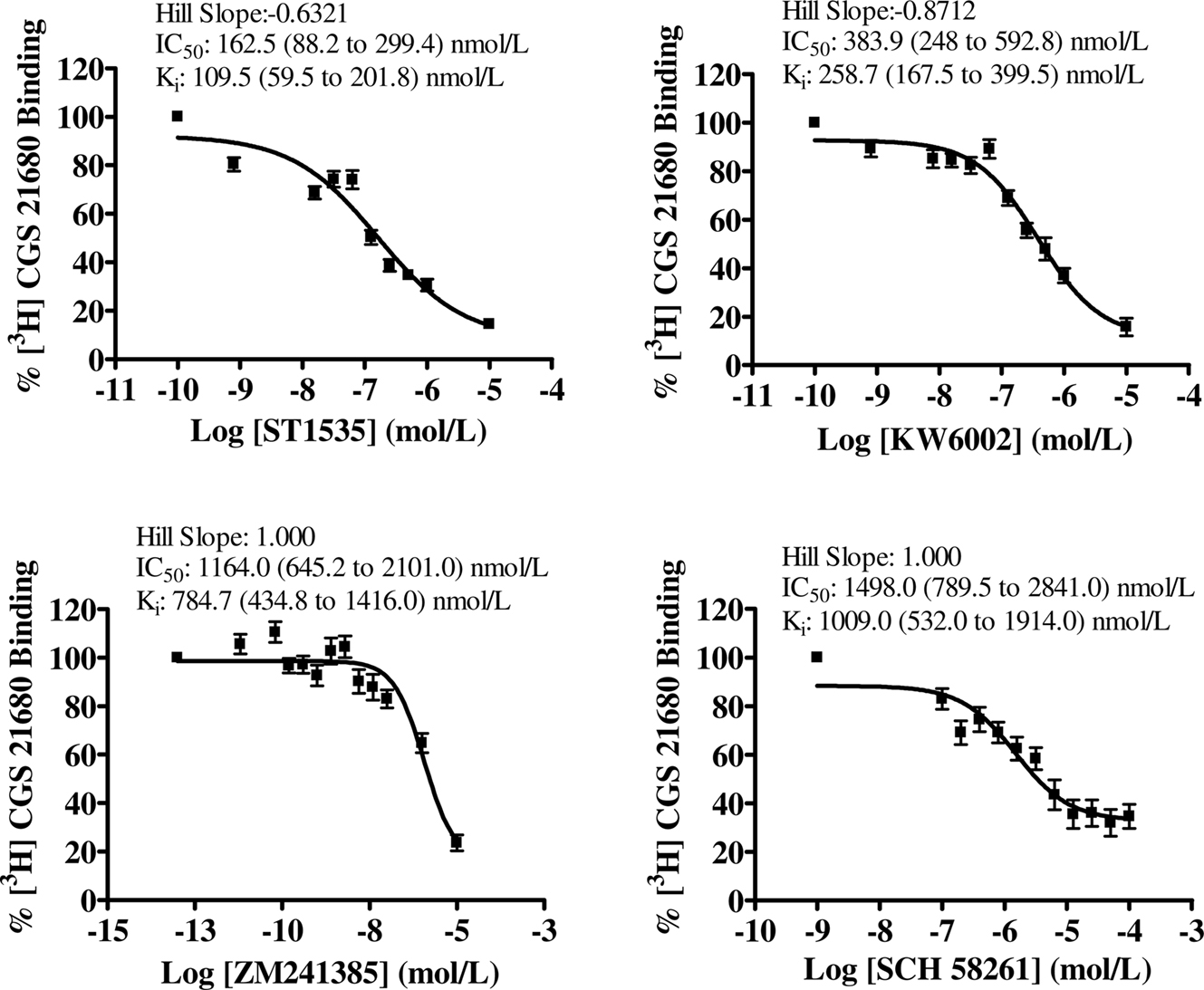
Figure 4. Dose–response curves of compound-mediated displacement of [3H]CGS21680 binding to rat hippocampus. The ordinates represent the specific binding of 30 nmol/L [3H]CGS21680 in the presence of different concentrations of compounds. Non-specific binding was determined with 100 μM 2-chloroadenosine. Curves were obtained from four independent experiments performed in duplicate. Values in brackets represent 95% confidence intervals.
In the striatum, the affinity values of both ST1535 and KW6002 for “total” and “typical” A2A receptors were similar and decreased from two to five fold for “atypical” binding sites (Table 1, Figures 1, 3 and 4). ZM241385 and SCH58261 displaced [3H]CGS21680 binding to “total” A2A receptors with affinities comparable to those previously published (Table 1, Figure 1). However, isolation of ”typical” binding site (Table 1, Figure 3) determined a 10-fold decrease in the affinity of ZM241385, a result in contrast with the literature (Cunha et al., 1996).
The lowest Hill coefficients, around 0.6, were found for ZM241385 in binding to “total” and “atypical” striatal adenosine A2A receptors (Figures 1 and 4). Hill coefficients for ST1535 were around the unit in binding “typical” and “atypical” receptors and 0.7 for “total” receptors (Figures 1, 3 and 4). Hill coefficients for KW6002 in binding “atypical” receptors were 1.4, and 0.7–0.8 for “total” and “typical” receptors, respectively (Figures 1, 3 and 4).
In hippocampus, [3H]CGS21680 specific binding represented about 50% of the total binding. All compounds bound to “total” adenosine A2A receptors (Table 2, Figure 2) with affinity values lower than in the striatum. In the hippocampus, the use of DPCPX completely abolished [3H]CGS21680 specific binding, possibly because of saturation of all binding sites by DPCPX. As a consequence, data obtained in hippocampus with the use of DPCPX were not evaluable (Table 2). In the presence of CSC, ST1535 was the only compound which displaced some affinity, while no binding of KW6002 and ZM241385 was observable. For ZM241385 and SCH58261, calculation of IC50 in the hippocampus was possible only by imposing a slope equal to 1, while ST1535 and KW6002 had Hill coefficients around 0.6–0.8 (Figures 2 and 5).
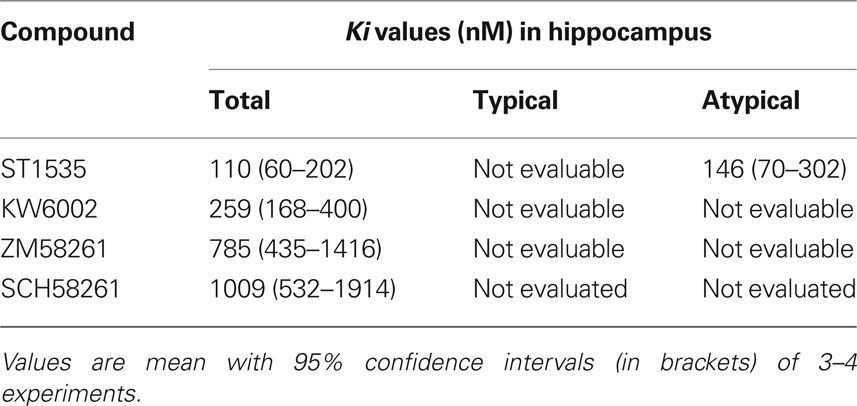
Table 2. Displacement of [3H]CGS 21680 binding from total, typical and atypical adenosine A2A receptors in rat hippocampal membranes.
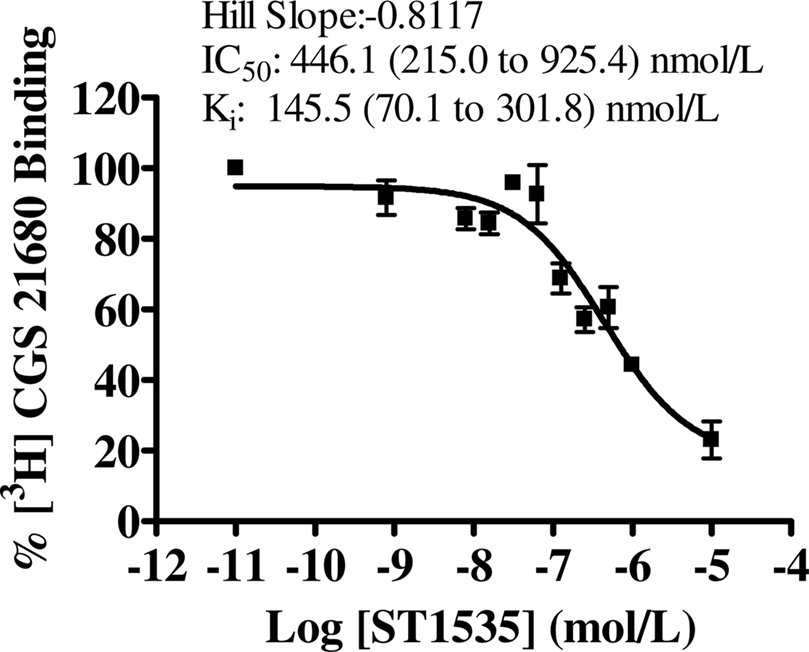
Figure 5. Dose–response curves of compound-mediated displacement of [3H]CGS21680 binding to rat hippocampus in the presence of 200 nmol/L CSC, to isolate atypical adenosine A2A receptors. The ordinates represent the specific binding of 30 nmol/L [3H]CGS21680 in the presence of different concentrations of compounds and a fixed saturating concentration of CSC. Non-specific binding determined with 100 μM 2-chloroadenosine. The curve was obtained from three independent experiments performed in duplicate. Values in brackets represent 95% confidence intervals.
Discussion
All compounds bound better to A2A in the striatum than in hippocampus, confirming some published data in literature (Wan et al., 1990; Johansson et al., 1993; Kirk and Richardson, 1995; Cunha et al., 1996). As repeatedly shown, using different methods, adenosine A2A receptors are highly enriched in caudate putamen, nucleus accumbens and tuberculum olfactorium, and expression levels elsewhere are lower (Jarvis and Williams, 1989; Dixon et al., 1996). However, it seems conceivable that differences in affinity values might not be due to different number of adenosine A2A receptors in the two tissues, since the ratio between the affinity values in hippocampus and striatum was not a constant but variable among the different compounds we used. In fact, if one could have hypothesized that the small difference in affinity values for ST1535 (about 5-fold) and KW6002 (about 10-fold) between striatum and hippocampus could have been attributed to a different number of receptors, the same hypothesis cannot explain the greater difference in affinity values for ZM241385 (about 1100-fold) and SCH58261 (about 200-fold). The affinity values of the various compounds for total adenosine A2A striatal receptors are in agreement with published data (Lindströom et al., 1996; Cunha et al., 1997; Ongini et al., 1999; Stasi et al., 2006; Yang et al., 2007; Pinna, 2009). Our results, however, are in contrast with ZM241385 binding data by Cunha et al. (1996), who did not show evident difference between striatal and hippocampal adenosine A2A receptors. On the contrary, we found that ZM241385 displayed over 1,000-fold affinity for A2A receptors in striatum compared to hippocampus. In addition, in striatum, ZM241385 also bound with higher affinity to “atypical” than to “typical” receptors, and did not recognize “atypical” receptors in the hippocampus, in contrast with Cunha et al. (1996). We can just hypothesize that these contrasting results with Cunha are due to different receptor preparation or binding methods or, alternatively, with the use of different animal strain and age. Similarly to ZM241385, KW6002 occupied “atypical” adenosine A2A receptors in striatum but not in hippocampus. Despite the evidence that [3H]CGS21680 displays different affinity values for adenosine A1 and A2A receptors (Hutchison et al., 1989), at the concentrations we used it may bind to sites associated with adenosine A1 receptors (O’Kane and Stone, 1998; Lopes et al., 2002; Halldner et al., 2004). Therefore, binding affinity of ST1535, KW6002, ZM241385 and SCH58261 in the hippocampus may reflect their interaction on A1 receptors. In fact, the ratio between A1 and A2A receptors in transfected cells is about 10-fold for ST1535 (Minetti et al., 2005), 30- to 70-fold for KW6002 (Mihara et al., 2007; Yang et al., 2007), 400- to 6000-fold for ZM241385 (Linden et al., 1999; Fredholm et al., 2001; Minetti et al., 2005), and 100- to 500-fold for SCH58261 (Ongini and Fredholm, 1996; Zocchi et al., 1996; Yang et al., 2007; Pinna, 2009). These ratios are in the range of those we observed between the Kivalues in striatum and hippocampus. Dual affinity of ST1535 for rat adenosine A2A and A1 receptors may be the reason why it was the only adenosine A2A antagonist which displaced [3H]CGS21680 from “atypical” sites in hippocampus.
Moreover, the fact that adenosine A2A and A1 receptors may dimerize in striatum (Ciruela et al., 2006) may explain our data in this region, as a consequence of a stereodynamic equilibrium among the two receptors, the radioligand and the compound.
Another player in this equilibrium is the adenosine A2B receptor, with which ST1535, KW6002 and ZM214385 interact at concentrations only moderately higher then those for adenosine A2A receptors: of about 3-fold for ST1535 (Stasi et al., 2006), 1- to 18-fold for KW6006 (Stasi et al., 2006; Yang et al., 2007), and 22-fold for ZM214385 (Linden et al., 1999). On this line, there is evidence of interaction between A2A, A2B and A1 receptors in striatum (Okada et al., 1996). Even if CGS21860 apparently has very low affinity for adenosine A2B receptors (Linden et al., 1999), the fact that ST1535, KW6002 and ZM214385 interact with A1 and A2B receptors, and thus influencing A2A receptor binding, may explain the binding to “atypical” sites by CGS21860 in striatum. Receptor cross-talk, as demonstrated for A1 and A2A receptors (Lopes et al., 1999), and physical interactions among receptors able to create entities with their own pharmacological characteristics, as shown for association between A1 and P2Y receptors (Yoshioka et al., 2001), should also be considered.
In conclusion, we suggest that the apparent “atypicality” of [3H]CGS21680 binding may depend on unselectivity of the compounds used.
The implications of such results on humans are difficult to predict since it has been reported that CGS2168 in humans is less potent and selective than in rats and that differences exist in binding affinities and potency of some agonists between human and rat (Fredholm et al., 2001).
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationship that could be construed as a potential conflict of interest.
References
Bastia, E., Xu, Y.-H., Scibelli, A. C., Day, Y.-J., Linden, J., Chen, J.-F., and Schwarzschild, M. A. (2005). A crucial role for forebrain adenosine A2A receptors in amphetamine sensitization. Neuropsychopharmacology 30, 891–900.
Ciruela, F., Casado, V., Rodriguez, R. J., Lujan, R., Burgueno, J., Canals, M., Borycz, J., Reboòa, N., Goldberg, S. R., Mallol, J., Cortes, A., Canela, E. I., Lopez-Gimemez, J. F., Milligan, G., Lluis, C., Cunha, R. A., Ferré, S., and Franco, R. (2006). Presynaptic control of striatal glutamatergic neurotransmission by adenosine A1-A2A receptor heteromers. J. Neurosci. 26, 2080–2087.
Cunha, R. A., Constantino, M. D., and Ribeiro, J. A. (1997). ZM241385 is an antagonist of the facilitatory responses produced by the A2A adenosine receptor agonists CGS21680 and HENECA in the rat hippocampus. Br. J. Pharmacol. 122, 1279–1284.
Cunha, R. A., Johansson, B., Constantino, M. D., Sebastião, A. M., and Fredholm, B. B. (1996). Evidence for high-affinity binding sites for the adenosine A2A receptor agonist [3H] CGS 21680 in the rat hippocampus and cerebral cortex that are different from striatal A2A receptors. Naunyn-Schmiedebergs Arch. Pharmacol. 353, 261–271.
Dixon, A. K., Gubitz, A. K., Sirinathsinghji, D. J., Richardson, P. J., and Freeman, T. C. (1996). Tissue distribution of adenosine receptor mRNAs in the rat. Br. J. Pharmacol. 118, 1461–1468.
Ferré, S., Diamond, I., Goldberg, S. R., Yao, L., Hourani, S. M. O., Huang, Z. L., Urade, Y., and Kitchen, I. (2007). Adenosine A2A receptors in ventral striatum, hypothyalamus and nociceptive circuitry. Impications for drug addiction, sleep and pain. Prog. Neurobiol. 83, 332–347.
Fredholm, B. B., IJzerman, A. P., Jacobson, K. A., Klotz, K. N., and Linden, J. (2001). International Union of Pharmacology. XXV. Nomenclature and classification of adenosine receptors. Pharmacol. Rev. 53, 527–552.
Halldner, L, Lopes, L. V., Daré, E., Lindstrom, K., Johansson, B., Ledent, C., Cunha, R., and Fredholm, B. B. (2004). Binding of adenosine receptor ligands to brain of adenosine receptor knock-out mice: evidence that CGS21680 binds to A1 receptors in hippocampus. Naunyn-Schmiedebergs Arch. Pharmacol. 370, 270–278.
Hutchison, A. J., Webb, R. L., Oei, H. H., Ghai, G. R., Zimmerman, M. B., and Williams, M. (1989). CGS21680C, an A2 selective adenosine receptor agonist with preferential hypothensive activity. J. Pharmacol. Exp. Ther. 251, 47–55.
Ikeda, K., Kurokawa, M., Aoyama, S., and Kuwana, Y. (2002). Neuroprotection by adenosine A2A receptor blockade in experimental models of Parkinson’s disease. J. Neurochem. 80, 262–270.
Jacobson, K. A., Nikodijevicć, O., Padgett, W. L., Gallo-Rodriguez, C., Maillard, M., and Daly, J. W. (1993). 8-(3-Chlorostyryl)caffeine (CSC) is a selective A2-adenosine antagonist in vitro and in vivo. FEBS Lett. 323, 141–144.
Jarvis, M. F., and Williams, M. (1989). Direct autoradiographic localization of adenosine A2 receptors in the rat brain using the A2 selective agonist, [3H]CGS 21680. Eur. J. Pharmacol. 168, 243–246.
Johansson, B., Georgiev, V., Parkinson, F. E., and Fredholm, B. B. (1993). The binding of the adenosine A2 receptor selective agonist [3H]CGS 21680 to rat cortex differs from its binding to rat striatum. Eur. J. Pharmacol. 247, 103–110.
Kase, H., Aoyama, S., Ichimura, M., Ikeda, K., Ishii, A., Kanda, T., Koga, K., Koike, N., Kurokawa, M., Kuwana, Y., Mori, A., Nakamura, J., Nonaka, H., Ochi, M., Saki, M., Shimada, J., Shindou, T., Shiozaki, S., Suzuki, F., Takeda, M., Yanagawa, K., Richardson, P. J., Jenner, P., Bedard, P., Borrelli, E., Hauser, R. A., and Chase, T. N. (2003). KW-6002 US-001 Study Group. Progress in pursuit of therapeutic A2A antagonists: the adenosine A2A receptor selective antagonist KW6002: research and development toward a novel nondopaminergic therapy for Parkinson’s disease. Neurology 61, S97–S100.
Kirk, I. P., and Richardson, P. J. (1995). Further characterization of [3H]-CGS 21680 binding sites in the rat striatum and cortex. Br. J. Pharmacol. 114, 537–543.
Linden, L., Thai, T., Figler, H., Jin, X., and Robeva, A. S. (1999). Characterization of human A2B adenosine receptors: radioligand binding, western blotting, and coupling to Gq in human embryonic kidney 293 cells and HMC-1 mast cells. Mol. Pharmacol. 56, 705–713.
Lindström, K., Ongini, E., and Fredholm, B. B. (1996). The selective adenosine A2A receptor antagonist SCH 58261 discriminates between two different binding sites for [3H]-CGS 21680 in the rat brain. Naunyn-Schmiedebergs Arch. Pharmacol. 354, 539–541.
Lohse, M. J., Klotz, K. N., Lindenborn-Fotinos, J., Reddington, M., Schwabe, U., and Olsson, R. A. (1987). 8-Cyclopentyl-1,3-dipropylxanthine (DPCPX)-a selective high affinity antagonist radioligand for A1 adenosine receptors. Naunyn-Schmiedebergs Arch. Pharmacol. 336, 204–210.
Lopes, L. V., Cunha, R. A., Kull, B., Fredholm, B. B., and Ribeiro, J. A. (2002). Adenosine A(2A) receptor facilitation of hippocampal synaptic transmission is dependent on tonic A(1)receptor inhibition. Neuroscience 112, 319–329.
Lopes, L. V., Cunha, R. A., and Ribeiro, J. A. (1999). Cross talk between A(1) and A(2A) adenosine receptors in the hippocampus and cortex of young adult and old rats. J. Neurophysiol. 82, 3196–3203.
Mayer, C. A., Haxhiu, M. A., Martin, R. J., and Wilson, C. G. (2005). Adenosine A2A receptors mediate GABAergic inhibition of respiration in immature rats. J. Appl. Physiol. 100, 91–97.
Mihara, T., Mihara, K., Yarimizu, J., Mitani, Y., Matsuda, R., Yamamoto, H., Aoki, S., Akahane, A., Iwashita, A., and Matsioka, N. (2007). Pharmacological characterization of a novel, potent adenosine A1 and A2A receptor dual antagonists, 5-5-amino-3-(4-fluorphenyl)pyrazin-2-yl]1-1-isopropylpyridine-2(1H)-one (ASP5854), in models of Parkinson’s disease and cognition. J. Pharmacol. Exp. Ther. 323, 708–719.
Minetti, P., Tinti, M. O., Carminati, P., Castorina, M., Di Cesare, M. A., Di Serio, S., Gallo, G., Ghirardi, O., Giorgi, F., Giorgi, L., Piersanti, G., Bartoccini, F., and Tarzia, G. (2005) 2-n-Butyl-9-methyl-8-[1,2,3]triazol-2-yl-9H-purin-6-ylamine and analogues as A2A adenosine receptor antagonists. Design, synthesis, and pharmacological characterization. J. Med. Chem. 48, 6887–6896.
Morelli, M., Carta, A. R., and Jenner, P. (2009). Adenosine A2A receptors and Parkinson’s disease. Handb. Exp. Pharmacol. 193, 589–615.
Nonaka, H., Ichimura, M., Takeda, M., Nonaka, Y., Shimada, J,. Suzuki, F., Yamaguchi, K., and Kase, H. (1994). KF17837 ((E)-8-(3,4-dimethoxystyryl)-1,3-dipropyl-7-methylxanthine), a potent and selective adenosine A2 receptor antagonist. Eur. J. Pharmacol. 267, 335–341.
Okada, M., Mizuno, K., and Kaneko, S. (1996). Adenosine A1 and A2 receptors modulate extracellular dopamine levels in rat striatum. Neurosci. Lett. 212, 53–56.
O’Kane, E. M., and Stone, T. W. (1998). Interaction between adenosine A1 and A2 receptor-mediated responses in the rat hippocampus in vitro. Eur. J. Pharmacol. 362, 17–25.
Ongini, E., Dionisotti, S., Gessi, S., Irenius, E., and Fredholm, B. B. (1999). Comparison of CGS15943, ZM 241385 and SCH 58261 as antagonists at human adenosine receptors. Naunyn-Schmiedebergs Arch. Pharmacol. 359, 7–10.
Ongini, E., and Fredholm, B. B. (1996). Pharmacology of adenosine A2A receptors. Trends Pharmacol. Sci. 17, 364–372.
Pinna, A. (2009). Novel investigational adenosine A2A receptor antagonists for Parkinson’s disease. Expert Opin. Investig. Drugs 18, 1619–1631.
Sebastião, A. M., and Ribeiro, J. A. (2009). Adenosine receptors and the central nervous system. Handb. Exp. Pharmacol. 193, 471–534.
Stasi, M. A., Borsini, F., Varani, K., Vincenzi, F., Di Cesare, M. A., Minetti, P., Ghirardi, O., and Carminati, P. (2006). ST 1535: a preferential A2A adenosine receptor antagonist. Int. J. Neuropsychopharmacol. 9, 575–584.
Wan, W., Sutherland, G. R., and Geiger, J. D. (1990). Binding of the adenosine A2 receptor ligand [3H]CGS 21680 to human and rat brain: evidence for multiple affinity sites. J. Neurochem. 55, 1763–1771.
Yang, M., Soohoo, D., Spelaiman, S., Kalla, R., Zablochi, J., Chu, N., Leung, K., Yao, L., Diamond, I., Belardinelli, L., and Shrycock, K. C. (2007). Characterization of the potency, selectivity, and pharmacokinetic profile for six adenosine A2A receptor antagonists. Naunyn-Schmiedebergs Arch. Pharmacol. 375, 133–144.
Yoshioka, K., Saitoh, O., and Nakata, H. (2001). Heteromeric association creates a P2Y-like adenosine receptor. Proc. Natl. Acad. Sci. U.S.A. 98, 7617–7622.
Yu, L., Shen, H. Y., Coelho, J. E., Araújo, I. M., Huang, Q. Y., Day, Y. J., Rebola, N., Canas, P. M., Rapp, E. K., Ferrara, J., Taylor, D., Müller, C. E., Linden, J., Cunha, R. A., and Chen, J. F. (2008). AdenosineA2A receptor antagonists exert motor and neuroprotective effects by distinct cellular mechanisms. Ann. Neurol. 63, 338–346.
Keywords: adenosine, receptors, atypical, typical, ST1535, KW6002, ZM241385, SCH58261
Citation: Riccioni T, Leonardi F and Borsini F (2010) Adenosine A2A receptor binding profile of two antagonists, ST1535 and KW6002: consideration on the presence of atypical adenosine A2A binding sites. Front. Psychiatry 1:22. doi: 10.3389/fpsyt.2010.00022
Received: 23 March 2010;
Paper pending published: 08 June 2010;
Accepted: 03 July 2010;
Published online: 06 August 2010
Edited by:
Martine Hascoet, University of Nantes, FranceReviewed by:
Micaela Morelli, University of Cagliari, ItalyMichel Bourin, University of Nantes, France
Copyright: © 2010 Riccioni, Leonardi and Borsini. This is an open-access article subject to an exclusive license agreement between the authors and the Frontiers Research Foundation, which permits unrestricted use, distribution, and reproduction in any medium, provided the original authors and source are credited.
*Correspondence: Teresa Riccioni, C&PNS and General Pharmacology, Research and Development, Sigma-Tau Industrie Farmaceutiche Riunite S.p.A., Via Pontina, Km.30,400, I-00040 Pomezia, Rome, Italy. e-mail:dGVyZXNhLnJpY2Npb25pQHNpZ21hLXRhdS5pdA==


