- 1Institute of Neuroscience, Newcastle upon Tyne, UK
- 2Northumberland, Tyne and Wear NHS Foundation Trust, Newcastle upon Tyne, UK
Greater intra-individual variability (IIV) in reaction time (RT) on a sustained attention task has been reported in patients with bipolar disorder (BD) compared with healthy controls. However, it is unclear whether IIV is task specific, or whether it represents general cross-task impairment in BD. This study aimed to investigate whether IIV occurs in sustained attention tasks with different parameters. Twenty-two patients with BD (currently euthymic) and 17 controls completed two sustained attention tasks on different occasions: a low target frequency (~20%) Vigil continuous performance test (CPT) and a high target frequency (~70%) CPT version A-X (CPT-AX). Variability measures (individual standard deviation and coefficient of variation) were calculated per participant, and ex-Gaussian modeling was also applied. This was supplemented by Vincentile analysis to characterize RT distributions. Results indicated that participants (patients and controls) were generally slower and more variable when completing the Vigil CPT compared with CPT-AX. Significant group differences were also observed in the Vigil CPT, with euthymic BD patients being more variable than controls. This result suggests that IIV in BD demonstrates some degree of task specificity. Further research should incorporate analysis of additional RT distributional models (drift diffusion and fast Fourier transform) to fully characterize the pattern of IIV in BD, as well as its relationship to cognitive processes.
Introduction
Bipolar disorder (BD) is a severe and heterogeneous mood disorder (1). The disorder is associated with marked neurocognitive problems during mood episodes (2, 3), and in full symptomatic recovery. Processing speed, executive functioning, and sustained attention appear to be particularly affected [e.g., Ref. (4–7)]. Indeed, neurocognitive problems are considered a core feature of the disorder (8) and are related to lower quality of life (9). As such, there is interest in developing our understanding of potential cognitive endophenotypes (10), which may have benefits for diagnosis and treatment.
Sustained attention – the ability to maintain concentration over a period of time (11) – is considered a potential cognitive endophenotype of BD (12). To date, studies of sustained attention in BD have tended to rely on global measures of behavioral performance (e.g., overall hits, commission and omission errors). However, there is increasing interest in going beyond these basic measures and examining additional indices of performance such intra-individual variability (IIV).
Attentional IIV refers to extent to which individual reaction time (RT) responses vary during a task (13). It is typically assessed using individual standard deviation (iSD) or the coefficient of variation (CoV), represented by iSD/mean RT (14). IIV may be an informative behavioral marker in BD. Evidence from healthy aging suggests that variability is biologically meaningful; it is heritable (15, 16), is sensitive to the effects of age [i.e., increased IIV with age; (17, 18)], and is a strong behavioral correlate of reduced white matter integrity (19, 20). Furthermore, increased IIV is associated with poorer cognitive functioning (21) and can predict mortality longitudinally (22). IIV may also have clinical utility; for instance, increased IIV is a proposed cognitive marker for prodromal Alzheimer’s disease at the mild cognitive impairment stage (23).
There are statistical and theoretical caveats in simple summary measures of IIV. Indices, such as iSD and CoV, represent pooled data and thus assume that the RT distribution is Gaussian (24). However, empirical RT distributions are typically skewed (25) due to a subset of excessively slow RTs among responses in a normal range (26). A more representative analysis of IIV can be achieved by fitting RT data to an ex-Gaussian distribution (27). In the ex-Gaussian model, the distribution of the RTs is represented as the product (convolution) of two randomly distributed variables; one that is Gaussian (normally distributed), and another that is exponential. The latter distribution accounts for the positive skew generally observed in RT distributions (28). The ex-Gaussian distribution is described by three summary parameters; mu (μ) and sigma (σ) (the mean and SD of the Gaussian component), and tau (τ), which references the exponential component.
Conceptually, the distribution of faster responses is indexed by μ and σ. The infrequent, longer RTs which lengthen the tail of the distribution are indexed by τ and can be examined separately from mean RT (24, 29–31).
Analysis of RT distributions using the ex-Gaussian model has been applied successfully in diverse research fields. In general, application of the model has enabled researchers to specify where in a RT distribution groups or individuals differ. Such differences are obscured by examining mean RT in isolation, as excessively slow responding, albeit occasional, can skew this summary statistic (24). Within healthy aging, increases in σ and τ have been reported in older participants compared with a younger sample (29). Indeed, increases in τ may be sensitive to neurodegenerative processes (30). In attention deficit hyperactivity disorder (ADHD), faster overall responding (μ) as well increases in excessively slow responding (τ) has been described (31, 32). Increases in τ observed in ADHD have also been proposed as a candidate endophenotype for the disorder, in part, due to its proposed heritability (33).
Recently, the ex-Gaussian model has been applied in BD (34). The model was used to characterize the RT distribution from a task of sustained attention [Vigil CPT; (35)]. Greater positive skew (τ) in the RT distributions of both euthymic and depressed patients with BD was found – a pattern of responding consistent with the notion of fluctuating attentional task engagement in BD [see also Ref. (36)]. However, the need for replication was also noted in order to determine the influence of factors such as task parameters.
Sustained attention is typically assessed using continuous performance tests [CPTs; (37)]. In the original task, participants responded to an infrequent target “X” and in a later variant, to the “X” only after an “A” cue. Rosvold et al. (37) noted that the ability to correctly classify participants (into those with brain damage and those who were neurologically intact) improved with this increase in task difficulty. Consequently, CPT-AX is considered to be a general marker of brain health (11, 37).
Many CPTs have subsequently been developed, each with numerous procedural variations (38). Common procedural variants include increased/decreased target frequency (39); interstimulus interval (ISI), per block, or adjusted for individual accuracy (39, 40); overall task length, e.g., ranging from 3 min (41) to 30 min (42); and stimulus type, e.g., letters (43, 44) or numbers (45). Finally, the “quality” of the stimuli can be altered through degradation of stimulus integrity over the course of an experiment (46).
It is perhaps not surprising that manipulation in CPT task parameters can affect behavioral response characteristics, some of which are used as markers of the ability to maintain attention. For instance, higher target frequencies are associated with faster mean RTs, as well as increases in errors (47, 48). It is thought that this high target frequency manipulation induces a frequent response tendency (40). Underlying this effect may be an altered demand on response inhibitory and motor control mechanisms (thereby impulsivity), rather than sustained attention per se (49). In contrast, low target frequency manipulations result in a slower overall RT (39). Generally, this agrees with the historical definition of vigilance, whereby participants maintain a “vigil” or watch over a long period of time and respond to an infrequent event (50). It has been argued that a low target frequency presentation is therefore a more appropriate index of sustained attention (49). Varying the ISI in a task can also impact response characteristics. A short ISI (<500 ms) is associated with faster mean RTs, as well as increases in omission errors (39). Conversely, a longer ISI is associated with slower RTs, and with increased IIV as measured via the ex-Gaussian distribution (40).
As these CPT parameters impact behavioral outcome measures, which may have clinical utility they should be considered when investigating IIV in BD. The aim of this study was therefore to determine whether a similar pattern of IIV would be obtained using a sustained attention task with different parameters in patients with euthymic BD and in healthy controls. The CPT, version AX [CPT-AX; (36)], and the Vigil CPT (35) were utilized. Across both tasks, there are common parameters. Both tasks have a high event rate Parasuraman (50) (Vigil = 64 events/min; CPT-AX = 70 events/min) and both have a similar working memory load (both “1-back” cued target sequences). However, the tasks differ on target frequency. Target sequences are presented infrequently during the Vigil CPT (~20%) compared with CPT-AX (~70%). We predicted that both tasks would result in increased IIV, and with ex-Gaussian modeling, an increase in the τ parameter in patients with BD.
Materials and Methods
All participants included in the current analyses were from studies conducted within the Institute of Neuroscience at Newcastle University (6, 36). Data included in this study were collected between 2000 and 2003 (Thompson et al.) and 2001 and 2003 (Robinson et al.). Euthymia was confirmed for patients in both studies (see below). These participants were a subset of those reported in Gallagher et al. (34) for whom CPT-AX data were also available.
Participants
Twenty-two adult euthymic outpatients between the ages of 30 and 57 years (M = 43.13, SD = 7.78) with a SCID (51) confirmed diagnosis of BD were included in the analysis. Clinical interviews were conducted by psychiatrists trained in SCID administration. Recruitment was via services within the Northumberland, Tyne and Wear NHS Foundation Trust in the North East of England. Euthymia – defined as a score of ≤7 on the on the 21-item Hamilton Depression Rating Scale [HAMD; (52)] and the Young Mania Rating Scale [YMRS; (53)] – was prospectively verified over 1 month from the initial assessment.
During the verification month, patients completed the Beck Depression Inventory [BDI; (54)] and the Altman Mania Rating Scale [AMRS; (55)] weekly.
All patients were stable and taking psychotropic medication: 16 were prescribed lithium, 10 were prescribed antidepressants, and 5 were prescribed antipsychotics. Exclusion criteria for patients was as follows: (i) presence of another current Axis I diagnosis (except anxiety), (ii) neurological or medical condition, (iii) history of substance or alcohol abuse/dependence over the past 6 months, (iv) prescribed corticosteroids or antihyperintensive medication, and (v) electroconvulsive therapy (ECT) within the past year.
Twenty healthy controls, between the ages of 30 and 53 years (M = 43.55, SD = 6.67) were recruited through local advertisements. Controls did not have a psychiatric history (SCID confirmed) or have a first degree relative with a psychiatric disorder. Groups were well matched on sex, age, and premorbid IQ (National Adult Reading Test, NART) (56) and did not significantly differ in these characteristics (p > 0.05; Table 1). All study protocols were approved by the appropriate National Health Service Local Research Ethics Committee. Written informed consent was obtained prior to study participation.
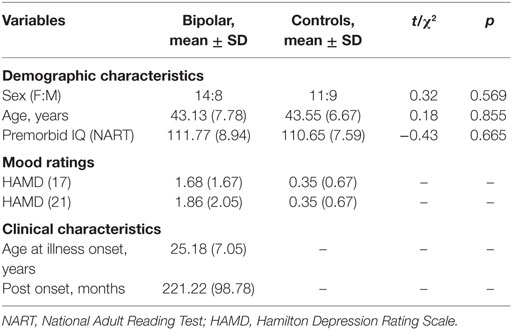
Table 1. Clinical and demographic characteristics of the euthymic BD patients (n = 22) and controls (n = 20).
Neurocognitive Testing: Sustained Attention Tasks
Continuous Performance Test, A–X
Single, randomized letters are presented sequentially on a computer screen and participants respond to a target sequence (36). Letters are presented in white, on a black background. In this task, participants respond to target “X,” only when it was presented after an “A” (“AX” target trial). This task uses a stimulus presentation time of 50 ms, and an ISI of 800 ms. In addition, participants respond to an increased number of target sequences.
Over 200 trials of paired stimuli (split into 4 blocks of 50 paired trials), 140 target pairs (35 per block) are presented in 6 min, with no breaks between blocks or practice trials. In both tasks, participants were asked to respond as quickly and accurately.
Vigil Continuous Performance Test
In this task, single, randomized letters are presented sequentially on a computer screen for 85 ms, followed by a 900-ms ISI (35). Letters are presented in white, on a black background. Participants respond when they view target “K,” only when cued by an earlier “A” stimulus (“AK” target sequence). Targets occur infrequently in this CPT. Over the course of 480 stimuli, 100 target sequences are presented in 8 min. These targets were pseudo-randomized, so that 25 target sequences are presented in 4 blocks (no breaks given between blocks).
Data Analysis
First, RTs below 100 ms were removed following established absolute cut-off principles (27, 57), which removed two responses. As the response window for the CPT-AX task could not be extended, the response window for Vigil was restricted to 850 ms.
The restriction was applied to ensure that differences in IIV were not simply due to task-related differences in the time participants had to respond before the next stimulus. A total of 4054 responses were analyzed, and the restriction of the response window removed eight responses (0.19% of total trials). Only correct trials (“hits”) occurring within the response windows were analyzed.
RT IIV Analysis
Analysis of RT IIV was applied to correct trials (hits). Common measures of IIV were calculated, which included iSD and CoV (the latter is expressed as a percentage). These measures were applied to all participants (patients n = 22, controls n = 20). The DISTRIB toolbox (58) in MATLAB® R2013b (59) (The MathsWorks, Inc., Natick, MA, USA) was used to fit the ex-Gaussian probability density function to the distribution of correct RTs.
Three parameters of the ex-Gaussian distribution are estimated per individual using this function; μ, σ, and τ. The algorithm failed to fit the distribution to three control participants, who were then removed from the analysis (patients n = 22, controls n = 17).
In addition, Vincentile plots were calculated as an overall graphical representation of the data. These plots can be calculated without prior assumptions regarding the theoretical shape of the distribution (60). To calculate Vincentiles, response times per individual are ranked from fastest to slowest into eight bins (each bin presents 12.5% of RTs), and then averaged.
Statistical Analysis
Data were analyzed using the Statistical Package for Social Sciences (SPSS), version 21 (61). Matching characteristics for study groups (age, sex, education) were analyzed using independent t-tests (continuous variables) and chi-square tests (categorical). Demographic and clinical characteristics were also analyzed between groups in the same manner. Behavioral outcome measures (IIV) were analyzed using repeated measures ANOVA, with task (CPT-AX vs. Vigil) as the within-subjects variable and group (patient vs. controls) as the between-subjects variable.
Results
Task
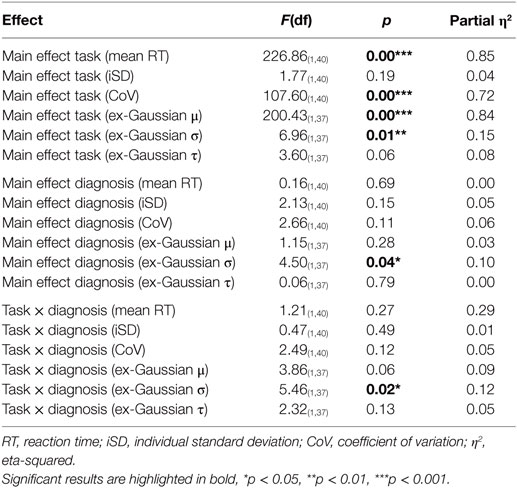
Average response time was slower for the Vigil CPT (mean RT for patients and controls together: M = 394.24, SE = 15.83; ex-Gaussian μ: M = 327.19, SE = 11.11) compared with the CPT-AX (mean RT: M = 299.90, SE = 13.42; ex-Gaussian μ: M = 218.51, SE = 7.32). Examination of the Vincentile plots (Figure 1) indicated that participants performed the Vigil task more slowly across the whole distribution (from V1 to V8) compared with CPT-AX (p < 0.05). Variability as measured by CoV was higher in the CPT-AX task (M = 26.33, SE = 1.38) compared with Vigil (M = 19.50, SE = 0.98). However, the ex-Gaussian σ parameter was higher in the Vigil task (M = 38.76, SE = 3.40) compared with the CPT-AX (M = 31.23, SE = 2.16). Task had no significant main effect on the remaining IIV parameters (iSD, and ex-Gaussian τ).
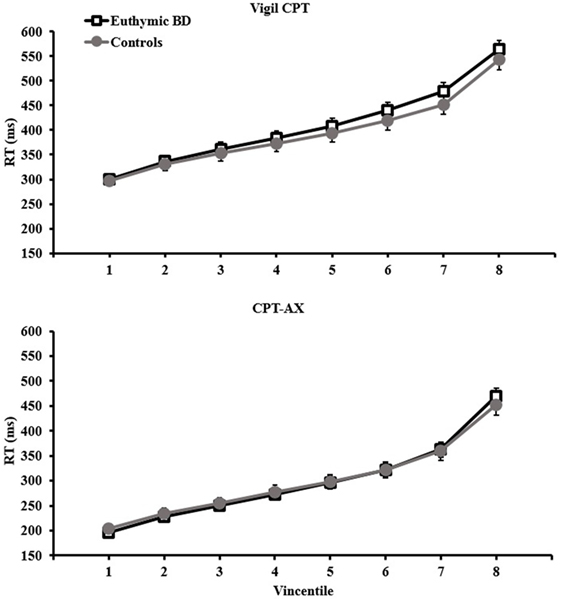
Figure 1. Vincentile plots (1–8) for euthymic BD (n = 22) and controls (n = 20) per task. Mean RTs are taken from the slowest 12.5% (1) to the fastest (8). Plots for Vigil CPT are represented in the top panel and CPT-AX in the bottom. Error bars represent SEM.
Group
The between-subjects effect for group was significant for ex-Gaussian σ (p < 0.001). Euthymic BD patients were more variable overall (ex-Gaussian σ: M = 40.24, SE = 3.26) compared with controls (ex-Gaussian σ: M = 29.75, SE = 3.71). No further between-subjects effects reached significance (see Table 2).
Task × Group Interaction
A significant interaction was observed between task and diagnosis for ex-Gaussian σ (p < 0.05; Table 2). The interaction was driven by differences in variability between the groups in the Vigil task [t(37) = −2.51, p < 0.05], but not the CPT-AX [t(37) = −0.882, p > 0.05]. Here, bipolar euthymic patients were more variable (M = 47.34, SE = 4.74) than controls (M = 30.18, SE = 4.73) (Figure 2). Results were also assessed separately per task, between patients and controls.
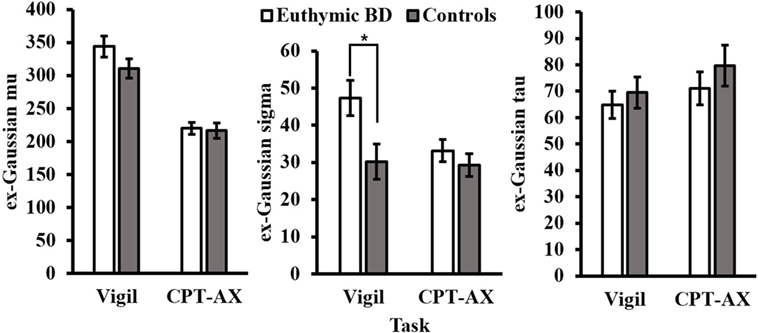
Figure 2. Ex-Gaussian parameters for euthymic BD and controls per task (CPT-AX and Vigil). Euthymic BD patients were more variable than controls, as indicated by ex-Gaussian σ (*p < 0.05), but only in the Vigil CPT. Error bar represent SEM.
Exploratory Analyses
For the Vigil and CPT-AX tasks, exploratory two-tailed Spearman’s correlations were performed between IIV variables (iSD, CoV, and ex-Gaussian parameters), clinical characteristics (HAMD21, months post onset, and age of onset), and demographic (age) for patients. Demographic information (age) was also investigated in controls. No significant correlations were observed between IIV indices and any clinical or demographic characteristic in either task (all p > 0.05; see Table S1 for Vigil CPT and Table S2 for CPT-AX in Supplementary Material) for patients.
Age did not significantly correlate (all p > 0.05) with any IIV index in either task for controls (see Table S3 in Supplementary Material).
Discussion
This study investigated RT IIV during two differing sustained attention tasks (CPT-AX and Vigil CPT) in euthymic BD patients and healthy controls. The sample overall (patients and controls) completed the low target frequency Vigil CPT more slowly than the high target frequency CPT-AX, as indicated by greater values of mean RT and ex-Gaussian μ. In addition, variability was higher for sample overall for ex-Gaussian σ, but only in the Vigil CPT. Variability as measured by iSD and ex-Gaussian τ between each task was similar for the sample overall. Between groups, however, euthymic BD patients exhibited greater values of σ compared with controls, but only during the Vigil CPT. All other indices of variability (iSD, CoV, and ex-Gaussian τ) were similar between patients and controls in the CPT-AX and Vigil.
The variability in responding demonstrated by euthymic BD patients suggests irregular attentional engagement. However, this inconsistency is only observed under the unique task conditions of the Vigil CPT. In Parasuraman’s (50) definition, both tasks included in this study had high event rates (more than 60 stimuli presented per minute). Such tasks tend to be taxing, resulting in a vigilance decrement (i.e., reduction in accuracy over time). The high event rate and low target frequency parameters of the Vigil CPT may have resulted in unique task conditions requiring more effortful processing. This level of processing would be required to maintain an adequate level of attentional task engagement in response to increased task demands (62, 63). It is possible that the euthymic BD patients in this study were more sensitive to the task conditions induced during the Vigil CPT, reflected by the greater variability in responding.
Working memory load should also be considered. It should be noted that CPTs generally involve a modest contribution of working memory, namely, goal maintenance (6, 36). However, as both tasks required participants to maintain a target sequence of equal length (“AX” for the CPT-AX and “AK” for the Vigil CPT), the contribution of working memory was controlled for. While target percentage was the primary interest of this study, other differences between the tasks, such as stimulus presentation and ISI may also have influenced results.
The results of this study may fit within an accelerated cognitive aging interpretation of disease progression in BD (64, 65), as increased IIV is associated with age-related processes (17, 18). The accelerated aging model highlights that many of the changes observed in BD such as altered brain structure, and cognitive impairments among others, mirror those observed in healthy aging (66).
Methodological considerations of the study should also be taken into account. In the data set analyzed in this study, we did not observe increased ex-Gaussian τ in the Vigil CPT in patients. We included a subsample of euthymic bipolar patients (n = 22) and controls (n = 17) from the larger sample (n = 86 per group) in Gallagher et al. (34), who completed both the Vigil CPT and the CPT-AX. It is possible that the results of this study may have been a cohort effect from using this subsample from the larger study, given that the observed increase in σ was only observed in BD depression, not in euthymic patients in the larger study. It is also possible that the results of this study were due to difference in the demographic and/or clinical characteristics between the samples. However, this is unlikely, given similar reported characteristics between the two studies (age, IQ, depression severity), as well as lack of relationship between these characteristics and any RT IIV parameters. As such, it is likely that the lack of comparison between the results of this study, and those of Gallagher et al. (34) may be due to smaller sample size, as opposed to study characteristics. The small sample size of this study can be considered a limitation.
It should be noted that a longer window for correct responses was included in the Gallagher et al. ex-Gaussian modeling of Vigil CPT responses. In addition to the full response window being used (985 ms), “late” responses of up to 1970 ms were included under certain circumstances.
This window was restricted in this study to 850 ms, which may have resulted in a shift within the fitted ex-Gaussian distributional parameters. However, in the present subsample, this only resulted in a very small number of responses that were excluded and thus is unlikely to have resulted in a large change in the group results. In Table S4 in Supplementary Material, we investigated the possibility that use of a restricted response window removed data contributing to positive skew (τ). Extension of the response window did not alter the results from analysis of the Vigil CPT, between patients and controls – ex-Gaussian σ remained the sole significant between-group difference.
Based on the results of this study, future research should clarify the role that other task parameters have upon RT distributions. CPT procedural variations, such as event rate, have also been shown to impact on mean RTs (39, 50, 67). Identifying the independent contributions of each task parameter (e.g., such as speed of stimulus presentation, working memory load, etc.) would be worthwhile, as CPTs generally manipulate more than one parameter. For instance, the CPT variants included in this study both used high event rates, yet varied on target frequency. Future work should clarify the conditions necessary for task-dependent variability in RT distributions.
A further point of discussion concerns the interpretation of the ex-Gaussian model. While authors have suggested that certain components of the model represent specific cognitive functions [e.g., increases in τ and attentional lapses; (28)], caution in utilizing this interpretation is warranted. As the model lacks solid theoretical underpinning, application of the ex-Gaussian model should be considered descriptive (68). Consequently, use of this model in isolation cannot account for the cognitive factors that drive behavioral performance (27, 69).
With these caveats considered, future research could combine theoretical RT models with ex-Gaussian distribution fitting. One candidate is the drift diffusion model (DDM) (70, 71). In brief, the model assumes that responses are made following accumulation of information that reaches a threshold (correct or incorrect decision boundary). After this threshold is reached, the participant responds. The model consists of three parameters; (i) drift rate (v), which is the rate of information acquired from a stimulus to make a response decision; (ii) boundary separation (a), which contains information about response biases (i.e., speed/accuracy trade-offs), and (iii) non-decision time (Ter), which indexes other processes that are different from decision-making (e.g., motor preparation). Combining ex-Gaussian analysis with the DDM may serve to strengthen the link between altered response profiles observed in psychiatric populations (such as BD), and underlying cognitive factors (e.g., decision processes), which may account for such differences.
Further research could also combine analysis of the ex-Gaussian distribution with examining the temporal components of RT IIV. With use of the Fast Fourier Transform (FFT), periodic patterns of responding that are specific to certain time scales (temporal frequency bands) can be detected; manifesting itself as peaks in the (spectral) power at the specific frequency band (72). Interestingly, analysis of FFT may indicate underlying abnormalities, such as inefficient processing in specific neural and/or resting-state networks [e.g., as used in ADHD; (31, 72)]. As altered network activity has been reported frequently in BD (73), analysis of the oscillatory pattern of response times warrants further investigation. This may shed light on the underlying pathophysiology associated with the disorder, which may provide novel targets for psychopharmacological interventions.
To conclude, while all participants were slower and more variable when completing the Vigil CPT compared with the CPT-AX, euthymic BD patients exhibited greater variability (σ) than controls in the Vigil CPT. In addition, the results also suggest IIV has a degree of task specificity. Future research should consider alternative RT models and analyses of temporal instability of RT concurrently with the ex-Gaussian.
Author Contributions
Data collection: LR, JT, SW, IF, and PG. Data analysis: RM and AF. Manuscript writing: RM, AF, LR, JT, SW, IF, and PG.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding
AF is supported through the Research Capability Funding from the Newcastle Tyne and Wear NHS Foundation Trust. The original research was funded by the Stanley Foundation.
Supplementary Material
The Supplementary Material for this article can be found online at http://journal.frontiersin.org/article/10.3389/fpsyt.2016.00106
References
1. Lim CS, Baldessarini RJ, Vieta E, Yucel M, Bora E, Sim K. Longitudinal neuroimaging and neuropsychological changes in bipolar disorder patients: review of the evidence. Neurosci Biobehav Rev (2013) 37(3):418–35. doi:10.1016/j.neubiorev.2013.01.003
2. Martínez-Arán A, Vieta E, Reinares M, Colom F, Torrent C, Sánchez-Moreno J, et al. Cognitive function across manic or hypomanic, depressed, and euthymic states in bipolar disorder. Am J Psychiatry (2004) 161(2):262–70. doi:10.1176/appi.ajp.161.2.262
3. Gallagher P, Gray JM, Watson S, Young AH, Ferrier IN. Neurocognitive functioning in bipolar depression: a component structure analysis. Psychol Med (2013) 44(5):961–74. doi:10.1017/S0033291713001487
4. Arts B, Jabben N, Krabbendam L, van Os J. Meta-analyses of cognitive functioning in euthymic bipolar patients and their first-degree relatives. Psychol Med (2008) 38(06):771–85. doi:10.1017/S0033291707001675
5. Torres IJ, Boudreau VG, Yatham LN. Neuropsychological functioning in euthymic bipolar disorder: a meta-analysis. Acta Psychiatr Scand (2007) 116:17–26. doi:10.1111/j.1600-0447.2007.01055.x
6. Thompson JM, Gallagher P, Hughes JH, Watson S, Gray JM, Ferrier IN, et al. Neurocognitive impairment in euthymic patients with bipolar affective disorder. Br J Psychiatry (2005) 186(1):32–40. doi:10.1192/bjp.186.1.32
7. Robinson LJ, Thompson JM, Gallagher P, Goswami U, Young AH, Ferrier IN, et al. A meta-analysis of cognitive deficits in euthymic patients with bipolar disorder. J Affect Disord (2006) 93(1–3):105–15. doi:10.1016/j.jad.2006.02.016
8. Green MF. Cognitive impairment and functional outcome in schizophrenia and bipolar disorder. J Clin Psychiatry (2006) 67(10):e12. doi:10.4088/JCP.1006e12
9. Brissos S, Dias VV, Carita AI, Martinez-Arán A. Quality of life in bipolar type I disorder and schizophrenia in remission: clinical and neurocognitive correlates. Psychiatry Res (2008) 160(1):55–62. doi:10.1016/j.psychres.2007.04.010
10. Gottesman II, Gould TD. The endophenotype concept in psychiatry: etymology and strategic intentions. Am J Psychiatry (2003) 160(4):636–45. doi:10.1176/appi.ajp.160.4.636
11. Riccio C, Reynolds CR, Lowe P, Moore JJ. The continuous performance test: a window on the neural substrates for attention? Arch Clin Neuropsychol (2002) 17(3):235–72. doi:10.1016/S0887-6177(01)00111-1
12. Ancín I, Santos JL, Teijeira C, Sánchez-Morla EM, Bescós MJ, Argudo I, et al. Sustained attention as a potential endophenotype for bipolar disorder. Acta Psychiatr Scand (2010) 122(3):235–45. doi:10.1111/j.1600-0447.2009.01532.x
13. Castellanos FX, Sonuga-Barke EJS, Scheres A, Di Martino A, Hyde C, Walters JR. Varieties of attention-deficit/hyperactivity disorder-related intra-individual variability. Biol Psychiatry (2005) 57(11):1416–23. doi:10.1016/j.biopsych.2004.12.005
14. Dykiert D, Der G, Starr JM, Deary IJ. Age differences in intra-individual variability in simple and choice reaction time: systematic review and meta-analysis. PLoS One (2012) 7(10):45759. doi:10.1371/journal.pone.0045759
15. Vernon AP. The heritability of measures of speed of information-processing. Pers Individ Dif (1989) 10(5):573–6. doi:10.1016/0191-8869(89)90040-8
16. Finkel D, Pedersen NL. Genetic and environmental contributions to the associations between intraindividual variability in reaction time and cognitive function. Aging Neuropsychol Cogn (2014) 21(6):746–64. doi:10.1080/13825585.2013.874523
17. Deary JI, Der G. Reaction time, age, and cognitive ability: longitudinal findings from age 16 to 63 years in representative population samples. Aging Neuropsychol Cogn (2005) 12(2):187–215. doi:10.1080/13825580590969235
18. Bielak A, Hultsch D, Strauss E, MacDonald S, Hunter M. Intraindividual variability in reaction time predicts cognitive outcomes 5 years later. Neuropsychology (2010) 24(6):731–41. doi:10.1037/a0019802
19. Fjell A, Lars M, Westlye T, Amlien IK, Walhovd KB. Reduced white matter integrity is related to cognitive instability. J Neurosci (2011) 31(49):18060–72. doi:10.1523/JNEUROSCI.4735-11.2011
20. Moy G, Millet P, Haller S, Baudois S, de Bilbao F, Weber K, et al. Magnetic resonance imaging determinants of intraindividual variability in the elderly: combined analysis of grey and white matter. Neuroscience (2011) 186:88–93. doi:10.1016/j.neuroscience.2011.04.028
21. Hultsch DF, MacDonald SWS, Dixon RA. Variability in reaction time performance of younger and older adults. J Gerontol B Psychol Sci Soc Sci (2002) 57(2):101–15. doi:10.1093/geronb/57.2.P101
22. MacDonald SWS, Nybegr L, Sandblom J, Fischer H, Bäckman L. Increased response-time variability is associated with reduced inferior parietal activation during episodic recognition in aging. J Cogn Neurosci (2008) 20(5):779–86. doi:10.1162/jocn.2008.20502
23. Kälin AM, Pflüge M, Gietl AF, Riese F, Jäncke L, Nitsch RM, et al. Intraindividual variability across cognitive tasks as a potential marker for prodromal Alzheimer’s disease. Front Aging Neurosci (2014) 6:147. doi:10.3389/fnagi.2014.00147
24. Buzy WM, Medoff DR, Schweitzer JB. Intra-individual variability among children with ADHD on a working memory task: an ex-Gaussian approach. Child Neuropsychol (2009) 15(5):441–59. doi:10.1080/09297040802646991
26. Castellanos FX, Sonuga-Barke EJS, Milham MP, Tannock R. Characterizing cognition in ADHD: beyond executive dysfunction. Trends Cogn Sci (2006) 10(3):117–23. doi:10.1016/j.tics.2006.01.011
27. Luce R. Response Times: Their Role in Inferring Elementary Mental Organization. (Vol. 3). New York: Oxford University Press (1986).
28. Leth-Steensen C, Elbaz ZK, Douglas VI. Mean response times, variability, and skew in the responding of ADHD children: a response time distributional approach. Acta Psychol (2000) 104(2):167–90. doi:10.1016/S0001-6918(00)00019-6
29. McAuley T, Yap M, Christ SE, White DA. Revisiting inhibitory control across the life span: insights from the ex-Gaussian distribution. Dev Neuropsychol (2006) 29(3):447–58. doi:10.1207/s15326942dn2903_4
30. Tse C-S, Balota D, Yap M, Duchek J, McCabe D. Effects of Healthy aging and early stage dementia of the Alzheimer’s type on components of response time distributions in three attention tasks. Neuropsychology (2010) 24(3):300–15. doi:10.1037/a0018274
31. Vaurio RG, Simmonds DJ, Mostofsky SH. Increased intra-individual reaction time variability in attention-deficit/hyperactivity disorder across response inhibition tasks with different cognitive demands. Neuropsychologia (2009) 47(12):2389–96. doi:10.1016/j.neuropsychologia.2009.01.022
32. Hervey AS, Epstein JN, Curry JF, Tonev S, Arnold LE, Conners CK, et al. Reaction time distribution analysis of neuropsychological performance in an ADHD sample. Child Neuropsychol (2006) 12(2):125–40. doi:10.1080/09297040500499081
33. Lin H, Hwang-Gu S, Gau SS-F. Intra-individual reaction time variability based on ex-Gaussian distribution as a potential endophenotype for attention-deficit/hyperactivity disorder. Acta Psychiatr Scand (2015) 132(1):39–50. doi:10.1111/acps.12393
34. Gallagher P, Finkelmeyer A, Goshawk M, Macritchie KA, Lloyd AJ, Thompson JM, et al. Neurocognitive intra-individual variability in mood disorders: effects on attentional response time distributions. Psychol Med (2015) 45(14):2985–97. doi:10.1017/S0033291715000926
35. Cegalis J, Bowlin J. VIGIL: Software for the Assessment of Attention. Nashua, NH: Forthought (1991).
36. Robinson LJ, Thompson JM, Gallagher P, Gray JM, Young AH, Ferrier IN. Performance monitoring and executive control of attention in euthymic bipolar disorder: employing the CPT-AX paradigm. Psychiatry Res (2013) 210(2):457–64. doi:10.1016/j.psychres.2013.06.039
37. Rosvold EH, Mirksy A, Sarason I, Bransome E, Beck L. A continuous performance test of brain damage. J Consult Psychol (1956) 20(5):343–50. doi:10.1037/h0043220
38. Greenberg LM, Waldmant ID. Developmental normative data on the test of variables of attention (T.O.V.A.TM). J Child Psychol Psychiatry (1993) 34(6):1019–30. doi:10.1111/j.1469-7610.1993.tb01105.x
39. Ballard JC. Assessing attention: comparison of response-inhibition and traditional continuous performance tests. J Clin Exp Neuropsychol (2001) 23(3):331–50. doi:10.1076/jcen.23.3.331.1188
40. Conners CK, Epstein JN, Angold A, Klaric J. Continuous performance test performance in a normative epidemiological sample. J Abnorm Child Psychol (2003) 31(5):555–62. doi:10.1023/A:1025457300409
41. Bremer DA. MINI-CPT: a continuous performance test program for the Tandy PC-8 pocket computer. Behav Res Methods Instrum Comput (1989) 21(1):11–4. doi:10.3758/BF03203866
42. Mansour CS, Haier RJ, Buchsbaum MS. Gender comparisons of cerebral glucose metabolic rate in healthy adults during a cognitive task. Pers Individ Dif (1996) 20(2):183–91. doi:10.1016/0191-8869(95)00167-0
43. Smid H, Lamain W, Hogeboom M, Mulder G, Mulder L. Psychophysiological evidence for continuous information transmission between visual search and response processes. J Exp Psychol Hum Percept Perform (1991) 17(3):696–714. doi:10.1037/0096-1523.17.3.696
44. Smid H, de Witte MR, Homminga I, van den Bosch RJ. Sustained and transient attention in the continuous performance task. J Clin Exp Neuropsychol (2006) 28(6):859–83. doi:10.1080/13803390591001025
45. Cornblatt BA, Lenzenweger MF, Erlenmeyer-Kimling N. The continuous performance test, identical pairs version: II. Contrasting attentional profiles in schizophrenic and depressed patients. Psychiatry Res (1989) 29(1):65–85. doi:10.1016/0165-1781(89)90188-1
46. Nuechterlein KH, Parasuraman R, Jiang Q. Visual sustained attention: image degradation produces rapid sensitivity decrement over time. Science (1983) 220(4594):327–9. doi:10.1126/science.6836276
47. Beale I, Matthew PJ, Oliver S, Corballis MC. Performance of disabled and normal readers on the continuous performance test. J Abnorm Child Psychol (1987) 15(2):229–38. doi:10.1007/BF00916351
48. Silverstein ML, Weinstein M, Turnbull A. Nonpatient CPT performance varying target frequency and interstimulus interval on five response measures. Arch Clin Neuropsychol? (2004) 19(8):1017–25. doi:10.1016/j.acn.2003.12.010
49. Carter L, Russell PN, Helton WS. Target predictability, sustained attention, and response inhibition. Brain Cogn (2013) 82(1):35–42. doi:10.1016/j.bandc.2013.02.002
50. Parasuraman R. Memory load and event rate control sensitivity decrements in sustained attention. Science (1979) 205(4409):924–7. doi:10.1126/science.472714
51. First MB, Gibbon M, Spitzer RL. User’s Guide for the Structured Clinical Interview for DSM-IV Axis II Personality Disorders: SCID-II. Washington, DC: American Psychiatric Publication (1997).
52. Hamilton M. A rating scale for depression. J Neurol Neurosurg Psychiatry (1960) 23(1):56–62. doi:10.1136/jnnp.23.1.56
53. Young RC, Biggs JT, Ziegler VE, Meyer DA. A rating scale for mania: reliability, validity and sensitivity. Br J Psychiatry (1978) 133(5):429–35. doi:10.1192/bjp.133.5.429
54. Beck AT, Steer RA, Brown GK. Beck Depression Inventory-II. San Antonio: Psychological Corporation (1996).
55. Altman EG, Hedeker D, Peterson JL, David JM. The Altman self-rating mania scale. Biol Psychiatry (1997) 42(10):948–55. doi:10.1016/S0006-3223(96)00548-3
56. Nelson HE. National Adult Reading Test (NART): For the Assessment of Premorbid Intelligence in Patients with Dementia: Test Manual. Windsor: NFER-Nelson (1982).
57. Ulrich R, Miller J. Effects of truncation on reaction time analysis. J Exp Psychol (1994) 123(1):34–80. doi:10.1037/0096-3445.123.1.34
58. Lacouture Y, Cousineau D. How to use MATLAB to fit the ex-Gaussian and other probability functions to a distribution of response times. Tutor Quant Methods Psychol (2008) 4(1):35–45.
60. Balota DA, Yap MJ, Cortese MJ, Watson JM. Beyond mean response latency: response time distributional analyses of semantic priming. J Mem Lang (2008) 59(4):495–523. doi:10.1016/j.jml.2007.10.004
61. Spss I. IBM SPSS Statistics Version 21. Boston, MA: International Business Machines Corp (2012).
62. Mackworth NH. The breakdown of vigilance during prolonged visual search. Q J Exp Psychol (1948) 1(1):6–21. doi:10.1080/17470214808416738
63. Mackworth NH. Researches on the Measurement of Human Performance. Vol. 268. London: Her Majesty’s Stationary Office (1950). 156 p.
64. Simon NM, Smoller JW, McNamara KL, Maser RS, Zalta AK, Pollack MH, et al. Telomere shortening and mood disorders: preliminary support for a chronic stress model of accelerated aging. Biol Psychiatry (2006) 60(5):432–5. doi:10.1016/j.biopsych.2006.02.004
65. Sodhi SK, Linder J, Chenard CA, Miller DD, Haynes WG, Fiedorowicz JG. Evidence for accelerated vascular aging in bipolar disorder. J Psychosom Res (2012) 73(3):175–9. doi:10.1016/j.jpsychores.2012.06.004
66. Rizzo LB, Costa LG, Mansur RB, Swardfager W, Belangero SI, Grassi-Oliveira, et al. The theory of bipolar disorder as an illness of accelerated aging: implications for clinical care and research. Neurosci Biobehav Rev (2014) 42:157–69. doi:10.1016/j.neubiorev.2014.02.004
67. Parasuraman R, Giambra L. Skill development in vigilance: effects of event rate and age. Psychol Aging (1991) 6(2):155–69. doi:10.1037/0882-7974.6.2.155
68. Schmiedek F, Oberauer K, Wilhelm O, Süß H-M, Wittmann WW. Individual differences in components of reaction time distributions and their relations to working memory and intelligence. J Exp Psychol (2007) 136(3):414. doi:10.1037/0096-3445.136.3.414
69. Heathcote A, Popiel SJ, Mewhort DJ. Analysis of response time distributions: an example using the Stroop task. Psychol Bull (1991) 109(2):340–7. doi:10.1037/0033-2909.109.2.340
70. Ratcliff R. A diffusion model account of response time and accuracy in a brightness discrimination task: fitting real data and failing to fit fake but plausible data. Psychon Bull Rev (2002) 9(2):278–91. doi:10.3758/BF03196283
71. Ratcliff R, McKoon G. The diffusion decision model: theory and data for two-choice decision tasks. Neural Comput (2008) 20(4):873–922. doi:10.1162/neco.2008.12-06-420
72. Johnson KA, Roberston IH, Kelly SP, Silk TJ, Barry E, Dáibhis A, et al. Dissociation in performance of children with ADHD and high-functioning autism on a task of sustained attention. Neuropsychologia (2007) 45(10):2234–45. doi:10.1016/j.neuropsychologia.2007.02.019
Keywords: bipolar disorder, attention, neuropsychology, ex-Gaussian distribution, variability
Citation: Moss RA, Finkelmeyer A, Robinson LJ, Thompson JM, Watson S, Ferrier IN and Gallagher P (2016) The Impact of Target Frequency on Intra-Individual Variability in Euthymic Bipolar Disorder: A Comparison of Two Sustained Attention Tasks. Front. Psychiatry 7:106. doi: 10.3389/fpsyt.2016.00106
Received: 29 January 2016; Accepted: 03 June 2016;
Published: 16 June 2016
Edited by:
Michael Noll-Hussong, University of Ulm, GermanyReviewed by:
Jared W. Young, University of California, San Diego, USAFelipe Ortuño, Clínica Universidad de Navarra, Spain
Caroline Gurvich, Monash University, Australia
Copyright: © 2016 Moss, Finkelmeyer, Robinson, Thompson, Watson, Ferrier and Gallagher. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Peter Gallagher, cGV0ZXIuZ2FsbGFnaGVyQG5ld2Nhc3RsZS5hYy51aw==
 Rachel Ann Moss
Rachel Ann Moss Andreas Finkelmeyer1
Andreas Finkelmeyer1 Stuart Watson
Stuart Watson Peter Gallagher
Peter Gallagher