- 1Biomedical Engineering, Hanyang University, Seoul, South Korea
- 2Psychiatry, National Health Insurance Service Ilsan Hospital, Goyang, South Korea
- 3Institute of Behavioral Science in Medicine, Yonsei University College of Medicine, Seoul, South Korea
- 4Psychiatry, Yonsei University College of Medicine, Seoul, South Korea
- 5Psychiatry, Eunpyeong Hospital, Seoul, South Korea
Internet gaming disorder (IGD) is characterized by a loss of control over gaming and a decline in psychosocial functioning derived from excessive gameplay. We hypothesized that individuals with IGD would show different autonomic nervous system (ANS) responses to the games than those without IGD. In this study, heart rate variability (HRV) was assessed in 21 young males with IGD and 27 healthy controls while playing their favorite Internet game. The subjects could examine the game logs to identify the most and least concentrated periods of the game. The changes in HRV during specific 5-min periods of the game (first, last, and high- and low-attention) were compared between groups via a repeated measures analysis of variance. Significant predictors of HRV patterns during gameplay were determined from stepwise multiple linear regression analyses. Subjects with IGD showed a significant difference from controls in the patterns of vagally mediated HRV, such that they showed significant reductions in high-frequency HRV, particularly during the periods of high attention and the last 5 min, compared with baseline values. A regression analysis showed that the IGD symptom scale score was a significant predictor of this reduction. These results suggest that an altered HRV response to specific gaming situations is related to addictive patterns of gaming and may reflect the diminished executive control of individuals with IGD while playing Internet games.
Introduction
Internet gaming disorder (IGD), one of the most studied forms of Internet addiction, is characterized by a difficulty in controlling excessive Internet game use despite negative psychosocial consequences (1). Although it has not yet been fully clarified, much effort has been devoted to elucidating the neurobiological background underlying IGD (2). Some researchers are interested in the physiological features of IGD, particularly in autonomic nervous system (ANS) dysfunction. ANS dysfunction has been associated with psychiatric disorders (3), including substance abuse and behavioral addiction (4, 5). As the ANS responds to internal and external stimuli to maintain homeostasis, its function is closely related to adaptive adjustments in behavior strategies (6). ANS dysfunction likely contributes to the development and maintenance of loss of control over gaming, as individuals with IGD are unable to adjust their behavior strategies despite negative outcomes.
ANS function can be assessed non-invasively by measuring heart rate variability (HRV). A study by Lin et al. found that school-aged children with Internet addictions had lower levels of total-power HRV than non-addicted children (7). The authors also indicated that children with Internet addiction had lower high-frequency (HF) percentages and higher low-frequency (LF) percentages than non-addicted children. Data from Kim et al. similarly showed that adolescents with IGD had lower total-power HRV, but both HF and LF values were significantly lower and their ratios did not differ between those with IGD and controls (8). However, there is not sufficient evidence, with mixed results reported in previous studies, to clarify the role of ANS function in IGD.
To more accurately assess ANS function, it may be useful to measure HRV responses to particular stimuli as well as during the resting state, as this better reflects the ability of the ANS to respond appropriately and adaptively to environmental change (9). A previous study on Internet addiction showed that individuals with problematic Internet use had a lower standard deviation of the R-R interval (SDNN, reflects overall HRV levels) than non-problematic users during rest but not during and after the Trier social stress test (TSST) (10). This measure could be applied to assess HRV responses to gaming-related stimuli rather than general stress stimuli. Our previous work suggested that individuals with IGD have HRV suppression during gameplay (11). As HRV suppression may represent inefficient executive control (12), we speculated that HRV suppression in response to gaming in individuals with IGD reflects an imbalance between enhanced reward-seeking and diminished executive control (13). However, our previous study only analyzed HRV data for the first 5 min of the game. These data do not encompass the full range of responses to the variety of situations that gaming comprises, such as those requiring high levels of attention with many points to consider and those with repetitive actions or that have little influence on the final outcome of the game, which require less attention. Our previous study also did not include the subjects' perceptions of these various situations in the game. Thus, further data are needed assessing the alterations of HRV throughout the duration of the game to determine the role of the ANS in IGD pathophysiology.
Numerous studies on addiction suggest that addictive behaviors begin from a voluntary and goal-directed pattern that gradually becomes habitual and compulsive (14, 15). This progression accompanies several neurobiological changes, including weakened prefrontal cortical control and strengthened dorsal striatal control (16, 17). With this in mind, addictive gaming may also be associated with habitual and compulsive behavioral patterns. Individuals with IGD may thus be less influenced by prefrontal control, even when playing games at a high level of attention, and consequently play games in a habitual or compulsive manner rather than a goal-directed manner. To appropriately interpret these behavioral patterns, the situational factors of game should be considered.
We hypothesized that the difference in gaming behaviors between individuals with and without IGD become apparent during situations demanding a high level of attention, reflecting an addictive pattern of habitual gameplay with weakened prefrontal cortical control. We also hypothesized that HRV response during periods of high attention would be significantly related to the severity of IGD rather than to other psychological problems (e.g., depression and anxiety). Unlike the previous studies of HRV in IGD, the current study analyzed the changes in HRV during the game in which gaming addicts were actually addicted, and included consideration of the situational factors of game. Thus, in this study, we analyzed HRV of young males with and without IGD while playing an Internet game at specific time periods, including the initial and final periods, as well as during periods when the gamer was and was not particularly focused. We examined how HRV values change during these different situations, whether they differ between individuals with and without IGD, and if they are associated with the severity of IGD.
Materials and Methods
Participants
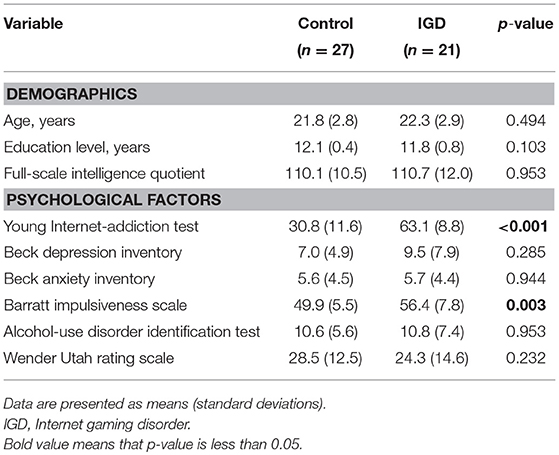
The Institutional Review Board approved the protocol for this study (HYI-16-044), and all subjects provided signed informed consent before participating. This study included 48 subjects, all right handed and aged between 16 and 27 years (mean age: 22.0 ± 2.8 years), who were recruited through online bulletin boards, flyers, and word of mouth. All subjects were assessed for their Internet-use patterns. All subjects in this study frequently played “League of Legends” (Riot Games, 2009), the most popular multiplayer online battle arena game in Korea. To control for the difference of hedonic sensation or familiarity of each subject for the game, only those who were in the same rank in the game were recruited. Participants who primarily used the Internet for gaming and scored above 50 on the Young's Internet-addiction test (Y-IAT) (18) were classified as subjects with IGD (n = 21; age, 22.3 ± 2.9 years). They were reassessed for IGD via a psychiatric interview based on the criteria in the Diagnostic and Statistical Manual of Mental Disorders, fifth edition (1). Subjects who scored below 50 on the Y-IAT were classified as controls (n = 27; age, 21.8 ± 2.8 years).
All subjects were screened via a four-item brief screening tool (19) (feeling nervous, anxious, or on edge; not being able to stop or control worrying; feeling down, depressed, or hopeless; little interest or pleasure in doing things) to ensure they had no clinically significant depression or anxiety symptoms during more than half of the last 2 weeks. The Korean version of the Diagnostic and Statistical Manual of Mental Disorders, fourth edition, was also used to assess the presence of major mental disorders other than IGD (20) and the Wechsler adult intelligence scale IV was used to assess intelligence quotients (21). All subjects completed the following respective self-report questionnaires regarding depression, anxiety, alcohol-related problems, childhood symptoms of attention-deficit/hyperactivity disorder, and impulsivity: the Beck depression inventory (22), the Beck anxiety inventory (23), the alcohol-use disorders identification test (24), the Wender Utah rating scale (25), and the Barratt impulsiveness scale, version 11 (26).
Subjects with a substantial psychiatric comorbid condition other than IGD (e.g., depression, psychotic disorder, or substance dependence), neurological or medical disorder that affected the HRV (e.g., cardiac disease or endocrine disease), low intelligence, or were taking drugs that affected the HRV (e.g., beta blockers or anticholinergics) were excluded from the study. All subjects were psychiatric medication naïve at the time of assessment.
Experimental Protocol
The experimental protocol for this study is presented in Figure 1. After a rest period of at least 10 min, resting-state electrocardiograms (ECGs) were recorded for 5 min while subjects were in a relaxed sitting position for the baseline HRV. The subjects then played the online game League of Legends three times, with a 5-min rest after each game. Each game lasted for at least 20 min. After each game ended, subjects checked the game log to identify 5-min periods of high attention and low attention. For each game, initial HRV (during the first 5 min), high- and low-attention HRV (during the 5-min periods of high and low attention, respectively), and last-time HRV (during the final 5 min of the game) signals were analyzed. HRV values for each period were averaged from 3 games. After all the games were over, HRV signals were measured in the resting state for 5 min for the post-game HRV.
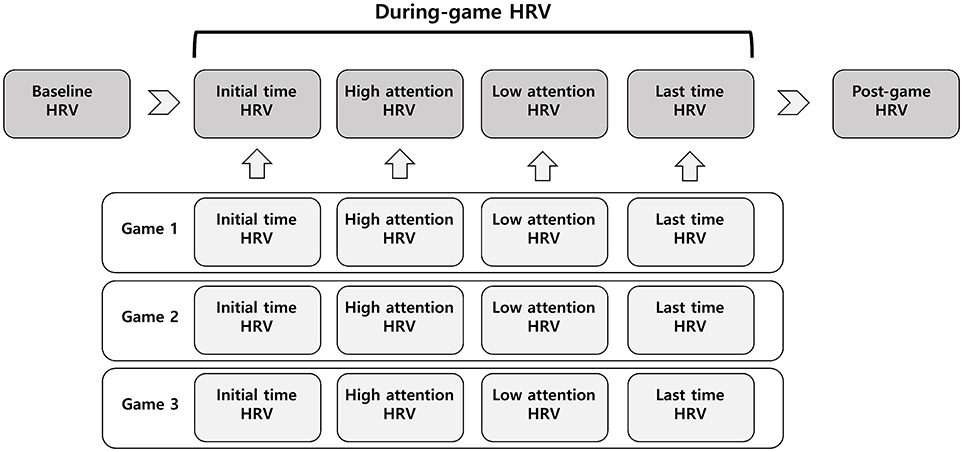
Figure 1. Experimental protocol of measurements for heart rate variability (HRV). Each period was 5 min.
HRV Analysis
Three ECG channels were connected to each of the subjects' chests to obtain ECG signals via an MP150 (BIOPAC Systems Inc., Santa Barbara, CA, USA). To eliminate noise from the subjects' movement, breathing, and muscle electrical activity, the data were preprocessed using third-order Butterworth high-pass filtering with a 0.1-Hz cutoff frequency, a sixth-order Butterworth notch filter, and third-order Butterworth low-pass filtering with a 15-Hz cutoff frequency (27, 28). All ECG signals were acquired at 200 Hz, and the Pan and Tomkins method was used to automatically detect R-R intervals (29).
HRV parameters were extracted from time and frequency-domains, using a HRV analysis software (30). The time-domain method measures the time between R-R intervals or the instantaneous heart rate at a specific time. The time-domain parameters used in this study were the SDNN and the root mean squared differences of successive N-N intervals (RMSSD). SDNN indicates overall HRV, whereas RMSSD indicates short-term changes in heart rate and is used to predict parasympathetic activity (31). For the frequency-domain analyses, we used a 20% filter to remove ectopic beats, and data were interpolated at 4 Hz (32). The frequency-domain parameters were transformed with an autoregressive model. The LF domain of HRV (0.04–0.15 Hz) reflects parasympathetic and sympathetic nerve activity, whereas the HF domain (0.15–0.4 Hz) mainly represents parasympathetic activity (33). The frequency-domain HRV parameters with skewed distributions were logarithmically transformed (34). The LF/HF ratio was used as an index of sympathovagal balance (35).
Statistical Analysis
Statistical analyses were performed with Statistical Package for the Social Sciences (SPSS), version 24.0 K (SPSS Inc., Chicago, IL, USA). A p-value of < 0.05 was considered statistically significant. Independent t-tests were used to compare demographic and psychometric characteristics or absolute values of HRV parameters between subjects with IGD and controls. Repeated measures analyses of variance (ANOVAs) were used to group differences in longitudinal changes of HRV, using the period (baseline, initial, high-attention, low-attention, last-time, and post-game) as the within-subjects factor and group (IGD subjects or controls) as the between-subjects factor. Among the various parameters of HRV (lnLF, lnHF, SDNN, RMSSD, and LF/HF), we tested which parameter was statistically significant in the interaction between group and period, and post-hoc comparisons were applied to test whether the differences between certain periods were statistically significant. Bonferroni's corrections (p < 0.05/15) were conducted to adjust for multiple comparisons (15 paired comparisons of six periods). These analyses were used to characterize the patterns of HRV during gameplay (changes of specific HRV parameters between specific periods in the game) in subjects with IGD. Stepwise multiple linear regression analyses were then conducted to identify significant predictors of these patterns. For these analyses, demographic or psychometric characteristics (Y-IAT, age, intelligence quotient, scores from self-report questionnaires) of all subjects were entered as independent variables.
Results
Clinical Characteristics of the Subjects
Subjects with IGD had Y-IAT scores that were significantly higher than the controls (p < 0.001; Table 1). Subjects with IGD and controls did not differ significantly in self-reported scale scores for depression, anxiety, alcohol-related problems, and childhood attention-deficit/hyperactivity symptoms. However, subjects with IGD scored significantly higher on tests of impulsivity than did controls (p = 0.003).
HRV Changes
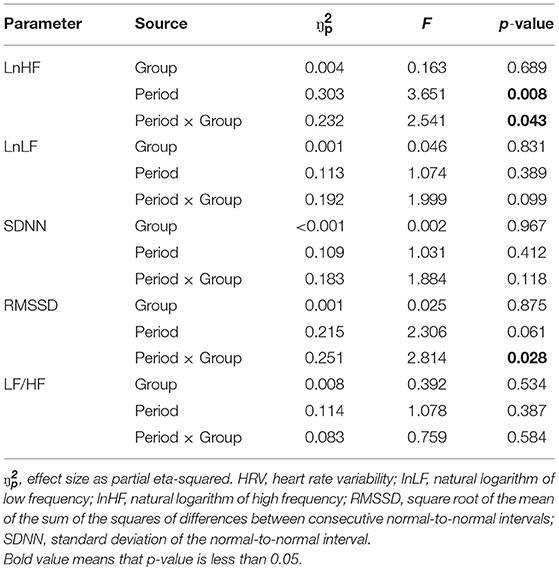
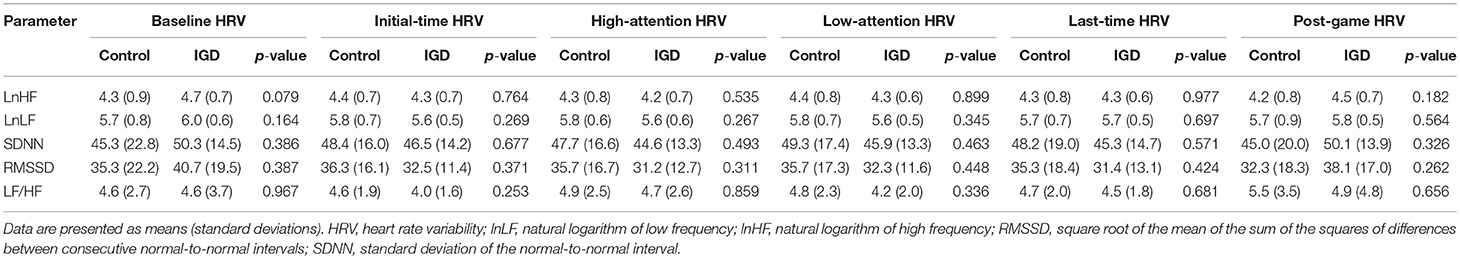
For all periods, there were no statistically significant differences between subjects with IGD and controls in any of the HRV parameters (Table 2). However, interaction effects between period and group were statistically significant for lnHF (p = 0.043) and RMSSD (p = 0.028) via repeated measures ANOVAs (Table 3).
Post-hoc comparisons showed that subjects with IGD had significant reductions in lnHF for high-attention HRV (p = 0.001) and last-time HRV (p = 0.003) compared with that for baseline HRV. Although not statistically significant after correction for multiple comparisons, subjects with IGD also had trends toward reductions in RMSSD for high-attention HRV (p = 0.007) compared with that for baseline HRV. Unlike subjects with IGD, subjects without IGD did not show significant differences in any of the HRV parameter between specific periods (p > 0.05).
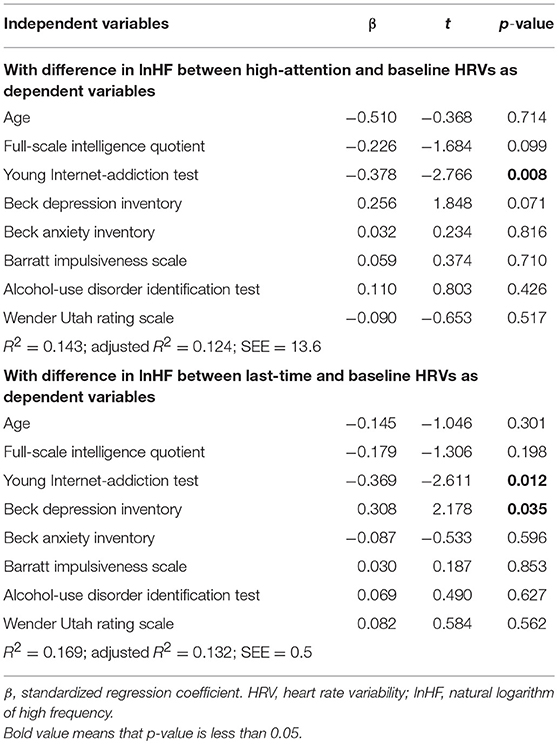
Stepwise multiple linear regression analyses were performed with the differences of lnHF for high-attention HRV and last-time HRV relative to baseline HRV as the dependent variables (Table 4). Of the independent variables, only the Y-IAT score was a significant predictor of the difference of lnHF between high-attention HRV and baseline HRV. However, the Y-IAT score and Beck depression inventory score were significant predictors of the difference in lnHF between last-time HRV and baseline HRV. All the variables used for multiple linear regression had a variance inflation factor of <5.
Discussion
In this study, we measured the changes in HRV in young males with IGD while they played an Internet game. Although the absolute values for HRVs did not differ between young males with IGD and controls, those with IGD had significant differences with respect to longitudinal changes of lnHF and RMSSD during gaming. Specifically, lnHF was reduced from baseline during periods of high attention and during the last 5 min of the game, supporting our hypothesis that a characteristic HRV response would become apparent during game periods requiring a high level of attention. Further analyses supported our second hypothesis, showing that the severity of the IGD, assessed by the Y-IAT score, predicted this characteristic reduction in lnHF.
The group differences in patterns of change in lnHF and RMSSD we observed during gameplay are consistent with a previous study, suggesting that HRV responses in subjects with IGD differ from those of controls while playing the game (11). HF and RMSSD are closely correlated and reflect vagal activity (35). The neurovisceral model suggests that vagally mediated HRV is an index that reflects executive control over affective and cognitive processes (36). According to this model, these differences are related to the difficulty in exerting executive control over excessive gaming in subjects with IGD.
Subjects with IGD showed significant reductions in lnHF for high-attention HRV and last-time HRV compared with that for baseline HRV. Although statistical significance was not high enough, they also showed trends toward reductions in RMSSD for high-attention HRV compared with that for baseline HRV. A large reduction of vagally mediated HRV is suggested to be a specialized HRV feature that reliably reflects weakened prefrontal neural function responsible for top-down executive control (37). In the present study, the subjects determined what period required high attention, and the gameplay during the final 5 min was a determinant of whether the game was won or lost. Thus, during these important periods, the reduction in HF-HRV suggests that top-down executive control was diminished when the demand for concentration on the game was increased. We infer that these characteristics of gameplay in young males with IGD are related to their habitual/compulsive game use.
Interestingly, the only predictor of the reduced lnHF during periods of high attention was the severity of the IGD, suggesting that an altered HRV response to gaming in individuals with IGD reflects the addictive features of gaming. Therefore, the current findings support a putative role of altered ANS response to gaming in the status of IGD. On the other hand, the severity of both IGD and depression predicted the reduction in lnHF during last 5 min of the game, when some of the gamers may have given up. We speculate that psychological characteristics of depression affect the extent to which subjects concentrate at the end of the game, which is associated with their HRV responses.
As psychological conditions (e.g., depression and anxiety) were assessed prior to participation, we ensured that the subjects with IGD and controls did not differ in this regard. This may explain why no significant differences in the resting-state HRVs were observed. However, subjects with IGD self-reported higher impulsiveness than controls, in agreement with previous studies in which high impulsivity is the prominent psychobehavioral feature in IGD (38, 39). Moreover, high impulsivity is related to diminished executive function (40). Nevertheless, impulsivity and executive function each contain various components, and the relationship between them is complex (41). Further research is needed to clarify the association between the high impulsivity of individuals with IGD and their HRV responses to gaming.
There are some limitations to this study. First, the sample size of this study is small, thus not all variables affecting HRV could be controlled. Second, the inclusion of indexes besides HRV is needed to fully reflect the subjects' physiological and autonomic responses to gaming. Third, the design of this study was cross-sectional; therefore, the changes in HRV responses observed during gameplay should be followed up in longitudinal studies of subjects with IGD. Such studies would validate the use of HRV as an important index reflecting the progress of treatments for IGD.
Despite its limitations, our current study is the first to analyze HRV data during the game, encompassing responses to the specific gaming situations. Young males with IGD showed vagally mediated HRV responses to gaming that were significantly different from those of healthy controls, particularly during periods requiring high attention. They showed a reduction in HF-HRV that was also related to the severity of their IGD. This alteration in their autonomic responses suggests that individuals with IGD are less influenced by executive control, even when playing games at a high level of attention. Our results suggest that the habitual gaming behaviors of individuals with loss of control over gaming reflects diminished prefrontal control.
Ethics Statement
All of the procedures involving human participants were performed in accordance with the ethical standards of the institutional and national research committees and with the 1964 Helsinki declaration and its later amendments. The experimental protocol was approved by the Institutional Review Board at Hanyang University (HYI-16-044).
Author Contributions
Y-CJ and IK contributed to the study design. JP and JEL collected the clinical and bio-signal data. SH and DL performed statistical analysis and wrote the first draft of the manuscript. KN, JL and DJ provided critical revision of the manuscript and important intellectual content. All authors contributed to manuscript revision, read, and approved the submitted version.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
This research was supported by the Original Technology Research Program for Brain Science through the National Research Foundation of Korea funded by the Ministry of Science, ICT and Future Planning (NRF-2015M3C7A1064906 and 2015M3C7A1064789).
References
1. Petry NM, O'brien CP. Internet gaming disorder and the DSM-5. Addiction (2013) 108:1186–7. doi: 10.1111/add.12162
2. Kuss DJ, Griffiths MD. Online gaming addiction in children and adolescents: a review of empirical research. J Behav Addict. (2012) 1:3–22. doi: 10.1556/JBA.1.2012.1.1
3. Alvares GA, Quintana DS, Hickie IB, Guastella AJ. Autonomic nervous system dysfunction in psychiatric disorders and the impact of psychotropic medications: a systematic review and meta-analysis. J Psychiatry Neurosci. (2016) 41:89–104. doi: 10.1503/jpn.140217
4. Ulrich N, Ambach W, Hewig J. Severity of gambling problems modulates autonomic reactions to near outcomes in gambling. Biol Psychol. (2016) 119:11–20. doi: 10.1016/j.biopsycho.2016.06.005
5. Quintana DS, Guastella AJ, McGregor IS, Hickie IB, Kemp AH. Heart rate variability predicts alcohol craving in alcohol dependent outpatients: further evidence for HRV as a psychophysiological marker of self-regulation. Drug Alcohol Depend. (2013) 132:395–8. doi: 10.1016/j.drugalcdep.2013.02.025
6. Porges SW. The polyvagal theory: new insights into adaptive reactions of the autonomic nervous system. Cleve Clin J Med. (2009) 76:S86–90. doi: 10.3949/ccjm.76.s2.17
7. Lin PC, Kuo SY, Lee PH, Sheen TC, Chen SR. Effects of internet addiction on heart rate variability in school-aged children. J Cardiovasc Nurs. (2014) 29:493–8. doi: 10.1097/JCN.0b013e3182a477d5
8. Kim N, Hughes TL, Park CG, Quinn L, Kong ID. Altered autonomic functions and distressed personality traits in male adolescents with internet gaming addiction. Cyberpsychol Behav Soc Netw. (2016) 19:667–73. doi: 10.1089/cyber.2016.0282
9. Laborde S, Mosley E, Thayer JF. Heart rate variability and cardiac vagal tone in psychophysiological research–recommendations for experiment planning, data analysis, and data reporting. Front Psychol. (2017) 8:213. doi: 10.3389/fpsyg.2017.00213
10. Moretta T, Buodo G. Autonomic stress reactivity and craving in individuals with problematic internet use. PLoS ONE (2018) 13:e0190951. doi: 10.1371/journal.pone.0190951
11. Lee D, Hong SJ, Jung YC, Park J, Kim IY, Namkoong K. Altered heart rate variability during gaming in internet gaming disorder. Cyberpsychol Behav Soc Netw. (2018) 21:259–67. doi: 10.1089/cyber.2017.0486
12. Park G, Thayer JF. From the heart to the mind: cardiac vagal tone modulates top-down and bottom-up visual perception and attention to emotional stimuli. Front Psychol. (2014) 5:278. doi: 10.3389/fpsyg.2014.00278
13. Brand M, Young KS, Laier C, Wölfling K, Potenza MN. Integrating psychological and neurobiological considerations regarding the development and maintenance of specific Internet-use disorders: an Interaction of Person-Affect-Cognition-Execution (I-PACE) model. Neurosci Biobehav Rev. (2016) 71:252–66. doi: 10.1016/j.neubiorev.2016.08.033
14. Everitt BJ, Dickinson A, Robbins TW. The neuropsychological basis of addictive behaviour. Brain Res. Rev. (2001) 36:129–38. doi: 10.1016/S0165-0173(01)00088-1
15. Everitt BJ, Robbins TW. Neural systems of reinforcement for drug addiction: from actions to habits to compulsion. Nat Neurosci. (2005) 8:1481–9. doi: 10.1038/nn1579
16. Everitt BJ, Robbins TW. From the ventral to the dorsal striatum: devolving views of their roles in drug addiction. Neurosci Biobehav Rev. (2013) 37:1946–54. doi: 10.1016/j.neubiorev.2013.02.010
17. Feil J, Sheppard D, Fitzgerald PB, Yücel M, Lubman DI, Bradshaw JL. Addiction, compulsive drug seeking, and the role of frontostriatal mechanisms in regulating inhibitory control. Neurosci Biobehav Rev. (2010) 35:248–75. doi: 10.1016/j.neubiorev.2010.03.001
18. Young KS. Caught in the Net: How to Recognize The Signs of Internet Addiction-and a Winning Strategy for Recovery. New York, NY: John Wiley & Sons (1998).
19. Kroenke K, Spitzer RL, Williams JB, Löwe B. An ultra-brief screening scale for anxiety and depression: the PHQ−4. Psychosomatics (2009) 50:613–21. doi: 10.1176/appi.psy.50.6.613
20. First MB, Spitzer RL, Gibbon M, Williams JB. Structured Clinical Interview for DSM-IV Axis I Disorders. New York, NY: New York State Psychiatric Institute (1995).
21. Wechsler D. Wechsler Adult Intelligence Scale-Fourth Edition (WAIS-IV). San Antonio, TX: The Psychological Corporation (2008).
22. Beck AT, Steer RA, Brown GK. Beck Depression Inventory-II. Vol. 78. San Antonio, TX: The Psychological Corporation (1996).
23. Beck AT, Epstein N, Brown G, Steer RA. An inventory for measuring clinical anxiety: psychometric properties. J Consult Clin Psychol. (1988) 56:893. doi: 10.1037/0022-006X.56.6.893
24. Reinert DF, Allen JP. The alcohol use disorders identification test (AUDIT): a review of recent research. Alcohol Clin Exp Res. (2002) 26:272–9. doi: 10.1111/j.1530-0277.2002.tb02534.x
25. Ward MF. The Wender Utah Rating Scale: an aid in the retrospective. Am J Psychiatry (1993) 1:885. doi: 10.1176/ajp.150.6.885
26. Patton JH, Stanford MS. Factor structure of the Barratt impulsiveness scale. J Clin Psychol. (1995) 51:768–74. doi: 10.1002/1097-4679(199511)51:6<768::AID-JCLP2270510607>3.0.CO;2-1
27. Zhao ZD, Chen YQ. A New Method for Removal of Baseline Wander and Power Line Interference in ECG Signals. Dalian: IEEE (2006), p. 4342–7.
28. Christov II, Daskalov IK. Filtering of electromyogram artifacts from the electrocardiogram. Med Eng Phys. (1999) 21:731–6. doi: 10.1016/S1350-4533(99)00098-3
29. Pan J, Tompkins WJ. A real-time QRS detection algorithm. IEEE Trans Biomed Eng. (1985) 32:230–6. doi: 10.1109/TBME.1985.325532
30. Ramshur JT. Design, Evaluation, and Application of Heart Rate Variability Analysis Software (HRVAS). Memphis, TN: University of Memphis (2010).
31. Mietus J, Peng C, Henry I, Goldsmith R, Goldberger A. The pNNx files: re-examining a widely used heart rate variability measure. Heart (2002) 88:378–80. doi: 10.1136/heart.88.4.378
32. Peretz A, Kaufman JD, Trenga CA, Allen J, Carlsten C, Aulet MR, et al. Effects of diesel exhaust inhalation on heart rate variability in human volunteers. Environ Res. (2008) 107:178–84. doi: 10.1016/j.envres.2008.01.012
33. Reyes del Paso GA, Langewitz W, Mulder LJ, Roon A, Duschek S. The utility of low frequency heart rate variability as an index of sympathetic cardiac tone: a review with emphasis on a reanalysis of previous studies. Psychophysiology (2013) 50:477–87. doi: 10.1111/psyp.12027
34. Boardman A, Schlindwein FS, Rocha AP. A study on the optimum order of autoregressive models for heart rate variability. Physiol Meas. (2002) 23:325–36. doi: 10.1088/0967-3334/23/2/308
35. Sztajzel J. Heart rate variability: a noninvasive electrocardiographic method to measure the autonomic nervous system. Swiss Med Wkly. (2004) 134:514–22.
36. Thayer JF, Lane RD. Claude Bernard and the heart–brain connection: further elaboration of a model of neurovisceral integration. Neurosci Biobehav Rev. (2009) 33:81–8. doi: 10.1016/j.neubiorev.2008.08.004
37. Thayer JF, Hansen AL, Saus-Rose E, Johnsen BH. Heart rate variability, prefrontal neural function, and cognitive performance: the neurovisceral integration perspective on self-regulation, adaptation, and health. Ann. Behav. Med. (2009) 37:141–53. doi: 10.1007/s12160-009-9101-z
38. Cao F, Su L, Liu T, Gao X. The relationship between impulsivity and internet addiction in a sample of Chinese adolescents. Eur Psychiatry (2007) 22:466–71. doi: 10.1016/j.eurpsy.2007.05.004
39. Choi SW, Kim H, Kim GY, Jeon Y, Park S, Lee JY, et al. Similarities and differences among Internet gaming disorder, gambling disorder and alcohol use disorder: a focus on impulsivity and compulsivity. J. Behav. Addict. (2014) 3:246–53. doi: 10.1556/JBA.3.2014.4.6
40. Bickel WK, Jarmolowicz DP, Mueller ET, Gatchalian KM, McClure SM. Are executive function and impulsivity antipodes? A conceptual reconstruction with special reference to addiction. Psychopharmacology (2012) 221:361–87. doi: 10.1007/s00213-012-2689-x
Keywords: autonomic nervous system, heart rate variability, internet gaming disorder, gameplay, addiction
Citation: Hong SJ, Lee D, Park J, Namkoong K, Lee JS, Jang DP, Lee JE, Jung Y-C and Kim IY (2018) Altered Heart Rate Variability During Gameplay in Internet Gaming Disorder: The Impact of Situations During the Game. Front. Psychiatry 9:429. doi: 10.3389/fpsyt.2018.00429
Received: 01 March 2018; Accepted: 20 August 2018;
Published: 11 September 2018.
Edited by:
Jintao Zhang, Beijing Normal University, ChinaReviewed by:
Seung-Hwan Lee, Inje University Ilsan Paik Hospital, South KoreaHee Chan Kim, Seoul National University, South Korea
Youngjoon Chee, University of Ulsan, South Korea
Copyright © 2018 Hong, Lee, Park, Namkoong, Lee, Jang, Lee, Jung and Kim. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Young-Chul Jung, ZXVnZW5lanVuZ0B5dWhzLmFj
In Young Kim, aXlraW1AaGFueWFuZy5hYy5rcg==
†These authors have contributed equally to this work
 Sung Jun Hong
Sung Jun Hong Deokjong Lee
Deokjong Lee Jinsick Park
Jinsick Park Kee Namkoong3,4
Kee Namkoong3,4 Dong Pyo Jang
Dong Pyo Jang Young-Chul Jung
Young-Chul Jung In Young Kim
In Young Kim