- 1Graduate Institute of Biomedical Sciences, College of Medicine, China Medical University, Taichung, Taiwan
- 2Division of Nephrology and Kidney Institute, China Medical University Hospital, Taichung, Taiwan
- 3Division of Pulmonary and Critical Care Medicine, China Medical University Hospital and China Medical University, Taichung, Taiwan
- 4Department of Family Medicine, China Medical University Hospital, Taichung, Taiwan
- 5Management Office for Health Data, China Medical University Hospital and Center of Augmented Intelligence in Healthcare, Taichung, Taiwan
- 6College of Medicine, China Medical University, Taichung, Taiwan
- 7Department of Nuclear Medicine, China Medical University Hospital, Taichung, Taiwan
- 8Department of Bioinformatics and Medical Engineering, Asia University, Taichung, Taiwan
Purpose: The association between neurodegenerative diseases and transfusion remains to be investigated.
Methods: The study population comprised 63,813 patients who underwent a blood transfusion and 63,813 propensity score-matched controls between 2000 and 2010. Data were obtained from the Taiwan National Health Insurance Research Database, which is maintained by the National Health Research Institutes. A Cox regression analysis was conducted to elucidate the relationship between blood transfusions and the risk of dementia.
Results: A multivariate Cox regression analysis of factors, such as age, sex, cardiovascular ischemia disease, and depression, revealed that patients who underwent a blood transfusion showed a 1.73-fold higher risk of dementia [95% confidence interval (CI) = 1.62-1.84] and a 1.37-fold higher risk of Alzheimer’s disease (AD) [95% CI = 1.13-1.66] than those who did not. Patients who received a transfusion of washed red blood cells showed a 2.37-fold higher risk of dementia (95% CI = 1.63-3.44) than those who did not.
Conclusion: Blood transfusion, especially transfusion of any type of red blood cells is associated with an increased risk of dementia.
Introduction
Anemia, defined as hemoglobin concentration <12 g/dl in women and <13 g/dl in men according to World Health Organization (WHO), is a prevalent condition worldwide (1). The burden of anemia is high, an is estimated to effect between 27% to 32.9% of the world population (2, 3). Anemia is associated with increasing mortality and morbidity (4). In addition to correct underlying diseases, transfusion is a frequent clinical procedure carried out in patients with anemia (5–8). However, transfusion itself is also related to adverse events and complications (9, 10). Iron overload is one of the inevitable and notorious complications of transfusion, especially in transfusion-dependent patients (11, 12). Clinical consequences of deregulated iron metabolism and iron overload has previously been reported including hemochromatosis, insulin resistance, peripheral inflammation, cirrhosis, heart failure, and decreased survival benefits (13–17).
Recently, decreased serum iron levels have been reported to be causally associated with an increased risk of developing Parkinson’s disease (PD) (18). Abnormal deposits of iron has been observed to pathologically accumulate in the substantia nigra in PD and in the cortex in AD (19). Lei et al. have also showed that Tau deficiency could induce parkinsonism with dementia by impairing Amyloid precursor protein-mediated iron exports (20). Duce et al. have found that iron-export ferroxidase activity of the β-amyloid precursor protein undergoes interference and inhibition in AD (21). The above studies clearly demonstrate that perturbations in iron homeostasis and ferroptotic signaling are tightly associated with the development of neurodegeneration (22). Furthermore, inflammation can be induced during blood transfusion (23) and has also been reported to be involved in the pathogenesis of neurodegeneration (24).
Pathogenesis of neurodegeneration also involves misfolding protein aggregation and prorogation through anatomical connections of peripheral neurons (25, 26). Given the evidence that peripheral inoculation of the misfolded proteins can induce the aggregation of aberrant proteins in mice models and vascular inflammation is associated with dementia (27–30), the horizontal transmissibility of neurodegenerative diseases might therefore be possible (31, 32).
Edgren et al. however, reported that AD and PD cannot be transmitted through blood transfusions with blood originating from donors with misfolded proteins (33). Whether transfusion is associated with an increased risk of neurodegeneration and dementia remains unknown and called for a nationwide longitudinal cohort study. In this study, we analyzed the association between blood transfusions and the risk of dementia, using data from the National Health Insurance Research Database (NHIRD), which contains claims data of most Taiwanese citizens (> 99% coverage rate) and thus provides valuable information for epidemiological investigations.
Methods
Data Source
The Taiwan National Health Insurance (NHI) program was implemented in March 1995 and covers approximately 99% of Taiwanese residents (34). This retrospective population-based cohort study was conducted using data from the NHIRD for the period from 2000 to 2011. The details of the database and the program are provided in previous studies (35).
Sampled Patients
In this study, we included patients aged ≥20 years who underwent a blood transfusion from 1 January 2000, to 31 December 2011. A blood transfusion was defined as a transfusion of packed red blood cells (RBCs), washed RBCs, frozen deglycerolized RBCs, leukocyte-poor RBCs, platelet concentrate, white blood cells (WBC) concentrate, plateletpheresis, WBCphresis, fresh frozen plasma, frozen plasma, cryoprecipitate, whole blood, and reduced leukocytes-platelets. The date of the blood transfusion was used as the index date. Patients with missing data on their date of birth and sex and those with preexisting dementia (ICD-9-CM codes 290, 331, 294.0, or 294.1) or AD (ICD-9-CM codes 290.1 and 331.0), were excluded. Further, to investigate the long-term effect of transfusion, we delayed the start of follow-ups for dementia by 2 years. Thus, persons with any dementia or AD diagnosed within 2 years after the index date were also excluded.
Every patient who underwent a blood transfusion was propensity score-matched to one randomly selected insurant without any history of blood transfusion, dementia, or AD at baseline. A logistic regression model was used to calculate propensity scores for patients in need of blood transfusion as a function of the background variables including age, sex, year of the index date, urbanization level, occupation, frequency of brain CT/MRI/per year, frequency of psychiatric outpatient visits/per year, and comorbidities. The comorbidities included in this study were diabetes (ICD-9-CM code 250), hypertension (ICD-9-CM code 401 to 405), hyperlipidemia (ICD-9-CM code 272), anxiety (ICD-9-CM code 300.00), depression (ICD-9-CM codes 296.2, 296.3, 300.4, and 311), obesity (ICD-9-CM code 278), bipolar disorder (ICD-9-CM code 296), schizophrenia (ICD-9-CM code 295), head injury (ICD-9-CM codes 800–804, 850–854, and 959.01), injury and poisoning (ICD-9-CM codes 800–999; excluding 800–804, 850–854, and 910–919), posttraumatic stress disorder (ICD-9-CM code 309.81), iron deficiency anemia (ICD-9-CM code 280.9), stroke (ICD-9-CM codes 430–438), and chronic kidney disease (CKD) and end-stage renal disease (ICD-9-CM code 585), GI bleeding (ICD-9-CM codes 530-535, 562.12, 562.13, 569.3, 569.85, 578, 455.2, 455.5, 455.8), and surgery (ICD-9-CM procedure codes 01-99). The medications included in this study were benzodiazepine (BZD), non-BZD, and antipsychotics; and the year of the index date listed in Table 1. The non-BZD included Zopiclone, Zolpidem hemitartrate, Zolpidem tartrate, and Zaleplon. Patients who had a blood transfusion were matched (1:1 ratio) with those who did not have a blood transfusion according to their propensity score through the nearest neighbor matching, initially to the eighth digit and then as required to the first digit. Therefore, matches were first made within a caliper width of 0.0000001, and then the caliper width was increased for unmatched cases to 0.1. We reconsidered the matching criteria and performed a rematch (greedy algorithm). The mean and median propensity scores were compared between the two cohorts.
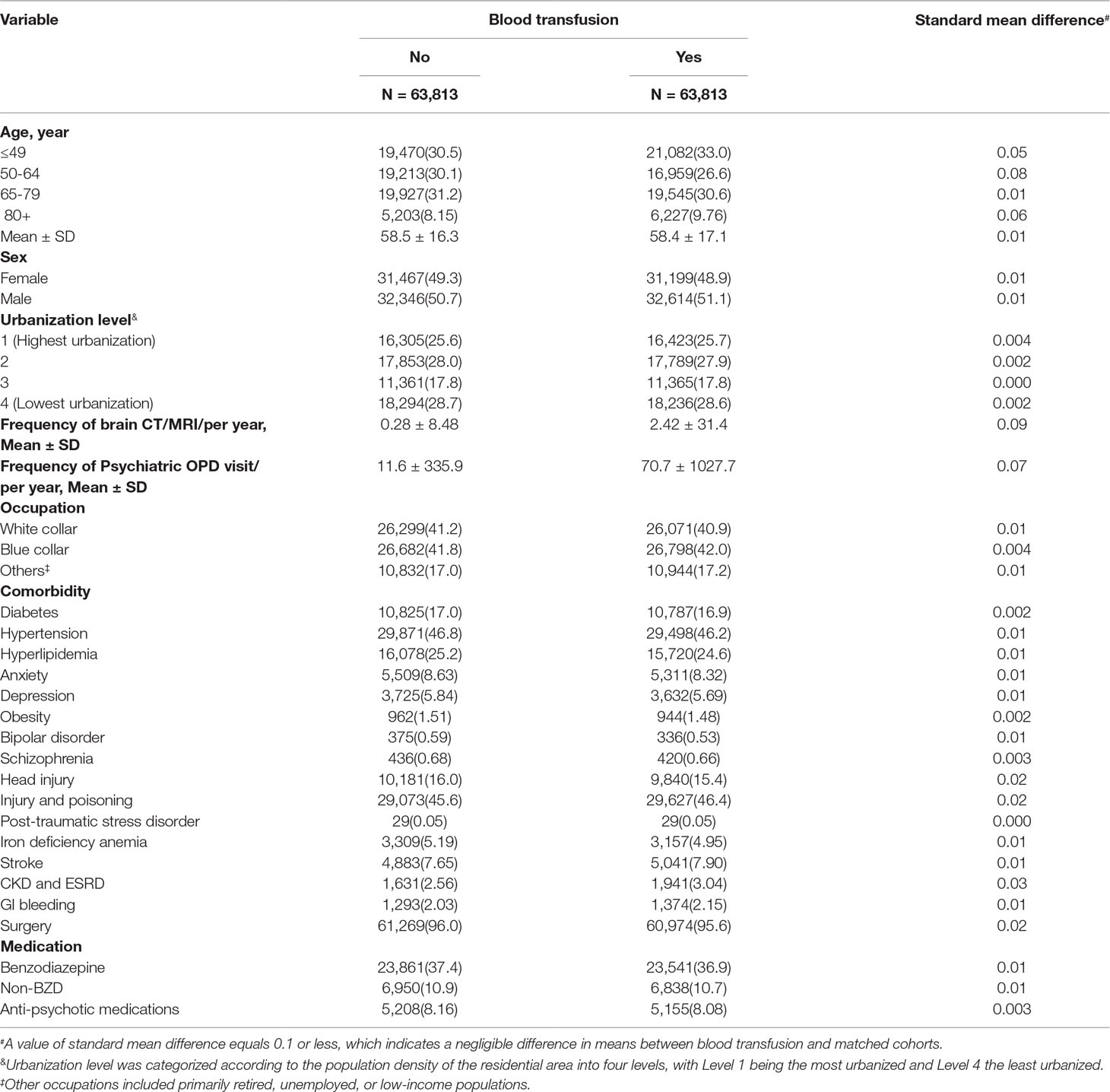
Table 1 Demographic characteristics and comorbidities in cohorts with and without blood transfusion.
Outcome
The primary outcome of this study was a diagnosis of dementia (ICD-9-CM codes 290, 331, 294.0, or 294.1) and AD (ICD-9-CM codes 290.1 and 331.0). Each study patient was followed up with until the primary outcome was diagnosed or were censored because of death, withdrawal from the NHI program, or if after 31 December 2011.
Statistical Analysis
The standard mean difference was used to examine the difference in categorical variables and continuous variables between the blood transfusion and matched cohorts. The standard mean difference value was 0.1 or less, which indicated a negligible mean difference between the blood transfusion and matched cohorts. The Kaplan–Meier method was used to estimate the cumulative incidence of subsequent dementia in the blood transfusion and matched cohorts, and significant differences were determined using the log-rank test. We calculated the incidence density rate of dementia according to different risk factors in analyses stratified by age, sex, urbanization level, occupation, comorbidities, and medication. Univariable and multivariable Cox proportional hazards models were used to estimate the hazard ratios (HRs) and 95% confidence intervals (CIs) to determine the association between blood transfusions and the risks of dementia or AD. The multivariable models were simultaneously adjusted for age, sex, urbanization level, occupation, frequency of brain CT/MRI/per year, and frequency of Psychiatric OPD visit/per year, and comorbidities of diabetes, hypertension, hyperlipidemia, anxiety, depression, obesity, bipolar disorder, schizophrenia, head injury, injury and poisoning, post-traumatic stress disorder, iron deficiency anemia, stroke, CKD and ESRD, GI bleeding, surgery, and medication of benzodiazepine, non-BZD, and anti-psychotic medications. A further analysis was performed to estimate the risk of dementia among patients receiving different types of blood transfusions. All statistical analyses were performed using SAS version 9.4 statistical software (SAS Institute, Inc., Cary, N.C., USA). The significance level for all analyses was set to a p value of 0.05.
Results
Demographic Characteristics, Comorbidities and Medications of Patients With Blood Transfusion and the Matched Cohort
A total of 63,813 patients were included in the blood transfusion cohort and 63,813 persons in the matched cohort, with similar distributions for age, sex, urbanization level, frequency of brain CT/MRI/per year, frequency of psychiatric outpatient visit/per year, occupation, comorbidities, and medication (Table 1). The median follow-up period was 3.86 ± 3.52 years in the study cohort and 5.49 ± 3.34 years in the matched cohort. In both cohorts, more than half of the patients were aged <65 years (59.6% vs 60.6%). The mean ages of the blood transfusion and matched cohorts at index date were 58.4 years (SD = 17.1) and 58.5 years (SD = 16.3), respectively. Both cohort groups mainly resided in highly urbanized areas (53.6% vs 53.6%) and were employed in blue collar jobs (42.0% vs 41.8%). The mean frequency of brain CT/MRI/per year in the matched and blood transfusion cohorts was 0.28 (SD = 8.48) and 2.42 (SD = 31.4) times per year, respectively. The major comorbidity was hypertension (46.2% vs 46.8%), followed by injury and poisoning (46.4% vs 45.6%) and hyperlipidemia (24.6% vs 25.2%). In both cohorts, the main medication used was BZD (36.9% vs 37.4%). In the category of injury and poisoning, there are 35 patients with carbon monoxide intoxication in blood transfusion cohort (0.05%; there are 29 patients with carbon monoxide intoxication in the matched cohort (0.05%)(p-value = 0.45).
Incidence Densities and Cox Model Analysis of Risk Factors Affecting Dementia Development
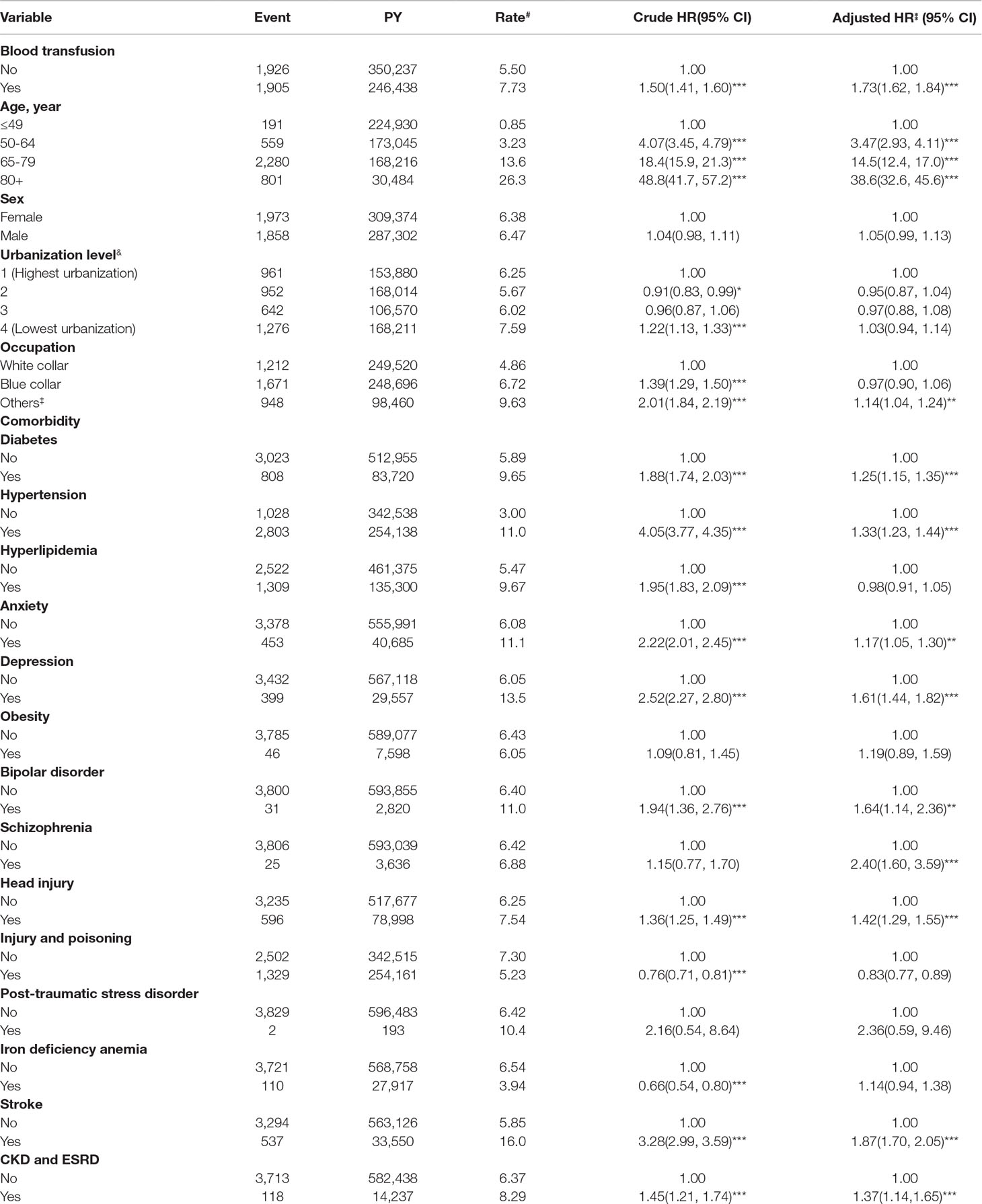
The overall incidence density rates of dementia were 5.50 and 7.73 per 1000 person-years in the matched and blood transfusion cohorts, respectively (Table 2). Compared with the matched cohort, the adjusted HRs were 1.73 (95% CI = 1.62–1.82) for the blood transfusion cohort. Compared with individuals aged ≤49 years, the risk of dementia was 3.47-fold higher in those aged between 50 and 64 years (95% CI = 2.93–4.11), 14.5-fold higher in those aged between 65 and 79 years (95% CI = 12.4–17.0), and 38.6-fold higher in those aged ≥80 years (95% CI 32.6–45.6). Patients employed in others had a higher risk of dementia than those employed in white collar jobs. The risk of dementia was higher in patients with comorbidities including diabetes, hypertension, anxiety, depression, bipolar disorder schizophrenia, head injury, stroke, and CKD and ESRD than those who did not. Individuals using BZDs had a 1.18-fold lower risk of dementia than those who did not (95% CI = 1.10–1.27). Individuals using non-BZDs had a 1.13-fold lower risk of dementia than those who did not (95% CI = 1.02–1.25). Individuals using antipsychotics had a 1.28-fold higher risk of dementia than those who did not (95% CI = 1.16–2.42).
Cox Model With Hazard Ratios and 95% Confidence Intervals of Alzheimer’s Disease Between the Blood Transfusion and Matched Cohorts
The overall incidence density rates of AD were 0.68 and 0.77 per 1000 person-years in the matched and blood transfusion cohorts, respectively (Table 3). Compared with the matched cohort, the HR of AD was 1.37 (95% CI = 1.13–1.66) in the blood transfusion cohort.
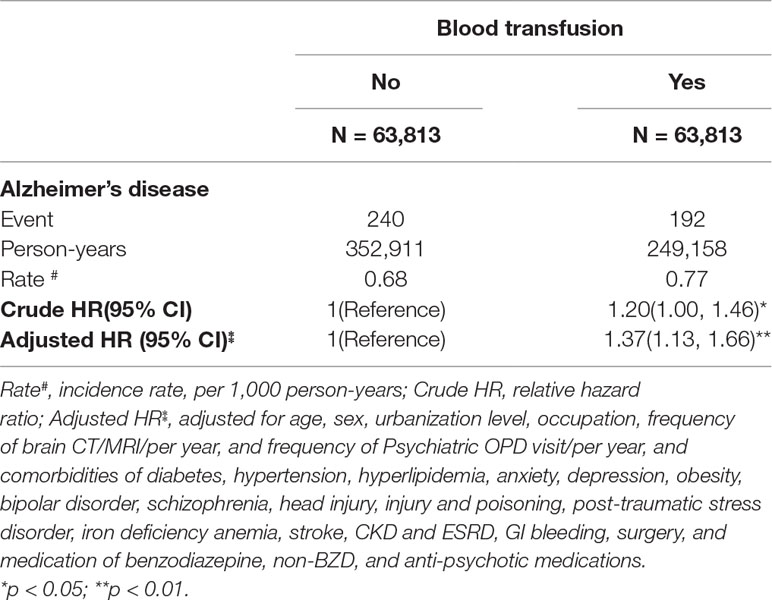
Table 3 Hazard ratio (HR) of Alzheimer’s disease for the blood transfusion cohort relative to the non-blood transfusion cohort.
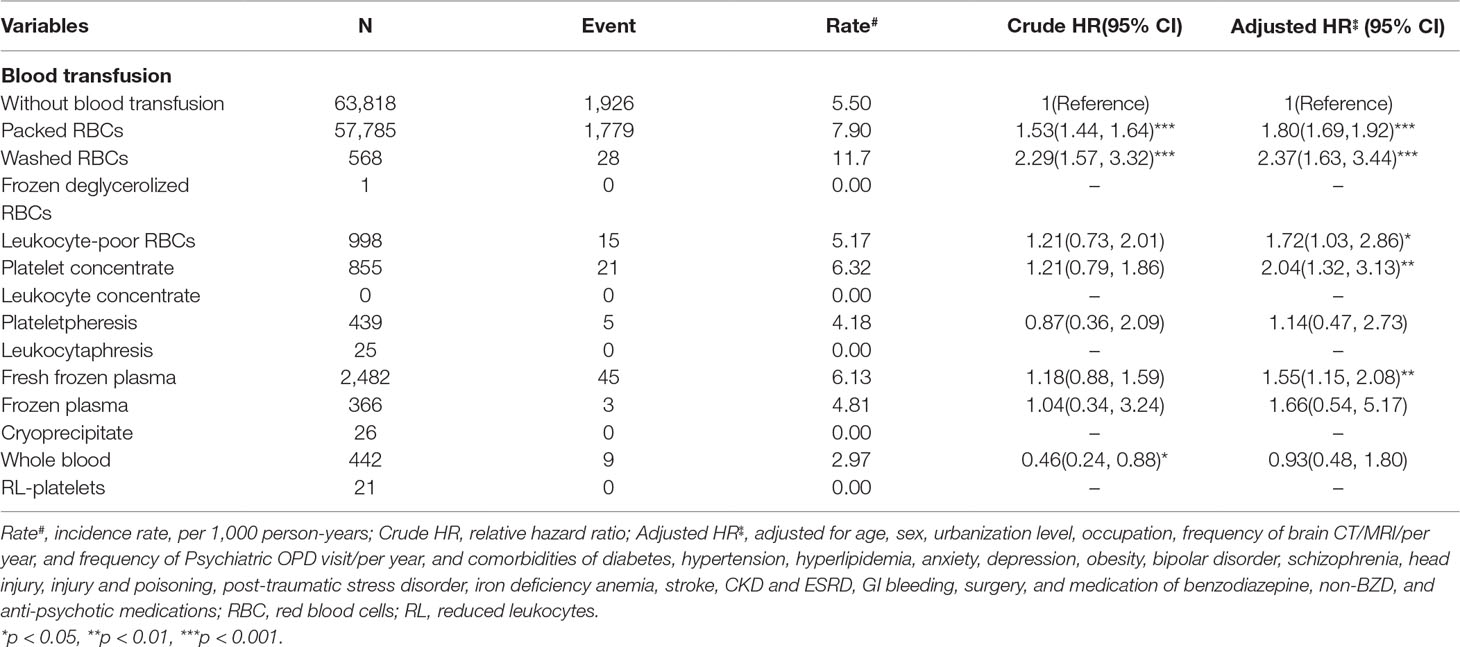
Incidence and HRs of Dementia Comparison Different Type Blood Transfusion and Matched Cohorts
Additionally, we analyzed the incidence and HR of dementia according to the different types of blood transfusions (Table 4). Patients who underwent packed RBCs transfusion, washed RBCs, Leukocyte-poor RBCs, platelet concentrate or FFP had a significantly higher risk of dementia than the matches.
Cumulative Incidence of Dementia and Dementia Event Risk According to Follow-Up Years in the Blood Transfusion and Matched Cohorts
The results indicated that at the end of the follow-up period, the cumulative incidence of dementia was higher in the blood transfusion cohort than in the matched cohort (log-rank test, p < 0.001) (Figure 1). The risk of dementia fell over time but persisted through the follow-up period (Table 5). The higher risk occurred during the first 3 y of the follow-up period (adjusted HR = 2.02, 95% CI = 1.74–2.35), and reduced with increasingly longer periods to 1.87 between 3 to 6 y of follow-ups. The risk remained for 6 y of follow-ups.
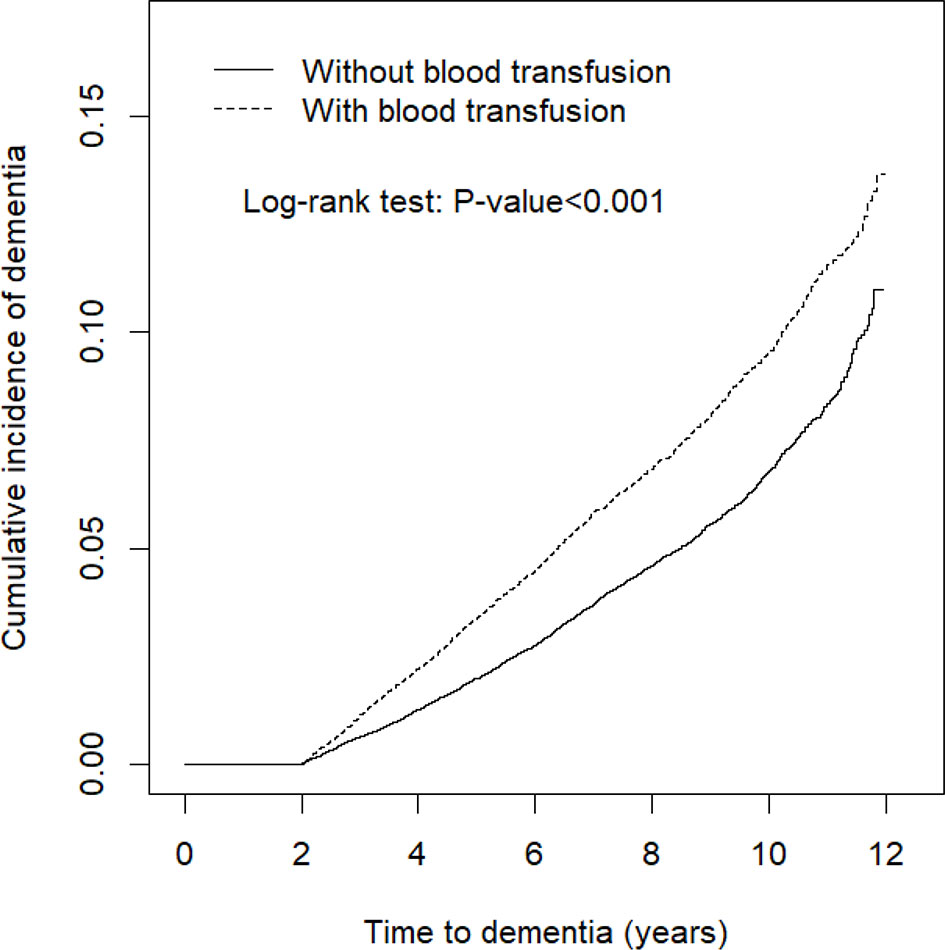
Figure 1 Cumulative incidence comparison of dementia for patients with (dashed line) or without (solid line) blood transfusion.
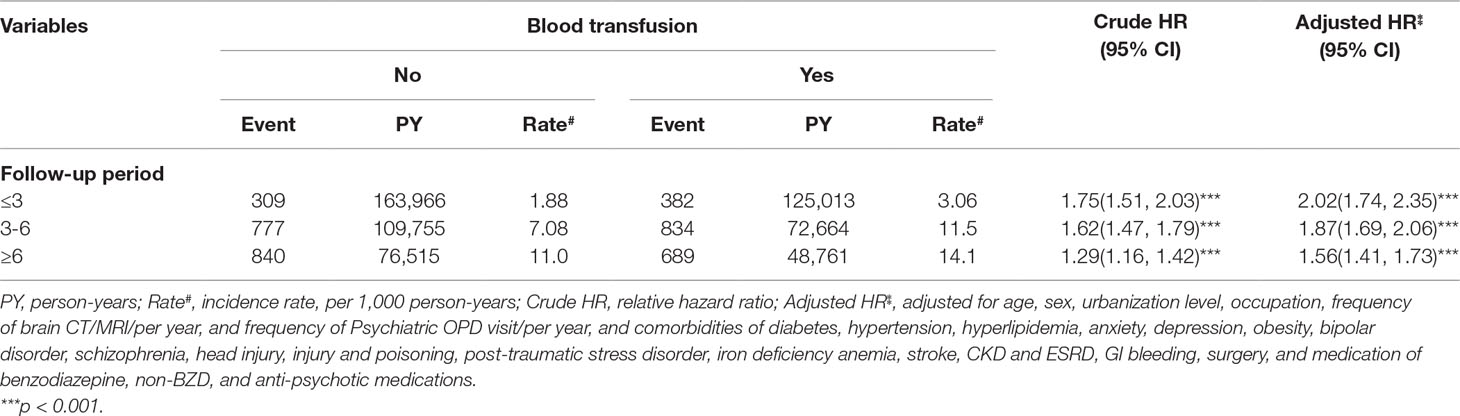
Table 5 Incidence of dementia by follow-up period and Cox model measured hazards ratio for patients with blood transfusions compared those without blood transfusions.
Relationship Between the Frequency of Blood Transfusion and the Risk of Dementia Development
The risk of dementia development was higher in patients presenting with 1, 2 or ≥3 blood transfusions compared with those in the matched cohort (p for trend <0.001) (Table 6).
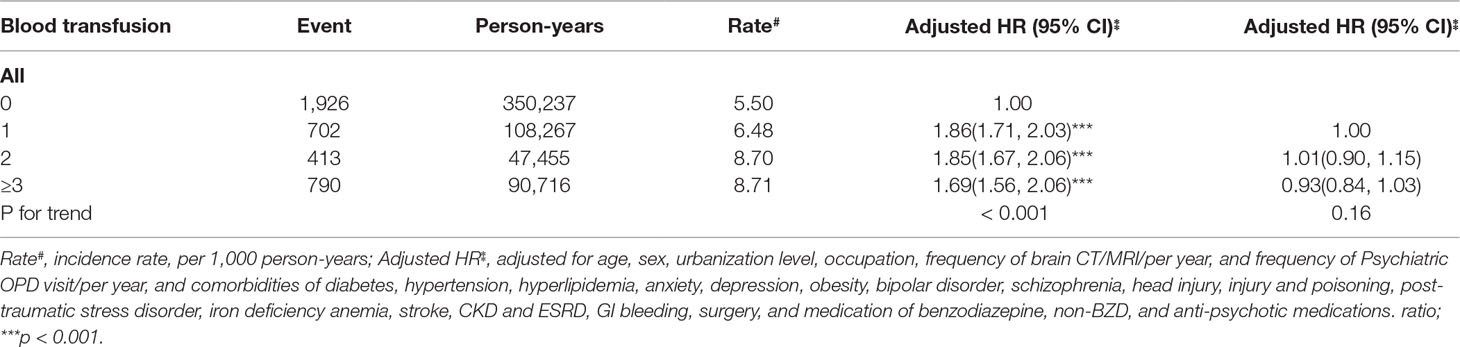
Table 6 The incidence rate and risk of dementia in patients with blood transfusions, stratified by frequency of blood transfusions.
Discussion
This study showed an association between blood transfusions and the risk of dementia. There are several possible explanations for our findings. First, transfusion might cause dysregulated iron metabolism in transfused patients. Lei et al. have also showed that Tau deficiency could induce parkinsonism with dementia by impairing the Amyloid precursor protein-mediated iron export (20). Duce et al. have found that iron-export ferroxidase activity of the β-amyloid precursor protein undergoes interference and is inhibited in AD (21). Transfusion has been linked with dysregulated iron metabolism in chronic transfused patients (14) and iron has been implicated in the pathology of many neurodegenerative diseases (36–39). Therefore, the dysregulated metabolism of iron as a result of transfusion might assist with the development of dementia in transfused patients. Second, transfusion-related inflammation might be involved in the pathogenesis of dementia. Patients who require transfusion are those with many comorbidities including diabetes mellitus, hypertension, chronic kidney disease, and stroke etc. It has been reported that transfusion could evoke inflammation and associated inflammatory cytokines. Cognasse et al. stated that microparticles originating from platelets, leukocytes, erythrocytes, and endothelial cells can trigger a pro-inflammatory message during transfusion (40). Chronic inflammation has been linked to neurodegenerative changes in the brain and the risks of dementia (41–44). Engelhart et al. also reported that increased levels of the plasma protein are associated with risks of dementia (45). Therefore, acute peripheral inflammation evoked during transfusion and chronic peripheral inflammation as a result of underlying comorbidities might both contribute to the increasing risk of dementia in transfused patients. Third, microparticles, e.g., exosomes or red blood cells in blood might play a role as carriers for molecules that may be related to the risk of dementia. Recent studies have demonstrated that neural-derived blood exosomes could predict or identify the development of dementia (46–49). Kapoginnias et al. identified that dysfunctional phosphorylated type 1 insulin receptor substrate in neural-derived blood exosomes could predict the development of dementia (48). This proposed mechanism might account for our findings that transfusion of different prepared blood products is associated with different risks of dementia. Transfusion of all types of RBC, platelet concentrate, and fresh frozen plasma were especially at risk of dementia. Further, Rieux et al. and Bu et al. have found that mutant huntingtin could be transferred via the bloodstream, cross the blood-brain-barrier and enter the brain parenchyma of mice (50, 51). However, since information about dementia of donors was unavailable, our study could not establish a direct relationship that neurodegenerative disease could be transmitted via blood transfusion. The mechanism remains unknown and future laboratory studied are needed to clarify the pathway.
Although both our study and the Edgren et al. study (33) investigated whether transfusion was associated with the risk of neurodegenerative disease, the two studies are not really comparable because the study designs were different. Several distinct differences in the present study and the Edgren et al. study should be mentioned here for future considerations and studies (33). First, Edgren et al. (33) reported that the risk of neurodegenerative diseases in recipients of blood transfusions from donors with a record of neurodegenerative disease was insignificant, while our study was an external comparison of the risk of neurodegenerative disease between recipients of blood from unknown donors and persons with no history of blood transfusion. Second, we have matched considerable dementia- associated comorbidities and medications, while Edgren et al. (33) considered age, gender, country, and diseases of donors. Chasse et al. investigated the association between blood donor age and sex with recipient survival (52). Chasse et al. considered many confounders in their study (52). From the study design of Chaase et al. (52), it is reasonable to surmise that confounders are critically important when investigating the consequences of blood transfusion.
Our data also showed that the risk of dementia was significantly higher, especially within 3 years following blood transfusion. Patients who require blood transfusion require more medical visits following the blood transfusion, thus providing more chances of finding dementia. Another supposed reason would be the competing risk that patients who require blood transfusions might have a shortened survival duration; thus, the risk of dementia seems decreased with years following transfusion.
Several limitations of this study should be mentioned. First, information on the apolipoprotein E genotype and the family history of each individual is unavailable. The influence of genetics could therefore not be evaluated in this study. Second, data on homocysteine levels (30), cholesterol levels, blood glucose levels, blood pressure, body mass index, smoking habits, alcohol consumption, sun exposure, and education levels, which are potential confounding factors of dementia, are unavailable in the NHIRD. However, to minimize the possible bias, we matched sex; occupation; associated comorbidities, including obesity, hypertension, diabetes, hyperlipidemia, and CKD (which is associated with a high homocysteine level); and medication associated with dementia. It should be noted that although we have utilized occupation as a proxy for educational level, occupation is not directly tightly associated with education level. Therefore, our study results showed that those with white collar jobs had a lower risk of dementia, which could not absolutely be caused by the higher level of education. Third, this study was conducted with registry data while dementia is often missed in registry data. Therefore, the events and risks of dementia might be underestimated. Fourth, the possibility of confounding through the indication of blood transfusion should also be considered. We have added gastrointestinal bleeding and surgery as the two circumstances that most warrant transfusion as variables to lessen the possible baseline bias. Fifth, information of variables including APOE, hippocampal volume or global atrophy or amnestic phenotype was unavailable in this study, which could potentially cause bias. Sixth, this is an external study and since those who need a transfusion might not be as likely as those who did not need a transfusion, a potential baseline bias could exist. However, we have done propensity matching to minimize this bias. Finally, whether blood donors had neurodegenerative diseases was unknown in this study. The medical follow up was short, which might not allow for enough time to assess for the incidence of AD or dementia cases in the young patient category. Further, each individual has a different post-transduction period and the number of doctor visits, in order to monitor his/her health status, was another limitation. The direct relationship between neurodegenerative diseases and blood transfusions could not be established in this study.
In conclusion, the results of the current study indicate that blood transfusions are associated with risks of dementia or AD. Transfusion is associated with the risk of dementia, regardless of transfusion frequency. Additional research is required to elucidate the biological mechanism underlying this association. This study promotes clinical alertness relating to the safety of blood transfusions, beyond previously known issues of hepatitis and HIV.
Data Availability
The dataset used in this study is held by the Taiwan Ministry of Health and Welfare (MOHW). The Ministry of Health and Welfare must approve the application to access this data. Any researcher interested in accessing this dataset can submit an application form to the Ministry of Health and Welfare requesting access and contact the staff of MOHW (Email:
Ethics Statement
The NHIRD encrypts patients’ personal information to protect privacy and provide researchers with anonymous identification numbers associated with relevant claims information, including sex, date of birth, medical services received, and prescriptions. Therefore, patient consent is not required to access the NHIRD. This study was approved to fulfill the condition for exemption by the Institutional Review Board (IRB) of China Medical University (CMUH-104-REC2-115-CR3). The IRB also specifically waived the consent requirement.
Author Contributions
S-YL and C-HK contributed to the conceptualization. Methodology, software, , and resources were handled by C-LL and C-HK. Validation, formal analysis, data curation, original draft preparation, review and editing, and visualization were handled by S-YL, C-LL, C-CL, H-CY, W-HH, and C-HK. Supervision, project administration, and funding acquisition were handled by C-HK.
Funding
This work was supported by grants from the Ministry of Health and Welfare, Taiwan (MOHW108-TDU-B-212-133004), the China Medical University Hospital (DMR-107-192); the Academia Sinica Stroke Biosignature Project (BM10701010021); the MOST Clinical Trial Consortium for Stroke (MOST 107-2321-B-039 -004-); the Tseng-Lien Lin Foundation, Taichung, Taiwan; and the Katsuzo and Kiyo Aoshima Memorial Funds, Japan. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript. No additional external funding was received for this study.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Abbreviations
CI, confidence interval; NHIRD, National Health Insurance Research Database; ICD-9-CM, International Classification of Diseases, Ninth Revision, Clinical Modification.
References
1. Patel KV. Epidemiology of anemia in older adults. In: Seminars in hematology: 2008. Bethesda, USA: Elsevier (2008). p. 210–7. doi: 10.1053/j.seminhematol.2008.06.006
2. Kassebaum NJ. The global burden of anemia. Hematol Oncol Clin (2016) 30(2):247–308. doi: 10.1016/j.hoc.2015.11.002
3. Kassebaum NJ, Jasrasaria R, Naghavi M, Wulf SK, Johns N, Lozano R, et al. A systematic analysis of global anemia burden from 1990 to 2010. Blood (2014) 123(5):615–24. doi: 10.1182/blood-2013-06-508325
4. Foley RN, Parfrey PS, Harnett JD, Kent GM, Murray DC, Barre PE. The impact of anemia on cardiomyopathy, morbidity, and mortality in end-stage renal disease. Am J Kidney Dis (1996) 28(1):53–61. doi: 10.1016/S0272-6386(96)90130-4
5. Madjdpour C, Spahn DR, Weiskopf RB. Anemia and perioperative red blood cell transfusion: a matter of tolerance. Crit Care Med (2006) 34(5):S102–8. doi: 10.1097/01.CCM.0000214317.26717.73
6. Shapiro MJ, Gettinger A, Corwin HL, Napolitano L, Levy M, Abraham E, et al. Anemia and blood transfusion in trauma patients admitted to the intensive care unit. J Trauma and Acute Care Surg (2003) 55(2):269–74. doi: 10.1097/01.TA.0000080530.77566.04
7. Smilowitz NR, Oberweis BS, Nukala S, Rosenberg A, Zhao S, Xu J, et al. Association between anemia, bleeding, and transfusion with long-term mortality following noncardiac surgery. Am J Med (2016) 129(3):315–323. e312. doi: 10.1016/j.amjmed.2015.10.012
8. Spinelli E, Bartlett RH. Anemia and transfusion in critical care: physiology and management. J Intensive Care Med (2016) 31(5):295–306. doi: 10.1177/0885066615571901
9. Jy W, Ricci M, Shariatmadar S, Gomez-Marin O, Horstman LH, Ahn YS. Microparticles in stored red blood cells as potential mediators of transfusion complications. Transfusion (2011) 51(4):886–93. doi: 10.1111/j.1537-2995.2011.03099.x
10. Slonim AD, Joseph JG, Turenne WM, Sharangpani A, Luban NL. Blood transfusions in children: a multi-institutional analysis of practices and complications. Transfusion (2008) 48(1):73–80.
11. Perrotta P, Snyder E. Non-infectious complications of transfusion therapy. Blood Rrev (2001) 15(2):69–83. doi: 10.1054/blre.2001.0151
12. Brittenham GM. Iron-chelating therapy for transfusional iron overload. N E J Med (2011) 364(2):146–56. doi: 10.1056/NEJMct1004810
13. Porter JB, de Witte T, Cappellini MD, Gattermann N. New insights into transfusion-related iron toxicity: implications for the oncologist. Crit Rev Oncol Hematol (2016) 99:261–71. doi: 10.1016/j.critrevonc.2015.11.017
14. Harmatz P, Butensky E, Quirolo K, Williams R, Ferrell L, Moyer T, et al. Severity of iron overload in patients with sickle cell disease receiving chronic red blood cell transfusion therapy. Blood (2000) 96(1):76–9. doi: 10.1046/j.1423-0410.2000.79402651.x
15. Dandona P, Hussain M, Varghese Z, Politis D, Flynn D, Hoffbrand A. Insulin resistance and iron overload. Ann Clin Biochem (1983) 20(2):77–9. doi: 10.1177/000456328302000203
16. Ozment CP, Turi JL. Iron overload following red blood cell transfusion and its impact on disease severity. BBA-Gen Subjects (2009) 1790(7):694–701. doi: 10.1016/j.bbagen.2008.09.010
17. Nicolas G, Chauvet C, Viatte L, Danan JL, Bigard X, Devaux I, et al. The gene encoding the iron regulatory peptide hepcidin is regulated by anemia, hypoxia, and inflammation. J Clin Invest (2002) 110(7):1037–44. doi: 10.1172/JCI15686
18. Pichler I, Fabiola Del Greco M, Gögele M, Lill CM, Bertram L, Do CB, et al. Serum iron levels and the risk of Parkinson disease: a mendelian randomization study. PLoS Med (2013) 10(6):e1001462. doi: 10.1371/journal.pmed.1001462
19. Zecca L, Youdim MB, Riederer P, Connor JR, Crichton RR. Iron, brain ageing and neurodegenerative disorders. Nat Rev Neurosci (2004) 5(11):863–73. doi: 10.1038/nrn1537
20. Lei P, Ayton S, Finkelstein DI, Spoerri L, Ciccotosto GD, Wright DK, et al. Tau deficiency induces parkinsonism with dementia by impairing APP-mediated iron export. Nat Med (2012) 18(2):291. doi: 10.1038/nm.2613
21. Duce JA, Tsatsanis A, Cater MA, James SA, Robb E, Wikhe K, et al. Iron-export ferroxidase activity of beta-amyloid precursor protein is inhibited by zinc in Alzheimer’s disease. Cell (2010) 142(6):857–67. doi: 10.1016/j.cell.2010.08.014
22. Masaldan S, Bush AI, Devos D, Rolland AS, Moreau C. Striking while the iron is hot: iron metabolism and ferroptosis in neurodegeneration. Free Radic Biol Med (2019) 133:221–33. doi: 10.1016/j.freeradbiomed.2018.09.033
23. Hod EA, Zhang N, Sokol SA, Wojczyk BS, Francis RO, Ansaldi D, et al. Transfusion of red blood cells after prolonged storage produces harmful effects that are mediated by iron and inflammation. Blood (2010) 115(21):4284–92. doi: 10.1182/blood-2009-10-245001
24. Glass CK, Saijo K, Winner B, Marchetto MC, Gage FH. Mechanisms underlying inflammation in neurodegeneration. Cell (2010) 140(6):918–34. doi: 10.1016/j.cell.2010.02.016
25. Knowles TP, Vendruscolo M, Dobson CM. The amyloid state and its association with protein misfolding diseases. Nat Rev Mol Cell Biol (2014) 15(6):384–96. doi: 10.1038/nrm3810
26. Brettschneider J, Del Tredici K, Lee VM, Trojanowski JQ. Spreading of pathology in neurodegenerative diseases: a focus on human studies. Nat Rev Neurosci (2015) 16(2):109–20. doi: 10.1038/nrn3887
27. Gupta A, Watkins A, Thomas P, Majer R, Habubi N, Morris G, et al. Coagulation and inflammatory markers in Alzheimer’s and vascular dementia. Int J Clin Pract (2005) 59(1):52–7. doi: 10.1111/j.1742-1241.2004.00143.x
28. Breteler MM. Vascular involvement in cognitive decline and dementia. Epidemiologic evidence from the Rotterdam Study and the Rotterdam Scan Study. Ann N Y Acad Sci (2000) 903:457–65. doi: 10.1111/j.1749-6632.2000.tb06399.x
29. Brandner S, Klein MA, Frigg R, Pekarik V, Parizek P, Raeber A, et al. Neuroinvasion of prions: insights from mouse models. Exp Physiol (2000) 85(6):705–12. doi: 10.1111/j.1469-445X.2000.02091.x
30. Eisele YS, Bolmont T, Heikenwalder M, Langer F, Jacobson LH, Yan Z-X, et al. Induction of cerebral β-amyloidosis: intracerebral versus systemic Aβ inoculation. Proc Natl Acad Sci (2009) 106(31):12926–31. doi: 10.1073/pnas.0903200106
31. Downes KA, Wilson E, Yomtovian R, Sarode R. Serial measurement of clotting factors in thawed plasma stored for 5 days. Transfusion (2001) 41(4):570. doi: 10.1046/j.1537-2995.2001.41040570.x
32. Bohnen NI, Warner MA, Kokmen E, Beard CM, Kurland LT. Prior blood transfusions and Alzheimer’s disease. Neurology (1994) 44(6):1159–60. doi: 10.1212/WNL.44.6.1159
33. Edgren G, Hjalgrim H, Rostgaard K, Lambert P, Wikman A, Norda R, et al. Transmission of neurodegenerative disorders through blood transfusion: a cohort study. Ann Intern Med (2016) 165(5):316–24. doi: 10.7326/M15-2421
34. Wang JC, Sung FC, Men M, Wang KA, Lin CL, Kao CH. Bidirectional association between fibromyalgia and gastroesophageal reflux disease: two population-based retrospective cohort analysis. Pain (2017) 158(10):1971–8. doi: 10.1097/j.pain.0000000000000994
35. Hsieh CY, Su CC, Shao SC, Sun SF, Lin SJ, Yang YHK, et al. Taiwan’s National Health Insurance Research Database: past and future. Clin Epi (2019) 11:349–58. doi: 10.2147/CLEP.S196293
36. Eid R, Arab NTT, Greenwood MT. Iron mediated toxicity and programmed cell death: a review and a re-examination of existing paradigms. BBA-Mol Cell Res (2017) 1864(2):399–430. doi: 10.1016/j.bbamcr.2016.12.002
37. Mena NP, Urrutia PJ, Lourido F, Carrasco CM, Nunez MT. Mitochondrial iron homeostasis and its dysfunctions in neurodegenerative disorders. Mitochondrion (2015) 21:92–105. doi: 10.1016/j.mito.2015.02.001
38. Belaidi AA, Bush AI. Iron neurochemistry in Alzheimer’s disease and Parkinson’s disease: targets for therapeutics. J Neurochem (2016) 139:179–97. doi: 10.1111/jnc.13425
39. Andersen HH, Johnsen KB, Moos T. Iron deposits in the chronically inflamed central nervous system and contributes to neurodegeneration. Cell Mol Life Sci (2014) 71(9):1607–22. doi: 10.1007/s00018-013-1509-8
40. Cognasse F, Hamzeh-Cognasse H, Laradi S, Chou M-L, Seghatchian J, Burnouf T, et al. The role of microparticles in inflammation and transfusion: a concise review. Transfus Apher Sci (2015) 53(2):159–67. doi: 10.1016/j.transci.2015.10.013
41. Leonard BE. Inflammation, depression and dementia: are they connected? Neurochem Res (2007) 32(10):1749–56. doi: 10.1007/s11064-007-9385-y
42. Schmidt R, Schmidt H, Curb JD, Masaki K, White LR, Launer LJ. Early inflammation and dementia: a 25-year follow-up of the Honolulu-Asia aging study. Ann Neurol (2002) 52(2):168–74. doi: 10.1002/ana.10265
43. Ravaglia G, Forti P, Maioli F, Chiappelli M, Montesi F, Tumini E, et al. Blood inflammatory markers and risk of dementia: the conselice study of brain aging. Neurobiol Aging (2007) 28(12):1810–20. doi: 10.1016/j.neurobiolaging.2006.08.012
44. De Luigi A, Fragiacomo C, Lucca U, Quadri P, Tettamanti M, De Simoni MG. Inflammatory markers in Alzheimer’s disease and multi-infarct dementia. Mech Ageing Dev (2001) 122(16):1985–95. doi: 10.1016/S0047-6374(01)00313-X
45. Engelhart MJ, Geerlings MI, Meijer J, Kiliaan A, Ruitenberg A, van Swieten JC, et al. Inflammatory proteins in plasma and the risk of dementia: the Rotterdam study. Arch Neurol (2004) 61(5):668–72. doi: 10.1001/archneur.61.5.668
46. Fiandaca MS, Kapogiannis D, Mapstone M, Boxer A, Eitan E, Schwartz JB, et al. Identification of preclinical Alzheimer’s disease by a profile of pathogenic proteins in neurally derived blood exosomes: a case-control study. Alzheimers Dement (2015) 11(6):600–607. e601. doi: 10.1016/j.jalz.2014.06.008
47. Winston CN, Goetzl EJ, Akers JC, Carter BS, Rockenstein EM, Galasko D, et al. Prediction of conversion from mild cognitive impairment to dementia with neuronally derived blood exosome protein profile. Alzheimers Dement (2016) 3:63–72. doi: 10.1016/j.dadm.2016.04.001
48. Kapogiannis D, Boxer A, Schwartz JB, Abner EL, Biragyn A, Masharani U, et al. Dysfunctionally phosphorylated type 1 insulin receptor substrate in neural-derived blood exosomes of preclinical Alzheimer’s disease. FASEB J (2014) 29(2):589–96. doi: 10.1096/fj.14-262048
49. Sun B, Dalvi P, Abadjian L, Tang N, Pulliam L. Blood neuron-derived exosomes as biomarkers of cognitive impairment in HIV. AIDS (2017) 31(14):F9–F17. doi: 10.1097/QAD.0000000000001595
50. Rieux M, Alpaugh M, Cicchetti F. The troubling story of blood-driven dementias. Nature Publishing Group (2019). doi: 10.1038/s41380-018-0225-z
51. Bu X, Xiang Y, Jin W, Wang J, Shen L, Huang Z, et al. Blood-derived amyloid-β protein induces Alzheimer’s disease pathologies. Mol Psychiatry (2018) 23(9):1. doi: 10.1038/mp.2017.204
Keywords: transfusion, dementia, Alzheimer’s disease, cohort study, Taiwan National Health Insurance Research Database
Citation: Lin S-Y, Hsu W-H, Lin C-C, Lin C-L, Yeh H-C and Kao C-H (2019) Association of Transfusion With Risks of Dementia or Alzheimer’s Disease: A Population-Based Cohort Study. Front. Psychiatry 10:571. doi: 10.3389/fpsyt.2019.00571
Received: 22 February 2019; Accepted: 22 July 2019;
Published: 16 August 2019.
Edited by:
Claudio Soto, University of Texas Health Science Center at Houston, United StatesReviewed by:
David Devos, Université de Lille, FranceRodrigo Morales, University of Texas Health Science Center at Houston, United States
Copyright © 2019 Lin, Hsu, Lin, Lin, Yeh and Kao. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Chia-Hung Kao, ZDEwMDQwQG1haWwuY211aC5vcmcudHc=
 Shih-Yi Lin1,2
Shih-Yi Lin1,2 Cheng-Li Lin
Cheng-Li Lin Chia-Hung Kao
Chia-Hung Kao