- 1Institute for Biomarker Research, Medical Diagnostic Laboratories, Genesis Biotechnology Group, Hamilton, NJ, United States
- 2Memorial Women’s Care, Houston, TX, United States
Background: Numerous bacteria are involved in the etiology of bacterial vaginosis (BV). Yet, current tests only focus on a select few. We therefore designed a new test targeting 22 BV-relevant species.
Methods: Using 946 stored vaginal samples, a new qPCR test that quantitatively identifies 22 bacterial species was designed. The distribution and relative abundance of each species, α- and β-diversities, correlation, and species co-existence were determined per sample. A diagnostic index was modeled from the data, trained, and tested to classify samples into BV-positive, BV-negative, or transitional BV.
Results: The qPCR test identified all 22 targeted species with 95 – 100% sensitivity and specificity within 8 hours (from sample reception). Across most samples, Lactobacillus iners, Lactobacillus crispatus, Lactobacillus jensenii, Gardnerella vaginalis, Fannyhessea (Atopobium) vaginae, Prevotella bivia, and Megasphaera sp. type 1 were relatively abundant. BVAB-1 was more abundant and distributed than BVAB-2 and BVAB-3. No Mycoplasma genitalium was found. The inter-sample similarity was very low, and correlations existed between key species, which were used to model, train, and test a diagnostic index: MDL-BV index. The MDL-BV index, using both species and relative abundance markers, classified samples into three vaginal microbiome states. Testing this index on our samples, 491 were BV-positive, 318 were BV-negative, and 137 were transitional BV. Although important differences in BV status were observed between different age groups, races, and pregnancy status, they were statistically insignificant.
Conclusion: Using a diverse and large number of vaginal samples from different races and age groups, including pregnant women, the new qRT-PCR test and MDL-BV index efficiently diagnosed BV within 8 hours (from sample reception), using 22 BV-associated species.
1 Introduction
Bacterial Vaginosis (BV), a condition in which the normal Lactobacillus-rich vaginal microbiome becomes dominated by polymicrobial anaerobic bacterial species under non-acidic pH, remains the most common cause of abnormal vaginal discharge in reproductive-age women, with an estimated prevalence rate of 29% in North America (Abou Chacra et al., 2021; Elnaggar et al., 2023). The normal vaginal microbiome is dominated by three major Lactobacillus species, L. crispatus, L. jensenii, and L. gasseri, with low abundance of L. acidophilus, which protects the vagina by producing lactic acid, hydrogen peroxide, and bacteriocins that suppress bacterial growth (Pacha-Herrera et al., 2022; Wu et al., 2022). Lactobacillus iners, however, is quite enigmatic as it occurs in both healthy and unhealthy vaginal environments; L. iners, unlike L. crispatus, L. jensenii, and L. gasseri, produces the human non-functional L form of lactic acid (Basavaprabhu et al., 2020).
To produce lactic acid, which is necessary to maintain an acidic pH of < 4.5, Lactobacillus sp. uses glycogen deposited unto the vaginal walls from the epithelial cells (Shen et al., 2022; Navarro et al., 2023). The acidic pH keeps away most microbial species and allows Lactobacillus sp. to proliferate, keeping the species diversity in the normal vaginal microbiota low. Lactic acid is metabolized from glycogen, whose production and deposition are mediated by circulating estrogen in a dose-dependent manner. In the absence of glycogen or lactic-acid–producing species, the pH rises, allowing other microbes to proliferate, colonize the vagina, and increase the microbiota diversity. Hence, physiological factors disrupting or increasing estrogen levels indirectly affect Lactobacillus sp. and BV-associated bacterial species’ prevalence (Shen et al., 2022; Navarro et al., 2023). The etiology and pathogenesis of BV are still not fully understood, making it important to study the involved microbes for timely diagnosis and treatment. Particularly, the interactions between species within the BV microbiome that cause pathologies, treatment failure and recurrence, or healing need further interrogations to enable a species biomarker-based diagnosis of BV.
The colonization of the vaginal microbiota by anaerobic bacterial and fungal species causes BV or vulvovaginal candidiasis, respectively, characterized by increased vaginal discharge with a fishy odor (Swidsinski et al., 2023). Dysbiosis also predisposes the vagina to sequelae of other sexually transmitted infections (STIs) and obstetric disorders. Although BV is asymptomatic in many women, it is associated with the development of common obstetric and gynecologic complications (Javed et al., 2019; Cheng et al., 2020; Gustin et al., 2021). It also increases the risk of contracting HIV and other STIs and pelvic inflammatory disease (PID) (Unemo et al., 2017). Notably, BV recurrence after treatment is common (estimated to affect 50% of women annually) (Hilbert et al., 2016; Coudray and Madhivanan, 2020; Coudray et al., 2020) and may be due to re-infections from sexual partners or failure of current treatment options (Coudray et al., 2020; Vodstrcil et al., 2021). Besides the health impacts of recurrence, BV treatment costs are increasing annually, specifically in the USA: this economic impact is particularly pronounced in BV-associated preterm births and other obstetric complications (Peebles et al., 2019; Watkins et al., 2024).
Species such as Gardnerella vaginalis, Fannyhessea vaginae, Prevotella bivia, Megasphaera sp., and BVAB are commonly implicated in the etiology of BV, with G. vaginalis and F. vaginae being common in most BV infections (Swidsinski et al., 2023) as sessiles. Swidsinksi et al. (2023) observed that vaginal epithelial cells from females with BV were covered with G. vaginalis biofilms that encapsulated other species, forming a polymicrobial biofilm, as well as by non-adherent planktonic bacterial cells. The biofilms associated with BV explain their ability to escape both the immune response and antimicrobial chemotherapy as biofilms prevent the immune cells and antimicrobial agents from reaching the pathogens (Cangui-Panchi et al., 2023; Swidsinski et al., 2023). This leads to high treatment failure and recurrence rates of about 50% (Muñoz-Barreno et al., 2021); indeed, the ability of these biofilm-coated polymicrobial species to spread into the uterus and fallopian tube explains their resistance to antimicrobials and the immune response (Swidsinski et al., 2023).
To diagnose BV, clinicians rely mostly on the classical clinical signs and symptoms outlined in Amsel’s criteria (Amsel et al., 1983) or on the microscopically based Nugent score (Nugent et al., 1991). While these standard diagnostic methods have been effective over the years, they have been confronted with limitations such as being labor-intensive and time-consuming, subjective and unable to accurately identify pathogens (Muzny et al., 2023). As recently discussed by Muzny et al. (2023), nucleic acid tests ease the burden of laboriously going through the clinical testing criteria, saving clinicians time and effort for other duties. The development of culture-independent molecular diagnostics has enabled the detection of non-cultivable bacterial species associated with BV and continues to revolutionize infectious disease diagnosis (Hilbert et al., 2016; van den Munckhof et al., 2019; Bhujel et al., 2021). Molecular tests such as real-time polymerase chain reaction (RT-PCR), combined with the traditional clinical diagnostic criteria, have greatly improved the sensitivity and specificity of detecting BV pathogens (Hilbert et al., 2016; van den Munckhof et al., 2019; Bhujel et al., 2021). They are also applicable in monitoring patient response to antibiotic therapy (Onderdonk et al., 2016; Elnaggar et al., 2023). This approach has proven more useful for identifying patients at risk for recurrent BV (Jones, 2019; Coudray and Madhivanan, 2020; Vodstrcil et al., 2021; Wu et al., 2022; Sobel and Vempati, 2024; Watkins et al., 2024).
Besides next-generation sequencing (NGS)-based metagenomics that target all genomes, current PCR diagnostics for BV mainly focus on a smaller spectrum of bacterial species. Hence, to increase the resolution and diagnostic power of PCR-based BV diagnostics (Balashov et al., 2014), we designed a new 22-species quantitative Real-Time PCR assay (qRT-PCR) that quantitatively detects 22 vaginal bacterial species within 8 hours (from sample reception). This newly designed proprietary qRT-PCR assay (Bacterial Vaginosis (with Lactobacillus Profiling) Panel®, Medical Diagnostic Laboratories, L.L.C. (MDL), New Jersey, USA) was used to screen 946 vaginal samples routinely obtained from different health centers across the United States. We further used the results, with assistance from machine-learning algorithms (Decision Trees and Random Forests) to design, train, and test an index (herein termed the MDL-BV index) that used a relative-abundance and species-based markers to classify the vaginal microbiome in three categories: BV-negative (healthy microbiome), transitional BV (between healthy and unhealthy vaginal microbiome), and BV-positive (abnormal microbiome).
2 Materials and methods
2.1 Specimen collection and processing
Clinical vaginal samples are routinely obtained from different healthcare centers across the United States for diagnostic processing at MDL. Historical vaginal specimens (n = 946) marked for disposal were received in OneSwab® (Copan Diagnostics, CA, USA) or ThinPrep® (Hologic, MA, USA) transport media in a Clinical Laboratory Improvement Amendments (CLIA)-certified infectious disease laboratory facility between January and June 2023 and stored at −80°C were selected randomly for this study. This included specimens from symptomatic, asymptomatic, pregnant, or non-pregnant females and from whom vaginal profiling was requested. For each biological specimen that arrives at the laboratory facility, specimen accessioning occurs that assigns a random MDL number to ensure the de-identification of specimens. To further the de-identification of specimens during this study, samples that matched our collection criteria were randomized and the MDL numbers associated with each sample were not recorded.
2.2 Targeted bacterial species
The newly designed quantitative real-time PCR (qRT-PCR) assay (Bacterial Vaginosis (with Lactobacillus Profiling) Panel®) is a BV diagnostic assay designed by MDL to qualitatively and quantitatively detect 22 bacterial species that are found in the eubiotic and dysbiotic vaginal microbiome. These species are L. crispatus, L. jensenii, L. gasseri, L. iners, L. acidophilus, Gardnerella vaginalis, Fannyhessea vaginae (Atopobium vaginae), Megasphaera sp. types 1 and 2, Prevotella bivia, Bacterial Vaginosis-Associated Bacterium (BVAB) 1–3, Ureaplasma urealyticum, Mycoplasma hominis, Mycoplasma genitalium, Mobiluncus curtisii, Mobiluncus mulieris, Sneathia sanguinegens, Bifidobacterium breve, Bacteroides fragilis, and Streptococcus anginosus (Onderdonk et al., 2016; Muzny et al., 2020; Mondal et al., 2023; Powell et al., 2023).
2.3 DNA preparation
DNA from the vaginal specimens was extracted according to validated in-house laboratory protocols using QIAamp® DNA Mini Kit (QIAGEN, Maryland, USA) and the X-tractor Gene® DNA workstation (QIAGEN, Maryland, USA) with slight modifications. Canine Herpes Virus DNA was spiked into the samples and subsequently detected as an internal extraction control.
To serve as amplification standards, plasmids for each of the 22 bacterial species were generated by whole-gene synthesis (Genewiz®, Azenta Life Sciences, Waltham, USA). Briefly, a species-specific region of each species was amplified, synthesized, and cloned into the pUC-GW-AMP plasmid vector with an ampicillin-resistance marker. These plasmids were transformed into chemically competent Escherichia coli cells (One Shot™ TOP10, ThermoFisher Scientific, New Jersey, USA) and grown in liquid Lysogeny Broth (LB) containing 50 µg/mL Ampicillin. Plasmid DNA was isolated from overnight cultures using Wizard® Plus SV Minipreps DNA Purification Systems (Promega, Wisconsin, USA) according to the manufacturer’s protocol. Extracted DNA was subsequently quantified spectrophotometrically using NanoDrop 1000 equipment (ThermoFisher Scientific, New Jersey, USA). Standard 10-fold serial dilutions of 108 to 101 DNA copies/µL were prepared for each bacterial species.
2.4 Identification of bacterial species by multiplex qRT-PCR
Each vaginal and plasmid control DNA sample was amplified in triplicates using species-specific primers and fluorescent probes on a CFX384 Touch Real-Time PCR Detection System (Bio-Rad, California, USA). The specific primer and probe sequences, as well as the multiplex RT-PCR conditions in the Bacterial Vaginosis (with Lactobacillus Profiling) Panel by Real-Time PCR assay are proprietary to MDL. An aliquot of 0.5μL of DNA was used as the template in a 4μL total PCR reaction mixture containing four primer sets, four probes, and the in-house—prepared mastermix of Taq DNA polymerase and dNTPs (MDL, New Jersey, USA) in multiplex PCR reactions. Human β-globulin DNA served as the internal control while no-template control was included to account for potential extraneous nucleic acid contamination. After amplification, a standard curve (fluorescence vs cycle number) was generated for each species and the target DNA copy for each sample was extrapolated. A 102 copies/μL concentration in each sample was established as the positive cutoff for a given bacterium. The relative percentage concentration of all the bacterial species identified in each vaginal specimen was then computed and tabulated (Supplementary Table S1).
2.5 Relative abundance- and species-based classification of BV
The mean, median, standard deviation, and concentration distribution of each species across samples as well as alpha-diversity, Shannon index, beta-diversity (Bray-Curtis Dissimilarity matrix), and relative abundance were calculated from the gDNA concentration data (obtained from the qRT-PCR amplification results of each species from the samples) and represented as box and Whisker plots, histograms, heatmaps, and charts. Using the distribution patterns in the data, a relative abundance or concentration-based cut-off criteria was established and used to define BV-positive (abnormal vaginal microbiota with bacterial vaginosis and depleted Lactobacillus sp.: ≤ 40% relative abundance), BV-negative (i.e., normal vaginal microbiota with ≥ 70% Lactobacillus sp. relative abundance), and transitional BV (transition between negative and positive BV microbiota with 30–50% Lactobacillus sp. relative abundance) status (see Table 1 for full definitions). Using the species-species correlation and co-existence data as well as previous research from literature, a species-based criteria was also established for determining BV-positive, BV-negative, and transitional BV status. A two-tier system was then created by synthesizing the species-based and concentration-based BV diagnosis criteria, which were used to interpret the PCR results. This criterion was called the MDL-BV index, which was further trained and tested on the qRT-PCR and demographics data using machine-learning algorithms (Decision Trees and Random Forests) in Python. The datasets were divided into two: one for training and one for testing. Python libraries such as numpy, pandas, scikitlearn, XGBoost, and matplotlib, were used for analysis and visualization.
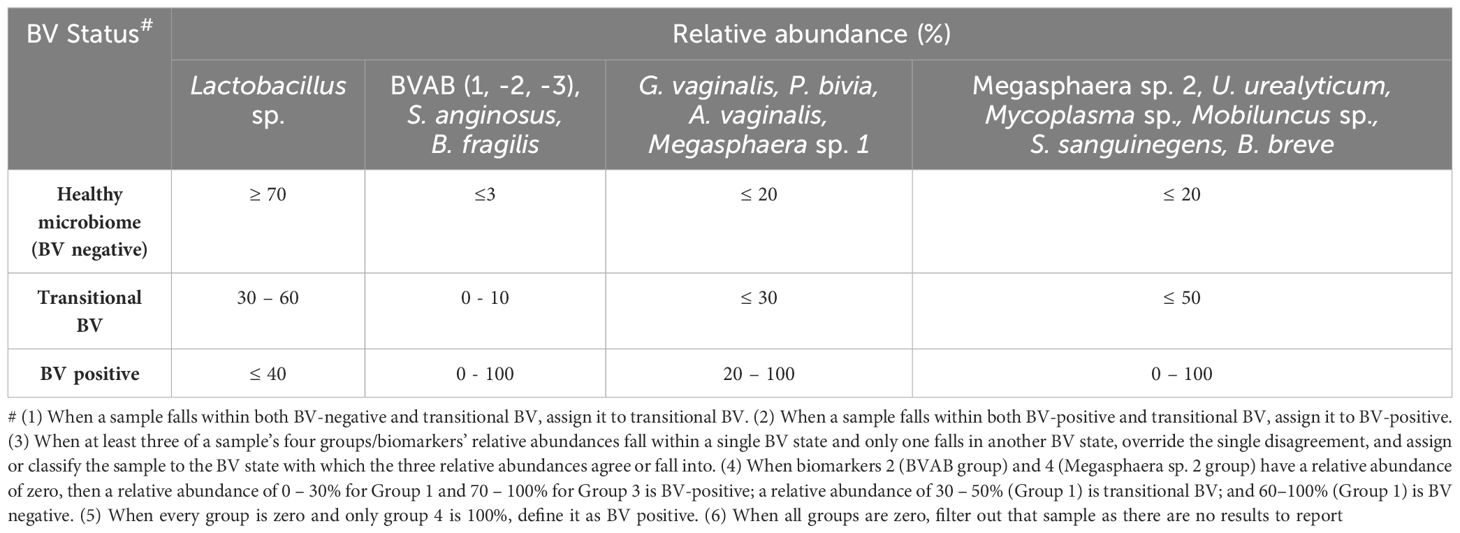
Table 1 MDL-BV index designed to interpret the new 22 species qRT-PCR results and diagnose vaginal samples as BV-positive, BV-negative, or transitional BV.
2.6 Demographics
The age, race, and pregnancy status of the females from whom the samples were taken were collected and used to stratify the data to compare the different rates, distributions, and relative abundance of the various species, BV-positive, BV-negative, and transitional BV according to age, race, and pregnancy status.
2.7 Statistical analysis
Statistical analyses were performed with Prism version 10.1.2 (Graph-Pad, California USA) or the R statistical package. Quantitative data obtained in this study were expressed as the mean ± standard error of the mean (SEM). To detect bacterial associations and correlations, Pearson’s correlation coefficient was chosen. The Kruskal–Wallis test was employed to determine any statistically significant differences in age distributions among BV classification groups. The data were parsed through Python (and Biopython) to calculate the significance of the coexistence between any two species using Chi-square and T-test.
Three types of groupings were used in this (Chi-square and T-test) analysis: (1) all Lactobacillus species were grouped into one and their absence/presence was compared with the absence/presence of each non-Lactobacillus species; (2) all Lactobacillus species except L. iners were grouped into one and their absence/presence was compared with the absence/presence of each non-Lactobacillus species; (3) the presence/absence of each Lactobacillus species was compared with that of each non-Lactobacillus species. The resulting data were tabulated in Supplementary Table S2, and the significant results were filtered out and used to generate heat maps. The results were only considered significant if P < 0.05.
3 Results
3.1 Demographics
The vaginal samples were obtained from 946 women whose average age was 34.8 years, with a standard deviation of 13.3 years. The ages ranged from 18 to 84 years. The median age was 32 years; hence, the age distribution is somewhat skewed towards younger women [Figure 1; Supplementary Table S1 (Demographics)]. Fifty-three women reported as pregnant while the rest were either not pregnant or unknown. Race data was obtained for 245 samples: White (n = 145 samples), Black (n = 79), “Other race” (n = 15), Asian (n = 4), and Native American (n = 2) [Figure 1; Supplementary Table S1 (Demographics)]. The “Other race” category refers to those not falling within any of the four races above: Hispanic/Latino, Native Hawaiian or Pacific Islander, and mixed races.
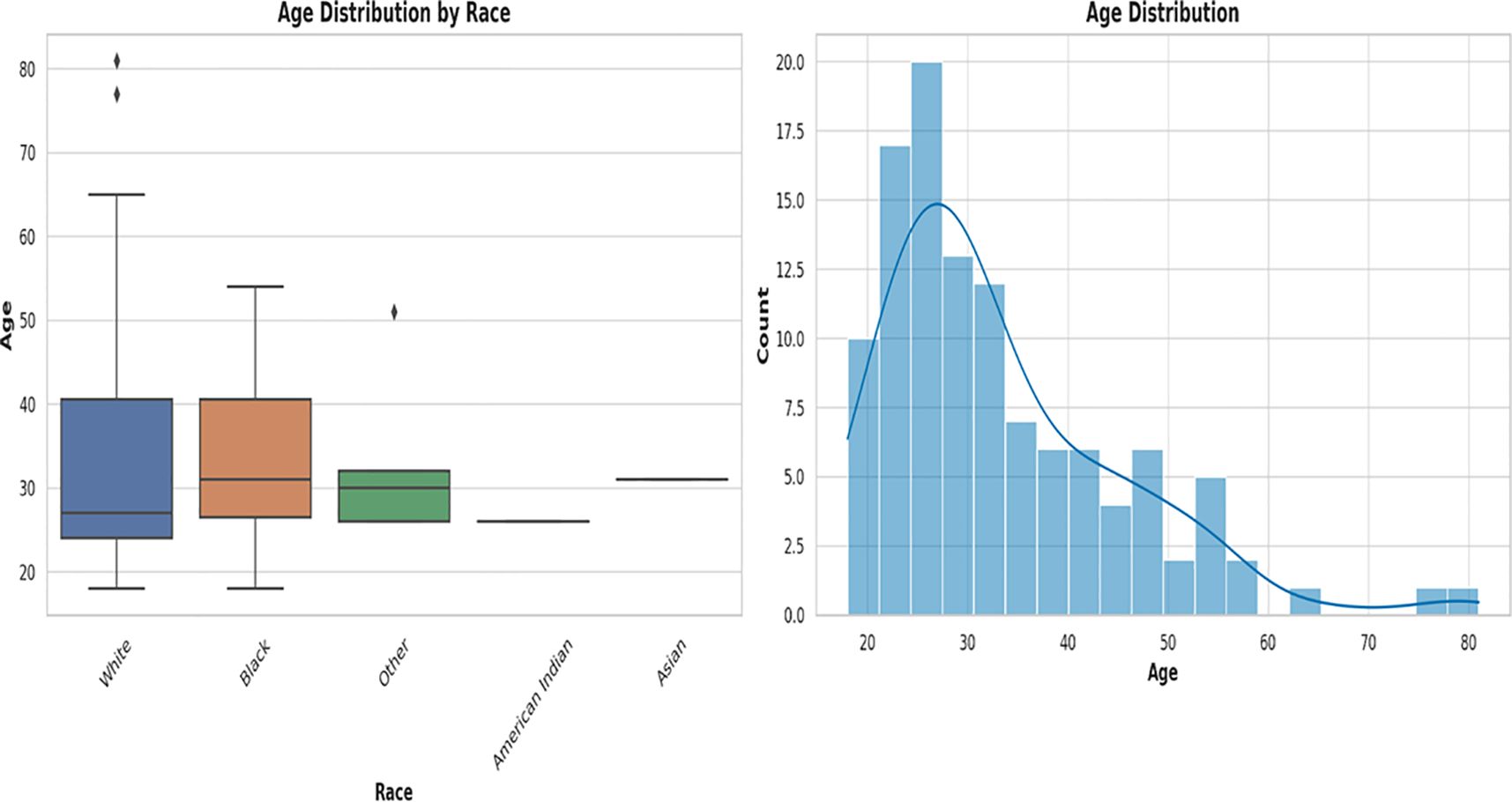
Figure 1 Box plot and histogram showing the age distribution among the races from which the vaginal samples were obtained. The box plot on the left, showing the age distribution among White people, Black people, Other (races), Native American or Alaska Natives, and Asians. White people were the largest population, followed by Black people. Other races are those who belong to none of the four races. The median ages among the races were very close within 28 and 35. An age distribution histogram on the right, shows the overall age distribution of participants in the study. The histogram includes a kernel density estimate (KDE) to show the smooth distribution of ages. The data is somewhat right-skewed, indicating a younger population with fewer older women. Most women fell into the 20–40 age range, with a peak around the late 20s to early 30s.
The age distribution of the races [Figure 1; Supplementary Table S1 (Demographics)] shows a broader distribution of ages within the White population (with outliers) than that of the other races, albeit the median ages across all races fell within a narrow range of 28 – 35 years. The age distribution of White people ranged from 18 – 83 years, with 50% falling within 28 – 40 years. Whereas the ages of Black people ranged from 18 to 55 years, 50% were within the same 28 – 40 years range. The “Other races”, which includes Hispanics/Latinos, had ages between 28 and 32 years and the median age was different among all the races [Figure 1; Supplementary Table S1 (Demographics)].
3.2 Identification and distribution of species across samples
The BV qRT-PCR assay efficiently identified the targeted 22 species with 95 – 100% sensitivity and specificity. Except for M. genitalium which was not detected in any sample, all the other species were present in at least one of the 946 samples. The percentage of the count of the identified species across all the samples are as follows: L. iners (75%), G. vaginalis (65%), F. vaginae (53%), Megasphaera sp. type 1 (44%), and P. bivia (41%) (Figure 2). These are followed by L. crispatus (29%), L. jensenii (23%), BVAB-1 (17%), U. urealyticum (13%), L. gasseri (12%), M. hominis (12%), M. curtisii (11%), and BVAB-3 (10%). The remaining species had less than 10% relative abundance across all samples combined (Figure 2).
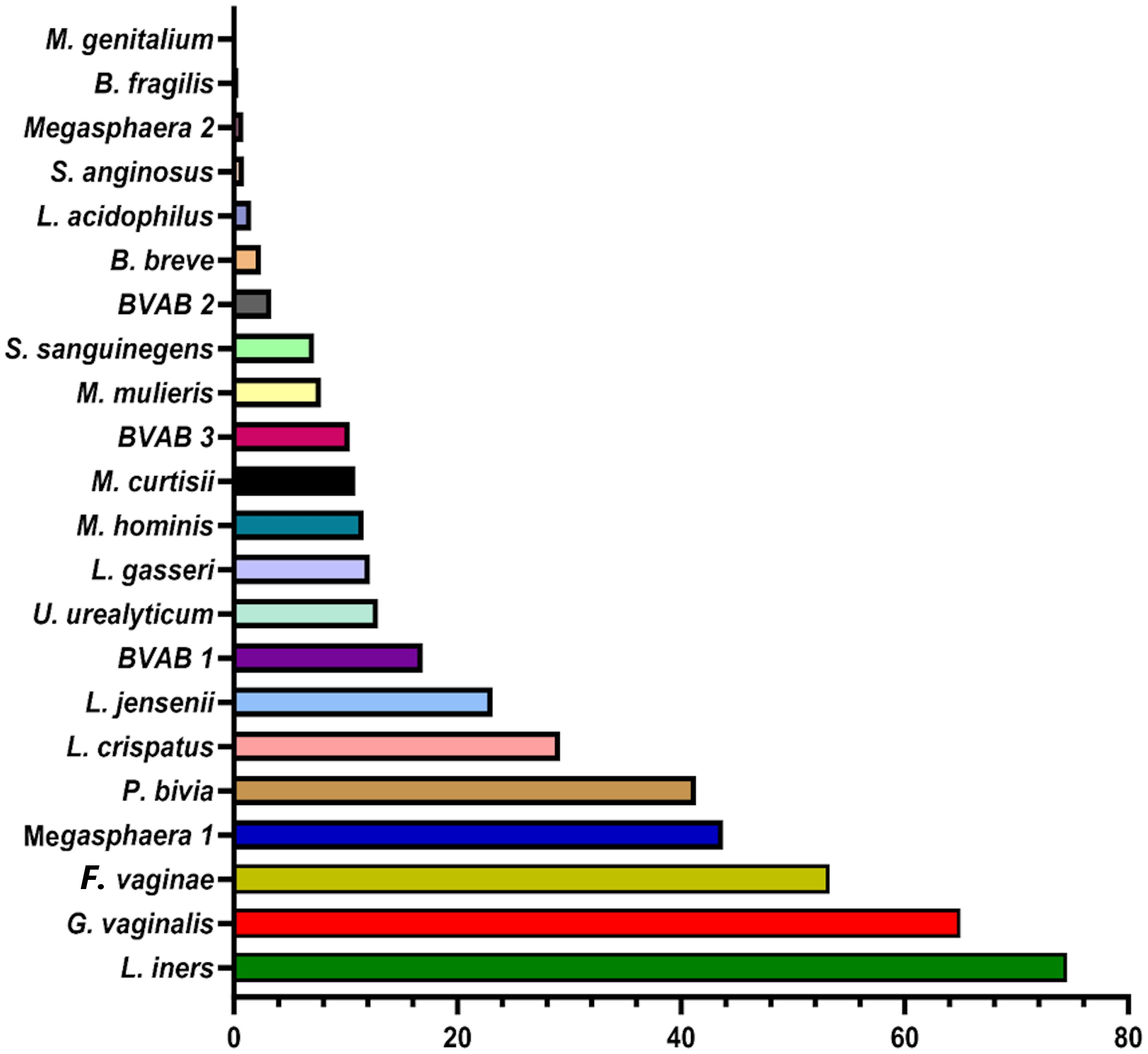
Figure 2 Percentage of the count of each species across all vaginal specimens (n = 946) as detected by qPCR. Among the 946 samples, it is observed that the most dominant species are Lactobacillus iners, Gardnerella vaginalis, Atopobium (Fannyhessea) vaginae, Megasphaera sp. type 1, Prevotella bivia, Lactobacillus crispatus, and Lactobacillus jensenii. The horizontal axis is the percentage of each species across all samples. No Mycoplasma genitalium was detected in any of all the samples.
To understand the distribution (spread) of the species and their concentrations across the samples, we used bar charts, histograms, and box and Whisker plots. L. iners, followed by G. vaginalis, F. vaginae, Megasphaera sp. type 1, P. bivia, and L. crispatus had better concentration distributions across the samples than the other species, with their median concentrations (102 and 106 gDNA copies/µL) being found in more than 100 samples. Indeed, other species were found in low concentrations across all the samples (Supplementary Figures S1–S22). The relative concentration for each identified species ranged between 102 and 108 genomic DNA copies/µL and the median range was between 102 and 106 copies/µL (Figure 3).
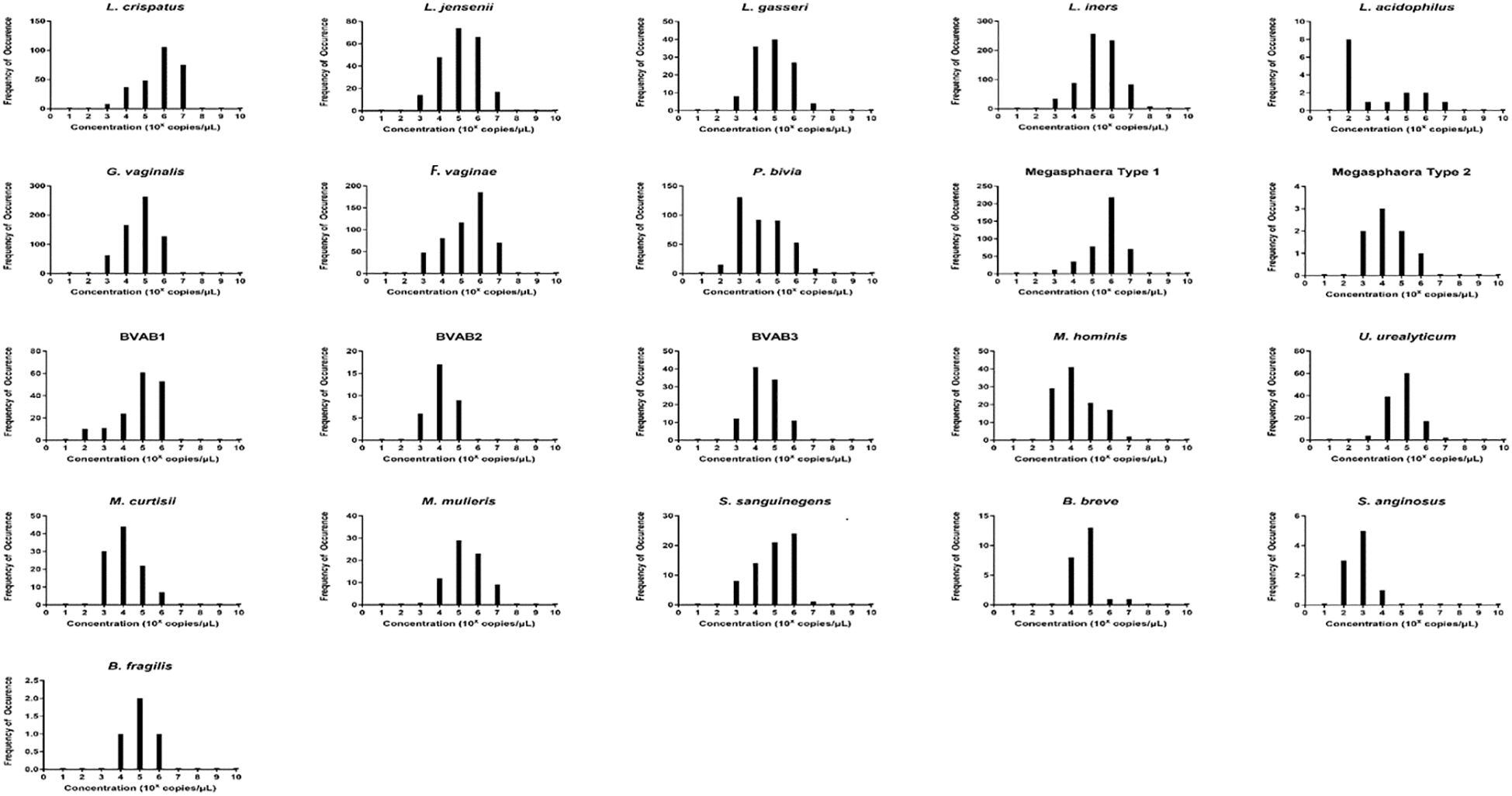
Figure 3 The relative concentration distribution of the identified bacterial species in vaginal swabs (n = 946). Each chart represents the concentration distribution per species; M. genitalium is not shown as was not detected in any of the samples. The vertical axis shows the count (frequency) of samples while the concentrations are shown on the horizontal axis. The median concentrations (102 and 106 gDNA copies/µL) of Lactobacillus iners and L. crispatus, Gardnerella vaginalis, Atopobium (Fannyhessea) vaginae, Prevotella bivia, and Megasphaera sp. type 1 were found in more than 100 samples. The charts also do not show the count of samples with zero concentrations but only counts of samples with higher concentrations.
The concentrations of the Lactobacillus species detected in this study were in the order, L. iners > L. crispatus > L. jensenii > L. gasseri > L. acidophilus (Figure 2). Additionally, F. vaginae and G. vaginalis occurred in similar amounts (~105 copies/µL) and were slightly more than the values obtained for P. bivia (104 copies/µL). BVAB-1 was the most predominant of the BVABs, and its relative concentration was 44% and 31% more than BVAB-3 and BVAB-2, respectively. Notably, over 98% of the Megasphaera species identified in this study belonged to type 1 with a mean concentration of ~106 copies/µL. Of the Mycoplasmas, U. urealyticum was found in higher concentrations than M. hominis. Although M. curtisii was 39% more abundant, it occurred in lower concentrations in vaginal swabs than M. mulieris.
3.3 Relative abundance, α- and β-diversities
We further investigated the per-sample species relative abundance and richness as well as inter-sample species diversity using alpha and beta diversities. The relative abundance of the species perfectly mirrored their concentration distribution across the samples (Figure 3; Supplementary Figures S1–S22), with L. iners, L. jensenii, P. bivia, G. vaginalis, Megasphaera sp. type 1, and F. vaginae being more abundant in many samples than the other species. The relative abundance, shown as a heatmap and a box plot (Figure 4), represents the abundance of each species across samples, normalized relative to the total concentration of microbial species within each sample. However, the other less abundant species also presented greater variability in relative abundance across the samples, as shown in the outliers (black stars) and absence of boxes (Figure 4B).
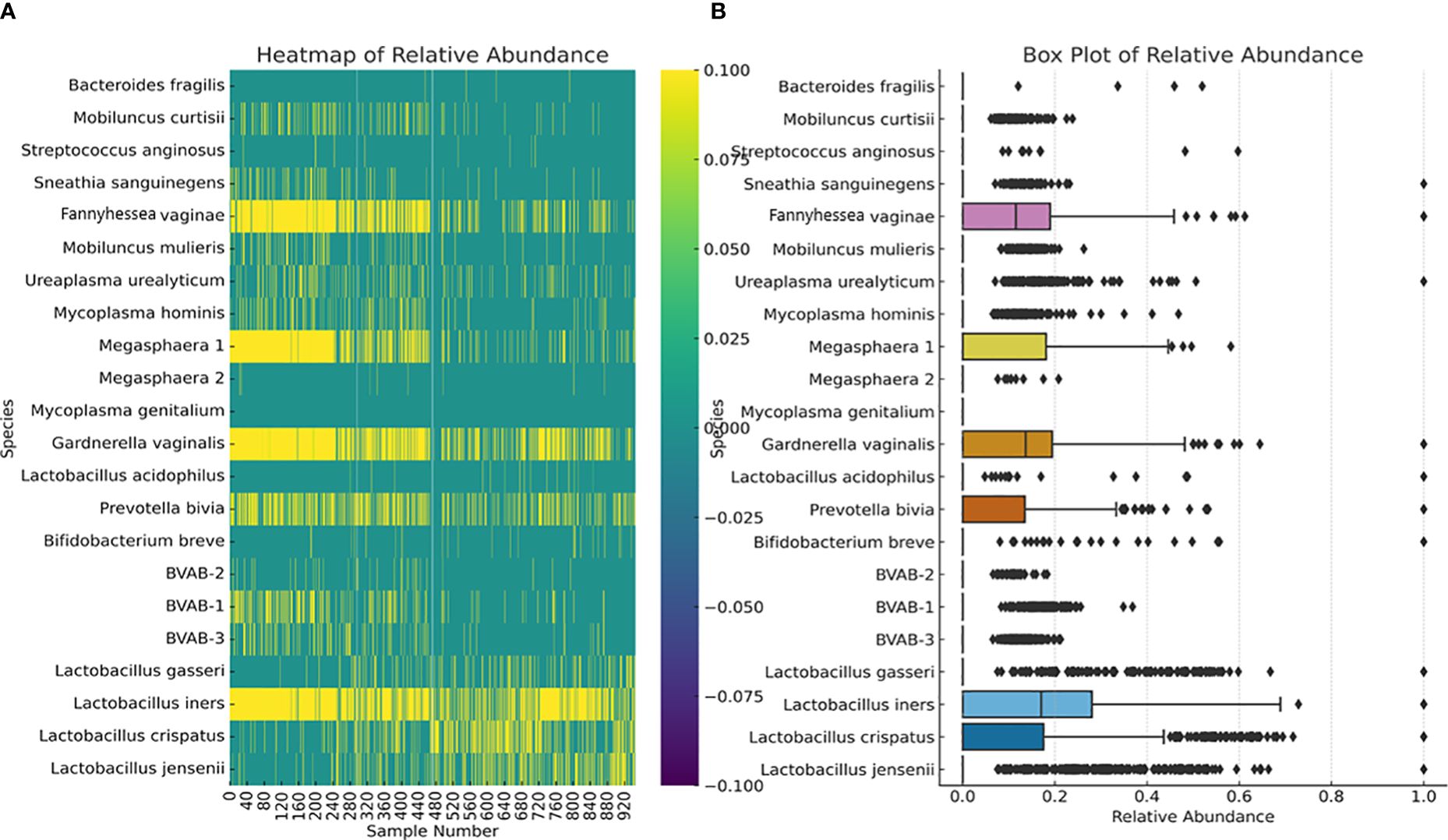
Figure 4 Relative abundance of the 22 species across all the 946 vaginal samples. The heatmap (A) highlights the distribution and intensity of species’ relative abundance across all samples, offering a color-coded representation that easily identifies the most prevalent species. The box plot (B) provides a statistical summary of each species’ relative abundance, including the median, interquartile range, and any outliers, offering insight into the variability and distribution of abundance for each species. This plot provides insights into the central tendency, spread, and outliers for the relative abundance of each microbial species within the dataset. Median values are represented by the line within each box. Interquartile range (IQR), indicating the middle 50% of the data, is shown by the box itself. Whisker extend to show the range of the data, i.e., 1.5 * IQR from the quartiles. Outliers are points outside the Whisker and are indicated as individual points.
The alpha diversity of the species within each sample was determined using species richness and Shannon diversity index (Figure 5). The species richness shows that 50% of the samples had 2 – 6 species, with 8 – 12 species occurring in 100 – 300 samples (Figures 5A, C). The Shannon index, which ranges from 0 (no diversity) to 5 (practically 3.5, i.e., most diverse), showed that 50% of the samples’ diversity index was between 0.7 and 1.8, with 200 – 280 samples having a diversity of > 2.0 (Figures 5B, D).
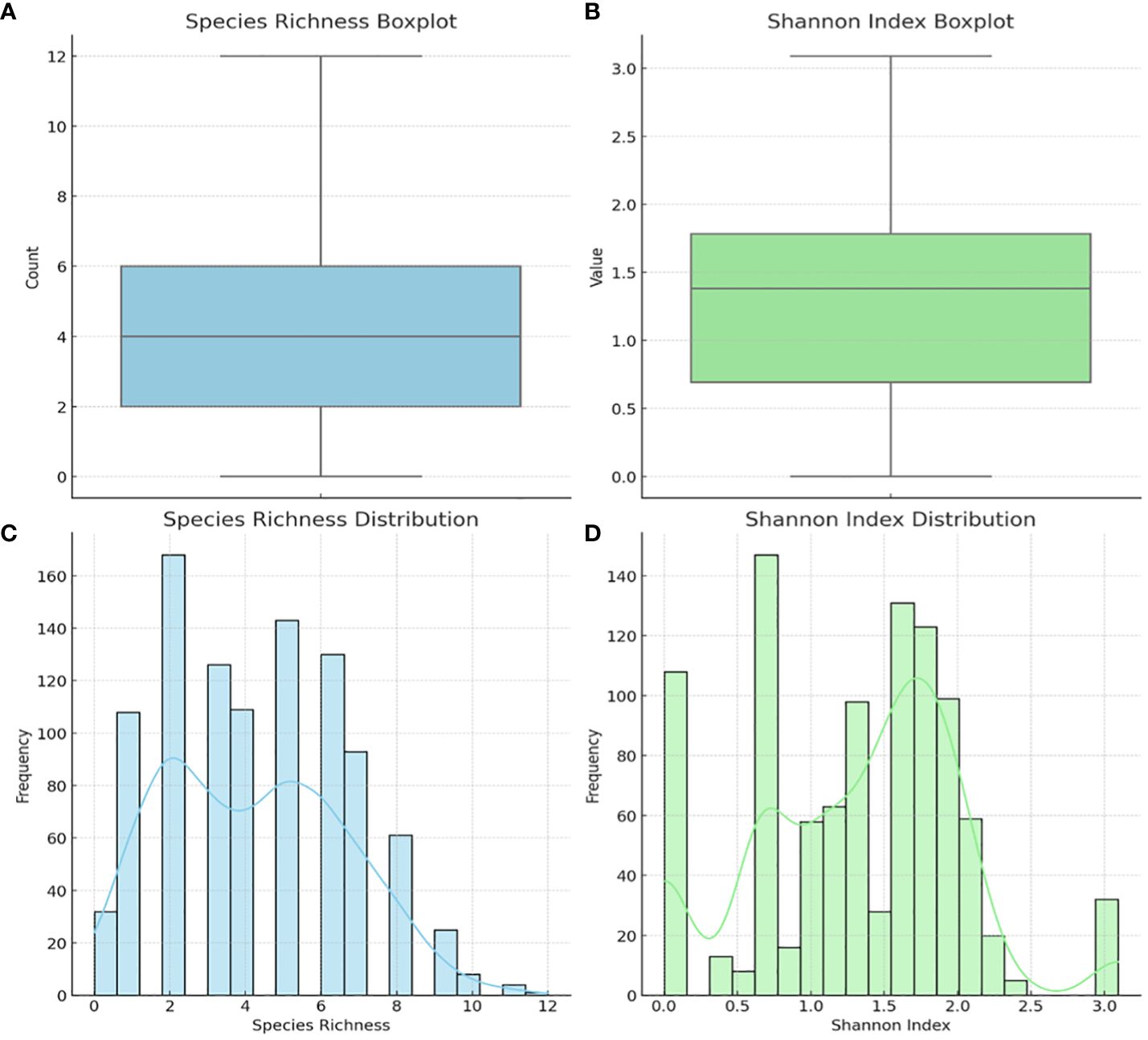
Figure 5 Alpha diversity of the 946 vaginal samples: species richness and Shannon index. Boxplot of Species Richness (A), showing the count of species present across samples. This boxplot provides a clear visualization of the central tendency and variability in species richness. Boxplot of the Shannon Index (B), indicating the diversity value that considers both abundance and evenness of species. Histogram of Species Richness Distribution (C), providing a view of the frequency distribution of species counts across samples. Histogram of Shannon Index (D) visualizes the distribution of the Shannon index across samples, reflecting both the abundance and evenness of species. The Shannon index is a more comprehensive measure of diversity, considering not just the presence of species but also their relative abundances.
Inter-sample (β-) diversity, using the Bray-Curtis dissimilarity index and associated principal component analysis (PCoA), showed that a few of the samples shared strong similarities (Figure 6). Samples with close similarity in species diversity are shown as blue (purple) while those with little similarity are shown as yellow. The abundance of yellow in the matrix shows how different most of the samples are from one another (Figure 6A). The PCoA chart also reflected this, with a few samples clustering together but most of the samples were spatially separated (Figure 6B).
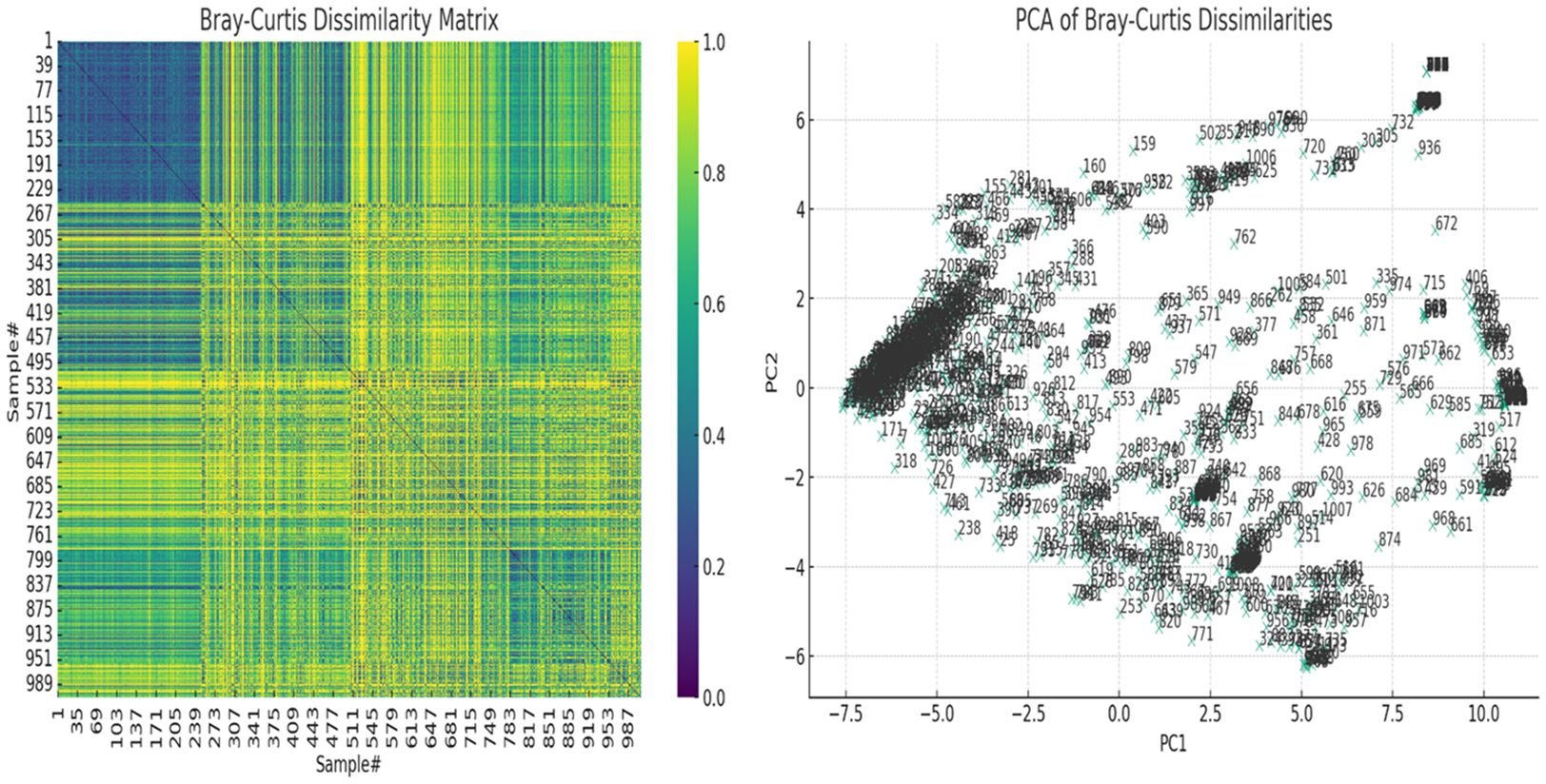
Figure 6 The Bray-Curtis dissimilarity matrix heatmap and associated principal component analysis (PCoA) for beta diversity. On the left, the Bray-Curtis Dissimilarity Matrix heatmap shows the pairwise dissimilarities between the samples. Higher values (closer to yellow, 1.0) indicate greater dissimilarity between samples, while lower values (closer to purple, 0.0) indicate greater similarity. On the right, the PCoA of Bray-Curtis Dissimilarities scatter plot visualizes the samples in a reduced two-dimensional space based on their dissimilarities. Each point represents a sample, and their positions reflect patterns of variation across the samples. Labels on the plot correspond to the sample numbers, helping to identify specific samples within the context of the PCoA. These show how dissimilar the samples were from each other.
3.4 Correlation and co-existence of species
We performed correlation, Chi-square and T-test analyses of the data to determine the significance of the species-species co-existence in the samples (Supplementary Table S2). A heat map of the significant results was generated for easy visualization of the data (Figure 7; Supplementary Table S2). Unlike the the Chi-square test (Figures 7A–C), the T-test provided a significant association between the non-Lactobacillus species (BVAB-1 was significantly affected by the presence/absence of L. iners only but not by the other Lactobacillus species)–Figure 7D and the presence/absence of the Lactobacillus species (without L. iners–Figure 7E or as a group–Figure 7F). In the absence of L. iners, the Lactobacillus species were not significantly associated with B. fragilis (which was only significantly associated with L. gasseri) but rather, with Megasphaera sp. type 2 (Figure 7B). Hence, the Chi-square test provided a more stringent cut-off than the T-test: individually, the Lactobacillus species were significantly associated with 14 species (Figure 7C).
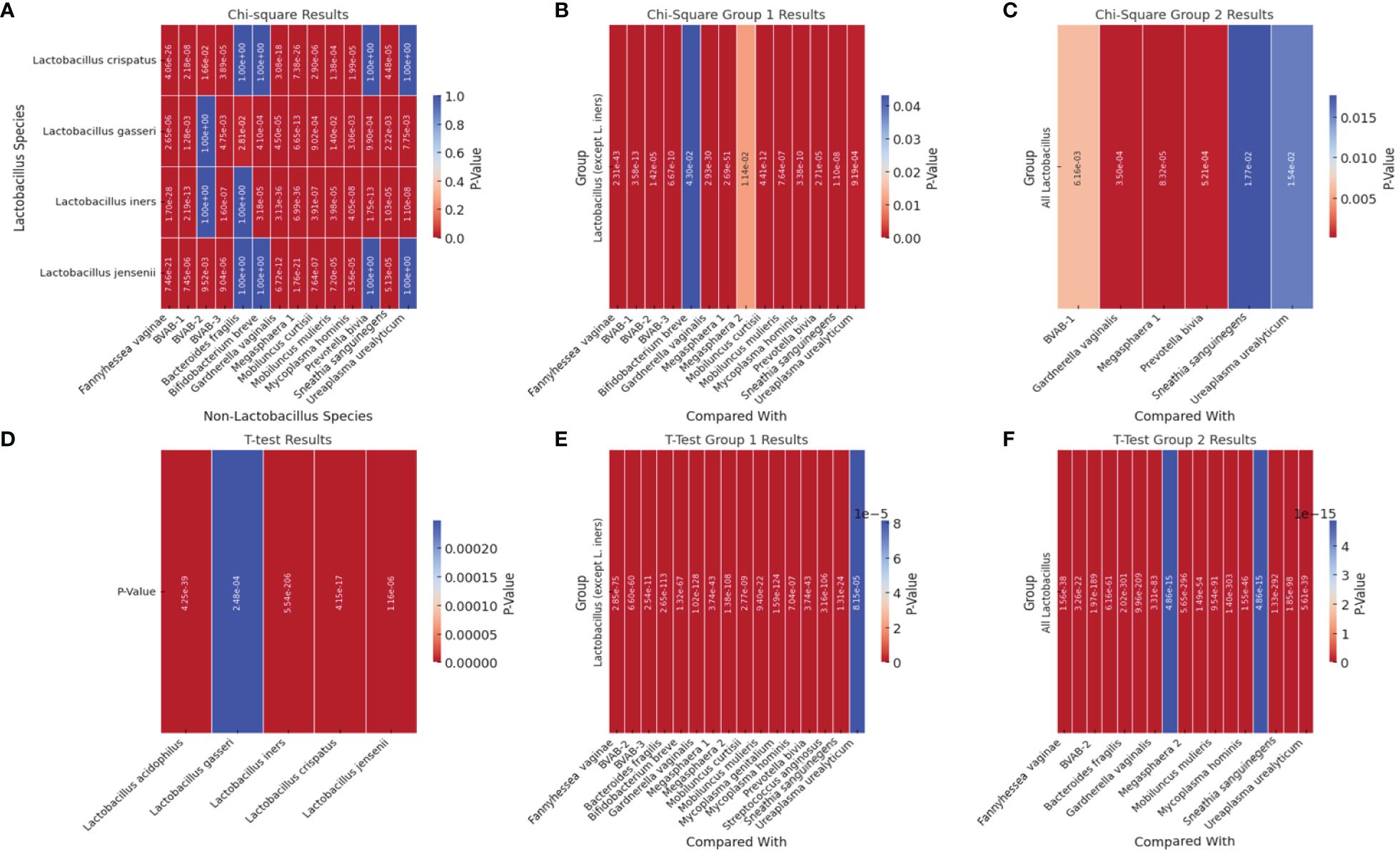
Figure 7 Heatmap of significant p-square values obtained from Chi-square and T-test analysis of pairwise comparisons between Lactobacillus and other non-Lactobacillus species within the samples. (A) visualizes the significant Chi-square p-values between Lactobacillus species and non-Lactobacillus species. (B) (Group 1) visualizes the significant Chi-square p-values of all Lactobacillus species (except L. iners) against each non-Lactobacillus species. (C) visualizes the significant Chi-square p-values from grouped comparisons of all Lactobacillus species against each non-Lactobacillus species. (D) shows the significant T-test p-values of all Lactobacillus species against all the other non-Lactobacillus species grouped together (T-test could not get individual species comparisons because of the structure of the data). (E) visualizes the significant T-test p-values from T-Test Group 1, which involves grouped comparisons of all Lactobacillus species (except L. iners) against each non-Lactobacillus species. (F) visualizes the significant T-test p-values from the “T-Test Group 2” sheet, which involves grouped comparisons of all Lactobacillus species against each non-Lactobacillus species. Rows represent different Lactobacillus species. Columns represent different non-Lactobacillus species. The color intensity indicates the level of significance, with cooler colors (towards blue) indicating lower p-values (higher significance) and warmer colors (towards red) indicating higher p-values (lower significance). Cells filled with a p-value of 1.0 represent non-significant results that were not included in the original significant results table and are shown for completeness.
Therefore, the presence of BVAB-1 and B. fragilis were independent of L. iners. Furthermore, L. crispatus and L. jensenii were significantly associated with the presence/absence of the same non-Lactobacillus species except B. fragilis, B. breve, P. bivia, and U. urealyticum (Figure 7). Although L. acidophilus was included in the Chi-square pairwise association test, it did not yield any significant results with any of the species (Supplementary Table S2).
Using Pearson’s correlation coefficient, a clear pattern was observed regarding the co-existence of the 21 species within the vaginal microbiota: all the Lactobacillus species, except L. iners, inversely correlated with most of the other non-Lactobacillus species while species such as BVAB (1–3), Megasphaera sp. type 1, P. bivia, G. vaginalis, F. vaginae, M. hominis, M. mulieris, M. curtisii, and S. sanguinegens were positively correlated with each other. Notably, some species had very little or no inverse correlation with the Lactobacillus sp. (except L. iners): S. anginosus, B. breve, U. urealyticum, M. hominis, Megasphaera sp. type 2, and B. fragilis. S. anginosus, B. breve, and B. fragilis were the only non-Lactobacillus species with an inverse correlation with L. iners; the other species had a positive correlation with L. iners. The BVAB species did not correlate with each other as BVAB -2 was less correlated with both BVAB-1 and BVAB-3; these latter two species, however, had a strong positive correlation (Supplementary Figure S23).
3.5 Relative abundance- and species-based diagnostic criteria
All the Lactobacillus sp. were bundled together as a marker of a normal vaginal microbiome (Group 1). Owing to the absence of a Nugent score or Amsel data for the samples, we used a species-based criteria to select species that are not found in normal vaginal microbiota: BVAB-1, -2, -3, B. fragilis, and S. anginosus. These five species were grouped into a species marker (Group 2) to identify BV-positive samples (Table 1). Owing to the strong co-existence association between G. vaginalis, P. bivia, Megasphaera sp. type 1, and A. vaginalis (Supplementary Figures S10, S23), they were tied together to serve as a marker (Group 3) to fine-tune our criteria in distinguishing between transitional BV, BV-positive, and BV-negative samples. Finally, the remaining eight species were also grouped into a marker (Group 4) to further distinguish between BV-positive, BV-negative, and transitional BV (Table 1).
The relative abundance distribution of the species within each of the four biomarker groups above was then used to select cut-off ranges for each group, incorporating a relative abundance-based criteria into the species-based criteria above. This resulted in a four-marker criterion, called the MDL-BV index, for diagnosing BV. We trained and tested this two-tier diagnostic MDL-BV index on our samples using machine-learning codes (Decision Trees and Random Forests) in Python, adjusting the ranges of each (species group) biomarker’s relative abundance until an optimal range per (species group) biomarker was found. The results of the MDL-BV index’s classifications were manually verified to ensure its veracity.
Observations made during the training and testing process made us include the following four instructions into the MDL-BV index code to enable categorization of all types of vaginal samples into their respective BV states: 1. When a sample falls within both BV-negative and transitional BV, assign it to transitional BV; 2. When a sample falls within both BV-positive and transitional BV, assign it to BV-positive; 3. When at least three of a sample’s four groups/biomarkers’ relative abundances fall within a single BV state and only one falls in another BV state, override the single disagreement, and assign or classify the sample to the BV state with which the three relative abundances agree; 4. When biomarkers 2 and 4 have a relative abundance of zero, then a relative abundance of 0 – 30% for Group 1 and 70 – 100% for Group 3 is BV-positive; a relative abundance of 30 – 50% (Group 1) is transitional BV and 60 – 100% (Group 1) is BV negative; 5. When every group is zero and only group 4 is 100%, define it as BV positive; 6. When all groups are zero, filter out that sample as there are no results to report (Table 1).
The final MDL-BV index was then tested on the data used in this study and 490 samples were classified as BV positive, 335 samples were classified as BV negative, and 151 samples were classified as Transitional BV. A manual verification of these classifications found them to be accurate, based on the MDL-BV index ranges. The relative abundance of each of the four biomarkers/groups and the final BV status classification based on these relative abundances, produced by the Python code, is shown in Supplementary Table S3 and summarized in Table 1.
3.6 Demographics affect BV
The effect of age, pregnancy status, and race on BV status was analyzed using a correlation heatmap (Supplementary Figure S24) and Box and Whisker distribution plots (Figure 8). Notably, almost all BV-negative cases were found within the White population. Transitional BV was found only among White and Black people, with BV-positive cases being widely distributed among the Black population aged 28 – 40; most White women who had BV were aged between 25 and 35. The age groups of the BV-positive and transitional BV populations were almost the same among Black people but very different among White people. The ‘Other” race (including Latinos/Hispanics) also had substantial BV, with their ages between a tight window of 28 to 32 years. Although the number of Asian and American Indian samples was relatively few, they were all BV-positive and fell within the median age range.
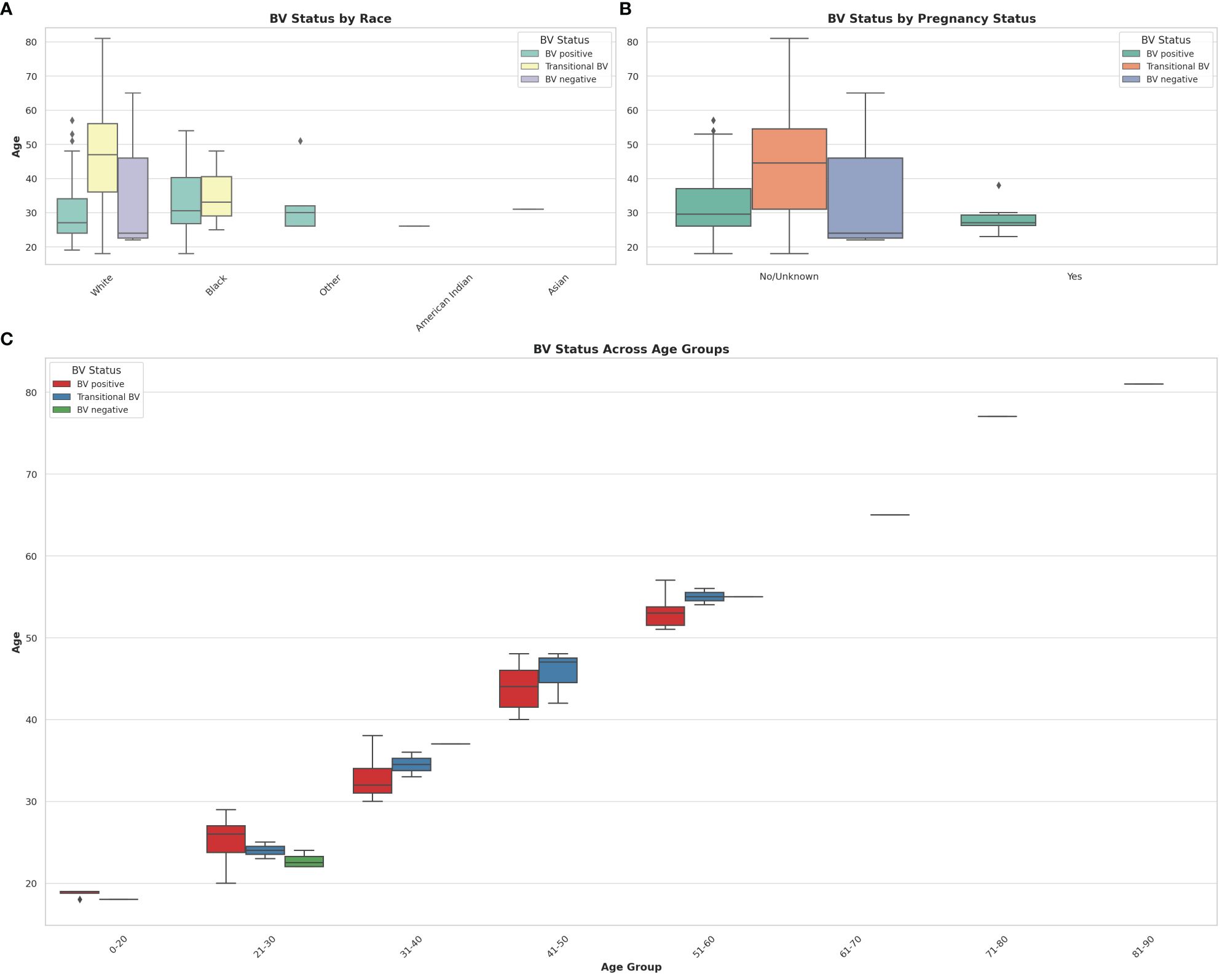
Figure 8 Box and Whisker plots showing the distribution of BV-positive, Transitional BV, and BV-negative samples across different races, ages, and pregnancy status. (A) shows the distribution of BV status across different races. (B) illustrates the distribution of BV status among pregnant and non-pregnant women. (C) visualizes the distribution of BV status across different age groups. The horizontal axes show the ages for the different classifications and distributions. The box colors are not the same for all the three plots and their respective keys in the upper right or left corners designate the correct interpretation of each color. The Whisker show the upper and lower quartiles while the boxes show the 25th and 75th percentile range (50% of the population) while the stars show outliers. Chi-square and Kruskal-Wallis tests respectively showed that there were no significant effect/association between race, pregnancy status, and age groups with BV.
It is notable that most women who were pregnant were also BV-positive, with no transitional and BV-negative status being found among them. Further, the age differences between BV-positive pregnant women (24 – 30 years) and non-pregnant women (28 – 38 years) were wide. The Box plot in 8C shows a similar occurrence of BV among women aged 21 – 60, with very little incidence being found among women aged above 60 and below 20. Notably, transitional BV cases were more common among women aged between 41 and 50 than among the other age groups. BV-negative cases were mostly found among women aged between 21 – 30 years.
Chi-square tests indicated that there was no statistically significant association between race and BV status (P = 0.217), and between pregnancy status and BV status (P = 0.527) at significance levels (P < 0.05). The Kruskal-Wallis test, which was used to assess whether there are statistically significant differences in age distributions between the BV positive and BV negative groups, found no statistically significant differences in the age distributions between the BV positive and BV negative groups (P = 0.820) at significance levels (P < 0.05). Therefore, age by itself, may not be a distinguishing factor between these two groups within the dataset analyzed.
3.7 Demographics affect variations in relative abundance
3.7.1 Variance of Lactobacillus shows lower abundance in BV-positive samples and higher abundance in BV-negative samples.
The relative abundance of Lactobacillus sp. showed that while L. acidophilus, L. jensenii, and L. crispatus had low mean abundance in BV-positive and transitional BV samples, and high mean abundance in BV-negative samples, L. iners and L. gasseri did not exhibit such phenomena. The mean abundance of L. gasseri was similar across all three BV states (marginal variance). L. iners was more prevalent in transitional BV samples and less abundant in both BV-negative and BV-positive samples; its mean relative abundance in both BV-positive and –negative species was similar. The greatest variance was observed in L. acidophilus’ relative abundance levels between BV-positive and BV-negative samples (Figure 9).
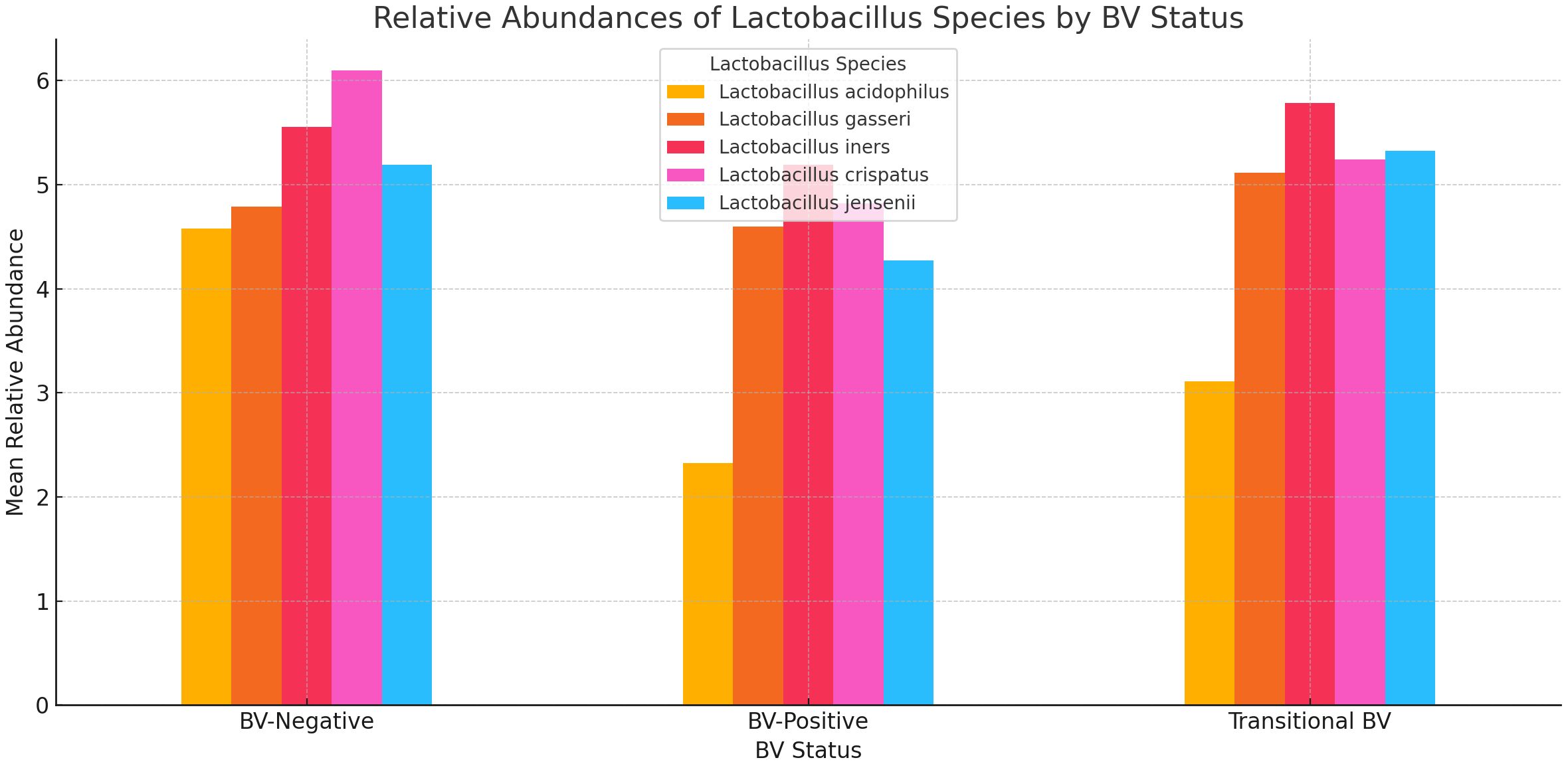
Figure 9 A bar chart displaying the relative abundances of five Lactobacillus species across three BV-Negative, BV-Positive, and Transitional BV statuses. Overall, Lactobacillus species tend to have higher mean abundances in BV-Negative samples, indicating their potential protective role against BV.
3.7.2 Variance of Lactobacillus and vaginal anaerobes show differences in pregnancy
The mean abundance of L. acidophilus, L. gasseri, and L. iners were slightly higher in pregnant women than in non-pregnant women. L. crispatus and L. iners had the highest relative abundance in non-pregnant and pregnant women, respectively. While L. jensenii’s mean relative abundance in both pregnant and non-pregnant women was similar, L. crispatus stood out as the only Lactobacillus with a slightly higher relative abundance in non-pregnant women than in pregnant women. Notably, the variance between the mean relative abundance of L. acidophilus in pregnant and non-pregnant women was markedly wider than the other species (Figure 10; Supplementary Figure S25). Except for L. crispatus, the mean relative abundance of the Lactobacillus sp. was marginally higher in non-pregnant women than in pregnant women (Supplementary Figure S25).
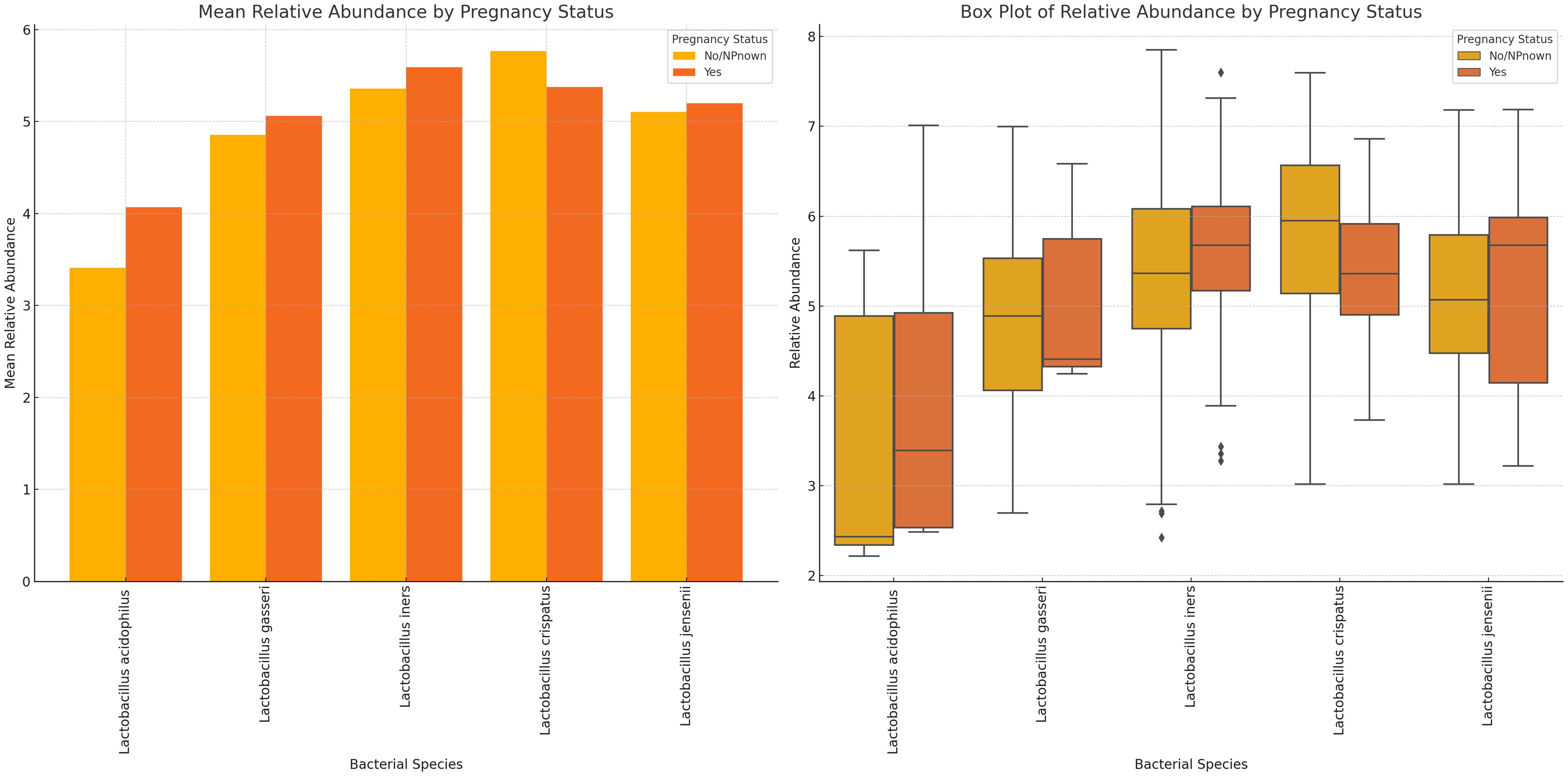
Figure 10 Plots displaying the relative abundances of five Lactobacillus species in vaginal samples collected from pregnant and non-pregnant women. L. crispatus was the highest in non-pregnant women while L. iners was highest in pregnant women. In all, the variance between these two cohorts were not so wide. L. acidophilus had a wider spread/distribution of relative abundances across the samples for the two cohorts.
The variance in mean relative abundance of the facultative and obligate anaerobes in pregnant and non-pregnant women differed per species (Figure 11; Supplementary Figure S26). Instructively, B. fragilis, Megasphaera sp. type 2, and B. breve were only present in non-pregnant women and absent in pregnant women. BVAB-3, P. bivia, M. curtisii, F. vaginae and M. mulieris had similar levels in both cohorts, with a slight decrease in four out of the five species for pregnant women; only M. curtisii was slightly higher in pregnant women. The differential abundance of BVAB-1, BVAB-2, G. vaginalis, Megasphaera sp. type 1, S. anginosus, and U. urealyticum was higher in pregnant women. S. sanguinegens alone was higher in non-pregnant women (Figure 11; Supplementary Figure S26). Non-pregnant women had higher diversity of anaerobes (n = 16 species) and generally lower mean relative abundance than pregnant women (n = 13 species) (Supplementary Figure S27).
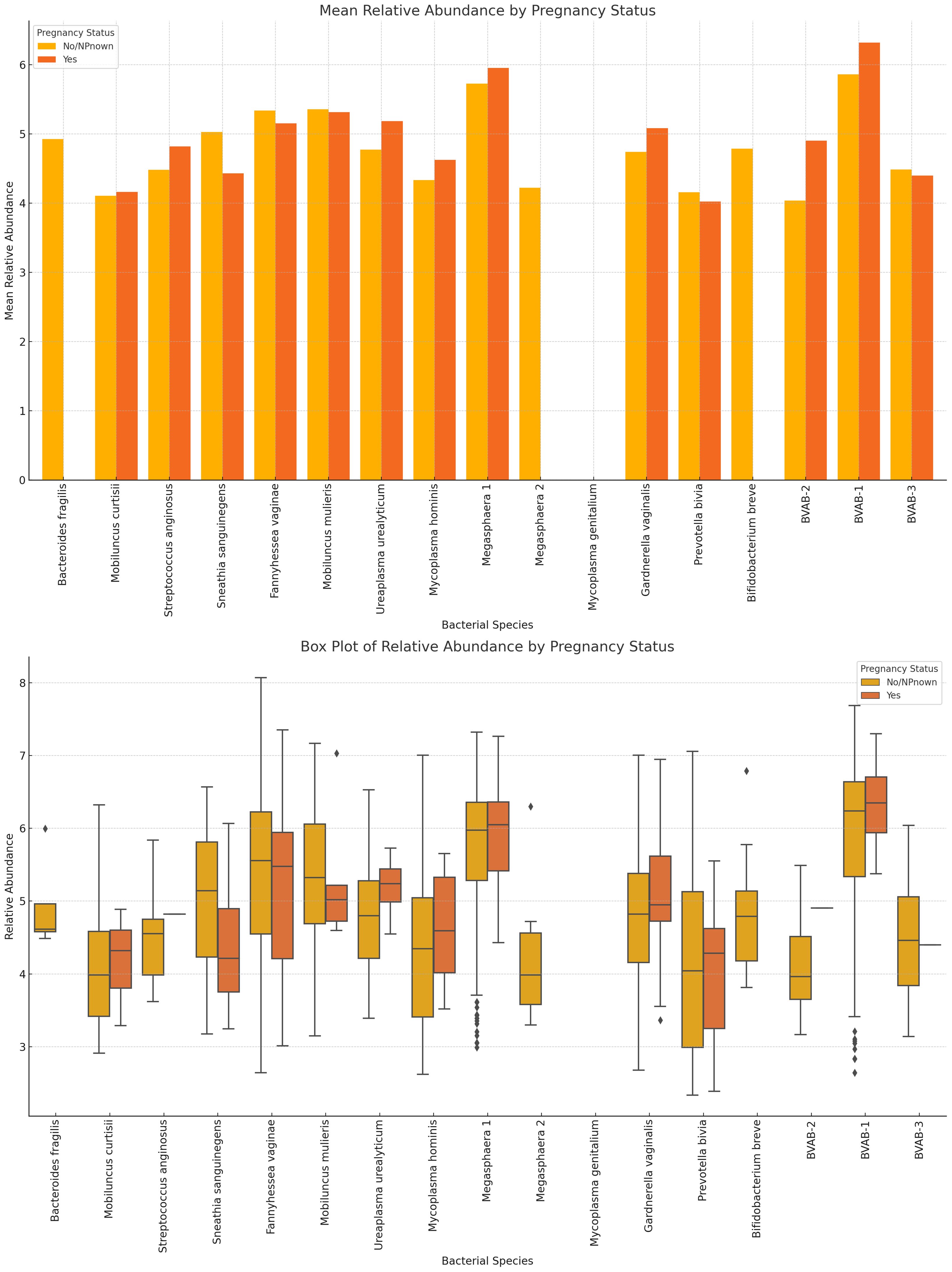
Figure 11 The mean and absolute relative abundances of various non-Lactobacillus bacterial species across pregnancy statuses (Pregnant and Not Pregnant). Species such as B. fragilis, Megasphaera sp. type 2, and B. breve were only present in non-pregnant women while the S. sanguinegens variance between pregnant and non-pregnant women was higher (in non-pregnant women). BVAB-3, P. bivia, F. vaginae, and M. mulieris had slightly higher mean abundances in non-pregnant women than in pregnant women. The rest had higher or slightly higher mean abundances in pregnant women than in non-pregnant women.
3.7.3 Variance of Lactobacillus and vaginal anaerobes show differences by age group
L. acidophilus was absent in females aged 61+ years and highest in those aged 41–50 years while L. crispatus was highest in those aged 61+ years. L. jensenii was most dominant in the 0–20 years’ cohort while L. gaserri had similar abundance levels across all age groups, except the 51–60 age group where it was slightly lower. L. iners was also most abundant in the 0–20 years group (Figure 12; Supplementary Figure S28).
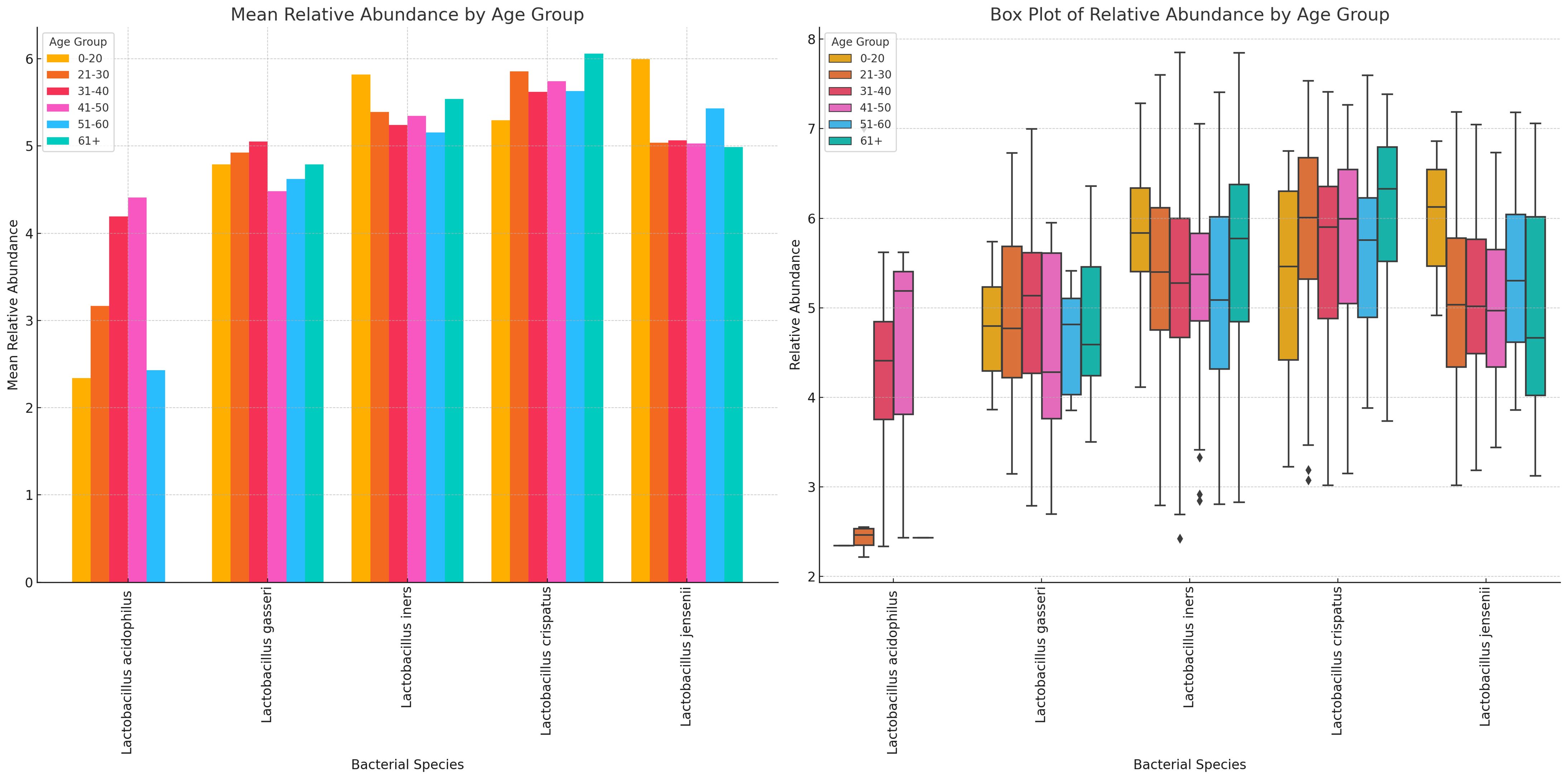
Figure 12 Charts displaying the mean and absolute relative abundances of five Lactobacillus species across different age groups (0–20, 21–30, 31–40, 41–50, 51–60, 61+). L. acidophilus was more abundant in age 41–50 group, followed by age 31–40 and 21–30 groups. L. crispatus was higher than the other species in all age groups except 0–20 group, where L. iners and L. jensenii were higher. Except. L. acidophilus, the other species had a wider distribution of relative abundances for the different age groups.
Likewise, the anaerobic bacterial species differed between age groups, with S. sanguinegens, F. vaginae, M. mulieris, Megasphaera sp. type 1, G. vaginalis, B. breve, and BVAB-3 being very dominant in females aged 61+ years. B. breve was very abundant in the 41–50 years group while Megasphaera sp. type 2 was also very prevalent in the 0–20-year group. B. fragilis, S. anginosus, M. mulieris, and Megasphaera sp. type 2 were also highly prevalent within the 31–40-year cohort. Notably, within the 21–30, 31–40, and 41–50 age groups, most of the species were mostly of similar or slightly higher relative abundance; the outliers are described above (Figure 13; Supplementary Figure S29).
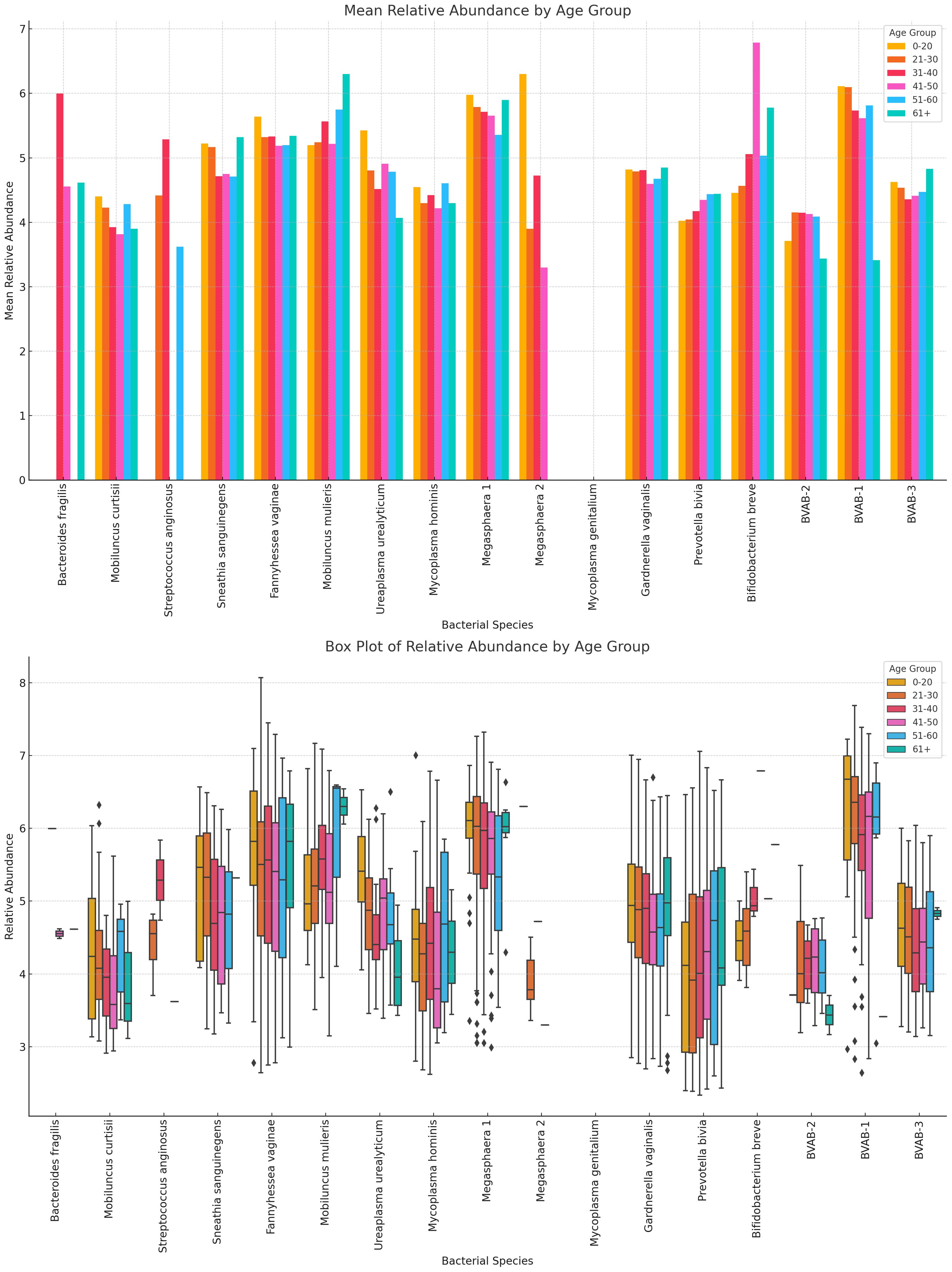
Figure 13 The bar chart displays the mean and absolute relative abundances of various non-Lactobacillus bacterial species across different age groups. Fourteen species were present in age 0–20 group, 15 were found in age 21–30, 41–50, and 51–60 groups, 16 was found in age 31–40 group, and 13 were found in 61+ age group. Species such as P. bivia, F. vaginae and S. sanguinegens had broader spread of relative abundances across the age groups. Overall, non-Lactobacillus bacterial species are primarily observed in the 21–30 age group, with little to no data available for other age groups. This indicates a higher microbial diversity in this age group compared to others.
There were 13 species in the 0–20 group, 15 in the 21–30, 41–50, and 51–60 groups, and 13 in the 61+ group, showing that the diversity increases after age 21, plateaus until age 60 and decreases after age 61. The highest relative abundances were mainly within the age 21–50 bracket for majority of the species (Figure 13; Supplementary Figure S29).
3.7.4 Variance of Lactobacillus and vaginal anaerobes show differences by age group
Variance of Lactobacillus and vaginal anaerobes show diversity and abundance vary by race Lactobacillus sp. and non-Lactobacillus sp. across the different racial groups. Specifically, L. iners, L. crispatus, and L. jensenii were present in most of the races, albeit L. crispatus was higher in most races and L. acidophilus was consistently lower than the other Lactobacilli. L. iners was very common in Asian people compared with the other Lactobacilli; L. acidophilus was higher in Others (Hispanics and Pacific Islanders) races and lower in the remaining races. L. gasseri was high in Asian, Black, and Others (Hispanics and Pacific Islanders) races across all races. Notably, L. crispatus was the highest Lactobacilli in White people (Figure 14; Supplementary Figures S30, S31).
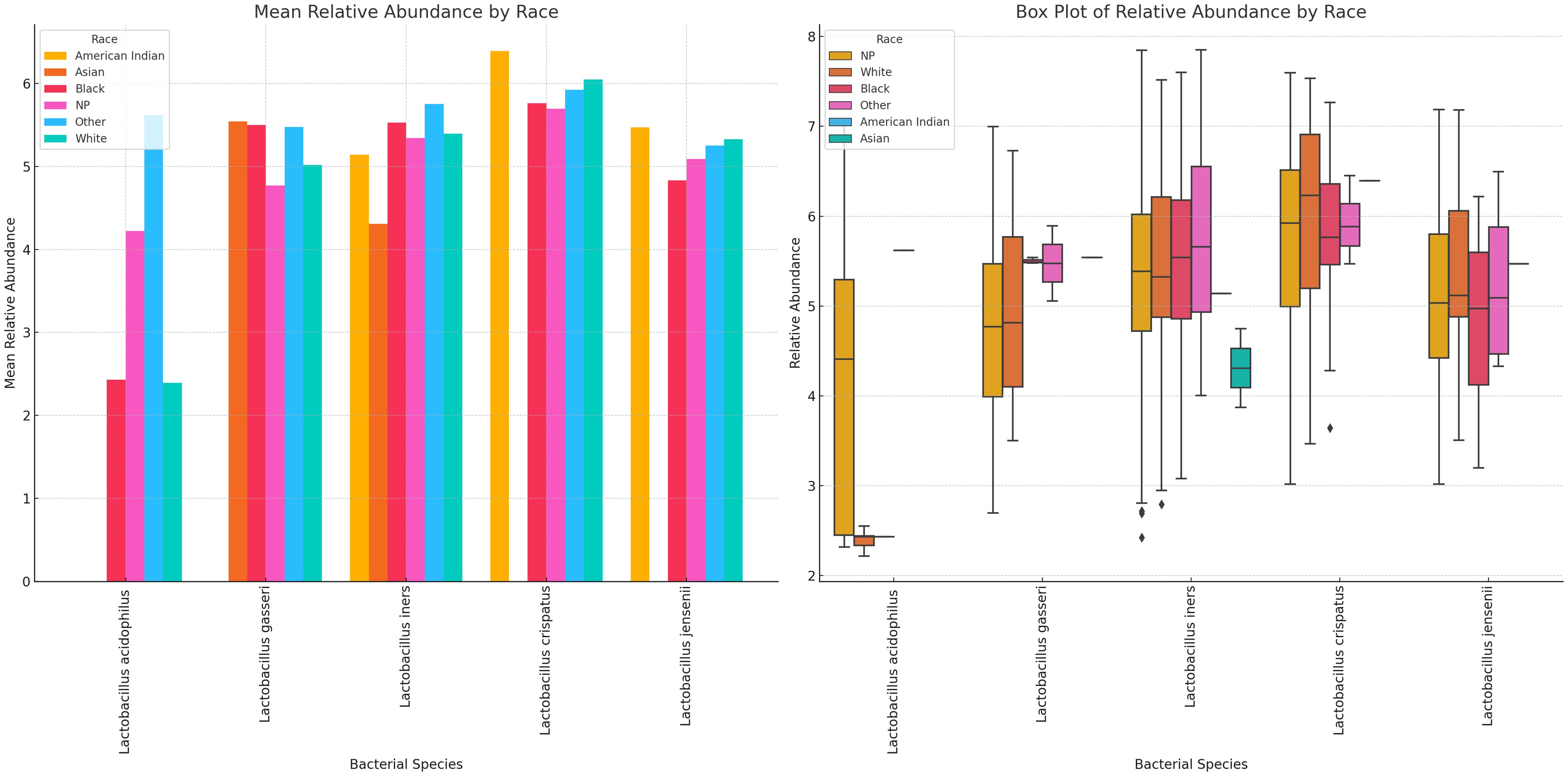
Figure 14 Mean and absolute relative abundance distribution of Lactobacillus species in different racial/ethnic groups. White, Black, and other races had more diversity of Lactobacillus sp. while Asian people had very little diversity. L. acidophilus was higher in other (Hispanics and Pacific Islanders) races than White and Black people. L. crispatus was higher in American Indians and White people while L. gasseri and L. iners were higher in Black and Other races. L. gasseri was also higher in Asian people. The relative abundance spread of the species differed per race. NP means not provided. The Whisker show the upper and lower quartiles while the boxes show the 25th and 75th percentile range (50% of the population) while the stars show outliers.
Racial variations in relative abundance were most obvious in B. fragilis, S. anginosus, M. hominis, Megasphaera sp. type 2, P. bivia, B. breve, and BVAB-2. For many of the species, there were a higher relative abundance among Black people than White people: Mobilincus sp., S. sanguinegens, U. urealyticum, G. vaginalis, Megasphaera sp., and BVAB-2. B. fragilis M. mulieris, G. vaginalis, BVAB-1 and BVAB-3 were highly abundant in Asian people. Among White people, F. vaginalis, M. hominis, S. sanguinegens, Megasphaera sp. type 1, G. vaginalis, P. bivia, B. breve, BVAB-2 and BVAB- 3 were dominant (Figure 15; Supplementary Figures S32, S33).
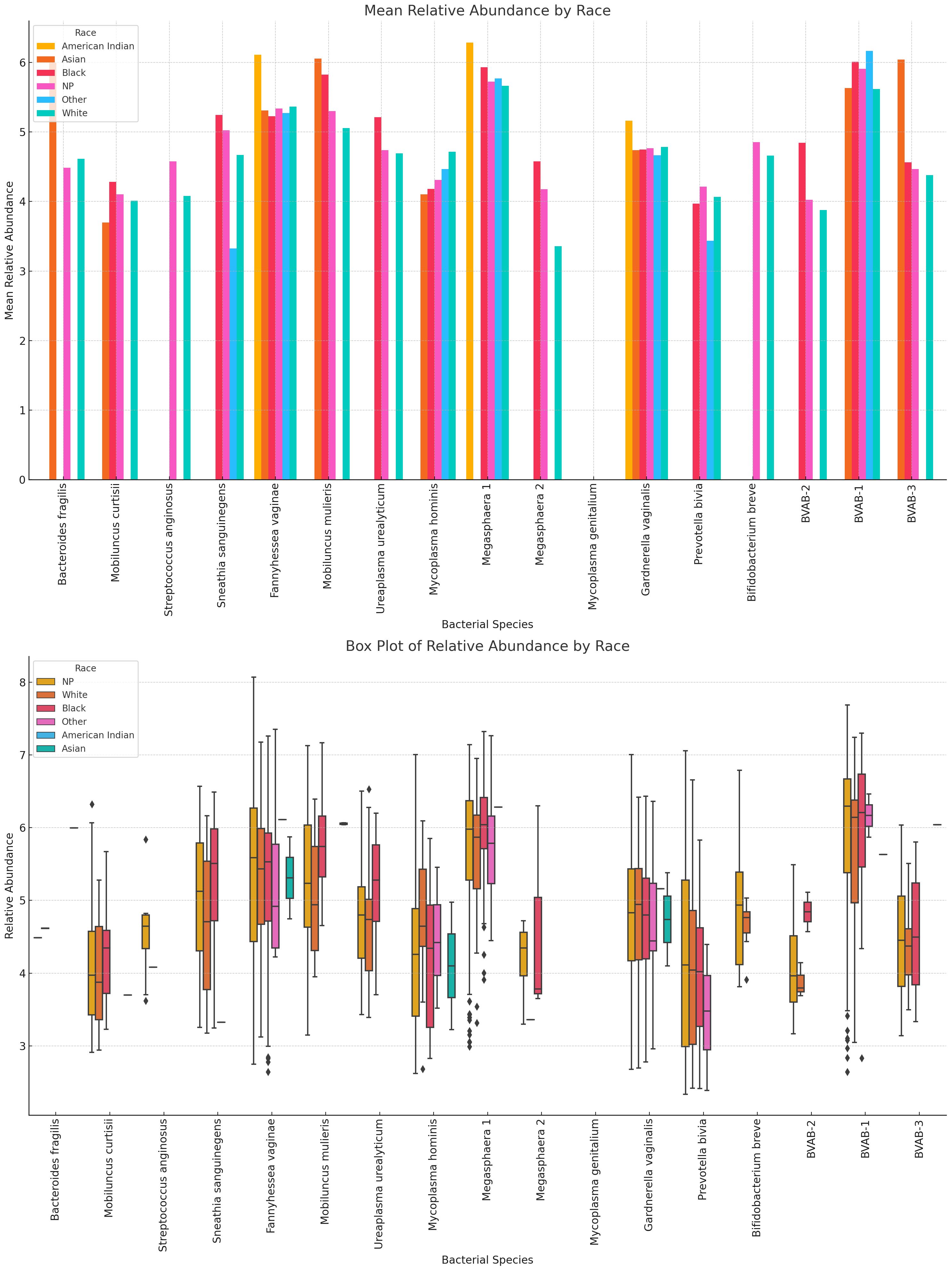
Figure 15 Relative abundance distribution of non-Lactobacillus species in different racial/ethnic groups. For many of the species, there were a higher relative abundance among Black people than White people, explaining the higher prevalence of BV in Black people than White people. F. vaginae, Megasphaera sp. type 1, and G. vaginalis were highest in American Indians. Relative abundance distribution of the species differed per species. The Whisker show the upper and lower quartiles while the boxes show the 25th and 75th percentile range (50% of the population) while the stars show outliers. NP means not provided.
4 Discussion
Unlike other infections with single etiological agents, BV is a polymicrobial infection with no clear consensus on the specific bacterial species responsible for the altered vaginal microbiota state (Mondal et al., 2023; Sobel and Vempati, 2024). The exact contributions of BV-associated microbes to the pathogenesis of BV, which may be relevant for accurate diagnosis and therapeutics, remain unresolved. Nevertheless, the occurrence of normal, transitional, and abnormal vaginal microbiota states is dependent on the composition and interactions of the different Lactobacillus and anaerobic species present (Onderdonk et al., 2016; Lynch et al., 2019; Chee et al., 2020; Drew et al., 2020). To address this gap between the vaginal microbiome dynamics and the current BV diagnostic tests’ limitations, a new qRT-PCR test that detects and measures the gDNA concentrations of 22 species found in all the various conditions of the vaginal microbiome was designed. The relatively shorter turnaround time of this test and the ease of adoption in a routine diagnostic laboratory makes it possibly more valuable than the current classical tests (Muzny et al., 2023; Swidsinski et al., 2023).
With such a broad spectrum of bacterial species, the resolution of the test is enhanced to efficiently distinguish between normal, abnormal, and transitional microbiota. The test has been validated with 95 – 100% sensitivity and specificity with a short turnaround time of 8 hours (from sample reception). In studies where results from PCR or qPCR studies targeting 2 – 13 species have been compared with Amsel’s criteria or Nugent’s score, the results have been highly sensitive and specific (Menard et al., 2008; Malaguti et al., 2015; Lynch et al., 2019; Drew et al., 2020; Vodstrcil et al., 2021). We are therefore confident that subsequent studies with our test, using Nugent score or Amsel’s criteria-diagnosed samples, may equally yield similar or better results with its larger bacteria target spectrum. Although this study is limited by the inability to compare the current results with clinical presentation data, its sensitivity and specificity in detecting the controls used confirm its efficiency.
In fact, the test’s diagnostic capability is further appreciated when the make-up and sample size of our vaginal specimens are considered: 946 vaginal samples from a wide spectrum of ages (18 – 83), races (White, Black, Asian, American Indian, and “Other (Hispanics and Pacific Islanders)”, not to mention the races of most women who failed to state their race), and pregnancy status. The Bray-Curtis dissimilarity matrix and the principal component analysis (PCoA) showed how dissimilar the samples were as few of the samples clustered together (Figure 6). The sample differences were further highlighted by the species richness (α-diversity) of the samples in which the number of species per sample ranged from 2 – 12, with 50% of the samples having 2 – 6 species per sample. Instructively, the Shannon index further clarified this observation by showing that the species diversity per sample was low, with most samples having an index between 0.8 and 1.8 and the most diverse sample having an index of 3.2 (out of 3.5).
Thus, although the samples were dissimilar in terms of composition, their diversity was relatively low, which could be characteristic of a healthy vaginal microbiome if the relative abundance of Lactobacillus sp. is high (Balashov et al., 2014). However, it is worth noting that the Shannon diversity is affected by the number of species sampled and the vaginal microbiome is naturally not as diverse as the gut microbiome (Javed et al., 2019; Muzny et al., 2020; Abou Chacra and Fenollar, 2021). Hence, although the 22 species used are more than any PCR test, it may not represent all the species in the vaginal microbiota. For instance, the alpha diversity of the samples was calculated through a direct count of the number of species observed in each sample without using any estimators or indices. Hence, it does not account for undetected species or attempt to estimate the total species richness in the samples, which methods like the Chao1 index aim to do. Notwithstanding, the representative nature of the samples used in this study is self-evident.
To our knowledge, no qPCR assay has the same broad-spectrum species target, making this assay an important innovation and the first to do so. Whereas other assays have used only two or up to 13 species (Malaguti et al., 2015), not all of the species were quantitatively detected as was done in this assay (Brotman and Ravel, 2008; Menard et al., 2008; Malaguti et al., 2015; Drew et al., 2020). This is despite the calls for increasing the bacterial spectrum for molecular BV diagnosis, focusing on Megasphaera, G. vaginalis, F. vaginae, and other anaerobes (Brotman and Ravel, 2008; Menard et al., 2008; Lynch et al., 2019). Moreover, owing to the biofilm-forming nature of these BV-associated species, which leads to immune evasion, high treatment failure rates and recurrence, a shorter-turnaround diagnostic tool is merited (Cangui-Panchi et al., 2023; Muzny et al., 2023). This study answers that call.
By both detecting and quantifying the 22 species in vaginal samples, we were able to obtain a clearer picture of the vaginal microbiome’s composition and make a better diagnosis of its condition; a feat only achievable with whole-genome metagenomics (Asante and Osei Sekyere, 2019; Osei Sekyere et al., 2020; Osei Sekyere et al., 2023). For instance, although BVAB-1, -2, and -3 were detected together in the same study (Fredricks et al., 2005), BVAB-2 has been commonly tested as a marker of BV positivity without BVAB-1 and -3. Yet, this study showed that BVAB-2 is the least prevalent/abundant among these three species, with BVAB-1, followed by BVAB-3, being the most common. A Brazilian study using 223 BV-positive samples also reported the same findings regarding the relative abundance of BVAB-1, BVAB-2, and BVAB-3, affirming our observations (Malaguti et al., 2015). A positive association between BV and high-risk Human Papilloma Virus (HPV) genotypes has been reported, owing to the occurrence of BVAB 1 and 3, and other BV-associated- bacteria in women co-infected with HIV and HPV (Naidoo et al., 2022). The same study also showed that the presence of BVAB 1 and 3 had an elevated likelihood of increasing the severity of cervical neoplasia in this population (Naidoo et al., 2022). Nevertheless, these species are not as closely related as initially thought as a recent phylogenetic analysis identified BVAB 1, 2, and 3 to be “Clostridiales genomosp.”, Oscillospiraceae bacterium strain CHIC02, and Mageeibacillus indolicus, respectively (Osei Sekyere et al., 2023).
Additionally, the relative abundance of the various species identified by this test mirrors what has been reported (Malaguti et al., 2015; Bayigga et al., 2019; Abou Chacra et al., 2021; Zhang et al., 2022; Powell et al., 2023), with Lactobacillus sp. (except L. acidophilus), G. vaginalis, A. (F.) vaginae, P. bivia, and Megasphaera sp. type 1 being the most dominant and widely distributed (Figures 2, 4; Supplementary Figures S1–S22). The absence of M. genitalium [which was found in low abundance in other studies using BV-positive samples (Malaguti et al., 2015)], and lower abundance and prevalence of L. acidophilus, M. hominis, U. urealyticum, Megasphaera sp. type 2, Mobiluncus sp., S. sanguinegens, B. breve, and S. anginosus are also consistent with other findings (Atassi et al., 2019; Chee et al., 2020; Argentini et al., 2022; Wu et al., 2022), further crediting the diagnostic efficiency of this test.
Overall, the Lactobacillus species had higher mean abundances in BV-negative samples, lower abundances in BV-positive samples, and in-between for transitional BV, indicating their potential protective role against BV (Figure 9). The higher relative abundance of L. iners in pregnant women and transitional BV samples could further suggest that L. iners play a crucial role in transitioning the vaginal microbiome from BV-negative to BV-positive. The transitioning effect of L. iners has been investigated and reported earlier and these observations further throws more light on the veracity of this conclusion (Zheng et al., 2021), albeit other studies provide conflicting findings and call for additional studies (Carter et al., 2023).
It was observed from the correlation, and co-existence analysis that the five Lactobacillus sp. mostly co-existed together while G. vaginalis, A. (F.) vaginae, P. bivia, and Megasphaera sp. type 1 also co-existed in the same samples (Figures 4, 7; Supplementary Figures S1–S23). The Chi-square and T-tests also largely agreed with the significant association between the Lactobacillus sp. and the non-Lactobacillus species, with few exceptions (Figure 7). This was used to form two separate groups of species-based biomarkers. Furthermore, the BVABs, S. anginosus, and B. fragilis, which are known to be mainly associated with BV microbiomes and absent in normal vaginal microbiomes (Weyers et al., 2009; Matu et al., 2010; Malaguti et al., 2015; Schmidt et al., 2015; Barrientos-Durán et al., 2020), were also teased from the remaining non-Lactobacillus species and grouped into another biomarker group. The remaining eight species were then also bundled together into another fourth group (Table 1). Using the relative abundance distributions of each species (Figure 4; Supplementary Figures S1–S22), the relative abundance range for each of the four species biomarker groups was set to form the MDL-BV index, which was then further trained and tested on the data using machine-learning algorithms (Decision Trees and Random Forests) to diagnose BV. A relative abundance-based approach was adopted over a nominal concentration value because concentrations vary from sample to sample, and the swabbing method of sample collection among clinicians is not standardized; hence, samples collected by each swab is not quantitative.
The MDL-BV index has a high cut-off range for negative BV status (≥70% Lactobacillus sp. relative abundance) to ensure that samples diagnosed as normal were BV-negative in verity. It also errs on the side of caution by classifying samples that meets the criteria of both BV-negative and transitional or transitional and BV-positive, as transitional BV or BV-positive respectively. The training of the model on the data further revealed certain intricacies and nuances in the distribution of the species and their relative abundances, which were used to refine it by including more details and rules. For instance, in situations where only two of the four biomarkers had a relative abundance score, the ratios were adjusted (Table 1). This strengthened the diagnostic efficiency of the MDL-BV index, resulting in a final classification of the samples as 491 BV-positive, 318 BV-negative, and 137 transitional BV. A manual verification of the results confirmed the veracity of the classifications (Supplementary Table S3).
Instead of using just two biomarkers involving all Lactobacillus sp. and all non-Lactobacillus species (Balashov et al., 2014), this index uses four biomarkers, which further refines the ability of the index to correctly distinguish between transitional BV or BV-positive microbiomes. Evidently, a correct molecular diagnosis of BV is critical to its treatment and monitoring, making this test and index very important in gynecology and obstetrics. The next stage of this research is to train the model on large datasets with Nugent scores/Amsel criteria as a means of further enhancing it and strengthening its proof of concept in BV diagnosis.
The distribution of BV among the different demographic variables viz., age, race, and pregnancy status showed important differences, albeit none of the differences were statistically significant (Figure 8). Specifically, although the number of vaginal samples from Black people was lower than that from White people, there were more BV-positive samples from the former than from the latter. While most of the vaginal samples from White people were BV-negative, those from Black people were either BV-positive or transitional. Notably, the samples from the “Other” races (which includes Hispanics/Latinos and Pacific Islanders), Asians, and Native American Indians, although fewer in number, were also BV-positive, with none being negative. Furthermore, the mean age and age range for White people with transitional BV were higher than those of all the other races, including the mean age of White people with abnormal vaginal microbiomes (Figure 8). Hence, although these differences were not statistically significant, they concur with other studies that show a higher prevalence of BV among Black and Hispanics than among White people and Asians (Peebles et al., 2019). This agreement between our data and other studies further confirms the accuracy of our MDL-BV index.
A further analysis of the racial vaginal microbiome composition showed that Black women had a higher prevalence of many of the anaerobes implicated in the BV pathogenesis, including G. vaginalis, F. vaginae, and Megasphaera sp., followed by Hispanics and Asian women who also had higher abundance of these BV-associated species than Caucasians (Figure 15; Supplementary Figures S32, S33). These findings are not singular as other studies have also identified the same. Particularly, Borgdoff et al. (2017) found a higher abundance of L. crispatus among Caucasians than Black and Asian women and a higher G. vaginalis and L. iners in Ghanaian and Asian (and Mediterranean) women, respectively (Borgdorff et al., 2017). Contrarily however, Roachford et al. (2022) also found a higher abundance of L. crispatus and L. iners in African Americans than in other ethnicities (Roachford et al., 2022); nevertheless, most studies agree that Africans have lower Lactobacillus-rich microbiomes compared with White women. These racial and ethnic variations were examined recently by Xin Wei et al. (2024) and found that the nucleotide identities of these vaginal denizens found in different ethnicities varied from each other, underscoring an evolutionary mechanism that explains these observed variations (Wei et al., 2024).
In effect, the genomes of the same species had differences when isolated from different races. While they also observed similarly higher abundance of BV-associated bacteria in Black pregnant women than other races (higher alpha diversity and Shannon index), they established that the alpha diversity remained fairly constant over gestational time and across ethnicities (Wei et al., 2024). This is similar to our finding in which the differences between pregnant and non-pregnant cohorts’ vaginal microbiome variations were marginal. Notwithstanding, this study brings out certain unique findings that merit further investigation: the absence of B. fragilis, Megasphaera sp. type 2, and B. breve in pregnant women; the higher differential abundance of G. vaginalis, BVAB-1 and -2, U. urealyticum, Megasphaera sp. type 1, and S. anginosus in pregnant women; and the higher abundance of S. sanguinegens in non-pregnant women compared with pregnant women.
Albeit pregnancy had no significant effect on BV, we posit that the observed marginal variations of higher diversity and higher relative abundance in pregnant women than in non-pregnant women can be due to the hormonal changes that occur in the former. Hormonal variations in pregnancy also explain the lower relative abundance of Lactobacillus sp. (except L. iners) and anaerobes among non-pregnant women. Notably, L. acidophilus presented the widest variations between cohorts, suggesting that it is more sensitive to these changes than the other species and could be adopted as a biomarker. Indeed, the higher abundance of L. crispatus among non-pregnant and menopausal women, the absence of L. acidophilus in menopausal women, and the higher abundance of L. iners among pregnant women further show how the variations in hormones affect these species’ relative abundance through changes in the hosts estrogen-medicated metabolisms (Auriemma et al., 2021). The higher abundance of L. crispatus and L. iners was been reported previously (Borgdorff et al., 2017; Wei et al., 2024).
Although BV among non-pregnant women was more prevalent than among pregnant women, BV-negative or transitional BV samples were not obtained from pregnant women (Figure 8). This is a concerning observation as BV in pregnancy is associated with preterm labor, low birth weight, premature rupture of membranes, miscarriage, chorioamnionitis, and birth asphyxia (Bayigga et al., 2019; Basavaprabhu et al., 2020; Vodstrcil et al., 2021). Although the pregnancy stage of the women from whom the samples were obtained is unknown, it is known that the first-trimester vaginal microbiome is similar to a BV-positive microbiome while the 2nd and 3rd trimesters are similar to a normal vaginal microbiome (Kaur et al., 2020). Hence, the stage of the pregnancy and vaginal symptoms should be considered in making a final therapeutic decision regarding pregnant women when using molecular-based tests. Furthermore, the difference in the number of pregnant women (n = 53) vis-a-viz the number of non-pregnant (or unknown pregnancy status) women is large and can skew the data. This is a major limitation in our data set and should be considered when analyzing the pregnancy data.
Although BV-positive samples were found in all age groups (18 – 83 years), they were mostly prevalent among women who were between 20 and 60 years, with a higher number of BV-positive and transitional BV samples being found among women between 41 – 50 years. The species variations per age group further support this observation as the anaerobic species diversity and abundance increase from after 20 years, plateau until age 50, and begin to decline again (Figures 12, 13; Supplementary Figures S28, S29). Indeed, the median age found in this study’s population is similar to what was found in Brazil (Malaguti et al., 2015) and in the USA (Watkins et al., 2024), with women above 21 years being more likely to suffer recurrence of BV (Coudray and Madhivanan, 2020; Coudray et al., 2020). Hence, the age groups found in our data concur with that of other studies, which confirms that BV is common among reproductive-age women and lower before puberty and after menopause (Auriemma et al., 2021). However, age was not significantly associated with BV occurrence in this study.
5 Conclusion
The new qRT-PCR assay developed by MDL is the first of its kind to quantitatively detect 22 species in the vaginal microbiome. We developed a novel classification system, the new MDL-BV index, which was trained on large sets of data and based on four species-based and relative abundance-based biomarkers. This multifaceted two-tier approach of using species and relative abundance provides a better diagnostic resolution for a polymicrobial infection such as BV. We are working to extend this approach to apply to other microbiome-based diagnostics to make disease diagnosis reflective of the clinical presentations. Our study is, however, limited by the absence of a clinical Nugent score or Amsel’s criteria; however, we are working to include these in the next studies that will involve the MDL-BV index and the 22-species qRT-PCR BV test. The clinical and socio-economic importance of this novel proprietary BV diagnostic test in obstetrics and gynecology is notable.
Data availability statement
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding authors.
Ethics statement
Ethics approval was not required for this study as the samples used were leftover, discarded samples provided by healthcare providers and were anonymized, posing no risk of identifying individual patients. According to the Common Rule (45 CFR 46) and HIPAA (45 CFR 164.514(b)(2)), research involving anonymized data does not constitute human subjects research and thus does not require IRB approval. Written informed consent was not obtained as the study used leftover, discarded samples, and the study did not involve direct interaction with patients.
Author contributions
AO: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Software, Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing. RH: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Software, Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing. JT: Investigation, Methodology, Supervision, Writing – review & editing. SF: Supervision, Validation, Writing – review & editing. EM: Funding acquisition, Resources, Supervision, Validation, Visualization, Writing – review & editing. MA: Funding acquisition, Project administration, Resources, Supervision, Validation, Visualization, Writing – review & editing. JO: Conceptualization, Data curation, Formal analysis, Investigation, Methodology, Project administration, Software, Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing.
Funding
The author(s) declare financial support was received for the research, authorship, and/or publication of this article. This work was funded by the Medical Diagnostics Laboratories, LLC.
Acknowledgments
The authors are grateful to the technicians at Medical Diagnostic Laboratories for their direct and indirect assistance. The material and financial resources provided by Medical Diagnostic Laboratories towards this project are warmly acknowledged and deeply appreciated. We are also grateful to Annette Daughtry for assisting with the review of the initial draft.
Conflict of interest
All the authors are scientists and staff of Medical Diagnostic Laboratories LLC.
The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fcimb.2024.1409774/full#supplementary-material
Supplementary Table 1 | Raw demographic and qPCR data showing the demographics of the women from whom the samples were obtained and the qPCR gDNA concentrations of each species within the 946 samples.
Supplementary Table 2 | Chi-square and T-test statistical data showing the significant and non-significant associations between Lactobacillus sp. and the other non-Lactobacillus species.
Supplementary Table 3 | BV status classification data generated by the Python algorithm trained on the MDL-BV index ranges. The relative abundance of each group of biomarkers are summed up and shown under their respective groups. The ratio of these relative abundances are used to infer the BV status of the sample.
Supplementary Figures 1–22 | Concentration distribution of each of the 22 bacterial species tested by the qPCR test across all the samples. Both boxplot and histograms charts are shown for each species.
Supplementary Figure 23 | A correlation matrix showing the correlation between the individual species found within the samples. Different color codes are used to show positive and negative correlations.
Supplementary Figure 24 | A correlation heatmap showing the correlation between age, pregnancy, race, and BV status.
References
Abou Chacra, L., Fenollar, F. (2021). Exploring the global vaginal microbiome and its impact on human health. Microb. Pathog. 160, 105172. doi: 10.1016/j.micpath.2021.105172
Abou Chacra, L., Fenollar, F., Diop, K. (2021). Bacterial vaginosis: What do we currently know? Front. Cell Infect. Microbiol. 11. doi: 10.3389/FCIMB.2021.672429
Amsel, R., Totten, P. A., Spiegel, C. A., Chen, K. C. S., Eschenbach, D., Holmes, K. K. (1983). Nonspecific vaginitis. Diagnostic criteria and microbial and epidemiologic associations. Am. J. Med. 74, 14–22. doi: 10.1016/0002-9343(83)91112-9
Argentini, C., Fontana, F., Alessandri, G., Lugli, G. A., Mancabelli, L., Ossiprandi, M. C., et al. (2022). Evaluation of modulatory activities of lactobacillus crispatus strains in the context of the vaginal microbiota. Microbiol. Spectr. 10, e02733–21. doi: 10.1128/spectrum.02733-21
Asante, J., Osei Sekyere, J. (2019). Understanding antimicrobial discovery and resistance from a metagenomic and metatranscriptomic perspective: Advances and applications. Environ. Microbiol. Rep. 11, 62–86. doi: 10.1111/1758-2229.12735
Atassi, F., Pho Viet Ahn, D. L., Lievin-Le Moal, V. (2019). Diverse Expression of Antimicrobial Activities Against Bacterial Vaginosis and Urinary Tract Infection Pathogens by Cervicovaginal Microbiota Strains of Lactobacillus gasseri and Lactobacillus crispatus. Front. Microbiol. 10. doi: 10.3389/fmicb.2019.02900
Auriemma, R. S., Scairati, R., del Vecchio, G., Liccardi, A., Verde, N., Pirchio, R., et al. (2021). The vaginal microbiome: A long urogenital colonization throughout woman life. Front. Cell Infect. Microbiol. 11. doi: 10.3389/fcimb.2021.686167
Balashov, S. V., Mordechai, E., Adelson, M. E., Sobel, J. D., Gygax, S. E. (2014). Multiplex quantitative polymerase chain reaction assay for the identification and quantitation of major vaginal lactobacilli. Diagn. Microbiol. Infect. Dis. 78, 321–327. doi: 10.1016/j.diagmicrobio.2013.08.004
Barrientos-Durán, A., Fuentes-López, A., de Salazar, A., Plaza-Díaz, J., García, F. (2020). Reviewing the composition of vaginal microbiota: inclusion of nutrition and probiotic factors in the maintenance of eubiosis. Nutrients 12, 419. doi: 10.3390/nu12020419
Basavaprabhu, H. N., Sonu, K. S., Prabha, R. (2020). Mechanistic insights into the action of probiotics against bacterial vaginosis and its mediated preterm birth: An overview. Microb. Pathog. 141, 104029. doi: 10.1016/j.micpath.2020.104029
Bayigga, L., Kateete, D. P., Anderson, D. J., Sekikubo, M., Nakanjako, D. (2019). Diversity of vaginal microbiota in sub-Saharan Africa and its effects on HIV transmission and prevention. Am. J. Obstet. Gynecol. 220, 155–166. doi: 10.1016/j.ajog.2018.10.014
Bhujel, R., Mishra, S. K., Yadav, S. K., Bista, K. D., Parajuli, K. (2021). Comparative study of Amsel’s criteria and Nugent scoring for diagnosis of bacterial vaginosis in a tertiary care hospital, Nepal. BMC Infect. Dis. 21, 825. doi: 10.1186/s12879-021-06562-1
Borgdorff, H., van der Veer, C., Van Houdt, R., Alberts, C. J., De Vries, H. J., Bruisten, S. M., et al. (2017). The association between ethnicity and vaginal microbiota composition in Amsterdam, the Netherlands. PLoS One 12, e0181135. doi: 10.1371/journal.pone.0181135
Brotman, R. M., Ravel, J. (2008). Ready or not: the molecular diagnosis of bacterial vaginosis. Clin. Infect. Dis. 47, 44–46. doi: 10.1086/588662
Cangui-Panchi, S. P., Ñacato-Toapanta, A. L., Enríquez-Martínez, L. J., Salinas-Delgado, G. A., Reyes, J., Garzon-Chavez, D., et al. (2023). Battle royale: Immune response on biofilms - host-pathogen interactions. Curr. Res. Immunol. 4, 100057. doi: 10.1016/j.crimmu.2023.100057
Carter, K. A., Fischer, M. D., Petrova, M. I., Balkus, J. E. (2023). Epidemiologic evidence on the role of lactobacillus iners in sexually transmitted infections and bacterial vaginosis: A series of systematic reviews and meta-analyses. Sex. Transm. Dis. 50, 224–235. doi: 10.1097/OLQ.0000000000001744
Chee, W. J. Y., Chew, S. Y., Than, L. T. L. (2020). Vaginal microbiota and the potential of Lactobacillus derivatives in maintaining vaginal health. Microb. Cell Fact 19, 203. doi: 10.1186/s12934-020-01464-4
Cheng, L., Norenhag, J., Hu, Y. O. O., Brusselaers, N., Fransson, E., Ährlund-Richter, A., et al. (2020). Vaginal microbiota and human papillomavirus infection among young Swedish women. NPJ Biofilms Microbiomes 6, 39. doi: 10.1038/s41522-020-00146-8
Coudray, M. S., Madhivanan, P. (2020). Bacterial vaginosis-A brief synopsis of the literature. Eur. J. Obstet. Gynecol. Reprod. Biol. 245, 143–148. doi: 10.1016/j.ejogrb.2019.12.035
Coudray, M. S., Sheehan, D. M., Li, T., Cook, R. L., Schwebke, J., Madhivanan, P. (2020). Factors associated with the recurrence, persistence, and clearance of asymptomatic bacterial vaginosis among young African American women: A repeated-measures latent class analysis. Sex. Transm. Dis. 47, 832–839. doi: 10.1097/OLQ.0000000000001256
Drew, R. J., Murphy, T., Broderick, D., O’Gorman, J., Eogan, M. (2020). An interpretation algorithm for molecular diagnosis of bacterial vaginosis in a maternity hospital using machine learning: proof-of-concept study. Diagn. Microbiol. Infect. Dis. 96, 114950. doi: 10.1016/j.diagmicrobio.2019.114950
Elnaggar, J. H., Ardizzone, C. M., Cerca, N., Toh, E., Łaniewski, P., Lillis, R. A., et al. (2023). A novel Gardnerella, Prevotella, and Lactobacillus standard that improves accuracy in quantifying bacterial burden in vaginal microbial communities. Front. Cell Infect. Microbiol. 13. doi: 10.3389/fcimb.2023.1198113
Fredricks, D. N., Fiedler, T. L., Marrazzo, J. M. (2005). Molecular identification of bacteria associated with bacterial vaginosis. N. Engl. J. Med. 353, 1899–1911. doi: 10.1056/NEJMoa043802
Gustin, A., Cromarty, R., Schifanella, L., Klatt, N. R. (2021). Microbial mismanagement: how inadequate treatments for vaginal dysbiosis drive the HIV epidemic in women. Semin. Immunol. 51, 101482. doi: 10.1016/j.smim.2021.101482
Hilbert, D. W., Smith, W. L., Paulish-Miller, T. E., Chadwick, S. G., Toner, G., Mordechai, E., et al. (2016). Utilization of molecular methods to identify prognostic markers for recurrent bacterial vaginosis. Diagn. Microbiol. Infect. Dis. 86, 231–242. doi: 10.1016/j.diagmicrobio.2016.07.003
Javed, A., Parvaiz, F., Manzoor, S. (2019). Bacterial vaginosis: An insight into the prevalence, alternative treatments regimen and it’s associated resistance patterns. Microb. Pathog. 127, 21–30. doi: 10.1016/j.micpath.2018.11.046
Jones, A. (2019). Bacterial vaginosis: A review of treatment, recurrence, and disparities. J. Nurse Pract. 15, 420–423. doi: 10.1016/j.nurpra.2019.03.010
Kaur, H., Merchant, M., Haque, M. M., Mande, S. S. (2020). Crosstalk between female gonadal hormones and vaginal microbiota across various phases of women’s gynecological lifecycle. Front. Microbiol. 11. doi: 10.3389/fmicb.2020.00551
Lynch, T., Peirano, G., Lloyd, T., Read, R., Carter, J., Chu, A., et al. (2019). Molecular diagnosis of vaginitis: comparing quantitative PCR and microbiome profiling approaches to current microscopy scoring. J. Clin. Microbiol. 57, e00300–19. doi: 10.1128/JCM.00300-19
Malaguti, N., Bahls, L. D., Uchimura, N. S., Gimenes, F., Consolaro, M. E. L. (2015). Sensitive detection of thirteen bacterial vaginosis-associated agents using multiplex polymerase chain reaction. BioMed. Res. Int. 2015, 645853. doi: 10.1155/2015/645853
Matu, M. N., Orinda, G. O., Njagi, E. N. M., Cohen, C. R., Bukusi, E. A. (2010). In vitro inhibitory activity of human vaginal lactobacilli against pathogenic bacteria associated with bacterial vaginosis in Kenyan women. Anaerobe 16, 210–215. doi: 10.1016/j.anaerobe.2009.11.002
Menard, J. P., Fenollar, F., Henry, M., Bretelle, F., Raoult, D. (2008). Molecular quantification of Gardnerella vaginalis and Atopobium vaginae loads to predict bacterial vaginosis. Clin. Infect. Dis. 47, 33–43. doi: 10.1086/588661
Mondal, A. S., Sharma, R., Trivedi, N. (2023). Bacterial vaginosis: A state of microbial dysbiosis. Med. Microecol. 16, 100082. doi: 10.1016/j.medmic.2023.100082
Muñoz-Barreno, A., Cabezas-Mera, F., Tejera, E., MaChado, A. (2021). Comparative effectiveness of treatments for bacterial vaginosis: A network meta-analysis. Antibiot (Basel Switzerland) 10, 978. doi: 10.3390/antibiotics10080978
Muzny, C. A., Cerca, N., Elnaggar, J. H., Taylor, C. M., Sobel, J. D., van der Pol, B. (2023). State of the art for diagnosis of bacterial vaginosis. J. Clin. Microbiol. 61, e00837-22. doi: 10.1128/jcm.00837-22
Muzny, C. A., Łaniewski, P., Schwebke, J. R., Herbst-Kralovetz, M. M. (2020). Host-vaginal microbiota interactions in the pathogenesis of bacterial vaginosis. Curr. Opin. Infect. Dis. 33, 59–65. doi: 10.1097/QCO.0000000000000620
Naidoo, K., Abbai, N., Tinarwo, P., Sebitloane, M. (2022). BV associated bacteria specifically BVAB 1 and BVAB 3 as biomarkers for HPV risk and progression of cervical neoplasia. Infect. Dis. Obstet. Gynecol. 2022, 9562937. doi: 10.1155/2022/9562937
Navarro, S., Abla, H., Delgado, B., Colmer-Hamood, J. A., Ventolini, G., Hamood, A. N. (2023). Glycogen availability and pH variation in a medium simulating vaginal fluid influence the growth of vaginal Lactobacillus species and Gardnerella vaginalis. BMC Microbiol. 23, 1–20. doi: 10.1186/s12866-023-02916-8
Nugent, R. P., Krohn, M. A., Hillier, S. L. (1991). Reliability of diagnosing bacterial vaginosis is improved by a standardized method of gram stain interpretation. J. Clin. Microbiol. 29, 297–301. doi: 10.1128/jcm.29.2.297-301.1991
Onderdonk, A. B., Delaney, M. L., Fichorova, R. N. (2016). The human microbiome during bacterial vaginosis. Clin. Microbiol. Rev. 29, 223–238. doi: 10.1128/CMR.00075-15
Osei Sekyere, J., Maningi, N. E., Fourie, P. B. (2020). Mycobacterium tuberculosis, antimicrobials, immunity, and lung–gut microbiota crosstalk: current updates and emerging advances. Ann. N. Y Acad. Sci. 1467, 21–47. doi: 10.1111/nyas.14300
Osei Sekyere, J., Oyenihi, A. B., Trama, J., Adelson, M. E. (2023). Species-specific analysis of bacterial vaginosis-associated bacteria. Microbiol. Spectr. 11, e04676-22. doi: 10.1128/spectrum.04676-22
Pacha-Herrera, D., Erazo-Garcia, M. P., Cueva, D. F., Orellana, M., Borja-Serrano, P., Arboleda, C., et al. (2022). Clustering analysis of the multi-microbial consortium by lactobacillus species against vaginal dysbiosis among Ecuadorian women. Front. Cell Infect. Microbiol. 12. doi: 10.3389/fcimb.2022.863208
Peebles, K., Velloza, J., Balkus, J. E., McClelland, R. S., Barnabas, R. V. (2019). High global burden and costs of bacterial vaginosis: A systematic review and meta-analysis. Sex. Transm. Dis. 46, 304–311. doi: 10.1097/OLQ.0000000000000972
Powell, A. M., Sarria, I., Goje, O. (2023). Microbiome and vulvovaginitis. Obstet. Gynecol. Clin. North Am. 50, 311–326. doi: 10.1016/j.ogc.2023.02.005
Roachford, O. S. E., Alleyne, A. T., Nelson, K. E. (2022). Insights into the vaginal microbiome in a diverse group of women of African, Asian and European ancestries. PeerJ 10, e14449. doi: 10.7717/peerj.14449
Schmidt, K., Cybulski, Z., Roszak, A., Grabiec, A., Talaga, Z., Urbański, B., et al. (2015). Combination of microbiological culture and multiplex PCR increases the range of vaginal microorganisms identified in cervical cancer patients at high risk for bacterial vaginosis and vaginitis. Ginekol Pol. 86, 328–334. doi: 10.17772/gp/2417
Shen, L., Zhang, W., Yuan, Y., Zhu, W., Shang, A. (2022). Vaginal microecological characteristics of women in different physiological and pathological period. Front. Cell Infect. Microbiol. 12. doi: 10.3389/fcimb.2022.959793
Sobel, J. D., Vempati, Y. S. (2024). Bacterial vaginosis and vulvovaginal candidiasis pathophysiologic interrelationship. Microorganisms 12, 108. doi: 10.3390/microorganisms12010108
Swidsinski, S., Moll, W. M., Swidsinski, A. (2023). Bacterial vaginosis—Vaginal polymicrobial biofilms and dysbiosis. Dtsch Arztebl Int. 120, 347. doi: 10.3238/arztebl.m2023.0090
Unemo, M., Bradshaw, C. S., Hocking, J. S., de Vries, H. J. C., Francis, S. C., Mabey, D., et al. (2017). Sexually transmitted infections: challenges ahead. Lancet Infect. Dis. 17, e235–e279. doi: 10.1016/S1473-3099(17)30310-9
van den Munckhof, E. H. A., van Sitter, R. L., Boers, K. E., Lamont, R. F., te Witt, R., le Cessie, S., et al. (2019). Comparison of Amsel criteria, Nugent score, culture and two CE-IVD marked quantitative real-time PCRs with microbiota analysis for the diagnosis of bacterial vaginosis. Eur. J. Clin. Microbiol. Infect. Dis. 38, 959–966. doi: 10.1007/s10096-019-03538-7
Vodstrcil, L. A., Muzny, C. A., Plummer, E. L., Sobel, J. D., Bradshaw, C. S. (2021). Bacterial vaginosis: drivers of recurrence and challenges and opportunities in partner treatment. BMC Med. 19, 1–12. doi: 10.1186/s12916-021-02077-3
Watkins, E., Chow, C. M., Lingohr-Smith, M., Lin, J., Yong, C., Tangirala, K., et al. (2024). Treatment patterns and economic burden of bacterial vaginosis among commercially insured women in the USA. J. Comp. Eff Res. 13, e230079. doi: 10.57264/cer-2023-0079
Wei, X., Tsai, M.-S., Liang, L., Rand, L., Snyder, M., Jiang, C., et al. (2024). Vaginal microbiomes show ethnic evolutionary dynamics and positive selection of Lactobacillus adhesins driven by a long-term niche-specific process. CellReports 43, 114078. doi: 10.1016/j.celrep.2024.114078
Weyers, S., Verstraelen, H., Gerris, J., Monstrey, S., Lopes dos Santos Santiago, G., Saerens, B., et al. (2009). Microflora of the penile skin-lined neovagina of transsexual women. BMC Microbiol. 9, 102. doi: 10.1186/1471-2180-9-102
Wu, S., Hugerth, L. W., Schuppe-Koistinen, I., Du, J. (2022). The right bug in the right place: opportunities for bacterial vaginosis treatment. NPJ Biofilms Microbiomes 81 2022, 8:1–811. doi: 10.1038/s41522-022-00295-y
Zhang, Q., Chen, R., Li, M., Zeng, Z., Zhang, L., Liao, Q. (2022). The interplay between microbiota, metabolites, immunity during BV. Med. Microecol 11, 100049. doi: 10.1016/j.medmic.2021.100049
Keywords: bacterial vaginosis (BV), MDL-BV index, vaginitis, qRT-PCR, BVAB, vaginal microbiome, machine learning
Citation: Oyenihi AB, Haines R, Trama J, Faro S, Mordechai E, Adelson ME and Osei Sekyere J (2024) Molecular characterization of vaginal microbiota using a new 22-species qRT-PCR test to achieve a relative-abundance and species-based diagnosis of bacterial vaginosis. Front. Cell. Infect. Microbiol. 14:1409774. doi: 10.3389/fcimb.2024.1409774
Received: 31 March 2024; Accepted: 07 June 2024;
Published: 28 June 2024.
Edited by:
Edwin Kamau, Tripler Army Medical Center, United StatesReviewed by:
António Machado, Universidad San Francisco de Quito, EcuadorAddy Cecilia Helguera-Repetto, Instituto Nacional de Perinatología (INPER), Mexico
Copyright © 2024 Oyenihi, Haines, Trama, Faro, Mordechai, Adelson and Osei Sekyere. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Ayodeji B. Oyenihi, YW95ZW5paGlAbWRsYWIuY29t; Jason Trama, anRyYW1hQGlici1nZW5ldGljcy5jb20=; John Osei Sekyere, am9zZWlzZWt5ZXJlQG1kbGFiLmNvbQ==
†These authors have contributed equally to this work
 Ayodeji B. Oyenihi
Ayodeji B. Oyenihi Ronald Haines
Ronald Haines Jason Trama1*
Jason Trama1* Sebastian Faro
Sebastian Faro John Osei Sekyere
John Osei Sekyere