- 1Clinical Laboratory Center, Shaoxing People’s Hospital, Shaoxing, Zhejiang, China
- 2Department of Hospital Management, Shaoxing People’s Hospital, Shaoxing, Zhejiang, China
- 3Department of Endocrine and Metabolism, Shaoxing People’s Hospital, Shaoxing, Zhejiang, China
Background: Hypoglycaemia has been linked to an increased risk of cardiac arrhythmias by causing autonomic and metabolic alterations, which may be associated with detrimental outcomes in individuals with diabetes(IWD), such as cardiovascular diseases (CVDs) and mortality, especially in multimorbid or frail people. However, such relationships in this population have not been thoroughly investigated. For this reason, we conducted a systematic review and meta-analysis.
Methods: Relevant papers published on PubMed, Embase, Cochrane, Web of Knowledge, Scopus, and CINHAL complete from inception to December 22, 2022 were routinely searched without regard for language. All of the selected articles included odds ratio, hazard ratio, or relative risk statistics, as well as data for estimating the connection of hypoglycaemia with cardiac arrhythmia, CVD-induced death, or total death in IWD. Regardless of the heterogeneity assessed by the I2 statistic, pooled relative risks (RRs) and 95% confidence intervals (CI) were obtained using random-effects models.
Results: After deleting duplicates and closely evaluating all screened citations, we chose 60 studies with totally 5,960,224 participants for this analysis. Fourteen studies were included in the arrhythmia risk analysis, and 50 in the analysis of all-cause mortality. Hypoglycaemic patients had significantly higher risks of arrhythmia occurrence (RR 1.42, 95%CI 1.21-1.68), CVD-induced death (RR 1.59, 95% CI 1.24-2.04), and all-cause mortality (RR 1.68, 95% CI 1.49-1.90) compared to euglycaemic patients with significant heterogeneity.
Conclusion: Hypoglycaemic individuals are more susceptible to develop cardiac arrhythmias and die, but evidence of potential causal linkages beyond statistical associations must await proof by additional specifically well planned research that controls for all potential remaining confounding factors.
Introduction
Diabetes, a non-communicable disease, is a serious public health concern worldwide. More than half a billion people (536.6 million) aged 20-79 were predicted to develop diabetes mellitus (DM) in 2021, with roughly 90% having type 2 DM (T2DM) (1). Diabetes is a serious, chronic disease that profoundly affects the lives and well-being of individuals, families, and communities throughout the world. Diabetes is one of the leading ten causes of adult mortality, with a total estimated global healthcare expenditure of 966 billion USD (1).
Cardiovascular diseases (CVDs) are the most common cause of morbidity and mortality in this populace (2). Numerous large epidemiological studies demonstrate that maintaining target glycemic control is the only proven strategy for preventing diabetic vascular complications (3). To accomplish this goal, active use of oral diabetes medications or insulin as well as lifestyle modifications in accordance with the individual situation shall be initiated after the diagnosis of diabetes (4–6). However, intensive glycaemic control with antidiabetic drugs inevitably exposes IWD to the common side effect of hypoglycaemia (7–10). Recent evidence confirms that a hypoglycaemic episode, irrespective of severity, is clinically important because of its link to increased arrhythmias (via effects on cardiac repolarisation and alterations in cardiac autonomic activity, hypokalemia due to excess of insulin and increased secretion of catecholamines, which might drive potassium into the cell during hypoglycemia, and fuel energy shortage at the level of the cardiomyocyte due to low availability of glucose despite stress and increased demand as well (11, 12)) and other cardiovascular (CV) events and mortality (13–18). Similarly, a recent systematic review (19) reports that hypoglycaemia leads to electrocardiogram changes associated with a greater likelihood of cardiac arrhythmias, which are related to increased CV events and mortality. Another systematic review (20) concludes that hypoglycaemia is a risk factor for adverse vascular events and death. Nevertheless, contradictory evidence exists regarding the association between hypoglycaemia and the risks of cardiac arrhythmias and mortality (21–24). The aforementioned reviews have numerous limitations, including a lack of subgroup analysis, and a failure to recognise heterogeneity. Specifically, some undetected confounders (e.g. study design, hypoglycaemia criteria, ignorance of model adjustment, and dissimilar comorbidity profile) that are ignored by model adjustment can result in bias. Meanwhile, some studies (25) show that only spontaneous hypoglycaemia is associated with increased mortality, while others (26, 27) show no effect. Based on the available evidences, several authors (28, 29) even suggest that hypoglycaemia is a marker of disease severity and/or comorbidity burden in hospitalised patients.
For these reasons, our aim was to assess if hypoglycaemia affects the risk of arrhythmia or mortality in IWD.
Methods
Search strategy and criteria for inclusion
Two separate reviewers searched PubMed, EMBASE, Wiley Cochrane Central Register of Controlled Trials(CENTRAL), Web of knowledge, Scopus and CINAHL complete up to December 22, 2022 as per applicable recommendations (30). Any disagreement between them was resolved through consensus. In addition to keywords, we used Medical Subject Headings terms in PubMed, EMTREE terms in EMBASE, CINAHL headings in CINAHL, and keywords in all included databases as a part of our strategy. The search terms were “hypoglycaemia”, “hypoglycemia” or “hypogly*” and “diabetes” or “diabet*”, and “arrhythmias” or “arrhythmia*” or “dysrhythmia*” or “mortality” or “mortalit*” or “death” (search technique shown in Additional file 1). The grey literature was found by manually searching reference lists from eligible research and associated reviews. Authors were contacted to obtain necessary data. Prior to the start of this investigation, no review protocol for this systematic review has been published or registered.
The inclusion criteria were: (i) observational studies (OSs) [cohort, case-control, cross-sectional, and longitudinal studies], post-hoc analysis or sub-analysis of randomised controlled trials (p-h/sa of RCTs); (ii) focusing on the relationship of any hypoglycaemia with arrhythmias or CVD-induced death or overall death in IWD, and reporting risk or prevalence or incidence of arrhythmias (mortality) for IWD with hypoglycaemia as compared with euglycaemia, and (iii) standardized mortality or incidence ratio (SMR or SIR), or incidence rate ratio (IRR), or odds ratios (OR), or hazard ratio (HR), or relative risk (RR), as well as pertinent relevant raw data for recalculation are all used to report effect estimates. Exclusion criteria were as follows: (I) emphasizing the connection of hypoglycaemia with adverse outcomes such as arrhythmias- or CVD-caused death or all-cause mortality in non-diabetic people; (II) case report, quasiexperiment (in which subjects are not assigned at random), editorial, remark, review, letter or unpublished study; (III) only published as abstract or conference proceeding. Diabetes studies that did not provide such estimations were also omitted. When a site-specific dataset was published multiple times, the study with the newest t publication or the largest sample size was generally picked.
Data extraction and quality evaluation
Data were collected about the research (design, first author’s name, title, year of publication, data source, country/region, baseline years, sample size, follow-up duration, diabetes clarity, endpoints, measure of relationship, number of observed and expected events), participants (mean age and the gender), analysis strategy (statistical models, adjustment factors), effect size (e.g., SMR or IRR, or SIR, or HRs, RRs or ORs), as well as pertinent raw data for recalculation.
Two researchers independently assessed the quality of each p-h/sa of RCT and enrolled study based on cohort or case-control design utilizing the 9-star Newcastle-Ottawa Scale (NOS) (31). A rating of above six stars (31) indicates high quality. The 11-item Agency for Healthcare Research and Quality (AHRQ) checklist is recommended for assessing the quality of cross-sectional or longitudinal studies (32, 33). Each item’s responses are “yes,” “no,” and “unclear”. If the response to each question is “yes”, one point is awarded; otherwise, no points are given. Studies with a total score of 0–5, 6–7, 8–11, respectively, were deemed to be of low, moderate and high quality. Disagreements were resolved through dialogue.
Statistical analysis
The main and second goals were the hazards of arrhythmias and overall death (CVD death) whereas the analysis period, respectively. OR, or RR with 95% confidence interval (CI) was used to summarize dichotomous outcomes. ORs are comparable to RRs since the absolute risk of arrhythmias or death is minimal in these populations and the two indices provide equivalent RR estimates (34). The I2 statistic was used to determine the fraction of variability between studies because of heterogeneity between studies, and with I2 values greater than 50%, 25-50%, and less than 25%, was classified as high, medium, and low respectively (35). The iterative non-central Chi-2 test was used to find a CI for I2 (36).
Concerning the antecedent discrepancy of p-h/sa of RCT and OSs, we performed subgroup analyses for arrhythmia by sample size (<1000, and ≥1000), diurnal differences (nocturnal hypoglycaemia, and daytime hypoglycaemia), severity of hypoglycaemia (severe, and total), clarity of diabetes (T1DM, T2DM, and total), and type of arrhythmia (incidence of QTc interval prolongation, and other arrhythmia). For all-cause mortality analysis, subgroup analyses were conducted with sample size, country/region (developed, developing, and total), study population (simple diabetes, and diabetes with other disease or high risk for CV disease), hypoglycaemia episode (1, and ≥2), clarity of diabetes, study design (OSs, and p-h/sa of RCT), severity of hypoglycaemia, follow-up duration (≤1 year [including in-hospital mortality, and ICU mortality], and >1 year), and methods for effect estimate extraction (reported and calculated). Any quantifiable source of heterogeneity was identified through sensitivity analysis by excluding each study individually.
Publication bias was examined using Begg’s and Egger’s tests when at least five studies were available for analysis, as well as by visually inspecting the asymmetry of funnel plots of estimated effects against standard errors (37). Any publication bias (P<0.10) was corrected using Duval & Tweedie’s trim-and-fill approach. All other analyses were conducted using STATA 14.0 (US) at P < 0.05 significance level.
Results
Study identification
From the 51,794 articles discovered through a systematic search, 137 were selected for further analysis (Figure 1). Two articles (24, 38) from one data source presented conflicting findings, and thus were both included. Four studies (22, 39–41) each two from the same teams/institutions and each reporting different outcomes for analysis were included. Finally, 60 articles provided data on the relationship between hypoglycaemia and arrhythmias or mortality (Table 1). The 60 studies included 11 p-h/sa RCTs (13, 21, 22, 41, 51, 75–77, 82, 86, 91), and 50 OSs (41 cohort studies (14, 23, 24, 39, 40, 43–45, 47–50, 52, 53, 55–65, 67–70, 72, 73, 78–81, 83, 85, 87–90) 3 (nested) case-control studies (54, 71, 84), 3 cross-sectional studies (38, 42, 74) 1 longitudinal study (66) and one study (46) including both cohort and nested case-control studies). The sample sizes ranged from 25 to 2,032,689 patients, and the mean age of patients ranged between 25.6 and 81.86 years. The female proportion ranged from 2.90% to 59.27%, and duration of DM was from 1.36 to 44.25 years.
Apart from 10 studies on multinational origins, other origins were mentioned developed countries or regions in 46 articles, and developing countries in 4 articles. More than 50% studies (34/60) reported severe hypoglycaemia. Sixteen articles reported IWD with comorbidities or at high risk of CVD. Definitions of hypoglycaemia varied widely and were mostly based on blood glucose concentration or the presence of symptoms necessitating the assistance of another individual or medical help. Ten, 46, and 15 articles presented data on arrhythmia risk only, all-cause mortality only, and CVD death only, respectively, and four studies reported data on both arrhythmia and all-cause mortality risk.
Quality evaluation
For cardiac arrhythmia analysis, quality analysis showed about 7 of 11 cohort studies and one case-control study were of high methodological quality, with ≥7 NOS scores (mean= 7.58; Table S1). One p-h/sa of RCT showed high quality according to NOS scores. Two cross-sectional studies were of moderate quality according to AHRQ (Table S1). When analysing the risk of all-cause mortality in 46 studies, 24 cohort studies and 2 case-control studies were of high methodological quality, with ≥7 NOS scores (mean= 7.73; Table S1). According to AHRQ, one cross-sectional study and one longitudinal research were of moderate quality. All 10 p-h/sas of RCTs were of high quality (mean NOS score= 8.50; Table S1).
Risk of cardiac arrhythmia
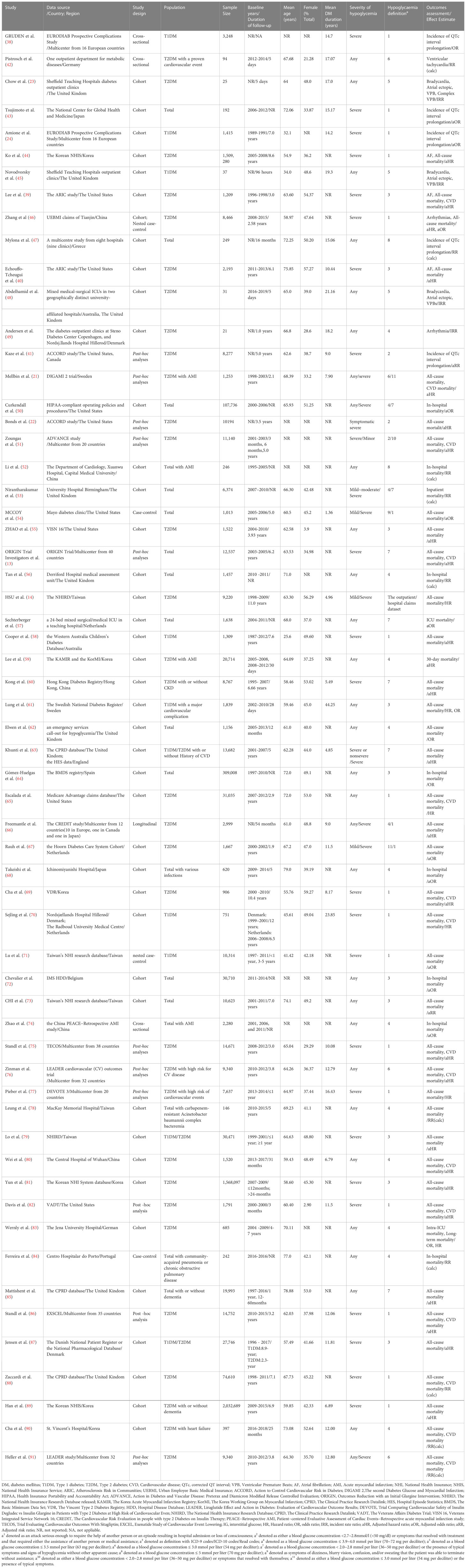
The analysis of the connection between hypoglycemia and the risk of arrhythmia involved fourteen relevant studies. Overall, the pooled RR showed a 42% greater incidence of cardiac arrhythmia in IWD with versus without hypoglycemia (RR,1.42; 95%CI,1.21-1.68; p<0.001), but there was evident heterogeneity between studies (I2 = 71.7%, p<0.001) (Figure 2). Sensitivity analysis revealed that heterogeneity did not vanish after single studies were removed. In the arrhythmia risk analysis, Funnel plots revealed no evidence of systematic bias (Begg’s test, P=0.443; Egger’s test, P=0.245) (Figure S1).
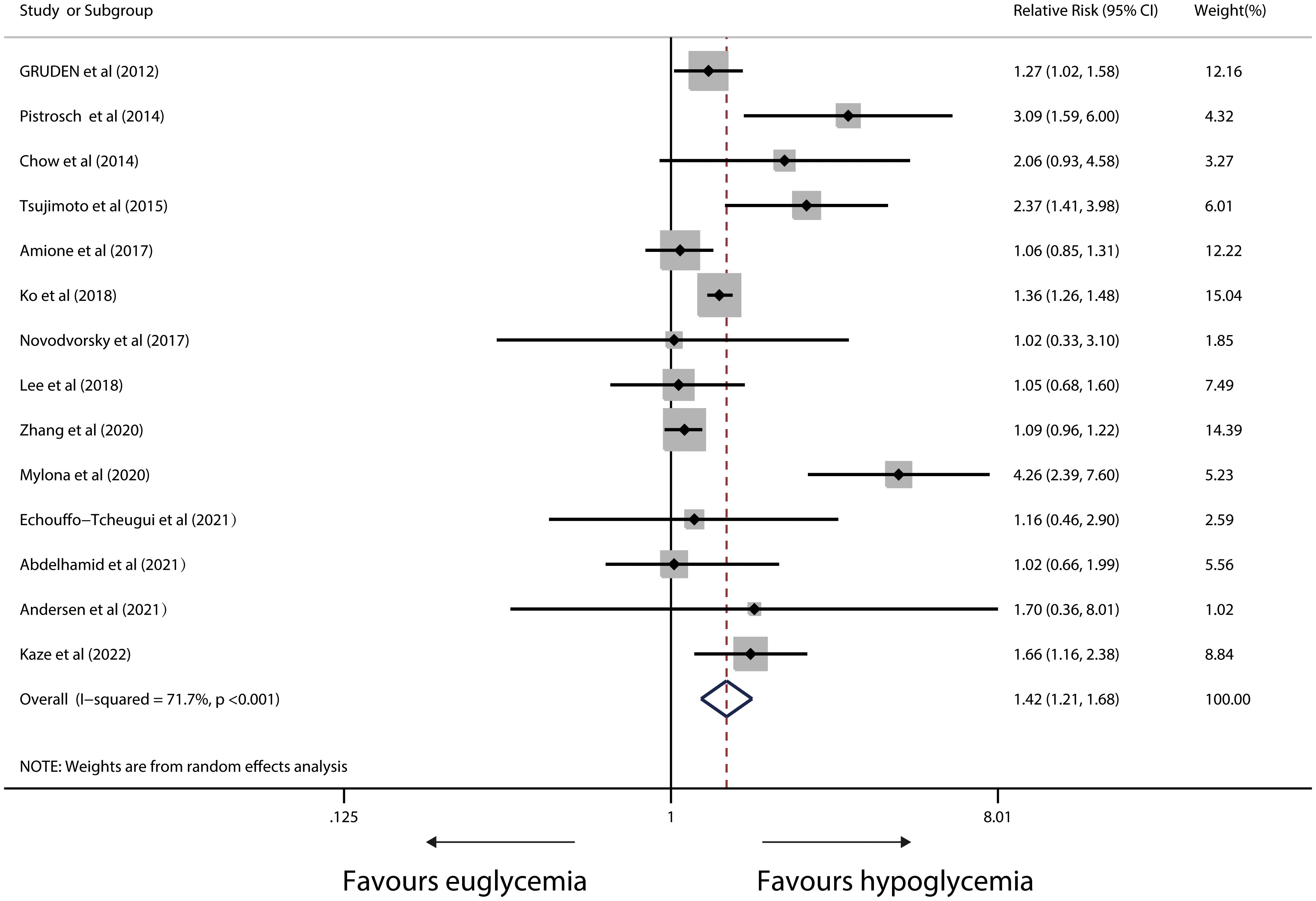
Figure 2 Forest plot for the association between hypoglycaemia with risk of cardiac arrhythmia in diabetic patients (X-axis: log scale; solid square: relative risk; horizontal lines: 95% CIs. The same in other figures).
The pooled RRs were essentially uniform regardless of the sample size (<1000 patients, P=0.001; ≥1000 patients, P=0.001), severity of hypoglycaemia (severe hypoglycaemia P=0.001; total hypoglycaemia, p=0.014), noctural hypoglycaemia (P=0.012), type of arrhythmia (incidence of QTc interval prolongation, P=0.004; other arrhythmia, P=0.007), T2DM (P =0.001), or total diabetes (P<0.001) (Figure 3).
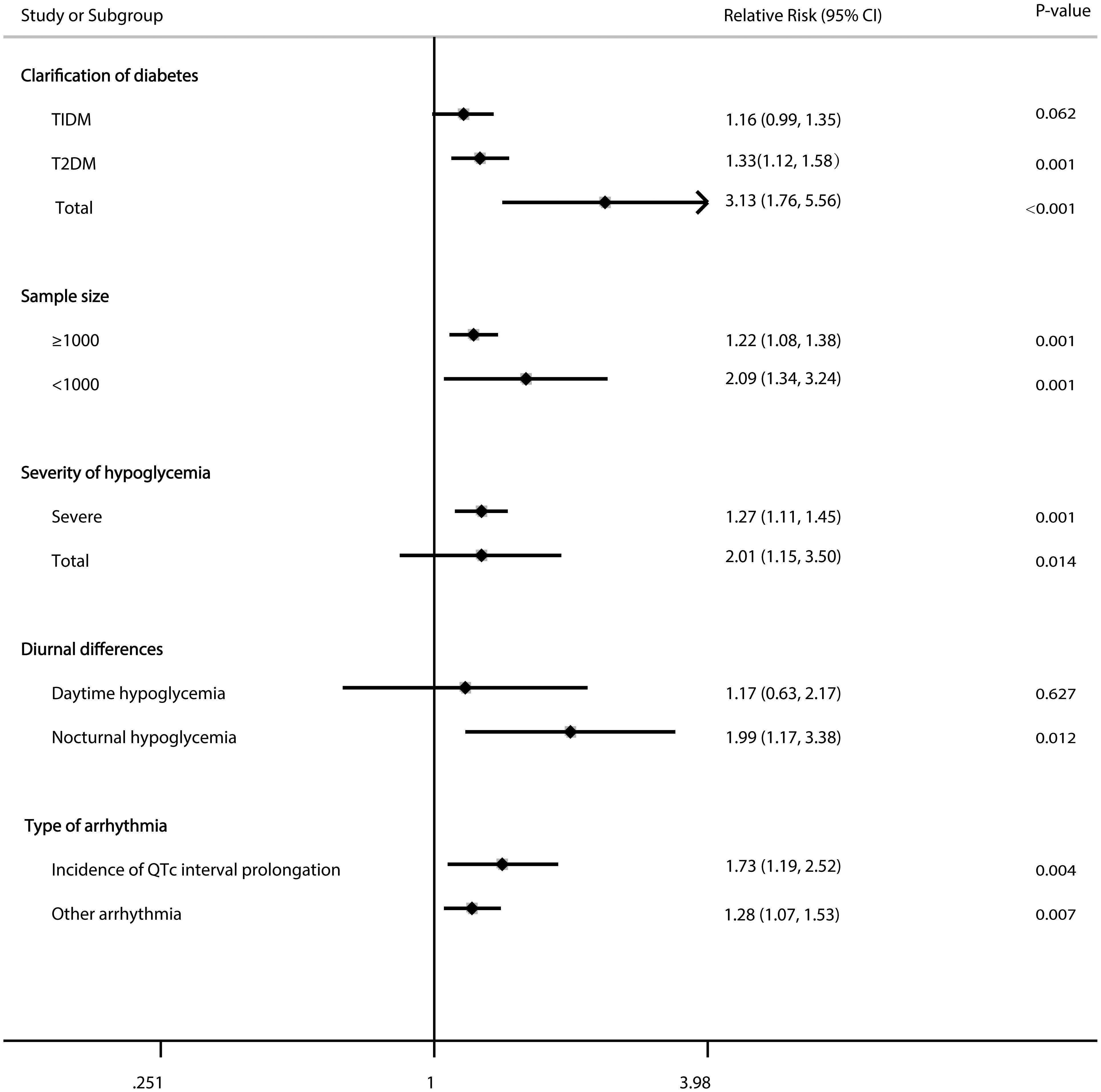
Figure 3 Forest plot for the association between hypoglycaemia with risk of cardiac arrhythmia in diabetic patients according to some clinically important variables.
Risk of all-cause mortality
Fifty studies reported a link between hypoglycemia and overall death in IWD. In a random-effects model, the pooled RR was 1.68 (95%CI, 1.49 to 1.90; P<0.001) with severe heterogeneity (I2 = 97.1%; P<0.001; Figure 4).
Figure 4 Forest plot for the association between hypoglycaemia with risk of all-cause mortality in diabetic patients.
A sensitivity analysis revealed that heterogeneity did not vanish after removing single studies. Neither Egger’s test (P = 0.514) nor visual inspection revealed significant publication bias (Figure S2).
Estimated sample size, follow-up duration, country/region, study population, clarity of diabetes, study design, severity of hypoglycaemia, hypoglycaemia episode and methodologies for effect estimate extraction yielded similar results (Figure 5).
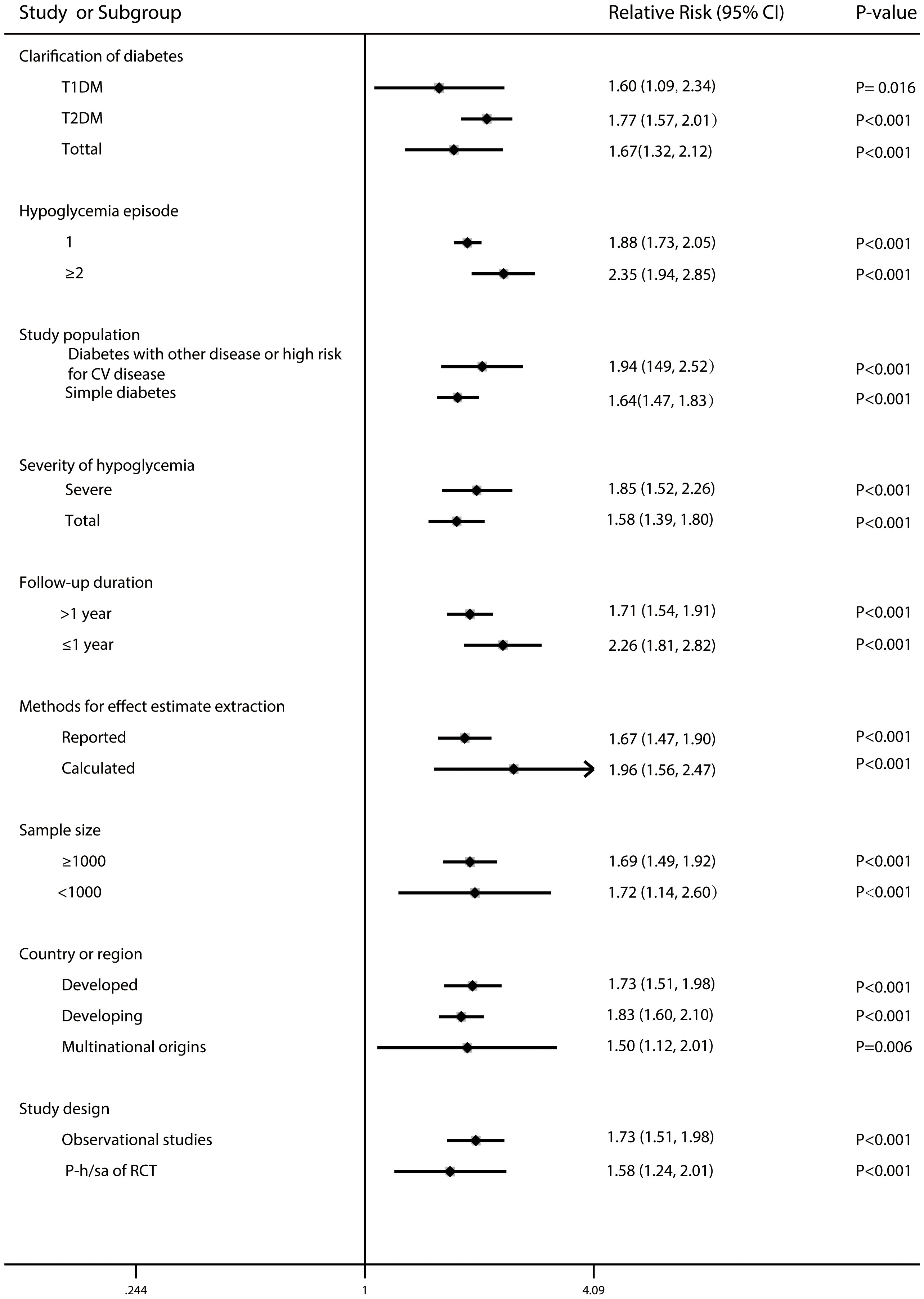
Figure 5 Forest plot for the association between hypoglycaemia with risk of all-cause mortality in diabetic patients according to some clinically important variables.
Risk of CVD death
A random-effects model exhibiting substantial heterogeneity (I2= 80.8%; P<0.001) from 15 relevant studies found that hypoglycemic versus euglycaemic IWD had a 59% significantly higher risk (pooled RR, 1.59; 95%CI, 1.24-2.04; P<0.001) in susceptibility to CVD-caused death. Figure 6 depicts meta-analysis forest plots. A sensitivity analysis revealed that heterogeneity did not vanish after single studies were removed. Tests by Begg’s and Egger’s tests showed no clear systematic bias in the CVD-induced death risk analyses (both P > 0.1; Figure S3).
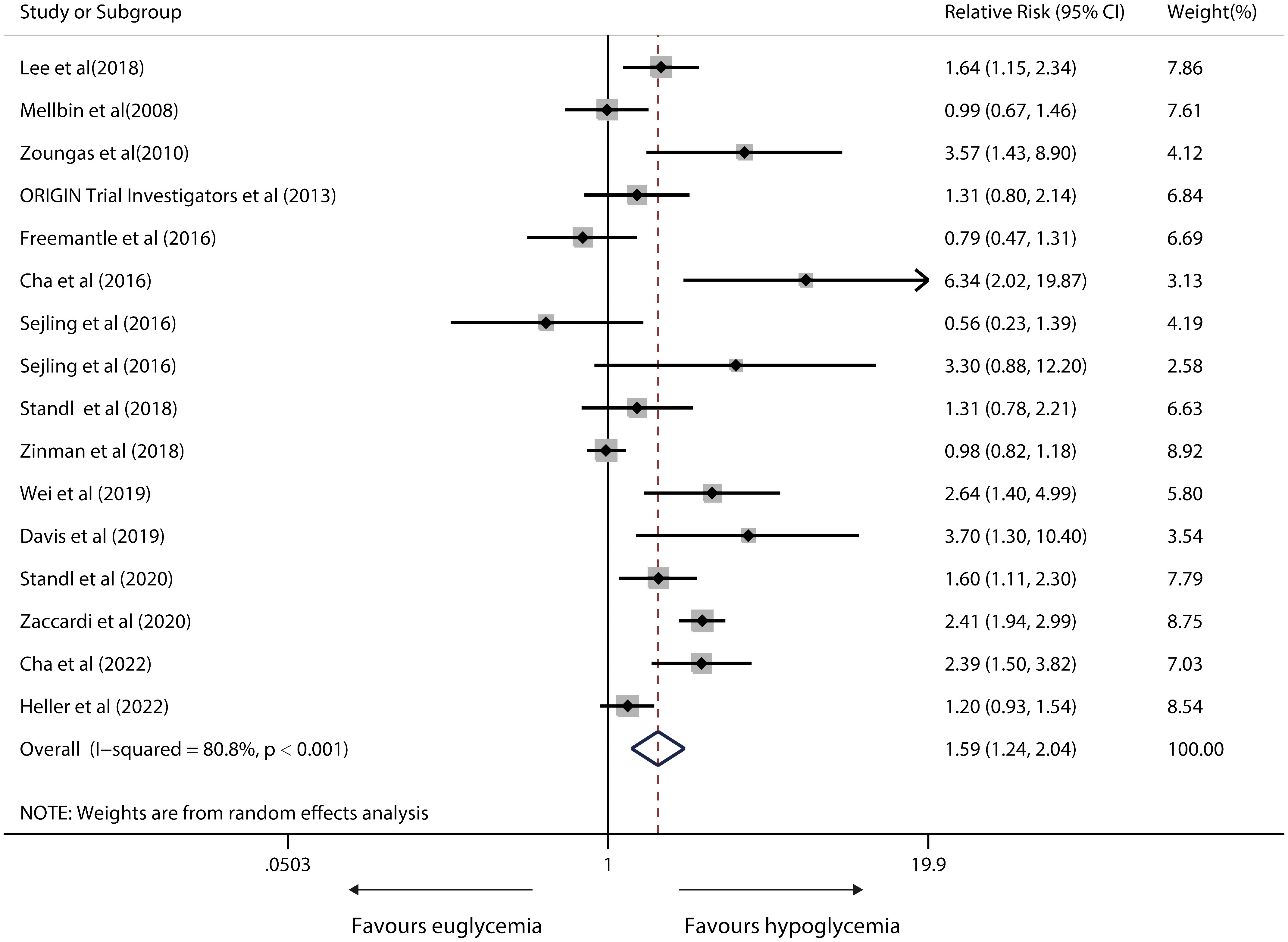
Figure 6 Forest plot for the association between hypoglycaemia with risk of CVD death in diabetic patients according to some clinically important variables.
Discussion
All current studies with 5,960,224 individuals were reviewed to find out evidence of a link between hypoglycemia and negative consequences, including cardiac arrhythmia, overall death, and CVD-induced death. We did not aim to establish a direct relationship, but did highlight the importance of hypoglycemia in relation to CV events and death. It is generally recognised that hypoglycemia, particularly severe hypoglycemia (SH), carries a greater proarrhythmic risk than euglycemia or hyperglycemia, as also observed in this study. The proarrhythmic mechanisms of hypoglycaemia, especially SH, may be various (92). Low glucose directly affects the human Ether-à-go-go Related Gene ion channel (93). Hypokalemia, and catecholamine both delay cardiac repolarisation, increasing the risk of early afterdepolarisations and ventricular arrhythmias. QT prolongation throughout the day may further trigger early afterdepolarisations. In the same way, activation of the sympathetic nervous system and an increase in calcium in the cytosol can lead to delayed afterdepolarisation and early heartbeats, which contribute to more ventricular ectopic activity during the day. Nevertheless, cardiac arrhythmias induced by hypoglycemia exhibit diurnal changes, which were also discovered in our study by subgroup analyses. Arrhythmia rates are even higher at night or early morning, when sympathoadrenal responses are suppressed (94). A sluggish sinus rate at a state of vagal dominance may disclose latent pacemakers (especially under situations of heightened automaticity), which causes excessive atrial and ventricular ectopic activity during nocturnal hypoglycaemia (45).
Even though the overall pooled RR suggests a 42% substantially higher likelihood of cardiac arrhythmia in hypoglycemic patients over euglycaemia patients, around 57% (8/14) recruited studies report no significant link between hypoglycemia and cardiac arrhythmia. There can be several reasons for this null result. First, the antagonistic autonomic nerve responses are a fundamental mechanism underlying the harmful effect of SH, but can be diminished in patients with long-standing diabetes, long follow-up, or established cardiovascular risks (95–97). Considering the involved patients here have relatively longer follow-up time, the effect of hypoglycemia is most likely obscured by the highly frequent cardiovascular and cerebrovascular comorbidities at baseline. Amione et al. (24) investigated the possible impact of repeated bouts of SH as a marker of QTc prolonged QTc interval over a seven-year period, and concluded that the impact of prediction could be due to adaptive mechanisms (98, 99). Second, earlier research revealed a dose-response or linear relationship between hypoglycemia and various unfavorable outcomes (71, 100), though only two studies (24, 41) were included. Third, individuals with repeated hypoglycemic episodes typically have a diminished awareness of hypoglycemia (101), which explains why there is no apparent temporal link between hypoglycemia and arrhythmia.
As for the mortality analysis, we did not aim to prove causality, but did highlight the associations between hypoglycemia and death. Due to pre-existing comorbidities, CVD-induced death usually accounted for the majority of deaths in the diabetes group. The relationship between hypoglycaemia and cardiovascular events is bidirectional in accordance with previous studies (75, 86). Firstly, we discovered a link between hypoglycemia and all-cause mortality in IWD, with a higher all-cause mortality in individuals with hypoglycemia compared to those without. The underlying biological mechanisms directly linking hypoglycaemia to death have yet to be elucidated. Nevertheless, some established scientific hypotheses suggest the fundamental mechanisms are primarily sympathoadrenal activation, cardiac or cerebral ischemia, abnormal cardiac repolarisation, QT prolongation, or fatal arrhythmia or autonomic impairment, increased thrombogenesis, inflammation, endothelial dysfunction, and vasoconstriction caused by aberrant endocrine or nervous reactions during recognised or unrecognised hypoglycaemic episodes (11, 102–113). All of these alterations can affect heart anatomy, cardiac stress, vascular hemodynamics, and function (13, 114–118). Episodes of asymptomatic or unreported symptomatic hypoglycaemia probably occur after the initial occurrence of SH, increasing the likelihood of recurrent incidents. Although these effects are true, they are more likely to explain a temporary increase in CV risk at the acute phase of hypoglycaemia rather than the long-term connection observed in another study (63).
Secondly, hypoglycaemia or SH is a plausible indicator instead of a direct cause of an elevated risk of unfavorable clinical outcomes, which reflects the influence of concomitant illnesses in addition to unmeasured or incompletely defined confounding variables (119) that independently increase the risk of mortality as opposed to being a risk factor causing these events. Other studies suggest that SH is only associated with cardiovascular events in individuals who already have a high cardiovascular risk (120, 121). Nonetheless, our subgroup analyses reveal a quantitative insignificant difference in the RR of all-cause mortality for those with low versus high cardiovascular risk (RR 1.62 vs. 1.94, respectively). This result suggests the effect may be more pronounced in in those with a greater cardiovascular risk.
The mechanisms that relate SH to non-cardiovascular fatalities remain unknown (22, 54). SH may be a signal for greater provider monitoring in clinical settings, or a sign of quickly deteriorating health. SH may be appropriate for clinicians to review the mental and physical states of a patient to see whether any therapy adjustment is required. Such actions may minimize the risks of future hypoglycaemia and CVDs.
Subgroup analyses were performed so as to clarify whether the incidence of cardiac arrhythmia or all-cause death differed among relevant characteristics. In the trials with T2DM patients, preliminary subgroup analysis revealed a substantial increase in overall death risk among hypoglycemic versus euglycaemic individuals. Because T1DM patients are frequently younger than T2DM patients, they have less risk factors for death. Despite this, SH is more likely to attack T1DM patients, raising the risk of arrhythmia. SH is more likely to assault T1DM patients, increasing the risk of arrhythmia. This is not the case, and the underlying reason is unknown (87). In terms of hypoglycemia episodes and severity, more episodes and severity lead to higher risks of arrhythmia and all-cause mortality rate, and may indicate quickly failing health. Shorter follow-up period is associated with a greater increase in all-cause mortality in hypoglycemic patients compared euglycaemic patients. It might be that the population with the shortest follow-up duration has the most co-morbidities or frailty, in whom severe hypoglycemia episodes may be a surrogate for the underlying severity of the overall advanced and complex illness condition. Subgroup analysis based on study design methodologies for extracting effect estimates calls for more well-designed research in the future.
No review or meta-analysis can avoid heterogeneity of OS due to the lack of standardisation of method for clarifying cases, study design and time, classification of endpoints, and the amount of inter-study confounding that has been adjusted. For example, definitions of hypoglycaemia differ greatly. In spite of the sensitivity testing, we were unable to account for the greatest between-study heterogeneity in the outcome of cardiac arrhythmia and death. Therefore, these results need confirmation from more research. Furthermore, the statistical methodologies used in p-h/sa of RCTs and OSs did not adequately address the influence of unmeasured confounding components regarding overall effect estimation. The origins of variability were attributed to differences in study populations and exposure, according to sensitivity analyses.
Some of the significant strengths of this meta-analysis include the detailed retrieval plan with Cochrane procedures, the broad search approach, the relatively large sample size, and the capacity to investigate the link between hypoglycemia and cardiac arrhythmia or death. Nevertheless, this study has some limitations. First, the avoidance of unreported reports can have influenced our findings. Second, there is also a wide range of exposure, including severe hypoglycemia and any hypoglycemia. Third, the analyses employed for arrhythmia source research have drawbacks as well. Some studies have presented incident rate ratios for arrhythmias, implying that an increased risk applies to the whole research group whereas, in fact, only a few individuals who are extremely vulnerable may be impacted (45). Fourth, because we did not test autonomic function, we were unable to investigate its potential contributions.
In summary, hypoglycemia, as opposed to euglycaemia, is associated with a higher risk of cardiac arrhythmia and mortality. Nevertheless, evidence of potential causal linkages beyond statistical associations must await proof by additional specifically well planned research that controls for all potential remaining confounding factors, including a unified definition of hypoglycemia (122) as recommended by the International Hypoglycaemia Study Group.
Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Author contributions
XW and GL conceived the conception and design of the study, the search of the relevant literature, the extraction and analysis of data, and the drafting and revision of the final manuscript. SZ and FZ interpreted the analysed data, reviewed the manuscript critically, and contributed to its drafting. The final manuscript was read and approved by all authors.
Funding
This study was funded by grants from Zhejiang Medical and Health Science and Technology Project (Grant Agreement No. 2021KY1148) and Zhejiang Chinese medicine technology project (Grant Agreement No. 2022ZB363).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fendo.2023.1222409/full#supplementary-material
References
1. Sun H, Saeedi P, Karuranga S, Pinkepank M, Ogurtsova K, Duncan BB, et al. IDF Diabetes Atlas: Global, regional and country-level diabetes prevalence estimates for 2021 and projections for 2045. Diabetes Res Clin Pract (2022) 183:109119. doi: 10.1016/j.diabres.2021.109119
2. Leon BM, Maddox TM. Diabetes and cardiovascular disease: Epidemiology, biological mechanisms, treatment recommendations and future research. World J Diabetes (2015) 6:1246–58. doi: 10.4239/wjd.v6.i13.1246
3. Kelly TN, Bazzano LA, Fonseca VA, Thethi TK, Reynolds K, He J. Systematic review: glucose control and cardiovascular disease in type 2 diabetes. Ann Intern Med (2009) 151:394–403. doi: 10.7326/0003-4819-151-6-200909150-00137
4. ElSayed NA, Aleppo G, Aroda VR, Bannuru RR, Brown FM, Bruemmer D, et al. 9. Pharmacologic approaches to glycemic treatment: standards of care in diabetes-2023. Diabetes Care (2023) 46:S140–140S157. doi: 10.2337/dc23-S009
5. ElSayed NA, Aleppo G, Aroda VR, Bannuru RR, Brown FM, Bruemmer D, et al. 6. Glycemic targets: standards of care in diabetes-2023. Diabetes Care (2023) 46:S97–97S110. doi: 10.2337/dc23-S006
6. ElSayed NA, Aleppo G, Aroda VR, Bannuru RR, Brown FM, Bruemmer D, et al. 1. Improving care and promoting health in populations: standards of care in diabetes-2023. Diabetes Care (2023) 46:S10–10S18. doi: 10.2337/dc23-S001
7. Cryer PE. Glycemic goals in diabetes: trade-off between glycemic control and iatrogenic hypoglycemia. Diabetes (2014) 63:2188–95. doi: 10.2337/db14-0059
8. McCoy RG, Lipska KJ, Yao X, Ross JS, Montori VM, Shah ND. Intensive treatment and severe hypoglycemia among adults with type 2 diabetes. JAMA Intern Med (2016) 176:969–78. doi: 10.1001/jamainternmed.2016.2275
9. Johansen MY, MacDonald CS, Hansen KB, Karstoft K, Christensen R, Pedersen M, et al. Effect of an intensive lifestyle intervention on glycemic control in patients with type 2 diabetes: A randomized clinical trial. JAMA (2017) 318:637–46. doi: 10.1001/jama.2017.10169
10. Mahoney GK, Henk HJ, McCoy RG. Severe hypoglycemia attributable to intensive glucose-lowering therapy among US adults with diabetes: population-based modeling study, 2011-2014. Mayo Clin Proc (2019) 94:1731–42. doi: 10.1016/j.mayocp.2019.02.028
11. Tsujimoto T, Yamamoto-Honda R, Kajio H, Kishimoto M, Noto H, Hachiya R, et al. Vital signs, QT prolongation, and newly diagnosed cardiovascular disease during severe hypoglycemia in type 1 and type 2 diabetic patients. Diabetes Care (2014) 37:217–25. doi: 10.2337/dc13-0701
12. Christensen TF, Baekgaard M, Dideriksen JL, Steimle KL, Mogensen ML, Kildegaard J, et al. A physiological model of the effect of hypoglycemia on plasma potassium. J Diabetes Sci Technol (2009) 3:887–94. doi: 10.1177/193229680900300436
13. ORIGIN Trial Investigators, Mellbin LG, Rydén L, Riddle MC, Probstfield J, Rosenstock J, et al. Does hypoglycaemia increase the risk of cardiovascular events? A report from the ORIGIN trial. Eur Heart J (2013) 34:3137–44. doi: 10.1093/eurheartj/eht332
14. Hsu PF, Sung SH, Cheng HM, Yeh JS, Liu WL, Chan WL, et al. Association of clinical symptomatic hypoglycemia with cardiovascular events and total mortality in type 2 diabetes: a nationwide population-based study. Diabetes Care (2013) 36:894–900. doi: 10.2337/dc12-0916
15. International Hypoglycaemia Study Group. Hypoglycaemia, cardiovascular disease, and mortality in diabetes: epidemiology, pathogenesis, and management. Lancet Diabetes Endocrinol (2019) 7:385–96. doi: 10.1016/S2213-8587(18)30315-2
16. Andersen A, Jørgensen PG, Knop FK, Vilsbøll T. Hypoglycaemia and cardiac arrhythmias in diabetes. Ther Adv Endocrinol Metab (2020) 11:2042018820911803. doi: 10.1177/2042018820911803
17. Amiel SA. The consequences of hypoglycaemia. Diabetologia (2021) 64:963–70. doi: 10.1007/s00125-020-05366-3
18. Hsieh YC, Liao YC, Li CH, Lin JC, Weng CJ, Lin CC, et al. Hypoglycaemic episodes increase the risk of ventricular arrhythmia and sudden cardiac arrest in patients with type 2 diabetes-A nationwide cohort study. Diabetes Metab Res Rev (2020) 36:e3226. doi: 10.1002/dmrr.3226
19. Fitzpatrick C, Chatterjee S, Seidu S, Bodicoat DH, Ng GA, Davies MJ, et al. Association of hypoglycaemia and risk of cardiac arrhythmia in patients with diabetes mellitus: A systematic review and meta-analysis. Diabetes Obes Metab (2018) 20:2169–78. doi: 10.1111/dom.13348
20. Yeh JS, Sung SH, Huang HM, Yang HL, You LK, Chuang SY, et al. Hypoglycemia and risk of vascular events and mortality: a systematic review and meta-analysis. Acta Diabetol (2016) 53:377–92. doi: 10.1007/s00592-015-0803-3
21. Mellbin LG, Malmberg K, Waldenström A, Wedel H, Rydén L, DIGAMI 2 investigators. Prognostic implications of hypoglycaemic episodes during hospitalisation for myocardial infarction in patients with type 2 diabetes: a report from the DIGAMI 2 trial. Heart (2009) 95:721–7. doi: 10.1136/hrt.2008.152835
22. Bonds DE, Miller ME, Bergenstal RM, Buse JB, Byington RP, Cutler JA, et al. The association between symptomatic, severe hypoglycaemia and mortality in type 2 diabetes: retrospective epidemiological analysis of the ACCORD study. BMJ (2010) 340:b4909. doi: 10.1136/bmj.b4909
23. Chow E, Bernjak A, Williams S, Fawdry RA, Hibbert S, Freeman J, et al. Risk of cardiac arrhythmias during hypoglycemia in patients with type 2 diabetes and cardiovascular risk. Diabetes (2014) 63:1738–47. doi: 10.2337/db13-0468
24. Amione C, Giunti S, Fornengo P, Soedamah-Muthu SS, Chaturvedi N, Fuller JH, et al. Incidence of prolonged QTc and severe hypoglycemia in type 1 diabetes: the EURODIAB Prospective Complications Study. Acta Diabetol (2017) 54:871–6. doi: 10.1007/s00592-017-1018-6
25. Boucai L, Southern WN, Zonszein J. Hypoglycemia-associated mortality is not drug-associated but linked to comorbidities. Am J Med (2011) 124:1028–35. doi: 10.1016/j.amjmed.2011.07.011
26. Egi M, Bellomo R, Stachowski E, French CJ, Hart GK, Taori G, et al. Hypoglycemia and outcome in critically ill patients. Mayo Clin Proc (2010) 85:217–24. doi: 10.4065/mcp.2009.0394
27. Garg R, Hurwitz S, Turchin A, Trivedi A. Hypoglycemia, with or without insulin therapy, is associated with increased mortality among hospitalised patients. Diabetes Care (2013) 36:1107–10. doi: 10.2337/dc12-1296
28. Lipska KJ, Kosiborod M. Hypoglycemia and adverse outcomes: marker or mediator. Rev Cardiovasc Med (2011) 12:132–5. doi: 10.3909/ricm0586
29. Carey M, Boucai L, Zonszein J. Impact of hypoglycemia in hospitalised patients. Curr Diabetes Rep (2013) 13:107–13. doi: 10.1007/s11892-012-0336-x
30. Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PloS Med (2009) 6:e1000097. doi: 10.1016/j.jclinepi.2009.06.005
31. Hu Y, Lei M, Ke G, Huang X, Peng X, Zhong L, et al. Metformin use and risk of all-cause mortality and cardiovascular events in patients with chronic kidney disease-A systematic review and meta-analysis. Front Endocrinol (Lausanne) (2020) 11:559446. doi: 10.3389/fendo.2020.559446
32. Zeng X, Zhang Y, Kwong JS, Zhang C, Li S, Sun F, et al. The methodological quality assessment tools for preclinical and clinical studies, systematic review and meta-analysis, and clinical practice guideline: a systematic review. J Evid Based Med (2015) 8:2–10. doi: 10.1111/jebm.12141
33. Ma LY, Zhao B, Ou YN, Zhang DD, Li QY, Tan L. Association of thyroid disease with risks of dementia and cognitive impairment: A meta-analysis and systematic review. Front Aging Neurosci (2023) 15:1137584. doi: 10.3389/fnagi.2023.1137584
34. Siristatidis C, Sergentanis TN, Kanavidis P, Trivella M, Sotiraki M, Mavromatis I, et al. Controlled ovarian hyperstimulation for IVF: impact on ovarian, endometrial and cervical cancer–a systematic review and meta-analysis. Hum Reprod Update (2013) 19:105–23. doi: 10.1093/humupd/dms051
35. Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med (2002) 21:1539–58. doi: 10.1002/sim.1186
36. Hedges LV, Pigott TD. The power of statistical tests in meta-analysis. Psychol Methods (2001) 6:203–17. doi: 10.1037/1082-989X.6.3.203
37. Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ (1997) 315:629–34. doi: 10.1136/bmj.315.7109.629
38. Gruden G, Giunti S, Barutta F, Chaturvedi N, Witte DR, Tricarico M, et al. QTc interval prolongation is independently associated with severe hypoglycemic attacks in type 1 diabetes from the EURODIAB IDDM complications study. Diabetes Care (2012) 35:125–7. doi: 10.2337/dc11-1739
39. Lee AK, Warren B, Lee CJ, McEvoy JW, Matsushita K, Huang ES, et al. The association of severe hypoglycemia with incident cardiovascular events and mortality in adults with type 2 diabetes. Diabetes Care (2018) 41:104–11. doi: 10.2337/dc17-1669
40. Echouffo-Tcheugui JB, Daya N, Lee AK, Tang O, Ndumele CE, Windham BG, et al. Severe hypoglycemia, cardiac structure and function, and risk of cardiovascular events among older adults with diabetes. Diabetes Care (2021) 44:248–54. doi: 10.2337/dc20-0552
41. Kaze AD, Yuyun MF, Erqou S, Fonarow GC, Echouffo-Tcheugui JB. Severe hypoglycemia and incidence of QT interval prolongation among adults with type 2 diabetes. J Clin Endocrinol Metab (2022) 107:e2743–2743e2750. doi: 10.1210/clinem/dgac195
42. Pistrosch F, Ganz X, Bornstein SR, Birkenfeld AL, Henkel E, Hanefeld M. Risk of and risk factors for hypoglycemia and associated arrhythmias in patients with type 2 diabetes and cardiovascular disease: a cohort study under real-world conditions. Acta Diabetol (2015) 52:889–95. doi: 10.1007/s00592-015-0727-y
43. Tsujimoto T, Yamamoto-Honda R, Kajio H, Kishimoto M, Noto H, Hachiya R, et al. High risk of abnormal QT prolongation in the early morning in diabetic and non-diabetic patients with severe hypoglycemia. Ann Med (2015) 47:238–44. doi: 10.3109/07853890.2015.1017528
44. Ko SH, Park YM, Yun JS, Cha SA, Choi EK, Han K, et al. Severe hypoglycemia is a risk factor for atrial fibrillation in type 2 diabetes mellitus: Nationwide population-based cohort study. J Diabetes Complications (2018) 32:157–63. doi: 10.1016/j.jdiacomp.2017.09.009
45. Novodvorsky P, Bernjak A, Chow E, Iqbal A, Sellors L, Williams S, et al. Diurnal differences in risk of cardiac arrhythmias during spontaneous hypoglycemia in young people with type 1 diabetes. Diabetes Care (2017) 40:655–62. doi: 10.2337/dc16-2177
46. Zhang J, He X, Zhang L, Wang K, Chen H, Wu J. Association of severe hypoglycemia with all-cause mortality and complication risks among patients with type 2 diabetes mellitus in China. Diabetes Res Clin Pract (2020) 170:108493. doi: 10.1016/j.diabres.2020.108493
47. Mylona M, Liatis S, Anastasiadis G, Kapelios C, Kokkinos A. Severe iatrogenic hypoglycaemia requiring medical assistance is associated with concurrent prolongation of the QTc interval. Diabetes Res Clin Pract (2020) 161:108038. doi: 10.1016/j.diabres.2020.108038
48. Ali Abdelhamid Y, Bernjak A, Phillips LK, Summers MJ, Weinel LM, Lange K, et al. Nocturnal hypoglycemia in patients with diabetes discharged from ICUs: A prospective two-center cohort study. Crit Care Med (2021) 49:636–49. doi: 10.1097/CCM.0000000000004810
49. Andersen A, Bagger JI, Sørensen SK, Baldassarre M, Pedersen-Bjergaard U, Forman JL, et al. Associations of hypoglycemia, glycemic variability and risk of cardiac arrhythmias in insulin-treated patients with type 2 diabetes: a prospective, observational study. Cardiovasc Diabetol (2021) 20:241. doi: 10.1186/s12933-021-01425-0
50. Curkendall SM, Natoli JL, Alexander CM, Nathanson BH, Haidar T, Dubois RW. Economic and clinical impact of inpatient diabetic hypoglycemia. Endocr Pract (2009) 15:302–12. doi: 10.4158/EP08343.OR
51. Zoungas S, Patel A, Chalmers J, de Galan BE, Li Q, Billot L, et al. Severe hypoglycemia and risks of vascular events and death. N Engl J Med (2010) 363:1410–8. doi: 10.1056/NEJMoa1003795
52. Li DB, Hua Q, Guo J, Li HW, Chen H, Zhao SM. Admission glucose level and in-hospital outcomes in diabetic and non-diabetic patients with ST-elevation acute myocardial infarction. Intern Med (2011) 50:2471–5. doi: 10.2169/internalmedicine.50.5750
53. NIrantharakumar K, Marshall T, Kennedy A, Narendran P, Hemming K, Coleman JJ. Hypoglycaemia is associated with increased length of stay and mortality in people with diabetes who are hospitalised. Diabetes Med (2012) 29:e445–8. doi: 10.1111/dme.12002
54. McCoy RG, Van Houten HK, Ziegenfuss JY, Shah ND, Wermers RA, Smith SA. Increased mortality of patients with diabetes reporting severe hypoglycemia. Diabetes Care (2012) 35:1897–901. doi: 10.2337/dc11-2054
55. Zhao Y, Campbell CR, Fonseca V, Shi L. Impact of hypoglycemia associated with antihyperglycemic medications on vascular risks in veterans with type 2 diabetes. Diabetes Care (2012) 35:1126–32. doi: 10.2337/dc11-2048
56. Tan HK, Flanagan D. The impact of hypoglycaemia on patients admitted to hospital with medical emergencies. Diabetes Med (2013) 30:574–80. doi: 10.1111/dme.12123
57. Sechterberger MK, Bosman RJ, Oudemans-van Straaten HM, Siegelaar SE, Hermanides J, Hoekstra JB, et al. The effect of diabetes mellitus on the association between measures of glycaemic control and ICU mortality: a retrospective cohort study. Crit Care (2013) 17:R52. doi: 10.1186/cc12572
58. Cooper MN, de Klerk NH, Jones TW, Davis EA. Clinical and demographic risk factors associated with mortality during early adulthood in a population-based cohort of childhood-onset type 1 diabetes. Diabetes Med (2014) 31:1550–8. doi: 10.1111/dme.12522
59. Lee SA, Cho SJ, Jeong MH, Kim YJ, Kim CJ, Cho MC, et al. Hypoglycemia at admission in patients with acute myocardial infarction predicts a higher 30-day mortality in patients with poorly controlled type 2 diabetes than in well-controlled patients. Diabetes Care (2014) 37:2366–73. doi: 10.2337/dc13-2856
60. Kong AP, Yang X, Luk A, Cheung KK, Ma RC, So WY, et al. Hypoglycaemia, chronic kidney disease and death in type 2 diabetes: the Hong Kong diabetes registry. BMC Endocr Disord (2014) 14:48. doi: 10.1186/1472-6823-14-48
61. Lung TW, Petrie D, Herman WH, Palmer AJ, Svensson AM, Eliasson B, et al. Severe hypoglycemia and mortality after cardiovascular events for type 1 diabetic patients in Sweden. Diabetes Care (2014) 37:2974–81. doi: 10.2337/dc14-0405
62. Elwen FR, Huskinson A, Clapham L, Bottomley MJ, Heller SR, James C, et al. An observational study of patient characteristics and mortality following hypoglycemia in the community. BMJ Open Diabetes Res Care (2015) 3:e000094. doi: 10.1136/bmjdrc-2015-000094
63. Khunti K, Davies M, Majeed A, Thorsted BL, Wolden ML, Paul SK. Hypoglycemia and risk of cardiovascular disease and all-cause mortality in insulin-treated people with type 1 and type 2 diabetes: a cohort study. Diabetes Care (2015) 38:316–22. doi: 10.2337/dc14-0920
64. Gómez-Huelgas R, Guijarro-Merino R, Zapatero A, Barba R, Guijarro-Contreras A, Tinahones F, et al. The frequency and impact of hypoglycemia among hospitalised patients with diabetes: A population-based study. J Diabetes Complications (2015) 29:1050–5. doi: 10.1016/j.jdiacomp.2015.07.018
65. Escalada J, Liao L, Pan C, Wang H, Bala M. Outcomes and healthcare resource utilisation associated with medically attended hypoglycemia in older patients with type 2 diabetes initiating basal insulin in a US managed care setting. Curr Med Res Opin (2016) 32:1557–65. doi: 10.1080/03007995.2016.1189893
66. Freemantle N, Danchin N, Calvi-Gries F, Vincent M, Home PD. Relationship of glycaemic control and hypoglycaemic episodes to 4-year cardiovascular outcomes in people with type 2 diabetes starting insulin. Diabetes Obes Metab (2016) 18:152–8. doi: 10.1111/dom.12598
67. Rauh SP, Rutters F, Thorsted BL, Wolden ML, Nijpels G, van der Heijden AA, et al. Self-reported hypoglycaemia in patients with type 2 diabetes treated with insulin in the Hoorn Diabetes Care System Cohort, the Netherlands: a prospective cohort study. BMJ Open (2016) 6:e012793. doi: 10.1136/bmjopen-2016-012793
68. Takeishi S, Mori A, Hachiya H, Yumura T, Ito S, Shibuya T, et al. Hypoglycemia and glycemic variability are associated with mortality in non-intensive care unit hospitalised infectious disease patients with diabetes mellitus. J Diabetes Investig (2016) 7:429–35. doi: 10.1111/jdi.12436
69. Cha SA, Yun JS, Lim TS, Hwang S, Yim EJ, Song KH, et al. Severe hypoglycemia and cardiovascular or all-cause mortality in patients with type 2 diabetes. Diabetes Metab J (2016) 40:202–10. doi: 10.4093/dmj.2016.40.3.202
70. Sejling AS, Schouwenberg B, Faerch LH, Thorsteinsson B, de Galan BE, Pedersen-Bjergaard U. Association between hypoglycaemia and impaired hypoglycaemia awareness and mortality in people with Type 1 diabetes mellitus. Diabetes Med (2016) 33:77–83. doi: 10.1111/dme.12810
71. Lu CL, Shen HN, Hu SC, Wang JD, Li CY. A population-based study of all-cause mortality and cardiovascular disease in association with prior history of hypoglycemia among patients with type 1 diabetes. Diabetes Care (2016) 39:1571–8. doi: 10.2337/dc15-2418
72. Chevalier P, Vandebrouck T, De Keyzer D, Mertens A, Lamotte M. Cost and co-morbidities associated with hypoglycemic inpatients in Belgium. J Med Econ (2016) 19:44–52. doi: 10.3111/13696998.2015.1086775
73. Chi MJ, Liang CK, Lee WJ, Peng LN, Chou MY, Chen LK. Association of new-onset diabetes mellitus in older people and mortality in Taiwan: A 10-year nationwide population-based study. J Nutr Health Aging (2017) 21:227–32. doi: 10.1007/s12603-016-0751-9
74. Zhao S, Murugiah K, Li N, Li X, Xu ZH, Li J, et al. Admission Glucose and In-hospital Mortality after Acute Myocardial Infarction in Patients with or without Diabetes: A Cross-sectional Study. Chin Med J (Engl) (2017) 130:767–75. doi: 10.4103/0366-6999.202733
75. Standl E, Stevens SR, Armstrong PW, Buse JB, Chan J, Green JB, et al. Increased risk of severe hypoglycemic events before and after cardiovascular outcomes in TECOS suggests an at-risk type 2 diabetes frail patient phenotype. Diabetes Care (2018) 41:596–603. doi: 10.2337/dc17-1778
76. Zinman B, Marso SP, Christiansen E, Calanna S, Rasmussen S, Buse JB, et al. Hypoglycemia, cardiovascular outcomes, and death: the LEADER experience. Diabetes Care (2018) 41:1783–91. doi: 10.2337/dc17-2677
77. Pieber TR, Marso SP, McGuire DK, Zinman B, Poulter NR, Emerson SS, et al. DEVOTE 3: temporal relationships between severe hypoglycaemia, cardiovascular outcomes and mortality. Diabetologia (2018) 61:58–65. doi: 10.1007/s00125-017-4422-0
78. Leung CH, Liu CP. Diabetic status and the relationship of blood glucose to mortality in adults with carbapenem-resistant Acinetobacter baumannii complex bacteremia. J Microbiol Immunol Infect (2019) 52:654–62. doi: 10.1016/j.jmii.2018.06.005
79. Lo SC, Yang YS, Kornelius E, Huang JY, Lai YR, Huang CN, et al. Early cardiovascular risk and all-cause mortality following an incident of severe hypoglycaemia: A population-based cohort study. Diabetes Obes Metab (2019) 21:1878–85. doi: 10.1111/dom.13746
80. Wei W, Zhao S, Fu SL, Yi L, Mao H, Tan Q, et al. The association of hypoglycemia assessed by continuous glucose monitoring with cardiovascular outcomes and mortality in patients with type 2 diabetes. Front Endocrinol (Lausanne) (2019) 10:536. doi: 10.3389/fendo.2019.00536
81. Yun JS, Park YM, Han K, Cha SA, Ahn YB, Ko SH. Severe hypoglycemia and the risk of cardiovascular disease and mortality in type 2 diabetes: a nationwide population-based cohort study. Cardiovasc Diabetol (2019) 18:103. doi: 10.1186/s12933-019-0909-y
82. Davis SN, Duckworth W, Emanuele N, Hayward RA, Wiitala WL, Thottapurathu L, et al. Effects of severe hypoglycemia on cardiovascular outcomes and death in the veterans affairs diabetes trial. Diabetes Care (2019) 42:157–63. doi: 10.2337/dc18-1144
83. Wernly B, Jirak P, Lichtenauer M, Franz M, Kabisch B, Schulze PC, et al. Hypoglycemia but not hyperglycemia is associated with mortality in critically ill patients with diabetes. Med Princ Pract (2019) 28:186–92. doi: 10.1159/000496205
84. Ferreira L, Moniz AC, Carneiro AS, MIranda AS, Fangueiro C, Fernandes D, et al. The impact of glycemic variability on length of stay and mortality in diabetic patients admitted with community-acquired pneumonia or chronic obstructive pulmonary disease. Diabetes Metab Syndr (2019) 13:149–53. doi: 10.1016/j.dsx.2018.08.028
85. Mattishent K, Richardson K, Dhatariya K, Savva GM, Fox C, Loke YK. The effects of hypoglycaemia and dementia on cardiovascular events, falls and fractures and all-cause mortality in older individuals: A retrospective cohort study. Diabetes Obes Metab (2019) 21:2076–85. doi: 10.1111/dom.13769
86. Standl E, Stevens SR, Lokhnygina Y, Bethel MA, Buse JB, Gustavson SM, et al. Confirming the bidirectional nature of the association between severe hypoglycemic and cardiovascular events in type 2 diabetes: insights from EXSCEL. Diabetes Care (2020) 43:643–52. doi: 10.2337/dc19-1079
87. Jensen MH, Dethlefsen C, Hejlesen O, Vestergaard P. Association of severe hypoglycemia with mortality for people with diabetes mellitus during a 20-year follow-up in Denmark: a cohort study. Acta Diabetol (2020) 57:549–58. doi: 10.1007/s00592-019-01447-x
88. Zaccardi F, Ling S, Lawson C, Davies MJ, Khunti K. Severe hypoglycaemia and absolute risk of cause-specific mortality in individuals with type 2 diabetes: a UK primary care observational study. Diabetologia (2020) 63:2129–39. doi: 10.1007/s00125-020-05223-3
89. Han E, Han KD, Lee BW, Kang ES, Cha BS, Ko SH, et al. Severe hypoglycemia increases dementia risk and related mortality: A nationwide, population-based cohort study. J Clin Endocrinol Metab (2022) 107:e1976–1976e1986. doi: 10.1210/clinem/dgab860
90. Cha SA, Yun JS, Kim GH, Ahn YB. Impact of hypoglycemia at the time of hospitalisation for heart failure from emergency department on major adverse cardiovascular events in patients with and without type 2 diabetes. Cardiovasc Diabetol (2022) 21:218. doi: 10.1186/s12933-022-01651-0
91. Heller SR, Geybels MS, Iqbal A, Liu L, Wagner L, Chow E. A higher non-severe hypoglycaemia rate is associated with an increased risk of subsequent severe hypoglycaemia and major adverse cardiovascular events in individuals with type 2 diabetes in the LEADER study. Diabetologia (2022) 65:55–64. doi: 10.1007/s00125-021-05556-7
92. Nordin C. The case for hypoglycaemia as a proarrhythmic event: basic and clinical evidence. Diabetologia (2010) 53:1552–61. doi: 10.1007/s00125-010-1752-6
93. Zhang Y, Han H, Wang J, Wang H, Yang B, Wang Z. Impairment of human ether-à-go-go-related gene (HERG) K+ channel function by hypoglycemia and hyperglycemia. Similar phenotypes but different mechanisms. J Biol Chem (2003) 278:10417–26. doi: 10.1074/jbc.M211044200
94. Jones TW, Porter P, Sherwin RS, Davis EA, O'Leary P, Frazer F, et al. Decreased epinephrine responses to hypoglycemia during sleep. N Engl J Med (1998) 338:1657–62. doi: 10.1056/NEJM199806043382303
95. Zammitt NN, Frier BM. Hypoglycemia in type 2 diabetes: pathophysiology, frequency, and effects of different treatment modalities. Diabetes Care (2005) 28:2948–61. doi: 10.2337/diacare.28.12.2948
96. Du YF, Ou HY, Beverly EA, Chiu CJ. Achieving glycemic control in elderly patients with type 2 diabetes: a critical comparison of current options. Clin Interv Aging (2014) 9:1963–80. doi: 10.2147/CIA.S53482
97. Yun JS, Ko SH. Avoiding or coping with severe hypoglycemia in patients with type 2 diabetes. Korean J Intern Med (2015) 30:6–16. doi: 10.3904/kjim.2015.30.1.6
98. Murphy NP, Ford-Adams ME, Ong KK, Harris ND, Keane SM, Davies C, et al. Prolonged cardiac repolarisation during spontaneous nocturnal hypoglycaemia in children and adolescents with type 1 diabetes. Diabetologia (2004) 47:1940–7. doi: 10.1007/s00125-004-1552-y
99. Robinson RT, Harris ND, Ireland RH, Macdonald IA, Heller SR. Changes in cardiac repolarisation during clinical episodes of nocturnal hypoglycaemia in adults with Type 1 diabetes. Diabetologia (2004) 47:312–5. doi: 10.1007/s00125-003-1292-4
100. Ng JM, Cox H, Longbotham D, Kilpatrick ES, Atkin SL, Allan BJ. Hypoglycemia and clinical outcomes in patients with diabetes hospitalised in the general ward: response to Turchin et al. Diabetes Care (2009) 32:e151. doi: 10.2337/dc09-1341
101. Cryer PE. Mechanisms of hypoglycemia-associated autonomic failure and its component syndromes in diabetes. Diabetes (2005) 54:3592–601. doi: 10.2337/diabetes.54.12.3592
102. Desouza C, Salazar H, Cheong B, Murgo J, Fonseca V. Association of hypoglycemia and cardiac ischemia: a study based on continuous monitoring. Diabetes Care (2003) 26:1485–9. doi: 10.2337/diacare.26.5.1485
103. Maser RE, Mitchell BD, Vinik AI, Freeman R. The association between cardiovascular autonomic neuropathy and mortality in individuals with diabetes: a meta-analysis. Diabetes Care (2003) 26:1895–901. doi: 10.2337/diacare.26.6.1895
104. Wright RJ, Frier BM. Vascular disease and diabetes: is hypoglycaemia an aggravating factor. Diabetes Metab Res Rev (2008) 24:353–63. doi: 10.1002/dmrr.865
105. Adler GK, Bonyhay I, Failing H, Waring E, Dotson S, Freeman R. Antecedent hypoglycemia impairs autonomic cardiovascular function: implications for rigorous glycemic control. Diabetes (2009) 58:360–6. doi: 10.2337/db08-1153
106. Gill GV, Woodward A, Casson IF, Weston PJ. Cardiac arrhythmia and nocturnal hypoglycaemia in type 1 diabetes–the 'dead in bed' syndrome revisited. Diabetologia (2009) 52:42–5. doi: 10.1007/s00125-008-1177-7
107. Robinson RT, Harris ND, Ireland RH, Lee S, Newman C, Heller SR. Mechanisms of abnormal cardiac repolarisation during insulin-induced hypoglycemia. Diabetes (2003) 52:1469–74. doi: 10.2337/diabetes.52.6.1469
108. Sommerfield AJ, Wilkinson IB, Webb DJ, Frier BM. Vessel wall stiffness in type 1 diabetes and the central hemodynamic effects of acute hypoglycemia. Am J Physiol Endocrinol Metab (2007) 293:E1274–9. doi: 10.1152/ajpendo.00114.2007
109. Frier BM, Schernthaner G, Heller SR. Hypoglycemia and cardiovascular risks. Diabetes Care (2011) 34 Suppl 2:S132–7. doi: 10.2337/dc11-s220
110. Iqbal A, Prince LR, Novodvorsky P, Bernjak A, Thomas MR, Birch L, et al. Effect of hypoglycemia on inflammatory responses and the response to low-dose endotoxemia in humans. J Clin Endocrinol Metab (2019) 104:1187–99. doi: 10.1210/jc.2018-01168
111. Chow E, Iqbal A, Walkinshaw E, Phoenix F, Macdonald IA, Storey RF, et al. Prolonged prothrombotic effects of antecedent hypoglycemia in individuals with type 2 diabetes. Diabetes Care (2018) 41:2625–33. doi: 10.2337/dc18-0050
112. Verhulst C, van Heck J, Fabricius TW, Stienstra R, Teerenstra S, McCrimmon RJ, et al. Sustained proinflammatory effects of hypoglycemia in people with type 2 diabetes and in people without diabetes. Diabetes (2022) 71:2716–27. doi: 10.2337/db22-0246
113. Ratter JM, Rooijackers HM, Tack CJ, Hijmans AG, Netea MG, de Galan BE, et al. Proinflammatory effects of hypoglycemia in humans with or without diabetes. Diabetes (2017) 66:1052–61. doi: 10.2337/db16-1091
114. Lee AK, McEvoy JW, Hoogeveen RC, Ballantyne CM, Selvin E. Severe hypoglycemia and elevated high-sensitivity cardiac troponin T in older adults with diabetes: the ARIC study. J Am Coll Cardiol (2016) 68:1370–1. doi: 10.1016/j.jacc.2016.06.049
115. Rezende PC, Everett BM, Brooks MM, Vlachos H, Orchard TJ, Frye RL, et al. Hypoglycemia and elevated troponin in patients with diabetes and coronary artery disease. J Am Coll Cardiol (2018) 72:1778–86. doi: 10.1016/j.jacc.2018.07.067
116. Gerich JE. Hypoglycaemia and counterregulation in type 2 diabetes. Lancet (2000) 356:1946–7. doi: 10.1016/S0140-6736(00)03303-1
117. Cryer PE. Severe hypoglycemia predicts mortality in diabetes. Diabetes Care (2012) 35:1814–6. doi: 10.2337/dc12-0749
118. Goto A, Arah OA, Goto M, Terauchi Y, Noda M. Severe hypoglycaemia and cardiovascular disease: systematic review and meta-analysis with bias analysis. BMJ (2013) 347:f4533. doi: 10.1136/bmj.f4533
119. MacMahon S, Collins R. Reliable assessment of the effects of treatment on mortality and major morbidity, II: observational studies. Lancet (2001) 357:455–62. doi: 10.1016/S0140-6736(00)04017-4
120. Leong A, Berkowitz SA, Triant VA, Porneala B, He W, Atlas SJ, et al. Hypoglycemia in diabetes mellitus as a coronary artery disease risk factor in patients at elevated vascular risk. J Clin Endocrinol Metab (2016) 101:659–68. doi: 10.1210/jc.2015-3169
121. Pistrosch F, Hanefeld M. Hypoglycemia and cardiovascular disease: lessons from outcome studies. Curr Diabetes Rep (2015) 15:117. doi: 10.1007/s11892-015-0678-2
122. International Hypoglycaemia Study Group. Glucose Concentrations of Less Than 3.0 mmol/L (54 mg/dL) Should Be Reported in Clinical Trials: A Joint Position Statement of the American Diabetes Association and the European Association for the Study of Diabetes. Diabetes Care (2017) 40:155–7. doi: 10.2337/dc16-2215
Keywords: hypoglycaemia, diabetes, arrhythmia, mortality, risk, systematic review, meta-analysis
Citation: Li G, Zhong S, Wang X and Zhuge F (2023) Association of hypoglycaemia with the risks of arrhythmia and mortality in individuals with diabetes - a systematic review and meta-analysis. Front. Endocrinol. 14:1222409. doi: 10.3389/fendo.2023.1222409
Received: 14 May 2023; Accepted: 24 July 2023;
Published: 14 August 2023.
Edited by:
Sen Li, Beijing University of Chinese Medicine, ChinaReviewed by:
Eberhard Standl, Helmholtz Zentrum München, GermanyAhmed Iqbal, The University of Sheffield, United Kingdom
Copyright © 2023 Li, Zhong, Wang and Zhuge. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Xingmu Wang, eG13MjAyMzEyM0AxNjMuY29t
†These authors have contributed equally to this work and share first authorship
 Gangfeng Li1†
Gangfeng Li1† Xingmu Wang
Xingmu Wang