- 1The First Clinical College, Shandong University of Traditional Chinese Medicine, Jinan, China
- 2Reproductive and Genetic Center, The Affiliated Hospital of Shandong University of Traditional Chinese Medicine, Jinan, China
Objective: There is no clear evidence of clinical significance of endometrial compaction, which can be measured by a reduction in endometrial thickness (EMT) during the follicular-luteal transition before the day of embryo transfer. In this study, we aim to determine whether endometrial compaction has an effect on in vitro fertilization (IVF) success.
Method(s): We searched PubMed, Cochrane, Embase, and Web of Science electronic databases for studies published in English up to March 2023. Heterogeneity between studies was assessed using the I2 statistic. The random effects model and fixed effects model was used to pool the risk ratio (RR) with a corresponding 95% confidence interval (CI). A subgroup analysis was performed based on different methods of ultrasonic measurement and different endometrial compaction rates (ECR). Stata 17.0 software was used for meta-analysis. Pregnancy outcomes, which included clinical pregnancy rate, ongoing pregnancy rate, live birth rate, and spontaneous abortion rate, were evaluated.
Result(s): In this study, 18 cohort studies were included, involving 16,164 embryo transfer cycles. Pooled results indicated that there was no significant difference between the endometrial compaction group and the non-compaction group in terms of clinical pregnancy rate (RR [95% CI]=0.98 [0.90,1.08]; I2 = 69.76%), ongoing pregnancy rate (RR [95% CI]=1.18 [0.95,1.47]; I2 = 78.77%), live birth rate (RR [95% CI]= 0.97 [0.92,1.02]; I2 = 0.00%) or spontaneous abortion rate (RR [95% CI]= 1.07[0.97,1.26]; I2 = 0.00%). According to the subgroup analysis of ultrasonic measurement methods, in the transvaginal ultrasound (TVUS) combined with abdominal ultrasonography (AUS) cycles of the endometrial compaction group, the rate of ongoing pregnancy (RR [95% CI] = 1.69 [1.26, 2.26]; I2 = 29.27%) and live birth (RR [95% CI] = 1.27 [1.00,1.61]; I2 = 62.28%) was significantly higher than that of the non-compaction group. Additionally, subgroup analysis based on ECR revealed a significantly higher rate of ongoing pregnancy when ECR ≥ 15% (RR [95% CI] = 1.99 [1.61, 2.47]; I2 = 0.00%).
Conclusion: Endometrial compaction has no adverse effect on clinical pregnancy rate, ongoing pregnancy rate, live birth rate, or spontaneous abortion rate. A possible explanation for the contradictory findings of previous studies lies in the method by which the EMT is measured.
Systematic review registration: https://www.crd.york.ac.uk/prospero/display_record.php?ID=CRD42023430511, identifier CRD42023430511.
Introduction
Infertility has become a major public health concern worldwide. Current evidence indicates a 15.5% prevalence of infertility, with 55.2% of couples seeking medical care (1). Medical advances have made assisted reproduction a viable option for infertile couples worldwide. Statistically, ongoing pregnancy rates following IVF vary between 8.6% and 46.2% per cycle (2), depending on embryo quality, endometrial receptivity (ER), and embryo-endometrial communication (3). Preimplantation genetic testing (PGT) has become increasingly common in recent years, which to some extent has improved the quality of implanted embryos and reduced the effects of aneuploid embryos on IVF. Yet, its positive predictive value is not higher than 50-60% (4). Hence, ER evaluation is essential before embryo transfer (ET).
Good ER enables the endometrium to provide an optimal environment for embryo development and placenta formation. EMT is the most widely recognized ER marker (5). EMT measurement by ultrasound is also routinely performed in clinical practice as a non-invasive method of ER evaluation. For ER, a “window of implantation” occurs when the endometrium is at its best to support trophoblast-endometrial interactions, which is thought to occur around days 22-24 of an ideal 28-day cycle (6). In previous studies, EMT has been studied on the day human chorionic gonadotropin (hCG) is triggered in fresh ET cycles, as well as on the day when estrogen is discontinued or progesterone is begun in frozen-thawed embryo transfer (FET) cycles (7, 8).
Nevertheless, there is still controversy regarding the predictiveness of EMT on pregnancy outcomes (9). In many cycles, ET has been postponed due to inadequate EMT. A recent meta-analysis demonstrated that a thin endometrium is associated with poor live birth rates (LBR) (10). LBRs are decreased when EMT is less than 8 mm on the day of hCG in a fresh ET cycle or when EMT is less than 7 mm on the day of progestogen initiation in a FET cycle (8). However, the study by Ata et al. (11) evaluated the effect of EMT and LBR after the transfer of 959 single euploid blastocysts without EMT cutoff. They could not find a threshold below which LBR decreased, and concluded that an endometrium of 3-4 mm has a similar LBR to a thicker endometrium.
Though the above studies have mainly examined EMT, which is measured before hCG administration or progesterone administration, few have looked at how EMT changes during the end of the follicular phase or the early luteal phase influence pregnancy outcomes. Haas et al. (12) reported that endometrial compaction (thinning of the EMT during the end of the follicular phase or early luteal phase) may lead to more favorable pregnancy outcomes, but subsequent studies have made conflicting findings (13, 14). In the present study, we sought to review the evidence from observational studies to explore whether endometrial compaction is predictive of pregnancy outcomes and to inform reproductive clinicians in their assessment of ER.
Materials and methods
The systematic review and meta-analysis was conducted according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) checklist (15).
Search strategy
PubMed, Cochrane, Embase, and Web of Science electronic databases were searched for studies published in English up to March 2023, using Medical Subject Headings [MeSH] and keywords related to the study, no restriction on the year of publication. The database’s specific strategy was (‘Embryo Transfer’ OR ‘Embryo Transfers’ OR ‘Blastocyst Transfer’ OR ‘Blastocyst Transfers’ OR ‘Fertilizations in Vitro’ OR ‘IVF’ OR ‘In Vitro Fertilization’ OR ‘In Vitro Fertilizations’ OR ‘ICSI’ OR ‘Intracytoplasmic Sperm Injection’ OR ‘Intracytoplasmic Sperm Injections’) AND (‘Endometrial compaction’ OR ‘Endometrial thickness change’ OR ‘change, endometrial thickness’ OR ‘Endometrial thickness decreased’ OR ‘Endometrial thickness compacted’) AND (‘Pregnancy Outcome’ OR ‘Pregnancy Outcomes’ OR ‘Clinical outcomes’ OR ‘Live birth’ OR ‘Clinical pregnancy’ OR ‘Ongoing pregnancy’). Additionally, references of all included articles were screened, and literature retrieval was finalized. The entire retrieved literature was screened by two independent reviewers, and the included literature was collated using EndNote X9.3.3. Any articles with uncertainties were resolved through discussion and, if necessary, group discussion with a third investigator to reach a final consensus.
Inclusion/exclusion criteria
Studies that met the following criteria were considered eligible for systematic review: (1) The study was an observational cohort study; (2) The study population was all women who underwent IVF; (3) Report on the relationship between EMT changes and pregnancy outcomes; (4) Studies had at least one of the following outcomes: clinical pregnancy, ongoing pregnancy, live birth, and spontaneous abortion; (5) Published in English.
Studies with the following conditions will be excluded: (1) No clinical outcome or no available data; (2) Overview, conference abstracts, case reports, and case series; (3) Articles without complete research strategy.
Outcome measures
Clinical pregnancy is defined as one or more gestational sacs seen in the uterine cavity detected by ultrasound. Ongoing pregnancy is defined as transvaginal ultrasound showing fetal heart activity at 12 weeks of gestation or later. Live birth is defined as surviving infants delivered at ≥ 24 weeks of gestation. Spontaneous abortion is considered to be the pregnancy loss of one or more gestational sacs previously observed before 24-week gestation.
Data extraction
According to the PRISMA guidelines, the following data were extracted independently by two authors from each eligible study: first author’s surname, publication year, country, study duration, study design, number of cycles, endometrial preparation protocol, endometrial measurement method, number of embryos transferred, embryo development stage, and pregnancy outcomes. Data from different subgroups within the same study were also extracted for possible synthesis. Disagreements between the two reviewers were resolved in the same manner as described above.
Quality assessment
The quality of the included cohort studies was assessed independently by two reviewers using the Newcastle-Ottawa Scale (NOS), which assigns a maximum of nine points to each study based on three broad dimensions: selection (4 points), comparability (2 points), and outcome (3 points). Scores ranged from 0 to 9 points, with studies ≥ 7 points being considered high quality, 4-6 points indicating moderate quality, and < 4 points indicating low quality. Disagreements between the two reviewers were resolved in the same manner as described above.
Statistical analysis
Pregnancy outcomes were counted as dichotomous variables and expressed as RR with a 95% CI. The degree of heterogeneity was quantified by I2 statistics, when I2 = 0%, considered no heterogeneity between studies, when I2 < 25%, considered mild heterogeneity between studies, when 25% ≤ I2 < 50%, considered moderate heterogeneity between studies, when I2 ≥ 50%, considered high heterogeneity between studies (16). According to heterogeneity, the results were calculated using a random effects model (Der Simonian-Laird) or fixed-effects model (Mantel-Haenszel). All statistical analyses were performed using Stata 17.0.
Results
Literature search and study features
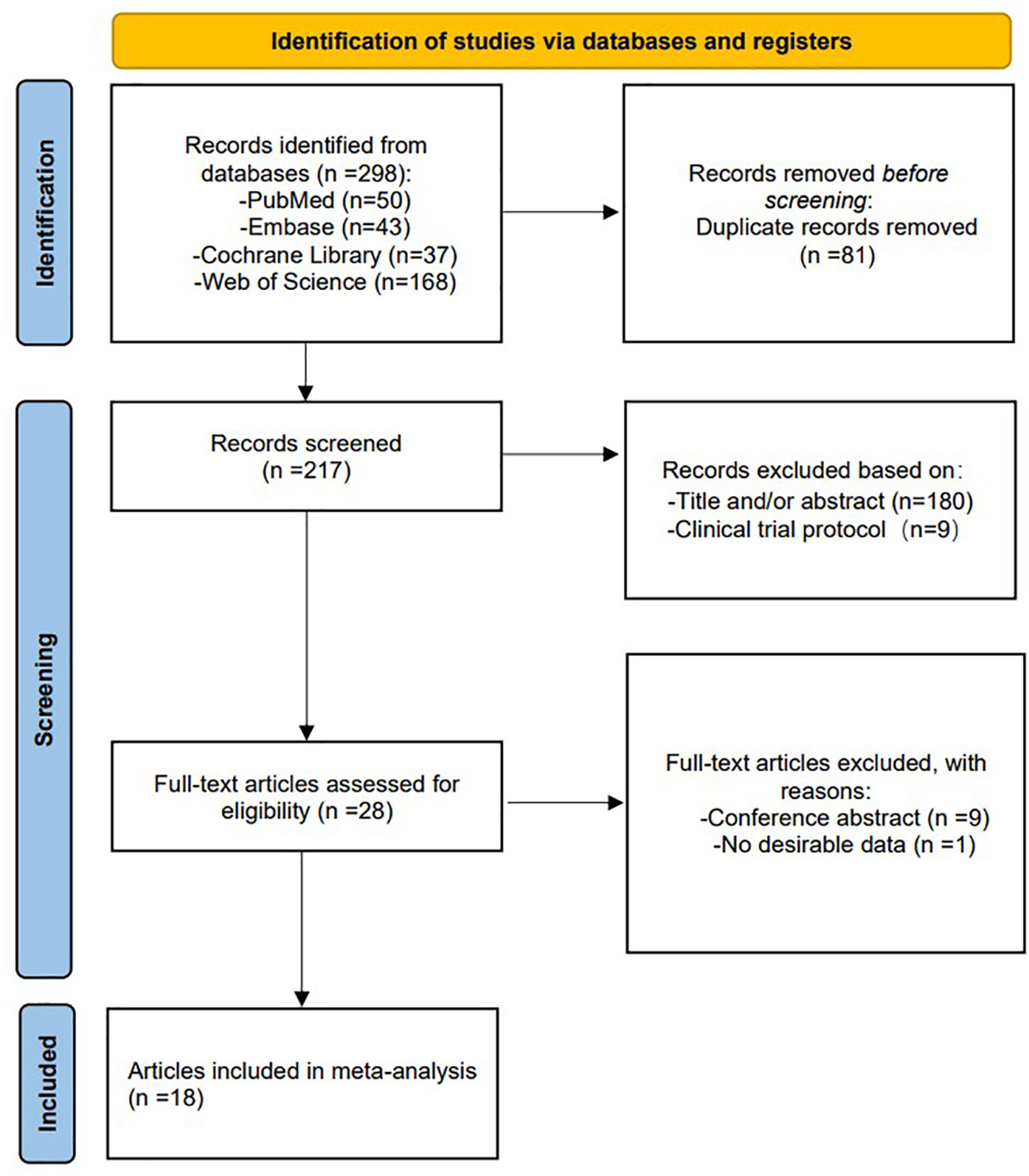
In total, 298 studies were retrieved and reviewed. 217 studies were retained after removing duplicates, with each title and abstract being evaluated by two reviewers. Subsequently, 28 full-text articles were screened for a full review, and 10 articles were excluded for the reasons outlined in the flowchart, leaving 18 studies (12–14, 17–31) that met the inclusion criteria. The PRISMA scheme for searching and selecting literature is shown in Figure 1.
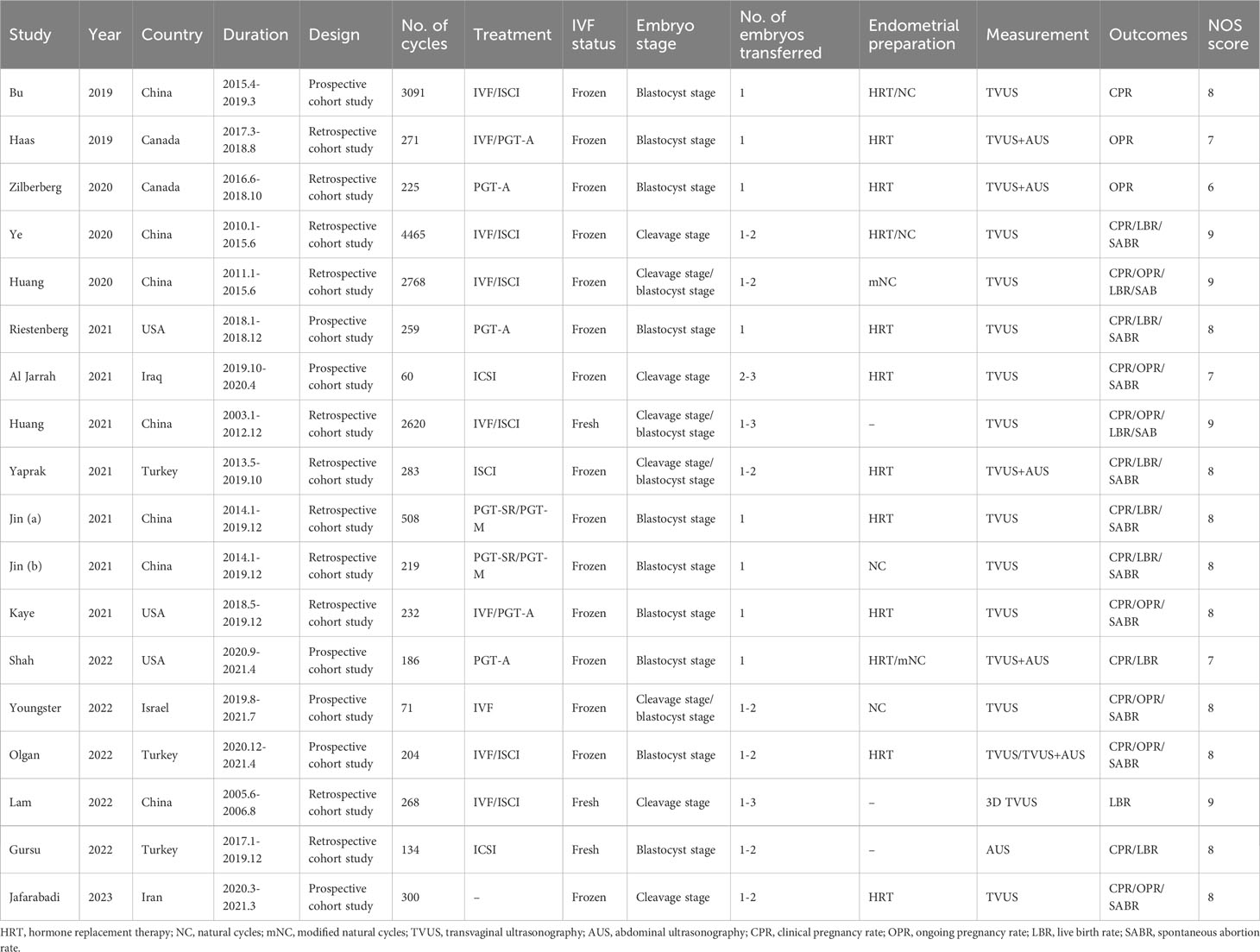
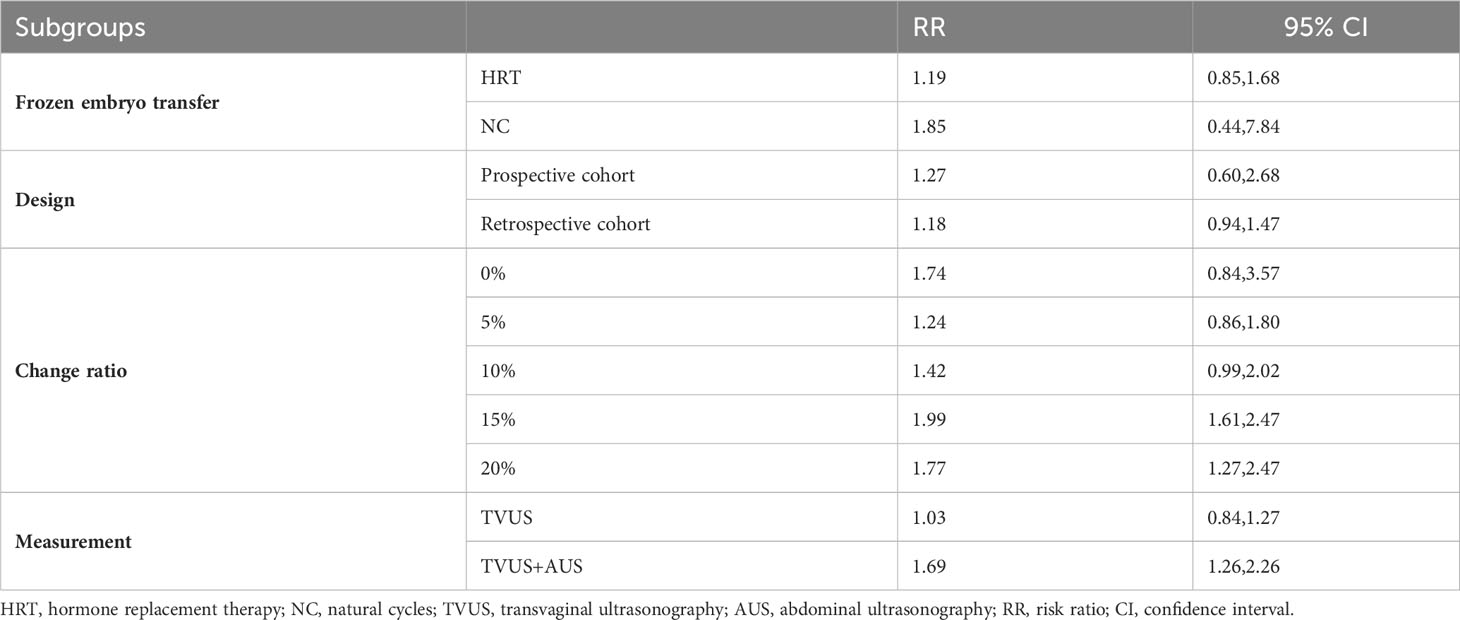
Table 1 shows the basic characteristics of the 18 studies, including first author’s surname, publication year, country, study duration, study design, number of cycles, endometrial preparation protocol, endometrial measurement method, number of embryos transferred, embryo development stage, outcome indicators, and quality score. Seven prospective studies and 11 retrospective studies were eligible for meta-analysis. Among the 18 included studies, 3 examined fresh ET, 15 examined FET. A total of 16,164 cycles were included, including 3022 fresh ET cycles, and 13,142 FET cycles (including 6481 hormone replacement therapy [HRT] cycles and 6661 natural cycles [NC] or modified natural cycles [mNC]).
In five studies of FET-HRT (13, 14, 22, 23, 31) and one study with fresh oocyte donation (26), ECR was defined as the rate of change in EMT between the day of progesterone administration and the day of ET. In 7 other FET-HRT studies (12, 18–20, 24, 25, 28), ECR was defined as the rate of change at which EMT changed from the end of estrogen-only phase to the ET or the day before the ET. In 2 fresh ETs (21, 27) and 2 FET-mNC (17, 29) studies, ECR refers to the rate at which EMT changes from hCG triggered to ET. Youngster et al. (30) described ECR as the rate of change from the day of ovulation to the day of ET.
Meta-analysis of clinical pregnancy rate
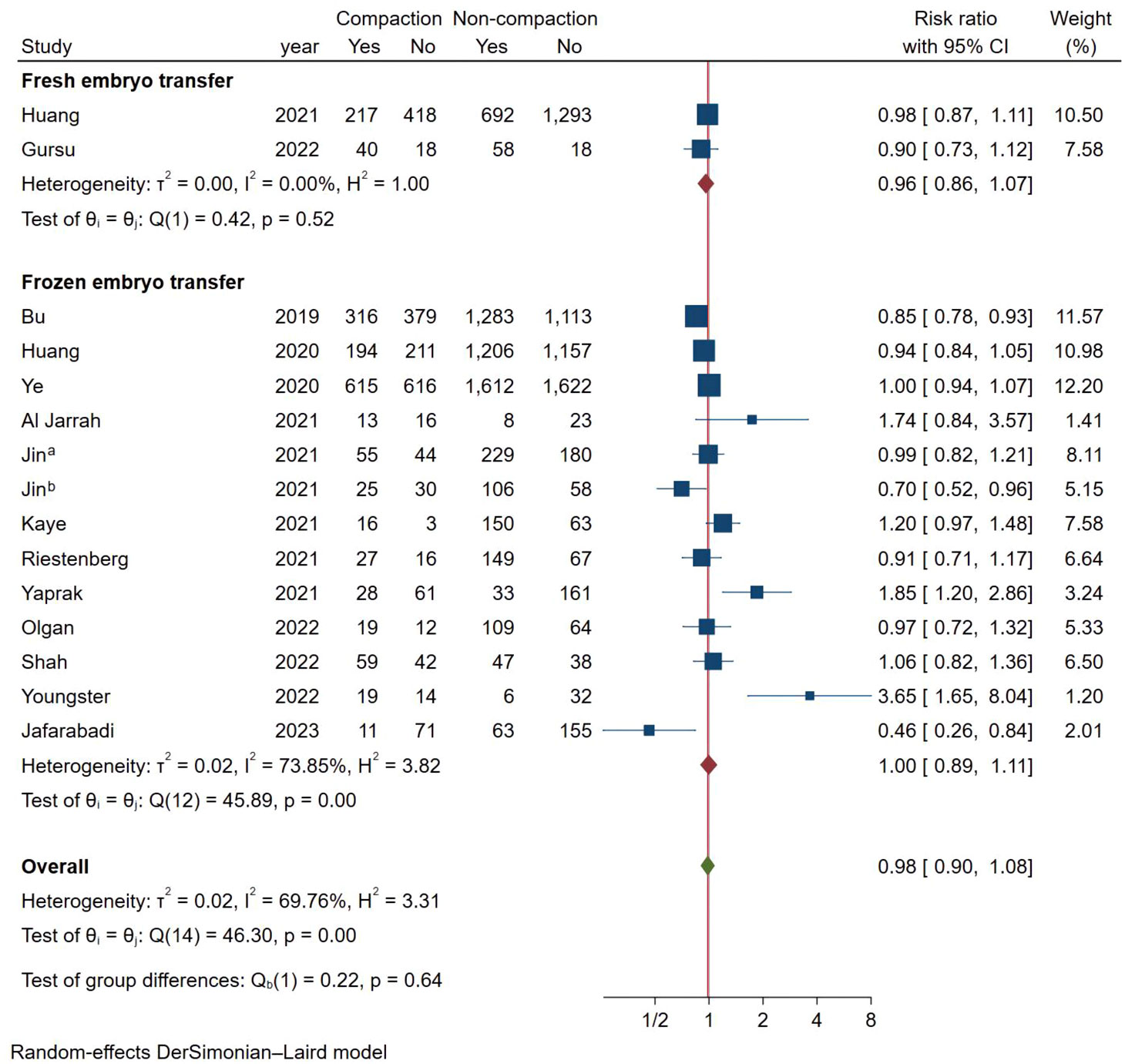
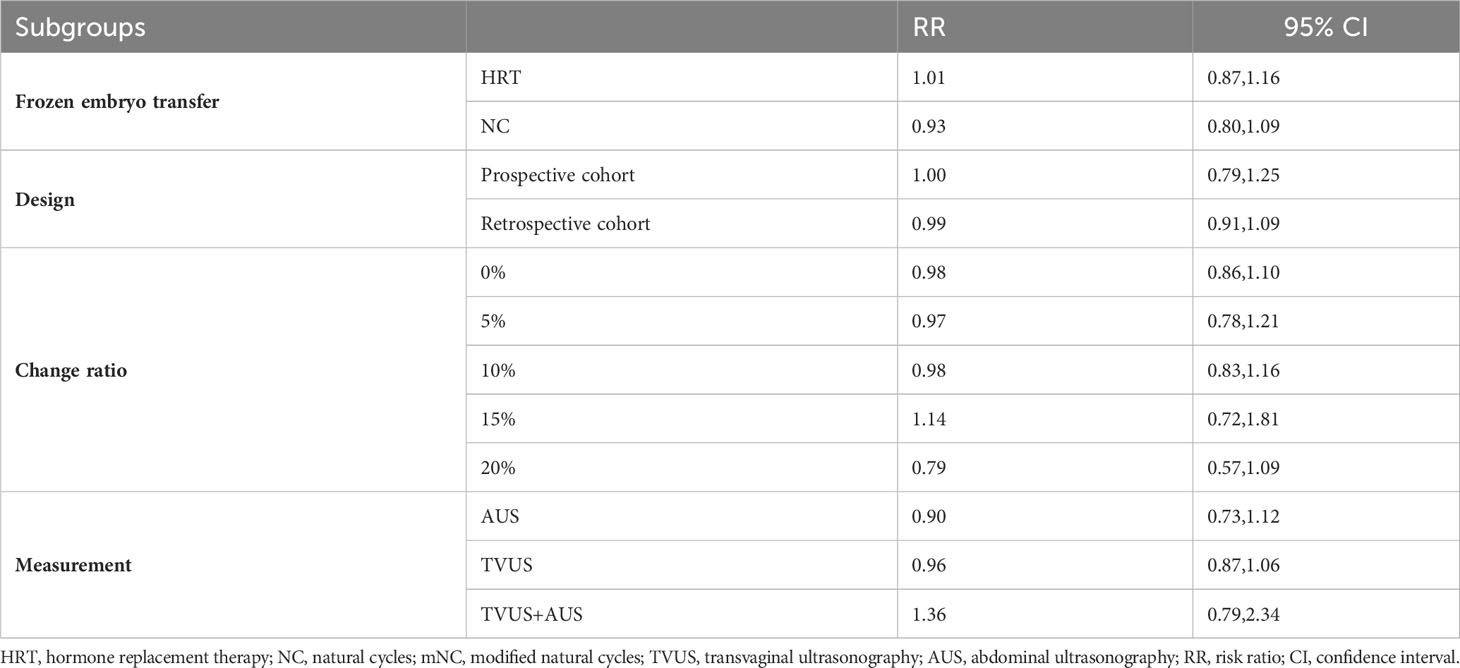
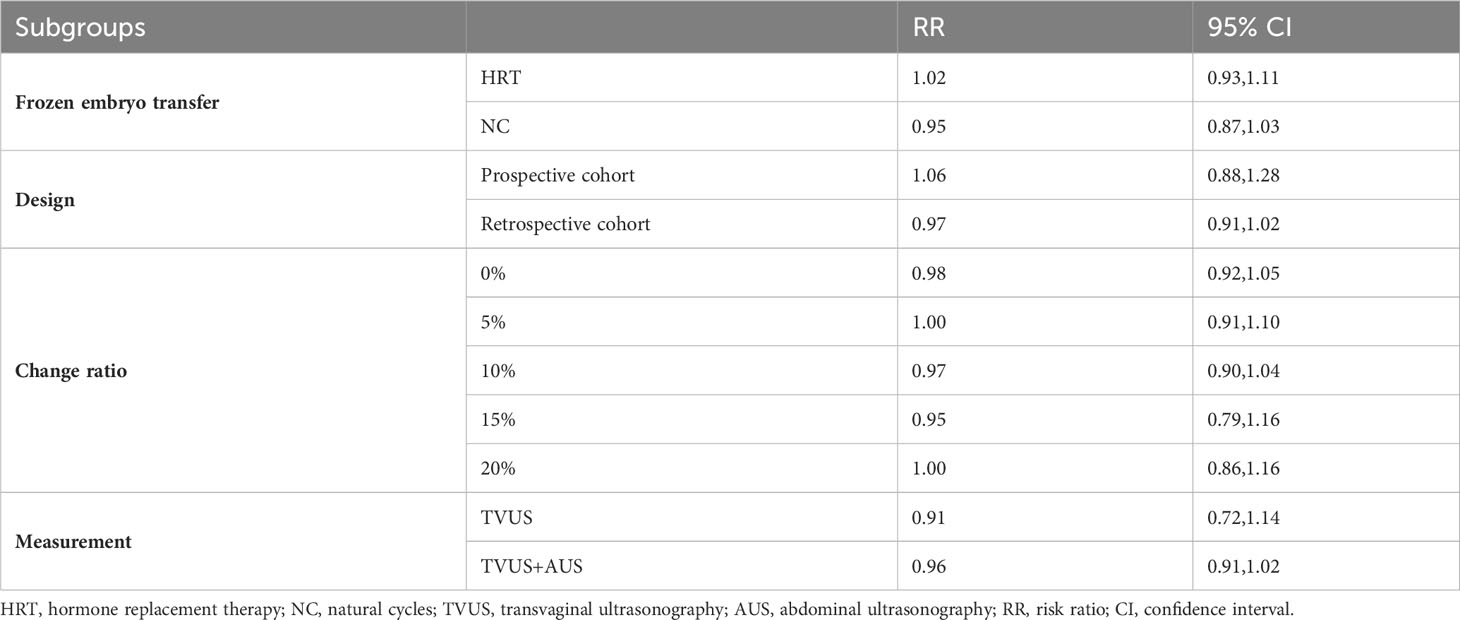
As shown in Figure 2, a total of 15 studies documented CPR, and the pooled results indicated that endometrial compaction was not associated with CPR (RR [95% CI] = 0.98 [0.90,1.08]; I2 = 69.76%), but there was high heterogeneity across the studies. Subgroup analysis revealed no statistically significant difference in CPR between endometrial compaction and non-compaction groups in the Fresh ET subgroup (RR [95% CI] = 0.96 [0.86,1.07]; I2 = 0.00%) or the FET subgroup (RR [95% CI] = 1.00 [0.89,1.11]; I2 = 73.85%). Moreover, we also performed subgroup analysis according to different preparation protocols, study design, measurement methods, and ECR. As shown in Table 2, there was no correlation between endometrial compaction and CPR.
Meta-analysis of ongoing pregnancy rate
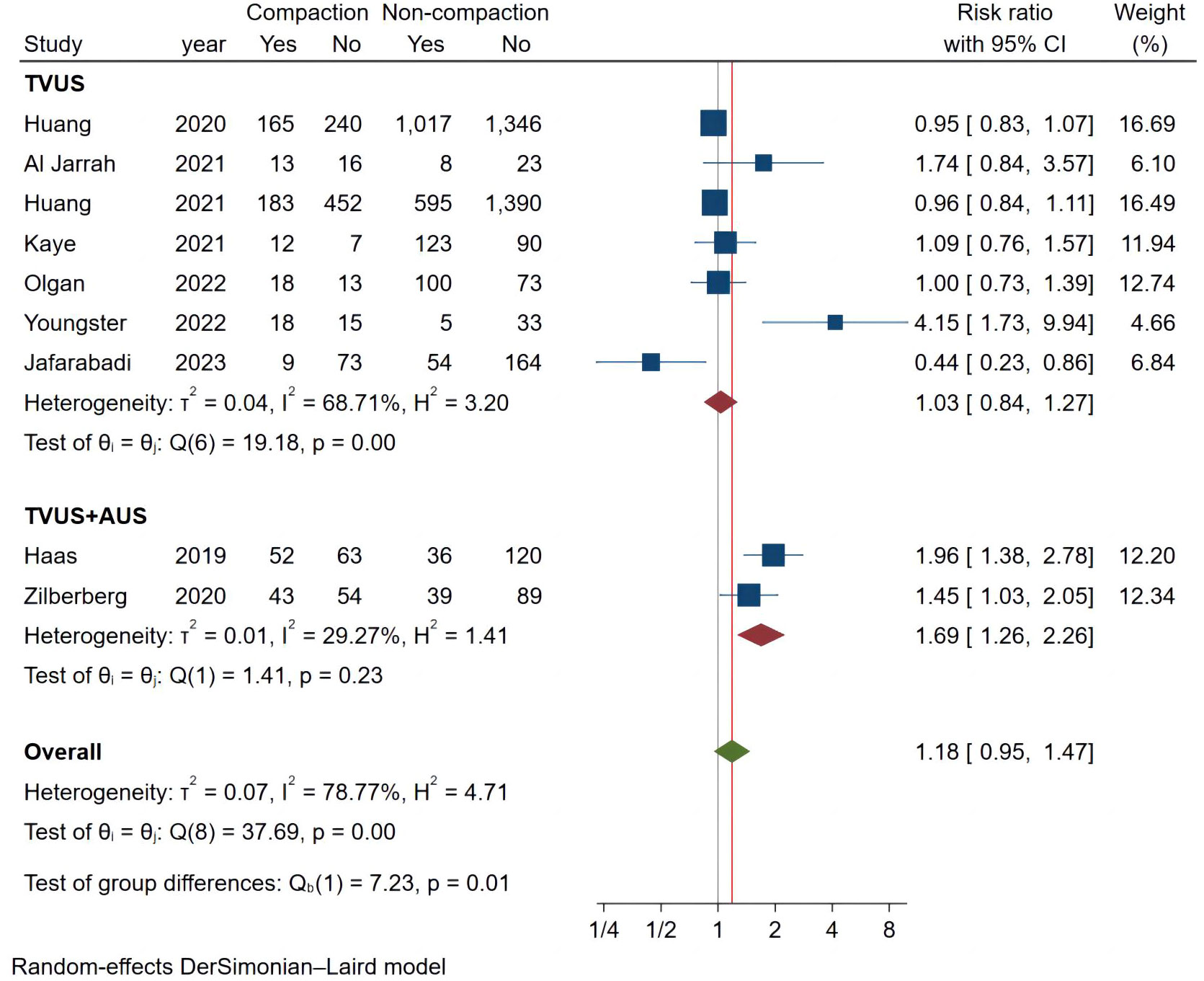
We analyzed 9 studies that met the requirements and the combined results are shown in Figure 3, where endometrial compaction was not associated with the OPR (RR [95% CI] = 1.18 [0.95,1.47]; I2 = 78.77%). There was high heterogeneity among studies, so we also performed a subgroup analysis with only one study in the Fresh ET group (RR [95% CI] = 0.96[0.84,1.11]; I2 = 0.00%) and 8 studies in the FET group (RR [95% CI] = 1.25 [0.93,1.68]; I2 = 80.64%), all of which showed no correlation between endometrial compaction and OPR.
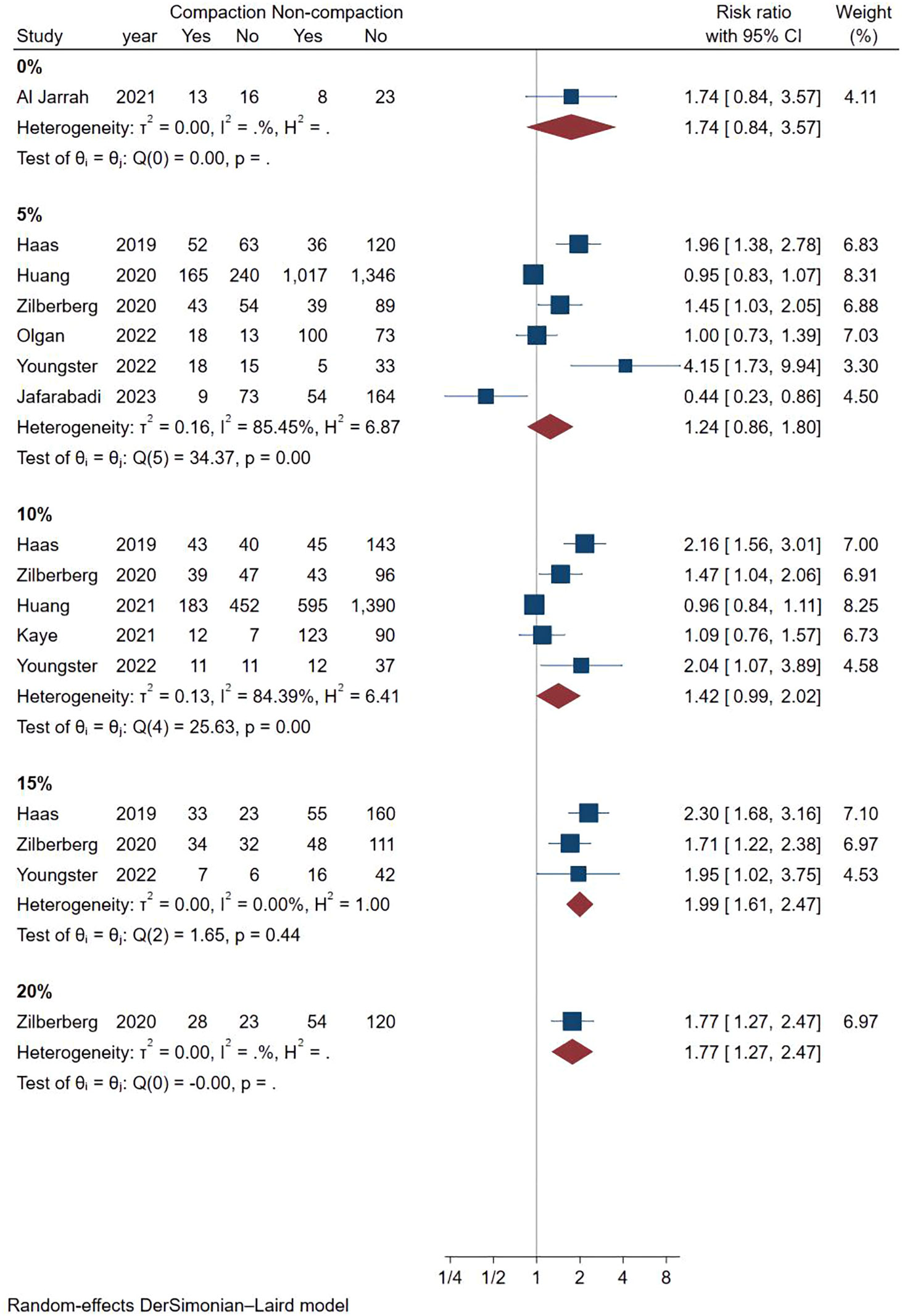
Considering the high heterogeneity between studies, we conducted more subgroup analyses. In a subgroup analysis of different ultrasound measurement methods, the OPR of the endometrial compaction group in the ET cycle was significantly higher than that of the non-compaction group (RR [95% CI]=1.69 [1.26, 2.26]; I2 = 29.27%), based on the combination of the first TVUS measurement and the second AUS measurement of EMT (see Figure 4). According to a subgroup analysis of different ECRs, the OPR was higher in the endometrial compaction group than in the non-compaction group (RR [95% CI] = 1.99 [1.61,2.47]; I2 = 0.00%). In the other subgroups, similar results were not observed, as shown in Figure 5. No statistical differences were found between the endometrial compaction and non-compaction groups in the subgroup analysis of other influencing factors, as shown in Table 3.
Meta-analysis of live birth
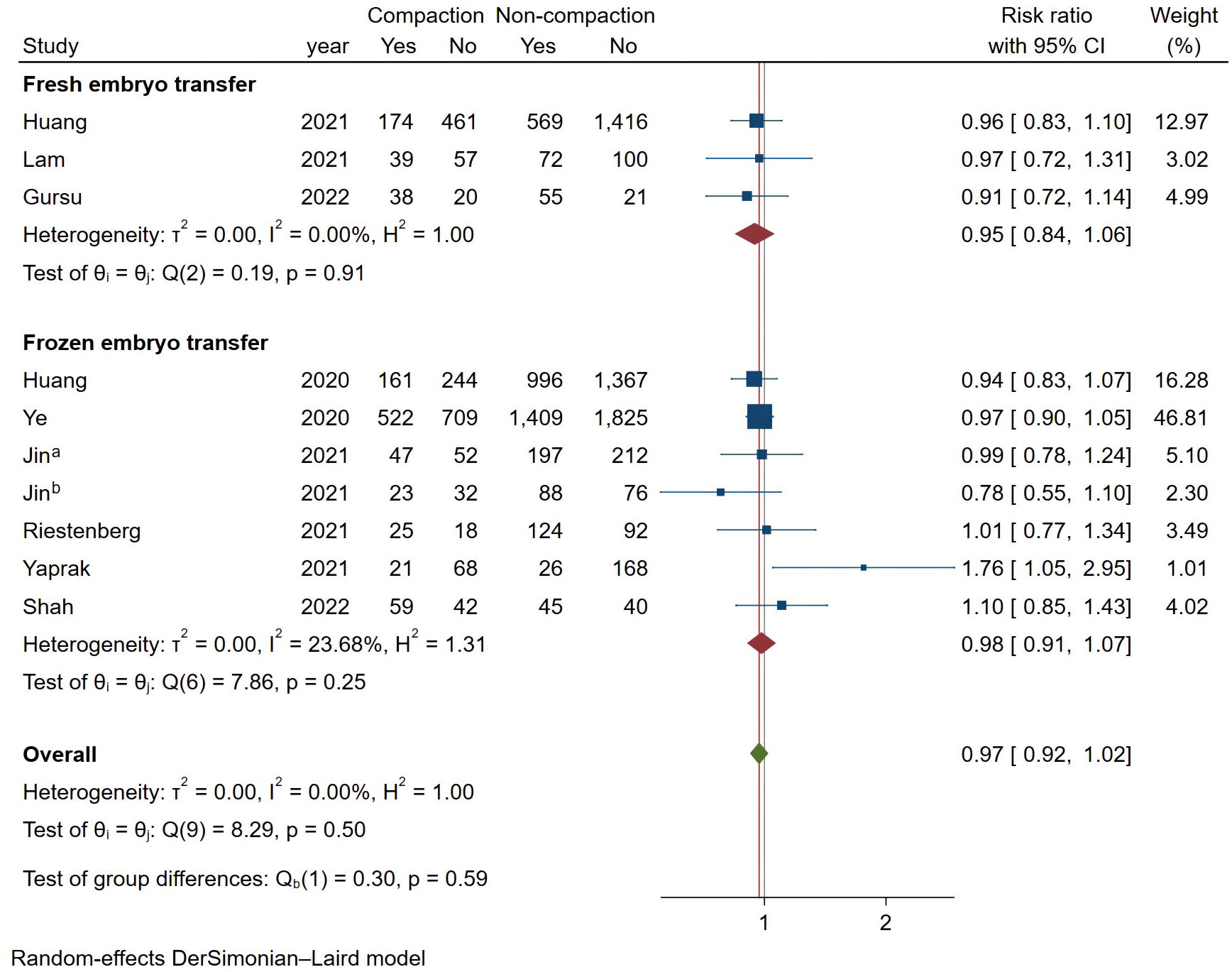
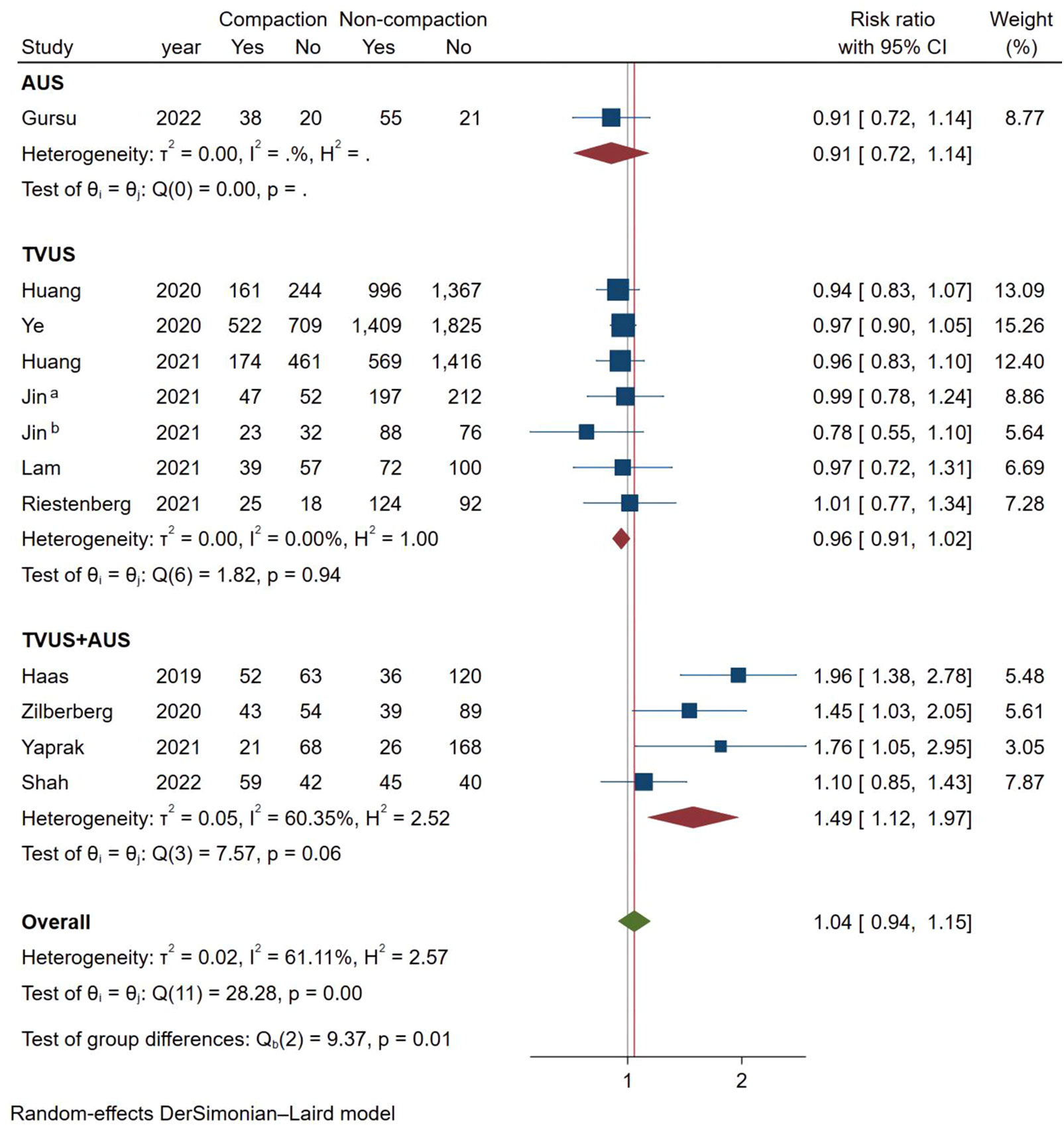
As shown in Figure 6, a total of 10 studies reported LBR using a fixed effects model (Mantel-Haenszel), and the combined results showed no statistically significant association between endometrial compaction and LBRs (RR [95% CI] = 0.97 [0.92,1.02]; I2 = 0.00%). In subgroup analysis, the Fresh ET group (RR [95% CI] = 0.95 [0.85,1.07]; I2 = 0.00%) and FET group (RR [95% CI] = 0.98 [0.92,1.04]; I2 = 23.68%) showed no significant difference between the endometrial compaction and non-compaction.
In the subgroup analysis of TVUS + AUS, the endometrial compaction group showed a slight improvement in LBR (RR [95% CI]=1.27 [1.00,1.61]; I2 = 62.28%). However, as shown in Figure 7, no significant statistical difference in LBR was detected when TVUS was used for both measurements prior to ET (RR [95% CI] = 0.96 [0.91,1.02]; I2 = 0.00%). As shown in Table 4, there were no significant differences in pooled RRs of LBR after conducting other subgroups analyses.
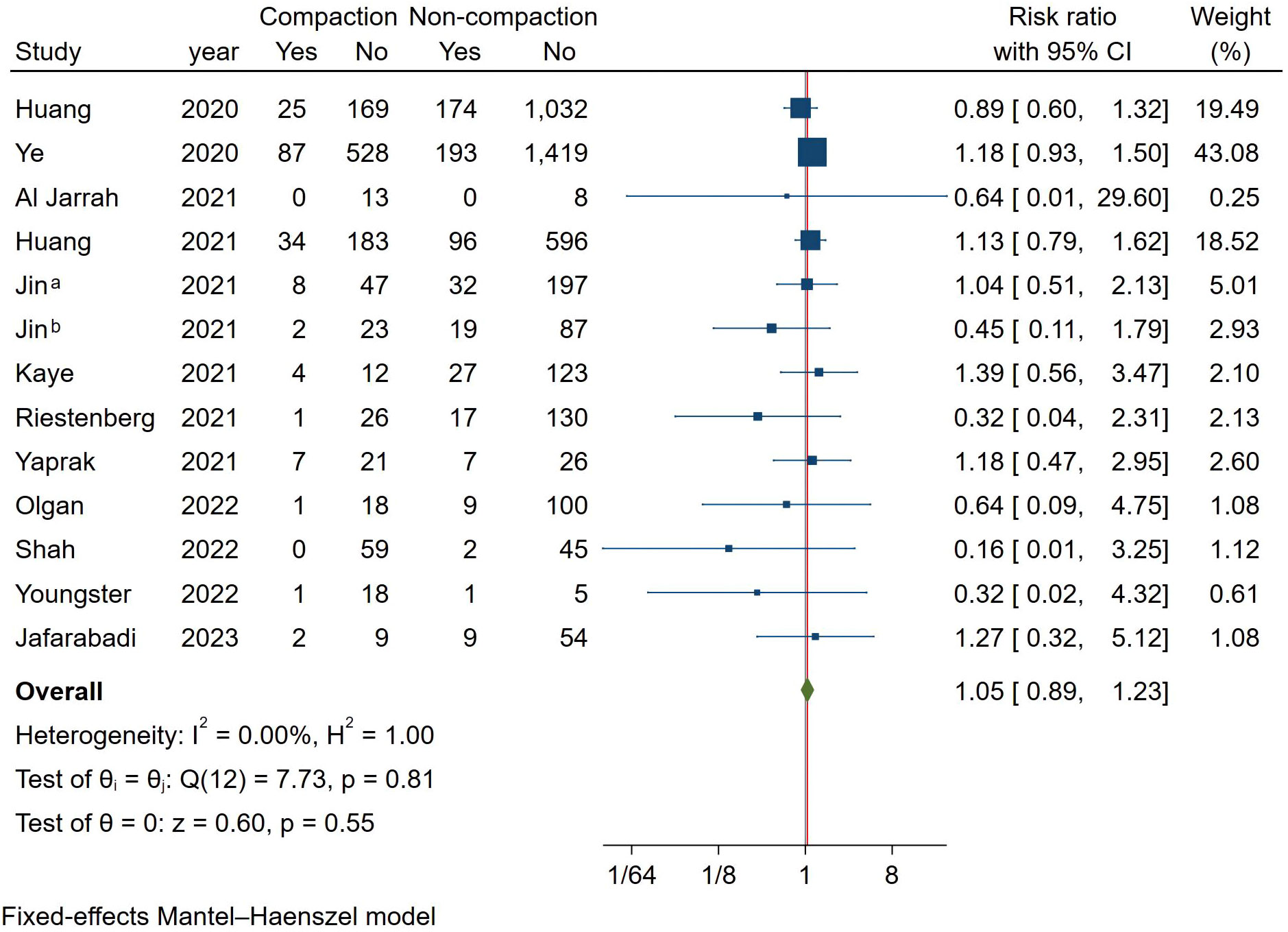
Meta-analysis of spontaneous abortion
As shown in Figure 8, we analyzed all studies that met the criteria. Because of the low heterogeneity between studies, a fixed-effect model with the Mantel-Haenszel method was used. Combined results showed that endometrial compaction was not associated with spontaneous abortion (RR [95% CI] = 1.05 [0.89,1.23]; I2 = 0.00%).
Publication bias
The funnel plot for each outcome was visually symmetric (see Supplementary Appendix 3-1, 3-2, 3-3, 3-4). Furthermore, the regression-based Egger test did not show statistical significance (P = 0.0512, 0.120, 0.4295 and 0.0727 for clinical pregnancy, ongoing pregnancy, live birth and spontaneous abortion, respectively), suggesting that there was no significant publication bias in the studies that were included.
Discussion
EMT is currently used widely in the field of IVF as a clinical predictor of pregnancy outcomes. Most previous studies focused on EMT’s effect on pregnancy outcomes in IVF. Generally, thinner endometrium is associated with poorer pregnancy outcomes (2, 32). In the last decade, only a few studies have focused on the effect of endometrial compaction on pregnancy outcomes, and the results are contradictory. Thus, we designed this systematic review to evaluate the impact of endometrial compaction on IVF outcomes. CPR, OPR, LBR, and spontaneous abortion rates were not significantly affected by endometrial compaction. However, a subgroup analysis showed that endometrial compaction may be associated with an increase in OPR and LBR when TVUS was used for the first measurement of EMT and AUS was used for the second measurement of EMT. Meanwhile, when ECR ≥ 15%, LBR also appeared to be better, despite significant heterogeneity between studies.
A total of 7 prospective studies and 11 retrospective studies were included. Of these, 11 studies (17, 18, 20–22, 24, 26–29, 31) did not find a statistically significant association between endometrial compaction and pregnancy outcomes, whereas in 5 studies (12, 19, 23, 25, 30) better pregnancy outcomes were reported. In contrast, Bu et al. (14) and Jin et al. (13) concluded that an increased endometrium after progesterone administration was associated with better pregnancy outcomes. In the present meta-analysis, combined results showed that endometrial compaction was not associated with CPR, OPR, LBR, or spontaneous abortion rate.
Because of the heterogeneity among studies, we performed subgroup analyses. The subgroup analysis according to the ultrasound measurement method indicated that using AUS at the time of ET resulted in higher OPR and LBR in the endometrial compaction group than in the non-compaction group. These findings are consistent with those reported by Haas (12), Zilberberg (19), and Yaprak (25). While continuous EMT monitoring using TVUS did not reveal a correlation between endometrial compaction and pregnancy outcome, in the subgroup analysis. It is generally accepted that EMT measured by TVUS are more accurate than those measured by AUS. According to our hypothesis, AUS measurement results in a thinner EMT as well as a higher incidence of endometrial compaction as a result of pressure placed on the abdomen while AUS measurements are taken. The study of Olgan et al. (28) compared two different measurements and the conclusions obtained were consistent with our conjecture. We believe that the data measured by sequential TVUS are more reliable than those measured by TVUS and AUS. Hence, TVUS may be a good choice for future researchers who wish to reduce measurement errors.
In this study, we also performed subgroup analysis according to different ECRs. Our study showed that endometrial compaction resulted in a higher OPR when 15% was used as a cut-off value for ECR. However, two of the three studies included combined transvaginal and transabdominal measurements. As a consequence of the small number of included studies, it is not possible to exclude the effect of measurement methods on study outcomes.
In the FET-HRT analysis, endometrial compaction did not significantly affect OPR and LBR. Nonetheless, this compares with Oliveira et al. ‘s findings (33), which found that endometrial compaction during FET-HRT cycles benefited OPR and LBR. In our study, we analyzed OPR and LBR separately and included data from more recent studies, which may explain the differences. Therefore, more well-designed clinical trials are needed to validate an innovative concept before it can be introduced into clinical practice.
Additionally, the researchers observed the changes in endometrium during the natural cycle and found that the thickness and volume increased rapidly during the follicular phase and decreased slightly after ovulation (34, 35). In the follicular phase, estrogen accelerates the proliferation and growth of endometrial glands and vessels, which exhibit a typical trilinear ultrasound sign. After ovulation, the endometrium becomes more curved and vascularized by progesterone, and the EMT becomes thinner or unchanged, which appears hyperechoic on ultrasound. Researchers (12, 19, 20, 23, 30, 36) reported that the degree of endometrial compaction can indicate the degree of endometrial response to progesterone and contribute to an assessment of ER. However, Olgan et al. (28) and Youngster et al. (30) demonstrated that progesterone levels and estrogen to progesterone ratio were not associated with endometrial compaction. Thus, further studies may identify factors affecting endometrial compaction and how endometrial compaction improves pregnancy outcomes from the perspective of progesterone resistance and progesterone receptor expression.
There are some limitations to this meta-analysis. First of all, most of the included literature is retrospective cohort studies. Secondly, there are differences in embryo culture methods, ET staging, EMT measurement methods, ECR thresholds, and clinical outcome evaluation. This results in significant heterogeneity between individual studies. Further, only three studies on fresh ET were included, including one on the oocyte donation cycle. Insufficient studies were included to obtain reliable results.
Conclusion
In summary, the present evidence suggests that endometrial compaction is not sufficient for predicting pregnancy outcomes. Also, the choice of endometrial measurement methods is a key factor influencing endometrial compaction assessment. Besides reducing unnecessary cycle cancellations, this finding may also provide an instructive basis for future studies. To confirm this finding, more well-designed, large-scale prospective studies should be conducted.
Data availability statement
The original contributions presented in the study are included in the article/supplementary material. Further inquiries can be directed to the corresponding author.
Author contributions
X-TC: Data curation, Formal Analysis, Writing – original draft. Z-GS: Conceptualization, Funding acquisition, Methodology, Writing – review & editing, Data curation. J-YS: Conceptualization, Software, Supervision, Writing – review & editing.
Funding
The author(s) declare that no financial support was received for the research, authorship, and/or publication of this article.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fendo.2023.1264608/full#supplementary-material
Supplementary Appendix 1 | Search strategy
Supplementary Appendix 2 | Newcastle-Ottawa Scale
Supplementary Appendix 3-1 | Funnel plot of clinical pregnancy.
Supplementary Appendix 3-2 | Funnel plot of ongoing pregnancy.
Supplementary Appendix 3-3 | Funnel plot of live birth.
Supplementary Appendix 3-4 | Funnel plot of spontaneous abortion.
References
1. Zhou Z, Zheng D, Wu H, Li R, Xu S, Kang Y, et al. Epidemiology of infertility in China: a population-based study. BJOG (2018) 125(4):432–41. doi: 10.1111/1471-0528.14966
2. Kasius A, Smit JG, Torrance HL, Eijkemans MJ, Mol BW, Opmeer BC, et al. Endometrial thickness and pregnancy rates after IVF: a systematic review and meta-analysis. Hum Reprod Update (2014) 20(4):530–41. doi: 10.1093/humupd/dmu011
3. Guzeloglu-Kayisli O, Kayisli UA, Taylor HS. The role of growth factors and cytokines during implantation: endocrine and paracrine interactions. Semin Reprod Med (2009) 27(1):62–79. doi: 10.1055/s-0028-1108011
4. Cimadomo D, Rienzi L, Conforti A, Forman E, Canosa S, Innocenti F, et al. Opening the black box: why do euploid blastocysts fail to implant? A systematic review and meta-analysis. Hum Reprod Update (2023) 29(5):570–633. doi: 10.1093/humrep/dead093.234
5. Craciunas L, Gallos I, Chu J, Bourne T, Quenby S, Brosens JJ, et al. Conventional and modern markers of endometrial receptivity: a systematic review and meta-analysis. Hum Reprod Update (2019) 25(2):202–23. doi: 10.1093/humupd/dmy044
6. Bergh PA, Navot D. The impact of embryonic development and endometrial maturity on the timing of implantation. Fertil Steril (1992) 58(3):537–42. doi: 10.1016/S0015-0282(16)55259-5
7. Mahutte N, Hartman M, Meng L, Lanes A, Luo ZC, Liu KE. Optimal endometrial thickness in fresh and frozen-thaw in vitro fertilization cycles: an analysis of live birth rates from 96,000 autologous embryo transfers. Fertil Steril (2022) 117(4):792–800. doi: 10.1016/j.fertnstert.2021.12.025
8. Liu KE, Hartman M, Hartman A, Luo ZC, Mahutte N. The impact of a thin endometrial lining on fresh and frozen-thaw IVF outcomes: an analysis of over 40 000 embryo transfers. Hum Reprod (2018) 33(10):1883–8. doi: 10.1093/humrep/dey281
9. Aboulghar MA, Aboulghar MM. Optimum endometrial thickness before embryo transfer: an ongoing debate. Fertil Steril (2023) 120(1):99–100. doi: 10.1016/j.fertnstert.2023.04.013
10. Gao G, Cui X, Li S, Ding P, Zhang S, Zhang Y. Endometrial thickness and IVF cycle outcomes: a meta-analysis. Reprod BioMed Online (2020) 40(1):124–33. doi: 10.1016/j.rbmo.2019.09.005
11. Ata B, Linan A, Kalafat E, Ruiz F, Melado L, Bayram A, et al. Effect of the endometrial thickness on the live birth rate: insights from 959 single euploid frozen embryo transfers without a cutoff for thickness. Fertil Steril (2023) 120(1):91–8. doi: 10.1097/01.ogx.0000979684.30759.bd
12. Haas J, Smith R, Zilberberg E, Nayot D, Meriano J, Barzilay E, et al. Endometrial compaction (decreased thickness) in response to progesterone results in optimal pregnancy outcome in frozen-thawed embryo transfers. FERTILITY AND STERILITY (2019) 112(3):503–+. doi: 10.1016/j.fertnstert.2019.05.001
13. Jin ZQ, Li JD, Yang ET, Shi H, Bu ZQ, Niu WB, et al. Effect of endometrial thickness changes on clinical pregnancy rates after progesterone administration in a single frozen-thawed euploid blastocyst transfer cycle using natural cycles with luteal support for PGT-SR- and PGT-M-assisted reproduction: a retrospective cohort study. Reprod Biol Endocrinol (2021) 19(1):154. doi: 10.1186/s12958-021-00841-x
14. Bu Z, Yang X, Song L, Kang B, Sun Y. The impact of endometrial thickness change after progesterone administration on pregnancy outcome in patients transferred with single frozen-thawed blastocyst. Reprod Biol Endocrinol (2019) 17(1):99. doi: 10.1186/s12958-019-0545-0
15. Page MJ, Moher D, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. PRISMA 2020 explanation and elaboration: updated guidance and exemplars for reporting systematic reviews. Bmj (2021) 372:n160. doi: 10.1136/bmj.n160
16. Hatala R, Keitz S, Wyer P, Guyatt G. Tips for learners of evidence-based medicine: 4. Assessing heterogeneity of primary studies in systematic reviews and whether to combine their results. Cmaj (2005) 172(5):661–5. doi: 10.1503/cmaj.1031920
17. Huang J, Lin J, Cai R, Lu X, Song N, Gao H, et al. Significance of endometrial thickness change after human chorionic gonadotrophin triggering in modified natural cycles for frozen-thawed embryo transfer. Ann Trans Med (2020) 8(23):1590. doi: 10.21037/atm-20-1459
18. Ye J, Zhang J, Gao HY, Zhu YW, Wang Y, Cai RF, et al. Effect of endometrial thickness change in response to progesterone administration on pregnancy outcomes in frozen-thawed embryo transfer: analysis of 4465 cycles. Front Endocrinol (Lausanne) (2020) 11:546232. doi: 10.3389/fendo.2020.546232
19. Zilberberg E, Smith R, Nayot D, Haas J, Meriano J, Barzilay E, et al. Endometrial compaction before frozen euploid embryo transfer improves ongoing pregnancy rates. FERTILITY AND STERILITY (2020) 113(5):990–5. doi: 10.1016/j.fertnstert.2019.12.030
20. Al Jarrah DM, Obaidi MTA, Al Asadi IJ. Endometrial compaction and serum progesterone measurements at the day of embryo transfer cannot predict pregnancy outcomes in frozen-thaw embryo transfer cycles. Int J Res Pharm Sci (2021) 12(1):407–15.
21. Huang JY, Lin JY, Gao HY, Zhu J, Lu XF, Song N, et al. Value of endometrial thickness change after human chorionic gonadotrophin administration in predicting pregnancy outcome following fresh transfer in vitro fertilization cycles. Arch OF GYNECOLOGY AND OBSTETRICS (2021) 303(2):565–72. doi: 10.1007/s00404-020-05763-4
22. Jin Z, Shi H, Lu M, Bu Z, Huo M, Zhang Y. Endometrial thickness changes after progesterone administration do not affect the pregnancy outcomes of frozen-thawed euploid blastocyst transfer: a retrospective cohort study. Fertil Steril (2021) 116(6):1502–12. doi: 10.1016/j.fertnstert.2021.08.008
23. Kaye L, Rasouli MA, Liu A, Raman A, Bedient C, Garner FC, et al. The change in endometrial thickness following progesterone exposure correlates with in vitro fertilization outcome after transfer of vitrified-warmed blastocysts. J OF ASSISTED Reprod AND Genet (2021) 38(11):2947–53. doi: 10.1007/s10815-021-02327-6
24. Riestenberg C, Quinn M, Akopians A, Danzer H, Surrey M, Ghadir S, et al. Endometrial compaction does not predict live birth rate in single euploid frozen embryo transfer cycles. J OF ASSISTED Reprod AND Genet (2021) 38(2):407–12. doi: 10.1007/s10815-020-02043-7
25. Yaprak E, Şükür YE, Özmen B, Sönmezer M, Berker B, Atabekoğlu C, et al. Endometrial compaction is associated with the increased live birth rate in artificial frozen-thawed embryo transfer cycles. Hum Fertil (Camb) (2023) 26(3):550–6. doi: 10.1080/14647273.2021.1966703
26. Gursu T, Goksever Celik H, Eraslan A, Angun B, Ozaltin S, Yeh J, et al. Impact of endometrial thickness change in response to progesterone on live birth rates embryo transfers with fresh oocyte donation cycles. J Obstet Gynaecol (2022) 42(7):3260–7. doi: 10.1080/01443615.2022.2112022
27. Lam MT, Li HR, Ng EHY. Impact of endometrial thickness and volume compaction on the live birth rate following fresh embryo transfer of in vitro fertilization. J OF ULTRASOUND IN Med (2022) 41(6):1455–63. doi: 10.1002/jum.15830
28. Olgan S, Dirican EK, Sakinci M, Caglar M, Ozsipahi AC, Gul SM, et al. Endometrial compaction does not predict the reproductive outcome after vitrified–warmed embryo transfer: a prospective cohort study. Reprod BioMedicine Online (2022) 45(1):81–7. doi: 10.1016/j.rbmo.2022.02.025
29. Shah JS, Vaughan DA, Dodge LE, Leung A, Korkidakis A, Sakkas D, et al. Endometrial compaction does not predict live birth in single euploid frozen embryo transfers: a prospective study. Hum Reprod (2022) 37(5):980–7. doi: 10.1093/humrep/deac060
30. Youngster M, Mor M, Kedem A, Gat I, Yerushalmi G, Gidoni Y, et al. Endometrial compaction is associated with increased clinical and ongoing pregnancy rates in unstimulated natural cycle frozen embryo transfers: a prospective cohort study. J OF ASSISTED Reprod AND Genet (2022) 39(8):1909–16. doi: 10.1007/s10815-022-02544-7
31. Jafarabadi MN, Dastjerdi MA, Rashidi BH, Shariat M, Haghollahi F. Does endometrial compaction predict clinical pregnancy rate after cleavage stage frozen embryo transfer? CRESCENT J OF Med AND Biol Sci (2023) 10:132–6. doi: 10.34172/cjmb.2023.22
32. Momeni M, Rahbar MH, Kovanci E. A meta-analysis of the relationship between endometrial thickness and outcome of in vitro fertilization cycles. J Hum Reprod Sci (2011) 4(3):130–7. doi: 10.4103/0974-1208.92287
33. Oliveira JBA, Petersen CG, Massaro FC, Petersen B, Vagnini L, Nicoletti A, et al. Endometrial compaction in artificial frozen-thawed embryo transfer cycles is associated with improvement in pregnancy outcomes: a meta-analysis. Hum Reprod (2022) 37:I183–4. doi: 10.1093/humrep/deac106.100
34. Jokubkiene L, Sladkevicius P, Rovas L, Valentin L. Assessment of changes in endometrial and subendometrial volume and vascularity during the normal menstrual cycle using three-dimensional power Doppler ultrasound. Ultrasound Obstet Gynecol (2006) 27(6):672–9. doi: 10.1002/uog.2742
35. Raine-Fenning NJ, Campbell BK, Clewes JS, Kendall NR, Johnson IR. Defining endometrial growth during the menstrual cycle with three-dimensional ultrasound. BJOG: Int J Obstetrics Gynaecology (2004) 111(9):944–9. doi: 10.1111/j.1471-0528.2004.00214.x
Keywords: endometrial compaction, transvaginal ultrasound, in vitro fertilization, frozen-thawed embryo transfer, endometrial thickness
Citation: Chen X-T, Sun Z-G and Song J-Y (2023) Does endometrial compaction before embryo transfer affect pregnancy outcomes? a systematic review and meta-analysis. Front. Endocrinol. 14:1264608. doi: 10.3389/fendo.2023.1264608
Received: 21 July 2023; Accepted: 30 October 2023;
Published: 14 November 2023.
Edited by:
Rong Li, Peking University Third Hospital, ChinaReviewed by:
Chuanlong Zhang, China Academy of Chinese Medical Sciences, ChinaZhiqin Bu, Zhengzhou University, China
Copyright © 2023 Chen, Sun and Song. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Jing-Yan Song, aGFubGluZ2p1emVpOTFAMTI2LmNvbQ==; Zhen-Gao Sun, c3Vuemhlbmdhbzc3QDEyNi5jb20=
 Xiao-Tong Chen
Xiao-Tong Chen Zhen-Gao Sun
Zhen-Gao Sun Jing-Yan Song
Jing-Yan Song