- 1College of Animal Science and Veterinary Medicine, Henan Agricultural University, Zhengzhou, China
- 2State Key Laboratory of Veterinary Etiological Biology, Key Laboratory of Veterinary Parasitology of Gansu Province, Lanzhou Veterinary Research Institute, Chinese Academy of Agricultural Sciences, Lanzhou, China
- 3Center for Animal Disease Control and Prevention of Henan Province, Zhengzhou, China
Toxoplasma gondii is an important cause of reproductive failure in small ruminants that also poses a risk to consumers who consume undercooked meat. However, little is known about sheep toxoplasmosis in China for the world. Therefore, this study was conducted to assess the prevalence of T. gondii infection in sheep from China, to isolate T. gondii via bioassay in mice and to evaluate the virulence of the isolated T. gondii based on vero cell invasion and mice. A total of 840 samples (304 unfrozen hearts and 536 sera) from sheep in China were collected from 2014 to 2016. Heart samples (n = 36) of T. gondii seropositive sheep (MAT, ≥25) were bioassayed in mice individually. DNA derived from cell cultured tachyzoites of the isolated T. gondii was characterized by PCR-RFLP of 10 loci (SAG1, SAG2, SAG3, BTUB, GRA6, c22-8, c29-2, L358, PK1, and Apico). The virulence of the T. gondii was evaluated based on the mortality and encystation in mice, as well as their growth characteristics in cell culture. Antibodies to T. gondii were found in 174 of 840 (20.71%, 304 hearts juice and 536 sera) sheep by the modified agglutination test (cut-off 1:25). Viable T. gondii was isolated from the hearts of two of 36 seropositive sheep hearts. Both genotypes of the sheep heart isolates were ToxoDB#9. The virulence of the two ToxoDB#9 isolations varied significantly. To the best of our knowledge, this is the first report of isolation of ToxoDB#9 strain of T. gondii from sheep in China.
Introduction
The parasite Toxoplasma gondii is a major cause of reproductive failure in small ruminants, including sheep. Veterinary Investigation Diagnosis Analysis data from 2014 showed that about 25% of ovine production problems were caused by T. gondii (www.gov.uk/government/statistics). Moreover, viable T. gondii has been isolated from goat meat, milk and cheese (Dubey et al., 2014a,b). T. gondii infection is widespread among humans. The prevalence of T. gondii is higher in Latin America than in North America and East Asia (Dubey, 2010); however, the cause of this difference is not known. T. gondii cause lymphadenopathy, retinochoroiditis, encephalitis, abortion, and the death of immunocompromised patients (Hide, 2016). In China, mutton is the main ingredient of hotpot, which often results in meat being undercooked. Therefore, the consumption of undercooked meat containing T. gondii tissue cysts could pose a health risk to consumers.
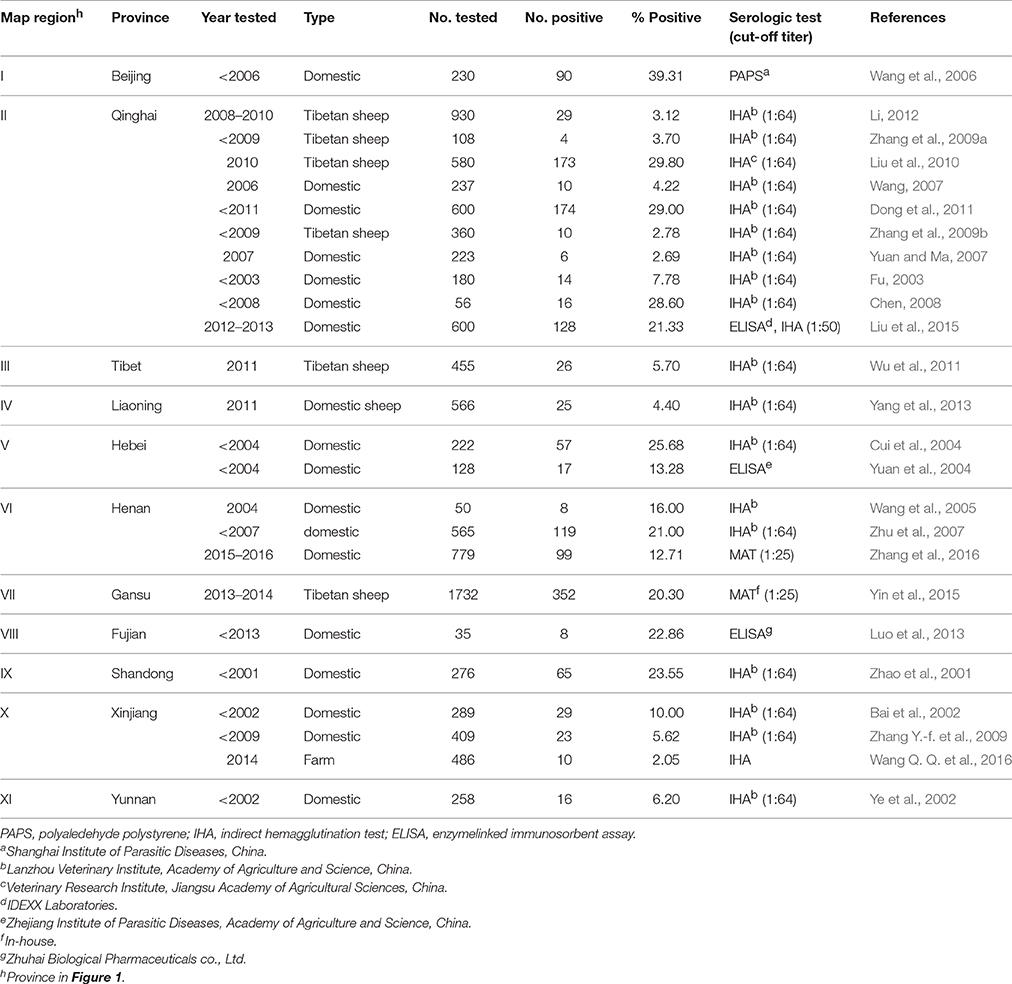
Isolation of viable T. gondii from feline has been the most successful model in China. Among 122 viable T. gondii isolates from animals and humans in China, 85 strains (69.7%) were isolated from tissue or fecal samples from cats. Moreover, 73 (85.9%) T. gondii isolates from cats were genotyped as ToxoDB#9, while 10 were genotyped as ToxoDB#1(1), ToxoDB#2(1), ToxoDB#10(1), ToxoDB#17(1), ToxoDB#18(2), and ToxoDB#205(4), and the genotypes of the other two strains were not determined (Dubey et al., 2007; Zhou et al., 2009; Chen et al., 2011; Qian et al., 2012; Wang et al., 2013a; Li et al., 2015; Yang et al., 2015; Wang D. et al., 2016; Wang Q. Q. et al., 2016). However, only one isolate of T. gondii has been obtained from sheep in Qinghai (type II) (Zhou et al., 2009), and no studies have reported isolation of viable T. gondii from other small ruminants in China.
China has the largest number of sheep in the world, with an estimated 187 million domestic sheep (https://top5ofanything.com/list/d4d1ef5e/Countries-With-the-Most-Sheep) and an unknown number of wild sheep. The clinical and economic importance of sheep toxoplasmosis remains uncertain, and most epidemiological literature was published in Chinese. Therefore, the present study was conducted to summarize these Chinese papers and present the sero-prevalence in sheep from different geographical areas. Further, the prevalence of T. gondii infections in sheep from China was investigated, and an attempt to isolate viable T. gondii was made.
Materials and Methods
Sheep Sample Collection
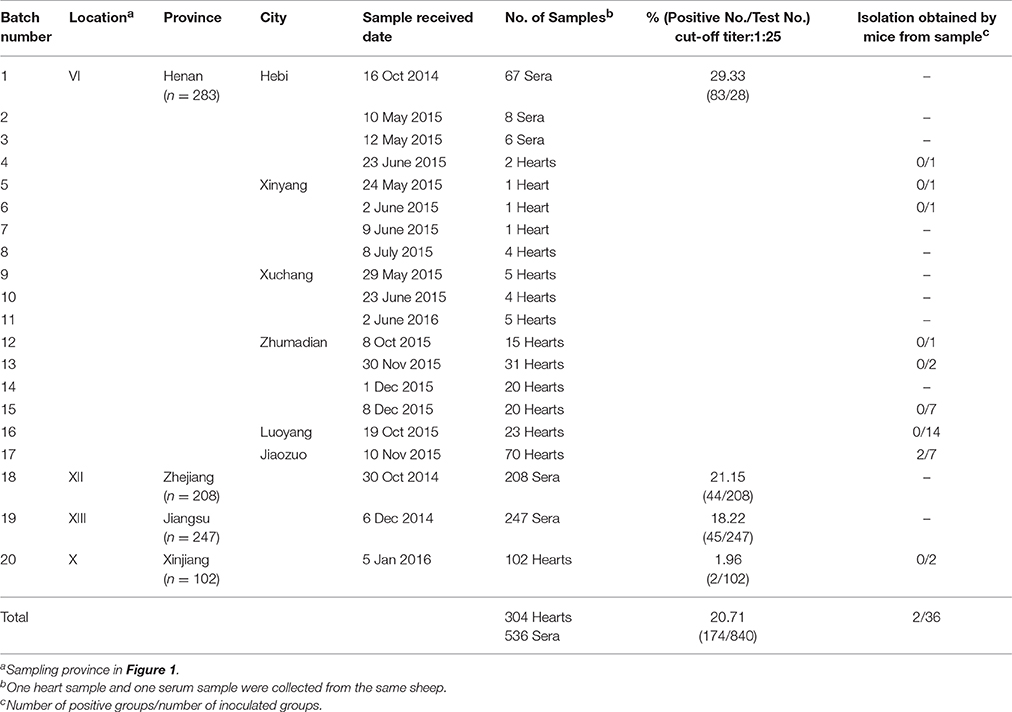
A total of 840 domestic sheep samples (304 fresh hearts and 536 sera) were collected from individual farms in Henan, Xinjiang, Zhejiang, and Jiangsu Province (Table 1, Figure 1). The climate of Henan Province (33°N, 113.30°E) is humid and subtropical, whereas Zhejiang Province (29.12°N, 120.30°E) is characterized by a subtropical monsoon climate. Jiangsu Province (Latitude 32.54°N, Longitude 119.48°E) is situated in the climatic transition belt between the warm-temperate and subtropical zones. Xinjiang (41°N, 85°E) has a semi-arid and desert climate. Domestic sheep in China are part of the farmers's household, and live with whatever other animals the farmers own, including cats. Unfrozen hearts and serum from sheep were collected between 2014 and 2016. In addition, juice was obtained from 304 hearts. Blood was obtained from jugular veins of 536 sheep. The juice or blood samples were allowed to clot, then centrifuged at 2000 × g for 10 min, after which the supernatant were separated and stored at −20°C until tested. Available background information is summarized in Table 1.
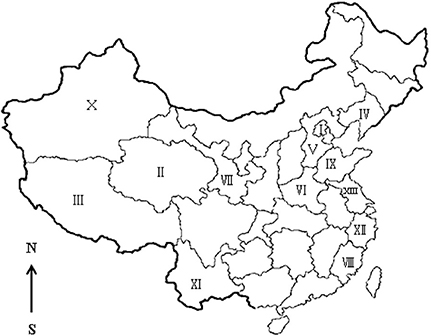
Figure 1. Seroepidemiology of Toxoplasma gondii in sheep in China. I, Beijing; II, Qinghai; III, Tibet; IV, Liaoning; V, Hebei; VI, Henan; VII, Gansu; VIII, Fujian; IX, Shandong; X, Xinjiang; XI, Yunnan; XII, Zhejiang; XIII, Jiangsu.
Serological Examination
Serum and heart juice samples from 840 sheep were tested for antibodies to T. gondii using the modified agglutination test (MAT) (Dubey and Desmonts, 1987). Whole formalin fixed RH T. gondii tachyzoites were kindly provided by Dr. J. P. Dubey (ARS, USDA). A titer of 1:25 was considered indicative of exposure to T. gondii. In addition, sera and heart juice were double diluted further with 0.01 M phosphate buffered saline (PBS), then tested for T. gondii parasites. Briefly, 100 mL 0.01 M PBS was amended with 8.5 g NaCl, 0.308 g NaH2PO4 (M.W. 120), and 1.08 g Na2HPO4 (M.W. 142), after which the pH was adjusted to 7.2 (Dubey, 2010).
Isolation of Viable T. gondii from Sheep Hearts by Bioassay in Mice
Specific-pathogen-free Kunming mice were supplied by the Zhengzhou University Laboratory Animal Center. Eight-week-old female Kunming mice were used in this study. Heart samples of T. gondii seropositive sheep (MAT, ≥25) were bioassayed in mice separately. The myocardium (50 g) was then homogenized, digested in pepsin (5.2 g pepsin, 10.0 g NaCl, 14 mL HCl, diluted to 1 l with deionized water, pH 1.1–1.2). The heart homogenate was subsequently incubated at 37°C in a shaking water bath for 60 min. After which, the sample was filtered by double gauze and centrifuged at 1200 × g for 10 min. The supernatant was then removed and the pellet was suspended in 0.01 M PBS (pH 7.2) and neutralized by mixing with 1.2% sodium bicarbonate. Following mixing, the sample was centrifuged at 1200 × g for 10 min, after which the supernatant was removed and 5–10 mL of saline containing 1000 units penicillin and 100 μg of streptomycin per ml was added. Myocardium digested liquid was then inoculated subcutaneously into four Kunming mice (1 mL per mouse) that had been maintained on drinking water supplemented with dexamethasone phosphate (DXM, 10 μg/ml) for 3 days before inoculation (Dubey, 2010). DXM treated mice were utilized as a control group, while DXM untreated mice were utilized as a blank group. Lung or brain impression smears of dead mice were examined for T. gondii tachyzoites or cysts. Survivors were bled on day 60 post-inoculation (DPI) and 120 DPI. 1:25 and 1:200 dilutions of sera from each mouse were tested for T. gondii antibodies with the MAT. Mice were killed 120 DPI and their brains were examined for tissue cysts after a squash preparation. All brains of survivors were homogenized and sub-passaged into new groups of mice subcutaneously.
In vitro Cultivation and Genotyping
Brain homogenates of T. gondii positive mice were seeded into vero cell culture flasks as previously described (Dubey, 2010). The number of cysts in the brains of mice were counted microscopically using the method reported by Dubey et al. (2012). Briefly, whole mouse brain was homogenized with 1 mL of saline (0.85% NaCl), tissue cysts were counted microscopically in 50 μl of the homogenate, and the count multiplied by 20 was the number of tissue cysts per brain. The time required for tachyzoites grow up in cell culture was recorded. DNA was extracted from cell culture derived tachyzoites using a commercial DNA extraction kit (Tiangen Biotec Company, DP304, China). The multiplex PCR of the T. gondii isolates was performed using 10 PCR-RFLP genetic markers, SAG1, SAG2 (5′–3′ SAG2, alt.SAG2), SAG3, BTUB, GRA6, c22-8, c29-2, L358, PK1, and Apico as previously described by Su et al. (2010). Reference T. gondii DNA was included in all batches.
Evaluation the Virulence of T. gondii Tachyzoites Isolated from Sheep by Mice
Fresh tachyzoites were collected from cell culture, counted in a disposable hemocytometer and diluted 10-fold from 10−1 to 10−6 to reach an end-point of <1 tachyzoite. Next, <1, 100, 101, 102, 103, or 104 tachyzoites were inoculated intraperitoneally into five Kunming mice for each dilution. Clinical symptoms, illness and death of mice were observed and recorded every day. Lung or mesenteric lymph node impression smears of dead mice were examined for T. gondii tachyzoites. At 30 DPI, sera from mice were analyzed for T. gondii antibody. The virulence was evaluated based on the percentage of dead mice among T. gondii positive mice.
Ethics Approval and Consent to Participate
This study was carried out in accordance with the recommendations of the institutional animal use committee of the Henan Agricultural University (China). The protocol was approved by the Beijing Association for Science and Technology (approval SYXK [Beijing] 2007-0023).
Statistical Analysis
Statistical analysis was performed using the Graph Pad Prism 4.0 software (Graph Pad Software Inc., San Diego, CA, USA). Data were analyzed by the chi-squared test or Fisher's exact test. A P < 0.05 was considered statistically significant.
Results
Seroepidemiology of T. gondii in Sheep
Antibodies to T. gondii were found in 174 of 840 (20.71%) sheep with titers of 1:25 in 48, 1:50 in 2, 1:100 in 24, and 1:200 or above in 100. Seropositivity rates varied with respect to source of sheep. The difference in seroprevalence of T. gondii in sheep from Henan Province (29.33%), Zhejiang Province (21.15%) and Jiangsu Province (18.22%) was not significant. However, the prevalence was higher in all of these regions than in Xinjiang (1.96%) (P < 0.05) (Table 1).
Detailed information was only available for 522 of the 840 samples. Female sheep (18.64%, 74/397) shows a tendency to be more susceptible to T. gondii (odds ratio = 1.278) than male sheep (15.20%, 19/125), but this difference was not significant. The seroprevalence of T. gondii in aborting sheep (41.30%, 38/92) was higher than that in non-aborting sheep (11.80%, 36/305), with an odds ratio of 5.258 (95% CI, 3.059–9.038), which was statistically significant (P < 0.0001). In this study, 16.22% (6/37) of sheep <1 year old were seropositive for T. gondii, while17.93% (87/485) of those >1 year old were seropositive. The risk of acquiring T. gondii infection in adult sheep (17.93%) tended to be higher when compared to that in lambs (16.22%), with an odds ratio of 1.129 (95% CI, 0.4571–2.7910); however, this difference was not statistically significant (P > 0.05).
Isolation and Virulence of T. gondii from Sheep
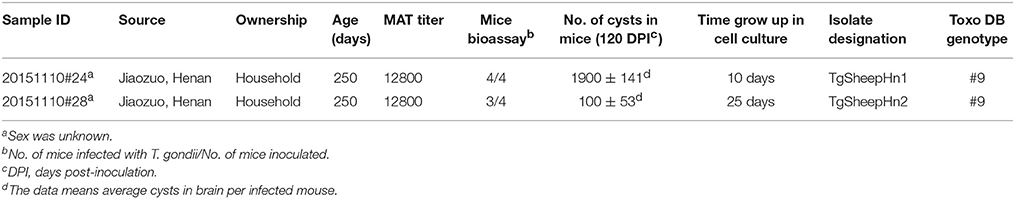
A total of 36 T. gondii seropositive sheep heart homogenates were bioassayed in four Kunming mice individually. T. gondii antibodies were only detected in 10 groups of mice at 60 DPI. Specifically, one mouse was positive in eight groups, three mice were positive in one group, and four mice were positive in one group. However, T. gondii antibodies were only detected in mice from the last two groups at 120 DPI, while the other eight groups were negative. Viable T. gondii were isolated from the two positive groups by Kunming mice (Table 1). All mice remained asymptomatic. T. gondii tissue cysts from the brain were detected in these mice when killed at 120 DPI. The average number of brain cysts in T. gondii infected mice was 1900 ± 141 from sheep heart sample 20151110#24, while 100 ± 53 cysts were observed in heart sample 20151110#28. The brain homogenates of T. gondii positive mice were sub-inoculated into mice, and seeded onto cell cultures for propagation of tachyzoites. Two isolates (TgSpHn1, TgSpHn2) were successfully propagated in cell culture and mice. Genetic typing of the isolates from sheep hearts revealed that they were all ToxoDB genotype #9 (Chinese 1) (Table 2).
Mice showed 100% mortality after inoculation with 100 T. gondii tachyzoites of TgSheepHn1. For TgSheepHn2, the 100% mortality dose was 1000 tachyzoites. However, loading with the highest level of 104 tachyzoites per mouse induced only 80% (TgSheepHn1) and 60% mortality (TgSheepHn2). The survival time post-inoculation with 104, 103, 102, 101, and 100 TgSheepHn1 parasites was 8, 9.5 days, 259 h, 20.5, and 17.5 days, while it was 8, 22 days, 343 h, above 30 days and above 30 days for TgSheepHn2, respectively (Table 3). The survival time post-inoculation with T. gondii tachyzoites differed between the two isolates from sheep.
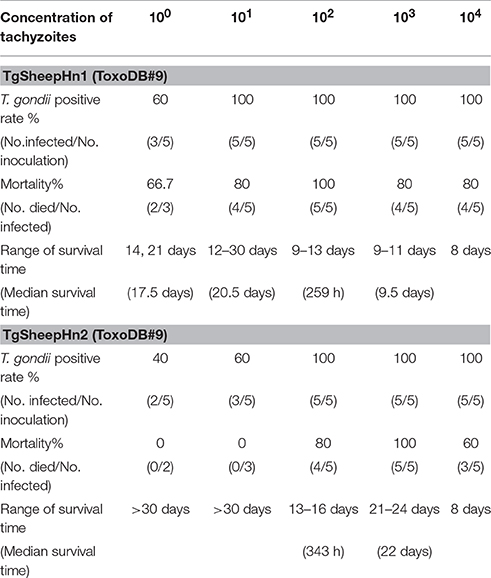
Table 3. Pathogenicity of the two isolated T. gondii tachyzoite strains with gradient dilution dosage on Kunming mice inoculated by intraperitoneal injection (30 DPI).
Discussion
In this study, the prevalence of antibodies and titers to T. gondii in sheep from Henan, Jiangsu, and Zhejiang province was higher than in sheep from Xinjiang (Table 1). The climate of Henan, Zhejiang, and Jiangsu is subtropical, whereas that of Xinjiang is semi-arid and desert. The arid climate of Xinjiang may contribute to the low prevalence of toxoplasmosis in the region. These results are consistent with those of other reports (Table 4, Figure 1). We have summarized available reports on sheep toxoplasmosis in Table 4. The prevalence of antibodies and titers to T. gondii was 2–39% in sheep from different parts of China (Table 4), indicating widespread environmental contamination with T. gondii oocysts. These findings are in accordance with those of our previous investigation of T. gondii in free-range chickens (Feng et al., 2016).
Oral ingestion of T. gondii oocysts is the main source of infection for sheep, and poses a risk for exogenous transplacental transmission in pregnant sheep (Innes et al., 2009). Moreover, a previous study confirmed that reactivation of T. gondii cysts in chronically infected sheep serves as another important risk for endogenous transplacental transmission in sheep during pregnancy (Williams et al., 2005; Hide, 2016). Additionally, mutton containing T. gondii tissue cysts poses a threat to human health. Mutton is the main ingredient of hot pots, instant-boiled mutton, dumpling and kebabs in China, however, these cooking methods are often not sufficient to eradicate T. gondii. Moreover, the high prevalence of T. gondii in aborting sheep observed in the present study indicate that T. gondii is a risk factor for abortion. Increasing rates of prevalence of T. gondii antibodies in older sheep indicated post-natal exposure of T. gondii infection, which agreed with the results of previous studies (Dubey, 2010).
T. gondii antibodies were detected in mice of 10 groups at 60 DPI, but were no longer present in eight groups at 120 DPI. Moreover, mice in eight groups were all negative for T. gondii after sub-passage in mice. After checking the T. gondii DNA of the eight injected sheep heart pepsin digested liquid by PCR, none of them were positive. We do not know the reason for this phenomenon, therefore, more studies investigating this interesting observation should be conducted.
The MAT we used has been extensively employed for the detection of T. gondii antibodies in many species, including humans and sheep (Dubey, 2010). Viable T. gondii was isolated from 100% (3/3) of sheep with MAT antibodies above 1:800 (Dubey et al., 2014a). The isolation of viable T. gondii is the gold standard for detecting live T. gondii parasites. However, the success of isolation depends on the tissues tested and the methods used. The density of T. gondii cysts in the heart has been shown to be higher than that in the brain or muscle, and the heart is the ideal choice for isolation of T. gondii (Dubey et al., 2015). Felid bioassay is the most sensitive method for identification of T. gondii (Dubey, 2010). For murine bioassay, the use of immunosuppressed mice facilitates early detection of T. gondii. Immunosuppression of mice by dexamethasone has been shown to be a useful method of isolating T. gondii (Qian et al., 2012). Moreover, viable ToxoDB#9 strains of T. gondii were isolated from cats, pigs, voles and humans from China in a previous study (Dubey et al., 2007; Chen et al., 2011; Wang et al., 2013a,b). Additionally, DNA from Hipposideros larvatus, sika deer, goat, Cebus apella and masked palm civets was genotyped and identified as ToxoDB#9 (Jiang et al., 2014; Chen et al., 2015; Li et al., 2015; Miao et al., 2015; Cong et al., 2016; Hou et al., 2016). When combined with our results, these findings indicate that ToxoDB#9 is predominant and widespread in animals from China, including sheep. These results indicate the limited genetic diversity of T. gondii from China.
The virulence of T. gondii was assessed based on their growth rates in cell culture and outbred mice after intraperitoneal injection of dilutions of tachyzoites. The pathogenicity, encystation and growth rate in cell culture of TgSheepHn1 were all stronger than those of TgSheepHn2. ToxoDB#9 isolates were previously reported to have different virulence and pathogenicity in mice (Cheng et al., 2015), which is in accordance with the virulence of the two isolates from sheep observed in the present study. Continued passages of a strain in mice or cell culture can alter the virulence (Dubey, 2010). In the present study, these factors were considered before making conclusions concerning the virulence of strains. Virulence and genome structure analyses of different ToxoDB#9 stains of T. gondii showed remarkable variation in ROP 16 and GRA15 (Li et al., 2014). The diversity of ToxoDB #9 stains of T. gondii may be connected to the invasion and immune response of this successful parasite.
The results of the present study showed that there is widespread exposure of sheep to T. gondii in China. Two viable ToxoDB#9 stains of T. gondii were isolated from sheep hearts and found to have different virulence. To the best of our knowledge, this is the first report of isolation of ToxoDB#9 from sheep in China. Because this organism remains present in the tissues of sheep and can therefore infected people via consumption of undercooked meat, sheep pose a risk of T. gondii infection and have the potential to impact public health.
Author Contributions
YY performed the data analysis and wrote the manuscript. YF performed the laboratory tests, data analysis. QY participated in the RFLP laboratory test. YW, YL helped in collecting samples. XZ, HL, and LZ helped in the writing of the manuscript.
Funding
This research project was financed by the Program for Science and Technology Innovation Talents in Universities of Henan Province (Grant No. 17HASTIT038) and China Postdoctoral Science Foundation (2016M600577).
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
The authors acknowledge J. P. Dubey (US Department of Agriculture, USA) for providing positive serum, antigen and DNA of T. gondii. We thank Oliver CHK (US Department of Agriculture, USA) for his linguistic assistance during the preparation of this manuscript.
Abbreviations
MAT, modified agglutination test; DPI, days post-inoculation; PCR, polymerase chain reaction; ROP, rhoptry protein; GRA, dense granule.
References
Bai, W. S., Chen, Y., Ba, L. T., and Mai, M. T. (2002). Seroprevalence of Toxoplasma gondii in domestic animal in Aksu (Sinkiang). Chin. J. Vet. Parasit. 10, 29–30. doi: 10.3969/j.issn.1674-6422.2002.02.011
Chen, C. Y. (2008). Prevalence of Toxoplasma gondii in domestic animal in Datong (Qinghai Province). Chin. Qinghai J. Anim. Vet. Sci. 38, 23.
Chen, R., Lin, X., Hu, L., Chen, X., Tang, Y., Zhang, J., et al. (2015). Genetic characterization of Toxoplasma gondii from zoo wildlife and pet birds in Fujian, China. Iran J. Parasitol. 10, 663–668.
Chen, Z. W., Gao, J. M., Huo, X. X., Wang, L., Yu, L., Halm-Lai, F., et al. (2011). Genotyping of Toxoplasma gondii isolates from cats in different geographic regions of China. Vet. Parasitol. 183, 166–170. doi: 10.1016/j.vetpar.2011.06.013
Cheng, W., Liu, F., Li, M., Hu, X., Chen, H., Pappoe, F., et al. (2015). Variation detection based on next-generation sequencing of type Chinese 1 strains of Toxoplasma gondii with different virulence from China. BMC Genomics 16:888. doi: 10.1186/s12864-015-2106-z
Cong, W., Qin, S. Y., Meng, Q. F., Zou, F. C., Qian, A. D., and Zhu, X. Q. (2016). Molecular detection and genetic characterization of Toxoplasma gondii infection in sika deer (Cervus nippon) in China. Infect. Genet. Evol. 39, 9–11. doi: 10.1016/j.meegid.2016.01.005
Cui, P., Fang, S. F., Wu, Z. Y., and Wu, B. Q. (2004). Epidemiological investigation of Toxoplasmosis in farm animals of Hebei Province. Chin. J. Vet. Sci. Technol. 34, 33–34.
Dong, Y. S., Luo, Z. Q., Zhang, G. C., and Liu, X. Q. (2011). Prevalence of Toxoplasmosis in sheep and cattle in Qinghai Province. Chin. J. Zoonoses. 27, 359–363. doi: 10.3969/j.issn.1002-2694.2011.04.023
Dubey, J. P. (2010). Toxoplasmosis of Animals and Humans, 2nd Edn. Boca Raton, FL: CRC Press; Taylor & Francis Group.
Dubey, J. P., Casey, S. J., Zajac, A. M., Wildeus, S. A., Lindsay, D. S., Verma, S. K., et al. (2014a). Isolation and genetic characterization of Toxoplasma gondii from alpaca (Vicugna pacos) and sheep (Ovis aries). J. Trop. Anim. Health 46, 1503–1507. doi: 10.1007/s11250-014-0652-z
Dubey, J. P., and Desmonts, G. (1987). Serological responses of equids fed Toxoplasma gondii oocysts. Equine Vet. J. 19, 337–339. doi: 10.1111/j.2042-3306.1987.tb01426.x
Dubey, J. P., Ferreira, L. R., Martins, J., and McLeod, R. (2012). Oral oocyst-induced mouse model of toxoplasmosis: effect of infection with Toxoplasma gondii strains of different genotypes, dose, and mouse strains (transgenic, out-bred, in-bred) on pathogenesis and mortality. Parasitology 139, 1–13. doi: 10.1017/S0031182011001673
Dubey, J. P., Lehmann, T., Lautner, F., Kwok, O. C., and Gamble, H. R. (2015). Toxoplasmosis in sentinel chickens (Gallus domesticus) in New England farms: seroconversion, distribution of tissue cysts in brain, heart, and skeletal muscle by bioassay in mice and cats. Vet. Parasitol. 214, 55–58. doi: 10.1016/j.vetpar.2015.09.004
Dubey, J. P., Verma, S. K., Ferreira, L. R., Oliveira, S., Cassinelli, A. B., Ying, Y., et al. (2014b). Detection and survival of Toxoplasma gondii in milk and cheese from experimentally infected goats. J. Food Prot. 77, 1747–1753. doi: 10.4315/0362-028X.JFP-14-167
Dubey, J. P., Zhu, X. Q., Sundar, N., Zhang, H., Kwok, O. C., and Su C, (2007). Genetic and biologic characterization of Toxoplasma gondii isolates of cats from China. Vet. Parasitol. 145, 352–356. doi: 10.1016/j.vetpar.2006.12.016
Feng, Y. J., Lu, Y. Y., Wang, Y. H., Liu, J., Zhang, L. X., and Yang, Y. R. (2016). Toxoplasma gondii and Neospora caninum in free-range chickens in Henan Province of China. Biomed. Res. Int. 2016:8290536. doi: 10.1155/2016/8290536
Fu, Y. J. (2003). Seroprevalence of Toxoplasma gondii in swine, cattle and sheep in Qinghai Province. Chin. J. Vet. Sci. Technol. 33, 68–69.
Hide, G. (2016). Role of vertical transmission of Toxoplasma gondii in prevalence of infection. Expert. Rev. Anti. Infect. Ther. 14, 335–344. doi: 10.1586/14787210.2016.1146131
Hou, G. Y., Zhao, J. M., Zhou, H. L., and Rong, G. (2016). Seroprevalence and genetic characterization of Toxoplasma gondii in masked palm civet (Paguma larvata) in Hainan province, tropical China. Acta Trop. 162, 103–106. doi: 10.1016/j.actatropica.2016.06.011
Innes, E. A., Bartley, P. M., Buxton, D., and Katzer, F. (2009). Ovine toxoplasmosis. Parasitology 136, 1887–1894. doi: 10.1017/S0031182009991636
Jiang, H. H., Qin, S. Y., Wang, W., He, B., Hu, T. S., Wu, J. M., et al. (2014). Prevalence and genetic characterization of Toxoplasma gondii infection in bats in southern China. Vet. Parasitol. 203, 318–321. doi: 10.1016/j.vetpar.2014.04.016
Li, M., Mo, X. W., Wang, L., Chen, H., Luo, Q. L., Wen, H. Q., et al. (2014). Phylogeny and virulence divergency analyses of Toxoplasma gondii isolates from China. Parasit. Vect. 7:133. doi: 10.1186/1756-3305-7-133
Li, W. C. (2012). Seroprevalence of Toxoplasma gondii, Chlamydia, Brucella in Tibetan sheep. Chin. J. Vet. Med. 48, 58–59.
Li, Y. N., Nie, X., Peng, Q. Y., Mu, X. Q., Zhang, M., Tian, M. Y., et al. (2015). Seroprevalence and genotype of Toxoplasma gondii in pigs, dogs and cats from Guizhou province, Southwest China. Parasit. Vect. 8, 214. doi: 10.1186/s13071-015-0809-2
Liu, Q., Ma, R., Zhao, Q., Shang, L., Cao, J., Wang, X., et al. (2010). Seroprevalence of Toxoplasma gondii infection in Tibetan sheep in Northwestern China. J. Parasitol. 96, 1222–1223. doi: 10.1645/GE-2601.1
Liu, Z. K., Li, J. Y., and Pan, H. (2015). Seroprevalence and risk factors of Toxoplasma gondii and Neospora caninum infections in small ruminants in China. Prev. Vet. Med. 118, 488–492. doi: 10.1016/j.prevetmed.2014.12.017
Luo, C. Q., Yuan, Y., Huang, J. M., and Huang, C. Q. (2013). Epidemic investigation of Toxoplasma gondii infection in cattle and sheep in part of Longyan city. Chin. Anim. Health 15, 13–16.
Miao, Q., Huang, S. Y., Qin, S. Y., Yu, X., Yang, Y., Yang, J. F., et al. (2015). Genetic characterization of Toxoplasma gondii in Yunnan black goats (Capra hircus) in southwest China by PCR-RFLP. Parasit. Vect. 8, 57. doi: 10.1186/s13071-015-0673-0
Qian, W., Wang, H., Su, C., Shan, D., Cui, X., Yang, N., et al. (2012). Isolation and characterization of Toxoplasma gondii strains from stray cats revealed a single genotype in Beijing, China. Vet. Parasitol. 187, 408–413. doi: 10.1016/j.vetpar.2012.01.026
Su, C., Shwab, E. K., Zhou, P., Zhu, X. Q., and Dubey, J. P. (2010). Moving towards an integrated approach to molecular detection and identification of Toxoplasma gondii. Parasitology 137, 1–11. doi: 10.1017/S0031182009991065
Wang, C. M., He, H. X., Qin, J. H., Yao, S. X., Wang, L. R., Liu, L. Y., et al. (2005). Investigation of toxoplasmosis in swine and sheep in Xinxiang city Henan Province. Chin. J. Parasit. Parasitic Dis. 23, 31.
Wang, D., Liu, Y., Jiang, T., Zhang, G., Yuan, G., He, J., et al. (2016). Seroprevalence and genotypes of Toxoplasma gondii isolated from pigs intended for human consumption in Liaoning province, northeastern China. Parasit. Vectors. 9, 248. doi: 10.1186/s13071-016-1525-2
Wang, L., Cheng, H. W., Huang, K. Q., Xu, Y. H., Li, Y. N., Du, J., et al. (2013b). Toxoplasma gondii prevalence in food animals and rodents in different regions of China: isolation, genotyping and mouse pathogenicity. Parasit. Vect. 6:273. doi: 10.1186/1756-3305-6-273
Wang, L., Chen, H., Liu, D., Huo, X., Gao, J., Song, X., et al. (2013a). Genotypes and mouse virulence of Toxoplasma gondii isolates from animals and humans in China. PLoS ONE 8:e53483. doi: 10.1371/journal.pone.0053483
Wang, Q. Q., Huang, Y. J., Zhang, W. Q., Sun, H., Chen, Y., Kuerbanni, T., et al. (2016). Serological survey of toxoplasmosis of sheep from Xinjiang nan jiang farm. Anim. Husb. Feed Sci. 37, 107–108.
Wang, W. L., Zhang, Y. X., Zhang, F. J., Zhao, S. S., and Chen, Y. X. (2006). Prevalence of Toxoplasma gondii in sheep in Beijing. Chin. J. Vet. Med. 42, 29–30. doi: 10.3969/j.issn.0529-6005.2006.09.016
Wang, X. Y. (2007). Seroprevalence of Toxoplasma gondii in sheep in Haiyan (Qinghai Province). Chin. Qinghai J. Anim. Vet. Sci. 37, 29.
Williams, R. H., Morley, E. K., Hughes, J. M., Duncanson, P., Terry, R. S., Smith, J. E., et al. (2005). High levels of congenital transmission of Toxoplasma gondii in longitudinal and cross-sectional studies on sheep farms provides evidence of vertical transmission in ovine hosts. Parasitology 130, 301–307. doi: 10.1017/s0031182004006614
Wu, S. M., Danba, C., Huang, S. Y., Zhang, D. L., Chen, J., Gong, G., et al. (2011). Seroprevalence of Toxoplasma gondii infection in Tibetan sheep in Tibet, China. J. Parasitol. 97, 1188–1189. doi: 10.1645/GE-2912.1
Yang, N., Li, H., He, J., Mu, M., and Yang, S. (2013). Seroprevalence of Toxoplasma gondii infection in domestic sheep in Liaoning Province, Northeastern China. J. Parasitol. 99, 174–175. doi: 10.1645/GE-3201.1
Yang, Y., Ying, Y., Verma, S. K., Cassinelli, A. B., Kwok, O. C., Liang, H., et al. (2015). Isolation and genetic characterization of viable Toxoplasma gondii from tissues and feces of cats from the central region of China. Vet. Parasitol. 211, 283–288. doi: 10.1016/j.vetpar.2015.05.006
Ye, Y. M., Wei, D. Q., and Tu, Y. F. (2002). Seroepidemiological survey of toxoplasmosis in animals from Yunan Province. Chin. J. Parasit. Parasitic Dis. 20, 255.
Yin, M. Y., Wang, J. L., Huang, S. Y., Qin, S. Y., Zhou, D. H., Liu, G. X., et al. (2015). Seroprevalence and risk factors of Toxoplasma gondii in Tibetan Sheep in Gansu Province, Northwestern China. BMC Vet. Res. 11:358. doi: 10.1186/s12917-015-0358-0
Yuan, W. Y., Ma, K., and Liu, C. Y. (2004). Seroepidemiological survey of toxoplasmosis in animals from Heibei Province. J. Med. Pest Control. 20, 80–82. doi: 10.3969/j.issn.1003-6245.2004.02.011
Yuan, Y. H., and Ma, L. Q. (2007). Seroprevalence of Toxoplasma gondii in small tail han sheep in Qinghai Province. Anim. Husb. Vet. Med. 39, 75. doi: 10.3969/j.issn.0529-5130.2007.09.031
Zhang, N., Wang, S., Wang, D., Li, C., Zhang, Z., Yao, Z., et al. (2016). Seroprevalence of Toxoplasma gondii infection and risk factors in domestic sheep in Henan province, central China. Parasite 23, 53. doi: 10.1051/parasite/2016064
Zhang, X. Q., Li, W. C., and Niu, X. Y. (2009a). Seroprevalence of Toxoplasma gondii in Tibetan sheep in Tianjun Qinghai Province. Chin. Qinghai J. Anim. Vet. Sci. 39, 29. doi: 10.3969/j.issn.1003-7950.2009.01.014
Zhang, X. Q., Lu, Y., and Li, W. C. (2009b). Seroprevalence of Toxoplasma gondii in Tibetan sheep in Tianjun Qinghai Province. Chin. J. Anim. Health Inspection. 29, 44–45. doi: 10.3969/j.issn.1005-944X.2009.07.022
Zhang, Y.-f., Liu, F.-y., Xu, X.-p., He, L.-x., Li, A.-q., and Zhang, X.-s. (2009). Toxoplasmosis serological test at Northern regions in Xinjiang. Grass Feeding Livestock 4, 22–24.
Zhao, Y. Q., Zhen, T. M., Wang, J. X., Fu, B., and Han, G. D. (2001). Seroepidemiological survey of toxoplasmosis in animals from Shandong Province. J. Prev. Med. 17, 185. doi: 10.3969/j.issn.1006-4028.2001.03.041
Zhou, P., Zhang, H., Lin, R.-Q., Zhang, D.-L., Song, H.-Q., Su, C., et al. (2009). Genetic characterization of Toxoplasma gondii isolates from China. Parasitol. Int. 58, 193–195. doi: 10.1016/j.parint.2009.01.006
Keywords: Toxoplasma gondii, seroepidemiology, isolation, genotype, virulence, sheep, China
Citation: Yang Y, Feng Y, Yao Q, Wang Y, Lu Y, Liang H, Zhu X and Zhang L (2017) Seroprevalence, Isolation, Genotyping, and Pathogenicity of Toxoplasma gondii Strains from Sheep in China. Front. Microbiol. 8:136. doi: 10.3389/fmicb.2017.00136
Received: 12 September 2016; Accepted: 19 January 2017;
Published: 03 February 2017.
Edited by:
Paul J. Brindley, George Washington University, USAReviewed by:
Henrik Vedel Nielsen, Statens Serum Institut, DenmarkXiangrui Li, Nanjing Agricultural University, China
Copyright © 2017 Yang, Feng, Yao, Wang, Lu, Liang, Zhu and Zhang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: YuRong Yang, eWFuZ3l1NzcxMkBzaW5hLmNvbQ==
†These authors have contributed equally to this work.
 YuRong Yang
YuRong Yang YongJie Feng
YongJie Feng QiuXia Yao2
QiuXia Yao2