- 1Department of Genetic Medicine, Weill Cornell Medicine, New York, NY, United States
- 2Department of Microbiology and Immunology, Weill Cornell Medicine, New York, NY, United States
- 3Laboratory of Genetic Medicine and Immunology, Weill Cornell Medicine-Qatar, Doha, Qatar
- 4Faculta di Medicina e Chirurgia, Universita Cattolica del Sacro Cuero, Rome, Italy
- 5Translational Genetics and Bioinformatics Section, Research Division, Sidra Medicine, Doha, Qatar
- 6Qatar Computing Research Institute, Hamad Bin Khalifa University, Doha, Qatar
- 7Department of Medical Oncology, National Center for Cancer Care and Research, Hamad Medical Corporation, Doha, Qatar
- 8Department of Population Sciences, City of Hope, Duarte, CA, United States
- 9Department of Laboratory Medicine at UCSF, San Francisco, CA, United States
- 10Helen Diller Family Comprehensive Cancer Center at UCSF, San Francisco, CA, United States
- 11Division of General Internal Medicine, Department of Medicine, Institute for Human Genetics at UCSF, San Francisco, CA, United States
- 12Service de Cancérologie Radiothérapie, CHU Farhat Hached, Sousse, Tunisia
- 13Laboratoire d'Immuno-Oncologie Moléculaire, Faculté de Médecine de Monastir, Université de Monastir, Monastir, Tunisia
- 14Tumor Biology Section, Research Division, Sidra Medicine, Doha, Qatar
Triple-negative breast cancer (TNBC) accounts for ~15–20% of breast cancer (BC) and has a higher rate of early relapse and mortality compared to other subtypes. The Chemokine (C-C motif) ligand 5 (CCL5) and its signaling pathway have been linked to TNBC. We aimed to investigate the susceptibility and prognostic implications of genetic variation in CCL5 signaling genes in TNBC in the present study. We characterized variants in CCL5 and that of six other CCL5 signaling genes (CCND1, ZMIZ1, CASP8, NOTCH2, MAP3K21, and HS6ST3) among 1,082 unrelated Tunisian subjects (544 BC patients, including 196 TNBC, and 538 healthy controls), assessed the association of the variants with BC-specific overall survival (OVS) and progression-free survival (PFS), and correlated CCL5 mRNA and serum levels with CCL5 genotypes. We found a highly significant association between the CCND1 rs614367-TT genotype (OR = 5.14; P = 0.004) and TNBC risk, and identified a significant association between the rs614367-T allele and decreased PFS in TNBC. A decreased risk of lymph node metastasis was associated with the MAP3K21 rs1294255-C allele, particularly in rs1294255-GC (OR = 0.47; P = 0.001). CCL5 variants (rs2107538 and rs2280789) were linked to CCL5 serum and mRNA levels. In the TCGA TNBC/Basal-like cohort the MAP3K21 rs1294255-G allele was associated with a decreased OVS. High expression of CCL5 in breast tumors was significantly associated with an increased OVS in all BC patients, but particularly in TNBC/Basal-like patients. In conclusion, genetic variation in CCL5 signaling genes may predict not only TNBC risk but also disease aggressiveness.
Introduction
Triple-negative breast cancer (TNBC) accounts for 15–20% of patients diagnosed breast tumors. TNBC tumors relapse early after standard chemotherapy treatments and often develop visceral metastases; it thus represents a subtype with the poorest prognosis among all breast cancers. Others and our group showed that Arab women might experience an early disease onset and a preponderance of aggressive breast cancer phenotypes (1). To some extent, high rates of TNBC may explain the poor prognosis of BC diagnosed at a younger age in Arab populations. Studies of Arab American women showed a similar BC pattern as Arab women living in the Arab world (2, 3), suggesting that a genetic component could be among the determining factors associated with TNBC onset.
Immunity plays a crucial role in TNBC biology (4). TNBC, and/or the basal-like subtype, is characterized by higher mutational burden and lymphocyte density as compared to other subtypes (and especially as compared with estrogen/progesterone receptor positive tumors) (5). In TNBC, the density of tumor infiltrating lymphocytes has been associated with favorable prognosis (6, 7). Recently, the immune checkpoint blockade Atezolizumab (monoclonal antibody against the protein programmed cell death-ligand 1) has been approved by the FDA for the treatment of metastatic TNBC based on the positive results of a phase III trial (8). Furthermore, post-treatment chemokine (C-C motif) ligand 5 (CCL5) increase has been associated with responsiveness to immunotherapy (Imiquimod) in breast cancer skin metastasis (9).
Our recent work demonstrated that CCL5, is strongly implicated in BC pathogenesis, particularly in TNBC progression (10). Hematopoietic CCL5 plays a pivotal role in recruiting immune cells into the tumors (11, 12). The circulating CCL5 level variations, observed in BC patients, could reflect a functional genetic variation in CCL5. Three Single Nucleotide Polymorphisms (SNPs) in CCL5, namely rs2107538, rs2280788, and rs2280789, are the most frequent SNPs associated with inflammatory diseases (13). Thus far, potential association between genetic variation in CCL5 and breast cancer has not been investigated. Integrative genomics analysis, combining information from GWAS studies on breast cancer involving over 400,000 cases and over 400,000 controls, was performed by Hicks et al. (14) to determine whether genes containing SNPs associated with an increased risk of developing breast cancer are associated with TNBC. Twelve out the 34 large-effect SNPs associated with TNBC are located within genes involved in the JNK, p38 MAPK, NF-κB, and cAMP/PKA signaling pathways, all of which regulate CCL5 levels in immune cells (15–17). These findings prompted us to hypothesize that through their effect on circulating CCL5 levels, functional polymorphism in both CCL5 and CCL5 signaling genes could be associated with TNBC.
In addition to the three SNPs of CCL5, we selected six SNPs from the twelve associated with TNBC and involved in the JNK, p38 MAPK, NF-κB, and cAMP/PKA signaling pathways. These SNPs are tagged to CCND1, ZMIZ1, CASP8, NOTCH2, MAP3K21, and HS6ST3. These genes encode proteins which regulate CCL5 expression or themselves are regulated by CCL5. CCND1 (cell-cycle regulator cyclin D1) encodes a cyclin protein that is critical for the cell cycle. The CCL5/STAT/CCND1 signaling pathway plays an important role in the crosstalk between epithelial cells and immune cells (18). ZMIZ1 (zinc finger MIZ-type containing 1) encodes a transcription factor which is a member of the Protein Inhibitor of Activated STAT (PIAS)-like family of coregulators. Zmiz1 is important for T-cell development and involved in NOTCH signaling (19), by which it could regulate CCL5 expression (20). CASP8 encodes the Caspase 8 protein which plays a major role in cell apoptosis and regulates NF-κB signaling (21). The functional association between CASP8 and CCL5 levels, secreted by immune cells, was shown in a knockout mice model (22). NOTCH2 encodes a member of NOTCH transmembrane receptor family. Dysregulation of NOTCH signaling was involved in several diseases, including BC (23). CCL5 expression is activated by NOTCH signaling in the tumor microenvironment, both in cancer cells (20) and tumor infiltrating lymphocytes (24, 25). MAP3K21 gene encodes a member of the MAPK pathway (also known as MLK4). MAP3K21 serves as an activator of NF-κB signaling (26); a major pathway for inducing CCL5 expression (27). HS6ST3 encodes a member of Heparan sulfate (HS) involved in several cellular and molecular processes, including cell proliferation and differentiation (28). By regulating IGFR1 expression, HS6ST3 could affect CCL5 expression (29, 30).
Based on the abundant evidence of the role of CCL5 in TNBC, we evaluated, in this study, the association of 9 SNPs, reflecting the genetic variation in CCL5 signaling genes, with TNBC susceptibility and prognosis.
Materials and Methods
Patients and Controls
A total of 1,082 unrelated subjects with high quality of genomic DNA, comprising 544 breast cancer patients, including 196 TNBC, and 538 healthy controls, were included in this study. Controls and patients were selected from the same ethnic group living in the middle coast of Tunisia. Only patient/control subjects who have ancestors up to three generations back who were natives of Tunisia and have lived for at least 10 years in Tunisia were included in the study. The participation rate for patients and controls exceeded 90 and 75%, respectively.
All patients included in this study had primary breast cancer, with unilateral breast tumors and no family history of the disease. The diagnosis of cancer was confirmed by histopathological analyses. The mean age of patients was 48.8 ± 10.9 years. After completion of treatment, patients had regular visits every 3–4 months in the first 2 years, every 6 months in the following 3 years and annually thereafter. At each visit patients were checked for symptoms and undergo a physical examination, mammography, chest X-ray and abdominal ultrasound were performed annually. During follow up, both locoregional and distant tumor recurrence were diagnosed as relapse based on clinical, radiological and histological findings. The median follow-up was 55 months (range, 1–175 months). Data on each patient were collected from the department of Radiation Oncology of Sousse Hospital (Sousse, Tunisia) between 2002 and 2016. A detailed description of the clinical/pathological characteristics is summarized in Table 1.
A total of 538 healthy women with a mean age of 48 ± 14 years were recruited as controls. They are typically either outpatients seen for vaccination or blood donation or friends or non-biological relatives of cancer patients with no evidence of any personal or family history of cancer (or other serious illness). Samples from healthy controls were collected consecutively between 2004 and 2016 and were age–group matched to the cases.
After a written consent was obtained, each participant (patient/control) had a blood sample taken and was interviewed using a questionnaire. The questionnaire has been tested, standardized and endorsed by the oncologists involved in the recruitment of patients with breast cancer. This questionnaire included a collection of epidemiological and lifestyle data and a “genetic” part examining consanguinity of parents and familial history with detailed data on all first and second-degree relatives with breast cancer (age, disease status). Detailed clinical-pathologic characteristics of the tumor are included as well. For all patients with breast cancer, early treatment response was assessed at 3- and 6-months post-treatment and graded as complete, or partial or poor/disease progression. The blood samples from patients were taken before initiation of any treatment.
Approval for the study was given by the Weill Cornell Medicine-Qatar Ethics Committee. Both patients and controls gave their written consent to participate in the study.
TCGA Affymetrix Human SNP 6.0 array genotyping calls of patient germline DNA were downloaded from NCI's Genomic Data Commons (https://gdc.cancer.gov). SNPs were filtered to eliminate >5% missing genotypes, and individuals with >5% missing genotypes were also removed. Individuals were clustered into ancestry groups using principal components analysis and PAM clustering, and samples which had heterozygosity values that were >3 standard deviations from the mean heterozygosity from the same ancestral populations were also eliminated. Only one sample per individual were included, with blood-derived samples and samples with higher call rates preferentially retained. SNPs that deviated from the expectation under Hardy-Weinberg equilibrium (p < 1 × 10−6) in analysis of the largest ancestral group were also dropped (though SNPs previously associated with any cancer as reported in the GWAS catalog with p < 5 × 10−8 were not eliminated since they may deviate from HWE in cancer cases). SNPs with minor allele frequency, MAF <0.5% were excluded, and duplicate SNPs with identical genomic first positions were removed. Palindromic SNPs were excluded prior to stranding. Finally, we checked for close relative pair-based analyses in PLINK1.9 and dropped one of each of these relative pairs. Variants were imputed through Minimac 3 selecting the panel of 1000 Genome Phase 3v5 using mixed population. The final dataset includes 163 Basal-like/TN breast cancer patients with available overall survival information, which were used in the present analysis. Annotated Germline TCGA Exome data (31) were used to retrieve CCR5 delta-32 information.
SNP Selection and Genotyping
We selected the three of the most studied SNPs of CCL5 located in the promoter region of the CCL5 gene. We also selected six SNPs from breast cancer GWAS studies (14). The six SNPs containing genes are all involved in signaling pathways that can regulate CCL5 levels. The SNP information is summarized in Table S1. Genotyping was performed using the TaqMan® SNP genotyping assays on Applied Biosystems QuantStudio 6 Flex Real-Time PCR System (Foster City, California, United States).
Enzyme-Linked Immunosorbent Assay (ELISA)
A specific ELISA (Human CCL5/RANTES Quantikine ELISA Kit, R&D Systems, Minneapolis, Minnesota, United States) was used to determine CCL5 levels in serum before cancer treatment with a calorimeter (CLARIOstar, BMG LABTECH, Ortenberg, Germany) at a wavelength of 405 nm. Three hundred-sixteen breast cancer patients whose serum were available, including 128 TNBC patients, were tested in this study.
In silico Analysis of CCL5 Gene Expression
The allele specific gene expression was assessed at Genotype-Tissue Expression Portal (GTExPortal) (https://gtexportal.org/home/) (32) and eQTL Catalog (https://eqtl.onderzoek.io) (33). The cancer survival analysis with TCGA data was performed using UCSC XENA (https://xena.ucsc.edu) and for that with the gene expression array data was performed using KM plotter (http://kmplot.com) (34). Correlation between CCL5 and markers of stemness (cancer stem cells and normal stem cells) and epithelial to mesenchymal transition (EMT) markers, was performed by using a normalized RNA matrix, described elsewhere (35). The selection of markers was based on current literature (36).
RNA Extraction and qPCR Quantification
RNA was extracted from frozen blood using PAXgene Blood RNA Kit (Qiagen, Germantown, Maryland, United States) using the following protocol (37). 300 μl of blood sample was dispensed into 830 μl PAXgene reagent. The mixture was incubated for overnight at room temperature and spun for 3 min at 5,000 g. After washing with 500 μl RNase-free water, the pellet was resuspended in 350 μl Buffer BR1 and incubated with 300 μl Buffer BR2 (binding buffer) and 40 μl proteinase K for 10 min at 55°C in a shaker-incubator at 1,400 rpm. The lysate was transferred into a PAXgene Shredder spin column placed in a 2 ml processing tube and centrifuged for 3 min at 18,000 g. The flow-through fraction was mixed with 350 μl ethanol, transferred to a PAXgene RNA spin column and centrifuges for 1 min at 18,000 g. After washing the column with Buffer BR3 (washing buffer I), samples were incubated with 80 μl of DNase I incubation mix (10 ul DNase I with 70 ul Buffer RDD) for 15 min at room temperature. PAXgene RNA spin columns were washed with Buffer BR3 and 2 times of Buffer BR4 (Washing buffer II). RNA was eluted with 40 μl Buffer BR5 (elution buffer).
Total RNA was reverse-transcribed into cDNA using SuperScript II Reverse Transcriptase (Invitrogen, Carlsbad, California, United States) following the manufacturer's protocol. CCL5 mRNA levels were measured by qPCR with a GoTaq® 2-Step RT-qPCR System (Promega, Madison, Wisconsin, United States) for SYBR Green-based detection. The qPCR primers for CCL5 are 5′-CCAGCAGTCGTCTTTGTCAC-3′ and 5′-CTCTGGGTTGGCACACACTT-3′. The HPRT1 gene was used as a reference and primer sequences are 5′-TGACACTGGCAAAACAATGCA-3′ and 5′-GGTCCTTTTCACCAGCAAGCT-3′. Two hundred and twenty-six mRNA samples were tested in this study.
Statistical Analyses
Hardy-Weinberg equilibrium in both patient and control groups were tested using the Chi-square test. According to the general genotype model, risk association between the genotypes and breast cancer susceptibility and tumor characteristics were estimated by a crude odds ratio (OR) with 95% confidence intervals (95% CI) using the unconditional logistic regression analysis with genotypes coded additively (as 0, 1, or 2 copies of one of the alleles). The association was then adjusted for age and menopausal status. A P-value of <0.05 was required for statistical significance.
The statistical analysis was performed using the Epi-Info statistical program (version 5.01; CDC, Atlanta, GA, USA). Breast cancer specific overall survival (OVS) was defined as the time from the date of diagnosis to death from breast cancer or to last contact. Progression-free survival (PFS) was defined as the time from the date of diagnosis to distant metastasis, relapse and death from BC or to last contact. The survival curves were plotted according to Kaplan and Meier. Differences between groups were calculated by the log rank test. For PFS survival of TNBC patients between rs614367 risk allele carriers and non-risk allele carriers, a multivariate Cox Regression analysis was performed for age, menopausal status, tumor stage, SBR grade, lymph node status and pathologic classification using SPSS statistic software (Version 25.0, IBM Corporation).
Results
Genetic Variation in CCL5 Signaling Genes as Risk Factors for Breast Cancer and for TNBC
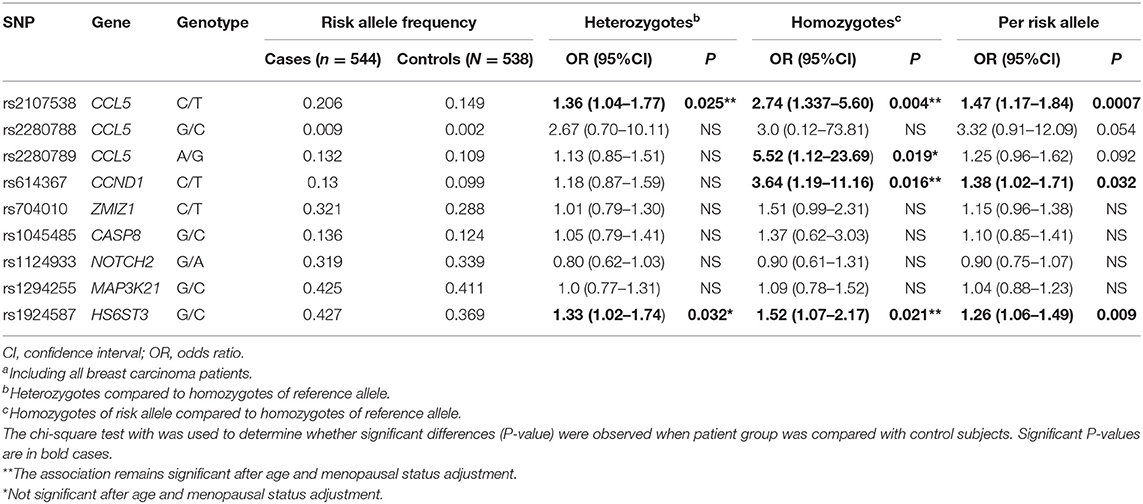
Genotype distribution of all 9 SNPs in controls did not deviate from Hardy-Weinberg equilibrium (Table S2, P > 0.05). The rs2280788-C allele is very rare in our study population compared to that found in the 1000 Genome and HapMap. The linkage disequilibrium (LD) between CCL5 rs2107538 and CCL5 rs2280789 was calculated using SHEsis (38). In controls, D′ is 0.763 and r2 is 0.407; in cases, D′ is 0.906 and r2 is 0.488. These results indicate a strong LD between these two loci. However, the allele frequencies between these two loci are quite different (Table 2). Therefore, we continued the analysis of these two loci separately.
A significant increase in the CCL5 rs2107538-T and HS6ST3 rs1924587-C allele frequencies was observed in the patient group compared to the controls (P = 0.0007 and 0.009, respectively, Table 2). The OR of breast cancer associated with the CCL5 rs2107538-T/T genotype was 2.74 (P = 0.004) and that of HS6ST3 rs1924587-C/C genotype was 1.52 (P = 0.021). The allelic frequency of the CCND1 rs614367-T was higher in patients with breast cancer compared to control subjects resulting in a significantly positive OR associated with this allele (OR = 1.38, P = 0.032). This association was found to be specifically confined to the CCND1 rs614367-T/T homozygous genotype (OR = 3.64, P = 0.016). In addition, a significant association was found between breast cancer and the CCL5 rs2280789-G/G homozygous genotype (OR = 5.52, P = 0.019). After adjustment for the potential confounders, including age and menopausal status, the association of CCL5 rs2107538-C/T genotype (P = 0.034), CCL5 rs2107538-T/T genotype (P = 0.011), CCND1 rs614367-T/T genotype (P = 0.021) and HS6ST3 rs1924587-C/C genotype (P = 0.019) with BC risk remained significant.
In order to identify a specific association between certain CCL5 signaling pathways and TNBC, we stratified the 544 patients with breast cancer according to the breast cancer subtype, namely TNBC and hormone receptor positive breast cancer (HRBC). A highly and significant increase in risk of TNBC compared to controls, but not of HRBC, was observed in subject carriers of the CCND1 rs614367-T/T homozygous genotype (OR = 5.14, P = 0.004, Table 3 and Table S3). Among the 9 SNPs, only the CCND1 rs614367-T/T genotype showed a strong and specific association with TNBC.
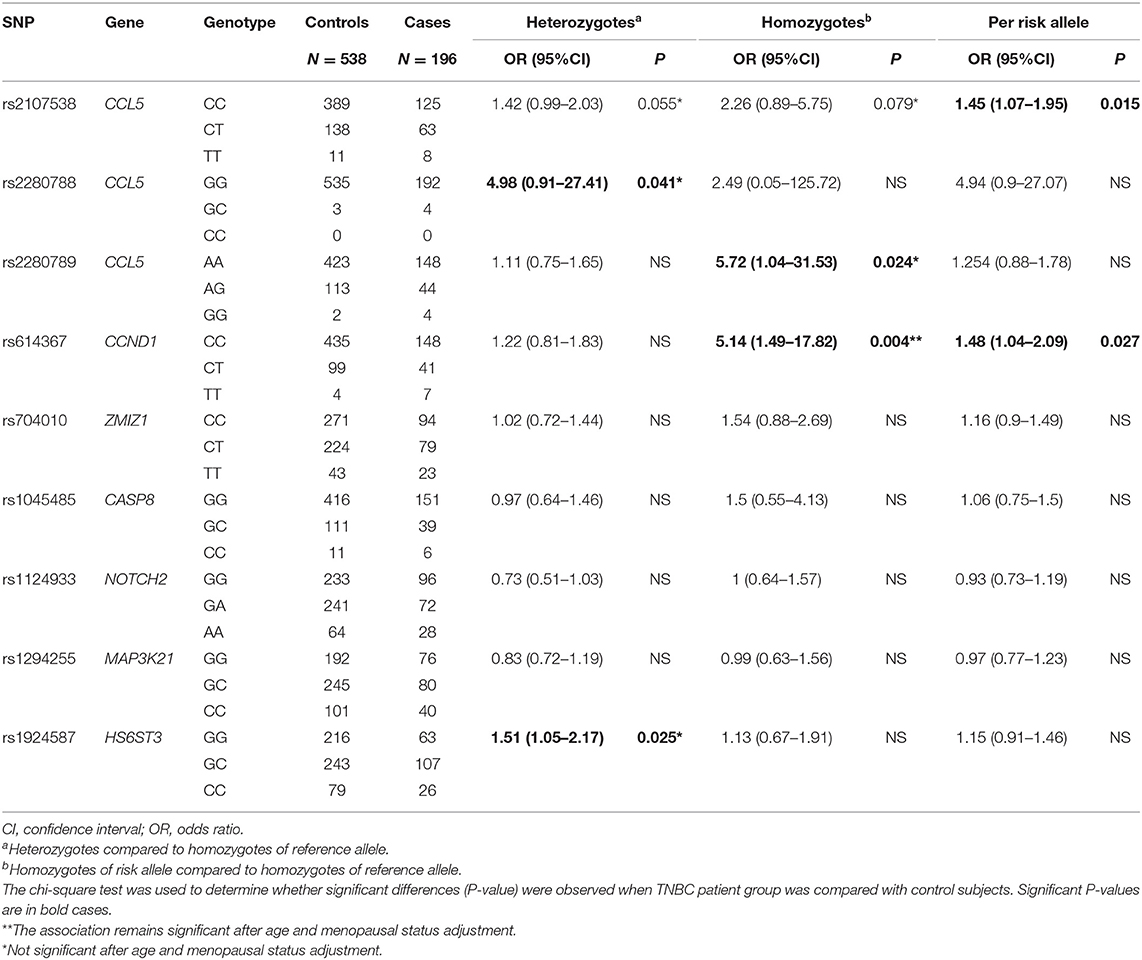
Table 3. Association of 9 SNPs of the CCL5 and CCL5 signaling pathway with triple negative breast cancer risk.
The CCL5 rs2107538-T allele was significantly associated with both TNBC and HRBC but the OR associated with HRBC was higher and more significant (OR = 1.45, P = 0.015 in TNBC compared to OR = 1.80, P = 0.0001 in HRBC). An increased risk of HRPC was found associated with the rs1924587-C allele (OR = 1.33, P = 0.006) and was confined to the homozygous C/C genotype (OR = 1.7, P = 0.012). A slightly significant association with TNBC was also observed in carriers of the heterozygous G/C genotype (OR = 1.51, P = 0.025).
Both CCL5 rs2280788-GC and CCL5 rs2280789-GG showed a slightly significant association with TNBC (OR = 4.98, P = 0.041; OR = 5.72, P = 0.024, respectively). Carriers of the rs2280789-G allele had a marginal increase in the risk for HRBC (OR = 1.36, P = 0.048).
After adjustment for the potential confounders, including age and menopausal status, the association of CCND1 rs614367-T/T genotype (P = 0.015) with TNBC risk and both of the association of CCL5 rs2107538-T/T genotype (P = 0.004) and HS6ST3 rs1924587-C/C genotype (P = 0.004) with HRBC risk remained significant, and the association of CCL5 rs2107538-C/T genotype (P = 0.049) with HRBC risk was suggestive.
Prognostic Significance of Genetic Variation in the CCL5 Signaling Pathway
Table S4 lists the clinical and pathological characteristics. The distribution of the clinical and pathological markers was in agreement with previously reported data, indicating that our cohort was representative of breast cancer patients. Progression-free survival (PFS) rates were estimated and compared by univariate analysis based on the clinical and pathological parameters. Significant associations were found for: clinical tumor size, lymph node status, and tumor grading with PFS. No significant differences were observed for age.
We investigated the potential association of 9 SNPs associated with the CCL5 signaling pathway and the clinical and pathological characteristics of breast cancer in our cohort. As shown in Tables S5, S6, the rs1294255-C allele was found more frequently in patients with no lymph-node metastasis; this resulted in a significantly negative OR associated with heterozygous rs1294255-G/C (OR = 0.47; P = 0.001 compared to GG genotype).
As shown in Figure 1A, the progression-free survival (PFS) was significantly shorter in the group of TNBC patients carrying the CCND1 rs614367-T allele. The estimated 5- and 10-years PFS rates in the groups of TNBC patients carrying or not the CCND1 rs614367-T allele were, respectively, 78 and 50% vs. 92 and 66% (log rank test, P = 0.014). Furthermore, a multivariate analysis was performed for age, menopausal status, tumor stage, SBR grade, lymph node status and pathologic classification. As shown in Figure S1, the survival rates showed a difference trend between patients with and without the CCND1 rs614367-T allele, but the difference did not reach statistical significance (P = 0.359).
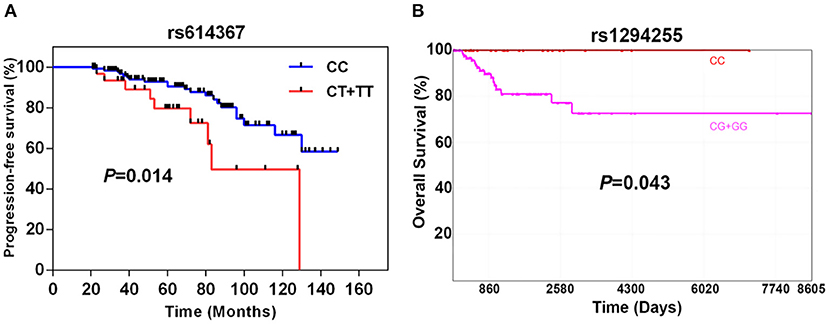
Figure 1. Breast cancer-specific progression-free survival of 196 TNBC patients according to the presence or absence of the CCND1 rs614367-T allele (A) and specific 10-years overall survival of 163 TNBC patients from the TCGA dataset according to the presence or absence of MAP3K21 rs1284255-G allele (B).
The limited number of breast cancer specific death events in our cohort hampered the search for a potential association between the 9 SNPs and OVS. The analysis of the TCGA dataset, which included 163 TNBC/Basal-like patients with available OS and genotype information, revealed a significant association between the MAP3K21 rs1294255-G allele and OVS. As shown in Figure 1B, the OVS was significantly shorter in the group of TNBC/Basal-like patients carrying the MAP3K21 rs1294255-G allele. The estimated 10-years OVS in the groups of TNBC patients with or without the MAP3K21 rs1294255-G allele was <80 and 100%, respectively (log rank test, P = 0.043). Similarly, CCL5 rs2107538-T allele showed a suggestive association with reduced OVS in TNBC patients (Figure S2). No statistically significant associations with OS were observed for the other seven polymorphisms.
Functional Implications for Breast Cancer and TNBC Risk Variants Associated With CCL5 Expression
To assess whether the 9 SNPs were associated with changes in the expression of CCL5 and/or their tagged genes, we performed eQTL (expression quantitative trait loci) analyses using GTEx and NESDA and NTR data. In a dose-dependent manner, the breast cancer risk alleles CCL5 rs2280789-G and CCL5 rs2107538-T were predictors of low levels of CCL5 expression in whole blood and lymphocytes of GTEx dataset (P = 7 × 10−6 and 6 × 10−8, respectively) (Figures S3A,B). The rs1294255-G allele was associated with low levels of MAP3K21 expression (Table S7). The association between CCL5 variants and CCL5 expression was confirmed by quantifying CCL5 mRNA in 226 blood samples from our study population. qPCR analysis suggested that CCL5 rs2280789-G and CCL5 rs2107538-T alleles associated with low levels of CCL5 mRNA (Figures 2A,B).
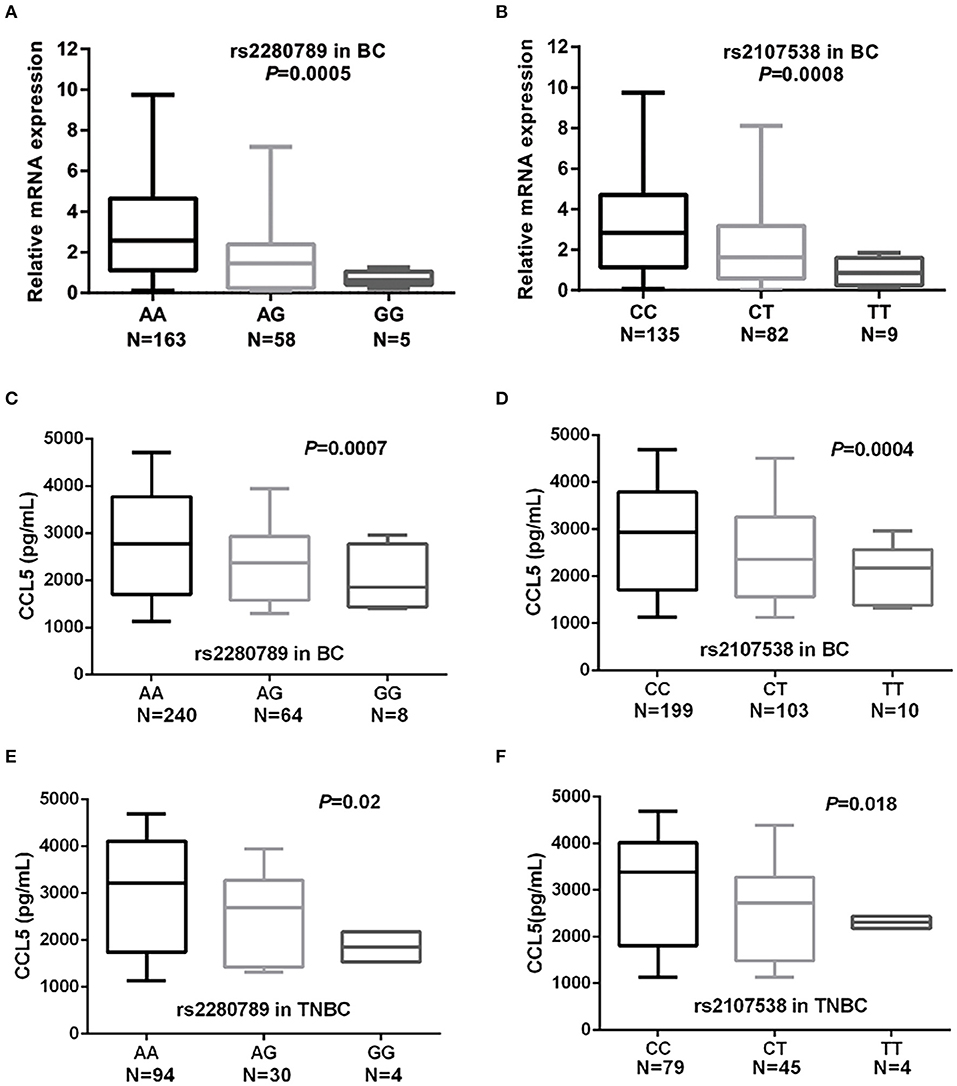
Figure 2. Quantification of CCL5 mRNA levels in blood cells and serum CCL5 in of breast cancer and TNBC patients. CCL5 mRNA levels in blood cells of breast cancer patients according to the genotypes of CCL5 rs2280789 (A) and CCL5 rs2107538 (B). CCL5 serum levels in 312 patients with breast cancer and in 128 patients with TNBC according to the genotypes of CCL5 rs2280789 (C,E) and CCL5 rs2107538 (D,F).
The CCL5 levels in serum before cancer treatment were correlated with patients' CCL5 genotypes. As shown in Figures 2C,D, the breast cancer risk alleles CCL5 rs2280789-G and CCL5 rs2107538-T were highly associated in a dose-dependent manner with low levels of CCL5 in sera of patients with breast cancer (all patients without subtype stratification). Similar findings were observed in sera of TNBC patients (Figures 2E,F). The size effect of CCL5 rs2107538 was higher in patients with TNBC than that observed in all patients with breast cancer. No significant difference in CCL5 levels was found between sera of control subjects and that of cancer patients (Figure S4).
Taken together, these findings suggest a potential impact of CCL5 expression levels on disease progression and clinical outcome. We addressed this hypothesis by assessing the correlation between CCL5 expression in breast tumors and disease survival using the TCGA dataset. As shown in Figure S5A, high levels of CCL5 expression predict a better survival in breast cancer patients (all subtypes included). This correlation did not reach statistical significance in cases with TNBC (likely duo to the limited sample size), but a trend toward higher survival rates was seen for TNBC tumors expressing high levels of CCL5 (Figure S5B). Further, we used the KM plotter platform (including a higher number of samples, i.e., the ones for which gene expression array were available either from TCGA or deposited in GEO) to evaluate an association between CCL5 expression and breast cancer and TNBC patients' survival. High levels of CCL5 expression correlated with a highly significant increase effect for TNBC patients' survival (Figure S5D). The estimated 5-years survival probabilities in the TNBC tumors expressing high or low levels of CCL5 were 0.88 and 0.63, respectively (log rank test, P = 0.00045). A similar observation was made when the analysis was performed on the unstratified breast cancer group (Figure S5C); here CCL5 expression had a less significant effect on the survival probabilities and the hazard ratio (HR) was higher compared to that observed in cases with TNBC (0.66 vs. 0.30). The estimated 5-years survival probabilities in the unstratified breast cancer tumors expressing high or low levels of CCL5 were 0.90 and 0.78, respectively (log rank test, P = 0.0093). Notably, the levels of CCL5 expression had no significant effect on non-TNBC patient survival (data not shown) in line with the predominant prognostic effect of tumor infiltrating lymphocytes observed prevalently in TNBC.
We next assessed in the TCGA cohort the potential association between CCL5 expression and markers of stemness and epithelial to mesenchymal transition (EMT) markers. As shown in Figure 3, CCL5 expression correlates at some extent with markers of stemness (cancer and normal stem cells) and with EMT markers in the analysis including all the intrinsic molecular breast cancer subtypes. However, such correlations were found particularly weak in the case of basal-like subtype, in which CCL5 was inversely correlated with mesenchymal markers. This might be explained by the pleiotropic role of CCL5, from one side being involved in TNBC pathogenesis, and from the other side mediating protective anti-tumor immunity via a positive immunologic loop resulting in lymphocyte recruitment through the interaction with its ligand CCR5 (co-expressed, together with CXCR3 by activated lymphocytes and NK cells). The inverse correlation between mesenchymal markers and CCL5 in the TNBC/basal-like subtype is also in line with the protective role of CCL5 observed in this group of tumors.
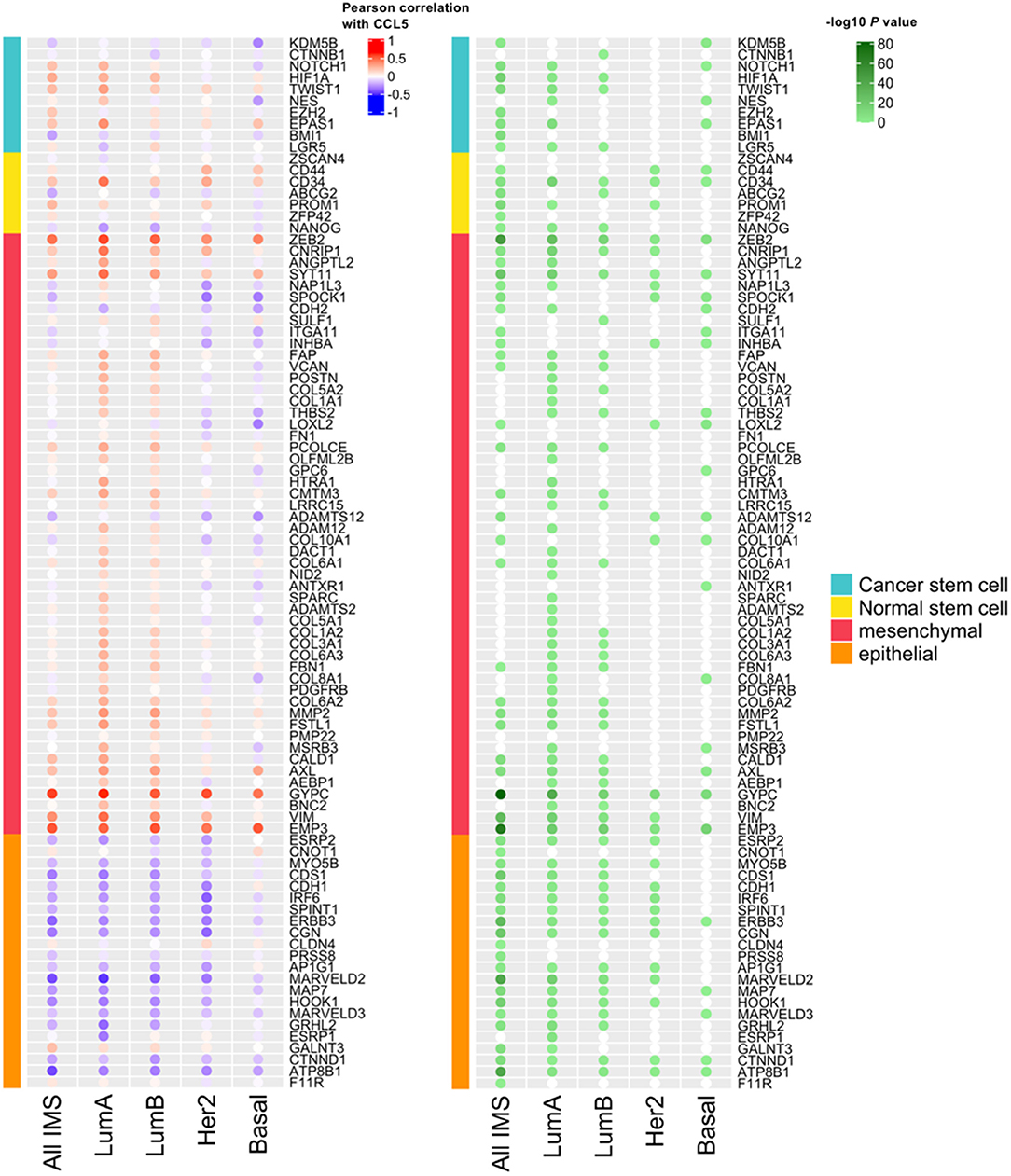
Figure 3. The correlation between CCL5 expression and the expression of markers of stemness and Epithelial-Mesenchymal Transition (EMT) markers. IMS, intrinsic molecular subtype; Lum, luminal.
We also assessed the survival impact of a functional deleterious germline polymorphisms of the CCL5 receptor CCR5 (Δ32 polymorphisms) in the TCGA cohort. The CCR5-Δ32 polymorphism consists of a 32-base deletion encoding a truncated protein, which is not expressed on the cell surface (11) and is strongly associated with HIV resistance (in the homozygous status) or disease progression (in the heterozygous status) (39). No differences or trends were observed in term of overall survival, either in the entire BC cohort or in the basal-like cohort (Figure S6), when patients were stratified according to the CCR5-Δ32 status.
Discussion
Given their crucial role in inflammatory diseases, chemokines were the subject of extensive study for several decades. Their role in recruiting immune cells to the tumor microenvironment made them an attractive potential target for cancer treatment. Among chemokines, CCL5 is relatively well-studied in cancers including breast cancer (40). However, knowledge of CCL5 implications with regard to cancer susceptibility remains elusive.
In the present study, we assessed the BC susceptibility of CCL5 polymorphisms, including rs2107538, rs2280788, and rs2280789. The BC susceptibility locus CCL5 rs2107538 in our cohort was significantly associated with the risk for both TNBC and HRBC; the other breast cancer susceptibility locus rs2280789 was associated with an increased risk for TNBC but only had a marginal effect on HRBC, while rs2280788 had a weak association with TNBC risk. We also assessed BC susceptibility of 6 SNPs in CCL5 signaling genes and found that CCND1 rs614367 was associated specifically with TNBC risk. HS6ST3 rs1924587 was preferentially associated with HRBC risk. MAP3K21 rs1294255 was associated with lymph-node invasion of breast tumors. For certain SNPs, we observed the association between BC and the allele risk in a dose-dependent manner. This is the case of rs2107538 and rs1924587. However, for other SNPs, the association was found confined either to the heterozygous or to the homozygous genotype. This could be linked to the low prevalence of the hetero/homozygous genotype in the study population. TNBC specific SNP rs614367 was associated with prognosis in a BC subgroup-TNBC but not in all breast cancer patients. We also showed the functional implication of CCL5 breast cancer susceptibility locus and correlated the risk alleles of CCL5 SNPs with low levels of serum CCL5. Furthermore, we performed in silico analysis and found that low CCL5 expression predicted a poor survival in BC and TNBC.
CCL5, which is originally known as RANTES (regulated on activation, normal T cell expressed and secreted), directs leukocytes to migrate into damaged or infected sites upon an inflammatory insult. In addition, CCL5 is essential to maintaining CD8 T cell function during chronic infection (41). It could be expected that high CCL5 levels at the tumor site would result in the recruitment of CD8+ T cells and sustain their cytotoxic capability against tumor cells. CCL5 expression is indeed associated with accumulation of CD8+ infiltrating T cells and associated with a favorable prognosis in colon cancer (42) and breast cancer (43). The risk allele of two SNPs in CCL5 gene corresponds with a lower CCL5 expression, which leads to a depletion of CD8+ infiltrating T cells, and, impairment of CD8+ T cell function and thus a higher risk of malignancy. Nevertheless, we did not observe any significant association between CCL5 polymorphism and survival or disease characteristics, except a suggestive effect of rs2107538-T allele on TNBC patient survival. This indicates that other type(s) of immune cells might play a dominant role in disease progression other than CD8+ T cells. Our in silico analyses, assessing the prognosis value of CCL5 expression, showed that low CCL5 expression predicted a poor survival in BC and TNBC. These findings are in agreement with certain previous reports (43, 44) but contradict others (13). These discrepancies could be due to several reasons including the size of the study population. Unlike most of the previous studies, in the current study we performed analyses on large public datasets (34). Furthermore, we assessed the CCL5 expression in tumor specimen harboring both tumor and stroma cells which provides a more accurate assessment of the CCL5 effects not only on tumor cells but also on tumor microenvironment and tumor-infiltrating immune cells. Conversely, several previous reports limited their assessment of the effects of CCL5 expression on tumor cell behavior which could lead to biased conclusions.
Our study is limited by the relatively few deaths in patients which limited the power for survival analyses. To address this limitation, we analyzed TCGA data which strengthened our findings and revealed a significant association between the MAP3K21 rs1294255-G allele and OVS in TNBC patients.
One of the goals of this study was to identify TNBC susceptibility SNPs given that immune pathways and activities were elevated in TNBC compared to non-TNBC. However, CCL5 polymorphisms were not preferentially associated with TNBC, indicating that CCL5 also plays an important role in non-TNBC. CCL5 has been implicated in luminal type breast cancer by modulating the phenotype of CD4+ T cells (45) and in ER-positive breast cancers by promoting macrophage influx (46). The clinically established BC Intrinsic subtype classifiers, PAM50 (47) and claudin-low predictor (48), do not contain CCL5 gene. All the above suggest a universal contribution of CCL5 in BC regardless of the subtypes. Tumor-derived CCL5 was detected in many clinical specimens and BC-derived cell lines, and were considered to promote BC aggressiveness (13). However, we and others showed that tumor-derived CCL5 expression did not significantly promote mammary carcinoma growth (10, 49), but the hematopoietic CCL5 played the major role in BC progression by regulating myeloid cells or macrophages in different subtype of BC (10, 50). Two recent studies suggested that intertumoral CCL5 level determined the recruitment of CD8+ T-cell in TNBC (43, 51). CCL5 might influence tumor growth by regulating different tumor-infiltrating immune cells in different subtypes of BC. Compared to CCL5 rs2107538, the association of CCL5 rs2280788 and CCL5 rs2280789 was not that significant. However, the OR of the latter two are often relatively high even if only a slight significance or a suggestive significance. Unlike common variants with low penetrance, these two variants seem to have a medium penetrance in breast cancer.
We showed that CCND1 rs614367 was associated with a strong and specific increased risk for TNBC, and also had prognostic value only for TNBC. CCND1 rs614367 is one of the strongest breast cancer risk loci identified by GWAS studies; however, its specific association with TNBC has not been identified. Replication of these findings in other populations will be of interest to determine whether the association of the CCND1 rs614367 with TNBC can be generalized. In GWAS studies, rs614367 was usually tagged to the CCND1 gene, which encodes cyclin D1, a central component in the cell cycle that could be regulated by estrogen. Although CCND1 is well-established as a cancer driver gene (52), it seems that CCND1 is an ER-positive breast cancer marker (53). FGF19 is one of its another nearby gene, and FGF19 and its receptor FGFR4 were strongly associated with the basal-like subtype of breast cancer (54). However, our in-silico analyses did not show any allele specific effect on these genes' expression. It will be of interest to further explore the functional implications of rs614367 like its potential long-range regulation or its association with non-coding RNA.
We also showed that HS6ST3 rs1924587 was preferentially associated with HRBC risk. HS6ST3 could promote breast cancer cell proliferation by upregulating IGF1R expression (29). It has been shown that IGF1R inhibits CCL5 expression and consequently reduces the chemotactic movement of CD8+ T lymphocytes (30). Taken together these findings suggest that through IGF1R, HS6ST3 could affect both CCL5 levels and tumor infiltrating CD8+ T lymphocytes behavior. This agrees with our finding showing that low levels of CCL5 expression associated with poor prognosis of BC. It is worth noting that IGF1R is abundant in HRBC but not in TNBC (55) which agrees with our current finding showing the preferential association between HS6ST3 rs1924587 and HRBC risk and could also explain the predominant role of CCL5 in HRBC.
Analysis of multiple datasets indicates that high expression of MAP3K21 is associated with a poor survival in breast cancer (56), and, the C allele of rs1294255 is associated with a low expression of MAP3K21 (Table S7). These findings are consistent with our analysis of the TCGA breast cancer dataset showing that rs1294255-G allele associated with poor prognosis. In the present study, we could not replicate the association, shown by GWAS, between rs1294255 and TNBC risk, but rather we observed that the rs1294255-C allele was found more frequently in patients with no lymph-node metastasis resulting in a significantly negative OR associated with the heterozygous rs1294255-CG.
Although the GWAS studies in Caucasians and the meta-analysis could lead to the discovery of SNPs associated with breast cancer, discovery studies and independent validation in different population are needed given the ethnic specificity and environmental exposure. For example, TNBC is over-expressed in African-American populations (57) as well as Arab populations. However, the correlated gene expression data and genotyping data are not available for these two populations.
In conclusion, the results of this study suggest that genetic variation in CCL5 and the CCL5 signaling pathway might have not only susceptibility but also prognostic implications for TNBC. Furthermore, this work adds to the evidence that CCL5 and its mediators play a critical role in the process of TNBC development and aggressiveness.
Data Availability Statement
The raw data supporting the conclusions of this manuscript will be made available by the authors, without undue reservation, to any qualified researcher.
Ethics Statement
The studies involving human participants were reviewed and approved by Weill Cornell Medicine-Qatar IRB. The patients/participants provided their written informed consent to participate in this study.
Author Contributions
SB, XM, DB, and LC contributed conception and design of the study. JS, AC, NB, YR, and SG performed the experiments. JS, YM, MS, RS, EZ, and JR analyzed the data. JS, MS, DB, and LC wrote the manuscript. All authors contributed to manuscript revision, read, and approved the submitted version.
Funding
This work was supported by Weill Cornell Medicine-Qatar and by grants from the Qatar National Research Fund (NPRP7-136-3-031 and NPRP10-0126-170262).
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
We thank Dr. Soha Roger Dargham for the help in statistical analyses. The publication of this article was funded by the Weill Cornell Medicine - Qatar Distributed eLibrary.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fonc.2019.01328/full#supplementary-material
References
1. Chouchane L, Boussen H, Sastry KS. Breast cancer in Arab populations: molecular characteristics and disease management implications. Lancet Oncol. (2013) 14:e417–24. doi: 10.1016/S1470-2045(13)70165-7
2. Nasseri K, Mills PK, Allan M. Cancer incidence in the Middle Eastern population of California, 1988–2004. Asian Pac J Cancer Prev. (2007) 8:405–11.
3. Hensley Alford S, Schwartz K, Soliman A, Johnson CC, Gruber SB, Merajver SD. Breast cancer characteristics at diagnosis and survival among Arab-American women compared to European- and African-American women. Breast Cancer Res Treat. (2009) 114:339–46. doi: 10.1007/s10549-008-9999-z
4. Disis ML, Stanton SE. Triple-negative breast cancer: immune modulation as the new treatment paradigm. Am Soc Clin Oncol Educ Book. (2015) 35:e25–30. doi: 10.14694/EdBook_AM.2015.35.e25
5. Hendrickx W, Simeone I, Anjum S, Mokrab Y, Bertucci F, Finetti P, et al. Identification of genetic determinants of breast cancer immune phenotypes by integrative genome-scale analysis. Oncoimmunology. (2017) 6:e1253654. doi: 10.1080/2162402X.2016.1253654
6. Salgado R, Denkert C, Demaria S, Sirtaine N, Klauschen F, Pruneri G, et al. The evaluation of tumor-infiltrating lymphocytes (TILs) in breast cancer: recommendations by an International TILs Working Group 2014. Ann Oncol. (2015) 26:259–71. doi: 10.1093/annonc/mdu450
7. Adams S, Gray RJ, Demaria S, Goldstein L, Perez EA, Shulman LN, et al. Prognostic value of tumor-infiltrating lymphocytes in triple-negative breast cancers from two phase III randomized adjuvant breast cancer trials: ECOG 2197 and ECOG 1199. J Clin Oncol. (2014) 32:2959–66. doi: 10.1200/JCO.2013.55.0491
8. Schmid P, Adams S, Rugo HS, Schneeweiss A, Barrios CH, Iwata H, et al. Atezolizumab and nab-paclitaxel in advanced triple-negative breast cancer. N Engl J Med. (2018) 379:2108–21. doi: 10.1056/NEJMoa1809615
9. Rozenblit M, Hendrickx W, Heguy A, Chiriboga L, Loomis C, Ray K, et al. Transcriptomic profiles conducive to immune-mediated tumor rejection in human breast cancer skin metastases treated with Imiquimod. Sci Rep. (2019) 9:8572. doi: 10.1038/s41598-019-42784-9
10. Zhang Y, Lv D, Kim HJ, Kurt RA, Bu W, Li Y, et al. A novel role of hematopoietic CCL5 in promoting triple-negative mammary tumor progression by regulating generation of myeloid-derived suppressor cells. Cell Res. (2013) 23:394–408. doi: 10.1038/cr.2012.178
11. Bedognetti D, Spivey TL, Zhao Y, Uccellini L, Tomei S, Dudley ME, et al. CXCR3/CCR5 pathways in metastatic melanoma patients treated with adoptive therapy and interleukin-2. Br J Cancer. (2013) 109:2412–23. doi: 10.1038/bjc.2013.557
12. Wang E, Bedognetti D, Marincola FM. Prediction of response to anticancer immunotherapy using gene signatures. J Clin Oncol. (2013) 31:2369–71. doi: 10.1200/JCO.2013.49.2157
13. Lv D, Zhang Y, Kim HJ, Zhang L, Ma X. CCL5 as a potential immunotherapeutic target in triple-negative breast cancer. Cell Mol Immunol. (2013) 10:303–10. doi: 10.1038/cmi.2012.69
14. Hicks C, Kumar R, Pannuti A, Backus K, Brown A, Monico J, et al. An integrative genomics approach for associating GWAS information with triple-negative breast cancer. Cancer Inform. (2013) 12:1–20. doi: 10.4137/CIN.S10413
15. Kumar D, Hosse J, von Toerne C, Noessner E, Nelson PJ. JNK MAPK pathway regulates constitutive transcription of CCL5 by human NK cells through SP1. J Immunol. (2009) 182:1011–120. doi: 10.4049/jimmunol.182.2.1011
16. Pocock J, Gómez-Guerrero C, Harendza S, Ayoub M, Hernández-Vargas P, Zahner G, et al. Differential activation of NF-kappa B, AP-1, and C/EBP in endotoxin-tolerant rats: mechanisms for in vivo regulation of glomerular RANTES/CCL5 expression. J Immunol. (2003) 170:6280–91. doi: 10.4049/jimmunol.170.12.6280
17. Qian X, Zhang J, Liu J. Tumor-secreted PGE2 inhibits CCL5 production in activated macrophages through cAMP/PKA signaling pathway. J Biol Chem. (2011) 286:2111–20. doi: 10.1074/jbc.M110.154971
18. Yang Y, Hu S, Liu J, Cui Y, Fan Y, Lv T, et al. CD8+ T cells promote proliferation of benign prostatic hyperplasia epithelial cells under low androgen level via modulation of CCL5/STAT5/CCND1 signaling pathway. Sci Rep. (2017) 7:42893. doi: 10.1038/srep42893
19. Pinnell N, Yan R, Cho HJ, Keeley T, Murai MJ, Liu Y, et al. The PIAS-like coactivator Zmiz1 Is a direct and selective cofactor of Notch1 in T cell development and leukemia. Immunity. (2015) 43:870–83. doi: 10.1016/j.immuni.2015.10.007
20. Colombo M, Mirandola L, Chiriva-Internati M, Basile A, Locati M, Lesma E, et al. Cancer cells exploit notch signaling to redefine a supportive cytokine milieu. Front Immunol. (2018) 9:1823. doi: 10.3389/fimmu.2018.01823
21. Duckett CS. Apoptosis and NF-kappa B: the FADD connection. J Clin Invest. (2002) 109:579–80. doi: 10.1172/JCI0215197
22. Alvarez-Diaz S, Dillon CP, Lalaoui N, Tanzer MC, Rodriguez DA, Lin A, et al. The pseudokinase MLKL and the kinase RIPK3 have distinct roles in autoimmune disease caused by loss of death-receptor-induced apoptosis. Immunity. (2016) 45:513–26. doi: 10.1016/j.immuni.2016.07.016
23. Mollen EWJ, Ient J, Tjan-Heijnen VCG, Boersma LJ, Miele L, Smidt ML, et al. Moving breast cancer therapy up a notch. Front Oncol. (2018) 8:518. doi: 10.3389/fonc.2018.00518
24. Liubomirski Y, Lerrer S, Meshel T, Morein D, Rubinstein-Achiasaf L, Sprinzak D, et al. Notch-mediated tumor-stroma-inflammation networks promote invasive properties and CXCL8 expression in triple-negative breast cancer. Front Immunol. (2019) 10:804. doi: 10.3389/fimmu.2019.00804
25. Choi K, Ahn YH, Gibbons DL, Tran HT, Creighton CJ, Girard L, et al. Distinct biological roles for the notch ligands Jagged-1 and Jagged-2. J Biol Chem. (2009) 284:17766–74. doi: 10.1074/jbc.M109.003111
26. Kim SH, Ezhilarasan R, Phillips E, Gallego-Perez D, Sparks A, Taylor D, et al. Serine/threonine kinase MLK4 determines mesenchymal identity in glioma stem cells in an NF-κB-dependent manner. Cancer Cell. (2016) 29:201–13. doi: 10.1016/j.ccell.2016.01.005
27. Liu J, Guan X, Ma X. Interferon regulatory factor 1 is an essential and direct transcriptional activator for interferon {gamma}-induced RANTES/CCl5 expression in macrophages. J Biol Chem. (2005) 280:24347–55. doi: 10.1074/jbc.M500973200
28. Habuchi H, Tanaka M, Habuchi O, Yoshida K, Suzuki H, Ban K, et al. The occurrence of three isoforms of heparan sulfate 6-O-sulfotransferase having different specificities for hexuronic acid adjacent to the targeted N-sulfoglucosamine. J Biol Chem. (2000) 275:2859–68. doi: 10.1074/jbc.275.4.2859
29. Iravani O, Bay BH, Yip GW. Silencing HS6ST3 inhibits growth and progression of breast cancer cells through suppressing IGF1R and inducing XAF1. Exp Cell Res. (2017) 350:380–9. doi: 10.1016/j.yexcr.2016.12.019
30. Wang X, Liu S, Cao L, Zhang T, Yue D, Wang L, et al. miR-29a-3p suppresses cell proliferation and migration by downregulating IGF1R in hepatocellular carcinoma. Oncotarget. (2017) 8:86592–603. doi: 10.18632/oncotarget.21246
31. Huang KL, Mashl RJ, Wu Y, Ritter DI, Wang J, Oh C, et al. Pathogenic germline variants in 10,389 adult cancers. Cell. (2018) 173:355–70.e14. doi: 10.1016/j.cell.2018.03.039
32. GTEx Consortium. The genotype-tissue expression (GTEx) project. Nat Genet. (2013) 45:580–5. doi: 10.1038/ng.2653
33. Jansen R, Hottenga JJ, Nivard MG, Abdellaoui A, Laport B, de Geus EJ, et al. Conditional eQTL analysis reveals allelic heterogeneity of gene expression. Hum Mol Genet. (2017) 26:1444–51. doi: 10.1093/hmg/ddx043
34. Györffy B, Lanczky A, Eklund AC, Denkert C, Budczies J, Li Q, et al. An online survival analysis tool to rapidly assess the effect of 22,277 genes on breast cancer prognosis using microarray data of 1,809 patients. Breast Cancer Res Treat. (2010) 123:725–31. doi: 10.1007/s10549-009-0674-9
35. Roelands J, Hendrickx W, Kuppen P, Mall R, Zoppoli G, Saad M, et al. Genomic landscape of tumor-host interactions with differential prognostic and predictive connotations. bioRxiv. (2019). doi: 10.1101/546069. [Epub ahead of print].
36. Malta TM, Sokolov A, Gentles AJ, Burzykowski T, Poisson L, Weinstein JN, et al. Machine learning identifies stemness features associated with oncogenic dedifferentiation. Cell. (2018) 173:338–54.e15. doi: 10.1016/j.cell.2018.03.034
37. Kim JH, Jin HO, Park JA, Chang YH, Hong YJ, Lee JK. Comparison of three different kits for extraction of high-quality RNA from frozen blood. Springerplus. (2014) 3:76. doi: 10.1186/2193-1801-3-76
38. Shi YY, He L. SHEsis, a powerful software platform for analyses of linkage disequilibrium, haplotype construction, and genetic association at polymorphism loci. Cell Res. (2005) 15:97–8. doi: 10.1038/sj.cr.7290272
39. Wheeler J, McHale M, Jackson V, Penny M. Assessing theoretical risk and benefit suggested by genetic association studies of CCR5: experience in a drug development programme for maraviroc. Antivir Ther. (2007) 12:233–45.
40. Yao M, Brummer G, Acevedo D, Cheng N. Cytokine regulation of metastasis and tumorigenicity. Adv Cancer Res. (2016) 132:265–367. doi: 10.1016/bs.acr.2016.05.005
41. Crawford A, Angelosanto JM, Nadwodny KL, Blackburn SD, Wherry EJ. A role for the chemokine RANTES in regulating CD8 T cell responses during chronic viral infection. PLoS Pathog. (2011) 7:e1002098. doi: 10.1371/journal.ppat.1002098
42. Zumwalt TJ, Arnold M, Goel A, Boland CR. Active secretion of CXCL10 and CCL5 from colorectal cancer microenvironments associates with GranzymeB+ CD8+ T-cell infiltration. Oncotarget. (2015) 6:2981–91. doi: 10.18632/oncotarget.3205
43. Araujo JM, Gomez AC, Aguilar A, Salgado R, Balko JM, Bravo L, et al. Effect of CCL5 expression in the recruitment of immune cells in triple negative breast cancer. Sci Rep. (2018) 8:4899. doi: 10.1038/s41598-018-23099-7
44. Pinto JA, Araujo J, Cardenas NK, Morante Z, Doimi F, Vidaurre T, et al. A prognostic signature based on three-genes expression in triple-negative breast tumours with residual disease. NPJ Genom Med. (2016) 1:15015. doi: 10.1038/npjgenmed.2015.15
45. Zhang Q, Qin J, Zhong L, Gong L, Zhang B, Zhang Y, et al. CCL5-mediated Th2 immune polarization promotes metastasis in luminal breast cancer. Cancer Res. (2015) 75:4312–21. doi: 10.1158/0008-5472.CAN-14-3590
46. Svensson S, Abrahamsson A, Rodriguez GV, Olsson AK, Jensen L, Cao Y, et al. CCL2 and CCL5 are novel therapeutic targets for estrogen-dependent breast cancer. Clin Cancer Res. (2015) 21:3794–805. doi: 10.1158/1078-0432.CCR-15-0204
47. Parker JS, Mullins M, Cheang MC, Leung S, Voduc D, Vickery T, et al. Supervised risk predictor of breast cancer based on intrinsic subtypes. J Clin Oncol. (2009) 27:1160–7. doi: 10.1200/JCO.2008.18.1370
48. Prat A, Parker JS, Karginova O, Fan C, Livasy C, Herschkowitz JI, et al. Phenotypic and molecular characterization of the claudin-low intrinsic subtype of breast cancer. Breast Cancer Res. (2010) 12:R68. doi: 10.1186/bcr2635
49. Jayasinghe MM, Golden JM, Nair P, O'Donnell CM, Werner MT, Kurt RA. Tumor-derived CCL5 does not contribute to breast cancer progression. Breast Cancer Res Treat. (2008) 111:511–21. doi: 10.1007/s10549-007-9802-6
50. Walens A, DiMarco AV, Lupo R, Kroger BR, Damrauer JS, Alvarez JV. CCL5 promotes breast cancer recurrence through macrophage recruitment in residual tumors. Elife. (2019) 8:e43653. doi: 10.7554/eLife.43653.029
51. Pantelidou C, Sonzogni O, De Oliveria Taveira M, Mehta AK, Kothari A, Wang D, et al. PARP inhibitor efficacy depends on CD8+ T-cell recruitment via intratumoral STING pathway activation in BRCA-deficient models of triple-negative breast cancer. Cancer Discov. (2019) 9:722–37. doi: 10.1158/2159-8290.CD-18-1218
52. Wilkerson PM, Reis-Filho JS. The 11q13-q14 amplicon: clinicopathological correlations and potential drivers. Genes Chromosomes Cancer. (2013) 52:333–55. doi: 10.1002/gcc.22037
53. Ahlin C, Lundgren C, Embretsén-Varro E, Jirström K, Blomqvist C, Fjällskog M. High expression of cyclin D1 is associated to high proliferation rate and increased risk of mortality in women with ER-positive but not in ER-negative breast cancers. Breast Cancer Res Treat. (2017) 164:667–78. doi: 10.1007/s10549-017-4294-5
54. Tiong KH, Tan BS, Choo HL, Chung FF, Hii LW, Tan SH, et al. Fibroblast growth factor receptor 4 (FGFR4) and fibroblast growth factor 19 (FGF19) autocrine enhance breast cancer cells survival. Oncotarget. (2016) 7:57633–5760. doi: 10.18632/oncotarget.9328
55. Farabaugh SM, Boone DN, Lee AV. Role of IGF1R in breast cancer subtypes, stemness, and lineage differentiation. Front Endocrinol. (2015) 6:59. doi: 10.3389/fendo.2015.00059
56. Lin CC, Lo MC, Moody R, Stevers N, Tinsley S, Gasparyan M, et al. Identification of MLK4 as a novel regulator of cancer stem cells in triple-negative breast cancer. Cancer Res. (2018) 78:Abstract nr 2004. doi: 10.1158/1538-7445.AM2018-2004
Keywords: CCL5, CCL5 signaling genes, triple negative breast cancer, prognosis, susceptibility
Citation: Shan J, Chouchane A, Mokrab Y, Saad M, Boujassoum S, Sayaman RW, Ziv E, Bouaouina N, Remadi Y, Gabbouj S, Roelands J, Ma X, Bedognetti D and Chouchane L (2019) Genetic Variation in CCL5 Signaling Genes and Triple Negative Breast Cancer: Susceptibility and Prognosis Implications. Front. Oncol. 9:1328. doi: 10.3389/fonc.2019.01328
Received: 29 July 2019; Accepted: 13 November 2019;
Published: 06 December 2019.
Edited by:
Laura Cerchia, Italian National Research Council, ItalyReviewed by:
Nella Prevete, University of Naples Federico II, ItalyPaola Ungaro, Italian National Research Council, Italy
Copyright © 2019 Shan, Chouchane, Mokrab, Saad, Boujassoum, Sayaman, Ziv, Bouaouina, Remadi, Gabbouj, Roelands, Ma, Bedognetti and Chouchane. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Lotfi Chouchane, bG9jMjAwOEBtZWQuY29ybmVsbC5lZHU=
 Jingxuan Shan
Jingxuan Shan Aziz Chouchane4
Aziz Chouchane4 Younes Mokrab
Younes Mokrab Salha Boujassoum
Salha Boujassoum Xiaojing Ma
Xiaojing Ma Lotfi Chouchane
Lotfi Chouchane