- 1Department of Gastroenterology, The Second Affiliated Hospital of Chongqing Medical University, Chongqing, China
- 2Department of Urology, The Second Affiliated Hospital of Chongqing Medical University, Chongqing, China
- 3Department of Pathology, The Second Affiliated Hospital of Chongqing Medical University, Chongqing, China
Peritoneal lymphomatosis is extremely rare and associated with poor prognosis. Most practitioners only pay more attention to peritoneal carcinomatosis. However, peritoneal lymphomatosis can be neglected and misdiagnosed. We report a teenager with 10 days of abdominal distension and pain accompanied by computed tomography scan suggesting diffuse thickening of the peritoneum and omentum and abdominopelvic effusion. Tuberculous peritonitis and peritoneal carcinomatosis were initially suspected. However, it was finally confirmed as non-Hodgkin’s B-cell lymphoma by omentum biopsies. He achieved complete remission after chemotherapy and autologous stem cell transplantation. But unfortunately, he suffered a relapse and died 10 months after diagnosis. Following a review of the literature, it can be concluded that the discovery of lymphomatosis in peritoneum is a rare finding. Lymphoma should be considered in the differential diagnosis of unexplained peritoneal thickening on computed tomography, and this case emphasizes the importance of early pathological diagnosis to make sure that the right treatment can be started opportunely.
Introduction
Non-Hodgkin’s lymphoma (NHL) includes more than 90 distinct genotypes of hematologic malignancies. About 90% of NHL cases are of B-cell origin, most commonly seen in diffuse large B-cell lymphoma (DLBCL) (1). DLBCL has been described in any site of the body, most occurring in nodal (usually cervical or abdominal) or extranodal disease, and extranodal involvement occurs in approximately 40% of DLBCLs (2, 3). However, peritoneal lymphomatosis (PL), which receives much less attention than peritoneal carcinomatosis and is more often misdiagnosed, usually related to diffuse B-cell lymphoma, refers to the seeding of parietal peritoneum and surface of the covered abdominal organs with lymphoma cells (4). The most frequent clinical symptoms of peritoneal lymphomatosis are abdominal pain, distension, and weight loss (5). We herein describe a case of a young teenager wherein a clinical picture resembling tuberculous peritonitis and peritoneal carcinomatosis was subsequently diagnosed as non-Hodgkin’s B-cell lymphoma with isolated peritoneal involvement.
Case Presentation
A 16-year-old boy was admitted to our hospital with a 10-day history of abdominal distension and epigastric pain, accompanied by anorexia, nausea, vomiting, dyspnea, palpitations, as well as low fever and night sweating. He had always been in good health with no significant medical or malignant family history.
On physical examination, the patient showed tachycardia, abdominal tension, tenderness, rebound tenderness in the upper quadrant, and shifting dullness. No palpable masses and no extremity edema were noted. Neither lymphadenopathy nor hepatosplenomegaly was detected.
Peripheral blood counts elevated in white blood cell 10.71×109/L (reference range: 3.50–9.50×109/L), neutrophils 8.03×109/L (reference range: 1.80–6.30×109/L), and monocytes 1.10×109/L (reference range: 0.10–0.60×109/L). The values of lymphocytes, hemoglobin, and platelets were normal. Fecal occult blood test was positive. The laboratory tests revealed a high uric acid level of 1,240.2 μmol/L (reference range: 208.0–506.0 μmol/L), aspartate aminotransferase value of 57 U/L (reference range: 15–46 U/L), and the serum carbohydrate antigen 125 value of 758 U/ml (reference range, <35 U/ml). Other values of blood urea nitrogen, creatinine, electrolytes, Epstein-Barr virus (EBV), human immunodeficiency virus (HIV), as well as other tumor markers (Carcinoembryonic antigen, Carbohydrate antigen 19−9, and Alpha fetoprotein), and hepatitis markers were negative. Assistant examinations associated with tuberculosis (Tuberculin skin test, T-SPOT test, and IgG antibody for tuberculosis) were negative. Additionally, abdominocentesis and ascitic fluid examination were performed as well. Orange-yellow turbid fluid with predominance of mononuclear cell indicated exudate.
Cervicothoracic and whole abdominal computed tomography (CT) scan without any intestinal involvement, tumor formation, or enlarged lymph nodes revealed diffuse thickening of peritoneum and omentum, thoracic, abdominal, and pelvic effusion (Figure 1), inflammation in the inferior lobe of the right lung, strip shadow of hepatic hilum and hepatogastric space, dilatation of intrahepatic bile duct, as well as edema and thickening of gallbladder wall. During the coronavirus disease 2019 (COVID-19) pandemic, since the patient’s condition deteriorated progressively, we failed to take any gastrointestinal endoscopies. The result of bone marrow biopsy was negative for malignancy. Finally, the patient underwent a surgical laparoscopy to determine the cause. A biopsy of the greater omentum showed diffuse peritoneal thickening and nodular omental infiltration, consistent with an aggressive type of non-Hodgkin’s B-cell lymphoma. Immunohistochemical stain subsequently showed positivity for CD10, CD20, PAX-5, and c-myc with proliferative fraction of 90% (Figure 2), negativity for CD3, CD56, CD138, and Mum-1, which was in favor of the diagnosis of diffuse large B-cell lymphoma, germinal center B-cell type, stage IV.
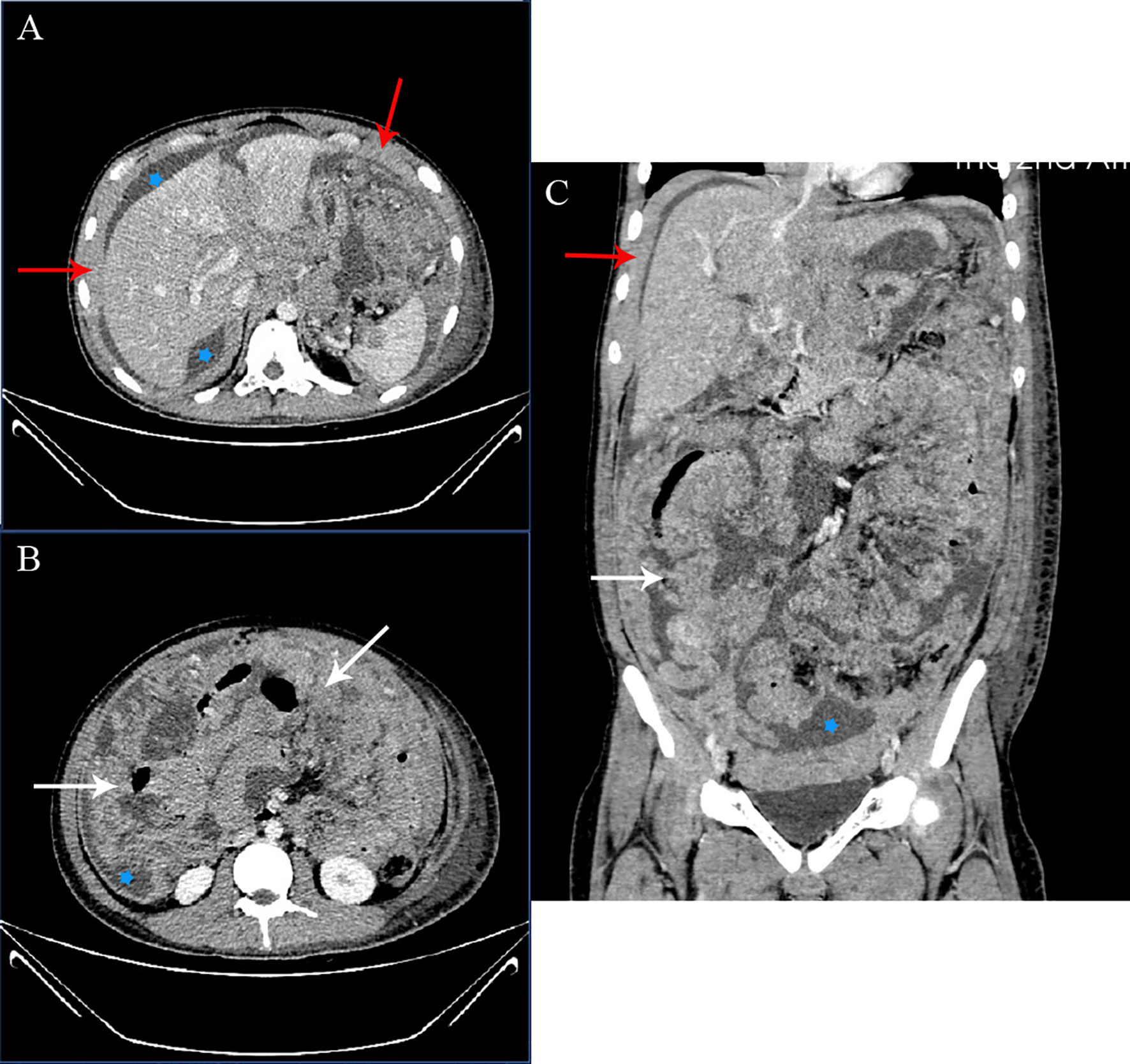
Figure 1 CT examination before-treatment. Axial (A), Axial (B), and coronal (C) enhanced CT images of the abdomen and pelvis in the portal phase. Extensive ascites (stars), thickening of the peritoneum (red arrow), and marked thickening of the omentum (white arrows) noted. No enlarged lymph nodes were noted.
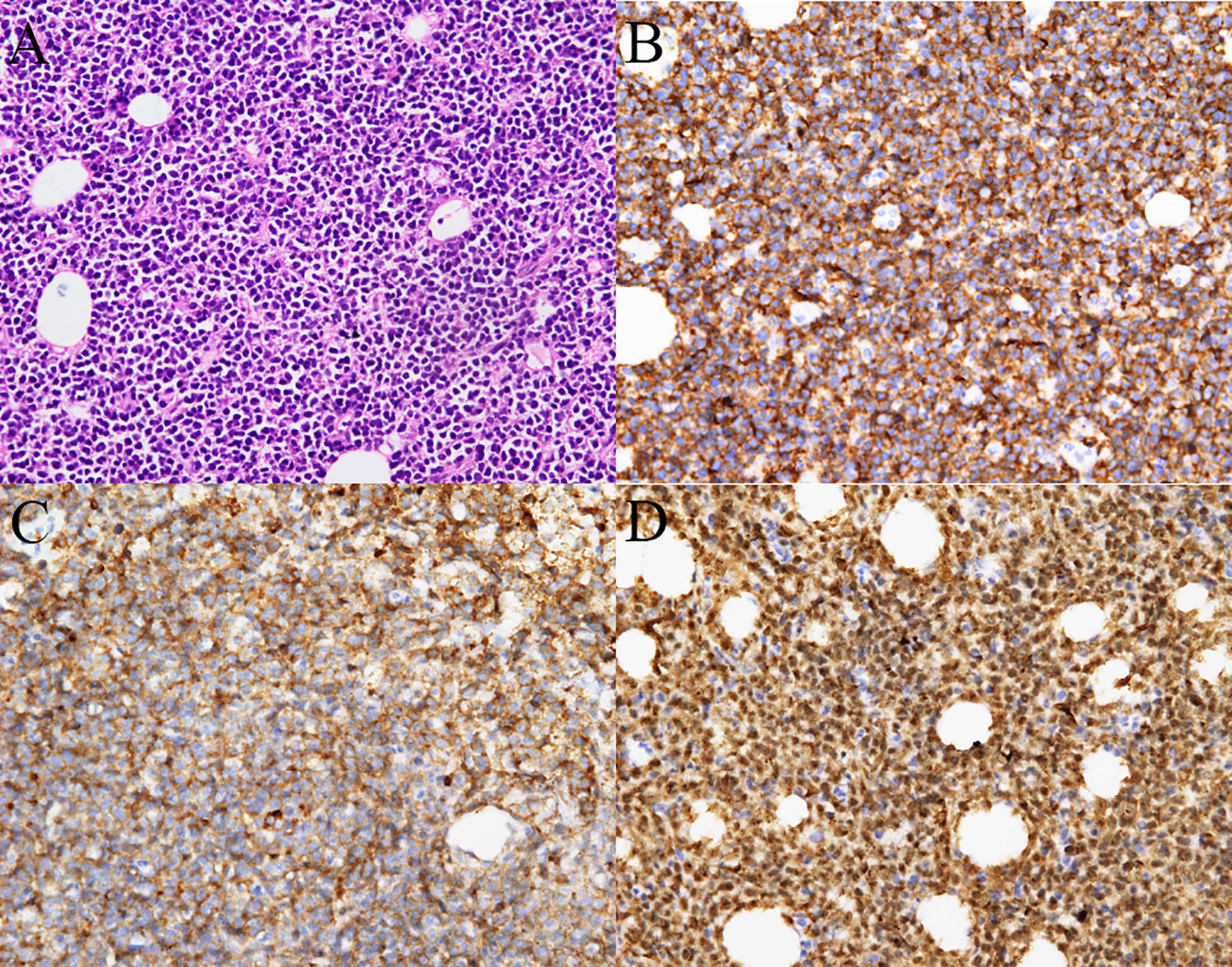
Figure 2 Histopathology from the omental thickening biopsy. (A) Hematoxylin-eosin staining showed diffuse infiltration of neoplastic lymphoid cells (400×). Tumor cells were immunoreactive for (B) CD20, (C) CD10, (D) and expressed high Ki-67 index (nearly 90%) (400×).
Considering the possibility of a diagnosis of peritoneal carcinomatosis, the patient firstly received three courses of hyperthermic intraperitoneal chemotherapy, followed by five cycles of cyclophosphamide, doxorubicin, vincristine, and Prednisone (CHOP) regimen with rituximab. During the first course of chemotherapy, the patient experienced signs and symptoms concerning for tumor lysis syndrome, including oliguria, hyperuricemia, hyperkalemia, hyperphosphatemia, and hypocalcemia. Follow-up CT reexamination demonstrated remission of his condition (Figure 3). Later autologous peripheral blood stem cell transplantation was completed 4 months after the chemotherapy. Unfortunately, the patient suffered a relapse 4 months after the blood stem cell transplantation and was given cisplatin, dexamethasone, and high-dose cytarabine (DHAP) regimen with rituximab, administered concurrently with intrathecal chemotherapy (methotrexate, cytarabine, dexamethasone). In the end, the patient suffered severe marrow suppression and gave up all the treatments for financial difficulty and died 10 months after initial diagnosis.
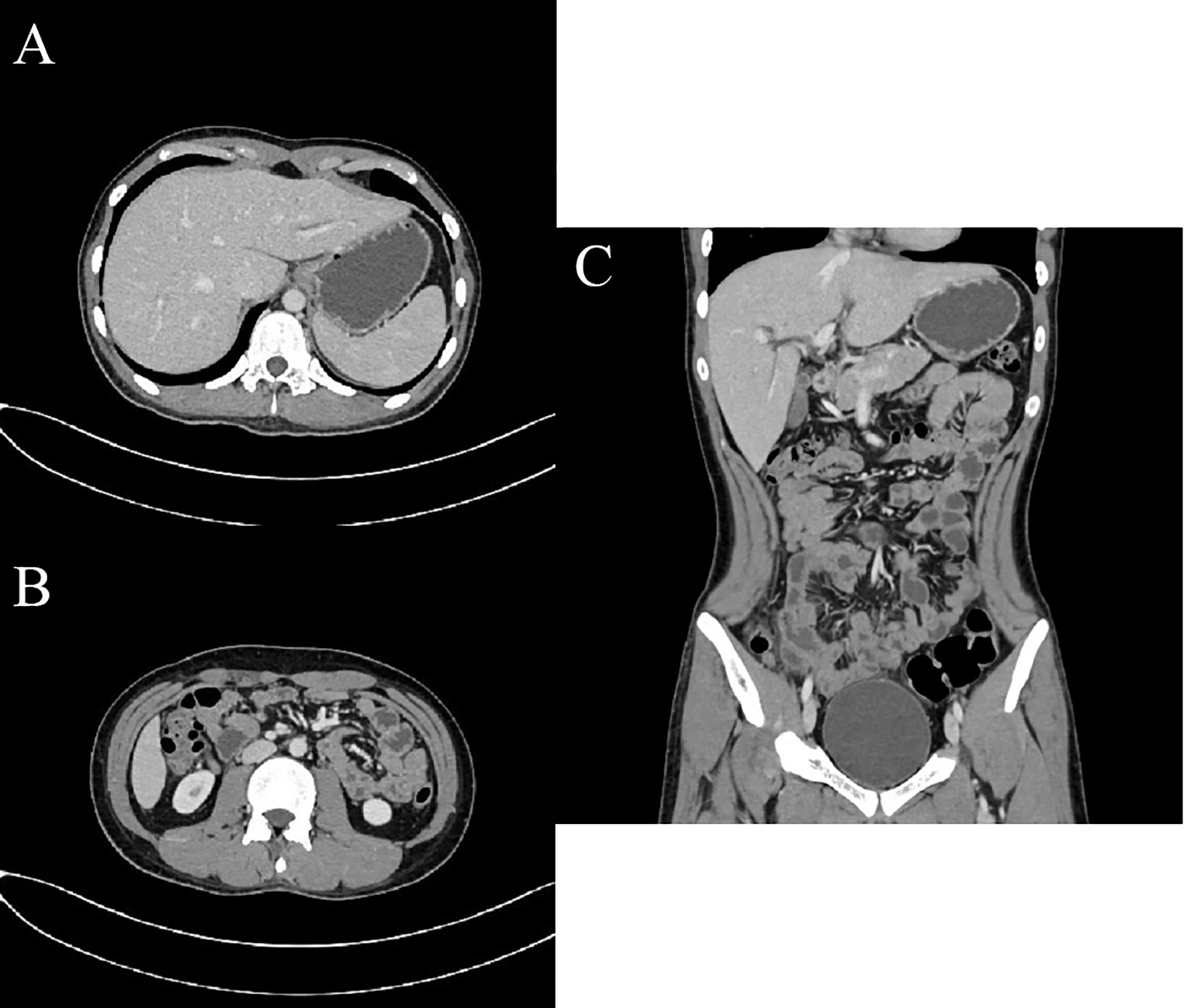
Figure 3 CT examination post-treatment. Axial (A), Axial (B), and coronal (C) enhanced CT images of the abdomen and pelvis in the portal phase post-treatment demonstrated resolution of ascites, thickening of peritoneum and omentum disappeared and turned to the normal shape.
Discussion
Peritoneal lymphomatosis (PL), which is described and explained in few past cases and studies (seen in Table 1), is always found in patients with DLBCL and Burkitt’s lymphoma and more often occurs in males. There is variation in age distribution but found mainly in children and the elderly (4, 5, 11–13). The youngest case reported until now was a 3-year-old infant (14). To our knowledge, this is the first report of isolated peritoneal lymphomatosis with DLBCL in a teenager. Similar to our case, most PL patients predominantly complain of abdominal pain, distension, and weight loss (5, 8, 11), though these clinical manifestations are non-specific.
There usually are two ways of peritoneal invasion in malignant lymphoma (10). First, primary effusion lymphoma (PEL), an uncommon B-cell malignancy, associated with Kaposi’s sarcoma-associated herpesvirus/Human herpesvirus 8 (KSHV/HHV-8) infection and coinfection with EBV or HIV, most often occurs in immunocompromised patients. The main characteristic of PEL is neoplastic effusions in body cavities without any detectable entity tumor (15–17). Second, the peritoneal affection can occur secondary to lesions in the digestive tract or abdominal lymph nodes (18). Moreover, since the omentum does not contain lymphoid tissue, it is uncommon to affect lymphoma. Though the pathway of dissemination is unclear, the possible routes are gastrocolic ligament, transverse mesocolon, and visceral peritoneal surfaces (12, 19). In our case, we discuss a young teenager presenting as isolated PL mainly considering his CT and follow-up imaging did not detect any evidence suggesting primary bowel and lymph node involvement. According to the classification, it refers to PEL, but the patient had no evidence of immunocompromise and infection of EBV or HIV. A limitation of this study is that our patient didn’t take any endoscopies because of the deterioration of the condition during the COVID-19 pandemic. Therefore, we cannot fully rule out the possibility of primary intestinal lymphoma infiltrating the peritoneum and omentum.
The differential diagnosis of peritoneal thickening includes peritoneal lymphomatosis, peritoneal carcinomatosis, tuberculous peritonitis, mesothelioma, and metastatic tumors, such as ovarian, breast, and gastrointestinal carcinomas (20, 21). PL has common imaging characteristics with peritoneal carcinomatosis and tuberculosis containing ascites, thickened peritoneum and omentum (22). An earlier study reported the distinguishing CT findings of peritoneal lymphomatosis including ascites without any loculation or septation and diffuse enlarged lymph nodes (21). Cabral et al. suggested that no gastrointestinal tract involvement is indicative of peritoneal lymphomatosis and that diffuse lymph node enlargement, smooth peritoneal soft tissue thickening or bulky homogeneous masses, as well as imaging findings of changeable extranodal lymphoma involvement were helpful signs of peritoneal lymphomatosis (19). Imaging findings of lymphomatosis rather than carcinomatosis include lymphadenopathy, large mesenteric mass, and splenomegaly (22). Positron emission tomography computed tomography (PET-CT) scan can be utilized to differentiate whether the ascites are caused by malignant lesions (23). It shows hypermetabolic activity of the peritoneal thickening may contribute to guide sampling, especially in DLBCL and Burkitt’s lymphoma (24, 25). CT/PET-CT may be the first choice to distinguish these kinds of diseases. In our case, as our patient was decided to undergo the omental biopsy, he didn’t take the PET-CT, which limited the certainty of the diagnoses regarding if the disease was isolated to the peritoneum or was a part of a more generalized lymphoma. His CT scan suggested diffuse thickening of the peritoneum and omentum with large-volume ascites, which reflected common imaging features of peritoneal lymphomatosis and peritoneal carcinomatosis. The image of our patient did not have any lymph node enlargement or splenomegaly in favor of lymphomatosis. Moreover, the gastrointestinal tract and digestive organs were normal, so we could not exclude either disease.
As imaging findings are non-specific, it has been proven that only pathology can make a definitive diagnosis (26). Aathira et al. reported a case of peritoneal tuberculosis that was confirmed as Burkitt’s lymphoma by omental and bone marrow biopsies (11). Another study reported a rare presentation of follicular lymphoma simulating peritoneal carcinomatosis, and the biopsy of the enlarged supraclavicular lymph node was reported in keeping with follicular lymphoma (8). Besides, histology is regarded as good criteria of diagnosis of peritoneal lymphomatosis, because cytology is not always available and lymphoma can elicit florid mesothelial hyperplasia, leading to inaccurate results and lengthening the diagnostic process (27, 28). Taking into account the history, the imaging findings, and the ascites analysis, the initial possibilities of our case included peritoneal tuberculosis and peritoneal carcinomatosis. Surprisingly, we took prompt omentum biopsies, and it was finally confirmed as non-Hodgkin’s B-cell lymphoma.
Peritoneal lymphomatosis is rare and aggressive. Based on these previous experiences and literature reviews, chemotherapy regimens for peritoneal lymphoma are similar to those for DLBCL using CHOP with or without rituximab based on staging (5, 10). Twenty-five percent of cases with peritoneal lymphomatosis were reported to have a good response to chemotherapy and prognosis (11, 29). Conversely, DLBCL behaves aggressively and has a poor prognosis. If untreated, the median survival is less than 1 year (30). It is therefore necessary to recognize this rare disease to obtain a pathological diagnosis as early as possible and make sure that the correct treatment can be started opportunely.
In conclusion, PL is a rare presentation of extranodal involvement of lymphoma with known poor prognosis. With no characteristic clinical presentations and specific imaging, PL is often misdiagnosed, and the delay of the diagnosis reduces the possibility for optimal treatment and chance of survival. In such ambiguous cases, early histological diagnosis is the primary tool of the rapid and effective treatment.
Data Availability Statement
The original contributions presented in the study are included in the article/supplementary material. Further inquiries can be directed to the corresponding authors.
Ethics Statement
Ethical review and approval was not required for the study on human participants in accordance with the local legislation and institutional requirements. Written informed consent to participate in this study was provided by the participants’ legal guardian/next of kin. Written informed consent was obtained from the individual(s), and minor(s)’ legal guardian/next of kin, for the publication of any potentially identifiable images or data included in this article.
Author Contributions
MZ did the data collection and wrote the original draft. ZW, ZY, BN, SY, and HY contributed to patient’s care and did a critical revision of the manuscript. XG contributed to the pathology description and images. HY and XG contributed to the final version of the manuscript. All authors contributed to the article and approved the submitted version.
Funding
This work was supported by Kuanren Talents Program of the Second Affiliated Hospital of Chongqing Medical University and Young Medical High-end Reserve Talents Program of Chongqing (to HY).
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Kareff S, Yin C, Feigert J. High-Grade B-Cell Lymphoma Masquerading as Peritoneal Lymphomatosis. BMJ Case Rep (2019) 12(8):e231238. doi: 10.1136/bcr-2019-231238
2. Møller M, Pedersen N, Christensen B. Diffuse Large B-Cell Lymphoma: Clinical Implications of Extranodal Versus Nodal Presentation–a Population-Based Study of 1575 Cases. Br J Haematol (2004) 124(2):151–9. doi: 10.1046/j.1365-2141.2003.04749.x
3. Mey U, Hitz F, Lohri A, Pederiva S, Taverna C, Tzankov A, et al. Diagnosis and Treatment of Diffuse Large B-Cell Lymphoma. Swiss Med Wkly (2012) 142:w13511. doi: 10.4414/smw.2012.13511
4. Curakova E, Genadieva-Dimitrova M, Misevski J, Caloska-Ivanova V, Andreevski V, Todorovska B, et al. Nonhodgkin’s Lymphoma With Peritoneal Localization. Case Rep Gastrointest Med (2014) 2014:723473. doi: 10.1155/2014/723473
5. Flores E, Aydin N, Vu D, Misra S. A Case Series of Diffuse Large B-Cell Lymphoma and Burkitt Lymphoma Presenting With Peritoneal Lymphomatosis. Int J Surg Case Rep (2016) 28:262–5. doi: 10.1016/j.ijscr.2016.10.007
6. Wong S, Sanchez T, Swischuk L, Huang F. Diffuse Peritoneal Lymphomatosis: Atypical Presentation of Burkitt Lymphoma. Pediatr Radiol (2009) 39(3):274–6. doi: 10.1007/s00247-008-1063-y
7. Oliveira C, Matos H, Serra P, Catarino R, Estevão A. Adult Abdominal Burkitt Lymphoma With Isolated Peritoneal Involvement. J Radiol Case Rep (2014) 8(1):27–33. doi: 10.3941/jrcr.v8i1.1400
8. Cunningham N, Ffrench-Constant S, Planche K, Gillmore R. Peritoneal Lymphomatosis: A Rare Presentation of Follicular Lymphoma Mimicking Peritoneal Carcinomatosis. BMJ Case Rep (2015) 2015:bcr2014207136. doi: 10.1136/bcr-2014-207136
9. Fei N, Shah N. Adult Sporadic Burkitt’s Lymphoma Presenting With Rapid Development of Peritoneal Lymphomatosis. Case Rep Oncol Med (2017) 2017:4789706. doi: 10.1155/2017/4789706
10. Liu E, Wang J, Yang WJM. Peritoneal Lymphoma With Ascites Mimicking Portal Hypertensive Ascites: A Case Report. Medicine (Baltimore) (2019) 98(8):e14583. doi: 10.1097/md.0000000000014583
11. Ravindranath A, Srivastava A, Seetharaman J, Pandey R, Sarma M, Poddar U, et al. Peritoneal Lymphomatosis Masquerading as Pyoperitoneum in a Teenage Boy. ACG Case Rep (2019) 6(6):e00116. doi: 10.14309/crj.0000000000000116
12. Sharifah M, Zamzami N, Rafeah T. Diffuse Peritoneal Lymphomatosis Simulating Peritoneal Carcinomatosis. Med J Malaysia (2011) 66(3):270–2.
13. Salemis N, Gourgiotis S, Tsiambas E, Karagkiouzis G, Nakos G, Karathanasis V. Diffuse Large B Cell Lymphoma of the Mesentery: An Unusual Presentation and Review of the Literature. J Gastrointest Cancer (2009) 40:79–82. doi: 10.1007/s12029-009-9114-7
14. Lu G, Qiao L, Li D, Liu Z, Zhao F, Yu DJM. Concurrent Lymphoma and Hemophilia B in a Pediatric Patient: A Case Report. Medicine (Baltimore) (2019) 98(19):e15474. doi: 10.1097/md.0000000000015474
15. Chen Y, Rahemtullah A, Hochberg E. Primary Effusion Lymphoma. Oncologist (2007) 12(5):569–76. doi: 10.1634/theoncologist.12-5-569
16. Carbone A, Gloghini AJC. PEL and HHV8-Unrelated Effusion Lymphomas: Classification and Diagnosis. Cancer (2008) 114(4):225–7. doi: 10.1002/cncr.23597
17. Shimada K, Hayakawa F, Kiyoi HJB. Biology and Management of Primary Effusion Lymphoma. Blood (2018) 132(18):1879–88. doi: 10.1182/blood-2018-03-791426
18. Glazer H, Lee J, Balfe D, Mauro M, Griffith R, Sagel SJR. Non-Hodgkin Lymphoma: Computed Tomographic Demonstration of Unusual Extranodal Involvement. Radiology (1983) 149(1):211–7. doi: 10.1148/radiology.149.1.6225145
19. Cabral F, Krajewski K, Kim K, Ramaiya N, Jagannathan J. Peritoneal Lymphomatosis: CT and PET/CT Findings and How to Differentiate Between Carcinomatosis and Sarcomatosis. Cancer Imaging (2013) 13:162–70. doi: 10.1102/1470-7330.2013.0018
20. Choi W, Kim J, Choi S, Park J, Park Y, Lim J, et al. Peritoneal Lymphomatosis Confused With Peritoneal Carcinomatosis Due to the Previous History of Gastric Cancer: A Case Report. Clin Imaging (2016) 40(5):837–9. doi: 10.1016/j.clinimag.2016.03.006
21. Kim Y, Cho O, Song S, Lee H, Rhim H, Koh B. Peritoneal Lymphomatosis: CT Findings. Abdom Imaging (1998) 23(1):87–90. doi: 10.1007/s002619900292
22. O’Neill A, Shinagare A, Rosenthal M, Tirumani S, Jagannathan J, Ramaiya N. Differences in CT Features of Peritoneal Carcinomatosis, Sarcomatosis, and Lymphomatosis: Retrospective Analysis of 122 Cases at a Tertiary Cancer Institution. Clin Radiol (2014) 69(12):1219–27. doi: 10.1016/j.crad.2014.06.019
23. Song J, Li H, Li Y, Yu H, Li X, Yu S, et al. Is PET-CT Scan an Effective Tool to Differentiate the Ascites Caused by Abdominal Malignancy and Peritoneal Tuberculosis? Clin Res Hepatol Gastroenterol (2014) 38(2):e41–3. doi: 10.1016/j.clinre.2013.08.009
24. Roy S, Parida G, Tripathy S, Singhal A, Shamim S, Tripathi M. Peritoneal Super Scan on F - FDG PET-CT in a Patient of Burkitt’s Lymphoma. Indian J Nucl Med (2017) 32(2):155–6. doi: 10.4103/0972-3919.202250
25. Wang X, Chen Z, Tang G, Zhang X. A Child With Burkitt Lymphoma With Pleural, Peritoneal, Mesenteric, Omental, and Renal Involvement: Diagnostics by FDG PET/CT. Clin Nucl Med (2011) 36(7):612–5. doi: 10.1097/RLU.0b013e318217af84
26. Mito M, Fukunari H, Watanabe A, Hayashi T. A Case of Peritoneal Lymphomatosis Diagnosed by Laparoscopic Biopsy. Gan To Kagaku Ryoho (2020) 47(13):1792–4.
27. Hazarika N, Dhabhar B, Saikia T. Highly Elevated Serum CA 125 in a Lady With Ascites and Retroperitoneal Mass–a Diagnostic Dilemma. J Assoc Physicians India (2008) 56:47–8.
28. Aslam M. Peritoneal Lymphomatosis, a Morphological Look Alike to Peritoneal Carcinomatosis: An Autopsy Report. J Clin Pathol (2009) 62(5):480. doi: 10.1136/jcp.2008.061671
29. Karaosmanoglu D, Karcaaltincaba M, Oguz B, Akata D, Ozmen M, Akhan O. CT Findings of Lymphoma With Peritoneal, Omental and Mesenteric Involvement: Peritoneal Lymphomatosis. Eur J Radiol (2009) 71(2):313–7. doi: 10.1016/j.ejrad.2008.04.012
Keywords: peritoneal lymphomatosis, non-Hodgkin’s lymphoma, peritoneal carcinomatosis, diagnosis, case report
Citation: Zhu M, Wu Z, Yang Z, Ning B, Yu S, Gu X and Yu H (2021) Non-Hodgkin’s Lymphoma Presenting as Isolated Peritoneal Lymphomatosis: A Case Report and Literature Review. Front. Oncol. 11:719554. doi: 10.3389/fonc.2021.719554
Received: 02 June 2021; Accepted: 09 August 2021;
Published: 02 September 2021.
Edited by:
Mohamed A. Yassin, Hamad Medical Corporation, QatarReviewed by:
Honar Cherif, Hamad Medical Corporation, QatarShereen Elazzazy, Hamad Medical Corporation, Qatar
Copyright © 2021 Zhu, Wu, Yang, Ning, Yu, Gu and Yu. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Xiling Gu, Njc2ODE2NDkzQHFxLmNvbQ==; Huihong Yu, eXVodWlob25nMTk5OUAxMjYuY29t
 Min Zhu
Min Zhu Zhixuan Wu1
Zhixuan Wu1