- 1School of Chinese Medicine, Shandong University of Traditional Chinese Medicine, Jinan, China
- 2Department of Obstetrics and Gynecology, Shandong Provincial Third Hospital, Jinan, China
- 3Department of Pathology, Shandong University of Traditional Chinese Medicine, Jinan, China
β-Sitosterol (SIT), a white powdery organic substance with a molecular formula of C29H50O, is one of the most abundant naturally occurring phytosterols in plants. With a chemical composition similar to that of cholesterol, SIT is applied in various fields such as medicine, agriculture, and chemical industries, owing to its unique biological and physicochemical properties. Modern pharmacological studies have elucidated good anti-tumor therapeutic effect activity of SIT, which mainly manifests as pro-apoptotic, anti-proliferative, anti-metastatic, anti-invasive, and chemosensitizing on tumor cells. In addition, SIT exerts an anti-tumor effect on multiple malignant tumors such as breast, gastric, lung, kidney, pancreatic, prostate, and other cancers. Further, SIT derivatives with structural modifications are promising anti-tumor drugs with significant anti-tumor effects. This review article focuses on recent studies relevant to the anti-tumor effects of SIT and summarizes its anti-tumor mechanism to provide a reference for the clinical treatment of malignant tumors and the development of novel anti-tumor drugs.
Introduction
Malignant tumors represent a global medical problem owing to their high incidence, difficult early diagnosis, variable biological characteristics, high recurrence and metastasis, and high mortality. Cancer is ranked as the second leading cause of death and morbidity worldwide (1). According to the World Health Organization, approximately 19.3 million patients were newly diagnosed with cancer and nearly 10 million patients died from cancer worldwide in 2020. Globally, 28.4 million new cases are estimated to occur by 2040, a 47% increase from 2020 (2). Cancer not only poses a serious threat to human life and health but also places a great burden on the public health system, resulting in a series of social problems. Therefore, the prevention and treatment of tumors have become the focus of medical research.
Phytosterols are natural bioactive compounds present in plant cell membranes, of which β-sitosterol (SIT) is the most abundant and broadly distributed in lipid-rich plant foods such as vegetables, nuts, seeds, grains, and olive oil (3–6). SIT has been experimentally demonstrated to exhibit multiple pharmacological properties, such as anti-diabetic (7), ameliorative effect on prostatic hyperplasia (8), anti-inflammatory (9), anti-atherosclerotic (10), lipid-lowering and hepatoprotective (11, 12), immune regulation (13), and protection against oxidative damage (14). Due to its efficacy, low toxicity, and good safety, SIT has attracted significant attention from researchers in recent years. With in-depth pharmacological investigation, SIT has been found to exhibit substantial anti-tumor activity and is a prospective drug for the treatment of malignant tumors. In this review, the research progress on the anti-tumor mechanism of SIT is summarized to provide new insights into solving the current problems of treatment with traditional anti-tumor drugs, such as poor efficacy, high toxicity, and drug resistance.
Induction of Tumor Cell Apoptosis After SIT Intervention
Apoptosis resistance is a common feature of human cancer cells and is generally correlated with resistance to anti-cancer therapy (15). Inhibiting the malignant growth of tumor cells and inducing tumor cell apoptosis are the main anti-tumor strategies. Apoptosis or programmed cell death is controlled by diverse signaling pathways involving various regulatory proteins, and the effect of SIT on these pathways has been demonstrated in several studies.
Tumor Suppressor Protein p53
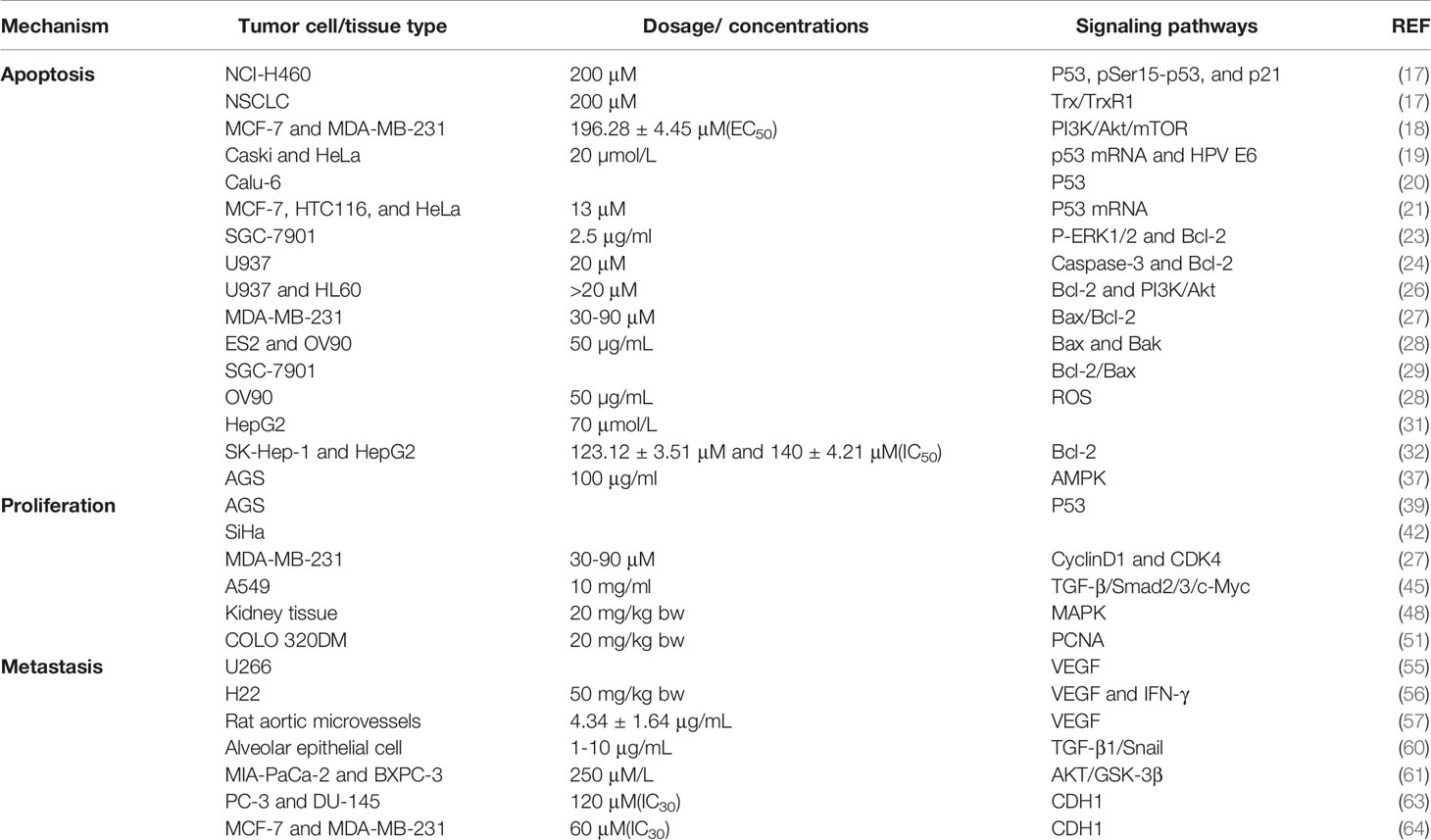
p53 is a tumor-suppressive protein that strictly regulates cell growth by promoting apoptosis and DNA repair under specific conditions. Mutated p53 can lead to abnormal cell proliferation and tumor development due to loss of function (16). A study conducted by Rajavel et al. illustrated that the levels of p53, pSer15-p53, and p21 were remarkably upregulated in human lung cancer NCI-H460 cells after 72 h of SIT intervention (17). The authors suggested that p53 activation is an important process in SIT-mediated apoptosis of non-small cell lung adenocarcinoma (NSCLC) cells, and that SIT elicits ROS-dependent apoptosis in NSCLC cells via downregulating the thioredoxin(Trx)/thioredoxin reductase (TrxR1) signaling pathway. In another study, Zhu et al. revealed that SIT impeded the viability of human breast cancer cells (MCF-7 and MDA-MB-231) through regulation of the PI3K/Akt/mTOR pathway, possibly the primary mechanism of its anti-tumor activity. SIT could markedly reduce the phosphorylation levels of Akt, B cell lymphoma-2 (Bcl-2)-associated agonist of cell death (Bad), p53, p38, proline-rich Akt substrate of 40 kDa (PRAS40), and glycogen synthase kinase 3β (GSK-3β) (18). Further, Cheng et al. reported the anti-proliferative effect of SIT, which was associated with increased p53 mRNA levels and reduced E6 transcripts of human papillomavirus (HPV) (19). The expression patterns of both p53 and HPV E6 proteins were similar to the corresponding transcriptional levels. In a similar report, Andrea et al. showed that SIT exhibits an anti-proliferative effect on cervical cancer HeLa cells, linked to an elevated level of p53 mRNA and a reduced level of oncogenic HPV E6 following SIT treatment (20). It has also been shown that SIT alters the morphology of human cervical cancer cells (CaSki and HeLa). Electron microscopy revealed reduced surface microvilli in SIT-treated cells with increased electron density in the cell membrane and decreased organelles. SIT intervention has been suggested to gradually impede the malignant characteristics of CaSki and HeLa cells. These results also demonstrate that the expression of proliferating cell nuclear antigen (PCNA) is decreased in CaSki and HeLa cells after SIT treatment, indicative of an anti-proliferative property. Thus, it can be inferred that SIT may limit DNA synthesis in CaSki and HeLa cells, thereby suppressing cell proliferation. Another study by Baeka et al. indicated that SIT could effectively reduce the viability of p53-deficient human lung cancer Calu-6 cells (21).
B Cell Lymphoma-2 (Bcl-2) Protein Family
Bcl-2 is a member of the Bcl-2 apoptosis-modulating protein family, and its impairment has been associated with a variety of cancers (22). Rajavel et al. characterized the expression of Bcl-2 and Bcl-2-associated X protein (Bax) after 72 h of SIT exposure (17). The results clearly showed significant downregulation of the Bcl-2 protein with increased Bax expression. In another report, Wang Juan et al. suggested that SIT-dependent activation of p-extracellular signal-regulated kinase (ERK)1/2 and Bcl-2 can enhance the activity of human monocytes and strengthen their ability to kill gastric cancer SGC-7901 cells (23). Additionally, PARK et al. illustrated that increased apoptosis induced by SIT is associated with downregulation of Bcl-2, protein degradation of poly (ADP-ribose) polymerase (PARP) and phospholipase C-γ1, and activation of cysteine-containing aspartate proteolytic enzyme (caspase-3) (24). SIT does not alter the expression of Bcl-xL and Bax in leukemia cells (U937) but selectively downregulates Bcl-2. This suggests a correlation between SIT-evoked apoptosis and caspase-3 activation with Bcl-2 downregulation. Sharmila et al. indicated that SIT treatment led to a significant decrease in the expression of cyclin-D1, Bcl-2, and vascular endothelial growth factor (VEGF) along with a substantial increase in the expression of caspase and Bax, and also inhibited toxicity of N-diethylnitrosamine (DEN) and ferric nitrilotriacetate (Fe-NTA) (25). In another study, SIT could boost the apoptosis of U937 and HL60 cells via mediating the Bcl-2 and PI3K/Akt signaling pathways (26). Similarly, SIT stimulated apoptosis in breast cancer MDA-MB-231 cells by increasing the Bax/Bcl-2 ratio and promoting mitochondrial membrane depolarization (27). Further, SIT elevated the expression level of Bax, Bcl-2 antagonist/killer (Bak), caspase-3 and -9, and cytochrome C in ovarian cancer cells (ES2 and OV90) in a dose-dependent manner (28). This data indicates that SIT upregulates pro-apoptotic signals in ovarian cancer cells. Zhao et al. proposed that SIT strikingly delays the growth of human gastric cancer SGC-7901 cells and facilitates their apoptosis in vitro through a mechanism possibly linked to decreased Bcl-2/Bax ratio and DNA damage (29). Ma et al. provided additional data demonstrating that SIT treatment restrained tumor growth in mice (30). Mechanistically, SIT treatment reduced PI3K/Akt expression, Bad activation, Bcl-xL expression, and cytochrome C release, resulting in caspase-3 and -9 activation, PARP cleavage, and apoptosis. Zhao Xiuhong et al. proposed that SIT can trigger apoptosis in HepG2 cells with a suggested mechanism related to the activation of mitochondria-controlled endogenous apoptotic pathway and death receptor-controlled exogenous apoptotic pathway (31). Another study showed that SIT expedites SK-Hep-1 and HepG2 cell death in a concentration-dependent manner, and the pro-apoptotic mechanism may be associated with Bcl-2-related proteins (32).
Reactive Oxygen Species
ROS represent a set of short-lived, highly reactive, oxygen-containing molecules capable of eliciting DNA damage and affecting the DNA damage response. Increased ROS production has been detected in various cancers with multiple roles. For instance, ROS can activate pro-tumorigenic signals, enhance cell survival and proliferation, and drive DNA damage and genetic instability. They can also induce tumor cell death triggered by oxidative stress (33, 34). Recently, the role of antioxidants in carcinogenesis has attracted considerable attention. SIT is a relatively mild-to-moderate antioxidant that exerts beneficial effects in vitro by reducing ROS levels. A study evaluated the antioxidant potential of SIT in 1,2-dimethylhydrazine (DMH)-induced colon carcinogenesis and detected the contents of enzymatic and non-enzymatic antioxidants and lipid peroxides in the colon and liver tissues (35). ROS generation exceeded the endogenous antioxidant capacity of the body, resulting in a severe imbalance in the cellular antioxidant defense mechanism. SIT could effectively attenuate DMH-induced elevation of hepatic lipid peroxide levels. Additionally, it exhibited a protective effect against DMH-induced antioxidant depletion in the colon and liver tissues of experimental animals. The results suggest that SIT can effectively attenuate DMH-induced oxidative stress in rats owing to its antioxidant potential and may serve as an effective chemopreventive drug against colon carcinogenesis. In another study, Bae et al. pretreated cells with N-acetylcysteine (NAC) for 1 h before SIT treatment to assess the relationship between SIT-induced mitochondrial dysfunction and ROS production (28). The results showed that NAC could partially inhibit SIT-induced loss of mitochondrial membrane potential (MMP) in ovarian cancer cells (OV90), suggesting that SIT mainly exerted adverse effects on mitochondria and indirectly disrupted ROS homeostasis. In addition, SIT treatment enhanced ROS production and calcium influx via activation of the endoplasmic reticulum-mitochondrial axis leading to MMP reduction and mitochondrial dysfunction in the two ovarian cancer cell lines. Significantly, in this study, SIT inhibited tumor development by promoting ROS generation, which seems to contradict with the previous researching result, whose reason we speculate is that SIT may have different biological effects on different tumor types or tumor cells without any clear specific mechanism. In summary, these findings demonstrate that SIT stimulates oxidative stress and activates pro-apoptotic signals, thereby promoting apoptosis and limiting the proliferation of ovarian cancer cells.
Adenosine 5′-Monophosphate -Activated Protein Kinase Pathway
AMPK is a key molecule in regulating bioenergy metabolism and plays a significant role in tumor progression. It is closely associated with cancer drug resistance through interaction with multiple known chemoresistance mechanisms. Hence, targeting AMPK has become a new strategy for cancer prevention and treatment (36). Eun et al. reported that SIT, depending on its concentration, strengthens the phosphorylation level of AMPK, and the enhanced activity of AMPK after SIT intervention affects the growth and apoptosis of tumor cells (37). Specifically, SIT-elicited apoptosis in gastric cancer AGS cells was achieved through the AMPK activation-mediated expression of phosphatase and tensin homolog (PTEN) gene.
Inhibition of Tumor Cell Proliferation in Response to SIT
Tumor cells exhibit abnormalities in morphology, metabolism, and function, with varied impaired abilities to differentiate and mature. Tumor cell lines or strains cultured in vitro have the characteristics of unlimited passage and lack apoptosis. Therefore, inhibiting tumor cell proliferation is a vital part of anti-tumor therapy, and success with SIT treatment has been documented in various studies.
Cell Cycle Blockade
Abnormal cell cycle activity almost occurs in all tumor types and provokes tumor cell proliferation. Targeting individual cell cycle components can be an effective anti-cancer strategy (38). In vitro experiments have illustrated that SIT impedes the proliferation of gastric cancer AGS cells by stimulating apoptosis and cell cycle arrest in the S phase, which may be linked to modulation of the p53 pathway (39). Zhou et al. reported that SIT triggered cell cycle arrest in the G2/M phase and apoptotic death in human lung adenocarcinoma A549 cells (40). Further, LU et al. showed that SIT impaired the proliferation of A549 cells in a dose-dependent manner (41). In another report, SIT markedly repressed the proliferation of human cervical squamous cell carcinoma SiHa cells and elicited cell cycle arrest in the S phase (rather than the G2/M phase) and mitotic phase (42). Laser confocal analysis showed that SIT could downregulate the expression of microtubule-associated protein 2 and microtubule-associated protein α. Moreover, SIT reduced the aggregation ratio of the microtubules in a time-dependent manner. These findings suggested that the anti-microtubule properties of SIT could contribute to inhibiting the proliferation of SiHa cells. In addition, Pradhan et al. demonstrated that SIT and its tubulin isotype potentially correlate with drug resistance and SIT can act as a potential tubulin-targeted anti-cancer drug (43). Shiying et al. showed that a high concentration of SIT arrested human breast cancer T47D cells in the G0/G1 phase because it augmented the proportion of cells in the G0/G1 phase while reducing their proportion in the S phase, consequently decreasing proliferation index and suppressing cell proliferation (44). In a similar report, Vundru et al. illustrated that SIT treatment led to G1 arrest in human breast cancer MDA-MB-231 cells corresponding to reduced levels of cyclinD1 and cyclin-dependent kinase (CDK) and increased levels of p21/Cip1 and p27/Kip1 proteins involved in inhibiting the kinase activity of CDK (27). Therefore, down-regulation of CyclinD1 and CDK4 may be related to SIT-induced G1 arrest in breast cancer cells. Further, cyclinD1 and CDK2 downregulation with a proportional increase in the number of cells in the G1 phase validates that the anti-cancer effect of SIT correlates with cell cycle arrest and apoptosis (17). Wang et al. revealed that SIT arrested A549 cells in the G0/G1 phase and repressed cell autophagy and proliferation, and its mechanism might be linked to inhibition of the transforming growth factor-β (TGF-β)/p-Smad2/3/c-Myc signaling pathway (45).
Mitogen-Activated Protein Kinase Pathway
The MAPK pathway has been implicated in cancer progression with involvement in a wide range of cellular processes encompassing differentiation, proliferation, and survival, and it is frequently altered in diseases (46, 47). Sharmila et al. elucidated that Fe-NTA-induced proliferation of rat renal cancer cells was reduced after SIT treatment via a mechanism pertaining to MAPK downregulation and attenuated phosphorylation of p38, ERK, and c-Jun N-terminal kinase (JNK) (48). SIT potentially blocked the MAPK pathway by inhibiting the ATP binding site and acting as a competitive inhibitor of ATP to prevent the activation of ERK1/2. Simultaneously, SIT could reduce the elevated levels of c-fos and c-jun genes caused by renal carcinogens. These results suggest that SIT can block cell proliferation, thereby preventing tumor invasion and angiogenesis. Different studies have shown the growth-inhibitory effects of SIT on various cancer cell lines, including oral, prostate, breast, colon, blood, gastric, and lung cancers.
PCNA Protein Pathway
PCNA is an important protein that affects tumor cell proliferation and is involved in diverse DNA metabolic processes, such as DNA replication and repair, chromatin organization and transcription, and condensation of sister chromatids (49). PCNA serves as an excellent inhibitory target to block highly proliferative cells, thus, contributing to the development of broad-spectrum anti-cancer therapy (50). Baskar et al. elucidated that SIT triggers dose-dependent growth inhibition of human colon cancer COLO 320D cells by scavenging ROS, inducing apoptosis, and inhibiting the expression of β-catenin and PCNA antigens in human colon cancer cells (51). Sharmila et al. concluded that SIT could lead to significant reductions in the protein levels of cyclinD1, PCNA, Bcl-2, and VEGF, noticeable increases in the levels of caspase and Bax, and impaired toxic effects of DEN and Fe-NTA (25).
Inhibition of Tumor Cell Metastasis and Invasion in Response to SIT
Migration and invasion are two essential steps in the metastatic cascade of cancer cells, and metastatic tumors are one of the main causes of cancer-associated deaths globally (52, 53). Metastasis of cancer cells often indicates deterioration of the disease, which greatly increases the difficulty of treatment. Therefore, suppression of tumor cell metastasis and invasion is of great significance in treating tumors, and SIT has shown potential in suppressing tumors by employing different mechanisms.
VEGF
Angiogenesis is essential for the development and growth of cancer. VEGF is a key signal for cancer angiogenesis and can be upregulated by a variety of growth factors (54). Anti-angiogenesis therapy is regarded as an effective approach for the treatment of many tumors. Therefore, the downregulation of VEGF expression shows a positive significance in anti-tumor therapy. Sook et al. demonstrated that SIT could diminish the expression of cyclooxygenase-2 and VEGF in human multiple myeloma U266 cells (55). A study by Lin Mingzhu et al. suggested tumor-suppressive activity of SIT in H22 tumor-bearing mice with a possible mechanism wherein SIT decreased VEGF levels and raised interferon-gamma (IFN-γ) levels in the serum (56). In addition, SIT could inhibit the regeneration of rat aortic microvessels, the underlying mechanism of which was likely linked to the downregulated VEGF expression (57).
Epithelial-Mesenchymal Transition
EMT is associated with tumorigenesis, invasiveness, metastasis, and therapeutic resistance (58). It can confer migratory and invasive properties to cells that can be selected by cancer cells during metastasis. Following EMT induction, cancer cells exhibit strengthened aggressive stem-like features and resistance to apoptosis (59). Park et al. revealed that SIT could reverse EMT in human alveolar epithelial cells by disrupting the TGF-β1/Snail signaling pathway (60). Qicao et al. illustrated that SIT repressed the migration and invasion properties of MIA-PaCa-2 and BXPC-3 cells and downregulated EMT markers and AKT/GSK-3β signaling pathway in pancreatic cancer (61). Downregulation of E-cadherin (CDH1) and EMT is crucial for tumor invasion and metastasis (62). It has been reported that CDH1 expression is notably elevated in prostate cancer cells (PC-3 and DU-145) upon treatment with SIT; the initiation and development of cancer correlate with the decrease in CDH1 expression, and the loss of function of this gene enhances invasive and metastatic capabilities, which in turn expedites tumor progression (63). Additional experiments by Pradhan et al. suggested that SIT upregulates CDH1 expression and effectively impedes the migration of MCF-7 and MDA-MB-231 cells in human breast cancer (64) (see Table 1).
Anti-Tumor Effects of SIT-Related Derivatives/Compounds
Although SIT has significant anti-tumor activity, natural phytosterols undergo auto-oxidation or enzymatic oxidation stimulated by ROS (such as ozone), O2, light, heat, or enzymes (65), resulting in the formation of phytosterol oxidation products (POPs) or oxygenated phytosterols. The beneficial and detrimental side effects of these compounds on human health remain controversial (5), and their poor solubility in water limits their bioavailability and therapeutic effect (66, 67). Therefore, scientists believe that modifying the structure or changing the dosage forms of monomeric compounds is necessary to improve drug release, solubility, targeting, and bioavailability (68). It is essential to develop phytosterol derivatives with significant anti-tumor effects as anti-tumor drugs.
Raj et al. assessed the cytotoxic potential of β-sitosterol-assisted silver nanoparticles (BSS-SNPs) in HepG2 cells, and the results showed that BSS-SNPs remarkably impaired the proliferation of HepG2 cells and augmented ROS levels (69). Treatment with BSS-SNPs induced upregulation of pro-apoptotic markers, such as Bax, p53, cytochrome c, caspase-3, and caspase-9, and downregulation of Bcl-2 expression along with apoptosis-relevant morphological changes. These findings support a theoretical hypothesis suggesting BSS-SNPs as potential drug candidates for hepatocellular carcinoma. Likewise, Shathviha et al. revealed that SIT-silver nanoparticles could effectively induce toxicity and early apoptosis in human colon cancer cells by enhancing p53 protein expression (70).
Tasyriq et al. demonstrated the biological activity of 7α-hydroxy-β-sitosterol in multiple tumor cell lines (71). The compound triggered G0/G1 cell cycle arrest by regulating the Bax/Bcl-2 imbalance and inactivating ERK1/2 and significantly restricted the proliferation of MCF-7 cells in other cancer cell lines.
Kha et al. suggested that SIT-glucoside could restrain the growth of hepatoma cells by stimulating the activity of caspase-3 and -9 via activating their pathways to induce cell apoptosis (72). In a similar report, Dolai et al. illustrated the apoptosis-inducing activity of SIT-glucoside in Ehrlich ascites cancer cells (73). Dose-dependent induction of DNA damage was observed after treatment with SIT-glucoside, and the expression of the apoptotic proteins p53 and p21 was enhanced, which constitute multiple downstream factors of the pro-apoptotic pathway. The increased caspase-3 and -9 activities suggest that caspases are key mediators of the SIT-glucoside-induced apoptosis pathway.
Maiyo et al. revealed significant cytotoxicity of β-sitosterol-3-O-glucoside against colon adenocarcinoma Caco-2 cells and its dose-dependent cytotoxicity in various cancer cell lines (74). The apoptosis experiments showed that, unlike untreated controls, cells treated with β-sitosterol-3-O-glucoside exhibited apoptotic characteristics, whereas no apoptosis was observed in non-cancer HEK293 cells. The results indicate the selective cytotoxic and pro-apoptotic activities of this compound.
Imanaka et al. showed that the metastatic rate of melanoma B16BL6 cells was significantly reduced following liposomal SIT treatment. Oral administration of liposomal SIT exerted a chemopreventive effect on tumor metastasis, potentially enhancing the host defense against metastatic tumor cells (75). The oral administration of liposomal SIT has been suggested to enhance mucosal immunity and strengthen the natural killer cell activity through the induction of interleukin (IL)-18 and IL-12.
Tilahun et al. synthesized stable, redox-sensitive, and bioreducible heparin-β-sitosterol (bHSC) conjugate micelles using heparin, SIT, and cysteamine, and confirmed their anti-metastatic effect (76). Given the advantages of high stability, low toxicity, good hemocompatibility, and high drug-loading capacity, bHSC micelles serve as a good candidate drug delivery system for the treatment of metastatic cancers.
Andima et al. prepared SIT-loaded poly(lactide-co-glycolic acid) nanoparticles using emulsification technology and experimentally verified the anti-proliferative effect of the particles (66). They suggested that the strength of nanoparticle formulations can be easily concentrated at disease sites.
Nisha et al. formulated SIT-loaded PEGylated (SIT-PEG) polymersomes which exhibited stronger in vitro anti-tumor activity than SIT in the cytotoxicity experiments (77). Further, estimation of apoptosis markers, such as caspase-3 and -9, using an enzyme-linked immunosorbent assay showed that the levels of caspase-3 and -9 markedly increased and returned to normal after SIT-PEG treatment. The polymer has been suggested to regulate the expression of caspase-3 and -9 at liver-specific cancer sites.
A newly isolated phytosterol, β-Sitosterol-D-glucoside (β-SDG), derived from sweet potato, may have strong anti-cancer activity. Xu et al. explored the effect of β-SDG on two breast cancer cell lines (MCF7 and MDA-MB-231) and MCF7 tumor-bearing nude mice (78). The results demonstrated the cytotoxic activity of β-SDG against MCF7 and MDA-MB-231 cells due to induced apoptosis and activated caspase proteases in these cells. In vivo experiments revealed that β-SDG regulated the expression of PI3K, p-Akt, Bcl-2 family members, and other factors involved in the PI3K/Akt-mediated mitochondrial pathway by up-regulating the expression of tumor suppressor miR-10a. These findings suggest that β-SDG inhibits tumor growth by disrupting the PI3K/Akt signaling pathway and can be developed as a potential therapeutic agent against MCF7 cell-associated breast cancer (illustrated in Table 2).
Effect of SIT on Other Factors Affecting Tumor Growth
Interleukin Family
Interleukins are key cytokines that affect immune cell function (79). Hao et al. observed a favorable in vivo anti-tumor effect of SIT on an established HepG2 tumor-bearing mouse model (80). SIT treatment remarkably suppressed the growth of solid tumors and enhanced immunity by increasing the thymus and spleen indices. In addition, the increased IL-2 and decreased IL-10 levels following SIT treatment suggest that SIT can also achieve anti-tumor effects by increasing the production of anti-tumor cytokines and reducing the release of immunosuppressive factors in the body.
Metabolic Reprogramming
Metabolic reprogramming can ensure continuous energy supply to tumor cells, and metabolic intermediates can participate in the generation of macromolecular substances in tumor cells. Therefore, targeting metabolic reprogramming has been a research hotspot for the prevention and treatment of tumors. Melanoma brain metastasis is known to be associated with mitochondrial complex I (CI). Terje et al. illustrated that SIT-targeted inhibition of CI resulted in inhibition of mitochondrial electron chain transmission and increased ROS generation, thus inducing melanoma cell apoptosis and repressing their brain metastasis (81). This process occurs only in tumor cells and does not affect normal cells. The resistance of melanoma cells to BRAF inhibitors can also be CI reversed by gene knockout or intervention with SIT. This suggests a clear therapeutic rationale for the application of SIT as a promising drug against melanoma metastasis. In vitro experiments revealed that SIT treatment suppressed the phosphorylation of many proteins related to tumorigenesis, such as AMPK, Akt, GSK, P38, and ERK1/2. It has been suggested that SIT may disrupt fundamental cellular functions such as energy metabolism and cell survival to restrain tumor cell growth, which is absent in normal melanocytes.
Enhanced Sensitivity to Chemotherapy
PLX4720 combined with SIT treatment notably decelerated tumor growth (81). In a similar report, Bae et al. treated human ovarian cancer cells (ES2 and OV90 cancer cells) with SIT alone or in combination with cisplatin or paclitaxel. SIT treatment could further enhance cisplatin- and paclitaxel-induced growth inhibition of cancer cells (28). Another study experimentally demonstrated that SIT combined with cyclophosphamide could effectively enhance anti-tumor effects (80). The nuclear factor kappa-B (NF-κB) signal transduction mechanism is involved in the progression of multiple cancers (82) and is essential for maintaining cell health by controlling fundamental cellular processes such as cell differentiation, growth, and survival (83). A recent study indicated that SIT positively modulates the NF-κB signaling pathway (84). Ziyuan et al. revealed that SIT could activate p53 by disrupting the p53-MDM2 interaction, resulting in increased nuclear translocation of p53 and blockade of the NF-κB pathway (85). This suggested that SIT mediated the p53/NF-κB/breast cancer resistance protein axis to modulate the response of colorectal cancer (CRC) cells to chemotherapy. Oxaliplatin (OXA) and SIT have shown synergistic tumor-suppressive effects in vivo, and the combined application of SIT and OXA may potentially improve CRC treatment. Atif et al. evaluated the effects of SIT and the anti-estrogen drug tamoxifen (TAM) on the growth and ceramide (CER) metabolism of MCF-7 and MDA-MB-231 cells in human breast cancer (86). Their study demonstrated that the combination of SIT and TAM suppressed the growth of both cell lines, and suppression was most pronounced in MDA-MB-231 cells. CER is a pro-apoptotic signal, and its level was augmented in both cell lines after treatment with SIT or TAM alone; however, the combined treatment induced a more significant increase in the cellular CER content. In addition, SIT and TAM increased CER levels in different ways. SIT efficiently activated de novo synthesis of CER in MCF-7 and MDA-MB-231 cells by stimulating serine palmitoyltransferase activity, whereas TAM promoted CER accumulation in the two cell lines by repressing CER glycosylation. These findings support the potential of a combined regimen of dietary SIT and TAM chemotherapy as an effective treatment for breast cancer. Gemcitabine (GEM) is one of the first-line drugs for the treatment of pancreatic cancer; however, its therapeutic effect is not durable due to long-term resistance. In a study by Qicao et al., SIT effectively limited the growth of pancreatic cancer cells through suppression of proliferation, promotion of G0/G1 phase arrest and apoptosis, inhibition of NF-κB activity, upregulation of Bax protein expression, and downregulation of Bcl-2 (87). Furthermore, SIT and GEM exhibited a significant synergistic effect in MIAPaCa-2 and BXPC-3 cells. More importantly, the combined treatment with SIT and GEM noticeably repressed the growth of pancreatic cancer xenografts (see Table 3).
Conclusion and Perspective
Plant-derived phytosterols have various beneficial physiological effects, including anti-hypercholesterolemic, anti-inflammatory, and anti-fungal activity. Considerable attention has been paid to the anti-cancer activity of these natural products with a low risk of side effects and anti-tumor resistance (28). Unlike currently available cancer chemotherapeutics, phytosterols are generally considered safe for human consumption and can be widely added to food matrices.
SIT is the most abundant phytosterol and has a broad spectrum of anti-tumor effects against lung cancer, breast cancer, prostate cancer, colorectal cancer, and leukemia. The anti-tumor effect of SIT is mainly achieved by promoting the apoptosis of tumor cells, inhibiting the malignant proliferation of tumors, and influencing the cell cycle. SIT regulates the malignant behavior of tumor cells through diverse pathways, as illustrated in Figure 1.
SITs are also usually considered non-toxic, with high safety observed in acute toxicity experiments, and this fact should not be ignored (88–92). Despite its well-tolerance and safety, most existing studies on SIT have focused on in vitro cell experiments, and there are very few in vivo studies. However, existing studies have confirmed that SIT has an in vivo antitumor effect (48, 56). Although many studies have suggested that SIT is harmless, some researchers believe it can affect cholesterol metabolism, and cholesterol plays a pivotal role in synapse formation, cell-cell interactions, and intracellular signaling. Meanwhile phytosterols can affect neuroinflammation, neurodegeneration, and disease progression in experimental animal models for different central nervous system disorders (93). But whether SIT has a similar effect is unclear. In many cases, good anti-cancer activity is observed; however, the mechanism of action has not been clearly explained. In addition, the low stability, poor water solubility, and short half-life of SIT also limit its bioavailability.
In summary, SIT has great potential for tumor inhibition, especially its derivatives after structural modification, which are prospective new anti-tumor drugs. It is believed that the anti-tumor pharmacological mechanism of SIT will be further developed and verified in the future.
Author Contributions
Conceptualization: LX and HRZ designed study; XXB and YNZ researched literature and wrote the manuscript; LX and HRZ reviewed. All authors contributed to the article and approved the submitted version.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Abbreviations
SIT, β-sitosterol; NSCLC, No-small cell lung adenocarcinoma; Trx, Thioredoxin; TrxR1, Thioredoxin reductase; HPV, Human papillomavirus; PCNA, Proliferating cell nuclear antigen; Bcl-2, B cell lymphoma-2; Bad, Bcl-2-associated associated agonist of cell death; Bak, Bcl-2 antagonist/killer; Bax, Bcl-2-associated X; PRAS40, Proline-rich Akt substrate of 40 kDa; GSK-3β, Glycogen synthase kinase 3β; DEN, Diethylnitrosamine; Fe-NTA, Ferric nitrilotriacetate; PARP, Poly (ADP-ribose) polymerase; ROS, Reactive oxygen species; VEGF, Vascular endothelial growth factor; DMH, Dimethylhydrazine; NAC, N-acetylcysteine; MMP, Mitochondrial membrane potential; AMPK, Adenosine 5′-monophosphate (AMP)-activated; protein kinase; PTEN, Phosphatase and tensin homolog; CDK, Cyclin-dependent kinase; TGF-β, Transforming growth factor-β; MAPK, Mitogen-activated protein kinase; ERK, Extracellular signal-regulated kinase; JNK, c-Jun N-terminal kinase; IFN-γ, Interferon-gamma; EMT, Epithelial-mesenchymal transition; CDH1, E-cadherin; POPs, Phytosterol oxidation products; BSS-SNP, IL, β-sitosterol-assisted silver nanoparticles; Interleukin; DHSC, Heparin-β-sitosterol conjugate; β-SDG, β-sitosterol-D-glucoside; IL, Interleukin; CI, Complex I; NF-κB, Nuclear factor kappa-B; OXA, Oxaliplatin; CRC, Colorectal cancer; TAM, Tamoxifen; CER, Ceramide; GEM, Gemcitabine.
Glossary
SIT: β-sitosterol
NSCLC: No-small cell lung adenocarcinoma
Trx: Thioredoxin
TrxR1: Thioredoxin reductase
HPV: Human papillomavirus
PCNA: Proliferating cell nuclear antigen
Bcl-2: B cell lymphoma-2
Bad: Bcl-2-associated associated agonist of cell death
Bak: Bcl-2 antagonist/killer
Bax: Bcl-2-associated X
PRAS40: Proline-rich Akt substrate of 40 kDa
GSK-3β,: Glycogen synthase kinase 3β
DEN: Diethylnitrosamine
Fe-NTA: Ferric nitrilotriacetate
PARP: Poly (ADP-ribose) polymerase
ROS: Reactive oxygen species
VEGF: Vascular endothelial growth factor
DMH: Dimethylhydrazine
NAC: N-acetylcysteine
MMP: Mitochondrial membrane potential
AMPK: Adenosine 5′-monophosphate (AMP)-activated, protein kinase
PTEN: Phosphatase and tensin homolog
CDK: Cyclin-dependent kinase
TGF-β: Transforming growth factor-β
MAPK: Mitogen-activated protein kinase
ERK: Extracellular signal-regulated kinase
JNK: c-Jun N-terminal kinase
IFN-γ: Interferon-gamma
EMT: Epithelial-mesenchymal transition
CDH1: E-cadherin
POPs: Phytosterol oxidation products
BSS-SNP: β-sitosterol-assisted silver nanoparticles
IL: Interleukin
DHSC: Heparin-β-sitosterol conjugate
β-SDG: β-sitosterol-D-glucoside
Interleukin: CI
Complex I: NF-κB
Nuclear factor kappa-B: OXA
Oxaliplatin: CRC
Colorectal cancer: TAM
Tamoxifen: CER
Ceramide: GEM
Gemcitabine:
References
1. Fitzmaurice C, Allen C, Barber RM, Barregard L, Bhutta ZA, Brenner H, et al. Global, Regional, and National Cancer Incidence, Mortality, Years of Life Lost, Years Lived With Disability, and Disability-Adjusted Life-Years for 32 Cancer Groups, 1990 to 2015: A Systematic Analysis for the Global Burden of Disease Study. JAMA Oncol (2017) 3(4):524–48. doi: 10.1001/jamaoncol.2016.5688
2. Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA: Cancer J Clin (2021) 71(3):209–49. doi: 10.3322/caac.21660
3. Babu S, Jayaraman S. An Update on β-Sitosterol: A Potential Herbal Nutraceutical for Diabetic Management. Biomed Pharmacother = Biomed Pharmacother (2020) 131:110702. doi: 10.1016/j.biopha.2020.110702
4. Cheng Y, Chen Y, Li J, Qu H, Zhao Y, Wen C, et al. Dietary β-Sitosterol Regulates Serum Lipid Level and Improves Immune Function, Antioxidant Status, and Intestinal Morphology in Broilers. Poultry Sci (2020) 99(3):1400–8. doi: 10.1016/j.psj.2019.10.025
5. Takayasu BS, Martins IR, Garnique AMB, Miyamoto S, Machado-Santelli GM, Uemi M, et al. Biological Effects of an Oxyphytosterol Generated by β-Sitosterol Ozonization. Arch Biochem Biophys (2020) 696:108654. doi: 10.1016/j.abb.2020.108654
6. Awad AB, Chinnam M, Fink CS, Bradford PG. Beta-Sitosterol Activates Fas Signaling in Human Breast Cancer Cells. Phytomed Int J Phytother Phytopharmacol (2007) 14(11):747–54. doi: 10.1016/J.PHYMED.2007.01.003
7. Ponnulakshmi R, Shyamaladevi B, Vijayalakshmi P, Selvaraj J. In Silico and In Vivo Analysis to Identify the Antidiabetic Activity of Beta Sitosterol in Adipose Tissue of High Fat Diet and Sucrose Induced Type-2 Diabetic Experimental Rats. Toxicol Mech Methods (2019) 29(4):276–90. doi: 10.1080/15376516.2018.1545815
8. Cicero AFG, Allkanjari O, Busetto GM, Cai T, Larganà G, Magri V, et al. Nutraceutical Treatment and Prevention of Benign Prostatic Hyperplasia and Prostate Cancer. Archivio Italiano di Urol Androl Organo Ufficiale [di] Soc Italiana di Ecografia Urol e Nefrol (2019) 91(3):139–52. doi: 10.4081/aiua.2019.3.139
9. Paniagua-Pérez R, Flores-Mondragón G, Reyes-Legorreta C, Herrera-López B, Cervantes-Hernández I, Madrigal-Santillán O, et al. Evaluation Of The Anti-Inflammatory Capacity Of Beta-Sitosterol In Rodent Assays. Afr J Tradit Complement Altern Medicines AJTCAM (2017) 14(1):123–30. doi: 10.21010/AJTCAM.V14I1.13
10. Tada H, Nohara A, Inazu A, Sakuma N, Mabuchi H, Kawashiri MA. Sitosterolemia, Hypercholesterolemia, and Coronary Artery Disease. J Atheroscl Thrombosis (2018) 25(9):783–9. doi: 10.5551/jat.RV17024
11. Yuan C, Zhang X, Long X, Jin J, Jin R. Effect of β-Sitosterol Self-Microemulsion and β-Sitosterol Ester With Linoleic Acid on Lipid-Lowering in Hyperlipidemic Mice. Lipids Health Dis (2019) 18(1):157. doi: 10.1186/s12944-019-1096-2
12. Abdou EM, Fayed MAA, Helal D, Ahmed KA. Assessment of the Hepatoprotective Effect of Developed Lipid-Polymer Hybrid Nanoparticles (LPHNPs) Encapsulating Naturally Extracted β-Sitosterol Against CCl(4) Induced Hepatotoxicity in Rats. Sci Rep (2019) 9(1):19779. doi: 10.1038/S41598-019-56320-2
13. Turck D, Castenmiller J, De Henauw S, Hirsch-Ernst KI, Kearney J, Knutsen HK, et al. A Combination of Beta-Sitosterol and Beta-Sitosterol Glucoside and Normal Function of the Immune System: Evaluation of a Health Claim Pursuant to Article 13(5) of Regulation (EC) No 1924/2006. EFSA J Eur Food Saf Authority (2019) 17(7):e05776. doi: 10.2903/J.EFSA.2019.5776
14. Lin F, Xu L, Huang M, Deng B, Zhang W, Zeng Z, et al. β-Sitosterol Protects Against Myocardial Ischemia/Reperfusion Injury via Targeting Pparγ/NF-κb Signalling. Evidence-Based Complement Altern Med eCAM (2020) 2020:2679409. doi: 10.1155/2020/2679409
15. Baguley BC. The Paradox of Cancer Cell Apoptosis. Front Biosci (Landmark edition) (2011) 16:1759–67. doi: 10.2741/3819
16. Kanapathipillai M. Treating P53 Mutant Aggregation-Associated Cancer. Cancers (2018) 10(6):154. doi: 10.3390/cancers10060154
17. Rajavel T, Packiyaraj P, Suryanarayanan V, Singh SK, Ruckmani K, Pandima Devi K. β-Sitosterol Targets Trx/Trx1 Reductase to Induce Apoptosis in A549 Cells via ROS Mediated Mitochondrial Dysregulation and P53 Activation. Sci Rep (2018) 8(1):2071. doi: 10.1038/s41598-018-20311-6
18. Zhu Y, Yao Y, Shi Z, Everaert N, Ren G. Synergistic Effect of Bioactive Anticarcinogens From Soybean on Anti-Proliferative Activity in MDA-MB-231 and MCF-7 Human Breast Cancer Cells In Vitro. Mol (Basel Switzerland) (2018) 23(7):1557. doi: 10.3390/molecules23071557
19. Cheng D, Guo Z, Zhang S. Effect of β-Sitosterol on the Expression of HPV E6 and P53 in Cervical Carcinoma Cells. Contemp Oncol (Poznan Poland) (2015) 19(1):36–42. doi: 10.5114/wo.2015.50011
20. Alvarez-Sala A, Attanzio A, Tesoriere L, Garcia-Llatas G, Barberá R, Cilla A. Apoptotic Effect of a Phytosterol-Ingredient and its Main Phytosterol (β-Sitosterol) in Human Cancer Cell Lines. Int J Food Sci Nutr (2019) 70(3):323–34. doi: 10.1080/09637486.2018.1511689
21. Baeka J, Rohb HS, Choi CI, Baekb KH, Kim KH. Raphanus Sativus Sprout Causes Selective Cytotoxic Effect on P53-Deficient Human Lung Cancer Cells In Vitro. Natural Product Commun (2017) 12(2):237–40. doi: 10.1177/1934578X1701200224
22. Ebrahim AS, Sabbagh H, Liddane A, Raufi A, Kandouz M, Al-Katib A. Hematologic Malignancies: Newer Strategies to Counter the BCL-2 Protein. J Cancer Res Clin Oncol (2016) 142(9):2013–22. doi: 10.1007/s00432-016-2144-1
23. Wang J, Liu JQ, Chen FX, Zhou ZH, Chen YQ, Li Y, et al. The Effect of β-Sitosterol on Cytotoxicity of Human Costimulatory Cells Against Gastric Cancer SGC-7901 Cells and its Mechanism. Immunol J (2014) 30(07):578–84. doi: 10.13431/J.CNKI.IMMUNOL.J.20140127
24. Park C, Moon DO, Rhu CH, Choi BT, Lee WH, Kim GY, et al. Beta-Sitosterol Induces Anti-Proliferation and Apoptosis in Human Leukemic U937 Cells Through Activation of Caspase-3 and Induction of Bax/Bcl-2 Ratio. Biol Pharm Bull (2007) 30(7):1317–23. doi: 10.1248/bpb.30.1317
25. Sharmila R, Sindhu G. Modulation of Angiogenesis, Proliferative Response and Apoptosis by β-Sitosterol in Rat Model of Renal Carcinogenesis. Indian J Clin Biochem IJCB (2017) 32(2):142–52. doi: 10.1007/s12291-016-0583-8
26. Moon DO, Kim MO, Choi YH, Kim GY. Beta-Sitosterol Induces G2/M Arrest, Endoreduplication, and Apoptosis Through the Bcl-2 and PI3K/Akt Signaling Pathways. Cancer Lett (2008) 264(2):181–91. doi: 10.1016/j.canlet.2008.01.032
27. Vundru SS, Kale RK, Singh RP. β-Sitosterol Induces G1 Arrest and Causes Depolarization of Mitochondrial Membrane Potential in Breast Carcinoma MDA-MB-231 Cells. BMC Complement Altern Med (2013) 13:280. doi: 10.1186/1472-6882-13-280
28. Bae H, Park S, Ham J, Song J, Hong T, Choi JH, et al. ER-Mitochondria Calcium Flux by β-Sitosterol Promotes Cell Death in Ovarian Cancer. Antioxid (Basel Switzerland) (2021) 10(10):1583. doi: 10.3390/antiox10101583
29. Zhao Y, Chang SK, Qu G, Li T, Cui H. Beta-Sitosterol Inhibits Cell Growth and Induces Apoptosis in SGC-7901 Human Stomach Cancer Cells. J Agric Food Chem (2009) 57(12):5211–8. doi: 10.1021/jf803878n
30. Ma H, Yu Y, Wang M, Li Z, Xu H, Tian C, et al. Correlation Between Microbes and Colorectal Cancer: Tumor Apoptosis is Induced by Sitosterols Through Promoting Gut Microbiota to Produce Short-Chain Fatty Acids. Apoptosis an Int J Programmed Cell Death (2019) 24(1-2):168–83. doi: 10.1007/s10495-018-1500-9
31. Zhao XH. Effects and Mechanism of β-Sitosterol on Proliferation and Apoptosis of HpeG2 Cells. [D] Lanzhou: Lanzhou University (2018).
32. Anwar MA, Tabassam S, Gulfraz M, Sheeraz Ahmad M, Raja GK, Arshad M. Isolation of Oxyberberine and β-Sitosterol From Berberis Lycium Royle Root Bark Extract and In Vitro Cytotoxicity Against Liver and Lung Cancer Cell Lines. Evidence-Based Complement Altern Med eCAM (2020) 2020:2596082. doi: 10.1155/2020/2596082
33. Moloney JN, Cotter TG. ROS Signalling in the Biology of Cancer. Semin Cell Dev Biol (2018) 80:50–64. doi: 10.1016/j.semcdb.2017.05.023
34. Srinivas US, Tan BWQ, Vellayappan BA, Jeyasekharan AD. ROS and the DNA Damage Response in Cancer. Redox Biol (2019) 25:101084. doi: 10.1016/j.redox.2018.101084
35. Baskar AA, Al Numair KS, Gabriel Paulraj M, Alsaif MA, Muamar MA, Ignacimuthu S. β-Sitosterol Prevents Lipid Peroxidation and Improves Antioxidant Status and Histoarchitecture in Rats With 1,2-Dimethylhydrazine-Induced Colon Cancer. J Med Food (2012) 15(4):335–43. doi: 10.1089/jmf.2011.1780
36. Wang Z, Wang N, Liu P, Xie X. AMPK and Cancer. Experientia Supplementum (2012) . 2016:107. doi: 10.1007/978-3-319-43589-3_9
37. Shin EJ, Choi HK, Sung MJ, Park JH, Chung MY, Chung S, et al. Anti-Tumour Effects of Beta-Sitosterol are Mediated by AMPK/PTEN/HSP90 Axis in AGS Human Gastric Adenocarcinoma Cells and Xenograft Mouse Models. Biochem Pharmacol (2018) 152:60–70. doi: 10.1016/j.bcp.2018.03.010
38. Suski JM, Braun M, Strmiska V, Sicinski P. Targeting Cell-Cycle Machinery in Cancer. Cancer Cell (2021) 39(6):759–78. doi: 10.1016/j.ccell.2021.03.010
39. Zhong JY, Chen HB, Ye DZ, Deng ZJ, Shao JJ, Han JW, et al. [Molecular Mechanism of Ganoderma Against Gastric Cancer Based on Network Pharmacology and Experimental Test]. Zhongguo Zhong Yao Za Zhi = Zhongguo Zhongyao Zazhi = China J Chin Mater Med (2022) 47(1):203–23. doi: 10.19540/J.CNKI.CJCMM.20210902.701
40. Zhou LY, Xu C, Wang LQ, WEI SY, Lu L, Lian XM. Effect of β-Sitosterol on Proliferation and Apoptosis of Lung Cancer Cell Line A549. Int J Lab Med (2016) 37(07):865–7. doi: 10.3969/J.ISSN.1673-4130.2016.07.001
41. Shi L, Tu YJ, Ye SQ, Xia Y, Ma CZ, Peng XZ, et al. A Potential Anti-Cancer Compound Separated From the Chloroform Extract of the Chinese Medicine Formula Shenqi San. Curr Med Sci (2020) 40(1):138–44. doi: 10.1007/s11596-020-2157-5
42. Wang L, Yang YJ, Chen SH, Ge XR, Xu CJ, Gui SQ. [Effects of Beta-Sitosterol on Microtubular Systems in Cervical Cancer Cells]. Zhonghua Yi Xue Za Zhi (2006) 86(39):2771–5. doi: 10.1038/ONC.2013.318
43. Pradhan M, Suri C, Choudhary S, Naik PK, Lopus M. Elucidation of the Anticancer Potential and Tubulin Isotype-Specific Interactions of β-Sitosterol. J Biomol Struct Dynamics (2018) 36(1):195–208. doi: 10.1080/07391102.2016.1271749
44. Tao SY, Niu JZ, Wang JF, Hao QX, Zhao PW, Yang MJ, et al. Effect of β-Sitosterol on T47D Cell Proliferation and Cell Cycle and Its Mechanisms. Modernization Traditional Chin Med Mater Medica-World Sci Technol (2015) 17(02):362–6. doi: 10.11842/WST.2015.02.012
45. Wang X, Li M, Hu M, Wei P, Zhu W. BAMBI Overexpression Together With β-Sitosterol Ameliorates NSCLC via Inhibiting Autophagy and Inactivating TGF-β/Smad2/3 Pathway. Oncol Rep (2017) 37(5):3046–54. doi: 10.3892/or.2017.5508
46. Lee S, Rauch J, Kolch W. Targeting MAPK Signaling in Cancer: Mechanisms of Drug Resistance and Sensitivity. Int J Mol Sci (2020) 21(3):1102. doi: 10.3390/ijms21031102
47. Masliah-Planchon J, Garinet S, Pasmant E. RAS-MAPK Pathway Epigenetic Activation in Cancer: miRNAs in Action. Oncotarget (2016) 7(25):38892–907. doi: 10.18632/oncotarget.6476
48. Sharmila R, Sindhu G. Evaluate the Antigenotoxicity and Anticancer Role of β-Sitosterol by Determining Oxidative DNA Damage and the Expression of Phosphorylated Mitogen-Activated Protein Kinases', C-Fos, C-Jun, and Endothelial Growth Factor Receptor. Pharmacognosy Magazine (2017) 13(49):95–101. doi: 10.4103/0973-1296.197634
49. Cardano M, Tribioli C, Prosperi E. Targeting Proliferating Cell Nuclear Antigen (PCNA) as an Effective Strategy to Inhibit Tumor Cell Proliferation. Curr Cancer Drug Targets (2020) 20(4):240–52. doi: 10.2174/1568009620666200115162814
50. Horsfall AJ, Abell AD, Bruning JB. Targeting PCNA With Peptide Mimetics for Therapeutic Purposes. Chembiochem Eur J Chem Biol (2020) 21(4):442–50. doi: 10.1002/cbic.201900275
51. Baskar AA, Ignacimuthu S, Paulraj GM, Al Numair KS. Chemopreventive Potential of Beta-Sitosterol in Experimental Colon Cancer Model–an In Vitro and In Vivo Study. BMC Complement Altern Med (2010) 10:24. doi: 10.1186/1472-6882-10-24
52. Zeeshan R, Mutahir Z. Cancer Metastasis - Tricks of the Trade. Bosnian J Basic Med Sci (2017) 17(3):172–82. doi: 10.17305/bjbms.2017.1908
53. Zeng A, Hua H, Liu L, Zhao J. Betulinic Acid Induces Apoptosis and Inhibits Metastasis of Human Colorectal Cancer Cells In Vitro and In Vivo. Bioorg Med Chem (2019) 27(12):2546–52. doi: 10.1016/j.bmc.2019.03.033
54. Carmeliet P. VEGF as a Key Mediator of Angiogenesis in Cancer. Oncology (2005) 69 Suppl 3:4–10. doi: 10.1159/000088478
55. Sook SH, Lee HJ, Kim JH, Sohn EJ, Jung JH, Kim B, et al. Reactive Oxygen Species-Mediated Activation of AMP-Activated Protein Kinase and C-Jun N-Terminal Kinase Plays a Critical Role in Beta-Sitosterol-Induced Apoptosis in Multiple Myeloma U266 Cells. Phytother Res PTR (2014) 28(3):387–94. doi: 10.1002/ptr.4999
56. Lin MZ, Zhao Y, Cai EB, Zhu HY, Gao YG, He ZM, et al. Anti-Tumor Effect of β-Sitosterol in H22 Tumor-Bearing Mice. Chin J Public Health (2017) 33(12):1797–800. doi: 10.11847/ZGGGWS2017-33-12-31
57. Taleb Agha M, Baharetha HM, Al-Mansoub MA, Tabana YM, Kaz Abdul Aziz NH, Yam MF, et al. Proapoptotic and Antiangiogenic Activities of Arctium Lappa L. On Breast Cancer Cell Lines. Scientifica (2020) 2020:7286053. doi: 10.1155/2020/7286053
58. Pastushenko I, Blanpain C. EMT Transition States During Tumor Progression and Metastasis. Trends Cell Biol (2019) 29(3):212–26. doi: 10.1016/j.tcb.2018.12.001
59. Suarez-Carmona M, Lesage J, Cataldo D, Gilles C. EMT and Inflammation: Inseparable Actors of Cancer Progression. Mol Oncol (2017) 11(7):805–23. doi: 10.1002/1878-0261.12095
60. Park YJ, Bang IJ, Jeong MH, Kim HR, Lee DE, Kwak JH, et al. Effects of β-Sitosterol From Corn Silk on TGF-β1-Induced Epithelial-Mesenchymal Transition in Lung Alveolar Epithelial Cells. J Agric Food Chem (2019) 67(35):9789–95. doi: 10.1021/acs.jafc.9b02730
61. Cao ZQ, Wang XX, Lu L, Xu JW, Li XB, Zhang GR, et al. Corrigendum: β-Sitosterol and Gemcitabine Exhibit Synergistic Anti-Pancreatic Cancer Activity by Modulating Apoptosis and Inhibiting Epithelial-Mesenchymal Transition by Deactivating Akt/GSK-3β Signaling. Front Pharmacol (2020) 11:565535. doi: 10.3389/fphar.2020.565535
62. Kroepil F, Fluegen G, Totikov Z, Baldus SE, Vay C, Schauer M, et al. Down-Regulation of CDH1 is Associated With Expression of SNAI1 in Colorectal Adenomas. PLoS One (2012) 7(9):e46665. doi: 10.1371/journal.pone.0046665
63. Pradhan N, Parbin S, Kausar C, Kar S, Mawatwal S, Das L, et al. Paederia Foetida Induces Anticancer Activity by Modulating Chromatin Modification Enzymes and Altering Pro-Inflammatory Cytokine Gene Expression in Human Prostate Cancer Cells. Food Chem Toxicol an Int J Published Br Ind Biol Res Assoc (2019) 130:161–73. doi: 10.1016/j.fct.2019.05.016
64. Pradhan N, Parbin S, Kar S, Das L, Kirtana R, Suma Seshadri G, et al. Epigenetic Silencing of Genes Enhanced by Collective Role of Reactive Oxygen Species and MAPK Signaling Downstream ERK/Snail Axis: Ectopic Application of Hydrogen Peroxide Repress CDH1 Gene by Enhanced DNA Methyltransferase Activity in Human Breast Cancer. Biochim Biophys Acta Mol Basis Dis (2019) 1865(6):1651–65. doi: 10.1016/j.bbadis.2019.04.002
65. Zhang B, Yu T, Li L, Meng XH. Recent Advances in Plant Sterol. Food Fermentation Industries (2015) 41(01):190–5. doi: 10.13995/J.CNKI.11-1802/TS.201501036
66. Andima M, Costabile G, Isert L, Ndakala AJ, Derese S, Merkel OM. Evaluation of β-Sitosterol Loaded PLGA and PEG-PLA Nanoparticles for Effective Treatment of Breast Cancer: Preparation, Physicochemical Characterization, and Antitumor Activity. Pharmaceutics (2018) 10(4):232. doi: 10.3390/pharmaceutics10040232
67. Dai Y, Row KH. Imidazole-Modified C(6) -Chitosan Derivatives Used to Extract β-Sitosterol From Edible Oil Samples With a Microwave-Assisted Solid Phase Extraction Method. J Separation Sci (2021) 44(21):3924–32. doi: 10.1002/jssc.202100503
68. Xiong FL, Wu SS, Li XY, Zhang XQ. Research Progress in Antitumor Activity of Dandelion. China Pharmacist (2016) 19(07):1363–6. doi: 10.3969/J.ISSN.1008-049X.2016.07.045
69. Raj RK, E D, R S. β-Sitosterol-Assisted Silver Nanoparticles Activates Nrf2 and Triggers Mitochondrial Apoptosis via Oxidative Stress in Human Hepatocellular Cancer Cell Line. J Biomed Mater Res Part A (2020) 108(9):1899–908. doi: 10.1002/jbm.a.36953
70. Shathviha PC, Ezhilarasan D, Rajeshkumar S, Selvaraj J. β-Sitosterol Mediated Silver Nanoparticles Induce Cytotoxicity in Human Colon Cancer HT-29 Cells. Avicenna J Med Biotechnol (2021) 13(1):42–6. doi: 10.18502/AJMB.V13I1.4577
71. Tasyriq M, Najmuldeen IA, In LL, Mohamad K, Awang K, Hasima N. 7α-Hydroxy-β-Sitosterol From Chisocheton Tomentosus Induces Apoptosis via Dysregulation of Cellular Bax/Bcl-2 Ratio and Cell Cycle Arrest by Downregulating ERK1/2 Activation. Evidence-Based Complement Altern Med eCAM (2012) 2012:765316. doi: 10.1155/2012/765316
72. Vo TK, Ta QTH, Chu QT, Nguyen TT, Vo VG. Anti-Hepatocellular-Cancer Activity Exerted by β-Sitosterol and β-Sitosterol-Glucoside From Indigofera Zollingeriana Miq. Mol (Basel Switzerland) (2020) 25(13):3021. doi: 10.3390/molecules25133021
73. Dolai N, Kumar A, Islam A, Haldar PK. Apoptogenic Effects of β-Sitosterol Glucoside From Castanopsis Indica Leaves. Natural Product Res (2016) 30(4):482–5. doi: 10.1080/14786419.2015.1023201
74. Maiyo F, Moodley R, Singh M. PHYTOCHEMISTRY, CYTOTOXICITY AND APOPTOSIS STUDIES OF B-SITOSTEROL-3-O-GLUCOSIDE AND B -AMYRIN FROM PRUNUS AFRICANA. Afr J Tradit Complement Altern Medicines AJTCAM (2016) 13(4):105–12. doi: 10.21010/ajtcam.v13i4.15
75. Imanaka H, Koide H, Shimizu K, Asai T, Kinouchi Shimizu N, Ishikado A, et al. Chemoprevention of Tumor Metastasis by Liposomal Beta-Sitosterol Intake. Biol Pharm Bull (2008) 31(3):400–4. doi: 10.1248/bpb.31.400
76. Debele TA, Mekuria SL, Tsai HC. Synthesis and Characterization of Redox-Sensitive Heparin-β-Sitosterol Micelles: Their Application as Carriers for the Pharmaceutical Agent, Doxorubicin, and Investigation of Their Antimetastatic Activities In Vitro. Mater Sci Eng C Mater Biol Appl (2017) 75:1326–38. doi: 10.1016/j.msec.2017.03.052
77. Nisha R, Kumar P, Gautam AK, Bera H, Bhattacharya B, Parashar P, et al. Assessments of In Vitro and In Vivo Antineoplastic Potentials of β-Sitosterol-Loaded PEGylated Niosomes Against Hepatocellular Carcinoma. J Liposome Res (2021) 31(3):304–15. doi: 10.1080/08982104.2020.1820520
78. Xu H, Li Y, Han B, Li Z, Wang B, Jiang P, et al. Anti-Breast-Cancer Activity Exerted by β-Sitosterol-D-Glucoside From Sweet Potato via Upregulation of MicroRNA-10a and via the PI3K-Akt Signaling Pathway. J Agric Food Chem (2018) 66(37):9704–18. doi: 10.1021/acs.jafc.8b03305
79. Huang Y, Li D, Qin DY, Gou HF, Wei W, Wang YS, et al. Interleukin-Armed Chimeric Antigen Receptor-Modified T Cells for Cancer Immunotherapy. Gene Ther (2018) 25(3):192–7. doi: 10.1038/gt.2017.81
80. Chen H, Chen WB, Zheng JH. To Study the Mechanism of Sculellaria Barbata β-Sitosterol on Anti-Liver Tumor. Pharm Today (2017) 27(06):380–14. doi: 10.12048/J.ISSN.1674-229X.2017.06.004
81. Sundstrøm T, Prestegarden L, Azuaje F, Aasen SN, Røsland GV, Varughese JK, et al. Inhibition of Mitochondrial Respiration Prevents BRAF-Mutant Melanoma Brain Metastasis. Acta Neuropathol Commun (2019) 7(1):55. doi: 10.1186/S40478-019-0712-8
82. Rasmi RR, Sakthivel KM, Guruvayoorappan C. NF-κb Inhibitors in Treatment and Prevention of Lung Cancer. Biomed Pharmacother = Biomed Pharmacother (2020) 130:110569. doi: 10.1016/j.biopha.2020.110569
83. Pflug KM, Sitcheran R. Targeting NF-κb-Inducing Kinase (NIK) in Immunity, Inflammation, and Cancer. Int J Mol Sci (2020) 21(22):8470. doi: 10.3390/ijms21228470
84. Gangwar V, Garg A, Lomore K, Korla K, Bhat SS, Rao RP, et al. Immunomodulatory Effects of a Concoction of Natural Bioactive Compounds-Mechanistic Insights. Biomedicines (2021) 9(11):1522. doi: 10.3390/biomedicines9111522
85. Wang Z, Zhan Y, Xu J, Wang Y, Sun M, Chen J, et al. β-Sitosterol Reverses Multidrug Resistance via BCRP Suppression by Inhibiting the P53-MDM2 Interaction in Colorectal Cancer. J Agric Food Chem (2020) 68(12):3850–8. doi: 10.1021/acs.jafc.0c00107
86. Awad AB, Barta SL, Fink CS, Bradford PG. Beta-Sitosterol Enhances Tamoxifen Effectiveness on Breast Cancer Cells by Affecting Ceramide Metabolism. Mol Nutr Food Res (2008) 52(4):419–26. doi: 10.1002/mnfr.200700222
87. Cao ZQ, Wang XX, Lu L, Xu JW, Li XB, Zhang GR, et al. β-Sitosterol and Gemcitabine Exhibit Synergistic Anti-Pancreatic Cancer Activity by Modulating Apoptosis and Inhibiting Epithelial-Mesenchymal Transition by Deactivating Akt/GSK-3β Signaling. Front Pharmacol (2018) 9:1525. doi: 10.3389/fphar.2018.01525
88. Bin Sayeed MS, Ameen SS. Beta-Sitosterol: A Promising But Orphan Nutraceutical to Fight Against Cancer. Nutr Cancer (2015) 67(8):1214–20. doi: 10.2174/0929867325666180607093111
89. Blanco-Vaca F, Cedó L, Julve J. Phytosterols in Cancer: From Molecular Mechanisms to Preventive and Therapeutic Potentials. Curr Med Chem (2019) 26(37):6735–49. doi: 10.2174/0929867325666180607093111
90. Mahaddalkar T, Suri C, Naik PK, Lopus M. Biochemical Characterization and Molecular Dynamic Simulation of β-Sitosterol as a Tubulin-Binding Anticancer Agent. Eur J Pharmacol (2015) 760:154–62. doi: 10.1016/j.ejphar.2015.04.014
91. Rajavel T, Mohankumar R, Archunan G, Ruckmani K, Devi KP. Beta Sitosterol and Daucosterol (Phytosterols Identified in Grewia Tiliaefolia) Perturbs Cell Cycle and Induces Apoptotic Cell Death in A549 Cells. Sci Rep (2017) 7(1):3418. doi: 10.1038/s41598-017-03511-4
92. Al-Hazmi GH, Awaad AS, Alothman MR, Alqasoumi SI. Anticandidal Activity of the Extract and Compounds Isolated From Cyperus Conglomertus Rottb. Saudi Pharm J SPJ Off Publ Saudi Pharm Soc (2018) 26(6):891–5. doi: 10.1016/j.jsps.2018.03.007
Keywords: β-Sitosterol, tumor, cancer, derivative, mechanism of action
Citation: Bao X, Zhang Y, Zhang H and Xia L (2022) Molecular Mechanism of β-Sitosterol and its Derivatives in Tumor Progression. Front. Oncol. 12:926975. doi: 10.3389/fonc.2022.926975
Received: 23 April 2022; Accepted: 16 May 2022;
Published: 08 June 2022.
Edited by:
Kevin J Ni, St George Hospital, AustraliaReviewed by:
Mariano Martinez-Vazquez, Universidad Nacional Autónoma de México, MexicoSyed Jafar Mehdi, University of Arkansas for Medical Sciences, United States
Copyright © 2022 Bao, Zhang, Zhang and Xia. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Lei Xia, cGF0aG9sb2d5MDAxQHNpbmEuY29t; Hairong Zhang, c2R6aGhyNzIxMUAxNjMuY29t
†These authors have contributed equally to this work and share first authorship
 Xingxun Bao
Xingxun Bao Yanan Zhang
Yanan Zhang Hairong Zhang2*
Hairong Zhang2* Lei Xia
Lei Xia