- 1David Geffen School of Medicine, University of California, Los Angeles, Los Angeles, CA, United States
- 2Department of Pathology, Brigham and Women’s Hospital and Harvard Medical School, Boston, MA, United States
- 3Department of Pathology, Yale School of Medicine, New Haven, CT, United States
- 4Department of Medicine, Memorial Sloan-Kettering Cancer CenterNew York, NY, United States
- 5Department of Thoracic and Cardiovascular Surgery, The University of Texas MD Anderson Cancer Center, Houston, TX, United States
- 6Loxo@Lilly, Eli Lilly and Company, Warsaw, Poland
- 7Loxo@Lilly, Eli Lilly and Company, Indianapolis, IN, United States
- 8Loxo@Lilly, Eli Lilly and Company, New York, NY, United States
- 9Department of Medicine, Weill Cornell Medical College, New York, NY, United States
The LIBRETTO-001 trial demonstrated the activity of the selective rearrangement during transfection (RET) inhibitor selpercatinib in advanced RET fusion-positive non-small cell lung cancer (NSCLC) and resulted in the drug’s approval for this indication. A cohort that included neoadjuvant and adjuvant selpercatinib was opened on LIBRETTO-001 for early-stage RET fusion-positive NSCLC with the primary endpoint of major pathologic response. A patient with a stage IB (cT2aN0M0) KIF5B-RET fusion-positive NSCLC received 8 weeks of neoadjuvant selpercatinib at 160 mg twice daily followed by surgery. While moderate regression in the primary tumor (stable disease, Response Evaluation Criteria in Solid Tumors (RECIST) guidelines version 1.1) was observed radiologically, assessment via an Independent Pathologic Review Committee revealed a pathologic complete response (0% viable tumor). This consensus assessment by three independent pathologists was aided by RET fluorescence in situ hybridization testing of a reactive pneumocyte proliferation showing no rearrangement. Neoadjuvant selpercatinib was well-tolerated with only low-grade treatment-emergent adverse events. The activity of prospective preoperative selpercatinib in this case establishes proof of concept of the potential utility of RET inhibitor therapy in early-stage RET fusion-positive NSCLC.
1 Introduction
Although cytotoxic chemotherapy and immunotherapy remain cornerstones of perioperative therapy in non-small cell lung cancer (NSCLC), many patients experience recurrent disease and limited survival. In trials randomizing epidermal growth factor receptor (EGFR)-mutant NSCLC to targeted therapy versus chemotherapy, neoadjuvant EGFR tyrosine kinase inhibitor (TKI) achieved high objective response rates (42%–62%), major pathologic response (MPR; 8%–24%), and prolonged progression-free survival (1). Neoadjuvant anaplastic lymphoma kinase TKI therapy was likewise found to be active in anaplastic lymphoma kinase fusion-positive NSCLC, including in patients for whom pathologic complete response (pCR) was achieved (2).
We thus prospectively explored the neoadjuvant use of selpercatinib, a highly selective and potent rearrangement during transfection (RET) kinase inhibitor with central nervous system activity approved in multiple countries for the treatment of RET-altered advanced or metastatic lung or thyroid cancers. The LIBRETTO-001 study (NCT03157128; which provided registration data resulting in the aforementioned approvals) was formally amended in 2021 to include neoadjuvant and adjuvant selpercatinib for early-stage RET fusion-positive NSCLC (3) with the primary endpoint of MPR (Figure 1).
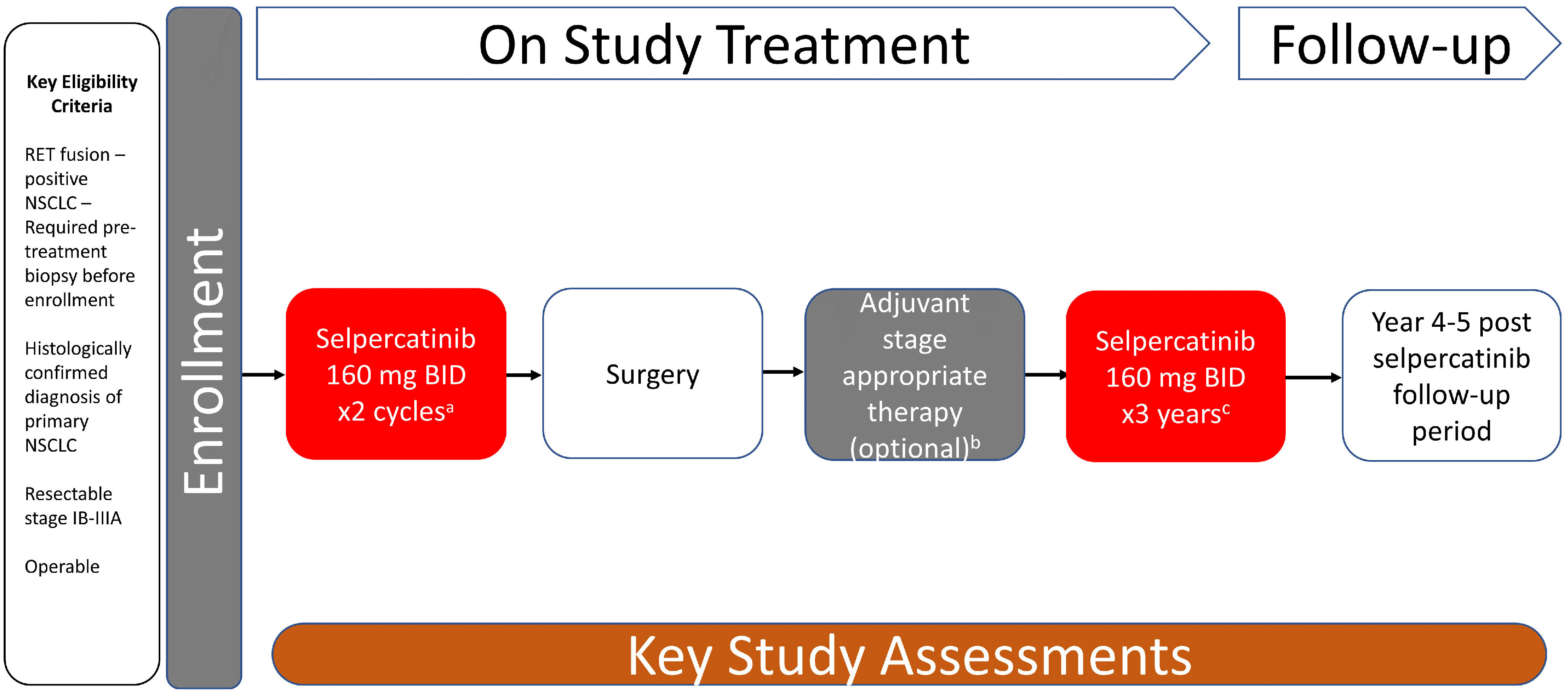
Figure 1 Study design. LIBRETTO-001 trial is a phase 2, open-label, single-arm study of selpercatinib for patients with RET-dependent cancer. The study design of the neoadjuvant-adjuvant cohort (cohort 7) of the trial is summarized in this figure. a Each cycle is 28 days. b Treating physician’s decision. c Up to 3 years of treatment or disease recurrence, patient withdrawal, intolerable toxicity, or death, whichever occurs first. BID, twice daily; NSCLC, non-small cell lung cancer; RET, rearrangement during transfection.
2 Case report
A 76-year-old Caucasian woman with a remote, five pack-year history of former smoking was diagnosed with clinical stage IB (T2aN0M0) adenocarcinoma of the lung at the University of California, Los Angeles, in September 2021. Baseline radiologic tumor assessments identified a left upper lobe tumor (3.1 cm, Figure 2) and no evidence of lymph node or distant metastasis; no F-fluorodeoxyglucose avidity was observed. A transbronchial tumor biopsy showed adenocarcinoma of the lung (Figure 3). DNA- and RNA-based next-generation sequencing of 648 tumor-associated genes (Tempus xT™ Version 4) identified a KIF5B-RET fusion and no other activating genomic aberrations.
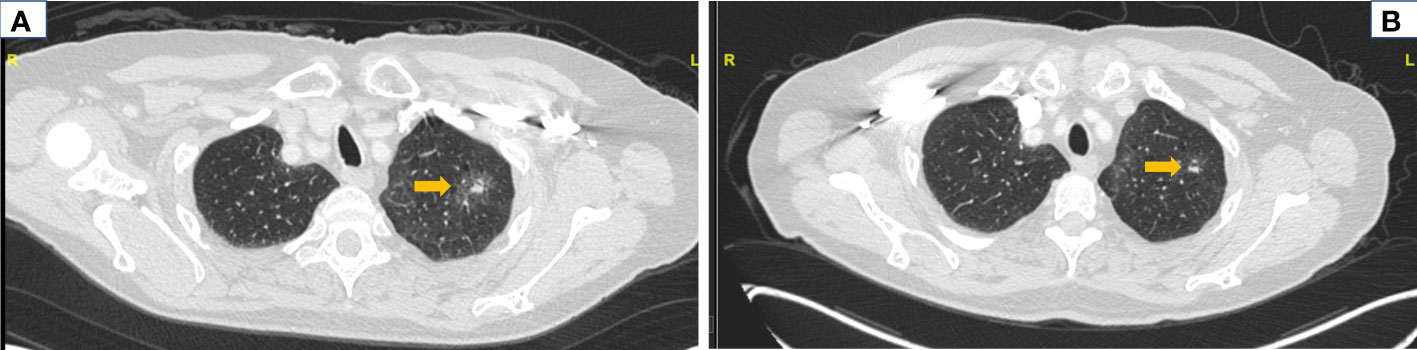
Figure 2 Radiologic response to neoadjuvant selpercatinib. Computed tomography (CT) imaging of chest before and after neoadjuvant selpercatinib treatment is shown. (A) A baseline CT of the chest before selpercatinib initiation revealed a left lung mass measuring 3.1 cm (arrow). (B) A follow-up CT of the chest after 8 weeks of selpercatinib showed disease regression to 2.9 cm in the same mass (arrow). This was classified as stable disease by RECIST v1.1 per investigator assessment. RECIST, Response Evaluation Criteria in Solid Tumors.
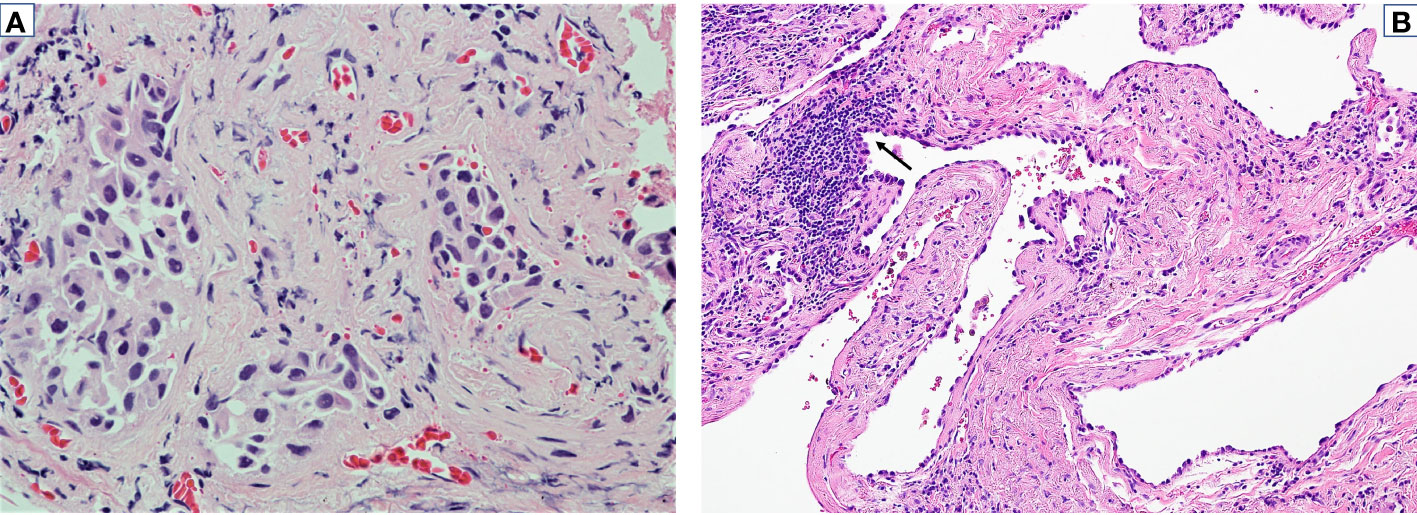
Figure 3 Pathologic response to neoadjuvant selpercatinib. Tumor specimens before and after neoadjuvant selpercatinib treatment are shown. (A) Pathologic assessment of a bronchoscopic biopsy with H&E staining before selpercatinib treatment identified a lung adenocarcinoma (×400). (B) Resected tumor after 8 weeks of selpercatinib treatment showed no evidence of viable tumor cells (×200). Reactive pneumocyte proliferation is denoted by an arrow. Cytogenetic testing of this specimen via RET FISH was negative for a RET fusion. FISH, fluorescence in situ hybridization.
Resectability was confirmed by a thoracic surgeon, and the patient consented to LIBRETTO-001 in October 2021, including the consent to publish her study-related information while keeping her identity confidential. In November 2021, neoadjuvant selpercatinib was initiated at the recommended dose of 160 mg twice daily for two 28-day cycles. Treatment was well tolerated; the only treatment-emergent adverse events (TEAEs) were grade 1 fatigue and diarrhea and grade 2 hypertension. During this time, one dose was missed inadvertently, and there was no dose modification planned.
After the completion of neoadjuvant selpercatinib, the tumor decreased from 3.1 to 2.9 cm (Response Evaluation Criteria in Solid Tumors (RECIST) 1.1 stable disease, investigator assessment, Figure 2). In early January 2022, the patient underwent left upper lobe apical and posterior segmentectomies. The surgery was performed as planned; no surgery-related adverse events were documented. One lymph node in levels 5–6, one lymph node in level 7, and fragments of lymph node(s) from left levels 11, 12, and 13 stations were all negative for carcinoma. The patient declined adjuvant selpercatinib due to low-grade TEAEs during neoadjuvant therapy.
The primary endpoint of the LIBRETTO-001 cohort was MPR. Pathologic assessment of the resected primary tumor in hematoxylin and eosin (H&E) staining was conducted by three independent pathologists including two from the Independent Pathologic Review Committee. On initial assessment, 0%, 10%, and 30% viable tumor cells were found.
To adjudicate the presence or absence of viable tumor cells, dual-color break-apart RET (10q11) fluorescence in situ hybridization (FISH) was performed (University of California, Los Angeles, Clinical Laboratory), which did not detect aberrations in the 200 nuclei examined. Alignment on the pathologic assessment of 0% viable tumor cells (pCR) was then reached among the three pathologists. The consensus was atypical cells represented a reactive pneumocyte proliferation (instead of carcinoma) secondary to treatment effect (Figure 3).
Plasma circulating tumor DNA samples were collected. KIF5B-RET was undetectable (Guardant360) in plasma throughout the trial (pre-selpercatinib, pre-operatively, and post-operatively) likely due to the fact that fusions are less well-detected by circulating tumor DNA and early-phase disease may have limited shedding. No other significant alterations were identified; a platelet-derived growth factor receptor alpha V193I variant of uncertain significance was found pre-selpercatinib (0.19% allele frequency) but not thereafter. This mutation was not detected by next-generation sequencing of the pre-selpercatinib lung cancer biopsy.
3 Discussion
In this patient with a KIF5B-RET-positive stage IB lung adenocarcinoma, prospective treatment with neoadjuvant selpercatinib in the LIBRETTO-001 trial achieved pCR. This case provides the first proof of concept of the clinical activity of a RET TKI in the neoadjuvant setting and sets a precedent for ongoing and future investigations in early-stage disease. While this cohort was closed after this patient was treated (for reasons unrelated to safety or activity), a separate trial continues to explore the utility of selpercatinib as adjuvant therapy in RET fusion-positive NSCLC (LIBRETTO-432; NCT04819100) (4).
pCR is an accepted regulatory endpoint that has supported the approval for neoadjuvant systemic therapy in NSCLC. Neoadjuvant nivolumab and chemotherapy were approved based on pCR as a main outcome measure (5). While it remains to be seen if RET inhibition will eventually be approved as perioperative therapy for RET fusion-positive NSCLC, neoadjuvant TKI therapy has already been approved for another fusion-positive subset of non-metastatic cancer (i.e., tropomyosin receptor kinase (TRK) inhibition for NTRK fusion-positive cancer, including NSCLC, one of whose approved indications is the setting where surgical resection likely results in severe morbidity) (6).
Several other factors are notable in this case. The discordance between radiologic (stable disease) and pathologic (pCR) assessments, conducted within a week of each other, highlights the inherent limitations of RECIST and the importance of pathologic evaluation in fully demonstrating the tumor response to effective therapy, especially in the short timelines of neoadjuvant therapy. Pathologic response in the setting of minimal radiologic response may need to be considered in other settings as well where surgical resection is not performed.
This case also highlights interobserver variation in the interpretation of pathologic response due to the challenge of using standard H&E staining to determine reactive versus neoplastic epithelium, especially in the setting of lepidic proliferations. To mitigate this challenge, molecular adjudication via FISH was utilized (7). A negative RET FISH result provided evidence of the lack of residual tumor (at least related to the original clone) and helped determine that the initially perceived “viable tumor cells” by two of three pathologists represented a benign reactive proliferation (8). The absence of driver alterations suggests a lack of residual tumor since driver alterations are typically retained during the course of cancer evolution (9). The scenario where the original driver is “lost” likely indicates that an independent clone (not related to the tumor in question) has become dominant. This scenario is unlikely in the present case but cannot be entirely ruled out; however, limited tissue precludes further analysis to address this question.
In summary, neoadjuvant selpercatinib was found to be active for a patient with RET fusion-positive NSCLC. Neoadjuvant therapy resulted in low-grade TEAEs but did not preclude or interfere with the performance of definitive surgical therapy. The activity of preoperative selpercatinib in this prospective case establishes proof of concept of the potential utility of selpercatinib in early-stage RET fusion-positive NSCLC.
Data availability statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Ethics statement
The studies involving human participants were reviewed and approved by UCLA Institutional Review Board (UCLA IRB) – Office of the Human Research Protection Program (OHRPP). The patients/participants provided their written informed consent to participate in this study. Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
Author contributions
This study was designed jointly by all authors. The sponsor (Loxo Oncology) collected, analyzed, and interpreted the trial data in collaboration with the authors. All authors were involved in the writing of the manuscript and its revisions. All the authors vouch for the completeness and accuracy of the clinical data and analyses and for the adherence of the trial to the protocol. All authors contributed to the article and approved the submitted version.
Acknowledgments
Preethi Govindarajan and Dana Schamberger from Syneos Health provided medical writing assistance and editorial support.
Conflict of interest
Author JG receives research funding from Eli Lilly and Company. LS consults with Astra Zeneca, Genentech, Eli Lilly and Company, and GV20 Therapeutics. She receives research funding from Bristol Myers Squibb and Genentech. SD is a consultant with Astra Zeneca and receives honoraria from Janssen, Takeda, and Merck. YM-G’s work was supported in part by a National Cancer Institute/ National Institutes of Health Cancer Center Support Grant (P30CA008748) to Memorial Sloan Kettering Cancer Center. YM-G reports travel, accommodation, and expenses from AstraZeneca and Loxo Oncology/Eli Lilly; food/beverages from Endeavor Biomedicines; and honoraria from Virology Education and Projects in Knowledge for a CME program funded by an educational grant from Amgen. She acknowledges associated research funding to the institution from Mirati Therapeutics, Loxo Oncology at Eli Lilly and Company, Elucida Oncology, Taiho Oncology, Hengrui USA, Ltd./Jiangsu Hengrui Pharmaceuticals, Luzsana Biotechnology, and Endeavor Biomedicines. She acknowledges royalties from Rutgers University Press and Wolters Kluwer. YM-G acknowledges receipt of funding from the Andrew Sabin Family Foundation as well as training through an institutional K30 grant from the NIH CTSA UL1TR00457. She has received funding from a Kristina M. Day Young Investigator Award from Conquer Cancer, the ASCO Foundation, endowed by Dr. Charles M. Baum and Carol A. Baum. She is also funded by the Fiona and Stanley Druckenmiller Center for Lung Cancer Research and a Paul Calabresi Career Development Award for Clinical Oncology NIH/NCI K12 CA184746. RR is a consultant with Johnson and Johnson. SS, AS, and BC are employed by Eli Lilly and Company. AD has stocks in TreeLine Bio. He receives honoraria from Medscape, OncLive, PeerVoice, Physicians Education Resources, Targeted Oncology, Research to Practice, Axis, PeerView Institute, Paradigm Medical Communications, WebMD, MJH Life Sciences, Med Learning, Imedex, Answers in CME, Clinical Care Options, EPG Health, JNCC/Harborside, Liberum, and Remedica Ltd. He is in a consulting or advisory role at Ignyta/Genentech/Roche, Loxo/ Bayer/Lilly, Takeda/Ariad/Millenium, TP Therapeutics, AstraZeneca, Pfizer, Blueprint Medicines, Helsinn, BeiGene, BerGenBio, Hengrui Therapeutics, Exelixis, Tyra Biosciences, Verastem, MORE Health, AbbVie, 14ner/Elevation Oncology, ArcherDX, Monopteros, Novartis, EMD Serono, Medendi, Repare RX, Nuvalent, Merus, Chugai Pharmaceutical, Remedica Ltd., mBrace, AXIS, EPG Health, Harborside Nexus, Liberum, RV More, Ology, Amgen, TouchIME, Janssen, Entos, Treeline Bio, Prelude, Applied Pharmaceutical Science, Inc., AiCME, i3 Health, and MonteRosa. He acknowledges research funding from Pfizer, Exelixis, GlaxoSmithKline, Teva, Taiho, and PharmaMar. His patents, royalties, and other intellectual property include Selpercatinib-Osimertinib filed/pending, Wolters Kluwer, Merck, Puma, Merus, and Boehringer Ingelheim. AD is also supported by grants from the National Cancer Institute/National Institutes of Health (P30CA008748, 1R01CA251591001A1, 1R01CA273224-01) and Lungevity.
The remaining author declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The authors declare that this study received funding from Loxo Oncology, which is now called Loxo@Lilly. The funder had the following involvement in the study: collected, analyzed, and interpreted the trial data in collaboration with the authors.
The remaining author declares that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Abbreviations
EGFR, epidermal growth factor receptor; FISH, fluorescence in situ hybridization; H&E, hematoxylin and eosin; MPR, major pathologic response; NSCLC, non-small cell lung cancer; pCR, pathologic complete response; RET, rearrangement during transfection; TEAEs, treatment-emergent adverse events; TKI, tyrosine kinase inhibitor.
References
1. Reyes R, Reguart N. Neoadjuvant treatment of stage IIIA-N2 in EGFR-Mutant/ALK-rearranged non-small cell lung cancer. Transl Lung Cancer Res (2021) 10:607–21. doi: 10.21037/tlcr-20-780
2. Zenke Y, Yoh K, Sakakibara-Konishi J, Daga H, Hosomi Y, Nogami N, et al. P1.18-04 neoadjuvant ceritinib for locally advanced non-small cell lung cancer with ALK rearrangement: SAKULA trial. J Thorac Oncol (2019) 14:S626–7. doi: 10.1016/j.jtho.2019.08.1320
3. Drilon A, Oxnard GR, Tan DSW, Loong HHF, Johnson M, Gainor J, et al. Efficacy of selpercatinib in RET fusion-positive non-small-cell lung cancer. N Engl J Med (2020) 383:813–24. doi: 10.1056/NEJMoa2005653
4. Tsuboi M, Goldman JW, Wu YL, Johnson ML, Paz-Ares L, Yang JC, et al. LIBRETTO-432, a phase III study of adjuvant selpercatinib or placebo in stage IB-IIIA RET fusion-positive non-small-cell lung cancer. Future Oncol (2022) 18:3133–41. doi: 10.2217/fon-2022-0656
5. FDA Approves neoadjuvant nivolumab and platinum-doublet chemotherapy for early-stage non-small cell lung cancer. Available at: https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-neoadjuvant-nivolumab-and-platinum-doublet-chemotherapy-early-stage-non-small-cell-lung (Accessed October 12, 2022).
6. FDA Approves larotrectinib for solid tumors with NTRK gene fusions . Available at: https://www.fda.gov/drugs/fda-approves-larotrectinib-solid-tumors-ntrk-gene-fusions (Accessed October 12, 2022).
7. Radonic T, Geurts-Giele WRR, Samsom KG, Roemen GMJM, Von der Thüsen JH, Thunnissen E, et al. RET fluorescence In situ hybridization analysis is a sensitive but highly unspecific screening method for RET fusions in lung cancer. J Thor Onc (2021) 16(5):798–806. doi: 10.1016/j.jtho.2021.01.1619
8. Baker JA, Sireci AN, Marella N, Cannon HC, Marquart TJ, Holzer TR, et al. Analytical accuracy of RET fusion detection by break-apart fluorescence in situ hybridization. Arch Pathol Lab Med (2022) 146:351–9. doi: 10.5858/arpa.2020-0376-OA
Keywords: RET fusion, selective RET inhibitor, NSCLC, neoadjuvant, major pathologic response, pathologic complete response, case report
Citation: Goldman JW, Sholl LM, Dacic S, Fishbein MC, Murciano-Goroff YR, Rajaram R, Szymczak S, Szpurka AM, Chao BH and Drilon A (2023) Case Report: Complete pathologic response to neoadjuvant selpercatinib in a patient with resectable early-stage RET fusion-positive non-small cell lung cancer. Front. Oncol. 13:1178313. doi: 10.3389/fonc.2023.1178313
Received: 02 March 2023; Accepted: 03 May 2023;
Published: 18 May 2023.
Edited by:
Min Li, Central South University, ChinaReviewed by:
Lorenzo Belluomini, University of Verona, ItalyMelina Elpi Marmarelis, University of Pennsylvania, United States
Copyright © 2023 Goldman, Sholl, Dacic, Fishbein, Murciano-Goroff, Rajaram, Szymczak, Szpurka, Chao and Drilon. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Jonathan W. Goldman, SldHb2xkbWFuQG1lZG5ldC51Y2xhLmVkdQ==
 Jonathan W. Goldman
Jonathan W. Goldman Lynette M. Sholl
Lynette M. Sholl Sanja Dacic
Sanja Dacic Michael C. Fishbein
Michael C. Fishbein Yonina R. Murciano-Goroff
Yonina R. Murciano-Goroff Ravi Rajaram5
Ravi Rajaram5