- 1Institute of Experimental Medicine, Czech Academy of Sciences, Prague, Czechia
- 2Institute of Biology and Medical Genetics, First Faculty of Medicine, Charles University, Prague, Czechia
- 3Biomedical Center, Faculty of Medicine in Pilsen, Charles University, Pilsen, Czechia
- 4Institute of Medical Biometry and Informatics, University of Heidelberg, Heidelberg, Germany
Editorial on the Research Topic
Current understanding of genomic and chromosomal instabilities in solid malignancies
A fundamental precondition of life is the highly precise transfer of genetic information and the preservation of genomic integrity. Hanahan and Weinberg, in their seminal review, had defined the hallmarks of cancer as acquired functional alterations that enable cancer cells to survive, proliferate, and disseminate with the development of genomic instability in cancer cells as a significant prerequisite for malignant transformation. Genomic instability, as defined, includes genetic changes that include mutations, chromosomal rearrangements, and telomerase function (1). The genomic instability in transforming epithelial cells into malignant cells remains paramount in tumorigenesis, as augmented by the accumulated data (2).
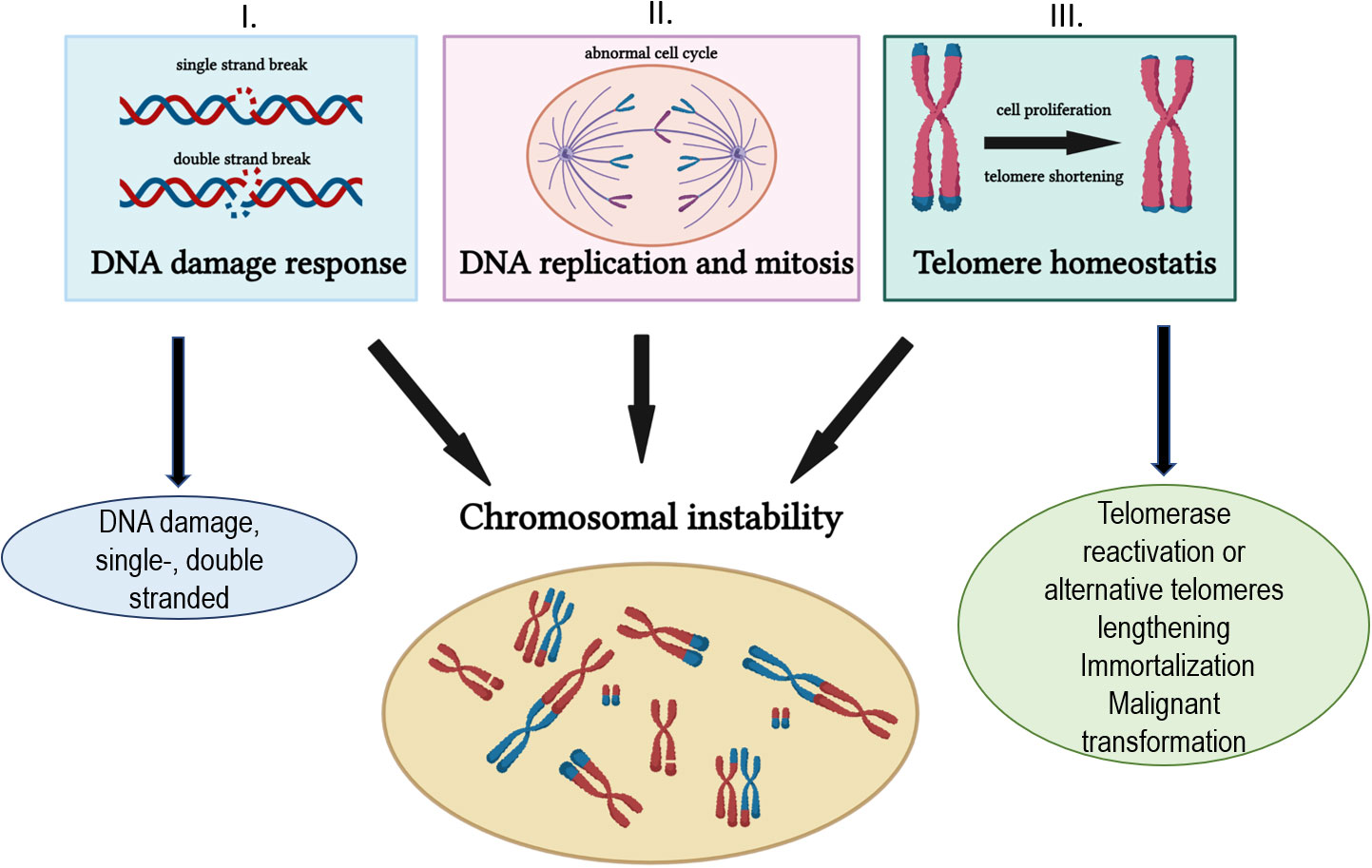
In general, multiple pathways are implicated in the maintenance of genomic integrity and in the prevention of its instability. Those pathways (Figure 1) predominantly include (I) DNA damage response and DNA repair mechanisms, (II) DNA replication and mitosis, and (III) telomere maintenance. The accumulation of various DNA damage due to the altered DNA repair capacity and telomere shortening disrupt the genomic integrity and pave the way to malignant transformation. DNA damage response (DDR), a signaling network that processes DNA damage, is critical for cancer progression and chemotherapy outcomes. DDR, a complex process, that detects DNA lesions and activate signaling pathways, such as cell cycle checkpoint induction, DNA repair, and or induction of cell death (3–5).
In solid cancers, impaired DNA repair, deficiencies in mitotic regulation, and altered telomere length (TL) homeostasis are vital aberrations in cancer initiation, progression, and dissemination. As is generally believed, suboptimal DNA repair results in a broad range of critical mutations, leading to genomic instability. The functional impairment of most DDR processes is widely pervasive in different cancers (6). The familial cancers exhibit high penetrance germline mutations in DNA repair genes: BRCA1/BRCA2 in breast cancer, MMR and polymerase deficiency (MLH1, MSH2, MSH6, PMS2, and POLE genes) in colorectal and ovarian cancers (OC), deleterious mutations in RAD51C and RAD51D and BRCA1 mutation in OC exemplify that paradigm (reviewed by 6). Chromosomal instability (CIN) in cancers often occurs due to improper regulations of mitotic fidelity and faithful chromosome segregation. These processes impact mitosis and its alteration results in dysfunctional centrosomes that trigger and maintain CIN (7). Functional aberrations in the mitotic checkpoint, comprising mutations and changes in gene expression, result in abnormal chromosome content and/or aneuploidy, important players in cancer development. An improper checkpoint response is also involved in developing drug resistance to microtubule poisons employed in treating various solid malignancies for a long time (8).
Telomeres maintain genomic integrity by protecting chromosomal ends (9). However, due to the intrinsic inability to completely replicate lagging DNA strands, telomeres become progressively shortened during successive cell division. Endogenous and exogenous DNA-damaging genotoxicants, including anticancer drugs, also affect the telomere length. Because of increased proliferation, telomeric attrition is considerably faster in tumor cells than in non-malignant somatic cells. Telomere shortening doubles as a robust tumor-suppressing mechanism, limiting the lifespan of cells to prevent uncontrolled growth. On the contrary, cancer cells often develop a mechanism to surpass telomere attrition through telomerase rejuvenation that stabilizes telomeres for continued proliferation (10). The rejuvenated telomerase preferentially stabilizes the shortest telomeres and critically short telomeres, which can lead to the formation of anaphase bridges through breakage–fusion–bridge cycles that contribute to CIN (11). Therefore, ubiquitous in advanced solid cancers, telomerase is fundamental to cell immortalization.
Human solid neoplasms often exhibit structural and numerical chromosomal instability (CIN) (4, 12, 13). CIN creates abnormal aneuploid karyotypes or continually expands phenotypic heterogeneity because of the consecutive cell divisions of tumor cell populations. For instance, CIN studied in colorectal carcinoma disclosed losses predominantly on chromosomes 1p, 5q, 8p, 17p, 18p, 18q, 20p, and 22q, whereas gains mainly were identified on chromosomes 1q, 8q, 12q, 13q, and 20q. These regions are likely to harbor oncogenes or tumor suppressor genes. Several signaling pathways and genes are associated with CIN, including APC, Wnt/β-catenin, p53, TGF-β/SMAD, KRAS, BRAF, and PIK3CA. These pathways are closely linked with the progression and metastasis of colorectal carcinoma (14).
The present Research Topic includes a collection of eight articles based on experimental data and a literature survey of genomic instability in various solid malignancies. Since genomic instability is a complex and heterogeneous process, the authors have logically addressed various aspects, including genetic characteristics, new genetic markers in the process, the role of the p53 family, the complex molecular profile of DNA repair during therapy, the prognostic significance of microsatellite instability and loss of heterozygosity. Furthermore, one study analyzed clinical perspectives of microsatellite instability in metastatic colorectal cancer, and one manuscript reports the process of chromothripsis, catastrophic rearrangements in a limited number of chromosomes, vis-a-vis tumor immunity. The collection of manuscripts indicates the complexity of the topic, and further functional studies in the area are warranted. The topic of genomic instability in cancer remains far from entirely explored and understood, despite the advent of new technologies.
Author contributions
PV and RK are Guest Editors for this Research Topic, they wrote the text, LV and MK contributed with writing, editing and critical reading. All authors contributed to the article and approved the submitted version.
Funding
Supported by the project National Institute for Cancer Research (Programme EXCELES, No. LX22NPO5102) and projects AZV NU21-03-00145, NU21-07-00247, NU22J-03-00028, GAČR: 21-27902S.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. Cell (2011) 144:646–73. doi: 10.1016/j.cell.2011.02.013
2. Senga SS, Grose RP. Hallmarks of cancer-the new testament. Open Biol (2021) 11:200358. doi: 10.1098/rsob.20.0358
3. Tomasova K, Kroupa M, Forsti A, Vodicka P, Vodickova L. Telomere maintenance in interplay with DNA repair in pathogenesis and treatment of colorectal cancer. Mutagenesis (2020) 35(3):261–71. doi: 10.1093/mutage/geaa005
4. Vodicka P, Andera L, Opattova A, Vodickova L. The interaction of DNA repair, telomere homeostasis, and p53 mutational status in solid cancers: risk, prognosis and prediction. Cancers (Basel) (2021) 13(3):479. doi: 10.3390/cancers13030479
5. Vodicka P, Vodenkova S, Opattova A, Vodickova L. DNA damage and repair measured by comet assay in cancer patients. Mutat Res Gen Tox En (2019) 843:95–110. doi: 10.1016/j.mrgentox.2019.05.009
6. Pearl LH, Schierz AC, Ward SE, Al-Lazikani B, Pearl FM. Therapeutic opportunities within the DNA damage response. Nat Rev Cancer (2015) 15:166–80. doi: 10.1038/nrc3891
7. Piemonte KM, Anstine LJ, Keri RA. Centrosome aberrations as drivers of chromosomal instability in breast cancer. Endocrinology (2021) 162(12):bqab208. doi: 10.1210/endocr/bqab208
8. Sarkar S, Sahoo PK, Mahata S, Pal R, Ghosh D, Mistry T, et al. Mitotic checkpoint defects: en route to cancer and drug resistance. Chromosome Res (2021) 29(2):131–44. doi: 10.1007/s10577-020-09646-x
9. Srinivas N, Rachakonda S, Kumar R. Telomeres and telomere length: A general overview. Cancers (Basel) (2020) 12(3):558. doi: 10.3390/cancers12030558
10. Heidenreich B, Kumar R. TERT promoter mutations in telomere biology. Mutat Res Rev Mutat Res (2017) 771:15–31. doi: 10.1016/j.mrrev.2016.11.002
11. Kroupa M, Rachakonda SK, Liska V, Srinivas N, Urbanova M, Jiraskova K, et al. Relationship of telomere length in colorectal cancer patients with cancer phenotype and patient prognosis. Br J Cancer (2019) 121(4):344–50. doi: 10.1038/s41416-019-0525-3
12. Hemminki K, Rachakonda S, Musak L, Vymetalkova V, Halasova E, Försti A, et al. Telomere length in circulating lymphocytes: association with chromosomal aberrations. Genes Chromosomes Cancer (2015) 54(3):194–6. doi: 10.1002/gcc.22225
13. Wang H, Ni J, Guo X, Xue J, Wang X. Effects of folate on telomere length and chromosome stability of human fibroblasts and melanoma cells in vitro: A comparison of folic acid and 5-methyltetrahydrofolate. Mutagenesis (2023) 38(3):160–8. doi: 10.1093/mutage/gead007
Keywords: DNA damage response, mitotic regulation, telomere homeostasis, solid malignancies, genomic instability
Citation: Vodicka P, Kroupa M, Vodickova L and Kumar R (2023) Editorial: Current understanding of genomic and chromosomal instabilities in solid malignancies. Front. Oncol. 13:1245087. doi: 10.3389/fonc.2023.1245087
Received: 23 June 2023; Accepted: 09 August 2023;
Published: 24 August 2023.
Edited and Reviewed by:
Claudio Sette, Catholic University of the Sacred Heart, Rome, ItalyCopyright © 2023 Vodicka, Kroupa, Vodickova and Kumar. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Pavel Vodicka, cGF2ZWwudm9kaWNrYUBpZW0uY2FzLmN6
 Pavel Vodicka
Pavel Vodicka Michal Kroupa
Michal Kroupa Ludmila Vodickova
Ludmila Vodickova Rajiv Kumar
Rajiv Kumar