- Department of Thoracic Surgery, Beijing Aerospace General Hospital, Beijing, China
Objectives: The purpose of this study was to assess the association between preoperative neutrophil-to-lymphocyte ratio (NLR) and the survival outcomes of esophageal cancer patients who underwent esophagectomy, the latest and comprehensive systematic review performed.
Methods: Related literature retrieved from PubMed, Web of Science, Embase, and Cochrane before January 2024, according to the inclusion criteria. Outcomes measured were overall survival (OS), disease-free survival (DFS), relapse-free survival (RFS), and cancer-specific survival (CSS).
Results: Eighteen studies with 6,119 esophageal cancer patients were retained for analysis. Meta-analysis demonstrated that OS (HR: 1.47; 95% CI: 1.29, 1.67; P < 0.00001), DFS (HR: 1.62; 95% CI: 1.29, 2.05; P < 0.0001), and CSS (HR: 1.62; 95% CI: 1.29, 2.05; P < 0.0001) were significantly shorter in the high NLR group compared with the low NLR group. In addition, meta-analysis revealed a similar RFS (HR: 1.47; 95% CI: 0.92, 2.35; P = 0.10) among the two groups. Subgroup analysis of OS and DFS based on mean/median age, NLR cutoff, and region found that all subgroups remained significant difference between two groups.
Conclusion: Among esophageal cancer patients who underwent esophagectomy, preoperative NLR can be used as prognostic factor independently. High-preoperative NLR is associated with poor prognosis. More large-scale, multicenter prospective clinical studies are needed to further validate the relationship between preoperative NLR and prognosis of esophageal cancer.
Introduction
As an increasingly global health problem, esophageal cancer significantly affects human life and survival. Based on the latest data, esophageal cancer ranks the sixth among cancer-related causes of death across the world (1), resulting in over 500,000 cancer deaths annually. With a 60-fold difference between high- and low-incidence regions, esophageal cancer demonstrates a significantly varied distribution, contributing to 5.3% of all cancer-related deaths globally (2, 3). Surgery is currently the preferred treatment for patients with esophageal cancer, often supplemented by adjuvant chemoradiotherapy or immune and targeted therapy (4). However, the overall prognosis and life quality of esophageal cancer patients are poor, with a long-term survival rate of less than 25%, largely because symptoms or signs typically appear when the disease has already advanced to a late stage (5, 6). Esophageal cancer patients experiencing poor prognosis primarily due to the hidden symptoms experienced by early stage patients, the absence of clear tumor specific markers in clinical practice, and the lack of specific and sensitive screening programs (7, 8). Therefore, the prognosis of esophageal cancer patients is unfavorable, and there is an urgent demand for specific indicators capable of predicting tumor occurrence and prognosis (9).
Since Virchow first proposed the potential correlation between inflammation and tumors in the 19th century (10), more and more epidemiological data and molecular biology laboratory tests have revealed the close relationship between the tumor microenvironment and tumor development. C-reactive protein, neutrophils, and lymphocytes within the tumor microenvironment can induce cellular oxidative stress, DNA mutations, proliferation, invasion, metastasis, and other associated reactions. This process ultimately fosters tumor initiation and progression, thereby influencing patient prognosis (11–14). Emerging studies on inflammatory complex indexes, such as the neutrophil-to-lymphocyte ratio (NLR), are shedding light on their correlation with tumor occurrence, proliferation, invasion, and prognosis as research progresses (15–17). Several studies have confirmed the significant predictive value of an elevated NLR before surgery or chemoradiotherapy in solid tumors, such as gastric cancer (18), lung cancer (19), breast cancer (20), liver cancer (21), pancreatic cancer (22), and kidney cancer (23). Among them, Nakamura et al. (24) showed that high NLR in patients with esophageal cancer predicted poor survival outcomes. Changes in inflammatory response and immune system regulation may improve the survival outcome of esophageal cancer patients. NLR is a simple and low-cost independent prognostic marker (24). The study of Sharaiha et al. (25) also found that NLR reflected the systemic inflammatory response generated by tumors and affected the invasive and metastatic tendency of tumors. Numerous studies have demonstrated the association of NLR with the effectiveness and prognosis of radiotherapy, chemotherapy, immunotherapy, and targeted therapy for esophageal cancer (26–28).
Although numerous studies reported the value of NLR in predicting esophageal cancer patients’ prognosis (29–35), there is no conclusive evidence to confirm whether preoperative NLRs are significantly associated with the long-term prognosis of esophageal cancer patients underwent esophagectomy due to differences in the optimal cutoff, esophageal cancer type, detection timing, and other factors between studies. Therefore, the study aimed at exploring the potential association of preoperative NLR with the long-term prognosis after radical surgery for esophageal cancer through systematic literature search and meta-analysis and to provide evidence-based medical evidence for the construction of accurate prediction models for patients with esophageal cancer.
Methods
Literature search
This meta-analysis was performed according to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analysis) 2020 statement (36) and has been prospectively registered in the PROSPERO (CRD42024524824). We conducted a systematic literature search via PubMed, Embase, Web of Science, and Cochrane up to January 2024 for studies that evaluated the prognostic value of preoperative NLR for the survival outcomes of patients with esophageal cancer. We searched the literature through the following terms: “esophageal neoplasms,” “lymphocytes,” and “neutrophil.” The detailed search strategies are as follows: ((((“Neutrophils”[Mesh]) OR ((((((((((Neutrophil) OR (Polymorphonuclear Neutrophils)) OR (Neutrophil, Polymorphonuclear)) OR (Polymorphonuclear Neutrophil)) OR (LE Cells)) OR (Cell, LE)) OR (LE Cell)) OR (Neutrophil Band Cells)) OR (Band Cell, Neutrophil)) OR (Neutrophil Band Cell))) AND ((“Lymphocytes”[Mesh]) OR (((((Lymphocyte) OR (Lymphoid Cells)) OR (Cell, Lymphoid)) OR (Cells, Lymphoid)) OR (Lymphoid Cell)))) AND (ratio)) AND ((“Esophageal Neoplasms”[Mesh]) OR (((((((((Esophageal Neoplasm) OR (Esophagus Neoplasm)) OR (Esophagus Neoplasms)) OR (Cancer of Esophagus)) OR (Cancer of the Esophagus)) OR (Esophagus Cancer)) OR (Esophagus Cancers)) OR (Esophageal Cancer)) OR (Esophageal Cancers))). Furthermore, we manually screened the bibliography lists of all included studies. Two authors (X.W. and S.J.L.) retrieved and assessed eligible articles independently. Any differences in literature retrieval were resolved by discussion.
Inclusion and exclusion criteria
Articles were eligible when meeting the following standards: (1) study design was randomized controlled trial, cohort, or case control; (2) studies were performed in patients with esophageal cancer; (3) studies evaluated the prognostic value of preoperative NLR for the survival outcomes of patients with esophageal cancer; (4) at least one survival outcome [overall survival (OS), disease-free survival (DFS), relapse-free survival (RFS), progression-free survival (PFS), cancer-specific survival (CSS), etc.] was evaluated; (5) complete data to analyze risk ratio (RR), odds ratio (OR), or hazard ratio (HR). We excluded study protocols, unpublished studies, non-original studies (including letters, comments, abstracts, correction, and reply), studies without sufficient data, and reviews.
Data abstraction
Data abstraction was conducted by two authors (X.W. and S.J.L.) severally. Any differences were settled by another author (F.W.L.). The following information extracted from eligible studies: first author name, published year, study duration, study country, study design, research population, sample size, age, gender, TNM stage, NLR cutoff, HR for OS, DFS, RFS, and CSS. If the research data were insufficient, corresponding authors were contacted to request complete data, if available.
Quality evaluation
The Newcastle-Ottawa Scale (NOS) was applied for assessing the quality of included cohort studies (37), and studies with 7–9 points were considered as high quality (38). Studies with NOS scores below 7 were not included for quantitative analysis. Two authors (X.W. and S.J.L.) severally assessed the quality of all included studies, and any disagreement was settled by discussion.
Statistical analysis
Meta-analysis was conducted by using Review Manager 5.4.1. HR was used for the synthesis of survival data. Metrics were presented with 95% confidential intervals (CIs). The chi-squared (χ2) test (Cochran’s Q) and inconsistency index (I2) were applied for the evaluation of the heterogeneity (39). χ2 p < 0.1 or I2 more than 50% were regarded as high heterogeneity. Random-effects model was applied to calculate the total HR for outcomes with significant heterogeneity (χ2 p < 0.1 or I2 more than 50%). Or else, fixed-effects model used. In addition, subgroup analyses performed for outcomes with five or more studies included to evaluate the possible confounders, if data were sufficient. In addition, sensitivity analysis was conducted to assess the influence of every included study on the total HR for results with three or more studies included. Egger’s regression tests and funnel plots (40) performed by Review Manager 5.4.1 and Stata 15.1 (Stata Corp, College Station, Texas, USA) to evaluate the publication bias. p < 0.05 represents statistically significant publication bias.
Results
Literature retrieval, study characteristics, and baseline
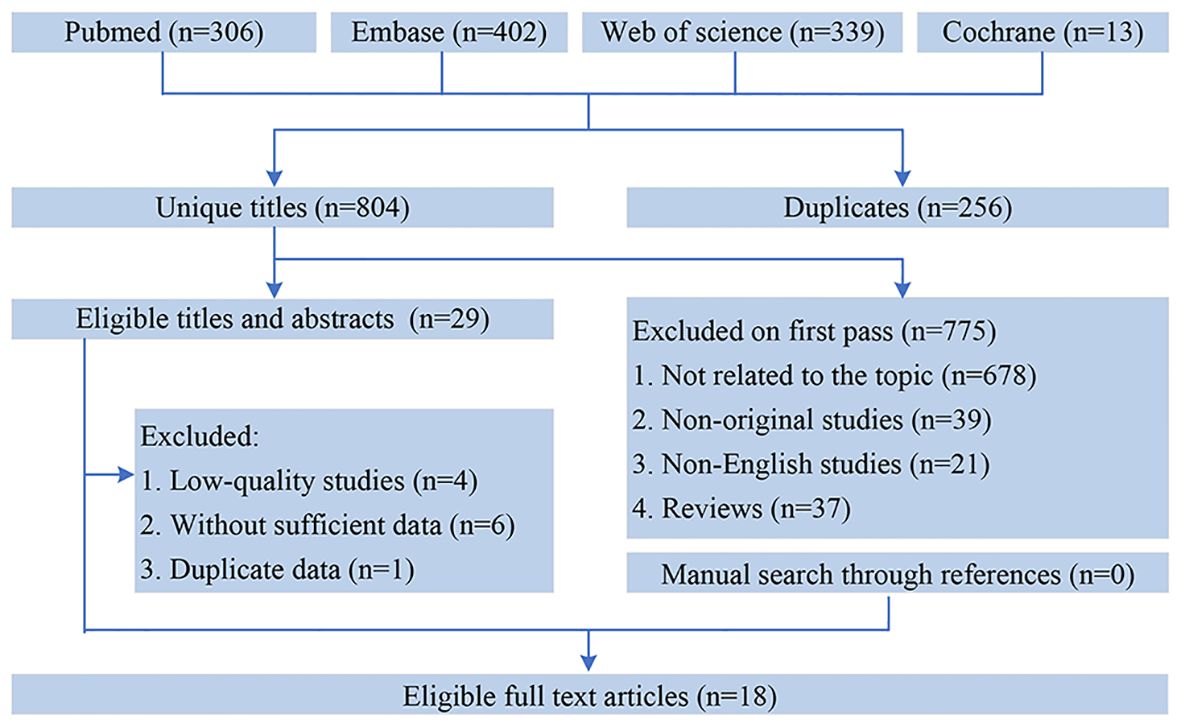
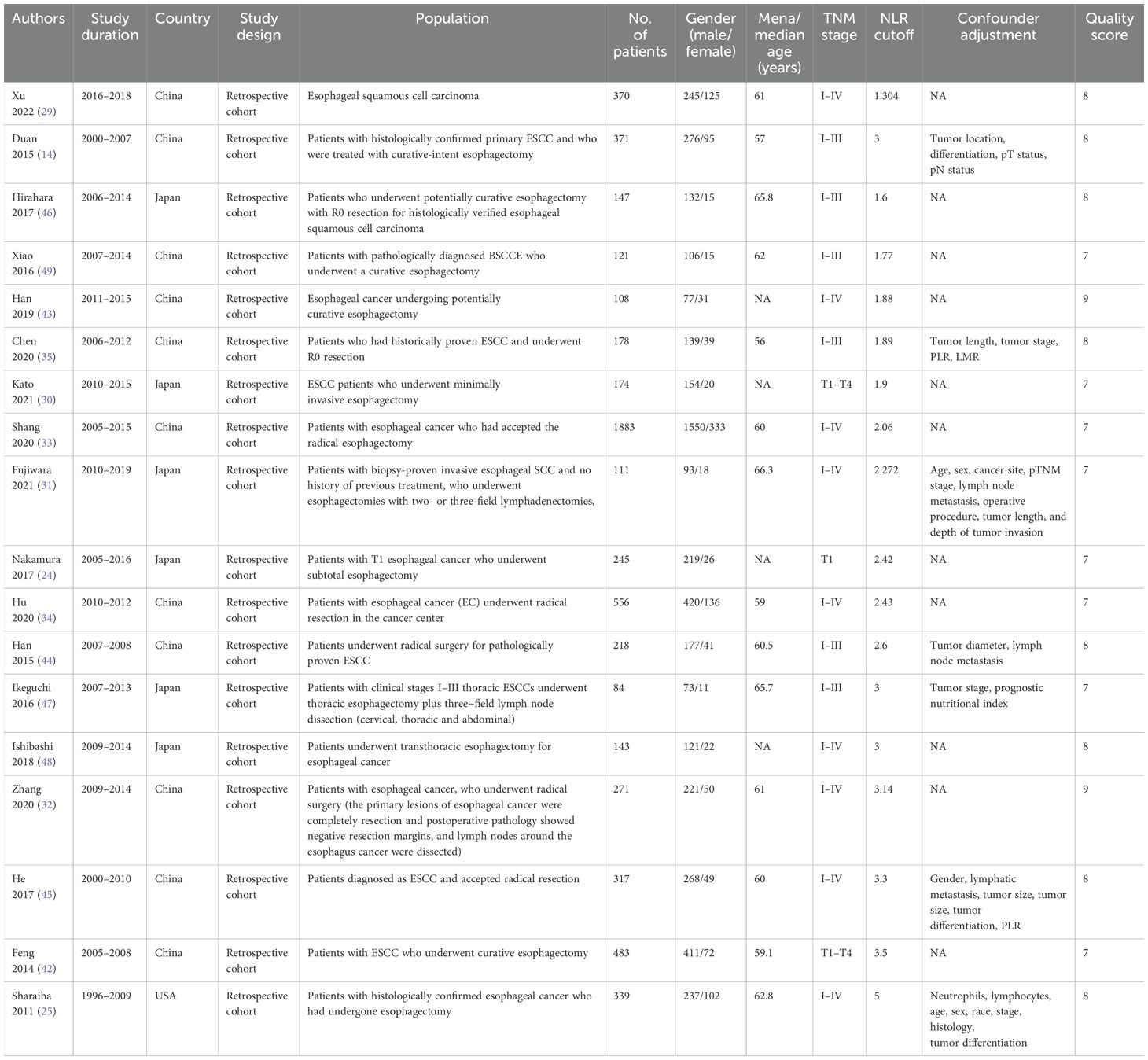
Figure 1 showed the flowchart of the literature retrieval and selection process. A total of 1,060 related studies in PubMed (n = 306), Embase (n = 402), Web of Science (n = 339), and Cochrane (n = 13) were identified. A total of 804 literature retained after removing duplicate studies. Eventually, 18 retrospective cohort studies with 6,119 patients were included (24, 25, 29–35, 41–49). Table 1 presents the characteristics and quality evaluation of each eligible cohort study.
OS
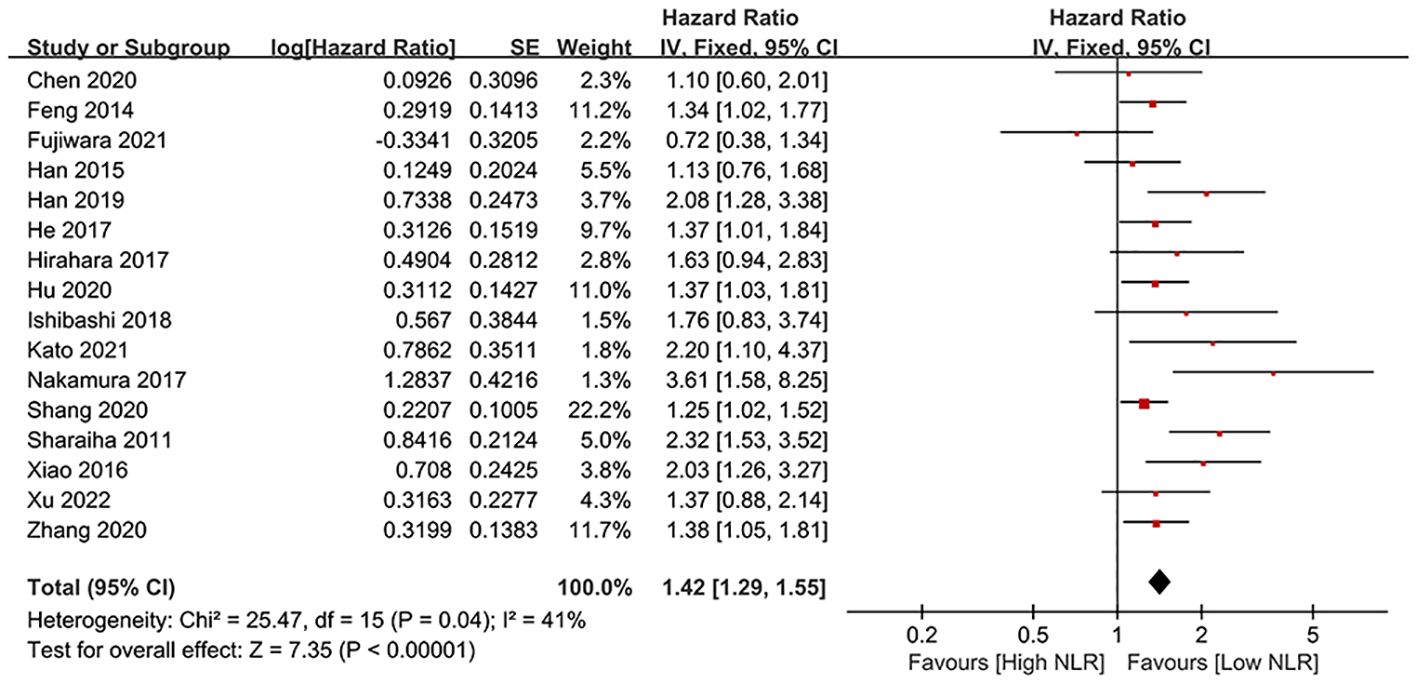
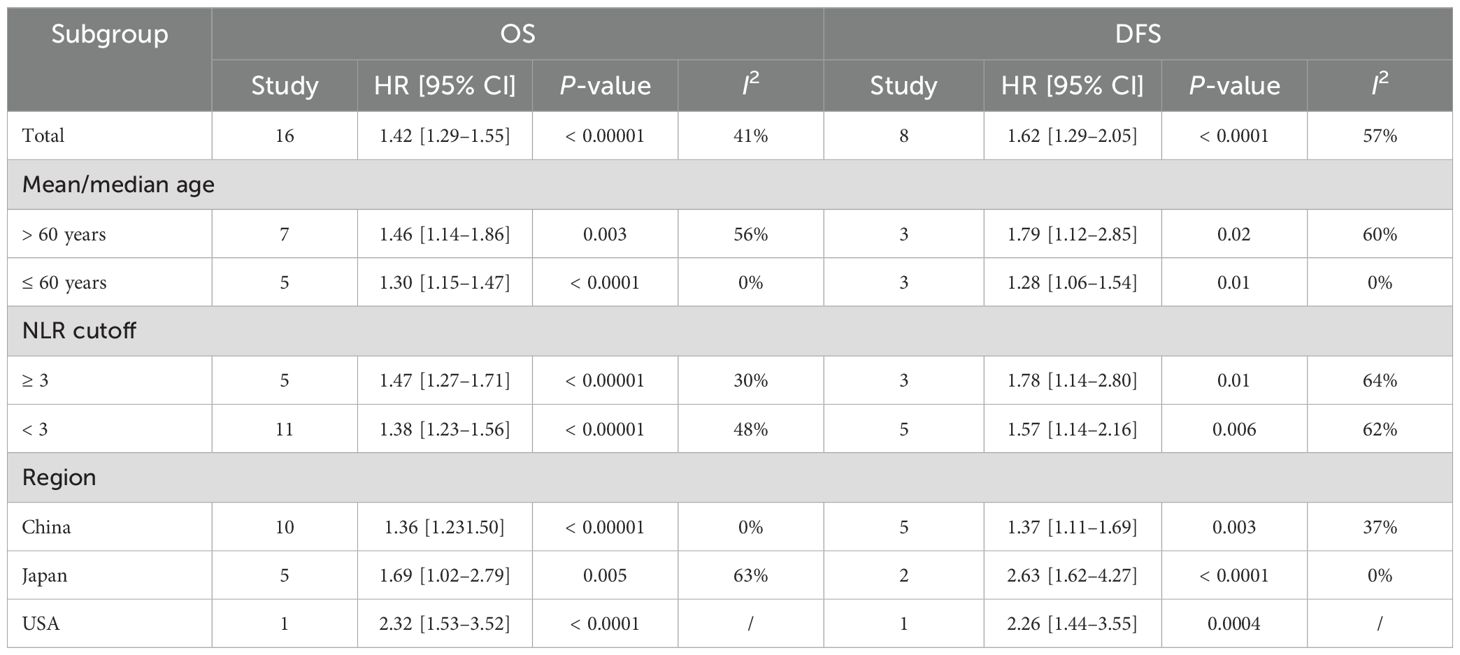
Results of OS were synthesized from 16 cohort studies (24, 25, 29–35, 42–46, 48, 49), and meta-analysis revealed a significantly shorter OS in high NLR group (HR: 1.47; 95% CI: 1.29, 1.67; P < 0.00001). There was no significant heterogeneity (I2 = 41%, P = 0.04) (Figure 2). Subgroup analysis based on mean/median age, NLR cutoff and region revealed that difference remained significant in all subgroups (Table 2).
DFS
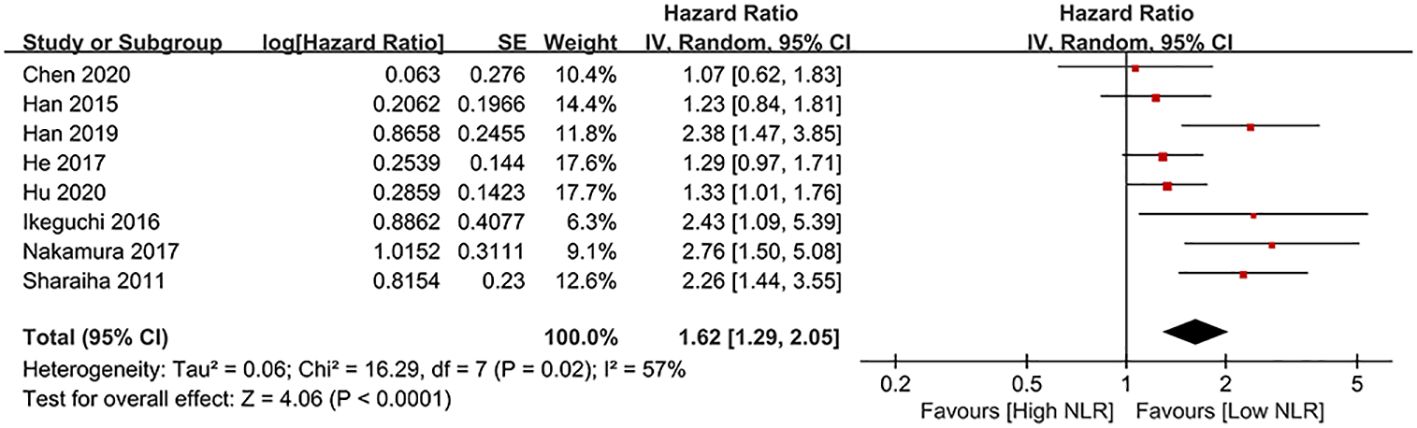
Results of DFS were synthesized from eight cohort studies (24, 25, 34, 35, 43–45, 47), and exhibited a significantly shorter DFS in high NLR group (HR: 1.62; 95% CI: 1.29, 2.05; P < 0.0001). Significant heterogeneity exists (I2 = 57%, P = 0.02) (Figure 3). Subgroup analysis based on mean/median age, NLR cutoff and region showed significant difference in all subgroups (Table 2).
RFS
Results of RFS were synthesized from three cohort studies (31, 41, 49), and meta-analysis revealed similar RFS exists in all groups (HR: 1.47; 95% CI: 0.92, 2.35; P = 0.10). Significant heterogeneity shown (I2 = 68%, P = 0.04) (Figure 4).
CSS
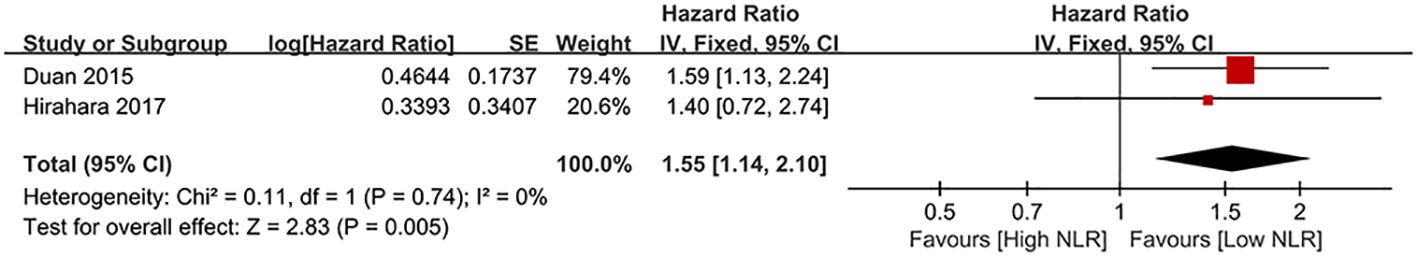
Results of CSS were synthesized from two cohort studies (41, 46), and meta-analysis found a significantly shorter CSS in high NLR group (HR: 1.62; 95% CI: 1.29, 2.05; P < 0.0001). No significant heterogeneity (I2 = 0%, P = 0.74) (Figure 5).
Publication bias and sensitivity analysis
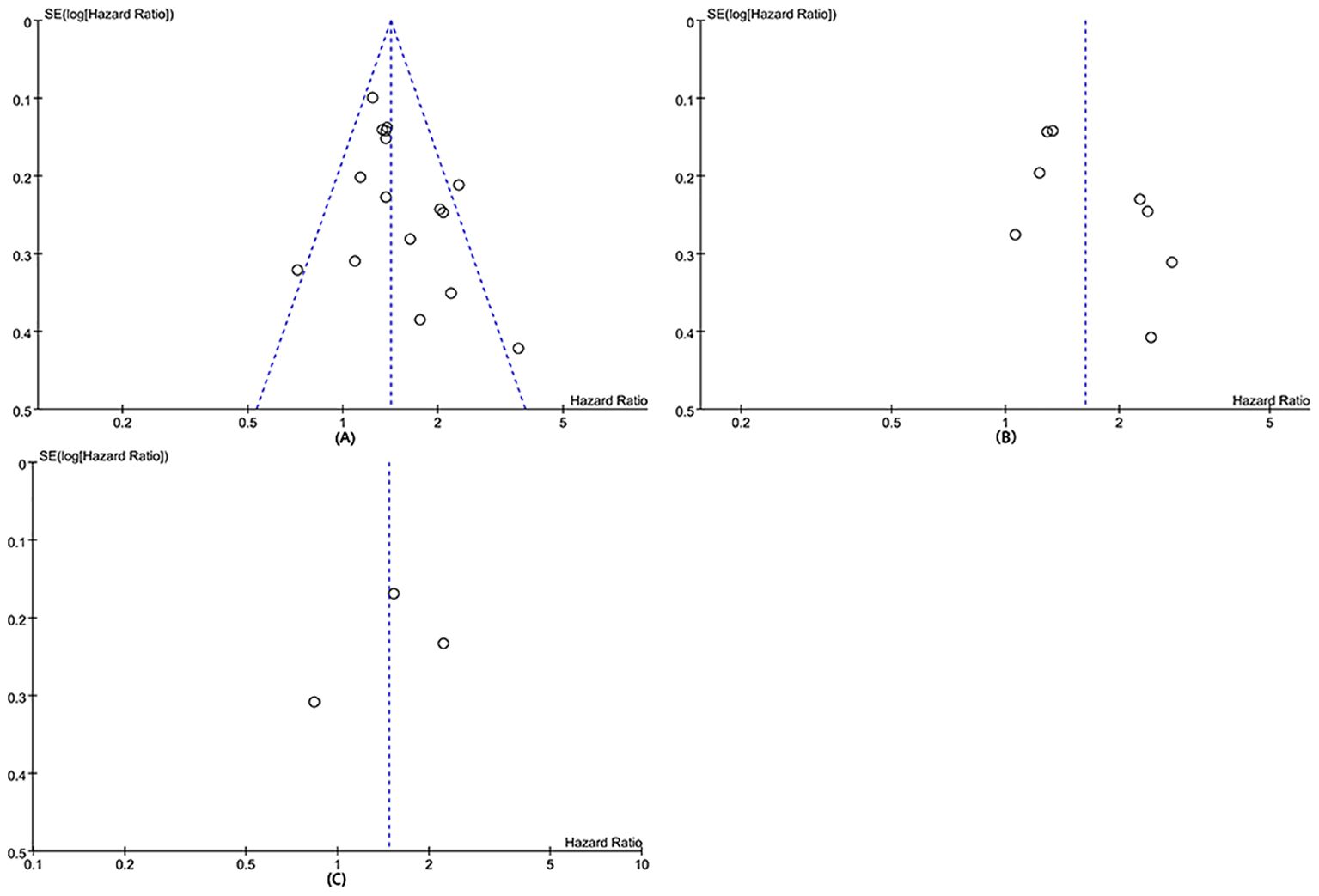
The potential publication bias of OS, DFS, RFS were assessed through funnel plots and Egger’s regression tests. No publication bias existed in OS (Egger’s test P = 0.098) (Figure 6A), DFS (Egger’s test P = 0.093) (Figure 6B), and RFS (Egger’s test P = 0.708) (Figure 6C). In addition, sensitivity analysis was used to assess the effect of each cohort study on the total HR. By excluding eligible studies one by one, the sensitivity analysis of OS, DFS, and RFS was performed.
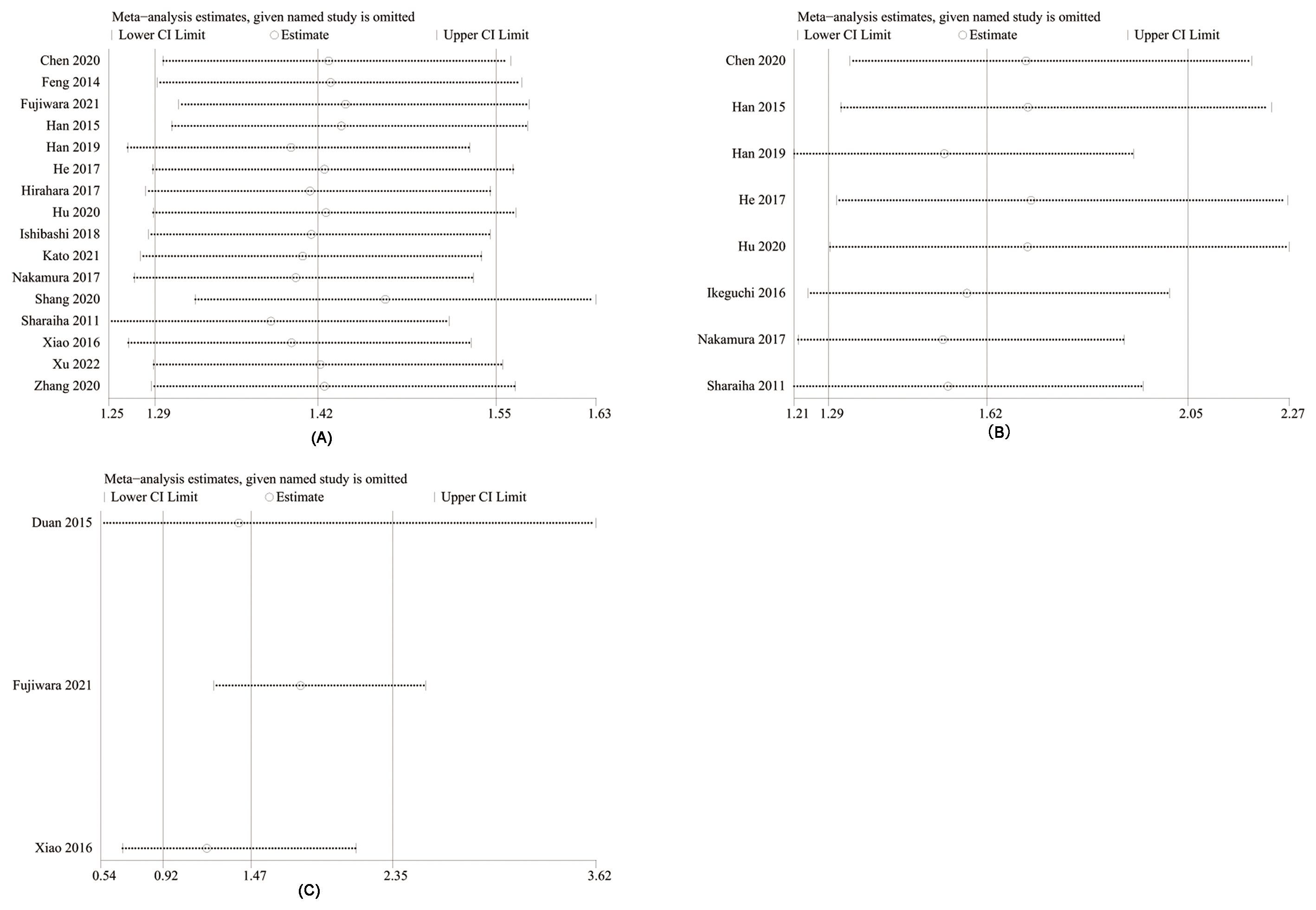
Sensitivity analysis exhibited that total HR kept stable after removing of each cohort study for OS (Figure 7A), DFS (Figure 7B), and RFS (Figure 7C), which implied the low sensitivity and stability of results.
Discussion
At present, the comprehensive treatment method mainly based on the radical resection, which is generally applied for patients occurred esophageal cancer (50). Several studies reported notable survival rate and recurrence disparities among esophageal cancer patients across different stages, with stages I and II patients displaying significantly superior 5-year postoperative survival rates compared to stages III and IV patients (51, 52). Therefore, the selection of effective indicators for preoperative clinical staging diagnosis, postoperative recurrence, and metastasis assessment of patients with esophageal cancer is conducive to formulating the best treatment for patients and intervention programs for high-risk groups of recurrence and metastasis. More attention focused on the relationship between NLR and tumor occurrence, development, prognosis, with the deepening researches on tumor and tumor microenvironment. Studies have proved that the higher level of preoperative NLR predicts, the worse prognosis (53–56). Dong et al. (57) found that the OS in patients with high-preoperative NLR was significantly shorter than the low, and CSS in patients with high NLR was also significantly shortened. Multivariate Cox models implied that preoperative NLR was affected by OS and CSS, after adjusting the clinical and pathological features. Meanwhile, clinical and pathological characteristics have no effect on the prognosis of esophageal cancer patients (43). Therefore, NLR can be used as an independent prognostic factor in esophageal cancer patients.
Here, several survival outcomes analyzed for assessing the value of preoperative NLR among patients occurred esophageal cancer. Our results revealed a significantly shorter OS, DFS, and CSS in high NLR group. In addition, subgroup analysis of OS and DFS based on mean/median age, NLR cutoff and region reveled significant difference exists in all subgroups, suggesting that preoperative NLR was significantly associated with the prognosis of esophageal cancer patients underwent esophagectomy, more attention should be paid to the clinical treatment of esophageal cancer. Our findings support most of the previously published research (57–59). It is worth mentioning that whether tumor stage affects the prognostic value of NLR for esophageal cancer is still controversial. The Global Esophageal Cancer Collaboration provided data on 22,654 patients with epithelial esophageal cancer. Of these, 13,300 patients who had not been treated before surgery were pathologically evaluated after esophagectomy or endoscopic therapy. Risk-adjusted survival was calculated for each patient using randomized survival forest analysis to identify relevant pathological stage groups, where survival decreased as the group increased, with significant differences between groups and homogeneity within groups. Although pTis had similar survival rates to stage pT1N0M0 cancer, it was placed in stage 0 alone. Thus, the final 0–IV stage is obtained with different stages and different prognoses (60). Studies have shown that preoperative NLR is significantly correlated with preoperative treatment and T stage (P = 0.0005 and P < 0.0001, respectively), but preoperative NLR is not significantly correlated with tumor stage (P = 0.16) or other evaluation characteristics (61, 62). In addition, a number of studies have shown that preoperative NLR level has no significant correlation with T stage and N stage, and the preoperative NLR level is not affected by T stage and N stage (31, 35, 43). In addition, another study showed that the impact of prognostic outcomes in patients with esophageal cancer does not change based on clinical and pathological features (43). In conclusion, most of the existing evidence supports that NLR can be used as an independent prognostic factor for patients with esophageal cancer, and its predictive value is not affected by confounding factors such as tumor stage. The cost of obtaining this parameter is relatively low; it is easy to evaluate in most medical centers and as an objective indicator, the bias caused by NLR is relatively limited between laboratories.
Lymphocytes play a vital role in inhibiting the proliferation, metastasis, cytotoxicity, and cell death of tumor cells, while neutrophils can produce cell growth factors, chemokines, and proteases, which can promote the growth of tumor cells. It can promote the proliferation and differentiation of tumor cells by remodeling extracellular matrix framework and inhibiting immune function (63, 64). Therefore, NLR is a balance index reflecting the anti-tumor immunity, tumor cell growth, and inflammation (65, 66). The tumors invasion into lymph nodes can result in the loss of immune function, less lymphocytes, and higher NLR value (29). This study showed that the OS, DFS, and CSS of the high NLR group were significantly shorter than the low. Studies have shown that NLR affects the prognosis of esophageal cancer patients significantly because tumor-associated neutrophils contain and secrete factors. These factors, such as human leukocyte elastase, peroxidase, matrix metalloproteinase, heparin-binding growth factor, and vascular permeability factor can promote angiogenesis and inhibit cell adsorption (65). Therefore, the increase of tumor-associated neutrophils was positively correlated with tumor progression. Increased tumor-associated neutrophils can inhibit the anticancer activity of lymphocytes, natural killer cells, and activated T cells (3, 67). Finally, progressive decline in nutritional status, biological function, and other adverse events resulted from systemic inflammatory responses among patients with cancer (68). Therefore, high NLR may have an indirect adverse effect on tumor patients, resulting in adverse patient outcomes.
To some extent, this study has revealed the significance of NLR, a commonly used hematological index in clinical practice, for the prognosis of esophageal cancer patients, but there are still some limitations. First, there was no standard method of electing and calculating the NLR best truncation value currently, and the common methods are ROC curve and median. Due to the different calculation methods of the NLR truncation values in the literatures included, the difference between the number of cases and the NLR truncation values were large, resulting in a certain degree of heterogeneity in the results. Second, although the subjects included were all patients with esophageal cancer, the basic characteristics, clinical stage, pathological type, degree of tumor invasion, and postoperative treatment of patients with esophageal cancer in each literature were different. Due to the relatively small sample size, all confounding factors could not be taken into account in the subgroup analysis. Moreover, the studies were all retrospective studies with a relatively small sample size, and most of them are single-center data from Asian countries (mainly China and Japan), which leads to a certain degree of unavoidable bias.
Under the situation, this meta-analysis is the latest and largest study to report the prognostic value of preoperative NLR for the survival outcomes of esophageal cancer patients underwent esophagectomy. The findings of this study support the importance of paying attention to the level changes of NLR in the clinical treatment of esophageal cancer patients underwent esophagectomy and building a more valuable prediction model based on inflammatory indicators including NLR to improve the prognosis and life quality of esophageal cancer patients.
Conclusion
Meta-analysis demonstrated that preoperative NLR can be used as an independent prognostic factor among esophageal cancer patients underwent esophagectomy. High-preoperative NLR is associated with poor prognosis in patients with esophageal cancer. Considering the limitations of retrospective studies, potential selection bias, and unavoidable heterogeneity, more large-scale, multicenter prospective clinical studies are needed to further validate the relationship between preoperative NLR and prognosis of esophageal cancer.
Author contributions
XW: Conceptualization, Data curation, Formal analysis, Methodology, Resources, Software, Validation, Visualization, Writing – original draft, Writing – review & editing. SL: Conceptualization, Data curation, Formal analysis, Methodology, Software, Validation, Writing – review & editing. FL: Conceptualization, Data curation, Formal analysis, Methodology, Software, Writing – review & editing. YC: Conceptualization, Funding acquisition, Investigation, Project administration, Resources, Supervision, Writing – review & editing.
Funding
The author(s) declare that no financial support was received for the research, authorship, and/or publication of this article.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Bray F, Laversanne M, Sung H, Ferlay J, Siegel RL, Soerjomataram I, et al. Global cancer statistics 2022: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. (2024) 74:229–63. doi: 10.3322/caac.21834
2. Song Q, Yang W, Meng Z, Wang J. Protocol for a systematic review and meta-analysis of Kang-ai injection for patients with oesophageal cancer. Med (Baltimore). (2020) 99:e22148. doi: 10.1097/MD.0000000000022148
3. Uhlenhopp DJ, Then EO, Sunkara T, Gaduputi V. Epidemiology of esophageal cancer: update in global trends, etiology and risk factors. Clin J Gastroenterol. (2020) 13:1010–21. doi: 10.1007/s12328-020-01237-x
4. Kato H, Nakajima M. Treatments for esophageal cancer: a review. Gen Thorac Cardiovasc Surg. (2013) 61:330–5. doi: 10.1007/s11748-013-0246-0
5. Domper Arnal MJ, Ferrández Arenas Á, Lanas Arbeloa Á. Esophageal cancer: Risk factors, screening and endoscopic treatment in Western and Eastern countries. World J Gastroenterol. (2015) 21:7933–43. doi: 10.3748/wjg.v21.i26.7933
6. Smyth EC, Lagergren J, Fitzgerald RC, Lordick F, Shah MA, Lagergren P, et al. Oesophageal cancer. Nat Rev Dis Primers. (2017) 3:17048. doi: 10.1038/nrdp.2017.48
7. Morgan E, Soerjomataram I, Rumgay H, Coleman HG, Thrift AP, Vignat J, et al. The global landscape of esophageal squamous cell carcinoma and esophageal adenocarcinoma incidence and mortality in 2020 and projections to 2040: new estimates from GLOBOCAN 2020. Gastroenterology. (2022) 163:649–658.e2. doi: 10.1053/j.gastro.2022.05.054
8. Thrift AP. Global burden and epidemiology of Barrett oesophagus and oesophageal cancer. Nat Rev Gastroenterol Hepatol. (2021) 18:432–43. doi: 10.1038/s41575-021-00419-3
9. Shah MA, Altorki N, Patel P, Harrison S, Bass A, Abrams JA. Improving outcomes in patients with oesophageal cancer. Nat Rev Clin Oncol. (2023) 20:390–407. doi: 10.1038/s41571-023-00757-y
10. Balkwill F, Mantovani A. Inflammation and cancer: back to Virchow? Lancet. (2001) 357:539–45. doi: 10.1016/S0140-6736(00)04046-0
11. Lin EW, Karakasheva TA, Hicks PD, Bass AJ, Rustgi AK. The tumor microenvironment in esophageal cancer. Oncogene. (2016) 35:5337–49. doi: 10.1038/onc.2016.34
12. Shen S, Gong J, Yang Y, Qin S, Huang L, She S, et al. Molecular mechanism of C-reaction protein in promoting migration and invasion of hepatocellular carcinoma cells in vitro. Int J Oncol. (2017) 50:1289–98. doi: 10.3892/ijo.2017.3911
13. Singh R, Mishra MK, Aggarwal H. Inflammation, immunity, and cancer. Mediators Inflammation. (2017) 2017:6027305. doi: 10.1155/2017/6027305
14. Wang L, Han H, Wang Z, Shi L, Yang M, Qin Y. Targeting the microenvironment in esophageal cancer. Front Cell Dev Biol. (2021) 9:684966. doi: 10.3389/fcell.2021.684966
15. Cupp MA, Cariolou M, Tzoulaki I, Aune D, Evangelou E, Berlanga-Taylor AJ. Neutrophil to lymphocyte ratio and cancer prognosis: an umbrella review of systematic reviews and meta-analyses of observational studies. BMC Med. (2020) 18:360. doi: 10.1186/s12916-020-01817-1
16. Proctor MJ, McMillan DC, Morrison DS, Fletcher CD, Horgan PG, Clarke SJ. A derived neutrophil to lymphocyte ratio predicts survival in patients with cancer. Br J Cancer. (2012) 107:695–9. doi: 10.1038/bjc.2012.292
17. Sharma N, Jha S. NLR-regulated pathways in cancer: opportunities and obstacles for therapeutic interventions. Cell Mol Life Sci. (2016) 73:1741–64. doi: 10.1007/s00018-015-2123-8
18. Nikolić I, Kukulj S, Samaržija M, Jeleč V, Žarak M, Orehovec B, et al. Neutrophil-to-lymphocyte and platelet-to-lymphocyte ratio help identify patients with lung cancer, but do not differentiate between lung cancer subtypes. Croat Med J. (2016) 57:287–92. doi: 10.3325/cmj.2016.57.287
19. Ethier JL, Desautels D, Templeton A, Shah PS, Amir E. Prognostic role of neutrophil-to-lymphocyte ratio in breast cancer: a systematic review and meta-analysis. Breast Cancer Res. (2017) 19:2. doi: 10.1186/s13058-016-0794-1
20. Hirahara T, Arigami T, Yanagita S, Matsushita D, Uchikado Y, Kita Y, et al. Combined neutrophil-lymphocyte ratio and platelet-lymphocyte ratio predicts chemotherapy response and prognosis in patients with advanced gastric cancer. BMC Cancer. (2019) 19:672. doi: 10.1186/s12885-019-5903-y
21. Zheng J, Cai J, Li H, Zeng K, He L, Fu H, et al. Neutrophil to lymphocyte ratio and platelet to lymphocyte ratio as prognostic predictors for hepatocellular carcinoma patients with various treatments: a meta-analysis and systematic review. Cell Physiol Biochem. (2017) 44:967–81. doi: 10.1159/000485396
22. Iwai N, Okuda T, Sakagami J, Harada T, Ohara T, Taniguchi M, et al. Neutrophil to lymphocyte ratio predicts prognosis in unresectable pancreatic cancer. Sci Rep. (2020) 10:18758. doi: 10.1038/s41598-020-75745-8
23. Cordeiro MD, Ilario EN, Abe DK, Carvalho PA, Muniz DQB, Sarkis AS, et al. Neutrophil-to-lymphocyte ratio predicts cancer outcome in locally advanced clear renal cell carcinoma. Clin Genitourin Cancer. (2022) 20:102–6. doi: 10.1016/j.clgc.2021.10.009
24. Nakamura K, Yoshida N, Baba Y, Kosumi K, Uchihara T, Kiyozumi Y, et al. Elevated preoperative neutrophil-to-lymphocytes ratio predicts poor prognosis after esophagectomy in T1 esophageal cancer. Int J Clin Oncol. (2017) 22:469–75. doi: 10.1007/s10147-017-1090-5
25. Sharaiha RZ, Halazun KJ, Mirza F, Port JL, Lee PC, Neugut AI, et al. Elevated preoperative neutrophil:lymphocyte ratio as a predictor of postoperative disease recurrence in esophageal cancer. Ann Surg Oncol. (2011) 18:3362–9. doi: 10.1245/s10434-011-1754-8
26. Al Lawati Y, Cools-Lartigue J, Ramirez-GarciaLuna JL, Molina-Franjola JC, Pham D, Skothos E, et al. Dynamic alteration of neutrophil-to-lymphocyte ratio over treatment trajectory is associated with survival in esophageal adenocarcinoma. Ann Surg Oncol. (2020) 27:4413–9. doi: 10.1245/s10434-020-08521-7
27. Barbetta A, Nobel TB, Sihag S, Hsu M, Tan KS, Bains MS, et al. Neutrophil to lymphocyte ratio as predictor of treatment response in esophageal squamous cell cancer. Ann Thorac Surg. (2018) 106:864–71. doi: 10.1016/j.athoracsur.2018.04.007
28. Guo JC, Lin CC, Lin CY, Hsieh MS, Kuo HY, Lien MY, et al. Neutrophil-to-lymphocyte ratio and use of antibiotics associated with prognosis in esophageal squamous cell carcinoma patients receiving immune checkpoint inhibitors. Anticancer Res. (2019) 39:5675–82. doi: 10.21873/anticanres.13765
29. Xu X, Jing J. Inflammation-related parameter serve as prognostic biomarker in esophageal squamous cell carcinoma. Front Oncol. (2022) 12:900305. doi: 10.3389/fonc.2022.900305
30. Kato T, Oshikiri T, Goto H, Urakawa N, Hasegawa H, Kanaji S, et al. Preoperative neutrophil-to-lymphocyte ratio predicts the prognosis of esophageal squamous cell cancer patients undergoing minimally invasive esophagectomy after neoadjuvant chemotherapy. J Surg Oncol. (2021) 124:1022–30. doi: 10.1002/jso.26611
31. Fujiwara Y, Higashida M, Kubota H, Okamoto Y, Mineta S, Endo S, et al. Perioperative predictive markers for recurrence of esophageal cancer after esophagectomy. Gastrointest Tumors. (2021) 8:87–95. doi: 10.1159/000513961
32. Zhang J, Zhang Y, Yv X, Song M, Zhang F, Tian X, et al. Prognostic value of combined preoperative prognostic nutritional index and neutrophil to lymphocyte ratio in esophageal squamous cell carcinoma. Transl Cancer Res. (2020) 9:5117–27. doi: 10.21037/tcr-19-2397
33. Shang QX, Yang YS, Hu WP, Yuan Y, He Y, Zhao JY, et al. Clinical and prognostic significance of preoperative lymphocyte-monocyte ratio, neutrophil-lymphocyte ratio and neutrophil-monocyte ratio on esophageal squamous cell carcinoma patients. Transl Cancer Res. (2020) 9:3903–14. doi: 10.21037/tcr-19-2777
34. Hu J, Chen D, Wu S, Chen Y, Li R, Miao H, et al. Prognostic significance of neutrophil-to-lymphocyte ratio in middle thoracic esophageal squamous cell carcinoma patients undergoing radical esophagectomy. J Thorac Dis. (2020) 12:363–74. doi: 10.21037/jtd.2020.01.31
35. Chen C, Chen G, Wu Y, Li J. Preoperative lymphocyte-monocyte ratio is not an independent prognostic factor in M0 (stage I-III) esophageal squamous cell carcinomas. Transl Cancer Res. (2020) 9:3483–90. doi: 10.21037/tcr.2020.03.75
36. Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. Bmj. (2021) 372:n71. doi: 10.1136/bmj.n71
37. Peterson J, Welch V, Losos M, Tugwell P. The Newcastle-Ottawa scale (NOS) for assessing the quality of nonrandomised studies in meta-analyses. Ottawa: Ottawa Hosp Res Institute. (2011) 2:1–12.
38. Kim SR, Kim K, Lee SA, Kwon SO, Lee JK, Keum N, et al. Effect of red, processed, and white meat consumption on the risk of gastric cancer: an overall and dose−response meta-analysis. Nutrients. (2019) 11:3. doi: 10.3390/nu11040826
39. Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. (2002) 21:1539–58. doi: 10.1002/sim.1186
40. Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. Bmj. (1997) 315:629–34. doi: 10.1136/bmj.315.7109.629
41. Duan H, Zhang X, Wang FX, Cai MY, Ma GW, Yang H, et al. Prognostic role of neutrophil-lymphocyte ratio in operable esophageal squamous cell carcinoma. World J Gastroenterol. (2015) 21:5591–7. doi: 10.3748/wjg.v21.i18.5591
42. Feng JF, Huang Y, Chen QX. Preoperative platelet lymphocyte ratio (PLR) is superior to neutrophil lymphocyte ratio (NLR) as a predictive factor in patients with esophageal squamous cell carcinoma. World J Surg Oncol. (2014) 12:58. doi: 10.1186/1477-7819-12-58
43. Han F, Liu Y, Cheng S, Sun Z, Sheng C, Sun X, et al. Diagnosis and survival values of neutrophil-lymphocyte ratio (NLR) and red blood cell distribution width (RDW) in esophageal cancer. Clin Chim Acta. (2019) 488:150–8. doi: 10.1016/j.cca.2018.10.042
44. Han LH, Jia YB, Song QX, Wang JB, Wang NN, Cheng YF. Prognostic significance of preoperative lymphocyte-monocyte ratio in patients with resectable esophageal squamous cell carcinoma. Asian Pac J Cancer Prev. (2015) 16:2245–50. doi: 10.7314/APJCP.2015.16.6.2245
45. He YF, Luo HQ, Wang W, Chen J, Yao YW, Yan Y, et al. Preoperative NLR and PLR in the middle or lower ESCC patients with radical operation. Eur J Cancer Care (Engl). (2017) 26:4. doi: 10.1111/ecc.12445
46. Hirahara N, Matsubara T, Kawahara D, Nakada S, Ishibashi S, Tajima Y. Prognostic significance of preoperative inflammatory response biomarkers in patients undergoing curative thoracoscopic esophagectomy for esophageal squamous cell carcinoma. Eur J Surg Oncol. (2017) 43:493–501. doi: 10.1016/j.ejso.2016.11.018
47. Ikeguchi M, Kouno Y, Kihara K, Suzuki K, Endo K, Nakamura S, et al. Evaluation of prognostic markers for patients with curatively resected thoracic esophageal squamous cell carcinomas. Mol Clin Oncol. (2016) 5:767–72. doi: 10.3892/mco.2016.1073
48. Ishibashi Y, Tsujimoto H, Hiraki S, Kumano I, Yaguchi Y, Horiguchi H, et al. Prognostic value of preoperative systemic immunoinflammatory measures in patients with esophageal cancer. Ann Surg Oncol. (2018) 25:3288–99. doi: 10.1245/s10434-018-6651-y
49. Xiao Q, Zhang B, Deng X, Wu J, Wang H, Wang Y, et al. The preoperative neutrophil-to-lymphocyte ratio is a novel immune parameter for the prognosis of esophageal basaloid squamous cell carcinoma. PloS One. (2016) 11:e0168299. doi: 10.1371/journal.pone.0168299
50. Zhang X, Jain D. Updates in staging and pathologic evaluation of esophageal carcinoma following neoadjuvant therapy. Ann N Y Acad Sci. (2020) 1482:163–76. doi: 10.1111/nyas.14462
51. Pather K, Mobley EM, Guerrier C, Esma R, Kendall H, Awad ZT. Long-term survival outcomes of esophageal cancer after minimally invasive Ivor Lewis esophagectomy. World J Surg Oncol. (2022) 20:50. doi: 10.1186/s12957-022-02518-0
52. Kitasaki N, Hamai Y, Emi M, Kurokawa T, Yoshikawa T, Hirohata R, et al. Prognostic factors for patients with esophageal squamous cell carcinoma after neoadjuvant chemotherapy followed by surgery. In Vivo. (2022) 36:2852–60. doi: 10.21873/invivo.13025
53. Qi Q, Song Q, Cheng Y, Wang N. Prognostic significance of preoperative prognostic nutritional index for overall survival and postoperative complications in esophageal cancer patients. Cancer Manag Res. (2021) 13:8585–97. doi: 10.2147/CMAR.S333190
54. Yu X, Wen Y, Lin Y, Zhang X, Chen Y, Wang W, et al. The value of preoperative Glasgow Prognostic Score and the C-Reactive Protein to Albumin Ratio as prognostic factors for long-term survival in pathological T1N0 esophageal squamous cell carcinoma. J Cancer. (2018) 9:807–15. doi: 10.7150/jca.22755
55. Zhang H, Shang X, Ren P, Gong L, Ahmed A, Ma Z, et al. The predictive value of a preoperative systemic immune-inflammation index and prognostic nutritional index in patients with esophageal squamous cell carcinoma. J Cell Physiol. (2019) 234:1794–802. doi: 10.1002/jcp.27052
56. Zheng Z, Yang C, Cai C, Zhu H. The preoperative neutrophil lymphocyte ratio and platelet lymphocyte ratio predicts disease-free survival in resectable esophageal squamous cell carcinoma. Cancer Manag Res. (2021) 13:7511–6. doi: 10.2147/CMAR.S321326
57. Pirozzolo G, Gisbertz SS, Castoro C, van Berge Henegouwen MI, Scarpa M. Neutrophil-to-lymphocyte ratio as prognostic marker in esophageal cancer: a systematic review and meta-analysis. J Thorac Dis. (2019) 11:3136–45. doi: 10.21037/jtd.2019.07.30
58. Li B, Xiong F, Yi S, Wang S. Prognostic and clinicopathologic significance of neutrophil-to-lymphocyte ratio in esophageal cancer: an update meta-analysis. Technol Cancer Res Treat. (2022) 21:15330338211070140. doi: 10.1177/15330338211070140
59. Jiang Y, Xu D, Song H, Qiu B, Tian D, Li Z, et al. Inflammation and nutrition-based biomarkers in the prognosis of oesophageal cancer: a systematic review and meta-analysis. BMJ Open. (2021) 11:e048324. doi: 10.1136/bmjopen-2020-048324
60. Rice TW, Chen LQ, Hofstetter WL, Smithers BM, Rusch VW, Wijnhoven BP, et al. Worldwide Esophageal Cancer Collaboration: pathologic staging data. Dis Esophagus. (2016) 29:724–33. doi: 10.1111/dote.2016.29.issue-7
61. Wang J, Yang Y, Shaik MS. Prognostic significance of the number of lymph nodes dissection in esophageal adenocarcinoma patients. Transl Cancer Res. (2020) 9:3406–15. doi: 10.21037/tcr-19-2802
62. Guo XW, Zhou JY, Jiang W, Ji L, Liu YC, Yin XX. The combination of preoperative nutritional risk screening-2002 and neutrophil-to-lymphocyte ratio is a useful prognostic marker in patients with esophageal squamous cell carcinoma. Nutr Cancer. (2021) 73:588–95. doi: 10.1080/01635581.2020.1766090
63. Yin C, Okugawa Y, Kitajima T, Shimura T, Ma R, Kawamura M, et al. Clinical significance of the preoperative inflammatory burden index in esophageal cancer. Oncology. (2024) 102:556–64. doi: 10.1159/000535727
64. Yang J, Zheng J, Qiu J, Zhang M, Liu L, Wang Z, et al. Systemic immune-inflammatory index, tumor-infiltrating lymphocytes, and clinical outcomes in esophageal squamous cell carcinoma receiving concurrent chemoradiotherapy. J Immunol Res. (2023) 2023:4275998. doi: 10.1155/2023/4275998
65. Kemuriyama K, An J, Motoyama S, Nagaki Y, Yamaguchi T, Sato Y, et al. Squamous cell carcinoma-derived G-CSF promotes tumor growth and metastasis in mice through neutrophil recruitment and tumor cell proliferation, associated with poor prognosis of the patients. Genes Cells. (2023) 28:573–84. doi: 10.1111/gtc.13051
66. Zhou J, Lin HP, Xu X, Wang XH, Rong L, Zhang Y, et al. The predictive value of peripheral blood cells and lymphocyte subsets in oesophageal squamous cell cancer patients with neoadjuvant chemoradiotherapy. Front Immunol. (2022) 13:1041126. doi: 10.3389/fimmu.2022.1041126
67. Mjaess G, Chebel R, Karam A, Moussa I, Pretot D, Abi Tayeh G, et al. Prognostic role of neutrophil-to-lymphocyte ratio (NLR) in urological tumors: an umbrella review of evidence from systematic reviews and meta-analyses. Acta Oncol. (2021) 60:704–13. doi: 10.1080/0284186X.2021.1886323
Keywords: neutrophil, lymphocyte, NLR, esophageal cancer, esophagectomy, meta-analysis
Citation: Wu X, Liu S, Li F and Chen Y (2024) Association between preoperative neutrophil-to-lymphocyte ratio and the survival outcomes of esophageal cancer patients underwent esophagectomy: a systematic review and meta-analysis. Front. Oncol. 14:1404711. doi: 10.3389/fonc.2024.1404711
Received: 21 March 2024; Accepted: 26 July 2024;
Published: 19 August 2024.
Edited by:
Philip Rosenberg, National Cancer Institute (NIH), United StatesReviewed by:
Jesus Rico-Feijoo, Hospital Universitario Río Hortega, SpainGina Ney, National Cancer Institute (NIH), United States
Copyright © 2024 Wu, Liu, Li and Chen. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: YingTai Chen, WWluZ3RhaWM3OEAxMjYuY29t
 Xun Wu
Xun Wu SiJie Liu
SiJie Liu