- 1Department of Pathology, West China Second University Hospital, Sichuan University, Chengdu, China
- 2Key Laboratory of Birth Defects and Related Diseases of Women and Children, Sichuan University, Ministry of Education, Chengdu, China
NTRK-rearranged spindle cell neoplasm is a rare subtype of soft tissue sarcoma that occasionally arises in the lower female genital tract. Accurate diagnosis is clinically important, as these tumors generally display low-grade malignant behavior and may respond to TRK inhibitor therapy. We report a rare case of cervical NTRK-rearranged spindle cell neoplasm in a 40-year-old woman who presented with abnormal vaginal bleeding. An initial biopsy of the cervical mass suggested a diagnosis of sarcoma. The patient subsequently underwent a total hysterectomy with bilateral salpingo-oophorectomy. Histological examination revealed spindle-shaped tumor cells diffusely infiltrating the cervical stroma in a solid growth pattern. Immunohistochemistry demonstrated diffuse expression of TRK and CD34. Fluorescence in situ hybridization (FISH) detected rearrangement of the NTRK1 gene, and RNA-based next-generation sequencing confirmed a TPM3::NTRK1 fusion gene. To date, only 61 cases of NTRK-rearranged spindle cell neoplasm in the female genital tract have been reported. Here, we present a new case with a detailed description of the clinical presentation, histopathological and immunophenotypic characteristics, molecular findings, and clinical outcome. Comparative analysis with previously reported cases suggests a possible correlation between NTRK fusion type and patient prognosis. Specifically, tumors with NTRK1 fusions tend to present at earlier stages and are associated with more favorable outcomes. These findings highlight the potential value of tailoring clinical management strategies based on fusion type.
Introduction
Uterine sarcomas represent a heterogeneous group of malignant mesenchymal tumors, with endometrial stromal sarcoma and leiomyosarcoma being the most common subtypes. Advances in molecular pathology have led to the identification of several novel sarcoma entities, including NTRK-rearranged spindle cell neoplasms (1), which can also occur in the female genital tract, particularly in the cervix (2–6). It is characterized by a “fibrosarcoma-like” spindle cell morphology and recurrent rearrangements involving the NTRK gene family. The histological features often overlap with those of other soft tissue tumors, posing significant diagnostic challenges, particularly in the absence of molecular genetic testing. Although recent studies have broadened the morphological and immunophenotypic spectrum of these tumors, prognostically relevant features remain undefined, and their biological behavior is still not well understood (7). Accurate identification holds clinical significance, as patients with recurrent or metastatic disease may benefit from targeted therapy using TRK inhibitors (8, 9). In this report, we describe a rare case of cervical NTRK-rearranged spindle cell neoplasm with detailed clinicopathological, immunophenotypic, and molecular features, aiming to improve diagnostic recognition and provide insights into the biological and therapeutic implications of this tumor type.
Case presentation
A 40-year-old woman was admitted to the hospital with abnormal vaginal bleeding. Ultrasound imaging revealed an irregular, weakly echogenic mass in the cervix, measuring approximately 7.9 cm×6.7 cm×8.0 cm, with indistinct margins and prominent internal vascularity. Surgical excision of the cervical tumor was performed via a transvaginal approach. Gross examination revealed a mass predominantly located in the anterior lip of the cervix. Histopathological evaluation suggested a diagnosis of sarcoma. The patient subsequently underwent total hysterectomy with bilateral salpingo-oophorectomy, pelvic lymphadenectomy, and para-aortic lymph node sampling.
Materials and methods
Histopathological examination was independently conducted by two gynecological pathologists. Details of the primary antibodies used for immunohistochemical (IHC) staining are provided in Supplementary Table 1. Immunostaining procedures were carried out according to standardized laboratory protocols and antibody manufacturer instructions, including the routine use of appropriate positive and negative controls.
Fluorescence in situ hybridization (FISH) analysis was carried out using a commercial dual-color break-apart probe kit targeting the NTRK1, NTRK2, and NTRK3 genes (Guangzhou LBP Medicine Science & Technology Co., Ltd., Guangzhou, China). Formalin-fixed paraffin-embedded tissue sections were deparaffinized and digested with pepsin at 37°C for 9 minutes. Co-denaturation of tissue sections and probes was performed at 85°C for 5 minutes, followed by overnight hybridization at 37°C. After stringent post-hybridization washes, nuclei were counterstained with 4′,6-diamidino-2-phenylindole (DAPI), and slides were mounted with coverslips. A tumor cell was considered positive for gene rearrangement if distinct red and green fluorescent signals (break-apart pattern) were observed, indicating disruption of the NTRK gene locus. At least 100 tumor cells were evaluated per case, and a specimen was considered positive when >10% of cells exhibited break-apart signals.
RNA-based next-generation sequencing (RNA-NGS) was conducted by GenePlus Technology (Beijing, China). Total RNA was extracted using the RNeasy FFPE Kit (Qiagen, Cat. No. 73504) according to the manufacturer’s protocol. RNA integrity and concentration were assessed with the Agilent 2100 Bioanalyzer (Agilent Technologies, WA, USA). Sequencing libraries were constructed and subjected to 150 bp paired-end sequencing using the DNBSEQ-T7 platform (GenePlus, Beijing, China). Raw sequencing data were filtered to remove low-quality reads and adapter sequences. High-quality reads were subsequently aligned to the human reference genome (hg19) using the STAR aligner. Fusions were detected by a customized version of Arriba 1.1.0 and annotated by in house software annoFilterArriba (version:1.0.0) with NCBI release 104 database. A gene fusion event was confirmed when ≥5 high-quality, unique reads spanned the fusion breakpoint, and ≥3 of those reads had unique start sites. All final candidate fusions were manually verified with the integrative genomics viewer browser. A series of quality control metrics was computed by using RNA-SeQC assessment. A threshold of ≥ 80 million mapped reads and ≥ 10 million junction reads per sample was set. The genome was visualized using IGV software (GeneVis v1.2.3) (10, 11).
A literature review was conducted to identify previously published cases of NTRK-rearranged uterine sarcoma. English-language articles were retrieved from the PubMed database using the keywords “NTRK” combined with “uterus”, “uterine”, “cervix”, “cervical”, or “female genital tract”.
Statistical analysis of the literature-derived data was performed using SPSS software (version 25.0). Data are presented as mean ± standard deviation (). For comparisons of means between two groups, the independent-sample t-test was used if assumptions of normality and homogeneity of variance were met. Otherwise, the nonparametric Wilcoxon rank-sum test was applied. A two-sided p-value < 0.05 was considered statistically significant.
Results
Gross examination revealed a solid, greyish-yellow tumor measuring 9.5 cm×7.3 cm×5.5 cm, and the tumor infiltrated approximately 50% of the cervical stroma (Figure 1). Microscopically, the tumor exhibited a diffuse, solid growth pattern with scattered residual endocervical glands. The tumor cells were relatively uniform, displaying spindle- or oval-shaped nuclei with mild nuclear atypia. Mitotic activity was brisk, with up to 25 mitoses per 10 high-power fields (HPFs). The tumor demonstrated variable-sized intratumoral blood vessels and focal lymphocytic infiltration (Figure 2). A total of 25 lymph nodes were dissected, including 12 from the left pelvic region, 12 from the right pelvic region, and 1 from the para-aortic region. No evidence of metastasis was identified.
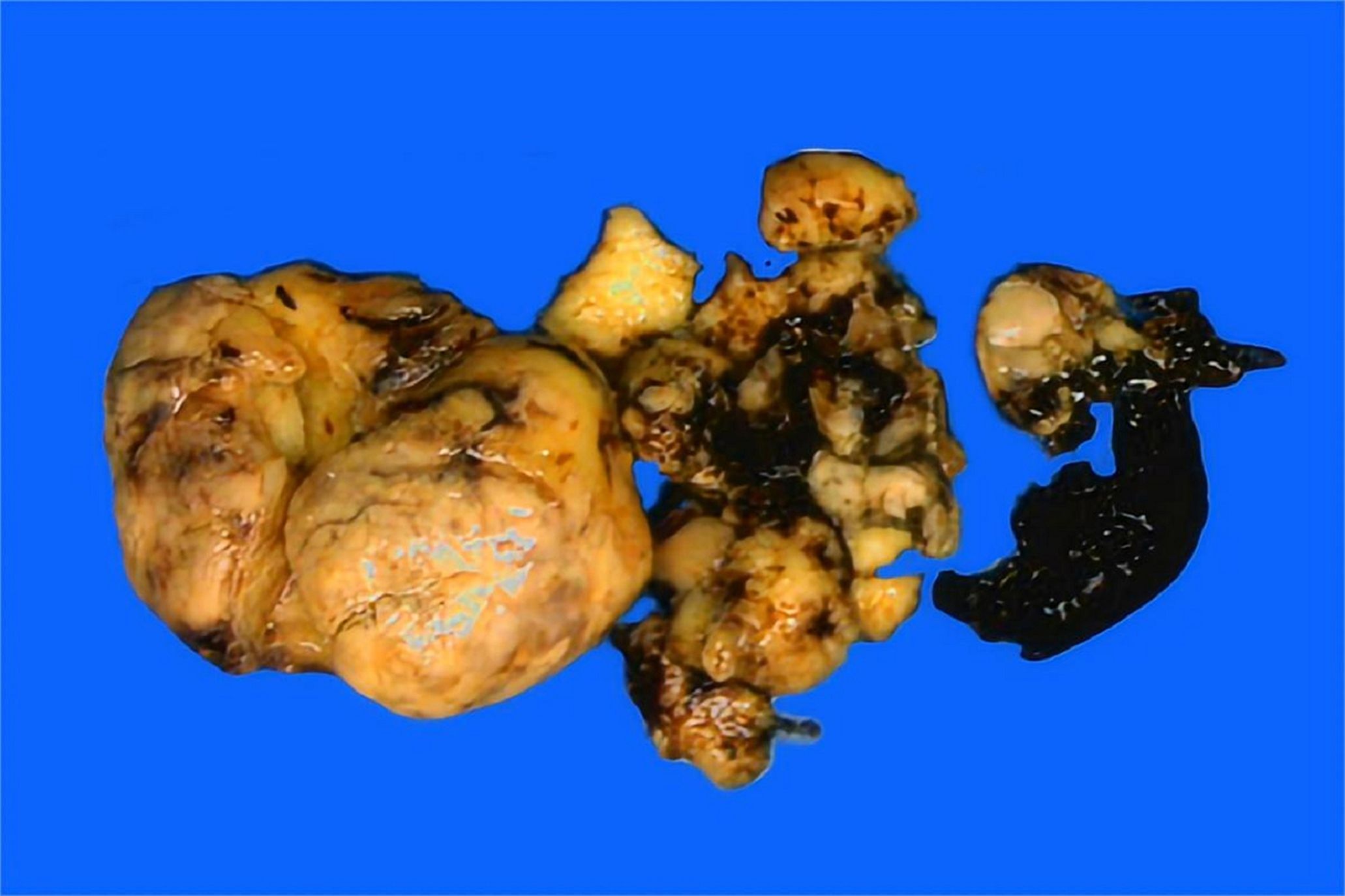
Figure 1. Grossly, the solid tumor presented a greyish-yellow cut surface with focal necrosis and hemorrhage.
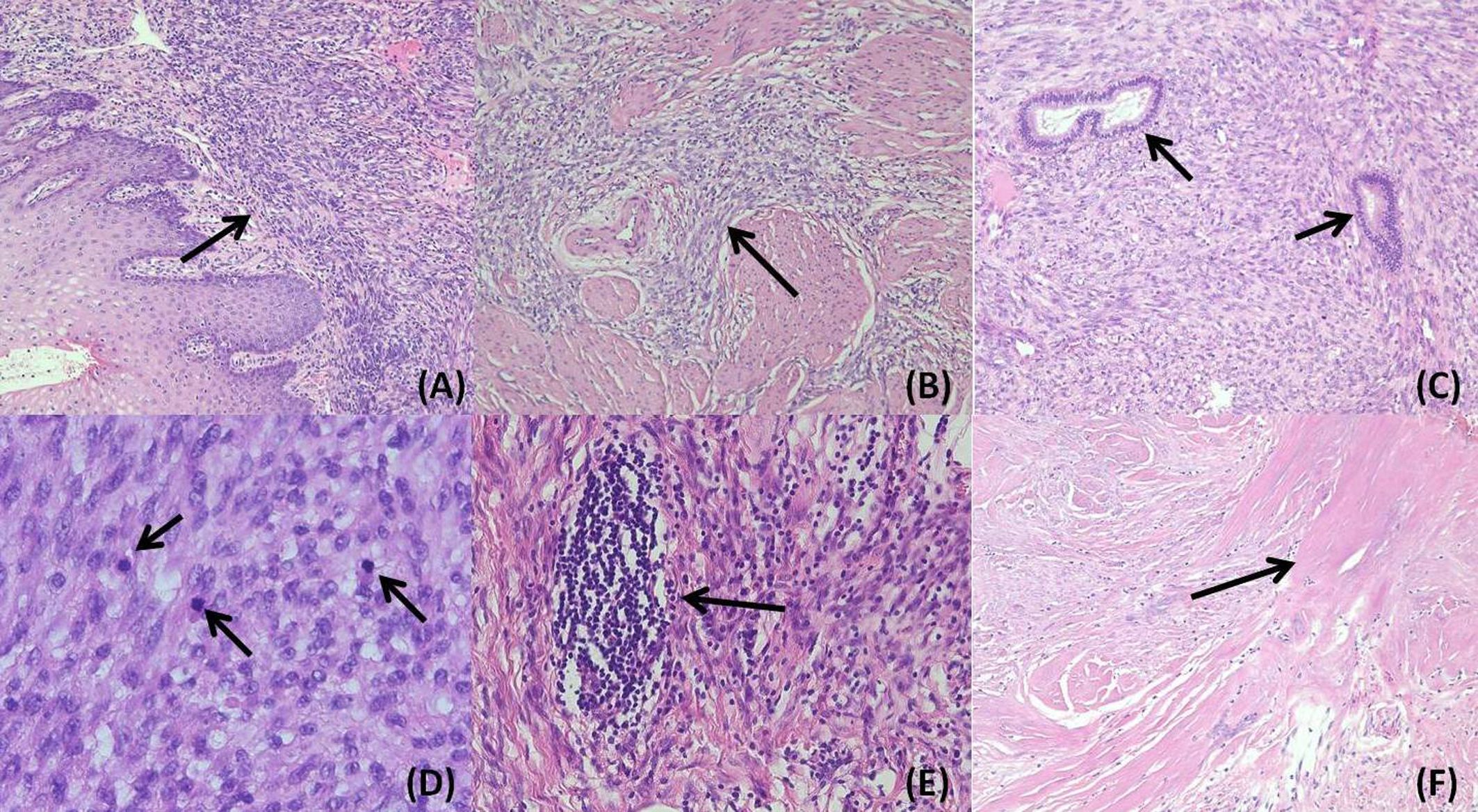
Figure 2. In the hematoxylin&eosin slide, the tumor exhibited a sheet-like growth pattern underneath the cervical mucosa (A) 100×magnification) and infiltrated the cervical stroma (B) magnification:100×). Residual endocervical glands were found among the tumor components (C) magnification:100×). The tumor consisted of spindle or oval cells with mild atypia and abundant mitoses (D) magnification:400×). Lymphocyte aggregation (E) magnification:200×) and necrosis (F) magnification:100×) could be found focally.
Immunohistochemical analysis showed diffuse and strong expression of TRK and CD34 in tumor cells. Focal and weak positivity was observed for cyclin D1, desmin, smooth muscle actin (SMA), CD10, estrogen receptor (ER), and progesterone receptor (PR). Staining for S-100, caldesmon, calponin, MyoD1, myogenin, BCOR, and ALK was negative (Figure 3). P53 staining showed a wild-type pattern, and the Ki67 proliferation index was approximately 15%. Based on the immunophenotype, uterine leiomyosarcoma, endometrial stromal sarcoma (ESS), rhabdomyosarcoma, and inflammatory myofibroblastic tumor (IMT) were excluded.
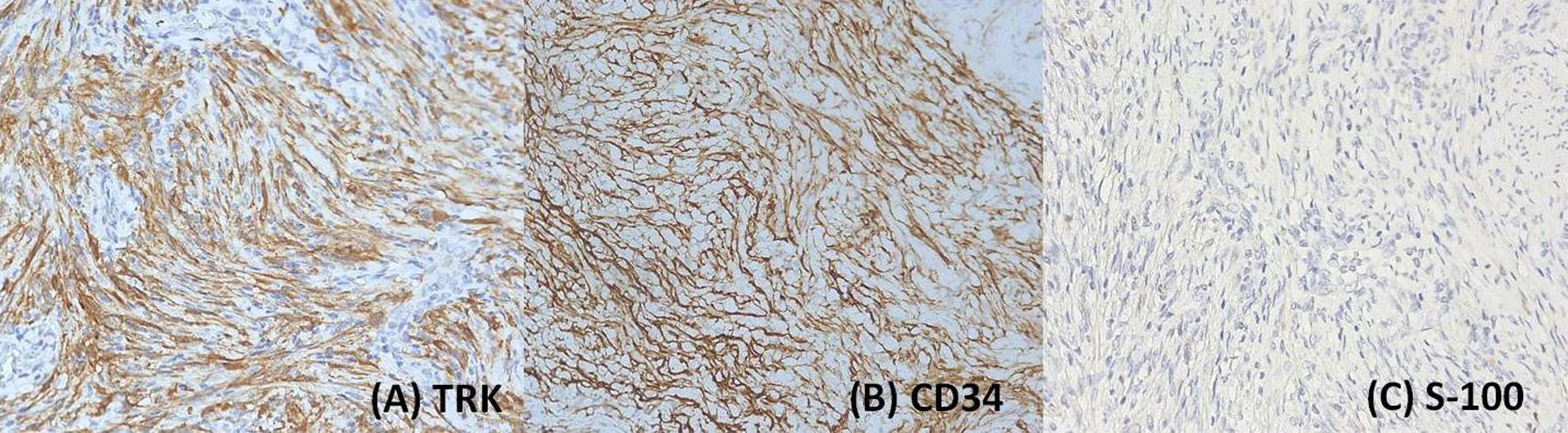
Figure 3. Tumor cells exhibited diffuse and strong immunostaining for TRK (A) magnification:200×) and CD34 (B) magnification:200×), while showing negative staining for S-100 (magnification: (C) 200×).
The FISH analysis demonstrated separated red and green fluorescence signals for the NTRK1 probe in tumor cells, indicating a rearrangement of the NTRK1 gene (Figure 4A). Targeted RNA sequencing was used to assess rearrangements involving a panel of 555 genes associated with tumorigenesis and tumor progression.A TPM3::NTRK1 gene fusion (NM_001043353.1/NM_002529.3) was detected in the tumor sample, with the specific fusion breakpoint located at exon 7 of TPM3 and exon 10 of NTRK1 (Figure 4B). A total of 224 supporting reads were identified (Figure 4C). This fusion is considered actionable for targeted therapy.
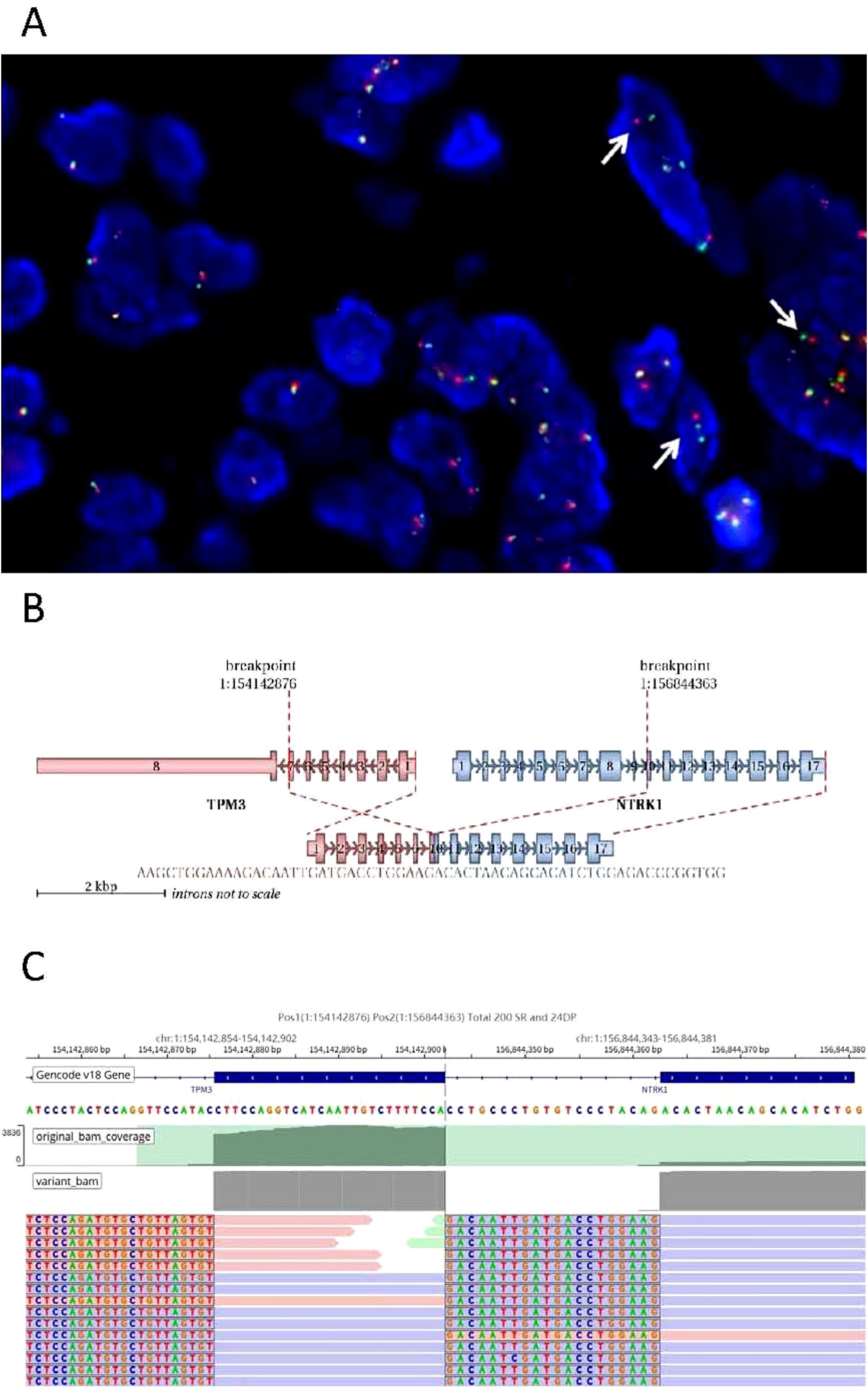
Figure 4. TPM3-NTRK1 fusion analysis. (A) Breakage of the NTRK1 gene was detected by FISH, which showed separate red and green signals in some tumor cells (arrow). (B) Schematic diagram of detailed fusion site of TPM3 and NTRK1. (C) NGS results showed a breakpoint of fusion. original bam coverge (Raw depth, including both mutated and non-mutated alleles); variant bam (Raw depth, including only mutated alleles); SR (Split Read): This refers to a sequencing read whose segments are mapped to different genomic locations. Such mapping indicates that the read spans a potential breakpoint, which may be caused by structural variations such as deletions, insertions, inversions, or translocations, resulting in discontinuity in the genomic sequence; DP (Discordant Pair): This refers to a pair of paired-end sequencing reads whose mapping does not conform to the expected insert size or orientation (e.g., the reads are oriented in opposite directions, or are mapped at an abnormal distance apart). A typical instance is when each read of a pair maps to two different genes, strongly suggesting the presence of a structural variant such as a translocation between these genes. SR and DP represent the most commonly used and critical types of bioinformatic evidence for the detection of genomic structural variations (SVs).
According to the 2018 International Federation of Gynecology and Obstetrics (FIGO) staging system, the tumor was classified as stage IB3 (pT1b3N0M0). The patient received adjuvant chemotherapy consisting of ifosfamide (1 g/m²/day, days 1-3), epirubicin (20 mg/m²/day, days 1-3), and cisplatin (20 mg/m²/day, days 1-3), all administered via intravenous infusion. The patient is currently undergoing adjuvant radiotherapy. No targeted therapy has been administered.
Literature review and survival analysis
A total of 20 publications encompassing 61 cases of NTRK-rearranged spindle cell neoplasms in the female genital tract were included in the analysis. Among these, 54 cases were located in the uterine cervix and 7 in the uterine corpus. The clinicopathological features of all 61 patients are summarized in Supplementary Table 2. Data from the literature were statistically analyzed using SPSS software version 25.0, with a p-value of <0.05 considered statistically significant.
The mean age at diagnosis was 39 years (range: 13–69 years), and the average tumor size was 7.0 cm (range: 1.3–23.0 cm). At the time of diagnosis, most tumors were confined to the uterus, with 88% of patients classified as stage IA or IB according to the FIGO system. The average mitotic count was 12 per 10 HPFs, ranging from 0 to 50. Tumor necrosis was present in 40% of cases, and lymphovascular invasion was identified in 20%. Follow-up data were available for 50 patients. Among them, 26 patients (52%) had no evidence of disease (NED), while 7 patients (14%) died of disease (disease-specific death, DSD). In stage IA patients, 83% (10/12) were NED, and 17% (2/12) were alive with disease (AWD); notably, two patients in this group developed distant metastases involving the pleura and bone. Among patients at stage IB, 57% (12/21) were NED, 19% (4/21) were AWD, and 24% (5/21) died of disease. Only five patients were diagnosed at more advanced stages, including stage IIA (n = 2), stage IIB (n = 2), and stage IVB (n = 1) (Table 1).
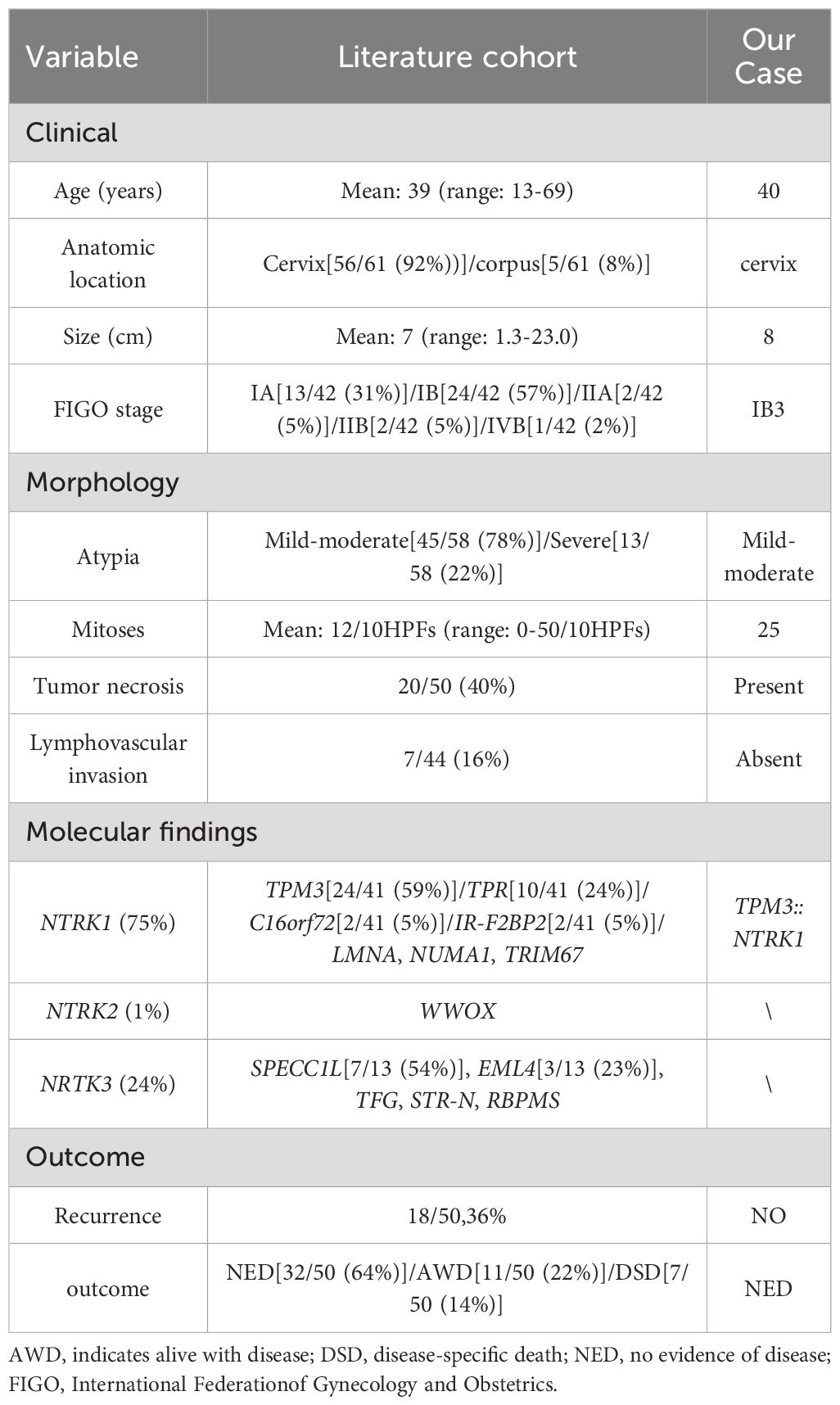
Table 1. Summary of the clinical, morphologic, and molecular findings in our own case and the literature cohort (n=61).
Molecular detection revealed that NTRK1 fusions were the most common type (44/59, 75%), followed by NTRK3 fusions (14/59, 24%) and a single NTRK2 fusion (1%). Various fusion types were identified (Table 1), among which TPM3::NTRK1 was the most common type (24/41, 59%). The comparative analysis demonstrated that tumors harboring NTRK3 fusions were significantly larger than those with NTRK1 fusions (mean diameter: 10.4 cm vs. 5.5 cm, p = 0.011) and more often presented at advanced FIGO stages (>IB vs. ≤IB, p = 0.016). Furthermore, chi-square analysis revealed a significantly higher disease-specific mortality rate in patients with NTRK3 fusions compared to those with NTRK1 fusions (33% vs. 6%, p = 0.028), suggesting a poorer prognosis associated with NTRK3 fusion tumors (Table 2). Although NTRK3 fusion tumors also exhibited a higher mitotic index and increased recurrence rates, these differences did not reach statistical significance. No significant differences were observed between the NTRK1 and NTRK3 fusion groups in terms of patient age, presence of lymphovascular invasion, or tumor necrosis.
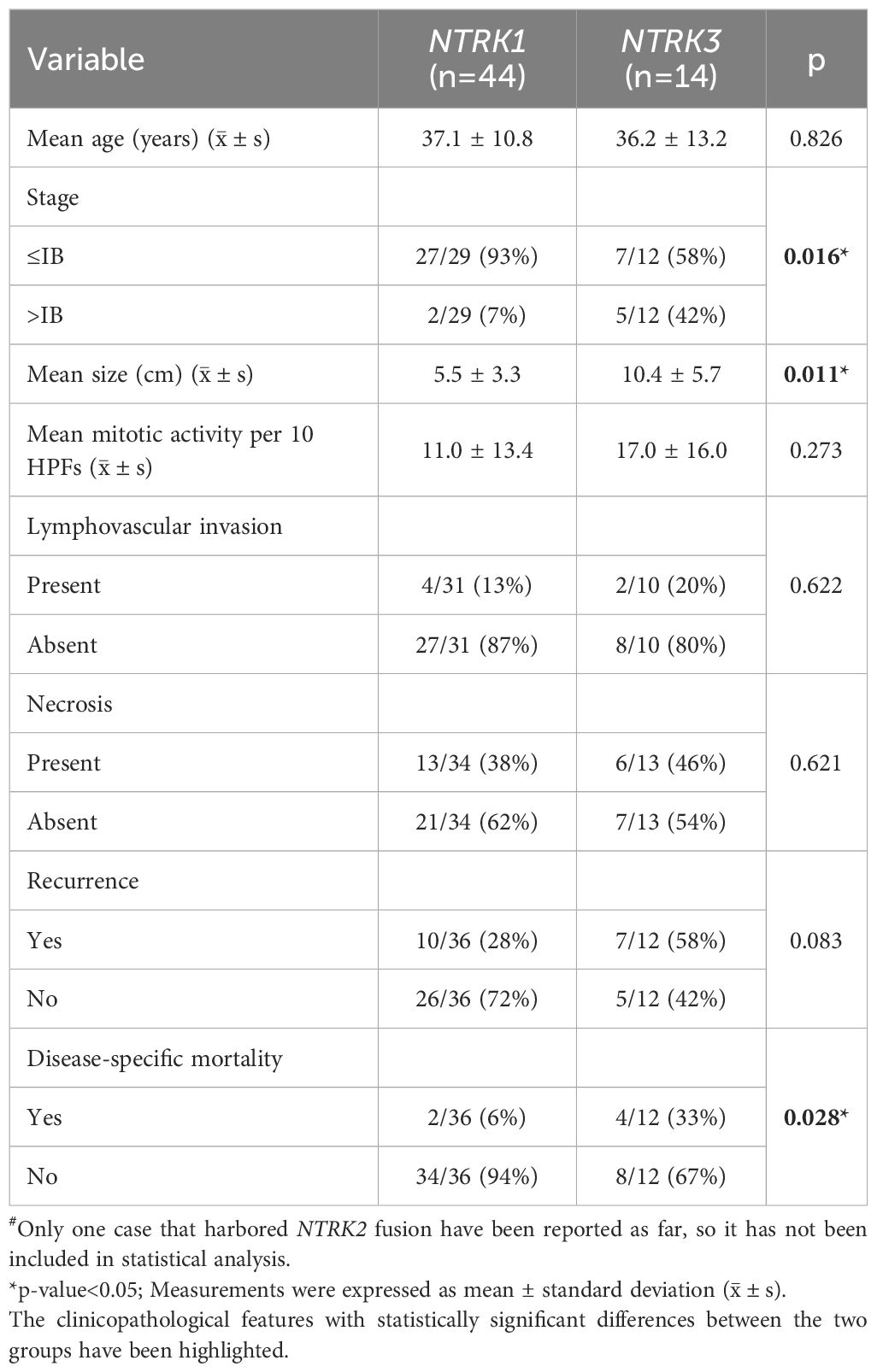
Table 2. Comparison of clinicopathologic features between NTRK1-rearranged group(n=44) and NTRK3-rearranged group (n=14)#.
Discussion
In this study, we presented a rare case of NTRK-rearranged spindle cell neoplasm of the uterine cervix and performed a literature review encompassing 62 cases of this tumor entity in the female genital tract. Based on published reports, we summarized the clinicopathological characteristics, molecular alterations, and clinical outcomes, including our own case (1, 2, 6–8, 12–26). We believe that the significance of reporting this case is to expand the database of such tumors in the female reproductive tract, as the number of reported cases is small and the data available for analysis of pathological diagnosis, treatment, and prognosis are very limited.
Given the rarity of this tumor and the limited data currently available, documenting such cases is essential to expand the collective knowledge base regarding diagnosis, therapeutic implications, and prognostic evaluation in the female reproductive system.
Pathogenesis
The three members of the neurotrophic tyrosine receptor kinase (NTRK) family (NTRK1, NTRK2, and NTRK3) encode TRKA, TRKB, and TRKC proteins, respectively (12). These receptors are physiologically expressed in the peripheral and central nervous systems and play critical roles in neural development and function (13–15). Upon ligand binding, the extracellular domains of NTRK receptors undergo dimerization, leading to autophosphorylation and activation of downstream signaling cascades, including the MAPK, PI3K, and PKC pathways. These pathways regulate essential cellular processes such as proliferation, differentiation, and survival (1, 27). NTRK gene rearrangements in tumors can result from either intrachromosomal or interchromosomal translocations. These rearrangements typically involve the fusion of the 5’ end of an NTRK gene with the 3’ end of a partner (chaperone) gene, producing a chimeric fusion protein. When the chaperone gene encodes a domain that facilitates dimerization, the resulting fusion protein can undergo constitutive dimerization without ligand binding. This ligand-independent activation leads to persistent stimulation of the downstream signaling pathways mediated by the TRK kinase domain, thereby promoting oncogenic transformation. This mechanism represents a key driver of tumorigenesis in NTRK-altered malignancies. Frequently reported fusion partners include LMNA, TPM3, PAN3, and ETV6 (14, 15). TRK receptor activation via NTRK fusion is recognized as a pan-cancer oncogenic mechanism, with an estimated incidence of 0.68% to 1% in adult soft tissue sarcomas (1, 8, 14, 27). NTRK rearrangements have been documented in a variety of soft tissue tumors in both adults and children, including infantile fibrosarcoma and congenital mesoblastic nephroma. However, such genetic alterations are exceedingly rare in gynecologic tumors.
Pathological diagnosis
To date, few distinct morphological features have been validated as reliable diagnostic indicators for NTRK-rearranged spindle cell neoplasms. Therefore, definitive diagnosis largely depends on IHC analysis and molecular testing.
Among the IHC markers, TRK, CD34 and S-100(especially TRK) are considered valuable for raising suspicion of this tumor type and guiding further molecular investigations. Tumor cells typically exhibit diffuse cytoplasmic positivity for TRK. An exception to this pattern has been reported in a TRK-negative case harboring a SPECC1L::NTRK3 fusion gene, which also demonstrated a loss of SMARCB1 (INI1) expression (23). In addition to the classic cytoplasmic staining, alternative TRK expression patterns have been described. For example, tumors with LMNA::NTRK1 fusions may show nuclear membrane localization, while tumors with TPM3::NTRK1 or ETV6::NTRK3 fusions can exhibit membranous staining patterns (1, 14, 28). Although TRK immunostaining is a useful screening tool, it is important to note that TRK positivity does not always indicate NTRK gene rearrangements. Cases with TRK expression must be confirmed by molecular assays. For instance, some high-grade endometrial stromal sarcomas (ESS) have shown TRK positivity despite lacking any NTRK fusions (22). Therefore, confirmatory testing such as FISH, reverse transcription PCR (RT-PCR), or next-generation sequencing (NGS) is essential to establish the diagnosis. Additionally, the tumor frequently expresses S-100 and CD34 (29). S-100 expression was reported in 43 of 49 cases (88%) and CD34 in 34 of 45 cases (76%) (Supplementary Table 2). Thus, we recommend the use of TRK, CD34, and S-100 immunostaining as an accessible and cost-effective initial screening strategy for all uterine spindle cell tumors, particularly those lacking definitive endometrial stroma or smooth muscle differentiation, to facilitate the identification of NTRK-rearranged neoplasms.
Differential diagnosis
As previously discussed, Many mesenchymal tumors of the uterus present with similar spindle cell morphology, making differential diagnosis based solely on conventional histopathology challenging. In this context, immunohistochemistry is an essential and practical tool to support accurate pathological classification. In our case, a broad range of differential diagnoses need to be considered based on the histological features, including low-grade and high-grade ESS, leiomyosarcoma, and IMT, among others. Low-grade ESS typically expresses CD10 with estrogen and progesterone receptors, whereas high-grade ESS may show immunoreactivity for cyclin D1 and/or BCOR. In contrast, our case was negative for myogenic markers such as desmin, caldesmon, calponin, and MyoD1, as well as for the IMT-associated marker ALK, supporting the exclusion of leiomyosarcoma, rhabdomyosarcoma and IMT. Adenosarcoma with sarcomatous overgrowth should also be excluded, as residual benign endocervical glands were observed among the tumor components in our case, but the characteristic periglandular sarcomatous cuffing typical of adenosarcoma was absent. For rare uterine malignant peripheral nerve sheath tumors (MPNSTs), which may show focal positivity for S-100 and CD34, the key distinction lies in their usual expression of neural markers. Some tumors previously diagnosed as MPNSTs have been reclassified as NTRK-rearranged spindle cell tumors by molecular testing (22).
Prognosis
A recent study by Costigan (7) suggests that several indicators, including both morphological and genetic features, may predict poor prognosis, such as lymphovascular invasion, necrosis, mitotic counts ≥8 per 10 HPFs, and NTRK3 fusion. Interestingly, tumors with NTRK3 fusions were generally larger and exhibited higher mitotic activity compared to those with NTRK1 fusions. They also tended to present at more advanced stages and had a higher likelihood of recurrence, suggesting a poorer outcome for these patients. Tumors exhibiting one or more of these features should be considered high-risk, whereas those lacking all of them may be classified as low-risk (7). According to these criteria, our case should be regarded as high-risk due to the presence of necrosis and active mitosis, and warrants prolonged follow-up despite no evidence of recurrence to date. In the female genital tract, 75% of reported cases harbored NTRK1 fusions, with fusion partners including TPM3, TPR, IRF2BP2, C16ORF72, LMNA, NUMA1, and TRIM67. Notably, all patients with TPM3::NTRK1 fusions (accounting for 59% of cases) were alive at the time of follow-up. These findings suggest that fusion type may correlate with survival outcomes, although further investigation is warranted.
Furthermore, it has been proposed that some NTRK-rearranged sarcomas with high-grade nuclear features may harbor TP53 mutations (7). In general, TP53 mutations result in a mutant-type immunostaining pattern, which may serve as a surrogate marker for underlying genetic alteration. TP53 mutations have been associated with high-grade nuclear features and poor prognosis in other tumors, such as endometrial carcinoma. However, the relationship between p53 expression patterns and prognosis remains unclear in NTRK-rearranged sarcomas of the female genital tract, highlighting the need for further studies exploring the prognostic significance of different p53 staining patterns.
Treatment
Most patients with NTRK-rearranged spindle cell neoplasms underwent surgical resection followed by adjuvant chemotherapy, with or without radiotherapy. As these tumors are typically negative for hormone receptors and rarely involve the ovaries, ovarian preservation does not appear to adversely affect prognosis (7, 12). The necessity of oophorectomy in young patients with NTRK-rearranged sarcomas remains uncertain. Studies have demonstrated that TRK inhibitors, such as entrectinib and larotrectinib, are highly effective, particularly in patients with advanced or metastatic disease. According to reports in the literature, 109 patients with NTRK gene fusions received larotrectinib treatment, achieving an investigator-assessed overall response rate of 81% (95% CI: 72%–88%). Among them, 63% experienced partial responses, while 17% achieved complete responses. Based on these results, the U.S. Food and Drug Administration (FDA) approved larotrectinib in 2018 for the treatment of solid tumors harboring NTRK gene fusions (4, 6, 10, 16, 17, 30). In our case, the patient underwent radical hysterectomy followed by adjuvant chemotherapy and radiotherapy, but has not received targeted therapy, as the disease was diagnosed at an early stage and no recurrence has been observed to date. Therefore, we are currently unable to assess the efficacy of TRK inhibitors in this case. It is worth noting that follow-up data for this tumor type remain limited, highlighting the need for larger, long-term studies to further evaluate treatment strategies.
Conclusion
In summary, primary NTRK-rearranged spindle cell neoplasm in the female genital tract is rare. Diagnosis based solely on clinical presentation or routine histopathology is challenging. Accurate pathological identification requires immunohistochemistry and molecular testing. Although the number of reported cases remains limited, affected patients may benefit from treatment with TRK inhibitors. Therefore, precise diagnosis is crucial for both gynecologists and pathologists in optimizing patient management.
Data availability statement
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding author.
Ethics statement
Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
Author contributions
LF: Conceptualization, Investigation, Writing – original draft. LL: Data curation, Writing – review & editing. YH: Data curation, Writing – review & editing. WJ: Supervision, Validation, Visualization, Writing – review & editing.
Funding
The author(s) declare that no financial support was received for the research and/or publication of this article.
Acknowledgments
The authors appreciate the invaluable support provided by gynecologists and other colleagues.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fonc.2025.1525722/full#supplementary-material
References
1. Chiang S, Cotzia P, Hyman DM, Drilon A, Tap WD, Zhang L, et al. NTRK fusions define a novel uterine sarcoma subtype with features of fibrosarcoma. Am J Surg Pathol. (2018) 42:791–8. doi: 10.1097/PAS.0000000000001055
2. Gatalica Z, Xiu J, Swensen J, and Vranic S. Molecular characterization of cancers with NTRK gene fusions. Mod Pathol. (2019) 32:147–53. doi: 10.1038/s41379-018-0118-3
3. Chiang S. S-100 and pan-trk staining to report NTRK fusion-positive uterine sarcoma: proceedings of the ISGyP companion society session at the 2020 USCAP annual meeting. Int J Gynecol Pathol. (2021) 40:24–7. doi: 10.1097/PGP.0000000000000702
4. Croce S, Hostein I, and McCluggage WG. NTRK and other recently described kinase fusion positive uterine sarcomas: A review of a group of rare neoplasms. Genes Chromosomes Cancer. (2021) 60:147–59. doi: 10.1002/gcc.22910
5. Hodgson A, Pun C, Djordjevic B, and Turashvili G. NTRK-rearranged cervical sarcoma: expanding the clinicopathologic spectrum. Int J Gynecol Pathol. (2021) 40:73–7. doi: 10.1097/PGP.0000000000000669
6. Nilforoushan N, Wethington SL, Nonogaki H, Gross J, Vang R, and Xing D. NTRK-fusion sarcoma of the uterine cervix: report of 2 cases with comparative clinicopathologic features. Int J Gynecol Pathol. (2022) 41:642–8. doi: 10.1097/PGP.0000000000000834
7. Costigan DC, Nucci MR, Dickson BC, Chang MC, Song S, Sholl LM, et al. NTRK -rearranged uterine sarcomas: clinicopathologic features of 15 cases, literature review, and risk stratification. Am J Surg Pathol. (2022) 46:1415–29. doi: 10.1097/PAS.0000000000001929
8. Croce S, Devouassoux-Shisheboran M, Pautier P, Ray-Coquard I, Treilleux I, Neuville A, et al. Uterine sarcomas and rare uterine mesenchymal tumors with Malignant potential. Diagnostic guidelines of the French Sarcoma Group and the Rare Gynecological Tumors Group. Gynecol Oncol. (2022) 167:373–89. doi: 10.1016/j.ygyno.2022.07.031
9. Dufresne A, Pissaloux D, Ngo C, Penel N, Le Cesne A, Macagno N, et al. Natural history and treatment efficacy in an ambispective case series of NTRK-rearranged mesenchymal tumors. ESMO Open. (2023) 8:101202. doi: 10.1016/j.esmoop.2023.101202
10. Kang N, Zhang Y, Guo S, Chen R, Kong F, Wang S, et al. Genomic and transcriptomic characterization revealed the high sensitivity of targeted therapy and immunotherapy in a subset of endometrial stromal sarcoma. Cancer Res Treat. (2023) 55:2022. doi: 10.4143/crt.2022.1647
11. Xiao G, Li L, Tanzhu G, Liu Z, Gao X, Wan X, et al. Heterogeneity of tumor immune microenvironment of EGFR/ALK-positive tumors versus EGFR/ALK-negative tumors in resected brain metastases from lung adenocarcinoma. J ImmunoTherapy Cancer. (2023) 11:2022–006243. doi: 10.1136/jitc-2022-006243
12. Dang X, Xiang T, Zhao C, Tang H, and Cui P. EML4-NTRK3 fusion cervical sarcoma: A case report and literature review. Front Med (Lausanne). (2022) 9:832376. doi: 10.3389/fmed.2022.832376
13. Rabban JT, Devine WP, Sangoi AR, Poder L, Alvarez E, Davis JL, et al. NTRK fusion cervical sarcoma: a report of three cases, emphasising morphological and immunohistochemical distinction from other uterine sarcomas, including adenosarcoma. Histopathology. (2020) 77:100–1. doi: 10.1111/his.14069
14. Wong DD, Vargas AC, Bonar F, Maclean F, Kattampallil J, Stewart C, et al. NTRK-rearranged mesenchymal tumours: diagnostic challenges, morphological patterns and proposed testing algorithm. Pathology. (2020) 52:401–9. doi: 10.1016/j.pathol.2020.02.004
15. Goulding EA, Morreau P, De Silva M, Watson M, van Vliet C, Leung B, et al. Case report: NTRK1-rearranged cervical sarcoma with fibrosarcoma like morphology presenting in a 13-year-old managed with a neo-adjuvant TRK-inhibitor and surgical excision. Gynecol Oncol Rep. (2021) 37:100845. doi: 10.1016/j.gore.2021.100845
16. Boyle W, Williams A, Sundar S, Yap J, Taniere P, Rehal P, et al. TMP3-NTRK1 rearranged uterine sarcoma: A case report. Case Rep Womens Health. (2020) 28:e00246. doi: 10.1016/j.crwh.2020.e00246
17. Croce S, Hostein I, Longacre TA, Mills AM, Perot G, Devouassoux-Shisheboran M, et al. Uterine and vaginal sarcomas resembling fibrosarcoma: a clinicopathological and molecular analysis of 13 cases showing common NTRK-rearrangements and the description of a COL1A1-PDGFB fusion novel to uterine neoplasms. Mod Pathol. (2019) 32:1008–22. doi: 10.1038/s41379-018-0184-6
18. Devereaux KA, Weiel JJ, Mills AM, Kunder CA, and Longacre TA. Neurofibrosarcoma revisited: an institutional case series of uterine sarcomas harboring kinase-related fusions with report of a novel FGFR1-TACC1 fusion. Am J Surg Pathol. (2021) 45:638–52. doi: 10.1097/PAS.0000000000001644
19. Grant L, Boyle W, Williams S, Pascoe J, and Ganesan R. Uterine neurotrophic tyrosine receptor kinase rearranged spindle cell neoplasms: three cases of an emerging entity. Int J Gynecol Pathol. (2024) 43:326–34. doi: 10.1097/PGP.0000000000000988
20. Huang HJ, Wang C, Fan DG, He YH, Chen X, and Zheng SL. NTRK-rearranged uterine sarcoma: a clinicopathological analysis of seven cases. Zhonghua Bing Li Xue Za Zhi. (2024) 53:189–91. doi: 10.3760/cma.j.cn112151-20230728-00036
21. Michal M, Hajkova V, Skalova A, and Michal M. STRN-NTRK3-rearranged mesenchymal tumor of the uterus: expanding the morphologic spectrum of tumors with NTRK fusions. Am J Surg Pathol. (2019) 43:1152–4. doi: 10.1097/PAS.0000000000001292
22. Mills AM, Karamchandani JR, Vogel H, and Longacre TA. Endocervical fibroblastic Malignant peripheral nerve sheath tumor (neurofibrosarcoma): report of a novel entity possibly related to endocervical CD34 fibrocytes. Am J Surg Pathol. (2011) 35:404–12. doi: 10.1097/PAS.0b013e318208f72e
23. Razack R, Butt JL, Hostein I, Velasco V, Croce S, Olory C, et al. SMARCB1 (INI1) deficient tumours of the uterine cervix: report of two cases, including one associated with an NTRK fusion. Int J Gynecol Pathol. (2024) 43:464–71. doi: 10.1097/PGP.0000000000001014
24. Szalai L, Vereczkey I, Szemes M, Rokusz A, Csernak E, Toth E, et al. NTRK-rearranged spindle cell sarcoma of the uterine cervix with a novel NUMA1::NTRK1 fusion. Virchows Arch. (2024) 484:527–31. doi: 10.1007/s00428-023-03724-1
25. Tsai JW, Lee JC, Hsieh TH, Huang SC, Lee PH, Liu TT, et al. Adult NTRK-rearranged spindle cell neoplasms of the viscera: with an emphasis on rare locations and heterologous elements. Mod Pathol. (2022) 35:911–21. doi: 10.1038/s41379-021-01005-3
26. Wells AE, Mallen AM, Bui MM, Reed DR, and Apte SM. NTRK-1 fusion in endocervical fibroblastic Malignant peripheral nerve sheath tumor marking eligibility for larotrectinib therapy: A case report. Gynecol Oncol Rep. (2019) 28:141–4. doi: 10.1016/j.gore.2019.04.006
27. Lanman T, Hayden Gephart M, Bui N, Toland A, and Nagpal S. Isolated leptomeningeal progression in a patient with NTRK fusion+ Uterine sarcoma: A case report. Case Rep Oncol. (2021) 14:1841–6. doi: 10.1159/000521158
28. Hechtman JF, Benayed R, Hyman DM, Drilon A, Zehir A, Frosina D, et al. Pan-trk immunohistochemistry is an efficient and reliable screen for the detection of NTRK fusions. Am J Surg Pathol. (2017) 41:1547–51. doi: 10.1097/PAS.0000000000000911
29. Moura MS, Costa J, Velasco V, Kommoss F, Oliva E, Le Loarer F, et al. Pan-TRK immunohistochemistry in gynaecological mesenchymal tumours: diagnostic implications and pitfalls. Histopathology. (2024) 84:451–62. doi: 10.1111/his.15082
Keywords: cervix, tumor, NTRK, NGS, prognosis
Citation: Feng L, Li L, He Y and Jiang W (2025) NTRK-rearranged spindle cell neoplasm of the female genital tract: case report and literature review. Front. Oncol. 15:1525722. doi: 10.3389/fonc.2025.1525722
Received: 10 November 2024; Accepted: 08 August 2025;
Published: 27 August 2025.
Edited by:
Yu Ligh Liou, The First Affiliated Hospital of Guangdong Pharmaceutical University, ChinaReviewed by:
Joanna Przybyl, McGill University, CanadaSandrine Oziel-Taieb, Institute Paoli Calmettes, France
Copyright © 2025 Feng, Li, He and Jiang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Wei Jiang, amVydmlzQDEyNi5jb20=
 Lulu Feng
Lulu Feng Lei Li
Lei Li Yanmei He1,2
Yanmei He1,2 Wei Jiang
Wei Jiang