- 1Institute for Studies in Medical History, Persian and Complementary Medicine, Iran University of Medical Sciences, Tehran, Iran
- 2Department of Traditional Medicine, School of Persian Medicine, Iran University of Medical Sciences, Tehran, Iran
- 3Student Research Committee, Iran University of Medical Sciences, Tehran, Iran
- 4Department of Traditional Pharmacy, School of Persian Medicine, Iran University of Medical Sciences, Tehran, Iran
- 5Department of Photodynamic, Medical Laser Research Center, Yara Institute, Academic Center for Education, Culture, and Research (ACECR), Tehran, Iran
Purpose: Tiryāq (Theriac) refers to a single or compound medication historically utilized as a general antidote against numerous poisons in several ethnomedical traditions, especially in traditional Persian medicine (PM). This study aims to summarize the traditional uses, phytochemistry, and pharmacological activities of medicinal plants with tiryāq properties, with a particular focus on their anti-hepatotoxic, hepatoprotective, neuroprotective, and cardioprotective activities.
Methods: Classical texts of traditional PM were broadly reviewed to extract information about tiryāq and its mechanisms. In addition, a detailed search of scientific databases was performed to validate the pharmacological properties of plants traditionally recognized for their antidotal effects.
Results: Thirty-one medicinal plants with antidote properties were identified. The primary function of tiryāq, as described in PM, is to neutralize toxins and bolster the immune system. These plants have cardiotonic, hepatoprotective, and neuroprotective properties. In addition to their antidotal applications, tiryāq remedies were traditionally used to manage chronic cough, stomachache, asthma, colic, and other ailments. Modern pharmacological studies support these applications, highlighting the plants’ antiviral, immunomodulatory, and antioxidant properties, especially against acute respiratory viral infections and other inflammatory circumstances.
Conclusion: Tiryāq plays a pivotal role in fortifying essential organs, including the heart, brain, and liver. Its prophylactic use during epidemics, along with its antioxidant and immune-stimulating properties, underscores its therapeutic potential. Further research is needed to conclusively determine the efficacy and broader therapeutic applications of medicinal plants with tiryāq properties.
1 Introduction
Poisoning is one of the common medical emergencies and represents an important global public health challenge. It comprises the most prevalent causes of morbidity and mortality, which is the second largest reason for morbidity worldwide (Alnasser et al., 2022). Harmful substances and toxins in the air, soil, food, fruits, vegetables or even medicinal and cosmetic products can harm the human body through inhalation, ingestion or direct contact (Alsherbiny et al., 2019). Ingestion is considered one of the main routes of exposure to poisons. Ingested natural or chemical toxins can injure or impair body functions through damage or death of living tissues (Alnasser et al., 2022; Nasiri et al., 2023). Exposure to toxicants has been regarded to be one of the main contributors to disorders, including autoimmune diseases (Pollard et al., 2010), fatty liver disease (Al-Eryani et al., 2015), neural dysfunction-related disorders such as Alzheimer’s disease and Parkinson’s disease, thyroid and ovarian problems, breast cancer, and obesity (Jamshidi et al., 2020).
Antidotes are therapeutic substances that neutralize the toxic effects of a drug, poison or toxin. Antidotes can mediate various mechanisms, including preventing the absorption and binding of poison, neutralizing poison directly, antagonizing the effect of poison on the end-organ, and inhibiting the transformation of poison into more toxic metabolites (Chacko and Peter, 2019; Aruwa et al., 2020). In recent years, the importance of using herbal antidotes in the management of poisonings has been highlighted. The evidence-based benefits of the phyto-antidotes against adverse chemical agents have also been confirmed (Aruwa et al., 2020; Lysiuk et al., 2020). Different studies have revealed antidote and protective effects on natural or chemical toxicities the following administration of medicinal plants and their main constituents, including turmeric, black cumin, milk thistle, cinnamon, barberry and green tea (Alsherbiny et al., 2019). Additionally, recent studies have emphasized the importance of understanding the molecular mechanisms through which these plants exert their antidotal effects, which could further validate their use in modern medicine. The concept of antidotes has a long history in traditional remedies and different substances of plant, animal, and mineral origin were used as antidotes (Magowska, 2021).
In the traditional Persian medicine (PM) textbooks, many materia medica have been introduced called “tiryaq” (equivalent of antidote in current nomenclature), which have played an important role in the treatment of some diseases and ailments, including poisoning (Al-ahwazi, 2007; Avicenna, 2009). Tiryāq is a therapeutic agent that has a plant, mineral or animal origin or a combination of these substances. Meanwhile, medicinal plants have always played an important role as antidotes (Sonneville et al., 2011; Bénéchet et al., 2019). The primary role of the tiryāq in the response to poisoning is to neutralize the toxic effects of toxicants on the human body and normally to create immunity (Aleem et al., 2020). Moreover, it was used for other medical purposes, including management of chronic cough, stomach discomfort, respiratory problems, fever, colic, some neurologic ailments, and so on (Sonneville et al., 2011; Bénéchet et al., 2019; Aleem et al., 2020; Taghizadieh et al., 2020). Tiryāq strengthens the main organs such as the heart (Al-ahwazi, 2007). Other uses of tiryāqs include prophylaxis in epidemics (Ansari et al., 2020; Mahroozade et al., 2021), antioxidant effects (Khan et al., 2020) and stimulating the immune system (Nigar and Itrat, 2013; Khan et al., 2020).
In traditional medicine, the antidote properties of medicinal plants against all kinds of poisons have been mentioned, however, they have been less investigated and identified in pharmacological studies. Many pharmacological studies still lack sufficient scientific rigor or fail to explore the full therapeutic potential of these plants. This study aims to introduce some medicinal plants with antidote properties and their medical uses, according to the main textbooks of traditional PM. Furthermore, this review aims to critically analyze the existing evidence on the pharmacological properties of these plants and discuss the potential gaps in the current research landscape.
2 Methods
In this study, we surveyed the most famous textbooks of ancient traditional PM, including Al-Qānūn fī al-Ṭibb (The Canon of Medicine) by Avicenna (Bénéchet et al., 2019) and the most comprehensive pharmacopeia, namely, Makhzan al-Adwīyah by Aghīlī Shīrazī (Sonneville et al., 2011), Al-Shamil fī al-Ṣinaʿa al-Ṭibbīyah by Ibn al-Nafīs (Nikhat and Fazil, 2023), Tuḥfah al-Muminīn by Muḥammad Mumin Tunkabunī (Salehi et al., 2018), Qarābādīn Kabīr by Aghīlī Shīrazī (Aghili Khorasani, 1970) to find all mentioned medicinal plants with tiryāq properties, their used parts, and their traditional uses and effects. Then, scientific names of the plants were obtained using herbal textbooks. The search terms were the scientific names and common names of each plant combined with “antidote,” “teryāgh,” “tiryāq,” “taryāq,” “teryāghiat,” “pādzahr,” “cardioprotective,” hepatoprotective,” “neuroprotective,” “immunomodulatory” “antioxidant,” “anti-inflammatory,” “antibacterial,” “antiviral” and “antifungal,” “antidote,” “detoxification.” To this end, electronic databases, including PubMed, Scopus, Google Scholar, Web of Science and other relevant ones were searched for studies describing ethnomedicinal uses and pharmacological and phytochemical aspects of the medicinal plant. Data were collected from May 1980 up to May 2023. Only published articles in English language were included in this review. Finally, we collected and classified the items related to the subject.
3 Potential therapeutic roles of antidotal plants
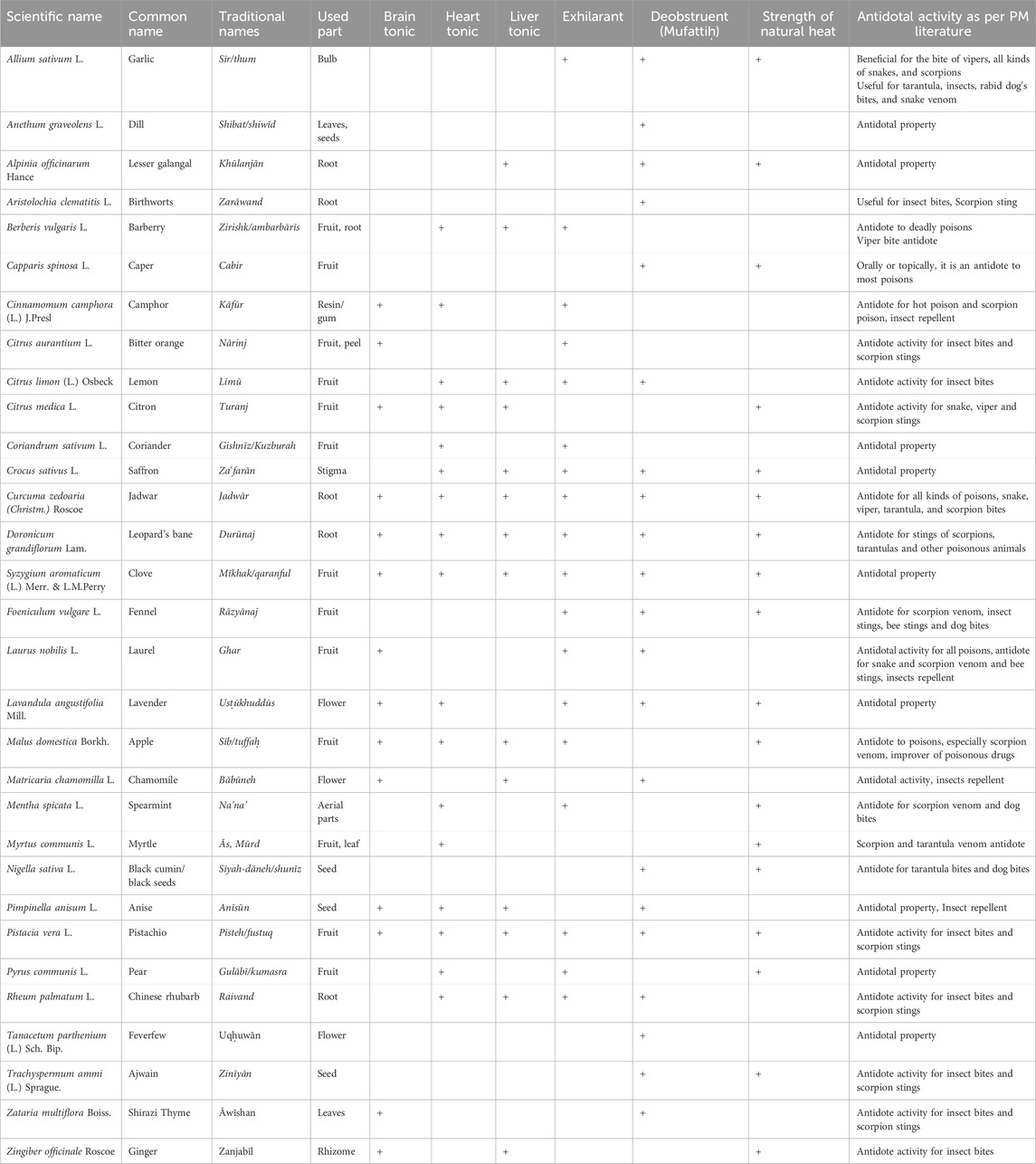
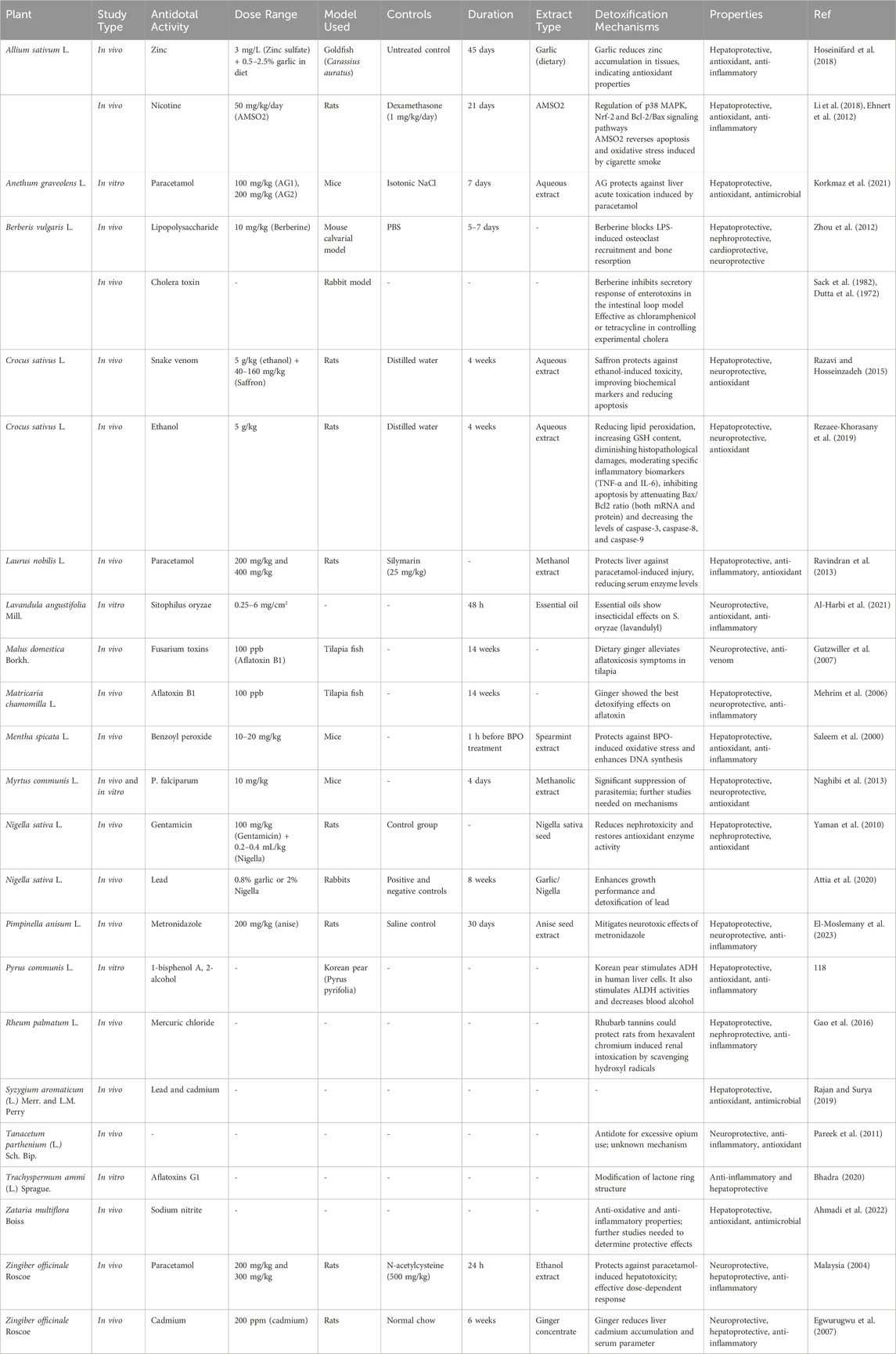
Tiryāq has been widely used in traditional medicine, and one of the most important of them is the antidote of various poisons and the treatment of poisoning (Nikhat and Fazil, 2023; Aghili Khorasani, 2009; Avicenna, 2009). Poison refers to any edible or drinkable substance that is not compatible with human life and after entering the body affects it and as a result causes human fatality by destroying the innate heat (Avicenna, 2009; Tabib Gilani, 2015). The antidote can protect the body against poison/toxic substances that disturb its equilibrium state, as well as expel them that enter the body. Moreover, other applications of an antidote included potential effects in the prevention and even treatment of disorders related to human exposure to contaminated food and air toxic agents (Aghili Khorasani, 2009; Avicenna, 2009). In the traditional PM, various antidotes have been mentioned that have been used individually or in combination. The antidotes act on poisons and remove them from the body or protect against the effects of poisons on the body. The antidote has protective effects on various organs of the human body, including the principal organs (brain, heart, and liver) and strengthens them (Aghili Khorasani, 2009; Avicenna, 2009). Avicenna believed that an antidote is any medicine that has the ability to maintain the strength and health of the soul and through it, the soul repels the harmful effects of the poison (Vaghasloo et al., 2017; Bénéchet et al., 2019; Nasiri et al., 2023). Traditional data on medicinal plants with antidote properties recommended in PM are shown in Table 1. Moreover, their pharmacological activities are summarized in Table 2 and their antidote and detoxification activities are described in Table 3. In this section, we selected ten plants, which people use in abundance such as fennel, nigella, citron, apple, saffron, ajwain, pistachio, garlic, coriander, and ginger. The biological activities of their main phytochemicals in related studies are also discussed.
3.1 Fennel (Foeniculum vulgare L.)
According to the traditional PM, fennel has been used for the treatment of a variety of eye, respiratory, liver and kidney diseases. It has exhilarating properties, and tonic for stomach and eyes. It can strengthen innate heat. Fennel is an antidote to animal poisons and is useful for poisonous animals’ stings such as scorpions, bees and the like. Consumption of fennel decoction is also recommended for rabid dog bites (Aghili Khorasani, 1970; Aghili Khorasani, 2009; Avicenna, 2009). In addition, it has shown protective effects on humor infection and is useful in prolonged fevers (Nikhat and Fazil, 2023). The primary phytoconstituents of this plant have been identified as phenols, phenolic glycosides, and volatile fragrance compounds such as trans-anethole, estragole, and fenchone. Fennel has shown many pharmacological properties, including antifungal, antibacterial, antioxidant, anti-inflammatory, hepatoprotective and neuroprotective (Rahimi and Ardekani, 2012), and antiviral (Orhan et al., 2012) activities. This plant also has immunomodulatory (Darzi et al., 2018), and cardioprotective effects (Natarajan and Grace, 2019). The antioxidant activity of fennel is attributed to phenolic compounds, while the volatile fragrance compounds make it a great flavoring agent (Rather et al., 2016).
3.2 Black seed (Nigella sativa L.)
Nigella sativa L. from the Ranunculaceae family, is considered a widely used medicinal plant all over the world. There are many therapeutic uses of this plant in several traditional medicine systems such as Unani, Ayurveda and Siddha (Ahmad et al., 2013). In traditional PM, black seed is widely used in the treatment of various ailments and diseases. It is considered antidote to cold poisons. Moreover, in combination with lukewarm water, it is useful to treat someone who has been attacked by a rabid dog or tarantula. The smoke from burning black seeds is an effective way to repel insects (Aghili Khorasani, 1970; Aghili Khorasani, 2009). It has been demonstrated that black seed has a wide range of biological activities, including antioxidant, antimutagenic, antidiabetic, analgesic, antibacterial, immunomodulatory, anti-inflammatory, spasmolytic, bronchodilator (Ahmad et al., 2013; Tavakkoli et al., 2017), and antiviral (Esharkawy et al., 2022) properties. The bioactive component, thymoquinone (TQ), present in the essential oil of black seed, is chiefly responsible for these therapeutic effects (Al-Ali et al., 2008).
Numerous animal studies have highlighted the protective effects of black seed and TQ on the heart (Yesmin et al., 2013), brain, lung, kidney and liver have been shown against some toxic agents either natural or chemical toxins (Ahmad et al., 2013; Tavakkoli et al., 2017). Moreover, preclinical research has documented its potential as an antidote for various poisoning, including mycotoxins, endotoxins, metals (Al, lead, mercury, and cadmium), pesticides (imidacloprid, propoxur, acetamiprid, chlorpyrifos, fenitrothion), solvents (ethanol, CCl4, toluene), and environmental pollutants (such as Bisphenol A). However, human studies are still needed to confirm the effectiveness of black seed as an antidote against human poisoning (Tavakkoli et al., 2017).
3.3 Citron (Citrus medica L.)
The citron (Citrus medica L.) belongs to the herbal family of Rutaceae. In Unani and Ayurvedic medicine, citron has been used for therapeutic purposes, the effects of its peel and whole fruit have been mentioned to improve flatulence, and strengthen the stomach and heart (Al-Yahya et al., 2013). Based on traditional PM textbooks, this plant is a heart, liver and brain tonic (Aghili Khorasani, 1970; Aghili Khorasani, 2009; Avicenna, 2009). Moreover, the citron has been introduced as an effective antidote to every kind of poison. Its consumption together with honey repels the harm of all poisons. Drinking citron extract has been used as an antidote against snake bites in humans. Citron oil also has antidote properties. Inhaling citron oil has been considered a remedy for conditions such as air pollution and epidemics (Aghili Khorasani, 2009). The poultice of this plant has been used for scorpion and horned snake bites. Citron seeds are used in all oral poisonings and bites. In all animal poisons, the use of peeled citron seeds orally or as a topical preparation is an alternative to “Tiryāq Fārūq” and is stronger than “Tiryāq Kabīr.” Moreover, its consumption along with hot water has been experienced in scorpion bites. Anointing with citron seed oil will keep the scorpion away (Aghili Khorasani, 2009).
In Greek texts, the use of different parts of citrus fruits as an antidote for “poison and venom” is also mentioned. Taking citron before meals is an antidote for any type of poison and is recommended orally and topically for scorpion and viper bites. Regarding its efficacy, it has been stated that citron peel perfumes the breath and its scent calms the spirit (Arias and Ramón-Laca, 2005). This plant has antioxidant, anti-inflammatory, antimutagenic, antibacterial, antifungal, antiviral (El Hawary et al., 2022), immunostimulatory (Peng et al., 2019), cardioprotective (Al-Yahya et al., 2013), hepatoprotective, and neuroprotective activities (Pooja et al., 2021). Citron is known as a potent antioxidant due to the presence of bioactive components, mainly phenolics, flavanones, vitamin C, and pectin. The main components of fruit peel oil include iso-limonene, citral, and limonene, and the fruit is rich in vitamins and minerals, especially vitamin A, vitamin C, niacin, and thiamin (Chhikara et al., 2018). In an animal study, methanolic extract of citrus peel showed a protective effect on acute cyanide poisoning-induced seizures and oxidative stress. These neuroprotective effects may be explained by inhibiting the excessive production of oxidative stress as well as maintaining antioxidant defense mechanisms (Abdel Moneim, 2014).
3.4 Apple (Malus domestica Borkh.)
Apple is one of the nutrient-rich and most popular fruits. This fruit has many benefits for the human body in terms of having a lot of vitamin C, fiber, antioxidants, etc. (Acquavia et al., 2021). From the viewpoint of traditional PM, it is very effective in treating poisons, especially scorpion venom. It also has a protective role in epidemics and air pollution and is an ameliorator of toxic drugs. It is recommended to use apple or its leaf extract for scorpion venom or other poisons, either when drunk or applied externally (Aghili Khorasani, 2009), In addition to the fruit, its leaf extract also has these properties (Avicenna, 2009). Sour apple is astringent, and relieves vomiting and thirst (Aghili Khorasani, 1970; Aghili Khorasani, 2009). This fruit is tonic for the brain, heart and liver, and also has exhilarant properties (Aghili Khorasani, 2009). Apples are highly consumed and nutritious fruit and contain various bioactive compounds, including pectins, dietary fibers, vitamins, oligosaccharides, triterpenic acids, and phenolic compounds. The high antioxidant properties of apples are due to the higher content of these phenolic compounds (Asma et al., 2023). There is now substantial scientific evidence that these bioactive compounds found in apples and their peels have the potential to promote human health by reducing the risk of cardiovascular disease, diabetes mellitus, inflammation, and cancer (Patocka et al., 2020). Some studies have also shown that apple has immunity enhancement, anti-inflammatory, antioxidant, antibacterial, cardioprotective, hepatoprotective (Patocka et al., 2020), and neuroprotective effects (Lu et al., 2019).
3.5 Ajwain (Trachyspermum ammi (L.) Sprague.)
Trachyspermum ammi (L.) Sprague. belongs to Apiaceae family (Zarshenas et al., 2013). Ajwain has been traditionally administered for the management of various ailments such as febrile conditions, cough, respiratory distress, fatigue, neuropathic and chronic pains (Kamalinejad et al., 2021). Ajwain has protective effects against cardiovascular issues, hepatotoxicity, and oxidative stress, showing promise in treating various types of poisoning. In the traditional PM, the antidote potency is explained to Ajwin. Its use is considered an antidote to poisons and insect and animal bites. Consumption of ajwain is useful in eliminating the harms of opium use and also for quitting. A medicinal rinse of ajwain is fast-acting to relieve scorpion venom (Aghili Khorasani, 2009). It is very useful in chronic fevers (Avicenna, 2009). Thymol is the main component in ajwain, which has antiseptic, antifungal, antibacterial, antioxidant, and anti-inflammatory properties. In addition, thymol has the ability to reduce the levels of C-reactive protein (CRP), Interleukin-1 beta (IL-1β), IL-6, tumor necrosis factor-alpha (TNF-α), TNF-β, and matrix metalloproteinase 9 (MMP9) (Korani and Jamshidi, 2020). Administration of dietary ajwain extract in rat models could attenuate the hexachlorocyclohexane-induced oxidative stress and hepatotoxicity (Anilakumar et al., 2009). The aqueous extract of ajwain showed antimicrobial effects on several bacterial strains, including Enterococcus faecalis, Staphylococcus aureus, Escherichia coli, P. aeruginosa, S. typhimurium, and Shigella flexneri (Kaur and Arora, 2009). It has protective effects on the cardiovascular system, including calcium channel blocking effect, which leads to a decrease in heart rate and blood pressure, positive ionotropic and negative chronotropic effects, cholinomimetic effects, which could cause bradycardia, lipid-lowering effect, and protective effect on body weight gain (Boskabady et al., 2014). Antifungal activity of different types of ajwain extract is also documented against Aspergillus species, Epidermophyton floccosum, Microsporum canis, Trichophyton mentagrophytes, Candida albicans, and C. utilis (Boskabady et al., 2014). Due to the calcium channel blocking effect, ajwain seed extract showed antihypertensive, antispasmodic, bronchodilator and hepatoprotective activities. Oral administration of ajwain in an animal model of liver injury induced by carbon tetrachloride (CCL4) and lethal dose of paracetamol (1 g/kg), showed protective effects on paracetamol- and CCL4-induced hepatic injuries and reduced liver enzymes (Gilani et al., 2005).
3.6 Garlic (Allium sativum L.)
Garlic (Allium sativum L.) is one of the most widely used medicinal plants for flavor and spice, which has many health benefits. It belongs to the Liliaceae family (Bhandari, 2012; Verma et al., 2023). This medicinal plant and its components have capabilities such as scavenging free radicals, anti-inflammatory, anti-cholesterol, anti-gastric ulcer, anti-bacterial, anti-cancer and antioxidant (Koscielny et al., 1999).
In traditional PM, garlic has antidote and exhilarant properties (Aghili Khorasani, 2009; Avicenna, 2009), and repels damage caused by air pollution, epidemics and incoherent waters (Aghili Khorasani, 2009). The use of large quantities of garlic and wine is of great benefit in the bites of vipers, snakes, and scorpions, to a point where there is almost no equal. If it is applied to the bite site, it is of great benefit by absorbing the poison (Nikhat and Fazil, 2023). Consumption of garlic is also useful for tarantula, insects, rabid dog’s bites, and snake venom. Garlic alone or in combination with fig leaves, cumin, olive oil and wine has been used to absorb all the toxins and relieve the discomfort associated with them (Aghili Khorasani, 2009).
Garlic consists of sulfur-containing plant compounds such as alliin, allicin, ajoenes, vinyldithiins, and flavonoid compounds such as quercetin. Extracts and isolated compounds of garlic have shown various pharmacological activities, including antibacterial, antiviral, antifungal, antioxidant, anti-inflammatory, and anticancer, antidiabetic, antihypertensive, anti-obesity, and antithrombotic activities (El-Saber Batiha et al., 2020). According to the available evidence, garlic can be considered an antidote or protective plant against many toxic agents. This plant has the ability to modulate the activity of several metabolizing enzymes. A number of in vitro and animal pre-clinical studies showed protective effects of garlic and its major components against cardiotoxicity, hematotoxicity, neurotoxicity, hepatotoxicity, nephrotoxicity, intestinal toxicity, pulmonary toxicity, bone marrow toxicity, and reproductive toxicity/teratogenicity induced by natural and chemical agents. The protective effect of garlic against natural and chemical toxins is attributed to its various properties, including free radical scavenging, antioxidant effect, reduction of lipid peroxidation, anti-inflammatory effect, a chelating agent in heavy metal poisoning, cytoprotective activities, increased protein production in damaged tissues, and suppression of apoptosis (Dorrigiv et al., 2020). Phospholipase A2 (PLA2) enzyme is one of the major components of snake venom. The antidotal effects of garlic against snake venom have been demonstrated through inhibition or inactivation of the PLA2 enzyme (Asad et al., 2013; Asad et al., 2014).
3.7 Pistachio (Pistacia vera L.)
Pistachio belongs to the Anacardiaceae family. In traditional PM, pistachio has been used for different ailments. It is considered a mind, brain, heart, liver and stomach tonic. Consuming pistachio alone or with wine is useful for getting rid of scorpion poisons and other insect bites (Aghili Khorasani, 2009). Especially, the decoction of pistachio in wine is very beneficial for insect bites (Avicenna, 2009). In other words, Pistachio is an antidote to insect bites and poisons (Tonekaboni, 2007). Also, consuming pistachio along with sugar improves the side effects of air pollution (Aghili Khorasani, 2009). Based on modern phytotherapy, it has antioxidant, anti-inflammatory, cardioprotective (Ersöz, 2023), hepatoprotective (Iranmanesh et al., 2017), antimicrobial, antiviral, antifungal and neuroprotective activities (Mandalari et al., 2021). Pistachio is considered a rich source of protein, fiber, monounsaturated fatty acids, minerals (copper, manganese, potassium, phosphorous, chromium, magnesium, iron, zinc, and selenium) and vitamins (vitamin B6, thiamin, vitamins E,riboflavin, and folate) as well as carotenoids, phenolic acids, flavonoids and anthocyanins (Mandalari et al., 2021).
Pistachio kernel contains antioxidants such as tocopherols, phylloquinone, carotenoids, chlorophyll and flavonoids, therefore pistachio is among the foods with high total antioxidant capacity (Liu et al., 2014). Some of the beneficial properties of pistachio can be partially related to its antioxidant metabolites (Gentile et al., 2007). In an animal study investigating the cardioprotective effect of pistachio skin extract, it was shown that this extract has antioxidant activity and can reduce apoptosis and deoxyribonucleic acid damage in heart damage (Ersöz, 2023). A recent systematic review and meta-analysis showed that pistachio has a beneficial effect on some cardiometabolic risk factors. Based on the findings of 11 clinical trials, pistachio consumption significantly reduced blood glucose (FBS and HbA1C), serum lipids, systolic blood pressure, and inflammatory biomarkers (Ghanavati et al., 2020). In an animal study, it was shown that treatment with pistachio supplementation eliminated spatial memory disorders due to neurotoxicity of cisplatin and vincristine, and improved cognitive and motor functions (Golchin et al., 2015). Other animal studies have shown the neuroprotective potential of pistachio, including reducing anxiety-like behavior, improving memory, cognitive and motor impairments, and increasing working memory and physical power (Mandalari et al., 2021). Pistachio had shown bactericidal effects on a wide range of Gram-positive bacteria, including Listeria monocytogenes, S. aureus and methicillin-resistant clinical isolates of S. aureus (MRSA) (Bisignano et al., 2013). Chronic gavage of pistachio hydro-alcoholic extract showed hepatoprotective activity against experimentally induced liver damage by CCL4 in rats. Treatment of different doses of pistachio extract decreased AST and ALT levels as well as plasma LDL concentration (Iranmanesh et al., 2017). Polyphenols from pistachio showed anti-herpetic effects on herpes simplex virus type 1 (HSV-1). Treatment with polyphenol-rich extracts of natural shelled (NPRE) pistachio kernels could reduce the expression of the viral proteins and viral DNA synthesis (Musarra-Pizzo et al., 2020).
3.8 Saffron (Crocus sativus L.)
According to the traditional PM, saffron has an exhilarant property and is a tonic for heart and liver and innate heat (Aghili Khorasani, 2009; Avicenna, 2009). In addition, it was the reformer of phlegm infection and prevents and protects it from change and corruption (Aghili Khorasani, 1970; Aghili Khorasani, 2009). Saffron, obtained from the dried stigma of Crocus sativus L is used as a spice, coloring and flavoring agent in foods and cosmetics worldwide (Xing et al., 2021). Biologically active metabolites present in this plant such as carotenoids, flavonoids, terpenoids, amino acids, glycosides, starch, mineral matter and alkaloids have shown potential medicinal properties (Bukhari et al., 2018; Xing et al., 2021; Abu-Izneid et al., 2022). In modern pharmacological studies, these components exhibited several properties such as anti-inflammatory, antioxidant, anxiolytic (Bukhari et al., 2018), antiviral, antitumor, antianxiety, hypoglycemic, hypolipidemic, memory-enhancing, immunomodulatory (Xing et al., 2021), antidepressant, antitussive, chelating metal, antifungal (Ouahhoud et al., 2021), cardioprotective (Bukhari et al., 2018; Xing et al., 2021), neuroprotective (Xing et al., 2021), hepatoprotective (Ouahhoud et al., 2021), and antibacterial (Naim et al., 2022) activities. Saffron and its active metabolites have demonstrated cardiac protective effects through modulating oxidative stress, inflammation, blood pressure, lipid and glycemic profiles or direct anti-atherogenic effects (Kadoglou et al., 2021). Saffron’s antioxidant activity, largely attributed to safranal, crocin, and carotene, plays a major role in its protective effects against oxidative stress, inflammation, and cardiovascular diseases (Bukhari et al., 2018; Abu-Izneid et al., 2022). The protective effects of phenolic metabolites in saffron petal extract against bacteria that potentially cause food-borne diseases were evaluated. Based on the findings, these phenolic compounds showed antibacterial effects on S. aureus, Salmonella typhimurium, E. coli and Listeria monocytogenes (Naim et al., 2022).
3.9 Coriander (Coriandrum sativum L.)
Coriander is a popular medicinal plant that belongs to Apiaceae family. It has several biological activities, including antioxidant, hypoglycemic, hypolipidemic, analgesic, anti-inflammatory, anti-cancer, immunomodulatory (Laribi et al., 2015), antiviral (Perera et al., 2022) antifungal, antibacterial, hepatoprotective and lead-detoxifying effects (Sahib et al., 2013). Moreover, neuroprotective effects such as anxiolytic, sedative-hypnotic and anticonvulsant activities were reported for the seeds and leaves of the plant (Jakubczyk et al., 2020). Several cardiovascular benefits of this botanical drug such as antihypertensive, anti-atherogenic, antiarrhythmic as well as cardioprotective effects have been mentioned (Mahleyuddin et al., 2021). In PM, Coriander has exhilarant properties and is a brain and heart tonic (Aghili Khorasani, 1970; Aghili Khorasani, 2009). Coriander volatile metabolites were evaluated for in vitro and in vivo antioxidant activity and hepatoprotective effects on carbon tetrachloride damage. In vitro antioxidant activity as free radical scavenging capacity, as inhibitory activity of essential oils on 2,2-diphenyl-1-picrylhydrazyl (DPPH) and OH radicals and effects on lipid peroxidation (LP) was measured. Its essential oils could reduce stable DPPH radicals in a dose-dependent manner and neutralize H2O2 (Pandey et al., 2011). The essential oil and aqueous extract of coriander leaves showed inhibitory activity against Bacillus subtilis, S. aureus, Klebsiella pneumonia and a pathogenic fungus, C. albicans (Foudah et al., 2021).
3.10 Ginger (Zingiber officinale Roscoe)
Ginger belongs to the Zingiberaceae family (Mao et al., 2019). According to traditional PM, ginger strengthens memory, digestion, stomach, and it is liver and brain tonic (Aghili Khorasani, 1970; Aghili Khorasani, 2009). In addition, this plant strengthens the natural heat (Aghili Khorasani, 2009). Ginger is also useful for removing animal toxins (Aghili Khorasani, 1970; Aghili Khorasani, 2009) and insect bites (Avicenna, 2009).
Accumulated studies have revealed that ginger indicates many biological activities, such as antibacterial (Sebiomo et al., 2011), antifungal (Nerilo et al., 2020), antiviral (Kaushik et al., 2020), immunomodulatory (Harun and Mohamad, 2022), anti-inflammatory (Abdi et al., 2021), antioxidant activity, cardiovascular protective (Abdi et al., 2021), neuroprotective (Yousuf et al., 2023), and hepatoprotective effects (Fahmi et al., 2019). Long-term studies have shown that ginger and its various active compounds have anti-inflammatory activity. It was initially suggested that the anti-inflammatory activity of ginger is mainly related to its inhibitory effect on prostaglandin and leukotriene synthesis. Both fresh ginger (consisting mainly of gingerols) and dried ginger extracts (the main source of shogaols) inhibit lipopolysaccharide (LPS)-induced prostaglandin E2 (PGE2) production (Ozkur et al., 2022). Phenolic metabolites in ginger, such as gingerol, have a protective effect on ischemic heart disease in rats (Dissanayake et al., 2020). Ginger plant extract can modulate immune responses that intensify the inflammation process at the cellular level. The active components of this plant have antioxidant activity and have effects similar to non-steroidal anti-inflammatory drugs and stop the metabolism of arachidonic acid by inhibiting cyclooxygenase and lipoxygenase pathways (Dissanayake et al., 2020). A botanical drug derived from fresh ginger rhizome has been proven to have an antiviral effect on human respiratory syncytial virus (HRSV) infection by reducing HRSV-induced plaque formation in respiratory mucosal cell lines. Therefore, high concentrations of ginger can stimulate mucosal cells to secrete IFN-β, which helps fight viral infections by reducing virus adhesion and internalization. This effect is very useful in the management of colds and fever with mucous secretions and the management of complications from cough and asthmatic conditions. Lyophilized water extract of ginger has an antiviral effect on hepatitis C virus infection. In a specific study, ginger has been proven to inhibit viral replication within HepG2 cells infected with hepatitis C virus by affecting viral RNA, and another study detailed that ginger is effective in reducing these cells. Hepatitis C virus loads, α-fetoprotein level and markers related to liver function such as aspartate aminotransferase (AST) and alanine aminotransferase (ALT) are prescribed in HCV patients (Dissanayake et al., 2020).
4 Future perspectives
The high toxicity of various modern chemicals and their pervasive potency to pollute the environment and diffuse within the human body necessitate effective preventive measures against acute and chronic poisoning. One such approach is the investigation of antitoxins derived from natural sources, especially medicinal plants, traditionally utilized for therapeutic purposes across various cultures.
In this regard, Sardari et al. investigated the therapeutic properties of caper plant, which is one of the types of antidotes. The caper plant, whose scientific name is Capparis spinosa, is one of the prominent examples of anti-toxic food. This plant affects a wide range of different body systems, including the digestive system, nervous system, respiratory system, spleen, and reproductive system. It can serve as an antidote for most poisons (Sardari et al., 2018). “Tiryaq Arba” has been introduced as a polyherbal formulation that has the potential to prevent acute respiratory infections. They showed that it has antiviral properties against different viruses, including coronavirus, adenovirus, respiratory syncytial virus, and para-influenza virus. Additionally, this antidote is an excellent medicine in epidemics. It is one of the heart- and brain-tonic and protective drugs. It is also used as a solvent and antidote in acute respiratory infections and is a potential prophylactic drug used for COVID-19 (Ansari et al., 2020). In another study, Khan et al. introduced Tiryaq-e-Wabai for prophylaxis during cholera, plague, and other epidemics, which also has antioxidant and immune system-stimulating effects. The properties of the plants that make up this antidote have already been investigated in an animal model, and their antiviral, antitussive, and expectorant activities are a solid basis for the use of prophylaxis in COVID-19 (Khan et al., 2020). Kalam et al. investigated the various therapeutic effects of Tiryaq-e-Wabai compounds and stated that Tiryaq-e-Wabai was used as an antidote, prophylaxis, anti-epidemic, and poisoning caused by bites (Kalam et al., 2020).
Emami et al. examined the opinions of Iranian medical sages about the nutrition of the elderly, and stated that an antidote is one of the recommended foods or medicines in PM, which is made from abundant amounts of natural substances and strengthens the heart and stomach, senses and stimulates the appetite (Emami et al., 2014). In a study conducted by Shamsi-Baghbanan et al., liver-protecting plants from the point of view of Ibn Sina and introduced Teriyaq Kabir as one of the combined drugs that protect the liver (Shamsi-Baghbanan et al., 2014). In another study conducted by Nigar and Itrat at the Greek Medicine Hospital in Bangalore, the effectiveness of an antidote as an immune system stimulant in the elderly (Nigar and Itrat, 2013).
Similarly, Teriyaq Kabir has been highlighted for its liver-protective properties (Emami et al., 2014). Clinical trials have further substantiated the immune-boosting potential of these antidotes, particularly in vulnerable populations such as the elderly (Nigar and Itrat, 2013). The present study investigated the mechanisms of medicinal plants with possible beneficial effects on poisoning and related organs based on the suggestions of PM. Key pathways include modulation of NF-κB and Nrf2 signaling, balancing pro-inflammatory and anti-inflammatory cytokines, and activating enzymatic and non-enzymatic antioxidant defenses. These mechanisms provide broad-spectrum protection across various tissues, consistent with PM’s holistic view that treatments should enhance overall health rather than targeting isolated organs (Bahramsoltani and Rahimi, 2020). For example, the role of pro-inflammatory cytokines (IL-1β, IL-6, and TNF-α) is well-documented in poisoning cases. Exposure to toxins like lead and carbon monoxide can induce chronic inflammation, oxidative stress, and damage to organs like the liver. Medicinal plants counteract these effects by reducing inflammation and oxidative stress, as demonstrated in studies on compounds like garlic polysaccharides, which suppress the expression of inflammatory factors (Feldmann et al., 2020; Shao et al., 2020).
The primary advantages of plant-based antidotes include their affordability, broad-spectrum efficacy, and minimal side effects (Lysiuk et al., 2020; Kenari et al., 2021). Edible medicinal plants, such as apple, lemon, saffron, pear, and pistachio, are particularly noteworthy for their safety, antioxidant properties, and ability to modulate the immune system. These plants also exhibit protective effects on vital organs such as the brain, heart, and liver through mechanisms such as reducing inflammation and enhancing cellular defense pathways. Their widespread daily use underscores their potential for therapeutic application. Emerging evidence highlights the potential of specific plants for targeted therapies. For instance, pistachio’s cardioprotective and neuroprotective activities are linked to its rich antioxidant composition (Ersöz et al., 2023). Active components of saffron, e.g., crocin and safranal, are effective in modulating oxidative stress and inflammation, as well as protecting against cardiovascular and neurological damage (Xing et al., 2021; Abu-Izneid et al., 2022). Similarly, ginger’s bioactive compounds inhibit inflammatory pathways and show antiviral effects on hepatitis C and respiratory syncytial viruses (Dissanayake et al., 2020).
The protective effects of these plants align with PM’s concept of enhancing the body’s “instinctive heat” or internal force as a means to combat illness. By stimulating cellular defense mechanisms, these plants not only prevent damage but also promote recovery, offering a comprehensive strategy for managing poisoning and its complications. The study’s limitations consist of a reliance on preclinical evidence, variability in plant preparation methods, studies with small sample sizes, limited demographic diversity, and the lack of standardized, placebo-controlled clinical trials in some studies. Additionally, long-term safety and efficacy data are insufficient, emphasizing the need for rigorous future research to validate these findings.
5 Conclusion
Medicinal plants suggest a promising attitude for developing safe and effective antidotes, indicating a natural resource in modern pharmacotherapy. Although traditional and contemporary evidence underscores their therapeutic potential, it is essential to address the existing methodological and translational gaps to fully shed light into their beneficial and detrimental effects. Future research that integrates the holistic principles of phytomedicine with rigorous scientific methodologies may revolutionize the management of poisoning and related circumstances. Also, systematic investigations into these plants and their active metabolites should be prioritized to identify novel therapeutic agents. Such studies may provide a deeper understanding of the pharmacological properties of these botanicals, paving the way for therapeutic innovations. Clinical trials are critical to validating the efficacy and safety profiles of these compounds, ensuring that they meet the rigorous standards required for therapeutic use. Additionally, mechanistic studies will further elucidate the molecular targets and pathways involved, enhancing our understanding of how these natural products exert their therapeutic effects. By bridging the gap between traditional and modern medicine, we can unravel the full potential of medicinal plants, ultimately resulting in improved health outcomes and novel strategies for managing poisoning and other related conditions. The integration of interdisciplinary approaches will be essential in advancing this field, fostering collaborations between ethnobotanists, pharmacologists, and clinicians to translate these findings into clinical practice.
Author contributions
HM: Investigation, Conceptualization, Methodology, Writing – original draft. MD: Writing – original draft, Supervision, Writing – review and editing. BY: Writing – review and editing. FS: Writing – review and editing. FE: Writing – review and editing, Investigation, Supervision.
Funding
The author(s) declare that no financial support was received for the research and/or publication of this article.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
Abd-Elkareem, M., Abd El-Rahman, M. A., Khalil, N. S. A., and Amer, A. S. (2021). Antioxidant and cytoprotective effects of Nigella sativa L. seeds on the testis of monosodium glutamate challenged rats. Sci. Rep. 11 (1), 13519. doi:10.1038/s41598-021-92977-4
Abdel Moneim, A. E. (2014). Citrus peel extract attenuates acute cyanide poisoning-induced seizures and oxidative stress in rats. CNS Neurol. Disord. Drug Targets 13 (4), 638–646. doi:10.2174/1871527312666131206095142
Abdi, T., Mahmoudabady, M., Marzouni, H. Z., Niazmand, S., and Khazaei, M. (2021). Ginger (Zingiber Officinale Roscoe) extract protects the heart against inflammation and fibrosis in diabetic rats. Can. J. Diabetes 45 (3), 220–227. doi:10.1016/j.jcjd.2020.08.102
Abu-Izneid, T., Rauf, A., Khalil, A. A., Olatunde, A., Khalid, A., Alhumaydhi, F. A., et al. (2022). Nutritional and health beneficial properties of saffron (Crocus sativus L): a comprehensive review. Crit. Rev. food Sci. Nutr. 62 (10), 2683–2706. doi:10.1080/10408398.2020.1857682
Acquavia, M. A., Pascale, R., Foti, L., Carlucci, G., Scrano, L., Martelli, G., et al. (2021). Analytical methods for extraction and identification of primary and secondary metabolites of apple (Malus domestica) fruits: a review. Separations 8 (7), 91. doi:10.3390/separations8070091
Aghili Khorasani, M. (2009). Makhzan-ol-Adviyah [storehouse of medicaments]. Tehran: Tehran University of Medical Sciences.
Aghili Khorasani, S. M. (1970). Qarabadin-e-Kabir [great pharmacopeia]. 1st ed. Tehran: Mahmoudi Press.
Ahmad, A., Husain, A., Mujeeb, M., Khan, S. A., Najmi, A. K., Siddique, N. A., et al. (2013). A review on therapeutic potential of Nigella sativa: a miracle herb. Asian Pac. J. Trop. Biomed. 3 (5), 337–352. doi:10.1016/S2221-1691(13)60075-1
Ahmadi, F., Monfared, A. L., and Shakarami, N. J. A. J. O.P. (2022). Protective effect of Zataria multiflora Boiss against sodium nitrite-induced hepatotoxicity in rats. 12(3), 213.
Ajarem, J. S., Maodaa, S. N., El-Serehy, H. A., Altoom, N., Allam, A. A., Hernandez-Bautista, R., et al. (2021). Anethum graveolens prevents liver and kidney injury, oxidative stress and inflammation in mice exposed to nicotine perinatally. Coatings 11 (7), 838. doi:10.3390/coatings11070838
Al-ahwazi, A. (2007). Kamel al-sanae al-tibb. Qom: Institute of Natural Medicine’s Revival, Jalal-aldin Publication.
Al-Ali, A., Alkhawajah, A. A., Randhawa, M. A., and Shaikh, N. A. (2008). Oral and intraperitoneal LD50 of thymoquinone, an active principle of Nigella sativa, in mice and rats. J. Ayub Med. Coll. Abbottabad 20 (2), 25–27.
Aleem, M., Khan, M. I., Danish, M., and Ahmad, A. (2020). History and traditional uses of tiryaq (theriac): an important formulation in Unani medicine. J. Phytopharm. 9, 429–432. doi:10.31254/phyto.2020.9608
Al-Eryani, L., Wahlang, B., Falkner, K., Guardiola, J., Clair, H., Prough, R., et al. (2015). Identification of environmental chemicals associated with the development of toxicant-associated fatty liver disease in rodents. Toxicol. Pathol. 43 (4), 482–497. doi:10.1177/0192623314549960
Al-Harbi, N. A., Al Attar, N. M., Hikal, D. M., Mohamed, S. E., Abdel Latef, A. A. H., Ibrahim, A. A., et al. (2021). Evaluation of insecticidal effects of plants essential oils extracted from basil, black seeds and lavender against Sitophilus oryzae. Plants (Basel). 10 (5), 829. doi:10.3390/plants10050829
Alnasser, S. M., Kordi, T. S., Asiri, A. A., Gupta, D. K., Alfadl, A. A., and Hussain, A. S. M. (2022). Epidemiology of chemical poisoning among adults in qassim region: an eight-year study. Toxics 10 (11), 709. doi:10.3390/toxics10110709
Alsherbiny, M. A., Abd-Elsalam, W. H., El badawy, S. A., Taher, E., Fares, M., Torres, A., et al. (2019). Ameliorative and protective effects of ginger and its main constituents against natural, chemical and radiation-induced toxicities: a comprehensive review. Food Chem. Toxicol. 123, 72–97. doi:10.1016/j.fct.2018.10.048
AlTamimi, S., El-Banna, H. A., Elzorba, H. Y., and Hassan, N. F. (2020). The potential cardioprotective effect of Matricaria chamomilla extract against diabetes-induced oxidative stress in rats. Farmacia 68, 269–279. doi:10.31925/farmacia.2020.2.12
Al-Yahya, M. A., Mothana, R. A., Al-Said, M. S., El-Tahir, K. E., Al-Sohaibani, M., and Rafatullah, S. (2013). Citrus medica “Otroj”: attenuates oxidative stress and cardiac dysrhythmia in isoproterenol-induced cardiomyopathy in rats. Nutrients 5 (11), 4269–4283. doi:10.3390/nu5114269
Amalraj, A., Kuttappan, S., and Varma, K. (2022). Chemistry, biological activities and therapeutic applications of medicinal plants in Ayurveda. London: The Royal Society of Chemistry. Available online at: https://books.google.com/books?hl=en&lr=&id=Vb2cEAAAQBAJ&oi=fnd&pg=PP1&dq=Amalraj,+A.,+Kuttappan,+S.,+and+Varma,+K.+(2022).+Chemistry,+biological+activities+and+therapeutic+applications+of+medicinal+plants+in+Ayurveda.+The+Royal+Society+of+Q15+Chemistry.&ots=Q5e3oERKTs&sig=kw4C3aDmzWSvGN7v_4hPfUQI4eA#v=onepage&q=Amalraj%2C%20A.%2C%20Kuttappan%2C%20S.%2C%20and%20Varma%2C%20K.%20(2022).%20Chemistry%2C%20biological%20activities%20and%20therapeutic%20applications%20of%20medicinal%20plants%20in%20Ayurveda.%20The%20Royal%20Society%20of%20Q15%20Chemistry.&f=false
Amir Rawa, M. S., Mazlan, M. K. N., Ahmad, R., Nogawa, T., and Wahab, H. A. (2022). Roles of syzygium in anti-cholinesterase, anti-diabetic, anti-inflammatory, and antioxidant: from alzheimer’s perspective. Plants 11 (11), 1476. doi:10.3390/plants11111476
Amrullah, A., and Fachrial, E. (2021). Cardiac protection activity of ethanol extract of white curcumin (Curcuma zedoaria), against cyclophosphamid induced cardiovascular complications in male rat. J. Pharm. Res. Int. 33 (41A), 248–256. doi:10.9734/jpri/2021/v33i41a32324
Andrys, D., Kulpa, D., Grzeszczuk, M., Bihun, M., and Dobrowolska, A. (2017). Antioxidant and antimicrobial activities of Lavandula angustifolia Mill. field-grown and propagated in vitro. Folia Hortic. 29 (2), 161–180. doi:10.1515/fhort-2017-0016
Anilakumar, K., Saritha, V., Khanum, F., and Bawa, A. (2009). Ameliorative effect of ajwain extract on hexachlorocyclohexane-induced lipid peroxidation in rat liver. Food Chem. Toxicol. 47 (2), 279–282. doi:10.1016/j.fct.2008.09.061
Ansari, S., Ahmad, I., Ali, M., and Maaz, M. (2020). “Tiryaq arba”(a polyherbal Unani formulation) as prophylactic medicine against epidemics of acute respiratory viral infections. Middle East J. Rehabilitation Health Stud. 7 (3). doi:10.5812/mejrh.102965
Anser, H., Imam, S., Khatoon, H., Sarfaraz, S., Iffat, W., Fatima, S., et al. (2021). Determination of anxiolytic and antidepressant like activity of hydro alcoholic fruit extract of Pyrus communis (L.) and its impact on the memory after chronic dosing in animal models. Pak. J. Pharm. Sci. 34, 1261–1269. doi:10.36721/PJPS.2021.34.3.SP.1261-1269.1
Arab, Z., Hosseini, M., Marefati, N., Beheshti, F., Anaeigoudari, A., Sadeghnia, H. R., et al. (2022). “Neuroprotective and memory enhancing effects of Zataria multiflora in lipopolysaccharide-treated rats,” in Veterinary research forum: faculty of veterinary medicine (Urmia, Iran: Urmia University), 101.
Arcusa, R., Villaño, D., Marhuenda, J., Cano, M., Cerdà, B., and Zafrilla, P. (2022). Potential role of ginger (Zingiber officinale Roscoe) in the prevention of neurodegenerative diseases. Front. Nutr. 374. doi:10.3389/fnut.2022.809621
Arias, B. A., and Ramón-Laca, L. (2005). Pharmacological properties of citrus and their ancient and medieval uses in the Mediterranean region. J. Ethnopharmacol. 97 (1), 89–95. doi:10.1016/j.jep.2004.10.019
Aruwa, C. E., Mukaila, Y. O., Ajao, A.A.-N., and Sabiu, S. (2020). An appraisal of antidotes’ effectiveness: evidence of the use of phyto-antidotes and biotechnological advancements. Molecules 25 (7), 1516. doi:10.3390/molecules25071516
Asad, M. H., Durr, E. S., Yaqab, T., Murtaza, G., Hussain, M. S., Hussain, M. S., et al. (2014). Phospholipases A2: enzymatic assay for snake venom (Naja naja karachiensis) with their neutralization by medicinal plants of Pakistan. Acta Pol. Pharm. 71 (4), 625–630.
Asad, M. H. H. B., Razi, M. T., Najamus-Saqib, Q., Nasim, S. J., Murtaza, G., Hussain, I., et al. (2013). Anti-venom potential of Pakistani medicinal plants: inhibition of anticoagulation activity of Naja naja karachiensis toxin. Curr. Sci., 1419–1424.
Asadi, Z., Ghazanfari, T., and Hatami, H. (2020). Anti-inflammatory effects of Matricaria chamomilla extracts on BALB/c mice macrophages and lymphocytes. Iran. J. Allergy, Asthma Immunol. 19, 63–73. doi:10.18502/ijaai.v19i(s1.r1).2862
Asma, U., Morozova, K., Ferrentino, G., and Scampicchio, M. (2023). Apples and apple by-products: antioxidant properties and food applications. Antioxidants 12 (7), 1456. doi:10.3390/antiox12071456
Atrooz, O., Al-Nadaf, A., Uysal, H., Kutlu, H. M., and Sezer, C. V. (2023). Biosynthesis of silver nanoparticles using Coriandrum sativum L. extract and evaluation of their antibacterial, anti-inflammatory and antinociceptive activities. South Afr. J. Bot. 157, 219–227. doi:10.1016/j.sajb.2023.04.001
Attia, K. M., Assar, M. H., Farouk, Z. M., and Basuney, H. A. J. A. J. f.V. S. (2020). Possible protective effects of black seed (Nigella sativa) or garlic (Allium sativum) against lead-induced toxicity in growing rabbits. Alex. J. Vet. Sci. 64 (2), 52. doi:10.5455/ajvs.66077
Aykac, A., Ozbeyli, D., Uncu, M., Ertaş, B., Kılınc, O., Şen, A., et al. (2019). Evaluation of the protective effect of Myrtus communis in scopolamine-induced Alzheimer model through cholinergic receptors. Gene 689, 194–201. doi:10.1016/j.gene.2018.12.007
Azadedel, S., Hanachi, P., and Saboora, A. (2021). Antioxidant activity and phenolic compounds profile of pistachio skins (Pistacia vera L., cultivars Kallehghuchi and Ohadi). Hormozgan Med. J. 25 (4), 180–186. doi:10.5812/hmj.106093
Badalamenti, N., Modica, A., Ilardi, V., and Bruno, M. (2021). Chemical constituents and biological properties of genus Doronicum (Asteraceae). Chem. and Biodivers. 18 (12), e2100631. doi:10.1002/cbdv.202100631
Bahramsoltani, R., and Rahimi, R. (2020). An evaluation of traditional Persian medicine for the management of SARS-CoV-2. Front. Pharmacol. 11, 571434. doi:10.3389/fphar.2020.571434
Bartha, G. S., Tóth, G., Horváth, P., Kiss, E., Papp, N., and Kerényi, M. (2019). Analysis of aristolochlic acids and evaluation of antibacterial activity of Aristolochia clematitis L. Biologia Futura. Biol. Futur. 70 (4), 323–329. doi:10.1556/019.70.2019.36
Belabdelli, F., Piras, A., Bekhti, N., Falconieri, D., Belmokhtar, Z., and Merad, Y. (2020). Chemical composition and antifungal activity of Foeniculum vulgare Mill. Chem. Afr. 3, 323–328. doi:10.1007/s42250-020-00130-x
Bénéchet, A. P., De Simone, G., Di Lucia, P., Cilenti, F., Barbiera, G., Le Bert, N., et al. (2019). Dynamics and genomic landscape of CD8+ T cells undergoing hepatic priming. Nature 574 (7777), 200–205. doi:10.1038/s41586-019-1620-6
Bhadra, P. J. I. J. o.N. S. (2020). An overview of ajwain (Trachyspermum ammi). Indian J. Nat. Sci. 10 (59), 18466–182474.
Bhandari, P. R. (2012). Garlic (Allium sativum L.): a review of potential therapeutic applications. Int. J. Green Pharm. (IJGP) 6 (2), 118. doi:10.4103/0973-8258.102826
Bhat, R. (2021). Bioactive compounds of rhubarb (rheum species). Bioact. Compd. Underutilized Veg. Legumes, 239–254. doi:10.1007/978-3-030-57415-4_14
Bisignano, C., Filocamo, A., Faulks, R. M., and Mandalari, G. (2013). In vitro antimicrobial activity of pistachio (Pistacia vera L.) polyphenols. FEMS Microbiol. Lett. 341 (1), 62–67. doi:10.1111/1574-6968.12091
Boskabady, M. H., Alitaneh, S., and Alavinezhad, A. (2014). Carum copticum L.: a herbal medicine with various pharmacological effects. BioMed Res. Int. 2014, 569087. doi:10.1155/2014/569087
Bukhari, S. I., Manzoor, M., and Dhar, M. K. (2018). A comprehensive review of the pharmacological potential of Crocus sativus and its bioactive apocarotenoids. Biomed. and Pharmacother. 98, 733–745. doi:10.1016/j.biopha.2017.12.090
Chacko, B., and Peter, J. V. (2019). Antidotes in poisoning. Indian J. Crit. Care Med. 23 (Suppl. 4), S241–s249. doi:10.5005/jp-journals-10071-23310
Chbili, C., Maoua, M., Selmi, M., Mrad, S., Khairi, H., Limem, K., et al. (2020). Evaluation of daily Laurus nobilis tea consumption on lipid profile biomarkers in healthy volunteers. J. Am. Coll. Nutr. 39 (8), 733–738. doi:10.1080/07315724.2020.1727787
Chen, J., Tang, C., Zhang, R., Ye, S., Zhao, Z., Huang, Y., et al. (2020). Metabolomics analysis to evaluate the antibacterial activity of the essential oil from the leaves of Cinnamomum camphora (Linn.) Presl. J. Ethnopharmacol. 253, 112652. doi:10.1016/j.jep.2020.112652
Chhikara, N., Kour, R., Jaglan, S., Gupta, P., Gat, Y., and Panghal, A. (2018). Citrus medica: nutritional, phytochemical composition and health benefits–a review. Food and Funct. 9 (4), 1978–1992. doi:10.1039/c7fo02035j
Darzi, S. E., Khazraei, S. P., and Amirghofran, Z. (2018). The immunoinhibitory and apoptosis-inducing activities of Foeniculum vulgare on human peripheral blood lymphocytes. Res. Pharm. Sci. 13 (2), 103–110. doi:10.4103/1735-5362.223792
Dėnė, L., Steinkellner, S., and Valiuškaitė, A. (2021). “Antifungal properties of Coriandrum sativum extracts on Fusarium spp. in vitro,” in Proceedings of the international scientific conference “rural development”, 19–22.
Devkota, H. P., Logesh, R., and Das, N. (2022). “Health-promoting activities of clove (Syzygium aromaticum),” in Clove (Syzygium aromaticum) (Elsevier), 195–201.
Dissanayake, K. G. C., Waliwita, W., and Liyanage, R. P. (2020). A review on medicinal uses of Zingiber officinale (ginger). Int. J. Health Sci. Res. 10 (6), 142–148.
Dorrigiv, M., Zareiyan, A., and Hosseinzadeh, H. (2020). Garlic (Allium sativum) as an antidote or a protective agent against natural or chemical toxicities: a comprehensive update review. Phytotherapy Res. 34 (8), 1770–1797. doi:10.1002/ptr.6645
Dutta, N., Marker, P., and Rao, N. J. B. J. o.P. (1972). Berberine in toxin-induced experimental cholera. Br. J. Pharmacol. 44 (1), 153–159. doi:10.1111/j.1476-5381.1972.tb07247.x
Egwurugwu, J., Ufearo, C., Abanobi, O., Nwokocha, C., Duruibe, J., Adeleye, G., et al. (2007). Effects of ginger (Zingiber officinale) on cadmium toxicity. Afr. J. Biotechnol. 6 (18), 2078–2082. doi:10.5897/ajb2007.000-2321
Ehnert, S., Braun, K., Buchholz, A., Freude, T., Egana, J., Schenck, T., et al. (2012). Diallyl-disulphide is the effective ingredient of garlic oil that protects primary human osteoblasts from damage due to cigarette smoke. Food Chem. X. 132 (2), 724–729. doi:10.1016/j.foodchem.2011.11.008
Elbestawy, M. K., El-Sherbiny, G. M., and Moghannem, S. A. (2023). Antibacterial, antibiofilm and anti-inflammatory activities of eugenol clove essential oil against resistant Helicobacter pylori. Molecules 28 (6), 2448. doi:10.3390/molecules28062448
El Hawary, S. S. E., Hashim, F. A., Ibrahim, N. A., Matloub, A. A., ElSayed, A. M., Farid, M. A., et al. (2022). A comparative study of chemical compositions, antimicrobial and antiviral activities of essential oils for Citrus medica var. sarcodactylis swingle fruits and leaves along with Limonia acidissima L. Leaves. Egypt. J. Chem. 65 (8), 263–270. doi:10.21608/EJCHEM.2021.108413.4958
El-Moslemany, A. M., Abd-Elfatah, M. H., Tahoon, N. A., Bahnasy, R. M., Alotaibi, B. S., Ghamry, H. I., et al. (2023). Mechanistic assessment of anise seeds and clove buds against the neurotoxicity caused by metronidazole in rats: possible role of antioxidants, neurotransmitters, and cytokines. Neurotransmitters, Cytokines 11 (9), 724. doi:10.3390/toxics11090724
El-Saber Batiha, G., Magdy Beshbishy, A., Wasef, G., Elewa, Y. H., Al-Sagan, A., Abd El-Hack, M. E., et al. (2020). Chemical constituents and pharmacological activities of garlic (Allium sativum L.): a review. Nutrients 12 (3), 872. doi:10.3390/nu12030872
El-Zahar, K. M., Al-Jamaan, M. E., Al-Mutairi, F. R., Al-Hudiab, A. M., Al-Einzi, M. S., and Mohamed, A.A.-Z. (2022). Antioxidant, antibacterial, and antifungal activities of the ethanolic extract obtained from berberis vulgaris roots and leaves. Molecules 27 (18), 6114. doi:10.3390/molecules27186114
Emamat, H., Zahedmehr, A., Asadian, S., Tangestani, H., and Nasrollahzadeh, J. (2020). Effect of barberry (Berberis vulgaris) consumption on blood pressure, plasma lipids, and inflammation in patients with hypertension and other cardiovascular risk factors: study protocol for a randomized clinical trial. Trials 21 (1), 986–989. doi:10.1186/s13063-020-04918-7
Emami, M., Nazarinia, M. A., Rezaeizadeh, H., and Zarshenas, M. M. (2014). Standpoints of traditional Persian physicians on geriatric nutrition. J. evidence-based complementary and Altern. Med. 19 (4), 287–291. doi:10.1177/2156587214543289
Ersöz, E., Aydın, M. S., Hacanlı, Y., Kankılıç, N., Koyuncu, İ., Güldür, M. E., et al. (2023). Cardioprotective effect of Pistacia vera L. (Green pistachio) hull extract in wistar albino rats with doxorubicin-induced cardiac damage. Anatol. J. Cardiol. 27 (3), 135–145. doi:10.14744/AnatolJCardiol.2022.2452
Ersöz, E., Aydın, M. S., Hacanlı, Y., Kankılıç, N., Koyuncu, İ., Güldür, M. E., et al. (2023). Cardioprotective effect of Pistacia vera L. (green pistachio) hull extract in wistar albino rats with doxorubicin-induced cardiac damage. Anatol. J. Cardiol. 27 (3), 135–145. doi:10.14744/AnatolJCardiol.2022.2452
Esharkawy, E. R., Almalki, F., and Hadda, T. B. (2022). In vitro potential antiviral SARS-CoV-19-activity of natural product thymohydroquinone and dithymoquinone from Nigella sativa. Bioorg. Chem. 120, 105587. doi:10.1016/j.bioorg.2021.105587
Fahmi, A., Hassanen, N., Abdur-Rahman, M., and Shams-Eldin, E. (2019). Phytochemicals, antioxidant activity and hepatoprotective effect of ginger (Zingiber officinale) on diethylnitrosamine toxicity in rats. Biomarkers 24 (5), 436–447. doi:10.1080/1354750X.2019.1606280
Feldmann, M., Maini, R. N., Woody, J. N., Holgate, S. T., Winter, G., Rowland, M., et al. (2020). Trials of anti-tumour necrosis factor therapy for COVID-19 are urgently needed. Lancet 395 (10234), 1407–1409. doi:10.1016/S0140-6736(20)30858-8
Foudah, A. I., Alqarni, M. H., Alam, A., Salkini, M. A., Ahmed, E. O. I., and Yusufoglu, H. S. (2021). Evaluation of the composition and in vitro antimicrobial, antioxidant, and anti-inflammatory activities of Cilantro (Coriandrum sativum L. leaves) cultivated in Saudi Arabia (Al-Kharj). Saudi J. Biol. Sci. 28 (6), 3461–3468. doi:10.1016/j.sjbs.2021.03.011
Gao, D., Zeng, L.-N., Zhang, P., Ma, Z.-J., Li, R.-S., Zhao, Y.-L., et al. (2016). Rhubarb anthraquinones protect rats against mercuric chloride (HgCl2)-Induced acute renal failure. Molecules 21 (3), 298. doi:10.3390/molecules21030298
Gentile, C., Tesoriere, L., Butera, D., Fazzari, M., Monastero, M., Allegra, M., et al. (2007). Antioxidant activity of Sicilian pistachio (Pistacia vera L. var. Bronte) nut extract and its bioactive components. J. Agric. Food Chem. 55 (3), 643–648. doi:10.1021/jf062533i
Ghanavati, M., Rahmani, J., Clark, C. C. T., Hosseinabadi, S. M., and Rahimlou, M. (2020). Pistachios and cardiometabolic risk factors: a systematic review and meta-analysis of randomized controlled clinical trials. Complement. Ther. Med. 52, 102513. doi:10.1016/j.ctim.2020.102513
Ghlissi, Z., Kallel, R., Krichen, F., Hakim, A., Zeghal, K., Boudawara, T., et al. (2020). Polysaccharide from Pimpinella anisum seeds: structural characterization, anti-inflammatory and laser burn wound healing in mice. Int. J. Biol. Macromol. 156, 1530–1538. doi:10.1016/j.ijbiomac.2019.11.201
Gilani, A., Jabeen, Q., Ghayur, M., Janbaz, K., and Akhtar, M. (2005). Studies on the antihypertensive, antispasmodic, bronchodilator and hepatoprotective activities of the Carum copticum seed extract. J. Ethnopharmacol. 98 (1-2), 127–135. doi:10.1016/j.jep.2005.01.017
Golchin, L., Shabani, M., Harandi, S., and Razavinasab, M. (2015). Pistachio supplementation attenuates motor and cognition impairments induced by cisplatin or vincristine in rats. Adv. Biomed. Res. 4, 92. doi:10.4103/2277-9175.156656
Gutzwiller, A., Czegledi, L., Stoll, P., Bruckner, L. J. J. o.A. P., and Nutrition, A. (2007). Effects of Fusarium toxins on growth, humoral immune response and internal organs in weaner pigs, and the efficacy of apple pomace as an antidote. J. Anim. Physiol. Anim. Nutr. Berl. 91 (9-10), 432–438. doi:10.1111/j.1439-0396.2006.00672.x
Haleshi, C., Rao, M. M., and Danapur, V. (2022). Pharmacognostic studies on a well known cardio protective plant Pistacia vera. Int. J. Pharm. Bio Med. Sci. 2 (5), 104–108. doi:10.47191/ijpbms/v2-i5-04
Harun, N. H., and Mohamad, M. F. (2022). The immunomodulatory effects of Zingiber officinale (ginger): a systematic review. Res. J. Pharm. Technol. 15 (8), 3776–3781. doi:10.52711/0974-360x.2022.00634
Hoseinifard, S. M., Omidzahir, S., Hoseini, S. M., and Beikaee, H. J. C. C. P. (2018). Protective effect of garlic (Allium sativum) against zinc poisoning in the testicular tissue of goldfish (Carassius auratus). Comp. Clin. Path. 27 (2), 357–361. doi:10.1007/s00580-017-2599-8
Hu, C., Zhu, W., Lu, Y., Ren, Y., Gu, J., Song, Y., et al. (2023). Alpinia officinarum mediated copper oxide nanoparticles: synthesis and its antifungal activity against Colletotrichum gloeosporioides. Environ. Sci. Pollut. Res. 30 (11), 28818–28829. doi:10.1007/s11356-022-24225-9
Iranmanesh, F., Mousaei Amin, A., Rahnama, A., Malaki Rad, A., and Shmasizadeh, A. (2017). Protective effect of hydro-alcoholic extract of Pistacia vera on liver enzymes following induction of hepatotoxicity in rats. J. Sabzevar Univ. Med. Sci. 24 (3), 149–156.
Jafari-Sales, A., and Pashazadeh, M. (2020). Antibacterial effect of methanolic extract of saffron petal (Crocus sativus L.) on some standard gram positive and gram negative pathogenic bacteria in vitro. Curr. Perspect. Med. Aromatic Plants (CUPMAP) 3 (1), 1–7. doi:10.38093/cupmap.692879
Jakubczyk, A., Karaś, M., Rybczyńska-Tkaczyk, K., Zielińska, E., and Zieliński, D. (2020). Current trends of bioactive peptides—new sources and therapeutic effect. Foods 9 (7), 846. doi:10.3390/foods9070846
Jamshidi, A. H., Eghbalian, F., Mahroozade, S., Mohammadi Kenari, H., Ghobadi, A., and Yousefsani, B. S. J. J. o.M. P. (2020). Recommended natural products in Alzheimer's disease based on traditional Persian medicine. medicine 19 (75), 17–29. doi:10.29252/jmp.19.75.17
Kačániová, M., Galovičová, L., Ivanišová, E., Vukovic, N. L., Štefániková, J., Valková, V., et al. (2020). Antioxidant, antimicrobial and antibiofilm activity of coriander (Coriandrum sativum L.) essential oil for its application in foods. Foods 9 (3), 282. doi:10.3390/foods9030282
Kadoglou, N. P., Christodoulou, E., Kostomitsopoulos, N., and Valsami, G. (2021). The cardiovascular-protective properties of saffron and its potential pharmaceutical applications: a critical appraisal of the literature. Phytotherapy Res. 35 (12), 6735–6753. doi:10.1002/ptr.7260
Kalam, M. A., Haseeb, A., Mushtaq, M. S., and Hussain, Z. (2020). Taryaq-i-Waba ‘i: a review on Potent compound formulation of Unani medicine with special reference to epidemic/pandemic diseases. J. Drug Deliv. Ther. 10 (4), 234–237. doi:10.22270/jddt.v10i4.4223
Kalantari, H., Foruozandeh, H., Khodayar, M. J., Siahpoosh, A., Saki, N., and Kheradmand, P. (2018). Antioxidant and hepatoprotective effects of Capparis spinosa L. fractions and Quercetin on tert-butyl hydroperoxide-induced acute liver damage in mice. J. traditional complementary Med. 8 (1), 120–127. doi:10.1016/j.jtcme.2017.04.010
Kalmarzi, R. N., Naleini, S. N., Ashtary-Larky, D., Peluso, I., Jouybari, L., Rafi, A., et al. (2019). Anti-inflammatory and immunomodulatory effects of barberry (Berberis vulgaris) and its main compounds. Oxidative Med. Cell. Longev. 2019, 1–10. doi:10.1155/2019/6183965
Kamalinejad, M., Sarmadian, H., Shokouhi, F., Dadmehr, M., Bahrami, M., Dehnad, A., et al. (2021). The clinical efficacy of Tiban syrup as adjuvant treatment in patients with COVID-19: a randomized, double blind clinical trial. Iran. J. Pharm. Sci. 17 (3), 49–62. doi:10.22037/ijps.v17.40273
Karimi, A., and Meiners, T. (2021). Antifungal activity of Zataria multiflora Boiss. essential oils and changes in volatile compound composition under abiotic stress conditions. Industrial crops Prod. 171, 113888. doi:10.1016/j.indcrop.2021.113888
Kaur, G. J., and Arora, D. S. (2009). Antibacterial and phytochemical screening of Anethum graveolens, Foeniculum vulgare and Trachyspermum ammi. BMC complementary Altern. Med. 9 (1), 30–10. doi:10.1186/1472-6882-9-30
Kaushik, S., Jangra, G., Kundu, V., Yadav, J. P., and Kaushik, S. (2020). Anti-viral activity of Zingiber officinale (Ginger) ingredients against the Chikungunya virus. Virusdisease 31, 270–276. doi:10.1007/s13337-020-00584-0
Kehili, S., Boukhatem, M. A., Belkadi, A., Boulaghmen, F., Ferhat, M. A., and Setzer, W. N. (2020). Spearmint (Mentha spicata L.) essential oil from tipaza (Algeria): in vivo anti-inflammatory and analgesic activities in experimental animal models. Acta Pharm. Hung. 90 (1), 15–26. doi:10.33892/aph.2020.90.15-26
Kenari, H. M., Kordafshari, G., Moghimi, M., Eghbalian, F., and TaherKhani, D. J. J. o.p. (2021). Review of pharmacological properties and chemical constituents of Pastinaca sativa. J. Pharmacopuncture 24 (1), 14–23. doi:10.3831/KPI.2021.24.1.14
Khan, A. A., Afrin, Z., and Kumar, P. (2020). Overview of covid 19 and Unani formulation tiryaq-e-wabai. J. Drug Deliv. Ther. 10 (5-s), 205–208. doi:10.22270/jddt.v10i5-s.4337
Khan, J., Sakib, S. A., Mahmud, S., Khan, Z., Islam, M. N., Sakib, M. A., et al. (2022). Identification of potential phytochemicals from Citrus limon against main protease of SARS-CoV-2: molecular docking, molecular dynamic simulations and quantum computations. J. Biomol. Struct. Dyn. 40 (21), 10741–10752. doi:10.1080/07391102.2021.1947893
Khodja, Y. K., Bachir-bey, M., Ladjouzid, R., Katiac, D., and Khettal, B. (2021). In vitro antioxidant and antibacterial activities of phenolic and alkaloid extracts of Laurus nobilis. South Asian J. Exp. Biol. 11 (3), 345–354. doi:10.38150/sajeb.11(3).p345-354
Klimek-Szczykutowicz, M., Szopa, A., and Ekiert, H. (2020). Citrus limon (Lemon) phenomenon—a review of the chemistry, pharmacological properties, applications in the modern pharmaceutical, food, and cosmetics industries, and biotechnological studies. Plants 9 (1), 119. doi:10.3390/plants9010119
Koçak, Ö. F., and Yılmaz, F. (2022). Use of Alpinia officinarum rhizome in textile dyeing and gaining simultaneous antibacterial properties. J. Nat. Fibers 19 (5), 1925–1936. doi:10.1080/15440478.2021.1889441
Kolniak-Ostek, J., Kłopotowska, D., Rutkowski, K. P., Skorupińska, A., and Kruczyńska, D. E. (2020). Bioactive compounds and health-promoting properties of pear (Pyrus communis L.) fruits. Molecules 25 (19), 4444. doi:10.3390/molecules25194444
Korani, M., and Jamshidi, M. (2020). The effect of aqueous extract of trachyspermum ammi seeds and ibuprofen on inflammatory gene expression in the cartilage tissue of rats with collagen-induced arthritis. J. Inflamm. Res. 13, 133–139. doi:10.2147/JIR.S236242
Korkmaz, S., Koç, F. J. I. j.o.a., and Sciences, N. (2021). The protective effects of dill (Anethum graveolens L.) in paracetamol-induced acute toxication in mice. 14(1), 47–60.
Koscielny, J., Klüssendorf, D., Latza, R., Schmitt, R., Radtke, H., Siegel, G., et al. (1999). The antiatherosclerotic effect of Allium sativum. Atherosclerosis 144 (1), 237–249. doi:10.1016/s0021-9150(99)00060-x
Kumar, A., and Yadav, G. (2022). Potential role of medicinal plants for their immunomodulatory activity-A review. Ann. Clin. Pharmacol. and Toxicol.
Laribi, B., Kouki, K., M'Hamdi, M., and Bettaieb, T. (2015). Coriander (Coriandrum sativum L.) and its bioactive constituents. Fitoterapia 103, 9–26. doi:10.1016/j.fitote.2015.03.012
Lee, D. W., Lee, M.-Y., Koh, S., and Yang, M. J. M. (2020a). Micropillar/microwell chip assessment for detoxification of bisphenol A with Korean pear (Pyrus pyrifolia). pyrus pyrifolia 11 (10), 922. doi:10.3390/mi11100922
Lee, E.-H., Park, H.-J., Kim, B.-O., Choi, H.-W., Park, K.-I., Kang, I.-K., et al. (2020b). Anti-inflammatory effect of Malus domestica cv. Green ball apple peel extract on Raw 264.7 macrophages. J. Appl. Biol. Chem. 63 (2), 117–123. doi:10.3839/jabc.2020.016
Li, A., Liu, Y., Zhu, X., Sun, X., Feng, X., Li, D., et al. (2018). Protective effect of methylallyl sulfone in the development of cigarette smoke extract-induced apoptosis in rats and HFL-1 cells. Biochem. Biophys. Res. Commun. 498 (3), 627–632. doi:10.1016/j.bbrc.2018.03.033
Li, L., and Wang, F. (2022). Wound healing and anti-inflammatory effects of Anethum graveolens extract loaded in PVA fibers: an in vitro and in vivo study. J. Bioact. Compatible Polym. 37 (4), 299–315. doi:10.1177/08839115221104074
Li, X., Chu, S., Liu, Y., and Chen, N. (2019). Neuroprotective effects of anthraquinones from rhubarb in central nervous system diseases. Evidence-Based Complementary Altern. Med. 2019, 3790728. doi:10.1155/2019/3790728
Liu, H., Wang, X., Shi, Q., Li, L., Zhang, Q., Wu, Z.-L., et al. (2020). Dimeric diarylheptanoids with neuroprotective activities from rhizomes of Alpinia officinarum. ACS omega 5 (17), 10167–10175. doi:10.1021/acsomega.0c01019
Liu, Y., Blumberg, J. B., and Chen, C. Y. (2014). Quantification and bioaccessibility of California pistachio bioactives. J. Agric. Food Chem. 62 (7), 1550–1556. doi:10.1021/jf4046864
Lu, Y., Du, Y., Qin, X., Wu, H., Huang, Y., Cheng, Y., et al. (2019). Comprehensive evaluation of effective polyphenols in apple leaves and their combinatory antioxidant and neuroprotective activities. Industrial crops Prod. 129, 242–252. doi:10.1016/j.indcrop.2018.12.013
Luo, Q., Tian, Z., Zheng, T., Xu, S., Ma, Y., Zou, S., et al. (2022). Terpenoid composition and antioxidant activity of extracts from four chemotypes of Cinnamomum camphora and their main antioxidant agents. Biofuels, Bioprod. Biorefining 16 (2), 510–522. doi:10.1002/bbb.2320
Lysiuk, R., Oliynyk, P., Antonyak, H., and Voronenko, D. (2020). Development of phyto-antidotes against adverse chemical agents. Poisonous Plants Phytochemicals Drug Discov., 249–268. doi:10.1002/9781119650034.ch12
Magowska, A. (2021). The natural history of the concept of antidote. Toxicol. Rep. 8, 1305–1309. doi:10.1016/j.toxrep.2021.06.019
Mahleyuddin, N. N., Moshawih, S., Ming, L. C., Zulkifly, H. H., Kifli, N., Loy, M. J., et al. (2021). Coriandrum sativum L.: a review on ethnopharmacology, phytochemistry, and cardiovascular benefits. Molecules 27 (1), 209. doi:10.3390/molecules27010209
Mahmoudi, T., Lorigooini, Z., Rafieian-Kopaei, M., Arabi, M., Rabiei, Z., Bijad, E., et al. (2020). Effect of Curcuma zedoaria hydro-alcoholic extract on learning, memory deficits and oxidative damage of brain tissue following seizures induced by pentylenetetrazole in rat. Behav. Brain Funct. 16 (1), 7–12. doi:10.1186/s12993-020-00169-3
Mahroozade, S., Mohammadi Kenari, H., Eghbalian, F., Ghobadi, A., and Yousefsani, B. S. J. C. M. R. (2021). Avicenna’s points of view in epidemics: some advice on coronavirus 2 (COVID-19). Complement. Med. Res. 28 (2), 175–176. doi:10.1159/000509398
Malaysia, J. (2004). Protective effect of the ethanol extract of Zingiber officinale Roscoe on paracetamol induced hepatotoxicity in rats. J. Sains Kesihat. Malays. 2(2), 85–95.
Mandalari, G., Barreca, D., Gervasi, T., Roussell, M. A., Klein, B., Feeney, M. J., et al. (2021). Pistachio nuts (Pistacia vera L.): production, nutrients, bioactives and novel health effects. Plants 11 (1), 18. doi:10.3390/plants11010018
Mao, Q.-Q., Xu, X.-Y., Cao, S.-Y., Gan, R.-Y., Corke, H., Beta, T., et al. (2019). Bioactive compounds and bioactivities of ginger (Zingiber officinale Roscoe). Foods 8 (6), 185. doi:10.3390/foods8060185
Mehrim, A., Abdelhamid, A., Abou-Shousha, A., Salem, M., and El-Sharawy, M. J. J. o.t.A. A. s. (2006). Nutritious attempts to detoxify aflatoxic diets of tilapia fish: 2-Clinical, biochemical and histological parameters. J. Arabian Aquac. Soc. 1 (2), 69–90.
Moroz, Y., Batsazova, V., Pohodina, L., and Shatalova, O. (2021). Experimental study of anti-inflammatory activity of the aristolochia clematitis extracts.
Mousavi, S. H., Hosseini, A., Bakhtiari, E., and Rakhshandeh, H. (2016). Capparis spinosa reduces Doxorubicin-induced cardio-toxicity in cardiomyoblast cells. Avicenna J. phytomedicine 6 (5), 488–494.
Musarra-Pizzo, M., Pennisi, R., Ben-Amor, I., Smeriglio, A., Mandalari, G., and Sciortino, M. T. (2020). In vitro anti-HSV-1 activity of polyphenol-rich extracts and pure polyphenol compounds derived from pistachios kernels (Pistacia vera L.). Plants (Basel) 9 (2), 267. doi:10.3390/plants9020267
Naghibi, F., Esmaeili, S., Abdullah, N. R., Nateghpour, M., Taghvai, M., Kamkar, S., et al. (2013). In vitro and in vivo antimalarial evaluations of myrtle extract, a plant traditionally used for treatment of parasitic disorders. Biomed. Res. Int. 2013, 316185. doi:10.1155/2013/316185
Naim, N., Bouymajane, A., Oulad El Majdoub, Y., Ezrari, S., Lahlali, R., Tahiri, A., et al. (2022). Flavonoid composition and antibacterial properties of Crocus sativus L. Petal extracts. Molecules 28 (1), 186. doi:10.3390/molecules28010186
Nasiri, E., Orimi, J. R., Hashemimehr, M., Aghabeiglooei, Z., Rezghi, M., and Amrollahi-Sharifabadi, M. (2023). Avicenna’s clinical toxicology approach and beneficial materia medica against oral poisoning. Archives Toxicol. 97 (4), 981–989. doi:10.1007/s00204-023-03464-w
Natarajan, P., and Grace, J. (2019). Effect of Mill. on Isoproterenol induced myocardial infarction in Foeniculum vulgare albino rats. Asian J. Pharm. Pharmacol. 5 (2), 259–265. doi:10.31024/ajpp.2019.5.2.7
Nerilo, S. B., Romoli, J. C. Z., Nakasugi, L. P., Zampieri, N. S., Mossini, S. A. G., Rocha, G. H. O., et al. (2020). Antifungal activity and inhibition of aflatoxins production by Zingiber officinale Roscoe essential oil against Aspergillus flavus in stored maize grains. Ciência Rural. 50. doi:10.1590/0103-8478cr20190779
Ni, J., Chen, H., Zhang, C., Luo, Q., Qin, Y., Yang, Y., et al. (2022). Characterization of Alpinia officinarum Hance polysaccharide and its immune modulatory activity in mice. Food and Funct. 13 (4), 2228–2237. doi:10.1039/d1fo03949k
Nigar, Z., and Itrat, M. (2013). Evaluation of a Unani polyherbal formulation (Tiryaqe wabai) as an immunostimulator in elderly persons. Anc. Sci. life 33 (2), 119–122. doi:10.4103/0257-7941.139054
Nikhat, S., and Fazil, M. (2023). Critical review and mechanistic insights into the health-protective and Immunomodulatory activity of Tiryāq (Theriac) from the purview of Unani medicine. Brain Behav. Immun. Integr. 4, 100021. doi:10.1016/j.bbii.2023.100021
Noumi, E., Ahmad, I., Adnan, M., Merghni, A., Patel, H., Haddaji, N., et al. (2023). GC/MS profiling, antibacterial, anti-quorum sensing, and antibiofilm properties of Anethum graveolens L. essential oil: molecular docking study and in-silico ADME profiling. Plants 12 (10), 1997. doi:10.3390/plants12101997
Ojueromi, O. O., Oboh, G., and Ademosun, A. O. (2022). Effect of black seeds (Nigella sativa) on inflammatory and immunomodulatory markers in Plasmodium berghei-infected mice. J. Food Biochem. 46 (11), e14300. doi:10.1111/jfbc.14300
Orhan, İ., Ozçelİk, B., Kartal, M., and Kan, Y. (2012). Antimicrobial and antiviral effects of essential oils from selected Umbelliferae and Labiatae plants and individual essential oil components. Turkish J. Biol. 36 (3), 239–246. doi:10.3906/biy-0912-30
Ouahhoud, S., Touiss, I., Khoulati, A., Lahmass, I., Mamri, S., Meziane, M., et al. (2021). Hepatoprotective effects of hydroethanolic extracts of Crocus sativus tepals, stigmas and leaves on carbon tetrachloride induced acute liver injury in rats. Physiology and Pharmacol. 25 (2), 178–188. doi:10.32598/ppj.25.2.30
Oueslati, S., Mansour, R., Medini, F., Ksouri, R., and Megdiche, W. (2020). Effect of the extraction method on the content of phenolic compounds and the antioxidant and antibacterial potential of Mentha spicata L. J. New Sci. 77, 4503–4509.
Özbeyli, D., Yarimbas, G., Ertaş, B., Şen, A., Sakarcan, S., and Sener, G. (2020). Myrtus communis extract ameliorates high-fat diet induced brain damage and cognitive function. J. Res. Pharm. 24 (6), 865–873. doi:10.35333/jrp.2020.245
Ozkur, M., Benlier, N., Takan, I., Vasileiou, C., Georgakilas, A. G., Pavlopoulou, A., et al. (2022). Ginger for healthy ageing: a systematic review on current evidence of its antioxidant, anti-inflammatory, and anticancer properties. Oxidative Med. Cell. Longev., 2022. doi:10.1155/2022/4748447
Pandey, A., Bigoniya, P., Raj, V., and Patel, K. (2011). Pharmacological screening of Coriandrum sativum Linn. for hepatoprotective activity. J. Pharm. Bioallied Sci. 3 (3), 435–441. doi:10.4103/0975-7406.84462
Pandur, E., Balatinácz, A., Micalizzi, G., Mondello, L., Horváth, A., Sipos, K., et al. (2021). Anti-inflammatory effect of lavender (Lavandula angustifolia Mill.) essential oil prepared during different plant phenophases on THP-1 macrophages. BMC Complementary Med. Ther. 21, 287–317. doi:10.1186/s12906-021-03461-5
Pareek, A., Suthar, M., Rathore, G. S., and Bansal, V. J. P. R. (2011). Feverfew (Tanacetum parthenium L.). A Syst. Rev. 5 (9), 103–110. doi:10.4103/0973-7847.79105
Park, Y. J., Lee, K. H., Jeon, M. S., Lee, Y. H., Ko, Y. J., Pang, C., et al. (2020). Hepatoprotective potency of chrysophanol 8-O-glucoside from Rheum palmatum L. against hepatic fibrosis via regulation of the STAT3 signaling pathway. Int. J. Mol. Sci. 21 (23), 9044. doi:10.3390/ijms21239044
Patocka, J., Bhardwaj, K., Klimova, B., Nepovimova, E., Wu, Q., Landi, M., et al. (2020). Malus domestica: a review on nutritional features, chemical composition, traditional and medicinal value. Plants 9 (11), 1408. doi:10.3390/plants9111408
Peng, B., Luo, Y., Hu, X., Song, L., Yang, J., Zhu, J., et al. (2019). Isolation, structural characterization, and immunostimulatory activity of a new water-soluble polysaccharide and its sulfated derivative from Citrus medica L. var. sarcodactylis. Int. J. Biol. Macromol. 123, 500–511. doi:10.1016/j.ijbiomac.2018.11.113
Perera, R., Senadheera, S. M., and Hewapathirana, S. (2022). “Computational study on SARS-CoV-2 viral protein interaction with natural compounds of Coriandrum sativum L,” in 2022 2nd international Conference on advanced Research in computing ICARC (IEEE), 320–325.
Pollard, K. M., Hultman, P., and Kono, D. H. (2010). Toxicology of autoimmune diseases. Chem. Res. Toxicol. 23 (3), 455–466. doi:10.1021/tx9003787
Pontes, V. C. B., Rodrigues, D. P., Caetano, A., and Gamberini, M. T. (2019). Preclinical investigation of the cardiovascular actions induced by aqueous extract of Pimpinella anisum L. seeds in rats. J. Ethnopharmacol. 237, 74–80. doi:10.1016/j.jep.2019.03.050
Pooja, S., Shetty, P., Kumari, N., and Shetty, K. (2021). Radioprotective and antioxidant potential of Tanacetum parthenium extract and synthetic parthenolide in Swiss albino mice exposed to electron beam irradiation. Int. J. Radiat. Res. 19 (1), 145–154. doi:10.29252/ijrr.19.1.145
Rahimi, R., and Ardekani, M. R. S. (2012). Medicinal properties of Foeniculum vulgare Mill. in traditional Iranian medicine and modern phytotherapy. Chin. J. Integr. Med. 19 (1), 73–79. doi:10.1007/s11655-013-1327-0
Rajan, D. P., and Surya, A. J. A. i.A. R. (2019). Studies on clove as a potential antidote for metal intoxication in Tilapia mossambica. Adva. Appl. Rese. 11 (1), 50–52. doi:10.5958/2349-2104.2019.00008.1
Rather, M. A., Dar, B. A., Sofi, S. N., Bhat, B. A., and Qurishi, M. A. (2016). Foeniculum vulgare: a comprehensive review of its traditional use, phytochemistry, pharmacology, and safety. Arabian J. Chem. 9, S1574–S1583. doi:10.1016/j.arabjc.2012.04.011
Ravindran, C. A., Murugaiyah, V., Khiang, P., Xavior, R. J. A. J. o.P., and Research, C. (2013). Hepatoprotective activity of leaf of methanol extract of Laurus nobilis L. against paracetamol induced hepatotoxicity in rats. Asian J. Pharm. Clin. Res. 6 (4), 153–157.
Razavi, B. M., and Hosseinzadeh, H. J. D. J. o.P. S. (2015). Saffron as an antidote or a protective agent against natural or chemical toxicities. DARU J. Pharm. Sci. 23, 31–39. doi:10.1186/s40199-015-0112-y
Recinella, L., Chiavaroli, A., di Giacomo, V., Antolini, M. D., Acquaviva, A., Leone, S., et al. (2020). Anti-Inflammatory and neuromodulatory effects induced by tanacetum parthenium water extract: results from in silico, in vitro and ex vivo studies. Molecules 26 (1), 22. doi:10.3390/molecules26010022
Rezaee-Khorasany, A., Razavi, B. M., Taghiabadi, E., Yazdi, A. T., and Hosseinzadeh, H. J. J. o.e. (2019). Effect of saffron (stigma of Crocus sativus L.) aqueous extract on ethanol toxicity in rats: a biochemical. Histopathol. Mol. study 237, 286–299. doi:10.1016/j.jep.2019.03.048
Roozitalab, G., Yousefpoor, Y., Abdollahi, A., Safari, M., Rasti, F., and Osanloo, M. (2022). Antioxidative, anticancer, and antibacterial activities of a nanoemulsion-based gel containing Myrtus communis L. essential oil. Chem. Pap. 76 (7), 4261–4271. doi:10.1007/s11696-022-02185-1
Rouf, R., Uddin, S. J., Sarker, D. K., Islam, M. T., Ali, E. S., Shilpi, J. A., et al. (2020). Antiviral potential of garlic (Allium sativum) and its organosulfur compounds: a systematic update of pre-clinical and clinical data. Trends food Sci. and Technol. 104, 219–234. doi:10.1016/j.tifs.2020.08.006
Sack, R. B., and Froehlich, J. L. J. I. (1982). Berberine inhibits intestinal secretory response of Vibrio cholerae and Escherichia coli enterotoxins. Infect. Immun. 35 (2), 471–475. doi:10.1128/iai.35.2.471-475.1982
Sahib, N. G., Anwar, F., Gilani, A.-H., Hamid, A. A., Saari, N., and Alkharfy, K. M. (2013). Coriander (Coriandrum sativum L.): a potential source of high-value components for functional foods and nutraceuticals- A review. Phytotherapy Res. 27 (10), 1439–1456. doi:10.1002/ptr.4897
Saleem, M., Alam, A., Sultana, S. J. F., and Toxicology, C. (2000). Attenuation of benzoyl peroxide-mediated cutaneous oxidative stress and hyperproliferative response by the prophylactic treatment of mice with spearmint (Mentha spicata). Food Chem. Toxicol. 38 (10), 939–948. doi:10.1016/s0278-6915(00)00088-0
Salehi, B., Valussi, M., Jugran, A. K., Martorell, M., Ramírez-Alarcón, K., Stojanović-Radić, Z. Z., et al. (2018). Nepeta species: from farm to food applications and phytotherapy. Trends food Sci. and Technol. 80, 104–122. doi:10.1016/j.tifs.2018.07.030
Sardari, S., Fallahiarezoudar, F., Emadi, F., Davati, A., Faramarzi, E., Fesharaki, M. G., et al. (2018). The use of caper plant in pharmaceutical ingredients: in traditional medicine and modern medicine. Indo Am. J. Pharm. Sci. 5 (1), 24–32.
Sari, S., Ginting, S. F., Chiuman, L., and Ferdinan Ginting, S. (2022). Effect of ethanol extract of white turmeric (curcuma zedoaria) as hepatoprotector in male rats induced by Cuso4 pentahydrate. J. Res. Soc. Sci. Econ. Manag. 1 (6), 747–753. doi:10.36418/jrssem.v1i6.94
Sebiomo, A., Awofodu, A., Awosanya, A., Awotona, F., and Ajayi, A. (2011). Comparative studies of antibacterial effect of some antibiotics and ginger (Zingiber officinale) on two pathogenic bacteria. J. Microbiol. Antimicrob. 3 (1), 18–22.
Sfar, M., Souid, G., Mzoughi, Z., Le Cerf, D., and Majdoub, H. (2023). Hepatoprotective effect against cadmium-induced liver toxicity in rats of Foeniculum vulgare seed polysaccharides. Chem. Afr. 6, 1753–1765. doi:10.1007/s42250-023-00621-7
Shamsi-Baghbanan, H., Sharifian, A., Esmaeili, S., and Minaei, B. (2014). Hepatoprotective herbs, avicenna viewpoint. Iran. Red Crescent Med. J. 16 (1), e12313. doi:10.5812/ircmj.12313
Shao, X., Sun, C., Tang, X., Zhang, X., Han, D., Liang, S., et al. (2020). Anti-inflammatory and intestinal microbiota modulation properties of Jinxiang garlic (Allium sativum L.) polysaccharides toward dextran sodium sulfate-induced colitis. J. Agric. Food Chem. 68 (44), 12295–12309. doi:10.1021/acs.jafc.0c04773
Sharma, L., Agarwal, D., Saxena, S., Kumar, H., Kumar, M., Verma, J., et al. (2018). Antibacterial and Antifungal activity of ajwain (Trachyspermum ammi) in different solvent. J. Pharmacogn. Phytochemistry 7 (3), 2672–2674.
Shen, C.-Y., Jiang, J.-G., Li, M.-Q., Zheng, C.-Y., and Zhu, W. (2017). Structural characterization and immunomodulatory activity of novel polysaccharides from Citrus aurantium Linn. variant amara Engl. J. Funct. Foods 35, 352–362. doi:10.1016/j.jff.2017.05.055
Shu, Y., He, D., Li, W., Wang, M., Zhao, S., Liu, L., et al. (2020). Hepatoprotective effect of Citrus aurantium L. against APAP-induced liver injury by regulating liver lipid metabolism and apoptosis. Int. J. Biol. Sci. 16 (5), 752–765. doi:10.7150/ijbs.40612
Singh, S., Bajpai, M., and Mishra, P. (2021). Pharmacognostic profile, in-vitro antioxidant and hepatoprotective potential of ethanolic fruit extract of Pyrus communis linn. Curr. Bioact. Compd. 17 (10), 49–59. doi:10.2174/1573407217666210308160905
Sobhy, S., Al-Askar, A. A., Bakhiet, E. K., Elsharkawy, M. M., Arishi, A. A., Behiry, S. I., et al. (2023). Phytochemical characterization and antifungal efficacy of camphor (Cinnamomum camphora L.) extract against phytopathogenic fungi. Separations 10 (3), 189. doi:10.3390/separations10030189
Sonneville, R., Mirabel, M., Hajage, D., Tubach, F., Vignon, P., Perez, P., et al. (2011). Neurologic complications and outcomes of infective endocarditis in critically ill patients: the ENDOcardite en REAnimation prospective multicenter study. Crit. care Med. 39 (6), 1474–1481. doi:10.1097/CCM.0b013e3182120b41
Stanojevic, L. P., Marjanovic-Balaban, Z. R., Kalaba, V. D., Stanojevic, J. S., and Cvetkovic, D. J. (2016). Chemical composition, antioxidant and antimicrobial activity of chamomile flowers essential oil (Matricaria chamomilla L.). J. Essent. oil Bear. plants 19 (8), 2017–2028. doi:10.1080/0972060x.2016.1224689
Taghizadieh, A., Mohammadinasab, R., Ghazi-Sha’rbaf, J., Michaleas, S. N., Vrachatis, D., and Karamanou, M. (2020). Theriac in the Persian traditional medicine. Erciyes Med. J. 42 (2), 235–239. doi:10.14744/etd.2020.30049
Tavakkoli, A., Ahmadi, A., Razavi, B. M., and Hosseinzadeh, H. (2017). Black seed (Nigella sativa) and its constituent thymoquinone as an antidote or a protective agent against natural or chemical toxicities. Iran. J. Pharm. Res. 16 (Suppl. l), 2–23.
Timalsina, B., Haque, M. N., Choi, H. J., Dash, R., and Moon, I. S. (2023). Thymol in Trachyspermum ammi seed extract exhibits neuroprotection, learning, and memory enhancement in scopolamine-induced Alzheimer's disease mouse model. Phytotherapy Res. 37, 2811–2826. doi:10.1002/ptr.7777
Vaghasloo, M. A., Naghizadeh, A., and Babashahi, N. (2017). The concept of the haar-re-gharizi and hararate gharizi: the innate hot [substance] and heat. Traditional Integr. Med., 3–8.
Velazhahan, R., Vijayanandraj, S., Vijayasamundeeswari, A., Paranidharan, V., Samiyappan, R., Iwamoto, T., et al. (2010). Detoxification of aflatoxins by seed extracts of the medicinal plant, Trachyspermum ammi (L.) Sprague ex Turrill – structural analysis and biological toxicity of degradation product of aflatoxin G1. Microbiol. Res. 21 (5), 719–725. doi:10.1016/j.foodcont.2009.10.014
Verma, T., Aggarwal, A., Dey, P., Chauhan, A. K., Rashid, S., Chen, K.-T., et al. (2023). Medicinal and therapeutic properties of garlic, garlic essential oil, and garlic-based snack food: an updated review. Front. Nutr. 10, 1120377. doi:10.3389/fnut.2023.1120377
Wang, C., Ma, C.-N., Liu, X.-L., Sun, Q., Zhang, Q., Lin, Y.-Y., et al. (2023). Apple consumption protects against acute ethanol-induced liver injury in rats. Appl. Sci. 13 (8), 5112. doi:10.3390/app13085112
Xiao, S., Yu, H., Xie, Y., Guo, Y., Fan, J., and Yao, W. (2021). The anti-inflammatory potential of Cinnamomum camphora (L.) J. Presl essential oil in vitro and in vivo. J. Ethnopharmacol. 267, 113516. doi:10.1016/j.jep.2020.113516
Xing, B., Li, S., Yang, J., Lin, D., Feng, Y., Lu, J., et al. (2021). Phytochemistry, pharmacology, and potential clinical applications of saffron: a review. J. Ethnopharmacol. 281, 114555. doi:10.1016/j.jep.2021.114555
Yadav, Y. C., Singh, A., Kannaujia, S. K., and Yadav, R. (2022). Neuroprotective effect of Citrus limon juice against scopolamine induced amnesia in Wistar rats: role of cholinergic neurotransmission monitoring and beta-actin signaling. Pharmacol. Research-Modern Chin. Med. 5, 100191. doi:10.1016/j.prmcm.2022.100191
Yaman, İ., Balikci, E. J. E., and Pathology, T. (2010). Protective effects of Nigella sativa against gentamicin-induced nephrotoxicity in rats. Exp. Toxicol. Pathol. 62(2), 183–190. doi:10.1016/j.etp.2009.03.006
Yesmin, F., Rahman, Z., Dewan, J. F., Helali, A. M., Rahman, N. I. A., Alattraqchi, A. G., et al. (2013). Hepato-protective role of the aqueous and n-hexane extracts of Nigella sativa Linn. in experimental liver damage in rats. Asian J. Pharm. Clin. Res. 6 (3), 205–209.
Yousuf, R., Verma, P. K., Sharma, P., Sood, S., Bhatti, M. A., and Bhat, Z. F. (2023). Neuroprotective effect of quercetin and Zingiber officinale on sodium arsenate-induced neurotoxicity in rats. Food Sci. and Nutr. 11, 2964–2973. doi:10.1002/fsn3.3278
Zarshenas, M. M., Moein, M., Samani, S. M., and Petramfar, P. (2013). An overview on ajwain (Trachyspermum ammi) pharmacological effects; modern and traditional. J. Nat. Remedies 14 (1), 98–105.
Zhong, K., Wang, R.-X., Qian, X.-D., Yu, P., Zhu, X.-Y., Zhang, Q., et al. (2020). Neuroprotective effects of saffron on the late cerebral ischemia injury through inhibiting astrogliosis and glial scar formation in rats. Biomed. and Pharmacother. 126, 110041. doi:10.1016/j.biopha.2020.110041
Keywords: antidote, tiryāq, medicinal plants, Persian medicine, detoxification
Citation: Mirhosseini H, Dadmehr M, Yousefsani BS, Seif F and Eghbalian F (2025) Tiryāq in traditional Persian medicine: a survey of antidotal plants and their modern pharmacological potential. Front. Pharmacol. 16:1503149. doi: 10.3389/fphar.2025.1503149
Received: 28 September 2024; Accepted: 14 March 2025;
Published: 02 April 2025.
Edited by:
Michał Tomczyk, Medical University of Bialystok, PolandReviewed by:
Nilufer Orhan, Independent Researcher, Austin, TX, United StatesAlaadin E. El-Haddad, October 6 University, Egypt
Copyright © 2025 Mirhosseini, Dadmehr, Yousefsani, Seif and Eghbalian. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Fatemeh Eghbalian, ZHJlZ2hiYWxpYW5AZ21haWwuY29t
 Haniye Mirhosseini1,2,3
Haniye Mirhosseini1,2,3 Majid Dadmehr
Majid Dadmehr Farhad Seif
Farhad Seif Fatemeh Eghbalian
Fatemeh Eghbalian