- 1Department of Mental Health, Homerton University Hospital, East London Foundation Trust, London, United Kingdom
- 2Psychopharmacology, Drug Misuse, and Novel Psychoactive Substances Research Unit, School of Life and Medical Sciences, University of Hertfordshire, Hatfield, United Kingdom
- 3Swansea University Medical School, Institute of Life Sciences 2, Swansea University, Swansea, United Kingdom
- 4Psychiatry Unit, Department of Clinical and Experimental Medicine, University of Catania, Catania, Italy
- 5Department of Health Sciences, University of Milan, Milan, Italy
- 6Department of Mental Health, Addictions’ Observatory (ODDPSS), Rome, Italy
- 7Department of Mental Health, Guglielmo Marconi” University, Rome, Italy
- 8Department of Mental Health, ASL Roma 2, Rome, Italy
Background: There is growing availability of novel psychoactive substances (NPS), including cognitive enhancers (CEs) which can be used in the treatment of certain mental health disorders. While treating cognitive deficit symptoms in neuropsychiatric or neurodegenerative disorders using CEs might have significant benefits for patients, the increasing recreational use of these substances by healthy individuals raises many clinical, medico-legal, and ethical issues. Moreover, it has become very challenging for clinicians to keep up-to-date with CEs currently available as comprehensive official lists do not exist.
Methods: Using a web crawler (NPSfinder®), the present study aimed at assessing psychonaut fora/platforms to better understand the online situation regarding CEs. We compared NPSfinder® entries with those from the European Monitoring Centre for Drugs and Drug Addiction (EMCDDA) and from the United Nations Office on Drugs and Crime (UNODC) NPS databases up to spring 2019. Any substance that was identified by NPSfinder® was considered a CE if it was either described as having nootropic abilities by psychonauts or if it was listed among the known CEs by Froestl and colleagues.
Results: A total of 142 unique CEs were identified by NPSfinder®. They were divided into 10 categories, including plants/herbs/products (29%), prescribed drugs (17%), image and performance enhancing drugs (IPEDs) (15%), psychostimulants (15%), miscellaneous (8%), Phenethylamines (6%), GABAergic drugs (5%), cannabimimetic (4%), tryptamines derivatives (0.5%), and piperazine derivatives (0.5%). A total of 105 chemically different substances were uniquely identified by NPSfinder®. Only one CE was uniquely identified by the EMCDDA; no CE was uniquely identified by the UNODC.
Conclusions: These results show that NPSfinder® is helpful as part of an Early Warning System, which could update clinicians with the growing numbers and types of nootropics in the increasingly difficult-to-follow internet world. Improving clinicians’ knowledge of NPS could promote more effective prevention and harm reduction measures in clinical settings.
Introduction
Cognitive enhancement may be defined as “the amplification or extension of core capacities of the mind through improvement or augmentation of internal or external information processing systems” (1). Both non-pharmacological and pharmacological enhancers are sought by the general public in order to improve performance during studying and at work by increasing concentration, motivation and accuracy, via physical, behavioral and biochemical activities (2).
Cognitive enhancer drugs (CEs) are also known as “nootropics” (from the Greek ‘nous’ meaning ‘mind’ and ‘trepein’ meaning ‘turning/bending’), a term initially penned by Corneliu Giurgea when piracetam was found to exhibit memory-enhancing properties in clinical trials (3, 4). Cognitive enhancer drugs such as modafinil improve cognition in very specific ways such that it enhances “pattern recognition memory, digit span recall, and mental digit manipulation” (5).
Cognitive Enhancers, Historical Perspective and State of the Art
Historically, CEs have been used to treat conditions related to cognition deficits such as Alzheimer’s disease, psychiatric disorders such as schizophrenia (6), stroke or attention deficit hyperactivity disorder (ADHD) (7–9). These phenomena commonly occur with aging (7–9). It was found that some CEs also improve cognitive functions in healthy subjects, such as memory, executive functions, creativity, and motivation (10). Their use has become more and more prevalent among college, high school, and university students as well as in the military (11–13).
The world of CEs is multifaceted and complex, with different molecules acting with different modes of actions and on different (and often multiple) receptors in the central nervous system (CNS). “Natural” enhancers such as nicotine (14–17) and caffeine (18) are generally accepted as substances that help us by improving focus, alertness, and productivity. Food-based antioxidants, herbal, and other food-derived nootropic agents have become increasingly popular in recent times after there have been suggestions of associations between cognition and diet (19). Prescription drugs, such as modafinil, amphetamine, and methylphenidate are used off-label by healthy people who do not have specific deficits but want to improve their standards of intellectual and cognitive performance (20). Cognitive enhancers also include many drugs which have never reached the market as they have been discontinued in Phase II or III clinical trials (7–9). The many dimensions of cognitive enhancement are described and disentangled in a recent review (2). Dresler and colleagues (2) pointed out how cognitive enhancement is not a monolithic phenomenon and how there are a great variety of interventions that can be classified and clustered into biochemical, physical, and behavioural enhancement strategies.
Misuse of Cognitive Enhancers
The most prevalent CEs that are currently abused/misused include diverted prescription medicines such as those used for the treatment of attention deficit hyperactivity disorder (ADHD) i.e. methylphenidate (MPH) and amphetamine/dextroamphetamine (Adderall—most common brand); “wakefulness-promoting agents” with psychostimulant effects such as modafinil (21–23); illicit psychostimulants such as amphetamine, and drugs that act on the glutamatergic AMPA receptors, the so-called ampakines or “glutamate activators” (24). While the benefits of medications, such as MPH or modafinil, in patients suffering from specific diagnosed conditions (such as ADHD or narcolepsy) have been studied and evaluated, the potential benefits of these substances in heathy individuals remain unclear. The use of CEs in healthy individuals poses significant concerns due to the lack of clinical evidence regarding their safety, effectiveness, and social consequences, especially with long-term use.
Urban and Gao (24) emphasized that these newly misused drugs, i.e. MPH, may in fact improve cognition by acting on the memory and learning circuits, thus exciting the dopamine/glutamate/noradrenergic neurons. The modulation of these neurotransmitters in healthy individuals seeks to enhance their cognitive functions beyond baseline levels, but may also lead to paradoxical effects, particularly in children’s and adolescent’s growing brains (25). In these cases, glutamate modulation may impair behavior flexibility, which may facilitate addictive behaviors. Conversely, dopamine and norepinephrine reuptake inhibition may lead to a hyperdopamin-/hypernoradrenalin-ergic state, which may induce a cognition decline because the relationship between the prefrontal cortex cognition enhancement and the levels of both dopamine and noradrenaline is non-linear and actually an inverted U-curve (25–27). Urban et al. (28) have also emphasized that the use of CEs such as MPH and modafinil can have short- and long-term impacts on plasticity in the pre-frontal cortex that may affect the potential for plastic learning especially in children and adolescents.
Like many other NPS, nootropics have become increasingly easily available on the internet over the last 20 years. According to the United Nations Office on Drugs and Crime (UNODC) Early Warning Advisory (EWA) on new psychoactive substances (NPS), NPS have been reported from over 100 countries and territories from all regions of the world (29–32). In addition, the European Monitoring Centre for Drugs and Drug Addiction (EMCDDA) has been monitoring more than 700 NPS that have appeared on Europe’s drug market in the last 20 years, of which almost 90% have appeared in the last decade (33, 34). The European Database on New Drugs (EDND) of the EMCDDA records the notifications of new substances and the detection of NPS in Europe (35). Although many of these identified NPS might be used by healthy people as CEs, there are limited data on how many or which substance is, nor are CEs classified as a specific category. Despite being a challenging task in view of the pharmacological differences of CEs, producing a formal classification of these substances is crucial in order to further develop scientific research on the topic as well as regulate and monitor their use and effects.
Previous Findings and Current Challenges
Scientific data regarding NPS used or misused as CEs are lacking. Recent research papers mostly focus on the misuse of specific and well-known CEs such as methylphenidate analogs (36, 37), designer benzodiazepines, phenmetrazine, modafinil, novel synthetic opioids (37), and MPH (38). More literature is available on CEs which are potentially able to address cognitive deficits in specific patient groups. Froestl and Maitre (39) have classified these molecules into 19 categories based on their pharmacodynamics. Some of these molecules could not be classified based on their pharmacodynamics and hence were classified based on their chemical structure or their origin i.e. as natural products or endogenous molecules (39). Many of these drugs were clinically tested for their potential to improve cognitive function. Although they all might have a potential for being misused by the general public looking to enhance their cognitive abilities, the vast majority of these molecules have never reached the market as most of them have been discontinued in Phase II or III clinical trials (7–9).
A comprehensive literature review completed by Froestl et al. (7–9) proposed a description and a classification of 1,705 molecules as “nootropic agents or CEs” in the Thomson Reuters Pharma database, which were studied for their potential to counter cognitive deficits in Alzheimer’s disease. The large number of CEs, reported in the latter review, is attributed to the fact that it contains a high proportion (42%) of molecules that were tested for the treatment of dementia and molecules which were discontinued. Many CEs were described as groups or families (i.e. beta-amyloid aggregation inhibitors). These CEs were not identified by either the EDND, EWA, or NPSfinder® as this is not part of the remit of any of the NPS early identification systems. In particular, many categories of CEs described by Froestl et al. (7–9) such as “Drugs interacting with Cytokines”, “Drugs interacting with Gene Expression”, “Drugs interacting with Heat Shock Proteins”, “Drugs interacting with Hormones”, “Drugs interacting with Ion Channels (different from receptors)”, “Drugs interacting with Nerve Growth Factors”, “Drugs interacting with Transcription Factors”, “Metal Chelators”, “Drugs preventing amyloid-beta aggregation”, “Drugs preventing amyloid-beta aggregation”, “Drugs interacting with tau”, “Stem Cells” include molecules specifically targeted for Alzheimer’s disease and, therefore, less likely to be relevant for the NPS early identification systems.
Apart from the known families of CEs (historically derivatives of MPH, modafinil, and racetams), psychonauts (subjects who experience intentionally drug-induced altered states of consciousness (40) have been experimenting with a variety of commonly prescribed drugs as well as illicit substances, often finding subjective evidence of cognitive enhancement and sharing their knowledge within the dark web sites and surface internet fora. At present, a comprehensive, up-to-date list of currently available CEs does not exist. Moreover, CEs are not described as a specific category/family within the EDND or EWA databases; this is because many substances, with many different (complex and, sometimes, not fully understood) pharmacological mechanisms, have the potential of improving aspects of cognition. Finally, some of these substances are not illegal (i.e. prescribed medication, food supplements, natural remedies etc.). For these reasons, it is difficult to create an early identification system which is able to keep professionals up-to-date with the CEs which are currently available to the general public via the online market.
Aims of the Study
In this study, the aims were to (a) identify and categorize the number of CEs collected by the NPSfinder® web crawler from a range of psychonaut, NPS-related, online sources; (b) compare the NPSfinder® cognitive enhancers’ list with related findings from the UNODC’s EWA and the EMCDDA’s EDND.
Materials and Methods
NPSfinder®, a Tool for the Early Recognition of NPS
NPSfinder® is a crawling/navigating software which was designed to facilitate the early recognition of the continuously growing amount of NPS that are available on the internet. At present, NPSfinder® is a password protected proprietary software, which allows registered researchers only to screen and classify the substances that are identified by the software. An open access part, which will allow the general public to have free access to the substances, is under development.
NPSfinder® automatically scans the web for new/novel/emerging NPS, including CEs, via the identification of psychonauts’ websites/fora. Every time a new website is identified, all its items are scanned and compared with the online existing ones. When a novel substance is found, this is added to the growing NPSfinder® database. NPSfinder® screening process is tailored to each website, and no specific keywords are used by the software. This proprietary method, which was created by trained software engineers, allows to map, on a 24/7 basis, the large variety of psychoactive molecules mentioned/discussed within a range of representative online psychonauts’ web sites/fora. This list is continuously growing (the current, full list of these sites is available upon request).
NPSfinder® was designed to extract a range of information regarding NPS, including: chemical and street names; chemical formulae; three-dimensional images and anecdotally reported clinical/psychoactive effects.
Identification of Cognitive Enhancers by NPSfinder®
NPSfinder® has been already successfully used to identify other types of NPS, including synthetic cathinones (41), novel psychedelics (42), and novel opioids (43). In each paper, the comparison with international or European NPS databases has shown that NPSfinder® is able to identify substances which were not previously described by the existent early detection systems. Raising awareness of novel substances has important implications from both a legislative and a clinical perspective.
Between 26 November 2017 and 31 May 2019, NPSfinder® carried out a range of open web crawling identification activities focusing on a large range of psychonaut-based, specialized, multilingual sources with a specific focus on new/traditional psychoactive substances of likely recreational interest. Although the language most typically used in these websites was English, further languages analyzed by NPSfinder® included: Dutch, French, German, Italian, Russian, Spanish, Swedish, and Turkish. With the help of an ad hoc check control panel, all data were manually examined by four medically/psychiatrically-trained professionals (i.e. FN, DA, CZ, and LG). In this way, a full assessment and editing of each NPSfinder® data item were conducted, and the range of unique CEs presented here was identified.
The collection of further information was completed by consulting a range of open libraries and chemistry databases referring to the index item, if existing. These data were then stored in an online, restricted access/password-controlled database located within firewall protected, highly secure, and consistently performing servers.
When any new item was detected during the automated web scan, the system sent an e-mail notification/alert to the core researchers’ mailing list. Data were then screened for relevance and possible duplications.
The identified psychoactive substances were classified as CEs when a cognitive enhancing ability of any kind (such as improved attention, concentration, alertness, and memory) was mentioned in the description and/or among the effects of the psychoactive substance. The used terms for the search were “nootropic”, “cognitive enhancers”, “cognition enhancement”, “smart drugs”, “memory enhancers”, “concentration enhancers”, “attention enhancers”, “neuro enhancers”, and “intelligence enhancers”. Therefore, it is to be noted that these identified CEs are thought by psychonauts as having cognitive enhancing properties according to their subjective and anecdotical experience rather than due to any pharmacological analysis.
When a substance that was identified by NPSfinder® was not explicitly described as able to enhance cognitive abilities but was listed as a known CE within the comprehensive review by Froestl et al. (7–9), it was still included among the list of NPSfinder® CEs.
Identification and Classification of Cognitive Enhancers
The NPSfinder® CE results (updated to May 2019) were compared with those reported by the UNODC’s EWA on NPS (updated by April 2019) and the EMCDDA’s EDND (updated by April 2019).
Using chemical structure identification and other published information (i.e. published research papers and official databases), researchers assigned each molecule to its drug class, using the classification described by Schifano et al. (44, 45) for NPS. This classification includes the following families: synthetic cannabimimetics, synthetic cathinones, novel psychostimulants, novel derivatives of classic psychedelics phenethylamines/MDMA-like drugs, synthetic opioids, synthetic cocaine substitutes, novel tryptamines derivatives, GABAergic drugs, phencyclidine-like dissociative drugs, piperazine derivatives, herbs/plants, prescribed drugs, and image and performance enhancing drugs (IPEDs).
Results
Identification and Classification of CEs
After about 18 months of operation, the number of substances identified by the web crawler activities was 5,922. By the time of writing (January 2020), 4,204 unique NPS substances were included in the database, and 1,718 out of 5,922 (29.0%) remaining substances were found to be false positives or duplicates. The most common NPS mentioned in psychonauts’ fora included: psychedelic phenethylamines (30.1%); synthetic cannabimimetics (29.8%); and opioids (10.1%).
A total of 142 unique CEs was identified by NPSfinder® (Table A1). Of these, 35 were explicitly described as having nootropic properties by psychonauts; the remaining 107 molecules were classified as CEs as also present in the comprehensive review on CEs written by Froestl et al. (7–9).
Using the classification described by Schifano and colleagues (44, 45), the CEs identified by NPSfinder® (n = 142) were divided into 10 categories; the majority of these substances were classified as plants/herbs/products (29%), prescribed drugs (17%), image and performance enhancing drugs (IPEDs) (15%), and psychostimulants (15%); in addition, there were substances classified as miscellaneous (8%), phenethylamines (6%), GABAergic drugs (5%), cannabimimetic (4%), tryptamines derivatives (0.5%), and piperazine derivatives (0.5%) (Table 1).

Table 1 CEs identified by NPSfinder® using Schifano et al.’s (44) classification (n=142).
Comparison of NPSfinder® Findings With EU and UN NPS-Related Databases
Current NPSfinder® results were compared with the EMCDDA and the UNODC databases in order to ascertain which molecules were also detected and listed by the official European and United Nation early identification systems.
Out of the 142 molecules identified as CEs by NPSfinder®, a total of 105 chemically different substances were uniquely identified by NPSfinder®; of the remaining 37 molecules, 22 were also listed in both the EDND and EWA databases, 15 of which were reported in both the NPSfinder® and in either the EMCDDA (n = 11) or the UN databases (n = 4) (Table A1).
Only one CE was uniquely identified by the EDND (MIQ-001, also called meta-IQ); no CE was uniquely identified by the EWA database.
CEs Identified According to Their Identification Source
Figure 1 shows the number of CEs identified by each source (including NPSfinder®, EDND and EWA database) as well as the ones identified by more than one source. A full list of the CEs is available upon request.
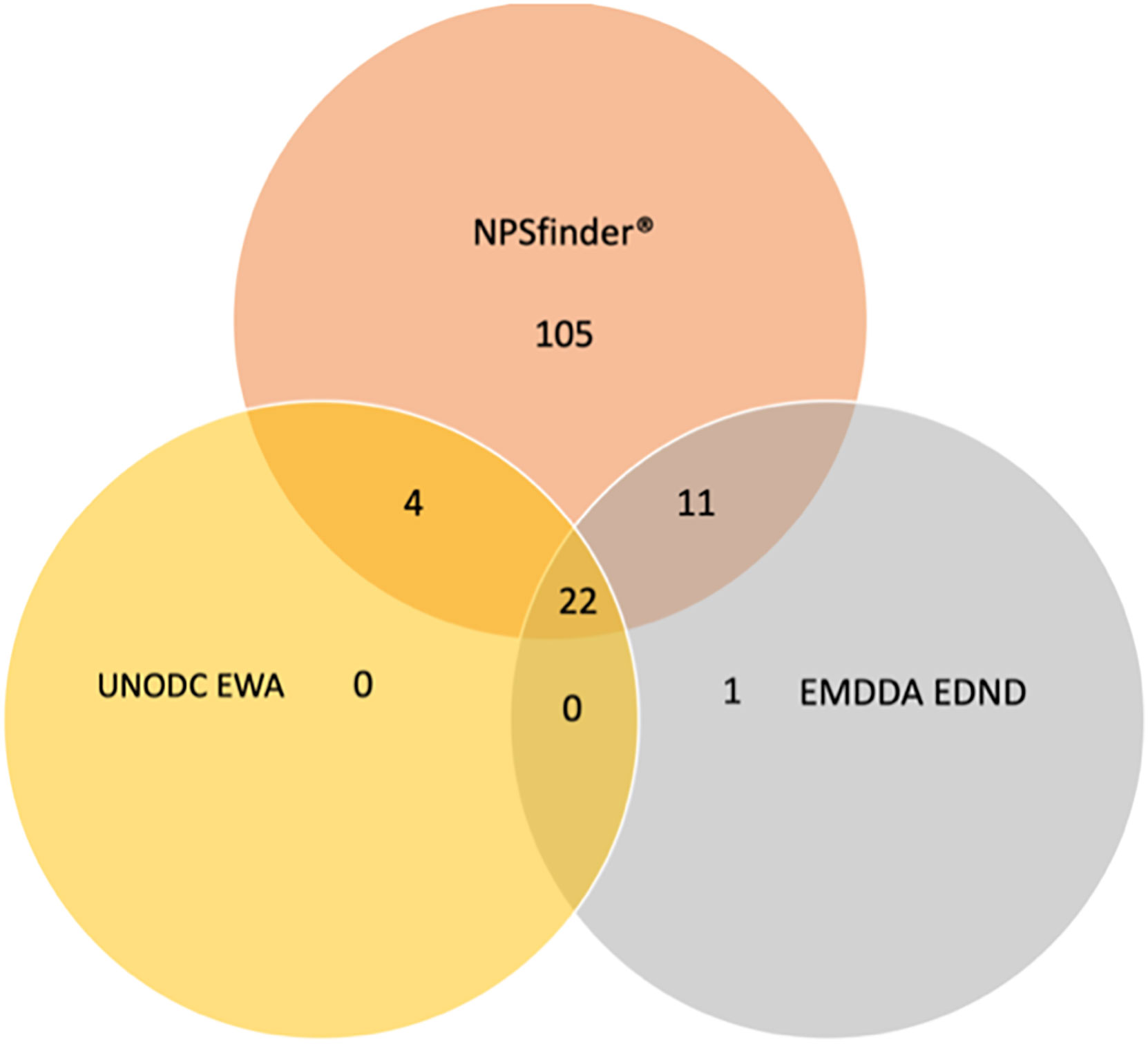
Figure 1 Number of CEs identified by EMCDDA, UNODC, and NPSfinder® according to their identification source (n = 1,785).
Discussion
In this paper, we aimed to evaluate whether the innovative crawling software NPSfinder® can be employed as a helpful tool in the early identification and prediction of CEs. In order to achieve this goal, findings from NPSfinder® were cross-checked with two official sources (EMCDDA’s EDND and UNODC’s EWA). To the best of our knowledge, this is an unprecedented list of drugs which are described as CEs and, therefore, with a potential for recreational misuse by healthy individuals.
NPSfinder® identified 35 molecules (out of the total of 4,204) that were described by psychonauts as having cognitive enhancing effects, such as improved memory, alertness, attention, and concentration. A further 107 molecules were previously described as CE (7–9), although psychonauts did not explicitly describe them as CE. Since psychonauts experiment with novel substances in order to intentionally experience altered states of consciousness, it is to be expected that their interest also extends to the world of CEs. Among the CEs that they have been discussing online, there are mostly molecules that are known to have nootropic properties, are not illegal, and are likely to be easily available on the market (such as racetam compounds, modafinil and its derivatives, methylphenidate and its derivatives and food supplements). Our results showed that NPSfinder® could be employed as an Early Warning System tool to help clinicians with keeping their knowledge up-to-date with the growing numbers and types of nootropics in the increasingly difficult-to-follow online market.
It is not surprising that the included sources (i.e. NPSfinder®, EDND, and EWA) have identified mis-matching numbers and types of CEs, as they differ in their methodology and purposes of CE identification. In fact, the EDND was created in order to allow the European Union to rapidly detect, assess, and respond to health and social threats caused by NPS (35). The UNODC EWA on NPS provides access to basic information on new psychoactive substances, including trend data, chemical details on individual substances, supporting documentation on laboratory analysis and legislative responses (30). Specifically, the EDND and EWA focus on illegal drugs and do not look at websites that contain patented medications, while NPSfinder® looks at websites whose contributors might have accessed sources containing patent medications.
NPSfinder® Findings
The large number of molecules that are both identified by NPSfinder® and described by Froestl et al. (7–9) leads us to believe that nowadays psychonauts are discussing (and likely using) substances that have been considered or used for the treatment of the Alzheimer’s disease over seven years ago, and they are doing so in order to improve their cognitive performances in the absence of clinical reasons.
Among the CEs that have been subjectively identified by psychonauts as able to improve certain aspects of their cognition, there are molecules whose objective cognitive enhancing properties have not been established by research studies, such as the selective serotonin re-uptake inhibitors (SSRIs), melatonin and many others.
Comparison of NPSfinder® Findings With EDND or EWA Databases
The large number of unique molecules that were uniquely identified by NPSfinder® can be explained with the innovative methodology that NPSfinder® used for the early identification of all NPS, including CEs (41–43). Being a dynamic software, NPSfinder® is able to automatically scan the web for new/novel/emerging NPS on a 24/7 basis. This is indeed an effective mechanism for the early identification of (potential) NPS, which are being discussed on the psychonauts’ websites and fora.
Description and Classification of CEs Identified by NPSfinder®
The CEs identified by NPSfinder® (n = 142) were divided into 10 categories as shown in Table 1.
Plants/herbs/product:
The NPSfinder® family of “Plants/herbs/product” contains a list of plant-based substances with a variety of psychoactive ingredients (Table 2).
In this group, there are many well-known substances such as: caffeine, nicotine, cinnamon, ginger root extracts, curcumin, ginseng, coumarins, menthol, St John’s wort, Yerba mate, Bacopa monnieri, Areca nut (and its main active ingredient arecoline), Lemon balm, Mucuna pruriens, Peganum harmala, harmaline, harmalol, and lobeline, some of which are commonly used by ayurvedic traditional medicine or in other branches of alternative medicine to improve memory and/or to treat various diseases. Flavonoids such as quercetin and naringin, as well as vitamins A, B, and D are also part of this group.
There are studies on the cognitive enhancing properties of caffeine (18), nicotine (14–17), curcumin (46–48), St John’s wort (Hypericum perforatum) (49), Bacopa monnieri (50), and many others. Perry and Howes (51) completed an informative review on medicinal plants in dementia, pointing out the potential cognitive benefits of a significant variety of plants and herbs. A recent systematic review has found that tyrosine and caffeine could enhance cognitive performance when healthy young adults are sleep-deprived in a military context (52).
Prescribed drugs:
Methylphenidate is undoubtedly the most prescribed CE, and being indicated for the treatment of ADHD in many countries, it is described, in this paper, within the “prescribed drugs” group. The non-medical use of methylphenidate as a CE, which involves an attempt to improve memory, increase mental concentration, control anxiety, and stimulate motivation and creativity, is rising worldwide (38, 53). Many other prescribed drugs are being talked about in psychonauts’ blogs and fora (Table 3).
Among the “prescribed drugs” family described by NPSfinder® the SSRIs are also listed as a class. Research studies have often failed to demonstrate that SSRIs can have cognitive enhancing properties (54, 55). For example, neither sertraline (54) nor citalopram (55) appeared to be superior to placebo in improving cognition in patients with Alzheimer’s disease and comorbid depression. It was also suggested that any cognitive benefits of SSRIs were likely to be secondary to their effect on mood or behavioral disturbances. However, a more recent review on the topic concluded that the lack of evidence for SSRIs as CEs or disease modifiers in Alzheimer’s disease is more the result of omissions in clinical trial design, as opposed to reports of negative evidence (56). Interestingly, both fluoxetine and methylphenidate potentiate gene regulation in the striatum, and their combination seems to mimic cocaine effects, with related increased risk for substance use disorder (57).
It is possible that many prescribed drugs are currently being misused by the general public but not picked up by the regulatory bodies because the vast majority of them are not classified as illegal. It is important that more studies and cross-sectional surveys are conducted as well as that the current pharmacovigilance systems focus on determining current patterns and quantifying current usage of these drugs by healthy people.
Image and Performance Enhancing Drugs (IPEDs):
Racetam compounds, which are classically one of the major CE family (58), are identified by NPSfinder® and listed within the IPEDs sub-group (Table 4).
Piracetam enhances cognitive function without causing sedation or stimulation (3). This drug is also being used in clinical practice for the treatment of several diseases (59–62) although its mechanism of action remains not fully understood.
NPSfinder® identified aniracetam, coluracetam, fasoracetam, nefiracetam, oxiracetam, phenylpiracetam, piracetam, and pramiracetam. Although all these substances have been mentioned in the psychonauts’ fora as having nootropic properties, research studies have not always succeeded in demonstrating their cognitive enhancing qualities. For example, recent studies failed in showing that aniracetam improves working memory in pigeons (63), learning and memory in rats (64), or cognitive and affective behavior in mice (65). Moreover, nefiracetam did not prove to be more efficacious than placebo in ameliorating apathy in stroke (66) despite some positive pre-clinical results (67, 68). One old study on pramiracetam has failed to demonstrate any cognitive benefit from its administration to patients suffering from Alzheimer’s disease (69). There are no available studies on coluracetam, fasoracetam, and phenylpiracetam.
Psychostimulant drugs:
Among the psychostimulant CEs are described many derivatives of methylphenidate and modafinil (Table 5). These have been listed in this group when not licensed as prescribed drugs.
Methylphenidate is a prescription drug with medical restrictions in several countries, therefore, many illegal analogues have emerged on the internet and darknet drug markets during the last few years (53). The derivatives of methylphenidate that have been identified by NPSfinder® include: 3,4-dichloromethylphenidate, 4-fluoromethylphenidate, 4-methylmethylphenidate, dexmethylphenidate, ethylphenidate, methylmorphenate, and methylnaphthidate.
The derivatives of modafinil include: adrafinil, fladrafinil, flmodafinil, and N-methyl-4,4′-difluoro-modafinil.
Miscellaneous:
The categories “miscellaneous” include amino-acids such as tryptophan and L-tryptophan, 5-hydroxytryptophan, phenylalanine, and theanine, as well as man-made chemicals such as vinpocetine and sulbutiamine and other various molecules such as beta-asarone, PRE-084, and RO-4491533. No research studies are available regarding the misuse of these molecules by healthy subjects in order to ameliorate their cognitive function (Table 6).
Phenethylamines:
The phenethylamines-related compounds that have been identified by NPSfinder® are listed in Table 7.
These are stimulant, entactogenic, and hallucinogenic substances that share similar chemical structures with amphetamine, catecholamines, synthetic cathinones, and other molecules (70).
Phenethylamines are known to enhance mood and empathy in healthy subjects. Substituted phenethylamines also include substituted amphetamines, which have been used as CEs to promote learning and memory but can ultimately lead to addiction (20). Dolder et al. (20) found that MDMA-induced subjective, emotional, sexual, and endocrine effects that were clearly distinct from those of methylphenidate and modafinil. To the best of our knowledge, there are no research studies or case reports focusing on the misuse of specific phenethylamines as CEs by healthy subjects.
GABAergic drugs:
GABAergic drugs are chemicals that produce their effects via interactions with the GABA system, such as by stimulating or blocking neurotransmission (71).
Among these, GABA, tolibut, picamilon, phenibut, and f-phenibut were discussed in the psychonauts’ fora as having tranquillizing as well as nootropic properties (Table 8). There is increasing evidence suggesting that phenibut, a potent psychoactive substance with GABA-B agonist properties which is often sold as a “dietary supplement”, can induce withdrawal and physical dependence which makes its use dangerous (72–76). f-phenibut, which is closely related to phenibut, is a central nervous system depressant (72); tolibut is a GABA analog that was developed in Russia (77), similarly to picamilon, which is formed by a synthetic combination of niacin and γ-aminobutyric acid (GABA). Picamilon was developed in the Soviet Union in 1969 (78) and further studied in both Russia (79) and Japan (80) as a prodrug of GABA.
Cannabimimetic:
Among Cannabimimetic drugs there are the synthetic cannabimimetics that are designer drugs that target the same receptors to which cannabinoids in cannabis plants, tetrahydrocannabinol (THC) and cannabidiol (CBD) bind (81, 82). dexanabinol, drinabant, Dronabinol, JZL-184, rimonabant, and URB-597 were the six CEs belonging to this group that were identified by the NPSfinder® (Table 9).
The use of cannabimimetics as CEs seems counter-intuitive as both pre-clinical and human studies have found a link between consumption of cannabinoids and long-term deficits of cognitive functions, especially high-order cognitive functions (83–88). However, recent pre-clinical studies have found that delta-9-THC can improve cognitive performances in rats (89) and mice (90). THC, in fact, appears to promote hippocampal neurogenesis to prevent neurodegenerative processes occurring in animal models of Alzheimer’s disease, to protect from inflammation-induced cognitive damage, and to restore memory and cognitive function in old mice (91).
Tryptamines derivatives:
5-Methoxytryptamine (5-MT, also called mexamine) (Table 10) was the only tryptamine derivative identified by NPSfinder® (as well as by the EWA). This is a tryptamine derivative closely related to both the serotonin and melatonin neurotransmitters (92). To the best of our knowledge, there are no studies, surveys, or case reports that identified 5-MT as a drug used by healthy people in order to improve their cognitive abilities. Jansen et al. (93) reviewed the efficacy of melatonin in addressing cognitive impairment in dementia but found the evidence for this to be inconclusive.
Piperazine derivatives:
Fipexide (also known as attentil and vigilor) (Table 11) is the only substitute piperazine that has been identified by NPSfinder® as a CE. This was initially developed in Italy in 1983 (94) and used as a CE in Italy and France for the treatment of dementia (95). Fipexide is no longer in use due to the occurrence of rare side-effects (96, 97). On psychonauts’ fora it is described as a molecule able to improve short term memory, attention, learning, and cognition.
Ethical, Clinical and Legal Issues
Ethical issues raised by cognitive enhancement have been debated for over a decade (98), and many experts have identified multiple ethical concerns including risks to mental and health safety (99). While CEs hold significant benefits in improving cognitive impairments in several neuropsychiatric disorders such as Alzheimer’s disease (7–9) and schizophrenia (100), the use of nootropics by healthy individuals clearly poses ethical, clinical, and legal issues, as well as the need to develop a practical policy framework.
Mohamed and Sahakian (101) pointed out that CEs’ use in healthy people might have some advantages, such as: helping reduce disparity in society by mitigating the adverse environmental effects (like poverty) on the brain; improving the performances of people who need to perform at the best of their abilities in every situation (such as surgeons or pilots); finally CEs might also be used by people with undiagnosed disorders (such as ADHD) who might be therefore self-medicating with stimulant medications.
On the other hand, it is of concern that the safety and efficacy of these drugs in healthy individuals in the long-term are still unclear. While some CEs have been studied and research data on their mechanism of action and potential benefit are available, the action, the beneficial effects, and the potential side-effects of the majority of them have yet to be fully described and understood. Furthermore, CEs’ effects (if present at all) seem to be temporary, lasting until their metabolism and elimination (102). Some of these drugs can cause dependence and have a significant range of harmful effects; they can be particularly dangerous to young people as their brains are not fully developed. Studies producing null results and some evidence of task-specific impairments should be also noted (103).
The limited evidence of effectiveness as well as the potential side-effects should be cautiously considered by relevant legislative and regulatory bodies. In 2015, the US Presidential Commission for the Study of Bioethical Issues (104) released a report on CE, reporting up-to-date findings and providing recommendations for clinicians (104). The Australian Alcohol and Drug Foundation has recently raised doubts about the actual cognitive benefits of most CEs, indicating that scientific studies showed only little to no benefits for cognitive enhancement in healthy individuals, while the associated side-effects do pose significant risks to health and safety of the general public (105).
While further research is needed on the topic, the early identification of CEs that are most commonly discussed on the internet will increase clinicians’ awareness of this phenomenon and potentially help them make clinical decisions for patients presenting with psychiatric symptoms or physical health problems related to these substances. NPSfinder® could also be an important tool for analytical toxicologists to focus their efforts on the detection of the most recently misused substances (106, 107).
Limitations
In the online world, a significant variety of molecules/substances are described as CEs by anecdotal report or unofficial sources; it is important to note that the list of CEs is constantly evolving and changing. An official, up-to-date, comprehensive list of CEs is not currently available in the literature. The Early Warning Systems fail in the early detection of these substances as they are mostly legal products such as food supplements or prescribed medication, which are misused by healthy individuals to improve their cognitive abilities.
In addition, there is a lack of an official classification of CEs in families/categories. We based our classification on the one described by Schifano et al. (44). We noted that another type of classification, such as the one described by Froestl et al. (7–9) which is based on substances’ pharmacodynamics properties, is also relevant and useful and could be used when further data on NPS pharmacological properties will be available.
In fact, many CEs do not have a fully understood mechanism of action, which makes it difficult to link them to a specific category; other CEs have multiple mechanisms of actions (i.e. might target several different receptors), and they could therefore belong to more than one category; for example, one CE might belong both to the “prescription drugs” and the “GABAergic drugs” groups. Some of the categories can themselves be very broad and have different types of molecules belonging to it, for example “IPEDs”.
Furthermore, it is important to note that a limited number of languages were used for the screening of molecules on the web, and although many substances were first identified in seizures in Asia, only European languages are used. For all these reasons, forming a comprehensive and definite classification of CEs remains a complex challenge.
Conclusions
In this paper, three different databases, including the innovative crawling software (NPSfinder®) and two official sources (EMCDDA’s EDND, UNODC’s EWA) were cross-checked.
CEs are a wide and diverse group of molecules, constantly growing in terms of numbers as well as availability among the general public and especially via online platforms. CEs differ for pharmacological activity, time, and mode of action, targeted cognitive domain, pharmacodynamic and pharmacokinetic properties, as well as possible short- and long-term side-effects. The popularity of chemicals that are potentially able to augment brain functions is not surprising in a society which constantly demands for increasingly high cognitive performances.
For the current official Early Warning Systems, it is challenging to identify and monitor the use of CEs as they are often sold as legal food supplements or as prescribed medication for a number of medical conditions. Due to its innovative methodology, NPSfinder® has demonstrated its ability to identify a higher number of CEs than the official EMCDDA’s EDND and UNODC’s EWA (108). For this reason, NPSfinder® can be considered as a helpful systematic tool which could update clinicians with the growing numbers and types of nootropics in the increasingly difficult-to-follow internet world.
Previously, Arillotta and colleagues (43) have identified 176 novel opioids which were not listed in either international or European NPS databases, such as EMCDDA or UNODC. This information is useful to stakeholders such as enforcement agents, emergency department, scientific community, prevention program setters, and other regulatory agencies. The same applies to CEs; in particular, the early identification of substances that are misused as CEs and the discovery of novel CEs that were never reported or identified before are crucial to raise the awareness of regulatory bodies. The identification of a drug is key to the treatment of its potential physical and psychiatric effects; if the drug is novel, its description may shed some light on its pharmacokinetics and toxicodynamics, which would in turn inform treatment decision-making in clinical settings.
Improving clinicians’ knowledge of CEs is of paramount importance, and further research in order to clarify mechanism of actions, as well as short- and long-term effects of many CEs is also needed. The early identification and better understanding of the distribution and effects of CEs could promote both more effective prevention and harm reduction measures in clinical settings, including emergency departments, mental health and general practice clinics.
Data Availability Statement
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Ethics Statement
The current study involving human participants were reviewed and approved by the University of Hertfordshire Ethics’ Committee; protocol number: aLMS/SF/UH/02951(3). Written informed consent from the patients/participants OR patients'/participants' legal guardian/next of kin was not required to participate in this study in accordance with the national legislation and the institutional requirements.
Author Contributions
FS and AV have conceived the idea of the manuscript and have coordinated the whole project. FN, CZ, DA, and LG have actually carried out the process of both data collection and systematization. FN performed the literature searching, the analysis of data and drafted the manuscript. FS, JC, and AG supervised the manuscript and contributed to the final version of the manuscript. FS approved the final content of the manuscript. JC provided data from the EMCDDA and UNODC databases for the purposes of this research. FS, JC, and AG have provided relevant epidemiological data and have contributed as well to the drafting and checking of the paper itself.
Conflict of Interest
None of the authors of this paper was directly involved with the website development. AV has conceived the idea of a new early detention software for NPS, which was developed by the professionals at Damicom srl, a small enterprise from Rome (Italy). FS and AV have coordinated the testing of the web crawler. FN, CZ, and DA have suggested minor changes to the software which have made the screening process more precise and efficient.
The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The reviewer, SC, declared a shared affiliation, though no collaboration, with several of the authors, FN, FS, JMC, AG, DA, CZ, and AV to the handling editor.
Acknowledgments
The authors are grateful to Damicom srl, a small enterprise from Rome (Italy), whose professionals have developed the NPSfinder® web crawler and so generously have allowed here the testing of its potential.
The authors are also grateful to the EMCDDA and UNODC for being able to use their databases for the purposes of this research.
Finally, we thank the European Commission for funding the EU-MADNESS and EPS/NPS projects.
Abbreviations
ADHD, attention deficit hyperactivity disorder; CEs, cognitive enhancers; EDND, European Database on New Drugs; EMCDDA, European Monitoring Centre for Drugs and Drug Addiction; EWA, Early Warning Advisory; GABA, gamma-aminobutyric acid; IPEDs, imaging and performance enhancing drugs; MPH, methylphenidate; NPS, novel psychoactive substances; SSRIs, Selective Serotonin Re-uptake Inhibitors; UNODC, United Nations Office on Drugs and Crime.
References
1. Bostrom N, Sandberg A. Cognitive enhancement: methods, ethics, regulatory challenges. Sci Eng Ethics (2009) 15:311–41. doi: 10.1007/s11948-009-9142-5
2. Dresler M, Sandberg A, Bublitz C, Ohla K, Trenado C, Mroczko-Wąsowicz A, et al. Hacking the Brain: Dimensions of Cognitive Enhancement. ACS Chem Neurosci (2019) 10:1137–48. doi: 10.1021/acschemneuro.8b00571
3. Giurgea C. Pharmacology of integrative activity of the brain. Attempt at nootropic concept in psychopharmacology. Actual Pharmacol (Paris) (1972) 25:115–56.
4. Giurgea C. The “nootropic” approach to the pharmacology of the integrative activity of the brain. Cond Reflex (1973) 8:108–15.
5. Turner DC, Robbins TW, Clark L, Aron AR, Dowson J, Sahakian BJ. Cognitive enhancing effects of modafinil in healthy volunteers. Psychopharmacol (Berl) (2003) 165:260–9. doi: 10.1007/s00213-002-1250-8
6. Millan MJ, Agid Y, Brune M, Bullmore ET, Carter CS, Clayton NS, et al. Cognitive dysfunction in psychiatric disorders: Characteristics, causes and the quest for improved therapy. Nat Rev Drug Discovery (2012) 11:141–68. doi: 10.1038/nrd3628
7. Froestl W, Muhs A, Pfeifer A. Cognitive enhancers (nootropics). Part 1: drugs interacting with receptors. J Alzheimer’s Dis (2012) 32:793–887. doi: 10.3233/JAD-2012-121186
8. Froestl W, Muhs A, Pfeifer A. Cognitive enhancers (nootropics). Part 2: drugs interacting with enzymes. J Alzheimers Dis (2013a) 33:547–658. doi: 10.3233/JAD-2012-121537
9. Froestl W, Pfeifer A, Muhs A. Cognitive enhancers (nootropics). Part 3: drugs interacting with targets other than receptors or enzymes. disease-modifying drugs. J Alzheimers Dis (2013b) 34:1–114. doi: 10.3233/JAD-121729
10. Fond G, Micoulaud-Franchi JA, Macgregor A, Richieri R, Miot S, Lopez R, et al. Neuroenhancement in Healthy Adults, Part I: Pharmaceutical Cognitive Enhancement: A Systematic Review. J Clinic Res Bioeth (2015) 6:2. doi: 10.4172/2155-9627.1000213
11. Franke AG, Bonertz C, Christmann M, Huss M, Fellgiebel A, Hildt E, et al. Non-medical use of prescription stimulants and illicit use of stimulants for cognitive enhancement in pupils and students in Germany. Pharmacopsychiatry (2011) 44:60–6. doi: 10.1055/s-0030-1268417
12. Taylor GP Jr, Keys RE. Modafinil and Management of Aircrew Fatigue. United States Department of the Air Force: Washington, DC (2003).
14. Mirza NR, Bright JL. Nicotine-induced enhancements in the five-choice serial reaction time task in rats are strain-dependent. Psychopharmacology (2001) 154:8–12. doi: 10.1007/s002130000605
15. Mirza NR, Stolerman IP. Nicotine enhances sustained attention in the rat under specific task conditions. Psychopharmacology (1998) 138:266–74. doi: 10.1007/s002130050671
16. Rezvani AH, Levin ED. Nicotine-alcohol interactions and attentional performance on an operant visual signal detection task in female rats. Pharmacol Biochem Behav (2003) 76:75–83. doi: 10.1016/S0091-3057(03)00193-X
17. Rezvani AH, Kholdebarin E, Dawson E, Levin ED. Nicotine and clozapine effects on attentional performance impaired by the NMDA antagonist dizocilpine in female rats. Int J Neuropsychopharmacol (2008) 11:63–70. doi: 10.1017/S1461145706007528
18. Cappelletti S, Piacentino D, Sani G, Aromatario M. Caffeine: cognitive and physical performance enhancer or psychoactive drug? Curr Neuropharmacol (2015) 13:71–88. doi: 10.2174/1570159X13666141210215655
19. Onaolapo AY, Obelawo AY, Onaolapo OJ. Brain Ageing, Cognition and Diet: A Review of the Emerging Roles of Food-Based Nootropics in Mitigating Age-Related Memory Decline. Curr Aging Sci (2019) 12:2–14. doi: 10.2174/1874609812666190311160754
20. Dolder PC, Müller F, Schmid Y, Borgwardt SJ, Liechti ME. Direct comparison of the acute subjective, emotional, autonomic, and endocrine effects of MDMA, methylphenidate, and modafinil in healthy subjects. Psychopharmacol (Berl) (2018) 235:467–79. doi: 10.1007/s00213-017-4650-5
21. Johnston LD, O’Malley PM, Bachman JG, Schulenberg JE. Monitoring the Future national results on adolescent drug use: overview of key findings. The University of Michigan, Institute for Social Research: Ann Arbor, MI (2011).
22. Substance Abuse and Mental Health Services (SAMHSA). Results from the 2010 National Survey on Drug Use and Health: Summary of National Findings. Rockville, MD: SAMHSA (2011).
23. Swanson JM, Volkow ND. Increasing use of stimulants warns of potential abuse. Nature (2008) 453:586. doi: 10.1038/453586a
24. Urban KR, Gao WJ. Performance enhancement at the cost of potential brain plasticity: neural ramifications of nootropic drugs in the healthy developing brain. Front Syst Neurosci (2014) 8:38. doi: 10.3389/fnsys.2014.00038
25. Arnsten AF, Li BM. Neurobiology of executive functions: catecholamine influences on prefrontal cortical functions. Biol Psychiatry (2005) 57:1377–84. doi: 10.1016/j.biopsych.2004.08.019
26. Arnsten AF. Stress signalling pathways that impair prefrontal cortex structure and function. Nat Rev Neurosci (2009a) 10:410–22. doi: 10.1038/nrn2648
27. Arnsten AF. Toward a new understanding of attention-deficit hyperactivity disorder pathophysiology: an important role for prefrontal cortex dysfunction. CNS Drugs (2009b) 1(Suppl. 1):33–41. doi: 10.2165/00023210-200923000-00005
28. Urban KR, Li YC, Gao WJ. Treatment with a clinically-relevant dose of methylphenidate alters NMDA receptor composition and synaptic plasticity in the juvenile rat prefrontal cortex. Neurobiol Learn Mem (2013) 101:65–74. doi: 10.1016/j.nlm.2013.01.004
29. United Nations Office on Drugs and Crime [UNODC]. Conventions (2019a). Available at: https://www.unodc.org/unodc/en/commissions/CND/conventions.html (Accessed August 12, 2019).
30. United Nations Office on Drugs and Crime [UNODC]. UNODC Early Warning Advisory (EWA) on New Psychoactive Substances (NPS) (2019b). Available at: https://www.unodc.org/LSS/Home/NPS (Accessed August 8, 2019).
31. United Nations Office on Drugs and Crime [UNODC]. World Drug Report 2019: 35 Million People Worldwide Suffer from Drug Use Disorders While Only 1 in 7 People Receive Treatment. 2019 (2019c). Available at: https://www.unodc.org/unodc/en/frontpage/2019/June/world-drug-report-2019_-35-million-people-worldwide-suffer-from-drug-use-disorders-while-only-1-in-7-people-receive-treatment.html (Accessed August 9, 2019).
32. UNODC Early Warning Advisory on New Psychoactive Substances [UNODC EWA NPS]. News: February 2019 – UNODC-SMART: Almost 900 NPS Reported to UNODC from 119 Countries and Territories (2019d). Available at: https://www.unodc.org/LSS/Announcement/Details/eff8dc38-7ab0-42b0-8cd9-753b89953fcc (Accessed September 30, 2019).
33. European Monitoring Centre for Drugs and Drug Addiction [EMCDDA]. (2018). Luxembourg: European drug report: trends and developments. Publications Office of the European Union (Accessed 11 Jun 2018).
34. European Monitoring Centre for Drugs and Drug Addiction [EMCDDA]. European Drug Report 2019 (2019). Available at: http://www.emcdda.europa.eu/edr2019 (Accessed August 9, 2019).
35. European Database on New Drugs [EDND]. EDND - Login Page (2019). Available at: https://ednd2.emcdda.europa.eu/ednd/login (Accessed August 28, 2019).
36. Carlier J, Giorgetti R, Varì MR, Pirani F, Ricci G, Busardò FP. Use of cognitive enhancers: methylphenidate and analogs. Eur Rev Med Pharmacol Sci (2019) 23(1):3–15. doi: 10.26355/eurrev_201901_16741
37. Batisse A, Eiden C, Peyriere H, Djezzara S, the French Addictovigilance Network. Use of new psychoactive substances to mimic prescription drugs: The trend in France. NeuroToxicology (2020) 79:20–4. doi: 10.1016/j.neuro.2020.03.015
38. Frati P, Kyriakou C, Del Rio A, Marinelli E, Vergallo GM, Zaami S, et al. Smart drugs and synthetic androgens for cognitive and physical enhancement: revolving doors of cosmetic neurology. Curr Neuropharmacol (2015) 13:5–11. doi: 10.2174/1570159X13666141210221750
39. Froestl W, Maitre L. The families of cognition enhancers. Pharmacopsychiatry (1989) 22(Suppl 2):54–100. doi: 10.1055/s-2007-1014626
40. Orsolini L, Papanti GD, Francesconi G, Schifano F. Mind navigators of chemicals’ experimenters? A Web-based Descript E-psychonauts Cyberpsychol Behav Soc Netw (2015) 18:296–300. doi: 10.1089/cyber.2014.0486
41. Schifano F, Napoletano F, Arillotta D, Zangani C, Gilgar L, Guirguis A, et al. The clinical challenges of synthetic cathinones. Br J Clin Pharmacol (2019a) 86:410–9. doi: 10.1111/bcp.14132
42. Schifano F, Napoletano F, Chiappini S, Orsolini L, Guirguis A, Corkery JM, et al. New psychoactive substances (NPS), psychedelic experiences, and dissociation: clinical pharmacological issues. Curr Addict Rep (2019b) 6:140–52. doi: 10.1007/s40429-019-00249-z
43. Arillotta D, Schifano F, Napoletano F, Chiappini S, Orsolini L, Guirguis A, et al. Novel opioids: systematic web crawling within the e-psychonauts’ scenario. Front Neurosci (2020) 14:149. doi: 10.3389/fnins.2020.00149
44. Schifano F, Orsolini L, Duccio Papanti G, Corkery JM. Novel psychoactive substances of interest for psychiatry. World Psychiatry (2015) 14:15–26. doi: 10.1002/wps.20174
45. Schifano F, Napoletano F, Chiappini S, Guirguis A, Corkery JM, Bonaccorso S, et al. New/emerging psychoactive substances and associated psychopathological consequences. Psychol Med (2019c) 22:1–13. doi: 10.1017/S0033291719001727
46. Lim GP, Chu T, Yang F, Beech W, Frautschy SA, Cole GM. The curry spice curcumin reduces oxidative damage and amyloid pathology in an Alzheimer transgenic mouse. J Neurosci (2001) 21:8370–7. doi: 10.1523/JNEUROSCI.21-21-08370.2001
47. Yang F, Lim GP, Begum AN, Ubeda OJ, Simmons MR, Ambegaokar SS, et al. Curcumin inhibits formation of amyloid beta oligomers and fibrils, binds plaques, and reduces amyloid in vivo. J Biol Chem (2005) 280:5892–901. doi: 10.1074/jbc.M404751200
48. Cole GM, Teter B, Frautschy SA. Neuroprotective effects of curcumin. Adv Exp Med Biol (2007) 595:197–212. doi: 10.1007/978-0-387-46401-5_8
49. Ben-Eliezer D, Yechiam E. Hypericum perforatum as a cognitive enhancer in rodents: A meta-analysis. Sci Rep (2016) 6:35700. doi: 10.1038/srep35700
50. Aguiar S, Borowski T. Neuropharmacological review of the nootropic herb Bacopa monnieri. Rejuvenation Res (2013) 16:313–26. doi: 10.1089/rej.2013.1431
51. Perry E, Howes MJ. Medicinal plants and dementia therapy: herbal hopes for brain aging? CNS Neurosci Ther (2011) 17(6):683–98. doi: 10.1111/j.1755-5949.2010.00202.x
52. Pomeroy DE, Tooley KL, Probert B, Wilson A, Kemps E. A Systematic Review of the Effect of Dietary Supplements on Cognitive Performance in Healthy Young Adults and Military Personnel. Nutrients (2020) 12(2):pii: E545. doi: 10.3390/nu12020545
53. Busardò FP, Kyriakou C, Cipolloni L, Zaami S, Frati P. From clinical application to cognitive enhancement: the example of methylphenidate. Curr Neuropharmacol (2016) 14:17–27. doi: 10.2174/1570159X13666150407225902
54. Lyketsos CG, DelCampo L, Steinberg M, Miles Q, Steele CD, Munro C, et al. Treating depression in Alzheimer disease: efficacy and safety of sertraline therapy, and the benefits of depression reduction: the DIADS. Arch Gen Psychiatry (2003) 60:737–46. doi: 10.1001/archpsyc.60.7.737
55. Nyth AL, Gottfries CG. The clinical efficacy of citalopram in treatment of emotional disturbances in dementia disorders. A Nordic multicentre study. Br J Psychiatry (1990) 157:894–901. doi: 10.1192/bjp.157.6.894
56. Chow TW, Pollock BG, Milgram NW. Potential cognitive enhancing and disease modification effects of SSRIs for Alzheimer’s disease. Neuropsychiatr Dis Treat (2007) 3:627–36.
57. Steiner H, Van Waes V, Marinelli M. Fluoxetine potentiates methylphenidate-induced gene regulation in addiction-related brain regions: concerns for use of cognitive enhancers? Biol Psychiatry (2010) 67:592–4. doi: 10.1016/j.biopsych.2009.10.004
58. Wilms W, Woźniak-Karczewska M, Corvini PF, Chrzanowski Ł. Nootropic drugs: Methylphenidate, modafinil and piracetam - Population use trends, occurrence in the environment, ecotoxicity and removal methods - A review. Chemosphere (2019) 233:771–85. doi: 10.1016/j.chemosphere.2019.06.016
59. Khani YA, Andermann F, Andermann E. Antimyoclonic efficacy of piracetam in idiopathic generalized epilepsy. Epilepsia (2005) 46:1145–6. doi: 10.1111/j.1528-1167.2005.04105.x
60. Leuner K, Kurz C, Guidetti G, Orgogozo JM, Müller WE. Improved mitochondrial function in brain aging and Alzheimer disease – the new mechanism of action of the old metabolic enhancer piracetam. Front Neurosci (2010) 4:44. doi: 10.3389/fnins.2010.00044
61. Waegemans T, Wilsher CR, Danniau A, Ferris SH, Kurz A, Winblad B. Clinical efficacy of piracetam in cognitive impairment: a meta-analysis. Dement Geriatr Cogn Disord (2002) 13:217–24. doi: 10.1159/000057700
62. Verma DK, Gupta S, Biswas J, Joshi N, Singh A, Gupta P, et al. New therapeutic activity of metabolic enhancer piracetam in treatment of neurodegenerative disease: Participation of caspase independent death factors, oxidative stress, inflammatory responses and apoptosis. Emerg Med Clin North Am (2018) 32:1–28. doi: 10.1016/j.emc.2013.09.001
63. Phillips H, McDowell A, Mielby BS, Tucker IG, Colombo M. Aniracetam does not improve working memory in neurologically healthy pigeons. PloS One (2019) 14:e0215612. doi: 10.1371/journal.pone.0215612
64. Reynolds CD, Jefferson TS, Volquardsen M, Pandian A, Smith GD, Holley AJ, et al. Oral aniracetam treatment in C57BL/6J mice without pre-existing cognitive dysfunction reveals no changes in learning, memory, anxiety or stereotypy. Vers 3 F1000Res (2017) 6:1452. doi: 10.12688/f1000research.11023.3
65. Elston TW, Pandian A, Smith GD, Holley AJ, Gao N, Lugo JN. Aniracetam does not alter cognitive and affective behavior in adult C57BL/6J mice. PloS One (2014) 9:e104443. doi: 10.1371/journal.pone.0104443
66. Starkstein SE, Brockman S, Hatch KK, Bruce DG, Almeida OP, Davis WA, et al. A Randomized, Placebo-Controlled, Double-Blind Efficacy Study of Nefiracetam to Treat Poststroke Apathy. J Stroke Cerebrovasc Dis (2016) 25:1119–27. doi: 10.1016/j.jstrokecerebrovasdis.2016.01.032
67. Lu XC, Dave JR, Chen Z, Cao Y, Liao Z, Tortella FC. Nefiracetam attenuates post-ischemic nonconvulsive seizures in rats and protects neuronal cell death induced by veratridine and glutamate. Life Sci (2013) 92:1055–63. doi: 10.1016/j.lfs.2013.04.004
68. Fu CY, He XY, Li XF, Zhang X, Huang ZW, Li J, et al. Nefiracetam Attenuates Pro-Inflammatory Cytokines and GABA Transporter in Specific Brain Regions of Rats with Post-Ischemic Seizures. Cell Physiol Biochem (2015) 37:2023–31. doi: 10.1159/000438562
69. Claus JJ, Ludwig C, Mohr E, Giuffra M, Blin J, Chase TN. Nootropic drugs in Alzheimer’s disease: symptomatic treatment with pramiracetam. Neurology (1991) 41:570–4. doi: 10.1212/WNL.41.4.570
70. Nelson ME, Bryant SM, Aks SE. Emerging drugs of abuse. Emerg Med Clin North Am (2014) 32:1–28. doi: 10.1016/j.emc.2013.09.001
71. Froestl W. An historical perspective on GABAergic drugs. Future Med Chem (2011) 3:163–75. doi: 10.4155/fmc.10.285
72. Lapin I. Phenibut (beta-phenyl-GABA): a tranquilizer and nootropic drug. CNS Drug Rev (2001) 7:471–81. doi: 10.1111/j.1527-3458.2001.tb00211.x
73. Samokhvalov AV, Paton-Gay CL, Balchand K, Rehm J. Phenibut dependence. BMJ Case Rep (2013), bcr2012008381. doi: 10.1136/bcr-2012-008381
74. Brunner E, Levy R. Case report of physiologic phenibut dependence treated with a phenobarbital taper in a patient being treated with buprenorphine. J Addict Med (2017) 11:239–40. doi: 10.1097/ADM.0000000000000303
75. Owen DR, Wood DM, Archer JR, Dargan PI. Phenibut (4-amino-3-phenyl-butyric acid): availability, prevalence of use, desired effects and acute toxicity. Drug Alcohol Rev (2016) 35:591–6. doi: 10.1111/dar.12356
76. Van Hout MC. A narrative review of the naturally occurring inhibitory neurotransmitter gamma-aminobutyric acid (GABA) called phenibut in dietary supplements. Perform Enhance Health (2018) 6:33–5. doi: 10.1016/j.peh.2018.02.001
77. Dmitriev AV, Andreev N. [The spectrum of analgesic activity of baclofen and tolibut]. Farmakol Toksikol (1987) 50:24–7.
78. Kopelevich VM, Gunar VI. Some approaches to the directed search for new drugs based on nicotinic acid. Pharmaceut Chem J (1999) 33:177–87. doi: 10.1007/BF02509934
79. Mirzoian RS, Gan’shina TS. [The new cerebrovascular preparation pikamilon]. Farmakol I Toksikol Russian) (1989) 52:23–6.
80. Matsuyama K, Yamashita C, Noda A, Goto S, Noda H, Ichimaru Y, et al. Evaluation of isonicotinoyl-gamma-aminobutyric acid (GABA) and nicotinoyl-GABA as pro-drugs of GABA. Chem Pharm Bull (1984) 32:4089–95. doi: 10.1248/cpb.32.4089
81. Macher R, Burke TW, Owen SS. Synthetic Marijuana. Law Enforcement Bulletin: FBI (2018). Retrieved 2018-05-04.
82. De Luca MA, Di Chiara G, Cadoni C, Lecca D, Orsolini L, Papanti D, et al. Cannabis; Epidemiological, Neurobiological and Psychopathological Issues: An Update. CNS Neurol Disord Drug Targ (2017) 16:598–609. doi: 10.2174/1871527316666170413113246
83. Volkow ND, Swanson JM, Evins AE, DeLisi LE, Meier MH, Gonzalez R, et al. Effects of cannabis use on human behavior, including cognition, motivation and psychosis: A review. JAMA Psychiatry (2016) 73:292–7. doi: 10.1001/jamapsychiatry
84. Curran HV, Freeman TP, Mokrysz C, Lewis DA, Morgan CJ, Parsons LH. Keep off the grass? Cannabis, cognition and addiction. Nat Rev Neurosci (2016) 17:293–306. doi: 10.1038/nrn.2016.28
85. Papanti D, Schifano F, Botteon G, Bertossi F, Mannix J, Vidoni D, et al. “Spiceophrenia”: A systematic overview of “Spice”-related psychopathological issues and a case report. Hum Psychopharmacol (2013) 28:379–89. doi: 10.1002/hup.2312
86. Cohen K, Kapitány-Fövény M, Mama Y, Arieli M, Rosca P, Demetrovics Z, et al. The effects of synthetic cannabinoids on executive function. Psychopharmacology (2017) 234:1121–34. doi: 10.1007/s00213-017-4546-4
87. Crean RD, Crane NA, Mason BJ. An evidence-based review of acute and long-term effects of cannabis use on executive cognitive functions. J Addict Med (2011) 5:1–8. doi: 10.1097/ADM.0b013e31820c23fa
88. Pattij T, Wiskerke J, Schoffelmeer ANM. Cannabinoid modulation of executive functions. Eur J Pharmacol (2008) 585:458–63. doi: 10.1016/j.ejphar.2008.02.099
89. Suliman NA, Taib CNM, Moklas MAM, Basir R. Delta-9-Tetrahydrocannabinol (Δ9-THC) Induce Neurogenesis and Improve Cognitive Performances of Male Sprague Dawley Rats. Neurotox Res (2018) 33:402–11. doi: 10.1007/s12640-017-9806-x
90. Bilkei-Gorzo A, Albayram O, Draffehn A, Michel K, Piyanova A, Oppenheimer H, et al. A chronic low dose of Δ9-tetrahydrocannabinol (THC) restores cognitive function in old mice. Nat Med (2017) 23:782–7. doi: 10.1038/nm.4311
91. Calabrese EJ, Rubio-Casillas A. Biphasic effects of THC in memory and cognition. Eur J Clin Invest (2018) 48:e12920. doi: 10.1111/eci.12920
92. Mashkovskii MD, Arutiunian GS. [On the pharmacology of 5-methoxytryptamine hydrochloride (Mexamine)]. Farmakol Toksikol (1963) 26:10–7.
93. Jansen SL, Forbes DA, Duncan V, Morgan DG. Melatonin for cognitive impairment. Cochrane Database Syst Rev (2006) 25:CD003802. doi: 10.1002/14651858.CD003802.pub2
94. Missale C, Pasinetti G, Govoni S, Spano PF, Trabucchi M. [Fipexide: a new drug for the regulation of dopaminergic system at the macromolecular level]. Art Italian Boll Chim Farm (1983) 122:79–85.
95. Bompani R, Scali G. Fipexide, an effective cognition activator in the elderly: a placebo-controlled, double-blind clinical trial. Curr Med Res Opin (1986) 10:99–106. doi: 10.1185/03007998609110426
96. Guy C, Blay N, Rousset H, Fardeau V, Ollagnier M. [Fever caused by fipexide. Eval Natl Pharmacovigil Survey Ther (1990) 45:429–31. French.
97. Guy C, Rousset H, Cathebras P, Vial N, Ode P, Ollagnier M. [Fever after taking fipexide hydrochloride]. Presse Med (1989) 18:1076. French.
98. Farah MJ, Illes J, Cook-Deegan R, Gardner H, Kandel E, King P, et al. Neurocognitive enhancement: What can we do and what should we do? Nat Rev Neurosci (2004) 5:421–5. doi: 10.1038/nrn1390
99. Zaami S, Tagliabracci A, Berretta P, Busardò FP, Marinelli E. Use of Methylphenidate Analogues as Cognitive Enhancers: The Prelude to Cosmetic Neurology and an Ethical Issue. Front Psychiatry (2020) 10:1006:1006. doi: 10.3389/fpsyt.2019.01006
100. Lees J, Michalopoulou PG, Lewis SW, Preston S, Bamford C, Collier T, et al. Modafinil and cognitive enhancement in schizophrenia and healthy volunteers: the effects of test battery in a randomised controlled trial. Psychol Med (2017) 47:2358–68. doi: 10.1017/S0033291717000885
101. Mohamed AD, Sahakian BJ. The ethics of elective psychopharmacology. Int J Neuropsychopharmacol (2012) 15:559–71. doi: 10.1017/S146114571100037X
102. De Jongh R, Bolt I, Schermer M, Olivier B. Botox for the brain: enhancement of cognition, mood and pro-social behavior and blunting of unwanted memories. Neurosci Biobehav Rev (2008) 32:760–76. doi: 10.1016/j.neubiorev.2007.12.001
103. Hall WD, Lucke JC. The enhancement use of neuropharmaceuticals: more scepticism and caution needed. Addiction (2010) 105:2041–3. doi: 10.1111/j.1360-0443.2010.03211.x
104. Bioethics Commission: Presidential Commission for the Study of Bioethical Issues. Gray Matters: Topics at the Intersection of Neuroscience, Ethics, and Society (2015). Available at: https://bioethicsarchive.georgetown.edu/pcsbi/node/4704.html (Accessed October 2019).
105. Alcohol and Drug Foundation. Nootropics (2019). Available at: https://adf.org.au/drug-facts/cognitive-enhancers/ (Accessed October 2019).
106. Marchei E, Palmi I, Pichini S, Pacifici R, Anton Airaldi IR, Costa Orvay JA, et al. Segmental hair testing to disclose chronic exposure to psychoactive drugs. Adicciones (2016) 28:158–62. doi: 10.20882/adicciones.825
107. Barceló B, Gomila I, Rotolo MC, Marchei E, Kyriakou C, Pichini S, et al. Intoxication caused by new psychostimulants: analytical methods to disclose acute and chronic use of benzofurans and ethylphenidate. Int J Legal Med (2017) 131:1543–53. doi: 10.1007/s00414-017-1648-9
108. Schifano F. Analyzing the Open/Deep Web to Better Understand the New/Novel Psychoactive Substances (NPS) Scenarios: Suggestions from CASSANDRA and NPS.Finder Research Projects. Brain Sci (2020) 10:146. doi: 10.3390/brainsci10030146
Appendix.
Keywords: cognitive enhancers, nootropics, novel psychoactive substances, novel psychoactive substances, screening, early warning systems
Citation: Napoletano F, Schifano F, Corkery JM, Guirguis A, Arillotta D, Zangani C and Vento A (2020) The Psychonauts’ World of Cognitive Enhancers. Front. Psychiatry 11:546796. doi: 10.3389/fpsyt.2020.546796
Received: 29 March 2020; Accepted: 17 August 2020;
Published: 11 September 2020.
Edited by:
Simona Pichini, National Institute of Health (ISS), ItalyReviewed by:
Stefania Chiappini, University of Hertfordshire, United KingdomJeremy Carlier, Sapienza University of Rome, Italy
Copyright © 2020 Napoletano, Schifano, Corkery, Guirguis, Arillotta, Zangani and Vento. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Fabrizio Schifano, Zi5zY2hpZmFub0BoZXJ0cy5hYy51aw==
 Flavia Napoletano
Flavia Napoletano Fabrizio Schifano
Fabrizio Schifano John Martin Corkery
John Martin Corkery Amira Guirguis
Amira Guirguis Davide Arillotta
Davide Arillotta Caroline Zangani
Caroline Zangani Alessandro Vento6,7,8
Alessandro Vento6,7,8









