Abstract
Background:
Sleep constitutes approximately one-third of human life and is vital for health maintenance. Although previous studies have established an association between physical activity (PA) and sleep quality, research on the effect of PA in improving objective sleep indices remains scarce. This study investigates the impact of PA levels on sleep quality and provides direct experimental evidence to support sleep quality interventions among college students.
Methods:
PA and sleep parameters were assessed using sleep diaries, actigraphy, and polysomnography (PSG). The spectral analysis was performed on PSG-acquired electroencephalographic (EEG) data to examine power distribution variations across distinct sleep stages. Concurrently, venous blood samples were collected for quantitative analysis of serum brain-derived neurotrophic factor (BDNF) levels using enzyme-linked immunosorbent assay (ELISA). Cognitive functions were assessed using the Psychomotor vigilance task (PVT).
Results:
Actigraphy-derived 7-day average daily steps demonstrated significant positive correlations with PSG-measured sleep efficiency (SE) and non-rapid eye movement sleep stage 2 (N2), while exhibiting negative correlations with wake after sleep onset (WASO) and wake stage. The results of multiple linear regression analysis showed that with the increase in the average daily steps, SE and N2 time increased, and wake stage and WASO decreased. Spectral analysis of sleep EEG data demonstrated that average daily steps positively correlated with the mean power of slow sigma and fast sigma during NREM sleep, as well as mean theta power during REM sleep. Additionally, this study revealed that higher average daily steps correlated with elevated BDNF concentrations and reduced reaction time in the PVT task.
Conclusions:
This study demonstrates that there is an association between PA and the modulation of sleep architecture and cognitive function among college students. These findings gain additional support from sleep EEG microstructural analyses, with the observed improvements potentially mediated through PA-mediated modulations in circulating BDNF levels.
Introduction
Sleep is one of the most fundamental biological processes, essential for maintaining both physical and psychological health. Adequate and high-quality sleep plays a crucial role in restoring energy and physical strength, protecting the brain, and regulating emotional states (1, 2). As a vital component of a healthy lifestyle, sleep quality has been closely linked to heart health, metabolic efficiency, cognitive function, and overall mental well-being. However, rapid socioeconomic development and resulting changes in lifestyle and work patterns over recent decades have led to a dramatic increase in sedentary behavior, particularly among younger populations such as college students. At the same time, the prevalence of insufficient or poor-quality sleep has steadily risen, raising public health concerns about its short- and long-term consequences (3, 4).
Physical activity (PA) is defined as any bodily movement produced by skeletal muscles that results in energy expenditure. It includes a wide range of activities such as occupational work, sports, exercise, and household chores (5). The strong interrelationship between PA and sleep health has been recognized in various studies; it is widely accepted that regular and moderate PA may improve sleep onset, duration, and depth (6, 7). For college students, a population facing academic stress, lifestyle irregularities, and frequent use of electronic devices, sleep disturbance and insufficient activity levels are particularly prevalent. Identifying accessible and effective strategies to promote better sleep in this group thus has significant importance.
Despite the established association between PA and sleep, there remain several critical research gaps. First, most studies have relied on subjective self-report questionnaires to evaluate sleep quality, which, although convenient, are prone to bias and may not fully capture objective changes in sleep architecture. Second, there is a lack of consensus on the most valid and user-friendly indicator of daily PA for predicting sleep outcomes in real-world settings; while measures such as calorie consumption or metabolic equivalents are commonly used, they often require complex calculations or are less intuitive for the general public. In contrast, the daily steps, an easily understood and widely accessible parameter, has been proposed as a practical metric for assessing PA in diverse populations (8–10). Higher daily steps have been associated with fewer depressive symptoms in both cross-sectional and longitudinal studies conducted among the general adult population (11). A population-based prospective cohort study further revealed that walking 10,000 steps per day might be linked to reduced mortality rates, as well as lower incidences of cancer and cardiovascular diseases (9). With the advent of portable monitoring technology, such as actigraphy, it has become possible to non-invasively and reliably track both daily movement and sleep-wake patterns (12).
The mechanisms linking PA and sleep require further exploration. Evidence indicates that evening exercise increases nocturnal non-rapid eye movement sleep (NREM) latency, particularly by enhancing slow-wave activity on the electroencephalogram (EEG) (13). This slow-wave sleep promotes bodily and neural recovery, facilitates the clearance of metabolic waste, and supports memory consolidation (14). Sleep has also been found to be involved in the regulation of neuroendocrine factors. Brain-derived neurotrophic factor (BDNF), abundantly present in brain regions critical for learning and memory, is essential for neuronal survival, differentiation, and synaptic plasticity, and also plays a key role in sleep regulation (15). Studies show that moderate aerobic exercise significantly raises BDNF levels in both the central and peripheral nervous systems, with the magnitude of this effect influenced by exercise intensity and duration. Elevated BDNF contributes to greater brain plasticity and offers protection against depression and anxiety (16, 17).
To address these gaps, the present study investigated the effect of PA, measured by daily steps, on sleep quality among college students using both subjective and objective methods. By employing actigraphy for continuous PA assessment, sleep diary recording, and polysomnography (PSG)-the gold standard for sleep measurement-as well as serum BDNF sampling across the sleep-wake cycle, this research aims to not only delineate the relationships among PA, sleep quality, cognitive function, and BDNF secretion, but also provide practical evidence for clinical interventions. By focusing on a homogeneous sample of college students and rigorously excluding individuals with clinically significant anxiety or depression, this study also seeks to minimize confounding factors and clarify the potential of increasing average daily steps as an accessible strategy for improving sleep quality.
In summary, this study aims to investigate the relationship between the amount of daily physical activity and both nighttime sleep quality and daytime functioning, as well as to explore the neural mechanisms that may mediate the associations among physical activity, sleep architecture, and cognitive function. The research results may provide information for sleep management and health promotion strategies in college campuses and other settings, and offer a scientific basis for promoting daily PA as a means to improve the sleep and overall health of young people.
Materials and methods
Subjects
Twenty-three college students were recruited via advertisements. All participants confirmed maintaining a stable sleep-wake cycle (bedtime: 23:00 ± 30 minutes; wake time: 07:00 ± 30 minutes) for a minimum of six consecutive months prior to study enrollment, as verified through one-week sleep diary. Exclusion criteria included: (1) current or past medical illness; (2) regular use of psychoactive substances (caffeine, alcohol, tobacco); (3) any medication within 6 months; and (4) recent shift work or trans-meridian travel (in the past 6 months). Eligible candidates were screened, including a structured clinical interview and physical examination based on DSM-IV. The study protocol (Chinese Clinical Trial Registry ID: ChiCTR-ROC-17012112) received ethical approval from Peking University Sixth Hospital’s Institutional Review Board. All participants provided written informed consent prior to enrollment. Complete demographic and baseline characteristics are presented in Table 1.
Table 1
| Characteristic | Sample amount | Characteristic | Sample amount | ||
|---|---|---|---|---|---|
| Male (%) | 10 (43.47%) | BMI | 21.77 ± 2.09 | ||
| Age (years) | 22.70 ± 2.36 | HAMD | 1.26 ± 1.42 | ||
| Undergraduate degree | 16.35 ± 1.85 | HAMA | 1.65 ± 1.23 | ||
| Average daily steps | 11085.63 ± 1786.40 | ACT | TAK | 2.50 ± 0.77 | |
| Sleep diary | TIB | 496.24 ± 20.38 | PSG | TST | 446.17 ± 40.13 |
| TST | 466.30 ± 22.80 | SE (%) | 89.72 ± 7.92 | ||
| SOL | 19.86 ± 7.39 | SOL | 17.63 ± 25.41 | ||
| NAK | 0.62 ± 0.64 | WASO | 33.39 ± 35.53 | ||
| ACT | TST | 428.38 ± 31.11 | W | 43.30 ± 37.53 | |
| SE (%) | 85.87 ± 5.36 | R | 81.09 ± 17.32 | ||
| SOL | 8.61 ± 5.58 | N1 | 37.85 ± 12.64 | ||
| WASO | 62.59 ± 27.25 | N2 | 208.93 ± 34.20 | ||
| NAK | 24.11 ± 7.25 | N3 | 118.30 ± 30.26 | ||
General clinical data of subjects (n=23).
The data are expressed as the mean ± SD or numbers (%). HAMD, Hamilton depression rating scale; HAMA, Hamilton anxiety rating scale; BMI, mean body index. ACT, actigraphy; PSG, polysomnography; TIB, total time in bed; TST, total sleep time; SOL, sleep onset latency; NAK, number of awakenings; SE, sleep efficiency; WASO, wake time after sleep onset; TAK, average time of awakenings; W, wake stage; REM, rapid eye movement sleep time; N1, duration of non-rapid eye movement sleep stage 1; N2, duration of non-rapid eye movement sleep stage 2; N3, duration of non-rapid eye movement sleep stage 3.
Experimental design
Prior to the formal experiment, comprehensive demographic data collection and standardized psychometric assessments were conducted for participant screening. Qualified participants were instructed to maintain stable PA regimens and habitual sleep-wake cycles throughout the study period, with significant deviations prohibited. All subjects underwent continuous wrist-actigraphy monitoring and maintained detailed sleep diaries for seven consecutive days to establish baseline activity-rest patterns. To control for potential first-night effects and screen for sleep pathologies, initial PSG recordings were obtained on Night 6, with primary PSG data collected on Night 7 for subsequent analysis. Fasting venous blood samples were acquired immediately following the final PSG monitoring (Figure 1).
Figure 1

Flow chart of the study. Prior to experimental commencement, participants underwent demographic data collection and psychometric assessment. All subjects completed sleep diary and actigraphy records for 7 consecutive days. Overnight PSG was conducted on nights 6 and 7, with fasting venous blood samples collected following the final PSG monitoring. PSG, polysomnography.
Questionnaire and scale assessment
Prior to the experiment, all participants underwent a comprehensive screening process, including a detailed interview and baseline assessments such as age, gender, years of education, height, and weight. Participants provided detailed self-reported information about their daily activities and medical history, with particular attention to any mental or physical illnesses, especially sleep-related disorders. The study employed the 24-item Hamilton depression rating scale (HAMD) to evaluate the severity of depression in participants over the past week (18). Similarly, the Hamilton anxiety rating scale (HAMA) was used to assess anxiety levels during the same period (19). To minimize confounding effects, participants with HAMD scores ≥8 or HAMA scores ≥7 were excluded from the study.
Subjective sleep assessment
Participants maintained a sleep diary for one week, documenting bedtime (lights-off time), sleep onset latency (SOL), nocturnal awakenings, morning awakening and rising times, total sleep time (TST), post-awakening subjective feelings, and additional factors such as caffeine intake, medication use, and PA levels. Total time in bed (TIB), TST, SOL, and number of awakenings (NAK) were computed to assess participants’ subjective sleep quality.
Wrist actigraphy for PA and sleep monitoring
Actigraphy (wGT3x-BT, Actigraphy, ACT) is a device for assessing activity levels and objectively measuring PA and sleep patterns (20). Following data collection, ActiLife 6 and MATLAB software are used to process the data and generate a clinical report based on sleep diary records. The recorded data includes subject name, wearing time, daily steps, TST, sleep efficiency (SE), SOL, wake time after sleep onset (WASO), NAK, and average time of awakenings (TAK). To minimize variability in average daily steps, participants were required to wear the device for 7 consecutive days with a minimum wearing time of 2 hours per day (21). Wearing time during sleep must exceed 1 hour, otherwise, the data is deemed invalid (22). Only data meeting the criteria for at least 4 days was included in the statistical analysis.
PSG to monitor sleep parameters and spectral-frequency analysis
The PSG recordings were performed using a Compumedics Grael series PSG device (Compumedics Sleep Study System, Melbourne, Australia). This system continuously monitored participants’ sleep architecture throughout the night, acquiring EEG signals from F3, F4, C3, C4, O1, and O2 channels, along with electrooculogram (EOG) and electromyography (EMG) signals. Simultaneously, it monitored snoring, oral/nasal airflow, and blood oxygen saturation. The recording parameters were standardized as follows: a sampling frequency of 256 Hz with high-pass and low-pass filters set at 0.3 Hz and 35 Hz, respectively. Prior to monitoring, electrode impedances were verified for all channels, and any location exceeding 5 kΩ was repositioned to meet the recording standard. PSG recordings were conducted in an environment with controlled temperature and humidity to minimize noise interference.
The PSG data were interpreted and analyzed using Compumedics Profusion PSG3 sleep analysis software. Sleep stages were scored in 30-second epochs, while limb movements and respiratory events were analyzed in 2-minute epochs. The following sleep parameters were derived and reported: TST, SE, SOL, wake stage (W), non-rapid eye movement sleep stage 1 (N1), non-rapid eye movement sleep stage 2 (N2), non-rapid eye movement sleep stage 3 (N3), rapid eye movement sleep (REM) stage and WASO.
For power spectral analysis of EEG signals, all preprocessed sleep EEG data underwent rigorous manual artifact inspection to EOG and EMG, thereby ensuring high data quality. Spectral analysis of artifact-free EEG data was performed using the Welch method (fast Fourier transform, FFT) in MATLAB (The MathWorks Inc., Natick, MA). Parameters included a 5-second Hanning window applied to consecutive non-overlapping segments of 30-second epochs with 50% overlap, providing a frequency resolution of 0.25 Hz. Average power for specific frequency bands was then calculated separately for NREM and REM stage (23). Relative power was computed for seven clinically relevant frequency bands: slow oscillations (SO, 0.5-1.0 Hz), slow-wave activity (SWA, 0.5-4.0 Hz), delta (1.0-4.0 Hz), theta (4.0-8.0 Hz), slow sigma (9.0-12.0 Hz), fast sigma (12.0-15.0 Hz), and beta (16.0-30.0 Hz) (24–26).
Enzyme-linked immunosorbent assay (ELISA)
Blood samples were collected within 30 minutes of waking on day 7. Samples were clotted at room temperature for 60 min, centrifuged at 1000 × g for 15 min, and the serum was immediately aliquoted and stored at −80°C until analysis. Serum BDNF levels were quantified using ELISA kits (DBD00, R&D Systems, Minneapolis, MN, USA).
Psychomotor vigilance task
After the PSG recording on the second night of this study, the participants were arranged to take the test in a quiet and comfortable environment. The PVT protocol, implemented using E-Prime software (version 1.0), presented participants with a red fixation square centrally positioned on the display. Following variable interstimulus intervals (ranging from 1,000 to 9,000 ms), a millisecond counter (yellow digits) appeared, initiating from 0 ms. Participants were instructed to respond to stimulus onset as rapidly as possible via left mouse button press within the 30,000 ms response window. Successful responses immediately halted the counter and triggered visual feedback (yellow box) displaying the achieved reaction time (RT) for 2,000 ms before trial termination. The complete assessment comprised 90 trials with an approximate total duration of 10 minutes. Post-test analysis classified trials according to conventional PVT performance metrics: lapses (RT ≥ 500 ms) and false starts (RT ≤ 150 ms), with valid responses defined as 150 ms < RT < 500 ms.
Statistical analysis
All statistical analyses were conducted using SPSS 26.0 (IBM Corp., Armonk, NY, USA) and GraphPad Prism 9.0 (GraphPad Software, San Diego, CA, USA). Continuous variables are expressed as mean ± standard deviation (SD). Normality of data distribution was assessed using Shapiro-Wilk tests. Based on the normality test results, we employed Pearson correlation analysis for normally distributed data and Spearman correlation analysis for non-normally distributed data. To evaluate the relationship between PA and sleep quality, multiple linear regression analysis was performed. Statistical significance was set at P < 0.05.
Results
Demographics and clinical characteristics of the subjects
Table 1 presents the demographic and clinical characteristics of all participants. This study comprised 10 males and 13 females (mean age = 22.70 ± 2.36 years; mean BMI = 21.77 ± 2.09 kg/m²). All participants scored within normal ranges on depression and anxiety questionnaires. Actigraphy data revealed average daily steps of 11,085.63 ± 1,786.40. The study enrollment process is illustrated in Figure 1.
Correlation between average daily steps and sleep parameters by sleep diary, ACT, and PSG
Subjective sleep parameters were documented via standardized sleep diary, contrasting with the objective physiological measurements obtained through ACT and PSG. The correlation analyses were employed to investigate potential covariation between habitual PA (operationalized as average daily steps) and these distinct sleep assessment paradigms. The results showed that the average daily steps were significantly associated with the SOL (r = -0.43, P = 0.039), but no significant association was observed with TIB, TST, or NAK in sleep diary (Figure 2). ACT data revealed that average daily steps exhibited a positive correlation with TST (r = 0.46, P = 0.028) and SE (r = 0.42, P = 0.044), but no significant correlation with SOL, WASO, NAK, or TAK (Figure 2). Additionally, our study demonstrated that average daily steps were positively associated with SE (r = 0.42, P = 0.048), and N2 stage (r = 0.48, P = 0.021), and inversely associated with WASO (r = -0.56, P = 0.005) and wake stage (r = -0.49, P = 0.018). Notably, no significant correlation was observed with TST, SOL, REM, N1, or N3 stage in the PSG data (Figure 2). To comprehensively investigate the relationship between PA and sleep quality among college students, we conducted a multivariate linear regression analysis using stepwise entry method. The model incorporated gender, age, education level, BMI, HAMD and HAMA scores as covariates, with average daily steps (measured in 1000-step increments) as the primary predictor and PSG-derived sleep parameters as dependent variables. Our regression analysis revealed the subjects increased average of 1,000 steps per day, and the sleep parameters SE and N2 stage increased by 1.85% and 9.16 minutes, and WASO and wake stage decreased by 11.34 minutes and 10.35 minutes recorded by PSG, respectively (Table 2). Our data show that increased PA may enhance sleep quality by both prolonging restorative N2 stage and reducing nocturnal awakenings. Notably, this study reveals measurable discrepancies between subjective self-reported sleep data and objective sleep measurements, highlighting the importance of differentiating these assessment methods when evaluating sleep quality.
Figure 2
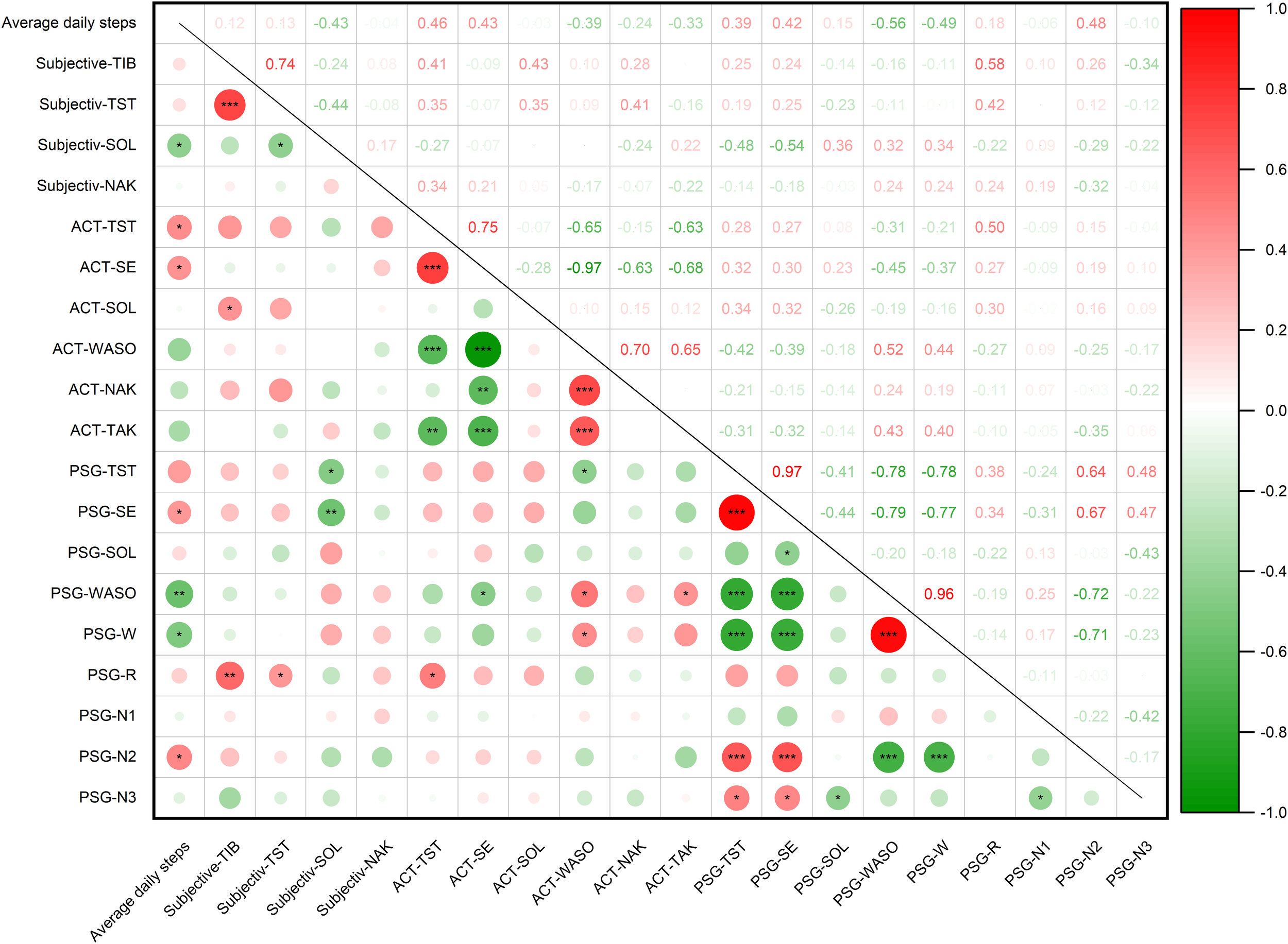
The correlation between the average daily steps and sleep parameters by sleep diary, ACT and PSG. TIB, total time in bed; TST, total sleep time; SOL, sleep onset latency; NAK, number of awakenings; SE, sleep efficiency; WASO, wake time after sleep onset; TAK, average time of awakenings; W, wake stage; REM, rapid eye movement sleep stage; N1, duration of non-rapid eye movement sleep stage 1; N2, duration of non-rapid eye movement sleep stage 2; N3, duration of non-rapid eye movement sleep stage 3. *P < 0.05, **P < 0.01, ***P < 0.001.
Table 2
| Variable | B | SEM | β | t | P | R2 |
|---|---|---|---|---|---|---|
| SE | 1.85 | 0.98 | 0.42 | 2.10 | 0.048* | 0.17 |
| WASO | -11.34 | 3,61 | -0.56 | -3.13 | 0.005** | 0.32 |
| W | -10.35 | 4.02 | -0.49 | -2.56 | 0.018* | 0.24 |
| N2 | 9.16 | 3.67 | 0.48 | 2.50 | 0.021* | 0.23 |
Multiple linear regression analysis of average daily steps and sleep parameters by PSG.
SE, sleep efficiency; WASO, wake time after sleep onset; W, wake stage; N2, duration of non-rapid eye movement sleep stage 2. *P < 0.05, **P < 0.01.
Correlations between average daily steps and EEG spectral power
Spearman correlation analysis revealed positive associations between average daily steps and the mean power of theta (r = 0.56, P = 0.006), slow sigma (r = 0.54, P = 0.007), and fast sigma (r = 0.43, P = 0.039) activity during NREM sleep, and the mean power of theta (r = 0.49, P = 0.019) during REM sleep (Tables 3, 4). These findings suggest that increased average daily steps may contribute to enhanced sleep continuity, deeper sleep, and improved memory function.
Table 3
| SO | SWA | Delta | Theta | Slow sigma | Fast sigma | Beta | |
|---|---|---|---|---|---|---|---|
| r | 0.09 | 0.18 | 0.19# | 0.56 | 0.54 | 0.43 | 0.12 |
| P | 0.673 | 0.412 | 0.380 | 0.006** | 0.007** | 0.039* | 0.590 |
The correlation between the average daily steps and EEG spectral power in NREM.
#Represents Pearson correlation, and the rest are Spearman correlation. SO, slow oscillations; SWA, slow-wave activity. *P < 0.05, **P < 0.01
Table 4
| SO | SWA | Delta | Theta | Slow sigma | Fast sigma | Beta | |
|---|---|---|---|---|---|---|---|
| r | 0.08# | 0.35 | 0.29 | 0.49 | 0.31 | 0.21 | 0.22 |
| P | 0.715 | 0.101 | 0.173 | 0.019* | 0.157 | 0.335 | 0.312 |
The correlation between the average daily steps and EEG spectral power in REM.
#Represents Pearson correlation, and the rest are Spearman correlation. SO, slow oscillations; SWA, slow-wave activity. *P < 0.05.
Correlation between average daily steps and cognitive function
We found that increasing average daily steps were significantly associated with reduced RT in the PVT task (r = -0.47, P = 0.025) (Figures 3A, B). Additionally, the data demonstrated a positive relationship between average daily steps and blood BDNF levels (r = 0.50, P = 0.016) (Figure 3C). Our findings demonstrate that enhanced PA levels are associated with both improved sleep quality and attenuated next-day cognitive impairment, potentially mediated through upregulated BDNF production.
Figure 3

(A, B) The correlation between the average daily steps and the number of lapses (A) and RT (B) in the PVT. (C) The correlation between the average daily steps and serum BDNF level. RT, reaction time; PVT, Psychomotor vigilance task.
Discussion
Our study systematically investigated the relationship between habitual PA and multidimensional sleep architecture among college students, incorporating both subjective and objective sleep assessments, alongside relevant physiological biomarkers and neurophysiological measurements (EEG power). Our results demonstrate that even moderate increments in average daily steps are associated with improvements in objective sleep parameters, particularly nocturnal SE and N2 stage duration, as well as reductions in wake stage and WASO. In parallel, greater PA predicted higher cognitive function and BDNF levels, and enhanced sleep EEG spectral features-findings that jointly support the hypothesis that PA may promote sleep-driven restoration and next-day functional capacity through both biological and neurophysiological pathways.
Sleep is a fundamental biological process, essential for cognitive health, emotional regulation, and overall somatic restoration (27, 28). The growing prevalence of insufficient or poor sleep quality-especially among young adults frequently engaged in sedentary or irregular lifestyles-underscores the need for easily implementable, non-pharmacological interventions (29). PA is widely recognized as one of the most robust, accessible health behaviors for promoting better sleep. Previous studies have highlighted that interventional exercise programs-including yoga, tai chi, and aerobic exercise-can ameliorate insomnia symptoms and improve both subjective and objective sleep quality by shortening SOL, enhancing sleep continuity, increasing TST, and minimizing nocturnal awakenings (30–32).
Differentiating itself from traditional interventional studies, our research systematically analyzed the real-world habitual PA-sleep architecture relationship. By leveraging continuous, objective ACT over a 7-day window paired with gold-standard PSG, we found that, among college students, higher average daily steps not only improved total nocturnal SE but also significantly increased N2 stage while shrinking WASO and wake stage. Multivariate regression further supported that daily steps positively predicted nocturnal SE and N2 time, and negatively predicted wake time. These findings corroborate and extend earlier evidence from intervention studies to more ecologically valid, everyday context among young adults. Given that N2 sleep comprises 50-60% of total sleep time across the night and serves as a crucial transitional stage from light (N1) to deeper sleep, its promotion is particularly relevant for the consolidation of cognitive and emotional functions (33–35). In our study, increased habitual PA was robustly correlated with greater N2 duration, underscoring the value of even modest increases in routine activity for optimizing restorative sleep microstructure. It is well-established that poor sleep often contributes to diminished daytime function, including impaired mood, cognitive processing, memory, and psychomotor vigilance (36). Conversely, improvements in nocturnal sleep quality can generate notable benefits for next-day performance. Our findings-demonstrating that more daily steps are linked to not only better sleep at night but also improved next-day psychomotor vigilance-strengthen this bidirectional framework (37). Research has proposed that the neurorestorative and memory-consolidating aspects of sleep depend critically on the quality and continuity of specific sleep stages, with N2 being prominent among them. Furthermore, our correlational analysis revealed that increased PA was associated with amplified power in EEG theta and sigma bands during NREM and REM sleep; these neuroelectrical patterns are closely tied to sleep stability, memory reactivation, and synaptic remodeling (38–40). Thus, daily PA, by promoting more coherent and restorative nighttime sleep, may help set the stage for better daytime alertness, learning, and productivity.
Despite the consistent association between increased PA and improved sleep, the precise underlying mechanisms remain multifactorial and incompletely understood. PA increases cerebral metabolic demand, resulting in the accumulation of sleep-promoting substances (notably adenosine) in the brain. Elevated adenosine facilitates sleep initiation and depth-its levels rise after exercise and decrease following recovery sleep, indicating its pivotal role in homeostatic sleep pressure (41). Exercise-induced elevations in core body temperature are followed by physiological cooling at sleep onset, which has been shown to promote deeper slow-wave sleep (42). Concurrently, habitual PA fosters parasympathetic dominance during sleep, particularly prominent in the second NREM cycle, reducing heart rate and supporting nocturnal autonomic stability (33). Research demonstrates that regular PA significantly elevates circulating BDNF levels, a key neurotrophin regulating neurogenesis, synaptic plasticity, and neural resilience (43). Mechanistically, exercise-induced lactate surges activate downstream CREB transcription factors through upregulated intracellular Ca2+ concentrations, thereby promoting BDNF biosynthesis; concurrently, enhanced neuroelectrical activity in prefrontal-hippocampal networks stimulates BDNF secretion via TrkB receptor-mediated PI3K/Akt and MAPK/ERK signaling phosphorylation cascades (44, 45). Our study’s observation that higher daily steps correlate with elevated serum BDNF supports the argument that PA-driven trophic support at the neurochemical level may underlie some of the sleep and cognitive benefits observed. Enhanced BDNF has been directly linked with better learning and cognitive performance, likely by supporting hippocampal circuitry engaged during sleep-dependent memory consolidation (46). The association we report between greater PA and enhanced NREM theta/sigma and REM theta power is notable. The sigma frequency band during NREM sleep corresponds to sleep spindles-hallmark electrophysiological features of NREM sleep that mediate memory consolidation and sleep stability maintenance (47, 48). Long-term exercise promotes increased spindle density and incidence, manifesting as elevated sigma power spectral density; concurrently, theta oscillations during REM sleep facilitate emotional memory processing and cognitive integration (49). Exercise-induced augmentation of REM sleep duration and intensity further elevates theta band power, potentially through enhanced neurometabolic activity involving lactate-mediated signaling cascades (50, 51). Thus, our data suggest that PA may serve as a modulator of brain activity patterns that both reflect and reinforce sleep’s cognitive and restorative functions. Bringing these lines of evidence together, we propose a plausible mechanistic model whereby increased PA during the day triggers favorable metabolic, neurotrophic, and neurophysiological changes, which in turn bolster sleep quality. Improved sleep, most notably via N2 and stabilized sleep microstructure, then enables better daytime cognitive and emotional functioning, potentially closing a virtuous feedback loop between waking activity, sleep, and neurobehavioral health.
An important yet frequently underappreciated issue highlighted by our study is the potential discrepancy between subjective and objective evaluations of sleep. In our study, sleep diaries did not consistently align with ACT or PSG findings (particularly regarding SOL and fragmentation), echoing concerns raised in previous literature (52). The discrepancy between subjective and objective sleep measures may arise from altered glucose metabolism in sensory processing and consciousness-related cortical regions, or from pathological high-frequency beta wave activity during NREM sleep inducing hallucinations of wakefulness (53). Furthermore, this psychophysiological dissociation often signals underlying psychological comorbidities, including anxiety, depression, and neuroticism, necessitating individualized clinical profiling based on patient-specific traits (54). Advancing the understanding of these mechanisms requires refined electrophysiological assessment methodologies, such as high-density EEG or combined neuroimaging approaches. Such discrepancies underscore the limitations of self-reported measures, which may be influenced by bias, inaccurate recall, or sleep misperception, and highlight the necessity for multimodal, objective data collection in both clinical and research contexts (55, 56). While our study offers novel, multi-layered insights into the link between PA, sleep quality, and neurobiological processes in a real-world cohort of young adults, certain limitations merit consideration. First, the study’s limited sample size and homogeneity in participant age and background constrain the generalizability of findings. Future investigations should incorporate middle-aged and elderly cohorts to comprehensively evaluate the effects of PA on sleep architecture and cognitive function. Second, we did not stratify types or intensities of PA beyond steps, nor did we capture long-term activity patterns or specific exercise regimens; future work should explore whether different modes or durations of PA differentially impact sleep architecture and next-day functioning. Third, the design was observational and correlative; longitudinal and interventional approaches will be essential to establish causality and to better characterize the temporal sequence of PA, sleep improvement, and daytime benefit. Fourth, while we identified BDNF and EEG power as putative mediators, direct experimental manipulations or mechanistic animal studies are needed to more definitively map the underlying pathways.
Nevertheless, our results suggest that even moderate increases in daily activity-readily achievable by most college students through lifestyle modifications or minimal behavioral interventions-can yield salutary effects on sleep and subsequent daytime cognitive function. In populations at risk for sleep complaints but not meeting diagnostic thresholds for insomnia disorder, PA emerges as an attractive, low-risk preventive and therapeutic strategy (57). Our findings indicate that increasing habitual PA, even at moderate levels, is associated with meaningful enhancements in nocturnal sleep quality among college students. Improvements are especially prominent in SE and N2 duration, accompanied by modulation of BDNF and EEG spectral markers, and better next-day psychomotor vigilance. These results provide experimental and theoretical foundation for incorporating PA as a practical, non-pharmacological adjunct in the management of poor sleep and its daytime consequences. Moreover, these research results may suggest that BDNF and the optimization of sleep neurophysiology may serve as key mechanistic bridges linking daytime activity, sleep, cognitive function, and next-day function (58, 59). Future research should expand sample diversity and size, dissect PA modalities and intensities, employ randomized controlled trials designs and integrate multi-omics and neuroimaging approaches to fully unravel the causal chains and molecular substrates involved. Ultimately, these efforts will not only inform the prevention and treatment of sleep problems in vulnerable populations, but also advance our understanding of the dynamic, bi-directional interplay between active waking, restorative sleep, and optimal daytime functioning.
Statements
Data availability statement
The original contributions presented in the study are included in the article/supplementary material. Further inquiries can be directed to the corresponding authors.
Ethics statement
The studies involving humans were approved by Peking University Sixth Hospital's Institutional Review Board. The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided written informed consent to participate in this study.
Author contributions
YJ: Data curation, Writing – original draft, Methodology, Writing – review & editing, Project administration, Formal Analysis. BW: Software, Methodology, Writing – review & editing. XS: Project administration, Validation, Supervision, Writing – review & editing, Conceptualization, Visualization. ZW: Conceptualization, Supervision, Funding acquisition, Writing – review & editing, Writing – original draft. WC: Funding acquisition, Writing – review & editing, Project administration, Conceptualization.
Funding
The author(s) declare that financial support was received for the research and/or publication of this article. This work was supported by National Natural Science Foundation of China (no. 81901347), China Postdoctoral Science Foundation (no. 2024M750133), Postdoctoral Fellowship Program of China Postdoctoral Science Foundation (no. GZC20240080), Zhejiang Provincial Natural Science Foundation of China under Grant (no. ZCLY24H0901), National Key Research and Development Program of China (no. 2021YFC2501400), Pioneer R&D Program of Zhejiang (no. 2025C01119).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be constructed as a potential conflict of interest.
The reviewer YZ declared a shared parent affiliation with the authors YJ and XS to the handling editor at the time of review.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1
Krause AJ Simon EB Mander BA Greer SM Saletin JM Goldstein-Piekarski AN et al . The sleep-deprived human brain. Nat Rev Neurosci. (2017) 18:404–18. doi: 10.1038/nrn.2017.55
2
Besedovsky L Lange T Haack M . The sleep-immune crosstalk in health and disease. Physiol Rev. (2019) 99:1325–80. doi: 10.1152/physrev.00010.2018
3
Centers for Disease Control and Prevention . QuickStats: percentage of adults who reported an average of ≤6 hours of sleep per 24-hour period, by sex and age group—United states, 1985 and 2004. JAMA. (2005) 294:2692. doi: 10.1001/jama.294.21.2692
4
Zheng YB Huang YT Gong YM Li MZ Zeng N Wu SL et al . Association of lifestyle with sleep health in general population in China: a cross-sectional study. Transl Psychiatry. (2024) 14:320. doi: 10.1038/s41398-024-03002-x
5
Hills AP Dengel DR Lubans DR . Supporting public health priorities: recommendations for physical education and physical activity promotion in schools. Prog Cardiovasc Dis. (2015) 57:368–74. doi: 10.1016/j.pcad.2014.09.010
6
Sejbuk M Mirończuk-Chodakowska I Witkowska AM . Sleep quality: A narrative review on nutrition, stimulants, and physical activity as important factors. Nutrients. (2022) 14. doi: 10.3390/nu14091912
7
Hartescu I Morgan K Stevinson CD . Increased physical activity improves sleep and mood outcomes in inactive people with insomnia: a randomized controlled trial. J Sleep Res. (2015) 24:526–34. doi: 10.1111/jsr.12297
8
Banach M Lewek J Surma S Penson PE Sahebkar A Martin SS et al . The association between daily step count and all-cause and cardiovascular mortality: a meta-analysis. Eur J Prev Cardiol. (2023) 30:1975–85. doi: 10.1093/eurjpc/zwad229
9
Del Pozo Cruz B Ahmadi MN Lee IM Stamatakis E . Prospective associations of daily step counts and intensity with cancer and cardiovascular disease incidence and mortality and all-cause mortality. JAMA Internal Med. (2022) 182:1139–48. doi: 10.1001/jamainternmed.2022.4000
10
Ekelund U Sanchez-Lastra MA Dalene KE Tarp J . Dose-response associations, physical activity intensity and mortality risk: A narrative review. J Sport Health Sci. (2024) 13:24–9. doi: 10.1016/j.jshs.2023.09.006
11
Bizzozero-Peroni B Díaz-Goñi V Jiménez-López E Rodríguez-Gutiérrez E Sequí-Domínguez I Núñez de Arenas-Arroyo S et al . Daily step count and depression in adults: A systematic review and meta-analysis. JAMA Netw Open. (2024) 7:e2451208. doi: 10.1001/jamanetworkopen.2024.51208
12
Martin JL Hakim AD . Wrist actigraphy. Chest. (2011) 139:1514–27. doi: 10.1378/chest.10-1872
13
Stutz J Eiholzer R Spengler CM . Effects of evening exercise on sleep in healthy participants: A systematic review and meta-analysis. Sports Med. (2019) 49:269–87. doi: 10.1007/s40279-018-1015-0
14
Chellappa SL Aeschbach D . Sleep and anxiety: From mechanisms to interventions. Sleep Med Rev. (2022) 61:101583. doi: 10.1016/j.smrv.2021.101583
15
Rahmani M Rahmani F Rezaei N . The brain-derived neurotrophic factor: missing link between sleep deprivation, insomnia, and depression. Neurochem Res. (2020) 45:221–31. doi: 10.1007/s11064-019-02914-1
16
Harder JA Fichorova RN Srivastava A Wiley A Burdick KE Locascio JJ et al . Brain-derived neurotrophic factor and mood in perimenopausal depression. J Affect Disord. (2022) 300:145–9. doi: 10.1016/j.jad.2021.12.092
17
Farrukh S Habib S Rafaqat A Sarfraz A Sarfraz Z Tariq H . Association of exercise, brain-derived neurotrophic factor, and cognition among older women: A systematic review and meta-analysis. Arch Gerontol Geriatr. (2023) 114:105068. doi: 10.1016/j.archger.2023.105068
18
Hamilton M . A rating scale for depression. J Neurol Neurosurg Psychiatry. (1960) 23:56–62. doi: 10.1136/jnnp.23.1.56
19
Hamilton M . The assessment of anxiety states by rating. Br J Med Psychol. (1959) 32:50–5. doi: 10.1111/j.2044-8341.1959.tb00467.x
20
Imboden MT Nelson MB Kaminsky LA Montoye AH . Comparison of four Fitbit and Jawbone activity monitors with a research-grade ActiGraph accelerometer for estimating physical activity and energy expenditure. Br J Sports Med. (2018) 52:844–50. doi: 10.1136/bjsports-2016-096990
21
Master L Nye RT Lee S Nahmod NG Mariani S Hale L et al . Bidirectional, daily temporal associations between sleep and physical activity in adolescents. Sci Rep. (2019) 9:7732. doi: 10.1038/s41598-019-44059-9
22
Marinac CR Quante M Mariani S Weng J Redline S Cespedes Feliciano EM et al . Associations between timing of meals, physical activity, light exposure, and sleep with body mass index in free-living adults. J Phys Act Health. (2019) 16:214–21. doi: 10.1123/jpah.2017-0389
23
Shi Y Nie Y Hao F Feng X Zhang Y Sanford LD et al . EEG spectral analysis of nighttime sleep and daytime MSLTs and neurocognitive evaluations in subjects with co-morbid insomnia and OSA. Respir Res. (2025) 26:139. doi: 10.1186/s12931-025-03193-x
24
Tabarak S Zhu X Li P Weber FD Shi L Gong Y et al . Temporal dynamics of negative emotional memory reprocessing during sleep. Transl Psychiatry. (2024) 14:434. doi: 10.1038/s41398-024-03146-w
25
Dong Y Cheng Y Wang J Ren Z Lu Y Yuan K et al . Abnormal power and spindle wave activity during sleep in young smokers. Front Neurosci. (2025) 19:1534758. doi: 10.3389/fnins.2025.1534758
26
Shi Y Tian X Li T Hu Y Xie Y Li H et al . The influence of transcranial alternating current stimulation on EEG spectral power during subsequent sleep: A randomized crossover study. Sleep Med. (2025) 126:185–93. doi: 10.1016/j.sleep.2024.12.011
27
Lotti S Moretton M Bulgari M Costantini L Dall’Asta M De Amicis R et al . Association between shift work and eating behaviours, sleep quality, and mental health among Italian workers. Eur J Nutr. (2025) 64:97. doi: 10.1007/s00394-025-03600-5
28
Yang YB Zheng YB Sun J Yang LL Li J Gong YM et al . To nap or not? Evidence from a meta-analysis of cohort studies of habitual daytime napping and health outcomes. Sleep Med Rev. (2024) 78:101989. doi: 10.1016/j.smrv.2024.101989
29
Sewell KR Erickson KI Rainey-Smith SR Peiffer JJ Sohrabi HR Brown BM . Relationships between physical activity, sleep and cognitive function: A narrative review. Neurosci Biobehav Rev. (2021) 130:369–78. doi: 10.1016/j.neubiorev.2021.09.003
30
Lowe H Haddock G Mulligan LD Gregg L Fuzellier-Hart A Carter LA et al . Does exercise improve sleep for adults with insomnia? A systematic review with quality appraisal. Clin Psychol Rev. (2019) 68:1–12. doi: 10.1016/j.cpr.2018.11.002
31
McKenna S Tierney M O’Neill A Fraser A Kennedy N . Sleep and physical activity: a cross-sectional objective profile of people with rheumatoid arthritis. Rheumatol Int. (2018) 38:845–53. doi: 10.1007/s00296-018-4009-1
32
Philbrook LE El-Sheikh M . Associations between neighborhood context, physical activity, and sleep in adolescents. Sleep Health. (2016) 2:205–10. doi: 10.1016/j.sleh.2016.05.008
33
Sandercock GR Bromley PD Brodie DA . Effects of exercise on heart rate variability: inferences from meta-analysis. Med Sci Sports Exerc. (2005) 37:433–9. doi: 10.1249/01.MSS.0000155388.39002.9D
34
Wei Y Colombo MA Ramautar JR Blanken TF van der Werf YD Spiegelhalder K et al . Sleep stage transition dynamics reveal specific stage 2 vulnerability in insomnia. Sleep. (2017) 40. doi: 10.1093/sleep/zsx117
35
Rosenberg RS Van Hout S . The American Academy of Sleep Medicine inter-scorer reliability program: sleep stage scoring. J Clin Sleep Med. (2013) 9:81–7. doi: 10.5664/jcsm.2350
36
Gardani M Bradford DRR Russell K Allan S Beattie L Ellis JG et al . A systematic review and meta-analysis of poor sleep, insomnia symptoms and stress in undergraduate students. Sleep Med Rev. (2022) 61:101565. doi: 10.1016/j.smrv.2021.101565
37
Matsangas P Shattuck NL . Sleep quality, occupational factors, and psychomotor vigilance performance in the U.S. Navy sailors. Sleep. (2020) 43. doi: 10.1093/sleep/zsaa118
38
Buchmann A Ringli M Kurth S Schaerer M Geiger A Jenni OG et al . EEG sleep slow-wave activity as a mirror of cortical maturation. Cereb Cortex. (2011) 21:607–15. doi: 10.1093/cercor/bhq129
39
Kurth S Ringli M Lebourgeois MK Geiger A Buchmann A Jenni OG et al . Mapping the electrophysiological marker of sleep depth reveals skill maturation in children and adolescents. NeuroImage. (2012) 63:959–65. doi: 10.1016/j.neuroimage.2012.03.053
40
Sun L Zhou H Cichon J Yang G . Experience and sleep-dependent synaptic plasticity: from structure to activity. Philos Trans R Soc London Ser B Biol Sci. (2020) 375:20190234. doi: 10.1098/rstb.2019.0234
41
Dworak M Diel P Voss S Hollmann W Struder HK . Intense exercise increases adenosine concentrations in rat brain: implications for a homeostatic sleep drive. Neuroscience. (2007) 150:789–95. doi: 10.1016/j.neuroscience.2007.09.062
42
Mcginty D Szymusiak R . Keeping cool - a hypothesis about the mechanisms and functions of slow-wave sleep. Trends Neurosci. (1990) 13:480–7. doi: 10.1016/0166-2236(90)90081-K
43
Schmitt K Holsboer-Trachsler E Eckert A . BDNF in sleep, insomnia, and sleep deprivation. Ann Med. (2016) 48:42–51. doi: 10.3109/07853890.2015.1131327
44
Reycraft JT Islam H Townsend LK Hayward GC Hazell TJ Macpherson REK . Exercise intensity and recovery on circulating brain-derived neurotrophic factor. Med Sci Sports Exerc. (2020) 52:1210–7. doi: 10.1249/MSS.0000000000002242
45
Ceylan HI Silva AF Ramirez-Campillo R Murawska-Cialowicz E . Exploring the effect of acute and regular physical exercise on circulating brain-derived neurotrophic factor levels in individuals with obesity: A comprehensive systematic review and meta-analysis. Biol (Basel). (2024) 13. doi: 10.3390/biology13050323
46
Riboldi JG Correa J Renfijes MM Tintorelli R Viola H . Arc and BDNF mediated effects of hippocampal astrocytic glutamate uptake blockade on spatial memory stages. Commun Biol. (2024) 7:1032. doi: 10.1038/s42003-024-06586-8
47
Schönauer M Pöhlchen D . Sleep spindles. Curr Biol: CB. (2018) 28:R1129–r30. doi: 10.1016/j.cub.2018.07.035
48
Maier JG Kuhn M Mainberger F Nachtsheim K Guo S Bucsenez U et al . Sleep orchestrates indices of local plasticity and global network stability in the human cortex. Sleep. (2019) 42. doi: 10.1093/sleep/zsy263
49
Pronier E Morici JF Girardeau G . The role of the hippocampus in the consolidation of emotional memories during sleep. Trends Neurosci. (2023) 46:912–25. doi: 10.1016/j.tins.2023.08.003
50
Daume J Gruber T Engel AK Friese U . Phase-amplitude coupling and long-range phase synchronization reveal frontotemporal interactions during visual working memory. J Neurosci. (2017) 37:313–22. doi: 10.1523/JNEUROSCI.2130-16.2016
51
Daume J Graetz S Gruber T Engel AK Friese U . Cognitive control during audiovisual working memory engages frontotemporal theta-band interactions. Sci Rep. (2017) 7:12585. doi: 10.1038/s41598-017-12511-3
52
Liebich T Lack L Hansen K Zajamšek B Lovato N Catcheside P et al . A systematic review and meta-analysis of wind turbine noise effects on sleep using validated objective and subjective sleep assessments. J Sleep Res. (2021) 30:e13228. doi: 10.1111/jsr.13228
53
Rezaie L Fobian AD McCall WV Khazaie H . Paradoxical insomnia and subjective-objective sleep discrepancy: A review. Sleep Med Rev. (2018) 40:196–202. doi: 10.1016/j.smrv.2018.01.002
54
Herzog R Crosbie F Aloulou A Hanif U Chennaoui M Leger D et al . A continuous approach to explain insomnia and subjective-objective sleep discrepancy. Commun Biol. (2025) 8:423. doi: 10.1038/s42003-025-07794-6
55
McCarter SJ Hagen PT St Louis EK Rieck TM Haider CR Holmes DR et al . Physiological markers of sleep quality: A scoping review. Sleep Med Rev. (2022) 64:101657. doi: 10.1016/j.smrv.2022.101657
56
Sun J Lu T Shao X Han Y Xia Y Zheng Y et al . Practical AI application in psychiatry: historical review and future directions. Mol Psychiatry. (2025) 30:4399–408. doi: 10.1038/s41380-025-03072-3
57
Peluso MA Guerra de Andrade LH . Physical activity and mental health: the association between exercise and mood. Clinics (Sao Paulo). (2005) 60:61–70. doi: 10.1590/S1807-59322005000100012
58
Villamil-Parra W Moscoso-Loaiza L . Effects of physical exercise on Irisin and BDNF concentrations, and their relationship with cardiometabolic and mental health of individuals with Metabolic Syndrome: A Systematic Review. Exp Gerontol. (2024) 198:112640. doi: 10.1016/j.exger.2024.112640
59
Lim DC Najafi A Afifi L Bassetti C Buysse DJ Han F et al . The need to promote sleep health in public health agendas across the globe. Lancet Public Health. (2023) 8:e820–e6. doi: 10.1016/S2468-2667(23)00182-2
Summary
Keywords
physical activity, sleep architecture, spectral analysis, cognitive function, BDNF
Citation
Ji Y, Wang B, Sun X, Wang Z and Chen W (2025) The impact of physical activity on sleep architecture and cognitive function among college students. Front. Psychiatry 16:1656278. doi: 10.3389/fpsyt.2025.1656278
Received
29 June 2025
Accepted
07 August 2025
Published
29 August 2025
Volume
16 - 2025
Edited by
Jianyu Que, Xiamen Xianyue Hospital, China
Reviewed by
Sizhi Ai, The First Affiliated Hospital of Xinxiang Medical University, China
Yong-Bo Zheng, Shandong University, China
Updates
Copyright
© 2025 Ji, Wang, Sun, Wang and Chen.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Xiulian Sun, xiulians@gmail.com; Zhong Wang, wangzhong@bjmu.edu.cn; Wenhao Chen, chenwenhao@zju.edu.cn
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.