- 1Department of Dermatology, Beijing Changping Hospital of Integrated Chinese and Western Medicine, Beijing, China
- 2Key Laboratory of Exercise and Physical Fitness, Beijing Sport University, Ministry of Education, Beijing, China
- 3Department of Physical Fitness and Health, School of Sport Science, Beijing Sport University, Beijing, China
Background: Androgenetic alopecia (AGA) is a prevalent chronic condition with health burden, yet its association with psychological well-being remains inconsistent. This study systematically examines differences in psychological well-being between AGA patients and non-AGA controls, focusing on symptoms of anxiety, symptoms of depression, stress, and other psychological issues.
Methods: Following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines, a systematic search was conducted across 7 databases (CNKI, Wanfang, PubMed, Web of Science, Cochrane, EMBASE, EBSCO series) from their inception to June 30, 2025. A total of 13 studies were included, comprising 2,737 AGA patients and 17,382 controls. Study quality was assessed using the Agency for Healthcare Research and Quality (AHRQ) tool for cross-sectional studies and Newcastle-Ottawa Scale (NOS) for case-control studies. Effect sizes were pooled using a random-effects model, supplemented by sensitivity analysis and publication bias assessment to ensure the robustness of the results.
Results: Compared to non-AGA controls, AGA patients demonstrated significantly higher levels of generalized anxiety symptoms (pooled SMD = -0.50, 95% CI [-0.99, 0.00], p = 0.05), social anxiety symptoms (pooled SMD = -0.50, 95% CI [-0.84,-0.16], p = 0.004), depression symptoms (pooled SMD = -0.38, 95% CI [-0.65,-0.12], p = 0.004), and perceived stress (pooled SMD = -1.09, 95% CI [-1.43,-0.74], p < 0.001). However, no significant difference was observed in general distress (pooled SMD = -0.01, 95% CI [-0.39,0.37], p = 0.97). Qualitatively, AGA patients also exhibited reduced self-esteem, life satisfaction, and emotional intelligence dimensions, along with greater body image dissatisfaction, somatization, interpersonal sensitivity, and psychoticism.
Conclusion: AGA patients experience increased levels of symptoms of anxiety, symptoms of depression, stress, body image dissatisfaction, somatization, interpersonal sensitivity, and psychoticism, as well as decreased self-esteem, life satisfaction, emotional intelligence, self-actualization, problem-solving ability, optimism, and happiness. In the future, clinical treatment should focus on patient psychological well-being through assessments and timely interventions, and further research should be conducted to provide precise clinical guidance.
Systematic Review Registration: [https://www.crd.york.ac.uk/PROSPERO/], identifier [CRD420250655604].
1 Introduction
Hair, a vital epidermal appendage of the head and face, performs essential physiological functions such as scalp protection, sensory perception, and sebum regulation. Additionally, its biophysical and biochemical properties serve as health indicators for disease screening (1, 2). As a core component of body image, it also influences psychological well-being (3, 4), a multidimensional concept that, in the context of our study, includes dimensions that encompass both the absence of psychological illness or distress and positive states and functions. Among hair disorders, androgenetic alopecia (AGA) is clinically significant due to its high prevalence and substantial health burden. It is a non-scarring hair loss that usually begins in puberty, characterized by gradual hair loss and miniaturization of hair follicles. In men, it shows as a receding frontal hairline and/or thinning hair on the top of the head. In women, it shows as thinning hair on the top of the head, but the hairline usually remains unchanged (5). Epidemiological studies show that the incidence rate of AGA is higher in men than in women at 24.88% and 5.06% respectively (6). Specifically, the prevalence is about 20% in Chinese men and ranges from 22.6% to 65% in African populations. Moreover, the incidence rate increases significantly with age—reaching approximately 50% by age 50 and 80% by age 80 in European men, increasing to 29% to 42% in women aged 70 years and over (5, 7, 8). Pathologically, AGA arises from genetic predisposition, dysregulated androgen metabolism, growth factor imbalances, and follicular microenvironment disruption—a process that drives progressive hair miniaturization while being associated with several systemic diseases such as cardiovascular disease and metabolic syndrome (8). Psychologically, AGA also profoundly impacts psychological health, frequently leading to diminished self-esteem, anxiety, and depression, which significantly compromise quality of life and social functioning (4).
Research on the psychological impact of AGA on patients dates back to the 1990s. Early studies revealed that, compared with non-balding individuals and those with low hair loss, patients with advanced hair loss experienced significantly greater stress related to socioemotional functioning, self-esteem, positive life events, and coping behaviors (4). Subsequent studies have shown that AGA patients may experience psychological problems such as anxiety, helplessness, decreased self-esteem, and even depression due to their dissatisfaction with their appearance and concerns about negative evaluations or social rejection from others (3, 4, 9). For instance, a 2023 study involving 1,230 male patients diagnosed with AGA found that 95% experienced stress and 78% felt embarrassed by their hair loss (10). Lin et al. investigated 530 employees at a medical institution and found that, compared with individuals without hair loss, AGA patients exhibited higher levels of somatization, depression, and hostility (11). Although previous studies have explored the relationship between AGA and various dimensions of psychological well-being, the findings remain a matter of debate. Regarding depression specifically, Noha et al., employing the Beck Depression Inventory, found that women with AGA had higher depression levels than healthy female volunteers of the same age (12). Conversely, He et al., using the Patient Health Questionnaire, observed no significant difference in depression levels between students with AGA and those without AGA among the freshmen of the two universities (13). Sinikumpu et al. also found that hair loss severity was not significantly associated with depression levels among 46-year-old men in the Northern Finland 1966 Birth Cohort, as assessed by the Beck Depression Inventory (14). Additionally, a systematic review and meta-analysis concluded that the link between AGA and depression was not statistically significant (3). These inconsistent results highlight the need for further research.
In addition to the contradictory conclusions drawn from prior research across diverse regions and populations, current research on the psychological well-being of the AGA group still has several limitations, mainly in the following aspects: (a) existing studies on AGA mostly focus on physiology and drug treatments, neglecting psychological well-being factors or only considering them as part of quality of life studies; (b) previous studies often examine a single psychological well-being issue in AGA populations, lacking comprehensive comparisons with non-AGA individuals; (c) past studies have limited sample sizes, and the research results are inconsistent, failing to provide sufficient research evidence; (d) previous systematic reviews lack Chinese data, and focus on a limited range of dimensions. For example, the study by Huang et al. (9) primarily utilized data from Western populations, with insufficient representation from China and other Asian countries, and lacked a non-AGA control group for comparison. The research by Frith et al. (3) focused exclusively on males and conducted quantitative analysis solely on depression and quality of life within the patient population, which limited the scope of their analysis. The review by Aukerman et al. (4) was qualitative in nature and did not include quantitative analysis. Therefore, this study compares the psychological well-being of AGA and non-AGA populations by including a broader range of studies from multiple databases, integrating data from multiple scales, incorporating data from China, and examining both male and female patients, thereby providing a theoretical basis for the prevention and intervention of AGA.
2 Materials and methods
This study adhered strictly to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. The registration number of this meta-analysis on PROSPERO is CRD420250655604 (Available from https://www.crd.york.ac.uk/PROSPERO/view/CRD420250655604).
2.1 Literature search
The electronic databases CNKI, Wanfang, PubMed, Web of Science, Cochrane, EMBASE, and EBSCO series were searched to find all literature related to AGA and psychological well-being. Search for the following keywords and their extensions using the corresponding search formula: “androgenetic alopecia/seborrheic alopecia (in Chinese)/pattern alopecia/hair loss”, “mental disorders/psychology/psychological/emotion/psyche/quality of life/anxiety/depression/stress/self-concept/self-esteem/risk factors” and “questionnaire/scale/inventory/index”. The search period was from the establishment of the databases to June 30, 2025. The language was limited to Chinese and English. Specific search formulas can be viewed in Supplementary Material.
2.2 Inclusion and exclusion criteria
Inclusion criteria: Studies were included if they (a) were classified as observational studies (Cross-sectional study, case-control study, cohort study), thereby avoiding the potential influence of interventions on the true association between the variables; (b) involved populations in the AGA group and the non-AGA group diagnosed by professionals; (c) clearly specified the tools for assessing psychological well-being and presented the data from the assessment scales; (d) reported mean and standard deviation results or provided data that could be calculated.
Exclusion criteria: Studies were excluded if they (a) were randomized controlled trial studies; (b) involved subjects with clinically diagnosed mental disorders, such as anxiety disorders and depressive disorders; (c) lacked a non-AGA group or did not report relevant results of the control group population; (d) were not reported in Chinese or English.
In our study, psychological well-being dimensions include: (a) Emotional States: Covers both positive emotions (such as optimism, sense of happiness, and life satisfaction) and negative emotions (such as anxiety, depression, stress, somatization, and obsessive-compulsive symptoms); (b) Self-Awareness and Self-Evaluation: Includes self-esteem, self-actualization, self-regard, and emotional self-awareness; (c) Interpersonal Relationships: Involves good interpersonal relationships, empathy, and social support; (d) Psychological Resilience and Adaptability: Includes the ability to cope with stress, impulse control, reality testing, problem-solving, stress tolerance, and the ability to adapt to environmental changes; (e) Psychological Growth: Involves self-awareness, self-development, and self-actualization; (f) Life Satisfaction: Overall satisfaction with and sense of happiness in life; (g) Psychosocial Functioning: Includes social adaptability and social participation; (h) Cognitive and Behavioral Health: Covers clear thinking, good judgment, focused attention, appropriate behavior, and the absence of significant behavioral problems (such as impulsive behavior and obsessive-compulsive behavior); (i) Overall Psychological Health Assessment: Includes the Global Severity Index (GSI) and assessments using psychological health scales, etc.
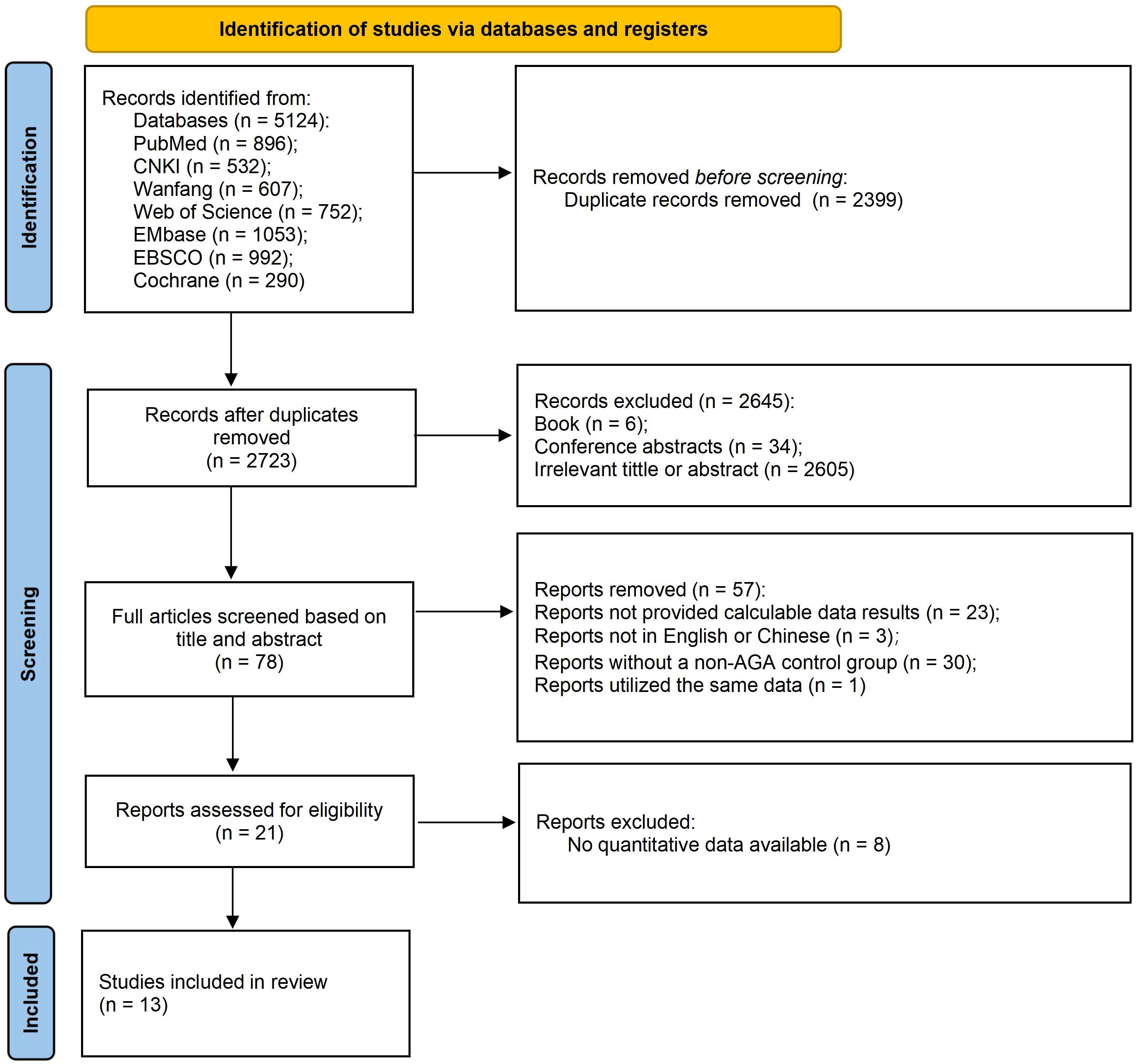
Two researchers independently performed a literature search and screening across 7 databases, using a customized search strategy for each. They screened titles and abstracts based on predefined criteria, excluded ineligible materials like conference abstracts, books and patents, and removed duplicates. The researchers then assessed the full texts of potentially relevant studies against inclusion criteria related to methodology, participant characteristics, and study data. Additionally, they reviewed the reference lists of selected articles and relevant reviews to identify further potential studies. In the final phase, a third researcher reviewed the articles simultaneously. In case of any conflicts, the third researcher made the final decision.
2.3 Data extraction
Data extraction was extracted by one researcher and confirmed by another. Using Excel, information such as the first author, publication year, country, ethnicity, study type, participants resource, sample size, participants’ gender and age, psychological assessment scales, and the reported mean and standard deviation of the scales were extracted. If a single study contains multiple independent AGA or non-AGA groups, each group will be considered as an independent study for analysis. If relevant data for the meta-analysis were missing from the paper, the authors were contacted to obtain the necessary data.
2.4 Quality assessment
The quality of the included studies was assessed using the Agency for Healthcare Research and Quality (AHRQ) tool for cross-sectional studies and the Newcastle-Ottawa Scale (NOS) for case-control or cohort studies. Any discrepancies were resolved by a third author.
The AHRQ consists of 11 items, with options of “Yes”, “No”, or “Unclear”. Items rated as “No” or “Unclear” receive a score of “0,” while items rated as “Yes” receive a score of “1.” The maximum score is 11, categorized into three quality levels: 0-3 (low quality); 4-7 (moderate quality); 8-11 (high quality) (15). The NOS evaluates study quality through three main aspects: Selection, Comparability, and Outcome. The total score is 9, and the score for each aspect can be adjusted according to the specifics of the study. Typically, studies scoring 7–9 are considered high quality, 4–6 as medium quality, and less than 4 as low quality (15). Medium and high quality literature is sufficient to be included in the meta-analysis.
2.5 Statistical analysis
When the number of studies in a particular dimension reached 3 or more, we conducted a meta-analysis on that dimension. Ultimately, 5 dimensions were included in the meta-analysis: symptoms of generalized anxiety (10 studies), symptoms of social anxiety (3 studies), symptoms of depression (8 studies), perceived stress (6 studies), and general distress (5 studies). These dimensions were categorized based on the assessment content and characteristics of the respective scales used in the studies (16).
The standardized mean difference (SMD) is used as an effect size to compare the psychological well-being status between AGA group and non-AGA group. The SMD is calculated as the difference in means between the two groups divided by the pooled standard deviation. When the mean and standard deviation (SD) were not directly reported, the SD was derived from the standard error of the mean (SEM) or 95% confidence intervals (CI) using the formulas specified in the Cochrane Handbook for Systematic Reviews of Interventions [ or (17). Additionally, when scores were presented as medians along with the 25th and 75th percentiles, they were transformed into mean ± SD using the method described by Wan et al. in the Cochrane Handbook (18). Subsequently, the standardized mean difference (d) is converted to Hedges’s g to correct for small sample bias. Following this, the pooled effect is calculated using either a fixed-effect model or a random-effects model, depending on the results of the heterogeneity analysis. Heterogeneity is assessed using I², with a fixed-effect model employed when I² ≤ 50% and a random-effects model when I² > 50%. Additionally, sensitivity analysis is conducted to test the stability of the results, and funnel plots as well as Egger’s and Begg’s tests are used to estimate publication bias, with a significance threshold of p < 0.05. In this process, Review Manager 5.4 is utilized for meta-analysis and heterogeneity assessment, while Stata 17.0 is used for sensitivity analysis and publication bias testing.
3 Results
3.1 Study characteristics and quality assessment
The initial search yielded 5,124 related articles in the database. After excluding 5,111 articles, a total of 13 articles were finally included, comprising 17,382 non-AGA controls and 2,737 AGA patients. 9 studies were conducted in China (13, 19–26), 2 in Egypt (12, 27), 1 in America (28) and 1 in Canada (29). Each study was confirmed based on the clinical criteria for AGA. Of these, 9 studies were diagnosed by specialist doctors in the dermatology or alopecia clinic (12, 13, 20, 23–25, 27–29), and 4 studies were diagnosed as AGA by the researchers based on the AGA diagnosis guidelines (19, 21, 22, 26).
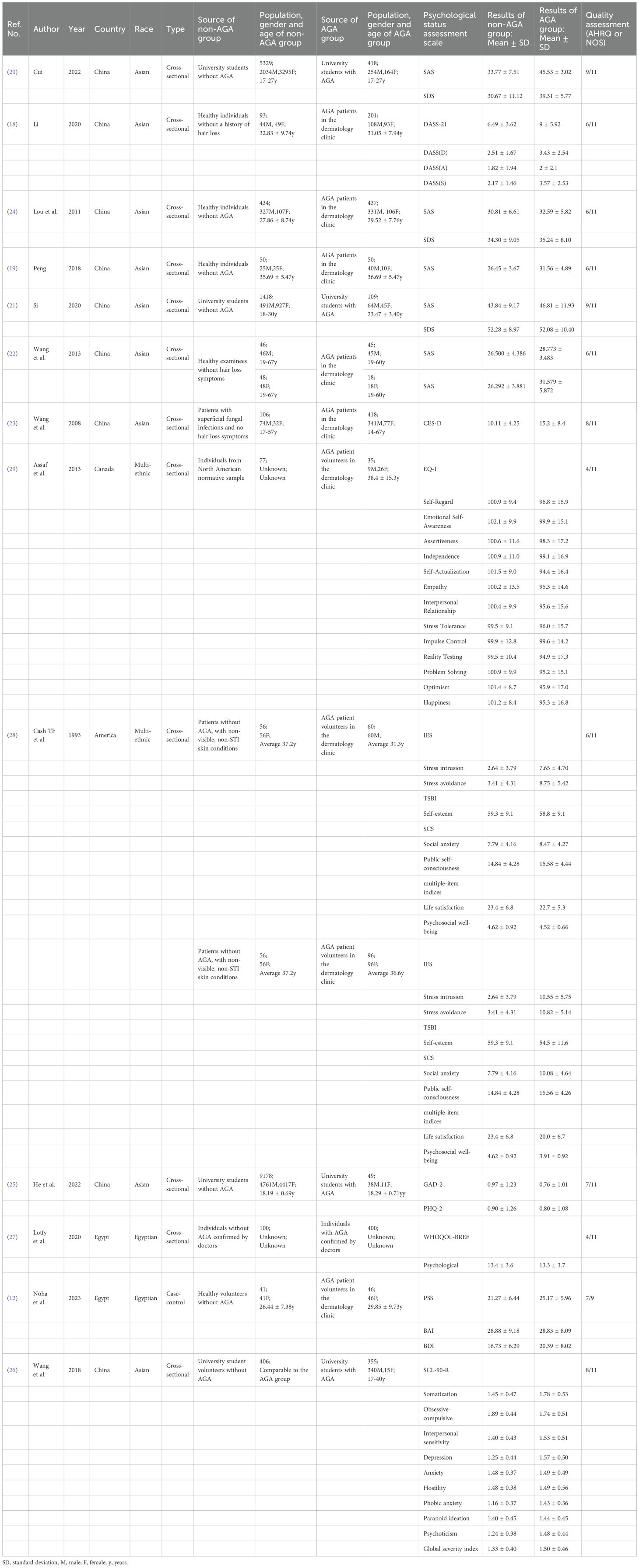
In the included studies, researchers used various validated psychological well-being questionnaires to assess psychological states over different time ranges, focusing on current psychological conditions. For symptoms of anxiety, the Self-Rating Anxiety Scale (SAS) was used in five studies (20–23, 25), while the Beck Anxiety Inventory (BAI) (12) and the 2-item Generalized Anxiety Disorder scale (GAD-2) (13) were also employed. For symptoms of depression, the Self-Rating Depression Scale (SDS) was used in three studies (21, 22, 25), and other tools included the Beck Depression Inventory (BDI) (12), the PHQ-2 (13), and the Center for Epidemiologic Studies Depression scale (CES-D) (24). For stress, the Perceived Stress Scale (PSS) (12) and the Impact of Event Scale (IES) (28) were used.
In multidimensional assessments, tools like the Depression, Anxiety, and Stress Scales (DASS-21) (19), the Revised Symptom Checklist-90 (SCL-90-R) (26), the Emotional Quotient Inventory (EQ-I) (29), and the World Health Organization Quality of Life Brief version (WHOQOL-BREF) (27) were employed to evaluate comprehensive psychological health. Additionally, Cash TF et al. (28) used the Texas Social Behavior Inventory (TSBI), the Self-Consciousness Scale (SCS), and two multi-item indices to assess individuals’ social behavior, self-awareness, and psychological status.
1 case-control study and 12 cross-sectional studies were included. The case-control study received a quality score of 7, while the quality scores of the cross - sectional studies ranged from 4 to 9. All studies were deemed to be of relatively medium quality and thus eligible for inclusion in the meta-analysis. The detailed screening procedure is depicted in Figure 1. Table 1 lists the basic characteristics of the included studies, the observed populations, scale tools, outcome indicators, and quality assessment results.
3.2 Association between AGA and symptoms of anxiety
In the analysis of symptoms of generalized anxiety, a total of 10 studies were included from 9 articles (12, 13, 19–23, 25, 26), involving 17,043 participants in the non-AGA group and 1,728 participants in the AGA group. We employed a random effects model to synthesize the findings from multiple independent studies. The overall effect showed an SMD of -0.50 with a 95% CI of [-0.99, -0.00], indicating that patients with AGA have significantly higher scores on anxiety symptoms compared to those without AGA (Figure 2).
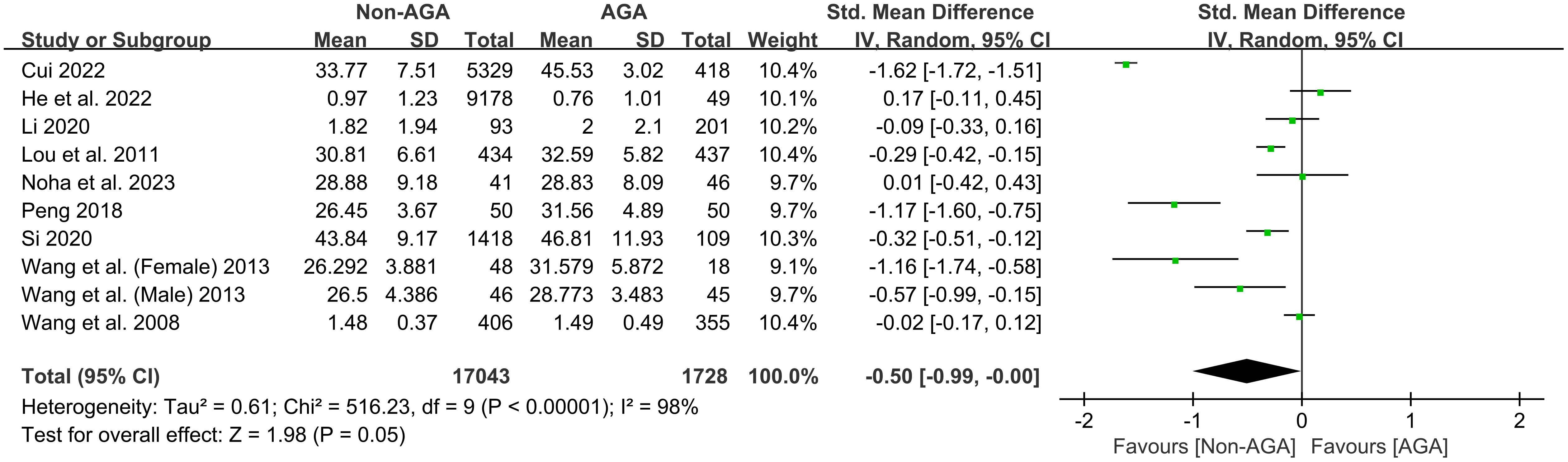
Figure 2. Forest plot of the meta-analysis on the relationship between AGA and symptoms of generalized anxiety.
As for symptoms of social anxiety, a total of 3 studies in 2 articles (26, 28) were included in the analysis, involving 518 non-AGA individuals and 511 AGA patients. A random effects model was utilized in this study to integrate the results. The pooled SMD was -0.50, with a 95% CI of [-0.84, -0.16], which similarly indicates that patients with AGA have significantly higher scores on symptoms of social anxiety compared to non-AGA patients (Figure 3).
![Forest plot showing the standard mean difference between non-AGA and AGA groups from three studies: Cash TF et al. (Female) 1993, Cash TF et al. (Male) 1993, and Wang et al. 2018. The plot includes means, standard deviations, and total participants for each group. Confidence intervals for each study and overall effect are indicated. Overall, the plot favors non-AGA with a mean difference of -0.50 [-0.84, -0.16]. Heterogeneity is shown with Tau² = 0.07, Chi² = 8.94, and an I² of 78%.](https://www.frontiersin.org/files/Articles/1705957/fpsyt-16-1705957-HTML-r1/image_m/fpsyt-16-1705957-g003.jpg)
Figure 3. Forest plot of the meta-analysis on the relationship between AGA and symptoms of social anxiety.
3.3 Association between AGA and symptoms of depression
8 studies (12, 13, 19, 21, 22, 24–26) were analyzed, including 17,005 non-AGA and 2,033 AGA participants. The results from multiple independent studies were integrated using a random effects model. The overall effect had an SMD of -0.38, with a 95% CI of [-0.65, -0.12], indicating that patients with AGA had significantly higher scores on depression symptoms compared to those without AGA (Figure 4).
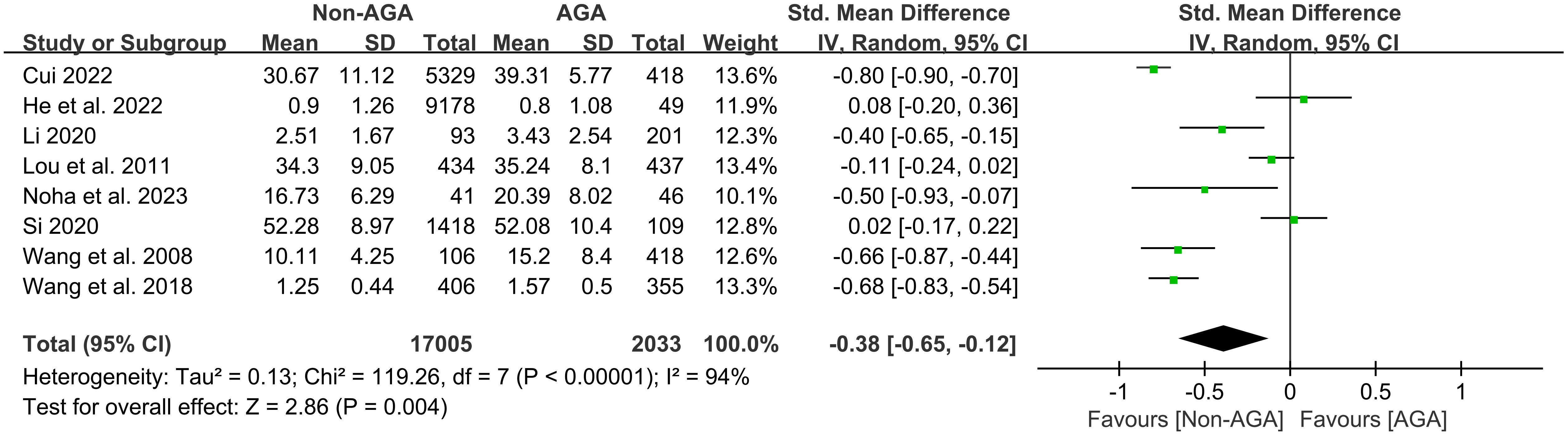
Figure 4. Forest plot of the meta-analysis on the relationship between AGA and symptoms of depression.
3.4 Association between AGA and perceived stress
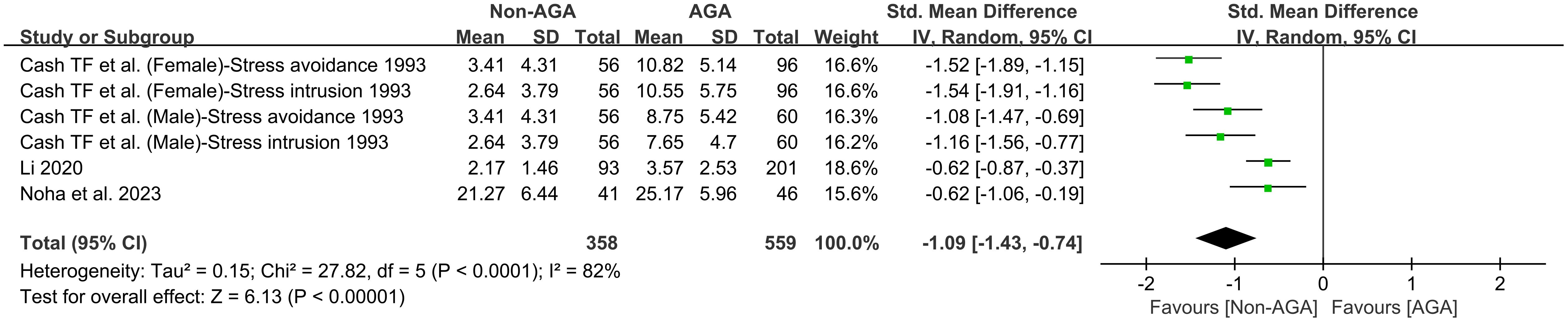
This meta-analysis included 6 studies (12, 19, 28), with a total of 358 participants in the non-AGA group and 559 in the AGA group. A random effects model was used to pool the results. The pooled effect size, represented by an SMD of -1.09, with a 95% CI ranging from -1.43 to -0.74, suggests that individuals with AGA reported significantly elevated levels of perceived stress in comparison to those without AGA (Figure 5).
3.5 Association between AGA and general distress
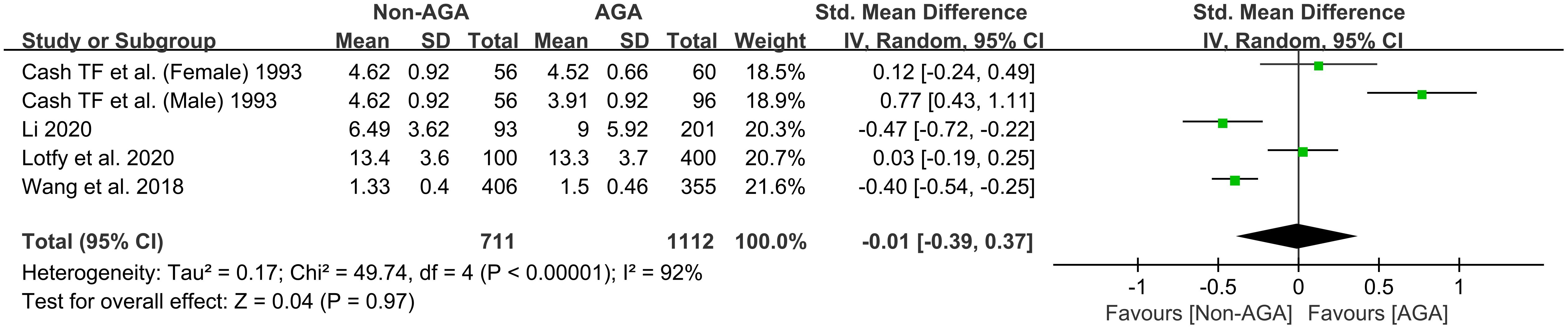
The meta-analysis incorporated 5 studies (19, 26–28) involving a total of 711 participants from the non-AGA group and 1,112 from the AGA group. Results were pooled using a random effects model. The pooled SMD was -0.01, with a 95% CI ranging from -0.39 to 0.37, indicating that there is no significant difference in the scores of general distress between the AGA group and the non-AGA group (Figure 6).
3.6 Association between AGA and other conditions of psychological well-being
Cash et al. (28) compared women with AGA, men with AGA, and a female control group, finding that AGA patients, especially women, exhibited more severe body image dissatisfaction, lower self-esteem, and lower life satisfaction. Assaf et al. (29) discovered that AGA patients had lower levels of Self-Actualization, Problem Solving, Optimism, and Happiness in terms of emotional intelligence. Wang et al. (26) conducted a study on 355 Chinese college students and found that the AGA group had higher levels of Somatization, Interpersonal Sensitivity, and Psychoticism, but lower levels of Obsessive-Compulsive behavior.
3.7 Publication bias and sensitivity analysis
Publication bias analysis was conducted for each dimension, with the funnel plot results illustrated in Figure 7. Both Egger’s regression test and Begg’s rank correlation test yielded p > 0.05, indicating minimal publication bias across the studies.
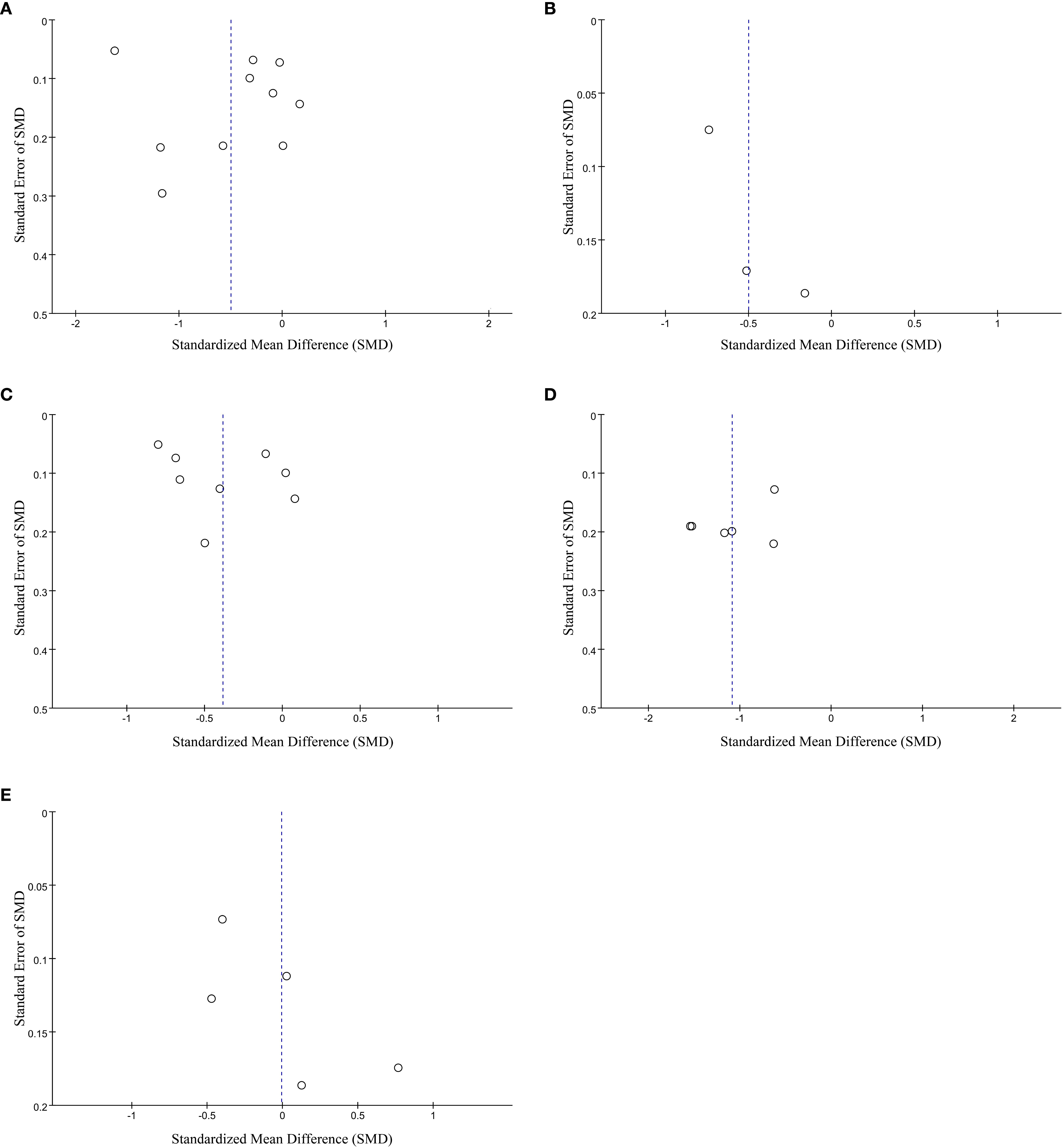
Figure 7. (A) Funnel plots of the meta-analysis on the relationship between AGA and symptoms of generalized anxiety. (B) Funnel plots of the meta-analysis on the relationship between AGA and symptoms of social anxiety. (C) Funnel plots of the meta-analysis on the relationship between AGA and symptoms of depression. (D) Funnel plots of the meta-analysis on the relationship between AGA and general distress. (E) Funnel plots of the meta-analysis on the relationship between AGA and general distress.
A leave-one-out sensitivity analysis was performed to evaluate the influence of individual studies on the pooled effect and heterogeneity. The exclusion of any single study did not result in significant alterations to the overall effect size estimate or its 95% confidence interval for each meta-analysis, suggesting that the results are not overly reliant on any particular study.
4 Discussion
4.1 AGA and symptoms of anxiety
We have explored the relationship between AGA and anxiety and found that individuals with AGA tend to have higher levels of anxiety, including both generalized anxiety and social anxiety. Previous research has shown that most men with AGA believe hair loss negatively impacts their social life, and that the more severe the hair loss, the higher the level of anxiety experienced by the patients (30, 31). And it is worth noting that previous studies have identified moderate to severe anxiety as a risk factor for AGA (32). This suggests a possible bidirectional relationship between anxiety and AGA, where anxiety can trigger hair loss, and in turn, hair loss can exacerbate anxiety.
4.2 AGA and symptoms of depression
Depression, as a complex and potentially severe psychological well-being state, warrants our attention regarding its association with AGA. The results of this study indicate that the level of depression symptoms in the AGA group is significantly higher. This is similar to the findings of previous studies. For instance, a study involving medical students in India found that the more severe the hair loss, the higher the level of depression (33). A study by Liu et al. in China found that the level of depression in the androgenic alopecia group was significantly higher than that in the non-AGA control group (34).
4.3 AGA and stress
Our study reveals that AGA patients have significantly higher stress levels than non-AGA controls. The link between stress and AGA has emerged as a key research focus in the medical field, with multiple studies identifying stress as a significant risk factor for AGA (32, 35).
4.4 AGA and general distress
General distress encompasses a range of comprehensive evaluation indicators designed to assess an individual’s overall psychological distress without necessitating a specific psychiatric diagnosis. The meta-analysis results on general distress reveal that, unlike the findings from meta-analyses focusing on specific dimensions, there is no significant difference in overall psychological well-being between the AGA and non-AGA populations. Similarly, Lin et al.’s study also found no significant difference in general distress between these two groups (11). This lack of difference may be attributed to several factors: (a)Limitations of the study sample or methods: The sample size or the specific methodologies employed in the studies may not have been sufficient to detect a significant difference; (b)Indirect impact of AGA on general distress: The influence of AGA on general distress may be mediated by other factors, such as social support or personal coping strategies; (c)Complexity of the concept of general distress: “General distress” is a complex, multidimensional concept that may be challenging to measure accurately with a single indicator.
4.5 AGA and other psychological well-being
In this study, AGA patients were found to have lower levels of self-esteem, life satisfaction, and emotional intelligence dimensions such as self-actualization, problem-solving, optimism, and happiness. They also exhibited higher levels of obsessive-compulsive behavior, body image dissatisfaction, somatization, interpersonal sensitivity, and psychoticism. However, due to the limited number of studies focusing on single symptoms, a meta-analysis could not be conducted across these dimensions.
4.6 Potential mechanisms underlying the relationship between AGA and psychological well-being
AGA is a multifactorial condition that not only affects hair growth but also has significant psychosocial and biological implications. The potential psychosocial and biological mechanisms underlying the association between AGA and psychological well-being are complex and interconnected.
4.6.1 Psychosocial mechanisms
a. Self-image and social anxiety: The visible changes in appearance caused by AGA can create a significant cognitive dissonance in how patients perceive themselves. This can lead to heightened concerns about appearance and the potential stigma associated with hair loss. As a result, patients may become overly preoccupied with negative social evaluations, which in turn can trigger social anxiety (4, 36). This heightened self-awareness and fear of negative judgment can significantly impact an individual’s social interactions and overall mental health;
b. The influence of disease cognition: AGA is a condition that can profoundly affect a patient’s sense of control over their health. The lack of understanding of the disease’s pathology and the uncertainty surrounding its progression can lead to feelings of helplessness. Patients who feel they have little control over their condition are more likely to experience higher levels of psychological distress, including increased anxiety and depression (37, 38);
c. The role of stress and quality of life: People suffering from hair loss often bear a heavier burden of stress (12, 19), which is further exacerbated by concerns about their hair loss itself (39). This stress not only deteriorates their mental health (40, 41), but also has a significant negative impact on their quality of life. Research has shown a significant positive correlation between quality of life scores and depression symptom scores (24), indicating that a decline in quality of life may worsen depressive symptoms. Moreover, stress, as a state of homeostatic imbalance, often coexists with psychological disorders such as anxiety and depression (40–43), suggesting a potential comorbidity between AGA and adverse psychological states.
4.6.2 Biological mechanisms
a. Activation of the hypothalamic-pituitary-adrenal (HPA) axis: Chronic stress activates the HPA axis, leading to persistently elevated cortisol levels that increase the sensitivity of hair follicles to dihydrotestosterone (DHT) and indirectly worsen androgenic damage through neurosteroid synthesis (44–46);
b. Inflammatory response: Stress induces the release of pro-inflammatory factors, inhibits the proliferation of hair follicle stem cells, disrupts the hair follicle cycle, and suppresses regenerative signaling in the Wnt pathway (47, 48);
c. Oxidative stress: The accumulation of reactive oxygen species (ROS) disrupts the antioxidant defenses of hair follicles, damages the DNA of dermal papilla cells, inhibits the expression of key follicular proteins, and accelerates miniaturization (45, 49);
d. Reduction in growth supply: Stress releases epinephrine and norepinephrine, causing vasoconstriction that reduces blood supply to hair follicles. It also alters neurotrophic factor levels, disrupting follicle function (12, 47, 50, 51);
e. Genetic amplification effect: Individuals with androgen receptor (AR) gene variants are more sensitive to stress-induced androgen signals, and epigenetic regulation further amplifies genetic risk (45, 52).
4.6.3 Impact of treatment drugs
Existing research indicates that AGA treatment drugs (such as finasteride) themselves may induce psychological side effects such as anxiety, depression, and even suicidal tendencies (53, 54). The confounding effect of the treatment drug should be taken seriously. The potential for treatment-induced psychological side effects underscores the importance of careful monitoring and management of patients undergoing AGA treatment.
In fact, the reasons for the correlation between AGA and a range of mental health issues are complex and multifaceted. This complexity stems not only from the inherent complexity of psychological well-being but also from the influence of sociocultural contexts and personal cognition. For example, in a study by Lin et al. (11), an evaluation of medical staff in a Chinese hospital found that AGA patients exhibited more severe somatization and higher hostility, although they did not have higher levels of interpersonal sensitivity, psychoticism, or lower obsessive-compulsive behavior. And given the variability in environments and self-awareness, AGA patients in specific populations—such as those in certain professions, individuals with high educational levels, unmarried individuals, younger patients, individuals identifying as sexual minority, and those seeking treatment—are particularly prone to psychological well-being issues (4, 26, 55–58). Moreover, it is important to note that some of these symptoms often co-occur and interact, leading to a more complex state of overall psychological well-being for the patients (41, 59). Therefore, research into the mechanisms underlying the relationship between AGA and psychological well-being should be further advanced, and the psychological well-being of AGA patients’ needs to be assessed from multiple dimensions, especially in certain specific populations.
Overall, the relationship between AGA and psychological well-being is multidimensional, complex, and interactive. Firstly, there is a significant association between AGA and various psychological well-being issues, with the mechanisms of influence being multifaceted and intricate. Secondly, this relationship is influenced by the social environment and personal cognition. Last but not least, AGA and psychological well-being are bidirectionally related, as poor psychological well-being can increase the risk of AGA, and hair loss can lead to adverse psychological well-being states in patients.
Future research and clinical practice should focus on the psychological health of AGA patients. First, psychological assessment should be incorporated into the routine clinical process. Standardized psychological scales should be used to comprehensively evaluate patients’ anxiety, depression, stress, and other psychological states, so as to detect and intervene potential problems in time. Second, multidisciplinary collaboration should be strengthened. Dermatologists should work together with psychologists to provide comprehensive treatment plans for patients. For example, patients with significant anxiety and depression symptoms should be referred to psychologists for cognitive-behavioral therapy or pharmacotherapy.
In addition, future research should focus on long-term follow-up of AGA and psychological states. Long-term tracking studies should be conducted to further explore the long-term impact of psychological interventions on the progression of AGA. At the same time, the mechanisms of interaction between AGA and psychological states should be further investigated, including genetic variations and neuroendocrine pathways. Targeted studies should be conducted on specific groups, such as college students and office workers, to analyze the relationship between psychological stress and AGA and explore effective intervention measures. Through these measures, the psychological health of AGA patients can be better improved in the future, and their quality of life can be enhanced.
4.7 Strengths and limitations
This systematic review and meta-analysis have several strengths: (a) It is the first systematic review and meta-analysis to examine the relationship between AGA and multiple dimensions of psychological well-being, strictly following PRISMA guidelines; (b) Unlike prior studies that only targeted individuals with AGA, this one systematically compared AGA and non-AGA groups, with both a qualitative review and quantitative analyses; (c) A comprehensive search was conducted across seven databases, including Chinese-language studies, which provided support from Chinese data.
However, this meta-analysis also has some limitations: (a) The studies were restricted to Chinese and English, and the majority of participants were male, resulting in a gender imbalance; (b) Most studies included used a cross-sectional design, with causality requiring further confirmation; (c) The limited number of eligible studies made it challenging to conduct subgroup analyses to explore the sources of heterogeneity and the impact of potential confounding factors; (d) The studies included in this research were conducted across multiple regions, which may introduce potential influences on the results due to cultural differences and self-reporting biases.
5 Conclusion
This systematic review and meta-analysis comprehensively evaluated the association between AGA and psychological well-being across multiple dimensions. The findings indicate that AGA is significantly associated with heightened levels of symptoms of generalized and social anxiety, symptoms of depression, perceived stress, body image dissatisfaction, somatization, interpersonal sensitivity, and psychoticism. Additionally, AGA is linked to diminished self-esteem, life satisfaction, emotional intelligence, self-actualization, problem-solving ability, optimism, and happiness. In the future, it is crucial to prioritize patient psychological well-being through assessments and timely interventions. Meanwhile, further research should be conducted to provide precise clinical guidance.
Data availability statement
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding author.
Author contributions
YK: Conceptualization, Data curation, Funding acquisition, Investigation, Software, Writing – original draft, Writing – review & editing. YS: Conceptualization, Data curation, Formal Analysis, Investigation, Software, Writing – original draft, Writing – review & editing. LZ: Conceptualization, Data curation, Methodology, Project administration, Supervision, Writing – original draft.
Funding
The author(s) declare financial support was received for the research and/or publication of this article. This work was supported by the Project of Beijing Changping Hospital of Integrated Chinese and Western Medicine (Grant Nos. YJKT-20250029 and YJKT-20250082).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fpsyt.2025.1705957/full#supplementary-material
References
1. Su Q, Zhou C, He C, Jiao Q, Wang Z, and Jia Y. Research progress in composition, classification and influencing factors of hair. Asian J Beauty Cosmetol. (2023) 21:503–16. doi: 10.20402/ajbc.2023.0001
2. Wu X, Xu P, Zhang Y, and Zhang Z. Dinguishing human characteristics based on hair metabolomics and proteomics: A review. Chin J Biotech. (2022) 38:3638–47. doi: 10.13345/j.cjb.220526
3. Frith H and Jankowski GS. Psychosocial impact of androgenetic alopecia on men: A systematic review and meta-analysis. Psychol Health Med. (2024) 29:822–42. doi: 10.1080/13548506.2023.2242049
4. Aukerman EL and Jafferany M. The psychological consequences of androgenetic alopecia: A systematic review. J Cosmet Dermatol. (2023) 22:89–95. doi: 10.1111/jocd.14983
5. Blume-Peytavi U, Blumeyer A, Tosti A, Finner A, Marmol V, Trakatelli M, et al. S1 guideline for diagnostic evaluation in androgenetic alopecia in men, women and adolescents. Br J Dermatol. (2011) 164:5–15. doi: 10.1111/j.1365-2133.2010.10011.x
6. Oiwoh SO, Akinboro AO, Olasode OA, and Onayemi EO. Androgenetic alopecia: prevalence and clinical characteristics in a south-west Nigerian population. Nigerian J Med. (2021) 30:507–13. doi: 10.4103/NJM.NJM_102_21
7. Li G and Ding J. Research progress on the pathogenesis of androgenetic alopecia and the western medicine dermal treatment. China Med Her. (2017) 14:43–6+70.
8. Oiwoh SO, Enitan AO, Adegbosin OT, Akinboro AO, and Onayemi EO. Androgenetic alopecia: A review. Niger Postgrad Med J. (2024) 31:85–92. doi: 10.4103/npmj.npmj_47_24
9. Huang C-H, Fu Y, and Chi C-C. Health-related quality of life, depression, and self-esteem in patients with androgenetic alopecia a systematic review and meta-analysis. JAMA Dermatol. (2021) 157:963–70. doi: 10.1001/jamadermatol.2021.2196
10. Almashali MA, Alotaibi MA, Alkhanani AH, Al Dera NM, Alwadany MM, Almousa AS, et al. The psychosocial burden of androgenetic alopecia in Saudi Arabia: A cross-sectional study. J Family Med Prim Care. (2023) 12:3374–9. doi: 10.4103/jfmpc.jfmpc_1151_23
11. Lin X, Wei Y, Lin C, and Liu C. Investigation and analysis of the current situation of alopecia and its related factors among medical staff in jieyang. J Shantou Univ Med Coll. (2024) 37:58–62. doi: 10.13401/j.cnki.jsumc.2024.01.013
12. Mohamed NE, Soltan MR, Galal SA, El Sayed HS, Hassan HM, and Khatery BH. Female pattern hair loss and negative psychological impact: possible role of brain-derived neurotrophic factor (Bdnf). Dermatol Pract conceptual. (2023) 13. doi: 10.5826/dpc.1303a139
13. He F, Shen M, Zhao Z, Liu Y, Zhang S, Tang Y, et al. Epidemiology and disease burden of androgenetic alopecia in college freshmen in China: A population-based study. PloS One. (2022) 17:e0263912. doi: 10.1371/journal.pone.0263912
14. Sinikumpu SP, Jokelainen J, Auvinen J, Timonen M, and Huilaja L. Association between psychosocial distress, sexual disorders, self-esteem and quality of life with male androgenetic alopecia: A population-based study with men at age 46. BMJ Open. (2021) 11. doi: 10.1136/bmjopen-2021-049855
15. Zeng J, He J, Chen M, and Li J. Association between mean platelet volume and obstructive sleep apnea-hypopnea syndrome: A systemic review and meta-analysis. PloS One. (2024) 19:e0297815. doi: 10.1371/journal.pone.0297815
16. Linardon J, Cuijpers P, Carlbring P, Messer M, and Fuller-Tyszkiewicz M. The efficacy of app-supported smartphone interventions for mental health problems: A meta-analysis of randomized controlled trials. World Psychiatry. (2019) 18:325–36. doi: 10.1002/wps.20673
17. Higgins JPT TJ, Chandler J, Cumpston M, Li T, Page MJ, and Welch VA eds. Cochrane Handbook for Systematic Reviews of Interventions Version 6.5. In: Cochrane.
18. Wan X, Wang W, Liu J, and Tong T. Estimating the sample mean and standard deviation from the sample size, median, range and/or interquartile range. BMC Med Res Methodol. (2014) 14:135. doi: 10.1186/1471-2288-14-135
19. Li Q. A Correlation Study of Negative Emotions and Chinese Medicine Syndrome of Androgenic Alopecia. China: Guangzhou University of Chinese Medicine (2020).
20. Peng D. Research on the relationship between various alopecia diseases, sleep quality and emotional factors. World Latest Med Inf. (2018) 18:164+8. doi: 10.19613/j.cnki.1671-3141.2018.58.080
21. Cui W. Investigation and Analysis of Androgenetic Alopecia and Related Factors among College Students in Jiamusi University. China: Jiamusi University (2022).
22. Si Y. An Investigation on the Prevalence of Androgenetic Alopecia and Its Related Factors in Students of Ningxia Medical University. China: Ningxia Mdedical University (2020).
23. Wang D, Xu Q, Lei X, Wu X, and Wu J. Correlation between the three kinds of common hair loss diseases, sleep quality and emotional factors. J Pract Dermatol. (2013) 6:339–42.
24. Wang L, Fan W, Cao L, Xu W, Liu L, Li Z, et al. The life quality of patients with alopecia. J Clin Dermatol. (2008) 37:417–9. doi: 10.3969/j.issn.1000-4963.2008.07.002
25. Lou W and Yang Q. A preliminary study on the psychological and psychiatric situation and quality of life on the patients with alopecia. China J Leprosy Skin Dis. (2011) 27:219–20. doi: 10.3969/j.issn.1009-1157.2011.03.041
26. Wang X, Xiong C, Zhang L, Yang B, Wei R, Cui L, et al. Psychological assessment in 355 chinese college students with androgenetic alopecia. Med (United States). (2018) 97. doi: 10.1097/MD.0000000000011315
27. Elsaie LT, Elshahid AR, Hasan HM, Soultan FAZM, Jafferany M, and Elsaie ML. Cross sectional quality of life assessment in patients with androgenetic alopecia. Dermatologic Ther. (2020) 33:e13799. doi: 10.1111/dth.13799
28. Cash TF, Price VH, and Savin RC. Psychological effects of androgenetic alopecia on women: comparisons with balding men and with female control subjects. J Am Acad Dermatol. (1993) 29:568–75. doi: 10.1016/0190-9622(93)70223-g
29. Monselise A, Bar-On R, Chan L, Leibushor N, McElwee K, and Shapiro J. Examining the relationship between alopecia areata, androgenetic alopecia, and emotional intelligence. J cutaneous Med Surg. (2013) 17:46–51. doi: 10.2310/7750.2012.12003
30. Cash TF. The psychological effects of androgenetic alopecia in men. J Am Acad Dermatol. (1992) 26:926–31. doi: 10.1016/0190-9622(92)70134-2
31. Hwang HW, Ryou S, Hyeong Jeong J, Lee JW, Lee KJ, Lee SB, et al. The quality of life and psychosocial impact on female pattern hair loss. Ann Dermatol. (2024) 36:44–52. doi: 10.5021/ad.23.082
32. Feng J, Zhang Z, Xie J, and Song J. Investigation of influencing factors of androgenetic alopecia in wuhan city. Med J Wuhan Univ. (2023) 44:65–9. doi: 10.14188/j.1671-8852.2022.0520
33. Prasanna H, Rajashekar TS, Kumar KS, Athish KK, Kiran M, and Reddy M. The association of depression, loneliness, and internet addiction levels in male bachelor of medicine, bachelor of surgery (Mbbs) students with androgenetic alopecia male pattern baldness in a medical college in kolar, India. Cureus J Med Sci. (2023) 15. doi: 10.7759/cureus.35607
34. Liu S, Gu H, Ji R, Shi W, Liu F, Xie H, et al. The mediation role of sleep on the relationship between drinks behavior and female androgenetic alopecia. PeerJ. (2024) 12:e18647. doi: 10.7717/peerj.18647
35. Ye X, Song X, Liu X, Zhou W, Wang F, and Chen H. Analysis of influencing factors of female androgenetic alopecia in young women. Chongqing Med J. (2024) 53:69–72+8.
36. Creadore A, Manjaly P, Li SJ, Tkachenko E, Zhou G, Joyce C, et al. Evaluation of stigma toward individuals with alopecia. JAMA Dermatol. (2021) 157:392–8. doi: 10.1001/jamadermatol.2020.5732
37. Alotaibi HM, Idris RB, Alajlan AH, Alghufaili AA, Barakeh M, Alobaid SA, et al. Illness perception, psychological distress, and quality of life in patients with alopecia: A cross-sectional study from riyadh, Saudi Arabia. Arch Dermatol Res. (2025) 317:646. doi: 10.1007/s00403-025-04093-2
38. Yu NL, Tan H, Song ZQ, and Yang XC. Illness perception in patients with androgenetic alopecia and alopecia areata in China. J Psychosom Res. (2016) 86:1–6. doi: 10.1016/j.jpsychores.2016.04.005
39. Li Y, Li J, Xie Z, Zhou L, Liu Y, Wang W, et al. The relationship between uncertainty stress and perceived anxiety on hair loss among college students in guangzhou. Chin J Soc Med. (2024) 41:72–6. doi: 10.3969/j.issn.1673-5625.2024.01.018
40. Hou Y, Shang M, Yu X, Gu Y, Li H, Lu M, et al. Joint effects of recent stressful life events and adverse childhood experiences on perinatal comorbid anxiety and depression. BMC Pregnancy Childbirth. (2023) 23:41. doi: 10.1186/s12884-023-05375-1
41. Davies MR, Glen K, Mundy J, ter Kuile AR, Adey BN, Armour C, et al. Factors associated with anxiety disorder comorbidity. J Affect Disord. (2023) 323:280–91. doi: 10.1016/j.jad.2022.11.051
42. Lu S, Wei F, and Li G. The evolution of the concept of stress and the framework of the stress system. Cell Stress. (2021) 5:76–85. doi: 10.15698/cst2021.06.250
43. Weinmann T, Wibowo R, Forster F, Gerlich J, Wengenroth L, Weinmayr G, et al. Association of chronic stress during studies with depressive symptoms 10 years later. Sci Rep. (2025) 15:2379. doi: 10.1038/s41598-025-85311-9
44. Craft TK and Devries AC. Vulnerability to stroke: implications of perinatal programming of the hypothalamic-pituitary-adrenal axis. Front Behav Neurosci. (2009) 3:54.2009. doi: 10.3389/neuro.08.054.2009
45. Lolli F, Pallotti F, Rossi A, Fortuna MC, Caro G, Lenzi A, et al. Androgenetic alopecia: A review. Endocrine. (2017) 57:9–17. doi: 10.1007/s12020-017-1280-y
46. McArdle CJ, Arnone AA, Heaney CF, and Raab-Graham KF. A paradoxical switch: the implications of excitatory gabaergic signaling in neurological disorders. Front Psychiatry. (2023) 14:1296527. doi: 10.3389/fpsyt.2023.1296527
47. Vishnyakova KS, Vetkova LG, Jasko MV, Aliper AM, Buzdin AA, Popov KV, et al. Hair growth stimulation by a natural remedy: animal studies. Madridge J Dermatol Res. (2018) 3:37–44. doi: 10.18689/mjdr-1000109
48. Qi J and Garza LA. An overview of alopecias. Cold Spring Harb Perspect Med. (2014) 4. doi: 10.1101/cshperspect.a013615
49. Diab HM, Soltan MY, El-Khazragy N, and Atef Raafat AM. Assessment of the oxidative stress status in androgenetic alopecia in Egyptian men. QJM: Int J Med. (2021) 114. doi: 10.1093/qjmed/hcab093.006
50. Huan N, Yu Y, Wang P, and Wang C. Research progress regarding the diagnosis and treatment of mental stress-induced myocardial ischemia. Anatol J Cardiol. (2020) 24:126–36. doi: 10.14744/AnatolJCardiol.2020.69447
51. Cheng Y, Lv L-J, Cui Y, Han X-M, Zhang Y, and Hu C-X. Psychological stress impact neurotrophic factor levels in patients with androgenetic alopecia and correlated with disease progression. World J Psychiatry. (2024) 14:1437–47. doi: 10.5498/wjp.v14.i10.1437
52. Sadasivam IP, Sambandam R, Kaliyaperumal D, and Dileep JE. Androgenetic alopecia in men: an update on genetics. Indian J Dermatol. (2024) 69:282. doi: 10.4103/ijd.ijd_729_23
53. Trüeb RM, Gavazzoni Dias MFR, and Dutra Rezende H. Suicidality and psychological adverse events in patients treated with finasteride. Skin Appendage Disord. (2021) 7:524–6. doi: 10.1159/000514365
54. Lyakhovitsky A, Amichai B, Galili E, Cohen A, Kridin K, Segal Z, et al. The risk of psychiatric disorders in finasteride users with benign prostatic hyperplasia and androgenetic alopecia: A population-based case-control study. Australas J Dermatol. (2024) 65:621–9. doi: 10.1111/ajd.14359
55. Adamowicz R, Załęcki P, Dukiel A, and Nowicka D. Association between androgenetic alopecia and psychosocial disease burden: A cross-sectional survey among polish men. Dermatol Res Pract. (2022) 2022:1845044. doi: 10.1155/2022/1845044
56. Zucchelli F, Mathews A, Sharratt N, Montgomery K, and Chambers J. The psychosocial impact of alopecia in men: A mixed-methods survey study. Skin Health Dis. (2024) 4:e420. doi: 10.1002/ski2.420
57. Tas B, Kulacaoglu F, Belli H, and Altuntas M. The tendency towards the development of psychosexual disorders in androgenetic alopecia according to the different stages of hair loss: A cross-sectional study. Bras Dermatol. (2018) 93:185–90. doi: 10.1590/abd1806-4841.20185658
58. Moorthy SK, Yu L, Peng L, Shen L, Han Y, Zhang Z, et al. Quality of life and its association with androgenetic alopecia patients in shanghai: A cross-sectional study. Clinical Cosmetic Investigational Dermatol. (2022) 15:2883–93. doi: 10.2147/ccid.s393633
Keywords: androgenetic alopecia, psychology, anxiety, depression, stress, meta
Citation: Kong Y, Shang Y and Zhang L (2025) Association between androgenetic alopecia and psychological well-being: a systematic review and meta-analysis. Front. Psychiatry 16:1705957. doi: 10.3389/fpsyt.2025.1705957
Received: 15 September 2025; Accepted: 10 November 2025; Revised: 06 November 2025;
Published: 26 November 2025.
Edited by:
Shinsuke Hidese, Teikyo University, JapanReviewed by:
Marije Van Dalen, Erasmus Medical Center, NetherlandsElnaz Pourgholi, Shahid Beheshti University of Medical Sciences, Iran
Copyright © 2025 Kong, Shang and Zhang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Liuwei Zhang, MjQ2NkBic3UuZWR1LmNu
†These authors have contributed equally to this work and share first authorship
 Yuan Kong1†
Yuan Kong1† Liuwei Zhang
Liuwei Zhang