- 1Department of Population Medicine and Diagnostic Sciences, College of Veterinary Medicine, Cornell University, Ithaca, NY, United States
- 2Animal Health Diagnostic Center, College of Veterinary Medicine, Cornell University, Ithaca, NY, United States
A Corrigendum on
Dogs with acute myeloid leukemia have clonal rearrangements in T and B cell receptors
by Stokol, T., Nickerson, G. A., Shuman, M., and Belcher, N. (2017). Front. Vet. Sci. 4:76. doi: 10.3389/fvets.2017.00076
In the published article, there was an error in Figure 1, Table 1, Table 2, Table 3, and Table 6 as published. CD1c in the Figures and Tables should be CD1a. The authors discovered this error after publication of the article when the source of the antibody sent a notification stating that the antigen was not CD1c as originally indicated. The corrected Figure and Tables and their captions appear below.
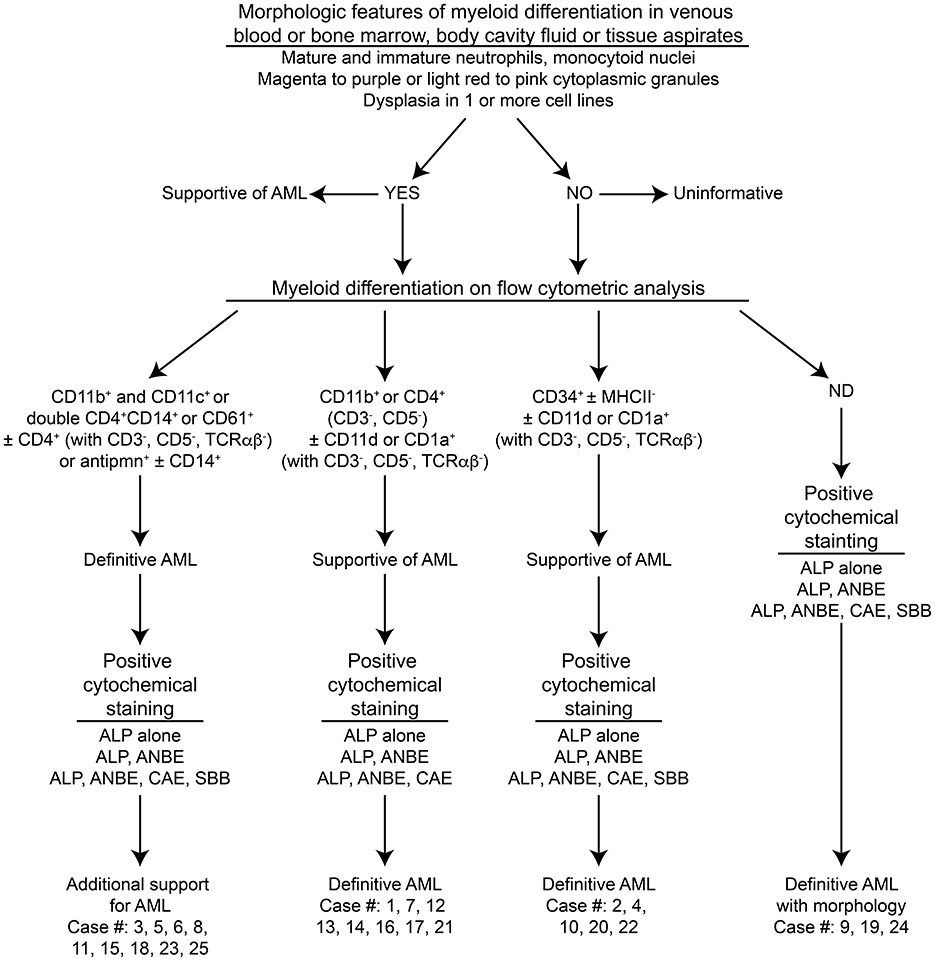
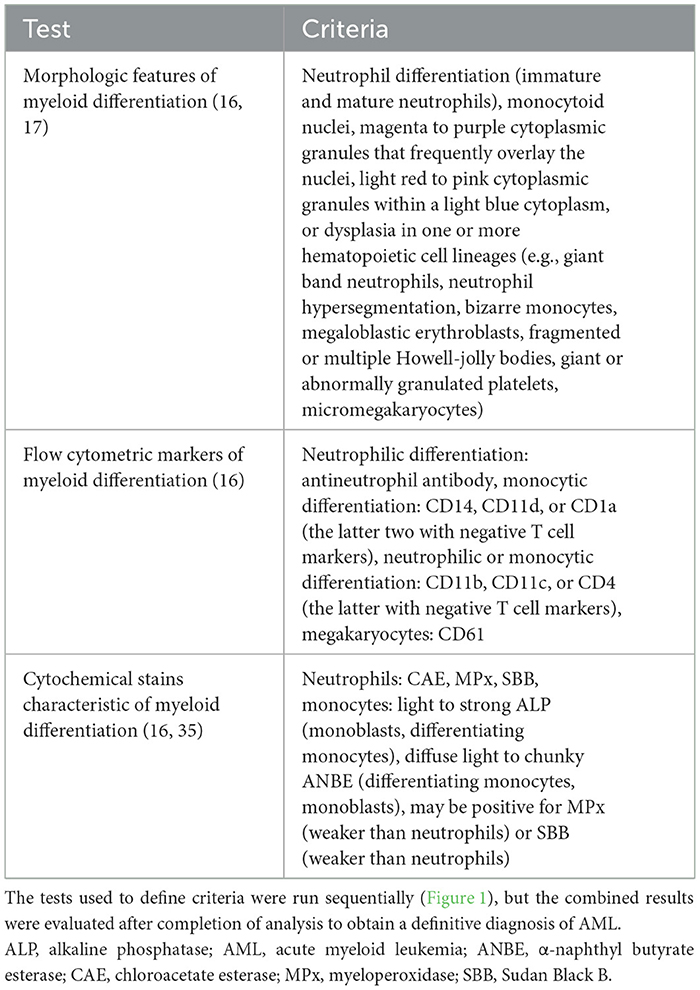
Figure 1. Algorithm used to diagnose acute myeloid leukemia (AML) in the 25 dogs of this study. This diagnostic algorithm was based on the order in which tests were generally performed in our laboratory, i.e., morphologic assessment of blood, bone marrow, or body cavity fluid or tissue aspirates, followed by flow cytometric analysis (performed routinely twice a week), followed by cytochemical staining (performed as needed). After completion of all the tests, the results were reevaluated, and a diagnosis of AML was based on the combined data. The path used to diagnose each case (#) is also shown. More details on the criteria are provided in Table 1.
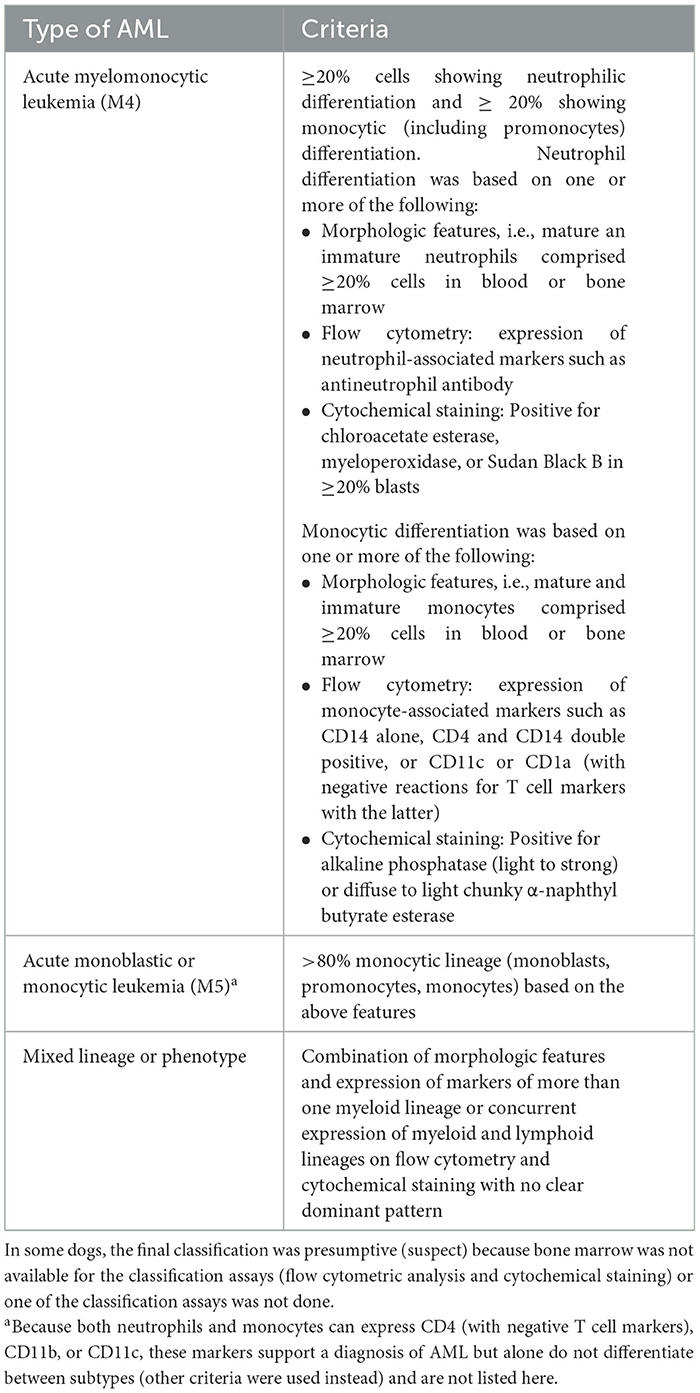
Table 2. Criteria for classification of the subtype of acute myeloid leukemia (AML) based on the World Health Organization scheme – not otherwise specified (17, 18).
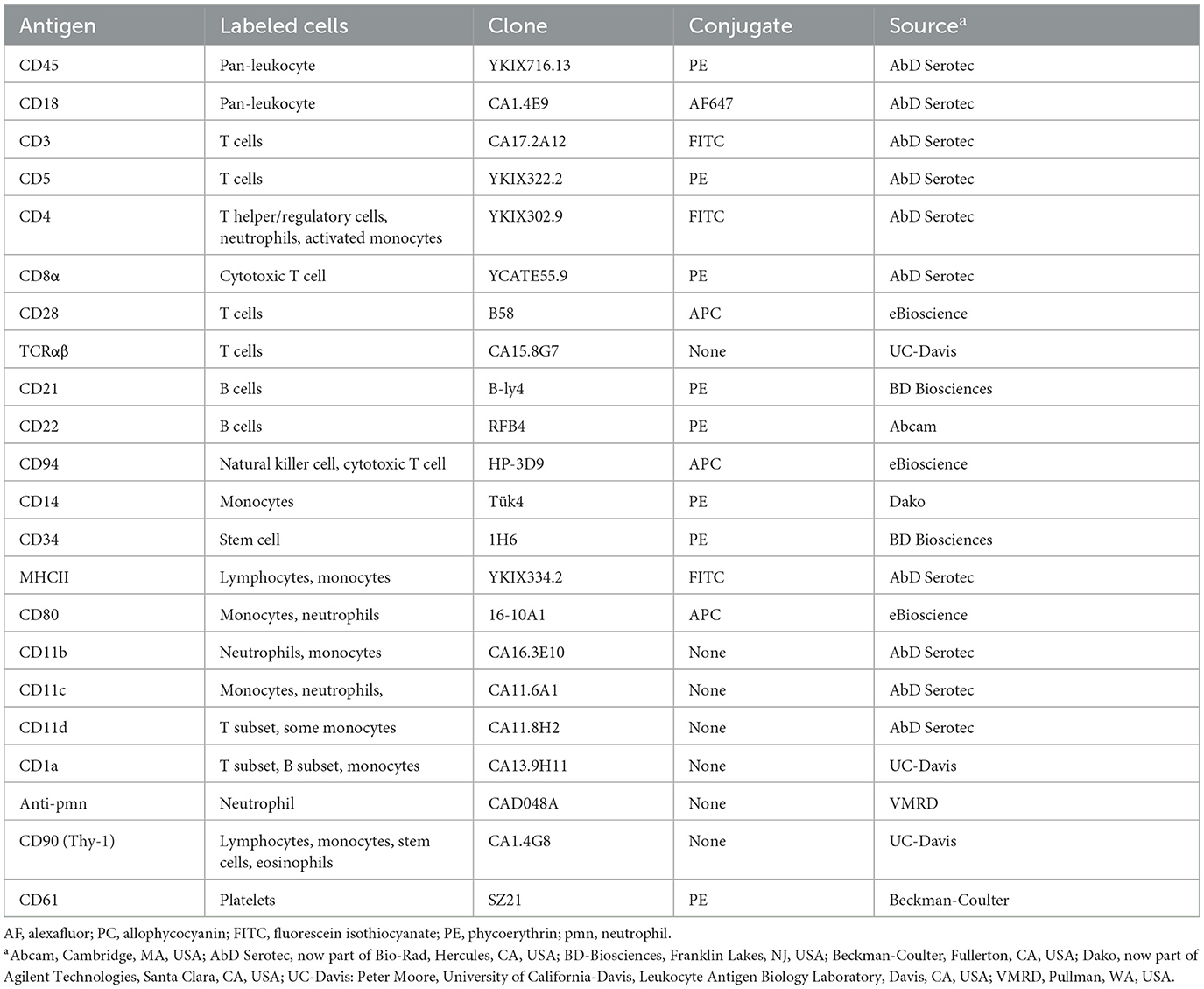
Table 3. Antibodies used in flow cytometry at Cornell University to label antigens on tumor cells in liquid samples (blood or bone marrow, body cavity fluid, or tissue aspirates) from dogs with acute myeloid leukemia.
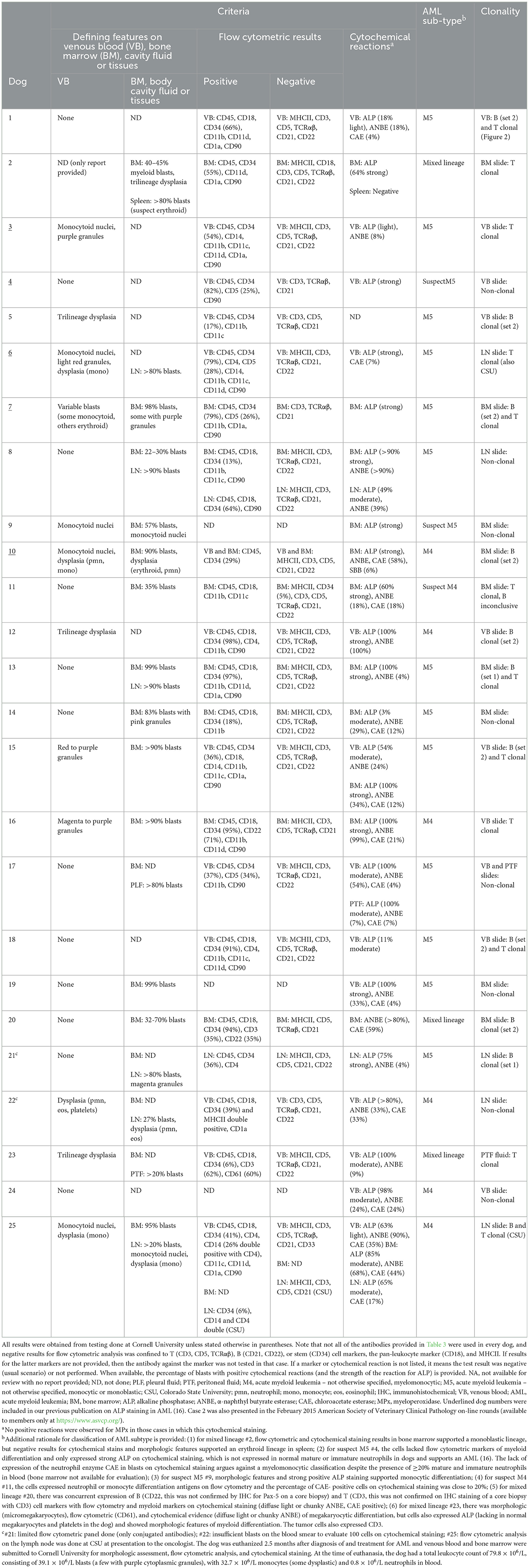
Table 6. Morphologic findings from blood or cytologic smears and results from flow cytometric labeling, cytochemical staining, and clonality testing in 25 dogs with AML.
The authors apologize for this error and state that this does not change the scientific conclusions of the article in any way. The original article has been updated.
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Keywords: acute myelogenous leukemia, canine, polymerase testing for antigen receptor rearrangements, clonality testing, phenotyping, leukemia, flow cytometry, cytochemistry
Citation: Stokol T, Nickerson GA, Shuman M and Belcher N (2025) Corrigendum: Dogs with acute myeloid leukemia have clonal rearrangements in T and B cell receptors. Front. Vet. Sci. 11:1533729. doi: 10.3389/fvets.2024.1533729
Received: 24 November 2024; Accepted: 03 December 2024;
Published: 10 January 2025.
Edited and reviewed by: Stefano Comazzi, University of Milan, Italy
Copyright © 2025 Stokol, Nickerson, Shuman and Belcher. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Tracy Stokol, dHJhY3kuc3Rva29sQGNvcm5lbGwuZWR1
 Tracy Stokol
Tracy Stokol Gabrielle A. Nickerson2
Gabrielle A. Nickerson2