Abstract
The Arctic is becoming warmer at a high rate, and contractions in the extent of sea ice are currently changing the habitats of marine top-predators dependent on ice. Polar bears (Ursus maritimus) depend on sea ice for hunting seals. For these top-predators, longer ice-free seasons are hypothesized to force the bears to hunt for alternative terrestrial food, such as eggs from colonial breeding birds. We analyzed time-series of polar bear observations at four locations on Spitsbergen (Svalbard) and one in east Greenland. Summer occurrence of polar bears, measured as the probability of encountering bears and the number of days with bear presence, has increased significantly from the 1970/80s to the present. The shifts in polar bear occurrence coincided with trends for shorter sea ice seasons and less sea ice during the spring in the study area. This resulted in a strong inverse relationship between the probability of bear encounters on land and the length of the sea ice season. Within, 10 years after their first appearance on land, polar bears had advanced their arrival dates by almost 30 days. Direct observations of nest predation showed that polar bears may severely affect reproductive success of the barnacle goose (Branta leucopsis), common eider (Somateria mollissima) and glaucous gull (Larus hyperboreus). Nest predation was strongest in years when the polar bears arrived well before hatch, with more than 90% of all nests being predated. The results are similar to findings from Canada, and large-scale processes, such as climate and subsequent habitat changes, are pinpointed as the most likely drivers in various parts of the Arctic. We suggest that the increasing, earlier appearance of bears on land in summer reflects behavioral adaptations by a small segment of the population to cope with a reduced hunting range on sea ice. This exemplifies how behavioral adaptations may contribute to the cascading effects of climate change.
Introduction
Understanding causes and consequences of climate-related shifts in ecosystem functioning, as well as the role of focal species in these processes, is currently a dominant theme in ecology. In the Arctic, temperature has increased at a rate two to three times faster than at southerly latitudes (Post et al., 2009). As a consequence, the rapid environmental changes that are taking place can be directly attributed to global warming (Gilg et al., 2012). The decrease in the extent of sea ice is on the order of 5% per decade in the Arctic (Liu et al., 2004; Serreze et al., 2007), and it is expected that this trend of disappearing sea ice will continue with the strongest losses predicted in the southern Arctic seas, including the Barents Sea area. Changes in the physical environment linked with global warming have become evident in the marine food web, for example, by shifts in the breeding phenology of sea birds (Barbraud and Weimerskirch, 2006; Moe et al., 2009). The Arctic terrestrial ecosystem has been affected as well, which includes changes in ecosystem functioning such as increased primary production (Madsen et al., 2011; Sistla et al., 2014).
One of the species directly affected by global warming is the polar bear (Ursus maritimus) whose life history is closely tied to sea ice (Lunn and Stirling, 1985; Ramsay and Hobson, 1991). Evidence is accumulating that polar bears are suffering from a warming climate and associated loss of sea ice habitat (Stirling et al., 1999; Derocher et al., 2004; Stirling and Derocher, 2012). It is expected that continued sea ice reductions will severely affect polar bear populations (Durner et al., 2009), which will force them into terrestrial ecosystems during the summer months in search of food (Stempniewicz, 1993; Drent and Prop, 2008; Rockwell and Gormezano, 2009; Smith et al., 2010; Hanssen et al., 2013; Prop et al., 2013; Iverson et al., 2014). Several studies have documented that polar bears on land can potentially have a large impact on their prey, in particular when bears feed on bird eggs (Drent and Prop, 2008; Smith et al., 2010; Prop et al., 2013; Iverson et al., 2014).
The occurrence of polar bears on land raises two fundamental questions. Firstly, what are the underlying causes of this shift? A major cause could be related to changes in sea ice conditions, as suggested by Iverson et al. (2014). Secondly, what is the role of polar bears as a novel top-predator in terrestrial habitats near the coast? To examine the role of polar bears in these terrestrial habitats, direct observations are needed, and these should be carried out including the periods before and after the appearance of bears.
In this paper, we document changes in the summer distribution of polar bears by exploring their relative presence at several locations on Spitsbergen and one location in Greenland. Based on the patterns of occurrence, which were collected over a period of >40 years, we assess the factors that may have contributed to the summer range expansion of polar bears. We test the hypothesis that the bear incursions on land are related to changes in sea ice conditions. Furthermore, we explore the effects of polar bears on the reproductive success of colonial breeding birds at one of our study locations.
Methods
Study areas
Observations were collected along the west coast of Spitsbergen, which is the largest island in the Svalbard archipelago, and the east coast of Greenland. The study areas on Spitsbergen are composed of flat tundra stretches of up to 15 km wide, delineated by steep mountains and glaciers (Hisdal, 1998). Large fjords intersect the area, and islands are scattered along the coast, with many of these areas hosting breeding colonies of barnacle geese (Branta leucopsis), common eiders (Somateria mollissima) and glaucous gulls (Larus hyperboreus). Further bird nesting aggregations occur on tundra flats [e.g., colonies of pink-footed geese (Anser brachyrhynchus)], shore cliffs, and mountains (Kovacs and Lydersen, 2006).
Traill Island is part of the North East Greenland coastal fringe that is deeply indented by a network of long fjords. At the outer coast, the extent of annual landfast ice is delineated by a sharp ice edge that separates it from southward moving drift ice of varying extent, originating from the Arctic Ocean. The coastal zones contain breeding habitats for several bird species (Boertmann, 1994). In contrast to Svalbard, barnacle geese breed here exclusively on cliffs, and this applies also to small aggregations of glaucous gulls. Arctic terns (Sterna paradisaea), common eiders and long-tailed ducks (Clangula hyemalis) mainly nest on small rocky islands. These species may delay onset of breeding until ice breakup in mid-July.
We compiled data from a total of five locations in Svalbard and Greenland: Hornsund, Bellsund, Nordenskiöldkysten, and Kongsfjorden on the west coast of Spitsbergen (77–79°N); and Traill Island on the east coast of Greenland (72–73°N) (Figure 1). Descriptions of these locations are in Supplement 1.
Figure 1
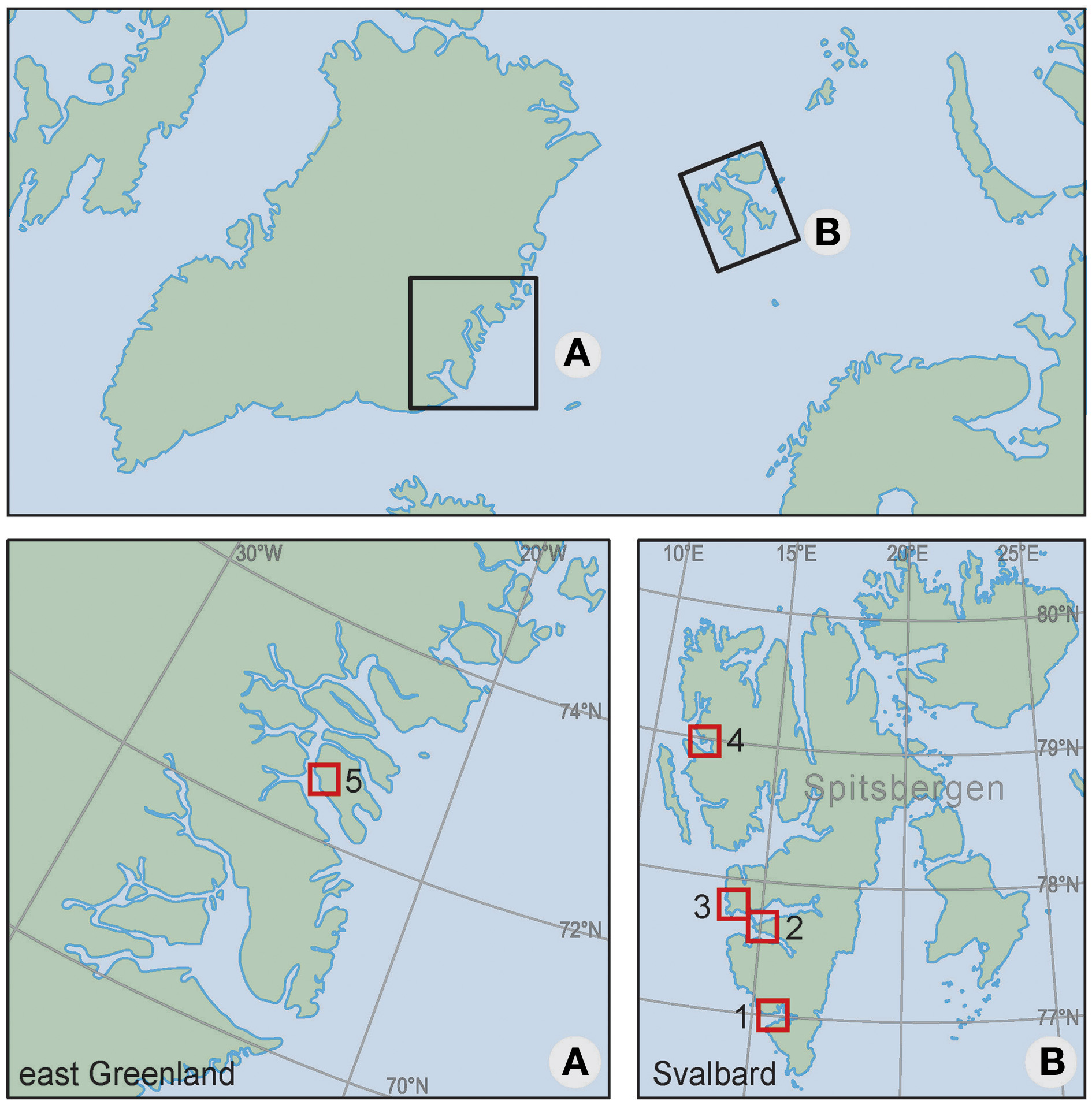
Location of the study areas: (1) Hornsund, (2) Bellsund, (3) Nordenskiöldkysten, (4) Kongsfjorden (1–4 on Spitsbergen, Svalbard) and (5) Traill Island (east Greenland). Supplement 1 provides detailed maps of each of the areas (Figure S1), a photographic impression of the study animals (Figure S2), and a map indicating the locations of sea ice extractions (Figure S3).
Biological data
Bear occurrence
Observations were initiated in the 1970s or 1980s and continued until recently (Table 1). We restricted our analyses to June–July on Spitsbergen and 15 June–15 August in Greenland, where coastal ice breaks up later. The time spent by the biologists in the field averaged over the years and locations was 44 days, but this varied among years and locations (Supplement 1, Table S1).
Table 1
| Nordenskiöldkysten | Kongsfjorden | Traill Island | Hornsund | Bellsund | |
|---|---|---|---|---|---|
| BIOLOGICAL DATA DURING SUMMER | |||||
| Bear presence (0 or 1) | 1977–2014 (25) | 1982–2014 (26) | 1988–2013 | 1972–2012 (30) | 1987–2010 |
| Bear days | 1977–2014 (25) | 1982–2014 (26) | 1988–2013 | ||
| Bear occurrence dates | 2004–2014 (9) | ||||
| Bear predation rates on birds | 2004–2014 (9) | ||||
| SEA ICE DATA | |||||
| Large-scale, at sea | 1979–2013 | 1979–2013 | 1979–2013 | 1979–2013 | 1979–2013 |
| Fine-scale, fjords/coastal | 2004–2014 (9) | 2008–2013 | 2008–2013 | 2008–2013 | 2008–2013 |
Summary of variables on biological data and sea ice acquired for the different locations.
Listed are the time periods for when data was available. For incomplete time series, numbers of years with data are given in parenthesis. Locations are listed in order of data extent. See Methods and Supplement 1 for a full description of the variables. For location of the ice data areas, see Supplement 1, Figures S3 and S4.
The five locations differed in the way the observations were recorded and which data were obtained (Table 1). From all the locations, we had information on “annual bear presence,” which is a binary variable denoting whether at least one bear had been observed or not. However, all polar bear observations were recorded in Nordenskiöldkysten, Kongsfjorden and Traill Island, which enabled us to calculate the number of bear days (e.g., two bear days may result from either two individuals being present for 1 day or one bear staying for 2 days).
Study localities may have differed in observation effort and detectability of bears. On Nordenskiöldkysten, daily observations were collected from several vantage points, including an observation tower that provided an extensive overview over the wide landscape. No observation towers were used at the other locations, but the nearby surroundings were scanned for polar bears as a measure of field safety. In all cases, the observations were done by experienced observers while doing fieldwork, typically on breeding birds. The spatial and temporal scale of the fieldwork was comparable between successive years within locations. We expect that any differences in detection probabilities were consistent over the years; thus, they should not affect any of the trends that we report.
Predation rates on birds
Polar bear predation rates on birds were obtained on Nordenskiöldkysten during the years 2004–2014 (all years except 2005–2006). On the island Diabasøya, which hosts the main bird colony of the area (Supplement 1, Figure S1), nests were monitored 6–16 h per day during the period that nesting geese or eiders were present. An observation tower on the mainland provided a good view of the island, which was 100 m off shore. Nests in view of the tower were mapped on high-resolution photographs of the island, which enabled us to assess the breeding history of individual birds. The species concerned were the barnacle goose, common eider and glaucous gull. As gull nests were not restricted to the island, all nests on rocky outcrops in adjacent bays visible from the observation tower were also recorded. Daily nest records included the following categories: settling (nest owners exhibit territorial behavior, and/or extensive nest building), incubating, nest predated (eggs or chicks taken by polar bear), nest successful (at least one egg hatching), and nest abandoned (nest owners abandon the nest territory, usually associated with removal of eggs by glaucous gulls). After the breeding season, all nests in the colony were mapped to determine the total number of nest sites for each species. Geese and eiders were only vulnerable to polar bear predation during the egg phase and during the first day after hatching, after which they departed from the colony; glaucous gulls had an extended period of predation as unfledged chicks stayed in the colony. Further details are given in Supplement 1 (Methods—Establishing predation rates).
We calculated two measures of predation intensity. (1) For each observation year (2004, 2007–2014), the closely monitored nests were used to calculate the proportion of predated nest attempts for species S as PNPs = (number of nest predationss)/(number of nest attemptss). We obtained this measure only for barnacle geese and glaucous gulls as the number of common eider nests in view was too low to assume an unbiased sample. (2) In 2009–2014, we determined the number of bear predations by non-stop records of polar bear behavior in the breeding colony. For these years, we obtained a measure of predation intensity, which is different from the previous measure by considering the number of (physical) nest sites rather than nest attempts: the predation ratio PRs = (number of predationss)/(number of nests locationss). This ratio may exceed 1.0 as a nest site may be used by several successive individuals, and thus potentially predated multiple times through the season. Further details are given in Supplement 1 (Methods—Establishing predation rates).
Sea ice data
Large-scale conditions at sea
To assess large-scale conditions of sea ice, we downloaded data on sea ice concentrations from the website of the University of Colorado [see Supplement 1 (Methods—Large-scale sea ice data) for link]. We extracted daily sea ice concentrations from the period 1979–2013 (Table 1) from four 25 × 25 km cells at each of six different locations: southwest Spitsbergen, west Spitsbergen, northwest Spitsbergen, north Spitsbergen, east Spitsbergen, and east Greenland (Supplement 1, Figure S3). Sea ice formation and disappearance follow a seasonal pattern with maximum concentrations in late winter (usually March) and minimum in autumn (usually September). We therefore structured each year of data as the period from 1 September to 31 August (Supplement 2, Figures S1–S6). By using a threshold sea ice concentration of 30% based on work in the Canadian Arctic (Iverson et al., 2014), we then calculated two sea ice indices. The length of the ice season was calculated as the number of days from the first day with sea ice >30% to the last day with sea ice >30%. The latter was also used to define the start of the ice-free season. When sea ice concentrations were never above 30% during a year, start of the ice-free season was set at 0 (1 January). We also calculated monthly mean sea ice concentrations for April, May, June and July.
Fine-scale conditions in fjords and coastal sites
As opposed to the large-scale data, fine-scale data on sea ice conditions were only available for the most recent years (Table 1). We downloaded ice maps for 2004 and 2007–2014 (April–July) from www.met.no/Hav_og_is to describe ice conditions in coastal areas in relation to the timing of bird breeding seasons. The maps are high-resolution sea ice concentration charts that are mainly based on weather-independent images from the Radarsat-2 satellite. The spatial resolution is sufficiently high (approximately 50 m) to analyze ice conditions in coastal areas, including fjords. Ice concentrations are classified by six categories. For the purpose of our study, we selected the three densest categories: dense drift ice (70–90% cover), very dense drift ice (90–100%), and fast ice. Ice data were analyzed for the coastal areas of Hornsund (southwest Spitsbergen), Bellsund/Van Mijenfjorden (west Spitsbergen), Kongsfjorden/Krossfjorden (northwest Spitsbergen), Woodfjorden (north Spitsbergen), Isbukta, Kvalvågen, Dunérbukta, and Sørporten (east Spitsbergen), and Traill Island (east Greenland) (see Supplement 1, Figure S4, for locations). Annual ice availability was characterized in two ways: (1) the ice data were aggregated by season to estimate the average amount of ice; and (2) the sea ice data were subjected to a non-linear (4-parameter) logistic regression (Sebaugh, 2011) to estimate the date of peak ice melt. To do this, we extracted the inflection point of the curves (the steepest part of the curve, midway the lower and upper asymptotes) in these regressions. In addition, the ice data were aggregated by 5-day periods to describe location-specific melt patterns.
Statistics
We used the program R (version 3.1.1, R Development Core Team, 2014) for statistical analyses. Linear models (lm) were used to test for temporal trends of the ice indices. Annual numbers of bear days were analyzed as a function of year and location by generalized linear models with a Poisson distribution. To account for overdispersion in the data, a quasi-GLM (glm, family = quasipoisson, link = log) was fitted to correct the standard errors (Zuur et al., 2009). Non-significant terms (P > 0.05) were dropped from the model to refit the model with the remaining terms.
Logistic regression (glm, family = binomial, link = logit) models were used to analyze annual polar bear presence (binary response, 0 = no bears observed, 1 = at least one bear observed) as a function of year, location and sea ice conditions. In order to choose one index among the closely related variables for large-scale sea ice conditions, we followed Iverson et al. (2014) and started with comparing models that related bear presence to location and the various sea ice indices (Supplement 4, Table S1). Length of the ice season was the ice index most strongly related to bear presence, and we used this variable in subsequent analyses. To test to what extent polar bear presence was associated with ice conditions, the first logistic regression model that we generated included length of ice season and location as explanatory variables. Length of the sea ice season and year were closely correlated (Supplement 3, Table S1), which precluded testing their effects simultaneously. Instead, we tested if polar bears responded to deviations in ice conditions from long-term trends by calculating detrended values of length of the ice season, using the residuals from the regression of length of the ice season with year. The second model, therefore, included year, location, and detrended length of ice season as explanatory variables.
To test for trends over the years in within-season patterns, we analyzed date of polar bear observations by a linear mixed model (lme) with year (centered at the mean) as covariate for the fixed variables. To account for within-year dependencies of observations, year was included as a random factor. Analysis was restricted to observations on Nordenskiöldkysten, the area with most detailed bear observations. A similar analysis was performed to test if bear occurrence was associated with variation in the date of peak ice melt as a measure of ice phenology (see above) in nearby fjords (Bellsund/Van Mijenfjord, indicated as W in Supplement 1, Figure S4).
The ratios of the numbers of predations and numbers of nests were compared among species by a Two-Way ANOVA including species and year as factors.
Results
Temporal trends in sea ice conditions
Large-scale sea ice conditions changed substantially during 1979–2013 in the study areas (Figure 2, Supplement 2, Figures S1–S6). Monthly sea ice cover dropped significantly over time for almost all months and all areas (Supplement 3, Table S1). In east Greenland, sea ice cover did not decrease over time in April or May but did so in June and July.
Figure 2
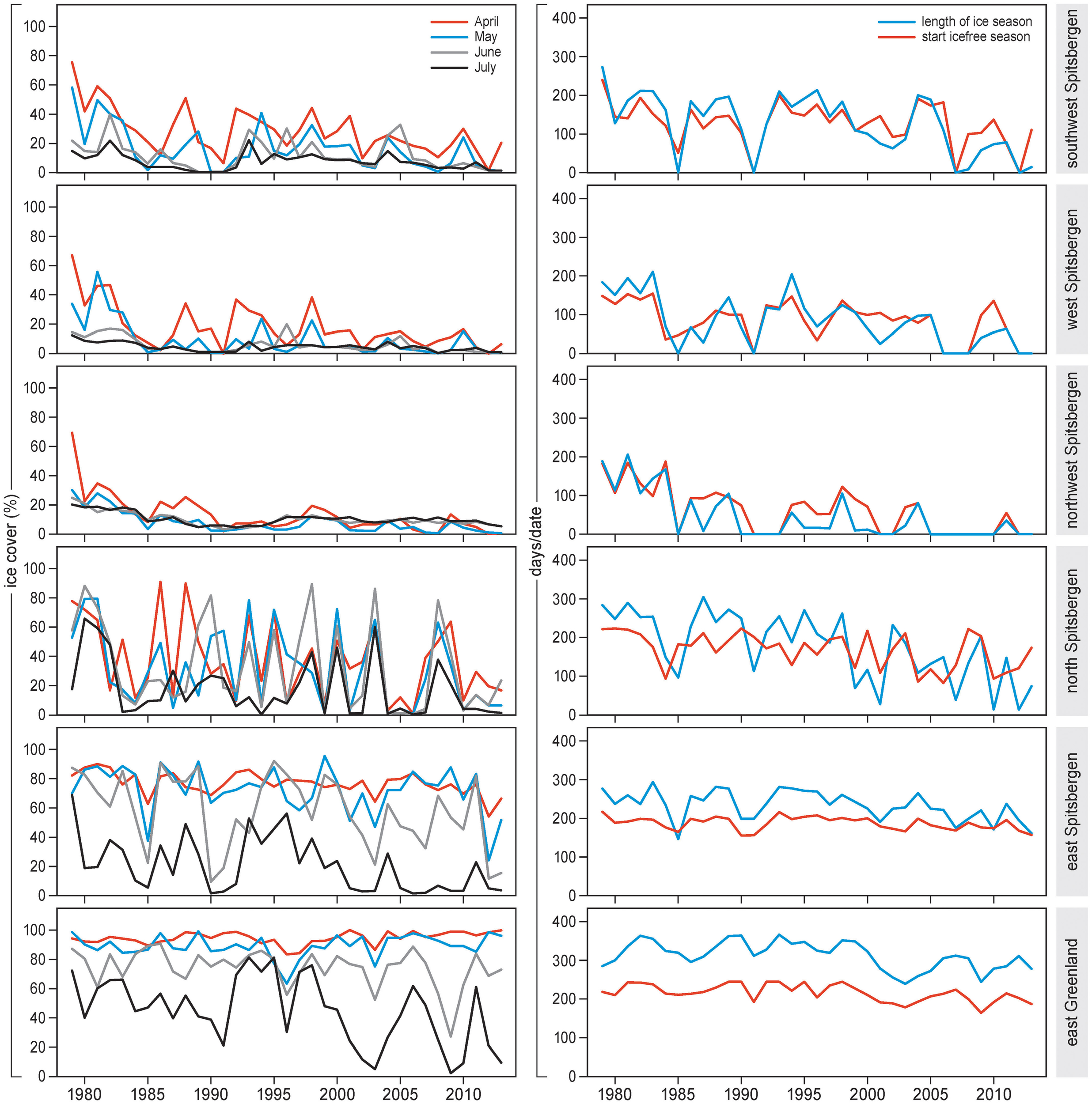
Sea ice concentration (% cover) (left), length of the ice season (days) and start of the ice-free season (days from 1 January) (right) for the period 1979–2013 in selected plots off Spitsbergen (E, SW, W, NW, N) and Greenland (E). Ice concentrations are given for April, May, June and July separately.
Length of sea ice season dropped over the years, on average by 3.5 days/year (Supplement 3, Table S1). The strongest decrease was in north Spitsbergen (–5.5) and southwest Spitsbergen (–4.2), and the weakest was in east Spitsbergen (–1.7) and east Greenland (–1.8).
Start of the ice-free season advanced by 2.0 days/year (see Supplement 3, Table S1). The strongest advancement was in northwest Spitsbergen (3.9 d earlier per year) and west Spitsbergen (2.3), and weakest in east Spitsbergen (0.6) and east Greenland (1.1).
Seasonal timing of the sea ice breakup varied among areas (Figure 3). Sea ice dropped below a concentration of 30% during the bird nesting season (=June to mid-July) in the open seas and coastal areas of north and east Spitsbergen and in east Greenland, and in the coastal area of west Spitsbergen. In other areas, sea ice breakup occurred before the nesting season.
Figure 3
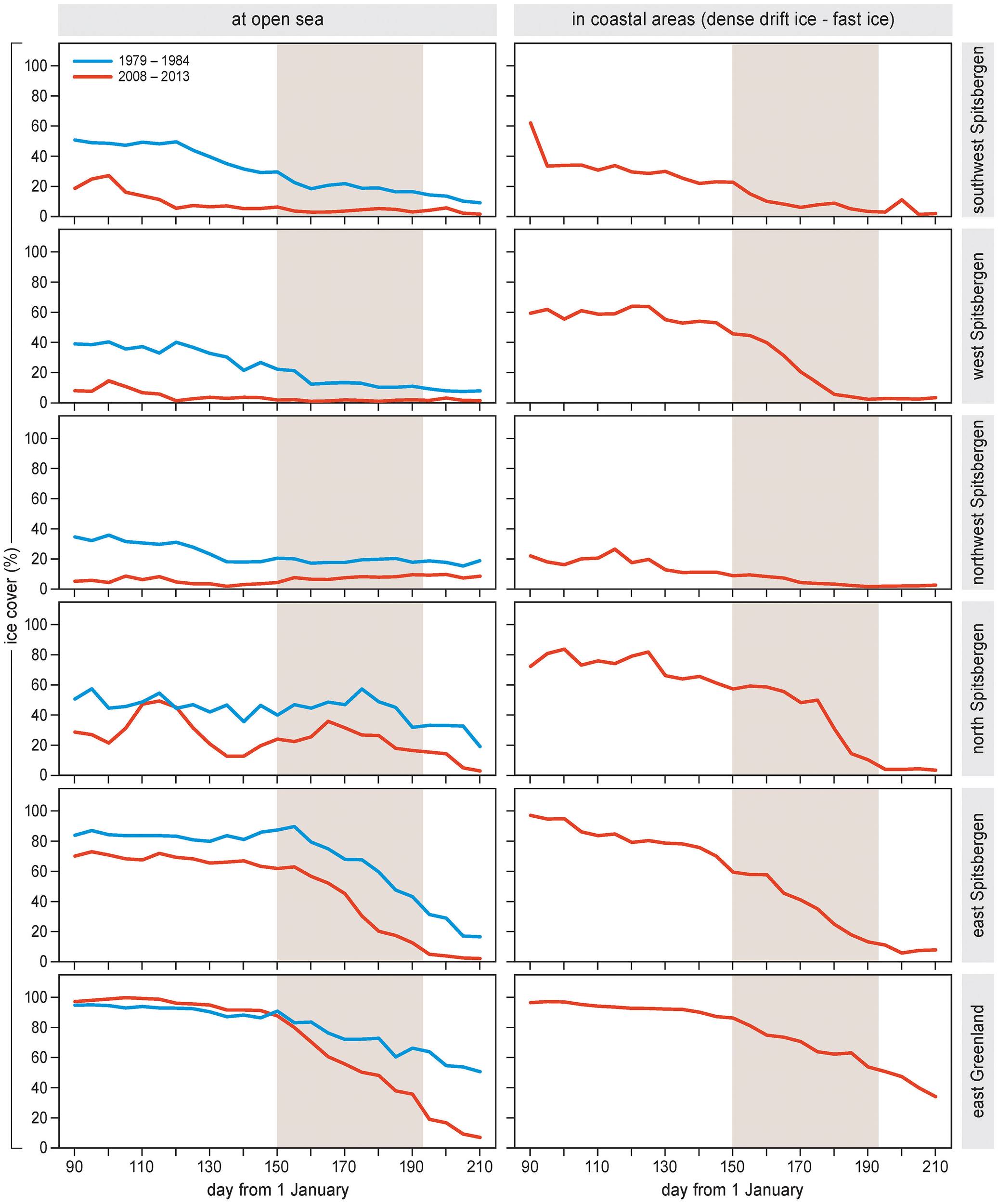
Left panels. Large-scale sea ice concentration (% cover) throughout the months of April–July in areas off the coast of Spitsbergen and Greenland. Separately indicated are averages for 1979–1984 and 2008–2013. Right panels. Local sea ice cover (area covered by fast ice or drift ice denser than 70%) throughout April–July in the fjords or coastal sites of Spitsbergen and Greenland (2008–2013). Shading indicates the period of sea bird nesting (approximately 1 June–15 July for all areas).
Bear observations
Annual number of polar bear days
Polar bears were only occasionally seen in the summers of 1980–2000 in the study areas on the west coast of Spitsbergen and east coast of Greenland. This changed drastically from the early 2000s onwards, when polar bears became regular summer visitors (Figure 4). Annual numbers of bear days increased exponentially, on average, by 15% each year (Table 2), a rate which did not differ between locations (the interaction term between year and location was not significant; Table 2). Bear numbers differed among locations, with those in Kongsfjorden and Traill Island being 31% and 61% of numbers on Nordenskiöldkysten (Table 2).
Figure 4
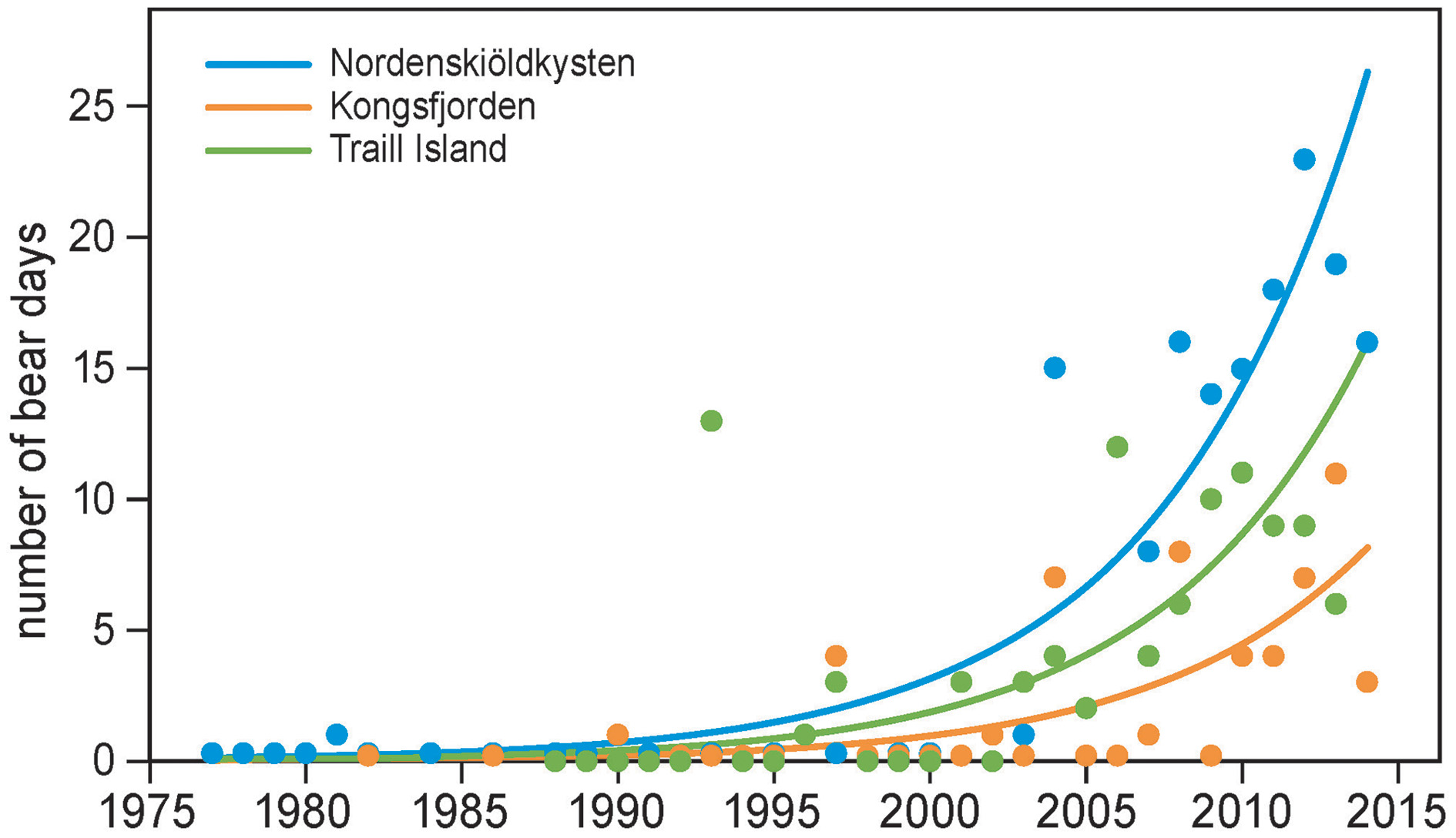
The number of polar bear days in summer as a function of year and location. Locations are Nordenskiöldkysten and Kongsfjorden on Spitsbergen, and Traill Island in Greenland. The observation periods in summer were June–July on Spitsbergen and 15 June–15 August on Traill Island. The curves are based on the model in Table 2.
Table 2
| Parameter | Estimate | SE | t-value | P |
|---|---|---|---|---|
| Intercept | 1.08 | 0.305 | 3.53 | <0.001 |
| Year | 0.15 | 0.025 | 5.92 | <0.001 |
| Location-NK | 0 | |||
| Location-KF | –1.16 | 0.386 | –3.01 | 0.004 |
| Location-TI | –0.50 | 0.310 | –1.61 | 0.112 |
Modeling the number of polar bear days in June and July by study location and year (1972–2014).
Locations are Nordenskiöldkysten (NK), Kongsfjorden (KF), and Traill Island (TI). Data were analyzed by Poisson GLM and corrected for overdispersion by a factor 5.30. Non-significant terms were dropped from the final model. Model results are on a log scale. Location Nordenskiöldkysten was used as the reference category.
Residual deviance 200.21, df = 69.
Terms not included in the final model: Year × Location.
Year was centered at the mean value and the intercept must be interpreted accordingly.
Annual probability of polar bear presence
The probability of polar bears being present was negatively related to length of the ice season (Figure 5A, Table 3A), indicating that more bears were encountered on land in years when the ice season was short. The intercept of the regression differed among locations, which underlines the additional importance of spatial variation. Polar bear presence was not related to detrended values of the length of the ice season (Table 3B). We, thus, did not find evidence that bears respond to year-to-year fluctuations in sea ice deviating from the long-term trend.
Figure 5
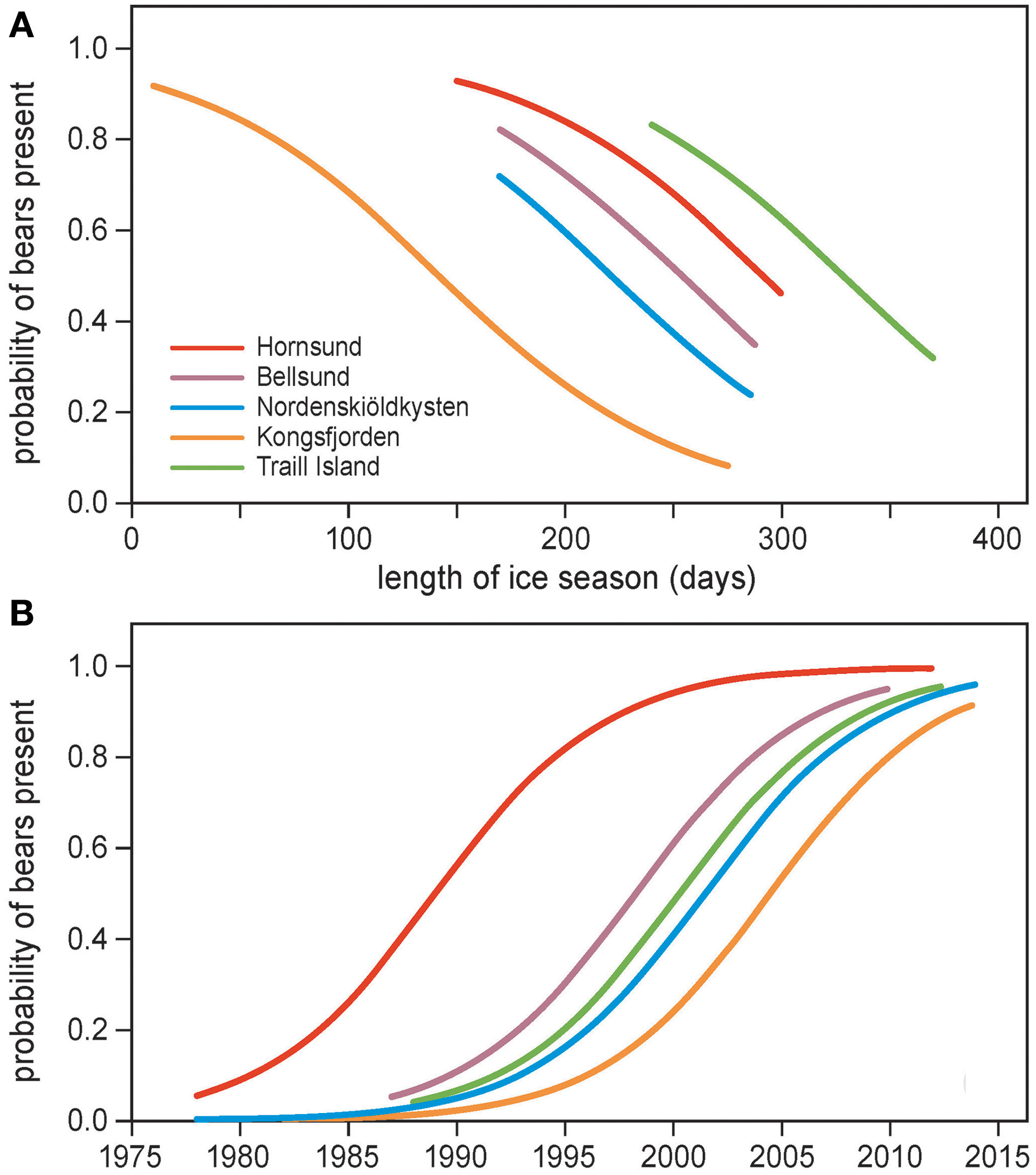
Probability of polar bear encounter in summer as a function of (A) length of the ice season and location, and (B) year and location. Locations are Hornsund, Bellsund, Nordenskiöldkysten and Kongsfjorden on Spitsbergen, and Traill Island in Greenland. The observation periods were limited to June–July on Spitsbergen and 15 June–15 August on Traill Island. The curves are based on the logistic regression models in Table 3.
Table 3
| Parameter | Estimate | SE | z-value | P | |
|---|---|---|---|---|---|
| (A) | |||||
| Intercept | 5.294 | 1.281 | 4.13 | <0.001 | |
| Length of ice season | −0.018 | 0.005 | −3.72 | <0.001 | |
| Location-HS | 0 | ||||
| Location-BS | −0.686 | 0.634 | −1.08 | 0.279 | |
| Location-NK | −1.264 | 0.649 | −1.95 | 0.051 | |
| Location-KF | −2.710 | 0.801 | −3.38 | 0.001 | |
| Location-TI | 0.671 | 0.708 | 0.95 | 0.343 | |
| Residual deviance = 144.5, df = 116. | |||||
| (B) | |||||
| Intercept | 2.413 | 0.796 | 3.03 | 0.002 | |
| Year | 0.253 | 0.045 | 5.65 | <0.001 | |
| Detrended length of ice season | −0.001 | 0.006 | −0.20 | 0.845 | |
| Location-HS | 0 | ||||
| Location-BS | −2.319 | 0.969 | −2.39 | 0.017 | |
| Location-NK | −3.136 | 1.116 | −2.81 | 0.005 | |
| Location-KF | −3.918 | 1.097 | −3.57 | <0.001 | |
| Location-TI | −2.843 | 1.008 | −2.82 | 0.005 | |
| Residual deviance = 94.6, df = 115. | |||||
Modeling the probability of polar bear presence in June and July during 1979–2013 by (A) length of the ice season and study location, and (B) year, detrended length of the ice season and study location.
Locations are Hornsund (HS), Bellsund (BS), Nordenskiöldkysten (NK), Kongsfjorden (KF), Traill Island (TI). Data were analyzed by GLM with a binomial distribution. Location Hornsund was the reference category in the analyses.
Associated with the ice effects, the probability of polar bears being present in summer increased steeply over the years (Figure 5B, Table 3B), with a similar rate of increase across locations (the interaction term between year and location was not significant). Notably, the significantly different intercepts for the locations (Table 3B) showed that polar bears appeared in each of the areas at different times (Figure 5B). Bears appeared first in Hornsund in the 1990s. Later, polar bears appeared in Bellsund, Traill Island, Nordenskiöldkysten, and Kongsfjorden (Figure 5B).
Within-season occurrence of polar bears
On Nordenskiöldkysten, polar bear occurrence shifted forward by 3.0 days/year (SE = 0.83; df = 7, t = −3.64, P = 0.008) during 2004–2014 (Figure 6A). With respect to the date of peak ice melt in adjacent fjords, the advancement was by 2.1 days/year (SE = 1.36; df = 7, t = −1.52, P = 0.17). In a year-by-year comparison, bear occurrence was not related to the date of peak ice melt (df = 7, t = 0.31, P = 0.76).
Figure 6
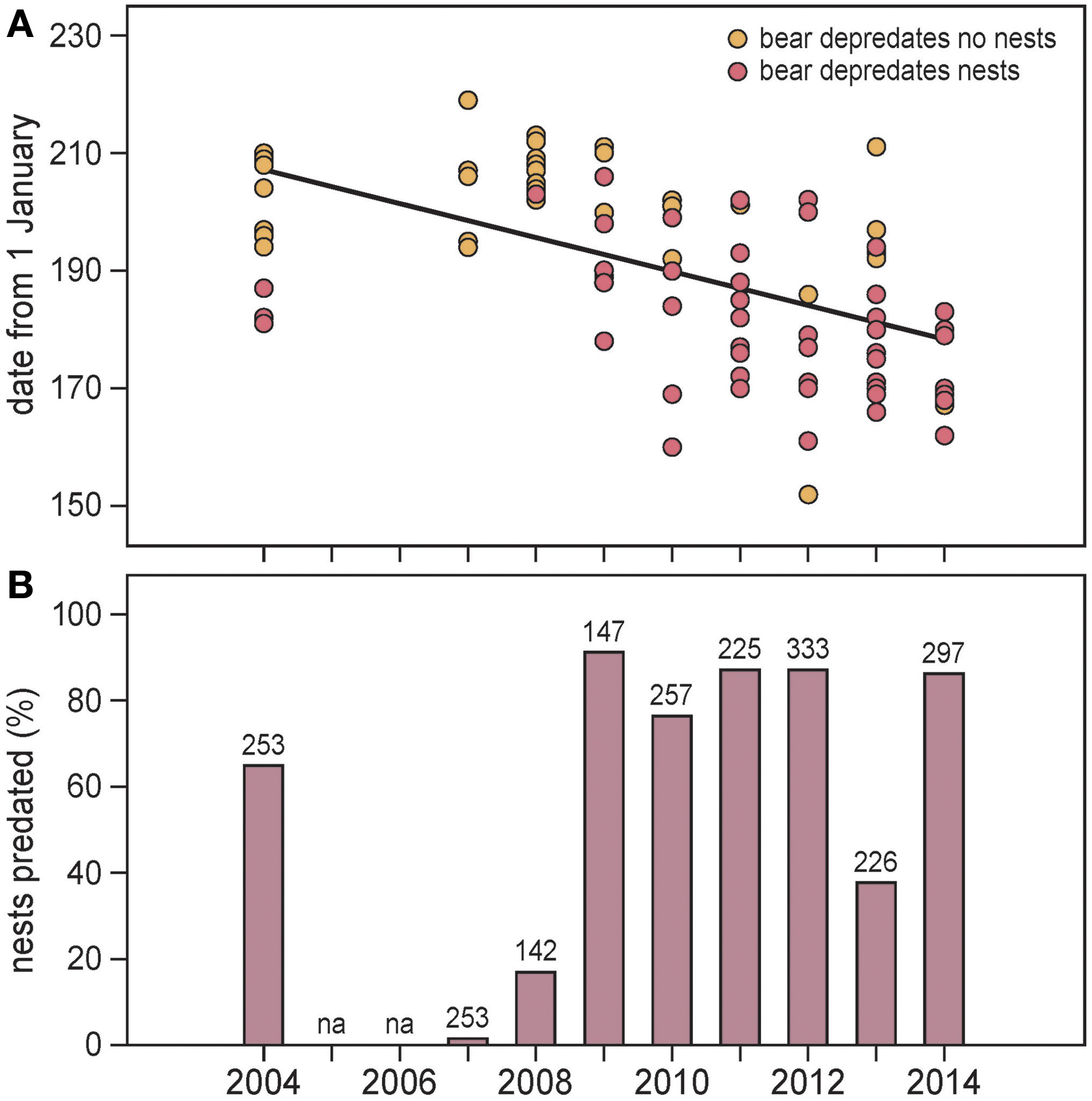
(A) Occurrence of polar bears on Nordenskiöldkysten (west Spitsbergen) in summers 2004–2014 (no observations in 2005 and 2006). 95% of all nest predations took place between 9 June and 9 July (dates 160–190). Separately indicated are polar bears that predated bird nests and those that were not observed eating eggs. The regression line (based on all records in a linear mixed model) is y = 190.1 – 3.01 × Year; df = 7, t = −3.64, P = 0.008; Year centered at the mean. (B) The proportions of barnacle goose nest attempts aborted by polar bear predation in the main study colony on Nordenskiöldkysten, 2004–2014. Numbers represent the sample size of intensively monitored barnacle goose nests included in the analyses. na means that no observations were available.
Polar bear predation in bird colonies
The number of barnacle goose nests in the main study colony on Nordenskiöldkysten fluctuated between 340 (2004) and 520 (2012), without a significant trend over the years (y = 413.3 + 10.37 × Year (centered at the mean), R2 = 0.241, P = 0.18). During this period, the number of successful nests varied between 0 and 85, with the percentage of nests predated by polar bears ranging from 0 to 91% (Figure 6B). Highest predation occurred in the years when the first bears appeared in June, well before bird eggs hatched (Pearson correlation coefficient between predation rate and arrival date of the first bear = –0.75, n = 9, P = 0.021). The average predation ratio of barnacle goose nests (the ratio of number of predations and number of nest sites) was 0.93 (SD 0.28) (Figure 7).
Figure 7
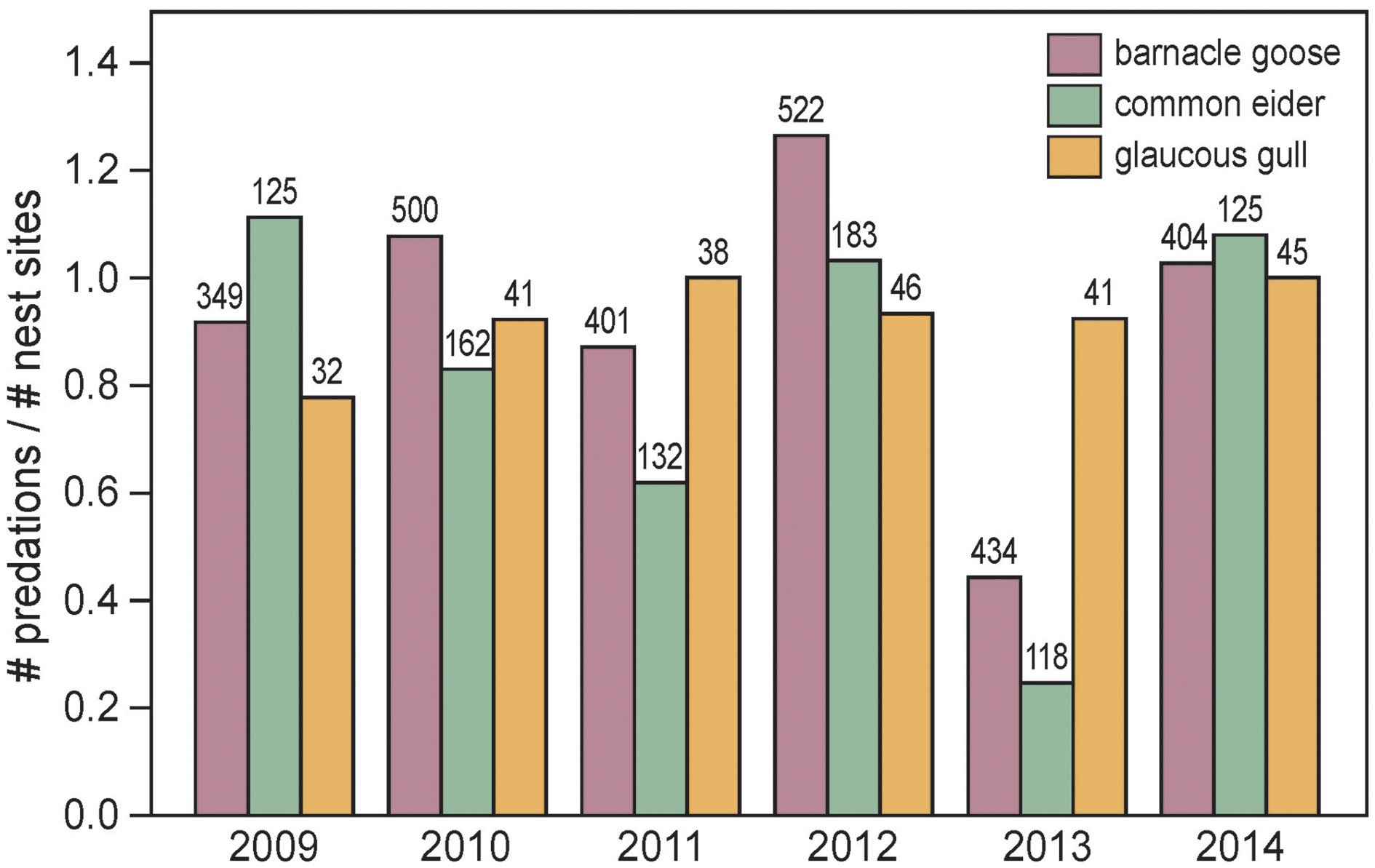
Intensity of polar bear predation on barnacle goose, common eider and glaucous gull nests in the main study colony on Nordenskiöldkysten during 2009–2014, as measured by the ratio of numbers of predations and numbers of nest sites. A nest site may be used by successive nest owners, and, therefore, nests were predated up to three times within a single season. Indicated are the numbers of nests in the colony. Ratios do not differ among species [F(2, 10) = 0.58, P = 0.57, while accounting for year effects].
The average number of common eider nests was 140 (SD 25.8) without a significant trend over the years 2009–2014 (y = 144.9 – 2.31 × Year (centered at the mean), R2 = 0.028, P = 0.75). The predation ratio was 0.82 (SD 0.33), which is similar to that in the barnacle goose (Figure 7).
The average number of glaucous gull nests in the main study colony and surroundings was 45 (SD = 2.8). Gulls hatched their eggs approximately 1 week earlier than barnacle geese and common eiders, but this did not make them less vulnerable to polar bear predation. Gull chicks, which were reared in the colony, were easy prey for bears. During 2009–2014, the predation ratio was 0.93 (SD 0.08).
Discussion
When a new predator enters the scene, the delicate balance between predators and their prey may be perturbed (Bell et al., 2010; Sih et al., 2010). Polar bears colonizing coastal areas, from which they have been absent for a long time, is an example of this situation. Below, we discuss why polar bears have recently extended their summer range toward the terrestrial habitats of west Spitsbergen and east Greenland. We propose that the increased number of bears on land predating bird nests is due to the lack of sea ice on which bears can hunt seals in summer. This process may have been strengthened in west Spitsbergen by a recent expansion into areas that had been occupied by bears year round before humans arrived but thereafter became devoid of bears in summer. Lønø (1970) refers to an expedition led by Jonas Poole in June–July in 1610 to western Spitsbergen, where 27 polar bears were killed and 5 cubs were captured alive (confirming the account of the Willem Barentsz' voyage in 1596; de Veer, 1598). Although polar bears must have been common in summer in the area then, Lønø argues based on later reports from Norwegian trappers that they were rarely encountered in summer on the west coast in the 19th century. He also argues that a larger local stock of bears may have been present year round before the collapse of the walrus (Odobenus rosmarus) population.
Polar bears on land
The west coast of Spitsbergen has little sea ice in most years, and summer observations of bears in the periods free of sea ice have been relatively rare at least in the later decades of the 20th century. The east coast of Greenland hosts a polar bear population that used to roam the extensive drift ice offshore, only infrequently showing up at coastal tundra sites (Born et al., 1997; Wiig et al., 2003; Laidre et al., 2013). The bears that we observed during the past years were either in transit or they were extensively feeding on eggs in bird colonies. Incidentally, polar bears have also been observed feeding on carcasses of muskox (Ovibos moschatus) (Greenland) or reindeer (Rangifer tarandus) (see also Gormezano and Rockwell, 2013; Stempniewicz et al., 2014). The presence of polar bears in areas that used to be devoid of bears, often feeding on items that are regarded as relatively unimportant energy sources compared to their staple food (Derocher et al., 2004; Stirling, 2011), fits well with records from other parts of the Arctic (Schliebe et al., 2008; Smith et al., 2010; Iverson et al., 2014). This suggests that polar bears more frequently occurring on land is a circumpolar phenomenon.
We hypothesized that occurrence of polar bears on land is related to sea ice conditions. In line with earlier findings (Serreze et al., 2007; Moe et al., 2009) we report a high rate of sea ice disappearing. The large spatial variation in sea ice abundance and rate of disappearance over the years corresponds to patterns found at the scale of the entire Arctic (Cavalieri and Parkinson, 2012; Meier et al., 2014). Average annual reduction in length of the ice season, a proxy of polar bear habitat availability, dropped by 3.5 days per year. Within our selection of study areas, the smallest reductions in ice season length occurred in east Spitsbergen and east Greenland. The area of east Spitsbergen is very important to the Barents Sea polar bear population (Løøo, 1970) due to the cold water and good access to sea ice much of the year. However, in summer and autumn, many of the bears that use this area in winter and spring will be further east and north on the pack ice (Mauritzen et al., 2001; Aars et al., 2009).
The number of bear days on land showed a consistent increase after polar bears colonized a specific area, although the data suggest a deceleration during the last couple of years of the study period. Given the high degree of seasonal site fidelity in polar bears (Mauritzen et al., 2001; Stirling et al., 2004; Zeyl et al., 2009; Lone et al., 2013), we suppose that a more or less permanent group of bears roamed the west coast of Spitsbergen in summer. If so, this group may be on the order of a few dozen individuals, based on the number of bears reported every year to the marine mammal sighting database (http://mms.data.npolar.no/) managed by the Norwegian Polar Institute. This number is also in accordance with a survey in August 2004 when 31 bears were counted along the coast of Spitsbergen (Aars et al., 2009), although numbers on land may have increased since then. We, thus, suspect that the number of polar bears along the west coast consists of a modest fraction of the Barents Sea population estimated at 2650 (Aars et al., 2009).
While some of the bears are likely local to the west coast of Spitsbergen year round, others may be visitors from the fjords in north Spitsbergen, and some are from east Spitsbergen. Visual observations and telemetry data show that polar bears move with drifting sea ice around the southern tip of Spitsbergen from east to west, and often eastwards into Hornsund where they may stay or cross the glacier back to Storfjorden on the east coast (Lydersen et al., 2002; unpublished data, Norwegian Polar Institute). This migration route linking the areas in east and west likely explains why Hornsund was the area where bears occurred earliest in the bird colonies.
Polar bear presence on land showed a strong negative relationship with the length of the ice season when accounting for variation among locations. This is on par with recent studies that showed a clear link between bear movements to terrestrial habitats and sea ice conditions (Cherry et al., 2013; Iverson et al., 2014; but see Stirling et al., 2004). That is not to say that sea ice conditions are the only factor determining polar bear numbers and distribution. We found that year of observation was a stronger predictor of bear presence than length of the ice season, even though both were closely correlated. This indicates that other factors contributed to the trend of increasing numbers of bears on land.
An important reason for an increasing number of bears on the west coast may be due to bears recolonizing areas where they used to be present before they were heavily hunted for about a hundred years, until 1973 (Lønø, 1970). Maternity denning in areas of west Spitsbergen has not been described in earlier literature despite several dedicated studies but has been recorded several times in recent years (Andersen et al., 2012). A recolonization may also be triggered by a likely increase in the population size at least until recently (Derocher, 2005). It seems, therefore, that an increasing number of bears have started using the west coast, searching land for food when sea ice is absent.
The population in east Greenland is still hunted, although not at the intensity as in Svalbard in former days (Rosing-Asvid, 2002), and the increased occurrence on land is, thus, not likely to be explained by recolonization. Instead, a shift in habitat use may be associated with a decrease in sea ice habitat; thus, bears are forced onto land for longer periods than in earlier years. It seems, therefore, that large-scale processes, such as climate and subsequent habitat changes, are a common driver but that population history plays an additional role.
Habitat choice depends on the quality of each of the habitats considered (Fretwell and Lucas, 1970), and any change in quality is reflected in allocation of time over the habitats (Mauritzen et al., 2003; Schreiber, 2012). Thus, the changes in polar bear numbers that we observed might be due to—or strengthened by—developments in the recently colonized coastal habitat. Use of bird eggs as a food source for polar bears was already described by Lønø (1970) in Svalbard. It has been suggested that eggs allow individual bears to have a high energy intake (Rockwell and Gormezano, 2009). During the past decades, this potential food source became more widespread in the coastal zones of Spitsbergen (Drent et al., 1998) and Greenland (Madsen et al., 2011). Concurrent with the growth of the Svalbard goose populations, goose numbers in coastal areas have increased by a factor of three (barnacle geese) or four (pink-footed geese) (Mehlum, 1998; Goosemap et al., 2013; Black et al., 2014). Other alternative food sources may also further increase the value of the summer habitat to these bears. Increasing numbers of harbor seals (Phoca vitulina) have been spreading along the coasts of Spitsbergen in later years (Lydersen et al., 2002). Walrus haul-out sites are also increasing, due to a fast growing population (Kovacs et al., 2014). In Greenland, muskox carcasses may also be a source of energy for polar bears on land. Due to the increasing frost–melt events in winter during the last decades (Gilg et al., 2009), the number of available carcasses has increased (BS, unpublished data).
Earlier arrival on land by polar bears
Polar bears occurred at the coastal site of Nordenskiöldkysten earlier each year, and we have obtained evidence that bears arrived progressively earlier relative to the date of peak sea ice melt. Due to this rapid advancement, polar bear occurrence has undergone a striking change relative to the breeding season of colonial birds, in particular geese and eider. Initially, bears used to show up from the end of incubation onwards, whereas in most recent years, the first bears arrived during the early nest phase. This suggests that bears have adapted seasonal movements after experiences with new food resources. The phenomenon of bears returning to familiar, profitable sites is in agreement with the observation that polar bears repeatedly visit areas where they have been before (Mauritzen et al., 2001; Zeyl et al., 2009; Andersen et al., 2012; Lone et al., 2013). Our observations additionally suggest that polar bears adapt quickly once they have discovered new hunting opportunities. Indeed, there is strong pressure for early exploitation: (1) to hit the peak of egg availability; (2) to arrive before competing bears; and (3) to arrive before birds abandon their nests en masse as may happen at any time during inclement weather, which is what occurred during the breeding season of 2013.
Polar bear predation on eggs
Polar bear predation was particularly severe on offshore island colonies of barnacle geese, common eiders and glaucous gulls. Some polar bears also preyed on aggregations of pink-footed goose nests further inland (Prop et al., 2013). Earlier arrival by bears in the colonies resulted in higher proportions of predated nests. Predation often led to nest territories being occupied by goose pairs that otherwise might have had difficulties in obtaining a nest site (Black et al., 2014), which increased the number of eggs available to bears later in the season.
Adult survival is a main determinant of population growth rates in long-lived species (Caswell, 2001). Reproductive costs are usually high due to impaired survival of successful pairs (Black et al., 2014). As such, a single predation event may not reduce the lifetime reproduction of an individual much because of the chances to reproduce another year. Breeding failure in Arctic-nesting birds is common, due to inclement weather or adverse snow conditions (Prop and de Vries, 1993; Yannic et al., 2014), and birds rely on the most favorable (and consequently most productive) years. However, polar bear predation will lower the reproductive outputs during favorable years. When possibilities to compensate an annual 10–15% mortality (Wilson et al., 2007; Allard et al., 2010; Black et al., 2014) are lost in any year, maintaining the local population at a constant level is unlikely. Surprisingly, the numbers of nests in the colony for either species remained relatively stable (Figure 7), which suggests that recruitment took place by birds arriving from elsewhere.
Polar bears act as top-predators in marine ecosystems or coastal ecosystems between sea and land. They have been documented to play a role in the population dynamics of light-bellied brent geese (Branta bernicla hrota) by visiting breeding locations in eastern Svalbard in years with dense sea ice in summer (Madsen et al., 1998). Our observations indicate that the ecological amplitude of polar bears in the Arctic ecosystem has broadened. We witnessed: (1) polar bears extending their range toward the coastal areas of west Spitsbergen and east Greenland; and (2) a persistent effect of polar bears on colonial breeding birds, including barnacle geese, common eiders and glaucous gulls, by high predation rates. We obtained evidence that these changes were driven by shifts in sea ice conditions, but we cannot rule out the possibility of a complex set of corroborating factors. Nevertheless, the number of polar bears exploiting the coastal habitat was small, and we suspect that there is little scope for further increase in numbers. This underlines the importance of the sea ice habitat to support current population levels of polar bears.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Statements
Acknowledgments
We sincerely thank the people involved in fieldwork and data collection, in particular: Krzysztof Adamski, Mateusz Barcikowski, Krzysztof Czajka, Lech Iliszko, Dariusz Jakubas, Jacek Jania, Liliana Keslinka, Dorota Kidawa, Leszek Kolondra, Anna Kowalska, Krzysztof Migała, Mateusz Moskalik, Andrzej Pachuta, Jerzy Pereyma, Witold Szczuciński, Jan Marcin Węsławski, Katarzyna Wojczulanis-Jakubas, Barbara Wojtasik, and Marek Zajączkowski in Hornsund; Louis Nielsen on Eholmen in Bellsund; Tom van Spanje, Roeland Bom, Oebele Dijk, Arjen Drost, Larry Griffin, Brian Morrell, Annette Scheepstra, and Ronald Visser on Nordenskiöldkysten; Francois Criscuolo, Olivier Chastel, Tore Nordstad, Anette Fenstad, Vegard Bråthen, Dagfinn Breivik Skomsø, Solveig Nilsen, Elise Skottene, Nora Bjørnlid, Heidi Kilen, Elise Biersma, Maarten Loonen, and Kjell Tore Hansen in Kongsfjorden. Many thanks to Martin Biuw for developing scripts for ice extraction, Dick Visser for preparing the figures, Esther Chang (eScribe) for editorial support, Christiane Hübner and Arthur Glaser (Mountain Hardwear-NL) for logistic help. We are grateful to the late Rudi Drent for guidance. We thank the Governor of Svalbard for permissions to do field work, Svalbard Environmental Protection Fund for funding the analyses, and Arctic Field Grant (Svalbard Science Forum, Research Council of Norway) and FRAM—High North Research Centre on Climate and the Environment for funding field work. We also thank the many private donators, Huib Kluijver Fund (NOU), Willem Barentsz Polar Institute (Groningen), and the INNO Fund of WWF-NL for supporting the work on Nordenskiöldkysten. For the study in Greenland, we sincerely acknowledge the Government of Greenland (Ministry of Domestic Affairs, Nature and Environment) in Nuuk for granting access and research permits to do field work in the National Park on a yearly basis. Likewise, generous support was received from University of Freiburg as well as from the GREA (Groupe de Recherche en Ecologie Arctique, http://grearctique.free.fr/). We also thank two anonymous reviewers and the Editor for their valuable comments on the manuscript.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Supplementary material
The Supplementary Material for this article can be found online at: http://www.frontiersin.org/journal/10.3389/fevo.2015.00033/abstract
References
1
Aars J. Marques T. A. Buckland S. T. Andersen M. Belikov S. Boltunov A. et al . (2009). Estimating the Barents Sea polar bear subpopulation size. Mar. Mamm. Sci. 25, 35–52. 10.1111/j.1748-7692.2008.00228.x
2
Allard K. A. Gilchrist H. G. Breton A. R. Gilbert C. D. Mallory M. L. (2010). Apparent survival of adult Thayer's and Glaucous Gulls nesting sympatrically in the Canadian high Arctic. Ardea98, 43–50. 10.5253/078.098.0106
3
Andersen M. Derocher A. E. Wiig Ø. Aars J. (2012). Polar bear (Ursus maritimus) maternity den distribution in Svalbard, Norway. Polar Biol. 35, 499–508. 10.1007/s00300-011-1094-y
4
Barbraud C. Weimerskirch H. (2006). Antarctic birds breed later in response to climate change. Proc. Natl. Acad. Sci. U.S.A. 103, 6248–6251. 10.1073/pnas.0510397103
5
Bell C. P. Baker S. W. Parkes N. G. Brooke M. D. Chamberlain D. E. (2010). The role of the Eurasian Sparrowhawk (Accipiter nisus) in the decline of the House Sparrow (Passer domesticus) in Britain. Auk127, 411–420. 10.1525/auk.2009.09108
6
Black J. M. Prop J. Larsson K. (2014). The Barnacle Goose. London: T & AD Poyser/Bloomsbury.
7
Boertmann D. (1994). An annotated checklist to the birds of Greenland. Medd. Grønland Biosci. 38, 1–63.
8
Born E. W. Wiig Ø. Thomassen J. (1997). Seasonal and annual movements of radio-collared polar bears (Ursus maritimus) in northeast Greenland. J. Mar. Syst. 10, 67–77. 10.1016/S0924-7963(96)00072-3
9
Caswell H. (2001). Matrix Population Models. Sunderland, MA: Sinauer Associates.
10
Cavalieri D. J. Parkinson C. L. (2012). Arctic sea ice variability and trends, 1979–2010. Cryosphere6, 881–889. 10.5194/tc-6-881-2012
11
Cherry S. G. Derocher A. E. Thiemann G. W. Lunn N. J. (2013). Migration phenology and seasonal fidelity of an Arctic marine predator in relation to sea ice dynamics. J. Anim. Ecol. 82, 912–921. 10.1111/1365-2656.12050
12
Derocher A. E. (2005). Population ecology of polar bears at Svalbard, Norway. Popul. Ecol. 47, 267–275. 10.1007/s10144-005-0231-2
13
Derocher A. E. Lunn N. J. Stirling I. (2004). Polar bears in a warming climate. Integr. Comp. Biol. 44, 163–176. 10.1093/icb/44.2.163
14
de Veer G. (1598). Waerachtighe Beschrijvinghe van drie Seylagien, ter werelt noyt soo vreemt ghehoort. Transl. by RoeperV.WildemanD. in 2011. Amsterdam: Athenaeum – Polak & Van Gennep.
15
Drent R. H. Black J. M. Loonen M. J. J. E. Prop J. (1998). Barnacle geese Branta leucopsis on Nordenskiöldkysten, western Spitsbergen - in thirty years from colonisation to saturation. Norsk Polarinstitutt Skrifter200, 105–114.
16
Drent R. H. Prop J. (2008). Barnacle goose Branta leucopsis survey on Nordenskiöldkysten, west Spitsbergen 1975–2007: breeding in relation to carrying capacity and predator impact. Circumpolar Stud. 4, 59–83.
17
Durner G. M. Douglas D. C. Nielson R. M. Amstrup S. C. McDonald T. L. Stirling I. et al . (2009). Predicting 21st-century polar bear habitat distribution from global climate models. Ecol. Monogr. 79, 25–58. 10.1890/07-2089.1
18
Fretwell S. D. Lucas H. L. (1970). On territorial behavior and other factors influencing habitat distribution in birds. Acta Biotheor. 19, 16–36. 10.1007/BF01601953
19
Gilg O. Kovacs K. M. Aars J. Fort J. Gauthier G. Gremillet D. et al . (2012). Climate change and the ecology and evolution of Arctic vertebrates. Year Ecol. Conserv. Biol. 1249, 166–190. 10.1111/j.1749-6632.2011.06412.x
20
Gilg O. Sittler B. Hanski I. (2009). Climate change and cyclic predator-prey population dynamics in the high Arctic. Glob. Change Biol. 15, 2634–2652. 10.1111/j.1365-2486.2009.01927.x
21
Gormezano L. J. Rockwell R. F. (2013). Dietary composition and spatial patterns of polar bear foraging on land in western Hudson Bay. BMC Ecol. 13:51. 10.1186/1472-6785-13-51
22
Goosemap. (2013). Site-Specific Information for Geese Occurring on Svalbard. Available online at: http://goosemap.nina.no/
23
Hanssen S. A. Moe B. Bardsen B. J. Hanssen F. Gabrielsen G. W. (2013). A natural antipredation experiment: predator control and reduced sea ice increases colony size in a long-lived duck. Ecol. Evol. 3, 3554–3564. 10.1002/ece3.735
24
Hisdal V. (1998). Svalbard - Nature and History. Oslo: Norwegian Polar Institute.
25
Iverson S. A. Gilchrist H. G. Smith P. A. Gaston A. J. Forbes M. R. (2014). Longer ice-free seasons increase the risk of nest depredation by polar bears for colonial breeding birds in the Canadian Arctic. Proc. R. Soc. B281:20133128. 10.1098/rspb.2013.3128
26
Kovacs K. M. Aars J. Lydersen C. (2014). Walruses recovering after 60+ years of protection in Svalbard, Norway. Polar Res. 33:26034. 10.3402/polar.v33.26034
27
Kovacs K. M. Lydersen C. (2006). Birds and Mammals of Svalbard. Tromsø: Norwegian Polar Institute.
28
Laidre K. L. Born E. W. Gurarie E. Wiig Ø. Dietz R. Stern H. (2013). Females roam while males patrol: divergence in breeding season movements of pack-ice polar bears (Ursus maritimus). Proc. Biol. Sci. 280:20122371. 10.1098/rspb.2012.2371
29
Liu J. P. Curry J. A. Hu Y. Y. (2004). Recent Arctic Sea Ice Variability: Connections to the Arctic Oscillation and the ENSO. Geophys. Res. Lett. 31:L09211. 10.1029/2004GL019858
30
Lone K. Aars J. Ims R. A. (2013). Site fidelity of Svalbard polar bears revealed by mark-recapture positions. Polar Biol. 36, 27–39. 10.1007/s00300-012-1235-y
31
Lønø O. (1970). The polar bear (Ursus maritimus Phipps) in the Svalbard area. Norsk Polarinstitutt Skrifter149, 1–103.
32
Lunn N. J. Stirling I. (1985). The significance of supplemental food to polar bears during the ice-free period of Hudson-Bay. Can. J. Zool. 63, 2291–2297. 10.1139/z85-340
33
Lydersen C. Krafft B. A. Andersen M. Kovacs K. M. (2002). Marine mammals in the Bellsund-Van Keulenfjord area. Norsk Polarinstitutt Rapport121, 1–36.
34
Madsen J. Bregnballe T. Frikke J. Kristensen J. B. (1998). Correlates of predator abundance with snow and ice conditions and their role in determining timing of nesting and breeding success in Svalbard light-bellied brent geese Branta bernicla hrota. Norsk Polarinstitutt Skrifter200, 221–234.
35
Madsen J. Jaspers C. Tamstorf M. Mortensen C. E. Riget F. (2011). Long-Term Effects of Grazing and Global Warming on the Composition and Carrying Capacity of Graminoid Marshes for Moulting Geese in East Greenland. Ambio40, 638–649. 10.1007/s13280-011-0170-4
36
Mauritzen M. Belikov S. E. Boltunov A. N. Derocher A. E. Hansen E. Ims R. A. et al . (2003). Functional responses in polar bear habitat selection. Oikos100, 112–124. 10.1034/j.1600-0706.2003.12056.x
37
Mauritzen M. Derocher A. E. Wiig Ø. (2001). Space-use strategies of female polar bears in a dynamic sea ice habitat. Can. J. Zool. 79, 1704–1713. 10.1139/z01-126
38
Mehlum F. (1998). Areas in Svalbard important for geese during the pre-breeding, breeding and post-breeding periods. Norsk Polarinstitutt Skrifter200, 41–55.
39
Meier W. N. Hovelsrud G. K. van Oort B. E. H. Key J. R. Kovacs K. M. Michel C. et al . (2014). Arctic sea ice in transformation: A review of recent observed changes and impacts on biology and human activity. Rev. Geophys. 52, 185–217. 10.1002/2013RG000431
40
Moe B. Stempniewicz L. Jakubas D. Angelier F. Chastel O. Dinessen F. et al . (2009). Climate change and phenological responses of two seabird species breeding in the high-Arctic. Mar. Ecol. Prog. Ser. 393, 235–246. 10.3354/meps08222
41
Post E. Forchhammer M. C. Bret-Harte M. S. Callaghan T. V. Christensen T. R. Elberling B. et al . (2009). Ecological dynamics across the Arctic Associated with recent climate change. Science325, 1355–1358. 10.1126/science.1173113
42
Prop J. de Vries J. (1993). Impact of snow and food conditions on the reproductive performance of barnacle geese Branta leucopsis. Ornis Scandinavica24, 110–121. 10.2307/3676360
43
Prop J. Oudman T. van Spanje T. M. Wolters E. H. (2013). Patterns of predation of pink-footed goose nests by polar bear. Ornis Norvegica36, 38–46. 10.15845/on.v36i0.439
44
Ramsay M. A. Hobson K. A. (1991). Polar bears make little use of terrestrial food webs - evidence from stable-carbon isotope analysis. Oecologia86, 598–600. 10.1007/BF00318328
45
R Development Core Team. (2014). R: A Language and Environment for Statistical Computing. Vienna: R Foundation for Statistical Computing.
46
Rockwell R. F. Gormezano L. J. (2009). The early bear gets the goose: climate change, polar bears and lesser snow geese in western Hudson Bay. Polar Biol. 32, 539–547. 10.1007/s00300-008-0548-3
47
Rosing-Asvid A. (2002). The Polar Bear Hunt in Greenland. Nuuk: Greenland Institute of Natural Resources. Technical Report No. 45.
48
Schliebe S. Rode K. D. Gleason J. S. Wilder J. Proffitt K. Evans T. J. et al . (2008). Effects of sea ice extent and food availability on spatial and temporal distribution of polar bears during the fall open-water period in the Southern Beaufort Sea. Polar Biol. 31, 999–1010. 10.1007/s00300-008-0439-7
49
Schreiber S. J. (2012). The evolution of patch selection in stochastic environments. Am. Nat. 180, 17–34. 10.1086/665655
50
Sebaugh J. L. (2011). Guidelines for accurate EC50/IC50 estimation. Pharm. Stat. 10, 128–134. 10.1002/pst.426
51
Serreze M. C. Holland M. M. Stroeve J. (2007). Perspectives on the Arctic's shrinking sea-ice cover. Science315, 1533–1536. 10.1126/science.1139426
52
Sih A. Bolnick D. I. Luttbeg B. Orrock J. L. Peacor S. D. Pintor L. M. et al . (2010). Predator-prey naivete, antipredator behavior, and the ecology of predator invasions. Oikos119, 610–621. 10.1111/j.1600-0706.2009.18039.x
53
Sistla S. A. Rastetter E. B. Schimel J. P. (2014). Responses of a tundra system to warming using SCAMPS: a stoichiometrically coupled, acclimating microbe-plant-soil model. Ecol. Monogr. 84, 151–170. 10.1890/12-2119.1
54
Smith P. A. Elliott K. H. Gaston A. J. Gilchrist H. G. (2010). Has early ice clearance increased predation on breeding birds by polar bears?Polar Biol. 33, 1149–1153. 10.1007/s00300-010-0791-2
55
Stempniewicz L. (1993). The polar bear Ursus maritimus feeding in a seabird colony in Frans Josef Land. Polar Res. 12, 33–36. 10.1111/j.1751-8369.1993.tb00420.x
56
Stempniewicz L. Kidawa D. Barcikowski M. Iliszko L. (2014). Unusual hunting and feeding behaviour of polar bears on Spitsbergen. Polar Record50, 216–219. 10.1017/S0032247413000053
57
Stirling I. (2011). Polar Bears: The Natural History of a Threatened Species. Markham, ON: Fitzhenry & Whiteside.
58
Stirling I. Derocher A. E. (2012). Effects of climate warming on polar bears: a review of the evidence. Glob. Change Biol. 18, 2694–2706. 10.1111/j.1365-2486.2012.02753.x
59
Stirling I. Lunn N. J. Iacozza J. (1999). Long-term trends in the population ecology of polar bears in western Hudson Bay in relation to climatic change. Arctic52, 294–306. 10.14430/arctic935
60
Stirling I. Lunn N. J. Iacozza J. Elliott C. Obbard M. (2004). Polar bear distribution and abundance on the Southwestern Hudson Bay Coast during open water season, in relation to population trends and annual ice patterns. Arctic57, 15–26. 10.14430/arctic479
61
Wiig Ø. Born E. W. Pedersen L. T. (2003). Movements of female polar bears (Ursus maritimus) in the East Greenland pack ice. Polar Biol. 26, 509–516. 10.1007/s00300-003-0513-0
62
Wilson H. M. Flint P. L. Moran C. L. Powell A. N. (2007). Survival of breeding Pacific common eiders on the Yukon-Kuskokwim delta, Alaska. J. Wildl. Manag. 71, 403–410. 10.2193/2005-776
63
Yannic G. Aebischer A. Sabard B. Gilg O. (2014). Complete breeding failures in ivory gull following unusual rainy storms in North Greenland. Polar Res. 33:22749. 10.3402/polar.v33.22749
64
Zeyl E. Aars J. Ehrich D. Wiig Ø. (2009). Families in space: relatedness in the Barents Sea population of polar bears (Ursus maritimus). Mol. Ecol. 18, 735–749. 10.1111/j.1365-294X.2008.04049.x
65
Zuur A. F. Ieno E. N. Walker N. J. Saveliev A. A. Smith G. M. (2009). Mixed Effects Models and Extensions in Ecology with R. New York, NY: Springer. 10.1007/978-0-387-87458-6
Summary
Keywords
cascading effects, colonial breeding birds, depredation, global warming, polar bear, seabirds, sea ice
Citation
Prop J, Aars J, Bårdsen B-J, Hanssen SA, Bech C, Bourgeon S, de Fouw J, Gabrielsen GW, Lang J, Noreen E, Oudman T, Sittler B, Stempniewicz L, Tombre I, Wolters E and Moe B (2015) Climate change and the increasing impact of polar bears on bird populations. Front. Ecol. Evol. 3:33. doi: 10.3389/fevo.2015.00033
Received
30 November 2014
Accepted
11 March 2015
Published
25 March 2015
Volume
3 - 2015
Edited by
Morten Frederiksen, Aarhus University, Denmark
Reviewed by
Andrew Edward Derocher, University of Alberta, Canada; Paul Allen Smith, Environment Canada, Canada
Copyright
© 2015 Prop, Aars, Bårdsen, Hanssen, Bech, Bourgeon, de Fouw, Gabrielsen, Lang, Noreen, Oudman, Sittler, Stempniewicz, Tombre, Wolters and Moe.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Jouke Prop, Arctic Centre, University of Groningen, PO Box 716, 9700 AS Groningen, Netherlands jouke.prop@wxs.nl
This article was submitted to Interdisciplinary Climate Studies, a section of the journal Frontiers in Ecology and Evolution
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.