- 1Department of Zoology, George S. Wise Faculty of Life Sciences, Tel Aviv University, Tel Aviv, Israel
- 2The Steinhardt Museum of Natural History, Israel National Center for Biodiversity Studies, Tel-Aviv University, Tel Aviv, Israel
The solitary ascidian Microcosmus exasperatus is globally distributed in tropical and sub-tropical waters. In the Mediterranean it is considered an invasive species introduced through the Suez Canal, with a restricted distribution in the eastern basin. In order to understand the potential of this species to establish sustainable communities at additional sites in the Mediterranean, we studied its reproduction cycle over a 2-year period in relation to seawater temperature and chlorophyll-a data. Although M. exasperatus reproduces seasonally, with significantly greater activity in summer and early fall, mature oocytes occur throughout the year, suggesting multiple spawning periods. We found that reproductive effort significantly correlated with seawater temperature, while chlorophyll-a showed a low and insignificant explanatory power. A combined regression model of both parameters yielded the highest explained variance, suggesting a synergic effect of these two factors. Such a prolonged reproductive activity period enables repeated recruitment events. In view of the anticipated rise in seawater temperature, we predict that this species will gradually expand its distribution further across the Mediterranean.
Introduction
Increasing in world shipping trade, as well as of recreational boating, has not only benefited the world's global economy but has also contributed to the ever-increasing rate of transport of invasive species around the world (Ruiz et al., 1997; Cohen and Carlton, 1998; Mack et al., 2000; Hewitt et al., 2004; Zenetos et al., 2012; Seebens et al., 2013). The consequences of bioinvasions are broad, from the fundamental alteration of physical and biotic ecosystem features, to their impact on human health (Mooney and Drake, 1986; Williamson, 1996; Ruiz et al., 1997; Mack et al., 2000; Shine et al., 2000; Çinar et al., 2011). Invasive organisms can cause significant economic losses (U.S Congress OTA, 1993; Galil and Zenetos, 2002; Streftaris and Zenetos, 2006), and are considered a major cause of loss of biodiversity, second only to habitat destruction (Wilcove et al., 1998; Mack et al., 2000; Clavero and García-Berthou, 2005). The Mediterranean Sea is known as a “hot spot” of invasive species (Coll et al., 2010; Zenetos et al., 2010; Seebens et al., 2013; Galil et al., 2014b), with the Suez Canal playing a major role as a corridor for alien introductions (Galil, 2006, 2012; Coll et al., 2010; Zenetos et al., 2012). Since its opening in 1869, hundreds of invasive species have crossed the Canal and established populations in the Mediterranean, mostly along the Levant Basin (Galil and Goren, 2014). With the recent expansion of the Suez Canal (Galil et al., 2014a) the rate of introduction of invasive species of tropical origin into the Mediterranean is anticipated to increase significantly.
Ascidians (Phylum: Chordata, Class: Ascidiacea) are well known for their ability to invade new regions and cause significant damage to the indigenous fauna and aquaculture facilities (Bullard et al., 2007; Bullard and Carman, 2009; Daigle and Herbinger, 2009; Davis and Davis, 2010). As sessile filter-feeding invertebrates, they are a dominant component of fouling communities world-wide (Shenkar and Swalla, 2011). Some of the ascidian species are aggressive competitors, with advantageous life-history characteristics that contribute to their ability to become successful invaders (Lambert, 2005a,b; Shenkar and Loya, 2008; Bullard and Carman, 2009). Invasive ascidians, among many other features, characterized by rapid growth rates, early sexual maturation, and a high reproductive effort manifested in prolonged reproduction seasons (Lambert, 2001, 2002). All of this, combined with their broad environmental tolerance (Naranjo et al., 1997; Lowe, 2002; Lambert, 2005a; Pineda et al., 2012), has enabled invasive ascidians to establish thriving populations worldwide. Out of 33 ascidian species with introduction records in the Mediterranean (Coll et al., 2010), 7 alien ascidians have been reported from the coast of Israel (Shenkar and Loya, 2009), and only few studies have focused on their reproductive biology (Shenkar and Loya, 2008). The solitary ascidian Microcosmus exasperatus Heller 1878 (order: Stolidobranchia, family: Pyuridae, Figure 1A) has a wide global distribution and it very common in tropical and sub-tropical waters (VanName, 1945; Tokioka, 1967; Millar, 1977; Monniot, 1983, 2002; Goodbody, 1984; Kott, 1985; Rocha et al., 2012). Its presence has been reported since the 1960s in different locations from the Mediterranean Sea (Pérès, 1958; Monniot, 1981; Turon, 1987), but a re-examination of the samples given by Turon et al. (2007) has re-classified most of them as the closely-related species M. squamiger. Currently, the distribution of M. exasperatus is restricted to the eastern Mediterranean (Turon et al., 2007; Izquierdo-Muñoz et al., 2009; Ramos-Esplá et al., 2013; Gewing et al., 2016). This distribution, in addition to previous reports from the Red Sea (Monniot, 2002), indicates that the most likely introduction route of this species into this area is through the Suez Canal (Por, 1978; Turon et al., 2007). M. exasperatus is currently widely distributed along the Israeli coast forming massive aggregations on both natural and artificial substrates (Shenkar and Loya, 2009). Such large populations of invasive ascidians may have severe consequences for the local benthic community, which can lead to modification of the entire habitat structure (Castilla et al., 2004; Blum et al., 2007; Bullard et al., 2007; Dijkstra et al., 2007), and even exert major economic effects (Robinson et al., 2005; Ramsay et al., 2008; Daigle and Herbinger, 2009; Rius et al., 2011). Although M. exasperatus is globally widespread, research on its ecology and biology is scarce, and the available publications are usually related to its geographic distribution and taxonomic morphology (Turon et al., 2007; Ramos-Esplá et al., 2013). The accumulation of knowledge on the biological aspects of this species, such as its reproductive cycle, is a crucial step in the prediction of its potential to spread to new habitats, specifically to the western Mediterranean. Moreover, uncovering these aspects can also contribute to the accumulating but still sparse knowledge on the biological and ecological features of introduced ascidians in general (Whitlatch et al., 1995; Lowe, 2002; Bourque et al., 2007; Rius et al., 2008, 2009; Pineda et al., 2013), and of Lessepsian ascidian introductions specifically (Shenkar and Loya, 2008; Gewing et al., 2014). The aim of the current study was to determine the reproductive cycle of M. exasperatus along the Mediterranean coast of Israel, and to understand its association with seawater temperature and of chlorophyll-a as indicator of primary production and potential food availability. We hypothesized that, as a widespread tropical and sub-tropical species, M. exasperatus would exhibit a high reproductive effort over a wide range of temperatures, potentially contributing to its ability to establish sustainable populations at additional sites in the Mediterranean Sea.

Figure 1. (A) Microcosmus exasperatus. Note the heavy epibiont cover on its tunic (B) A dissected specimen. The left gonad is partially enveloped by the gut; (GO) gonads, (DG) digestive gland, and (DT) digestive tract. Scale bars = 1 cm.
Materials and Methods
Study Site and Sampling
The study was conducted on a monthly basis from Oct 2012 to Sep 2014 in ‘Akko Bay’ (32°55′17.23″N, 35°04′22.08″E), northern Mediterranean coast of Israel. Each month 5–9 individuals (excluding October, November 2012, February, April, May, and July 2013 n = 3–4) were randomly collected from a submerged shipwreck at a depth of 0.5–1.5 m. The samples were immediately narcotized with menthol crystals and transported to the lab within 3 h. In the lab, the samples were fixed in 4% formaldehyde diluted with seawater, following Shenkar and Loya (2008), and stored at room temperature until used for histological and gonad index analyses.
Seawater temperature at the study site was recorded every 4 h using an HOBO® data logger (Onset, MA, ± 0.53°C), located at 1 m depth at the study site. In addition, from November 2013 monthly means of chlorophyll-a concentrations (mg/m3, 1 km resolution) at the study site were generated using MyOcean Products. Initially, attempts were made to investigate the recruitment patterns of M. exasperatus population at the study site using settlement plates. However, the high wave exposure at the study site resulted in repeated loss and breakage of the plates.
Gonad Index and Histological Analyses
The Gonad Index (GI, in percentage) was calculated as the ratio between gonad wet weight and whole individual wet weight including the tunic. Epibionts were carefully removed from the specimens before weighing, in order to avoid error. In addition, a cut was performed along the lateral side of the siphons to enable the draining of any water still present in the ascidian body.
The histological slides were prepared from the right gonad only, as the left gonad in M. exasperatus is wrapped around the gut and its separation is more complicated and may cause damage or loss of oocytes. The left gonad was therefore preserved in 70% ethanol for future use. The gonads for the histology examination were dehydrated, embedded in paraffin, sectioned using a microtome (Shandon MIR by S.C.I.L.) to 7 μm thickness, and stained in hematoxylin-eosin. Three sections were prepared from each gonad, with the gap between sections being at least 40 μm.
The histological slides were examined and photographed using a Nikon eclipse 90i light microscope equipped with Nikon Digital Sight DS-L. For each gonad, diameter of 100 oocytes was measured in oocytes sectioned through the nucleolus (Bingham, 1997). Diameter measurements were performed using NIS Elements D 3.2 software (Nikon, NY), synchronized with the light microscope and camera.
The oocytes were classified into 50 μm size classes according to the developmental stage (Becerro and Turon, 1992; Pineda et al., 2013): <50 μm (pre-vitellogenic), 50–100 μm and 100–150 μm (vitellogenic), and >150 μm (mature).
Maturity of the testis was also examined and classified according to three (subjective) developmental stages: I-Immature (only spermatogonia), II-Mature (spermatozoa), III-Mature and spawned. We defined the third stage as mature and spawned because spawned sperm follicles were always observed together with mature sperm follicles (although the opposite did not always occur, Figure 3E).
Statistics
All statistical analyses were performed using R software for statistical computing (R Core Team, 2013). As the data violated the assumptions of normal distribution, non-parametric or permutation analyses were carried out. lmPerm (Wheeler, 2010) and coin (Zeileis et al., 2008) packages were used to perform permutation tests (perm = “Exact”) for data analyses.
Permutation nested design ANOVA (oocyte diameter nested within individuals that were nested within the study months), followed by non-parametric multi-comparison (Kruskalmc) post-hoc test, were carried out to examine differences in oocyte diameters among the study months.
Non-parametric Kruskal-Wallis test was performed to detect differences in gonad index between the study months, followed by non-parametric multi-comparison (Kruskalmc) post-hoc test using Pgirmess packages (Giraudoux, 2012).
In addition, non-parametric ANCOVA, with gonad weight as the response variable and total body weight as the covariate, was used to ascertain the statistical validity of the differences in gonad index ratios between months (Grant and Tyler, 1983). These additional analyses of gonad indices were important in order to prevent erroneous conclusions deriving from applying variance tests on ratios (Packard and Boardman, 1988). Multiple non-parametric regression model was applied to estimate the relationship between chlorophyll-a and seawater temperature (independent variables) on the gonad index (dependent variable, separately).
Results
Histological Development of the Gonad
Microcosmus exasperatus is typically characterized by two gonads, one on either side of the body. The gonads are elongated and consist of 3–4 lobes (Figure 1B). Histological cross-sections revealed that the female follicles and oocytes are located in the core of the gonad, grouped around the ciliated oviduct (Figure 2A), while the male follicles, when present, are situated in the periphery of the gonad surrounding them (Figure 2B).
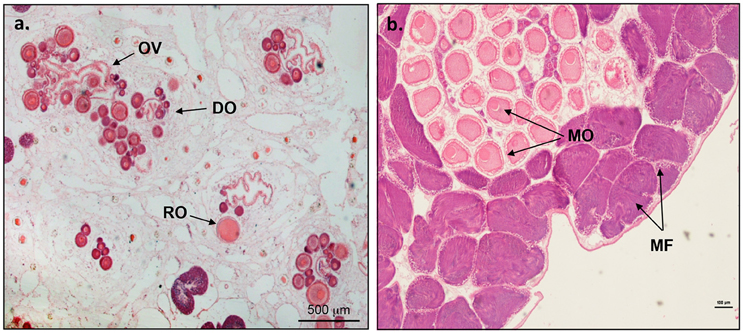
Figure 2. Microcosmus exasperatus. Close-up of gonad histology cross-sections taken under light microscope and stained with hematoxylin and eosin. (A) Developing oocytes (DO) grouped around the ciliated oviduct (OV) and residual oocytes (RO) from the previous reproductive cycle. Scale bar = 500 μm, and (B) Mature gonad close- up. The male follicles (MF) are situated in the periphery of the gonad, surrounding the oocytes (MO- mature oocytes). Scale bar = 100 μm.
Examination of gonad histology revealed an annual pattern (in regard to oocyte development), with spawning events occurring in the autumn months (September-December, Figure 3F), followed by a new reproductive cycle beginning in the winter months (January-February, Figure 3B). Development of the oocytes was continuous throughout the year, with the smallest ovaries in January and February, containing mainly previtellogenic oocytes. Mature ovaries (Figure 3D), which were larger in size and filled with vitellogenic and mature oocytes, appeared from March to December, but were most abundant between June and August. Between September and December the majority of individuals presented ovaries in spent condition (after spawning, Figure 3F), which also appeared in June and August 2013 and in June 2014.
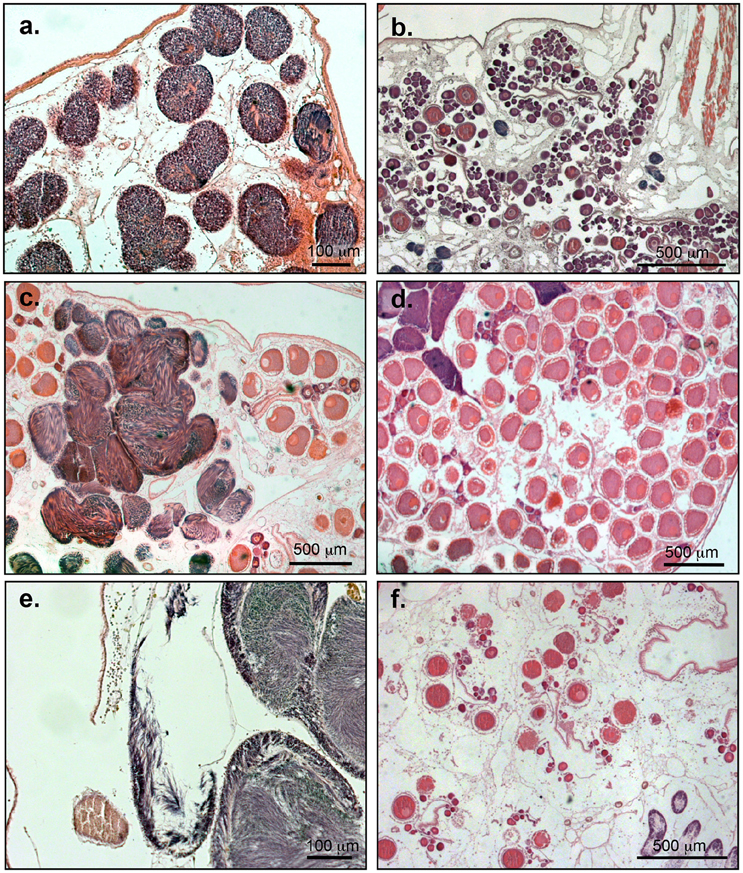
Figure 3. Microcosmus exasperatus. Light micrograph of the gonad developmental stages. (A) Immature sperm follicles stage I, scale bar = 100 μm. (B) Developing ova with various developmental stages of oocytes. (C) Male follicles at ripe stage II filled with spermatozoa. (D) Ripe ova with full-size oocytes. (E) Spawned sperm follicles, partially empty, stage III. (F) Ova after spawning, immature oocytes can be found beside residual oocytes.
Immature, mature, and spawned sperm follicles (Figures 3A,C,E) were present year round. However, as there was considerable variability in the sperm maturation within months and even within a single individual, we were unable to determine a clear annual pattern synchronized with oocyte maturation or with seawater temperature. Testis developmental stages throughout the study are given in Figure 4B.
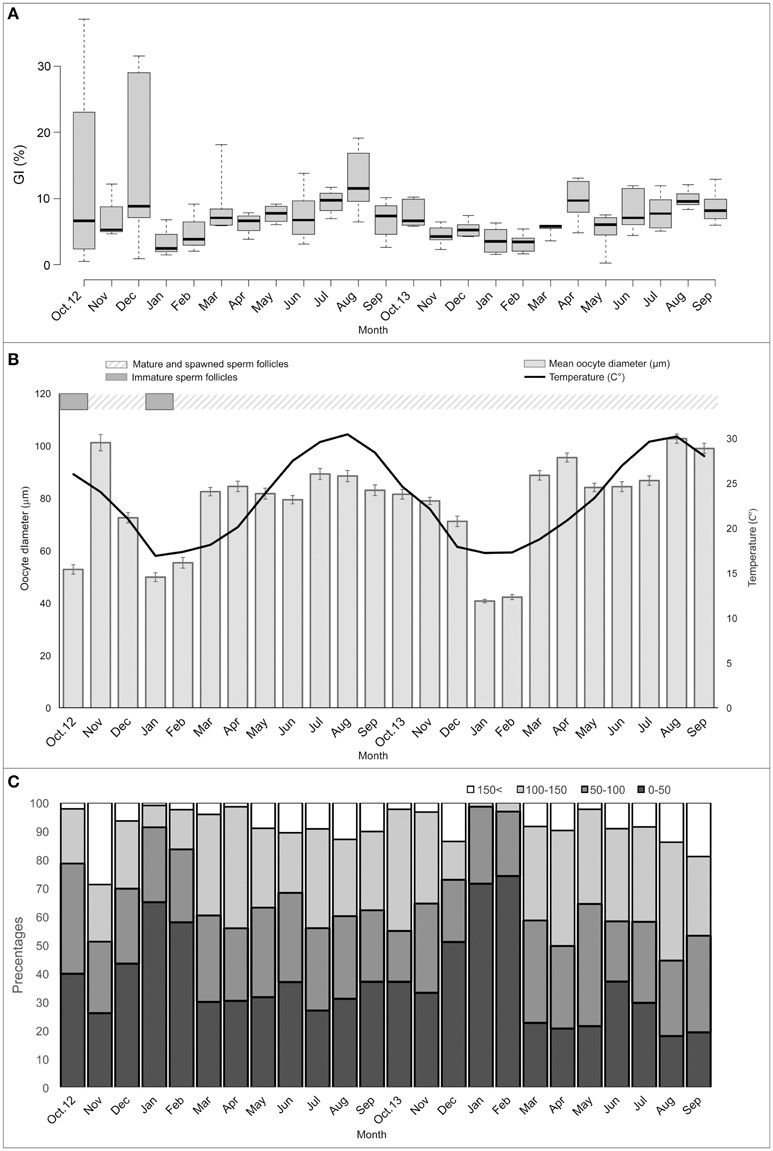
Figure 4. Reproductive cycle of Microcosmus exasperatus. (A) Monthly mean values of Gonad Index (percentages). (B) Mean oocyte diameter and sea surface temperature (1 m) as measured at the study site. Male follicle stages are indicated in the bar above. (C) Monthly relative percentages of each oocyte size-class: pre-vitellogenic <50 μm; 50–100 μm; vitellogenic 100–150 μm; mature >150 μm; (n = 3–9, +SEs).
Description of Reproductive Cycle by Quantity Measurements
The results of the quantity measurements confirmed the reproductive annual pattern obtained from the histological examinations. Both mean oocyte diameter and GI showed a seasonal pattern, with peak values in the summer followed by a large reduction when the spawning period began in the autumn (Figures 4A,B). However, findings from the first 3 months of the study (Oct-Dec of 2012) deviated from the reproductive pattern presented above, with the variance in those months also being considerably high, possibly due to the low sampling number of individuals collected during this period (n = 3–4).
The results of mean oocyte diameter, GI, and oocyte monthly size frequency distribution are given in Figure 4, together with sea surface temperature.
Mean oocyte diameter significantly differed over the 2 years of the study (permutation nested ANOVA, p < 0.01), with the lowest values (40.69–55.29 μm) in the winter months (January-February), increasing from March, and reaching the highest values (79.35–102.64 μm) in the summer months (June-August). In September the mean oocyte diameter decreased, reaching minimum values again in the winter. With the exception of inter-annual differences, the general pattern of mean oocyte diameter cycle was similar in the 2 consecutive years of the study. Post-hoc test (multiple comparison test after Kruskal-Wallis) revealed a significant difference (p < 0.05) between the winter months and the other months of the year. However, the winter months did not significantly differ from each other. GI values were also the lowest in the winter months and differed significantly from those in the summer (Kruskal–Wallis test, χ2 = 53.59, followed by post-hoc multiple comparison, p < 0.05), in which the values were the highest. Permutation ANCOVA (p < 0.001) confirmed that the differences in GI values throughout the year derived from the variance between the study months, taking into consideration the variance in the individual's body weight.
The multiple regression model, in which the GI was defined as the dependent variable and seawater temperature and chlorophyll-a as the independent variables, revealed high explanatory power (permutation linear regression, = 0.8979, p < 0.001). Chlorophyll-a showed a low and insignificant explanatory power ( = 0.250, p > 0.05), while seawater temperature was found to act as a good predictor of GI values ( = 0.7646, p < 0.005).
The oocyte size frequencies showed that the highest proportion of mature oocytes (>150 μm) appeared in August (2013 and 2014), followed by a large decrease from September to the winter months, while in the winter of 2014 no mature oocytes were present at all. Immature oocytes (<50 μm) were present year round, but were especially abundant in January-February of both 2013 and 2014.
The measured decline in oocyte quantity after the summer months indicates that a main spawning event, lasting from September to December, is followed by the beginning of a new gametogenesis cycle in the winter months, with the appearance of immature oocytes. Nonetheless, additional reductions in quantity throughout the year, such as between April and June of 2013 or April and May of 2014, suggest that an additional gamete release occurs during the year.
Discussion
The reproduction cycle of the invasive ascidian Microcosmus exasperatus along the Israeli Mediterranean coast is distinctly seasonal, but relatively prolonged in comparison to other invasive ascidians of tropical origin (Shenkar and Loya, 2008). All quantitative measurements indicate that the main reproduction period takes place at the end of the summer and during the autumn months (September to December). Immature oocytes were present year round, with a peak in the winter months. Histological observations confirmed that all individuals sampled during those months presented a spent or early gonad development condition, indicating that a spawning event had taken place prior to the winter months and that a new oogenesis cycle had begun. These results are consistent with the change in population density of M. exasperatus along the Israeli coast, as reported in Nagar and Shenkar (2016). The highest densities were recorded during the summer months (21.5 individuals/m2), implying a high reproductive effort, while during the winter months, the population density was the lowest (4.09 individuals/m2).
The presence of mature oocytes year round (excluding January and February of 2014) suggests that smaller spawning events may occur in the course of subsequent months following the main gamete release period. Moreover, the significant variance in oocyte diameter among individuals implies that different developmental stages can be found concomitantly in the same month: e.g., while some individuals were at a mature stage, others were redeveloping gonads. Multiple developmental stages of the gonads were also reported in populations of the invasive ascidian Styela clava in Ireland (Parker et al., 1999), New Zealand (Wong et al., 2011), and Canada (Davidson et al., 2005). Considering the tendency of M. exasperatus to form large aggregations (Shenkar and Loya, 2009; Nagar and Shenkar, 2016), it is possible that the asynchronous development of the gonads provides a mechanism for avoiding self-fertilization of the population, as suggested by Wong et al. (2011) for a Styela clava population in New Zealand.
As emphasized previously (Becerro and Turon, 1992; Bingham, 1997; Wong et al., 2011), the GI alone is not a sufficiently accurate parameter by which to evaluate reproduction activity. GI changes can also derive from the accumulation or reduction of nutritive tissues in the gonads. In M. exasperatus, the GI followed the same trend as that of the oocyte diameter variations (Figure 4), and as reflected in the histological observations. The compatibility of the GI with other reproduction parameters has also been reported from other species in the genus Microcosmus, such as in M. sabatieri (Becerro and Turon, 1992) and M. squamiger (Rius et al., 2009). Nonetheless, minor anomalies were found between the GI values and the mean oocyte diameter values, such as between March and April of 2013. In this case, the mean oocyte diameter increased, while the GI values decreased (Figures 4A,B). Such variation can be explained by the distinct distribution of oocyte size-classes within those months (Figure 4C), which indicates on the timing of spawning events. The percentage of the largest oocyte size-class (>150 μm) decreased between March to April (4.094%, n = 6 and 1.405%, n = 4, respectively), possibly due to a spawning event. Release of the largest oocytes may result in a decrease in GI values. Between March and April the total percentage of oocytes measuring above 100 μm increased (39.52 and 44.02%, respectively), which was also reflected in an increase in mean oocyte diameter but not in the total gonad weight, which affects the gonad index. However, both parameters showed the lowest values in the winter months, which then increased in the course of the spring, reaching their highest values in the summer.
In contrast to the prolonged reproductive cycle of M. exasperatus in the eastern Mediterranean, in the South China Sea the breeding season of this species lasts only 2 months, from February to April (Cole and Vorontsova, 1998). This is not surprising, given that ascidians are known to differ in their life-history traits among subpopulations in different seas (Millar, 1952; Davis, 1989; Rocha et al., 1999; Shenkar and Loya, 2008), or even among subpopulations existing in the same area (Rinkevich et al., 1993; Durante and Sebens, 1994). For example, Herdmania momus, a Lessepsian invader in the Levant basin, reproduces in its native range (Red Sea) continuously throughout the year; while in the Mediterranean it has a markedly short seasonal reproduction, taking place twice a year, when the sea temperature matches that of its native range (Shenkar and Loya, 2008). Furthermore, subpopulations of M. sabatieri demonstrate a different time lag in their reproduction periods in the Mediterranean. While M. sabatieri populations from the eastern basin reproduce mainly in the winter months (Vafidis et al., 2008), those from the western basin reproduce in the autumn (Becerro and Turon, 1992).
Numerous studies have suggested that seawater temperature is the main factor (among many other factors) controlling sexual reproduction in ascidians (Millar, 1971; Berrill, 1975; Turon, 1988). Indeed, the results of this study have revealed that seawater temperature acts as a good predictor of GI values ( = 0.7646, p < 0.005) and, therefore, that seawater temperature may play a main role in controlling the reproduction cycle also in the case of M. exasperatus.
Food availability too is known to have an important influence in regulating reproduction cycles in ascidians (Yamaguchi, 1975; Bingham, 1997; Wong et al., 2011), and low concentrations of food availability can even lead to gamete absorption and restrict reproductive activity (Goodbody, 1961; Berrill, 1975). Nonetheless, it was found in Potter Cove, Antarctica, that the solitary ascidian Cnemidocarpa verrucosa reproduces in the winter months, which are considered as the months with the lowest energy availability of the year (Sahade et al., 2004). In the case of M. exasperatus, chlorophyll-a alone (as an estimate of primary production, and food availability) had low and insignificant explanatory power when considering GI values ( = 0.250, p > 0.05, against GI values). Combining the two predictors together, temperature and chlorophyll-a, produced the best regression model with the highest explained variance (permutation multiple regression = 0.8979, p < 0.001). This indicates that chlorophyll-a alone is not a good predictor of the reproductive activity of M. exasperatus, but that it may have a synergic effect on the reproductive activity if temperature is also taken into account. As Sahade et al. (2004) noted, it is possible that high chlorophyll-a levels reflect favorable production conditions, which may increase the biomass of filter-feeding organisms, such as M. exasperatus. Current results show that the reproduction cycle of M. exasperatus is almost continuous throughout the year, excluding the winter months (Figure 4). Thus, this observed interruption of breeding could be a result of the combined low values of temperature and chlorophyll-a during this season. However, climate change, one of the most serious global environmental threats (Walther et al., 2002), may in the future alter the current reproduction pattern of the invasive ascidian M. exasperatus in the eastern Mediterranean. Sea surface temperature in this area is predicted to rise by about 3°C over the next century (Somot et al., 2006), favoring a continuous reproduction of this species and increasing its potential to spread into new areas in the Mediterranean. The consequences of global warming on the establishment of invasive species, and of invasive ascidians in particular, has been examined previously (Dukes and Mooney, 1999; Hobbs, 2000; Stachowicz et al., 2002; Occhipinti-Ambrogi, 2007), and has indicated that ocean warming is favoring the invasive species over the local fauna, and will facilitate the establishment and distribution of the former, especially those originating from warmer seas.
In the Mediterranean, benthic invertebrates such as sponges, mollusks, and cnidarians usually present a seasonal reproductive cycle. This is also true regarding colonial and solitary ascidians (Coma et al., 2000). Intensive reproduction pulses against a background of continuous gamete release has been reported also in other populations of ascidians from temperate seas, such as Ascidia mentula along the Swedish west coast (Svane and Lundälv, 1981) and Metandrocarpa taylori in California (Haven, 1971). In the Mediterranean, a seasonal reproductive pattern was described in Styela plicata (Pineda et al., 2013), M. squamiger (Rius et al., 2009) from the western basin and in M. savignyi from the eastern basin (Panagiotou et al., 2008).
A prolonged reproduction cycle, particularly in the summer months, constitutes a significant advantage to invasive species when invading new habitats. The relatively low food availability (Coma et al., 2000) and the high densities of algae competing for substrate on which to settle (Ballesteros, 1989), make the summer season unfavorable for most sessile invertebrates to reproduce in temperate seas. As it is generally assumed that tropical ascidians reproduce year-round (Goodbody, 1961; Shenkar and Loya, 2008), it seems that invasive populations of M. exasperatus exhibit great plasticity and adaptability in reproduction patterns. Such an ability to change reproductive period is known from other Lesspesian invaders such as fish (Golani, 1990), mollusks (Atad, 2005), and polychaetes (Arias et al., 2013).
The prolonged reproduction pattern that M. exasperatus exhibits in the eastern Mediterranean can provide this invasive species with a competitive advantage over the native species. In addition, continuous reproduction may allow invasive species to exploit temporal windows of unfavorable conditions (Pineda et al., 2013), either for settlement or for the spread of larvae. The high densities that M. exasperatus has established along the Israeli coast on artificial as well as on natural substrates (Shenkar and Loya, 2009; Nagar and Shenkar, 2016), together with its high reproductive abilities, suggest that it may have severe effects on the local fauna and possibly act as an ecosystem engineer (Castilla et al., 2004). The increasing rate of Lessepsian introductions of ascidians (Shenkar and Loya, 2008; Izquierdo-Muñoz et al., 2009; Çinar et al., 2011), as well as the accumulating evidence of the negative impact of invasive ascidians on the local fauna (Cohen et al., 2005), emphasizes the need to acquire greater knowledge on the biology and reproductive traits of these organisms, in order to better understand the potential of these invasive species to spread to new locations, like the western Mediterranean, especially in light of the anticipated rise in seawater temperature and increased anthropogenic activity, and thus also to develop the necessary and efficient management tools.
Author Contributions
Both authors, NS and LR, designed, performed, and analyzed the research. LR wrote the manuscript under the supervision of NS.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
We thank the NS team for technical support in the field, I. Brickner for histological sections, and O. Bronstein, O. Givan, and M. Novosolov for assistance in statistical analysis. We are grateful to the Akko Maritime Officers School for their support, and to N. Paz and V. Wexler for editorial assistance. Research funding was provided by The FP7 Marie Curie CIG grant number 321547 to NS.
References
Arias, A., Richter, A., Anadón, N., and Glasby, C. J. (2013). Revealing polychaetes invasion patterns: identification, reproduction and potential risks of the Korean ragworm, Perinereis linea (Treadwell), in the Western Mediterranean. Estuar. Coast. Shelf. Sci. 131, 117–128. doi: 10.1016/j.ecss.2013.08.017
Atad, I. (2005). The Limpet Cellana Rota As a Lessepsian Migrant Model: Arrival, Establishment and Competition with the Mediterranean Limpet, Patella Caerulea. MSc thesis dissertation, Tel Aviv University.
Ballesteros, E. (1989). Production of seaweeds in Northwestern Mediterranean marine communities: its relation with environmental factors. Sci. Mar. 53, 357–364.
Becerro, M. A., and Turon, X. (1992). Reproductive cycles of the ascidians Microcosmus sabatieri and Halocynthia papillosa in the Northwestern Mediterranean. Mar. Ecol. 133, 63–373. doi: 10.1111/j.1439-0485.1992.tb00360.x
Berrill, N. J. (1975). “Chordata: tunicata,” in Reproduction of Marine Invertebrates, Vol. II. Entoprocts and Lesser Coelomates, eds A. C. Giese and J. S Pearse (New York, NY: Academic Press), 241–282.
Bingham, B. L. (1997). Light cycles and gametogenesis in three temperate ascidian species. Invertebr. Biol. 166, 61–70. doi: 10.2307/3226925
Blum, J. C., Chang, A. L., Liljesthröm, M., Schenk, M. E., Steinberg, M. K., and Ruiz, G. M. (2007). The non-native solitary ascidian Ciona intestinalis (L.) depresses species richness. J. Exp. Mar. Biol. Ecol. 342, 5–14. doi: 10.1016/j.jembe.2006.10.010
Bourque, D., Davidson, J., MacNair, N. G., Arsenault, G., LeBlanc, A. R., Landry, T., et al. (2007). Reproduction and early life history of an invasive ascidian Styela clava Herdman in Prince Edward Island, Canada. J. Exp. Mar. Biol. Ecol. 342, 78–84. doi: 10.1016/j.jembe.2006.10.017
Bullard, S. G., and Carman, M. R. (2009). “Current trends in invasive ascidian research,” in Invasive Species: Detection, Impact and Control, eds C. P. Wilcox and R. B.Turpin (New York, NY: Nova Science Publishers), 57–79.
Bullard, S. G., Lambert, G., Carman, M. R., Byrnes, J., Whitlatch, R. B., Ruiz, G., et al. (2007). The colonial ascidian Didemnum sp. A: current distribution, basic biology and potential threat to marine communities of the northeast and west coasts of North America. J. Exp. Mar. Biol. Ecol. 342, 99–108. doi: 10.1016/j.jembe.2006.10.020
Castilla, J. C., Lagos, N. A., and Cerda, M. (2004). Marine ecosystem engineering by the alien ascidian Pyura praeputialis on a mid-intertidal rocky shore. Mar. Ecol. Prog. Ser. 268, 119–130 doi: 10.3354/meps268119
Çinar, M. E., Bilecenoglu, M., Öztürk, B., Katağan, T., Yokeş, M. B., Aysel, V., et al. (2011). An updated review of alien species on the coasts of Turkey. Medit. Mar. Sci. 12, 257–315. doi: 10.12681/mms.34
Clavero, M., and García-Berthou, E. (2005). Invasive species are a leading cause of animal extinctions. Trends. Ecol. Evol. 20, 110. doi: 10.1016/j.tree.2005.01.003
Cohen, A. N., and Carlton, J. T. (1998). Accelerating invasion rate in a highly invaded estuary. Science 279, 555–558. doi: 10.1126/science.279.5350.555
Cohen, A., Harris, L. H., Bingham, B. L., Carlton, J. T., Chapman, J. W., Lambert, C. C., et al. (2005). Rapid assessment survey for exotic organisms in southern California bays and harbors, and abundance in port and non-port areas. Biol. Invasions 7, 995–1002. doi: 10.1007/s10530-004-3121-1
Cole, L., and Vorontsova, M. (1998). Species of pyuridae (Ascidiacea) from South Vietnam. B. Mar. Sci. Lawrence. 62, 1–6.
Coll, M., Piroddi, C., Steenbeek, J., Kaschner, K., Lasram, F. B. R., Aguzzi, J., et al. (2010). The biodiversity of the Mediterranean Sea: estimates, patterns, and threats. PLoS ONE 5:e11842. doi: 10.1371/journal.pone.0011842
Coma, R., Ribes, M., Gili, J. M., and Zabala, M. (2000). Seasonality in coastal benthic ecosystems. Trends. Ecol. Evol. 15, 448–453. doi: 10.1016/S0169-5347(00)01970-4
Daigle, R. M., and Herbinger, C. M. (2009). Ecological interactions between the vase tunicate Ciona intestinalis and the farmed blue mussel Mytilus edulis in Nova Scotia, Canada. Aquat. Invasions 4, 177–187. doi: 10.3391/ai.2009.4.1.18
Davidson, J., Arsenault, G., MacNair, N., Landry, T., and Bourque, D. (2005). Reproduction, Epidemiology and Control of the Clubbed Tunicate, Styela clava, in Prince Edward Island Waters. Aquaculture and Fisheries Research Initiative Report Number 043AR15.
Davis, A. (1989). Contrasting population dynamics and life histories in two populations of the colonial subtidal ascidian Podoclavella moluccensis. Mar Ecol. Prog. Ser. 51, 107–119. doi: 10.3354/meps051107
Davis, M., and Davis, M. (2010). The impact of the ascidian Styela clava Herdman on shellfish farming in the Bassin de Thau, France. J. Appl. Ichthyol. 26, 12–18. doi: 10.1111/j.1439-0426.2010.01496.x
Dijkstra, J., Sherman, H., and Harris, L. G. (2007). The role of colonial ascidians in altering biodiversity in marine fouling communities. J. Exp. Mar. Biol. Ecol. 342, 199–171. doi: 10.1016/j.jembe.2006.10.035
Dukes, J. S., and Mooney, H. A. (1999). Does global change increase the success of biological invaders? Trends. Ecol. Evol. 14, 135–139. doi: 10.1016/S0169-5347(98)01554-7
Durante, K. M., and Sebens, K. P. (1994). Reproductive ecology of the ascidians Molgula Citrina (Alder and Hancock 1848) and Aplidium Glabrum (verrill 1871) from the gulf of maine, USA. Ophelia 3, 1–21. doi: 10.1080/00785326.1994.10429898
Galil, B. S. (2006). “The marine caravan–the Suez Canal and the Erythrean invasion,” in Bridging Divides: Maritime Canals as Invasion Corridors, eds S. Gollasch, B. S. Galil, and A. N. Cohen (Dordrecht: Springer-Verlag), 207–300.
Galil, B. S. (2012). Truth and consequences: the bioinvasion of the Mediterranean Sea. Integr. Zool. 7, 299–311. doi: 10.1111/j.1749-4877.2012.00307.x
Galil, B. S., and Goren, M. (2014). “Metamorphoses: bioinvasions in the Mediterranean Sea,” in The Mediterranean Sea: Its History and Present Challenges, eds S. Goffredo and Z. Dubinsky (Dordrecht: Springer), 463–478.
Galil, B. S., and Zenetos, A. (2002). “A sea change- exotics in the Eastern Mediterranean Sea,” in Invasive Aquatic Species of Europe. Distribution, Impacts and Management, eds E. Leppäkoski, S. Gollasch, and S. Olenin (Dordrecht: Kluwer Academic Publishers), 325–336.
Galil, B. S., Boero, F., Campbell, M. L., Carlton, J. T., Cook, E., Fraschetti, S., et al. (2014a). ‘Double trouble’: the expansion of the Suez Canal and marine bioinvasions in the Mediterranean Sea. Biol. Invasions 17, 973–976. doi: 10.1007/s10530-014-0778-y
Galil, B. S., Marchini, A., Occhipinti-Ambrogi, A., Minchin, D., Narščius, A., Ojaveer, H., et al. (2014b). International arrivals: widespread bioinvasions in European Seas. Ethol. Ecol. Evol. 26, 2–3. doi: 10.1080/03949370.2014.897651
Gewing, M.-T., Bronstein, O., Nagar, L. R., Granot, I., Frid, O., and Shenkar, N. (2016). First record of the non-indigenous ascidian Microcosmus exasperatus, Heller 1878, in Cyprus. Mar. Biodivers. Rec. 1–5. doi: 10.1007/s12526-015-0442-5. Available online at: http://link.springer.com/article/10.1007/s12526-015-0442-5
Gewing, M.-T., Rothman, S. B., Nagar, L. R., and Shenkar, N. (2014). Early stages of establishment of the non-indigenous ascidian Herdmania momus (Savigny, 1816) in shallow and deep water environments on natural substrates in the Mediterranean Sea. Bioinvasions Rec. 3, 77–81. doi: 10.3391/bir.2014.3.2.04
Giraudoux, P. (2012). Pgirmess: Data analysis in Ecology. R package version 1.5.1. Available online at: http://perso.orange.fr/giraudoux/
Golani, D. (1990). Environmentally—induced meristic changes in Lessepsian fish migrants, a comparison of source and colonizing populations. Bull. Instit. Oceanogr. Monaco No. special 7, 143–152.
Goodbody, I. (1961). Continuous breeding in three species of tropical ascidian. Proc. Zool. Soc. Lond. 136, 403–409. doi: 10.1111/j.1469-7998.1961.tb05882.x
Goodbody, I. (1984). Ascidians from Caribbean shallow water localities. Stud. Fauna Curaçao Caribb. Isl. 47, 62–76.
Grant, A., and Tyler, P. A. (1983). The analysis of data in studies of invertebrate reproduction.1. Introduction and statistical analysis of gonad indexes and maturity indexes. Int. J. Inver. Rep. Dev. 6, 259–269. doi: 10.1080/01651269.1983.10510052
Haven, N. D. (1971). Temporal patterns of sexual and asexual reproduction in the colonial ascidian Metandrocarpa taylori Huntsman. Biol. Bull. 140, 400–415. doi: 10.2307/1540277
Hewitt, C. L., Campbell, M. L., Thresher, R. E., Martin, R. B., Boyd, S., Cohen, B. F., et al. (2004). Introduced and cryptogenic species in Port Phillip Bay, Victoria, Australia. Mar. Biol. 144, 183–202. doi: 10.1007/s00227-003-1173-x
Izquierdo-Muñoz, A., Díaz-Valdés, M., and Ramos-Esplá, A. Á. (2009). Recent non-indigenous ascidians in the Mediterranean Sea. Aquat. Invasions 4, 59–64. doi: 10.3391/ai.2009.4.1.5
Kott, P. (1985). The Australian Ascidiacea, part 1. Phlebobranchia and Stolidobranchia. Mem. Queensl. Mus. 23, 1–440.
Lambert, C. C. (2005a). Historical introduction, overview, and reproductive biology of the protochordates. Can. J. Zool. 83, 1–7. doi: 10.1139/z04-160
Lambert, G. (2005b). Ecology and natural history of the protochordates. Can. J. Zool. 83, 34–50. doi: 10.1139/z04-156
Lambert, G. (2001). “A global overview of ascidian introductions and their possible impact on the endemic fauna,” in The Biology of Ascidians, eds H. Sawada, H. Yokosawa, and C. C. Lambert (Tokyo: Springer-Verlag), 249–257.
Lambert, G. (2002). Nonindigenous ascidians in tropical waters. Pac. Sci. 56, 291–298. doi: 10.1353/psc.2002.0026
Lowe, A. (2002). Microcosmus Squamiger; A Solitary Ascidian Introduced to Southern California Harbors and Marinas: Salinity Tolerance and Phylogenetic Analysis. Dissertation/Master's thesis, Fullerton, CA: California State University.
Mack, R. N., Simberloff, D., Lonsdale, W. M., Evans, H., Clout, M., and Bazzaz, F. A. (2000). Biotic invasions: causes, epidemiology, global consequences, and control. Ecol. Appl. 10, 689–710. doi: 10.1890/1051-0761(2000)010[0689:BICEGC]2.0.CO;2
Millar, R. (1952). The annual growth and reproductive cycle in four ascidians. J. Mar. Biol. Assoc. UK. 31, 41–61. doi: 10.1017/S0025315400003672
Millar, R. (1971). The biology of ascidians. Adv. Mar. Biol. 9, 1–100. doi: 10.1016/S0065-2881(08)60341-7
Millar, R. (1977). Ascidians (Tunicata: Ascidiacea) from the northern and north-eastern Brazilian shelf. J. Nat. Hist. 11, 169–223. doi: 10.1080/00222937700770131
Monniot, C. (1981). Apparition de l'ascidie Microcosmus exasperatus dans les ports Méditerranéens. Téthys 10, 59–62.
Monniot, C. (1983). Ascidies littorales de Guadeloupe. VI: Pyuridae et Molgulidae. Bull. Mus. Nat. His. Nat. 5A, 1021–1044.
Monniot, C. (2002). Stolidobranch ascidians from the tropical western Indian Ocean. Zool. J. Linn. Soc. 135, 65–120. doi: 10.1046/j.1096-3642.2002.00017.x
Mooney, H. A., and Drake, J. A. (1986). Ecology of Biological Invasions of North America and Hawaii. New York, NY: Springer-Verlag.
Nagar, R. L., and Shenkar, N. (2016). Temperature and salinity sensitivity of the invasive ascidian Microcosmus exasperatus Heller, 1878. Aquat. Invasions 11, 33–43. doi: 10.3391/ai.2016.11.1.04
Naranjo, S., Carballo, J., and Garcia-Gomez, J. (1997). Effects of environmental stress on ascidian populations in Algeciras Bay (southern Spain). Possible marine bioindicators? Mar. Ecol. Prog. Ser. 144, 119–131. doi: 10.3354/meps144119
Occhipinti-Ambrogi, A. (2007). Global change and marine communities: alien species and climate change. Marine. Poll. Bull. 55, 342–352. doi: 10.1016/j.marpolbul.2006.11.014
Packard, G. C., and Boardman, T. J. (1988). The misuse of ratios, indices, and percentages in ecophysiological research. Physiol. Zool. 61, 1–9. doi: 10.1086/physzool.61.1.30163730
Panagiotou, M., Antoniadou, C., and Chintiroglou, C. (2008). Population dynamics and reproductive status of Microcosmus savignyi Monniot, 1962 (Thermaikos Gulf, Eastern Mediterranean): a preliminary assessment. J. Nat. Hist. 42, 545–558. doi: 10.1080/00222930701835522
Parker, L., Culloty, S., O'Riordan, R., Kelleher, B., Steele, S., and Van der Velde, G. (1999). Preliminary study on the gonad development of the exotic ascidian Styela clava in Cork Harbour, Ireland. J. Mar. Biol. Assoc. UK. 79, 1141–1142. doi: 10.1017/S0025315499001472
Pérès, J. (1958). Ascidies recoltées sur les côtes Méditerranéennes d'Israel. Bull. Res. Counc. Isr. 7B, 143–150.
Pineda, M. C., López-Legentil, S., and Turon, X. (2013). Year-round reproduction in a seasonal sea: biological cycle of the introduced ascidian Styela plicata in the Western Mediterranean. Mar. Biol. 160, 221–230. doi: 10.1007/s00227-012-2082-7
Pineda, M. C., Turon, X., and López-Legentil, S. (2012). Stress levels over time in the introduced ascidian Styela plicata: the effects of temperature and salinity variations on hsp70 gene expression. Cell. Stress Chaperon. 17, 435–444. doi: 10.1007/s12192-012-0321-y
Por, F. D. (1978). Lessepsian Migration: The Influx of Red Sea Biota into the Mediterranean by Way of the Suez Canal. Berlin: Springer-Verlag.
R Core Team (2013). R: A Language and Environment for Statistical Computing. Vienna: R Foundation for Statistical Computing. Available online at: http://www.R-project.org
Ramos-Esplá, A. A., Izquierdo, A., and Çinar, M.E. (2013). Microcosmus exasperatus (Ascidiacea: Pyuridae), current distribution in the Mediterranean Sea. Mar Biodivers. Rec. 6, 1–5. doi: 10.1017/S1755267213000663
Ramsay, A., Davidson, J., Landry, T., and Arsenault, G. (2008). Process of invasiveness among exotic tunicates in Prince Edward Island, Canada. Biol. Invasions 10, 1311–1316. doi: 10.1007/s10530-007-9205-y
Rinkevich, B., Shlemberg, Z., Lilker-Levav, T., Goren, M., and Fishelson, L. (1993). Life history characteristics of Botrylloides (Tunicata) populations in Akko Bay, Mediterranean coast of Israel. Isr. J. Zool. 39, 197–212.
Rius, M., Heasman, K. G., and McQuaid, C. D. (2011). Long-term coexistence of non-indigenous species in aquaculture facilities. Mar. Poll. Bull. 62, 2395–2403. doi: 10.1016/j.marpolbul.2011.08.030
Rius, M., Pascual, M., and Turon, X. (2008). Phylogeography of the widespread marine invader Microcosmus squamiger (Ascidiacea) reveals high genetic diversity of introduced populations and non-independent colonizations. Divers. Distrib. 14, 818–828. doi: 10.1111/j.1472-4642.2008.00485.x
Rius, M., Pineda, M. C., and Turon, X. (2009). Population dynamics and life cycle of the introduced ascidian Microcosmus squamiger in the Mediterranean Sea. Biol. Invasions 11, 2181–2194. doi: 10.1007/s10530-008-9375-2
Robinson, T., Griffiths, C., McQuaid, C., and Rius, M. (2005). Marine alien species of South Africa-status and impacts. Afr. J. Mar. Sci. 27, 297–306. doi: 10.2989/18142320509504088
Rocha, R. M., Bonnet, N. Y., Baptista, M. S., and Beltramin, F. S. (2012). Introduced and native Phlebobranch and Stolidobranch solitary ascidians (Tunicata: Ascidiacea) around Salvador, Bahia, Brazil. Zoologia 29, 39–53. doi: 10.1590/S1984-46702012000100005
Rocha, R. M., da Cruz Lotufo, T. M., and de Almeida Rodrigues, S. (1999). The biology of Phallusia nigra Savigny, 1816 (Tunicata: Ascidiacea) in southern Brazil: spatial distribution and reproductive cycle. B. Mar. Sci. 6, 77–88.
Ruiz, G. M., Carlton, J. T., Grosholz, E. D., and Hines, A. H. (1997). Global invasions of marine and estuarine habitats by non-indigenous species: mechanisms, extent, and consequences. Am. Zool. 37, 621–632. doi: 10.1093/icb/37.6.621
Sahade, R., Tatián, M., and Esnal, G. B. (2004). Reproductive ecology of the ascidian Cnemidocarpa verrucosa at Potter Cove, South Shetland Islands, Antarctica. Mar. Ecol. Prog. Ser. 272, 131–140. doi: 10.3354/meps272131
Seebens, H., Gastner, M., and Blasius, B. (2013). The risk of marine bioinvasion caused by global shipping. Ecol. Lett. 16, 782–790. doi: 10.1111/ele.12111
Shenkar, N., and Loya, Y. (2008). The solitary ascidian Herdmania momus: native (Red Sea) versus non-indigenous (Mediterranean) populations. Biol. Invasions 10, 1431–1439. doi: 10.1007/s10530-008-9217-2
Shenkar, N., and Loya, Y. (2009). Non-indigenous ascidians (Chordata: Tunicata) along the Mediterranean coast of Israel. Mar. Biodivers. Rec. 2:e166. doi: 10.1017/S1755267209990753
Shenkar, N., and Swalla, B. J. (2011). Global diversity of Ascidiacea. PLoS ONE 6:e20657. doi: 10.1371/journal.pone.0020657
Shine, C., Williams, N., and Gündling, L. (2000). A Guide to Designing Legal and Institutional Frameworks on Alien Invasive Species. Gland: IUCN.
Somot, S., Sevault, F., and Déqué, M. (2006). Transient climate change scenario simulation of the Mediterranean Sea for the twenty-first century using a high-resolution ocean circulation model. Clim. Dynam. 27, 851–879. doi: 10.1007/s00382-006-0167-z
Stachowicz, J. J., Terwin, J. R., Whitlatch, R. B., and Osman, R. W. (2002). Linking climate change and biological invasions: ocean warming facilitates non-indigenous species invasions. Proc. Natl. Acad. Sci. U.S.A. 99, 15497–15500. doi: 10.1073/pnas.242437499
Streftaris, N., and Zenetos, A. (2006). Alien Marine Species in the Mediterranean- the 100 ‘Worst Invasives’ and their Impact. Mediterr. Mar. Sci. 7, 87–118. doi: 10.12681/mms.180
Svane, I., and Lundälv, T. (1981). Reproductive patterns and population dynamics of Ascidia mentula O.F. Müller on the Swedish west coast. J. Exp. Mar. Biol. Ecol. 50, 163–182. doi: 10.1016/0022-0981(81)90048-4
Tokioka, T. (1967). Pacific tunicata of the United States. Bull. Am. Mus. Nat. Hist. 251, 1–247. doi: 10.5479/si.03629236.251.1
Turon, X. (1987). Estudio de las Ascidias de las Costas de Cataluña e Islas Baleares Barcelona. Dissertation/Ph.D. thesis, Barcelona, SP: University of Barcelona.
Turon, X. (1988). The ascidians of Tossa de Mar (Girona, NE of Spain). II. Biological cycles of the colonial species. Cah. Biol. Mar. 29, 407–418.
Turon, X., Nishikawa, T., and Rius, M. (2007). Spread of Microcosmus squamiger (Ascidiacea: Pyuridae) in the Mediterranean Sea and adjacent waters. J. Exp. Mar. Biol. Ecol. 342, 185–188. doi: 10.1016/j.jembe.2006.10.040
U.S Congress OTA (1993). Harmful Non-indigenous Species in the United States OTA-F565. Washington, DC: US Government Printing Office.
Vafidis, D., Antoniadou, C., and Chintiroglou, C. (2008). Population dynamics, allometric relationships and reproductive status of Microcosmus sabatieri (Tunicata: Ascidiacea) in the Aegean Sea. J. Mar. Biol. Assoc. UK. 88, 1043–1051. doi: 10.1017/S0025315408001811
Walther, G. R., Post, E., Convey, P., Menze, A., Parmesan, C., Beebee, T. J., et al. (2002). Ecological responses to recent climate change. Nature 416, 389–395. doi: 10.1038/416389a
Wheeler, B. (2010). lmPerm: Permutation Tests for Linear Models Linear Model Functions Using Permutation Tests. The Comprehensive R Archive Network. Available online at: https://cran.r-project.org/web/packages/lmPerm/index.html
Whitlatch, R. B., Osman, R. W., Frese, A., Malatesta, R., Mitchell, P., and Sedgwick, L. (1995). “The ecology of two introduced marine ascidians and their effects of epifaunal organism in Long Island Sound,” in Northeast Conference on Non-indigenous Aquatic Species, ed N. Balcolm (Groton, CT: Connecticut Sea Grant College Program, University of Connecticut, Publication No. CT-SG-9504), 29–48.
Wilcove, D. S., Rothstein, D., Dubow, J., Phillips, A., and Losos, E. (1998). Quantifying threats to imperiled species in the United States. Bioscience 48, 607–615. doi: 10.2307/1313420
Wong, N. A., McClary, D., and Sewell, M. A. (2011). The reproductive ecology of the invasive ascidian, Styela clava, in Auckland Harbour, New Zealand. Mar. Biol. 158, 2775–2785. doi: 10.1007/s00227-011-1776-6
Yamaguchi, M. (1975). Growth and reproductive cycles of the marine fouling ascidians Ciona intestinalis, Styela plicata, Botrylloides violaceus and Leptoclinum mitsukurii at Aburatsubo Moriso Inlet (Central Japan). Mar. Biol. 29, 253–259. doi: 10.1007/BF00391851
Zeileis, A., Wiel, M. A., Hornik, K., and Hothorn, T. (2008). Implementing a class of permutation tests: the coin package. J. Stat. Softw. 281, 1–23. doi: 10.18637/jss.v028.i08
Zenetos, A., Almogi, A., Ates, S., Azzurro, E., Ballesteros, E., Bianchi, C. N., et al. (2012). Alien species in the Mediterranean Sea by 2012. A contribution to the application of European Union's Marine Strategy Framework Directive (MSFD). Part 2. Trends in introduction and pathway/vector. Medit. Mar. Sci. 13, 328–352. doi: 10.12681/mms.327
Zenetos, A., Gofas, S., Verlaque, M., Çinar, M. E., Garci'a Raso, J. E., Bianchi, C. N., et al. (2010). Alien species in the Mediterranean Sea by 2010. A contribution to the application of European Union's Marine Strategy Framework Directive (MSFD). Part I. Spatial distribution. Medit. Mar. Sci. 11, 318–493. doi: 10.12681/mms.87
Keywords: tunicates, marine bioinvasions, lessepsian invasion, ascidian reproduction, Mediterranean Sea
Citation: Raijman Nagar L and Shenkar N (2016) From Tropical to Sub-Tropical: Prolonged Reproductive Activity of the Invasive Ascidian Microcosmus exasperatus in the Eastern Mediterranean. Front. Ecol. Evol. 4:102. doi: 10.3389/fevo.2016.00102
Received: 29 May 2016; Accepted: 10 August 2016;
Published: 25 August 2016.
Edited by:
Paul Snelgrove, Memorial University of Newfoundland, CanadaReviewed by:
Melih Ertan Çinar, Ege University, TurkeyMary Carman, Woods Hole Oceanographic Institution, USA
Copyright © 2016 Raijman Nagar and Shenkar. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Noa Shenkar, c2hlbmthcm5AcG9zdC50YXUuYWMuaWw=
 Lilach Raijman Nagar
Lilach Raijman Nagar Noa Shenkar
Noa Shenkar