Abstract
The harlequin ladybird Harmonia axyridis is now established as a model to test hypotheses explaining why some species become successfully invasive, while others, even closely related ones, do not. In this review, we evaluate behavioral and immunological features that may play a role in the invasive performance of this model species. We discuss the behavioral traits and associated semiochemicals that promote the invasive success of H. axyridis. In particular, we consider (1) the aggregative behavior and the particular role of long-chain hydrocarbons; (2) the importance of sex pheromones and non-volatile chemicals in mate location and selection; (3) the use of allelochemicals for prey location; and (4) the nature of chemicals that protect against natural enemies. We also highlight the superior immune system of H. axyridis, which encompasses a broader spectrum of antimicrobial peptides (and higher inducible expression levels) compared with native ladybird beetles such as Adalia bipunctata and Coccinella septempunctata. The chemical defense compound harmonine and the antimicrobial peptides are thought to confer resistance against the abundant microsporidia carried by H. axyridis. These parasites can infect and kill native ladybird species feeding on H. axyridis eggs or larvae, supporting the hypothesis that intraguild predation plays a role in the ability of H. axyridis to outcompete native ladybird species in newly-colonized areas.
Introduction
Biological invasions are attracting scientific interest because invasive species can cause extensive economic losses and negatively affect the biodiversity of newly-colonized areas. The harlequin ladybird Harmonia axyridis (Coleoptera: Coccinellidae), which is also known as the multicolored or Asian ladybird, has emerged as a powerful model in which to test hypotheses explaining why some species are successful invaders, while others, even closely related ones, are not. H. axyridis is native to continental, temperate and subtropical parts of East and Central Asia and has been introduced into North America, Europe and Russia since the beginning of the Twenty-fifth century as a biological control agent against aphid and/or coccid pests (Roy et al., 2016). It is an excellent example of an efficient invasive species, being one of the most abundant ladybird species in various agricultural ecosystems including corn, soybean, wheat, corn, broad bean, potato, and cotton (Gardiner et al., 2011; Vandereycken et al., 2013a,b). H. axyridis is highly fertile and the eggs show high viability (Santos et al., 2014). Multi-year surveys have documented the decline of native coccinellid species in concert with the establishment of H. axyridis (e.g., Bahlai et al., 2015). Such declines are partially explained by exploitative competition for shared prey and interference competition via intraguild predation (Smith and Gardiner, 2013). Further effects caused by the introduction of H. axyridis include aggregation in buildings and in some agricultural settings, such that the species is considered both an urban and agricultural pest. However, several reports also highlight the positive effects of H. axyridis on agriculture, mainly due to its spectacular voracity, predation capacity, and effectiveness in suppressing plant pests (Riddick, 2017).
H. axyridis is a widely-used model species for research, especially studies concerning the effects of invasive species when they are introduced into new environments (Roy and Wajnberg, 2008; Sloggett et al., 2011). Invading populations often undergo rapid evolutionary changes associated with their introduction, particularly life history traits (Lambrinos, 2004). Studies comparing H. axyridis with other coccinellid beetles help to determine the basis of its unique invasive success. This paper considers the immunological and behavioral traits that make H. axyridis a successful invasive species.
Behavioral features
Aggregation
Aggregation is one of the most basic social behaviors. It leads isolated individuals to gather at particular sites to exploit resources. Many insect species show aggregation behavior, which has a range of benefits including enhanced defense, better access to mates, food location and utilization, prevention of desiccation, and regulation of internal temperature. Many beetle species aggregate, and this behavior can be observed at any time of year in H. axyridis (Durieux et al., 2015). This behavior can be a nuisance to humans, e.g., in vineyards, adult H. axyridis aggregate on ripe grape clusters. If they are not removed, they can be processed with the grape juice, and can introduce unpleasant aroma and taste qualities to the resulting wines (Botezatu et al., 2013). In H. axyridis, aggregation behavior is observed both at the adult and the pupal stage, and both increase the general fitness of the gregarious individuals.
Aggregation in H. axyridis has been studied predominantly to investigate their overwintering behavior. The beetles tolerate cold by accumulating large quantities of polyols, and thus reducing their supercooling point (Watanabe, 2002). However, this physiological trait does not allow H. axyridis individuals to remain in their feeding habitat and survive low winter temperatures. Therefore, the beetles are solitary during spring and summer, but seek aggregation sites as temperature and sunlight decrease. In their native range, overwintering H. axyridis aggregate in natural shelters (caves, crevices and rocky depressions) with good exposure to the sun, to mitigate their exposure to freezing conditions. On their way to these natural shelters, they sometimes shelter in human residences for several days (Wang et al., 2011). When the temperature and sunlight increase in the spring, the aggregated individuals return to nearby agricultural habitats and resume their solitary life. Invasive populations behave similarly, but whereas most native individuals aggregate outside during winter, this is not possible for invasive individuals. In Canada for example, H. axyridis does not survive outside during winter, whereas the native species do. With lower proportions of lipids than native species, H. axyridis is thought to be physiologically ill-equipped to overwinter outside in North America. The migratory flight pattern of invasive H. axyridis has been well-documented, with individuals preferentially moving toward prominent and high-color-contrast elements (Nalepa et al., 2005). On sunny autumn days, pale exterior walls with southern exposure can be covered by hundreds of migrating individuals. A correlation between the ability to select a proper overwintering site and the subsequent invasive success of H. axyridis has been suggested (Labrie et al., 2008). Invasive populations of adult H. axyridis are considered urban pests because large aggregations often form inside buildings, including dwellings, offices, and wind turbines (Nalepa et al., 1996; Dudek et al., 2015). Dwellings can be invaded by thousands of individuals, which release an unpleasant smell, cause staining, and sometimes induce allergic reactions (Nalepa et al., 2004; Goetz, 2007).
Camazine et al. (2001) categorize aggregation behavior in the animal kingdom as social and non-social aggregation, according to whether or not individuals interact with each other to modulate the behavioral decisions of conspecifics. During aggregation, adult H. axyridis show mutual interactions, and should therefore be considered as social aggregators. In addition to the physical contacts between congeners (Durieux et al., 2014a), laboratory assays have shown that chemical markings are involved in this aggregation behavior (Durieux et al., 2012). Non-volatile long-chain hydrocarbons are passively deposited by walking males and females on the surfaces surrounding the aggregation site (Kosaki and Yamaoka, 1996; Durieux et al., 2012). The chemical composition of the marking varies with the season, being richer in unsaturated hydrocarbons in winter than in summer (Durieux et al., 2013). Overwintering individuals are also more likely to follow this chemical mark than individuals collected in spring or summer. The overwintering surface is also chemically marked with a similar chemical blend, comprising the same molecules but in different ratios (Durieux et al., 2012). This suggests there are two different blends of long-chain hydrocarbons, the first leading conspecifics toward aggregation sites and the second ensuring the cohesion of the aggregate. The first individuals reaching a potential overwintering site may use chemical tracks left earlier by conspecifics. The chemical marking of oviposition sites is not exclusive to H. axyridis, e.g., it is also observed in Adalia bipunctata (Majerus, 1997). Thanks to their low volatility and high stability, these long-chain hydrocarbons remain on surfaces for several weeks (Durieux et al., 2014b). However, chemical analysis has shown that unsaturated hydrocarbons were no longer detected after a year, whereas some saturated hydrocarbons were still present in large quantities, suggesting that the chemical markings left by conspecifics during a previous aggregation period in an overwintering site are not sufficient to induce the gathering of new individuals (Durieux et al., 2014b). Whether a volatile aggregation pheromone is used by gregarious H. axyridis remains unclear. It is possible that (–)-β-caryophyllene fulfills this role, because this sesquiterpene (1) was isolated from the headspace volatiles above overwintering H. axyridis individuals, (2) elicited electrophysiological responses from female and male antennae, and (3) attracted both genders in a laboratory bioassay (Verheggen et al., 2007).
The data summarized above suggest that H. axyridis aggregate inside buildings due to their intrinsic behavior and this is not an emergent trait of invasive populations. However, differences between native and invasive populations may have arisen in the context of chemical communication established during the aggregation process. It is unclear whether behavioral/chemical traits associated with aggregation behavior are promoted as a result of the invasion process. Finally, gregarious pupation may act as a defensive mechanism for H. axyridis (Roberge et al., 2016). In the field, H. axyridis was the coccinellid species showing the highest rate of gregarious pupation events. The mortality rate of pupae located in a group was lower than that of isolated pupae exposed to intraguild predators or cannibals.
Mate location, mating, and oviposition
For sexually-reproducing animals, finding and selecting mates is an essential step which determines their reproductive success. Ladybird species exhibit a range of behavioral traits associated with reproduction, and chemical cues are usually involved (Fassotte et al., 2016). Like other ladybeetle species, freshly emerged H. axyridis adults pass through a pre-mating period during which their gonads mature (Obata and Hidaka, 1987). Several behavioral observations suggest that males are attracted over a distance (e.g., Schaller and Nentwig, 2000; Omkar and Pervez, 2005), and H. axyridis is the only ladybird species in which a female volatile sex pheromone has been identified (Fassotte et al., 2014). In the presence of prey, virgin H. axyridis females display a typical calling behavior: they raise their elytra and squeeze their abdomen. This behavior is associated with the release of five chemicals that attract males: (–)-β-caryophyllene (the major constituent), β-elemene, methyl-eugenol, α-humulene and α-bulnesene. The trichoid sensilla of the male beetles are thought to act as pheromone receptors (Chi et al., 2009).
Following distance attraction, the courtship behavior of H. axyridis males involves four characteristic steps before copulation: getting close to the female, examining her at a distance, mounting and attempting to copulate (Obata, 1987). The relative importance of visual, tactile and olfactory cues in mate recognition is often the subject of debate, and is likely to vary among different species. In contrast to H. axyridis, A. bipunctata males do not examine a female before touching her body surface with their maxillary palps, and copulation occurs directly after mounting the female (Hemptinne et al., 1998). Additional behavioral movements are sometimes observed in males of other species, including C. sexmaculata and Anegleis cardoni, and these behaviors may encourage the female to remain still during copulation and post-copulation (Maisin et al., 1997; Omkar et al., 2013). After genital connection, H. axyridis males shake their body at constant intervals to allow sperm transfer (Obata, 1987). The mating receptivity of H. axyridis females is dependent on their physiological state. Sexually immature females avoid copulation by moving away from an insistent male or by shaking him off their abdomen (Obata, 1988). They are also more reluctant to mate when they are deprived of food. Not all H. axyridis males are of equal fitness value as mate, and both the elytra color and body size affect male mating success (Ueno et al., 1998). As in other ladybeetle species, multiple copulations occur and enhance the total number of eggs and the percentage of hatching (Ueno, 1996; Omkar and Pervez, 2005). Furthermore, H. axyridis females retain their eggs for longer after mating with less preferred males, allowing the females to partially replace stored sperm with that from a preferred male (Su et al., 2009).
Chemical signals are involved in the courtship behavior of many invertebrate species, and ladybeetles are no exception. A significant number of published reports highlight the role of cuticular chemicals (Fassotte et al., 2016). Indeed, the qualitative and quantitative profile of cuticular hydrocarbons (CHCs) tend to be species and gender specific, making them good candidates for mate recognition (Hemptinne et al., 1998). The dominant CHC may facilitate species recognition, whereas gender recognition is dependent on quantitative and/or qualitative differences (Pattanayak et al., 2014). The CHC profile differs between virgin and mated H. axyridis females (unpublished results). To overcome sperm competition and subsequently increase their fitness, males should select their mate based on the reproductive status of the female. However, H. axyridis males failed to discriminate between virgin and mated females based on their chemical profile during laboratory assays (unpublished results).
Following mate attraction and selection, oviposition is the next important behavioral step for female ladybeetles. The distribution of oviposition sites among conspecific females is of prime importance because it allows them to share resources by partitioning their niches (Sicsú et al., 2015). A few laboratory and semi-field studies suggest that H. axyridis is deterred from ovipositing in the presence of conspecifics, whereas heterospecific competitors do not influence oviposition site selection (Yasuda et al., 2000; Almohamad et al., 2010). Gravid H. axyridis females reduced their rates of oviposition when exposed to the feces of conspecifics, but not when exposed to the feces of heterospecifics (Propylea japonica) (Agarwala et al., 2003). However, the opposite is not true, i.e., P. japonica avoids sites contaminated with either heterospecific or conspecific feces. Chemical markings deposited by syrphid and coccinellid larvae did not deter H. axyridis females from laying eggs. Similar results were observed in other ladybeetle species, including C. septempunctata, Hippodamia convergens, and A. bipunctata, where oviposition was deterred in the presence of conspecific larvae, but not in presence of heterospecific competitors (Ruzicka, 1997; Doumbia et al., 1998; Michaud and Jyoti, 2007). These results suggest the presence of a species-dependent oviposition-deterring pheromone in ladybeetles, which remains to be characterized and compared among coccinellid species. Finally, the cluster size and the distance from the cluster to an aphid colony affect the proportion of cannibalized eggs, as suggested by laboratory and field observations of H. axyridis (Osawa, 2003).
When a species shifts its geographical range, invading individuals face new selective pressures that may affect their reproductive strategy. Indeed, reproduction-associated life history traits may be subject to rapid evolutionary shifts during invasions because they affect population dynamics and genetic parameters that can, in turn, have feedback effects on evolutionary processes (Laugier et al., 2013). Evolutionary changes in reproductive strategy associated with invasion have been highlighted. A comparison of the sex pheromone composition between native and invasive H. axyridis populations showed no qualitative differences, but females from invasive populations released up to three times as much of the sex pheromone compared to native individuals (unpublished results). It is unclear whether invasive individuals were selected during invasion due to their capacity to call and find sexual partners more effectively. Males and females from invasive populations are also more reproductively efficient, with both sexes showing a shorter pre-mating period and producing more offspring than native individuals (Laugier et al., 2013). Finally, H. axyridis males can identify the population of origin (native vs. invasive) of a female based on her CHC profile (unpublished results). The reproductive behavior of H. axyridis is certainly a trait that deserves more attention in terms of the potential evolutionary shifts that may have accompanied its invasive success.
Prey location
H. axyridis is a generalist predator that feeds preferentially on aphids, but also occasionally upon a wide range of other soft-bodied arthropods and plant products (Koch, 2003). This feeding practice is thought to enhance its ability to colonize various ecosystems. Studies directly comparing prey location and consumption between H. axyridis and more strictly aphidophagous coccinellids are scarce, but would allow a better understanding of the invasive success achieved by H. axyridis. Because aphid colonies are sporadically distributed and transient, efficient prey finding behavior is essential. Indeed, when prey are scarce, H. axyridis exhibits slower development and produces smaller larvae (Dmitriew and Rowe, 2007). Compared to other ladybeetle species, H. axyridis is reputed to be more strongly polyphagous and voracious (Koch, 2003). This reputation was confirmed in a laboratory experiment where Leppanen et al. (2012) found that H. axyridis find aphids more quickly and consume more of them compared to six other ladybeetle species. However, Reynolds and Cuddington (2012) found that H. axyridis was less able than the green lacewing to attach and maneuver on plants with few branches and edges, resulting in a lower aptitude to capture prey on such plants.
Although visual cues are likely to be involved (Lambin et al., 1996), olfactory cues are considered more important for prey location by H. axyridis (Obata, 1986; Mondor and Warren, 2000; Sloggett et al., 2011). When seeking prey, ladybeetles increase their walking speed and reduce their turning frequency. Like other aphidophagous predators, H. axyridis is attracted to volatile cues released by prey and infested plants (Verheggen et al., 2007, 2008). When getting closer to prey, the foraging behavior becomes more intensive, with lower walking speed and more directional changes (Pettersson et al., 2005). Olfactory cues include prey pheromones (Verheggen et al., 2007, 2010), host-plant volatiles (Leroy et al., 2012a), prey waste products such as honeydew (Leroy et al., 2012b), and conspecific-associated odors (Almohamad et al., 2010; Leroy et al., 2012a). Like other insects, H. axyridis larvae deposit chemical marks as they forage. Following the detection of such marks, they modify their foraging behavior to avoid areas already visited, hence marking individuals consume more prey than non-marking ones (Meisner and Ives, 2011). Both H. axyridis and C. septempunctata larvae avoid foraging in areas with conspecific chemical markings, to reduce the risk of cannibalism (Meisner et al., 2011). But whereas C. septempunctata also avoids H. axyridis larval tracks, H. axyridis does not avoid C. septempunctata larval tracks, demonstrating an asymmetry in response to larval tracks that parallels the asymmetry in aggressiveness between these species as intraguild predators. Finally, recent experiments have shown that H. axyridis beetles exposed to sub-lethal doses of pesticide fly for longer periods and cover greater distances than non-exposed beetles (Xiao et al., 2017). They are thought to follow the migration of their prey away from the contaminated ecosystem and may also have developed avoidance behavior in the presence of pesticides, both of which are likely to promote the fitness of H. axyridis (Desneux et al., 2007).
Intraguild predation
Native ladybeetle populations have declined in most areas where H. axyridis has been introduced (Camacho-Cervantes et al., 2017) and this is often blamed on interference competition via intraguild predation (Pell et al., 2008). More specifically, H. axyridis is considered a top-level predator in the aphidophagous guilds, reflecting its direct predation behavior toward eggs and larvae of native coccinellids (Ware and Majerus, 2008) as well as non-coccinellid aphidophagous species, such as hoverflies and lacewing (Wells et al., 2017). H. axyridis also practices indirect intraguild predation on aphid parasitoids, because it preferentially consumes parasitized aphids over uninfected ones (Meisner et al., 2011). Laboratory and field studies of intraguild predation involve both visual observations and, more recently, the screening of gut contents by DNA analysis (e.g., Gagnon et al., 2011; Rondoni et al., 2015). Such studies have repeatedly indicated that intraguild predation behavior is important for the invasion success of H. axyridis. Moreover, semi-field experiments directly comparing the frequency of intraguild predation events in coccinellid species confirm that H. axyridis is the most successful intraguild predator during heterospecific confrontations (Raak-van den Berg et al., 2012). After encountering a heterospecific competitor, H. axyridis also drops less easily from a plant leaf than other coccinellid species. Poorly-fed H. axyridis larvae feed more voraciously on intraguild competitors than well-nourished ones (Ingels et al., 2015; Mirande et al., 2015). Indeed, small and poorly-fed larvae may have more to gain, from a fitness perspective, than well-nourished larvae, for whom food is not critical for survival. Along with its aggressive behavior, H. axyridis has multiple other traits making it more competitive than native ladybeetle species, i.e., it has a relatively large body, and carries spines at the larval stage as well as chemical defenses (Ware and Majerus, 2008; Sloggett et al., 2011).
Phenotypic plasticity
Theory suggests that invasive populations should evolve toward greater phenotypic plasticity because they face diverse environments during the invasion process (Lombaert et al., 2008). The high degree of phenotypic plasticity observed in H. axyridis has enabled its populations to become successful invaders of new territories, where they dominate native coccinellid species (Alyokhin and Sewell, 2004; Lombaert et al., 2008). Lombaert et al. (2008) compared phenotypic traits related to fitness among different H. axyridis populations and found that invasive populations displayed higher survival and phenotypic plasticity when entering into quiescence at low temperatures, compared to populations commercialized for biological control.
H. axyridis is highly polymorphic in terms of color patterning (Dobzhansky, 1933). In ladybeetles, melanism is advantageous in winter but costly in summer, so species that can change color throughout the year can maximize their fitness (Michie et al., 2010). Laboratory and field observations suggest that H. axyridis demonstrates seasonal phenotypic plasticity related to melanism, the non-melanic morph being more abundant in spring, and the darker morphs being more abundant in autumn (Michie et al., 2011). Melanization in H. axyridis is predominantly controlled by temperature during larval development. Such seasonal phenotypic plasticity allows individuals to produce the level of melanin necessary to maintain activity at the temperatures encountered when they emerge (Michie et al., 2011).
Chemical defenses
Like all coccinellid beetle species, H. axyridis can synthesize several defensive secondary compounds which play an important role against a range of attackers and are especially effective in reducing the performance of predators. The chemical defense system of ladybirds is based mainly on repellent (and in some cases toxic) alkaloids, which tend to be produced during all life stages. These alkaloids are derived from simple fatty acids, and their remarkable diversity makes ladybird beetles pioneers in combinatorial chemistry. Some defensive alkaloids are extremely toxic, such as precoccinelline produced by the seven-spot ladybird C. septempunctata, which is a potent neurotoxin in both insects and mammals. In contrast, adaline produced by the two-spot ladybird A. bipunctata is toxic in many insects but has little effect in mammals. The chemical defenses of H. axyridis have been extensively reviewed (Sloggett et al., 2011). In the context of its invasive performance, we focus here on the chemical defensive alkaloid harmonine [(17R,9Z)-1,17-diaminooctadec-9-ene], which is not produced by C. septempunctata or A. bipunctata. This compound was found to be responsible for the high constitutive antibacterial activity in the hemolymph of H. axyridis beetles (Röhrich et al., 2012).
A synthetic analog of harmonine has been produced as a reference and has been used to screen the activity of the natural compound against pathogens and parasites. The harmonine concentration in the hemolymph increases during development, reaching 27 mM in adult beetles (Schmidtberg et al., 2013). Interestingly, harmonine is active against a broad spectrum of bacteria, particularly against mycobacteria. Harmonine was also active against both chloroquine-sensitive and chloroquine-resistant Plasmodium falciparum, which is responsible for the most severe form of malaria (Röhrich et al., 2012). In addition, harmonine was also found to inhibit Leishmania major, which causes leishmaniosis (Nagel et al., 2015). These findings suggest that harmonine may function as a broad-spectrum chemical weapon, providing protection against diverse pathogens and parasites that are encountered by H. axyridis in also newly-colonized environments. Furthermore, harmonine may help to regulate the abundant microsporidia found in H. axyridis (Vilcinskas et al., 2015). Microsporidia are spore-forming obligate parasites that are frequently associated with insects. The average concentration of microsporidia in the H. axyridis hemolymph was found to increase during development, reaching appr. 13 × 106 per ml. These parasites have been shown to kill A. bipuncata larvae feeding on microsporidia-infected eggs or larvae of H. axyridis, suggesting they can be transmitted from the invasive carrier to native ladybirds via intraguild predation (Vogel et al., 2017a). The potential role of these parasites as bioweapons against competing native ladybird is discussed in more detail below.
The superior immune system of H. axyridis
Insects lack the antibody-based adaptive immunity found in vertebrates and rely entirely on innate immunity, which encompasses cellular mechanisms such as the phagocytosis and multicellular encapsulation of pathogens and parasites as well as humoral mechanisms based on the synthesis of antimicrobials. In the latter context, antimicrobial peptides (AMPs) play a predominant role among the immunity-related effector molecules produced by insects, and a large spectrum of evolutionarily conserved and taxon-specific AMP families has been described in insects (Mylonakis et al., 2016). Theory predicts that invasive species should have a better or more flexible immune system than even closely related non-invasive species because they have to cope with pathogens and parasites in new habitats, meaning they cannot adapt to such threats by coevolution (Lee and Klasing, 2004; Vilcinskas, 2013). Accordingly, next-generation sequencing of the immunity-related H. axyridis transcriptome revealed almost 50 genes encoding putative AMPs, the highest number of AMPs found in any animal species investigated thus far (Vilcinskas et al., 2013a). Native ladybirds have far fewer AMP genes (Vogel et al., 2017a): 15 putative AMP genes were identified in C. septempunctata and only 12 in A. bipunctata (Figure 1). H. axyridis not only has more AMP genes than native ladybird species such as C. septempunctata and A. bipunctata, but these genes are induced much more strongly in H. axyridis when the immune system is challenged (Vilcinskas et al., 2013a) (Figure 1).
Figure 1
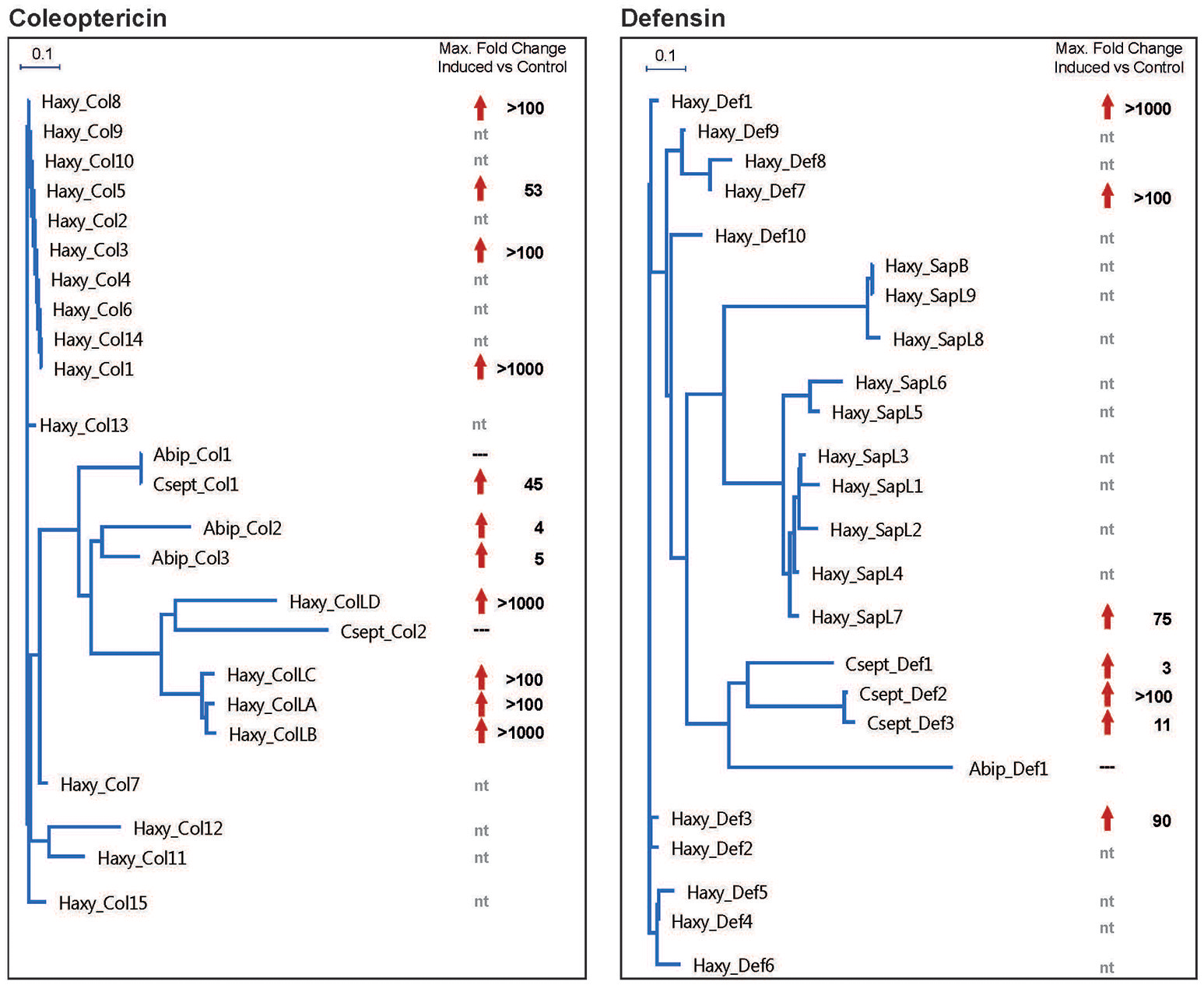
Coleoptericin and defensin gene trees and maximum induction levels after an immune challenge in three ladybeetle species. (A) Evolutionary relationships and gene expression changes among the coleoptericin proteins. (B) Evolutionary relationships and gene expression changes among the defensin proteins. The alignments were created using MAFFT and gene trees were built using the BioNJ algorithm implemented in MegAlignPro. The distance scale is shown at the top left. Differential gene expression values are shown for a selected set of coleoptericins and coleoptericin-like genes (A) or defensins and defensin-like genes (B) and are shown as fold changes following an immune challenge vs. untreated control beetles. Red arrows indicate higher gene expression levels in immune-challenged beetles whereas dashes indicate no change. Nt, not tested; Haxy, Harmonia axyridis; Abip, Adalia bipunctata; Csept, Coccinella septempunctata.
Phenotypic variation of immunity-related gene expression
A key question emerging from the studies described above is whether the differential induction of immunity-related genes in invasive and native ladybeetle species is related to the observed differences in immunity. The injection of bacteria caused a 100-fold induction of certain AMP genes (compared to untreated controls) in A. bipunctata, a 1,000-fold induction in C. septempunctata but a more than 10,000-fold induction in H. axyridis (Figure 2). Differences in induction spanning several orders of magnitude reflect unprecedented immunological differences between invasive and non-invasive species, which support the hypothesis that invasive success depends in part on a superior immune system (Lee and Klasing, 2004; Vilcinskas, 2013).
Figure 2
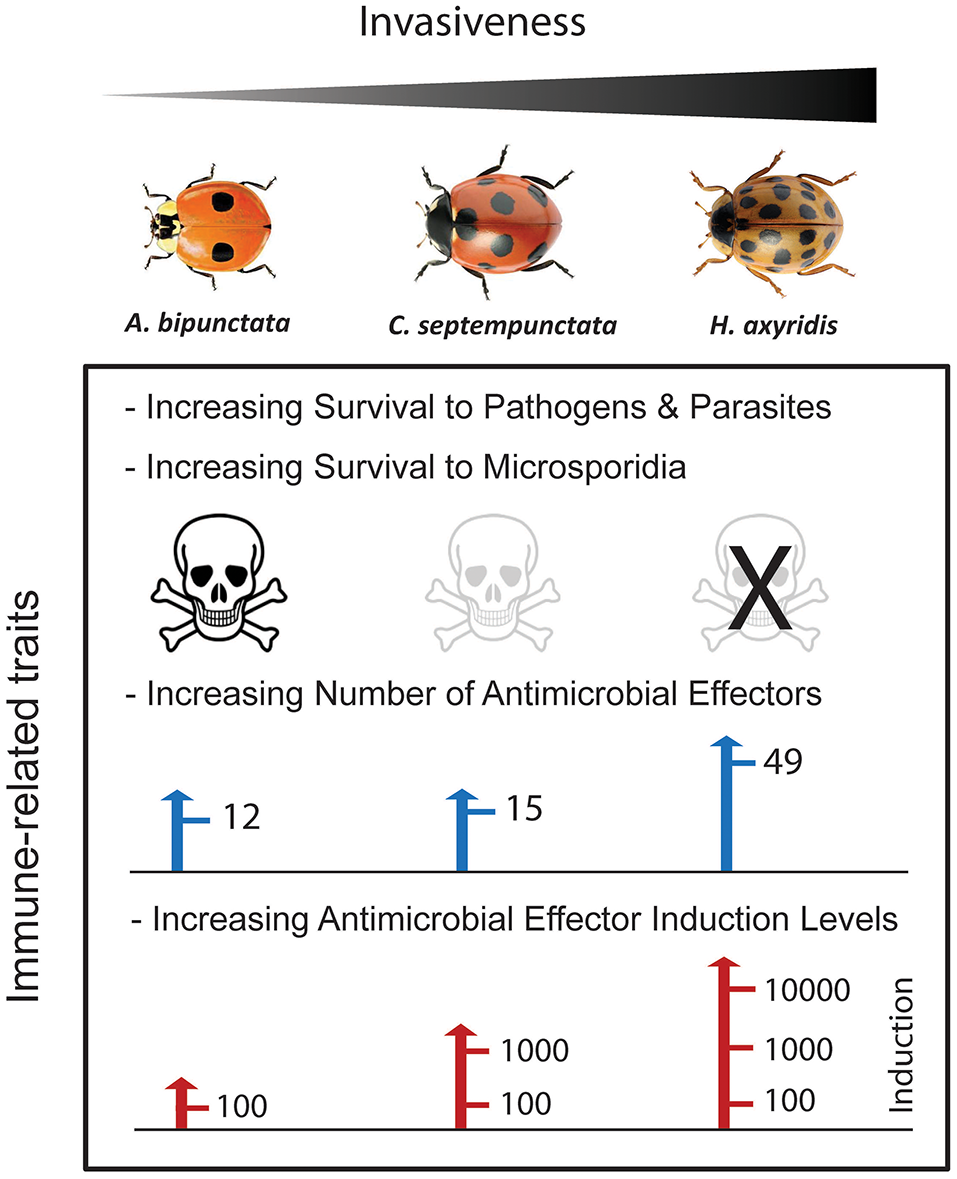
Relationship between immune-related traits, pathogen resistance and invasiveness in three ladybeetle species: A. bipunctata, C. septempunctata, and H. axyridis. As the number and inducible expression levels of AMPs increase, the greater the resistance against pathogens and parasites, and the greater the invasive performance.
The importance of diverse AMP repertoires and high induction levels became clearer when evidence emerged that insect AMPs show potentiating functional interactions against microbial pathogens (Rahnamaeian et al., 2015). For example, c-type-lysozymes from H. axyridis boost the antibacterial activity of co-expressed coleoptericins (Beckert et al., 2015), which are a family of AMPs restricted to the Coleoptera (Mylonakis et al., 2016). The diversity of the coleoptericin family has expanded during the evolution of H. axyridis (Vilcinskas et al., 2013a) and the genes are induced by up to 10,000-fold when the immune system is challenged (Vogel et al., 2017a). Taken together, these data suggest that coleoptericins play a key role in supporting the invasive performance of H. axyridis.
Figure 1 presents gene trees and maximum induction levels for the coleoptericin and defensin families in H. axyridis, C. septempunctata and A. bipunctata. The results show that H. axyridis induces these genes most strongly overall following an immune challenge, but even closely-related members of the coleoptericin and defensin families within each species display striking differences in fold-change values, showing that evolutionary relatedness is not a good predictor of AMP gene expression levels. Our latest data provide evidence of population-specific AMP gene expression, and the induction levels of individual AMPs indicate that AMP gene expression is dynamic, and may change more rapidly than previously thought (Gegner et al. unpublished results). These observations highlight the practical relevance of natural variability among AMP gene family members in terms of expression levels and induction among H. axyridis populations, which might add to the eventual success or failure of these populations when fighting off pathogens, especially in newly-colonized environments.
Changes in gene expression or gene regulation are thought to underlie many of the phenotypic differences between species, and may play an important role in adaptation to different environments. The evolution of dynamic gene expression profiles (and hence phenotypic plasticity) as different species or populations of the same species adapt to different environments is not understood in detail. Although gene expression variation in natural populations has been shown for multiple genes, the processes responsible for the maintenance of this variation as well as the benefits for the individual remain obscure.
Immunity and invasive performance
Given the abovementioned differences between the immune systems of three ladybird species differing in invasive propensity, a key question is how does a superior immune system translate into increased invasive success? One obvious explanation is that a strong immune system provides resistance against pathogens and parasites. Although some aphid symbionts have been shown to negatively impact the development and survival (Kovacs et al., 2017), indicating that certain prey-associated bacteria can evade the immune system of the Asian ladybird, both pupae and adults of H. axyridis were parasitized at a much lower rate than e.g., C. septempunctata populations from the same location (Comont et al., 2017). Accordingly, ladybird parasitoids parasitize H. axyridis only sporadically, and the beetles usually survive these attacks, with the parasitoids dying in the egg or at the larval stage. Compared to the native ladybird species C. septempunctata and A. bipunctata, H. axyridis is also more resistant to entomopathogenic nematodes and the entomopathogenic fungus Beauveria bassiana (Roy et al., 2008). Increased pathogen resistance mediated by a diverse spectrum of AMPs has also been reported in other insects challenged by pathogen-rich environments, including rat-tailed maggots of the drone fly Eristalis tenax, which can survive in contaminated aquatic habitats such as liquid manure storage pits (Altincicek and Vilcinskas, 2007), and the burying beetle Nicrophorus vespilloides, which feeds and reproduces on cadavers (Vogel et al., 2017b). However, the habitats colonized by H. axyridis are not particularly burdened with pathogens—indeed H. axyridis displaces native C. septempunctata and A. bipunctata populations that can survive perfectly well in such environments until H. axyridis arrives. So this raises the question, why has this invasive ladybird evolved a superior immune system?
We postulate that the invasive performance of H. axyridis may be directly and indirectly supported by its immune system (Figure 2). This invasive ladybird carries abundant microsporidia which it can tolerate, but which can infect and kill native ladybirds such as C. septempunctata and A. bipunctata when experimentally transferred or orally delivered upon feeding on its eggs or larvae (Vilcinskas et al., 2013b; Vogel et al., 2017a). As mentioned above, intraguild predation among predatory ladybirds may explain why H. axyridis can successfully outcompete native ladybirds (Gardiner et al., 2011). For example, A. bipunctata beetles die when feeding on H. axyridis eggs or larvae, but H. axyridis beetles suffer no ill effects when the relationship is reversed (Kajita et al., 2010). Accordingly, we found that microsporidia associated with H. axyridis kill A. bipunctata adults feeding on H. axyridis eggs (Vogel et al., 2017a). These spore-forming obligate parasites, which are distantly related to fungi, may function like biological weapons because they are tolerated by the invasive carrier, but can kill native competitors when transmitted (Vilcinskas, 2015). Our findings support previous studies highlighting the role of pathogens and parasites co-introduced with invasive species (Amsellem et al., 2017; Young et al., 2017). Taken together, these data suggest that the superior immune system in H. axyridis may have evolved so that this invasive species can safely carry microsporidia as biological weapons, unleashing them against defenseless competitors in newly-colonized habitats. It remains unclear whether the expanded spectrum of AMPs, the chemical defense molecule harmonine, or perhaps even both, contribute to the control of microsporidian propagation in the host (Vilcinskas et al., 2015). Although several mechanisms such as melanization, phagocytosis and AMPs have been discussed, there is thus far no clear evidence for any of the above being an effective defense mechanism against microsporidia (Kurtz et al., 2000; Hoch et al., 2004; Biron et al., 2005). However, the difference between being a pathogen, symbiont or mutualist can be gradual, and depends on both the host species and the environmental conditions, allowing for a remarkable degree of flexibility in host-parasite interactions. This level of flexibility was recently substantiated by identifying AMPs that maintain control over symbionts, which could otherwise turn traitor and cause disease in the host (Login et al., 2011).
Concluding remarks
The comparative analysis of the invasive ladybird H. axyridis with rather non-invasive coccinellid species enabled both to test hypotheses explaining the invasive success of particular species and to elucidate a considerable number of behavioral and immunological trait differences. However, it remains debatable which of these traits are important for the development of invasive performance. From the empiric point of view, those hypotheses appear more compelling which explain examples for biological invasions by other animals or even plants (Amsellem et al., 2017). H. axyridis has become a powerful model supporting the bioweapon theory claiming that pathogens or parasites co-introduced with invasive species can promote biological invasions if they harm or kill indigenous competitors (Vilcinskas, 2015). There are accumulating examples for biological invasions triggered by the same mechanism. The noble crayfish (Astacus astacus) declined in Europe upon the spread of the fungal pathogen Aphanomyces astaci which was co-introduced along with the signal crayfish Pacifastacus leniusculus from Northern America (Capinha et al., 2013). The replacement of native red squirrels (Sciurus vulgaris) by gray squirrels (S. carolinensis) in the United Kingdom has been attributed to Squirrel parapoxvirus co-introduced with gray squirrels from Northern America but killing only indigenous red squirrels (Collins et al., 2014).
The hypothesis that a superior immune system supports the performance of successful invaders is also reflected by examples from vertebrates. Invasive populations of the sparrows display higher surveillance against pathogens and a better immune status compared to non-invasive populations (Martin et al., 2014, 2017). A superior immune system may represent a general attribute of invasive species because it can confer resistance to both pathogens and parasites encountered in newly colonized habitats or carried and co-introduced as bioweapons against indigenous competitors.
Statements
Author contributions
All authors listed have made a substantial, direct, and intellectual contribution to the work, and approved it for publication.
Acknowledgments
The authors acknowledge funding provided by the German Research Foundation (VI 219/7-1, VO84171) and the Belgian Science Policy Office (BR/132/A1/EXOTIC-BE) via the BiodivERsA (Horizon 2020 ERA-NET COFUND scheme) grant “EXOTIC” and thank Richard M. Twyman for editing of the manuscript.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
1
AgarwalaB. K.YasudaH.KajitaY. (2003). Effect of conspecific and heterospecific feces on foraging and oviposition of two predatory ladybirds: role of fecal cues in predator avoidance. J. Chem. Ecol.29, 357–376. 10.1023/A:1022681928142
2
AlmohamadR.VerheggenF. J.FrancisF.HaubrugeE. (2010). Intraguild interactions between the predatory hoverfly Episyrphus balteatus (Diptera: Syrphidae) and the Asian ladybird, Harmonia axyridis (Coleoptera: Coccinellidae): effect of larval tracks. Eur. J. Entomol. 107, 41–45. 10.14411/eje.2010.004
3
AltincicekB.VilcinskasA. (2007). Analysis of the immune-related transcriptome from microbial stress resistant, rat-tailed maggots of the drone fly Eristalis tenax. BMC Genomics8:326. 10.1186/1471-2164-8-326
4
AlyokhinA.SewellG. (2004). Changes in a lady beetle community following the establishment of three alien species. Biol. Invasions6, 463–471. 10.1023/B:BINV.0000041554.14539.74
5
AmsellemL.BrouatC.DuronO.PorterS.VilcinskasA.FaconB. (2017). Importance of microorganisms to macroorganisms invasions: is the essential invisible to the eye? (The Little Prince, A. de Saint-Exupéry, 1943)Adv. Ecol. Res.57, 99–146. 10.1016/bs.aecr.2016.10.005
6
BahlaiC. A.Colunga-GarciaM.GageS. H.LandisD. A. (2015). The role of exotic ladybeetles in the decline of native ladybeetle populations: evidence from long-term monitoring. Biol. Invasions17, 1005–1024. 10.1007/s10530-014-0772-4
7
BeckertA.WiesnerA.BaumannA.PöppelA.-K.VogelH.VilcinskasA. (2015). Two c-type lysozymes boost the innate immune system of the invasive ladybird Harmonia axyridis. Dev. Comp. Immunol.49, 303–312. 10.1016/j.dci.2014.11.020
8
BironD. G.AgnewP.MarchéL.RenaultL.SidobreC.MichalakisY. (2005). Proteome of Aedes aegypti larvae in response to infection by the intracellular parasite vavraia culicis. Int. J. Parasit.35, 1385–1397. 10.1016/j.ijpara.2005.05.015
9
BotezatuA. I.KotseridisY.InglisD.PickeringG. J. (2013). Occurrence and contribution of alkyl methoxypyrazines in wine tainted by Harmonia axyridis and Coccinella septempunctata. J. Sci. Food Agr. 93, 803–810. 10.1002/jsfa.5800
10
Camacho-CervantesM.Ortega-IturrigaA.de-ValE. (2017). From effective biocontrol agent to successful invader: the harlequin ladybird (Harmonia axyridis) as an example of good ideas that could go wrong. Peer J. 5:e3296. 10.7717/peerj.3296
11
CamazineS.DeneubourgJ.FranksN.SneydJ.TheraulazG.BonabeauE. (2001). Self-Organization in Biological Systems. Princeton, NJ: Princeton University Press.
12
CapinhaC.LarsonE. R.TricaricoE.OldenJ. D.GherardiF. (2013). Effects of climate change, invasive species, and disease on the distribution of native European crayfishes. Conserv. Biol.27, 731–740. 10.1111/cobi.12043
13
ChiD. F.WangG. L.LiuJ. W.WuQ. Y.ZhuY. P. (2009). Antennal morphology and sensilla of Asian multicolored ladybird beetles, Harmonia axyridis Pallas (Coleoptera: Coccinellidae). Entomol. News120, 137–152. 10.3157/021.120.0203
14
CollinsL. M.WarnockN. D.ToshD. G.McInnesC.EverestD.MontgommeryW. I.et al. (2014). Squirrelpox virus: assessing prevalence, transmission and environmental degradation. PLoS ONE9:e89521. 10.1371/journal.pone.0089521
15
ComontR. F.PurseB. V.PhillipsW.KuninW. E.HansonM.LewisO. T.et al. (2017). Escape from parasitism by the invasive alien ladybird, Harmonia axyridis. Insect Conserv. Diver. 7, 334–342. 10.1111/icad.12060
16
DesneuxN.DecourtyeA.DelpuechJ.-M. (2007). The sublethal effects of pesticides on beneficial Arthropods. Ann. Rev. Entomol.52, 81–116. 10.1146/annurev.ento.52.110405.091440
17
DmitriewC.RoweL. (2007). Effects of early resource limitation and compensatory growth on lifetime fitness in the ladybird beetle (Harmonia axyridis). J. Evol. Biol.20, 1298–1310. 10.1111/j.1420-9101.2007.01349.x
18
DobzhanskyT. (1933). Geographical variation in lady beetles. Am. Nat.67, 97–126. 10.1086/280472
19
DoumbiaM.HemptinneJ.-L.DixonA. F. G. (1998). Assessment of patch quality by ladybirds: role of larval tracks. Oecologia113, 197–202. 10.1007/s004420050368
20
DudekK.DudekM.TryjanowskiP. (2015). Wind turbines as overwintering sites attractive to an invasive lady beetle, Harmonia axyridis Pallas (Coleoptera: Coccinellidae). Coleopt. Bull. 69, 665–669. 10.1649/0010-065X-69.4.665
21
DurieuxD.FassotteB.DeneubourgJ.-L.BrostauxY.VandereyckenA.JoieE.et al. (2015). Aggregation behavior of Harmonia axyridis under non-wintering conditions. Insect Sci.22, 670–678. 10.1111/1744-7917.12144
22
DurieuxD.FassotteB.FischerC.LognayG.HaubrugeE.VerheggenF. J. (2014a). Is contact between conspecifics involved in the cohesion of Harmonia axyridis (Pallas) (Coleoptera: Coccinellidae) aggregations?J. Insect Behav. 27, 1–13. 10.1007/s10905-013-9399-z
23
DurieuxD.FassotteB.VanderplanckM.BrostauxY.FischerC.LognayG.et al. (2014b). Is conspecific substrate marking a long-term external memory of previously colonized overwintering sites in Harmonia axyridis?J. Appl. Entomol. 138, 338–345. 10.1111/jen.12093
24
DurieuxD.FassotteB.VanderplanckM.DeneubourgJ.-L.FischerC.LognayG.et al. (2013). Substrate marking by an invasive Ladybeetle: seasonal changes in hydrocarbon composition and behavioral responses. PLoS ONE8:e61124. 10.1371/journal.pone.0061124
25
DurieuxD.FischerC.BrostauxY.SloggettJ. J.DeneubourgJ.-L.VandereyckenA.et al. (2012). Role of long-chain hydrocarbons in the aggregation behaviour of Harmonia axyridis (Pallas) (Coleoptera: Coccinellidae). J. Insect Physiol.58, 801–807. 10.1016/j.jinsphys.2012.03.006
26
FassotteB.FischerC.DurieuxD.LognayG.HaubrugeE.FrancisF.et al. (2014). First evidence of a volatile sex pheromone in lady beetles. PLoS ONE9:115011. 10.1371/journal.pone.0115011
27
FassotteB.FrancisF.VerheggenF. J. (2016). The scent of love: how important are semiochemicals in the sexual behavior of lady beetles?J. Pest Sci. 89, 347–358. 10.1007/s10340-016-0735-x
28
GagnonA.-È.DoyonJ.HeimpelG. E.BrodeurJ. (2011). Prey DNA detection success following digestion by intraguild predators: influence of prey and predator species. Mol. Ecol. Resources11, 1022–1032. 10.1111/j.1755-0998.2011.03047.x
29
GardinerM. M.O'NealM. E.LandisD. A. (2011). Intraguild predation and native lady beetle decline. PLoS ONE6:23576. 10.1371/journal.pone.0023576
30
GoetzD. W. (2007). Harmonia axyridis ladybug hypersensitivity in clinical allergy practice. Allergy Asthma Proc.28, 50–57. 10.2500/aap.2007.28.2956
31
HemptinneJ.LognayG.DixonA. (1998). Mate recognition in the two-spot ladybird beetle, Adalia bipunctata: role of chemical and behavioural cues. J. Insect Physiol.44, 1163–1171. 10.1016/S0022-1910(98)00081-X
32
HochG.SolterL. F.SchopfA. (2004). Hemolymph melanization and alterations in hemocyte numbers in Lymantria dispar larvae following infections with different entomopathogenic microsporidia. Entomol. Exp. Appl.113, 77–86. 10.1111/j.0013-8703.2004.00203.x
33
IngelsB.Van HasselP.Van LeeuwenT.De ClercqP. (2015). Feeding history affects intraguild interactions between Harmonia axyridis (Coleoptera: Coccinellidae) and Episyrphus balteatus (Diptera: Syrphidae). PLoS ONE10:128518. 10.1371/journal.pone.0128518
34
KajitaY.ObryckiJ. J.SlogettJ. J.HaynesK. F. (2010). Intraspecific alkaloid variation in ladybird eggs and its effects on con- and hetero-specific intraguild predators. Oecologia163, 313–322. 10.1007/s00442-009-1551-2
35
KochR. L. (2003). The multicolored Asian lady beetle, Harmonia axyridis: a review of its biology, uses in biological control, and non-target impacts. J. Insect Sci.3, 1–16. 10.1673/031.003.3201
36
KosakiA.YamaokaR. (1996). Chemical composition of footprints and cuticular lipids of three species of lady beetles. Jpn J. Appl. Entomol. Zool. 40, 47–53. 10.1303/jjaez.40.47
37
KovacsJ. L.WolfC.VoisinD.WolfS. (2017). Evidence of indirect symbiont conferred protection against the predatory lady beetle Harmonia axyridis in the pea aphid. BMC Ecol.17:26. 10.1186/s12898-017-0136-x
38
KurtzJ.NahifA. A.SauerK. P. (2000). Phagocytosis of vairimorpha sp. (microsporida, nosematidae) spores by Plutella xylostella and panorpa vulgaris hemocytes. J. Invertebr. Pathol. 75, 237–239. 10.1006/jipa.1999.4909
39
LabrieG.CoderreD.LucasÉ. (2008). Overwintering strategy of multicolored asian lady beetle (Coleoptera: Coccinellidae): cold-free space as a factor of invasive success. Ann. Entomol. Soc. Am.101, 860–866. 10.1603/0013-8746(2008)101[860:OSOMAL]2.0.CO;2
40
LambinM.FerranA.MauganK. (1996). Perception of visual information in the ladybird Harmonia axyridis Pallas. Entomol. Exp. Appl.79, 121–130. 10.1111/j.1570-7458.1996.tb00817.x
41
LambrinosJ. G. (2004). How interactions between ecology and evolution influence contemporary invasion dynamics. Ecology85, 2061–2070. 10.1890/03-8013
42
LaugierG. J. M.Le MoguédecG.TayehA.LoiseauA.OsawaN.EstoupA.et al. (2013). Increase in male reproductive success and female reproductive investment in invasive populations of the harlequin ladybird Harmonia axyridis. PLoS ONE8:77083. 10.1371/journal.pone.0077083
43
LeeK.KlasingK. (2004). A role of immunology ininvasion biology. Trends Ecol. Evol.19, 523–529. 10.1016/j.tree.2004.07.012
44
LeppanenC.AlyokhinA.GrossS. (2012). Competition for aphid prey between different lady beetle species in a laboratory arena. Psyche2012:890327. 10.1155/2012/890327
45
LeroyP. D.HeuskinS.SabriA.VerheggenF. J.FarmakidisJ.LognayG.et al. (2012b). Honeydew volatile emission acts as a kairomonal message for the Asian lady beetle Harmonia axyridis (Coleoptera: Coccinellidae). Insect Sci. 19, 498–506. 10.1111/j.1744-7917.2011.01467.x
46
LeroyP. D.SchillingsT.FarmakidisJ.HeuskinS.LognayG.VerheggenF. J.et al. (2012a). Testing semiochemicals from aphid, plant and conspecific: attraction of Harmonia axyridis. Insect Sci.19, 372–382. 10.1111/j.1744-7917.2011.01449.x
47
LoginF. H.BalmandS.VallierA.Vincent-MonégatC.VigneronA.Weiss-GayetM.et al. (2011). Antimicrobial peptides keep insect endosymbionts under control. Science334, 362–365. 10.1126/science.1209728
48
LombaertE.MalausaT.DevredR.EstoupA. (2008). Phenotypic variation in invasive and biocontrol populations of the harlequin ladybird, Harmonia axyridis. Biocontrol53, 89–102. 10.1007/s10526-007-9131-z
49
MaisinN.HassanS.HusseinM.SajapA. (1997). Mating behaviour of Menochilus sexmaculatus Fabr. (Coleoptera: Coccinellidae). Malays. Appl. Biol.26, 57–62.
50
MajerusM. E. N. (1997). How is Adalia bipunctata (Linn.) (Coleoptera: Coccinellidae) attracted to overwintering sites?Entomologist116, 212–217.
51
MartinL. B.CoonC. A. C.LieblA. L.SchreyA. W. (2014). Surveillance for microbes and range expansion in house sparrows. Proc. R. Soc. B281:20132690. 10.1098/rspb.2013.2690
52
MartinL. B.KilvitisH. J.BraceA. J.CoopeL.HaussmannM. F.MutatiA.et al. (2017). Costs of immunity and their role in the range expansion of the house sparrow in Kenya. J. Exp. Biol.220, 2228–2235. 10.1242/jeb.154716
53
MeisnerM. H.IvesA. R. (2011). Foraging efficiency and the fitness consequences of spatial marking by ladybeetle larvae. Oikos122, 1238–1246. 10.1111/j.1600-0706.2012.00224.x
54
MeisnerM. H.HarmonJ. P.HarveyC. T.IvesA. R. (2011). Intraguild predation on the parasitoid Aphidius ervi by the generalist predator Harmonia axyridis: the threat and its avoidance. Entomol. Exp. Appl. 138, 193–201. 10.1111/j.1570-7458.2010.01090.x
55
MichaudJ. P.JyotiJ. L. (2007). Repellency of conspecific and heterospecific larval residues to Hippodamia convergens (Coleoptera: Coccinellidae) ovipositing on sorghum plants. Eur. J. Entomol. 104, 399–405. 10.14411/eje.2007.059
56
MichieL. J.MallardF.MajerusM.JigginsF. (2010). Melanic through nature or nurture: genetic polymorphism and phenotypic plasticity in Harmonia axyridis. J. Evol. Biol.23, 1699–1707. 10.1111/j.1420-9101.2010.02043.x
57
MichieL. J.MassonA.WareR. L.JigginsF. M. (2011). Seasonal phenotypic plasticity: wild ladybirds are darker at cold temperatures. Evol. Ecol. 25, 1259–1268. 10.1007/s10682-011-9476-8
58
MirandeL.DesneuxN.HaramboureM.SchneiderM. I. (2015). Intraguild predation between an exotic and a native coccinellid in Argentina: the role of prey density. J. Pest Sci. 88, 155–162. 10.1007/s10340-014-0597-z
59
MondorE. B.WarrenJ. L. (2000). Unconditioned and conditioned responses to colour in the predatory coccinellid, Harmonia axyridis (Coleoptera: Coccinellidae). Eur. J. Entomol. 97, 463–467. 10.14411/eje.2000.071
60
MylonakisE.PodsiadlowskiL.MuhammedM.VilcinskasA. (2016). Diversity, evolution and medical applications of insect antimicrobial peptides. Philos. Trans. R. Soc. B371:20150290. 10.1098/rstb.2015.0290
61
NagelN.MasicA.SchurigtU.BolandW. (2015). Efficient synthesis of (R)-harmonine – the toxic principle of the multicolored Asian lady beetle (Harmonia axyridis). Org. Biomol. Chem.13, 5139–5146. 10.1039/C5OB00461F
62
NalepaC. A.KennedyG. G.BrownieC. (2004). Orientation of multicolored Asian lady beetles to buildings. Am. Entomol. 50, 165–166.
63
NalepaC. A.KennedyG. G.BrownieC. (2005). Role of visual contrast in the alighting behavior of Harmonia axyridis (Coleoptera: Coccinellidae) at overwintering sites. Environ. Entomol.34, 425–431. 10.1603/0046-225X-34.2.425
64
NalepaC. A.KiddK. A.AhlstromK. R. (1996). Biology of Harmonia axyridis (Coleoptera: Coccinellidae) in winter aggregations. Ann. Entomol. Soc. Am.89, 681–685. 10.1093/aesa/89.5.681
65
ObataS. (1986). Mechanisms of prey finding in the aphidophagous ladybird beetle, Harmonia axyridis (Coleoptera: Coccinellidae). Entomophaga31, 303–311. 10.1007/BF02373340
66
ObataS. (1987). Mating behavior and sperm transfer in the ladybird beetle, Harmonia axyridis (Pallas) (Coleoptera: Coccinellidae). Appl. Entomol. Zool. 22, 434–442. 10.1303/aez.22.434
67
ObataS. (1988). Mating refusal and its significance in females of the ladybird beetle, Harmonia axyridis. Physiol. Entomol. 13, 193–199.
68
ObataS.HidakaT. (1987). Ejection and ingestion of the spermatophore by the female ladybird beetle, Harmonia axyridis Pallas (Coleoptera: Coccinellidae). Can. Entomol.119, 603–604. 10.4039/Ent119603-6
69
Omkar PervezA. (2005). Mating behavior of an aphidophagous ladybird beetle, Propylea dissecta (Mulsant). Insect Sci.12, 37–44. 10.1111/j.1672-9609.2005.00006.x
70
OmkarJ.SahuJ.KumarG. (2013). Age specific mating incidence and reproductive behavior of the ladybird beetle, Anegleis cardoni (Weise) (Coleoptera: Coccinellidae). J. Asia Pac. Entomol.16, 263–268. 10.1016/j.aspen.2013.03.003
71
OsawaN. (2003). The influence of female oviposition strategy on sibling cannibalism in the ladybird beetle Harmonia axyridis (Coleoptera: Coccinellidae). Eur. J. Entomol. 100, 43–48. 10.14411/eje.2003.009
72
PattanayakR.MishraG.Omkar ChanotiyaC. S.RoutP. K.MohantyC. S. (2014). Does the volatile hydrocarbon profile differ between the sexes: a case study on five aphidophagous ladybirds. Arch. Insect Biochem. Physiol.87, 105–125. 10.1002/arch.21184
73
PellJ. K.BaverstockJ.RoyH. E.WareR. L.MajerusM. E. N. (2008). Intraguild predation involving Harmonia axyridis: a review of current knowledge and future perspectives. Biocontrol53, 147–168. 10.1007/s10526-007-9125-x
74
PetterssonJ.NinkovicV.GlinwoodR.BirkettM. A.PickettJ. A. (2005). Foraging in a complex environment—semiochemicals support searching behaviour of the seven spot ladybird. Eur. J. Entomol. 102, 365–370. 10.14411/eje.2005.053
75
Raak-van den BergC. L.de LangeH. J.van LenterenJ. C. (2012). Intraguild predation behaviour of ladybirds in semi-field experiments explains invasion success of Harmonia axyridis. PLoS ONE7:e40681. 10.1371/journal.pone.0040681
76
RahnamaeianM.CytrynskaM.Zdybicka-BarabasA.DobslaffK.WiesnerJ.TwymanR.et al. (2015). Insect antimicrobial peptides show potentiating functional interactions against Gram-negative bacteria. Proc. R. Soc. B282:20150293. 10.1098/rspb.2015.0293
77
ReynoldsP. G.CuddingtonK. (2012). Effects of plant gross morphology on predator searching behaviour. Environ. Entomol.41, 516–522. 10.1603/EN11179
78
RiddickE. W. (2017). Spotlight on the positive effects of the ladybird Harmonia axyridis on agriculture. Biocontrol62, 319–330. 10.1007/s10526-016-9758-8
79
RobergeC.FréchetteB.LabrieG.DumontF.LucasE. (2016). Gregarious pupation act as a defensive mechanism against cannibalism and intraguild predation. Insect Sci.23, 612–620. 10.1111/1744-7917.12209
80
RöhrichC. R.NgwaC. J.WiesnerJ.SchmidtbergH.DegenkolbT.KolleweC.et al. (2012). Harmonine, a defence compound from the harlequin ladybird, inhibits mycobacterial growth and demonstrates multi-stage antimalarial activity. Biol. Lett.8, 308–311. 10.1098/rsbl.2011.0760
81
RondoniG.AtheyK. J.HarwoodJ. D.ContiE.RicciC.ObryckiJ. J. (2015). Development and application of molecular gut-content analysis to detect aphid and coccinellid predation by Harmonia axyridis (Coleoptera: Coccinellidae) in Italy. Insect Sci.22, 719–730. 10.1111/1744-7917.12165
82
RoyH. E.BrownP. M. J.AdriaensT.BerkvensN.BorgesI.Clusella-TrullasS.et al. (2016). The harlequin ladybird, Harmonia axyridis: global perspectives on invasion history and ecology. Biol. Invasions18, 997–1044. 10.1007/s10530-016-1077-6
83
RoyH. E.BrownP. M.RotheryP.WareR. L.MajerusM. E. N. (2008). Interactions between the fungal pathogen Beauveria bassiana and three species of coccinelid: Harmonia axyridis, Coccinella septempunctata and Adalia bipunctata. Biocontrol53, 265–276. 10.1007/s10526-007-9122-0
84
RoyH. E.WajnbergE. (2008). From Biological Control to Invasion: The Ladybird Harmonia axyridis as a Model Species. Springer Netherlands. 10.1007/978-1-4020-6939-0
85
RuzickaZ. (1997). Recognition of oviposition-deterring allomones by aphidophagous predators (Neuroptera: Chrysopidae, Coleoptera: Coccinellidae). Eur. J. Entomol. 94, 431–434.
86
SantosA. A.AlmeidaL. M.Castro-GuedesC. F.PenteadoS. R. C. (2014). Life table analysis and consumption capacity for Harmonia axyridis (Coleoptera: Coccinellidae), feeding on Cinara atlantica (Hemiptera: Aphididae). Flor. Entomol. 97, 1702–1709. 10.1653/024.097.0445
87
SchallerM.NentwigW. (2000). Olfactory orientation of the sevenspot ladybird beetle, Coccinella septempunctata (Coleoptera: Coccinellidae): attraction of adults to plants and conspecific females. Eur. J. Entomol. 97, 155–160. 10.14411/eje.2000.029
88
SchmidtbergH.RöhrichC.VogelH.VilcinskasA. (2013). A switch from constitutive chemical defence to inducible innate immune responses in the invasive ladybird Harmonia axyridis. Biol. Lett.9:20130006. 10.1098/rsbl.2013.0006
89
SicsúP. R.MacedoR. H.SujiiE. R. (2015). Oviposition site selection structures niche partitioning among coccinellid species in a tropical ecosystem. Neotrop. Entomol.44, 430–438. 10.1007/s13744-015-0313-6
90
SloggettJ. J.MagroA.VerheggenF. J.HemptinneJ.-L.HutchisonW. D.RiddickE. W. (2011). The chemical ecology of Harmonia axyridis. Biocontrol56, 643–661. 10.1007/s10526-011-9376-4
91
SmithC. A.GardinerM. M. (2013). Biodiversity loss following the introduction of exotic competitors: does intraguild predation explain the decline of native lady beetles?PLoS ONE8:e84448. 10.1371/journal.pone.0084448
92
SuW.MichaudJ. P.RunzhiZ.FanZ.ShuangL. (2009). Seasonal cycles of assortative mating and reproductive behaviour in polymorphic populations of Harmonia axyridis in china. Ecol. Entomol.34, 483–494. 10.1111/j.1365-2311.2008.01075.x
93
UenoH. (1996). Estimate of multiple insemination in a natural population of Harmonia axyridis. Appl. Entomol. Zool. 31, 621–623. 10.1303/aez.31.621
94
UenoH.SatoY.TsuchidaK. (1998). Colour-associated mating success in a polymorphic ladybird beetle, Harmonia axyridis. Funct. Ecol.12, 757–761. 10.1046/j.1365-2435.1998.00245.x
95
VandereyckenA.BrostauxY.JoieE.HaubrugeE.VerheggenF. J. (2013b). Occurrence of Harmonia axyridis (Coleoptera: Coccinellidae) in field crops. Eur. J. Entomol. 110, 285–292. 10.14411/eje.2013.042
96
VandereyckenA.DurieuxD.JoieE.SloggettJ. J.HaubrugeE.VerheggenF. J. (2013a). Is the multicolored Asian ladybeetle, Harmonia axyridis, the most abundant natural enemy to aphids in agroecosystems?J. Insect Sci.. 13:158. 10.1673/031.013.15801
97
VerheggenF. J.ArnaudL.BartramS.GohyM.HaubrugeE. (2008). Aphid and plant volatiles induce oviposition in an aphidophagous hoverfly. J. Chem. Ecol.34, 301–307. 10.1007/s10886-008-9434-2
98
VerheggenF. J.FagelQ.HeuskinS.LognayG.FrancisF.HaubrugeE. (2007). Electrophysiological and behavioral responses of the Multicolored Asian Lady beetle, Harmonia axyridis Pallas, to sesquiterpene semiochemicals. J. Chem. Ecol.33, 2148–2215. 10.1007/s10886-007-9370-6
99
VerheggenF. J.HaubrugeE.MescherM. C. (2010). Alarm pheromones: chemical signaling in response to danger. Vitam. Horm. 83, 215–239. 10.1016/S0083-6729(10)83009-2
100
VilcinskasA. (2013). Evolutionary plasticity of insect immunity. J. Insect Physiol.59, 123–129. 10.1016/j.jinsphys.2012.08.018
101
VilcinskasA. (2015). Pathogens as biological weapons of invasive species. PLoS Pathog.11:e1004714. 10.1371/journal.ppat.1004714
102
VilcinskasA.MukherjeeK.VogelH. (2013a). Expansion of the antimicrobial peptide repertoire in the invasive ladybird Harmonia axyridis. Proc. R. Soc. B280:20122113. 10.1098/rspb.2012.2113
103
VilcinskasA.SchmidtbergH.EstoupA.TayehA.FaconB.VogelH. (2015). Evolutionary ecology of microsporidia associated with the invasive ladybird Harmonia axyridis. Insect Sci.22, 313–324. 10.1111/1744-7917.12159
104
VilcinskasA.StoeckerK.SchmidtbergH.RöhrichC. R.VogelH. (2013b). Invasive harlequin ladybird carries biological weapons against native competitors. Science340, 862–863. 10.1126/science.1234032
105
VogelH.SchmidtbergH.VilcinskasA. (2017a). Comparative transcriptomics of three ladybird species supports a role of immunity in invasion biology. Dev. Comp. Immunol. 67, 452–456. 10.1016/j.dci.2016.09.015
106
VogelH.ShuklaS.EnglT.WeissB.FischerR.SteigerS.et al. (2017b). The digestive and defensive basis of carcass utilization by the burying beetle and its microbiota. Nat. Comm. 8:15186. 10.1038/ncomms15186
107
WangS.MichaudJ. P.TanX. L.ZhangF.GuoX. J. (2011). The aggregation behavior of Harmonia axyridis in its native range in Northeast China. BioControl56, 193–206. 10.1007/s10526-010-9325-7
108
WareR. L.MajerusM. E. N. (2008). Intraguild predation of immature stages of British and Japanese coccinellids by the invasive ladybird Harmonia axyridis. Biocontrol53, 169–188. 10.1007/s10526-007-9135-8
109
WatanabeM. (2002). Cold tolerance and myo-inositol accumulation in overwintering adults of a lady beetle, Harmonia axyridis (Coleoptera: Coccinellidae). Eur. J. Entomol. 99, 5–9. 10.14411/eje.2002.002
110
WellsP. M.BaverstockJ.ClarkS. J.JigginsF. M.RoyH. E.PellJ. K. (2017). Determining the effects of life stage, shared prey density and host plant on intraguild predation of a native lacewing (Chrysoperla carnea) by an invasive coccinellid (Harmonia axyridis). Biocontrol62, 373–384. 10.1007/s10526-016-9775-7
111
XiaoD.TanX.WangW.ZhangF.DesneuxN.WangS. (2017). Modification of flight and locomotion performances, respiratory metabolism, and transcriptome expression in the lady beetle Harmonia axyridis through sublethal pesticide exposure. Front. Physiol.8:33. 10.3389/fphys.2017.00033
112
YasudaH.TakagiT.KogiK. (2000). Effects of conspecific and heterospecific larval tracks on the oviposition behavior of the predatory ladybird Harmonia axyridis (Coleoptera, Coccinellidae). Eur. J. Entomol. 97, 551–553. 10.14411/eje.2000.085
113
YoungH. S.ParkerI. M.GilbertG.GuerraA. S.NunnC. L. (2017). Introduced species, disease ecology, and biodiversity–disease relationships. Trends Ecol. Evol.32, 41–54. 10.1016/j.tree.2016.09.008
Summary
Keywords
biological invasions, ladybirds, innate immunity, chemical ecology, Harmonia axyridis
Citation
Verheggen FJ, Vogel H and Vilcinskas A (2017) Behavioral and Immunological Features Promoting the Invasive Performance of the Harlequin Ladybird Harmonia axyridis. Front. Ecol. Evol. 5:156. doi: 10.3389/fevo.2017.00156
Received
13 September 2017
Accepted
22 November 2017
Published
06 December 2017
Volume
5 - 2017
Edited by
Eric W. Riddick, Agricultural Research Service (USDA), United States
Reviewed by
Cesar Rodriguez-Saona, Rutgers University, United States; Jose A. Masero, Universidad de Extremadura, Spain
Updates
Copyright
© 2017 Verheggen, Vogel and Vilcinskas.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Andreas Vilcinskas andreas.vilcinskas@agrar.uni-giessen.de
This article was submitted to Behavioral and Evolutionary Ecology, a section of the journal Frontiers in Ecology and Evolution
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.