- 1Department of Pediatric Hematology and Oncology, Apollo Cancer Institutes, Chennai, India
- 2Department of Hematology, Christian Medical College, Vellore, India
- 3Pediatric Hemato-Oncology & BMT Unit, Medanta The Medicity, Gurgaon, India
- 4Department of Pediatric Hematology and Oncology, Narayana Health City, Bangalore, India
- 5Department of Pediatric Hematology and Oncology, BLK Super Specialty Hospital, New Delhi, India
- 6Department of Pediatric Hematology and Oncology, Fortis Memorial Research Institute, Gurugram, India
- 7Department of Pediatric Hematology and Oncology, Indraprastha Apollo Hospital, New Delhi, India
- 8Department of Biostatistics, Apollo Hospitals, Chennai, India
Background: Hematopoietic stem cell transplantation (HSCT) is the curative option for many primary immune deficiency disorders (PID). In the last 5 years, increased awareness, availability of diagnostics based on flow cytometry, genetic testing, improved supportive care, use of reduced toxicity conditioning, and success of haploidentical donor HSCT have improved access to HSCT for children with PID in India. We present results on children with PID who underwent HSCT across India and the factors that influenced outcome.
Patients and Methods: We collected retrospective data on the outcome of HSCT for PID from seven centers. We analyzed the impact of the type of PID, conditioning regimen, time period of HSCT- before or after January 2016, graft versus host disease prophylaxis, cause of mortality and overall survival.
Results: A total of 228 children underwent HSCT for PID at a median age of 12 months (range, 1 to 220 months) with a median follow up of 14.4 months. Infants accounted for 51.3% of the cohort and the male female ratio was 3:1. SCID (25%) and HLH (25%) were the more frequent diagnoses. Matched family donor was available in 36.4% and 44.3% children had a haploidentical HSCT. Reduced and myeloablative conditioning regimens were used with 64% children receiving a treosulfan based conditioning regimen. Peripheral blood stem cells were the predominant graft source at 69.3%. The survival in infants (60.2%) was inferior to children aged over 1 year (75.7% p value = 0.01). Children with Wiskott Aldrich syndrome (74.3%) and chronic granulomatous disease (82.6%) had the best outcomes. The survival was superior in children receiving HSCT from a matched sibling (78%) versus an alternate donor HSCT (61% p value = 0.04). In the cohort transplanted after January 2016 survival improved from 26.8% to 77.5% (p value = 0.00). Infection remains the main cause of mortality at in over 50% children. The 5-year overall survival rate was 68%.
Conclusion: Survival of children with PID undergoing HSCT in India has improved dramatically in last 5 years. Alternate donor HSCT is now feasible and has made a therapeutic option accessible to all children with PID.
Introduction
Hematopoietic stem cell transplantation (HSCT) is the curative option for many primary immune deficiency disorders (PID). Transplantation for PID requires early recognition and referral by the treating pediatrician. India has a population of over 500 million under the age of 18 years. Data from India is now available on the incidence of PID in India (1, 2). However, a significant number remain undiagnosed and would contribute to the infant and childhood mortality data. The Indian Society for Primary Immune Deficiency (ISPID) was established by a team of physicians involved in children’s care in this field in March 2011 (3). Each year, awareness programs have been arranged systematically across the country through ISPID and International collaborators like Foundation for Primary Immunodeficiency Diseases (FPID) to train pediatricians to recognize PID’s ten warning signs (4). In the last 5 years, centers of excellence for PID have been established to increase access to care across the country with diagnostic and management facilities at a subsidized cost. Access to technical information in this field, knowledge transfer from centers of excellence, and advances in molecular diagnosis has revolutionized care. Mutation screening is now possible for all children with PID. The first HSCT for PID in India was performed in 1998 at Vellore for a child with Wiskott Aldrich Syndrome (WAS). At present, there are over 75 centers in India performing HSCT with increasing numbers of pediatric transplant physicians leading the teams over the last 5 years, with over 1000 allogeneic transplant procedures performed each year. Advances in supportive care and access to generic drugs manufactured within the country have ensured that the programs are sustainable. Several governmental and non-governmental charitable organizations have joined hands to help children with rare diseases. Since January 2016, the number of HSCT for PID in India has increased due to high-resolution HLA typing, flow cytometry, molecular diagnosis, matched unrelated donor registries, haploidentical donors, and reduced toxicity conditioning medications being made available in the country. High-income countries report over 90% survival (5–7) in children undergoing HSCT and the survival % in India and other low to middle income countries ranges from 65% to 70 (8, 9).
We describe here, a multicenter study on the outcome of children with Primary immune deficiency that underwent hematopoietic stem cell transplantation across India.
Patients and Methods
We retrospectively collected outcome data of children with PID undergoing HSCT at seven HSCT centers from Chennai, Vellore, Delhi, Gurgaon, and Bangalore. Retrospective data included the age at HSCT, sex of the patient, the type of PID, donor and stem cell source, conditioning regimen, and graft-versus-host disease (GVHD) prophylaxis, type of T cell depletion, cause of death and overall survival. The cause of death was analyzed with a focus on infection-related mortality. The overall survival time was defined as the time between transplantation and death from any cause. The overall survival was analyzed with regards to age, sex, diagnosis, and type of PID.
Statistical Analysis
All normally distributed continuous variables were represented as mean ± SD. Normality of data assessed by Shapiro-Wilk’s test. Comparison of categorical variables was done by either Chi square test or Fisher’s exact test based on the number of observations. An independent sample t- test was used to compare the continuous variables between two groups. Overall survival curve was drawn and estimates were calculated by Kaplan-Meier (KM) method. Log rank test was used to compare survival between factors. Data entry was done in Microsoft Excel 2007. Data analysis and validation was done by IBM SPSS Statistics for Windows Version 25.0, Armonk, NY: IBM corp. All ‘p’ values <0.05 was considered as statistically significant. Minimum outcome data form developed by ISCTR (Indian Stem Cell Registry) was used to collect data. The hospital ethics committee approved of the study and written informed consent was obtained from all patient families.
Results
Demographic Data
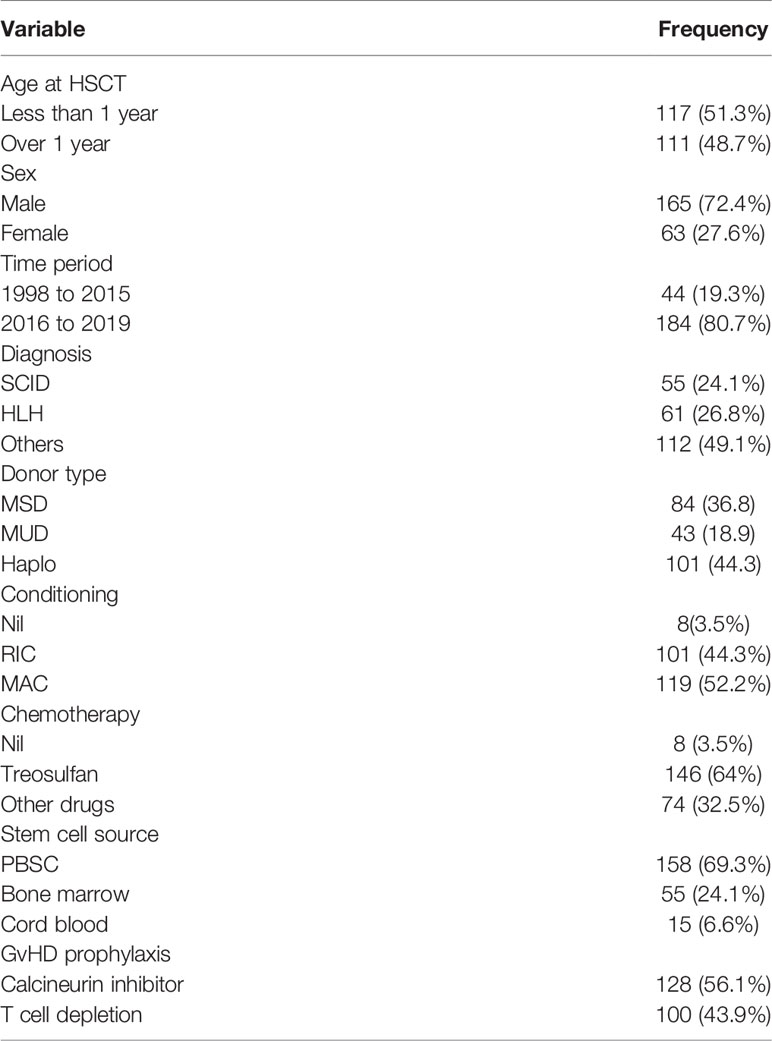
The patient characteristics with regards to demographic data have been represented in Table 1. The median age of the children who underwent HSCT is 12 months with a range from 1 to 220 months. There were more boys than girls in the cohort and girls represented only a quarter of the cohort. Half the children transplanted were under the age of 1 year. The most common indications for HSCT were SCID (n = 55, 24.2%) and familial HLH (n = 61, 26.9%) which accounted for 50% of the children. The other indications were Wiskott Aldrich syndrome (n= 35, 15.4%), chronic granulomatous disease (n = 23, 10.1%), leucocyte adhesion defect (n = 11, 4.5%), X linked agammaglobulinemia (n = 8, 3.5%), Mendelian susceptibility to mycobacterial disease (n = 7, 3.1%), Hyper IgM syndrome (n = 6, 2.6%), Hyper IgE syndrome (n = 5, 2.2%) and common variable immune deficiency (n = 2, 0.9%). Children with rare PIDs such as IPEX syndrome, heme oxygenase deficiency, LRBA deficiency and MHC Class II defects have also been transplanted in India (n = 15, 6.6%). Nearly 80% of the HSCT occurred in the period after January 2016 across all centers in India.
Details of HSCT
A fully matched family donor (n = 84, 36.8%) was available only for one third of the children and two thirds had an alternate donor source. Haploidentical family donor was the predominant donor source accounting for 101/228 (44.3%) of the transplantation. Despite access to matched unrelated donor registries, only 43 children (18.9%) had matched unrelated donor transplantation. Reduced intensity conditioning (n = 101, 43.3%) and myeloablative conditioning (n = 119, 52.2%) were equally represented and eight children had an unconditioned HSCT. Treosulfan based conditioning was used in 64% (n = 146) of the transplantation and in over 80% of the infants. Peripheral blood stem cells were used in over two thirds of the patients with a small cohort in the cord transplantation group. Notably, no cord blood transplantation has been performed after January 2016 in the country.
Graft versus host disease prophylaxis consisted predominantly of calcineurin inhibitors in 128 children (56.1%). Haploidentical HSCT was performed in 100 children (42.9%) with the use of post transplant cyclophosphamide in 57/100 (57%) and TCR alpha beta depletion in 43/100 (43%) children.
Variables Affecting HSCT Outcome
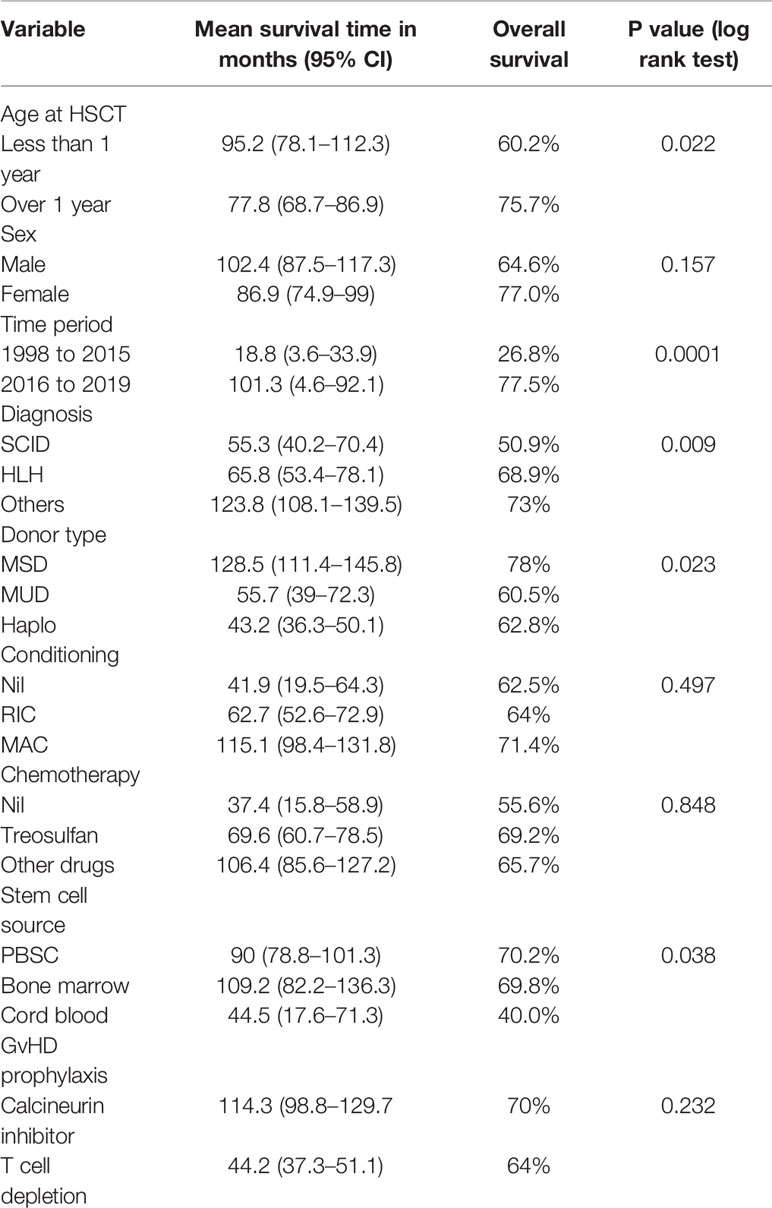
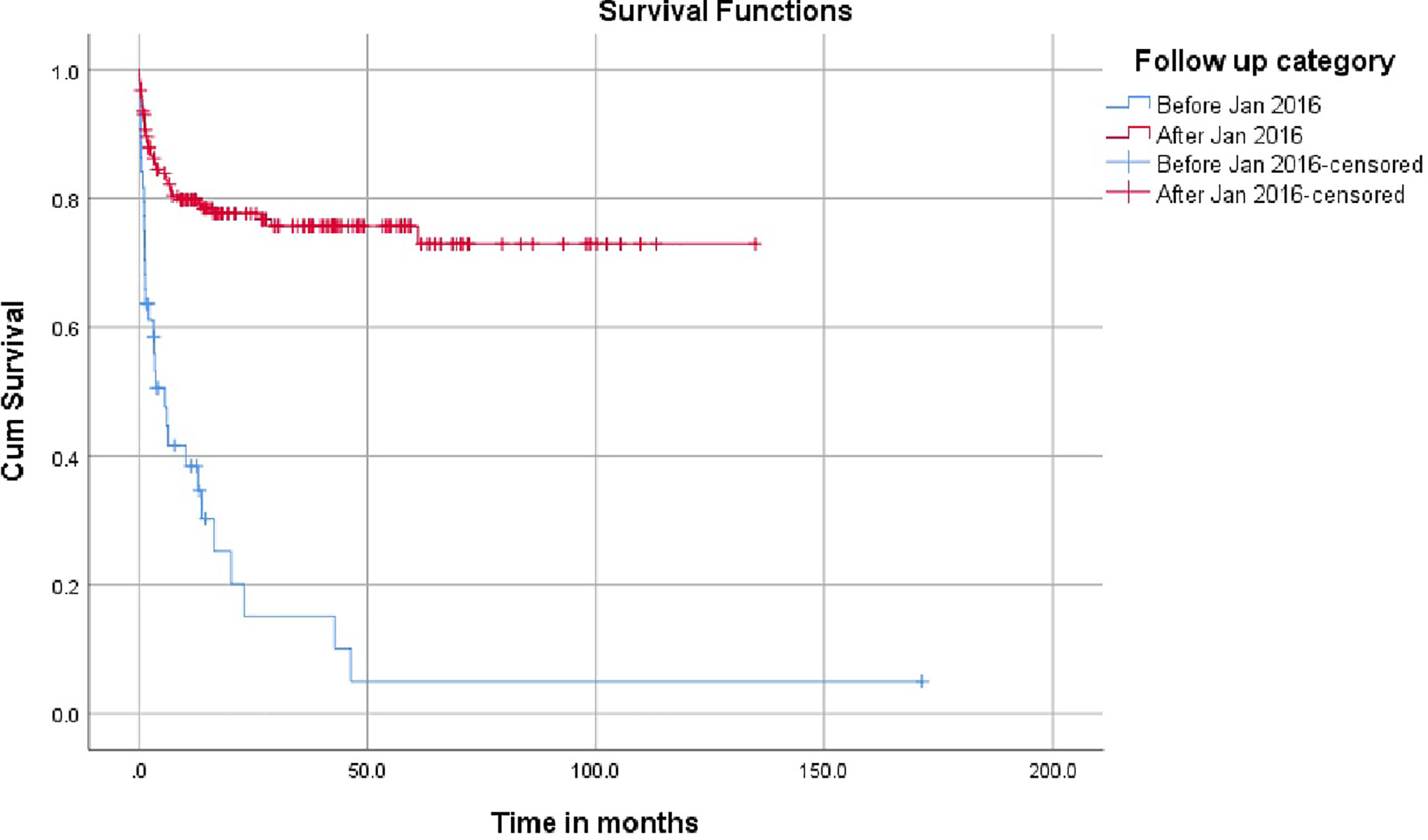
The impact of the demographics, donor source, conditioning regimen, and GvHD prophylaxis and have been represented in Table 2. The survival was superior in age over 1 year and the outcomes were similar in male and female children. Children with SCID had an inferior survival (50.9%) compared to children with Wiskott Aldrich syndrome, chronic granulomatous disease (CGD) and Hyper IgM (74.3%, 82.6% and 100% respectively). The most significant finding was the increase in the number of PID transplants from January 2016 in the country and the improved survival for children transplanted after January 2016 as shown in Figure 1.
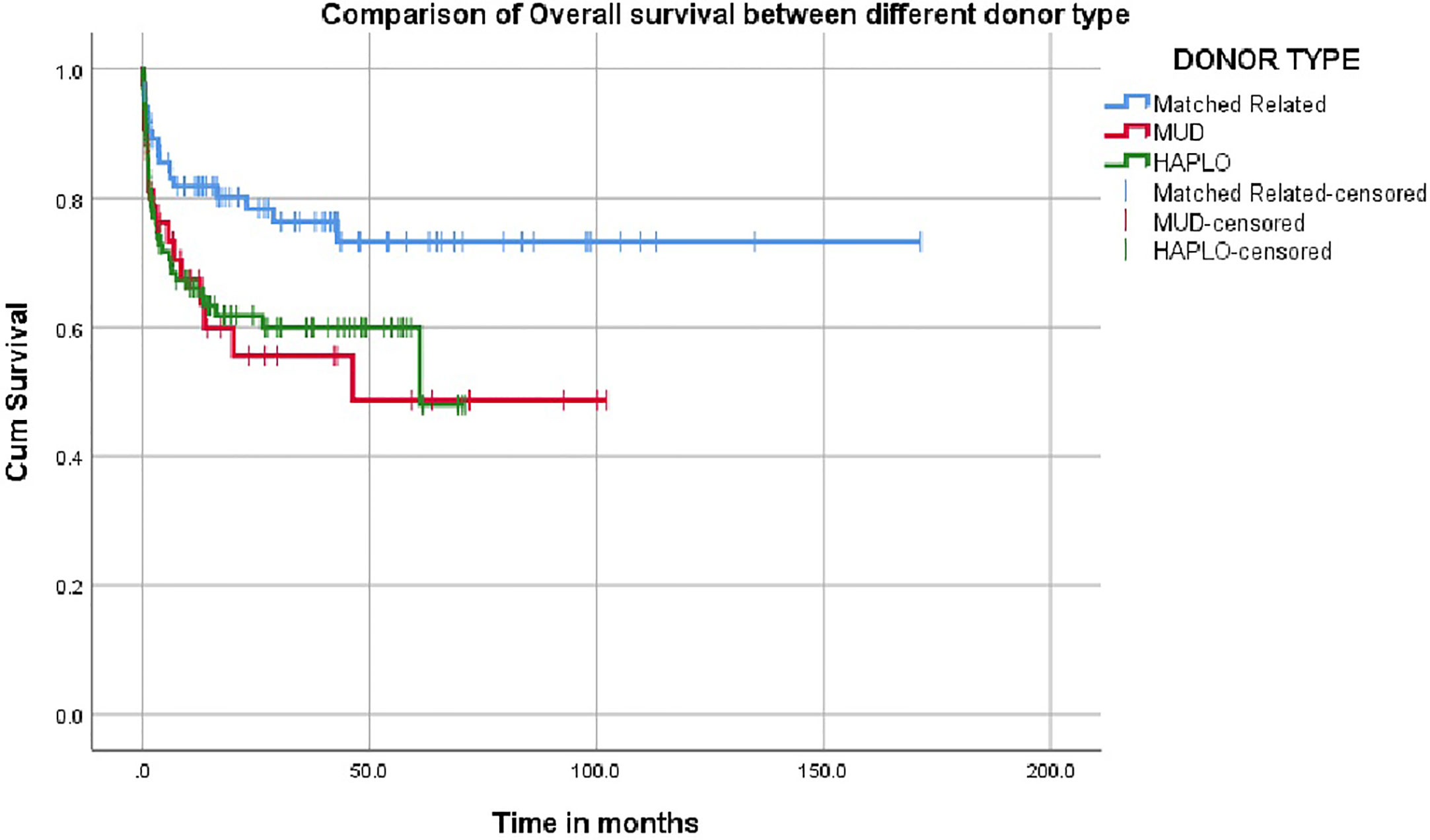
The overall survival was superior for children undergoing HSCT from matched family donors at 78% compared to alternate donor HSCT at 61% (Figure 2). In this mixed cohort, there was no difference in survival between myeloablative and reduced-intensity conditioning regimens. Cord as a source of stem cells resulted in poor survival of 40%. The survival in TCR alpha beta depletion 63.9% was equal to post-transplant cyclophosphamide at 64.9% (p value = 0.8) (Figure 3).
Infection was the leading cause of mortality at 53.9%, followed by graft versus host disease (GVHD) at 17%, graft rejection at 10.6%, and regimen related toxicity at 13.1%. A total of 11 children underwent a second HSCT for graft rejection and 8 children are alive following a second HSCT.
The most frequent presentation of disseminated BCG was a painful swelling or induration at the BCG vaccination site with axillary lymphadenitis. The other manifestations included intermittent fever, subcutaneous nodules, lesions in the liver and spleen, miliary involvement of the lung and osteomyelitis. These children were treated with a three drug regimen including isoniazid, ethambutol and levofloxacin during the transplant period. Calcineurin inhibitors interacted with rifampicin and this drug was introduced only when the immunosuppression was stopped. BCG infection is inherently resistant to pyrazinamide.
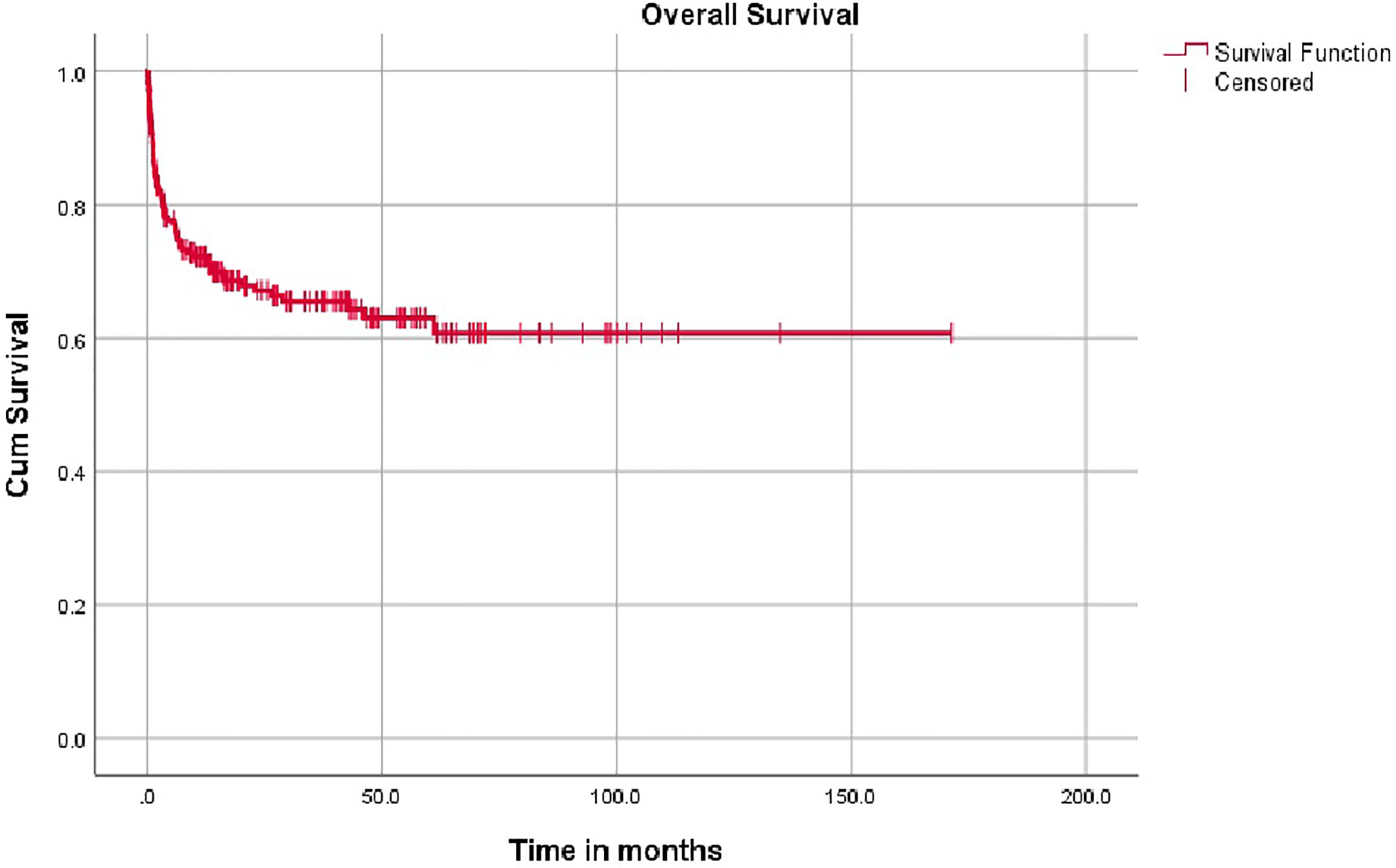
The overall mean survival time is 108 months (96.2–120.8). The 5 year overall survival rate is 68% (62%–74%) with a median follow up is 14.4 months (3.3–42.6) (IQR). Around 61% of patients had follow up of 2 years and 87.6% of patients had follow up of 5 years. Event free survival data was calculated on 118 patients with comprehensive data set was available and was 53.3%.
Discussion
Advances in the field of HSCT have resulted in improved survival for children with PID in high-income countries (10, 11). The impact of working together as a team has been demonstrated by the Primary Immune Deficiency Treatment Consortium (PIDTC) in North America, which has supported the diagnosis and management and research in the field of PID (12–15). Similar results have been seen in the EBMT (16–18) and Latin American groups (19) with data from focused groups showing improvement in overall survival. Significant challenges in early diagnosis, supportive care and financial constraints need to be overcome in low and middle income countries as reported by groups from Brazil, Jordan, China, Iran and Turkey (20–26).
In India, the field of PID has come up in leaps and bound over the past 5 years. The formation of the Indian Society for Primary Immune Deficiency (ISPID) resulted in regular meetings conducted with the help of experts from around the world. Training of physicians involved in the diagnosis and management of PID and knowledge transfer have resulted in improved survival seen from January 2016 (27). The progress has been slow but steady, and the introduction of haploidentical HSCT allowed many children to be transplanted (28–31). HSCT in India is poised at a critical juncture where collaborative trails and research is feasible with the formal launch of ISBMT – Indian Society for Blood and Marrow Transplantation. New centers focused on HSCT for PID have been established and have reported outcomes on 21 procedures over the last 2 years with an overall survival of 60% in critically ill children requiring haploidentical HSCT (oral communication Battad et al.).
This is the first report of a multicenter data on HSCT for PID in India. All children with PID now have access to molecular diagnosis and treatment (32, 33). Although only a third of the children have a matched family donor and unrelated registries like DATRI have a donor database of over 600,000 volunteers in the country, and this has made MUD transplants a reality (34). Unrelated cord transplantation has now been phased out in all HSCT centers as delayed engraftment results in unacceptably high transplant related mortality due to bacterial infections (oral communication ISBMT). Haploidentical HSCT with PTCY and TCR alpha beta depletion show promising results although the cost of HSCT is high with TCR alpha beta depletion (35, 36). Haploidentical HSCT with TCR alpha beta depletion will continue to hold a place in HSCT performed for infants and children with significant comorbidity despite the high costs involved. These numbers would also increase once newborn screening programs for SCID are established in the country.
Primary immunodeficiencies (PID) are disorders resulting from mutations in genes involved in immune host defense and immunoregulation. Replacement of hematopoietic stem cells with full or partial ablation of the recipient’s marrow with chemotherapy allows stable engraftment of donor-derived stem cell. The conditioning regimen is the key to reduce the risks of graft rejection and graft versus host disease (37–39). The use of treosulfan in 64% of the myeloablative conditioning has reduced mortality from sinusoidal obstruction syndrome. Nearly 70% of the infants undergoing HSCT were treated on the treosulfan. There are no laboratories to perform busulfan pharmacokinetics, and hence targeted busulfan therapy is not feasible and busulfan pharmacokinetics is unpredictable in infants. Generic forms of busulfan, melphalan, and thiotepa are now available in the country, which helps reduce the cost of conditioning drugs.
There has been a marked increase in the incidence of multidrug resistant bacterial infections since 2012 in the country. Multidrug resistant bacterial infections, late diagnosis and delayed referral contribute significantly to inferior outcomes in our country. Most infants have required intensive care support before transplantation due to infection as reported in published literature (40). Newer antifungal agents, such as anidulafungin and posaconazole, have helped combat fungal infections in this cohort. Ganciclovir, valganciclovir, and cidofovir are available for the treatment of CMV infection. Disseminated BCG remains a challenge as all babies are vaccinated with BCG at birth. Over 80% of the children with PID required prolonged antitubercular drugs to eliminate BCG infection with steroid cover during immune reconstitution syndrome (IRIS).
Graft versus host disease resulted in mortality in 17% of the children and graft rejection in 10.6%. Newer therapies like ruxolitinib and extracorporeal photopheresis are now available and will help reduce mortality from graft versus host disease. Follow-up of these children requires optimal communication with the shared care pediatrician for vaccination, early treatment of infections, and evaluation for late effects. Graft rejection, the need for immunoglobulin replacement and chronic graft versus host disease and poor immune reconstitution resulted in poor quality of life in about 15% of our survivors.
Female children comprised only 27.6% of the cohort and this reflects gender inequality issues that have persisted for decades in the community where expensive therapy like HSCT are preferentially offered to a male child. The survival in infants with SCID has been poor due to late presentation due to delayed diagnosis and lack of financial support. HSCT for children with XLA and CVID is on the increase in the country as lifelong immunoglobulin replacement is a major therapeutic challenge in all low income countries (41, 42). Newer options like gene therapy require specialized infrastructure and funding. Several groups in India are now engaged in gene therapy research to provide a financially viable option for families with rare genetic disorders (43, 44).
Conclusion
The survival of children with PID undergoing HSCT in India has improved dramatically in last 5 years. Alternate donor HSCT has made HSCT accessible to children with PID. The overall survival of 68% is lower than centers in high-income countries. There is an urgent need to improve our services in early diagnosis, newborn screening, supportive care and the introduction of novel technologies like indigenous gene therapy and gene editing.
Data Availability Statement
The original contributions presented in the study are included in the article/supplementary materials. Further inquiries can be directed to the corresponding author/s.
Ethics Statement
The studies involving human participants were reviewed and approved by the Apollo Speciality Hospital, Chennai, India. Written informed consent to participate in this study was provided by the participants’ legal guardian/next of kin.
Author Contributions
All authors listed have made a substantial, direct, and intellectual contribution to the work and approved it for publication. BR helped with data analysis and all other authors delivered patient care and contributed data.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
I would like to extend my sincere thanks to Dr. Sudhir Gupta, Dr. Paul Veys, Dr. Kanchan Rao and Dr. Neena Kapoor who have guided all the physicians involved in PID transplantation in India over the past 10 years and to Dr. Geeta Govindaraj and Dr. Abdulhakim Alrawas for pioneering early diagnosis and therapy for PID in their respective community.
References
1. Gupta S, Madkaikar M, Singh S, Sehgal S. Primary immunodeficiencies in India: a perspective. Ann N Y Acad Sci (2012) 1250:73–9. doi: 10.1111/j.1749-6632.2011.06353.x
2. Suri D, Rawat A, Singh S. X-linked Agammaglobulinemia. Indian J Pediatr (2016) 83(4):331–7. doi: 10.1007/s12098-015-2024-8
3. Madkaikar MR, Shabrish S, Kulkarni M, Aluri J, Dalvi A, Kelkar M, et al. Application of Flow Cytometry in Primary Immunodeficiencies: Experience From India. Front Immunol (2019) 10:1248. doi: 10.3389/fimmu.2019.01248
4. Madkaikar M, Mishra A, Ghosh K. Diagnostic Approach to Primary Immunodeficiency Disorders. Indian Pediatr (2013) 50:579–86. doi: 10.1007/s13312-013-0171-4
5. Norman M, David C, Wainstein B, Zeigler JB, Cohn R, Mitchell R, et al. Haematopoietic stem cell transplantation for primary immunodeficiency syndromes: A 5-year single-center experience. J Paediatr Child Health (2017) 53(10):988–94. doi: 10.1111/jpc.13643
6. Laberko A, Gennery AR. Clinical considerations in the hematopoietic stem cell transplant management of primary immunodeficiencies. Expert Rev Clin Immunol (2018) 14(4):297–306. doi: 10.1080/1744666X.2018.1459189
7. Gennery AR, Cant AJ. Advances in hematopoietic stem cell transplantation for primary immunodeficiency. Immunol Allergy Clin North Am (2008) 28(2):439–xi. doi: 10.1016/j.iac.2008.01.006
8. Ramya U, Dhaarani J, Meena S, Shivani P, Venkateswaran VS, Lakshmanan V, et al. Hematopoietic Stem Cell Transplantation for Primary Immunodeficiency Disorders: Experience from a Referral Center in India. Indian Pediatr (2018) 55:661–4. doi: 10.1007/s13312-018-1354-9
9. Kapoor N, Raj R. Hematopoietic Stem Cell Transplantation for Primary Immune Deficiency Disorders. Indian J Pediatr (2016) 83(5):450–4. doi: 10.1007/s12098-015-2012-z
10. Pai SY. Treatment of primary immunodeficiency with allogeneic transplant and gene therapy. Hematol Am Soc Hematol Educ Program (2019) 2019(1):457–65. doi: 10.1182/hematology.2019000052
11. Mitchell R. Hematopoietic Stem Cell Transplantation Beyond Severe Combined Immunodeficiency: Seeking a Cure for Primary Immunodeficiency. J Allergy Clin Immunol Pract (2019) 7(3):776–85. doi: 10.1016/j.jaip.2018.12.011
12. Cowan MJ. The Primary Immune Deficiency Treatment Consortium: how can it improve definitive therapy for PID? Expert Rev Clin Immunol (2016) 12(10):1007–9. doi: 10.1080/1744666X.2016.1216317
13. Griffith LM, Cowan MJ, Kohn DB, Notarangelo LD, Puck JM, Schultz KR, et al. Allogeneic hematopoietic cell transplantation for primary immune deficiency diseases: current status and critical needs. J Allergy Clin Immunol (2008) 122(6):1087–96. doi: 10.1016/j.jaci.2008.09.045
14. Griffith LM, Cowan MJ, Notarangelo LD, Kohn DB, Puck JM, Pai SY, et al. Primary Immune Deficiency Treatment Consortium (PIDTC) report. J Allergy Clin Immunol (2014) 133(2):335–47. doi: 10.1016/j.jaci.2013.07.052
15. Pai SY, Cowan MJ. Stem cell transplantation for primary immunodeficiency diseases: The North American experience. Curr Opin Allergy Clin Immunol (2014) 14(6):521–6. doi: 10.1097/ACI.0000000000000115
16. Veys P. Reduced intensity transplantation for primary immunodeficiency disorders. Pediatr Rep (2011) 3 Suppl 2(Suppl 2):e11. doi: 10.4081/pr.2011.s2.e11
17. Cavazzana M, Touzot F, Moshous D, Neven B, Blanche S, Fischer A. Stem cell transplantation for primary immunodeficiencies: the European experience. Curr Opin Allergy Clin Immunol (2014) 14(6):516–20. doi: 10.1097/ACI.0000000000000119
18. Chapel H, Prevot J, Gaspar HB, Espanol T, Bonilla FA, Solis L, et al. Primary immune deficiencies – principles of care. Front Immunol (2014) 5:627. doi: 10.3389/fimmu.2014.00627
19. Rodríguez-Baño J, Pérez-Moreno MA, Peñalva G, Garnacho-Montero J, Pinto C, Solis I, et al. Outcomes of the PIRASOA programme, an antimicrobial stewardship programme implemented in hospitals of the Public Health System of Andalusia, Spain: an ecologic study of time-trend analysis. Clin Microbiol Infect (2020) 26(3):358–65. doi: 10.1016/j.cmi.2019.07.009
20. Rousso SZ, Shamriz O, Zilkha A, Braun J, Averbuch D, Or R, et al. Hematopoietic Stem Cell Transplantations for Primary Immune Deficiencies: 3 Decades of Experience From a Tertiary Medical Center. J Pediatr Hematol Oncol (2015) 37(5):e295–300. doi: 10.1097/MPH.0000000000000352
21. Ikinciogullari A, Cagdas D, Dogu F, Tugrul T, Karasu G, Haskologlu S, et al. Clinical Features and HSCT Outcome for SCID in Turkey. J Clin Immunol (2019) 39(3):316–23. doi: 10.1007/s10875-019-00610-x
22. Wu J, Zhong W, Yin Y, Zhang H. Primary immunodeficiency disease: a retrospective study of 112 Chinese children in a single tertiary care center. BMC Pediatr (2019) 19(1):410. doi: 10.1186/s12887-019-1729-7
23. Erjaee A, Bagherpour M, Van Rooyen C, Van den Berg S, Kinnear CJ, Green RJ, et al. Primary immunodeficiency in Africa – a review. S Afr Med J (2019) 109(8b):3–11. doi: 10.7196/SAMJ.2019.v109i8b.13820
24. Patiroglu T, Akar HH, Unal E, Ozdemir MA, Karakukcu M. Hematopoietic Stem Cell Transplant for Primary Immunodeficiency Diseases: A Single-Center Experience. Exp Clin Transplant (2017) 15(3):337–43. doi: 10.6002/ect.2015.0233
25. Amayiri N, Al-Zaben A, Ghatasheh L, Frangoul H, Hussein AA. Hematopoietic stem cell transplantation for children with primary immunodeficiency diseases: single-center experience in Jordan. Pediatr Transplant (2013) 17(4):394–402. doi: 10.1111/petr.12081
26. Fernandes JF, Nichele S, Daudt LE, Tavares RB, Seber A, Kerbauy FR, et al. Transplantation of Hematopoietic Stem Cells for Primary Immunodeficiencies in Brazil: Challenges in Treating Rare Diseases in Developing Countries. J Clin Immunol (2018) 38(8):917–26. doi: 10.1007/s10875-018-0564-1
27. Yadav SP. Bone Marrow Transplantation for Primary Immune Deficiency Disorders in India: Past, Present and Future. Indian Pediatr (2018) 55(8):657–8.
28. Uppuluri R, Sivasankaran M, Patel S, Swaminathan VV, Ramanan KM, Ravichandran N, et al. Haploidentical Stem Cell Transplantation with Post-Transplant Cyclophosphamide for Primary Immune Deficiency Disorders in Children: Challenges and Outcome from a Tertiary Care Center in South India. J Clin Immunol (2019) 39(2):182–7. doi: 10.1007/s10875-019-00600-z
29. Sharma A, Rastogi N, Kapoor R, Chatterjee G, Yadav SP. Successful Haploidentical Stem Cell Transplant With Posttransplant Cyclophosphamide in Wiskott-Aldrich Syndrome With Myeloablative Conditioning [published online ahead of print, 2020 May 26]. J Pediatr Hematol Oncol (2020). doi: 10.1097/MPH.0000000000001841
30. Kohli S, Rastogi N, Nivargi S, Thakkar D, Katewa S, Yadav SP. Successful Haploidentical Stem Cell Transplant With Posttransplant Cyclophosphamide for Hemophagocytic Lymphohistiocytosis. J Pediatr Hematol Oncol (2019) 41(3):e158–60. doi: 10.1097/MPH.0000000000001265
31. Rastogi N, Katewa S, Thakkar D, Kohli S, Nivargi S, Yadav SP. Reduced-toxicity alternate-donor stem cell transplantation with posttransplant cyclophosphamide for primary immunodeficiency disorders. Pediatr Blood Cancer (2018) 65(1). doi: 10.1002/pbc.26783
32. Bandari AK, Muthusamy B, Bhat S, Govindaraj P, Rajagopalan P, Dalvi A, et al. A Novel Splice Site Mutation in IFNGR2 in Patients With Primary Immunodeficiency Exhibiting Susceptibility to Mycobacterial Diseases. Front Immunol (2019) 10:1964. doi: 10.3389/fimmu.2019.01964
33. Bucciol G, Meyts I. Recent advances in primary immunodeficiency: from molecular diagnosis to treatment. F1000Res (2020) 9:F1000 Faculty Rev-194. doi: 10.12688/f1000research.21553.1
34. Acevedo MJ, Wilder JS, Adams S, Davis J, Kelly C, Hilligoss D, et al. Outcomes of Related and Unrelated Donor Searches Among Patients with Primary Immunodeficiency Diseases Referred for Allogeneic Hematopoietic Cell Transplantation. Biol Blood Marrow Transplant (2019) 25(8):1666–73. doi: 10.1016/j.bbmt.2019.04.008
35. Shamriz O, Chandrakasan S. Update on Advances in Hematopoietic Cell Transplantation for Primary Immunodeficiency Disorders. Immunol Allergy Clin North Am (2019) 39(1):113–28. doi: 10.1016/j.iac.2018.08.003
36. Filipovich A. Hematopoietic cell transplantation for correction of primary immunodeficiencies. Bone Marrow Transplant (2008) 42(Suppl 1):S49–52. doi: 10.1038/bmt.2008.121
37. Pai SY, Logan BR, Griffith LM, Buckley RH, Parrott RE, Dvorak CC, et al. Transplantation outcomes for severe combined immunodeficiency, 2000-2009. N Engl J Med (2014) 371(5):434–46. doi: 10.1056/NEJMoa1401177
38. de la Morena MT, Nelson RP Jr. Recent advances in transplantation for primary immune deficiency diseases: a comprehensive review. Clin Rev Allergy Immunol (2014) 46(2):131–44. doi: 10.1007/s12016-013-8379-6
39. Lum SH, Hoenig M, Gennery AR, Slatter MA. Conditioning Regimens for Hematopoietic Cell Transplantation in Primary Immunodeficiency. Curr Allergy Asthma Rep (2019) 19(11):52. doi: 10.1007/s11882-019-0883-1
40. Cole TS, Johnstone IC, Pearce MS, Fulton B, Cant AJ, Gennery AR, et al. Outcome of children requiring intensive care following haematopoietic SCT for primary immunodeficiency and other non-malignant disorders. Bone Marrow Transplant (2012) 47(1):40–5. doi: 10.1038/bmt.2011.26
41. El-Sayed ZA, Abramova I, Aldave JC, Al-Herz W, Bezrodnik L, Boukari R, et al. X-linked agammaglobulinemia (XLA):Phenotype, diagnosis, and therapeutic challenges around the world. World Allergy Organ J (2019) 12(3):100018. doi: 10.1016/j.waojou.2019.100018
42. Leung D, Chua GT, Mondragon AV, Zhong Y, Nguyen-Ngoc-Quynh L, Imai K, et al. Current Perspectives and Unmet Needs of Primary Immunodeficiency Care in Asia Pacific. Front Immunol (2020) 11:1605. doi: 10.3389/fimmu.2020.01605
43. Chodisetty S, Nelson EJ. Gene therapy in India: a focus. J Biosci (2014) 39(3):537–41. doi: 10.1007/s12038-014-9431-2
Keywords: hematopoietic stem cell transplant, primary immune deficiency, conditioning, India, haploidentical
Citation: Raj R, Aboobacker FN, Yadav SP, Uppuluri R, Bhat S, Choudhry D, Dua V, Kharya G, Rastogi N, Sachdev M, Khandelwal V, Swaminathan V, Bakane A, Ramakrishnan B and George B (2021) Multicenter Outcome of Hematopoietic Stem Cell Transplantation for Primary Immune Deficiency Disorders in India. Front. Immunol. 11:606930. doi: 10.3389/fimmu.2020.606930
Received: 16 September 2020; Accepted: 03 November 2020;
Published: 08 January 2021.
Edited by:
Shobha Sehgal, Postgraduate Institute of Medical Education and Research, IndiaReviewed by:
Neena Kapoor, Children’s Hospital of Los Angeles, United StatesAnders Fasth, University of Gothenburg, Sweden
Copyright © 2021 Raj, Aboobacker, Yadav, Uppuluri, Bhat, Choudhry, Dua, Kharya, Rastogi, Sachdev, Khandelwal, Swaminathan, Bakane, Ramakrishnan and George. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Revathi Raj, cmV2YXJhakB5YWhvby5jb20=
 Revathi Raj
Revathi Raj Fouzia N. Aboobacker
Fouzia N. Aboobacker Satya Prakash Yadav
Satya Prakash Yadav Ramya Uppuluri1
Ramya Uppuluri1 Sunil Bhat
Sunil Bhat Dharma Choudhry
Dharma Choudhry Vikas Dua
Vikas Dua Gaurav Kharya
Gaurav Kharya Atish Bakane
Atish Bakane Balasubramaniam Ramakrishnan
Balasubramaniam Ramakrishnan Biju George
Biju George