- 1Wellcome Centre for Infectious Disease Research in Africa, Institute of Infectious Disease and Molecular Medicine, University of Cape Town, Cape Town, South Africa
- 2Department of Infectious Diseases, Imperial College London, London, United Kingdom
- 3Tuberculosis Laboratory, The Francis Crick Institute, London, United Kingdom
- 4Center for Infectious Disease and Vaccine Research, La Jolla Institute for Immunology, La Jolla, CA, United States
- 5Department of Medicine, University of California San Diego, La Jolla, CA, United States
- 6Immunobiology Section, Laboratory of Parasitic Diseases, National Institute of Allergy and Infectious Diseases, National Institutes of Health, Bethesda, MD, United States
- 7Inflammation and Innate Immunity Unit, Laboratory of Clinical Immunology and Microbiology, National Institute of Allergy and Infectious Diseases, National Institutes of Health, Bethesda, MD, United States
- 8T Lymphocyte Biology Section, Laboratory of Parasitic Diseases, National Institute of Allergy and Infectious Diseases, National Institutes of Health, Bethesda, MD, United States
- 9Department of Medicine, University of Cape Town, Cape Town, South Africa
- 10Division of Cardiology, Department of Medicine, University of Cape Town, Cape Town, South Africa
- 11Division of Medical Virology, Department of Pathology, University of Cape Town, Cape Town, South Africa
Studies of the immune response at the site of disease in extra-pulmonary tuberculosis (EPTB) disease are scarce. In this study, we compared the cellular profile of Mycobacterium tuberculosis (Mtb)-specific T cells in pericardial fluid and peripheral blood in patients with pericardial TB (PCTB). Whole blood and pericardial fluid (PCF) samples were collected at the time of diagnostic sampling, with repeat blood sampling after completion of anti-tubercular treatment (ATT) in 16 PCTB patients, most of them being HIV-1 infected (n=14). These samples were stimulated ex vivo and the phenotypic and functional cellular profile of PCF and blood was assessed by flow cytometry. We found that lymphocytes were the predominant cell type in PCF in PCTB, with a preferential influx of CD4 T cells. The frequencies of TNF-α producing Mtb-specific granulocytes and Mtb-specific CD4 T cells were significantly higher in PCF compared to blood. Mtb-specific CD4 T cells in PCF exhibited a distinct phenotype compared to those in blood, with greater GrB expression and lower CD27 and KLRG1 expression. We observed no difference in the production IFNγ, TNF or IL-2 by Mtb-specific CD4 T cells between the two compartments, but MIP-1β production was lower in the PCF T cells. Bacterial loads were not associated with alterations in the phenotype or function of Mtb-specific CD4 T cells. Upon ATT completion, HLA-DR, Ki-67 and GrB expression was significantly decreased, and relative IL-2 production was increased in peripheral Mtb-specific CD4 T cells. Overall, using an ex vivo assay to compare the immune response towards Mtb in PCF and in blood, we identified significant difference in the phenotypic profile of Mtb-specific CD4 T response between these two compartments. Moreover, we show that the activation profile of peripheral Mtb-specific CD4 T cells could be used to monitor treatment response in PCTB.
Introduction
Tuberculosis (TB) causes more deaths than any other bacterial disease. The WHO estimated that there were 1.2 million TB deaths in HIV uninfected individuals and 208 000 HIV/TB co-infected deaths in 2019 (1). Extra-pulmonary TB (EPTB) contributes 15% of the global TB incidence and presents diagnostic and therapeutic challenges (1). Meta-analyses of post-mortem studies have shown that in HIV/TB co-infected cases dissemination is frequent, with a pooled summary estimate of 87.9% of all TB cases (2). Importantly, 45.8% (95% CI 32.6–59.1%) of TB cases in HIV-1 infected persons were undiagnosed at time of death, highlighting the urgent need for improved rapid diagnostic tools for EPTB (2).
An important extra-pulmonary manifestation is pericardial TB (PCTB): the most common cause of pericardial disease in Africa, associated with debilitating complications and high mortality (3). PCTB disproportionately affects HIV-1 coinfected persons who are at high risk of hematogenous dissemination of TB. Although PCTB is a severe form of EPTB, our understanding of the broad range of clinicopathological PCTB phenotypes has not advanced since its first descriptions in the 1940s and 1960s (4–6). Moreover, studies of the immune response at the site of TB disease are limited.
The clinical presentation of PCTB is quite variable. At one extreme, pericardial fluid (PCF) contains a high bacillary load and represents a failure of immune control. At the other end of the spectrum, PCF can be paucibacillary with low yield of culture and polymerase chain reaction (PCR)-based tests. In this latter situation, a presumptive diagnosis of PCTB maybe made using a limited number of biomarkers with suboptimal specificity.
Despite the broad spectrum of disease phenotypes, the treatment approach is uniform and likely sub-therapeutic, as key sterilizing anti-tuberculosis drugs do not penetrate the pericardial space at sufficient concentrations to inhibit Mtb (7). This is of particular concern as both high bacillary load and a CD4 count <200 cells/mm3 in HIV-1 infected patients are predictors of mortality in PCTB (8). Interventions targeting both control of Mtb and HIV-1 are thus critical in HIV-1 coinfected persons with PCTB. However, regardless of ATT and anti-retroviral treatment (ART), severe complications such as re-accumulation of pericardial effusion after pericardiocentesis, compromised cardiac function due to tamponade and chronic pericardial inflammation leading to constriction (pericardial thickening with fibrosis) and death remain frequent (9). Thus, with few validated diagnostic, prognostic, and treatment monitoring biomarkers to guide clinicians in the management of PCTB, a better understanding of the immune response to PCTB represents an urgent unmet research priority to identify potential new clinical assessment tools (10).
Mtb control relies on a highly orchestrated immune response at the site of infection, and both innate and adaptive responses act synergistically to restrict Mtb growth. CD4 T cells, and in particular intact Th1 cellular responses are essential for control of Mtb (11). We have previously shown that a simple whole blood-based assay can be utilized to measure functional and phenotypic cellular markers that correlate with Mtb bacterial load, clinical disease severity and treatment response in PTB patients, regardless of HIV-1 status (12, 13).
Considering the importance of functional Mtb-specific CD4 T cells in Mtb control, in this study, we compared the frequency, polyfunctional capacity and phenotypic profile of Mtb-specific CD4 T cells in blood and PCF of patients with PCTB. We assessed whether the cellular profile associated with Mtb bacillary load and defined the effect of ATT on the peripheral Mtb-specific CD4 T cell response.
Material and methods
Study population
Participants with suspected PCTB recruited from the Groote Schuur Hospital Cardiology Unit. Only adults (≥ 18 years of age) who were undergoing pericardiocentesis as part of the routine management of their pericardial effusion and who had received no more than 3 doses of ATT prior were included. Pregnancy, severe anemia (Hemoglobin ≤ 7g dL-1), multi-drug resistant TB and severe concurrent opportunistic infection were exclusion criteria. All participants provided written informed consent and the study was approved by the University of Cape Town Human Research Ethics Committee (HREC: 050/2015, DMID protocol 15-0047). At the time of pericardiocentesis the study team collected PCF and paired blood samples for analysis. Only participants with definite (Mtb culture positive) or probable PCTB were included in the study. Probable PCTB was defined per criteria from Mayosi et al. where there was evidence of pericarditis with microbiologic confirmation of Mtb-infection elsewhere in the body and/or an exudative, lymphocyte predominant pericardial effusion with elevated adenosine deaminase (≥35 U L-1) (14). Study participants were followed up over the course of their ATT and up to one year.
Pericardial fluid, blood collection and stimulation assay
Pericardial fluid was obtained at the time of pericardiocentesis, placed in sterile Falcon tubes and transported to the laboratory at 4°C. Blood was collected in sodium heparin tubes at the time of pericardiocentesis. Both blood and pericardial fluid were processed within 3 hours of collection. The whole blood or whole PCF assay were adapted from the protocol described by Hanekom et al. (15). Briefly, 0.5 ml of whole blood or 1 ml of whole PCF were stimulated with a pool of 300 Mtb-derived peptides (Mtb300, 2 µg mL-1) (16) combined with γ-irradiated Mtb (H37Rv, 100 µg mL-1, obtained through BEI Resources, NIAID, NIH) at 37°C for 5 hours in the presence of the co-stimulatory antibodies, anti-CD28 and anti-CD49d (1 μg mL-1 each; BD Biosciences, San Jose, CA, USA) and Brefeldin-A (5 µg mL-1; Sigma-Aldrich, St Louis, MO, USA) and Monensin (5 µg mL-1, BD Biosciences, San Jose, CA, USA). The combination of Mtb peptides and inactivated whole bacteria allows the simultaneous detection of Mtb-specific T cell response and toll like receptor (TLR)-dependent innate response. Unstimulated cells were incubated with co-stimulatory antibodies and Brefeldin-A only. Red blood cells were then lysed in a 150 mM NH4Cl, 10 mM KHCO3, 1 mM Na4EDTA solution. Cells were then stained with a Live/Dead Near-InfraRed dye (Invitrogen, Carlsbad, CA, USA) and then fixed using a Transcription Factor Fixation buffer (eBioscience, San Diego, CA, USA), cryopreserved in freezing media (50% fetal bovine serum, 40% RPMI and 10% dimethyl sulfoxide) and stored in liquid nitrogen until use.
Cell staining and flow cytometry
Cryopreserved cells were thawed, washed and permeabilized with a Transcription Factor perm/wash buffer (eBioscience). Cells were then stained at room temperature for 45 minutes with the following antibodies: CD3 BV650 (OKT3; Biolegend, San Diego, CA, USA), CD4 BV785 (OKT4; Biolegend), CD8 BV510 (RPA-T8; Biolegend), CD27 PE-Cy5 (1A4CD27; Beckman Coulter, Brea, CA, USA), HLA-DR BV605 (L243; Biolegend), Ki67 PerCPcy5.5. (B56, BD), Granzyme B (GrB) BV421 (GB11, BD), Killer cell Lectin-like Receptor G1 (KLRG1) APC (13F12F2, eBioscience), IFN-γ BV711 (4S.B3; Biolegend), TNF-α PEcy7 (Mab11; Biolegend), IL-2 PE/Dazzle (MQ1-17H12, Biolegend), Mip-1β Alexa Fluor 488 (#24006, R&D systems, Minneapolis, MN, USA) and CD153 (R&D116614, R&D). Samples were acquired on a BD LSR-II and analyzed using FlowJo (v9.9.6, FlowJo LCC, Ashland, OR, USA). The granulocytes/monocytes population was defined based on their FSC/SSC characteristics. A positive cytokine response was defined as at least twice the background of unstimulated cells. To define the phenotype of Mtb300-specific cells, a cut-off of 30 events was used.
Statistical analysis
Statistical analysis of data was performed using GraphPad Prism (v9.1.2, San Diego, CA, USA). Differences between two groups (PCF/cult- and PCF/cult+) were evaluated using a non-parametric Mann-Whitney test. Correlations were evaluated using a non-parametric Spearman rank correlation. Differences in responses within the same individual measured in different sample types (blood vs PCF) or at two different time points (Baseline vs post-ATT) were evaluated using the Wilcoxon matched pairs signed rank test. P-values < 0.05 were considered significant.
Results
Study population
A total of 16 participants were included in the study, of which 9 were male, with a median age of 34 (Interquartile range (IQR): 28-43 years). Clinical characteristics of the study participants are listed in Table 1 and data for each individual patient presented in Supplemental Table 1. The majority (87.5% [14/16]) were HIV-1 infected, and 71.4% [10/14] had either not commenced or had defaulted ART at the time of enrolment into the study. The median peripheral CD4 count of the HIV-1 infected participants were 141 cells/mm3 (IQR: 45-188) and the median HIV-1 viral load was 47,907 mRNA copies/ml (IQR: 1,756-178,520). All participants had large pericardial effusions with the median volume of PCF aspirated being 950 ml (IQR: 480-1200). The PCF of 9 participants (56.2%) returned Mtb culture positive results, with a median time to culture positivity of 22 days (IQR: 13-26). All aspirated effusions were exudates, with high protein content and elevated adenosine deaminase (ADA) and lactate dehydrogenase (LDH). The majority showed a predominance of mononuclear cells (median: 81.7%) over polymorphonuclear cells (median: 18.3%) by PCF cell count. The diagnosis of pericardial TB in countries with high TB incidence is often made clinically as culture of pericardial fluid can take up to six weeks, and prompt treatment is required to prevent complications and mortality. Furthermore, only around 50% of those with pericardial TB will yield a positive pericardial fluid culture result in due course (17, 18). However, all participants included in our study met published diagnostic criteria for definite or probable pericardial TB (14), and was further differentiated by the Tygerberg Diagnostic Index Score (TBH DI Score). The TBH DI Score is a weighted score incorporating clinical symptoms and laboratory values designed to aid rapid diagnosis of pericardial TB (sensitivity 86%, specificity 84%) (17). The median TBH DI Score in our study was 10, with all participants scoring above the diagnostic cut-off of 6 for TB pericarditis. No difference in any of the measured clinical parameters were observed between PCTB patients with a positive PCF Mtb culture and PCTB patients with a negative PCF Mtb culture (Supplemental Table 2).
Cellular composition of pericardial fluid in PCTB
First, we examined the cellular composition of pericardial fluid (PCF) samples using flow cytometry (Figure 1A). As characteristically observed in PCTB (17, 19), we found lymphocytes to be the most abundant cell type in PCF (median 67%), while granulocytes/myeloid cells accounted for a median of 24% of the total live cell population (Figure 1B). However, it is notable that in six of the 16 studied participants, lymphocytes represented less than 50% of the total cell population. While not statistically significant, these participants tended to have a lower blood CD4 count and higher plasma HIV viral load compared to those with dominant lymphocytosis (median CD4: 92 vs 177 cells/mm3 and median HIV VL: 131,600 vs 31,540 mRNA copies/ml, respectively, data not shown). Further analysis comparing lymphocytic populations showed that the proportion of CD4 T cells was significantly higher in PCF compared to blood (median: 28.6 vs 20.8%, P = 0.029, respectively), while fewer CD3 negative cells were found in PCF compared to blood (median: 16.2 vs 28.5%, P = 0.049, respectively) (Figure 1C). This suggests a preferential recruitment of CD4 T cells into the pericardium during PCTB. This finding is further supported by a significantly higher CD4/CD8 ratio in PCF compared to blood (median: 0.8 vs 0.43, P = 0.006, respectively) (Figure 1D).
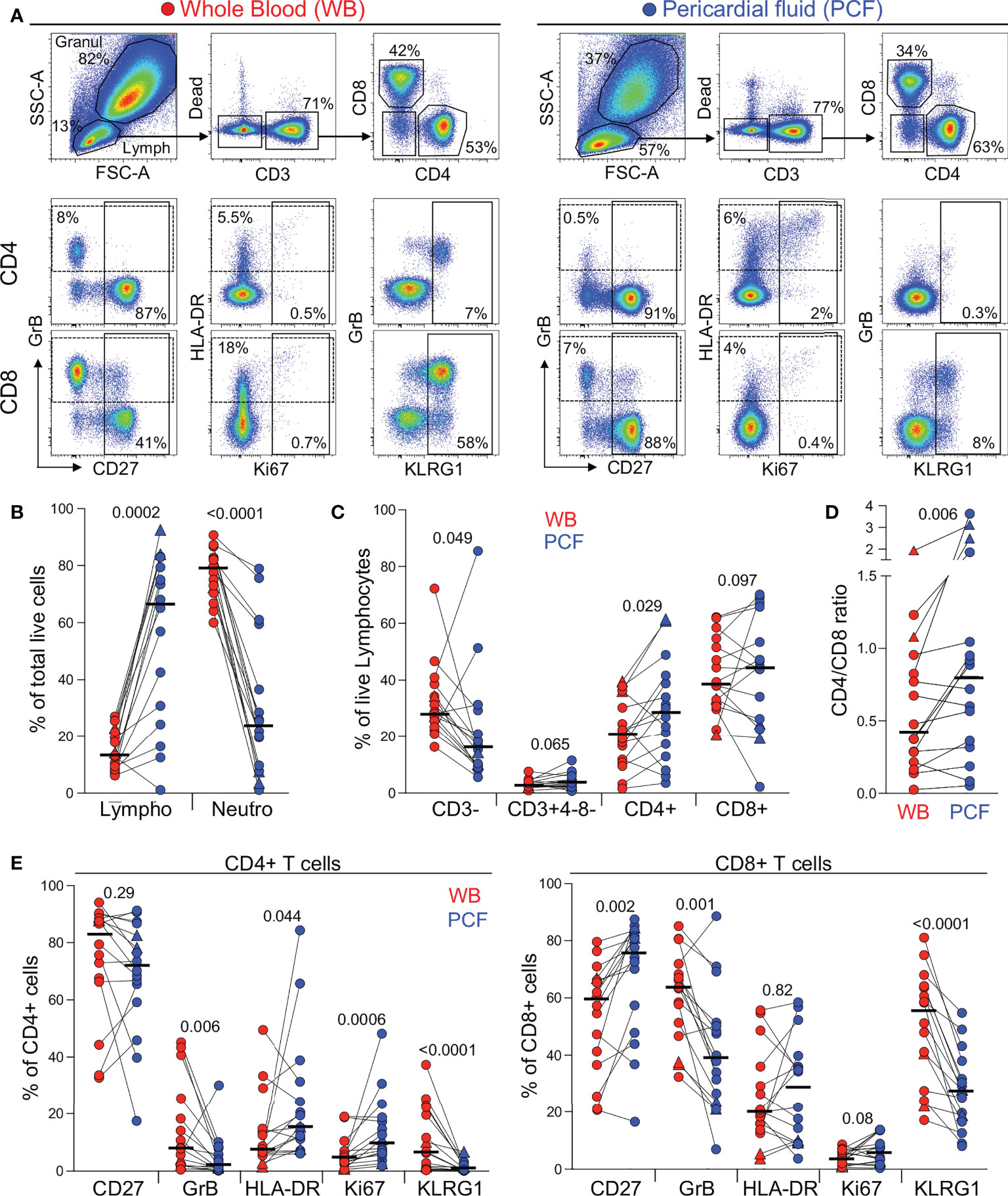
Figure 1 Comparison of the distribution of immune cells in whole blood and pericardial fluid (PCF) in patients with pericarditis. (A) Representative example of the distribution and phenotype of T cells in whole blood (red) and PCF (blue) using flow cytometry. (B) Proportion of lymphocyte (Lympho) and neutrophils (Neutro) defined according to their FSC/SSC profile and expressed as a percentage of total live cells in patients with pericarditis (n = 16). Triangles depict HIV-uninfected participants (n = 2). (C) Proportion of CD3-, CD3+CD4-CD8-, CD3+CD4+ and CD3+CD8+ cells, expressed as a percentage of total live lymphocytes. (D) CD4 to CD8 ratio in whole blood and PCF. (E) Expression of CD27, GrB, HLA-DR, Ki67 and KLRG1 in CD4 T cells (left panel) and CD8 T cells (right panel) from whole blood and PCF. Bars represent medians. Statistical comparisons were performed using a Wilcoxon rank test.
We next compared the phenotype of total CD4 and CD8 T cells between blood and PCF. CD4 T cells in PCF showed higher expression of Ki-67 and HLA-DR (median: 9.5% vs 4.4%, P = 0.0006 and 15.3% vs 7.4%, P = 0.044, respectively) and lower expression of GrB and KLRG1 (median: 1.8% vs 8%, P = 0.006 and 0.7% vs 6.5%, P < 0.0001, respectively) compared to peripheral CD4 T cells (Figure 1E, left panel). Similarly, CD8 T cells in PCF expressed less GrB and KLRG1 compared to peripheral CD8 T cells (median: 38.9% vs 63.5%, P = 0.001 and 27.4% vs 54.8%, P < 0.0001, respectively). Moreover, in PCF, CD8 T cells showed higher expression of CD27 compared to blood (median: 75.8% vs 59.5%, P = 0.002) (Figure 1E, right panel). The lower expression of KLRG1 on T cells found at the disease site is likely related to the poor ability of these cells to migrate to site of infection, as previously demonstrated in murine models (20, 21). Overall, these results suggest that infiltrating CD4 T cells were more activated and exhibited a more differentiated memory profile compared to peripheral cells. On the contrary, CD8 T cells in PCF had a lower cytotoxic potential and displayed features of an earlier differentiated phenotype compared to their blood counterparts.
Comparison of the Mtb-specific Immune response in paired blood and PCF
To measure the Mtb-specific immune response, we used an approach where ex vivo unprocessed whole blood or pericardial fluid samples. These samples were stimulated for a short time (5h) using an Mtb peptide pool combined with γ-irradiated Mtb (H37Rv) in the presence of protein transport inhibitors (added at the onset of the stimulation). The combination of peptides and whole bacteria allows the simultaneous detection of the Mtb-specific T cell response and toll like receptor (TLR)-dependent innate responses. Moreover, this type of assay has the advantage of preserving all cell subsets and soluble components present in vivo, thus maintaining a more physiologic environment (22).
First, although the flow cytometry panel used in this study was not designed to measure the innate immune response in detail, we were able to compare Mtb-induced production of TNF-α by peripheral and PCF granulocytes/myeloid cells (Figure 2A). Figure 2B shows that while Mtb stimulation led to TNF-α production in granulocytes/myeloid cells from both compartments, the frequencies of TNF-α producing cells was ~10 fold higher [IQR: 3.3 - 20.2] at the site of TB disease compared to blood (median: 1.1% vs 10.2%, respectively, P < 0.0001). TNF-α production was observed almost exclusively in the HLA-DR+ population, suggesting that Mtb-responding cells are likely classical (CD14++CD16-) or intermediate (CD14++CD16+) monocytes (23). No difference in the frequency of TNF-α responding granulocytes/myeloid cells in PCF was observed between patients with a positive or a negative PCF Mtb culture (Supplementary Figure 1A).
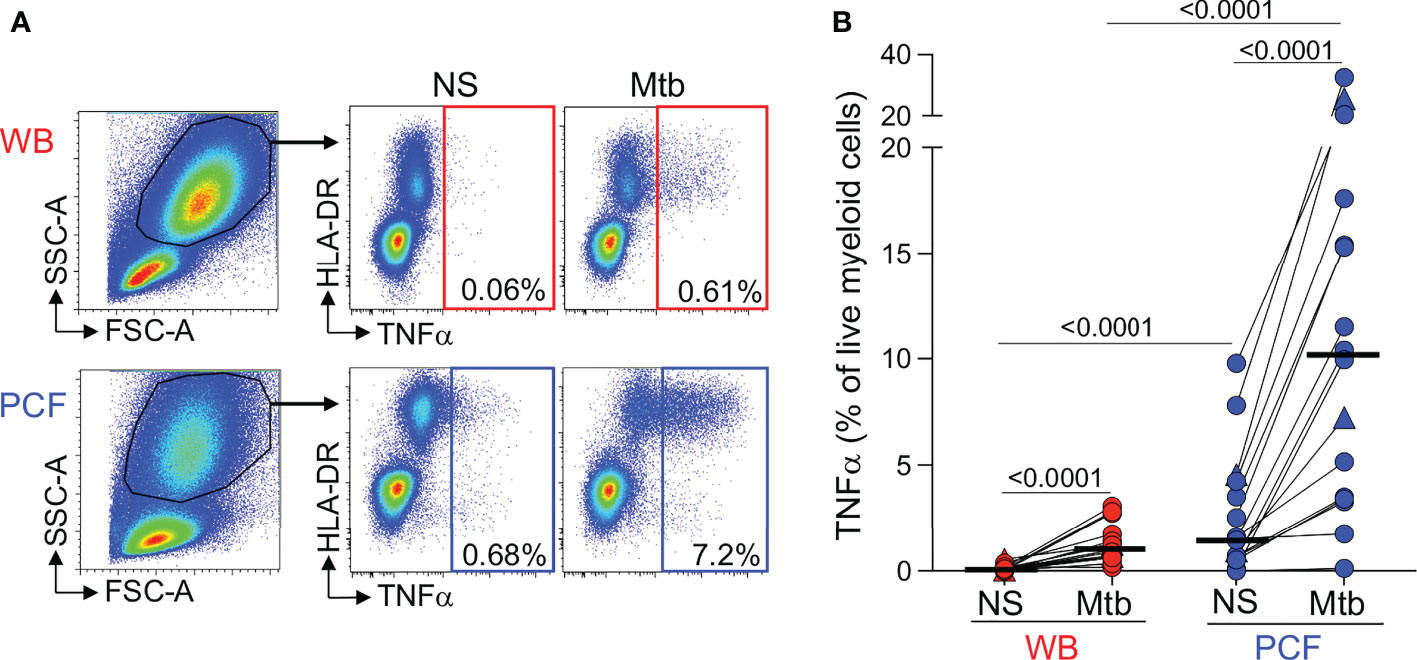
Figure 2 Comparison of TNFα production by granulocytes/myeloid cells in response to Mtb stimulation between whole blood and PCF. (A) Representative example of TNFα-producing cells in responses to Mtb antigen stimulation. Granulocytes/myeloid cells were identified based on their FCS/SSC profile. (B) Frequency of TNFα-producing granulocytes/myeloid cells in unstimulated (Uns) and Mtb-stimulated samples (Mtb) (n = 16). The triangles depict HIV-uninfected participants (n = 2). Bars represent medians. Statistical comparisons were performed using a Wilcoxon rank test for paired samples or a Mann-Whitney test for unpaired samples.
Next, to compare the adaptive immune response between blood and PCF, we measured IFN-γ, TNF-α and IL-2 expression in CD4 T cells in response to Mtb antigens (Figure 3A). Only one participant had no detectable Mtb-specific CD4 response in blood. The frequency of Mtb-specific CD4 T cells detected in PCF was significantly higher (~ 5-fold) than in blood for all measured cytokines (Figure 3B). There was no correlation between the frequency of Mtb-specific CD4 T cells in blood and PCF (P = 0.59, r = 0.14, Figure 3C). Moreover, we did not find any association between the frequency of Mtb-specific CD4 T cells and the number of absolute CD4 T cells in blood, nor between the frequencies of Mtb-specific CD4+ T cells and the percentage of total CD4 T cells in PCF (P = 0.11, r = -0.40 and P = 0.32, r = -0.26, respectively). Despite the major difference in magnitude of the Mtb-specific CD4 response between compartments, we did not observe any significant differences in polyfunctional capacity (i.e. expression of IFN-γ, TNF-α or IL-2) of Mtb-specific CD4 T cells between blood and PCF. Mtb-specific CD4 T cells exhibited a highly polyfunctional profile with a sizable proportion (median: 58% for blood and 55% for PCF) of cells co-expressing IFN-γ, TNF-α and IL-2 (Figure 3D). Additionally, Mtb culture positivity in PCF did not associate with the magnitude or polyfunctional capacities of Mtb-specific CD4 T cells in blood or in PCF (Supplementary Figure 1B, C).
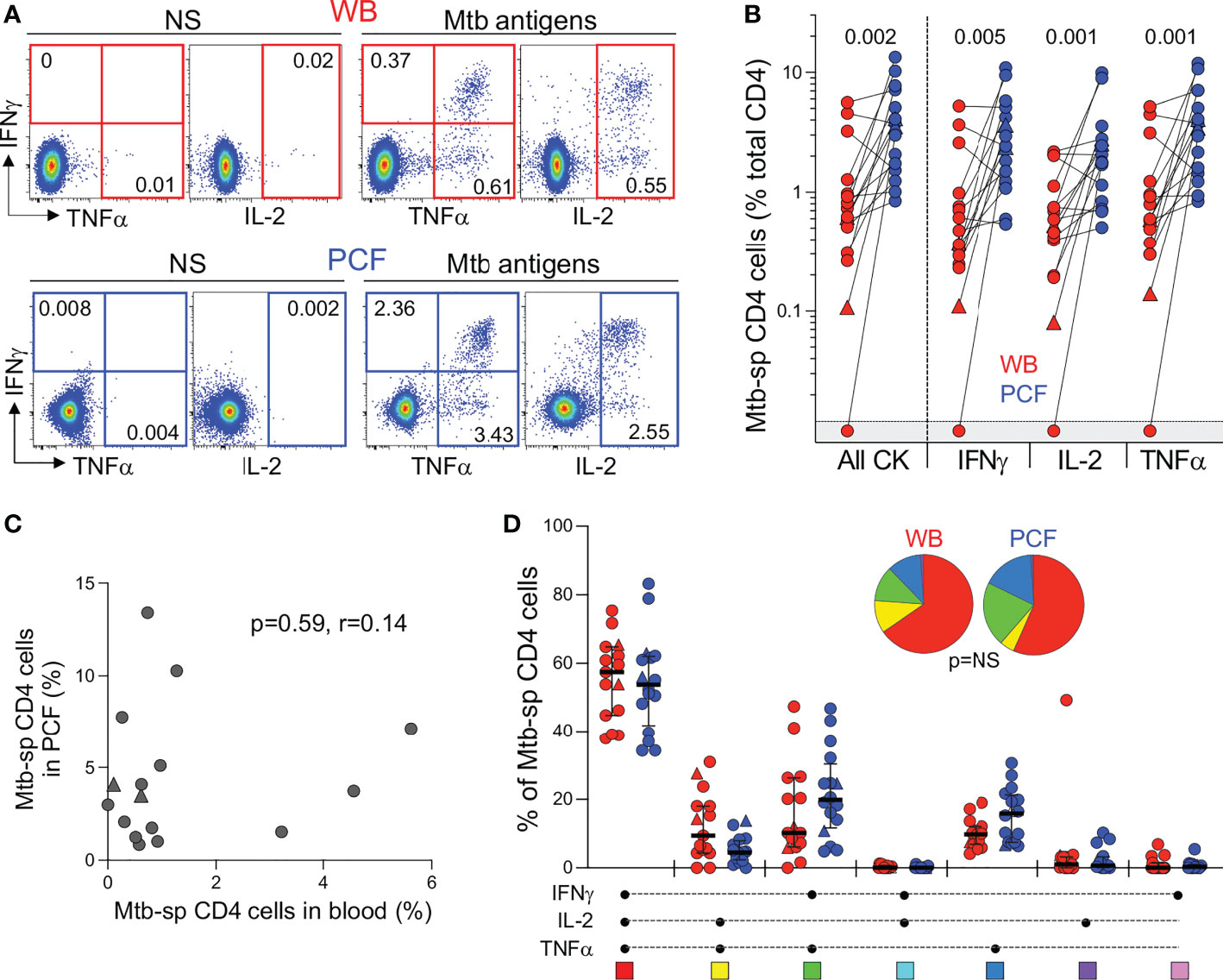
Figure 3 Comparison of the functional profile of Mtb-specific CD4 T cells between whole blood and PCF. (A) Representative example of IFNγ, IL-2, and TNFα production by CD4 T cells in unstimulated (unstim) and Mtb-stimulated whole blood and PCF samples. Number represents cytokine positive cells expressed as a percentage of total CD4 T cells. (B) Magnitude of Mtb-specific CD4 T cells in whole blood (red) and PCF (blue) (n = 16). The triangles depict HIV-uninfected participants (n = 2). Bars represent medians. Statistical comparisons were defined using a Wilcoxon rank test. (C) Correlation between the frequency of Mtb-specific CD4 T cells in blood and at site of disease (PCF). The triangles depict HIV-uninfected participants (n = 2). Correlation was tested by a two-tailed non-parametric Spearman rank test. (D) Polyfunctional profile of Mtb-specific CD4 T cells in whole blood (red) and PCF (blue). The x-axis displays all possible cytokine combinations (flavors), the composition of which is denoted with a dot for the presence of IL-2, IFNγ and TNFα. The proportion of each flavor contributing to the total Mtb-specific CD4 response per individual is shown. The median (black bar) and interquartile ranges (box) are shown. Each flavor is color‐coded, and data are summarized in the pie charts, representing the median contribution of each flavor to the total Mtb response. No statistically significant differences were observed using a Wilcoxon rank test to compare response patterns between groups and a permutation test to compare pies.
Detailed phenotyping was performed to define and compare the activation and maturation profile of Mtb-specific CD4 T cells from blood and PCF. Firstly, irrespective of the studied compartments, Mtb-specific CD4 T cells exhibited high expression of HLA-DR (ranging from 48 to 97%) and Ki67 (ranging from 3.6 to 79%) and low expression of CD27 (ranging from 9 to 46%); profiles characteristic of active TB disease, as previously reported (24–27) (Figure 4A). Paired comparison of blood and PCF revealed that Mtb-specific CD4 T cells in PCF had distinct characteristics compared to their peripheral counterparts, with elevated expression of GrB (medians: 36.6% vs 16%, respectively, P = 0.017) and lower expression of MIP-1β (36.6% vs 16%, P = 0.003), CD27 (16.2% vs 20%, P = 0.03) and KLRG1 (0.5% vs 3.8%, P = 0.002) (Figure 4A). However, despite differences in the level of expression of GrB and KLRG-1 between blood and PCF, the phenotype of Mtb-specific CD4 T cell in the blood associated with the profile of PCF Mtb-specific CD4 T cells for GrB, KLRG-1, CD153, Ki-67, and HLA-DR, with the strongest correlations being observed for GrB (P =0.0034, r = 0.74) and CD153 (P = 0.0034, r = 0.74) (Figure 4B). Additionally, the phenotypic profile of PCF Mtb-specific CD4 T cells was comparable regardless of PCF culture status (Figure 4C).
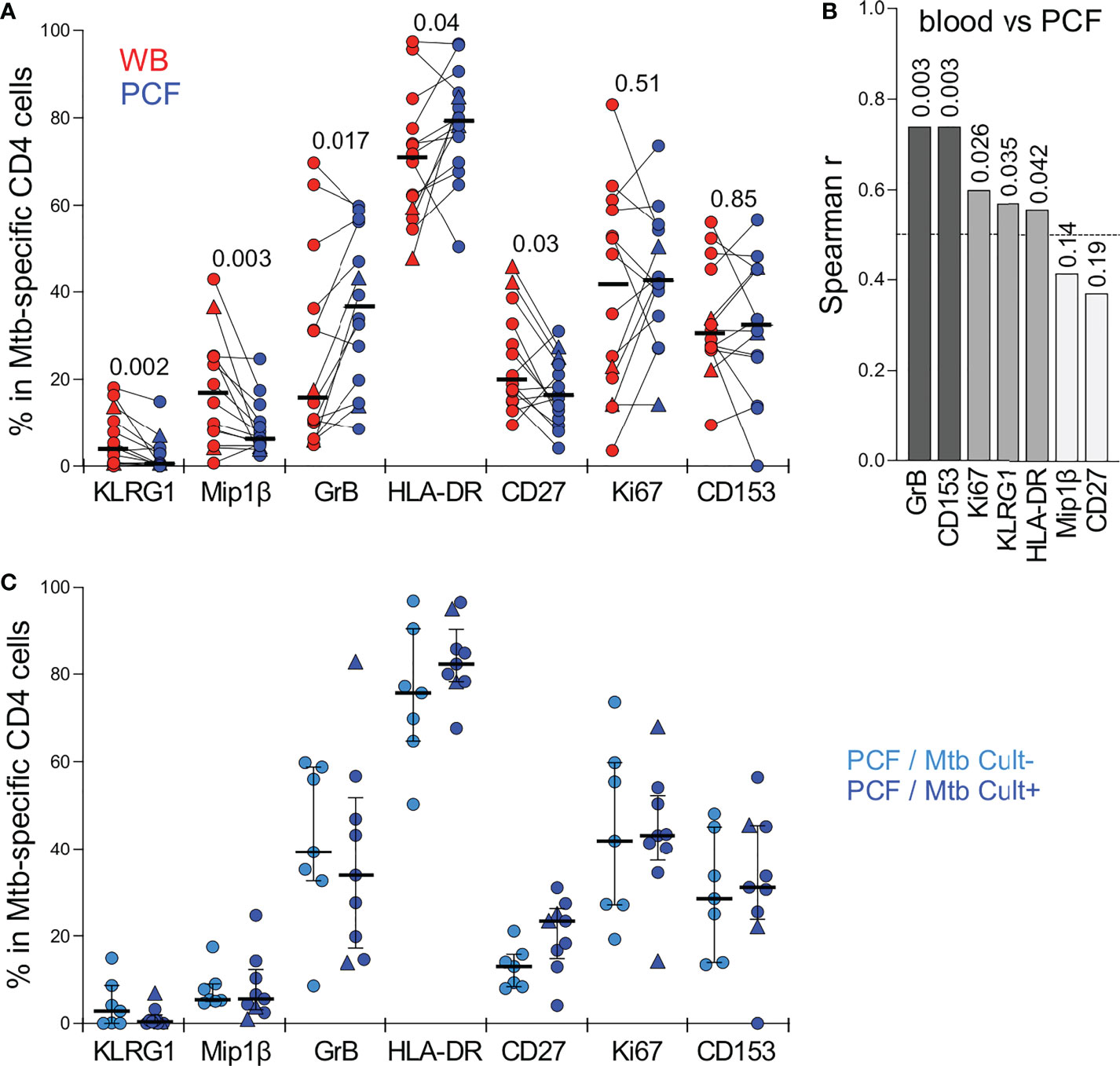
Figure 4 Comparison of the phenotypic profile of Mtb-specific CD4 T cells in blood and PCF. (A) Expression of GrB, CD27, HLA-DR, Ki67, KLRG1, CD153 and MIP1β in Mtb-specific CD4 T cells (i.e. producing any measured cytokine IFNγ, IL-2, and TNFα). Only paired samples with Mtb-specific responses >30 events are depicted (n=14). Bars represent medians. The triangles depict HIV-uninfected participants (n = 2). Statistical comparisons were performed using a Wilcoxon rank test. (B) Spearman correlation r values between indicated Mtb-specific CD4 T cell phenotype in blood and PCF. P values are indicated for each comparison. (C) Comparison of the phenotypic profile of Mtb-specific CD4 cells from PCF between patients who are PCF Mtb culture negative (PCF/Mtb Cult-, light blue, n=7) or PCF Mtb culture positive (PCF/Mtb Cult+, dark blue, n=9). Bars represent medians. Statistical comparisons were performed using the Mann-Whitney test.
We also investigated the frequency and phenotype of Mtb-specific CD8 T cells in these participants (Supplementary Figure 2). As previously described in TB patients (28), a CD8 T cell response was not observed in all participants. Mtb-specific CD8 T cells were detected in blood from 5 out of 16 patients (32%), while 8 of 16 patients (50%) had a detectable CD8 T cell response in PCF (Supplementary Figure 2B). As in the case of CD4 responses, we did not find any phenotypic differences between blood and PCF CD8 responses (Supplementary Figure 2C). Of note, compared to Mtb-specific CD4 T cells, CD8 T cells exhibited limited capacity to produce IL-2, expressed significantly higher level of GrB and Mip-1β; with CD153 expression being undetectable (Supplementary Figure 2D). The activation profile (defined with HLA-DR and Ki67 expression) were comparable between Mtb-specific CD4 and CD8 T cell responses in PCF and blood (Supplementary Figure 2D).
Impact of ATT on blood Mtb-specific CD4 T cell responses
In the context of pericardial TB (and extra-pulmonary TB, in general), the monitoring of the response to treatment is particularly challenging due to the lack of sensitive and specific tools. In this study, we defined the impact of ATT on the frequency, polyfunctional and phenotypic profile of peripheral Mtb-specific CD4 T cells pre- and post-ATT in a subset of patients (n=10) who had available follow-up blood samples at week 24 or 52. Successful ATT did not alter the magnitude of Mtb-responding CD4 T cells (Figure 5A). However, it significantly modified their functional capacity, with the proportion of IL-2 and TNF-α dual producing CD4 T cells becoming significantly expanded post-ATT, counterbalanced by a decrease of IFN-γ and TNF-α dual producing CD4 T cells (Figure 5B). More importantly, ATT led to major changes in the phenotype of the peripheral Mtb-specific CD4 response (Figures 5C, D). Post-ATT, HLA-DR, Ki67 and GrB expression by Mtb-specific CD4 T cells were significantly reduced compared to pre-treatment (medians: 15.7 vs 70% for HLA-DR; 2.8 vs 35% for Ki67 and 0.9 vs 10.9% for GrB, respectively). No changes were observed for CD153, CD27 or Mip-1β expression (Figure 5D). Overall, these results suggest that assessing the activation profile of Mtb-specific CD4 T cells could aid monitoring of treatment response in extra-pulmonary TB.
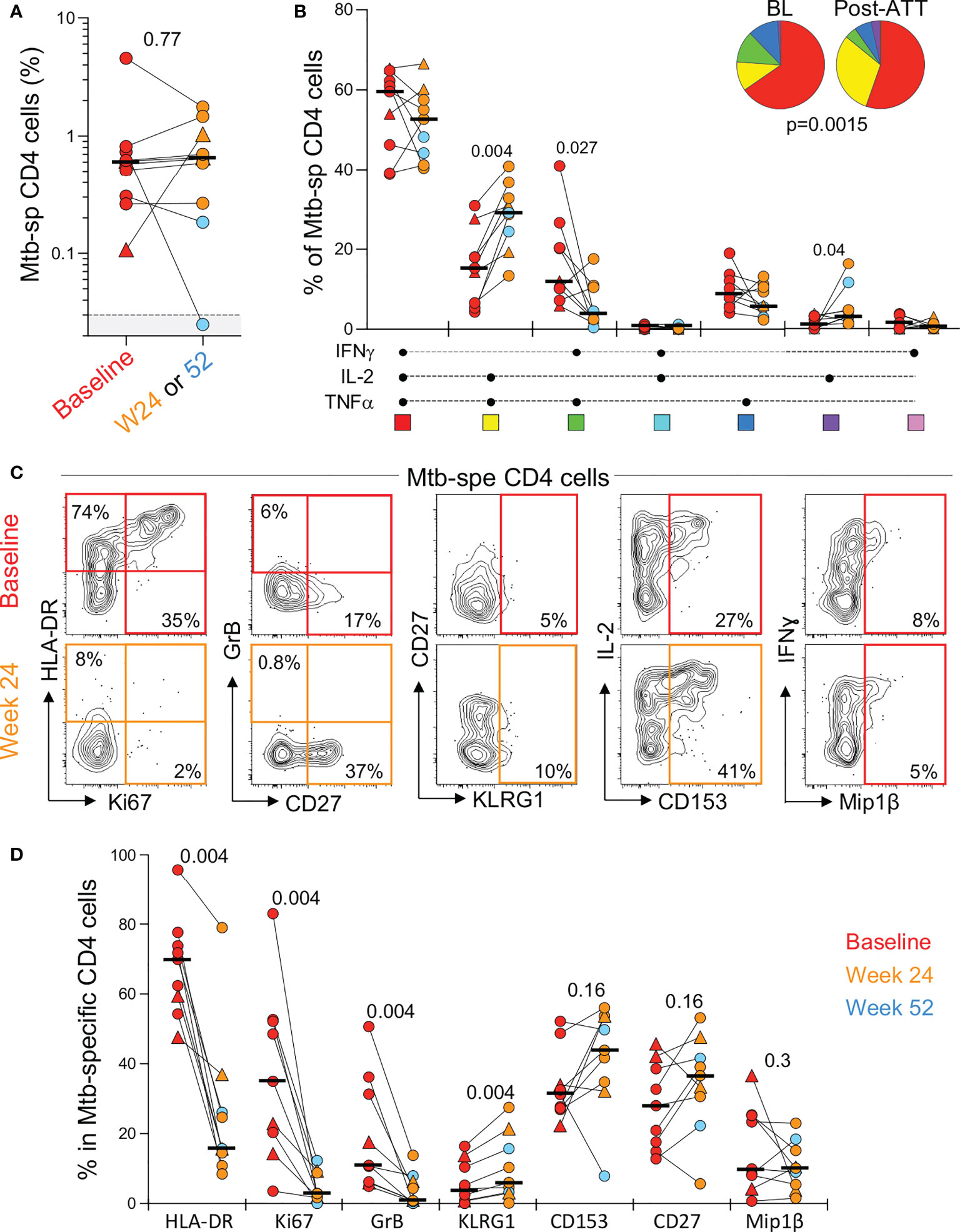
Figure 5 Evolution of the magnitude, polyfunctional capacity and phenotypic profile of Mtb-specific CD4 T cells response in whole blood from baseline (pre-ATT initiation) to ATT completion (24 or 52 weeks post-ATT). (A) Frequency of Mtb-specific CD4 T cells pre-ATT (Baseline, red) and post ATT (24 weeks, orange or 54 weeks, light blue) (n = 10 with available samples pre- and post-ATT). The triangles depict HIV-uninfected participants (n = 2). Bars represent medians. Statistical comparisons were performed using the Mann-Whitney test. (B) Polyfunctional profile of Mtb-specific CD4 T cells pre-ATT (BL) and post ATT (W24 or W52). Only paired samples, with a detectable response at both time points, are depicted (n=9). The median and interquartile ranges are shown. A Wilcoxon rank test was used to compare cytokine combination between groups and a permutation test to compare pies. (C) Representative flow plots of HLA-DR, Ki67, GrB, CD27, KLRG1, CD153 and MIP1β expression in Mtb-specific CD4 T cells pre- and post-ATT in one patient. (D) Summary graph of the expression of phenotypic markers measured. Only paired samples are depicted (n=9). Red symbols correspond to Baseline samples, orange to W24 samples and blue to W52 samples. The triangles depict HIV-uninfected participants (n = 2). Bars represent medians. Statistical comparisons were performed using the Wilcoxon rank test.
Discussion
Pericardial TB is an understudied and severe form of extra-pulmonary TB (10). Only two previous published studies have used flow cytometry to assess the cellular profile of PCF in PCTB (19, 29). Here, we demonstrate that a simple laboratory methodology (derived from a previously described whole blood assay (13)) can successfully be applied to PCF, revealing differences and similarities between the two compartments. Using this approach, we investigated the overall cellular profile of PCF, assessed whether PCF Mtb culture positivity associate with the Mtb specific cellular response and evaluated the evolution of peripheral Mtb-specific CD4 T cell responses in relation to ATT.
In accordance with previous reports, we found a significantly higher frequency of lymphocytes in PCF compared to blood, whereas neutrophils predominated in blood (17). Additionally, we observed significantly higher frequencies of CD4 T cells in PCF compared to blood, whereas preferential recruitment was not observed for CD8 T cells. This effect is likely to be even more pronounced in cohorts comprised of higher numbers of HIV-1 uninfected participants, given the findings by Reuter et al. showing lower frequencies of CD4 T cells and higher frequencies of CD8 T cells in PCF of HIV-1 co-infected compared to HIV-1 uninfected patients with PCTB (19).
Here, we report that Mtb-specific CD4 T cells are more abundant in PCF than in blood in HIV-1 infected individuals. This is in accordance with previous studies of other TB disease sites (e.g. lymph nodes, broncho-alveolar lavage and pleural fluid) (30–35). A previous study comparing PCF and blood of PCTB patients only observed increased PCF CD4 T cell responses in HIV-1 uninfected individuals using ESAT-6-specific IFN-γ ELISpot (29). This difference is likely attributable to the use of different laboratory assays (ELISpot versus flow cytometry), sample type (cryopreserved cells versus ex vivo whole blood or whole pericardial fluid) and stimuli (ESAT-6/CFP-10 versus Mtb300).
Despite markedly higher frequencies of Mtb-specific CD4 T cells in PCF compared to blood, only minor differences were observed in their functional capacity, with PCF CD4 response producing lower MIP1β and elevated GrB. Such differences may be explained by the higher degree of differentiation of Mtb-specific CD4 T cells at site compared to blood (36). The frequency of Th1 cytokine producing Mtb-specific CD4 T cells may play an important role for Mtb control, as demonstrated by a study involving low dose Mtb infection of non-human primates which compared T cell cytokine production at granuloma level in the lung to peripheral blood (37). They observed significant variability in cytokine production between different granulomas in the same animal, with sterile granulomas comprising a higher frequency of Th1 cytokine producing T cells than non-sterile granulomas. It is thus likely that the predominance of Mtb-specific CD4 T cells in PCF over blood reflects homing of these cells to the pericardium to attempt contribution to Mtb control.
We hypothesized that the presence of a higher Mtb load, as denoted by positive Mtb culture status, would enhance T cell proliferation (thus frequency), and may promote further differentiation and activation of responsive T cells (24, 38). However, we did not observe any major differences in the Mtb-specific T cell profile according to PCF Mtb culture status. It should be borne in mind that PCF Mtb culture is of poor sensitivity to diagnose PCTB (10). Nonetheless, this finding warrants further investigation, especially since time to culture positivity in PCF significantly predicts mortality in PCTB and considering the important established role of an efficient Th1 response in protecting against Mtb (8). Future studies of pericardial biopsy or pericardiectomy samples could potentially shed further light on this by analyzing T cell responses and Mtb load at the granuloma level.
Growing evidence points towards the necessity to elicit a balanced immune response to Mtb, with the predominance of either pro- or anti-inflammatory mediators being detrimental in the containment and elimination of Mtb (37, 39). IFN-γ and TNF-α are examples of essential pro-inflammatory cytokines that are required in the immune response to Mtb (40), especially through their capacity to activate infected macrophages to eliminate Mtb (41), but in excess, these cytokines may also exacerbate immunopathology (42–44). Excessive TNF-α can induce mitochondrial reactive oxygen species that lead to necrotic cell death and activate matrix metalloproteinases both of which have been associated with lung tissue damage during PTB (43, 45). We found PCF granulocytes to have significantly higher capacity to produce TNF-α in response to Mtb compared to blood granulocytes. We found no significant difference in frequency of Mtb-specific granulocytes producing TNF-α in culture positive compared to negative PCF. However, it remains to be investigated whether higher frequencies of TNF-α producing granulocytes are associated with enhanced immune pathology and complications such as pericardial fibrosis.
The clinical management of pericardial TB is challenging owing to the lack of sensitive and specific diagnostic and treatment monitoring tools. A definitive diagnosis of PCTB relies on the detection of Mtb in PCF or in pericardial tissue, and thus requires invasive sampling. The sensitivity of conventional PCF culture techniques is between 50-65%, whilst Xpert MTB/RIF of PCF has 66-66.7% sensitivity when assessed against a composite reference (46, 47). Measurement of IFN-γ in PCF has produced variable, but promising results as diagnostic tests (48, 49). However, such assays are currently limited to the research setting pending further validation against current reference standards. Moreover, few advances have recently been made in efforts to monitor treatment efficacy in EPTB. Indeed, PCTB treatment response is only assessed clinically owing to the impracticality of invasive sampling of the pericardium, contrasting with PTB where follow up sputum can be obtained to monitor Mtb clearance. While this study was not designed to identify diagnostic markers, it is worth mentioning that our findings show that i) in PCTB patients, the activation profile of peripheral Mtb-specific CD4 T cells is comparable to those observed in active PTB and ii) successfully treated PCTB is associated with significantly decreased expression of HLA-DR, Ki-67 and GrB and expansion of IL-2 producing Mtb-specific CD4 T cells compared to baseline. These results are in line with previous data showing HLA-DR as a robust biomarker to discriminate latent TB infection from PTB (13, 24, 25, 50) and EPTB (27), and to monitor PTB treatment response (13, 50, 51). However, further experiments including patients with non-tuberculous pericardial effusion will be necessary to further ascertain the specificity of HLA-DR expression on Mtb-specific T cells as a potential biomarker for PCTB diagnostic and treatment monitoring.
Our study was limited by small numbers of HIV-1 uninfected participants, reflective of how PCTB disproportionately affects HIV-1 coinfected persons in endemic countries. Further studies are needed to define whether similar T cell response patterns would be observed in immunocompetent subjects.
Nonetheless, in view of the dearth of knowledge on immune responses at TB disease site, our study demonstrates a novel and rapid experimental approach to measure the quantity and quality of T cell response ex vivo at site of disease. This technique allowed us to define key differences and similarities between the Mtb immune response in PCF and blood.
Data availability statement
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding author.
Ethics statement
The studies involving human participants were reviewed and approved by HREC: 050/2015 (University of Cape Town). The patients/participants provided their written informed consent to participate in this study.
Author contributions
CR, EB, DB, KM-B, MN, BM and RW designed the study. PH, MC, AJ and EB recruited the study participants. SR, EB, MC, and CR performed the whole blood and PCF assay. EB and CR performed the flow experiments. CR analysed and interpreted the data. ASe and CLA provided critical reagents. RW, ASh and CR obtained funding to support the project. CR and EB wrote the manuscript with all authors contributing to providing critical feedback.
Funding
This work was supported by grants from the National Institutes of Health (NIH) (U01AI115940 to RW and ASh) and (R21AI148027 to CR), the European and Developing Countries Clinical Trials Partnership EDCTP2 programme supported by the European Union (EU)’s Horizon 2020 programme (Training and Mobility Action TMA2017SF-1951-TB-SPEC to CR) and South Africa MRC Flagship Grant to MN. RW is supported by the Francis Crick Institute, which receives funds from Cancer Research UK (FC00110218), Wellcome (FC00110218) and the UK Medical Research Council (FC00110218). RW is also supported by Wellcome (203135), European and Developing clinical trials partnership (SRIA2015-1065) and the NIH (U01AI152103). EB is supported by a Harry Crossley Senior Clinical Fellowship. DB and ASh are supported by the Intramural Research Program of the National Institute of Allergy and Infectious Diseases at the NIH. This research was funded, in part, by Wellcome.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fimmu.2022.1009016/full#supplementary-material
References
1. World Health Organization. Global tuberculosis report 2019 ([amp]]lrm;2019). Available at: https://apps.who.int/iris/handle/10665/329368.
2. Gupta RK, Lucas SB, Fielding KL, Lawn SD. Prevalence of tuberculosis in post-mortem studies of HIV-infected adults and children in resource-limited settings: a systematic review and meta-analysis. AIDS (2015) 29(15):1987–2002. doi: 10.1097/QAD.0000000000000802
3. Noubiap JJ, Agbor VN, Ndoadoumgue AL, Nkeck JR, Kamguia A, Nyaga UF, et al. Epidemiology of pericardial diseases in Africa: a systematic scoping review. Heart (2019) 105(3):180–8. doi: 10.1136/heartjnl-2018-313922
4. Heimann HL, Binder S. Tuberculous pericarditis. Br Heart J (1940) 2(3):165–76. doi: 10.1136/hrt.2.3.165
6. Kreinin S. Tuberculous pericarditis observed from the stage of effusion to pericardial calcification. Am Rev Respir Dis (1960) 81:585–7. doi: 10.1164/arrd.1960.81.4.585
7. Shenje J, Ifeoma Adimora-Nweke F, Ross IL, Ntsekhe M, Wiesner L, Deffur A, et al. Poor penetration of antibiotics into pericardium in pericardial tuberculosis. EBioMedicine (2015) 2(11):1640–9. doi: 10.1016/j.ebiom.2015.09.025
8. Pasipanodya JG, Mubanga M, Ntsekhe M, Pandie S, Magazi BT, Gumedze F, et al. Tuberculous pericarditis is multibacillary and bacterial burden drives high mortality. EBioMedicine (2015) 2(11):1634–9. doi: 10.1016/j.ebiom.2015.09.034
9. Isiguzo G, Du Bruyn E, Howlett P, Ntsekhe M. Diagnosis and management of tuberculous pericarditis: What is new? Curr Cardiol Rep (2020) 22(1):2–9. doi: 10.1007/s11886-020-1254-1
10. Howlett P, Du Bruyn E, Morrison H, Godsent IC, Wilkinson KA, Ntsekhe M, et al. The immunopathogenesis of tuberculous pericarditis. Microbes Infect (2020) 22(4-5):172–81. doi: 10.1016/j.micinf.2020.02.001
11. Flynn JL, Chan J, Triebold KJ, Dalton DK, Stewart TA, Bloom BR. An essential role for interferon gamma in resistance to mycobacterium tuberculosis infection. J Exp Med (1993) 178(6):2249–54. doi: 10.1084/jem.178.6.2249
12. Du Bruyn E, Ruzive S, Lindestam Arlehamn CS, Sette A, Sher A, Barber DL, et al. Mycobacterium tuberculosis-specific CD4 T cells expressing CD153 inversely associate with bacterial load and disease severity in human tuberculosis. Mucosal Immunol (2021) 14(2):491–9. doi: 10.1038/s41385-020-0322-6
13. Riou C, Du Bruyn E, Ruzive S, Goliath RT, Lindestam Arlehamn CS, Sette A, et al. Disease extent and anti-tubercular treatment response correlates with mycobacterium tuberculosis-specific CD4 T-cell phenotype regardless of HIV-1 status. Clin Transl Immunol (2020) 9(9):e1176. doi: 10.1002/cti2.1176
14. Mayosi BM, Burgess LJ, Doubell AF. Tuberculous pericarditis. Circulation (2005) 112(23):3608–16. doi: 10.1161/CIRCULATIONAHA.105.543066
15. Hanekom WA, Hughes J, Mavinkurve M, Mendillo M, Watkins M, Gamieldien H, et al. Novel application of a whole blood intracellular cytokine detection assay to quantitate specific T-cell frequency in field studies. J Immunol Methods (2004) 291(1-2):185–95. doi: 10.1016/j.jim.2004.06.010
16. Lindestam Arlehamn CS, McKinney DM, Carpenter C, Paul S, Rozot V, Makgotlho E, et al. A quantitative analysis of complexity of human pathogen-specific CD4 T cell responses in healthy m. tuberculosis infected south africans. PloS Pathog (2016) 12(7):e1005760. doi: 10.1371/journal.ppat.1005760
17. Reuter H, Burgess L, van Vuuren W, Doubell A. Diagnosing tuberculous pericarditis. QJM (2006) 99(12):827–39. doi: 10.1093/qjmed/hcl123
18. Mayosi BM, Wiysonge CS, Ntsekhe M, Volmink JA, Gumedze F, Maartens G, et al. Clinical characteristics and initial management of patients with tuberculous pericarditis in the HIV era: the investigation of the management of pericarditis in Africa (IMPI Africa) registry. BMC Infect Dis (2006) 6:2. doi: 10.1186/1471-2334-6-2
19. Reuter H, Burgess LJ, Carstens ME, Doubell AF. Characterization of the immunological features of tuberculous pericardial effusions in HIV positive and HIV negative patients in contrast with non-tuberculous effusions. Tuberculosis (Edinb) (2006) 86(2):125–33. doi: 10.1016/j.tube.2005.08.018
20. Herndler-Brandstetter D, Ishigame H, Shinnakasu R, Plajer V, Stecher C, Zhao J, et al. KLRG1(+) effector CD8(+) T cells lose KLRG1, differentiate into all memory T cell lineages, and convey enhanced protective immunity. Immunity (2018) 48(4):716–29.e8. doi: 10.1016/j.immuni.2018.03.015
21. Sallin MA, Sakai S, Kauffman KD, Young HA, Zhu J, Barber DL. Th1 differentiation drives the accumulation of intravascular, non-protective CD4 T cells during tuberculosis. Cell Rep (2017) 18(13):3091–104. doi: 10.1016/j.celrep.2017.03.007
22. Hanekom WA, Dockrell HM, Ottenhoff TH, Doherty TM, Fletcher H, McShane H, et al. Immunological outcomes of new tuberculosis vaccine trials: WHO panel recommendations. PloS Med (2008) 5(7):e145. doi: 10.1371/journal.pmed.0050145
23. Smolen KK, Cai B, Kollmann TR. OMIP-038: Innate immune assessment with a 14 color flow cytometry panel. Cytometry A (2017) 91(10):966–8. doi: 10.1002/cyto.a.23109
24. Adekambi T, Ibegbu CC, Cagle S, Kalokhe AS, Wang YF, Hu Y, et al. Biomarkers on patient T cells diagnose active tuberculosis and monitor treatment response. J Clin Invest (2015) 125(9):1827–938. doi: 10.1172/JCI77990
25. Riou C, Berkowitz N, Goliath R, Burgers WA, Wilkinson RJ. Analysis of the phenotype of mycobacterium tuberculosis-specific CD4+ T cells to discriminate latent from active tuberculosis in HIV-uninfected and HIV-infected individuals. Front Immunol (2017) 8:968. doi: 10.3389/fimmu.2017.00968
26. Nikitina IY, Kondratuk NA, Kosmiadi GA, Amansahedov RB, Vasilyeva IA, Ganusov VV, et al. Mtb-specific CD27low CD4 T cells as markers of lung tissue destruction during pulmonary tuberculosis in humans. PloS One (2012) 7(8):e43733. doi: 10.1371/journal.pone.0043733
27. Silveira-Mattos PS, Barreto-Duarte B, Vasconcelos B, Fukutani KF, Vinhaes CL, Oliveira-de-Souza D, et al. Differential expression of activation markers by mycobacterium tuberculosis-specific CD4+ T-cell distinguishes extrapulmonary from pulmonary tuberculosis and latent infection. Clin Infect Dis (2019) 71:1905–11. doi: 10.1093/cid/ciz1070
28. Petruccioli E, Chiacchio T, Pepponi I, Vanini V, Urso R, Cuzzi G, et al. First characterization of the CD4 and CD8 T-cell responses to QuantiFERON-TB plus. J Infect (2016) 73(6):588–97. doi: 10.1016/j.jinf.2016.09.008
29. Matthews K, Ntsekhe M, Syed F, Scriba T, Russell J, Tibazarwa K, et al. HIV-1 infection alters CD4+ memory T-cell phenotype at the site of disease in extrapulmonary tuberculosis. Eur J Immunol (2012) 42(1):147–57. doi: 10.1002/eji.201141927
30. Chiacchio T, Petruccioli E, Vanini V, Butera O, Cuzzi G, Petrone L, et al. Higher frequency of T-cell response to m. tuberculosis latency antigen Rv2628 at the site of active tuberculosis disease than in peripheral blood. PloS One (2011) 6(11):e27539. doi: 10.1371/journal.pone.0027539
31. Jafari C, Ernst M, Strassburg A, Greinert U, Kalsdorf B, Kirsten D, et al. Local immunodiagnosis of pulmonary tuberculosis by enzyme-linked immunospot. Eur Respir J (2008) 31(2):261–5. doi: 10.1183/09031936.00096707
32. Nemeth J, Rumetshofer R, Winkler HM, Burghuber OC, Muller C, Winkler S. Active tuberculosis is characterized by an antigen specific and strictly localized expansion of effector T cells at the site of infection. Eur J Immunol (2012) 42(11):2844–50. doi: 10.1002/eji.201242678
33. Dieli F, Friscia G, Di Sano C, Ivanyi J, Singh M, Spallek R, et al. Sequestration of T lymphocytes to body fluids in tuberculosis: reversal of anergy following chemotherapy. J Infect Dis (1999) 180(1):225–8. doi: 10.1086/314852
34. Guyot-Revol V, Innes JA, Hackforth S, Hinks T, Lalvani A. Regulatory T cells are expanded in blood and disease sites in patients with tuberculosis. Am J Respir Crit Care Med (2006) 173(7):803–10. doi: 10.1164/rccm.200508-1294OC
35. Li L, Qiao D, Li Q, Zhang X, Lao S, Wu C, et al. Distinct polyfunctional CD4+ T cell responses to BGC, ESAT-6 and CFP-10 in tuberculous pleurisy. Tuberculosis (Edinb) (2012) 92(1):63–71. doi: 10.1016/j.tube.2011.11.004
36. Geldmacher C, Ngwenyama N, Schuetz A, Petrovas C, Reither K, Heeregrave EJ, et al. Preferential infection and depletion of mycobacterium tuberculosis-specific CD4 T cells after HIV-1 infection. J Exp Med (2010) 207(13):2869–81. doi: 10.1084/jem.20100090
37. Gideon HP, Phuah J, Myers AJ, Bryson BD, Rodgers MA, Coleman MT, et al. Variability in tuberculosis granuloma T cell responses exists, but a balance of pro- and anti-inflammatory cytokines is associated with sterilization. PloS Pathog (2015) 11(1):e1004603. doi: 10.1371/journal.ppat.1004603
38. Arrigucci R, Lakehal K, Vir P, Handler D, Davidow AL, Herrera R, et al. Active tuberculosis is characterized by highly differentiated effector memory Th1 cells. Front Immunol (2018) 9:2127. doi: 10.3389/fimmu.2018.02127
39. Etna MP, Giacomini E, Severa M, Coccia EM. Pro- and anti-inflammatory cytokines in tuberculosis: a two-edged sword in TB pathogenesis. Semin Immunol (2014) 26(6):543–51. doi: 10.1016/j.smim.2014.09.011
40. Keane J, Gershon S, Wise RP, Mirabile-Levens E, Kasznica J, Schwieterman WD, et al. Tuberculosis associated with infliximab, a tumor necrosis factor alpha-neutralizing agent. N Engl J Med (2001) 345(15):1098–104. doi: 10.1056/NEJMoa011110
41. Robinson CM, Jung JY, Nau GJ. Interferon-gamma, tumor necrosis factor, and interleukin-18 cooperate to control growth of mycobacterium tuberculosis in human macrophages. Cytokine (2012) 60(1):233–41. doi: 10.1016/j.cyto.2012.06.012
42. Tobin DM, Roca FJ, Oh SF, McFarland R, Vickery TW, Ray JP, et al. Host genotype-specific therapies can optimize the inflammatory response to mycobacterial infections. Cell (2012) 148(3):434–46. doi: 10.1016/j.cell.2011.12.023
43. Roca FJ, Whitworth LJ, Redmond S, Jones AA, Ramakrishnan L. TNF induces pathogenic programmed macrophage necrosis in tuberculosis through a mitochondrial-Lysosomal-Endoplasmic reticulum circuit. Cell (2019) 178(6):1344–61.e11. doi: 10.1016/j.cell.2019.08.004
44. Sakai S, Kauffman KD, Sallin MA, Sharpe AH, Young HA, Ganusov VV, et al. CD4 T cell-derived IFN-gamma plays a minimal role in control of pulmonary mycobacterium tuberculosis infection and must be actively repressed by PD-1 to prevent lethal disease. PloS Pathog (2016) 12(5):e1005667. doi: 10.1371/journal.ppat.1005667
45. Ravimohan S, Kornfeld H, Weissman D, Bisson GP. Tuberculosis and lung damage: from epidemiology to pathophysiology. Eur Respir Rev (2018) 27(147):170077. doi: 10.1183/16000617.0077-2017
46. Kohli M, Schiller I, Dendukuri N, Dheda K, Denkinger CM, Schumacher SG, et al. Xpert((R)) MTB/RIF assay for extrapulmonary tuberculosis and rifampicin resistance. Cochrane Database Syst Rev (2018) 8:CD012768. doi: 10.1002/14651858.CD012768.pub2
47. Hu X, Xing B, Wang W, Yang P, Sun Y, Zheng X, et al. Diagnostic values of xpert MTB/RIF, T-SPOT.TB and adenosine deaminase for HIV-negative tuberculous pericarditis in a high burden setting: a prospective observational study. Sci Rep (2020) 10(1):16325. doi: 10.1038/s41598-020-73220-y
48. Pandie S, Peter JG, Kerbelker ZS, Meldau R, Theron G, Govender U, et al. The diagnostic accuracy of pericardial and urinary lipoarabinomannan (LAM) assays in patients with suspected tuberculous pericarditis. Sci Rep (2016) 6:32924. doi: 10.1038/srep32924
49. Liu C, Cui YL, Ding CM, Wu YH, Li HL, Liu XF, et al. Diagnostic accuracy of interferon-gamma in pericardial effusions for tuberculous pericarditis: a meta-analysis. J Thorac Dis (2018) 10(2):854–60. doi: 10.21037/jtd.2017.12.107
50. Musvosvi M, Duffy D, Filander E, Africa H, Mabwe S, Jaxa L, et al. T-Cell biomarkers for diagnosis of tuberculosis: candidate evaluation by a simple whole blood assay for clinical translation. Eur Respir J (2018) 51(3):1800153. doi: 10.1183/13993003.00153-2018
Keywords: pericardial tuberculosis, site of disease, CD4 response, treatment response, whole blood, pericardial fluid
Citation: Du Bruyn E, Ruzive S, Howlett P, Cerrone M, Jacobs AJ, Arlehamn CSL, Sette A, Sher A, Mayer-Barber KD, Barber DL, Mayosi B, Ntsekhe M, Wilkinson RJ and Riou C (2022) Comparison of the frequency and phenotypic profile of Mycobacterium tuberculosis-specific CD4 T cells between the site of disease and blood in pericardial tuberculosis. Front. Immunol. 13:1009016. doi: 10.3389/fimmu.2022.1009016
Received: 01 August 2022; Accepted: 26 October 2022;
Published: 11 November 2022.
Edited by:
Christoph Hölscher, Research Center Borstel (LG), GermanyReviewed by:
Christof Geldmacher, LMU Munich University Hospital, GermanyAnand Kumar Maurya, India Institute of Medical Sciences Bhopal, India
Copyright © 2022 Du Bruyn, Ruzive, Howlett, Cerrone, Jacobs, Arlehamn, Sette, Sher, Mayer-Barber, Barber, Mayosi, Ntsekhe, Wilkinson and Riou. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Catherine Riou, Y3IucmlvdUB1Y3QuYWMuemE=
†These authors have contributed equally to this work
‡Deceased: Bongani Mayosi
 Elsa Du Bruyn
Elsa Du Bruyn Sheena Ruzive1
Sheena Ruzive1 Cecilia S. Lindestam Arlehamn
Cecilia S. Lindestam Arlehamn Alessandro Sette
Alessandro Sette Alan Sher
Alan Sher Katrin D. Mayer-Barber
Katrin D. Mayer-Barber Daniel L. Barber
Daniel L. Barber Bongani Mayosi
Bongani Mayosi Robert J. Wilkinson
Robert J. Wilkinson Catherine Riou
Catherine Riou