- 1Senior Department of Hematology, The Fifth Medical Center of People's Liberation Army (PLA), General Hospital, Beijing, China
- 2Department of Hematology, The Sixth Medical Center of People's Liberation Army (PLA), General Hospital, Beijing, China
- 3Department of Hematology, The First Affiliated Hospital of China Medical University, Shenyang, China
Background and aim: Given the paucity of evidence-based treatment recommendations, the most appropriate first-line regimen for adult Burkitt lymphoma is currently undefined. We aimed to identify the optimal treatment regimen containing rituximab for adult Burkitt lymphoma patients.
Methods: The PubMed, Embase, Web of Science, and Cochrane databases were searched in December 2021 (10). We included all studies for the treatment of Burkitt lymphoma including rituximab. We excluded studies of patients aged ≤14 years old and those with sample numbers ≤10 patients. Random-effects models were used to compare different chemotherapy regimens regarding estimated 2-year overall survival (OS) rate, 2-year progression-free survival (PFS) rate, and overall response rate (ORR).
Results: A total of 17 studies were included in this meta-analysis and divided into four groups: CODOX-M/IVAC, DA-EPOCH, GMALL-B-ALL/NHL2002, and Hyper-CVAD. DA-EPOCH was associated with a significantly higher 2-year OS rate [0.95, 95% confidence interval (CI) 0.86–1.00]. There was no significant difference in the 2-year PFS rates (0.81, 95% CI 0.76–0.85) and ORR (0.90, 95% CI 0.87–0.94) between these four treatment regimens.
Conclusions: The meta-analysis indicates that DA-EPOCH could be more effective in providing curative treatment for adult Burkitt lymphoma patients, especially without CNS and BM involvement considering OS time. Due to the types of studies and the limited number of included studies, bias should be acknowledged and a randomized controlled trial (RCT) needs to be performed to further identify the optimal treatment regimen for such patients.
Introduction
Burkitt lymphoma (BL) is a highly aggressive B-cell non-Hodgkin’s lymphoma, which often involves extranodal sites or is represented as leukemia (1). It is rare and characterized by rapid tumor cell proliferation, which one deemed related to EB virus infection and chromosomal translocation involving the c-myc oncogene (2, 3).
Current national guidelines recommend short-term, intensive, and multi-drug regimens as the first-line treatment option for adult BL patients (4), including CODOX-M/IVAC, DA-EPOCH, and Hyper-CVAD. GMALL-B-ALL/NHL2002 is also commonly used for the treatment of BL patients and has achieved a promising outcome in already published reports (5–7). Clinical trials and systematic reviews have demonstrated that the anti-CD20 monoclonal antibody rituximab in addition to chemotherapies further provides efficacy benefits in patients with BL in the advent of the immunotherapy era (8, 9). Rituximab has become a component of standard chemotherapy in the treatment of BL for years, and no comparative studies have been conducted to evaluate the efficacy of these first-line regimens so far. Here, we conducted this meta-analysis to identify the better regimen by accessing overall survival (OS), progression-free survival (PFS), and overall response rate (ORR) data in the literature up to December 2021.
Methods
Literature search strategy
The meta-analysis was performed in accordance with the Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) guidelines using four scientific databases, PubMed, Embase, Web of Science, and Cochrane Central Register of Controlled Trials (CENTRAL) Web of Science from 10 December 2021 to 5 May 2022 (10). The following search terms were used: (“Burkitt Lymphoma” [Mesh]) OR (((Burkitt lymphoma [Title/Abstract]) OR (Burkitt’s lymphoma [Title/Abstract])) OR (BL [Title/Abstract])) AND ((“Rituximab” [Mesh])) OR (Rituximab [Title/Abstract]). The detailed search terms are listed in Table S1.
Eligibility criteria
Because the aim of this study was to assess different regimens, inclusion criteria were as follows: (1) prospective and retrospective studies; (2) English language articles and full texts; (3) chemotherapy regimens containing rituximab; and (4) studies that reported survival and response for at least 2 years. Exclusion criteria were as follows: (1) children (patient’s age ≤ 14 years old); (2) sample number of fewer than 10 patients; and (3) article type: conference abstract, letter, comment, and other types that reported incomplete information. The detailed excluded studies for full-text screening are listed in Table S2.
Included prospective non-randomized studies were assessed using the MINORS index (11). Included retrospective studies were used to evaluate the quality using the JBI Critical Appraisal Checklist (12).
Data extraction
The following records about basic information of each study were extracted independently by two authors (XL and YL): study type, published journal, name of the first author, nations where trials were conducted, year of publication, chemotherapy regimen, the total number of patients, 2-year OS rate, 2-year PFS rate, ORR (CR and PR), and follow-up time. Patients’ characteristics including median age, gender, human immunodeficiency virus (HIV)-positive, LDH levels, the proportion of stage III–IV, International Prognostic Index (IPI) or Eastern Cooperative Oncology Group (ECOG) score >2, and the number of patients with central nervous system (CNS) and bone marrow (BM) involvement were also collected. The other authors (LQ and RL) resolved any inconsistencies in the data extraction process.
Statistical analysis
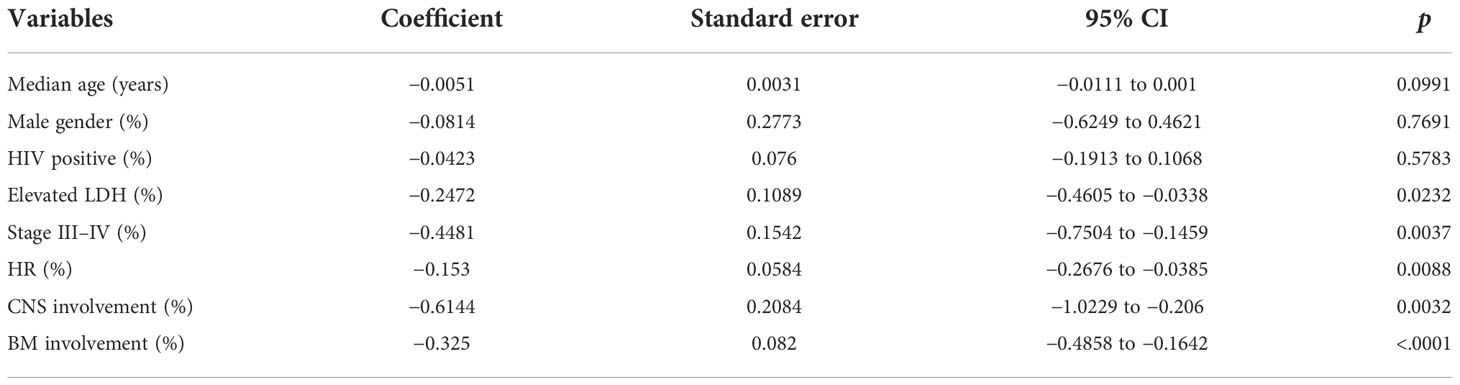
The primary outcome for efficacy was a 2-year OS rate; the second outcomes were a 2-year PFS rate and ORR. We used the software GetData Graph Digitizer 2.26.0.20 to digitize and extract data when the above-mentioned outcomes were presented only as a Kaplan–Meier survival graph. Heterogeneity was assessed using the I2 statistic. A forest plot was applied in the random-effects model if significant heterogeneity was observed (I2 > 50%); the fixed model would be applied otherwise. All of them used the DerSimonian and Laird method. The normality test results were used to determine whether the proportions should be applied in untransformed data or transformed with the Freeman–Tukey double arcsine transformation and confidence intervals were calculated using the Jackson method. A meta-regression was also performed to examine factors possibly related to outcomes, including median age, gender, HIV-positive, the proportion of elevated LDH, high risk, stage III–IV, CNS, and BM involvement. Potential publication bias was assessed by inspecting a funnel plot and Egger’s test. All statistical tests were two-tailed, and p values < 0.05 were considered significant. R software environment version 4.1.2 was used for statistical computing and graphics.
Results
Literature search
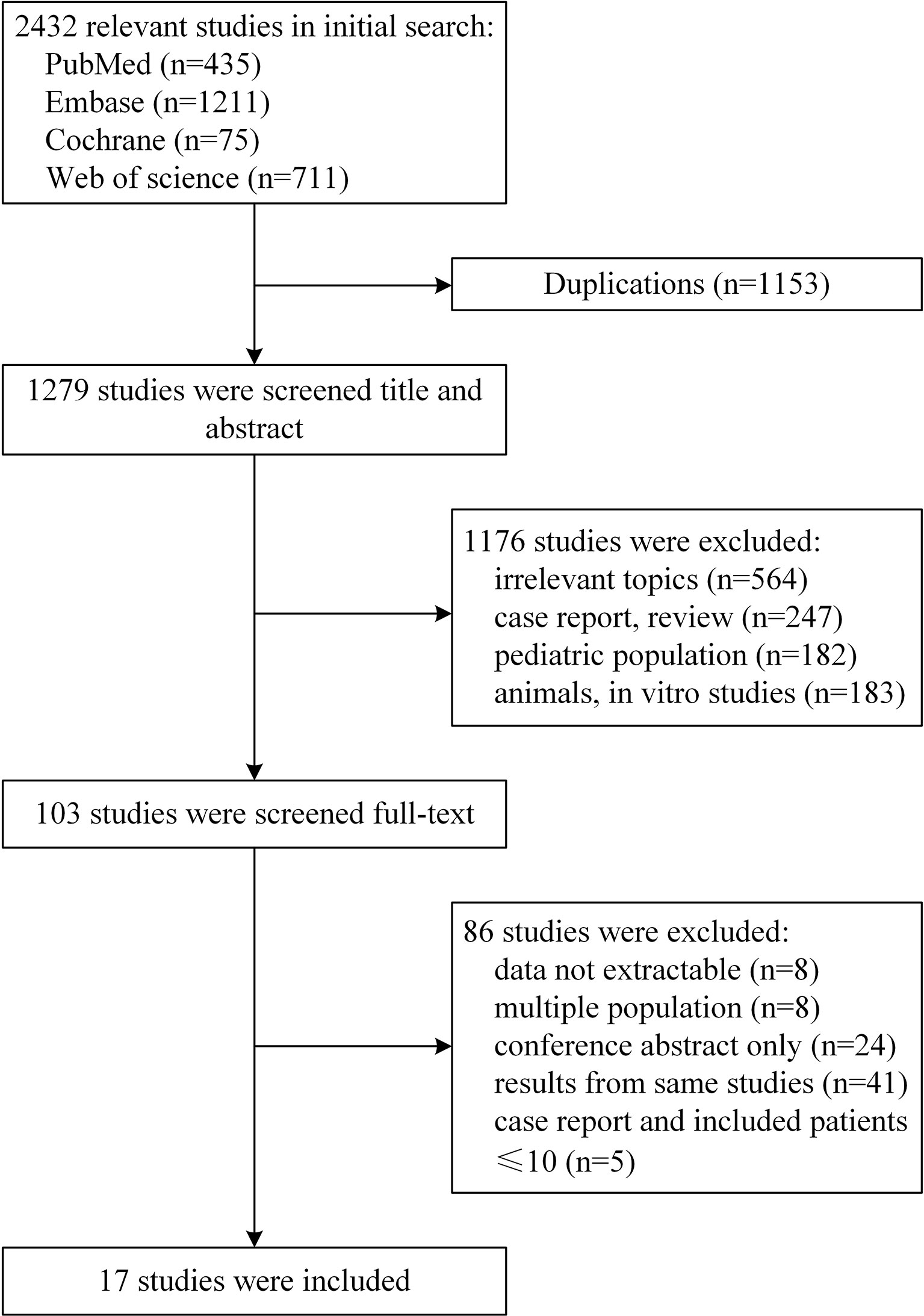
A total of 2,432 potentially relevant studies were identified for screening (Figure 1). After excluding 1,153 duplications, 103 studies remained by accessing titles and abstracts. Among the remaining full-text selections, 86 articles were removed because of conference posters, overlapping data, information being unable to extract, multiple cohorts, and small sample size. A total of 17 studies including altogether 1,258 patients were included in our final analysis (5–7, 13–26).
Study characteristics
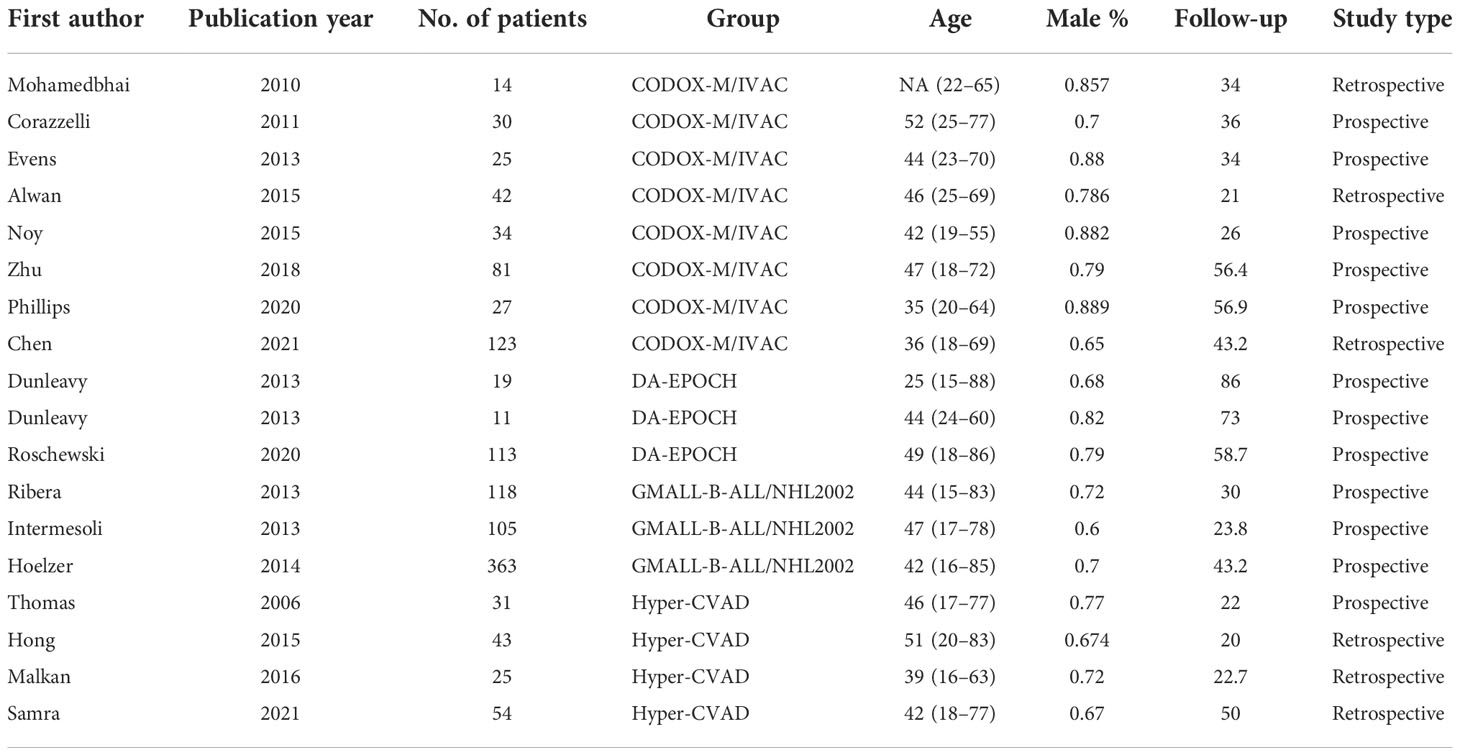
The baseline characteristics for the 17 studies are presented in Table 1. Twelve included articles were prospective studies, of which only one was a comparative study, and the remaining 11 were non-comparative and single-arm studies. In addition, six retrospective articles were included. No randomized controlled trial (RCT) was included. All of the articles were published between 2006 and 2021, comprising 1,258 patients. A total of 1,258 patients were included, with the number of patients in each study ranging from 14 to 363. The age range was 15 to 88 years (5, 19), with median ages between 25 and 52 years (19, 24). A relatively higher proportion of male patients was observed in the range from 0.6 to 0.889, which was consistent with the results of previous studies (27, 28). The included patients were followed up for 1.4 to 147.6 months. Five cohorts included only HIV-negative patients, three cohorts included only HIV-positive patients, and the proportion of HIV-positive patients in the remaining groups was 0.14 to 0.323. Four treatment groups were as follows: eight cohorts from eight studies with CODOX-M/IVAC (13–15, 17, 21, 22, 24, 25), three cohorts from two studies with DA-EPOCH (19, 20), three cohorts from three studies with GMALL-B-ALL/NHL2002 (5–7), and four cohorts from four studies with Hyper-CVAD, all of which used rituximab (16, 18, 23, 26). The included studies had a median follow-up time of 35 months (range, 20 to 86) (19, 23).
Study quality assessment
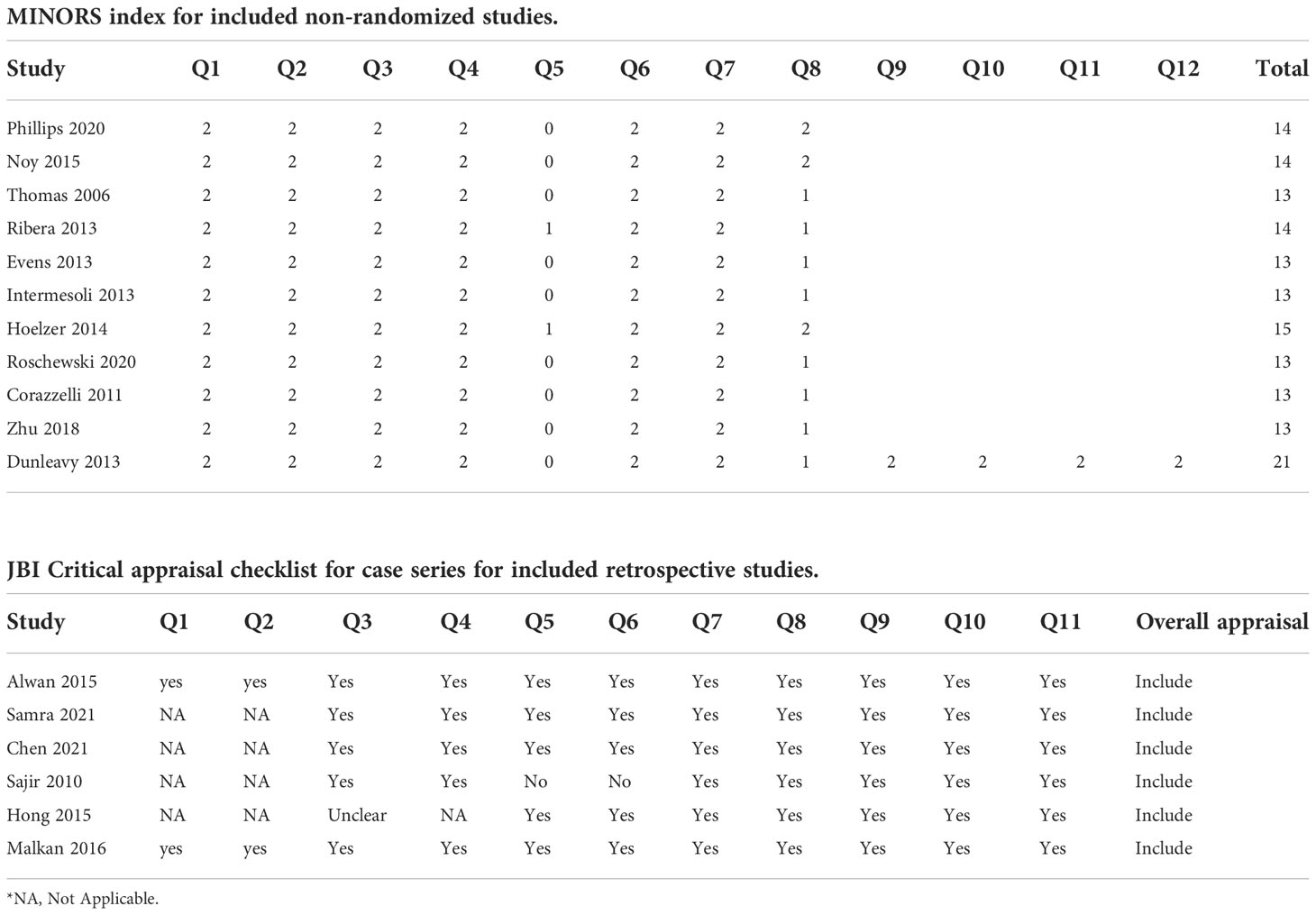
The summary of the quality assessment is outlined in Table 2. One study including two comparative groups scored 21 on the MINORS index (19). Ten prospective single-arm studies had generally moderate quality scoring between 13 and 15 points (5–7, 14–17, 19, 20, 24, 25). Six retrospective studies were determined to include after quality evaluation by the JBI Critical Appraisal Checklist (13, 18, 21–23, 26).
Pooled analyses and meta-regression
Primary outcome
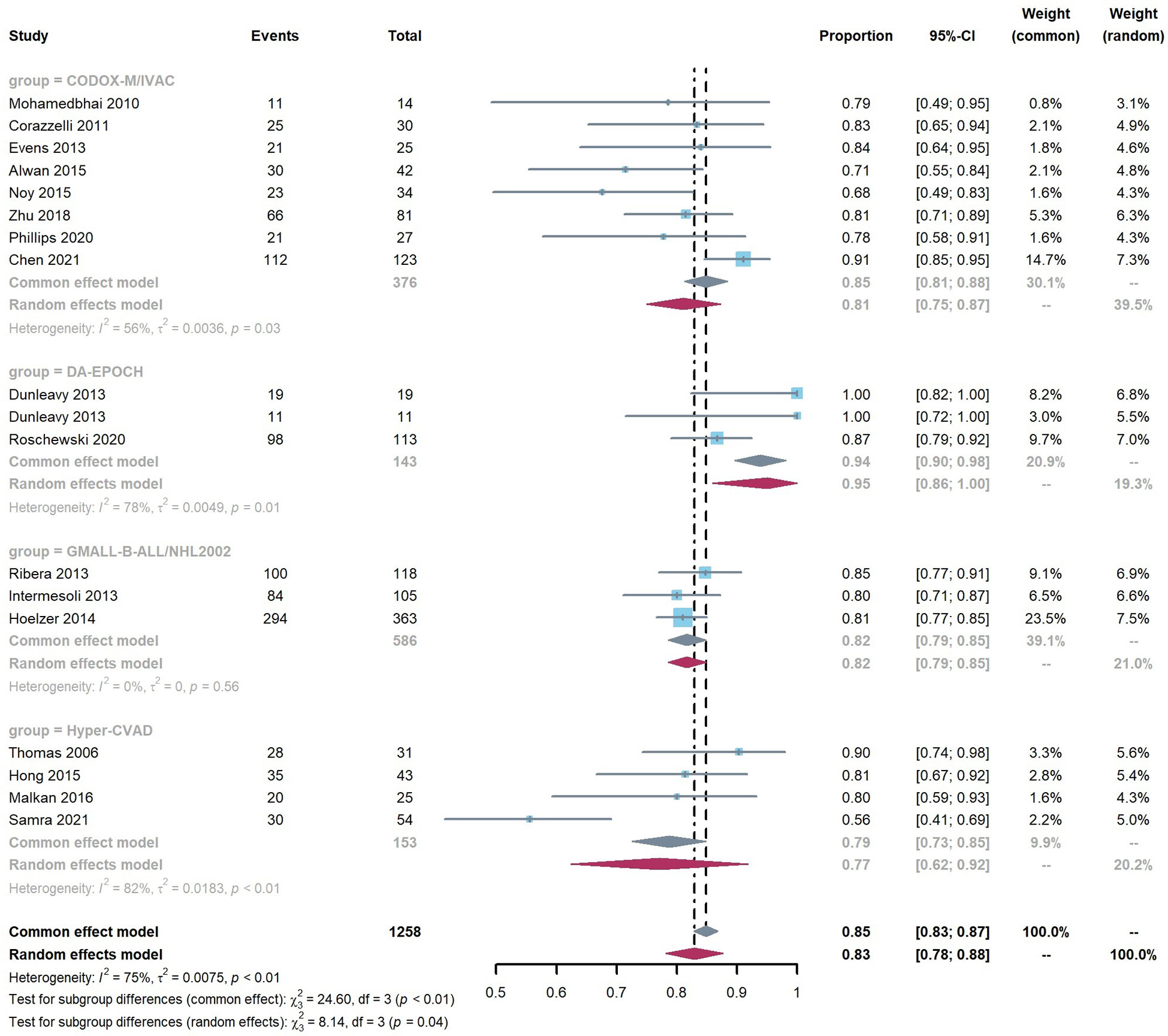
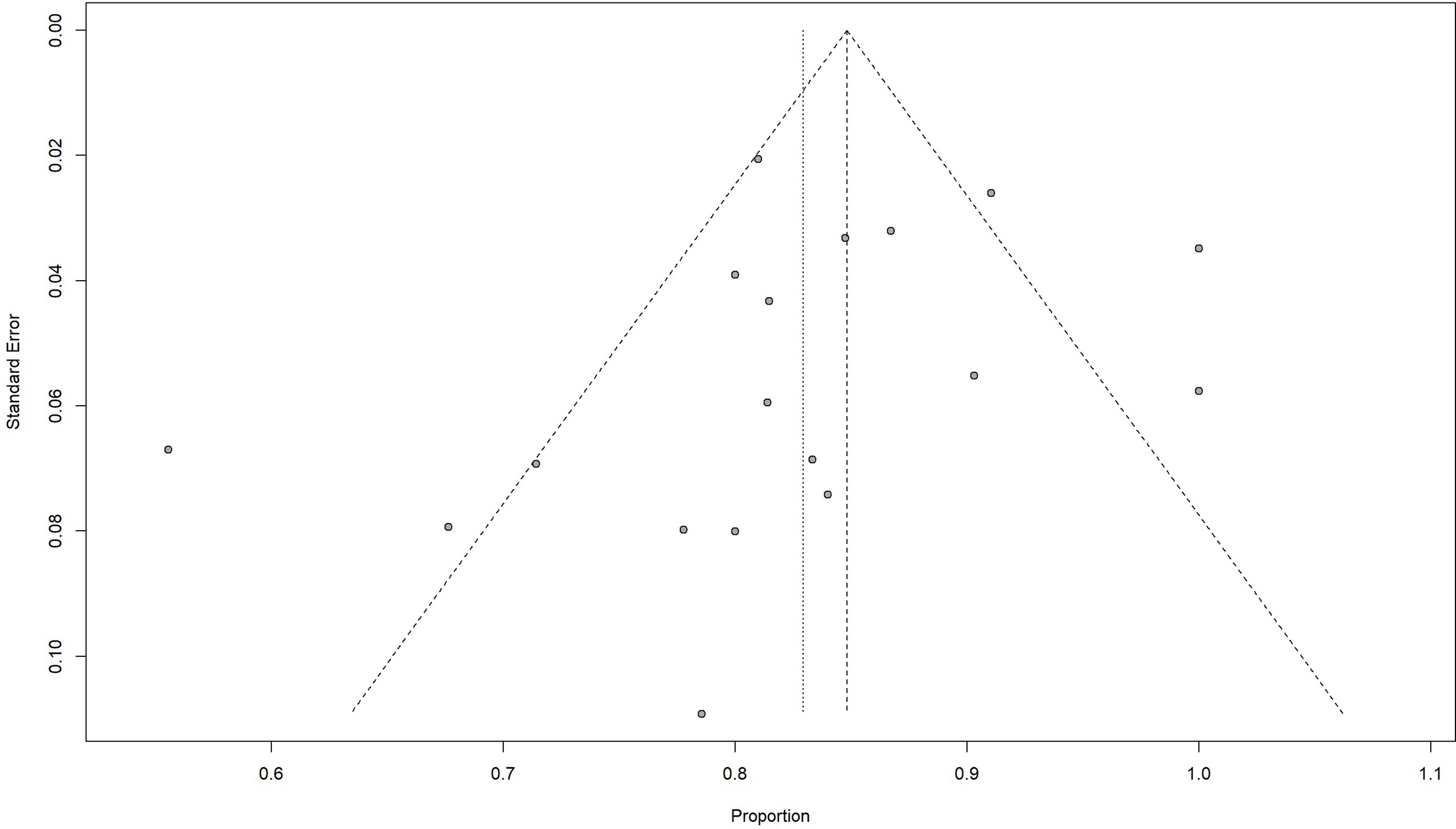
The overall pooled 2-year OS rate was 0.83 (95% CI 0.78–0.88). Our result showed a significant difference in the 2-year OS rate according to the chemotherapy regimen groups (p < 0.05). The results favored the DA-EPOCH group, which led to a higher OS rate (0.95, 95% CI 0.86–1.00), compared to CODOX-M/IVAC (0.81, 95% CI 0.75–0.87), Hyper-CVAD (0.77, 95% CI 0.62–0.92), and GMALL-B-ALL/NHL2002 (0.82, 95% CI 0.79–0.85) (Figure 2). Meta-regression with a mixed-effects model demonstrated that elevated LDH, the proportion of stage III-IV and high-risk patients, and the involvement of CNS or BM are associated with significant heterogeneity (I2 = 75%, p < 0.01) (Table 3). By contrast, no difference was found in age, sex, and HIV infection among different regimen groups. The funnel plot was considered roughly symmetric by inspection (Figure 3). Egger’s test was also performed to detect publication bias for included studies (p = 0.2519).
Secondary outcomes
The pooled ORR was 0.90 (95% CI 0.87–0.94) with significant heterogeneity (I2 = 69%, p < 0.01) in connection with the elevated LDH. There was no significant difference (p = 0.07) in ORR between DA-EPOCH, CODOX-M/IVAC, Hyper-CVAD, and GMALL-B-ALL/NHL2002 groups, with values of 0.99 (95% CI 0.93–1.00), 0.92 (95% CI 0.86–0.97), 0.89 (95% CI 0.79–0.99), and 0.86 (95% CI 0.79–0.94), respectively (Figure S1). A similar result was found in that these four groups DA-EPOCH (0.92, 95% CI 0.82–1.00), CODOX-M/IVAC (0.80, 95% CI 0.76–0.85), Hyper-CVAD (0.71, 95% CI 0.57–0.86), and GMALL-B-ALL/NHL2002 (0.79, 95% CI 0.76–0.83) have no obvious difference in the 2-year PFS rate (Figure S2). The roughly same factors as ORR were identified in the heterogeneity analysis of PFS (I2 = 61%, p < 0.01) using meta-regression, except for LDH. No publication bias was observed in both ORR and 2-year PFS rates using the funnel plot and Egger’s test (Figures S3, S4).
Discussion
BL is an aggressive and chemotherapy-sensitive B-cell non-Hodgkin’s lymphoma categorized into endemic, sporadic, and immunodeficiency-associated subtypes (29). The majority of BL patients obtained long-term survival after some intensive chemotherapy, and the prognosis has further improved owing to the advent of rituximab. Research showed that rituximab, the anti-CD20 monoclonal antibody, can not only prolong the time of disease progression but also extend OS for a variety of B-cell lymphomas, including BL (30). It is still uncertain which is the optimal regimen containing rituximab for BL patients, and we aim to answer this question through a meta-analysis. Previous studies found heterogeneity between pediatric and adult BL patients; thus, this meta-analysis focused on patients no less than 14 years of age (28). A total of 17 studies and 1,258 patients were finally identified through data search and literature screening and divided into four treatment groups as follows: DA-EPOCH, CODOX-M/IVAC, Hyper-CVAD, and GMALL-B-ALL/NHL2002. All included patients were sporadic or immunodeficiency-associated types, probably because endemic BL is prevalent in children of sub-Saharan Africa (29).
CODOX-M/IVAC, developed by Magrath, is a highly effective alternate and the most commonly used regimen as our result showed that patients in almost half of the included studies were treated with this chemotherapy program (31). B-NHL2002, a short intensive chemotherapy program based on a pediatric Berlin-Frankfurt-Münster protocol, was updated and improved several times by the German Multicenter Study Group for Adult ALL (GMALL). Three current reported large prospective trials showed substantial cure rates in adult BL/leukemia whether HIV-negative or HIV-positive (5–7). Hyper-CVAD (hyper-fractionated cyclophosphamide, doxorubicin, vincristine, and dexamethasone alternating with methotrexate plus cytarabine) was also a comparable treatment option among established dose-intensive regimens for BL (18, 23, 26). A retrospective study by Samra et al. reported that the Hyper-CVAD protocol showed highly promising efficacy and safety in high-risk patients with CNS or BM involvement (18). The DA-EPOCH (Risk-adapted etoposide, prednisone, vincristine, cyclophosphamide, and doxorubicin) protocol has been widely used in lymphoma and is well tolerated by BL patients of all ages and those with HIV infection (20). Based on the results of our meta-analysis, the DA-EPOCH regimen group might be considered a better treatment option for adult BL patients by comparing the 2-year OS rate among the four regimen groups. No significant difference was observed in the 2-year PFS rate and ORR among these four groups. Interestingly, one RCT performed by Chamuleau reported a similar estimated 2-year OS among R-CODOX-M/R-IVAC and DA-EPOCH-R for high-risk BL; this trial was suspended because of the slow accrual rate. The limited sample size and inclusion of only high-risk populations in this trial may have contributed to the discrepant results between this RCT and our analysis (32).
LDH levels, stage III–IV, CNS, and BM involvement have been widely used for risk stratification and considered as independent influential factors in the poor prognosis of BL. Our result displayed various sources of heterogeneity, including LDH levels, stage III–IV, CNS, and BM involvement, consistent with the previously reported results in strong association with OS rate, further confirming the reliability of our meta-analysis (33–35). Previous studies showed that BL is highly aggressive, especially when with extensive involvement. Patients with BM involvement using the Ann Arbor staging system were diagnosed at stage IV, and those with CNS involvement had no detailed description. The St Jude/Murphy system, which was much more commonly used for staging, suggested that BL patients with BM and/or CNS involvement were divided into stage IV and the high-risk group. Anyhow, the involvement of CNS and/or BM directly related to the advanced stage was a significant factor associated with inferior prognosis. As shown in our results, because of the different proportions of CNS and/or BM involvement in the included studies and the identification of the source of heterogeneity between patients with and without CNS and BM involvement by meta-regression, the conclusion that “the DA-EPOCH regimen had a higher OS rate compared with three other treatment groups” seems to be inapplicable to patients with BM or CNS involvement. Furthermore, studies have shown no difference in clinical and biochemical characteristics and treatment outcomes between HIV-positive and HIV-negative cohorts after rituximab-based chemotherapy (35–38). We agreed that HIV status does not affect the prognosis of adult BL patients. The prevalence of BL is higher in men than in women; however, we found no gender differences regarding prognosis.
In this meta-analysis, we compared the efficacy of different chemotherapy regimens by subgroup analysis and identified the factors that may influence the treatment outcomes. This is the first answer to the question of which is the best treatment option for BL in the rituximab era. The DA-EPOCH-R regimen was a less toxic regimen than other dose-intensive regimens for BL (e.g., CODOX-M-IVAC) (39). DA-EPOCH-R was a preferred option not only because it extends survival time but also because of its better safety and lower medical costs.
The following limitations of this study should be acknowledged. Firstly, included articles were performed by different study types, and most of the included studies were single-arm without RCT studies. Secondly, the small number of included studies and patients may not be representative enough of all adult BL patients; there were only less than 20 patients in some included studies. Thirdly, doses of chemotherapy agents modified by medicine centers and various manufacturers of rituximab cannot be overlooked. Also, the results of our heterogeneity analysis could be different from the real world owing to unavailable data from some cohorts. Furthermore, the applicability of our results in patients with BM and/or CNS involvement remains to be further validated due to the existence of heterogeneity.
Conclusion
In summary, a better treatment strategy for prolonging the survival time of adult BL without CNS and/or BM involvement was first identified through our meta-analysis. We found that DA-EPOCH had a greater OS rate in contrast with CODOX-M/IVAC, GMALL-B-ALL/NHL2002, and Hyper-CVAD groups. More studies, especially RCTs, need to be performed to identify the optimal chemotherapy regimen for patients with extensive organ involvement.
Data availability statement
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding author.
Author contributions
XL: Data acquisition, writing the original draft, and editing. YL: Data acquisition and analysis. RL: Resources, and data acquisition. JL: Manuscript revision and project administration. XY: Providing guidance, manuscript revision, and funding acquisition. LQ: Methodology, manuscript revision, funding acquisition and final approval of the last version. All authors contributed to the article and approved the submitted version.
Funding
This work was supported by a grant from the National Defense Science and Technology Innovation Special Zone Project-Spark Project (Grant No.20-163-00-TS-009-006-01) and a grant from the National Natural Science Foundation of China (Grant No. 81800180).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fonc.2022.1063689/full#supplementary-material
Supplementary Figure 1 | Pooled ORR according to regimen group
Supplementary Figure 2 | Pooled 2-year PFS rate according to regimen group
Supplementary Figure 3 | Funnel plot of ORR
Supplementary Figure 4 | Funnel plot of 2-year PFS rate
References
1. Molyneux EM, Rochford R, Griffin B, Newton R, Jackson G, Menon G, et al. Burkitt's lymphoma. Lancet (London England) (2012) 379(9822):1234–44. doi: 10.1016/s0140-6736(11)61177-x
2. Taub R, Kirsch I, Morton C, Lenoir G, Swan D, Tronick S, et al. Translocation of the c-myc gene into the immunoglobulin heavy chain locus in human burkitt lymphoma and murine plasmacytoma cells. Proc Natl Acad Sci United States America (1982) 79(24):7837–41. doi: 10.1073/pnas.79.24.7837
3. Pope JH, Horne MK, Scott W. Transformation of foetal human keukocytes in vitro by filtrates of a human leukaemic cell line containing herpes-like virus. Int J Cancer (1968) 3(6):857–66. doi: 10.1002/ijc.2910030619
4. Crombie J, LaCasce A. The treatment of burkitt lymphoma in adults. Blood (2021) 137(6):743–50. doi: 10.1182/blood.2019004099
5. Ribera JM, García O, Grande C, Esteve J, Oriol A, Bergua J, et al. Dose-intensive chemotherapy including rituximab in burkitt's leukemia or lymphoma regardless of human immunodeficiency virus infection status: Final results of a phase 2 study (Burkimab). Cancer (2013) 119(9):1660–8. doi: 10.1002/cncr.27918
6. Intermesoli T, Rambaldi A, Rossi G, Delaini F, Romani C, Pogliani EM, et al. High cure rates in burkitt lymphoma and leukemia: A northern Italy leukemia group study of the German short intensive rituximab-chemotherapy program. Haematologica (2013) 98(11):1718–25. doi: 10.3324/haematol.2013.086827
7. Hoelzer D, Walewski J, Döhner H, Viardot A, Hiddemann W, Spiekermann K, et al. Improved outcome of adult burkitt Lymphoma/Leukemia with rituximab and chemotherapy: Report of a Large prospective multicenter trial. Blood (2014) 124(26):3870–9. doi: 10.1182/blood-2014-03-563627
8. Nie M, Wang Y, Bi XW, Xia Y, Sun P, Liu PP, et al. Effect of rituximab on adult burkitt's lymphoma: A systematic review and meta-analysis. Ann Hematol (2016) 95(1):19–26. doi: 10.1007/s00277-015-2501-1
9. Minard-Colin V, Aupérin A, Pillon M, Burke GAA, Barkauskas DA, Wheatley K, et al. Rituximab for high-risk, mature b-cell non-hodgkin's lymphoma in children. New Engl J Med (2020) 382(23):2207–19. doi: 10.1056/NEJMoa1915315
10. Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, Ioannidis JP, et al. The prisma statement for reporting systematic reviews and meta-analyses of studies that evaluate health care interventions: Explanation and elaboration. Ann Internal Med (2009) 151(4):W65–94. doi: 10.7326/0003-4819-151-4-200908180-00136
11. Slim K, Nini E, Forestier D, Kwiatkowski F, Panis Y, Chipponi J. Methodological index for non-randomized studies (Minors): Development and validation of a new instrument. ANZ J Surg (2003) 73(9):712–6. doi: 10.1046/j.1445-2197.2003.02748.x
12. Moola S, Munn Z, Tufanaru C, Aromataris E, Sears K, Sfetcu R, et al. Joanna Briggs Institute reviewer’s manual. The Joanna Briggs Institute (2017). Available at: https://joannabriggs.org.
13. Alwan F, He A, Montoto S, Kassam S, Mee M, Burns F, et al. Adding rituximab to codox-M/Ivac chemotherapy in the treatment of hiv-associated burkitt lymphoma is safe when used with concurrent combination antiretroviral therapy. AIDS (London England) (2015) 29(8):903–10. doi: 10.1097/qad.0000000000000623
14. Phillips EH, Burton C, Kirkwood AA, Barrans S, Lawrie A, Rule S, et al. Favourable outcomes for high-risk burkitt lymphoma patients (Ipi 3-5) treated with rituximab plus codox-M/Ivac: Results of a phase 2 uk ncri trial. EJHaem (2020) 1(1):133–41. doi: 10.1002/jha2.3
15. Noy A, Lee JY, Cesarman E, Ambinder R, Baiocchi R, Reid E, et al. Amc 048: Modified codox-M/Ivac-Rituximab is safe and effective for hiv-associated burkitt lymphoma. Blood (2015) 126(2):160–6. doi: 10.1182/blood-2015-01-623900
16. Thomas DA, Faderl S, O'Brien S, Bueso-Ramos C, Cortes J, Garcia-Manero G, et al. Chemoimmunotherapy with hyper-cvad plus rituximab for the treatment of adult burkitt and burkitt-type lymphoma or acute lymphoblastic leukemia. Cancer (2006) 106(7):1569–80. doi: 10.1002/cncr.21776
17. Evens AM, Carson KR, Kolesar J, Nabhan C, Helenowski I, Islam N, et al. A multicenter phase ii study incorporating high-dose rituximab and liposomal doxorubicin into the codox-M/Ivac regimen for untreated burkitt's lymphoma. Ann Oncol (2013) 24(12):3076–81. doi: 10.1093/annonc/mdt414
18. Samra B, Khoury JD, Morita K, Ravandi F, Richard-Carpentier G, Short NJ, et al. Long-term outcome of hyper-Cvad-R for burkitt Leukemia/Lymphoma and high-grade b-cell lymphoma: Focus on cns relapse. Blood Adv (2021) 5(20):3913–8. doi: 10.1182/bloodadvances.2021004427
19. Dunleavy K, Pittaluga S, Shovlin M, Steinberg SM, Cole D, Grant C, et al. Low-intensity therapy in adults with burkitt's lymphoma. New Engl J Med (2013) 369(20):1915–25. doi: 10.1056/NEJMoa1308392
20. Roschewski M, Dunleavy K, Abramson JS, Powell BL, Link BK, Patel P, et al. Multicenter study of risk-adapted therapy with dose-adjusted epoch-r in adults with untreated burkitt lymphoma. J Clin Oncol (2020) 38(22):2519–29. doi: 10.1200/JCO.20.00303
21. Chen M, Wang Z, Fang X, Yao Y, Ren Q, Chen Z, et al. Modified r-Codox-M/Ivac chemotherapy regimens in Chinese patients with untreated sporadic burkitt lymphoma. Cancer Biol Med (2021) 18(3):833–40. doi: 10.20892/j.issn.2095-3941.2020.0314
22. Mohamedbhai SG, Sibson K, Marafioti T, Kayani I, Lowry L, Goldstone AH, et al. Rituximab in combination with codox-M/Ivac: A retrospective analysis of 23 cases of non-hiv related b-cell non-Hodgkin lymphoma with proliferation index >95%. Br J haemato (2011) 152(2):175–81. doi: 10.1111/j.1365-2141.2010.08447.x
23. Hong J, Kim SJ, Ahn JS, Song MK, Kim YR, Lee HS, et al. Treatment outcomes of rituximab plus hyper-cvad in Korean patients with sporadic burkitt or burkitt-like lymphoma: Results of a multicenter analysis. Cancer Res Treat (2015) 47(2):173–81. doi: 10.4143/crt.2014.055
24. Corazzelli G, Frigeri F, Russo F, Frairia C, Arcamone M, Esposito G, et al. Rd-Codox-M/Ivac with rituximab and intrathecal liposomal cytarabine in adult burkitt lymphoma and 'Unclassifiable' highly aggressive b-cell lymphoma. Br J haemato (2012) 156(2):234–44. doi: 10.1111/j.1365-2141.2011.08947.x
25. Zhu KY, Song KW, Connors JM, Leitch H, Barnett MJ, Ramadan K, et al. Excellent real-world outcomes of adults with burkitt lymphoma treated with codox-M/Ivac plus or minus rituximab. Br J haemato (2018) 181(6):782–90. doi: 10.1111/bjh.15262
26. Malkan ÜY, Güneş G, Göker H, Haznedaroğlu İC, Acar K, Eliaçık E, et al. The prognosis of adult burkitt’s cell leukemia in real-life clinical practice. Turkish J Hematol (2016) 33(4):281–5. doi: 10.4274/tjh.2015.0088
27. Mbulaiteye SM, Biggar RJ, Bhatia K, Linet MS, Devesa SS. Sporadic childhood burkitt lymphoma incidence in the united states during 1992-2005. Pediatr Blood Cancer (2009) 53(3):366–70. doi: 10.1002/pbc.22047
28. Boerma EG, van Imhoff GW, Appel IM, Veeger NJ, Kluin PM, Kluin-Nelemans JC. Gender and age-related differences in burkitt lymphoma–epidemiological and clinical data from the Netherlands. Eur J Cancer (Oxford Engl 1990) (2004) 40(18):2781–7. doi: 10.1016/j.ejca.2004.09.004
29. Noy A. Burkitt lymphoma - subtypes, pathogenesis, and treatment strategies. Clin lymphoma myeloma leukemia (2020) 20 Suppl 1:S37–s8. doi: 10.1016/s2152-2650(20)30455-9
30. Salles G, Barrett M, Foà R, Maurer J, O'Brien S, Valente N, et al. Rituximab in b-cell hematologic malignancies: A review of 20 years of clinical experience. Adv Ther (2017) 34(10):2232–73. doi: 10.1007/s12325-017-0612-x
31. Magrath I, Adde M, Shad A, Venzon D, Seibel N, Gootenberg J, et al. Adults and children with small non-Cleaved-Cell lymphoma have a similar excellent outcome when treated with the same chemotherapy regimen. J Clin Oncol (1996) 14(3):925–34. doi: 10.1200/JCO.1996.14.3.925
32. Chamuleau M, Stenner F, Chitu D, Novak U, Minnema M, Visser O, et al. (2022). R-CODOX-M/R-IVAC versus dose-adjusted(DA)-EPOCH-R in patients with newly diagnosed high-risk burkitt lymphoma; first results of a multi-center randomized HOVON/SAKK trial. EHA2022: Proceedings of the 27th European hematology association, Vienna, Austria: LB2370.
33. Evens AM, Danilov A, Jagadeesh D, Sperling A, Kim SH, Vaca R, et al. Burkitt lymphoma in the modern era: Real-world outcomes and prognostication across 30 us cancer centers. Blood (2021) 137(3):374–86. doi: 10.1182/blood.2020006926
34. Olszewski AJ, Jakobsen LH, Collins GP, Cwynarski K, Bachanova V, Blum KA, et al. Burkitt lymphoma international prognostic index. J Clin Oncol Off J Am Soc Clin Oncol (2021) 39(10):1129–38. doi: 10.1200/jco.20.03288
35. Tan JY, Qiu TY, Chiang J, Tan YH, Yang VS, Chang EWY, et al. Burkitt lymphoma - no impact of hiv status on outcomes with rituximab-based chemoimmunotherapy. Leukemia lymphoma (2022), 19:1–11. doi: 10.1080/10428194.2022.2027402
36. Barnes JA, Lacasce AS, Feng Y, Toomey CE, Neuberg D, Michaelson JS, et al. Evaluation of the addition of rituximab to codox-M/Ivac for burkitt's lymphoma: A retrospective analysis. Ann Oncol Off J Eur Soc Med Oncol (2011) 22(8):1859–64. doi: 10.1093/annonc/mdq677
37. Wang ES, Straus DJ, Teruya-Feldstein J, Qin J, Portlock C, Moskowitz C, et al. Intensive chemotherapy with cyclophosphamide, doxorubicin, high-dose Methotrexate/Ifosfamide, etoposide, and high-dose cytarabine (Codox-M/Ivac) for human immunodeficiency virus-associated burkitt lymphoma. Cancer (2003) 98(6):1196–205. doi: 10.1002/cncr.11628
38. Oriol A, Ribera JM, Bergua J, Giménez Mesa E, Grande C, Esteve J, et al. High-dose chemotherapy and immunotherapy in adult burkitt lymphoma: Comparison of results in human immunodeficiency virus-infected and noninfected patients. Cancer (2008) 113(1):117–25. doi: 10.1002/cncr.23522
Keywords: Burkitt lymphoma, meta-analysis, chemotherapy regimen, rituximab, single-arm
Citation: Lu X, Liu Y, Liu R, Liu J, Yan X and Qian L (2022) Comparison of chemotherapy regimens plus rituximab in adult Burkitt lymphoma: A single-arm meta-analysis. Front. Oncol. 12:1063689. doi: 10.3389/fonc.2022.1063689
Received: 07 October 2022; Accepted: 29 November 2022;
Published: 23 December 2022.
Edited by:
Alessandro Isidori, AORMN Hospital, ItalyReviewed by:
Prasanth Ganesan, Jawaharlal Institute of Postgraduate Medical Education and Research (JIPMER), IndiaMichele Merli, University of Insubria, Italy
Copyright © 2022 Lu, Liu, Liu, Liu, Yan and Qian. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Xiaojing Yan, eWFueGlhb2ppbmdfcHBAaG90bWFpbC5jb20=; Liren Qian, cWxyMjAwN0AxMjYuY29t
 Xiaoxuan Lu
Xiaoxuan Lu Yu Liu
Yu Liu Ruyu Liu1
Ruyu Liu1 Liren Qian
Liren Qian