- 1Department of Thoracic Surgery, The Second Hospital of Jilin University, Changchun, China
- 2Department of Hepatopancreatobiliary Medicine, The Second Hospital of Jilin University, Changchun, Jilin, China
- 3Department of Central Lab, The Second Hospital of Jilin University, Changchun, China
- 4Department of Pathology, The Second Hospital of Jilin University, Changchun, China
Lung cancer is one of the most prevalent cancer types and the leading cause of cancer-related deaths worldwide. Non-small cell lung cancer (NSCLC) accounts for 80-85% of all cancer incidences. Lung cancer therapy and prognosis largely depend on the disease’s degree at the diagnosis time. Cytokines are soluble polypeptides that contribute to cell-to-cell communication, acting paracrine or autocrine on neighboring or distant cells. Cytokines are essential for developing neoplastic growth, but they are also known to operate as biological inducers following cancer therapy. Early indications are that inflammatory cytokines such as IL-6 and IL-8 play a predictive role in lung cancer. Nevertheless, the biological significance of cytokine levels in lung cancer has not yet been investigated. This review aimed to assess the existing literature on serum cytokine levels and additional factors as potential immunotherapeutic targets and lung cancer prognostic indicators. Changes in serum cytokine levels have been identified as immunological biomarkers for lung cancer and predict the effectiveness of targeted immunotherapy.
1 Introduction
Lung cancer or lung carcinoma is the most common cause of cancer incidence and cancer-related deaths worldwide (1). Annually, over 2.2 million people are affected, and it causes around 1.8 million deaths worldwide. It has the highest incidence in developing nations, where tobacco use is most prevalent (2). Lung cancer mortality is projected to have grown by 18.3% between 2006 and 2016; in highly developed nations, attributable years of life lost came in second. New statistics show that in 2019, lung cancer was the seventeenth largest cause of disability-adjusted life years (DALYs) for all ages. However, it sharply increased to become the fifth and seventh leading cause for people aged 50 to 74 and 75 and older, respectively (3). Non-small-cell lung cancer (NSCLC) and small-cell lung cancer (SCLC) are two different types of lung cancer based on their histopathological characteristics (4). NSCLC is the most common type of lung cancer, accounting for 85% of cases. Adenocarcinoma (40%), squamous cell carcinoma (30%), and large-cell carcinoma (10%) are the three primary subtypes (5). Epidemiologic studies have shown that inflammation can induce tissue damage and help to develop lung cancer (6).
Cytokines are soluble chemical messengers that are critical in signaling immune responses. Th2 cytokines (IL-4, IL-5, and IL-10) steer the T-cell response away from a protective Th1 phenotype, while Th1 cytokines (IL-2, IFNγ, and TNFα) trigger cell-mediated responses. Malignant tumors have a skewed Th1/Th2 cytokine balance that encourages an immunosuppressive microenvironment (7). The primary regulators of the innate and adaptive immune systems, cytokines play a crucial role in regulating immunological responses to infections, autoimmune disorders, and cancer. Cytokines have a wide range of complex functions. In addition to providing protection, they can also contribute to developing cancer or autoimmune illnesses when overactive or severely deficient (8). Cytokines control the inflammatory response, which can have endocrine, paracrine, or autocrine actions. Besides producing inflammatory cytokines, human lung cancer cells can also do so (9). The different triggers that might result in the release of cytokines, which regulate numerous aspects of human physiology and disease locally, include interferons (IFNs), interleukins, colony-stimulating factors, chemokines, growth factors, and tumor necrosis factors (10). Recombinant cytokines have been used to treat cancer for a very long time because they are essential in developing, spreading, and eliminating cancer. However, the limited value of cytokines as therapeutics is due to their poor drug-like properties, complex biology, cytokine pleiotropy, severe dose-limiting toxicities, and drug-like characteristics of cytokines (11). Cytokines, however, offer the potential to enhance immunotherapy techniques for cancer treatment as they play a crucial role in innate and adaptive anti-tumor immunity. The classification of cytokines as inflammatory (released by Th-1 T helper cells) or anti-inflammatory (released by Th-2 T helper cells) is a frequent practice (12). The pathogenesis of cancer, particularly lung cancer, is thought to be significantly influenced by specific inflammatory cytokines, according to numerous research published to date (13).
According to many preclinical and clinical studies, immune cells that infiltrate tumors or tumor cells themselves may release cytokines and chemokines that alter the tumor microenvironment and encourage angiogenesis, growth, invasion, and metastasis. Additionally, cytokines work as a pro-tumor factor or a tumor cell growth inhibitor, respectively (14). According to reports, immunotherapy patients with metastatic melanoma with cytokine profiles evaluated throughout time had shown a correlation between the progression of irAEs and severe irAEs. In patients with advanced NSCLC receiving immunotherapy, recent studies have suggested that elevated levels of IL-1β and IFN-γ during treatment may indicate efficacy. In contrast, high levels of IL-6 during treatment may show less favorable results (15, 16).
Due to improvements in diagnostic and treatment methods and the fact that lung cancer is typically identified at a mature phase, patients’ prognoses are still direr for those with lung cancer than those with other types of cancer, with a 5-year comparative mortality risk of less than 20%. (17). The pillars of cancer treatment for a very long period included surgery, chemotherapy, and radiation therapy. The fourth pillar of the cancer care approach, immunotherapy, was recently added, giving patients their first opportunity to be treated for a possible treatment (18). Furthermore, in 2015, the Phase II Checkmate 063 study was the first significant study of immunotherapy showing activity in NSCLC (19). Immunotherapy is a cancer treatment employing the patient’s immune system. Immunotherapy can alter the immune system’s functioning to help it recognize and combat cancer cells (20). Chimeric antigen receptor (CAR) T-cell therapy, checkpoint inhibitors, cytokines, immunomodulators, and cancer vaccines are a few of the vital immunotherapy treatments for cancer currently being researched (21).
Over the past 40 years, much research has been done on cytokines and cytokine receptors as tumor targets and treatments. More excellent knowledge of the cancer ecosystem and the advancement of more efficient immunotherapies have led to the development of novel methods for employing cytokine channels in cancer therapy (22). Among these methods is the use of cytokine-based medicines to intensify other medications’ immune-related side effects and focus on tumors in their early stages (15). Elevated amounts of cytokines can be found in those with immune reactions to inflammation and cancer. Smoking is the primary risk factor for lung cancer, and smoke irritants and chemical irritants can change lung cancer cytokine levels by triggering an immune response (23).
Lung cancer still has an extremely poor prognosis despite all the advancements in treatment. This dismal outlook highlights the urgent requirement for the creation of new methods to prevent and cure this fatal illness successfully (24). The prognosis is the doctor’s best guess regarding how cancer will progress and respond to treatment. Numerous factors affect the prognosis and likelihood of survival. Performance status, age, and gender are examples of patient-related prognostic factors. Tumor-related prognostic indicators include clinical stage and histological type (25). Prognostic markers identify patient subpopulations with markedly varied expected outcomes who may benefit from various treatment modalities. However, a prognostic factor cannot provide information on a specific therapy’s advantages. In contrast, individuals with many unfavorable prognostic variables may require more active treatment to achieve the same lifespan as individuals without these qualities. Patients with many favorable predictive characteristics may require less intensive treatment to attain a cure.
In the earlier research, only a few cytokines were examined concurrently in the same subjects. The lack of prior studies reporting on the connection between lung cancer survival and circulating cytokine concentrations represents another significant gap in the evidence (26). The development of novel therapeutic targets and biomarkers can predict how well immune-based therapy for lung cancer will work, and a better understanding of these things is urgently needed.
2 Serum cytokine levels as possible immunotherapeutic targets
Small substances called serum cytokines play a crucial role in regulating other immune system cells’ development and function. When released, they give the immune system the go-ahead to function. All blood cell proliferation and the growth of other cells that support the body’s immunological and inflammatory systems are impacted by cytokines (27). By transmitting signals that can cause abnormal cells to die and normal cells to live longer, they also boost the body’s natural ability to fight cancer.
A chemokine is a special type of cytokine. Immune cells can be directed toward a target by chemokines. Chemokines come in various forms, including interleukins, interferons, tumor necrosis, and growth factors (28). In response to bacterial products and early response inflammatory mediators, resident lung cells secrete chemokines, which are small (~8–10 kD) heparin-binding proteins. These proteins are then preserved by matrix heparin sulfate proteoglycans at the site of inflammation, forming a chemokine gradient toward the inflammatory focus. Chemokines have chemotactic and activating actions on leukocyte subsets and serve as an essential stimulus for driving leukocytes to sites of injury. Chemokines are divided into four subfamilies based on the location of cysteine residues: CXC (α), CC (β), C (γ), and CX3C (δ) (29). The CXC (α) cysteine motif consists of the first two NH2-terminal cysteines separated by one nonconserved aa residue. Each subfamily stimulates a particular cell type by binding to specific receptors on inflammatory cells (30, 31).
The lung cancer chemokine axis that has attracted the most focus to date is CXCL12/CXCR4, which has been associated with increased invasiveness and metastatic risk (32). DARC-expressing A549 cells in lung cancer were found to have significantly decreased cellularity, higher necrosis, lower microvessel density, and less metastasis (33). D6 may slow the growth of human NSCLC by trapping specific chemokines such as CCL2, CCL4, and CCL5 (34). Furthermore, a phase 1 clinical trial of immunotherapy discovered that administering DCs expressing the CCR7 receptor ligand CCL21 into lung cancers could enhance immunity and cause the tumor to shrink (35). It was discovered that the CCR5 ligand CCL4 increased the expression of the stromal-derived factor-erythroid differentiation regulator 1 (ERDR1), which might assist in the survival of cancer cells (36). As a result, the CCL4/CCR5 axis appears responsible for the progression and proliferation of lung tumors. Future studies on individuals with lung cancer should be undertaken to assess the results.
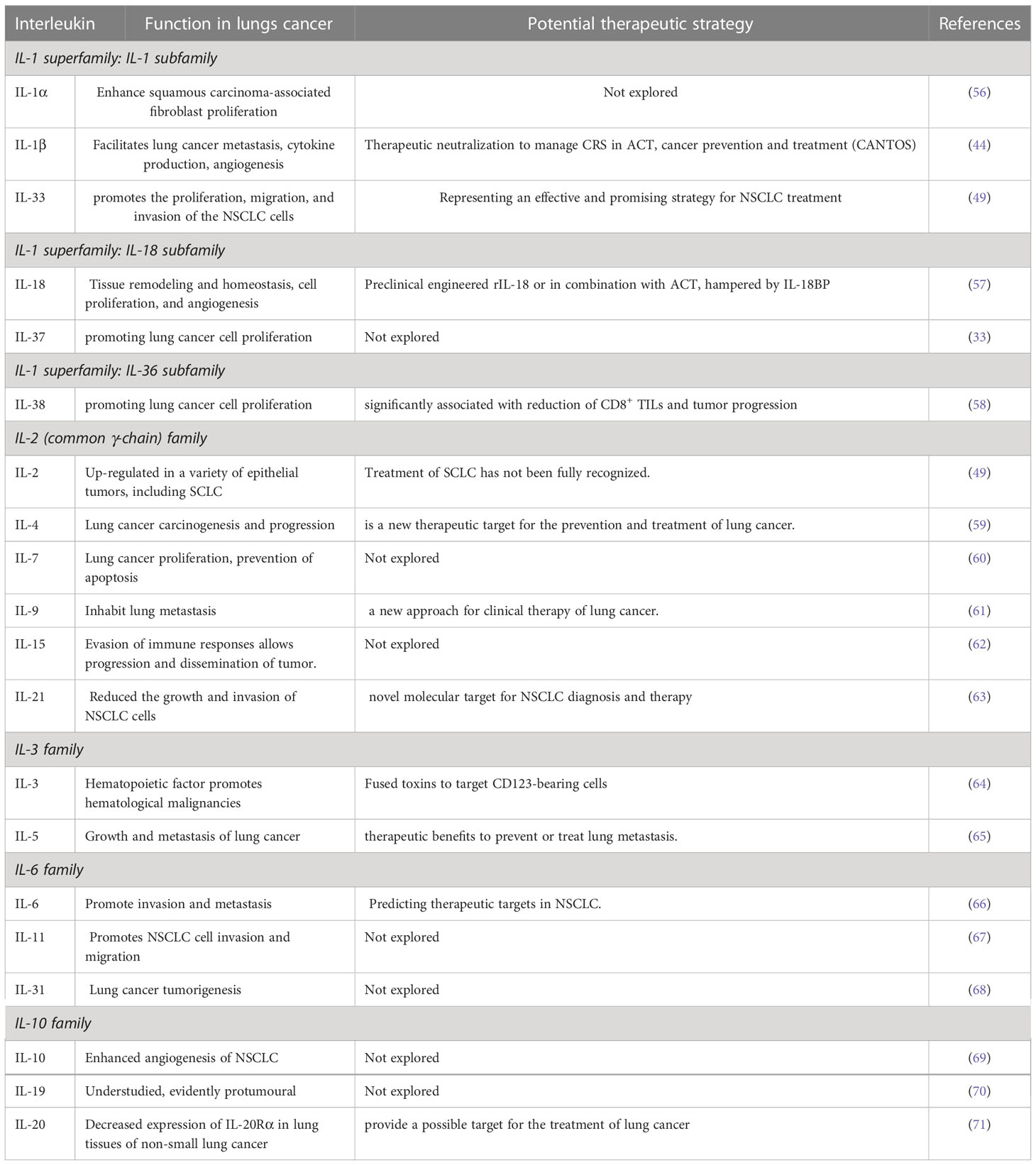
2.1 Interleukins
Important inflammatory cytokines include interleukins (ILs). In the human genome, more than 50 interleukins and associated proteins are encoded (37). The growth and occurrence of human malignant tumors are thought to be intimately tied to interleukins, which are also clinically used as diagnostic indicators for lung cancer (38). Previously, IL-1β (beta) and IL-1α (alpha) performed pro-tumorigenic functions in many malignancies and are double the primary serum mediators in the IL-1 family. Increased IL-1β promotes lung cancer spread by triggering angiogenesis, tumor growth, invasion, adhesion, cytokine production, and tumor epithelial-to-mesenchymal transition (39). In addition, the use of serum interleukin-1β (IL-1β), interleukin-6 (IL-6), and interleukin-8 (IL-8) in combination with carcinoembryonic antigen (CEA) as a biomarker panel for the detection and prediction of lung cancer metastasis are being studied (40). For example, several investigations have identified IL-1β as a prognostic factor in lung cancer, with elevated levels of IL-1β in blood or tumor tissue associated with poor existence (41).
In the tumor tissue and serum of NSCLC patients, IL-33 levels were noticeably elevated. The in vitro function investigation showed that IL-33 strongly encourages NSCLC cell proliferation, migration, and invasion. IL-33’s pro-tumor effect in NSCLC was further supported by in vivo research. Additionally, lung cancer has a bad prognosis when IL-33 is expressed at a lower level (42). Lung cancer was linked to elevated levels of the serum cytokines IL6 and IL8, and high levels of IL8 raised lung cancer risk. Research has been shown to elaborate on the potential effects of IL-37 on tumor growth, immune responses, and tumor angiogenesis. It has been discovered to play an anti-tumor function in various tumor types, including non-small cell lung cancer (43). The non-small cell lung cancer (NSCLC) patient’s potential IL-37 regulation mechanism is poorly understood (44). IL-38, a recently discovered anti-inflammatory factor in non-small cell lung cancer (NSCLC), makes up around 85% of all lung malignancies (45).
The FDA approved the use of high-dose interleukin-2 (IL-2) therapy in 1992 for metastatic renal cell carcinoma and in 1998 for the treatment of metastatic melanoma (46). A different meta-analysis demonstrates that IL-2 combination medication effectively cures non-small cell lung cancer (NSCLC), increases overall survival, and has few harmful side effects (47). In individuals with advanced NSCLC, IL-2 serum level determination by regression analysis was demonstrated to have independent predictive value; as a result, its potential application for outcome prediction is advocated (48).
Human interleukin-3 (IL-3) assesses the tolerance, hematologic effects, and safety in individuals with small-cell lung cancer (SCLC) before and following multi-agent antineoplastic therapy in a randomized, placebo-controlled, double-blind study (49). Human lung tumor cell lines express interleukin 4 (IL-4) receptors, and IL-4 can mediate mild to moderate antiproliferative action in vitro and in vivo in animal examples of human lung tumors (50). Ten blood cytokine levels, including; granulocyte-macrophage colony-stimulating factor, TNF (alpha), and IFN (gamma), IL1, IL6, IL12, IL4, IL8, IL10, IL5, were compared between 296 European-Americans and 170 African-Americans to determine their associations with lung cancer (51).
Bezel et al. measured 16 cytokine levels of tumor necrosis factor-alpha (TNF-α), GM-CSF, interferon-gamma (IFN-γ), interleukins (IL): IL-6, IL-7, IL-13, IL-17A, IL-23, IL-1b, IL-2, IL-8, IL-10, IL-12p70, IL-4, IL-5) and fractalkine, in bronchoalveolar lavage fluid (BALF) and serum of individuals with lung cancer compared to healthy persons (52). In preclinical investigations, GM-CSF, CCL21, interleukin-2 (IL-2), IL-21, IL-12, IL-15, IL-18, and type 1 interferon have been found to have anticancer action and encourage the development of CD4 T cells into Th1 cells that can secrete anti-tumor cytokines, such as IFN-γ and IL-2 (53).
Our findings may offer new support for preventing and managing lung tumor-associated IPF (idiopathic pulmonary fibrosis), which Zhang et al. first discovered to have higher levels of IL-22, IL-23, and IL-17 in the serum of patients with IPF (54). According to several recent studies, interleukin-33 has been linked to the advancement of lung cancer and can have opposing effects on the disease depending on the circumstances (3). Initially produced by regulatory T cells, IL-35 is a component of the interleukin-12 cytokines family. Significant evidence suggests that elevated blood IL-35 expression is linked to non-small cell pulmonary cancer in individuals with stage IV NSCLC (55).
According to Zhao and their colleagues, individuals with pulmonary cancer who experienced immune-related adverse events (AE) following immunotherapy had greater pre-treatment levels of IFN-α, IL-2, and IL-17 than those who experienced nonimmune-related adverse events (p=0.002, p=0.01, and p=0.02, respectively) (NAE). Before the second cycle of medication, individuals with AE compared to those with NAE had significantly higher variations in IFN-α (p=0.003), IFN-γ (p=0.012), and TNF-α (p=0.049), IL-2 (p=0.04), and IL-5 (p=0.007) levels (30) (See Table 1).
2.2 Interferons
When a pathological compromise occurs in our body, interferons, which are cytokines, are naturally created by our cells. These chemical messengers make the nearby normal cells resistant to the same type of infection (72). Beta IFN-β, generated by fibroblasts, macrophages, and epithelial cells; gamma IFN-y, produced by activated T-lymphocytes and natural killer lymphocytes; and alpha IFNα, produced by leukocytes, are the three forms of interferon (IFNβ) (73). Interferons are a good treatment option for cancer since they control angiogenesis, have immunomodulatory abilities, and so forth. Even in the advanced stages of both SCLC and NSCLC lung cancer, interferons have been proven beneficial (74).
An anti-HER2 monoclonal antibody has been shown to target IFN-β and increase the immunocytokine’s potency compared to either agent alone in a previous investigation. These new results imply that trastuzumab-IFN-mutein warrants clinical study as a new contender for anticancer treatments (75). IFNs may have biological effects on small cell lung cancer, according to preclinical and clinical research (SCLC). Non-small cell lung cancer studies have indicated that IFN-alpha is ineffective (NSCLC). Nonetheless, the potential specific potentiation of cisplatinum by IFN against human NSCLC xenografts is a fresh, encouraging discovery (76). These preliminary findings show that chemotherapy with IFN-alpha and cisplatin may be a beneficial alternative treatment for those with advanced NSCLC (77). The GO/G1 phase of the cell cycle looked to be where the cells accumulated due to IFNα, having an antiproliferative effect (78). IFN α & β (type I IFN) has been extensively tested in numerous clinical cases and experimental lung cancer models and is effective. However, other types and subtypes of interferons, such as IFN gamma, have also been tested in numerous lung cancer cases with varying degrees of success (79).
In a direct cytotoxic impact, IFNα and IFNy work together synergistically to promote the lysis of cancerous cells (80). All three types of interferon increase the expression of cell surface antigens, such as tumor necrosis factor (TNF) receptors and MHC antigens, making tumor cells easier for cytotoxic leukocytes or TNF, a cytokine with cytostatic solid and cytotoxic effects, to recognize (81). Like other cancers of neuroendocrine origin, SCLC cells exhibit low endogenous levels of class I antigen expression. They may therefore be able to avoid immune monitoring and spread more quickly in living things (82). IFNa and IFNy cause both SCLC and non-small cell lung cancer (NSCLC) cells to express HLA-A, B, C, and 2 microglobulin on their cell surfaces (83). IFNy can induce class II MHC antigens. Crawford has proven that the peripheral blood monocytes of patients with advanced NSCLC are activated by IFNy, as shown by a 50% or greater increase in the expression of the Fc receptor and 2 microglobulins by these cells (84).
2.3 Tumor necrosis factors
The multifunctional serum cytokine tumor necrosis factor (TNF) is crucial for various cellular processes, including cell proliferation, differentiation, survival, and death. Inflammatory cells release TNF, a cytokine that promotes inflammation and may contribute to cancer development (85). Nuclear factor b (NF-B) and c-Jun N-terminal kinase are (JNK). NF-κB is just two of the different signaling pathways that TNF uses to carry out its biological actions (JNK). While chronic JNK activation promotes cell death, NF-κB is an anti-apoptotic key cell survival signal (86). In determining cellular responses to TNF, NF-κB and JNK interact. TNF is duplicitous in cancer (87). TNF enhances tumor cell growth, invasion, proliferation, and tumor angiogenesis, as well as metastasis. Hence TNF may be an endogenous tumor promoter. TNF, however, might prevent cancer from developing (88). TNF can cause the death of cancer cells, making it a potential cancer treatment. However, much work is needed to lower its toxicity before TNF may be administered regularly (89).
Several tissues produce TNF, which is induced to express itself in response to inflammatory stimuli such as lipopolysaccharides (LPS). Notably, TNF is known to be released by cancerous cells in the tumor microenvironment (90). Toxicity and endotoxic shock have severely restricted the use of TNF for therapeutic purposes. Additionally, numerous investigations have revealed that TNF plays an oncogenic function in cancers linked to inflammation (91). TNF activates several inflammatory signaling networks by binding to its cognate receptors, TNF receptor 1 (TNFR1) or TNF receptor 2 (TNFR2) (92). TNFR1 is extensively distributed. However, TNFR2 is expressed mainly by immune and endothelial cells. Lung cancer has been found to have high levels of the cytokine and its receptors TNFR1 and TNFR2 (93).
Non-small cell lung cancer has a high expression of TNF and its receptors (NSCLC). The transcription factor NF-BNF-κB is significantly activated by TNF. According to studies, EGFR suppression causes TNF to be quickly increased in NSCLC, and this overexpression activates NF-NF-κB (94). The serum mediator of innate immunity responsible for causing hemorrhagic necrosis in tumors, TNF-alpha, was first discovered in the 1970s (95). TNFα levels were previously examined in 28 new patients with advanced-stage NSCLC before and after chemotherapy, as well as 15 healthy controls. The results showed that NSCLC patients had greater levels of TNF-α than controls (96). The first clinically usable TNF family-based signature for predicting prognosis and chemotherapy effectiveness for patients with SCLC was developed because little is known about TNF’s functions in small-cell lung cancer. The results presented here offer a novel approach to assessing the outlook of SCLC individuals and enhancing clinical care (97).
Treatment for 16 individuals with advanced non-small cell lung cancer included low-dose interleukin-2 and tumor necrosis factor. Enhanced lysis of autologous tumor in vitro was shown in four out of four patients throughout therapy, and all patients had increased lymphokine-activated killer and natural killer activity. We conclude that low-dose interleukin-2 and tumor necrosis factor-a immunotherapy can mediate tumor regression while posing manageable harm (98, 99). Such findings imply that tumor necrosis factors may impart pulmonary cancer cells’ resistance to future reactive oxygen species-based therapies. This resistance may result from these cells’ enhanced production of manganese superoxide dismutase. Clinical therapy failures could occur, mainly if a tumor necrosis factor is administered along with other medications (100).
2.4 Transforming growth factor-beta
There are three isoforms of transforming growth factor-beta (TGF-β). TGF-β is a versatile cytokine typically overexpressed in advanced cancers and associated with a poor prognosis. TGF-β’s function is context-dependent. TGF-β inhibits cellular proliferation, promotes cell apoptosis, and reduces inflammation, acting as a tumor suppressor for pre-malignant cells. For advanced malignancies, TGF-β encourages immune escape, treatment resistance, and distant metastasis. TGF-β may control the actions of a variety of immune cells, including regulating the development of regulatory T cells (Tregs), lowering the cytotoxicity of T cells and natural killer cells (NKs), and suppressing the antigen presentation of dendritic cells (DCs) (101). In addition, TGF-β limits immune cell infiltration by promoting the production of peritumoral collagen. Anti-PD-1/PD-L1 therapy had a minimal impact on TMEs with overactive TGF-β signaling. The TGFβ1 gene expression is higher in the tumor tissues of non-responders following anti-PD-1/PD-L1 treatments. In line with this, blocking PD-1/PD-L1 and TGF-β simultaneously produces an anti-tumor effect (102). Recently, a bispecific antibody (YM101) that targets both TGF-β and PD-L1 has been developed for cancer treatment (103).
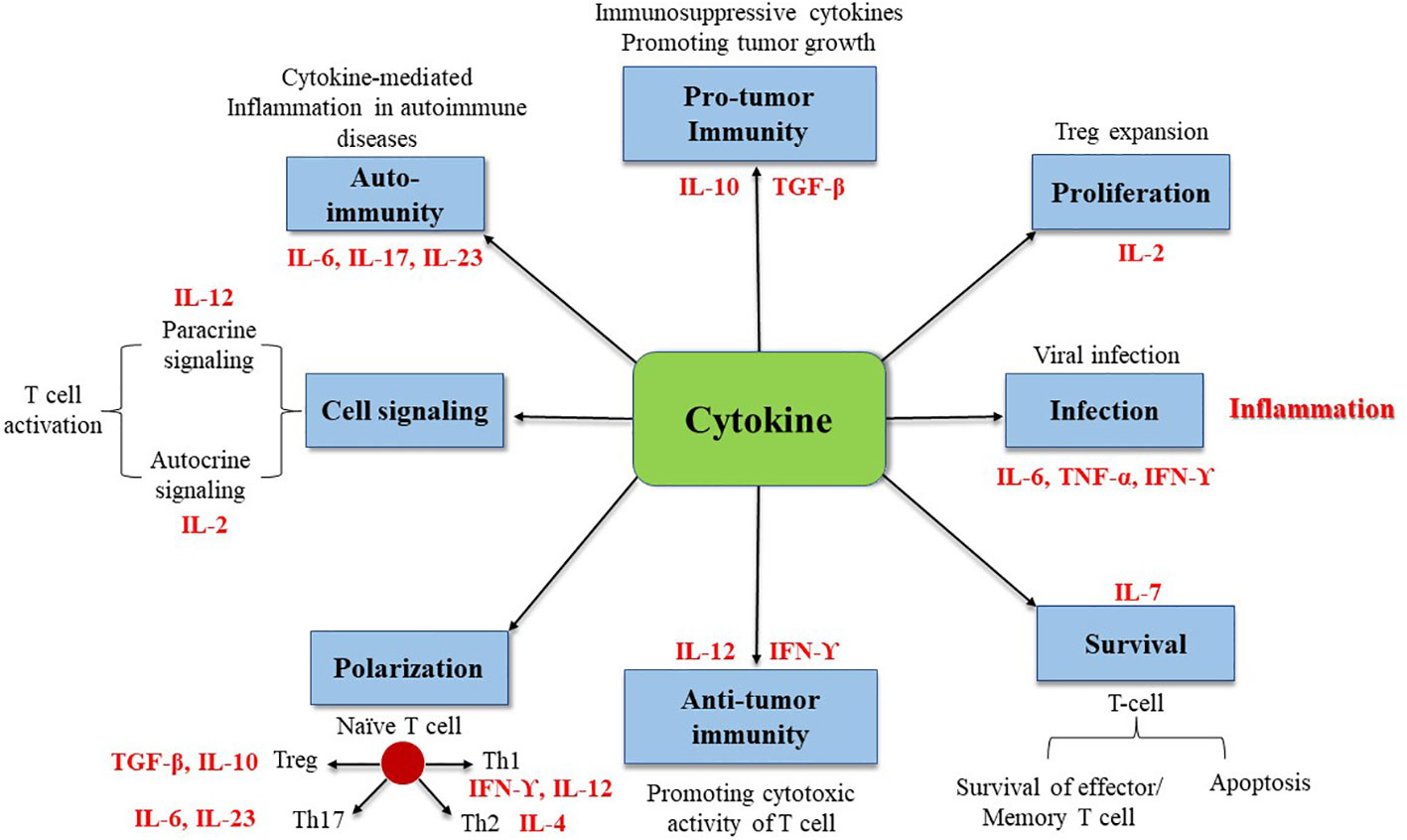
In summary, cytokines are the primary modulators of the innate and adaptive immune systems, primarily responsible for maintaining immunological homeostasis and regulating immune responses to infection, autoimmune diseases, and cancer. Cytokine roles are complicated and diverse. They can protect the body, but excessive activation or a severe lack can induce autoimmune disorders or promote cancer development (Figure 1).
3 Additional factors as prognostic indicators for lung cancer
Anorexia, performance status at the diagnosis, liver or skin metastases, and other conditions are additional indicators of a poor prognosis for NSCLC (104). According to molecular research, individuals with EGFR-activating mutations in adenocarcinoma had a better prognosis than patients without these mutations (105). Furthermore, the most significant prognostic factor for SCLC is the severity of the disease and the stage at presentation. The five-year mortality percentage for people with the restricted stage of cancer ranges from 10 to 13%, whereas the rate for those with the widespread phase of the infection is from 1 to 2%. (106)
It has been proposed that the following characteristics are linked to a better prognosis: Low microvessel count; low VEGF expression; absence of the KRAS mutation; no overexpression of c-erbB-2; No EGFR expression; TTF-1 positivity; high level of p16 expression; low-class III β-tubulin expression, low or no ERCC1 expression (advanced NSCLC treated with platinum-based chemotherapy); in individuals who had their tumors removed; low survivin expression. In a meta-analysis based on individual data, microvessel number was confirmed as a predictive factor for the prognostic value of angiogenesis, but only when it was estimated using the Chalkley procedure (107).
3.1 KLRK1
In a bioinformatics study, a Novel investigation found that KLRK1 is a prognostic and diagnostic biomarker for lung cancer (54). A homodimeric lectin-like receptor, KLRK1, encodes NKG2D. (108). KLRK1 is distinctive since it lacks inhibitory isoforms and functions as a cytotoxic and co-stimulatory molecule on T cells and NK cells. Two distinct KLRK1 ligands are found in the MIC and RAET1 gene families (109). KLRK1 is involved in regulating infection and tumor growth due to the frequent expression of its ligands on primary tumor cells (110). Phase I/II lung adenocarcinoma (P = 0.0094), older patients (P = 0.0072), male (P = 0.0033), and KLRK1 were all found to have substantial predictive significance. In phase I/II lung adenocarcinoma tumor individuals (P = 0.0025) and older patients (P = 0.012), KLRK1 was found to have substantial predictive significance (54).
3.2 ARL14
The expression of ARL14 in tumor tissues and standard samples was compared using immunohistochemistry (IHC). In NSCLC, we assessed ARL14’s predictive value. Higher ARL14 expression was linked to residual tumor in lung adenocarcinoma (P = 0.017), although it was related to age (P = 0.003) and N stage (P = 0.009) in lung squamous cell carcinoma. One hundred twenty patients with NSCLC provided similar outcomes. ARF7, FLJ22595, and ADP ribosylation factor-like GTPase 14 (ARL14) are protein-coding genes involved in GTP binding and signal transmission. ARL14 has recently been implicated in several biological activities, including vesicle-mediated and intracellular protein transport (111). Patients with bladder cancer with high levels of ARL14 methylation have a worse prognosis (112). ARF3, a homolog of ARL14, is said to express itself insufficiently in gastric cancer and may function as a possible biomarker for the prognosis of gastric cancer (113).
3.3 Gender as a prognostic factor
Women have a longer survival rate than men, according to numerous studies on gender as a prognostic factor in NSCLC. This may be related to biological variations between the sexes and/or confounding elements specific to women NSCLC individuals, such as smoking behaviors, histology, and young age (114). Individuals with small-cell lung cancer do not have gender-specific prognostic significance (SCLC). Eighty-eight women, or 14.9%, of the 591 patients with SCLC. Regarding minimal disease (48% vs. 37.8%) and no history of smoking (48.9% vs. 2.0%), women outnumbered males in likelihood. (52.3% vs. 62.8%) Men and women both had a progressive illness at the M stage. The complete cohort’s median survival times (MSTs) and 95% confidence intervals showed that women fared better than men in terms of survival (115). There were 144 NSCLC patients, all of whom had advanced disease. It was more common to see female patients. The most prevalent histologic type in both sexes (males, n = 54, 77.1%; females, n = 65, 87.8%) was adenocarcinoma (n = 119, 82.6%). (116).
3.4 TNM phase
According to the TNM classification, the clinical stage is the best indicator of prognosis in NSCLC (117). Distant metastases, tumor size, and nodal involvement are independently potent prognostic factors that are evaluated using the International Association for the Study of Lung Cancer (IASLC) international database of more than 80,000 lung cancer cases; the findings are thoroughly validated and frequently updated by the IASLC (118). However, within each stage group, there is much variation; some individuals advance their illness quickly, while others live for a very long time without experiencing a recurrence. Therefore, finding patient and tumor-related features is necessary to categorize patients according to their stage and individually tailor their treatments (119).
3.5 Age
The effects of age as a prognostic factor in NSCLC have been extensively explored. However, the outcomes are mixed. The medical features of lung tumors in individuals under the age of 45 are unusual and distinct from those in older people. Young individuals with advanced phase IIIB or IV (NSCLC) are being studied for their prognosis and outcomes. However, frequently, they are underrepresented in clinical trials, and older people (those over 65) with cancer have a high rate of cancer diagnoses (120). A randomized trial comparing the finest psychosocial support for individuals under 70 with adjuvant vinorelbine showed a substantial protective role and greater life satisfaction for the chemotherapy category (121).
3.6 Weight loss
The prognosis of many cancers, including NSCLC, is adversely affected by involuntary weight loss, a concerning indicator in oncology. Additionally, sarcopenia—the loss of skeletal muscle mass—is a potent predictor of poor prognosis distinct from weight loss (122). The proportion of body weight lost over a given period is a common way for writers to categorize weight reduction in their works. For patients with a BMI under 20, weight loss of more than 5% during the past six months (or more than 2%) qualifies as cancer cachexia (123). Patients who stabilize their weight after treatment have a better effect than those who continue to lose weight (124).
3.7 Performance status
Patients’ general health and everyday activities can be measured using their performance status. The ECOG score is more frequently used for performance status assessment in NSCLC because it is more widely utilized and easier to use than the KPS. It has also demonstrated higher predictive ability in patients with lung cancer (125). The inherent subjectivity of performance status and the weak-to-moderate connection in patient and doctor ratings are two issues that raise some doubts about its appropriateness. However, performance status is among the most well-researched prognostic markers, and its predictive value has been proven in several significant malignancies. The prevalence of low-Performance status in individuals with lung tumors compared to those with other cancers is not well understood, even though it is a common occurrence. Patients and clinicians estimated poor PS to be present in lung cancer patients at 34% and 48%, respectively (126).
3.8 Anemia
The typical definition of anemia is a reduction in the blood’s ability to carry oxygen due to a drop in hemoglobin (Hgb), a component of red blood cells. Hgb levels of less than 120 g/L for non-pregnant women and 130 g/L for men were used by a WHO study group in 1968 to identify anemia in adults (127). About 52% of tumor patients will become anemic during their illness, and 21% will need blood transfusions. Anemia is frequent in cancer patients (128). Anemia in cancer patients can be multifactorial and be brought on by cancer-related bleeding, cytokines produced by the tumor itself, or disruption of hematopoiesis brought on by metastatic bone marrow infiltration (129). Some studies suggest that pre-treatment anemia has a negative prognostic impact on NSCLC patients, but the function of anemia as a prognostic marker is debatable (130).
3.9 Leukocytosis
Patients with NSCLC typically experience leukocytosis, defined as an unusually high amount of WBCs in the peripheral blood. Infection, bone marrow metastases, and corticosteroids are some of the causes, either singularly or in combination (131). Nevertheless, other NSCLC patients exhibit leukocytosis for no apparent reason; tumor-related leukocytosis may be present (132). Interleukin-6, granulocyte-macrophage colony-stimulating factor (GM-CSF), and Granulocyte colony-stimulating factor (G-CSF) are three of the hematological cytokines that the tumor produces and secretes to create this paraneoplastic illness (133).
3.10 Thrombocytosis
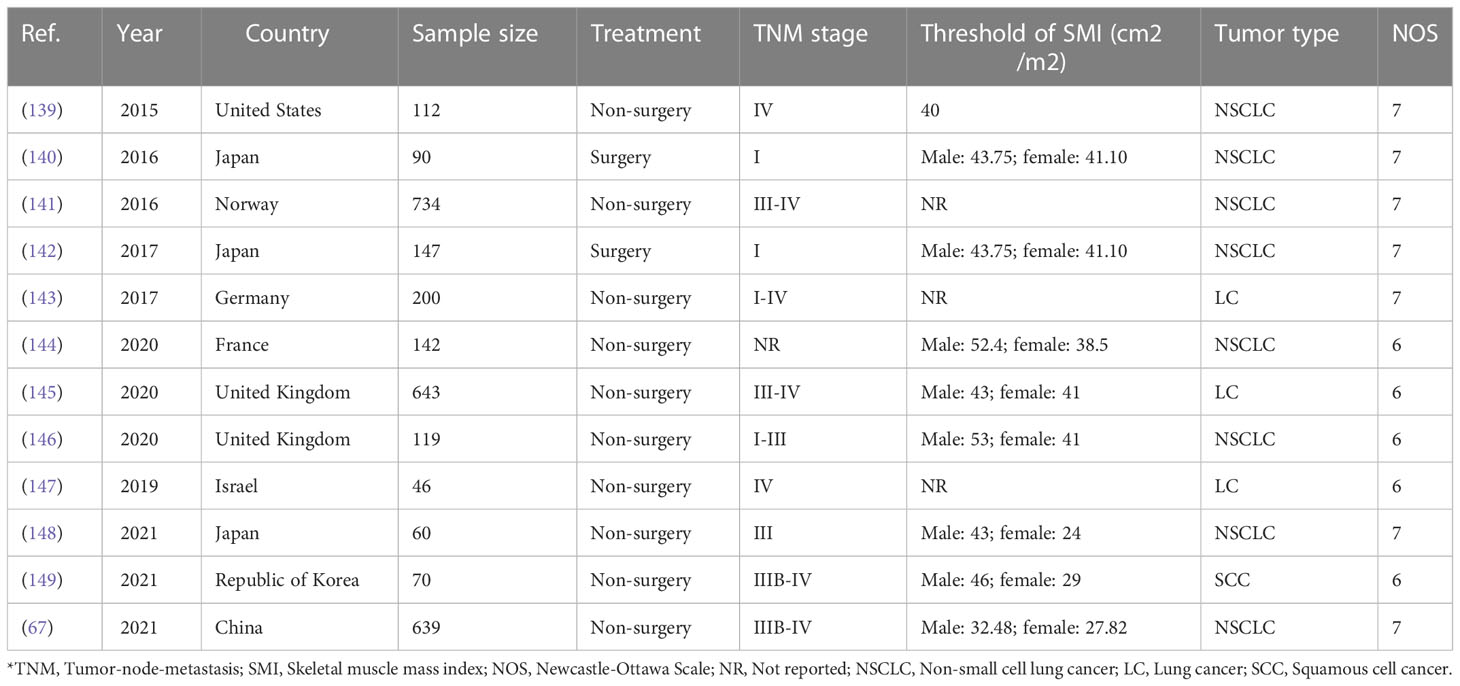
It has long been understood that thrombocytosis, an unusually high amount of platelets in blood circulation, is associated with malignant illness (134). In the same way that leukocytosis can be a paraneoplastic condition, thrombocytosis can be brought on by an increase in the tumor’s cytokine production. Numerous studies have found that it is a poor predictor of survival for people with lung cancer (135). However, some studies do not find this correlation, such as a pooled study of the NCCT (North Central Cancer Treatment Group) findings, including information from more than 1000 individuals with advanced-stage NSCLC (136). These findings suggested that platelet counts and several clinical-pathologic traits could be helpful prognostic variables in patients with unresectable NSCLC. The patients were 347 consecutive phase IIIB or IV NSCLC individuals treated in the medical oncology department between 2005 and 2009. (137). According to meta-analyses, 27% of individuals with lung cancer had thrombocytosis overall. This was divided into 22% for adenocarcinoma, 28% for squamous cell carcinoma (SCC), 36% for large cell carcinoma (LCC), and 30% for SCLC (138). Thrombocytosis and a bad prognosis have been linked, but the exact mechanism is unclear (Table 2).
4 Conclusion and future perspectives
Finally, we determined that serum cytokine significantly impacts patients with lung cancer. In a disease where tumor biomarkers studies are challenging, baseline levels and treatment changes may serve as efficient predictive biomarkers of success from adding new drugs to treatment. This comprehensive evaluation of the literature revealed significant relationships between many cytokines and metabolic tumor burden, but the findings will need to be verified in a larger cohort.
Author contributions
Study concept: YZ and LZ. Data collection: YZ, SJ, and KZ. Draft preparation: YZ, SJ, KZ, and LZ. Supervision: LZ. All authors read and agreed to the publication of this article.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fonc.2023.1064616/full#supplementary-material
References
1. Bray F, Ferlay J, Soerjomataram ISiegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates incidence and mortality worldwide for 36 cancers in 185 countries. Ca - Cancer J. Clin (2018) 68, 394–424.
2. Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin (2021) 71:209–49. doi: 10.3322/caac.21660
3. Yang J, Chen Z, Gong Z, Li Q, Ding H, Cui Y, et al. Immune landscape and classification in lung adenocarcinoma based on a novel cell cycle checkpoints related signature for predicting prognosis and therapeutic response. Front Genet (2022) 1209. doi: 10.3389/fgene.2022.908104
4. Lara MS, Brunson A, Wun T, Tomlinson B, Qi L, Cress R, et al. Predictors of survival for younger patients less than 50 years of age with non-small cell lung cancer (NSCLC): A California cancer registry analysis. Lung Cancer (2014) 85(2):264–9. doi: 10.1016/j.lungcan.2014.04.007
5. Herbst RS, Morgensztern D, Boshoff C. The biology and management of non-small cell lungCancer. Nature (2018) 553:446–54. doi: 10.1038/nature25183
6. Engels EA. Inflammation in the development of lung cancer: epidemiological evidence. Expert Rev Anticancer Ther (2008) 8(4):605–15. doi: 10.1586/14737140.8.4.605
7. Hardy-Werbin M, Rocha P, Arpi O, Taus Á., Nonell L, Durán X, et al. Serum cytokine levels as predictive biomarkers of benefit from ipilimumab in small cell lung cancer. Oncoimmunology (2019) 8(6):e1593810. doi: 10.1080/2162402X.2019.1593810
8. Kang JH, Bluestone JA, Young A. Predicting and preventing immune checkpoint inhibitor toxicity: targeting cytokines. Trends Immunol (2021) 42(4):293–311. doi: 10.1016/j.it.2021.02.006
9. Enewold L, Mechanic LE, Bowman ED, Zheng YL, Yu Z, Trivers G, et al. Serum concentrations of cytokines and lung cancer survival in African americans and caucasians. Cancer Epidemiol Biomarkers Prev (2009) 18(1):215–22. doi: 10.1158/1055-9965.EPI-08-0705
10. Liu C, Yang L, Xu H, Zheng S, Wang Z, Wang S, et al. Systematic analysis of IL-6 as a predictive biomarker and desensitizer of immunotherapy responses in patients with non-small cell lung cancer. BMC Med (2022) 20(1):1–15. doi: 10.1186/s12916-022-02356-7
11. Holder PG, Lim SA, Huang CS, Sharma P, Dagdas Y, Bulutoglu B, et al. Engineering interferons and interleukins for cancer immunotherapy. Advanced Drug Delivery Rev (2022) 114112. doi: 10.1016/j.addr.2022.114112
12. Fukuyama T, Ichiki Y, Yamada S, Shigematsu Y, Baba T, Nagata Y, et al. Cytokine production of lung cancer cell lines: Correlation between their production and the inflammatory/immunological responses in vivo and in vitro. Cancer Sci (2007) 98:1048 – 54. doi: 10.1111/j.1349-7006.2007.00507.x
13. Kaanane H, Senhaji N, Berradi H, Benchakroun N, Benider A, Karkouri M, et al. The influence of interleukin-6, interleukin-8, interleukin-10, interleukin-17, TNF-a, MIF, STAT3 on lung cancer risk in Moroccan population. Cytokine (2022) 151:155806. doi: 10.1016/j.cyto.2022.155806
14. Grivennikov SI, Greten FR, Karin M. Immunity, inflammation, and cancer. Cell (2010) 140(6):883–99. doi: 10.1016/j.cell.2010.01.025
15. Propper DJ, Balkwill FR. Harnessing cytokines and chemokines for cancer therapy. Nat Rev Clin Oncol (2022) 19(4):237–53. doi: 10.1038/s41571-021-00588-9
16. Shi Y, Liu X, Du J, Zhang D, Liu J, Chen M, et al. Circulating cytokines associated with clinical outcomes in advanced non-small cell lung cancer patients who received chemoimmunotherapy. Thorac Cancer (2022) 13(2):219–27. doi: 10.1111/1759-7714.14248
17. Garg A, Iyer H, Jindal V, Vashistha V, Ali A, Jain D, et al. Prognostic factors for treatment response and survival outcomes after first-line management of stage 4 non-small cell lung cancer: A real-world Indian perspective. Lung India: Off Organ Indian Chest Soc (2022) 39(2):102. doi: 10.4103/lungindia.lungindia_408_21
18. Garinet S, Laurent-Puig P, Blons H, Oudart JB. Current and future molecular testing in NSCLC, what can we expect from new sequencing technologies? J Clin Med (2018) 7(6):144. doi: 10.3390/jcm7060144
19. Rizvi NA, Mazières J, Planchard D, Stinchcombe TE, Dy GK, Antonia SJ, et al. Activity and safety of nivolumab, an anti-PD-1 immune checkpoint inhibitor, for patients with advanced, refractory squamous non-small-cell lung cancer (CheckMate 063): A phase 2, single-arm trial. Lancet Oncol (2015) 16:257–65. doi: 10.1016/S1470-2045(15)70054-9
20. American Cancer Society. Non-small cell lung cancer survival rates by stage (2016). Available at: http://www.cancer.org/cancer/lungcancernon-smallcell/detailedguide/non-small-cell-lung-cancer-survival-rates.
21. Brodsky AN. Cancer immunotherapy: The year in review and a look at the year ahead. Cancer Research Institute, New York, USA (2019). Available at: https://www.cancerresearch.org/blog/january2019/cancer-immunotherapy-2018-review-2019-predict.
22. Gupta M, Chandan K, Sarwat M. Natural products and their derivatives as immune check point inhibitors: Targeting Cytokine/Chemokine signalling in cancer. In: Seminars in cancer biology. Academic Press, UK (2022).
23. Yanbaeva DG, Dentener MA, Creutzberg EC, Wesseling G, Wouters EF. Systemic effects of smoking. Chest (2007) 131:1557 – 66. doi: 10.1378/chest.06-2179
24. Zhou X, Feng Y, Liu S, Li C, Teng Y, Li X, et al. IL-33 promotes the growth of non-small cell lung cancer cells through regulating miR-128-3p/CDIP1 signalling pathway. Cancer Manage Res (2021) 13:2379. doi: 10.2147/CMAR.S276297
25. Canadian cancer society CCS. (2020). Prognosis and survival of cancer. Available at: https://cancer.ca/en/cancer-information/cancer-types/lung/prognosis-and-survival.
26. Abidoye O, Ferguson MK, Salgia R. Lung carcinoma in African americans. Nat Clin Pract Oncol (2007) 4:118 – 29. doi: 10.1038/ncponc0718
27. Berraondo P, Sanmamed MF, Ochoa MaríaC, Etxeberria Iñaki, Aznar MA, Pérez-Gracia JoséL, et al. Cytokines in clinical cancer immunotherapy. Br J Cancer (2019) 120(1):6–15. doi: 10.1038/s41416-018-0328-y
28. Kany S, Vollrath JT, Relja B. Cytokines in inflammatory disease. Int J Mol Sci (2019) 20(23):6008. doi: 10.3390/ijms20236008
29. Bhatia M, Zemans RL, Jeyaseelan S. Role of chemokines in the pathogenesis of acute lung injury. Am J Respir Cell Mol Biol (2012) 46(5):566–72. doi: 10.1165/rcmb.2011-0392TR
30. Zhao N, Yi Y, Cao W, Fu X, Mei N, Li C. Corrigendum: Serum cytokine levels for predicting immune-related adverse events and the clinical response in lung cancer treated with immunotherapy. Front Oncol (2022) 12. doi: 10.3389/fonc.2022.1069999
31. Zhao J, Chen X, Herjan T, Li X. The role of interleukin-17 in tumor development and progression. J Exp Med (2020) 217(1):e20190297. doi: 10.1084/jem.20190297
32. Saintigny P, Burger JA. Recent advances in non-small cell lung cancer biology and clinical management. Discovery Med (2012) 13(71):287–97.
33. Addison CL, Belperio JA, Burdick MD, Strieter RM. Overexpression of the Duffy antigen receptor for chemokines (DARC) by NSCLC tumor cells results in increased tumor necrosis. BMC Cancer (2004) 4(1):1–15. doi: 10.1186/1471-2407-4-28
34. Wu FY, Fan J, Tang L, Zhao YM, Zhou CC. Atypical chemokine receptor D6 inhibits human non-small cell lung cancer growth by sequestration of chemokines. Oncol Lett (2013) 6(1):91–5. doi: 10.3892/ol.2013.1358
35. Sharma S, Zhu L, Srivastava MK, Harris-White M, Huang M, Lee JM, et al. CCL21 chemokine therapy for lung cancer. Int Trends Immun (2013) 1(1):10.
36. Mango RL, Wu QP, West M, McCook EC, Serody JS, van Deventer HW. CC chemokine receptor 5 on pulmonary mesenchymal cells promotes experimental metastasis via the induction of erythroid differentiation regulator 1ERDR1 promotes metastasis. Mol Cancer Res (2014) 12(2):274–82. doi: 10.1158/1541-7786.MCR-13-0164
37. Brocker C, Thompson D, Matsumoto A, Nebert DW, Vasiliou V. Evolutionary divergence and functions of the human interleukin (IL) gene family. Hum Genomics (2010) 5(1):30–555. doi: 10.1186/1479-7364-5-130
38. Liu W, Wang H, Bai F, Ding L, Huang Y, Lu C, et al. IL-6 promotes metastasis of non-small-cell lung cancer by up-regulating TIM-4 via NF-kappaB. Cell Prolif. (2020) 53(3):e12776. doi: 10.1111/cpr.12776
39. Li R, Ong SL, Tran LM, Jing Z, Liu B, Park SJ, et al. Chronic IL-1β-induced inflammation regulates epithelial-to-mesenchymal transition memory phenotypes via epigenetic modifications in non-small cell lung cancer. Sci Rep (2020) 10(1):377. doi: 10.1038/s41598-019-57285-y
40. Yan X, Han L, Zhao R, Fatima S, Zhao L, Gao F. Prognosis value of IL-6, IL-8, and IL-1β in serum of patients with lung cancer: A fresh look at interleukins as a biomarker. Heliyon (2022) 8(8):e09953. doi: 10.1016/j.heliyon.2022.e09953
41. Millares L, Barreiro E, Cortes R, Martinez-Romero A, Balcells C, Cascante M, et al. Tumor-associated metabolic and inflammatory responses in early-stage non-small cell lung cancer: local patterns and prognostic significance. Lung Cancer (2018) 122(January):124–30. doi: 10.1016/j.lungcan.2018.06.015
42. Yang M, Feng Y, Yue C, Xu B, Chen L, Jiang J, et al. A lower expression level of IL-33 is associated with a poor prognosis of pulmonary adenocarcinoma. PloS One (2018) 13(3):e0193428. doi: 10.1371/journal.pone.0193428
43. Mei Y, Liu H. IL-37: An anti-inflammatory cytokine with anti-tumor functions. Cancer Rep (2019) 2(2):e1151. doi: 10.1002/cnr2.1151
44. Jiang Y, Liao H, Zhang X, Cao S, Hu X, Yang Z, et al. IL-33 synergistically promotes the proliferation of lung cancer cells in vitro by inducing antibacterial peptide LL-37 and proinflammatory cytokines in macrophages. Immunobiology (2020) 225(6):152025. doi: 10.1016/j.imbio.2020.152025
45. Wang F, Zhang W, Wu T, Chu H. Reduced interleukin-38 in non-small cell lung cancer is associated with tumor progression. R Soc Open Biol (2018) 8(10):180132. doi: 10.1098/rsob.180132
46. Han L, Jiang Q, Yao W, Fu T, Zeng Q. Thoracic injection of low-dose interleukin-2 as adjuvant therapy improves the control of the malignant pleural effusions: A systematic review and meta-analysis based on Chinese patients. BMC Cancer (2018) 18(1):1–15. doi: 10.1186/s12885-018-4581-5
47. Dehghan R, Beig Parikhani A, Zeinali S, Shokrgozar M, Amanzadeh A, Ajdary S, et al. Efficacy and anti-tumor activity of a mutant type of interleukin 2. Sci Rep (2022) 12(1):1–7. doi: 10.1038/s41598-022-09278-7
48. Shah P, Forget MA, Frank ML, Jiang P, Sakellariou-Thompson D, Federico L, et al. Combined IL-2, agonistic CD3, and 4-1BB stimulation preserve clonotype hierarchy in propagated non-small cell lung cancer tumor-infiltrating lymphocytes. J immunotherapy Cancer (2022) 10(2). doi: 10.1136/jitc-2021-003082
49. Mroczko B, Szmitkowski M, Czygier M. Interleukin 3 (IL-3) in diagnosis and monitoring of non-small-cell lung cancer. Przeglad Lekarski (1999) 56(12):763–6.
50. Safi S, Yamauchi Y, Hoffmann H, Weichert W, Jost PJ, Winter H, et al. Circulating interleukin-4 is associated with a systemic T cell response against tumor-associated antigens in treatment-naïve patients with resectable non-Small-Cell lung cancer. Cancers (2020) 12(12):3496. doi: 10.3390/cancers12123496
51. Pine SR, Mechanic LE, Enewold L, Bowman ED, Ryan BM, Cote ML, et al. Differential serum cytokine levels and risk of lung cancer between African and European AmericansSerum cytokines, race, and lung cancer risk. Cancer Epidemiology Biomarkers Prev (2016) 25(3):488–97. doi: 10.1158/1055-9965.EPI-15-0378
52. Bezel P, Valaperti A, Steiner U, Scholtze D, Wieser S, Vonow-Eisenring M, et al. Evaluation of cytokines in the tumor microenvironment of lung cancer using bronchoalveolar lavage fluid analysis. Cancer Immunology Immunotherapy (2021) 70(7):1867–76. doi: 10.1007/s00262-020-02798-z
53. Atallah-Yunes SA, Robertson MJ. Cytokine based immunotherapy for cancer and lymphoma: Biology, challenges, and future perspectives. Front Immunol (2022) 13. doi: 10.3389/fimmu.2022.872010
54. Zhang Q, Tong L, Wang B, Wang T, Ma H. Diagnostic value of serum levels of IL-22, IL-23, and IL-17 for idiopathic pulmonary fibrosis associated with lung cancer. Ther Clin Risk Manage (2022) 18:429. doi: 10.2147/TCRM.S349185
55. Li Z, Zhu L, Zheng H, Jiang W, Wang Y, Jiang Z, et al. Serum IL-35 levels are a new candidate biomarker of cancer-related cachexia in stage IV non-small cell lung cancer. Thorac Cancer (2022) 13(5):716–23. doi: 10.1111/1759-7714.14307
56. Froidure A, Ladjemi MZ, Pilette C. Interleukin-1α: A key player for epithelial-to-mesenchymal signaling in COPD. Eur Respir J (2016) 48(2):301–4. doi: 10.1183/13993003.01180-2016
57. Senju H, Kumagai A, Nakamura Y, Yamaguchi H, Nakatomi K, Fukami S, et al. Effect of IL-18 on the expansion and phenotype of human natural killer cells: Application to cancer immunotherapy. Int J Biol Sci (2018) 14(3):331. doi: 10.7150/ijbs.22809
58. Kinoshita F, Tagawa T, Akamine T, Takada K, Yamada Y, Oku Y, et al. Interleukin-38 promotes tumor growth by regulating CD8+ tumor-infiltrating lymphocytes in the lung cancer tumor microenvironment. Cancer Immunology Immunotherapy (2021) 70(1):123–35. doi: 10.1007/s00262-020-02659-9
59. Fu C, Jiang L, Hao S, Liu Z, Ding S, Zhang W, et al. Activating the IL-4/STAT6 signaling pathway promotes lung cancer progression by increasing M2 myeloid cells. Front Immunol (2019) 10:2638. doi: 10.3389/fimmu.2019.02638
60. Lin J, Zhu Z, Xiao H, Wakefield MR, Ding VA, Bai Q, et al. The role of IL-7 in immunity and cancer. Anticancer Res (2017) 37(3):963–7.
61. Park SM, Lee JO, Lee H, Kim YS. Interleukin-9 inhibits lung metastasis of melanoma by stimulating anti-tumor M1 macrophages. Molecules Cells (2020) 43(5):479. doi: 10.14348/molcells.2020.0047
62. Adhami F, Steel JC, Morris JC. Interleukin-15 expression in lung cancer. Journal of Clinical Oncology (2012) 30(15). doi: 10.1200/jco.2012.30.15_suppl.e21064
63. Xue D, Yang P, Wei Q, Li X, Lin L, Lin T. IL-21/IL-21R inhibits tumor growth and invasion in non-small cell lung cancer cells via suppressing Wnt/β-catenin signaling and PD-L1 expression. Int J Mol Med (2019) 44(5):1697–706. doi: 10.3892/ijmm.2019.4354
64. Testa U, Pelosi E, Castelli G. CD123 as a therapeutic target in treating hematological malignancies. Cancers (2019) 11(9):1358. doi: 10.3390/cancers11091358
65. Zaynagetdinov R, Sherrill TP, Gleaves LA, McLoed AG, Saxon JA, Habermann AC, et al. Interleukin-5 facilitates lung metastasis by modulating the immune MicroenvironmentIL5 facilitates lung metastasis. Cancer Res (2015) 75(8):1624–34. doi: 10.1158/0008-5472.CAN-14-2379
66. Ke W, Zhang L, Dai Y. The role of IL-6 in immunotherapy of non-small cell lung cancer (NSCLC) with immune-related adverse events (irAEs). Thorac Cancer (2020) 11(4):835–9. doi: 10.1111/1759-7714.13341
67. Putoczki TL, Ernst M. IL-11 signaling is a therapeutic target for cancer. Immunotherapy (2015) 7(4):441–53. doi: 10.2217/imt.15.17
68. Ferretti E, Corcione A, Pistoia V. The IL-31/IL-31 receptor axis: general features and role in the tumor microenvironment. J Leukoc. Biol (2017) 102:711–7. doi: 10.1189/jlb.3MR0117-033R
69. Ouyang W. &O'Garra, a. IL-10 family cytokines IL-10 and IL-22: from basic science to clinical translation. Immunity (2019) 50:871–91. doi: 10.1016/j.immuni.2019.03.020
70. Chen YY, Li CF, Yeh CH, Chang MS, Hsing CH. Interleukin-19 in breast cancer. Clin Dev Immunol (2013) 2013:294320. doi: 10.1155/2013/294320
71. Baird AM, Al-Sarraf N, Gray S, O'Byrne K. The role of IL-20 in lung cancer-a new epigenetic target? Cancer Res (2007) 67(9_Supplement):263–3. doi: 10.1016/S0169-5002(08)70029-1
72. Fujie H, Tanaka T, Tagawa M, Kaijun N, Watanabe M, Suzuki T, et al. Anti-tumor activity of type III interferon alone or in combination with type I interferon against human non-small cell lung cancer. Cancer Sci (2011) 102(11):1977–90. doi: 10.1111/j.1349-7006.2011.02079.x
73. Hodge G, Barnawi J, Jurisevic C, Moffat D, Holmes M, Reynolds PN, et al. Lung cancer is associated with decreased expression of perforin, granzyme b, and interferon (IFN)-γ by infiltrating lung tissue T cells, natural killer (NK) T-like, and NK cells. Clin Exp Immunol (2014) 178(1):79–85. doi: 10.1111/cei.12392
74. Yan J, Jiang Y, Lu J, Wu J, Zhang M. Inhibiting proliferation, migration, and invasion in lung cancer induced by silencing interferon-induced transmembrane protein 1 (IFITM1). BioMed Res Int (2019) 2019. doi: 10.1155/2019/9085435
75. Lee CS, Milone M, Seetharamu N. Osimertinib in EGFR-mutated lung cancer: A review of the existing and emerging clinical data. OncoTargets Ther (2021) 14:4579. doi: 10.2147/OTT.S227032
76. Niiranen A, Holsti LR, Cantell K, Mattson K. Natural interferon-alpha is combined with conventional therapies in non-small cell lung cancer. Acta Oncol (1990) 29(7):927–30. doi: 10.3109/02841869009096391
77. Chao TY, Hwang WS, Yang MJ, Chang JY, Wang CC, Hseuh EJ, et al. Combination chemoimmunotherapy with interferon-alpha and cisplatin in advanced non-small cell lung cancer patients. Zhonghua Yi Xue za zhi= Chin Med Journal; Free China ed (1995) 56(4):232–8.
78. Lundblad D. Studies on the antiproliferative action of interferon: effects on proteins synthesized in the G1 and s phase of the cell cycle in 2 anchorage-dependent cell lines. (1991), (Doctoral dissertation, Umeå Universitet), Sweden. Available at: http://urn.kb.se/resolve?urn=urn:nbn:se:umu:diva-100575
79. Ghosh D, Parida P. Interferon therapy in lung cancer: Current perspectives. Curr Cancer Ther Rev (2016) 12(4):237–45. doi: 10.2174/1573394713666170316124158
80. Hirashima T, Kanai T, Suzuki H, Yoshida H, Matsushita A, Kawasumi H, et al. The levels of interferon-gamma release as a biomarker for non-small-cell lung cancer patients receiving immune checkpoint inhibitors. Anticancer Res (2019) 39(11):6231–40. doi: 10.21873/anticanres.13832
81. Boutsikou E, Domvri K, Hardavella G, Tsiouda D, Zarogoulidis K, Kontakiotis T. Tumor necrosis factor, interferon-gamma, and interleukins as predictive markers of anti-programmed cell-death protein-1 treatment in advanced non-small cell lung cancer: A pragmatic approach in clinical practice. Ther Adv Med Oncol (2018) 10. doi: 10.1177/1758835918768238
82. Yu R, Zhu B, Chen D. Type I interferon-mediated tumor immunity and its role in immunotherapy. Cell Mol Life Sci (2022) 79(3):1–24. doi: 10.1007/s00018-022-04219-z
83. Kanai T, Suzuki H, Yoshida H, Matsushita A, Kawasumi H, Samejima Y, et al. The significance of quantitative interferon-gamma levels in non-small-cell lung cancer patient's response to immune checkpoint inhibitors. Anticancer Res (2020) 40(5):2787–93. doi: 10.21873/anticanres.14251
84. Karachaliou N, Gonzalez-Cao M, Crespo G, Drozdowskyj A, Aldeguer E, Gimenez-Capitan A, et al. Interferon-gamma is an important marker of response to immune checkpoint blockade in non-small cell lung cancer and melanoma patients. Ther Adv Med Oncol (2018) 10. doi: 10.1177/1758834017749748
85. Wang X, Lin Y. Tumor necrosis factor and cancer, buddies or foes? 1. Acta Pharmacologica Sin (2008) 29(11):1275–88. doi: 10.1111/j.1745-7254.2008.00889.x
86. Qu F, Xu W, Deng Z, Xie Y, Tang J, Chen Z, et al. The fish c-jun n-terminal kinase (JNK) pathway is involved in bacterial MDP-induced intestinal inflammation. Front Immunol (2020) 11:459. doi: 10.3389/fimmu.2020.00459
87. Park HJ, Lee HJ, Choi MS, Son DJ, Song HS, Song MJ, et al. The JNK pathway inhibits inflammatory target gene expression and NF-kappaB activation by melittin. J Inflammation (2008) 5(1):1–13.
88. Sarzi-Puttini P, Atzeni F, Doria A, Iaccarino L, Turiel M. Tumor necrosis factor-a, biologic agents, and cardiovascular risk. Lupus (2005) 14(9):780–4. doi: 10.1191/0961203305lu2220oa
89. Perez-Ruiz E, Minute L, Otano I, Alvarez M, Ochoa MC, Belsue V, et al. Prophylactic TNF blockade uncouples efficacy and toxicity in dual CTLA-4 and PD-1 immunotherapy. Nature (2019) 569:428–32. doi: 10.1038/s41586-019-1162-y
90. Sethi G, Sung B, Aggarwal BB. TNF: A master switch for inflammation to cancer. Front Biosci (2008) 13:5094–107. doi: 10.2741/3066
91. Balkwill F. Tumour necrosis factor and cancer. Nat Rev Cancer (2009) 9:361–71. doi: 10.1038/nrc2628
92. Brenner D, Blaser H, Mak TW. Regulation of tumor necrosis factor signaling: live or let die. Nat Rev Immunol (2015) 15:362–74. doi: 10.1038/nri3834
93. Gong K, Guo G, Panchani N, Bender ME, Gerber DE, Minna JD, et al. EGFR inhibition triggers an adaptive response by co-opting antiviral signaling pathways in lung cancer. Nat Cancer (2020) 1:394–409. doi: 10.1038/s43018-020-0048-0
94. Gong K, Guo G, Beckley N, Zhang Y, Yang X, Sharma M, et al. Tumor necrosis factor in lung cancer: Complex roles in biology and resistance to treatment. Neoplasia (2021) 23(2):189–96. doi: 10.1016/j.neo.2020.12.006
95. Josephs SF, Ichim TE, Prince SM, Kesari S, Marincola FM, Escobedo AR, et al. Unleashing endogenous TNF-alpha as a cancer immunotherapeutic. J Trans Med (2018) 16(1):1–8. doi: 10.1186/s12967-018-1611-7
96. Topuz E, Tas F, Argon A, Yildiz DD, Oguz H, Yasasever V, et al. Serum levels of leptin and proinflammatory cytokines in advanced stage non-small cell lung cancer. J Clin Oncol (2005) 23(16_suppl):2594–4. doi: 10.1200/jco.2005.23.16_suppl.2594
97. Zhang Z, Wu P, Zhang C, Luo Y, Zhang G, Zeng Q, et al. Tumor necrosis factor family member profile predicts prognosis and adjuvant chemotherapy benefit for patients with small-cell lung cancer. Front Immunol (2021) 12. doi: 10.3389/fimmu.2021.745769
98. Zhang B, Xu A, Wu D, Xia W, Li P, Wang E, et al. ARL14 as a prognostic biomarker in non-small cell lung cancer. J Inflammation Res (2021) 14:6557. doi: 10.2147/JIR.S340119
99. Yang SC, Owen-Schaub L, Mendiguren-Rodriguez A, Grimm EA, Hong WK, Roth JA, et al. Combination immunotherapy for non-small cell lung cancer: results with interleukin-2 and tumor necrosis factor-α. J Thorac Cardiovasc Surg (1990) 99(1):8–13. doi: 10.1016/S0022-5223(19)35625-9
100. Pogrebniak HW, Prewitt TW, Matthews WA, Pass HI, Wallace RB. Tumor necrosis factor-α alters the response of lung cancer cells to oxidative stress. J Thorac Cardiovasc Surg (1991) 102(6):904–7. doi: 10.1016/S0022-5223(19)33941-8
101. Yi M, Zhang J, Li A, Niu M, Yan Y, Jiao Y, et al. The construction, expression, and enhanced anti-tumor activity of YM101: A bispecific antibody simultaneously targeting TGF-β and PD-L1. J Hematol Oncol (2021) 14(1):1–22. doi: 10.1186/s13045-021-01045-x
102. Liu CA, Wei HL, Hsin IM, Yua HC, Dueng YH, Wen CT, et al. Interstitial control-released polymer carrying a targeting small-molecule drug reduces PD-L1 and MGMT expression in recurrent high-grade gliomas with TMZ resistance. Cancers (2022) 14(4):1051. doi: 10.3390/cancers14041051
103. Yi M, Niu M, Wu Y, Ge H, Jiao D, Zhu S, et al. Combination of oral STING agonist MSA-2 and anti-TGF-β/PD-L1 bispecific antibody YM101: A novel immune cocktail therapy for non-inflamed tumors. J Hematol Oncol (2022) 15(1):1–21. doi: 10.1186/s13045-022-01363-8
104. Hoang T, Xu R, Schiller JH, Bonomi P, Johnson DH. Based on eastern cooperative oncology group data, a clinical model to predict survival in chemo-naive patients with advanced non-small-cell lung cancer treated with third-generation chemotherapy regimens. J Clin Oncol (2005) 23(1):175–83. doi: 10.1200/JCO.2005.04.177
105. D'Angelo SP, Janjigian YY, Ahye N, Riely GJ, Chaft JE, Sima CS, et al. The different clinical course of EGFR-mutant resected lung cancers: results of testing of 1118 surgical specimens and effects of adjuvant gefitinib and erlotinib. J Thorac Oncol (2012) 7(12):1815–22. doi: 10.1097/JTO.0b013e31826bb7b2
106. Sculier JP, Chansky K, Crowley JJ, Van Meerbeeck J, Goldstraw P. International staging committee and participating institutions. the impact of additional prognostic factors on survival and their relationship with the anatomical extent of disease is expressed by the 6th edition of the TNM classification of malignant tumors and the proposals for the 7th edition. J Thorac Oncol (2008) 3(5):457–66.
107. Paesmans M. Prognostic and predictive factors for lung cancer. Breathe (2012) 9(2):112–12. doi: 10.1183/20734735.006911
108. Lanier LL. Up on the tightrope: Natural killer cell activation and inhibition. Nat Immunol (2008) 9(5):495–502. doi: 10.1038/ni1581
109. Antoun A, Vekaria D, Salama RA, Pratt G, Jobson S, Cook M, et al. The genotype of RAET1L (ULBP6), a ligand for human NKG2D (KLRK1), markedly influences the clinical outcome of allogeneic stem cell transplantation. Br J Haematol (2012) 159(5):589–98. doi: 10.1111/bjh.12072
110. Jelenčić V, Šestan M, Kavazović I, Lenartić M, Marinović S, Holmes TD, et al. NK cell receptor NKG2D sets activation threshold for the NCR1 receptor early in NK cell development. Nat Immunol (2018) 19(10):1083–92. doi: 10.1038/s41590-018-0209-9
111. Ettinger DS, Douglas EW, Charu A, Dara LA, Wallace A, Jessica RB, et al. NCCN guidelines insights: Non–small cell lung cancer, version 1.2020: featured updates to the NCCN guidelines. Journal of the National Comprehensive Cancer Network (2019) 17(12):1464–72. doi: 10.6004/jnccn.2019.0059
112. Wang L, Jiazhong S, Yaqin H, Sha L, Jingqi Z, Hua D, et al. A six-gene prognostic model predicts overall survival in bladder cancer patients. Cancer cell international (2019) 19(1):1–15. doi: 10.1186/s12935-019-0950-7
113. Liu J, Liu S, Wu M, Deng J, Yao X, Liu F, et al. ARF3 inhibits proliferation and promotes apoptosis in gastric cancer by regulating AKT and ERK pathways. Acta biochimica Polonica (2021) 68(2):223–9. doi: 10.18388/abp.2020_5519
114. Baiu I, Titan AL, Martin LW, Wolf A, Backhus L. The role of gender in non-small cell lung cancer: A narrative review. J Thorac Dis (2021) 13(6):3816. doi: 10.21037/jtd-20-3128
115. Lim JH, Ryu JS, Kim JH, Kim HJ, Lee D. Gender as an independent prognostic factor in small-cell lung cancer: Inha lung cancer cohort study using propensity score matching. PloS One (2018) 13(12):e0208492. doi: 10.1371/journal.pone.0208492
116. Hsu CL, Chen KY, Shih JY, Ho CC, Yang CH, Yu CJ, et al. Advanced non-small cell lung cancer in patients aged 45 years or younger: outcomes and prognostic factors. BMC Cancer (2012) 12(1):1–7. doi: 10.1186/1471-2407-12-241
117. Shirasawa M, Fukui T, Kusuhara S, Hiyoshi Y, Ishihara M, Kasajima M, et al. Prognostic significance of the 8th edition of the TNM classification for patients with extensive disease small cell lung cancer. Cancer Manage Res (2018) 10:6039. doi: 10.2147/CMAR.S181789
118. Woodard GA, Jones KD, Jablons DM. Lung cancer staging and prognosis. Lung Cancer (2016), 47–75. doi: 10.1007/978-3-319-40389-2_3
119. Arriola E, Trigo JM, Sánchez-Gastaldo A, Navarro A, Perez C, Crama L, et al. Prognostic value of clinical staging according to TNM in patients with SCLC: A real-world surveillance epidemiology and end-results database analysis. JTO Clin Res Rep (2022) 3(1):100266. doi: 10.1016/j.jtocrr.2021.100266
120. Wong M., Gao J, Thanarajasingam G, Sloan JA, Dueck AC, Novotny PJ, et al. Expanding Beyond Maximum Grade: Chemotherapy Toxicity over Time by Age and Performance Status in Advanced Non‐Small Cell Lung Cancer in CALGB 9730 (Alliance A151729). Oncologist (2021) 26(3):e435–e444.
121. Tas F, Ciftci R, Kilic L, Karabulut S. Age is a prognostic factor affecting survival in lung cancer patients. Oncol Lett (2013) 6(5):1507–13. doi: 10.3892/ol.2013.1566
122. Moumtzi D, Lampaki S, Zarogoulidis P, Porpodis K, Lagoudi K, Hohenforst-Schmidt W, et al. Prognostic factors for long-term survival in patients with advanced non-small cell lung cancer. Ann Trans Med (2016) 4(9). doi: 10.21037/atm.2016.05.13
123. Mytelka DS, Li L, Benoit K. Post-diagnosis weight loss as a prognostic factor in non-small cell lung cancer. J Cachexia Sarcopenia Mscle (2018) 9(1):86–92. doi: 10.1002/jcsm.12253
124. Goksel T, Eser S, Guclu SZ, Karadag M, Cilli A, Ozlu T, et al. Prognostic factors affecting survival in cases with lung cancer [A lung cancer mapping project in Turkey (LCMPT)], Turkey. Eur Respiratory Soc (2013).
125. Sehgal K, Gill RR, Widick P, Bindal P, McDonald DC, Shea M, et al. Association of performance status with survival in patients with advanced non–small cell lung cancer treated with pembrolizumab monotherapy. JAMA network Open (2021) 4(2):e2037120–e2037120. doi: 10.1001/jamanetworkopen.2020.37120
126. Kawaguchi T, Takada M, Kubo A, Matsumura A, Fukai S, Tamura A, et al. Performance and smoking status are independent favorable prognostic factors for survival in non-small cell lung cancer: A comprehensive analysis of 26,957 patients with NSCLC. J Thorac Oncol (2010) 5(5):620–30. doi: 10.1097/JTO.0b013e3181d2dcd9
127. Kang HS, Shin AY, Yeo CD, Park CK, Kim JS, Kim JW, et al. Clinical significance of anemia as a prognostic factor in non-small cell lung cancer carcinoma with activating epidermal growth factor receptor mutations. J Thorac Dis (2020) 12(5):1895. doi: 10.21037/jtd-19-3932
128. Tanaka H, Ono T, Manabe Y, Kajima M, Fujimoto K, Yuasa Y, et al. Anemia is a prognostic factor for overall survival rate in patients with non-small cell lung cancer treated with stereotactic body radiation therapy. Cancer Manage Res (2021) 13:7447. doi: 10.2147/CMAR.S336044
129. Pirker R, Wiesenberger K, Pohl G, Minar W. Anemia in lung cancer: Clinical impact and management. Clin Lung Cancer (2003) 5(2):90–7. doi: 10.3816/CLC.2003.n.022
130. Pathak RS, Pantarotto JR, Cook G, Holmes O, Cross P, MacRae RM. Anemia is a poor prognostic factor for stage I non-small cell lung cancer (NSCLC) patients treated with stereotactic body radiation therapy (SBRT). Clin Trans Radiat Oncol (2019) 16:28–33. doi: 10.1016/j.ctro.2019.01.003
131. Holgersson G, Bergqvist M, Nilsson J, Thureson M, Harmenberg J, Bergstrom S. The predictive value of pre-treatment leukocytosis in patients with previously treated stage IIIB/IV non-small cell lung cancer treated with the IGF-1R pathway modulator AXL1717 or docetaxel; a retrospective analysis of a phase II trial. Asian Pacific J Cancer prevention: APJCP (2017) 18(6):1555. doi: 10.22034/APJCP.2017.18.6.1555
132. Kasuga I, Makino S, Kiyokawa H, Katoh H, Ebihara Y, Ohyashiki K. Tumor-related leukocytosis is linked with poor prognosis in patients with lung carcinoma. Cancer (2001) 92(9):2399–405. doi: 10.1002/1097-0142(20011101)92:9<2399::AID-CNCR1588>3.0.CO;2-W
133. Tavakkoli M, Wilkins CR, Mones JV, Mauro MJ. A novel paradigm between leukocytosis, G-CSF secretion, neutrophil-to-lymphocyte ratio, myeloid-derived suppressor cells, and prognosis in non-small cell lung cancer. Front Oncol (2019) 9:295. doi: 10.3389/fonc.2019.00295
134. Yuan Y, Zhong H, Ye L, Li Q, Fang S, Gu W, et al. Prognostic value of pre-treatment platelet counts in lung cancer: A systematic review and meta-analysis. BMC pulmonary Med (2020) 20(1):1–11. doi: 10.1186/s12890-020-1139-5
135. Maráz A, Furák J, Varga Z, Kahán Z, Tiszlavicz L, Hideghéty K. Thrombocytosis has a negative predictive value in lung cancer. Anticancer Res (2013) 33(4):1725–9.
136. Tomita M, Shimizu T, Hara M, Ayabe T, Onitsuka T. Prognostic impact of thrombocytosis in resectable non-small cell lung cancer. Interactive Cardiovasc Thorac Surg (2008) 7(4):613–5. doi: 10.1510/icvts.2007.174391
137. Davidov D. Thrombocytosis is a prognostic survival factor. J IMAB–Annual Proceeding Sci Papers (2014) 20(6):560–4. doi: 10.5272/jimab.2014206.560
138. Barlow M, Hamilton W, Ukoumunne OC, Bailey SE. The association between thrombocytosis and subtype of lung cancer: A systematic review and meta-analysis. Trans Cancer Res (2021) 10(3):1249. doi: 10.21037/tcr-20-3287
139. Jafri SH, Previgliano C, Khandelwal K, Shi R. Cachexia index in advanced non-small-cell lung cancer patients. Clin Med Insights: Oncol (2015) 9:CMO–S30891. doi: 10.4137/CMO.S30891
140. Kashima Y, Togashi Y, Fukuoka S, Kamada T, Irie T, Suzuki A, et al. Potentiality of multiple modalities for single-cell analyses to evaluate the tumor microenvironment in clinical specimens. Sci Rep (2021) 11(1):1–1. doi: 10.1038/s41598-020-79385-w
141. Sjøblom B, Grønberg BH, Wentzel-Larsen T, Baracos VE, Hjermstad MJ, Aass N, et al. Skeletal muscle radiodensity is prognostic for survival in patients with advanced non-small cell lung cancer. Clin Nutr (2016) 35(6):1386–93. doi: 10.1016/j.clnu.2016.03.010
142. Shoji F, Haratake N, Akamine T, Takamori S, Katsura M, Takada K, et al. The preoperative controlling nutritional status score predicts survival after curative surgery in patients with pathological stage I non-small cell lung cancer. Anticancer Res (2017) 37(2):741–7. doi: 10.21873/anticanres.11372
143. Nattenmüller J, Wochner R, Muley T, Steins M, Hummler S, Teucher B, et al. Prognostic impact of CT-quantified muscle and fat distribution before and after first-line chemotherapy in lung cancer patients. PloS One (2017) 12(1):e0169136. doi: 10.1371/journal.pone.0169136
144. Roch B, Coffy A, Jean-Baptiste S, Palaysi E, Daures JP, Pujol JL, et al. Cachexia-sarcopenia as a determinant of disease control rate and survival in non-small lung cancer patients receiving immune-checkpoint inhibitors. Lung Cancer. (2020) 143:19–26. doi: 10.1016/j.lungcan.2020.03.003
145. Abbas AE. Commentary: The value of close surveillance after lung cancer surgery: How absence of evidence is not evidence of absence. J Thorac Cardiovasc Surgery. (2021) 162(3):686–7. doi: 10.1016/j.jtcvs.2020.11.124
146. Dolan RD, McMillan DC. The prevalence of cancer-associated systemic inflammation: implications of prognostic studies using the Glasgow prognostic score. Crit Rev oncology/hematology. (2020) 150:102962. doi: 10.1016/j.critrevonc.2020.102962
147. Magri V, Gottfried T, Di Segni M, Urban D, Peled M, Daher S, et al. Correlation of body composition by computerized tomography and metabolic parameters with the survival of nivolumab-treated lung cancer patients. Cancer Manage Res (2019) 11:8201. doi: 10.2147/CMAR.S210958
148. Katsui K, Ogata T, Sugiyama S, Yoshio K, Kuroda M, Hiraki T, et al. Sarcopenia is associated with poor prognosis after chemoradiotherapy in patients with stage III non-small-cell lung cancer: A retrospective analysis. Sci Rep (2021) 11(1):1–0. doi: 10.1038/s41598-021-91449-z
Keywords: serum cytokines, targeted immunotherapy, prognosis, biomarkers, lung cancer
Citation: Zhao Y, Jia S, Zhang K and Zhang L (2023) Serum cytokine levels and other associated factors as possible immunotherapeutic targets and prognostic indicators for lung cancer. Front. Oncol. 13:1064616. doi: 10.3389/fonc.2023.1064616
Received: 08 October 2022; Accepted: 24 January 2023;
Published: 16 February 2023.
Edited by:
Federica Recine, Azienda Ospedaliera San Giovanni Addolorata, ItalyCopyright © 2023 Zhao, Jia, Zhang and Zhang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Lian Zhang, emhhbmdsaWFuMTEyNUAxNjMuY29t
 Yinghao Zhao
Yinghao Zhao Shengnan Jia2
Shengnan Jia2