- 1Division of Pediatric Nephrology and Hypertension, Stony Brook Children’s Hospital, School of Medicine, Stony Brook, NY, USA
- 2Stony Brook University, Stony Brook, NY, USA
- 3Division of Pediatric Cardiology, Stony Brook Children’s Hospital, School of Medicine, Stony Brook, NY, USA
Adults with arterial hypertension (HTN) have stroke, myocardial infarction, end-stage renal disease (ESRD), or die at higher rates than those without. In children, HTN leads to target organ damage, which includes kidney, brain, eye, blood vessels, and heart, which precedes “hard outcomes” observed in adults. Left ventricular hypertrophy (LVH) or an anatomic and pathologic increase in left ventricular mass (LVM) in response to the HTN is a pediatric surrogate marker for HTN-induced morbidity and mortality in adults. This mini review discusses current definitions, clinically relevant methods of LVM measurements and normalization methods, its epidemiology, management, and issue of reversibility in children with HTN. Pediatric definition of LVH and abnormal LVM is not uniformed. With multiple definitions, prevalence of pediatric HTN-induced LVH is difficult to ascertain. In addition while in adults cardiac magnetic resonance imaging is considered “the gold standard” for LVM and LVH determination, pediatric data are limited to “special populations”: ESRD, transplant, and obese children. We summarize available data on pediatric LVH treatment and reversibility and offer future directions in addressing LVH in children with HTN.
Introduction
Framingham Heart Study proved that adults with arterial hypertension (HTN) have increased mortality due to myocardial infarction and stroke compared to adults without HTN.1 These “hard outcomes” have not been clearly demonstrated in children with HTN. However, the Bogalusa Heart Study showed that children with HTN become hypertensive adults and that the major etiologies of adult heart disease (atherosclerosis, coronary heart disease, essential HTN) begin by 5–8 years of age.2 Since there are no “hard outcomes” of the deleterious effects of HTN in children, they are assessed by the “end organ” or “target organ” damage (TOD). Pediatric HTN-induced TOD manifests as an injury to several organs: (A) kidney: microalbuminuria/proteinuria, chronic kidney disease (1, 2), (B) eye: retinopathy (3), (C) vessels: increase in intima media thickness, atherosclerosis, reduced arterial compliance (2), (D) brain: cognitive impairment (4), and (E) heart: left ventricular hypertrophy (LVH) (5). TOD associated with uncontrolled HTN leads overtime to “hard outcomes” observed later in life (6). However, if HTN is controlled at least some of the TOD has been shown to be reversible (1, 7). Therefore, it is of paramount importance for pediatricians to be able to recognize presence of TOD as early as possible to act on it and thus prevent one of the “hard outcomes” in adulthood. LVH independently of blood pressure (BP) levels predicted poor prognosis in adults enrolled in the Framingham Heart Study. The relative risk of cardiovascular mortality for every 50 g increment in left ventricular mass (LVM) was 1.73 in men and 2.12 in women (8). The focus of this mini review is on definition, detection, epidemiology, management, and reversibility of LVH.
Definition
Left ventricular hypertrophy is defined as an increase in LVM in response to a disease state, due to either increase in left ventricular (LV) wall thickness or an increase in cavity size or both. These changes of LV diameters represent adaptive responses to a pathologic workload due to HTN or heart valve disease (as oppose to physiologic states of pregnancy or exercise/cardio-workout). However, LVH could also be related to infiltrative diseases of the myocardium or certain genetic disorders. HTN-induced LVH in children usually presents with an increase in wall thickness (concentric hypertrophy), without an increase in cavity size (eccentric hypertophy). Concentric LVH induced by HTN leads with time to LV dilatation, which results in a decline of the LV ejection fraction and eventually in “dilated cardiac failure” (9). Widening of the LV chamber creates circulatory difficulties and presents a vicious cycle to the cardiac muscle, increasing risk for cardiovascular complications (10). Both casual office BP readings and 24-h ambulatory blood pressure monitoring (ABPM) have a direct relationship to LVM/LVH: the higher office BP, the higher average BP recorded over the course of the day and night, or the higher ABPM BP loads (prevalence of abnormal elevated BP readings) the greater possibility for LVH (11). It is worth noting that long-standing white-coat HTN (normal ABMP and abnormal office BP) is associated with increased risks of cardiovascular complications in adults (12). However, this relationship is not consistent in number of pediatric patients with white-coat HTN (13). In summary, the definition of pediatric HTN-induced LVH pivots on an accurate assessment of LVM and determination of what normal versus abnormal is for a given individual. This in turn depends on the accuracy of LVM measurements, and LVH detection and normalization method used.
Detection Methods
Cardiac Magnetic Resonance (CMR)
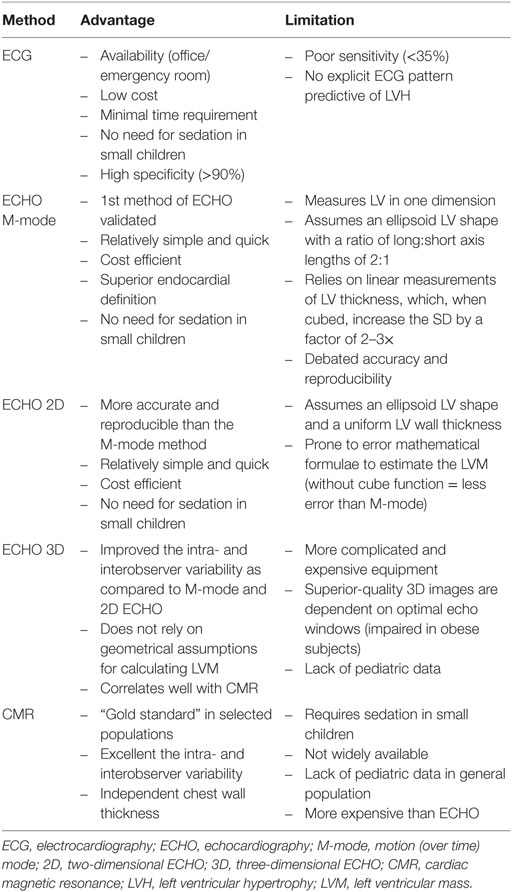
Cardiac magnetic resonance uses strong magnetic fields that stimulate the hydrogen nuclei (present in water or fat) to release radio waves that can be interpreted by computerized scanners and generate images of the heart (14). In adults, CMR imaging is considered as the gold standard for assessment of LVM and LVH (15, 16). In addition, CMR estimates of LVM have been shown to be closely correlated to actual heart weight determined at autopsy in both animal and human models (16). CMR yields high-quality images across the entire left ventricle that is not impeded by thoracic fat deposits and chest wall expansion. Furthermore, CMR is used in the evaluation of congenital heart disease, ant its use has been recently reported in children and young adults on maintenance dialysis (17, 18), and renal transplant recipients (19). Our group has described the utility of CMR in overweight hypertensive children and reported reproducibility (20). However, CMR is not yet widely available, is more expensive than echocardiography (ECHO), has disadvantages such as claustrophobia and the need for sedation in small children, and the data on its utilization in general pediatric population with HTN are lacking. Abnormal LVM and LVH in pediatric CMR is defined as a z score greater than +2.0 utilizing published review that calculated and tabulated pooled weighted mean values that are specific for age and sex (21).
Echocardiography
Although LVM determined by CMR is more accurate and reproducible, ECHO has lower cost and is a more accessible test compared with CMR. ECHO is an imaging technique that creates pictures of the heart utilizing high-frequency ultrasound waves. Whether it be two-dimensional, three-dimensional, or M-mode, ECHO is used to assess TOD and measure LVM. Echocardiographic studies determine the myocardial volume by subtracting the LV cavity volume from the volume of the correspondent epicardium. Upon obtaining the myocardial volume, multiplication by the myocardial density results in the LVM (22). The LVM can then be indexed to body surface area (BSA), or height2.7 to determine LVH (23). One of the challenges when using echocardiographic techniques to determine LVH is precisely finding the boundary between the cardiac blood pool and the endocardium (23). If this step was inaccurate due to, for example, poor acoustic window, or abundant chest fat tissue, there would be improper readings of the LV cavity volume and the epicardial volume. This in turn would result in inaccurate myocardial volumes when performing calculations and thus, inaccurate LVM levels and LVH indicators. For adults, a LVM index ≥51 g/m2.7 is used to define LVH based on a study by de Simone et al., which showed LVMI above this threshold is associated with more than four times increased risk of morbidity and mortality (24). The Fourth Report selected 51 g/m2.7 as their LVMI limit value to define LVH in children (25). However, this value does not adjust for growth and other potentially confounding factors. The Bogalusa Heart Study demonstrated that somatic growth is the strongest predictor of LVM (26). Therefore, LVM must be indexed to normalize the relationship without disregarding obesity. Foster et al. showed that normalizing LVM to BSA or height results in either underestimation or overestimation of LVM, respectively (27). They proposed lean body mass (LBM) as the ideal scaling variable for normalization. Although LBM can be measured by dual-energy X-ray absorptiometry, it is clinically difficult to ascertain (27). Foster et al. used LBM predictive equations and generated sex-specific LVM-for-LBM centile curves for children 5–18 years of age and defined LVH as LVMI-for-age >95th percentile (27). Despite this, most pediatric nephrologists index LVM to height2.7. Khoury et al. developed age- and sex-based LVMI (height2.7) centiles in 2009 (28). They observed little variation beyond age 9, suggesting their reference tables would only be needed for younger children. They defined LVH as LVM/height2.7 greater than 95th percentile for sex and age (28). According to their calculations after age 9 years, a constant 95th percentile value of 40 g/m2.7 (female), and 45 g/m2.7 (male) defines LVH (28). At present, it is challenging to say which indexing method is better because there is no one method without substantial limitations. Furthermore, ECHO cannot distinguish small but clinically significant changes in diastolic wall thickness from measurement error in individual children, even when measured by the same observer (29). Three-dimensional ECHO has also been utilized to quantify LVM and allows for LVM quantification using principles similar to CMR. LVM is determined by taking the difference between epicardial and endocardial volumes and may better account for ventricular morphology. Quantification of LVM by three-dimensional ECHO has been shown to be of use in the adult population; however, its use remains limited in pediatrics at this time (30).
Electrocardiography (ECG)
There is no explicit ECG pattern predictive of abnormal LVM. Instead, there are host of electrical abnormalities (voltage and non-voltage criteria) that are associated with LVH. The most commonly used are the Sokolow–Lyon criteria (31), and voltage criteria must be accompanied by non-voltage criteria to be considered diagnostic of LVH (32). However, HTN-induced LVH could be easily misclassified by using ECG; therefore, ECG should not be used alone in determining presence or absence of LVH. However, ECG still has a place in TOD assessment as it gives independent information on the cardiovascular risk even after adjusting for LVM (32). In children with HTN, although ECG has high specificity (>90%), it is a poor screening test with low sensitivity (<35%) in evaluation of abnormal LVM (33).
Left ventricular hypertrophy detection methods summary is presented in Table 1.
Epidemiology
The prevalence of LVH in children and adolescents is not precisely known partly because multiple definitions in different population exist. It varies from 4.8 to 50% in children with primary HTN (27, 28, 34–36) and has been reported as high as 55% in children after renal transplant (37) and 85% in children on dialysis (38). In children with chronic kidney disease, the following factors contribute to abnormalities in calculating LVM: male sex, higher body mass index, degree of anemia, fluid overload, and low-grade inflammation and can be more relevant than the effect of HTN (39).
In pediatric essential HTN, severe LVH defined as LVMI above 51 g/height2.7 has been found in 10–15% (34, 40, 41). The prevalence of LVH in children without HTN is unknown. In adults, it has been reported that obesity had significant independent associations with LVM and wall thickness (42), and this relationship has been confirmed in obese children and adolescents with normal office BPs (43, 44). In addition to body mass index, ethnicity contributes to differences in prevalence of LVH. A collaborative study of the International Pediatric Hypertension Association found that LVH and concentric hypertrophy occurred most frequently in hypertensive Hispanic children (35). In adults, LVH is more common in hypertensive African-Americans (AA) (50%) than in whites (33%) (45), and the adjusted risk of having LVH, whether indexed by height2.7 or by BSA, is greater for AA than for whites (46). In AA children with HTN, LVH prevalence was 49 versus 30% among non-AA (p < 0.05) (36). Children of AA descent with chronic kidney disease and high-risk APOL1 genotypes have both higher prevalence of LVH (53 versus 12%, p < 0.01) and obesity (48 versus 19%, p = 0.01) than their non-AA counterparts (47). Women were thought to have a lower prevalence of LVH than men for any given level of BP (8). However, LVH prevalence differences between sexes are dependent on LVM normalized to either height, height2.7, or BSA. LVM was identical in men and women when using LV mass/fat-free mass as a partition value of 4.1 g/kg (48). Since high dietary intake of sodium is associated with increase in BP and elevated BP is associated with LVH (49), salt restriction is associated with decrease in the incidence of LVH (50). In children with chronic kidney disease, differences in LVH prevalence between girls and boys were also dependent on LVH definition. When LVH was defined by LVM indexed to height, girls had higher prevalence of LVH (16 versus 9%, p = 0.01); when LVH was defined by LVM relative to estimated LBM, prevalence of LVH was similar between girls and boys (18 versus 17%, p = 0.92) (51). It has been known that adults with diabetes mellitus have higher LV mass, independent of HTN (52, 53). Adult type II diabetics have significantly higher prevalence of LVH in hypertensive women (54). Similarly, diabetic children have significant changes in LV dimensions, and girls are more affected than boys (55). Finally, there is strong evidence for the genetic influence on LVH prevalence across several study designs, datasets and ethnicities (56–60), and several genetic conditions, i.e., hypertrophic cardiomyopathy, caused by mutations in sarcomere genes (61), and RASopathies are causally associated with LVH (62). However, specific genetic influences on prevalence of HTN-associated LVH are currently unknown.
Management and Reversibility
Hypertension is a rare cause of heart failure in children and adolescents. In children, the rationale to treat LVH is based on adult data. Special attention should be given to children and adolescents with HTN and the following conditions: end-stage renal failure, diabetes, coarctation of the aorta, or Kawasaki disease. These patients are at increased risk of early cardiovascular events and heart failure (63). Non-pharmacologic hypertensive treatments such as dietary salt restriction, weight reduction, avoidance of smoking, and aerobic exercise have been effective in lowering cardiovascular risk factors in adults (64). In children with HTN, if BP does not improve by lifestyle modification or there is evidence of TOD, then pharmacologic treatment is indicated (65). In children with HTN, presence of LVH prompts treatment and LVH reversal becomes a major treatment goal. The baseline severity of LVH, and the degree and duration of HTN control determines slope of LVH reversal (40, 66, 67). In adults, any BP lowering agent lowers the risk of LVH (68). All anti-HTN classes are effective in lowering of BP in children; however, their differential effects on LVH have not been demonstrated in pediatric population (25). Angiotensin-converting enzyme (ACE) inhibitors and angiotensin receptor blockers (ARB) are especially effective in the regression of LVH due to host of additional BP-independent mechanisms: reduction of growth factors (TGF-beta), free oxygen radicals, inhibitory effects on myocyte growth, and collagen formation. In adults, treatment with ACE inhibitors or ARB increases insulin sensitivity (69, 70), whereas treatment with thiazides decreases insulin sensitivity (71, 72). In fact, ACE/ARB has been the drug of choice for over a decade for pediatric nephrologists who treat HTN (73). This preference comes not from pediatric data but from adult studies showing their beneficial, cardio and reno-protective effects in diabetes, HTN, and microalbuminuria (71, 72). In adults with HTN or with early-stage CKD, the addition of spironolactone to ACE/ARB reduced LVM and improved arterial stiffness (74). Spironolactone is usually used in children with congestive heart failure or as potassium sparring diuretic and not as first or second agent in HTN-induced LVH treatment. In adults, calcium channel antagonists are as effective as ACE inhibitors (such as Lisinopril) in their ability to reduce LVH and cardiac wall thickness (75). This is likely due to dominant effect of BP lowering over BP-independent effects. Another class of pharmacological agents that assist in the regression of LVH are beta receptor-blocking agents: both B1 receptor specific or non-selective that work through decreasing cardiac output and easing afterload facilitating LV remodeling. However, abnormalities in diastolic function associated with HTN-induced LVH are not reduced by reversal of LVH induced by antihypertensive treatment with beta-blockers (76). It is very interesting that in some children with primary HTN the main determinant of LVH regression is decrease in abdominal obesity with an increase in LBM rather than BP lowering (77). This conforms to recent adult study finding that higher BMI is associated with less reduction of hypertensive LVH: independent of BP control and of types of antihypertensive treatment (78). Although different classes of antihypertensive have different effects on LV mass in adults, the data regarding the specific effects of antihypertensive therapy on LVH in children are not adequate.
Future Directions
Left ventricular mass and LVH are associated with HTN and with increased BMI. Efforts to diminish pediatric cardiovascular risk factors and prevalence of HTN-induced LVH should thus be connected to the public campaign to reduce obesity and increase childhood physical activity. In addition, emergence of effective approaches to issue of medication adherence may result in better long-term BP control. Recognition of food addictions, full understanding of sugar and food additives effects on anxiety, depression, BP, and hyperactivity may lead to effective interventions of addressing the root cause of obesity/HTN, which in turn may result with time in a decrease in the prevalence of LVH. Increased use of advanced technologies such as CMR or multidimensional ECHO should lead to improved detection of LVH and better understanding of its pathophysiology and epidemiology. Our improved understanding of cardiac electrogenesis and electrical remodeling in TOD may allow for more targeted treatment of electrical abnormalities associated with HTN. Furthermore, randomized controlled studies in pediatric HTN populations are needed to answer the question if available adult data can really be translated to children.
Author Contributions
RW wrote the mini review and contributed to review of literature and design of the review. AK, LP, and KS-M contributed to review of literature and wrote the part of the review.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Footnotes
References
1. Assadi F. Effect of microalbuminuria lowering on regression of left ventricular hypertrophy in children and adolescents with essential hypertension. Pediatr Cardiol (2007) 28(1):27–33. doi: 10.1007/s00246-006-1390-4
2. Conkar S, Yılmaz E, Hacıkara Ş, Bozabalı S, Mir S. Is daytime systolic load an important risk factor for target organ damage in pediatric hypertension? J Clin Hypertens (Greenwich) (2015) 17(10):760–6. doi:10.1111/jch.12608
3. Skalina ME, Annable WL, Kliegman RM, Fanaroff AA. Hypertensive retinopathy in the newborn infant. J Pediatr (1983) 103:781–6. doi:10.1016/S0022-3476(83)80485-5
4. Lande MB, Batisky DL, Kupferman JC, Samuels J, Hooper SR, Falkner B, et al. Neurocognitive function in children with primary hypertension. J Pediatr (2017) 180:148–55.e1. doi:10.1016/j.jpeds.2016.08.076
5. Lai CC, Sun D, Cen R, Wang J, Li S, Fernandez-Alonso C, et al. Impact of long-term burden of excessive adiposity and elevated blood pressure from childhood on adult left ventricular remodeling patterns: the Bogalusa Heart Study. J Am Coll Cardiol (2014) 64(15):1580–7. doi:10.1016/j.jacc.2014.05.072
6. Ezzati M, Lopez AD, Rodgers A, Vander Hoorn S, Murray CJ. Comparative risk assessment collaborating group. Selected major risk factors and global and regional burden of disease. Lancet (2002) 360(9343):1347–60. doi:10.1016/S0140-6736(02)11403-6
7. Kupferman JC, Aronson Friedman L, Cox C, Flynn J, Furth S, Warady B, et al. BP control and left ventricular hypertrophy regression in children with CKD. J Am Soc Nephrol (2014) 25(1):167–74. doi:10.1681/ASN.2012121197
8. Levy D, Garrison RJ, Savage DD, Kannel WB, Castelli WP. Prognostic implications of echocardiographically determined left ventricular mass in the Framingham Heart Study. N Engl J Med (1990) 322:1561–6. doi:10.1056/NEJM199005313222203
9. Frohlich ED, Apstein C, Chobanian AV, Devereux RB, Dustan HP, Dzau V, et al. The heart in hypertension. N Engl J Med (1992) 327:998–1008. doi:10.1056/NEJM199210013271406
10. Maron BJ, McKenna WJ, Danielson GK, Kappenberger LJ, Kuhn HJ, Seidman CE, et al. American College of Cardiology/European Society of Cardiology clinical expert consensus document on hypertrophic cardiomyopathy. A report of the American College of Cardiology Foundation Task Force on Clinical Expert Consensus Documents and the European Society of Cardiology Committee for Practice Guidelines. J Am Coll Cardiol (2003) 42(9):1687–713.
11. Flynn JT, Daniels SR, Hayman LL, Maahs DM, McCrindle BW, Mitsnefes M, et al. Update: ambulatory blood pressure monitoring in children and adolescents a scientific statement from the American Heart Association. Hypertension (2014) 63:1116–35. doi:10.1161/HYP.0000000000000007
12. Tientcheu D, Ayers C, Das SR, McGuire DK, de Lemos JA, Khera A, et al. Target organ complications and cardiovascular events associated with masked hypertension and white-coat hypertension: analysis from the Dallas heart study. J Am Coll Cardiol (2015) 66(20):2159–69. doi:10.1016/j.jacc.2015.09.007
13. Lande MB, Meagher CC, Fisher SG, Belani P, Wang H, Rashid M. Left ventricular mass index in children with white coat hypertension. J Pediatr (2008) 153(1):50–4. doi:10.1016/j.jpeds.2008.01.025
14. Budoff MJ, Cohen MC, Garcia MJ, Hodgson JM, Hundley WG, Lima JA, et al. ACCF/AHA clinical competence statement on cardiac imaging with computed tomography and magnetic resonance: a report of the American College of Cardiology Foundation/American Heart Association/American College of Physicians Task Force on Clinical Competence and Training. J Am Coll Cardiol (2005) 46(2):383–402. doi:10.1016/j.jacc.2005.04.033
15. Bottini PB, Carr AA, Prisant LM, Flickinger FW, Allison JD, Gottdiener JS. Magnetic resonance imaging compared to echocardiography to assess left ventricular mass in the hypertensive patient. Am J Hypertens (1995) 8:221–8. doi:10.1016/0895-7061(94)00178-E
16. Sechtem U, Pflugfelder PW, Gould RG, Cassidy MM, Higgins CB. Measurement of right and left ventricular volumes in healthy individuals with cine MR imaging. Radiology (1987) 163:697–702. doi:10.1148/radiology.163.3.3575717
17. Lu JC, Nielsen JC, Morowitz L, Musani M, Mahani MG, Agarwal PP, et al. Use of a 1.0 Tesla open scanner for evaluation of pediatric and congenital heart disease: a retrospective cohort study. J Cardiovasc Magn Reson (2015) 25:39. doi:10.1186/s12968-015-0144-y
18. Schaefer B, Rusai K, Toth A, Pasti K, Ujszaszi A, Kreko M, et al. Cardiac magnetic resonance imagining in children with chronic kidney disease and renal transplantation. Pediatr Transplant (2012) 16:350–6. doi:10.1111/j.1399-3046.2012.01672.x
19. Malatesta-Muncher R, Wansapura J, Taylor M, Lindquist D, Hor K, Mitsnefes M. Early cardiac dysfunction in pediatric patients on maintenance dialysis and post kidney transplant. Pediatr Nephrol (2012) 27:1157–64. doi:10.1007/s00467-012-2124-x
20. Supe-Markovina K, Nielsen JC, Musani M, Panesar LE, Woroniecki RP. Assessment of left ventricular mass and hypertrophy by cardiovascular magnetic resonance imaging in pediatric hypertension. J Clin Hypertens (Greenwich) (2016) 18(10):976–81. doi:10.1111/jch.12808
21. Kawel-Boehm N, Maceira A, Valsangiacomo-Buechel ER, Vogel-Claussen J, Turkbey EB, Williams R, et al. Normal values for cardiovascular magnetic resonance in adults and children. J Cardiovasc Magn Reson (2015) 17:1–33. doi:10.1186/s12968-015-0111-7
22. Armstrong A, Gidding S, Gjesdal J, Wu C, Bluemke D, Lima J. LVM assessed by echocardiography and cardiac magnetic resonance, cardiovascular outcomes, and medical practice. JACC Cardiovasc Imaging (2012) 5(8):837–48. doi:10.1016/j.jcmg.2012.06.003
23. Stabouli S, Kotsis V, Rizos Z, Toumanidis S, Karagianni C, Constantopoulos A, et al. Left ventricular mass in normotensive, prehypertensive and hypertensive children and adolescents. Pediatr Nephrol (2009) 24:1545–51. doi:10.1007/s00467-009-1165-2
24. de Simone G, Devereux RB, Daniels SR, Koren MJ, Meyer RA, Laragh JH. Effect of growth on variability of left ventricular mass: assessment of allometric signals in adults and children and their capacity to predict cardiovascular risk. J Am Coll Cardiol (1995) 25(5):1056–62. doi:10.1016/0735-1097(94)00540-7
25. National High Blood Pressure Education Program Working Group on High Blood Pressure in Children and Adolescents. The fourth report on the diagnosis, evaluation, and treatment of high blood pressure in children and adolescents. Pediatrics (2004) 114(2 Suppl 4th Report):555–76. doi:10.1542/peds.114.2.S2.555
26. Urbina EM, Gidding SS, Bao W, Pickoff AS, Berdusis K, Berenson GS. Effect of body size, ponderosity, and blood pressure on left ventricular growth in children and young adults in the Bogalusa Heart Study. Circulation (1995) 91(9):2400–6. doi:10.1161/01.CIR.91.9.2400
27. Foster BJ, Khoury PR, Kimball TR, Mackie AS, Mitsnefes M. New reference centiles for left ventricular mass relative to lean body mass in children. J Am Soc Echocardiogr (2016) 29(5):441–7. doi:10.1016/j.echo.2015.12.011
28. Khoury PR, Mitsnefes M, Daniels SR, Kimball TR. Age-specific reference intervals for indexed left ventricular mass in children. J Am Soc Echocardiogr (2009) 22(6):709–14. doi:10.1016/j.echo.2009.03.003
29. Schoenmaker NJ, van der Lee JH, Groothoff JW, van Iperen GG, Frohn-Mulder IM, Tanke RB, et al. Low agreement between cardiologists diagnosing left ventricular hypertrophy in children with end-stage renal disease. BMC Nephrol (2013) 14:170. doi:10.1186/1471-2369-14-170
30. Chuang ML, Beaudin RA, Riley MF, Mooney MG, Mannin WJ, Douglas PS, et al. Three-dimensional echocardiographic measurement of left ventricular mass: comparison with magnetic resonance imaging and two-dimensional echocardiographic determinations in man. Int J Card Imaging (2000) 16:347–57. doi:10.1023/A:1026540809758
31. Sokolow M, Lyon TP. The ventricular complex in left ventricular hypertrophy as obtained by unipolar precordial and limb leads. Am Heart J (1949) 37(2):161–86. doi:10.1016/0002-8703(49)90562-1
32. Bacharova L, Schocken D, Estes EH, Strauss D. The role of ECG in the diagnosis of left ventricular hypertrophy. Curr Cardiol Rev (2014) 10(3):257–61. doi:10.2174/1573403X10666140514103220
33. Ramaswamy P, Patel E, Fahey M, Mahgerefteh J, Lytrivi ID, Kupferman JC. Electrocardiographic predictors of left ventricular hypertrophy in pediatric hypertension. J Pediatr (2009) 154(1):106–10. doi:10.1016/j.jpeds.2008.07.005
34. Daniels SR, Loggie JM, Khoury P, Kimball TR. Left ventricular geometry and severe left ventricular hypertrophy in children and adolescents with essential hypertension. Circulation (1998) 97:1907–11. doi:10.1161/01.CIR.97.19.1907
35. Hanevold C, Waller J, Daniels S, Portman R, Sorof J; International Pediatric Hypertension Association. The effects of obesity, gender, and ethnic group on left ventricular hypertrophy and geometry in hypertensive children: a collaborative study of the International Pediatric Hypertension Association. Pediatrics (2004) 113(2):328–33. doi:10.1542/peds.113.2.328
36. Pruette CS, Fivush BA, Flynn JT, Brady TM. Effects of obesity and race on left ventricular geometry in hypertensive children. Pediatr Nephrol (2013) 28(10):2015–22. doi:10.1007/s00467-013-2507-7
37. Mitsnefes MM, Kimball TR, Border WL, Witt SA, Glascock BJ, Khoury PR, et al. Abnormal cardiac function in children after renal transplantation. Am J Kidney Dis (2004) 43(4):721–6. doi:10.1053/j.ajkd.2003.12.033
38. Mitsnefes MM, Daniels SR, Schwartz SM, Meyer RA, Khoury P, Strife CF. Severe left ventricular hypertrophy in pediatric dialysis: prevalence and predictors. Pediatr Nephrol (2000) 14(10–11):898–902. doi:10.1007/s004670000303
39. Matteucci MC, Wühl E, Picca S, Mastrostefano A, Rinelli G, Romano C, et al. Left ventricular geometry in children with mild to moderate chronic renal insufficiency. J Am Soc Nephrol (2006) 17(1):218–26. doi:10.1681/ASN.2005030276
40. Sorof JM, Cardwell G, Franco K, Portman RJ. Ambulatory blood pressure and left ventricular mass in hypertensive children. Hypertension (2002) 39:903–8. doi:10.1161/01.HYP.0000013266.40320.3B
41. Litwin M, Niemirska A, Sladowska J, Antoniewicz J, Daszkowska J, Wierzbicka A, et al. Left ventricular hypertrophy and arterial wall thickening in children with essential hypertension. Pediatr Nephrol (2006) 21:811–9. doi:10.1007/s00467-006-0068-8
42. Lauer MS, Anderson KM, Levy D. Separate and joint influences of obesity and mild hypertension on left ventricular mass and geometry: the Framingham Heart Study. J Am Coll Cardiol (1992) 19(1):130–4. doi:10.1016/0735-1097(92)90063-S
43. Steinberger J, Jacobs DR, Moran A, Hong CP, Rocchini AP, Prineas RJ, et al. Relation of insulin resistance and body composition to left ventricular mass in children. Am J Cardiol (2002) 90:1177–80. doi:10.1016/S0002-9149(02)02795-9
44. Kharod AM, Ramlogan SR, Kumar S, Raghuveer T, Drake W, Dai H, et al. Childhood obesity increases left-ventricular mass irrespective of blood pressure status. Pediatr Cardiol (2014) 35:353–60. doi:10.1007/s00246-013-0782-5
45. Chapman JN, Mayet J, Chang CL, Foale RA, Thom SA, Poulter NR. Ethnic differences in the identification of left ventricular hypertrophy in the hypertensive patient. Am J Hypertens (1999) 12:437–42. doi:10.1016/S0895-7061(99)00027-8
46. Kizer JR, Arnett DK, Bella JN, Paranicas M, Rao DC, Province MA, et al. Differences in left ventricular structure between black and white hypertensive adults: the Hypertension Genetic Epidemiology Network study. Hypertension (2004) 43(6):1182–8. doi:10.1161/01.HYP.0000128738.94190.9f
47. Woroniecki RP, Ng DK, Limou S, Winkler CA, Reidy KJ, Mitsnefes M, et al. Renal and cardiovascular morbidities associated with APOL1 status among African-American and non-African-American children with focal segmental glomerulosclerosis. Front Pediatr (2016) 4:122. doi:10.3389/fped.2016.00122
48. Kuch B, Hense HW, Gneiting B, Döring A, Muscholl M, Bröckel U, et al. Body composition and prevalence of left ventricular hypertrophy. Circulation (2000) 102(4):405–10. doi:10.1161/01.CIR.102.4.405
49. Farquhar WB, Edwards DG, Jurkovitz CT, Weintraub WS. Dietary sodium and health: more than just blood pressure. J Am Coll Cardiol (2015) 65(10):1042–50. doi:10.1016/j.jacc.2014.12.039
50. Vaidya A, Bentley-Lewis R, Jeunemaitre X, Adler GK, Williams JS. Dietary sodium alters the prevalence of electrocardiogram determined left ventricular hypertrophy in hypertension. Am J Hypertens (2009) 22(6):669–73. doi:10.1038/ajh.2009.45
51. Ruebner RL, Ng D, Mitsnefes M, Foster BJ, Meyers K, Warady B, et al. Cardiovascular disease risk factors and left ventricular hypertrophy in girls and boys with CKD. Clin J Am Soc Nephrol (2016) 11(11):1962–8. doi:10.2215/CJN.01270216
52. Devereux RB, Roman MJ, Paranicas M, O’Grady MJ, Lee ET, Welty TK, et al. Impact of diabetes on cardiac structure and function: the strong heart study. Circulation (2000) 101(19):2271–6. doi:10.1161/01.CIR.101.19.2271
53. Bella JN, Devereux RB, Roman MJ, Palmieri V, Liu JE, Paranicas M, et al. Separate and joint effects of systemic hypertension and diabetes mellitus on left ventricular structure and function in American Indians (the strong heart study). Am J Cardiol (2001) 87(11):1260–5. doi:10.1016/S0002-9149(01)01516-8
54. Tenenbaum A, Fisman EZ, Schwammenthal E, Adler Y, Benderly M, Motro M, et al. Increased prevalence of left ventricular hypertrophy in hypertensive women with type 2 diabetes mellitus. Cardiovasc Diabetol (2003) 2:14. doi:10.1186/1475-2840-2-14
55. Suys BE, Katier N, Rooman RP, Matthys D, Op De Beeck L, Du Caju MV, et al. Female children and adolescents with type 1 diabetes have more pronounced early echocardiographic signs of diabetic cardiomyopathy. Diabetes Care (2004) 27(8):1947–53. doi:10.2337/diacare.27.8.1947
56. Arnett DK, Hong Y, Bella JN, Oberman A, Kitzman DW, Hopkins PN, et al. Sibling correlation of left ventricular mass and geometry in hypertensive African Americans and whites: the HyperGEN study. Hypertension Genetic Epidemiology Network. Am J Hypertens (2001) 14(12):1226–30. doi:10.1016/S0895-7061(01)02200-2
57. Bella JN, MacCluer JW, Roman MJ, Almasy L, North KE, Best LG, et al. Heritability of left ventricular dimensions and mass in American Indians: the strong heart study. J Hypertens (2004) 22(2):281–6. doi:10.1097/00004872-200402000-00011
58. Juo SH, Di Tullio MR, Lin HF, Rundek T, Boden-Albala B, Homma S, et al. Heritability of left ventricular mass and other morphologic variables in Caribbean Hispanic subjects: the Northern Manhattan Family Study. J Am Coll Cardiol (2005) 46(4):735–7. doi:10.1016/j.jacc.2005.05.025
59. Chien KL, Hsu HC, Su TC, Chen MF, Lee YT. Heritability and major gene effects on left ventricular mass in the Chinese population: a family study. BMC Cardiovasc Disord (2006) 6:37. doi:10.1186/1471-2261-6-37
60. Arnett DK, Meyers KJ, Devereux RB, Tiwari HK, Gu CC, Vaughan LK, et al. Genetic variation in NCAM1 contributes to left ventricular wall thickness in hypertensive families. Circ Res (2011) 108(3):279–83. doi:10.1161/CIRCRESAHA.110.239210
61. Maron BJ, Maron MS, Semsarian C. Genetics of hypertrophic cardiomyopathy after 20 years: clinical perspectives. J Am Coll Cardiol (2012) 60:705–15. doi:10.1016/j.jacc.2012.02.068
62. Sana ME, Quilliam LA, Spitaleri A, Pezzoli L, Marchetti D, Lodrini C, et al. A novel HRAS mutation independently contributes to left ventricular hypertrophy in a family with a known MYH7 mutation. PLoS One (2016) 11(12):e0168501. doi:10.1371/journal.pone.0168501
63. Rad EM, Assadi F. Management of hypertension in children with cardiovascular disease and heart failure. Int J Prev Med (2014) 5:10–6.
64. MacMahon SW, Wilcken DE, MacDonald GJ. The effect of weight reduction on left ventricular mass: a randomized controlled trial in young, overweight hypertensive patients. N Engl J Med (1986) 314:334–9. doi:10.1056/NEJM198602063140602
65. Lurbe E, Agabiti-Rosei E, Cruickshank JK, Dominiczak A, Erdine S, Hirth A, et al. 2016 European Society of Hypertension guidelines for the management of high blood pressure in children and adolescents. J Hypertens (2016) 34(10):1887–920. doi:10.1097/HJH.0000000000001039
66. McNiece KL, Gupta-Malhorta M, Samuels J, Bell C, Garcia K, Pottenberger T, et al. Left ventricular hypertrophy in hypertensive adolescents. Analysis of risk by 2004 National High Blood Pressure Education Program Working Group criteria. Hypertension (2007) 50:392–5. doi:10.1161/HYPERTENSIONAHA.107.092197
67. Richey PA, Disessa TG, Hastings MC, Somes GW, Akpert BS, Jones DP. Ambulatory blood pressure and increased left ventricular mass in children at risk for hypertension. J Pediatr (2008) 152:343–8. doi:10.1016/j.jpeds.2007.07.014
68. Miller AB, Reichek N, St John Sutton M, Iyengar M, Henderson LS, Tarka EA, et al. Importance of blood pressure control in left ventricular mass regression. J Am Soc Hypertens (2010) 4(6):302–10. doi:10.1016/j.jash.2010.09.003
69. Black HR, Davis B, Barzilay J, Nwachuku C, Baimbridge C, Marginean H. Metabolic and clinical outcomes in nondiabetic individuals with the metabolic syndrome assigned to chlorthalidone, amlodipine, or lisinopril as initial treatment for hypertension: a report from the antihypertensive and lipid-lowering treatment to prevent heart attack trial (ALLHAT). Diabetes Care (2008) 31:353–60. doi:10.2337/dc07-1452
70. Benson SC, Pershadsingh HA, Ho CI. Identification of telmisartan as a unique angiotensin II receptor antagonist with selective PPARγ-modulating activity. Hypertension (2004) 43:993–1002. doi:10.1161/01.HYP.0000123072.34629.57
71. Mathew J, Sleight P, Lonn E, Johnstone D, Pogue J, Yi Q, et al. Reduction of cardiovascular risk by regression of electrocardiographic markers of left ventricular hypertrophy by the angiotensin-converting enzyme inhibitor ramipril. Circulation (2001) 104(14):1615–21. doi:10.1161/hc3901.096700
72. Julius S, Nesbitt SD, Egan BM, Weber BM, Michelson EL, Kacirotti N, et al. Feasibility of treating prehypertension with an angiotensin receptor blocker. N Eng J Med (2006) 354:1685–97. doi:10.1056/NEJMoa060838
73. Woroniecki RP, Flynn JT. How are hypertensive children evaluated and managed? A survey of North American pediatric nephrologists. Pediatr Nephrol (2005) 20(6):791–7. doi:10.1007/s00467-004-1804-6
74. Edwards NC, Steeds RP, Stewart PM, Ferro CJ, Townend JN. Effect of spironolactone on left ventricular mass and aortic stiffness in early-stage chronic kidney disease: a randomized controlled trial. J Am Coll Cardiol (2009) 54(6):505–12. doi:10.1016/j.jacc.2009.03.066
75. Terpstra W, May J, Smit A, Graefft P, Havinga T, Veur E, et al. Long-term effects of amlodipine and lisinopril on left ventricular mass and diastolic function in elderly, previously hypertensive patients: the ELVERA trial. J Hypertens (2001) 19(2):303–9. doi:10.1097/00004872-200102000-00018
76. Trimarco B, De Luca N, Cuocolo A, Ricciardelli B, Rosiello G, Lembo G, et al. Beta blockers and left ventricular hypertrophy in hypertension. Am Heart J (1987) 114(4 Pt 2):975–83. doi:10.1016/0002-8703(87)90596-5
77. Litwin M, Niemirska A, Sladowska-Kozlowska J, Wierzbicka A, Janas R, Wawer ZT, et al. Regression of target organ damage in children and adolescents with primary hypertension. Pediatr Nephrol (2010) 25:2489–99. doi:10.1007/s00467-010-1626-7
Keywords: left ventricular hypertrophy, LVMI, target organ damage, childhood hypertension, cardiac magnetic resonance, echocardiography
Citation: Woroniecki RP, Kahnauth A, Panesar LE and Supe-Markovina K (2017) Left Ventricular Hypertrophy in Pediatric Hypertension: A Mini Review. Front. Pediatr. 5:101. doi: 10.3389/fped.2017.00101
Received: 28 February 2017; Accepted: 20 April 2017;
Published: 11 May 2017
Edited by:
Ibrahim F. Shatat, Medical University of South Carolina, USA; Sidra Medical and Research Center, QatarReviewed by:
Aftab S. Chishti, University of Kentucky, USAJuan C. Kupferman, Maimonides Medical Center, USA
Copyright: © 2017 Woroniecki, Kahnauth, Panesar and Supe-Markovina. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Robert P. Woroniecki, cm9iZXJ0Lndvcm9uaWVja2lAc3Rvbnlicm9va21lZGljaW5lLmVkdQ==
 Robert P. Woroniecki
Robert P. Woroniecki Andrew Kahnauth
Andrew Kahnauth Laurie E. Panesar
Laurie E. Panesar Katarina Supe-Markovina1
Katarina Supe-Markovina1