- 1East Midlands Congenital Heart Centre, University Hospitals of Leicester, Leicester, United Kingdom
- 2Cardiovascular Research Center, University of Leicester, Leicester, United Kingdom
Introduction: We reviewed our experience with decellularized porcine small intestine sub-mucosa (DPSIS) patch, recently introduced for congenital heart defects.
Materials and Methods: Between 10/2011 and 04/2016 a DPSIS patch was used in 51 patients, median age 1.1 months (5 days to 14.5 years), for aortic arch reconstruction (45/51 = 88.2%) or aortic coarctation repair (6/51 = 11.8%). All medical records were retrospectively reviewed, with primary endpoints interventional procedure (balloon dilatation) or surgery (DPSIS patch replacement) due to patch-related complications.
Results: In a median follow-up time of 1.5 ± 1.1 years (0.6–2.3years) in 13/51 patients (25.5%) a re-intervention, percutaneous interventional procedure (5/51 = 9.8%) or re-operation (8/51 = 15.7%) was required because of obstruction in the correspondence of the DPSIS patch used to enlarge the aortic arch/isthmus, with median max velocity flow at Doppler interrogation of 4.0 ± 0.51 m/s. Two patients required surgery after failed interventional cardiology. The mean interval between DPSIS patch implantation and re-intervention (percutaneous procedure or re-operation) was 6 months (1–17 months). While there were 3 hospital deaths (3/51 = 5.9%) not related to the patch implantation, no early or late mortality occurred for the subsequent procedure required for DPSIS patch interventional cardiology or surgery. The median max velocity flow at Doppler interrogation through the aortic arch/isthmus for the patients who did not require interventional procedure or surgery was 1.7 ± 0.57 m/s.
Conclusions: High incidence of re-interventions with DPSIS patch for aortic arch and/or coarctation forced us to use alternative materials (homografts and decellularized gluteraldehyde preserved bovine pericardial matrix).
Introduction
Aortic coarctation is quite frequently associated with aortic arch hypoplasia, requiring attention at the time of surgery. The criteria generally agreed to define the presence of aortic arch hypoplasia are: (a) aortic arch size mm < body weight kg + 1; (b) aortic arch diameter z-score < −2.0; (c) ratio of transverse arch diameter to descending aorta <50%.
Surgery for aortic coarctation with aortic arch reconstruction can be performed with various surgical approaches. Despite good outcomes reported with the technique of resection of the aortic coarctation and end-to-end anastomosis extended to the aortic arch through a postero-lateral thoracotomy (1–5), the currently preferred option is repair through median sternotomy with cardiopulmonary bypass. The approach with cardiopulmonary bypass through median sternotomy, necessary when associated heart malformations require surgical treatment in the same session, can be accomplished with various techniques of aortic arch reconstruction with or without the use of a patch enlargement.
After experimental (6–8) and clinical (9–11) studies, the decellularized porcine small intestine sub-mucosa (DPSIS) patch has been introduced with the commercial name of CorMatrix® (CorMatrix Cardiovascular, Roswell, GA) for the surgical treatment of congenital heart defects, with implantations performed for closure of septal defects, valve repair, and as vascular patch in both the systemic and pulmonary circulations (12–16).
Despite the initial positive comments, in the last few years several reports appeared with less than favorable medium and long-term outcomes (17–27).
We therefore decided to review our experience with DPSIS patch implantation.
Materials and Methods
Between October 2011 and April 2016, in 51 patients, median age 1.1 months (range 5 days to 14.5 years) and median weight 4.0 kg (range 2.2–50.2 kg), a DPSIS was used for either the reconstruction of the aortic arch (45/51 = 88.2%) or the repair of aortic coarctation (6/51 = 11.8%). All the initial operations have been performed by two surgeons, using exactly the same surgical technique for aortic arch enlargement.
In this group of 51 patients there were 19 patients (19/51 = 37%) with associated malformations: ten patients with ventricular septal defect (VSD), three neonates with hypoplastic left heart syndrome (HLHS), two patients with severe left ventricular outflow tract obstruction (LVOTO), two neonates with aortic arch interruption (AAI), one of whom with associated Transposition of the Great Arteries (TGA), one with complete atrio-ventricular septal defect (cAVSD) and one with congenitally corrected TGA.
All medical records were retrospectively reviewed, with primary endpoints being either interventional procedure (balloon dilatation) or surgery (DPSIS patch replacement or augmentation) due to the recurrent aortic arch/isthmus obstruction.
In all patients the consent for surgery and for the utilization of data for publication and/or presentation in scientific meetings was signed by one of the parents or by the legal guardian the day before surgery.
The retrospective study was approved by the Ethical Committee of the Children's Hospital, University Hospital of Leicester.
Results
While there were 3 hospital deaths (3/51 = 5.9%) not related to the patch implantation, no early or late mortality occurred for the subsequent procedure required for DPSIS patch interventional cardiology or surgery.
Among the 19 patients with associated malformations, in the same surgical procedure with aortic arch/isthmus reconstruction, 6 neonates underwent pulmonary artery banding (four with VSD, one with AVSD and one with congenitally corrected TGA), three had a Norwood procedure (HLHS), two a Damus-Kaye-Stansel procedure (severe LVOTO), and one arterial switch (TGA).
In a median follow-up time of 1.5 ± 1.1 years (range 7 months to 2.3 years) in 13/51 patients (25.5%), a re-intervention, either percutaneous interventional cardiology procedure (5/51 = 9.8%) or re-operation (8/51 = 15.7%) was required because of obstruction in the correspondence of the DPSIS patch used for enlargement of the aortic arch/isthmus. In these 13 patients the indication for re-intervention was given because of a max velocity flow at Doppler interrogation through the aortic arch/isthmus was recorded with a median value of 4.0 ± 0.51 m/s. In all these patients, after the demonstration of the presence of significant pressure gradient at Doppler, the morphology of the recurrent narrowing in the correspondence of the aortic arch/isthmus, shown with echocardiography, was confirmed with either cardiac CT scan or MRI with 3D reconstruction (Figure 1).
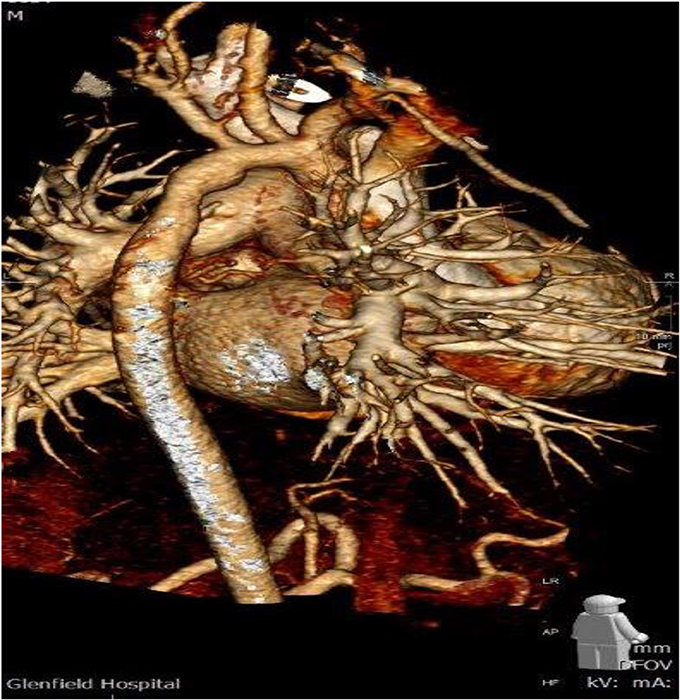
Figure 1. CT scan with 3D reconstruction showing the hypoplastic aortic arch two and half year after DPSIS patch arch reconstruction.
The 11 patients who required re-intervention (percutaneous procedure or re-operation) and the 2 waiting for surgical re-intervention had the DPSIS patch implantation at a median age of 8 days (range 4 days to 8 years). The initial diagnosis and procedure and the type of re-intervention are listed in Table 1.
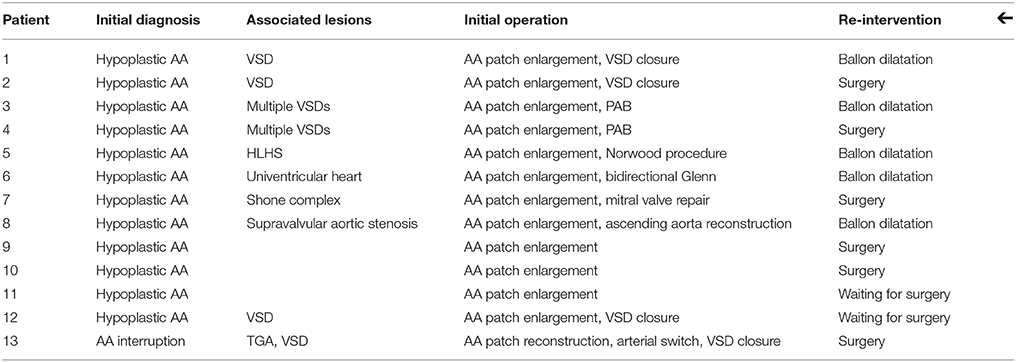
Table 1. AA, aortic arch; HLHS, hypoplastic left heart syndrome; PAB, pulmonary artery banding; TGA, transposition of the great arteries; VSD, ventricular septal defect.
The mean interval between the original surgery and the re-intervention was 6 months (range 10 days to 17 months). Two among these patients required surgery after failed attempt with interventional cardiology procedure.
No early and late mortality occurred for the DPSIS patch surgical implantation and also for the subsequent interventional procedure or surgery.
The max velocity flow at Doppler interrogation through the aortic arch/isthmus for the patients who did not require interventional procedure or surgery was recorder with a mean value of 1.7 ± 0.57 m/s.
No difference has been found between the group of patients who underwent re-intervention because aortic arch/isthmus obstruction and the patients who didn't require re-intervention, as the two groups were homogeneous for age, body weight, associated congenital heart defects and morphology of the aortic arch/isthmus.
No difference has been found between the two surgeons in relationship to the incidence of re-intervention.
In particular, the correlation between the geometrical morphology of the aortic arch and the incidence of re-interventions was investigated in relationship to the presence of gothic aortic arch (28–31). Despite the presence of gothic aortic arch was observed in the pre-operative CT scan investigation in 2/13 (= 15.4%) patients requiring re-intervention vs. only 2/38 (= 5.3%) patients who didn't require re-intervention, the difference didn't reach statistical difference at the T-test (P = 0.12).
The surgically explanted DPSIS patches, macroscopically very thick and rigid (Figure 2), underwent histologic evaluations, with these reports: (a) fragments of fibrous tissue containing cellular necrotic tissue surrounded by histiocytes and occasional multinucleate giant cells; (b) fibrous thickening with neovascularization; (c) presence of perivascular lymphocytes and plasma cells.
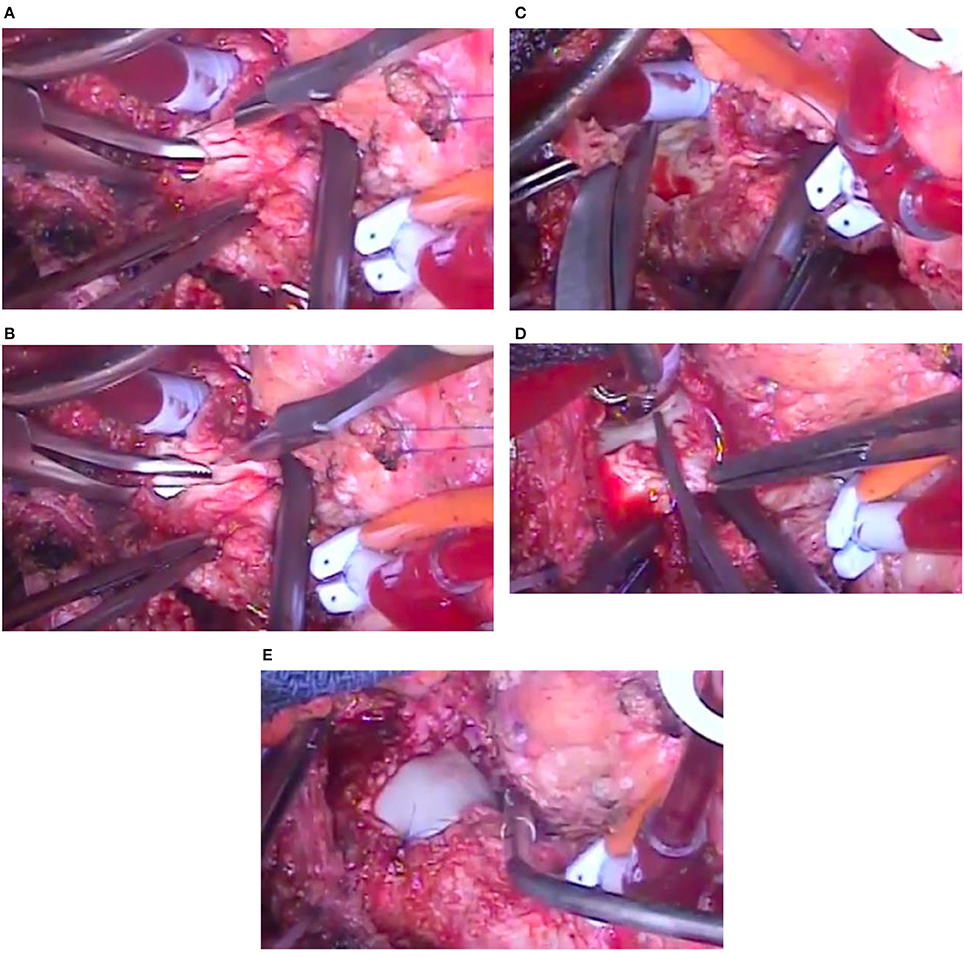
Figure 2. Intra-operative images of the knife incision of the patch, extended with the introduction of a surgical instrument (coming from the left side) through the opening in the narrowing of the aortic arch (A,B), removal by scissors of the thick patch superiorly (C) and inferiorly (D) and new aortic arch enlargement with another patch material (E) in the same patient of Figure 1.
Discussion
Surgery for aortic arch reconstruction with aortic coarctation can be performed with various surgical approaches for cardiopulmonary bypass. Deep hypothermia and circulatory arrest (32–34) has been progressively replaced by regional cerebral perfusion (35–38) with or without associated myocardial perfusion and beating heart (39, 40), and more recently with distal aortic cannulation for lower body perfusion (41–43). Our standard approach was to use regional cerebral and myocardial perfusion with aortic cross clamping and myocardial ischemia only for the repair of the associated intra-cardiac defects, but we do not consider this matter relevant for the surgical results related to this study.
The aortic arch reconstruction can be accomplished with different surgical techniques avoiding the use of a patch to enlarge the aortic narrowing: the ascending sliding arch aortoplasty and the aortic arch advancement technique are the most used (36, 44–47). Alternatively a patch is utilized to enlarge the aortic arch narrowing, with various synthetic and biological materials reported as patch enlargement, including polytetrafluoroethylene, aortic or pulmonary homografts, autologous or heterologous pericardium, autologous vascular patch from the pulmonary artery or the aorta (29, 37, 48–51). The advent of bioengineering has certainly expanded the horizon of materials potentially available as biological patch (52).
Recurrent obstruction in the correspondence of the aortic arch and/or isthmus, requiring either interventional procedures (balloon dilatation/stent) or surgery, is the most frequently reported complication, with an extremely variable incidence reported from 2 to 38%. The recurrent obstruction seems to be correlated with the surgical technique utilized, with the lowest incidence of re-intervention reported with ascending sliding arch aortoplasty and/or aortic arch advancement technique (2–3%), in comparison with the patch enlargement technique (18–38%), occasionally correlated with the patch material used (22, 29–31, 37, 45–51, 53).
In our unit the patch enlargement technique was introduced years ago, motivated by the desire to avoid the potential risk of left bronchus compression, and remained the first choice in the period of the current study, despite very low incidence (0.7 and 1.8%) of left bronchial compression respectively reported with the aortic advancement technique (46) and the end-to side anastomosis (47). With regard to the left bronchial compression surgical approaches have also been suggested either for the prevention of the complication, such as the anterior translocation of the right pulmonary artery (54), or for the treatment, such as the autograft aortic arch extension and sleeve resection (55).
The DPSIS patch was introduced in our unit on October 2011 for closure of septal defects, valve repair, and as vascular patch in both the systemic and pulmonary circulations after positive experimental (6) and clinical reports (12). The company (CorMatrix Cardiovascular, Roswell, GA) claimed, in the official advertising of the patch, the ability of the DPSIS patch to regrow and remodel, constituting a structurally support gradually replaced by native tissues, leaving no foreign material behind, encouraging the body's natural immune response, allowing capillary in-growth and infiltration of white blood cells into newly remodeled tissue, and avoiding inflammation and scarring (CorMatrix®).
Unfortunately the expectations have not been followed by the clinical evidence, with several clinical reports showing poor medium-term clinical outcomes (17–27).
In addition several histologic examinations of the explanted DPSIS patches showed an intense, predominantly eosinophilic inflammatory response with rapid degenerative changes and developing fibrosis (18, 20–22) and a high rate of intimal hyperplasia formation (19), not different from our observations; also aneurysm formation of the patch has been reported after aortic arch implantation (22).
Even in more contrast with the advantages claimed by the company, in no cases the histologic examination of the explanted DPSIS patches showed evidence of tissue integration or recellularization, with the patch acting as a bioscaffold for reconstitution of the native heart tissue ingrowth (20, 23, 27).
Our experience with the histology of the explanted patches confirmed the observations reported in the literature.
Limits of the Study
We are aware of the limits of our study, in particular:
a) this is a single center retrospective analysis. As a matter of fact, when the DPSIS patch has been introduced in our unit a prospective data collection was not started.
b) the results obtained with DPSIS patch have not been compared with other patch materials because the alternative patches have been used either in the years before or after this experience, and therefore the duration of follow-up would have been completely different, and would have not allowed a meaningful comparison.
c) the results obtained with DPSIS patch have not been compared with other surgical techniques because the techniques reported in the discussion, the ascending sliding arch aortoplasty and the aortic arch advancement technique, have been introduced only recently, and therefore the number of patients and follow-up are limited.
Conclusion
The high incidence of re-interventions with DPSIS patch for aortic arch and/or coarctation observed in our experience (25.5%) forced us to abandon the use of this material and replace it with alternative biological materials: homografts and decellularized gluteraldehyde preserved bovine pericardial matrix (CardioCel®, Admedus, Perth, Australia) (56). The preliminary results seem favorable, but a larger number of patients and longer follow-up will be required for a meaningful comparison with the DPSIS patch.
Author Contributions
AC provided a substantial contribution to the conception and design of the work, drafted the work and revised for the content, and agreed to be accountable for all aspects of the work. PS gave an important contribution to the acquisition and analysis of the data of the work. LB helped for the acquisition and analysis of the data of the work, as well as to the interpretation of the data. BM contributed to the interpretation of the data of the work, as well as the revision and approval of the content before submission for publication.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
The reviewer MC and handling Editor declared their shared affiliation.
References
1. Elliott MJ Coarctation of the aorta with arch hypoplasia: improvements on a new technique. Ann Thorac Surg. (1987) 44:321–3. doi: 10.1016/S0003-4975(10)62087-5
2. Vouhé PR, Trinquet F, Lecompte Y, Vernant F, Roux PM, Touati G, et al. Aortic coarctation with hypoplastic aortic arch. results of extended end-to-end aortic arch anastomosis. J Thorac Cardiovasc Surg. (1988) 96:557-63.
3. Barreiro CJ, Ellison TA, Williams JA, Durr ML, Cameron DE, Vricella LA. Subclavian flap aortoplasty: still a safe, reproducible, and effective treatment for infant coarctation. Eur J Cardiothorac Surg. (2007) 31:649–53. doi: 10.1016/j.ejcts.2006.12.038
4. Kotani Y, Anggriawan S, Chetan D, Zhao L, Liyanage N, Saedi A, et al. Fate of the hypoplastic proximal aortic arch in infants undergoing repair for coarctation of the aorta through a left thoracotomy. Ann Thorac Surg. (2014) 98:1386–93. doi: 10.1016/j.athoracsur.2014.05.042
5. Choo YM, Ramli N, Ali S, Hassan RAA, Wong AR, Corno AF. Congenital malformations of the Aortic Arch with associated intra-abdominal issues: when to intervene? World J Med Surg Case Rep. (2015) 4:5–10. Available online at: www.npplweb.com/wjmscr/content/4/2
6. Robotin-Johnson MC, Swanson PE, Johnson DC, Schuessler RB, Cox JL. An experimental model of small intestinal submucosa as a growing vascular graft. J Thorac Cardiovasc Surg. (1998) 116:805–11. doi: 10.1016/S0022-5223(98)00436-X
7. Wainwright JM, Hashizume R, Fujimoto KL, Remlinger NT, Pesyna C, Wagner WR et al. Right ventricular outflow tract repair with a cardiac biologic scaffold. Cells Tissues Organs (2012) 195:159–70. doi: 10.1159/000331400
8. Miller JR, Henn MC, Lancaster TS, Lawrance CP, Schuessler RB, Shepard M et al. Pulmonary valve replacement with small intestine submucosa-extracellular matrix in a porcine model. World J Pediatr Congenit Heart Surg. (2016) 7:475–83. doi: 10.1177/2150135116651113
9. Brinster DR, Patel JA. The use of CorMatrix extracellular matrix for aortic root enlargement. J Cardiothorac Surg. (2014) 9:178. doi: 10.1186/s13019-014-0178-5
10. Abu Saleh WK, Al Jabbari O, Grande-Allen J, Ramchandani M. Extracellular matrix scaffold as a tubular graft for ascending aorta aneurysm repair. J Card Surg. (2015) 30:648–50. doi: 10.1111/jocs.12583
11. Ali JM, Dunning J. Stented CorMatrix® conduit to bypass benign superior vena caval obstruction. Eur J Cardiothorac Surg. (2018) 53:472–74. doi: 10.1093/ejcts/ezx303
12. Scholl FB, Boucek MM, Chan KC, Valdes-Cruz L, Perryman R. Preliminary experience with cardiac reconstruction using decellularized porcine extracellular matrix scaffold: human applications in congenital heart disease. World J Pediatr Congenital Heart Surg. (2010) 1:132–6. doi: 10.1177/2150135110362092
13. Witt RG, Raff G, Van Gundy J, Rodgers-Ohlau M, Si MS. Short-term experience of porcine small intestinal submucosa patches in paediatric cardiovascular surgery. Eur J Cardiothorac Surg. (2013) 44:72–6. doi: 10.1093/ejcts/ezs638
14. Kalfa D, Bacha E New technologies for surgery of the congenital cardiac defect. Rambam Maimonides Med J. (2013) 4:1–14. doi: 10.5041/RMMJ.10119
15. Deorsola L, Pace Napoleone C, Abbruzzese PA Repair of an unusual aortic coarctation using an extracellular matrix patch. Ann Thorac Surg. (2014) 97:1059–61. doi: 10.1016/j.athoracsur.2013.06.109
16. Ashfad A, Brown T, Reemsten B Repair of complete atrioventricular septal defects with decellularized extracellular matrix: initial and midterm outcomes. World J Pediatr Congenit Heart Surg. (2017) 8:310–4. doi: 10.1177/2150135116684797
17. Zaidi AH, Nathan M, Emani S, Baird C, Del Nido PJ, Gauvreau K, et al. Preliminary experience with porcine intestinal submucosa (CorMatrix) for valve reconstruction in congenital heart disease: histologic evaluation of explanted valves. J Thorac Cardiovasc Surg. (2014) 148:2216–24. doi: 10.1016/j.jtcvs.2014.02.081
18. Rosario-Quinones F, Magid MS, Yau J, Pawale A, Nguyen K. Tissue reaction to porcine intestinal submucosa (CorMatrix) implants in pediatric cardiac patients: a single-center experience. Ann Thorac Surg. (2015) 99:1373–7. doi: 10.1016/j.athoracsur.2014.11.064
19. Hibino N. Preliminary experience in the use of an extracellular matrix (CorMatrix) as a tube graft: word of caution. Semin Thorac Cardiovasc Surg. (2015) 27:288–95. doi: 10.1053/j.semtcvs.2015.08.008
20. Woo JS, Fishbein MC, Reemtsen B Histologic examination of decellularized porcine intestinal submucosa extracellular matrix (CorMatrix) in pediatric congenital heart surgery. Cardiovasc Pathol. (2016) 25:12–7. doi: 10.1016/j.carpath.2015.08.007
21. Mosala Nezhad Z, Poncelet A, de Kerchove L, Gianello P, Fervaille C, El Khoury G. Small intestinal submucosa extracellular matric (CorMatrix® in cardiovascular surgery: a systematic review. Interact Cardiovasc Thorac Surg. (2016) 22:839–50. doi: 10.1093/icvts/ivw020
22. Erek E, Aydin S, Suzan D, Yildiz O, Demir IH, Odemis E. Early degeneration of extracellular matrix used for aortic reconstruction during Norwood operation. Ann Thorac Surg (2016) 101:758–60. doi: 10.1016/j.athoracsur.2015.04.051
23. Nelson JS, Heider A, Si MS, Ohye RG. Evaluation of explanted CorMatrix intracardiac patches in children with congenital heart disease. Ann Thorac Surg. (2016) 102:1329–35. doi: 10.1016/j.athoracsur.2016.03.086
24. Padalino MA, Castaldi B, Fedrigo M, Gallo M, Zucchetta F, Vida VL, et al. Porcine intestinal submucosa (CorMatrix) for semilunar valve repair in children: a word of caution after midterm results. Semin Thorac Cardiovasc Surg. (2016) 28:436–45. doi: 10.1053/j.semtcvs.2016.04.015
25. Kelley TM, Kashem M, Wang H, McCarthy J, Carroll ND, Moser GW et al. Anterior leaflet augmentation with CorMatrix porcine extracellular matrix in twenty-five patients: unexpected patch failures and histologic analysis. Ann Thorac Surg. (2017) 103:114–20. doi: 10.1016/j.athoracsur.2016.05.090
26. Dobrilovic N, Soukas P, Sadiq I, Goldstein L, Raman J. Early complications of biologic extracellular matrix patch after use for femoral artery repair. J Vasc Surg. (2017) 65:705–10. doi: 10.1016/j.jvs.2016.07.131
27. Hofmann M, Schmiady MO, Burkhardt BE, Dave HH, Huber M, Kretschmar O, et al. Congenital aortic valve repair using CorMatrix®: a histologic evaluation. Xenotransplantation (2017) 24:e12341. doi: 10.1111/xen.12341
28. Ou P, Mousseaux E, Celermajer DS, Pedroni E, Vouhe P, Sidi D, et al. Aortic arch shape deformation after coarctation surgery: effect on blood pressure response. J Thorac Cardiovasc Surg. (2006) 132:1105–11. doi: 10.1016/j.jtcvs.2006.05.061
29. Seo DM, Park J, Goo HW, Kim YH, Ko JK, Jhang WK. Surgical modification for preventing a gothic arch after aortic arch repair without the use of foreign material. Interact Cardiovasc Thorac Surg. (2015) 20:504–9. doi: 10.1093/icvts/ivu442
30. Bruse JL, Khushnood A, McLeod K, Biglino G, Sermesant M, Pennec X, et al. How successful is successful? Aortic arch shape after successful aortic coarctation repair correlates with left ventricular function. J Thorac Cardiovasc Surg. (2017) 153:418–27. doi: 10.1016/j.jtcvs.2016.09.018
31. Haller C, Chetan D, Saedi A, Parker R, van Arsdell GC, Honjo O. Geometry and growth of the reconstructed aorta in patients with hypoplastic left heart syndrome and variants. J Thorac Cardiovasc Surg. (2017) 153:1479-87 doi: 10.1016/j.jtcvs.2017.02.011
32. Pigula FA Arch reconstruction without circulatory arrest: scientific basis for continued use and application to patients with arch anomalies. Semin Thorac Cardiovasc Surg Pediatr Card Surg Annu. (2002) 5:104–15. doi: 10.1053/pcsu.2002.31480
33. Visconti KJ, Rimmer D, Gauvreau K, Del Nido PJ, Mayer JE, Hagino I, et al. Regional low-flow perfusion versus circulatory arrest in neonates: one-year neurodevelopmental outcome. Ann Thorac Surg. (2006) 82:2207–11. doi: 10.1016/j.athoracsur.2006.06.069
34. De Zelicourt D, Jung P, Horner M, Pekkan K, Kanter KR, Yoganathan AP. Cannulation strategy for aortic arch reconstruction using deep hypothermic circulatory arrest. Ann Thorac Surg. (2010) 94:614–20. doi: 10.1016/j.athoracsur.2012.03.053
35. Corno AF, Pozzi M Safe innominate artery cannulation for cardiopulmonary bypass in neonates. Asian Cardiovasc Thorac Ann. (2007) 15:528–30. doi: 10.1177/021849230701500619
36. McKenzie ED, Klysik M, Morales DL, Heinie JS, Fraser CD, Kovalchin J. Ascending sliding arch aortoplasty: a novel technique for repair of arch hypoplasia. Ann Thorac Surg. (2011) 91:805–10. doi: 10.1016/j.athoracsur.2010.10.038
37. Lamers LJ, Frommelt PC, Mussatto KA, Jaquiss RD, Mitchell ME, Tweddell JS. Coarctectomy combined with an interdigitation arch reconstruction results in a lower incidence of recurrent arch obstruction after the Norwood procedure than coarctectomy alone. J Thorac Cardiovasc Surg. (2012) 143:1098–102. doi: 10.1016/j.jtcvs.2011.09.037
38. Andropoulos DB, Easley RB, Brady K, Mckenzie ED, Heinie JS, Dickerson HA et al. Neurodevelopmental outcomes after regional cerebral perfusion with neuromonitoring for neonatal aortic arch reconstruction. Ann Thorac Surg. (2013) 95:648–54. doi: 10.1016/j.athoracsur.2012.04.070
39. Kotani Y, Ishino K, Kasahara S, Yoshizumi K, Honjo O, Kawada M, et al. Continuous cerebral and myocardial perfusion during aortic arch repair in neonates and infants. ASAIO J. (2008) 52:536–8. doi: 10.1097/01.mat.0000235276.77489.bb
40. Gates RN, Palafox BA, Parker B. Results with all blood microplegia as a myocardial protection strategy for complex neonatal arch reconstruction. ASAIO J. (2008) 54:451–3. doi: 10.1097/MAT.0b013e318184e66d
41. Hammel JM, Deptula JJ, Karamlou T, Wedemeyer E, Abdullah I, Duncan KF. Newborn aortic arch reconstruction with descending aortic cannulation improves postoperative renal function. Ann Thorac Surg. (2013) 96:1721–6. doi: 10.1016/j.athoracsur.2013.06.033
42. Abu-Omar Y, Ali JM, Colah S, Dunning JJ. Aortic arch replacement with a beating heart: a simple method using continuous 3-way perfusion. Perfusion (2014) 29:6–9. doi: 10.1177/0267659113497071
43. Raees MA, Morgan CD, Pinto VL, Westrick AC, Shannon CN, Christian KG et al. Neonatal aortic arch reconstruction with direct splanchnic perfusion avoids deep hypothermia. Ann Thorac Surg. (2017) 104:2054–63. doi: 10.1016/j.athoracsur.2017.04.037
44. Bleakney CA, Zafar F, Fraser CD. Circumflex right aortic arch with associated hypoplasia and coarctation: repair by aortic arch advancement and end-to-side anastomosis. Ann Thorac Surg. (2011) 91:624–6. doi: 10.1016/j.athoracsur.2010.05.056
45. Mery CM, Khan MS, Guzman-Prineda FA, Verm R, Umakanthan R, Watrin CH et al. Contemporary results of surgical repair of recurrent aortic arch obstruction. Ann Thorac Surg. (2014) 98:133–40. doi: 10.1016/j.athoracsur.2014.01.065
46. Mery CM, Guzman-Pruneda FA, Carberry KE, Watrin CH, McChesney GR, Chan JG et al. Aortic arch advancement for aortic coarctation and hypoplastic aortic arch in neonates and infants. Ann Thorac Surg (2014) 98:625–33. doi: 10.1016/j.athoracsur.2014.04.051
47. Lee MG, Brink J, Galati JC, Rakhra SS, Konstantinov IE, Cheung MM et al. End-to-side repair for aortic arch lesions offers excellent chances to reach adulthood without reoperation. Ann Thorac Surg. (2014) 98:1405–11. doi: 10.1016/j.athoracsur.2014.05.007
48. Bernabei M, Margaryan R, Arcieri L, Bianchi G, Pak V, Murzi B. Aortic arch reconstruction in newborns with an autologous pericardial patch: contemporary results. Interact Cardiovasc Thorac Surg. (2013) 16:282–5. doi: 10.1093/icvts/ivs510
49. Bechtold C, Purbojo A, Schwitulla J, Glockler M, Toka O, Dittrich S, et al. Aortic arch reconstruction in neonates with biventricular morphology: increased risk for development of recoarctation by use of autologous pericardium. Thorac Cardiovasc Surg. (2015) 63:373–9. doi: 10.1055/s-0034-1396532
50. Lee H, Yang JH, Jun TG, Cho YH, Kang IS, Huh J et al. Augmentation of the lesser curvature with an autologous vascular patch in complex aortic coarctation and interruption. Ann Thorac Surg. (2016) 101:2309–14. doi: 10.1016/j.athoracsur.2016.01.017
51. Vitanova K, Cleuziou J, Pabst von Ohain J, Burri M, Eicken A, Lange R. Recoarctation after Norwood I procedure for hypoplastic left heart syndrome: impact of patch material. Ann Thorac Surg. (2017) 103:617–21. doi: 10.1016/j.athoracsur.2016.10.030
52. Fraser CD Cardiac bioengineering for congenital heart disease: time for progress. J Cardiovasc Transl Res. (2011) 4:655–7. doi: 10.1007/s12265-011-9294-y
53. Chen PC, Cubberley AT, Reyes K Predictors of reintervention after repair of interrupted aortic arch with ventricular septal defect. Ann Thorac Surg. (2013) 96:621–8. doi: 10.1016/j.athoracsur.2013.04.027
54. Chang YH, Sung SC, Kim H, Choo KS, Lee HD, Park JA. Anterior translocation of the right pulmonary artery to avoid airway compression in aortic arch repair. Ann Thorac Surg. (2013) 96:2198–202. doi: 10.1016/j.athoracsur.2013.06.079
55. Mitchell MB, Campbell DN, Toews WH, Khan TZ. Autograft aortic arch extension and sleeve resection for bronchial compression after interrupted aortic arch repair. Ann Thorac Surg. (2002) 73:1969–71. doi: 10.1016/S0003-4975(02)03412-4
Keywords: aortic coarctation, aortic arch anomalies, aortic arch surgery, patch materials, aorta
Citation: Corno AF, Smith P, Bezuska L and Mimic B (2018) Is Decellularized Porcine Small Intestine Sub-mucosa Patch Suitable for Aortic Arch Repair? Front. Pediatr. 6:149. doi: 10.3389/fped.2018.00149
Received: 20 February 2018; Accepted: 04 May 2018;
Published: 30 May 2018.
Edited by:
Giovanni Biglino, University of Bristol, United KingdomReviewed by:
Massimo Caputo, University of Bristol, United KingdomElena Giulia Milano, University of Verona, Italy
Copyright © 2018 Corno, Smith, Bezuska and Mimic. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Antonio F. Corno, YWZjMTBAbGVpY2VzdGVyLmFjLnVr
 Antonio F. Corno
Antonio F. Corno Paul Smith1
Paul Smith1 Laurynas Bezuska
Laurynas Bezuska Branko Mimic
Branko Mimic