- 1Department of Clinical Nutrition, Hainan Maternal and Children's Medical Center, Haikou, China
- 2Department of Pediatrics, Zhongshan Hospital Affiliated to Dalian University, Dalian, China
- 3Department of Critical Medicine, Hainan Maternal and Children's Medical Center, Haikou, China
Background: Influences of fish oil supplementation on body weight and other cardiometabolic factors in overweight or obese children and adolescents remain not fully understood. We performed a systematic review and meta-analysis of randomized controlled trials (RCTs) to evaluate the role of fish oil for these children.
Methods: Relevant studies were obtained by search of PubMed, Embase, and Cochrane's Library databases. A random-effect model, which incorporates the potential heterogeneity of the included studies, was used to pool the results.
Results: Twelve RCTs including 1,028 overweight or obese children and adolescents were included. Compared to control, fish oil supplementation significantly reduced body mass index [BMI, mean difference (MD): −0.96 kg/m2, 95% confidence interval (CI): −1.69 to −0.23, P = 0.01] but did not significantly reduce body weight or waist circumference (P = 0.68 and 0.76). Moreover, fish oil supplementation significantly reduced serum triglyceride (MD: −0.24 mmol/L, 95% CI: −0.40 to −0.08, P = 0.004) but did not significantly affect serum total cholesterol and high-density or low-density lipoprotein cholesterol (P = 0.83, 0.42, and 0.31, respectively). Additionally, fish oil supplementation significantly lowered systolic blood pressure (SBP, MD: −2.46 mmHg, 95% CI: −4.93 to −0.01, P = 0.04) but did not significantly change diastolic blood pressure (P = 0.22). Supplementation with fish oil did not significantly affect fasting plasma glucose (P = 0.33).
Conclusions: In overweight or obese children and adolescents, supplementation with fish oil could reduce BMI, decrease serum triglyceride, and lower SBP, while serum cholesterol and fasting glucose may not be significantly affected.
Introduction
Obesity in children and adolescents has become an important public health problem (1, 2). A recent systematic review and meta-analysis of 103 studies including 477,620 children aged 2 to 13 years from 28 countries showed a prevalence of combined prevalence of overweight and obesity of up to 30% in 2016 (3). In the United States, about 10% of adolescents had severe obesity according to the NHANES 2013–2014 data (4). Moreover, a considerable increase in Class I obesity has been noticed in children of 2 to 5 years old in the 2015–2016 NHANES cycle (4). Childhood obesity has been associated with comorbidities of almost every body system (2). Notably, obesity in children and adolescents has been associated with increased prevalence of various cardiometabolic disorders, such as dyslipidemia, hypertension, diabetes, metabolic syndrome, etc., which could finally contribute to increased risk of adult cardiovascular diseases and mortality (5–7). Therefore, interventions targeting obesity and related cardiometabolic risk factors in overweight or obese children and adolescents are important to improve their lifetime health status (8, 9).
Nutritional intervention has become an important component of lifestyle intervention in this population (10). Fish oil, which mainly consists of the marine omega-3 polyunsaturated fatty acids (n-3 PUFAs) eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA), has been suggested to reduce weight in adults (11–13). However, previous randomized controlled trials (RCTs) evaluating the efficacy of fish oil supplementation on body weight in overweight or obese children and adolescents showed inconsistent results (14–25). Moreover, fish oil supplementation has been associated with other benefits in obese adults, such as lowering of blood pressure (BP) (26) and improving dyslipidemia (27). However, whether fish oil supplementation in overweight or obese children and adolescents also confers similar benefits on cardiometabolic risk factors including BP, lipid profile, etc. remains unknown. Therefore, in this study, we performed a meta-analysis of RCTs to evaluate the influences of fish oil on childhood obesity and related cardiometabolic risk factors.
Methods
The PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) statement (28) and the Cochrane Handbook guidelines (29) were followed during the designing and implementation of the study.
Search Strategy
PubMed, Embase, and the Cochrane Library (Cochrane Center Register of Controlled Trials) databases were searched for relevant studies with a combined strategy of: (1) “omega-3 fatty acids” OR “fish oil” OR fish-oil OR “polyunsaturated fatty acids” OR “marine oil” OR “eicosapentaenoic acid” OR “docosahexaenoic acid” OR “DHA” OR “EPA”; (2) “child” OR “children” OR “adolescent” OR “pediatric” OR “pediatric”; (3) “obese” OR “obesity” OR “overweight”; AND (4) “random” OR “randomly” OR “randomized” OR “randomized.” Only clinical studies published in English or Chinese were considered. The references of related reviews and original articles were also searched as a complementation. The latest database search was conducted on June 25, 2020.
Study Selection
Inclusion criteria were as follows: (1) peer-reviewed articles in English or Chinese; (2) designed as crossover or parallel-group RCTs; (3) included overweight or obese children AND adolescents who were randomly allocated to an intervention group with fish oil supplementation or a control group with placebo or no treatment; and (4) reported at least one of the following outcomes, including changes of body weight, body mass index (BMI), waist circumference, blood lipids [triglyceride (TG), total cholesterol (TC), high-density lipoprotein cholesterol (HDLC), and low-density lipoprotein cholesterol (LDL-C)], systolic or diastolic blood pressure (SBP or DBP), and fasting plasma glucose (FPG). Reviews, studies including adults, preclinical studies, observational studies, and repeated reports were excluded.
Data Extraction and Quality Assessment
Study search, data extraction, and quality evaluation were achieved by two independent authors. If disagreement occurred, it was resolved by consensus between the two authors. We extracted data regarding study information (first author, publication year, and study country), study design (blind or open-label, crossover, or parallel group), participant characteristics (number of participants, mean age, gender, and health status), regimens of fish oil and controls, and treatment durations. Quality evaluation was achieved using the Cochrane's Risk of Bias Tool (29) according to the following aspects: (1) random sequence generation, (2) allocation concealment, (3) blinding of participants and personnel, (4) blinding of outcome assessors, (5) incomplete outcome data, (6) selective outcome reporting, and (7) other potential bias.
Statistical Analysis
All endpoints were estimated based on the change from baseline to follow-up, and pooled effects were presented as mean differences (MDs) with 95% confidence interval (CI). We used the Cochrane's Q test to detect the heterogeneity, and significant heterogeneity was suggested if P < 0.10 (30). The I2 statistic was also calculated, and an I2 > 50% reflected significant heterogeneity. In view of the clinical heterogeneity among the included studies regarding the characteristics of participants and interventions, a random-effect model, which is considered as a conservative method by incorporating the heterogeneity among the included studies, was applied to pool the results (29). Subgroup analyses were performed to evaluate the influences of study characteristics on the outcomes, including study design, health status of the participants, dose of fish oil, and treatment durations. Medians of the continuous variables were chosen as the cutoff value for defining subgroups. Publication bias was evaluated by visual inspection of funnel plots and Egger's regression asymmetry test (31). P-values < 0.05 were considered statistically significant. The RevMan (Version 5.1; Cochrane, Oxford, UK) and Stata software (Version 12.0; Stata, College Station, TX) were applied for statistical analyses.
Results
Search Results
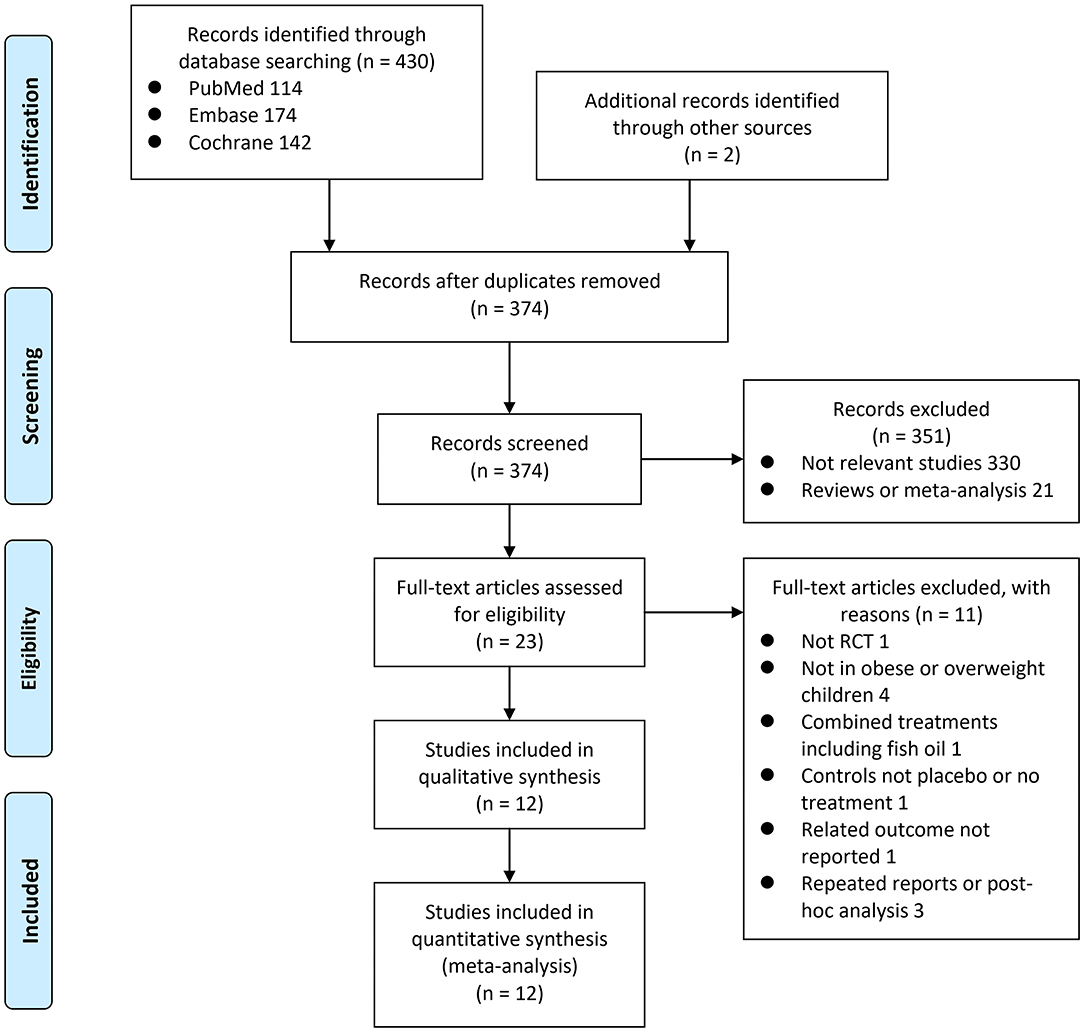
In summary, 432 studies were obtained through the initial database search. After exclusion of duplicate studies, 374 studies were screened. Among them, 351 studies were subsequently excluded based on titles and abstracts primarily because these studies were not irrelevant. Among the 23 potentially relevant articles, 11 were further excluded via full-text review based on reasons listed in Figure 1. Finally, 12 RCTs were included in the meta-analysis (14–25).
Study Characteristics and Quality Evaluation
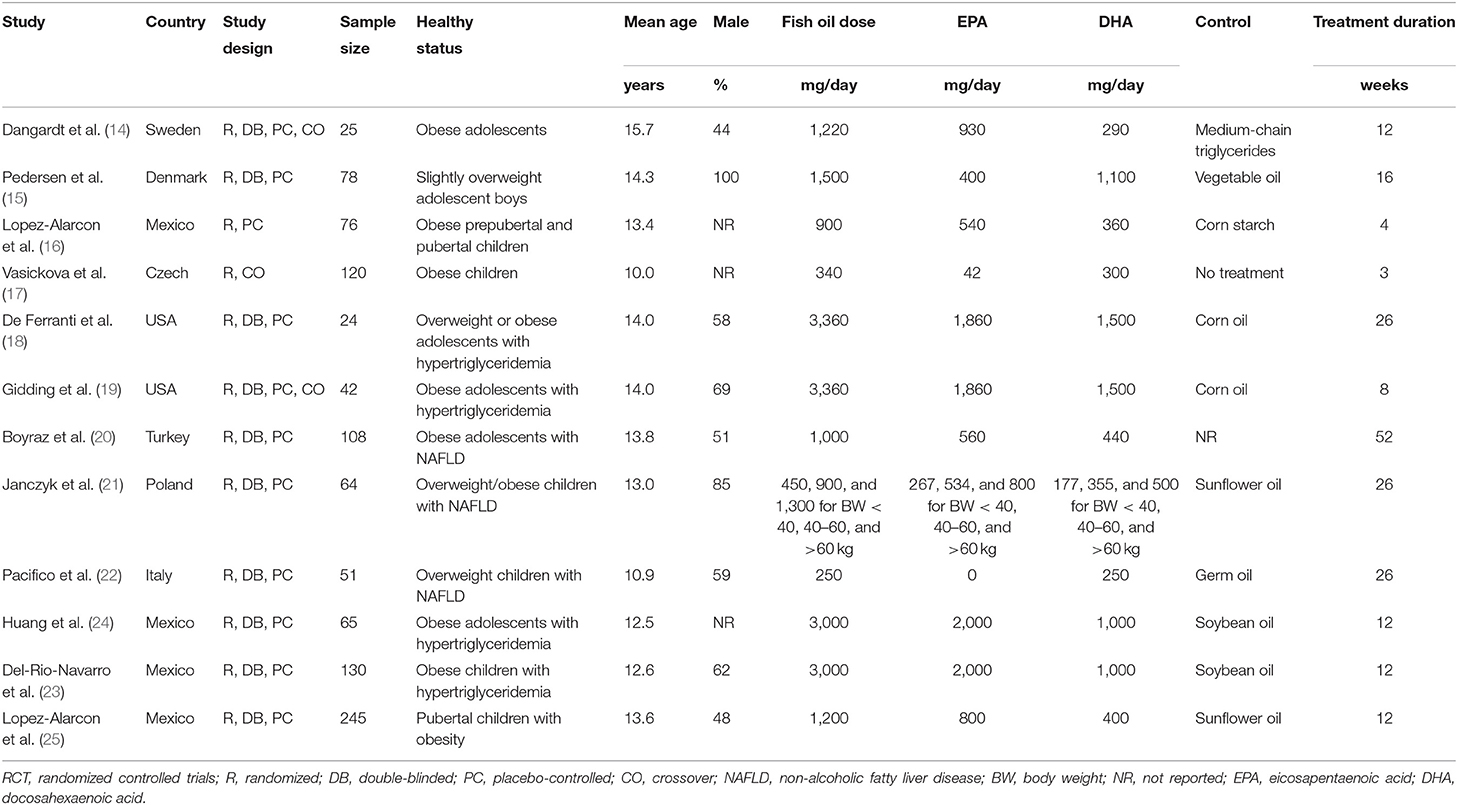
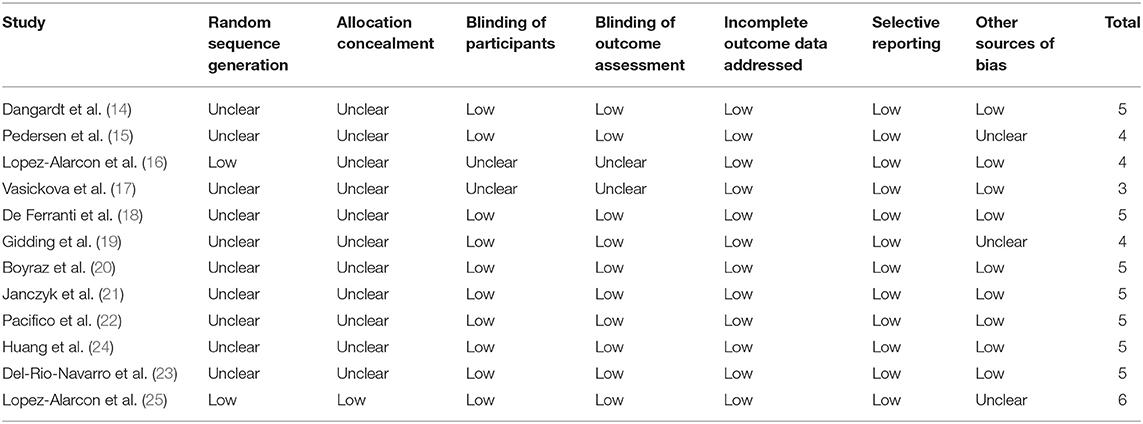
Table 1 shows the characteristics of the included studies. Overall, 12 RCTs including 1,028 overweight or obese children and adolescents were included. These studies were published between 2010 and 2019 and performed in Sweden (14), Denmark (15), Mexico (16, 23–25), the Czech Republic (17), the United States (18, 19), Turkey (20), Poland (21), and Italy (22). Three studies were crossover RCTs (14, 17, 19), while the remaining RCTs were of parallel-group design. All the RCTs included overweight or obese children and adolescents, among which four studies included those with hypertriglyceridemia (18, 19, 23, 24), and the other three included those with non-alcoholic fatty liver disease (NAFLD) (20–22). The mean ages of the included children and adolescents varied between 10 and 16 years. The total dose of fish oil varied from 250 to 3,000 mg/day, with EPA and DHA ranging within 0–2,000 and 177–1,500 mg/day, respectively. The treatment durations varied from 3 to 52 weeks. Table 2 shows the details of study quality evaluation. All of the included RCTs were double blinded except for two studies (16, 17). Methods of random sequence generation were reported in two studies (16, 25), and information of allocation concealment was reported in only one study (25). The overall quality score varied within 3 to 6.
Fish Oil Supplementation on Body Weight, BMI, and Waist Circumference
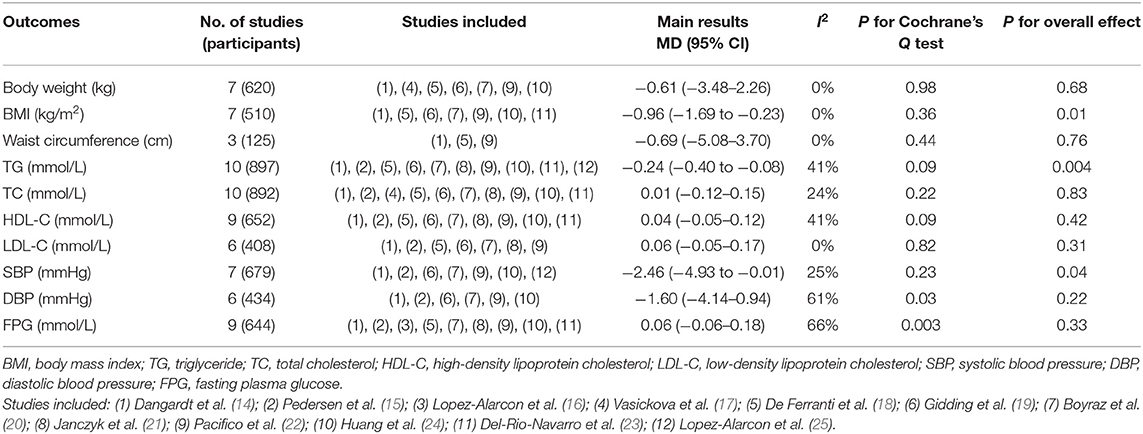
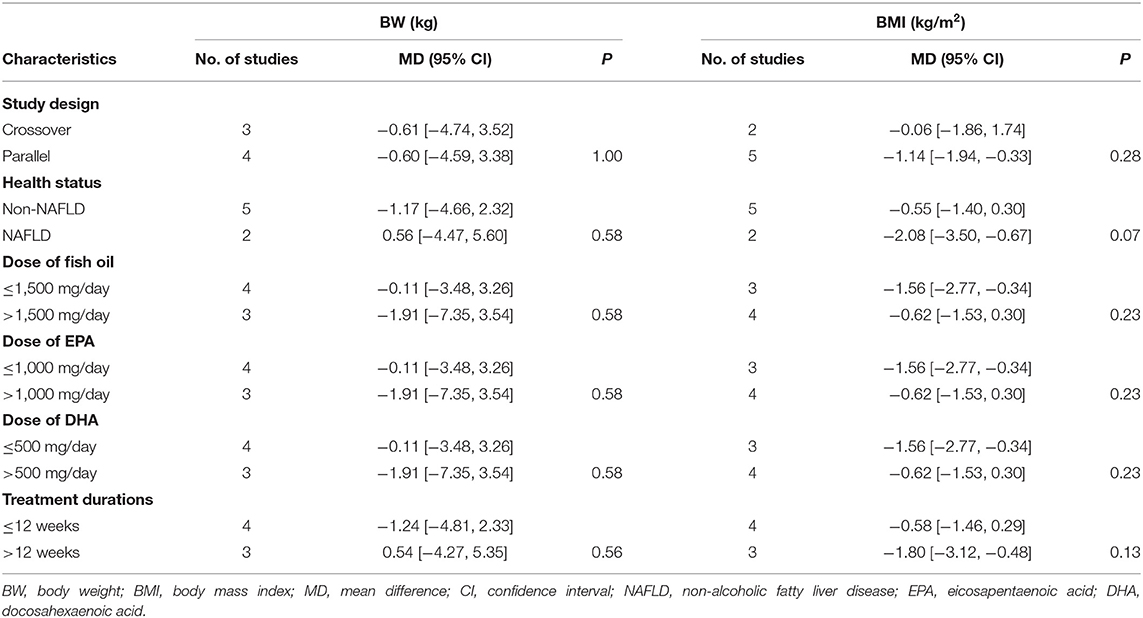
Meta-analysis of seven RCTs showed that compared to control, fish oil supplementation did not significantly reduce body weight of the overweight or obese children and adolescents (MD: −0.61 kg, 95% CI: −3.84–2.26, P = 0.68; I2 = 0%; Table 3). However, fish oil supplementation significantly reduced BMI in these participants (MD: −0.96 kg/m2, 95% CI: −1.69 to −0.23, P = 0.01; I2 = 0%; Table 3). Pooled results of three RCTs showed that fish oil supplementation did not significantly reduce waist circumference (MD: −0.69 cm, 95% CI: −5.08–3.70, P = 0.76; I2 = 0%; Table 3). Subgroup analyses suggested that study characteristics such as study design, health status of the participants, dose of fish oil, or treatment durations did not significantly affect the influences of fish oil supplementation on body weight or BMI in overweight or obese children and adolescents (P for subgroup difference all > 0.05; Table 4).
Fish Oil Supplementation on Blood Lipids
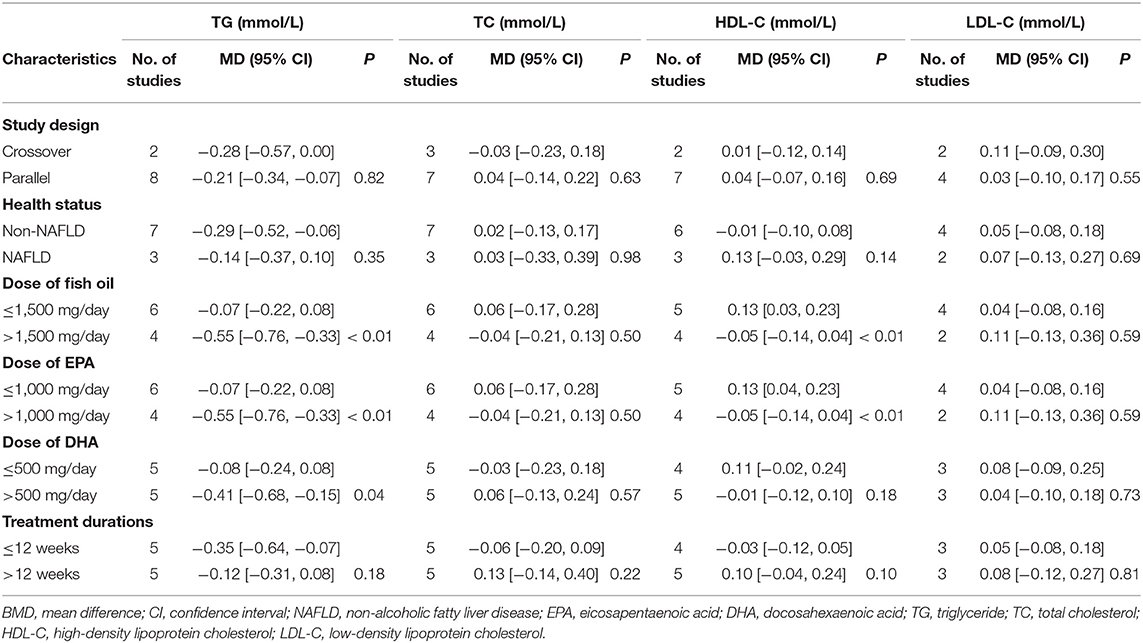
Meta-analysis of 10 RCTs showed that fish oil supplementation significantly reduced serum level of TG compared to control in overweight or obese children and adolescents (MD: −0.24 mmol/L, 95% CI: −0.40 to −0.08, P = 0.004; I2 = 41%; Table 3). Subgroup analysis showed that TG was reduced more remarkably after high-dose fish oil supplementation (fish oil > 1,500 mg/day, EPA > 1,000 mg/day, DHA > 500 mg/day) than that after low-dose fish oil supplementation (P for subgroup difference all < 0.05; Table 5). Compared to control, fish oil supplementation did not significantly affect serum TC (MD: 0.01 mmol/L, 95% CI: −0.12–0.15, P = 0.83; I2 = 24%; Table 3), HDL-C (MD: 0.04 mmol/L, 95% CI: −0.05–0.17, P = 0.42; I2 = 41%; Table 3), or LDL-C (MD: 0.06 mmol/L, 95% CI: −0.05–0.17, P = 0.31; I2 = 0%; Table 3) levels in these participants. Subgroup analysis suggested that study characteristics including study design, health status of the participants, dose of fish oil, or treatment durations did not significantly affect the influences of fish oil supplementation on TC or LDL-C (P for subgroup difference all > 0.05; Table 5). However, HDL-C may be increased in studies with low-dose supplementation (fish oil ≤ 1,500 mg/day, EPA ≤ 1,000 mg/day; P for subgroup difference < 0.05; Table 5).
Fish Oil Supplementation on BP and FPG
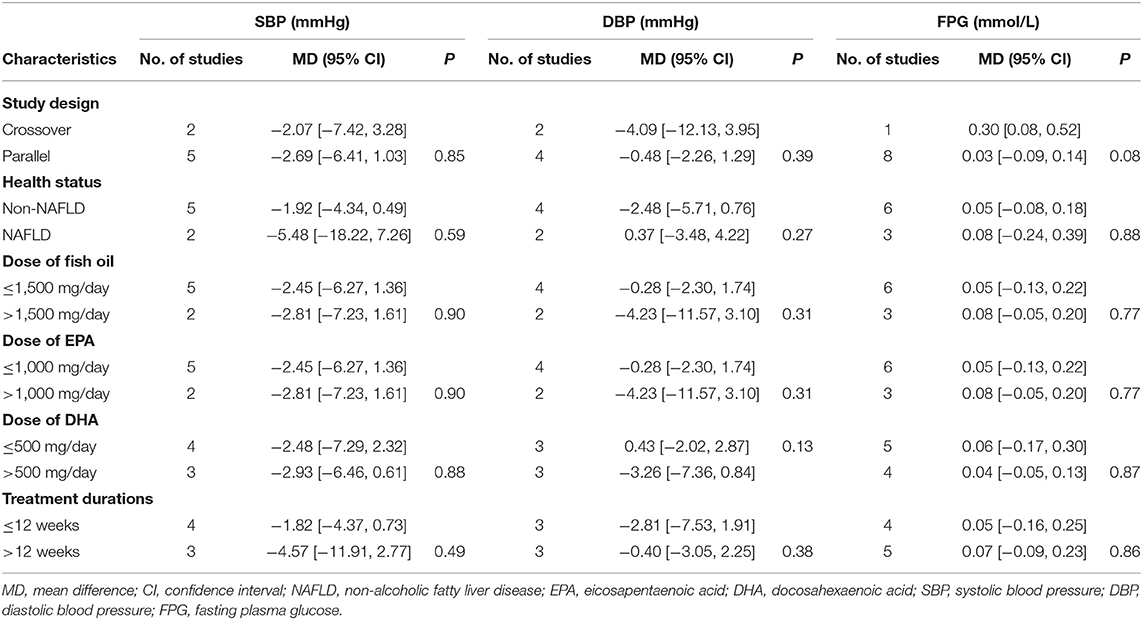
Meta-analysis of seven RCTs showed that fish oil supplementation significantly reduced SBP compared to control in overweight or obese children and adolescents (MD: −2.46 mmHg, 95% CI: −4.93 to −0.01, P = 0.04; I2 = 25%; Table 3), while DBP (MD: −1.60 mmHg, 95% CI: −4.14–0.94, P = 0.03; I2 = 61%; Table 3) or FPG (MD: 0.06 mmol/L, 95% CI: −0.06–0.18, P = 0.33; I2 = 66%; Table 3) was not significantly affected. Subgroup analysis suggested that study characteristics including study design, health status of the participants, dose of fish oil, or treatment durations did not significantly affect the influences of fish oil supplementation on SBP, DBP, and FPG in these participants (P for subgroup difference all > 0.05; Table 6).
Publication Bias
The funnel plots for the meta-analyses of the influences of fish oil supplementation on body weight, BMI, blood lipids, SBP, DBP, and FPG are shown in Figures 2A–I. The plots were symmetrical on visual inspection, suggesting low risk of publication biases. Egger's regression tests showed similar results (P all > 0.10). The potential publication bias underlying the meta-analysis of the influence of fish oil supplementation on waist circumference was undetermined since only three studies were included.
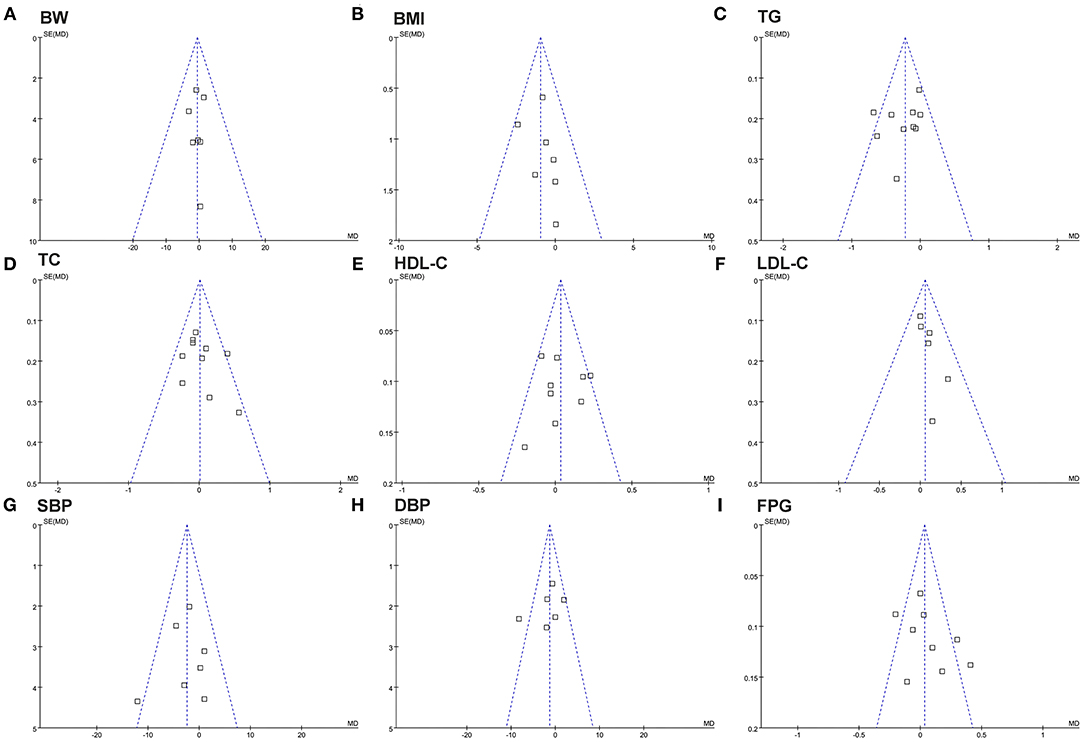
Figure 2. Funnel plots for the meta-analysis evaluating the influence of fish oil supplementation on obesity and other cardiometabolic risk factors in overweight or obese children. (A) Body weight (BW); (B) BMI; (C) TG; (D) TC; (E) HDL-C; (F) LDL-C; (G) SBP; (H) DBP; (I) FPG.
Discussion
In this systematic review and meta-analysis, by pooling the results of available RCTs, we found that fish oil supplementation significantly reduced BMI in overweight or obese children and adolescents, although body weight and waist circumference was not significantly affected. Moreover, fish oil significantly decreased serum TG in these participants, and the efficacy was more remarkable in studies with higher dose of fish oil supplementation. Serum TC, HDL-C, or LDL-C was not significantly affected. In addition, fish oil supplementation was associated with a moderate BP lowering efficacy, as evidenced by a reduction of SBP by 2.42 mmHg. Supplementation with fish oil was not associated with a significant change of FPG. Taken together, these results indicated that in overweight or obese children and adolescents, fish oil supplementation could significantly reduce BMI, decrease TG, and moderately lower SBP compared to controls, suggesting a potential beneficial influence of fish oil on cardiometabolic risk factors in these participants.
To the best of our knowledge, our meta-analysis is the first meta-analysis regarding the influences of fish oil supplementation on obesity and other cardiometabolic risk factors in overweight or obese children and adolescents. One important finding of the study was that fish oil supplementation significantly reduced BMI as compared with controls in overweight or obese children and adolescents. Further subgroup analysis showed that the effect of fish oil supplementation on BMI in these participants was independent of study characteristics such as study design, health status of the participants, dose of fish oil, or treatment durations, which further confirmed the robustness of the finding. Although fish oil was not shown to reduce body weight or waist circumference significantly in this meta-analysis, in view of the superiority of BMI in diagnosis of pediatric obesity to the other two parameters (32), our results confirmed a potential anti-obese efficacy of fish oil in these participants. Previous meta-analysis in obese adults also showed that fish oil supplementation significantly reduced BMI as compared with controls (11). An early cross-sectional study in children between 5 and 12 years from Australia showed that lower omega-3 index evidenced by the erythrocyte fatty acid composition was associated with increased BMI and obesity, as well as insulin resistance (33), indicating a potential inverse association between body long-chain omega-3 polyunsaturated fatty acid (n-3 PUFA) contents and risk of obesity. A subsequent study in subcutaneous adipose tissues of obese adolescents showed that fish oil supplementation modulated the expressions of genes related to lipid metabolism, oxidative stress, and hypoxia, including PPARa, SREBP1, and PGC-1a, accompanied by reduced BMI in these participants. These results suggested that fish oil supplementation may exert its anti-obese efficacy via directly modulating the genes involved in lipid metabolism in obese adolescents (34). Our meta-analysis did not show a lowering efficacy of fish oil supplementation on body weight or waist circumference in overweight or obese children and adolescents, which was not consistent with the findings in a previous meta-analysis in obese adults (11), which may be explained by potential differences between children and adults in terms of body composition and lipid metabolism (35). Besides, for the outcome of waist circumference, only three RCTs with 125 children were included. The influences of fish oil supplementation on waist circumference in overweight or obese children and adolescents should be validated in large-scale RCTs.
As for the influences of fish oil supplementation on blood lipids, our meta-analysis showed that fish oil significantly decreased serum TG in overweight or obese children and adolescents, and the efficacy was more remarkable in studies with higher dose of fish oil supplementation. However, serum TC, HDL-C, or LDL-C was not significantly affected. The TG-lowering efficacy of fish oil in obese children was consistent with the findings of previous studies in adult patients with various clinical conditions, such as diabetes (36) and end-stage kidney diseases (37, 38). Besides, results of subgroup analyses showed that HDL-C in obese adolescents may be increased in studies with low-dose supplementation (fish oil ≤ 1,500 mg/day, EPA ≤ 1,000 mg/day) and in studies with longer treatment durations (>12 weeks), which has also been observed in previous studies including adult participants (36–38). In view of the role of HDL-C in attenuating atherosclerosis, these findings may highlight other mechanisms underlying the potential cardiometabolic benefits of fish oil in overweight or obese children and adolescents.
In addition, fish oil supplementation was found to moderately lower SBP in the overweight or obese children and adolescents. An early study has confirmed that high n-3 PUFA levels in thin/normal weight children are associated with lower and therefore healthier BP (39), suggesting a potential role of n-3 PUFA in maintaining healthy BP. Besides, endothelial dysfunction has been considered as an initial factor for hypertension in children and adults (40). Fish oil supplementation has been shown to improve endothelial function in children (24, 41), which was consistent with the findings in adult populations (42). Moreover, our meta-analysis did not show a significant influence of fish oil supplementation on FPG in obese adolescents, which is consistent with previous findings in adult patients (36).
The strengths of our meta-analysis may include the following. Firstly, only RCTs were included, which minimized the possible biases caused by studies with other designs. Secondly, only RCTs with interventions of exclusive fish oil supplementation were included, which eliminated the potential confounding effects of other co-interventions. Thirdly, the age range of the participants of the included RCTs was not wide and was representative of school-age children and adolescent populations, which ensured the homogeneity of the study population. Besides, our study also has some limitations. First, characteristics of participants and fish oil treatment regimens were varied among the included studies, such as the age, sex, and health status of the children and adolescents, as well as the dosages and treatment durations of fish oil, which may contribute to the heterogeneity among the included studies. Secondly, although subgroup analyses were performed to explore the potential influences of variances in study characteristics on the outcome, results of subgroup analyses should be interpreted with caution because limited datasets were included for each subgroup. Thirdly, the dietary intake of fish and omega-3 fatty acids were generally not reported in the included studies. Differences in the dietary omega-3 fatty acid intake may affect the potential benefits of fish oil supplementation in these participants. Finally, clinical outcomes were not evaluated in this study. Whether fish oil supplementation could reduce morbidity and mortality in overweight or obese children and adolescents may be investigated in large-scale RCTs.
In conclusion, supplementation with fish oil could reduce BMI, decrease serum triglyceride, and lower SBP in overweight or obese children and adolescents, while serum cholesterol and fasting glucose may not be significantly affected. Findings of the meta-analysis should be validated in large-scale RCTs. Moreover, studies are warranted to evaluate the influences of fish oil supplementation on lifetime morbidity and mortality in overweight or obese children and adolescents.
Data Availability Statement
The original contributions generated for this study are included in the article/supplementary material, further inquiries can be directed to the corresponding author/s.
Author Contributions
SWu, CZ, XH, and ZL designed the study. SWu, CZ, and ZW performed database search, literature identification, study quality evaluation, and data extraction. SWa, PY, and TS performed statistical analysis. CZ, SWa, PY, and TS interpreted the results. SWu, CZ, XH, and ZL drafted the manuscript. All authors critically revised the manuscript and approved its submission.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
1. Kohut T, Robbins J, Panganiban J. Update on childhood/adolescent obesity and its sequela. Curr Opin Pediatr. (2019) 31:645–53. doi: 10.1097/MOP.0000000000000786
2. Morales Camacho WJ, Molina Diaz JM, Plata Ortiz S, Plata Ortiz JE, Morales Camacho MA, Calderon BP. Childhood obesity: aetiology, comorbidities, and treatment. Diabetes Metab Res Rev. (2019) 35:e3203. doi: 10.1002/dmrr.3203
3. Garrido-Miguel M, Cavero-Redondo I, Alvarez-Bueno C, Rodriguez-Artalejo F, Moreno LA, Ruiz JR, et al. Prevalence and trends of overweight and obesity in European children from 1999 to 2016: a systematic review and meta-analysis. JAMA Pediatr. (2019) 173:e192430. doi: 10.1001/jamapediatrics.2019.2430
4. Skinner AC, Ravanbakht SN, Skelton JA, Perrin EM, Armstrong SC. Prevalence of obesity and severe obesity in US children, 1999-2016. Pediatrics. (2018) 141:e20173459. doi: 10.1542/peds.2017-3459
5. Umer A, Kelley GA, Cottrell LE, Giacobbi PJr, Innes KE, Lilly CL. Childhood obesity and adult cardiovascular disease risk factors: a systematic review with meta-analysis. BMC Public Health. (2017) 17:683. doi: 10.1186/s12889-017-4691-z
6. Weihrauch-Bluher S, Schwarz P, Klusmann JH. Childhood obesity: increased risk for cardiometabolic disease and cancer in adulthood. Metabolism. (2019) 92:147–52. doi: 10.1016/j.metabol.2018.12.001
7. Lindberg L, Danielsson P, Persson M, Marcus C, Hagman E. Association of childhood obesity with risk of early all-cause and cause-specific mortality: a Swedish prospective cohort study. PLoS Med. (2020) 17:e1003078. doi: 10.1371/journal.pmed.1003078
8. Bleich SN, Vercammen KA, Zatz LY, Frelier JM, Ebbeling CB, Peeters A. Interventions to prevent global childhood overweight and obesity: a systematic review. Lancet Diabetes Endocrinol. (2018) 6:332–46. doi: 10.1016/S2213-8587(17)30358-3
9. Cardel MI, Atkinson MA, Taveras EM, Holm JC, Kelly AS. Obesity treatment among adolescents: a review of current evidence and future directions. JAMA Pediatr. (2020) 174:609–17. doi: 10.1001/jamapediatrics.2020.0085
10. Kim J, Lim H. Nutritional management in childhood obesity. J Obes Metab Syndr. (2019) 28:225–35. doi: 10.7570/jomes.2019.28.4.225
11. Bender N, Portmann M, Heg Z, Hofmann K, Zwahlen M, Egger M. Fish or n3-PUFA intake and body composition: a systematic review and meta-analysis. Obes Rev. (2014) 15:657–65. doi: 10.1111/obr.12189
12. Du S, Jin J, Fang W, Su Q. Does fish oil have an anti-obesity effect in overweight/obese adults? A meta-analysis of randomized controlled trials. PLoS ONE. (2015) 10:e0142652. doi: 10.1371/journal.pone.0142652
13. Zhang YY, Liu W, Zhao TY, Tian HM. Efficacy of Omega-3 polyunsaturated fatty acids supplementation in managing overweight and obesity: a meta-analysis of randomized clinical trials. J Nutr Health Aging. (2017) 21:187–92. doi: 10.1007/s12603-016-0755-5
14. Dangardt F, Osika W, Chen Y, Nilsson U, Gan LM, Gronowitz E, et al. Omega-3 fatty acid supplementation improves vascular function and reduces inflammation in obese adolescents. Atherosclerosis. (2010) 212:580–5. doi: 10.1016/j.atherosclerosis.2010.06.046
15. Pedersen MH, Molgaard C, Hellgren LI, Lauritzen L. Effects of fish oil supplementation on markers of the metabolic syndrome. J Pediatr. (2010) 157:395–400. doi: 10.1016/j.jpeds.2010.04.001
16. Lopez-Alarcon M, Martinez-Coronado A, Velarde-Castro O, Rendon-Macias E, Fernandez J. Supplementation of n3 long-chain polyunsaturated fatty acid synergistically decreases insulin resistance with weight loss of obese prepubertal and pubertal children. Arch Med Res. (2011) 42:502–8. doi: 10.1016/j.arcmed.2011.06.010
17. Vasickova L, Stavek P, Suchanek P. Possible effect of DHA intake on body weight reduction and lipid metabolism in obese children. Neuro Endocrinol Lett. (2011) 32(Suppl. 2):64–7.
18. De Ferranti SD, Milliren CE, Denhoff ER, Steltz SK, Selamet Tierney ES, Feldman HA, et al. Using high-dose omega-3 fatty acid supplements to lower triglyceride levels in 10- to 19-year-olds. Clin Pediatr. (2014) 53:428–38. doi: 10.1177/0009922814528032
19. Gidding SS, Prospero C, Hossain J, Zappalla F, Balagopal PB, Falkner B, et al. A double-blind randomized trial of fish oil to lower triglycerides and improve cardiometabolic risk in adolescents. J Pediatr. (2014) 165:497–503.e492. doi: 10.1016/j.jpeds.2014.05.039
20. Boyraz M, Pirgon O, Dundar B, Cekmez F, Hatipoglu N. Long-term treatment with n-3 polyunsaturated fatty acids as a monotherapy in children with nonalcoholic fatty liver disease. J Clin Res Pediatr Endocrinol. (2015) 7:121–7. doi: 10.4274/jcrpe.1749
21. Janczyk W, Lebensztejn D, Wierzbicka-Rucinska A, Mazur A, Neuhoff-Murawska J, Matusik P, et al. Omega-3 Fatty acids therapy in children with nonalcoholic Fatty liver disease: a randomized controlled trial. J Pediatr. (2015) 166:1358–63.e1351–1353. doi: 10.1016/j.jpeds.2015.01.056
22. Pacifico L, Bonci E, Di Martino M, Versacci P, Andreoli G, Silvestri LM, et al. A double-blind, placebo-controlled randomized trial to evaluate the efficacy of docosahexaenoic acid supplementation on hepatic fat and associated cardiovascular risk factors in overweight children with nonalcoholic fatty liver disease. Nutr Metab Cardiovasc Dis. (2015) 25:734–41. doi: 10.1016/j.numecd.2015.04.003
23. Del-Rio-Navarro BE, Miranda-Lora AL, Huang F, Hall-Mondragon MS, Leija-Martinez JJ. Effect of supplementation with omega-3 fatty acids on hypertriglyceridemia in pediatric patients with obesity. J Pediatr Endocrinol Metab. (2019) 32:811–9. doi: 10.1515/jpem-2018-0409
24. Huang F, Del-Rio-Navarro BE, Leija-Martinez J, Torres-Alcantara S, Ruiz-Bedolla E, Hernandez-Cadena L, et al. Effect of omega-3 fatty acids supplementation combined with lifestyle intervention on adipokines and biomarkers of endothelial dysfunction in obese adolescents with hypertriglyceridemia. J Nutr Biochem. (2019) 64:162–9. doi: 10.1016/j.jnutbio.2018.10.012
25. Lopez-Alarcon M, Inda-Icaza P, Marquez-Maldonado MC, Armenta-Alvarez A, Barbosa-Cortes L, Maldonado-Hernandez J, et al. A randomized control trial of the impact of LCPUFA-omega3 supplementation on body weight and insulin resistance in pubertal children with obesity. Pediatr Obes. (2019) 14:e12499. doi: 10.1111/ijpo.12499
26. Colussi G, Catena C, Novello M, Bertin N, Sechi LA. Impact of omega-3 polyunsaturated fatty acids on vascular function and blood pressure: relevance for cardiovascular outcomes. Nutr Metab Cardiovasc Dis. (2017) 27:191–200. doi: 10.1016/j.numecd.2016.07.011
27. Asbaghi O, Choghakhori R, Abbasnezhad A. Effect of Omega-3 and vitamin E co-supplementation on serum lipids concentrations in overweight patients with metabolic disorders: a systematic review and meta-analysis of randomized controlled trials. Diabetes Metab Syndr. (2019) 13:2525–31. doi: 10.1016/j.dsx.2019.07.001
28. Moher D, Liberati A, Tetzlaff J, Altman DG. Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. BMJ. (2009) 339:b2535. doi: 10.1136/bmj.b2535
29. Higgins J, Green S. Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0. The Cochrane Collaboration. (2011). Available online at: www.cochranehandbook.org (accessed April, 2020).
30. Higgins JP, Thompson SG. Quantifying heterogeneity in a meta-analysis. Stat Med. (2002) 21:1539–58. doi: 10.1002/sim.1186
31. Egger M, Davey Smith G, Schneider M, Minder C. Bias in meta-analysis detected by a simple, graphical test. BMJ. (1997) 315:629–34. doi: 10.1136/bmj.315.7109.629
32. Simmonds M, Llewellyn A, Owen CG, Woolacott N. Simple tests for the diagnosis of childhood obesity: a systematic review and meta-analysis. Obes Rev. (2016) 17:1301–15. doi: 10.1111/obr.12462
33. Burrows T, Collins CE, Garg ML. Omega-3 index, obesity and insulin resistance in children. Int J Pediatr Obes. (2011) 6:e532–9. doi: 10.3109/17477166.2010.549489
34. Mejia-Barradas CM, Del-Rio-Navarro BE, Dominguez-Lopez A, Campos-Rodriguez R, Martinez-Godinez M, Rojas-Hernandez S, et al. The consumption of n-3 polyunsaturated fatty acids differentially modulates gene expression of peroxisome proliferator-activated receptor alpha and gamma and hypoxia-inducible factor 1 alpha in subcutaneous adipose tissue of obese adolescents. Endocrine. (2014) 45:98–105. doi: 10.1007/s12020-013-9941-y
35. Cardoso Chaves O, Do Carmo Castro Franceschini S, Machado Rocha Ribeiro S, Ferreira Rocha Sant'ana L, Garcon De Faria C, Eloiza Priore S. Comparison of the biochemical, anthropometric and body composition variables between adolescents from 10 to 13 years old and their parents. Nutr Hosp. (2012) 27:1127–33. doi: 10.3305/nh.2012.27.4.5832
36. Gao C, Liu Y, Gan Y, Bao W, Peng X, Xing Q, et al. Effects of fish oil supplementation on glucose control and lipid levels among patients with type 2 diabetes mellitus: a Meta-analysis of randomized controlled trials. Lipids Health Dis. (2020) 19:87. doi: 10.1186/s12944-020-01214-w
37. Lim AK, Manley KJ, Roberts MA, Fraenkel MB. Fish oil treatment for kidney transplant recipients: a meta-analysis of randomized controlled trials. Transplantation. (2007) 83:831–8. doi: 10.1097/01.tp.0000258613.32993.84
38. Zhu W, Dong C, Du H, Zhang H, Chen J, Hu X, et al. Effects of fish oil on serum lipid profile in dialysis patients: a systematic review and meta-analysis of randomized controlled trials. Lipids Health Dis. (2014) 13:127. doi: 10.1186/1476-511X-13-127
39. Wolters M, Pala V, Russo P, Rise P, Moreno LA, De Henauw S, et al. Associations of Whole Blood n-3 and n-6 Polyunsaturated Fatty Acids with Blood Pressure in Children and Adolescents - results from the IDEFICS/I.Family Cohort. PLoS ONE. (2016) 11:e0165981. doi: 10.1371/journal.pone.0165981
40. Konukoglu D, Uzun H. Endothelial Dysfunction and Hypertension. Adv Exp Med Biol. (2017) 956:511–40. doi: 10.1007/5584_2016_90
41. Ciccone MM, Scicchitano P, Gesualdo M, Zito A, Carbonara S, Ricci G, et al. The role of omega-3 polyunsaturated fatty acids supplementation in childhood: a review. Recent Pat Cardiovasc Drug Discov. (2013) 8:42–55. doi: 10.2174/1574890111308010006
Keywords: fish oil, eicosapentaenoic acid, docosahexaenoic acid, children, adolescents, obesity
Citation: Wu S, Zhu C, Wang Z, Wang S, Yuan P, Song T, Hou X and Lei Z (2021) Effects of Fish Oil Supplementation on Cardiometabolic Risk Factors in Overweight or Obese Children and Adolescents: A Meta-Analysis of Randomized Controlled Trials. Front. Pediatr. 9:604469. doi: 10.3389/fped.2021.604469
Received: 10 September 2020; Accepted: 22 February 2021;
Published: 27 April 2021.
Edited by:
Consolato Sergi, University of Alberta Hospital, CanadaReviewed by:
Joseph Feulefack, University of Alberta, CanadaMarta Cristina Sanabria, National University of Asunción, Paraguay
Copyright © 2021 Wu, Zhu, Wang, Wang, Yuan, Song, Hou and Lei. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Xiaoli Hou, eGlhb2xpaG91ODkyMkAxMjYuY29t; Zhixian Lei, bGVpemhpeGlhbl84NTUyQDIxY24uY29t
 Shaojing Wu1
Shaojing Wu1 Xiaoli Hou
Xiaoli Hou