- 1 Centre for Socioeconomic Research, Cardiff University, Cardiff, UK
- 2 All Wales Adult Cystic Fibrosis Centre, Llandough Hospital, Cardiff, UK
Background: Health-related quality of life (HRQoL) is a rapidly growing area of expertise and the most commonly used patient-reported outcome (PRO). The impact of cystic fibrosis (CF) on HRQoL is liable to be great, making CF patients ideal candidates for the application of HRQoL instruments. The aims of this study were to assess the affect of CF on HRQoL, to ascertain the reliability and validity of the United Kingdom Sickness Impact Profile (UKSIP) and the Cystic Fibrosis Quality of Life Questionnaire (CFQoL) in the adult CF population, and to examine their role in the management of patients. Methods: Seventy participants were recruited from the All Wales Adult Cystic Fibrosis Centre at Llandough Hospital, UK. There were two stages to the study: self-report of the UKSIP and CFQoL; and completion of the same two questionnaires 7–10 days later. Results: The areas of HRQoL most impaired by CF were employment and concerns regarding the future. The UKSIP and CFQoL showed high internal consistency (rα = 0.89–0.93) and test–retest reliability (rs = 0.57–0.94, p < 0.005) in the CF population. Validity was variable with the UKSIP showing discrimination across socio-demographic factors, whilst the CFQoL showed increased sensitivity to clinical variables. Many parameters influenced patient-reported HRQoL, with the greatest correlations seen with the Borg score (p < 0.005). The use of a HRQoL instrument in CF annual reviews is recommended to provide holistic patient care. The results of this study underpin the value of HRQoL as a patient-reported outcome measure in the management of adult CF.
Introduction
Cystic fibrosis (CF) is the most common genetic disease in Caucasians (Sheppard and Nicholson, 2002). The outlook for CF patients has greatly improved over recent years and the life expectancy of a child born in the 2000s is predicted to be in excess of 40 years, double that of 20 years ago (Mason, 2005). The impact of CF on a patient’s Health-related quality of life (HRQoL) is likely to be great and cannot be assessed purely by physiological parameters, and therefore analysis of patient’s views on the wider impact of the disease is needed (Fallowfield, 1991). The nature of CF results in an increase in treatment burden, yet a decrease in physical and psychosocial health over time. This, complimentary to the fact that there is considerable variation in disease severity, makes CF patients ideal candidates for application of HRQoL instruments. QoL is also increasingly being recognized by major CF organizations as a priority area of investigation (WHO, 1999; Littlewood, 2001), to establish its relevance as a patient-reported outcomes (PROs) measure in the management of CF.
Quality of life research in CF patients has primarily focused on assessment of specific treatments, comparison with other populations, and comparison with some clinical variables (Hatziagorou et al., 2002). Wahl et al. (2005) commented that “little attention has been paid to patient satisfaction with broader life domains.” This study addresses this gap in research, and prioritizes areas of importance to the patient that may not have been predicted by the physician. Assessment in this area focuses on the patient and their needs; an invaluable resource for practitioners to develop a partnership of care with the patient, following the current thinking at the forefront of healthcare policy (Marshall et al., 2005). Analysis of present literature also identified a skewed coverage of QoL favoring adolescents with CF over adults (Jong et al., 1997a). As a demographic change toward an increasing number of adults with CF has taken place, this study focused on an adult population (WHO, 1999).
Hatziagorou et al. (2002) comment that much needs to be done to improve the quality of QoL measures. This study provides significant information about the suitability of the United Kingdom Sickness Impact Profile (UKSIP) and Cystic Fibrosis Quality of Life questionnaire (CFQoL; Gee et al., 2000; Quittner et al., 2009) as instruments to assess HRQoL in CF patients; by assessing their validity, reliability, and patient acceptability. The UKSIP is a gold standard generic questionnaire that has previously never been used in a CF population, and the CFQoL is a disease-specific questionnaire that has never been validated against a gold standard HRQoL instrument. Using this unique and highly comprehensive double-instrument technique also allows areas of HRQoL to be highlighted that may have been overlooked in previous studies using individual instruments.
Materials and Methods
Study Participants
The sample population was recruited from the Specialist Adult Centre for CF at Llandough Hospital, Cardiff over a 10-week period. Participants were over 16 years old, in a stable phase of their disease and had adequate understanding to complete the questionnaires. There were three separate cohorts recruited: outpatients; inpatients; and postal respondents.
Study Instruments
A double-instrument technique was used, including generic and disease-specific measures. This approach is highly favored as it is both robust and clinically advantageous (French, 1995; Bowling, 2002; Fitzpatrick, 2002). Before considering different applications of patient-based outcome measures, it is helpful to note a recommendation that has been made by some authors that the optimal strategy is to use a combination of types of measure in a clinical trial. Most commonly it is recommended that trialists include a generic together with a disease-specific measure (Guyatt et al., 1991; Fletcher et al., 1992; Bombardier et al., 1995; Abbott et al., 2011). The main argument for such an approach is that the two kinds of measures are likely to produce complementary evidence in showing change over time (Fitzpatrick et al., 1998).
The United Kingdom Sickness Impact Profile
The UKSIP is the British version of the American SIP developed in the early 1970s. It is a self-reported, tick box, weighted instrument consisting of 12 different categories: sleep and rest; eating; work; home management; recreation and pastimes; ambulation; mobility; body care and movement; social interaction; alertness behavior; emotional behavior; and communication (Schraufnagel, 1999). Scores are derived from a weighted scale, with increasing score indicating a greater impairment of HRQoL. The SIP has been shown to be of use in the chronically ill (Bowling, 2005), and its specific use in CF has also been demonstrated (Jong et al., 1997a). The UKSIP has the advantage of generating a single index score, summary indices and profile scores for the 12 categories. The UKSIP has an established validity and reliability in many disease areas (Salek and Luscombe, 1992), and has also been used as a generic HRQoL measure in a number of chronic conditions such as atopic dermatitis (Sowden et al., 1991; Salek et al., 1993a), acne (Salek et al., 1996), psoriasis (Salek et al., 2004), renal (Salek et al., 2003), Parkinson’s disease (Salek et al., 1993b, 1994), cardiovascular (Salek and VandenBurg, 1988), and arthritis (Salek, 1997).
Cystic Fibrosis Quality of Life Questionnaire
The CFQoL is a patient-derived self-reported instrument designed specifically to assess the needs of CF patients (Gee et al., 2000). It covers nine areas: physical functioning; social functioning; treatment issues; chest symptoms; emotional functioning; concerns for the future; interpersonal relationships; body image; and career concerns (Gee et al., 2003). It has a six-point Likert scale where the degree of agreement or frequency of occurrence is given a numerical value; with a decreasing score indicating a greater impairment on QoL. It has high reliability and validity, and robust discriminatory ability (Gee et al., 2000). This credibility coupled with its ease of use (about 10 min to complete and of UK origin) means that it has been utilized in several trials for CF (Gee et al., 2003, 2005; Esmond et al., 2006).
The CFQoL was selected for this study following careful examination of a number of QoL instruments such as the Cystic Fibrosis Questionnaire (CFQ14+; Henry et al., 2003) developed in France, Living with Cystic Fibrosis Questionnaire (LCFQ; Patterson et al., 2008) developed in the USA and EuroQol 5 Dimension for Children and Adolescents (EQ-5D-Y; Wille and Ravens-Sieberer, 2006; Wille et al., 2006a,b). Consequently, the CFQoL was considered to be the most robust, widely used and cross culturally translated CF specific QoL instrument (Debska and Mazurek, 2007; Monti et al., 2008).
Study Design
There were two stages to the study: the first stage (Test I) involved completion of the two questionnaires when delivered by the interviewer or received by post; the second stage (Test II) was completion of the same two questionnaires 7–10 days later, with a postal return.
The outpatients were recruited from the weekly CF clinics at Llandough hospital. The consultant assessed the stability of the patients’ health before recruitment to provide validation of the test–retest reliability analysis. This was achieved by ensuring all participants had no significant change in lung function (within 10% of their normal FEV1).
The inpatients were recruited on the day of discharge from hospital and were asked to complete the UKSIP whilst the researcher was present. The CFQoL was not completed at this stage due to its retrospective frame of reference. The remaining three questionnaires were then supplied to the participant to return by post.
A separate cohort of patients not scheduled to attend clinic within 10 weeks was identified. These 54 patients were sent the entire study by post. These participants were coded separately.
The three cohorts were included in the study in order to capture different degrees of severity and to secure a bigger sample size.
All participants were provided with self-addressed stamped envelopes to return the second stage, as well as covering letters specifying the dates of completion. To minimize training effect, order of the completion of the questionnaires was randomized for the outpatient and postal cohorts using computer generated random numbers. Those who did not respond in the postal cohort were contacted first by telephone and then by letter. If there was no subsequent response then they were classed as non-responders.
All clinical data was collected from the notes on the day of completion of the questionnaire or if this was unavailable, from the next closest date. This included data on lung function (FEV1, FVC, and PEFR) and nutritional adequacy (body mass index, BMI), as well as the use of oral and enteral supplementary feeds. Physical activity was assessed using the Borg score; an established known measure of the perception of dyspnea, and consequently exercise capacity in CF (Dodd et al., 2005; Debska and Mazurek, 2007).
Data Analysis
The data was not normally distributed and therefore was analyzed by descriptive and non-parametric tests using SPSS 18.0. The use of Mann–Whitney U tests established no significant difference between the mean scores of the postal and interviewer-delivered cohorts, therefore they were analyzed as one sample.
Differences in scores were analyzed with the socio-demographic and clinical status of the participants using Mann–Whitney U Tests. Associations between the HRQoL scores and the socio-demographic and clinical measures were examined using Spearman’s rank correlation coefficients. Reliability was assessed by internal consistency (Cronbach’s alpha) and test–retest reliability analysis (Spearman’s rank correlation coefficients). These are the standard reliability tests used to assess HRQoL instruments. Validity of the instruments was considered by concurrent, predictive, and face validity. The significance level was set at p < 0.05 for two tailed tests. All correlations were interpreted according to the guidelines set by Cohen in 1988, small (r = ±0.10 to 0.29), medium (r = ±0.30 to 0.49), and large (r = ± 0.50 to 1.0; Pallant, 2005).
Results
Sociodemographic and Clinical Characteristics of the Study Participants
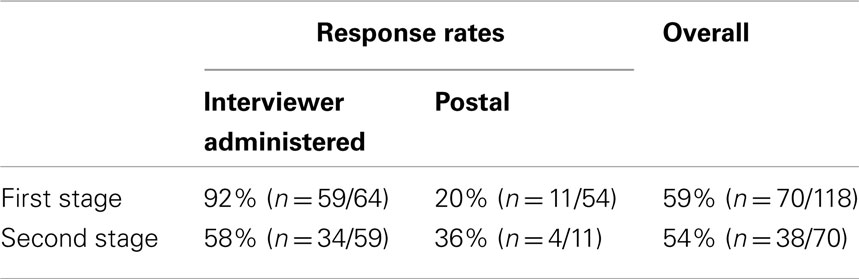
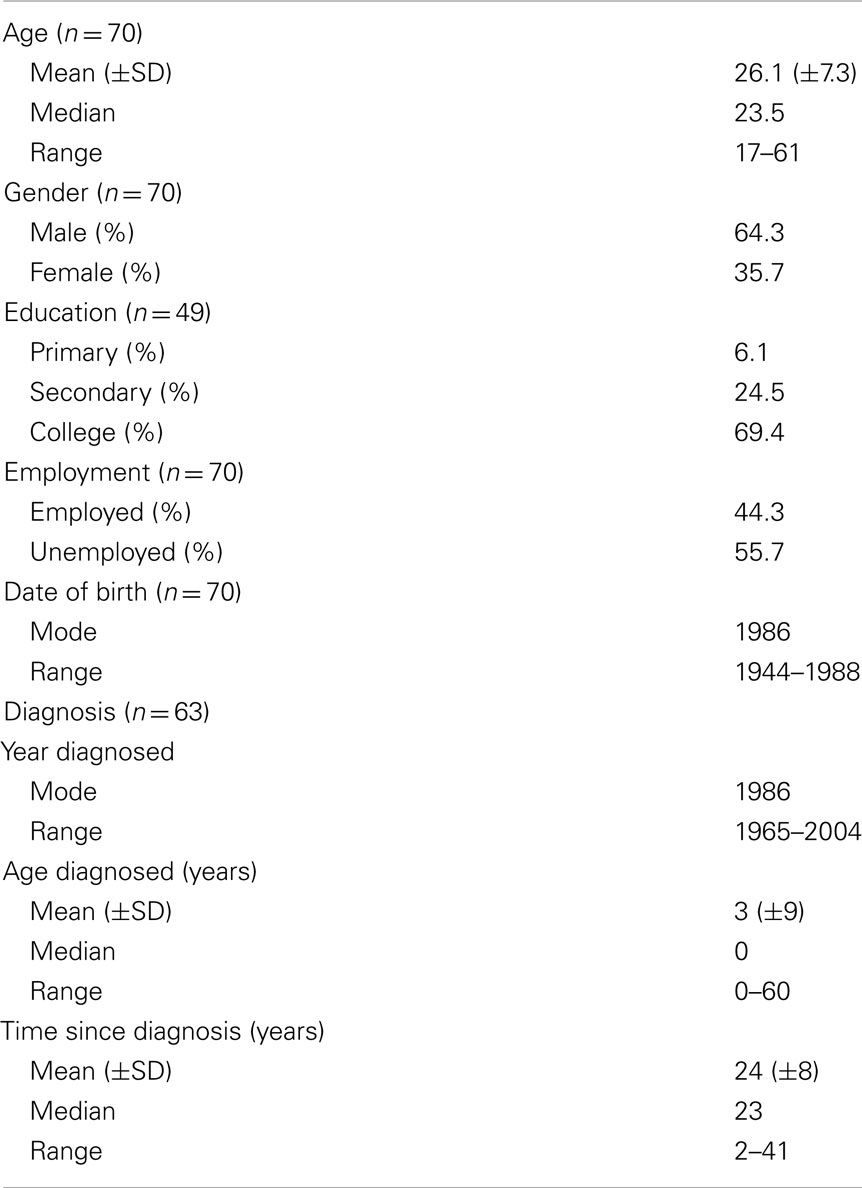
A total of 118 patients were recruited into the study of whom 70 took part (Table 1); an overall response rate of 59%. The patients mean age was 26 (median = 23; range 17–61 years) with 45 (64%) males (Table 2). Thirty-nine (56%) of the study participants were unemployed. Over half the study population reported that CF had stopped them working at some point, with 82% of those currently unemployed attributing it to their CF. Thirty-four (69%) of those participants responding had completed college education, with just three of the participants having only completed primary education. Clinical data is displayed in Table 1.
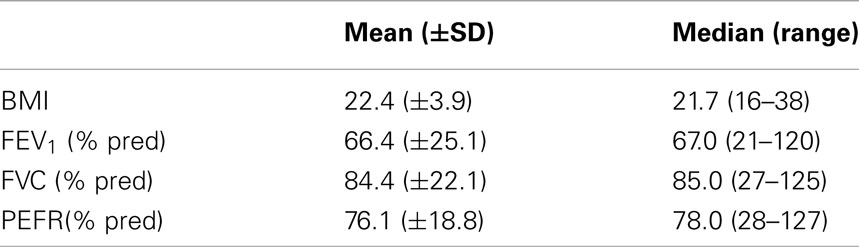
Table 3 shows the nutritional and lung function values of the study participants. As the scores were not normally distributed, both the mean and median have been reported as well as the ranges seen.
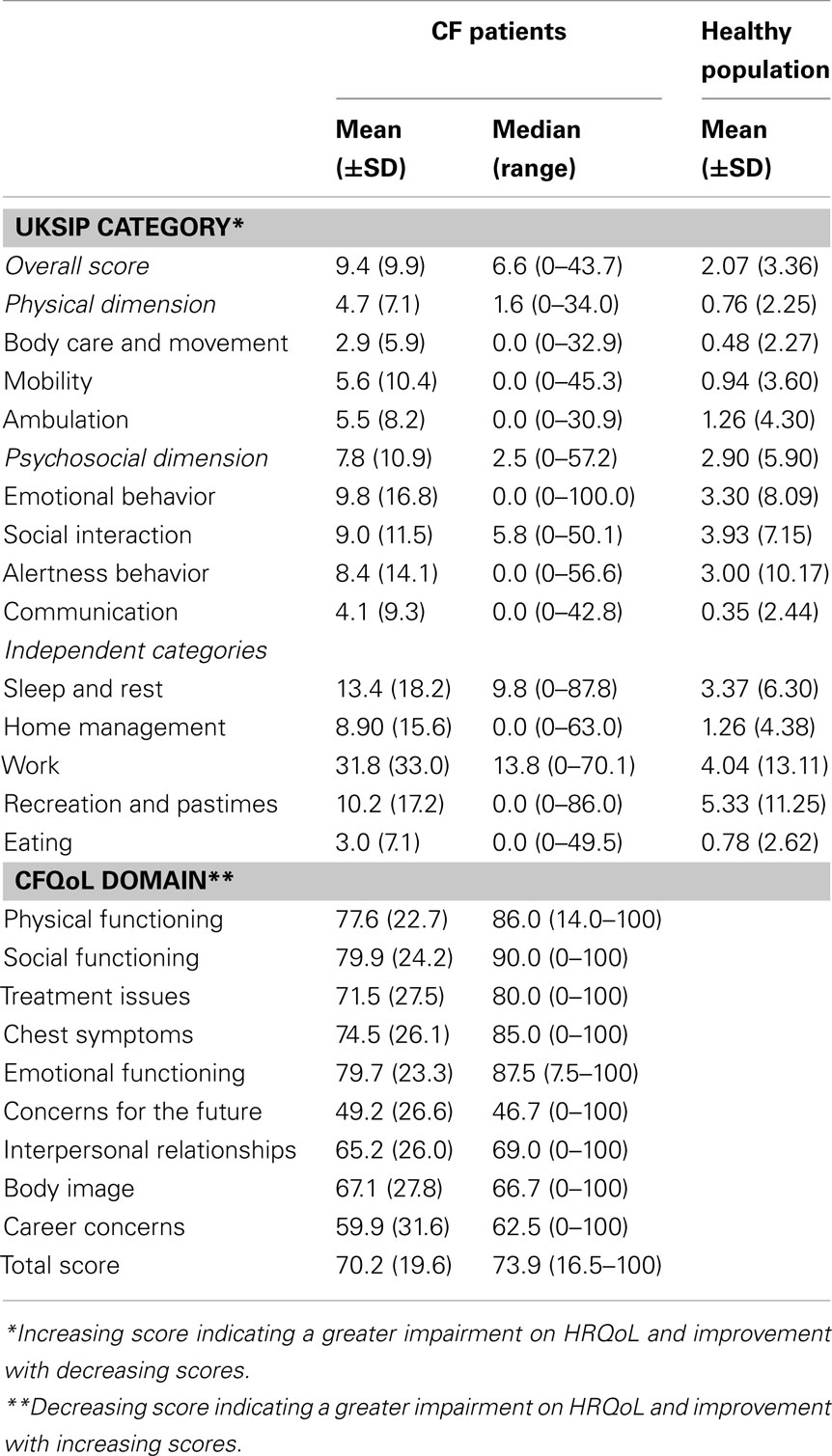
Table 4 highlights the mean HRQoL scores; showing that the poorest area on HRQoL was work for the UKSIP (M = 31.8) and concerns for the future in the CFQoL (M = 49.2).
Relationship between HRQoL Scores and Socio-Demographics
Moderate correlations were found between age and the patient-reported UKSIP categories recreation and pastimes (rs = 0.35, p = 0.003) and work (rs = 0.31, p = 0.01), as well as small correlations with overall score, ambulation and home management (rs = 0.25–0.29, p < 0.05). No correlations between the CFQoL scores and age were found.
Females reported a poorer HRQoL than males in all UKSIP categories except for communication, where males showed the greatest impairment (p = 0.38). A statistically significant poorer HRQoL was seen for females in the UKSIP categories, emotional behavior (p = 0.012) and eating (p = 0.033). No significant differences in CFQoL scores were found between males and females. Gender differences were not examined in detail as this has already been researched in the present literature (Gee et al., 2005).
Participants’ employment status resulted in differences in UKSIP scores in all areas except communication (p < 0.05), with the unemployed reporting a poorer HRQoL. Highly significant differences were found between employed and unemployed participants for all CFQoL domains except treatment issues and concerns for the future (p < 0.01).
Significant differences were found between those at secondary and college education levels in the UKSIP categories ambulation, alertness behavior, work, and physical dimension (p < 0.01); with the former showing the greater impairment on HRQoL. Significant differences were also found with overall UKSIP score, body care and movement, mobility and eating (p < 0.05). There were no significant differences with the study participants in the primary education group. One significant difference was found in the CFQoL categories across education, with those completing primary education showing an increased impairment on HRQoL in the career concerns than those in college education (p < 0.05).
Reliability of the UKSIP and CFQoL
The standard level for acceptability of Cronbach’s alpha values is >0.70. The internal consistency reliability of the UKSIP and CFQoL was high for both Test I and Test II; with Cronbach’s alpha coefficients of 0.87 and 0.90 (n = 70 and 38), and 0.90 and 0.93 (n = 57 and 30), respectively. Test–retest reliability for both instruments was high; UKSIP (rs = 0.57–0.84, p < 0.01, n = 38) and CFQoL (rs = 0.74–0.94, p < 0.01, n = 38) demonstrating an acceptable level of reproducibility for both measures.
Validity
Face validity
All participants found the CFQoL easy to understand (n = 68) and easy to complete (n = 68). Fifty-two participants (74%) were willing to complete it every time they came to clinic (n = 67), and completion time was approximately 9 min.
Concurrent validity
All categories except communication in the UKSIP showed significant correlations with the UKSIP patient-rated overall health question (rs = 0.26–0.55, p < 0.05). Similarly the CFQoL scores were significantly correlated in all domains except concerns for the future (rs = −0.27 to −0.69, p < 0.05). Similar domains (emotion, social interaction, physical, employment, and total scores) in the two questionnaires showed highly significant correlations (rs = −0.50 to −0.76, p < 0.01).
Predictive Validity
Disease severity
Disease severity was defined according to FEV1 (% predicted) as mild (≥70%), moderate (40–69%), and severe (<40%); n = 32, 25, and 11 respectively. No significant differences were found between UKSIP scores for severe and moderate disease. Significant differences in UKSIP scores between moderate and mild disease severities were found for ambulation, work, physical dimension, and overall score (p < 0.05). Severe disease showed the larger impairment on HRQoL than mild disease in the UKSIP categories physical dimension (p = 0.01) and eating (p < 0.05), as well as work, ambulation and overall UKSIP score (p < 0.05). Two highly significant differences were found between the severe and mild disease severities for the CFQoL categories chest symptoms (p < 0.01) and body image (p < 0.01). Significant differences were also found between severe and mild disease severities for the CFQoL categories physical functioning, chest symptoms, emotional functioning, body image, career concerns, and total score (p < 0.05). The severe disease groups also showed a greater impairment on emotional functioning than the moderate severity group (p < 0.05). Differences were also found between moderate and mild disease severities in physical functioning (p < 0.05) and chest symptoms (p < 0.05).
Body mass index. No significant differences were found between the UKSIP scores and BMI classifications (≤19 (underweight), 20–29 (normal), ≥30 (overweight), n = 11, 53, and 5 respectively. In the CFQoL scores the underweight participants reported a poorer physical functioning (p < 0.05) and body image (p < 0.01) than those in the normal group. They also reported a poorer HRQoL compared to the overweight group across physical functioning, treatment issues, body image, and career concerns (p < 0.05). Between the normal and overweight groups significant differences were found for treatment issues (p < 0.05) and career concerns (p < 0.05), with the greatest impairment in the normal group.
Supplementary feeding. A highly significant difference was found between oral supplementary feeding and enteral feeding groups in relation to the UKSIP categories sleep and rest (p < 0.01) and eating (p < 0.05). In the CFQoL scores, a highly significant difference was found across body image with those receiving enteral feeding reporting a poorer HRQoL than those not receiving any form of supplementary feeding (p < 0.01). Those receiving oral supplements reported an increased impairment on HRQoL in treatment issues (p < 0.05) and body image (p < 0.005).
The BORG scale. The Borg score showed highly significant correlations (p < 0.01) with 12 of the UKSIP categories, nine of these showed large correlations (rs = 0.50–0.61). With the CFQoL, correlations were found in all domains except treatment issues, eight domains were statistically significant (rs = −0.39 to −0.62, p < 0.05).
Discussion
This study has found that CF has a substantial impact on HRQoL in relation to work and concerns regarding the future. The percentage of unemployed in this study is comparable to recent survey statistics, where 53% were currently unemployed (Walters and Warren, 2001). Consideration of the large effects observed on HRQoL due to unemployment, in parallel to the fact that over three quarters of those unemployed reported CF as the cause, highlights that CF has a direct substantial impact on this domain of life. The negative implications of unemployment on HRQoL is a well reported phenomenon; both generally (Fallowfield, 1991; Scrambler, 1995) and specifically with regards to CF (Harris and Super, 1995; Jong et al., 1997a). Therefore a healthcare professional’s role in providing employment advice, with regards to health, could be key to improving HRQoL in this area. The increasing future concerns relating to fertility and transplant status are likely to cause HRQoL impairment in young adults, and as they progress through adulthood the prospect of an early death becomes more of a reality. This highlights a need in clinical practice for the role of counseling and psychology in a multidisciplinary approach to CF care, as has already been recommended in current guidelines (Kerem et al., 2005).
This study has also revealed that dyspnea could have a large impact on a CF patient’s HRQoL, as the Borg score correlated more significantly than any other clinical variable. This is consistent with other studies that have found that dyspnea is highly correlated to HRQoL, indicating the considerable effect that it has on the patient’s life (Schrier et al., 1990; Jong et al., 1997b). One theory of why dyspnea is so closely related to HRQoL scores is that the questions used in assessing physical HRQoL and the dyspnea value are similar, containing the same questions to assess both areas (e.g., Are you feeling breathless?; Schrier et al., 1990). In contrast, the findings in this study show HRQoL impairment across many of the categories in both questionnaires. Because of the relatively new use of the Borg score at Llandough Hospital not all patients had received an assessment, reducing the sample population involved. However the fact that such large statistical significance was shown indicates that this is an area that merits further investigation. The greater correlations seen with the UKSIP suggest that it may be the broader domains of HRQoL that are influenced by dyspnea.
The greater impairment seen on HRQoL with increasing age (using the UKSIP) is to be expected, due to the multiple pathological conditions that a CF patient faces as their disease progresses, and is reported in current literature (Quittner et al., 2005). However, the CFQoL did not show correlations with age, perhaps suggesting a lack of specificity of the CFQoL.
The UKSIP results, suggesting that women have a statistically lower HRQoL concerning emotional wellbeing than men, complement the present literature (Congleton et al., 1996; Gee et al., 2003). The decreased HRQoL with regards to eating in females compared to men is reported elsewhere (Abbott et al., 2000), and suggests a differing attitude between males and females in relation to food and can be linked to the increased likelihood of eating disorders seen in women (Walters, 2001). Males reported a poorer HRQoL in the CFQoL body image domain, a finding previously reported (Abbott et al., 2000; Gee et al., 2003). This could be explained by the fact that the low BMI often seen in CF patients may be considered undesirable to males but more appealing to women in relation to their ideal personal body type, irrespective of the health implications. The poorer overall HRQoL indicated in women compared to men may be because women are subject to a poorer prognosis than males in CF (Harris and Super, 1995; Abbott et al., 2000). The CFQoL did not detect any significant differences between males and females, which could indicate a lack of discriminative ability in this area; contrary to its use in other papers (Gee et al., 2003; Gobbi et al., 2005).
The physical dimension in the UKSIP was most affected by disease severity; this is as expected as a poorer lung function would limit physical activities. The CFQoL supports this with the greatest difference being shown in chest symptoms. Eating was also adversely affected highlighting the other major pathological effect of CF which is on the gastrointestinal system, where 95% of patients show exocrine pancreatic insufficiency with symptoms of malabsorption and constipation (Mason, 2005). No significant differences were found between the severe and moderate disease severities in the UKSIP and only one in the CFQoL, suggesting that HRQoL reduces significantly once a patient reaches moderate disease severity. However, it could be that neither of the instruments were sensitive enough to detect differences between these two disease groups, as other questionnaires in literature have demonstrated discriminatory ability in this area (Quittner et al., 2005).
The UKSIP showed no discrimination between the groupings by BMI, implicating the superiority of the CFQoL with regards to relationships with clinical measures. The fact that the underweight reported a poorer HRQoL again highlights the major impact that the failure to thrive caused by CF has on the HRQoL of the individual. Physical functioning was reduced in the underweight participants suggesting they have a poorer overall health and physical capacity. This may be because malnutrition is linked in a vicious circle to lung infection (Ratjen and Doring, 2003).
The UKSIP highlighted an impairment on sleep related HRQoL of those receiving enteral feeding, probably due to the fact that the feeds are usually given overnight (Mason, 2005). This is a very important issue as adequate sleep is regarded as a prime requirement to the QoL of any person (Salek, 1993). Body image in the CFQoL was also reduced in the enteral feeding group, possibly due to an increased self consciousness (Abbott et al., 1999). Those on oral supplements reported problems in the treatment issues domain suggesting that adding another dimension of treatment can impair a patient’s HRQoL. This could be of increased relevance in CF due to the polypharmacy involved and the compliance issues that patients are continually faced with. This could have clinical practice implications when weighing up the benefit risk ratio of adding a treatment to a patient’s regimen.
Both Test I and Test II alpha coefficients for the UKSIP and CFQoL were high, indicating their acceptable internal consistency reliability in this application. The reliability values for the CFQoL complement the current literature and were greater than those for the UKSIP (Gee et al., 2000). Not all participants replied within the 7- to 10-day period specified, however this would only reduce the correlation seen between the scores meaning that the reliability reported is probably lower than reality. The UKSIP has demonstrated good reliability and validity as a proxy assessment by informed carers (i.e., husbands, wives, siblings, etc.; Salek et al., 1995a,b,c). However, this was not examined in this study since it is not relevant to the CF population.
Face validity of the CFQoL was high, however some of the comments from the participants highlighted the need for a not applicable option on the questionnaire. The UKSIP also had some patient comments regarding the fact that there was no sometimes option, and some participants found the wording hard to understand, reducing its face validity. The response rate in this study was slightly above average (Walters and Warren, 2001; Gee et al., 2003). However, the UKSIP was developed as a generic measure for use in patients with different diseases and was extensively tested for its psychometric properties and showed excellent content and discriminant validity (Bergner et al., 1981).
Concurrent validity with the UKSIP was established through a strong association with the patient-rated overall health question. The fact that concerns for the future was not correlated is likely to be because the impairment in this domain is so great that it is not related to their present state of health but is continual throughout their life. This is backed by findings in another study that found poor scores for this domain irrespective of disease severity (Walters and Warren, 2001). The questionnaires also showed high correlations between their applicable domains, again indicating that they are both adequately measuring HRQoL. This validity complements the present literature for the CFQoL which has shown good concurrent validity with the SF-36 (Gee et al., 2000).
Predictive validity and sensitivity of the questionnaires was variable. The UKSIP was highly discriminative across socio-demographic areas with limited clinical distinction; and the opposite was true of the CFQoL. Accurate choice of a generic or disease-specific measure could therefore be fundamental when these questionnaires are used in research or clinical practice. Although not examined in this study, both instruments have shown to be capable of detecting change over time (Salek et al., 1993a). The number of assessment points in detecting change over time depends on the aims and objective of the study, but should not be used at assessment points of less than 4-week intervals to avoid under or over estimation.
Limitations
One limitation of this study was that there was a mixture of postal return and interviewer-administered questionnaires. Although this could be a potential source of bias, it is common practice to use a postal method to ensure practicality for the patient, and was essential to ensure that patients who did not attend clinic were included in the results. A further limitation is that many statistical tests were completed, increasing the likelihood of significant results due to chance. This was confounded by the fact that more than one factor could influence HRQoL, for example the results were not adjusted for age or gender when looking at disease severity. However, as there were few erratic or inexplicable results and high levels of clinical significance reported, this limitation is reduced. Finally, the differing protocols required for inpatients, outpatients, and postal cohorts could cause bias; however these were designed to limit their affect on the results.
Conclusion
The role of HRQoL measures as a PRO can offer “a new dimension” to multidisciplinary health care and complement clinical practice by the inclusion of a HRQoL measure in a CF annual review setting (Salek, 1993; Gee et al., 2000). This approach was identified as being beneficial by a participant who commented that they wished they could have completed a similar questionnaire earlier in their disease process to compare it with their results now. Increased consideration of HRQoL issues when considering clinical interventions and treatments is also important in the management of patients with CF.
This study highlights the detrimental impact that unemployment has on all areas of HRQoL. However, the influence of CF on working status goes beyond personal health status, to involve complicated and possibly sensitive issues surrounding social stigma and discrimination. Several participants mentioned problems gaining and maintaining employment that they attributed to their CF; resulting in some not revealing their CF to employers. These issues may also influence the patient’s perspective on whether they are fit to work which could have substantial implications on clinical practice with regards to: treatments that could facilitate CF patients to return to work; encouragement and explanation of the health benefits of working by health care professionals; and advice to patients during periods of unemployment.
Until recently no CF specific HRQoL measures were available, therefore assessment in this area is fairly novel. Despite their individual limitations, the two instruments used in this study offer reliability and validity in the CF population. As the life expectancy of the CF population continues to increase, the use of HRQoL measures should rise in accordance with this; both to maximize patient benefit and to allow effective partnership between the healthcare professional and the CF patient. The use of two questionnaires in a clinical setting is impractical, but the importance of assessment of both specific and broader domains was identified as paramount. With this in mind, creation of a HRQoL instrument for CF incorporating both the generic and specific influences recognized in this study, whilst maintaining the reliability and validity of the two instruments used, is recommended for enhancing the role of patient-reported outcomes in the management of patients with CF. In the meantime, clinicians are encouraged to use the most relevant UKSIP domains (i.e., emotional behavior, social interaction, sleep and rest, work, recreation, and pastimes) and CFQOL domains (i.e., concerns for the future, independent relationships, body image, and career concerns) in their routine clinical setting. This approach of course could be at the expense of compromised reliability and validity of the measures.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
Abbott, J., Conway, S., Etherington, C., Fitzjohn, J., Gee, L., Morton, A., Musson, H., and Webb, A. K. (1999). Enteral nutrition and body satisfaction in CF adults. Neth. J. Med. 54(Suppl.), S75.
Abbott, J., Conway, S. P., Etherington, C., Fitzjohn, J., Gee, L., Morton, A., Musson, H., and Webb, A. K. (2000). Perceived body image and eating behaviour in young adults with cystic fibrosis and their healthy peers. J. Behav. Med. 23, 501–517.
Abbott, J., Hart, A., Havermans, T., Matossian, A., Goldbeck, L., Barreto, C., Bergsten-Brucefors, A., Besier, T., Catastini, P., Lupi, F., and Staab, D. (2011). Measuring health-related quality of life in clinical trials in cystic fibrosis. J. Cyst. Fibros. 10(Suppl. 2), S82–S85.
Bergner, M., Bobbitt, R. A., Carter, W. B., and Gilson, B. S. (1981). The sickness impact profile: development and final revision of a health status measure. Med. Care 19, 787–805.
Bombardier, C., Melfi, C. A., Paul, J., Green, R., Hawker, G., Wright, J., and Coyte, P. (1995). Comparison of a generic and a disease-specific measure of pain and physical function after knee replacement surgery. Med. Care 33(Suppl.), AS131–AS144.
Bowling, A. (2002). Research Methods in Health: Investigating Health and Health Services, 2nd Edn. Buckingham: Open University Press.
Bowling, A. (2005). Measuring Health: A Review of Quality of Life Measurement Scales, 3rd Edn. Buckingham: Open University Press.
Congleton, J., Hodson, M., and Duncan-Skingle, F. (1996). Quality of life in adults with cystic fibrosis. Thorax 51, 936–940.
Debska, G., and Mazurek, H. (2007). Validation of Polish version of CFQoL in patients with cystic fibrosis. Pol. Merkur. Lekarski 23, 340–343.
Dodd, J. D., Barry, S. C., and Gallagher, C. G. (2005). Respiratory factors do not limit maximal symptom-limited exercise in patients with mild cystic fibrosis lung disease. Respir. Physiol. Neurobiol. 152, 176–185.
Esmond, G., Butler, M., and McCormack, A. M. (2006). Comparison of hospital and home intravenous antibiotic therapy in adults with cystic fibrosis. J. Clin. Nurs. 15, 52–60.
Fallowfield, L. (1991). The Quality of Life: The Missing Measurement in Health Care. London: Souvenir Press.
Fitzpatrick, R. (2002). Patient Reported Health Instruments. Available at: http://phi.uhce.ox.ac.uk/ [accessed 10/12/2005].
Fitzpatrick, R., Davey, C., Buxton, M. J., and Jones, D. R. (1998). Evaluating patient-based outcome measures for use in clinical trials. Health Technol. Assess. 2, i–iv, 1–74.
Fletcher, A., Gore, S., Jones, D., Fitzpatrick, R., Spiegelhalter, D., and Cox, D. (1992). Quality of life measures in health care. II: design, analysis, and interpretation. BMJ 305, 1145–1148.
Gee, L., Abbott, J., Conway, S. P., Etherington, C., and Webb, A. K. (2000). Development of a disease specific health related quality of life measure for adults and adolescents with cystic fibrosis. Thorax 55, 946–954.
Gee, L., Abbott, J., Conway, S. P., Etherington, C., and Webb, A. K. (2003). Quality of life in cystic fibrosis: the impact of gender, general health perceptions and disease severity. J. Cyst. Fibros. 2, 206–213.
Gee, L., Abbott, J., Hart, A., Conway, S. P., Etherington, C., and Webb, A. K. (2005). Associations between clinical variables and quality of life in adults with cystic fibrosis. J. Cyst. Fibros. 4, 59–66.
Gobbi, F., Lupi, F., Monti, F., and Miano, A. (2005). Health related quality of life in adults and adolescents with cystic fibrosis: differences of gender, age and disease severity. J. Cyst. Fibros. 4, S108–S118.
Guyatt, G., Feeny, D., and Patrick, D. (1991). Issues in quality-of-life measurement in clinical trials. Control. Clin. Trials 12(Suppl.), 81S–90S.
Harris, A., and Super, M. (1995). Cystic Fibrosis: The Facts, 3rd Edn. New York: Oxford University Press.
Hatziagorou, E., Karagianni, P., Vidalis, A., Bullinger, M., Tsanakas, I., and DISABKIDS Group. (2002). Quality of life in cystic fibrosis. Hippokratia 6(Suppl. 1), 67–71.
Henry, B., Aussage, P., Grosskopf, C., and Goehrs, J. M. (2003). Development of the Cystic Fibrosis Questionnaire (CFQ) for assessing quality of life in pediatric and adult patients. Qual. Life Res. 12, 63–76.
Jong, W. D., Kaptein, A. A., Schans, C. P. V. D., Mannes, G. P. M., Aalderen, W. M. Cv., Grevink, R. G., and Koëter, G. H. (1997a). Quality of life in patients with cystic fibrosis. Pediatr. Pulmonol. 23, 95–100.
Jong, W. D., Schans, C. P. V. D., Mannes, G. P. M., Aalderen, W. M. C. V., Grevink, R. G., and Koter, G. H. (1997b). Relationship between dyspnoea, pulmonary function and exercise capacity in patients with cystic fibrosis. Respir. Med. 91, 41–46.
Kerem, E., Conway, S., Elborn, S., and Heijerman, H. (2005). Standards of care for patients with cystic fibrosis: a European consensus. J. Cyst. Fibros. 4, 7–26.
Littlewood, D. J. (2001). Standards for the Clinical Care of Children and Adults with Cystic Fibrosis in the UK. Kent: Cystic Fibrosis Trust.
Marshall, S., Haywood, K., and Fitzpatrick, R. (2005). Patient Involvement and Collaboration in Shared Decision-Making: A Structured Review to Inform Chronic Disease Management. Report from the Patient-Reported Health Instruments Group to the Department of Health [cited 01/04/06]. Available at: http://phi.uhce.ox.ac.uk/
Monti, F., Lupi, F., Gobbi, F., Agostini, F., Miano, A., Gee, L., and Abbott, J. (2008). Validation of the Italian version of the Cystic Fibrosis Quality of Life Questionnaire (CFQoL), a disease specific measure for adults and adolescents with cystic fibrosis. J. Cyst. Fibros. 7, 116–122.
Patterson, J. M., Wall, M., Berge, J., and Milla, C. (2008). Gender differences in treatment adherence among youth with cystic fibrosis: development of a new questionnaire. J. Cyst. Fibros. 7, 154–164.
Quittner, A. L., Modi, A. C., Wainwright, C., Otto, K., Kirihara, J., and Montgomery, A. B. (2009). Determination of the minimal clinically important difference scores for the Cystic Fibrosis Questionnaire-Revised respiratory symptom scale in two populations of patients with cystic fibrosis and chronic Pseudomonas aeruginosa airway infection. Chest 135, 1610–1618.
Quittner, L., Buu, A., Messer, M. A., Modi, A. C., and Watrous, M. (2005). Development and validation of the cystic fibrosis questionnaire in the United States: a health-related quality-of-life measure for cystic fibrosis. Chest 128, 2347.
Salek, M. S. (1993). Health-related quality of life measurement: a new challenge for pharmacy practitioners. J. Pharm. Pharmacol. 45(Suppl. 1), 387–392.
Salek, M. S. (1997). Measuring the quality of life of patients with arthritis: validation and clinical applications of a self-administered health status instrument (UKSIP). J. Appl. Ther. 1, 205–222.
Salek, M. S., Finlay, A. Y., Luscombe, D. K., Allen, B. R., Berth-Jones, J., Camp, R. D. R., Graham-Brown, R. A. C., Khan, G. K., Marks, R., Motley, R. J., Ross, J. S., and Sowden, J. M. (1993a). Cyclosporin greatly improves the quality of life of adults with severe atopic dermatitis – a randomised double-blind, placebo controlled trial. Br. J. Dermatol. 129, 422–430.
Salek, M. S., Thomas, S., Luscombe, D. K., and Bayer, A. J. (1993b). The impact of memory clinic assessment on the quality of life of patients with cognitive decline: sensitivity of the UK Sickness Impact Profile. Pharm. Weekbl. 15(Suppl. 5), 15.
Salek, M. S., Finlay, A. Y., Lewis, J. J., and Sumner, M. J. (2004). Quality of life improvement in treatment of psoriasis with intermittent short course Cyclosporin (NeoralÒ). Qual. Life Res. 13, 91–95.
Salek, M. S., Griffith, A. R., Spiller, C., Luscombe, D. K., and Bayer, A. J. (1994). Assessment of quality of life in patients with dementia – which measure? Pharm. World Sci. 16 (Suppl. G), 12.
Salek, M. S., Khan, G. K., and Finlay, A. Y. (1996). Questionnaire techniques in assessing acne handicap: reliability and validity study. Qual. Life Res. 5, 131–138.
Salek, M. S., and Luscombe, D. K. (1992). Health-related quality of life assessment: a review. J. Drug Deliv. Sci. Technol. 5, 137–153.
Salek, M. S., Millar, B., Darby, E., Turpin, S., Coles, G. A., and Maggs, C. (2003). Health related quality of life of home dialysis patients and their carers. J. Appl. Ther. Res. 4, 11–18.
Salek, M. S., Turpin, S., Derby, E., Millar, B., and Maggs, C. (1995a). The experience of home dialysis and its impact on the informal carer. Pharm. World Sci. 17(Suppl. J), 18.
Salek, M. S., Turpin, S., Darby, E., Millar, B., and Maggs, C. (1995b). Home dialysis – quality of life of patients and impact on carers. Qual. Life Res. 4, 479–480.
Salek, M. S., Turpin, S., Darby, E., Millar, B., and Maggs, C. (1995c). “Evaluation of the quality of life of home dialysis patients and their carers: general versus renal-specific measure,” in Progress in Clinical Pharmacy. Advanced Activities in Pharmaceutical Care (Netherlands: European Society of Clinical Pharmacy), 123–129.
Salek, M. S., and VandenBurg, M. J. (1988). Measuring the quality of life in angina pectoris. J. Drug Ther. Res. 13, 186–191.
Schraufnagel, D. (1999). Sickness Impact Profile. Available from: http://www.atsqol.org/sick.asp [accessed 10/12/2005].
Schrier, A. C., Dekker, F. W., Kaptein, A. A., and Dijkman, J. H. (1990). Quality of life in elderly patients with chronic nonspecific lung disease seen in family practice. Chest 4, 894–898.
Sheppard, M. N., and Nicholson, A. G. (2002). The pathology of cystic fibrosis. Curr. Diagn. Pathol. 8, 50–59.
Sowden, J. M., Berth-Jones, J., Ross, J. S., Motley, R. J., Marks, R., Finlay, A. Y., Salek, M. S., Graham-Brown, R. A. C., Allen, B. R., and Camp, R. D. R. (1991). Double-blind controlled, crossover study of cyclosporin in adults with severe refractory atopic dermatitis. Lancet 338, 137–140.
Wahl, A. K., Rustoen, T., Hanestad, B. R., Gjengedal, E., and Moum, T. (2005). Living with cystic fibrosis: impact on global quality of life. Heart Lung 34, 324–331.
Walters, S. (2001). Sex differences in weight perception and nutritional behaviour in adults with cystic fibrosis. J. Hum. Nutr. Diet. 14, 83–91.
Walters, S., and Warren, R. (2001). Cystic Fibrosis – A Millennium Survey. Available at: http://www.cfstudy.com/sarah/Report2000_2.pdf [accessed 14/3/06].
WHO. (1999). Services for Adults with Cystic Fibrosis. Report of a Joint WHO/ICF(M)A/IACFA Meeting, 7–8 June, The Hague: WHO.
Wille, N., and Ravens-Sieberer, U. (2006). “Age-appropriateness of the EQ-5D adult and child-friendly version – testing the feasibility, reliability and validity in children and adolescents,” in 23rd Scientific Plenary Meeting of the EuroQol Group in Barcelona, Spain: September 14–16, ed. X. Badía (Barcelona: IMS Health), 217–229.
Wille, N., Ravens-Sieberer, U., and The Child-Friendly Task Force on Behalf of the EuroQol Group. (2006a). “Establishing definitions of the concepts included in CF-EQ-5D – a revision of the ‘definition of EQ-5D concepts’ for adults,” in 23rd Scientific Plenary Meeting of the EuroQol Group in Barcelona, Spain: September 14–16, ed. X. Badía (Barcelona: IMS Health), 233–256.
Wille, N., Ravens-Sieberer, U., and The Child-Friendly Task Force on Behalf of the EuroQol Group. (2006b). “Proposal on a core set of instruments for validation of the international child-friendly EQ-5D,” in 23rd Scientific Plenary Meeting of the EuroQol Group in Barcelona, Spain: September 14–16, ed. X. Badía (Barcelona: IMS Health), 259–276.
Keywords: cystic fibrosis, quality of life, health-related quality of life, cystic fibrosis quality of life questionnaire, United Kingdom sickness impact profile
Citation: Salek MS, Jones S, Rezaie M, Davies C, Mills R and Ketchell RI (2012) Do patient-reported outcomes have a role in the management of patients with cystic fibrosis? Front. Pharmacol. 3:38. doi: 10.3389/fphar.2012.00038
Received: 23 November 2011; Paper pending published: 12 December 2011;
Accepted: 21 February 2012; Published online: 12 March 2012.
Edited by:
Jean-Paul Deslypere, Proclin Therapeutic Research Pte Ltd., SingaporeReviewed by:
Nora Ibargoyen, Basque Office for Health Technology Assessment (Osteba), SpainSuyash Prasad, BioMarin Pharmaceutical, USA
Copyright: © 2012 Salek, Jones, Rezaie, Davies, Mills and Ketchell. This is an open-access article distributed under the terms of the Creative Commons Attribution Non Commercial License, which permits non-commercial use, distribution, and reproduction in other forums, provided the original authors and source are credited.
*Correspondence: M. Sam Salek, Centre for Socioeconomic Research, Cardiff University, Redwood Building, King Edward VII Avenue, Cardiff CF10 3XF, UK. e-mail:c2FsZWtzc0BjZi5hYy51aw==

 S. Jones1
S. Jones1