Abstract
Introduction: The population of young women who suffered from female pattern hair loss (FPHL) or female androgenic alopecia (AGA) is gradually increasing. Platelet-rich plasma is a novel and promising therapeutic method as a nonsurgical treatment for FPHL.
Objective: To summarize different preparation methods of PRP and treatment regimes in FPHL, qualitatively evaluate the current observations, and quantitively analyze the efficacy of PRP in FPHL treatment.
Methods: Six databases, MEDLINE, EMBASE, Web of Science, Cochrane Central Register of Controlled Trials, LILACS, and CNKI, were searched with terms “platelet-rich plasma,” synonyms for AGA and FPHL. Meta-analysis was conducted with enrolled observational studies and randomized controlled trials separately.
Results: We evaluated 636 studies and 12 trials from all searched databases. A total of 42 studies of 1,569 cases, including 776 female participants covering 16 randomized controlled trials and 26 observational trials, were included for qualitative synthesis study and systematic review. PRP showed positive efficacy in treating FPHL in hair density compared to the control groups with odds ratio (OR) 1.61, 95% CI 0.52–2.70, and compared to baseline with OR 1.11, 95% CI 0.86–1.37.
Conclusion: PRP showed excellent efficiency as a novel therapy of FPHL through hair density evaluation. Further studies are needed to define standardized protocols, and large-scale randomized trials still need to be conducted to confirm its efficacy.
Introduction
Female pattern hair loss (FPHL), with an alternative name female androgenic alopecia (AGA), is the most common type of hair loss affecting 6.0% of Chinese women, among which 3.6% were under 40-year-old (Wang et al., 2010). FPHL is characterized by progressive follicular miniaturization and the accompanying conversion of terminal follicles into vellus-like follicles (Trueb, 2002). Patients may present with hair density reduction, hair thinning, and widening of the area especially on the center of the scalp (Olsen, 2001). Those changes may lead to serious psychological impacts (van Zuuren et al., 2012) on one’s self-esteem, interpersonal relationships, and the social status (Cash, 2001). Although multiple nonsurgical therapeutic methods like topical minoxidil, oral finasteride, and low-level laser comb had been introduced to FPHL treatment, more large-scale–randomized controlled trials still need to be investigated to confirm their efficacy (van Zuuren et al., 2016). Despite decreased number of more actively proliferating progenitor cells, the current studies have presented an unaltered number of hair follicle stem cells in a hair loss scalp (Garza et al., 2011). Hence the application of autologous stem cells including autologous micro-grafts enriched of human follicle cells (HF-MSCs) as well as platelet-rich plasma has been explored and gradually been introduced to clinical use (Gentile et al., 2019).
Platelet-rich plasma (PRP) is a preparation of an enriched platelet autologous plasma in which the concentration of platelet is above normal contained in the whole blood (Wu et al., 2016). Platelets are able to secrete growth factors, adhesion molecules, and chemokines. After being activated, those effective factors interact with the local environment and promote cell differentiation, proliferation, and regeneration (Weibrich et al., 2002; Davì and Patrono, 2007; Sánchez-González et al., 2012). Published data also highlighted that PRP contains major growth factors, including basic fibroblast growth factor (bFCF), platelet-derived growth factor (PDGF), vascular endothelial growth factor (VEGF), epidermal growth factor (EGF), transforming growth factor-β (TGF-β), and insulin-like growth factor-1 (IGF-1) (Cervelli et al., 2012). In the past decade, PRP has been studied and widely used in alopecia, acne scarring, skin rejuvenation, chronic wounds, and vitiligo (Hesseler and Shyam, 2019). Comparing with traditional nonsurgical therapies and the surgical approaches such as hair transplantation, PRP is believed as a promising treatment of AGA with lower cost and fewer adverse effects. However, only a limited number of publications focus on the utilization of PRP in AGA treatment, and few studies or reviews about the application of PRP in FPHL have been performed.
In this study, we summarized different PRP preparation methods, reviewed the various treatment regime in FPHL that are reported in the previous studies, qualitatively evaluated the current observations, and quantitively analyzed the efficacy of PRP in FPHL treatment. This work may offer a reference to the clinical workers and associated FPHL patients.
Methods
This meta-analysis was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analysis (http://www.prisma-statement.org) and Meta-analysis Of Observation Studies in Epidemiology (MOOSE) statement (Stroup et al., 2000).
Literature Screening
Two investigators independently conducted a systematic search of studies published before October 29, 2020, using the databases MEDLINE via PubMed, embase, Web of Science via Ovid, Cochrane Central Register of Controlled Trials (CENTRAL), LILACS, and CNKI. The strategy of search terms was composed of at least one term from two search blocks: the term “platelet-rich plasma” and synonyms for androgenic alopecia (AGA) and female pattern hair loss (FPHL). All synonyms were found based on MESH and ENTRÉE (Supplementary Material S1).
Study Selection
Original studies include observational studies (i.e., case series, cross-sectional, case-control, and cohort) and randomized trials of platelet-rich plasma (PRP), and AGA in woman patients in English, German, Swedish, Norwegian, Spanish, Danish, Turkish, and Chinese were all eligible for inclusion. Exclusion criteria were reviews, studies only included male patients, abstracts, unpublished studies, and lack of raw data. Conference reports were also excluded for insufficient details for analysis. Two reviewers screened the titles and abstracts of the identified studies. If the information provided in the abstracts was not sufficient to access the eligibility, a full-text evaluation was conducted. All observational studies and randomized trials will be recruited for qualitative analysis, and only randomized controlled trials will be analyzed quantitively and enrolled in the meta-analysis in this work. Two authors also evaluated the quality of the included studies independently. Any disagreement was resolved through discussion or decided by a third person.
Data Extraction
Two reviewers completed the data extraction work independently with the following information: author, year of publication, journal name, country, methods of PRP preparation, treatment regime, and the number of cases/controls (total, exposed, nonexposed). In case of disagreement, a third person conducted a further assessment.
Endpoint Definition
The efficacy of PRP was evaluated by an increase in hair density, increment of hair count, improvement in the hair-pull test, satisfaction of patients from the questionaries, and changes of hair thickness compared with photos taken before and after the treatment sections. Given that various test methods were taken through the studies we included, only the most widely used methods would be set at the endpoints for all-pooled studies.
To access the safety of PRP, all-side effects, including local injection pain, headache, increasing scalp sensitivity, and any allergic effects, would be recorded.
Data Analysis
We conducted the meta-analysis with Stata statistical software 15.0 (Metrika Consulting, Stockholm, Sweden) using the “metan” command. A random-effect analysis with the method of Dorsmanin and Laird (Higgins et al., 2020) was taken, given the considerable heterogeneity. We chose 95% confidence intervals (CI) and I (Trueb, 2002) to access statistical heterogeneity (Higgins et al., 2003; Cook and Reed, 2015): 30–60% as moderate heterogeneity, 50–90% substantial heterogeneity, greater than 75% considerable heterogeneity (Higgins et al., 2020). Funnel plots and Egger tests were performed for publication bias assessment with asymmetry in the funnel plot and a p-value that was less than 0.1. Eggers’s test would overrule visual inspection if results diverged between the two methods above.
Clinical heterogeneity was measured by stratification, for example, subgroup analysis of the study types, i.e., randomized controlled trials and observational trials. The observational trials were further stratified into case series, cohort studies, cross-sessional studies, and case-control studies. We also conducted a subgroup analysis based on different endpoint definitions, e.g., hair density, hair count, and increment in hair density. Subgroup analysis with different PRP preparation methods and PRP concentrations were only performed with sufficient data.
The quality of each study was evaluated based on Joanna Briggs Institute Critical Appraisal tools for case-series studies (Munn et al., 2020), Newcastle-Ottawa Scale (Stang, 2010) for cohort studies and case-control studies, and Cochrane Risk of Bias Tool for Randomized Controlled Trials (Higgins et al., 2011). Three groups of study quality were defined: high, middle, and low. Rovis was used for the presentation of the quality of each study (McGuinness and Higgins, 2020).
Results
Literature Search
We found 636 literature and 12 trials from databases and three additional references by reference screening. Six hundred and three studies were excluded for reasons including duplicated, male patients only, and not in human subjects (Figure 1).
FIGURE 1
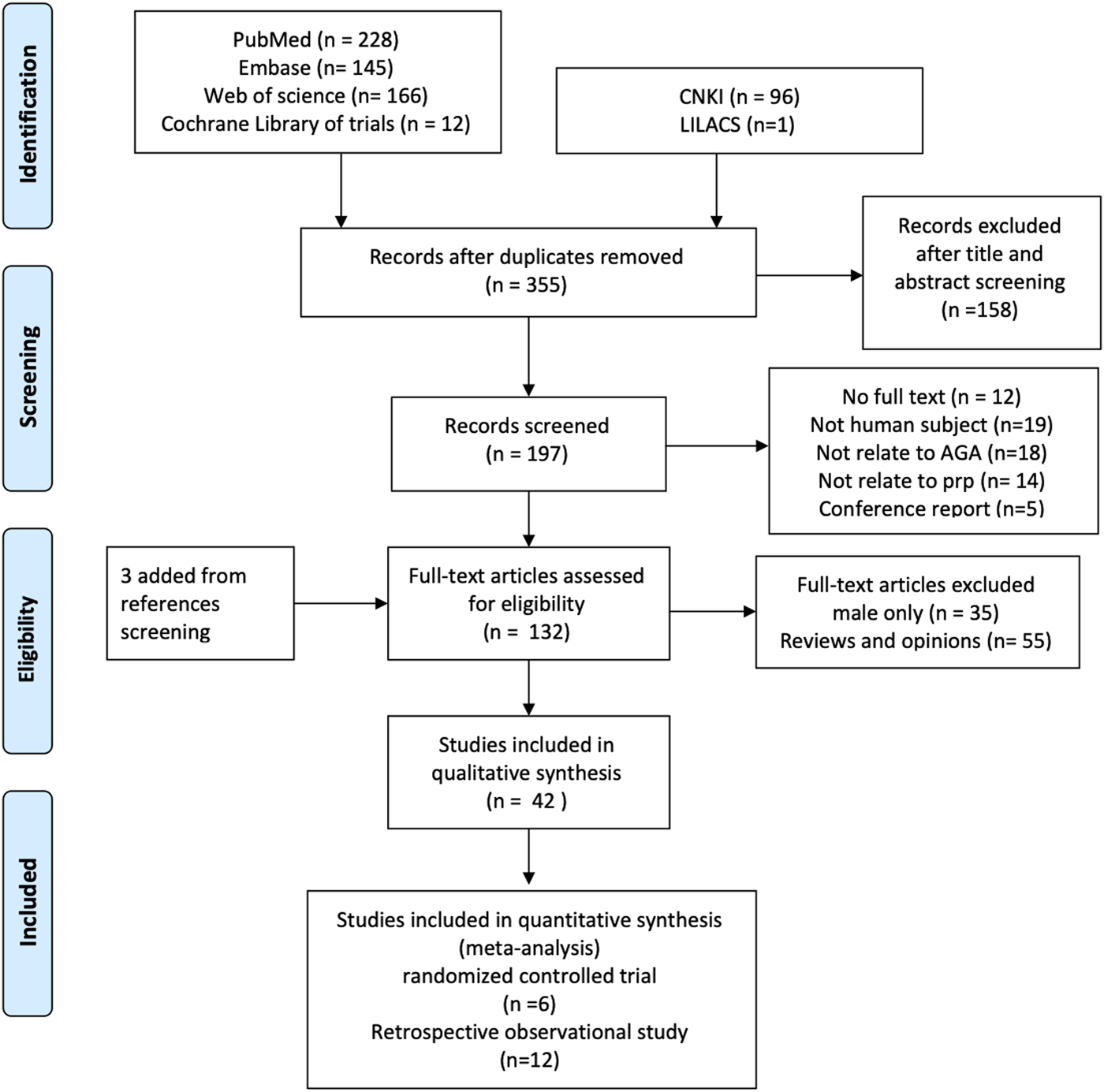
The process of literature screening.
Although only the PRP effect on a female would be evaluated in this study and given only a few pure women studies were found, studies involved in both female and male patients would also be pooled in this work. A total of 42 studies, 1,569 cases including 776 female participants covering 16 randomized controlled trials and 26 observational trials, were included for qualitative synthesis study and systematic review.
Study Characters
Studies with various group settings were enrolled (Table 1). Eight out of 16 randomized controlled trials investigated the effect of PRP comparing with placebo, in which 6 of which were done in half-headed comparation. Four studies evaluated the differences of the effect of PRP combination therapy with topical minoxidil, oral finasteride, laser, and polydeoxyribonucleotide (PDRN), while the other four accessed the different efficacy from treating strategy, i.e., PRP concentration, centrifugation protocol, treatment sessions, and treatment doses. The risk of bias of all-eight–randomized placebo-controlled trials was graded by two reviewers (Figures 2, 3).
TABLE 1
| Author/year | Country | Trial characters | Subject characters | Objectives |
|---|---|---|---|---|
| Abaroa 2016 | Mexico | RCT | 10 males and 6 females | Efficacy of APRP |
| Alves 2016 | Spain | RCT | 23 males and 12 females | Efficacy of APRP with half-headed |
| Alves 2018 | Spain | RCT | 13 males and 11 females | Efficacy of APRP combines with topical minoxidil and oral finasteride, half-headed |
| Anitua 2017 | Spain | UCT | 13 males and 6 females | Efficacy of APRP |
| Bruce 2019 | USA | RCT | 20 females | Efficacy of APRP with half-headed |
| Butt G 2020 | Pakistan | RCT | 17 males and 5 females | Efficacy of APRP with SVF-PRP |
| Butt G 2019 | Pakistan | UCT | 20 males and 10 females | Efficacy of APRP with minoxidil |
| Dina 2019 | USA | Case report | 2 females | Efficacy of APRP |
| Dubin 2020 | USA | RCT | 30 females | Efficacy of APRP with placebo |
| El-husseiny 2020 | Egypt | UCT | 13 males and 10 females | Efficacy of APRP in double-spin and single-spin, half-headed |
| Garg 2017 | India | UCT | 65 males and 50 females | Efficacy of APRP |
| Gentile a 2020 | Italy | UCT | 15 females | Efficacy of APRP combined with microneedling technique, low-level laser therapy |
| Gentile b 2020 | Italy | UCT | 63 males and 27 females | Efficacy of APRP activated APRP compared with nonactivated PRP |
| Gentile 2018 | Italy | UCT | 18 males and 5 females | Efficacy of APRP |
| Gkini 2014 | Greece | UCT | 20 males and 2 females | Efficacy of APRP |
| Hausauer 2018 | USA | RCT | 29 males and 10 females | Efficacy of APRP with half-headed |
| Ho 2020 | USA | UCT | 24 females | Efficacy of APRP with minoxidil |
| Juhasz 2020 | USA | UCT | 74 females and 30 males | Efficacy of APRP |
| Kang 2014 | South Korea | RCT | 15 males and 11 females | Efficacy of APRP with placebo |
| Laird 2018 | Spain | Patients survey | 41 females | Efficacy of APRP |
| Lee 2015 | South Korea | RCT | 40 females | Efficacy of APRP plus PDNR and PDNR monotherapy |
| Makki 2020 | Egypt | UCT | 13 males and 37 females | Efficacy of APRP |
| Paththinige 2018 | Siri lanka | UCT | 27 males and 1 females | Efficacy of APRP |
| Puig 2016 | USA | RCT | 16 females | Efficacy of APRP with placebo |
| Qu 2019 | China | UCT | 51 males and 37 females | Efficacy of APRP |
| Rossano 2017 | Italy | UCT | 25 males and 16 females | Efficacy of APRP |
| Schiavone 2018 | Italy | UCT | 102 males and 66 females | Efficacy of APRP |
| Schiavone 2014 | Italy | UCT | 42 males and 22 females | Efficacy of APRP |
| Sclafani 2014 | USA | UCT | 9 males and 6 females | Efficacy of APRP |
| Shapiro 2020 | USA | RCT | 18 males and 17 females | Efficacy of APRP with half-headed |
| Siah 2020 | United Kingdom | RCT | 1 males and 9 females | Efficacy of APRP |
| Singhal 2015 | India | UCT | 1 males and 2 females | Efficacy of APRP |
| Starace 2019 | Italy | UCT | 10 females | Efficacy of APRP |
| Takikawa 2011 | Japan | RCT | 16 males and 10 females | Efficacy of APRP with D/P MPS |
| Tan 2019 | Singapore | RCT | 33 males and 22 females | Efficacy of APRP with half-headed |
| Tawfik 2018 | Egypt | RCT | 13 females | Efficacy of APRP with placebo |
| Zolfaghari 2020 | Iran | UCT | 4 males and 9 females | Efficacy of APRP |
| Zhang 2018 | China | RCT | 28 males and 32 females | Efficacy of APRP with placebo |
| Navarro 2015 | Spain | UCT | 50 males and 50 females | Efficacy of APRP |
| DeVasconcelos 2015 | Brazil | UCT | 9 males and 7 women | Efficacy of APRP |
| Yang 2020 | China | RCT | 27 males and 5 females | Efficacy of APRP with minoxidil |
| Zhang 2020 | China | RCT | 36 males and 34 females | Efficacy of APRP with lacer and placebo |
The study characters.
RCT, randomized controlled trials; UCT, uncontrolled clinical trials; APRP, autogenous platelet-rich plasma; SVF, stromal vascular fraction; PDNR, polydeoxyribonucleotide; D/P MPs, dalteparin and prota-mine microparticles; USA, the United States of America.
FIGURE 2
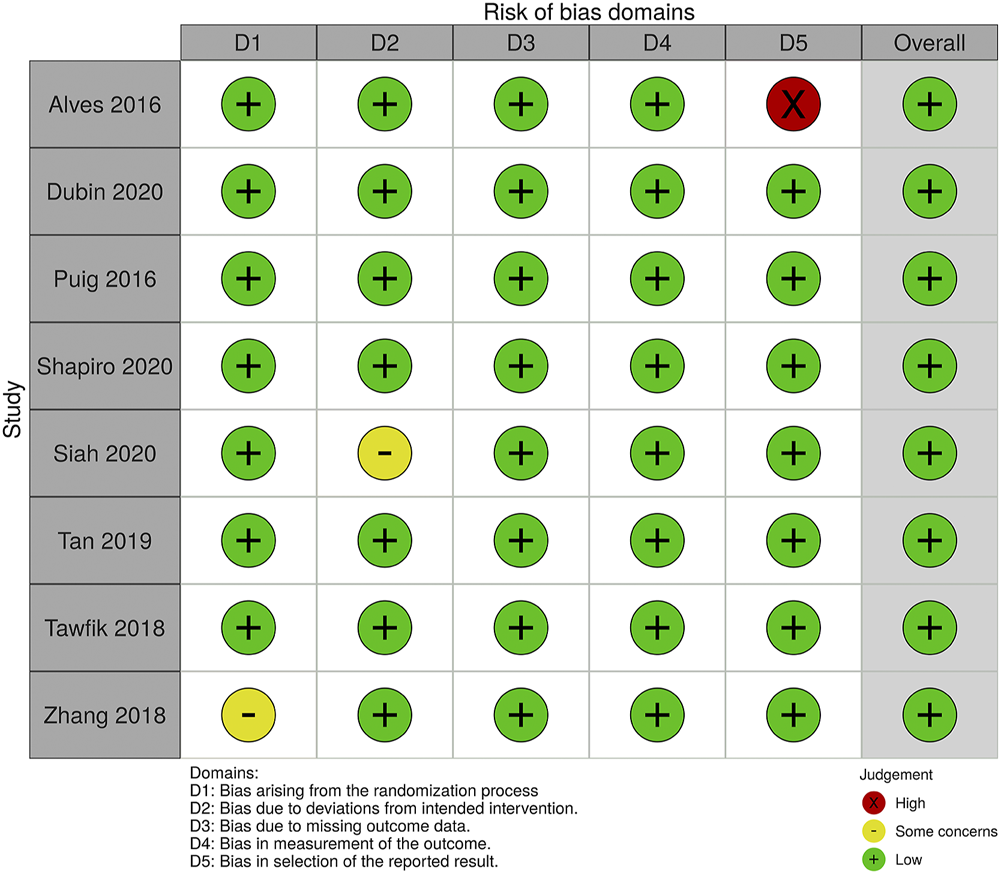
The Risk of Bias evaluation of randomized trials.
FIGURE 3
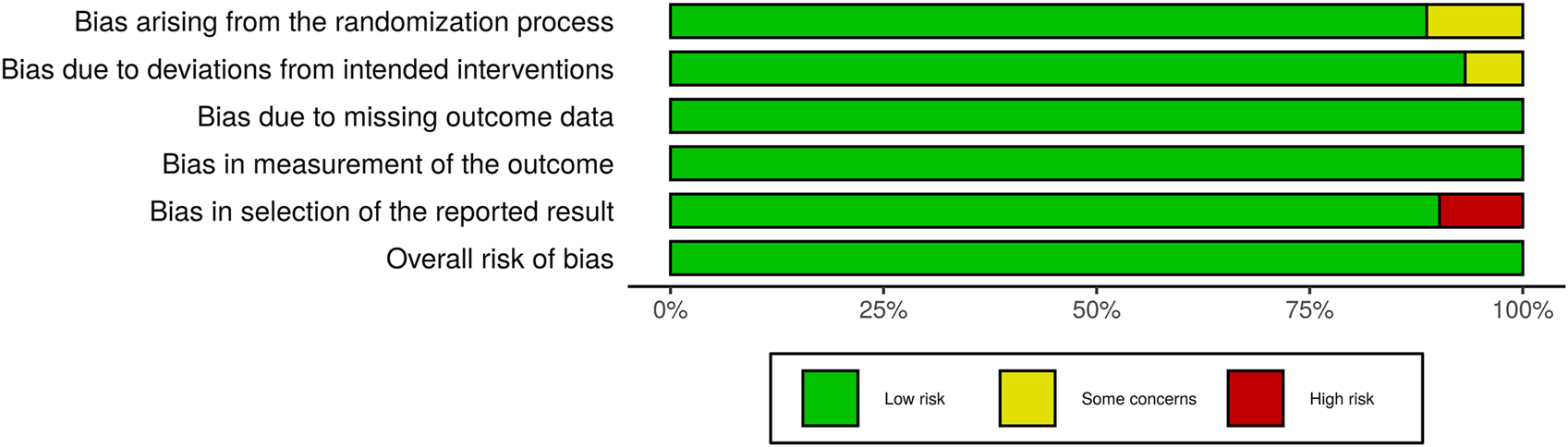
Summary of Risk of Bias in Randomized Controlled Trials.
Observation studies including 21 case series, 2 case reports, one patient survey, one treatment protocol, and one treatment experience were enrolled, while one case series containing only one female patient was abandoned. Quality of the total 20 case series was accessed, three of which were excluded for high risk after evaluation (Figure 4).
FIGURE 4

Risks of Bias evaluation of case series.
Study Subjects
Among all 42 studies, we only found seven studies that recruited only female participants. The mean age of the total-enrolled patients was above 18 years old and between 25 and 35 years old. Most female patients had a history of AGA for at least 3–5 years with type I to III FPHL on Ludwig scale (Harrison, 2017) or 1–5 on Sinclair score system (Kasprzak et al., 2019). Patients who had previous hair transplantation, a drug-taking history that could cause hair loss or any inflammations, scars, or erythema on the scale were excluded. Laboratory tests, including hemoglobin, platelet count, serum ferritin, liver function, thyroid function, and female hormone profile, were checked for excluding any other autoimmune or systematic diseases that could cause hair loss. Acute or chronic infections should also be tested before using PRP.
Platelet-Rich Plasma Preparation
The methods of PRP preparation vary in studies (Table 2). PRP kits were commonly used from a different company and centrifugation protocol. The choices of activators and anticoagulation vary depending on PRP kits and study purposes. In all-enrolled studies, calcium gluconate in a 10:1 ratio and sodium citrate were mainly added as activators and anticoagulants, respectively. Moreover, platelet concentration differs from 1.5 times to seven-fold as whole blood according to the PRP preparation protocols.
TABLE 2
| Author/year | Blood drawn | Centrifugation protocol | Activators | Anticoagulant | Platelet concentration |
|---|---|---|---|---|---|
| Abaroa 2016 | 20–30 ml | Double-spin method, 20–30 ml of cititrated blood, centrifuged at 1,800 rpm for 10 min, abandon buffy coat, 3,000 rpm for 10 min | Calcium gluconate in a 10:1 ratio | 6.8% sodium citrate | 200–400% over basal blood |
| Alves 2016 | 18 ml | 460 g for 8 min | 0.15 ml 10% calcium chloride immediately before application | 2 ml of 3.8% sodium citrate | Approcimately 3 times higher than whole blood |
| Alves 2018 | 18 ml | 460 g for 8 min | PRP kits | 2 ml of 3.8% sodium citrate | Approcimately 3 times higher than whole blood |
| Anitua 2017 | 18 ml | 580 g for 8 min | PRGF activbitor | 9 ml tubes 3.8% sodium citrate | |
| Bruce 2019 | 60 ml | Dual spin centrifugation, 1,500 rpm for 10 min, removal of red cell layer, additional centrifugation at 3,500 rpm for 10 min, G force 684, and radius of 50 | 8 ml citrate dextrose solution A | ||
| Butt G 2020 | 9 ml | 650 g for 10 min | PRP kit (tray life tube gel) comprising a mixture of polymers that sepatated plasma and sodium citrate | ||
| Butt G 2019 | 9 ml | 1,000 rpm for 10 min | PRP kit (tray life tube gel) comprising a mixture of polymers that sepatated plasma and sodium citrate | ||
| Dina 2019 | 9 ml | 1,100 g for 6 min | 0.5 m calcium chloride | Sodium citrate | |
| Dubin 2020 | 22 ml | Eclipse PRP system 3500 revolutions per minute for 10 min | |||
| El-husseiny 2020 | 10 ml | Double-spin: 800D at 1,12 g (100 rpm) for 10 min, plasma for 448 g units (2,000 rpm) for 10 min, single-spin: 1,372 g units (3,500 rpm) for 10 min | 1 ml calcium gluconate (1:9) | Tri-sodium citrate | |
| Garg 2017 | 13.5 ml | REMI [centrifuged for 4 min at 3,000 rpm (revolutions per minute)] | Y cell bio kit | 1.5 ml of ACD-A solution | It gives 5–7 times the concentration of the baseline platelet count |
| Gentile a 2020 | 55 ml | Three different kit: C-Punt, i-stem, MAG-18 | |||
| Gentile b 2020 | 55 ml or 18 ml for APRP, ANAPRP for 18 ml | A-PRP:C-PunT prepration system, 1,200 rpm per 10 min, followed 1,200 rpm for 5 min, MAG-18 PRP kit, 3,000 rpm for 6 min, then 3,000 rpm for 2 min, AA-PRP: Cascade-selphyl, 1,100 g per 10 min | Calcium gluconate in a 10:1 ratio | Sodium citrate (ACD) | |
| Gkini 2014 | 16 ml | RegenKit-BCT-3, 1,500 g for 5 min | Calcium gluconate | Dodium citrate solution | 5.8 times as whole blood |
| Hausauer 2018 | 22 ml | 3,500 revolutions per minute for 10 min | EclipsePRP kits | 4 to 6 times the platelet concentration of whole blood | |
| Ho 2020 | 8 ml | 1,500 g 5 min | RegenKit-BCT | 1.5 times, total 5 ml | |
| Juhasz 2020 | 8 or 16 ml | 1,500 g 5 min | regenkit-BCT | 1.6 times, total 5 ml | |
| Kang 2014 | 60 ml | SmartPReP | Sodium citrate solution | ||
| Laird 2018 | 8 ml | Double-spin cycle | 20 mg of acell MatriStem micro matrix + 1cc of 10% calcium gluconate | Dextrose solution A | |
| Lee 2015 | 60 ml | SmartPReP | 4% sodium citrate solution | ||
| Makki 2020 | 10 ml | 400 g for 10 min, upper part for 800 g 10 min | |||
| Paththinige 2018 | 18 ml | Male 3,000 rpm 4 min, female 3,000 rpm for 3 min; fastened PRP kit was for 3,200 rpm, 6 min | Calcium gluconate in a 10:1 ratio | Sodium citrate | |
| Puig 2016 | 60 ml | 2.75 to 3.4 times | |||
| Qu 2019 | 40 ml | 3,300 rpm for 4 min, then 3,200 rpm for 3 min | Tricell kit | ||
| Rossano 2017 | 16 ml | 1,500 g 5 min | RegenKit-BCT-3, calcium gluconate per 0.9 ml of PRP 1:10 ratio | Sodium citrate | Mean platelet counts were1,9*105 in whole blood and 5.5*105 platelets/µl in PRP, 5 TIMES, 0.1ml/m2 |
| Schiavone 2018 | 60–120 ml | Soft-spin, 1,500r 5 min | GLO PRP kit, and C-PunT | ACD-A | 4.5 fold |
| Sclafani 2014 | 18 ml | 1,100 g for 6 min | Calcium chloride | Thixotropic separator gel | |
| Shapiro 2020 | 10 ml | 1,500 g 5 min | Thicotropic gel for separating | ||
| Siah 2020 | 20 ml | 300 g 18C, 5 min, 700 g for 17 min | |||
| Starace 2019 | 10 ml | 2,500 rpm for 10 min | ACD-A acid-citrate-dextrose | ||
| Takikawa 2011 | 15 ml | 1,700 r, 15 min, then 3,000 rpm in 5 min | 0.2% sodium citrate | Platelet concentration in PRP (88.2 7 21.7 ? 104/1 mL, n = 15 persons) was significantly higher than that in whole blood (14.4 7 3.8 ? 104/1 mL, n = 15 persons) | |
| Tawfik 2018 | 10 ml | 1,200 g for 15 min, then 200 g for 10 min | 1:9 ratio, 0.1 ml calcium gluconate per 0.9 ml of PRP | Sodium citrate | |
| Zolfaghari 2020 | 40 ml | Women: 1,400 r 14 m, men 1,600 r, then 4 min at 4,000 rpm | PRGF activbitor | 3.8% sodium citrate | |
| Zhang 2018 | 16 ml | 1,500 G, 3,000 r, 10 min | |||
| Navarro 2015 | 18 ml | 580 g for 8 min | PRGF activbitor | 3.8% sodium citrate | |
| Zhang 2020 | 30 ml | 3,000 r/min, 10 min | 10% calciun cholirade | Sodium citrate | 6.34 ± 0.4 fold |
Preparation of PRP in studies.
Treatment Strategy
Despite all studies performed intradermal injections on the local alopecia area for AGA treatment, the whole procedure diverged (Table 3). 70% alcohol, spirit, or 0.1% octenidine hydrochloride spray were used for cleaning the local skin, and local anesthesia with the help of 2% lidocaine with 0.001% epinephrine were commonly used. two studies used anesthesia cream, one used soft head message, and one conducted cold air anesthesia before injections.
TABLE 3
| Author/year | PRP-injected (total, each injection) injection details | Treatment session arrangement | Anesthesia | |
|---|---|---|---|---|
| Abaroa 2016 | Total 3–5 ml to right half of the scales, intradermally, each puncture was made in spaces of 1cm, apply 0.2 ml in each puncture for 15 to 20 punctures | Twice a week for three weeks | At 72-h intervals over a three-week period | Not mentioned |
| Alves 2016 | 4 selected areas with 30-G needle, about 0.15 ml/cm2 | 1 month from each other for 3 months | None | |
| Alves 2018 | 4 selected areas with 30-G needle, about 0.15 ml/cm2 | 1 month from each other for 3 months | None | |
| Anitua 2017 | 3–4 cm3 of freshly activated RPGF injected to the hair-depleted area, with a 30-G needle | Every months for 3 sessions, 2 additional reminer injection doses were administered at months 4 and 7 after the start point | Not mentioned | |
| Bruce 2019 | The patient’s scalp was cleaned with 70% alcohol, and a grid was marked with dots approximately 1–2 cm apart, covering the affected area. A total of 5 ml of PRP was injected using a 30-gauge needle, approximately 0.1 ml per injection site and 50 injection points in the grid. The needle was angled close to 90°, targeting the transition between the dermis and subcutaneous layer | Cold air | ||
| Butt G 2020 | 3 ml PRP at 0.5 interval intradermally with insulin syringes | 2 sessions each after 4 weeks | Local anesthetic gel, 1 h before giving injections | |
| Butt G 2019 | Cleaned with spirit, 1 cm distance using nappage technique for 1.5–2.5 mm deep in the skin | Repeated after 4 weeks | Local anesthetic gel, 1 h before giving injections | |
| Dina 2019 | 4–5 ml of PRP to the scalp vertex and temporal hairline | 3 treatments, spaced 4 weeks apart | Not mentioned | |
| Dubin 2020 | 4.0 ml of PRP was injected 3–8 mm below the skin surface into the subdermal plane via a 30-gauge 0.5-inch needle. Each injection was comprised of 0.2 ml of PRP and spaced 1–2 cm apart | At week 0, 4, 8 | Not mentioned | |
| El-husseiny 2020 | Multiple small ingections in a linear pattern 1 cm apart over the right half of the scalp, intradermally | 3 treatments | 3 weeks apart | Not mentioned |
| Gentile a 2020 | 0.2 ml*cm2 with a 30-G needle and10 ml luer lock syringe for 5 mm deep | Not mentioned | ||
| Gentile b 2020 | 0.2 ml*cm2 with 0.5 mm sterilemicro-needling procedure | Repeated every 15 days for three times | None | |
| Gkini 2014 | Cleaned using 0.1% octenidine hydrochloride spray, PRP for 0.05–0.1 ml/cm2 with a 27-G needle for 1.5–2.5 mm deep | 3 treatment sessions with an interval of 3 weeks, a booster in 6 m | Local anethesia | |
| Hausauer 2018 | 0.2–0.5 ml, half-inch needle every 2–3 cm at balding areas with a 32-gauge needle, half-inch needle deep subdermally | group1: Received 4 total injections, the first 3 at monthly intervals and the last 3 months later, group2: Received 2 total injections, one at baseline and one at 3 months | 23% topical lidocaine or 7% tetracaine ointment | |
| Ho 2020 | 0.1 ml PRP and spaced 1 cm apart for approximately 1 cm deep | Monthly for four additional months for four followed by maintenance injections every 3–6 months | Not mentioned | |
| Juhasz 2020 | 0.1 ml PRP and spaced 1 cm apart for approximately 1 cm deep | 2 PRP sessions completed at 4–6 weeks intervals | ||
| Kang 2014 | Cleaned with 70% alcohol and interfollicular injection | 3 months interval | 2% lidocaine with 0.001% epinephrine 3–5 ml injection | |
| Laird 2018 | Using microneedle on the treatment area with derma rollar for a 22-G gauge needle deep dermis/upper cutis | 2% lidocaine with 0.001% epinephrine 3–5 ml injection | ||
| Lee 2015 | Cleaned with 70% alcohol, intra-perifollicular injection | PDNR weekly for 12 weeks | 2% lidocaine with 0.001% epinephrine 3–5 ml injection | |
| Makki 2020 | Cleaned and sterilized with spirit and povidone-iodine, 1 cm distance using nappage technique with the insulin syringe | 4 times at 4 weeks interval | Topical anesthesia | |
| Paththinige 2018 | Loaded with 1 ml syringes before injection, cleaned with 70% alcohol, nappage technique (injections 1 cm apart, in a linear manner). 1.5–2.5 mm deep, with a 25-G needle | 4 treatment sessions with initial three treatments in an interval of 3 weeks and a booster session performed at 14 weeks from the baseline treatment (2 months after the 3rd treatment session) | Local anethesia cream | |
| Puig 2016 | Anesthetized using a ring block method:achieved by injecting a 50:50 mixture of 2% lidocaine and 0.5% bupivicaine | |||
| Qu 2019 | With a 30-G needle | With a 1-month interval for 6 consecutive sessions | Not mentioned | |
| Rossano 2017 | Clean with 0.1% citidine chloride spray, injected with a 27-G needle and 1 ml syringes for 1.5–2.5 mm deep in the skin | 4sessions of PRP application each 40–60 days | Not mentioned | |
| Schiavone 2018 | 1–2 mm deep | 2 injection regimen with a 3-month interval between the 2 interventions | Not mentioned | |
| Schiavone 2014 | 4 injections would be on the vertices of a square with sides = 1 cm. The amount injected, per each injection, was approximately 0.2–0.3 ml. With a 22–24G needle for 1 mm deep | 2 injection regimen with a 3-month interval between the 2 interventions | Not mentioned | |
| Sclafani 2014 | 0.1 ml per injection spot, separate by 5–8 mm intradermally | Every 4weeks for 3 treatment sessions | Not mentioned | |
| Shapiro 2020 | 3–4 mm deep for 0.1–0.2 ml per injection/cm2 | 1-month intervals for 3 month with a final follow-up visit three months after the last treatment | Not mentioned | |
| Siah 2020 | 3 cm2 area with a volume of 0.1 ml/cm2 | 5 injections with a 2-week interval | Not mentioned | |
| Singhal 2015 | 8–12 ml of total volume | 3 months at an interval of 2–3 weeks. The treatment is repeated every 2 weeks for four sessions | Not mentioned | |
| Starace 2019 | 1 ml per injection point, with a 25G needle | Every 2 weeks for a total of 4 sessions | Anethesia cream | |
| Takikawa 2011 | A 25G needle | 5 injections at 0,2,4,6,9 weeks | Not mentioned | |
| Tan 2019 | 4 treatment sessions total 3 weeks apart for 9 weeks | Not mentioned | ||
| Zhang 2018 | 4 injections would be on the vertices of a square with sides = 1 cm. The amount injected, per each injection, was approximately 0.2–0.3 ml | Once a month, for 4 times | Not mentioned | |
| Navarro 2015 | A 30-G needle for total 2.5–3 ml, with a mesotherapy gun | 2 treatment sessions with 1 month of time period between them | Soft head message | |
| DeVasconcelos 2015 | Intradermally at a dose of 0.2 ml of on each point of the affexted region, with spaces of approximately 2 cm between these points with a 26G 1/2 needle | 3 injextions at 21 intervals | Not mentioned | |
| Yang 2020 | Inject with a 3 mm interval | |||
| Zhang 2020 | 35 spot for 0.1 ml each spot | Once a month for 4times | Not mentioned | |
Treatment protocols.
The majority of studies selected 22- to 30-G gauge needles with insulin syringes to perform the procedure, while sterilemicro-needling and DHN1 mesotherapy gun were also used in independent studies. Nappage technique (Modarressi, 2013) was taken by most studies with 1 to 3 cm distance between each injection point. 1cc PRP was injected within each grided injection point. The depth of intradermal injections was approximately 1.5–2.5 mm deep, but 0.5 mm deep for the sterilemicro-needling procedure. Intrafollicular injections and intra-perifollicular injections were also carried in several studies.
For treatment schedules, normally 3–5 treatment sessions with 4–6 intervals were performed. And the time of follow-up was commonly from 12 weeks to 9 months depending on the growth period of hairs (Lee et al., 2020).
Outcome Evaluation Methods and Adverse Effects
In addition to Ludwig and Sinclair scale, endpoints evaluation methods included biopsy with ki-67 immunochemistry stain, photographic evaluation, hair density, hair count, global photographs, and phototrichogram analysis (Ueki et al., 2003), physician global assessment score (PhGAS), patient global assessment score (PaGAS), and pull test. The satisfaction questionnaires and scales were taken from the perspective of patients and other observers were also used to evaluate the efficacy of PRP in some of the recruited studies (Table 4).
TABLE 4
| Author/year | Outcomes result and measurement | Adverse events | Outcomes on molecular levels |
|---|---|---|---|
| Abaroa 2016 | Biopsy and photographs at week 12 | Not mentioned | Not mentioned |
| Alves 2016 | Global photographs and phototrichogram analysis (vertex, grontal, and occipital) | Not mentioned | Not mentioned |
| Alves 2018 | Global photographs and phototrichogram analysis (vertex, grontal, and occipital) | Not mentioned | Not mentioned |
| Anitua 2017 | Phototrichogram analysis using the TrichoScope ASG and the TrichoSciencePro hair and scalp diagnostic software, for women, mild scalp and crown region, hair density, hair diameterm terminal/vellus-like hair ratio and thin/regular/thick hair shafts among terminal follicles, and standardized global macrophotographs, self-assessment questionnaire and rated satisfaction following a likert scale, biosy from 6 volunteers, with ki-67 | Not mentioned | Measuring TGFβ1, PDGF-AB, EGF, VEGF, TSP-1 and Ang-1 increasing by ELISA |
| Bruce 2019 | TrichoScan analysis and TrichoScan digital image analysis, hair count, hair density, cumulative thickness. QOL questionaire | weeks 4,8 and 12 of PRP treatment | Not mentioned |
| Butt G 2020 | Macroscopic photographs, pull test, trichoscopic photomicrographs, physician glob asseement score (PhGAS), patient global assessment score (PaGAS), hair density | Not mentioned | Not mentioned |
| Butt G 2019 | Macroscopic photographs, pull test, trichoscopic photomicrographs, physician glob asseement score (PhGAS), patient global assessment score (PaGAS) | Not mentioned | Not mentioned |
| Dubin 2020 | Hair density, caliber and blinded global photographic assessment | In PRP, mild headache 50%, scalp tightness 50%, swelling29, redness 14%, post-injection bleeding 7%, and tingling 7% | Not mentioned |
| El-husseiny 2020 | Patients' global photographs, Lugwig's and Sinclair's grading, questionaire of satisfacition, improvement in hair density, hair quality and pain injection and infection | 11 headache, all reported mild pain during injections | No significant difference was found between the median concentrations (ng/L) of VEGF in both nonactivated and activated single- and double-spin prepared PRP measuring by ELISA |
| Garg 2017 | Parameters which were observed on video-microscopy are hair count, diameter of hair, change in texture, multiplicity of hair, perifollicular halo, perifollicular pigmentation, increase in telogen hair and increase in vellus hair count | Not mentioned | Not mentioned |
| Gentile a 2020 | Photography, physician's and patient's global assessment scale, and standardized phototrichograms | Not mentioned | Not mentioned |
| Gentile b 2020 | Hair density by trichoscan, bioposi on anti-ki-67, anti-CD31 | Not mentioned | Not mentioned |
| Gkini 2014 | Hair density and patients' satisfaction, hair-pull test, dermoscopic photomicrographs, macroscopic photographs and a satisfaction questionnaire | 25% mild pain after application, 60% scalo sensitivity | Not mentioned |
| Hausauer 2018 | Folliscope and global photography, hair count, hair density, shaft caliber, Norwood-Hamilton or ludwig scale were determined, patient's satisfiaction | Not mentioned | Not mentioned |
| Ho 2020 | Hair density and diameter using folliscope | Not mentioned | Not mentioned |
| Juhasz 2020 | Hair density | Not mentioned | Not mentioned |
| Kang 2014 | Phototrichogram scalp on hair numbers, thickness with follioscope | Not mentioned | Flow cytometry was performed using PRP preparation and an equal amount of peripheral blood in two healthy volunteers. One participant presented 6.7 cells ⁄ lL of CD34 + cells in peripheral blood, whereas those in the autologous PRP preparation were 31.1 cells ⁄ lL |
| Laird 2018 | Satisfaction | Not mentioned | Not mentioned |
| Lee 2015 | Hair density, hair count, hair thickness | Not mentioned | Western blot analyses of PDGF-A revealed significant differences between PRP-treated skin samples and control skin samples |
| Makki 2020 | Photographs, quartile grading scale with two dermatologists, hair parameters, and hair density, patients satisfaction | Local injection pain | Not mentioned |
| Paththinige 2018 | Hair density, hair count under dermoscopic photographs | Not mentioned | Not mentioned |
| Puig 2016 | Hair count through photography, hair mass index (measured using the cohen hair check system); and patient survery | Not mentioned | Not mentioned |
| Qu 2019 | Global macroscopic photographs, standardized phototrichograms, hair-pull test and satisfaction questionaire | Mild pain when injection, headache | Not mentioned |
| Rossano 2017 | Gene type | Not mentioned | It showed a negative correlation. IL-1a could be used as a prognostic value for PRP efficacy in female pattern hair loss |
| Schiavone 2018 | Photographs by global physician assessment (GPA) score and questionaires | Bruise after 48–72 h and spontaneously resolved in the fourth to fifth postop day | Not mentioned |
| Schiavone 2014 | Photographs by global physician assessment (GPA) score and questionaires | Not mentioned | Not mentioned |
| Sclafani 2014 | Hair density | No | Not mentioned |
| Shapiro 2020 | Hair density | No | Not mentioned |
| Siah 2020 | Dermatoscope, photography, hair density counting and hair caliber measurement | At week8, treatment site having a higher hair density 129.3 comparing to placebo site 115.3 | PDGF-BB was the highest concentration of growth factor injected, and VEGF was tested for the lowest growth factor concentration |
| Singhal 2015 | Hair count, hair thickness, hair root strength, and overall alopecia | 3 mild head pain | Not mentioned |
| Starace 2019 | Pull test, blobal photographs, and trichoscan, hair measurement | Not mentioned | Not mentioned |
| Takikawa 2011 | Histological exam | Not mentioned | Not mentioned |
| Tan 2019 | Folliscope, questionnaire | Not mentioned | Not mentioned |
| Tawfik 2018 | Hair growth, hair density, hair diameter, photography, hair-pull test, patient's satisfaction scale, standardized phototrichograms, and patient’s satisfaction | Not mentioned | Not mentioned |
| Zhang 2018 | Biopsy ki-67, hair density | Not mentioned | Not mentioned |
| Navarro 2015 | Trichogram, photograpy, anagen, telogen | Not mentioned | Not mentioned |
| DeVasconcelos 2015 | Mann-whitnety test | No | Not mentioned |
| Yang 2020 | Dermascopy and photograph | Not mentioned | Not mentioned |
| Zhang 2020 | Photographs, satisfaction questionaire | Not mentioned | Not mentioned |
| Zolfaghari 2020 | Hair number and thickness | Not mentioned | Measuring TGFβ1, TGFβ2, PDGF, EGF, VEGF, and HGF increasing by ELISA |
Outcomes measurement and adverse events in enrolled studies, and the evaluation of growth factors.
All-included studies showed positive responses and an improvement compared with the baseline; a few pooled patients reported adverse effects, including bruise and mild pain on injection sites after 48–72 h, which would resolve spontaneously in the 4th or 5th postoperation day. Mild headache and scalp sensitivity were also reported in a few studies.
Labs
Apart from clinical evaluation, 7 out of 42 studies also did an assessment on platelet-rich growth factors (PRGF) (Table 4). 6 out of 7 focused on hair follicles regenerative factors including VEGF, PDGF, IGF, TGF-β, and HGF through enzyme-linked immunosorbent assay (ELISA), Western blot for mRNA expression, flow cytometry on CD34+, and animal models. One study reveals a negative correlation between individual genetic inflammatory profile and efficacy of PRP, which was different from PRP in males (Rossano et al., 2017).
Subgroup Meta-Analysis
PRP efficacy compared to placebo. Only endpoints evaluated in the same measurement methods would be pooled in the meta-analysis. Using the random-effects model, PRP showed positive efficacy in the treatment of FPHL in hair density comparing to the control groups with odds ratio [OR] 1.61; 95% CI 0.52–2.70 (Figure 5).
FIGURE 5
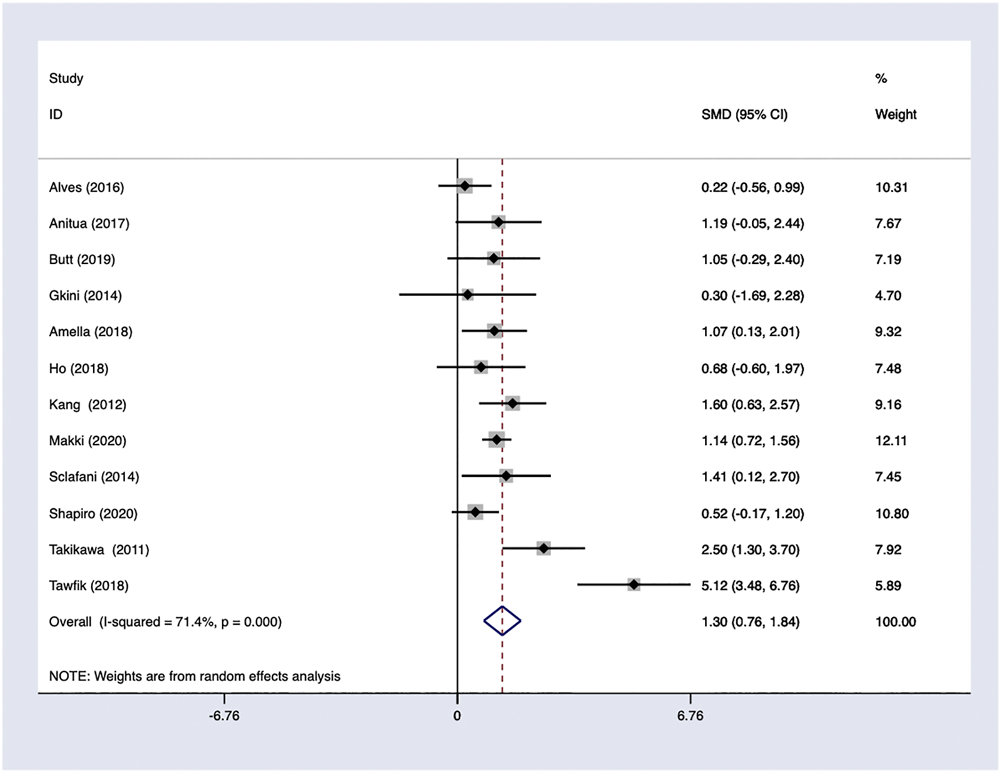
PRP efficacy compared to the baseline.
PRP efficacy compared to the baseline. Only studies with high quality would be pooled in the analysis. Using a random-effects model, PRP showed effectiveness and improvements on hair density comparing to baseline with OR 1.11; 95% CI 0.86–1.37 (Figure 6).
FIGURE 6

PRP efficacy compared to placebo.
Meta-analysis with other evaluation methods was not conducted due to limited studies.
Heterogeneity
In general, a meta-analysis of PRP efficacy comparing to the placebo had considerable statistical heterogeneity (I (Trueb, 2002) >75%), while a smaller substantial heterogeneity exists when comparing to the baseline. Visual inspection of funnel plots and Eggers tests were consistently in agreement with publication bias (Figure 7). Funnel plot that comparing with placebo suggested a tendency toward lack of large studies with both positive and negative results, two of the enrolled small studies showed little association (Figure 8). For the funnel plot in the baseline of the comparison group, a tendency toward a lack of small studies with negative and positive results was indicated (Figure 9).
FIGURE 7

Begg’s funnel plot of studies compared the efficacy complared to placebo.
FIGURE 8

Begg’s funnel plot of studies compared the efficacy complared to the baseline.
FIGURE 9
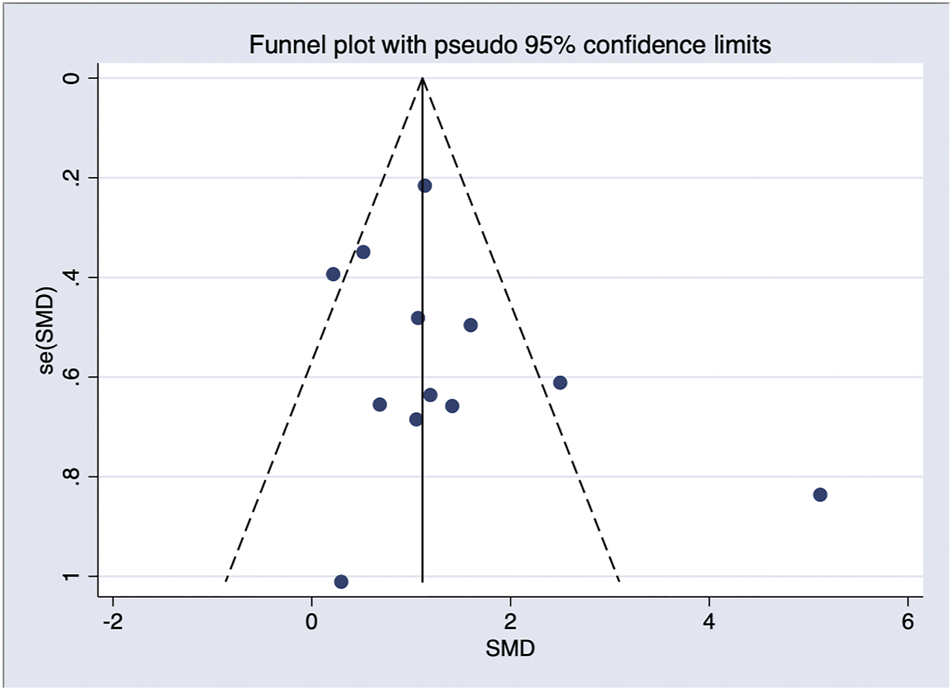
Funnel plot of studies compared the efficacy complared to the baseline.
Discussion
Our study demonstrated an effective response and relative safety for the application of PRP in the treatment of FPHL with the meta-analysis for RCT and observation studies. Especially for those patients with negative response to the tropical use of minoxidil, PRP offered an alternative treatment option. We only used hair density as an endpoint to perform meta-analysis for the diversity of endpoint settings among the enrolled studies, while hair density is the most commonly used one. This result is in accordance with previous systemic review (Torabi et al., 2020).
The major strengths of the study suggested a positive efficacy of PRP for female AGA and elucidated different PRP preparing methods and treatment regimes. Although there were several systemic reviews (Torabi et al., 2020) and meta-analysis on the application of PRP in AGA treatment (Evans et al., 2020), one mainly focus on female patients remain scarce.
In the author’s opinion, the need for larger scale randomized–controlled trials and extensive meta-analysis precedes a considerable heterogeneity challenge. Heterogeneity anticipated was mainly because of female’s nationalities and races, the difference treatment regime, PRP preparation methods, injection details, and PRP concentration. Subgroup analysis according to the different patient’s races and nationalities were not included in this study for the limited-published data and only a few studies included races information. Lower heterogeneity showed when studies of low quality were abandoned suggesting methodological heterogeneity contributed. Although efforts had been made to accommodate this issue, limited number of studies just based on women patients impeded methods like subgroup analysis, which resulted in a challenge to interpret the efficacy of PRP on female patients. As some studies measured patients of both gender but without separated data for female patients, we cannot prove strong evidence for the treating efficacy.
Selection bias is possibly addressed by group settings among studies. The prevalence of AGA is known to vary from the races (Paik et al., 2001; Gan and Sinclair, 2005; Khumalo et al., 2007). Enrolled studies were conducted by different countries, while all-pooled female participants were diverse in races. Subgroups regarding to ethnicity were not included in this study for the limited-enrolled studies. Whether divergences exist in the efficacy of PRP through races remain unclear. Besides, the majority of randomized studies were set in half-headed. Patients received PRP on half-scalp and placebo on the other half. Both injected spots showed improvement of hair growth or hair density. Although PRP-injected site showed a more obvious effect, the improvement of hair density on the placebo-injected sites may result in a smaller difference between PRP and placebo. Besides, whether PRP had a growth effect on the opposite side of the scalp remains obscure.
In aggregate, our study showed an efficacy of PRP in the therapy of FPHL through hair density evaluation. Although the mechanism of action on hair follicles is still under debate, it has proved to be a promising option for FPHL treatment. Given that current treatments differ from methodology and treatment technique, further studies are needed to define standardized protocols and large-scale–randomized trials still need to be conducted to confirm its efficacy.
Statements
Data availability statement
The original contributions presented in the study are included in the article/Supplementary Material; further inquiries can be directed to the corresponding author.
Author contributions
SZ and FQ contributed equally to this work. SZ and FQ did the conceptualization, CZ and YH screening the literatures, FQ and YG evaluated the quality of the included studies, SZ made the decision when any disagreement occurs. SZ and XY extracted the data. FQ and CZ did the meta-analysis and formal analysis, SZ and FQ wrote the original draft, SZ and YH reviewed and editing the draft. YH and CZ did the supervision of the whole work.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fphar.2021.642980/full#supplementary-material.
References
1
CashT. F. (2001). The psychology of hair loss and its implications for patient care. Clin. Dermatol.19 (2), 161–166. 10.1016/s0738-081x(00)00127-9
2
CervelliV.ScioliM. G.GentileP.DoldoE.BonannoE.SpagnoliL. G.et al (2012). Platelet-rich plasma greatly potentiates insulin-induced adipogenic differentiation of human adipose-derived stem cells through a serine/threonine kinase Akt-dependent mechanism and promotes clinical fat graft maintenance. Stem Cell Transl. Med. Mar.1 (3), 206–220. 10.5966/sctm.2011-0052
3
CookD. A.ReedD. A. (2015). Appraising the quality of medical education research methods. Acad. Med.90 (8), 1067–1076. 10.1097/ACM.0000000000000786
4
DavìG.PatronoC. (2007). Platelet activation and atherothrombosis. N. Engl. J. Med.357:(24), 2482–2494. 10.1056/NEJMra071014
5
EvansA. G.MwangiJ. M.PopeR. W.IvanicM. G.BotrosM. A.GlassmanG. E.et al (2020). Platelet-rich plasma as a therapy for androgenic alopecia: a systematic review and meta-analysis. J. Dermatol. Treat.26, 1–14. 10.1080/09546634.2020.1770171
6
GanD. C. C.SinclairR. D. (2005). Prevalence of male and female pattern hair loss in Maryborough. J. Invest. Dermatol. Symp. Proc.10 (3), 184–189. 10.1111/j.1087-0024.2005.10102.x
7
GarzaL. A.YangC.-C.ZhaoT.BlattH. B.LeeM.HeH.et al (2011). Bald scalp in men with androgenetic alopecia retains hair follicle stem cells but lacks CD200-rich and CD34-positive hair follicle progenitor cells. J. Clin. Invest.121 (2), 613–622. 10.1172/JCI44478
8
GentileP.ScioliM. G.BielliA.De AngelisB.De SioC.De FazioD.et al (2019). Platelet-rich plasma and micrografts enriched with autologous human follicle mesenchymal stem cells improve hair re-growth in androgenetic alopecia. biomolecular pathway analysis and clinical evaluation. Biomedicines7 (2), 27. 10.3390/biomedicines7020027
9
HarrisonJ. (2017). Advances in aesthetic therapies: plasma-rich protein procedure for the treatment of alopecia. Plast. Surg. Nurs.37 (2), 52–55. 10.1097/psn.0000000000000182
10
HesselerM. J.ShyamN. (2019). Platelet-rich plasma and its utility in medical dermatology: a systematic review. J. Am. Acad. Dermatol.81 (3), 834–846. 10.1016/j.jaad.2019.04.037
11
HigginsJ.ThomasJ.ChandlerJ.CumpstonM.LiT.PageM. J.et al (2020). Cochrane handbook for systematic reviews of interventions. version 6.1 (updated September 2020). Cochrane.
12
HigginsJ. P. T.AltmanD. G.GotzscheP. C.JuniP.MoherD.OxmanA. D.et al (2011) The Cochrane Collaboration's tool for assessing risk of bias in randomised trials. BMJ343, d5928. 10.1136/bmj.d5928
13
HigginsJ. P. T.ThompsonS. G.DeeksJ. J.AltmanD. G. (2003). Measuring inconsistency in meta-analyses. BMJ327 (7414), 557–560. 10.1136/bmj.327.7414.557
14
KasprzakM.SicińskaJ.SinclairR. (2019). The trichoscopy derived Sinclair scale: enhancing visual assessment through quantitative trichoscopy. Australas. J. Dermatol.60 (2), 134–136. 10.1111/ajd.12964
15
KhumaloN. P.JessopS.GumedzeF.EhrlichR. (2007). Hairdressing and the prevalence of scalp disease in African adults. Br. J. Dermatol.157 (5), 981–988. 10.1111/j.1365-2133.2007.08146.x
16
LeeJ.RabbaniC. C.GaoH.SteinhartM. R.WoodruffB. M.PflumZ. E.et al (2020) Hair-bearing human skin generated entirely from pluripotent stem cells. Nature582 (7812), 399–404. 10.1038/s41586-020-2352-3
17
McGuinnessL. A.HigginsJ. P. T. (2020). Risk-of-bias VISualization (robvis): an R package and Shiny web app for visualizing risk-of-bias assessments. Res. Synth. Methods12 (1), 55–61. 10.1002/jrsm.1411
18
ModarressiA. (2013). Platlet rich plasma (PRP) improves fat grafting outcomes. World J. Plast. Surg.2 (1), 6–13. 10.1097/prs.0000000000005676
19
MunnZ.BarkerT. H.MoolaS.TufanaruC.SternC.McArthurA.et al (2020). Methodological quality of case series studies: an introduction to the JBI critical appraisal tool. JBI Evid. Synth.18 (10), 2127–2133. 10.11124/JBISRIR-D-19-00099
20
OlsenE. A. (2001). Female pattern hair loss. J. Am. Acad. Dermatol.45 (3 Suppl. l), S70–S80. 10.1067/mjd.2001.117426
21
PaikJ.-H.YoonJ.-B.SimW.-Y.KimB.-S.KimN.-I. (2001). The prevalence and types of androgenetic alopecia in Korean men and women. Br. J. Dermatol.145 (1), 95–99. 10.1046/j.1365-2133.2001.04289.x
22
RossanoF.Di MartinoS.IodiceL.Di PaoloM.MissoS.TomeoR.et al (2017). Correlation between individual inflammation genetic profile and platelet rich plasma efficacy in hair follicle regeneration: a pilot study reveals prognostic value of IL-1a polymorphism. Eur. Rev. Med. Pharmacol. Sci.21 (22), 5247–5257. 10.26355/eurrev_201711_13848
23
Sánchez-GonzálezD. J.Méndez-BolainaE.Trejo-BahenaN. I. (2012). Platelet-rich plasma peptides: key for regeneration. Int. J. Peptides12, 1. 10.1155/2012/532519
24
StangA. (2010). Critical evaluation of the Newcastle-Ottawa scale for the assessment of the quality of nonrandomized studies in meta-analyses. Eur. J. Epidemiol.25 (9), 603–605. 10.1007/s10654-010-9491-z
25
StroupD. F.BerlinJ. A.MortonS. C.OlkinI.WilliamsonG. D.RennieD.et al (2000). Meta-analysis of observational studies in epidemiology. a proposal for reporting. JAMA283 (15), 2008–2012. 10.1001/jama.283.15.2008
26
TorabiP.BehrangiE.GoodarziA.RohaninasabM. (2020). A systematic review of the effect of platelet-rich plasma on androgenetic alopecia of women. Dermatol. Ther.33, e13835. 10.1111/dth.13835
27
TruebR. M. (2002). Molecular mechanisms of androgenetic alopecia. Exp. Gerontol.37 (8–9), 981–990. 10.1016/s0531-5565(02)00093-1
28
UekiR.TsuboiR.OgawaH.InabaY. (2003). Phototrichogram analysis of Japanese female subjects with chronic diffuse hair loss. J. Invest. Dermatol. Symp. Proc.8 (1), 116–120. 10.1046/j.1523-1747.2003.12184.x
29
van ZuurenE. J.FedorowiczZ.CarterB. (2012). Evidence-based treatments for female pattern hair loss: a summary of a Cochrane systematic review. Br. J. Dermatol. Nov167 (5), 995–1010. 10.1111/j.1365-2133.2012.11166.x
30
van ZuurenE. J.FedorowiczZ.SchoonesJ. (2016). Interventions for female pattern hair loss. Cochrane Database Syst. Rev.13 (5), CD007628. 10.1002/14651858.CD007628.pub4
31
WangT. L.ZhouC.ShenY. W.WangX. Y.DingX. L.TianS.et al (2010). Prevalence of androgenetic alopecia in China: a community-based study in six cities. Br. J. Dermatol. Apr162 (4), 843–847. 10.1111/j.1365-2133.2010.09640.x
32
WeibrichG.KleisW. K. G.HafnerG.HitzlerW. E. (2002). Growth factor levels in platelet-rich plasma and correlations with donor age, sex, and platelet count. J. Craniomaxillofac. Surg.30 (2), 97–102. 10.1054/jcms.2002.0285
33
WuP. I.-K.DiazR.Borg-SteinJ. (2016). Platelet-rich plasma. Phys. Med. Rehabil. Clin. North America27 (4), 825–853. 10.1016/j.pmr.2016.06.002
Summary
Keywords
platelet-rich plasma, female androgenic alopecia, female pattern hair loss, systematic review, meta-analysis
Citation
Zhou S, Qi F, Gong Y, Zhang C, Zhao S, Yang X and He Y (2021) Platelet-Rich Plasma in Female Androgenic Alopecia: A Comprehensive Systematic Review and Meta-Analysis. Front. Pharmacol. 12:642980. doi: 10.3389/fphar.2021.642980
Received
18 December 2020
Accepted
19 March 2021
Published
06 May 2021
Volume
12 - 2021
Edited by
Pietro Gentile, University of Rome Tor Vergata, Italy
Reviewed by
Kurt Neumann, Independent Researcher, Kerékteleki, Hungary
Raja Ahsan Aftab, Taylor’s University, Malaysia
Updates
Copyright
© 2021 Zhou, Qi, Gong, Zhang, Zhao, Yang and He.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Yanling He, heyanlingdermatology@gmail.com
†These authors have contributed equally to this work
This article was submitted to Drugs Outcomes Research and Policies, a section of the journal Frontiers in Pharmacology
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.