- 1Clinical Medicine Department, First Medical College, Lanzhou University, Lanzhou, China
- 2Critical Care Department, The First Hospital of Lanzhou University, Lanzhou, China
- 3Secretary’s Office, Gansu Province Maternity and Child Health Hospital, Gansu Province Central Hospital, Lanzhou, China
Background: Chronic obstructive pulmonary disease (COPD) is characterized by chronic hypoxia, inflammation, oxidative stress, and irreversible airflow limitations. Rhodiola L. is a genus of botanical drugs used in traditional medicine that may influence COPD.
Objective: A systematic review of the safety and efficacy of Rhodiola L. in patients with COPD.
Material and methods: We searched the PubMed, Embase, Cochrane Library, Web of Science, Scopus, China National Knowledge Infrastructure (CNKI), Chongqing VIP, Wanfang, and SinoMed databases. The search strategy used terms including “COPD” and “Rhodiola.” Two independent reviewers conducted the literature screening, data extraction, and risk of bias assessment, with a third reviewer involved to resolve disagreements. Statistical analysis was conducted in Review Manager (version 5.4.1), following the Cochrane Handbook.
Results: This review included nine studies, of which two focused on Rhodiola crenulata (Hook.f. and Thomson) H. Ohba (R. crenulata) and two on Rhodiola kirilowii (Regel) Maxim (R. kirilowii); the remaining five focused on Rhodiola wallichiana (Hook.) S.H.Fu (R. wallichiana). Compared with the placebo, patients who received Rhodiola L. presented no more adverse events (p = 0.65) but showed significant improvement in the percentage of forced expiratory volume in 1 s at prediction (FEV1%pred), forced expiratory volume in 1 s (FEV1), the ratio of forced expiratory volume in 1 s on forced vital capacity (FEV1/FVC), saturation of oxygen in arterial blood, partial pressure of oxygen in arterial blood (PaO2), partial pressure of carbon dioxide in arterial blood (PaCO2), systolic pulmonary arterial pressure, diastolic pulmonary arterial pressure, COPD assessment test, efficient rate, C-reactive protein, and N-terminal pro-B-type natriuretic peptide (all p < 0.01). Compared with ambroxol, R. kirilowii provided additional benefits to patients with COPD in FEV1%pred, FEV1, FEV1/FVC, PaO2, PaCO2, 8-iso-prostaglandin F2α, superoxide dismutase, glutathione, malondialdehyde, and total antioxidant capacity (all p < 0.01).
Conclusion: Among the Rhodiola L. genus, this review included R. wallichiana, R. crenulata, and R. kirilowii, which might be safe and effective in COPD. Although this study has several limitations, further RCTs are needed.
Systematic Review Registration: [https://www.crd.york.ac.uk/PROSPERO/ display_record.php?RecordID=302881], identifier [CRD42022361890].
1 Introduction
Chronic obstructive pulmonary disease (COPD) is the third-leading cause of mortality globally (GBD Chronic Respiratory Disease Collaborators, 2020). COPD is a chronic respiratory disease characterized by irreversible airflow limitations. It consists of complicated pathophysiological processes including hypoxia, oxidative stress, and sustained systematic or local inflammation (GOLD Report, 2023). Based on these changes, COPD leads to many typical manifestations including continuous dyspnea, cough, chronic fatigue, and impaired quality of life. The conventional treatment of COPD is inhaled bronchodilators or corticosteroids (GOLD Report, 2023). However, COPD is not just an acute local inflammatory disease; therefore, these treatments are insufficient and the control of deterioration by such methods is usually not satisfying in some patients.
Rhodiola L. is a genus of botanical drugs broadly used as adaptogens. It has a long history of use and has been applied in traditional medicine throughout Asia and Europe. It has also been recorded in the official pharmacopeia of several countries, including China and Russia, because of its widespread clinical application and its proven efficacy in clinical trials for a variety of diseases (Panossian et al., 2021). Rhodiola L. is widely distributed in the high altitudes of Asia and Europe and was generally used to relieve altitude sickness. In traditional Chinese medicine, Rhodiola L. is believed to tonify the Qi and benefit the lungs, while in modern medicine, Rhodiola L. reportedly alleviates dyspnea, chronic cough, and fatigue (Chinese Pharmacopoeia Commission, 2020), which are the major clinical manifestations of COPD (GOLD Report, 2023). Several bioactive substances have been separated from Rhodiola L., including salidroside, tyrosol, and ethyl gallate (Han et al., 2016). These ingredients provide anti-inflammation, anti-overoxidation, and anti-depression effects, among others. (Li et al., 2017). Based on these mechanisms, several studies confirmed the efficacy of Rhodiola L. in many diseases. For instance, a comprehensive supplementation including Rhodiola L. reduced stress levels in healthy individuals (Noah et al., 2022), (Boyle et al., 2022). Rhodiola L. also improved explosive physical training performance in healthy participants (Williams et al., 2021). Rhodiola L. also improved depression symptoms and the quality of life of depressant patients (Gao et al., 2020), (Mao et al., 2015). In patients with respiratory diseases other than COPD, for instance, in obstructive sleep apnea, Rhodiola L. supplementation not only improved patient mental conditions but also elevated serum anti-oxidant concentrations (Yu et al., 2019).
However, despite the hypothesis that Rhodiola L. might have some benefits in patients with COPD, there remains no consensus on whether Rhodiola L. is efficient and safe in this population. One challenge is several species of Rhodiola L. are applied in similar medical use. For example, in China, extracts of Rhodiola crenulata (Hook.f. and Thomson) H. Ohba (R. crenulata), Rhodiola wallichiana (Hook.) S.H.Fu (R. wallichiana), and Rhodiola kirilowii (Regel) Maxim (R. kirilowii) are marketed (“Rhodiola crenulata (Hook.f. and Thomson) H. Ohba.,” 2023; “Rhodiola kirilowii (Regel) Maxim.,” 2023; “Rhodiola wallichiana (Hook.) S.H.Fu.,” 2023). Several randomized controlled trials (RCTs) on this topic reported different outcomes. For instance, one study on the efficacy of R. crenulata in COPD assessed patient lung function, symptom score, and physical endurance, while another study focused on inflammatory biomarkers (Chen et al., 2015; Chuang et al., 2015). Moreover, contrary results have been reported. For example, two studies reported that the addition of R. wallichiana significantly elevated the efficient rate of COPD treatment (Qv et al., 2014; Li et al., 2016), while Zhu et al. (2015) observed no significant difference between the Rhodiola L. and placebo groups. These findings make it difficult to summarize the information on this topic. Therefore, we conducted this systematic review and meta-analysis to make conclusions based on existing evidence from RCTs.
2 Methods
This systematic review was conducted following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) statement and preliminarily registered on the International Prospective Register of Systematic Reviews (PROSPERO, #CRD 42022361890) (Page et al., 2021; Yu et al., 2023). This study was designed and conducted following the guidance of the Cochrane Handbook for Systematic Reviews of Interventions (Higgins et al., 2022). Moreover, this report followed the “Consensus statement on the Phytochemical Characterisation of Medicinal Plant extracts” (ConPhyMP) guideline (Heinrich et al., 2022).
2.1 Literature search and screening
The PubMed, Cochrane Library, Web of Science, Scopus, Embase, ClinicalTrials.gov, China National Knowledge Infrastructure, Chongqing VIP, Wanfang, and SinoMed databases were searched for potential literature studies. Major terms used in the literature search were “Rhodiola” and “COPD.” The detailed search strategy and results are presented in Supplementary Table S1. Articles written in English or Chinese and published from the inception of databases to 30 September 2022 were included. Two reviewers independently screened the included literature, and a third experienced reviewer combined the two screening results by discussing them with the other reviewers.
2.2 Eligibility criteria for the literature screening
The eligibility criteria used in the screening process were 1) participants: adult patients with spirometry or physician-confirmed COPD; 2) intervention: drugs in which Rhodiola L. was the main ingredient, regardless of administration type; 3) comparators: placebo or conventional treatment (including monotherapy or combined bronchodilator or corticosteroid therapy); 4) outcomes: as the outcomes were inconsistent, we did not set limits on this domain and performed a meta-analysis of any indicators reported by at least two studies; and 5) study design: only RCTs.
2.3 Data extraction and quality assessment
Two reviewers independently extracted data from the finally retrieved articles using a pre-designed data form. These items included the following: 1) basic study information: first author, publication year, and PICOS information; 2) the risk of bias information defined in Cochrane Review Manager 5.4.1; and 3) the data on efficacy and safety outcomes. We did not target specific outcomes. Instead, we comprehensively gathered every outcome and meta-analyzed any outcome that was reported by at least two studies.
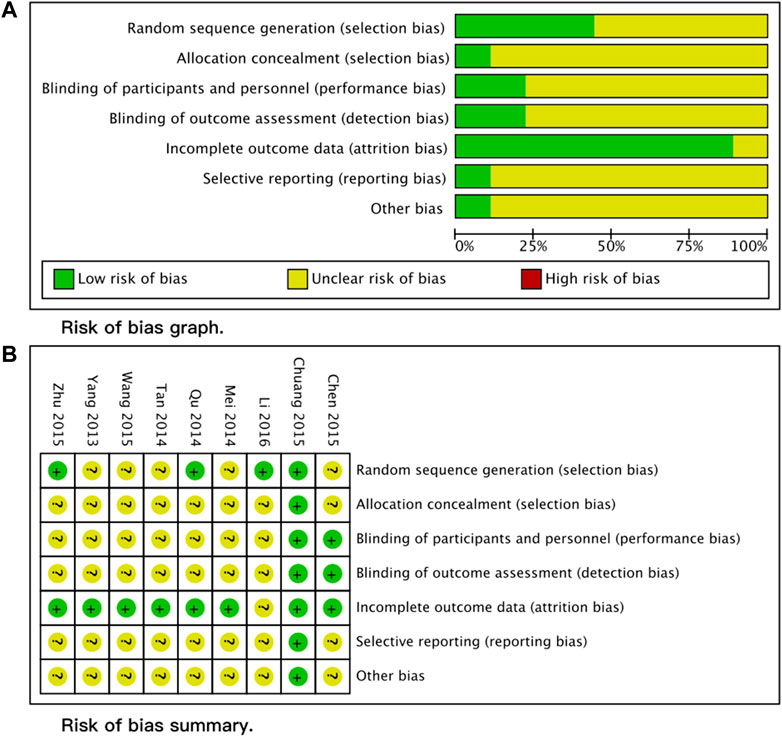
We assessed the quality of included studies using the Cochrane Risk of Bias (RoB) Tool (1.0) provided in RevMan 5.4. The RoB assessment contains seven domains: random sequence generation, allocation concealment, blinding of participants and personnel, blinding of outcome assessment, incomplete outcome, selective reporting, and other bias. Two different reviewers independently assessed all included studies, and a third experienced reviewer combined the two assessing results.
2.4 Data analysis
All data analysis was conducted in RevMan (version 5.4.1, The Cochrane Collaboration). We systematically reviewed and meta-analyzed two types of data (dichotomous and continuous). For dichotomous data, the risk ratio (RR) was calculated; for continuous data, the mean difference (MD) or standardized mean difference (SMD) was calculated. When the continuous data were tested using different methods and mechanisms or presented in different units, SMD was used; otherwise, the MD was used. Along with the pooling estimations, the 95% confidence interval (95%CI) was also. Heterogeneity was analyzed using the I2 test. When I2 > 50% and p < 0.05, a random-effects model (REM) was used; otherwise, a fixed-effects model (FEM) was used in the pooling process. For outcomes that were pooled by REM, we further conducted sensitivity analysis by omitting one study at a time. Although Egger et al. implied that the results of publication bias tests for outcomes with <10 included studies may be unreliable, we generated funnel plots for outcomes with >5 included studies (Egger et al., 1997).
3 Results
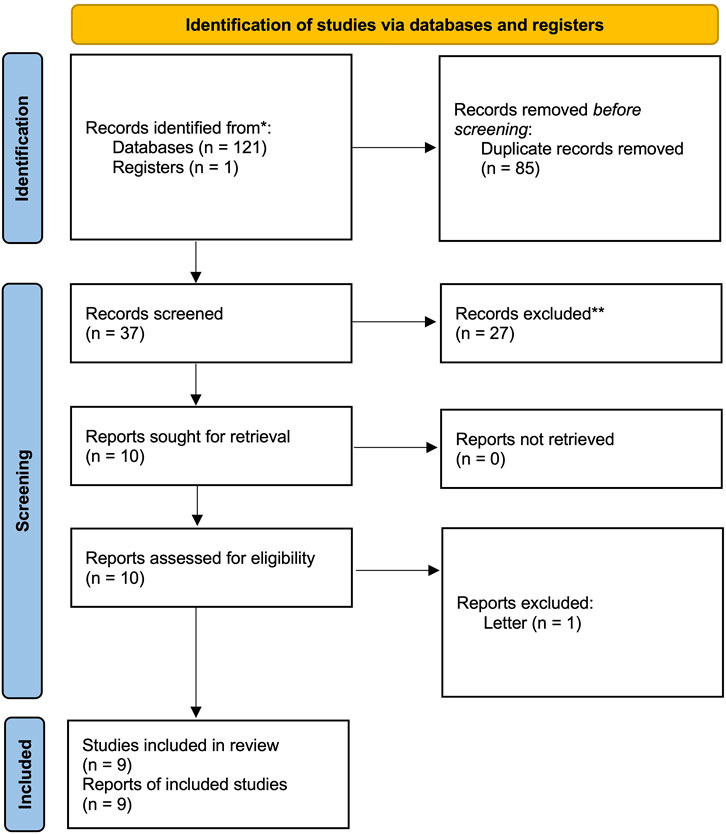
We identified 122 articles in the search process. After reading the titles and abstracts, we retrieved the full texts of 10 studies. One study was excluded after reading the full article because it was a letter, and no sufficient information could be extracted (Wang, 2010). Therefore, this study finally included nine studies (Wang, 2010; Yang et al., 2013; Mei et al., 2014; Qv et al., 2014; Tan, 2014; Chen et al., 2015; Chuang et al., 2015; Zhu et al., 2015; Li et al., 2016). The detailed screening process is presented in the PRISMA flow diagram (Figure 1). The overall quality of the included studies was medium, with the lack of detailed descriptions of randomization and masking the major reason impairing the quality of included studies. The detailed RoB assessment information is shown in Figure 2. Of all nine included articles, two were reported in English; the remaining seven were reported in Chinese, and all the studies were conducted in the Chinese population. Seven trials compared Rhodiola L. with placebo, while the other two trials compared R. kirilowii with ambroxol. Therefore, we performed separate analyses.
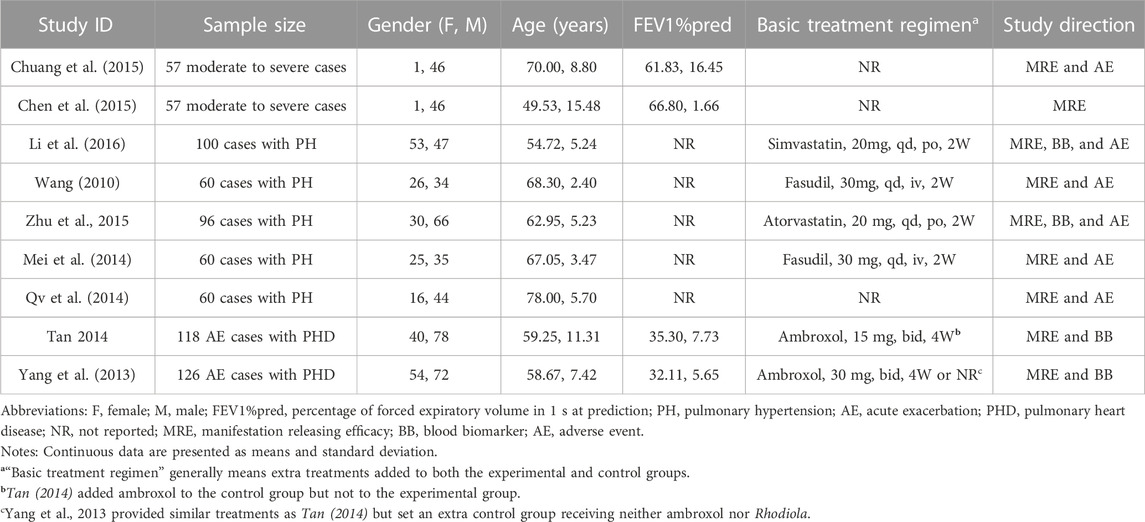
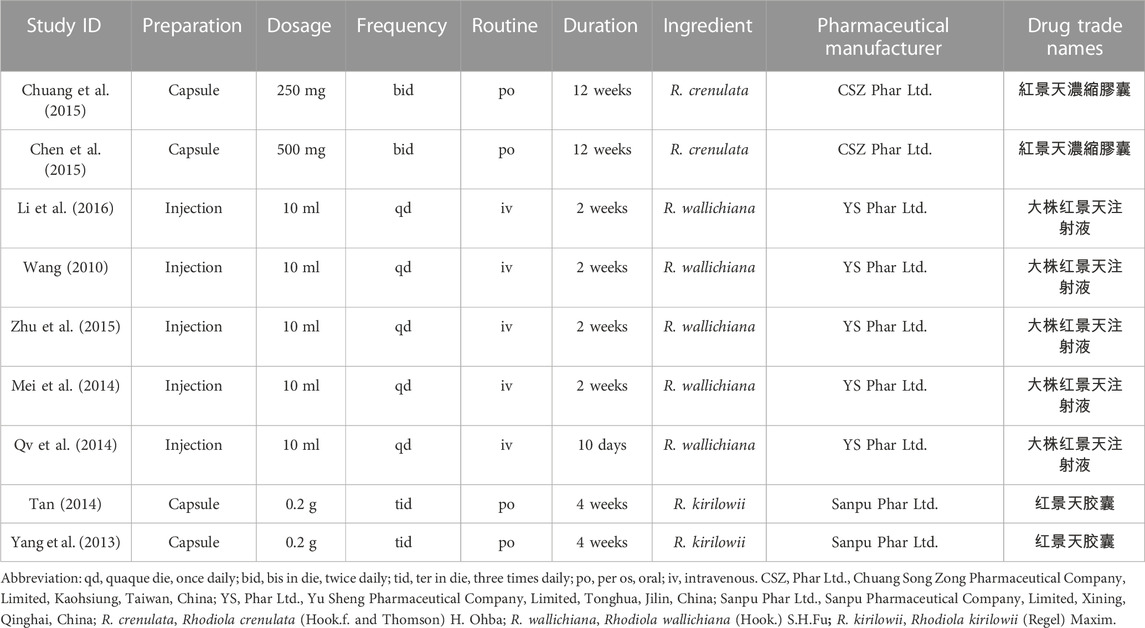
Two trials studied R. crenulata, two R. kirilowii, and five R. wallichiana. Detailed information on included studies is shown in Table 1. According to the ConPhyMP guidelines (Heinrich et al., 2022), all included studies focused on “type A” extracts (documented in national pharmacopeia with licensed application). Table 2 summarizes their preparations, dosage, routine, pharmaceutical manufacturer, etc.
Two comparisons were made in included articles: Rhodiola L. vs. control and Rhodiola L. (only R. kirilowii) vs. ambroxol. The outcomes reported in these articles could be divided into four domains: manifestation-releasing efficacy, serum biomarkers, and safety concerns. We pooled the MD for every continuous outcome because all of them were tested and reported in the same manner. The meta-analysis results are summarized in Figures 3, 4. The related forest plots are shown in Supplementary Figures Series S1, S2. Sensitivity analysis was conducted by omitting the result of one study at a time for those pooling estimations for which I2 > 50% and p < 0.05. All outcomes were robust to their heterogeneity. Subgroup analyses were conducted based on Rhodiola L. species.
3.1 Efficacy against COPD clinical manifestations
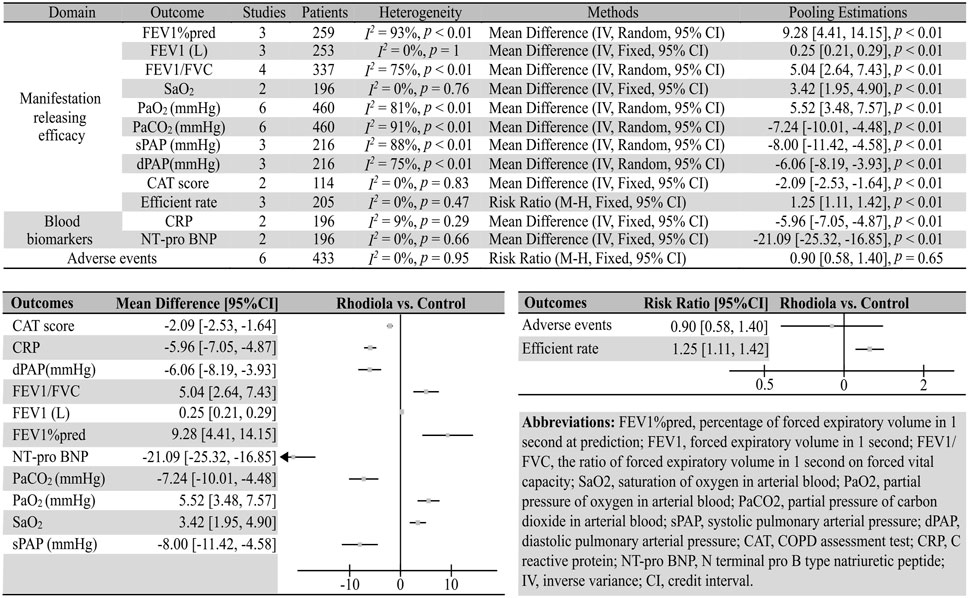
Compared with placebo, Rhodiola L. improved the FEV1%pred (three studies; 259 patients; I2 = 93%, p < 0.01; REM; MD [95%CI] = 9.28 [4.41, 14.15], p < 0.01), FEV1 (three studies; 253 patients; I2 = 0, p = 1; REM; MD [95%CI] = 0.25 [0.21, 0.29], p < 0.01), FEV1/FVC (four studies; 337 patients; I2 = 75%, p < 0.01, REM; MD [95%CI] = 5.04 [2.64, 7.43], p < 0.01), SaO2 (two studies, 196 patients; I2 = 0%, p = 0.76, FEM; MD [95%CI] = 3.42 [1.95, 4.90], p < 0.01), PaO2 (six studies, 460 patients; I2 = 81%, p < 0.01, REM; MD [95%CI] = 5.52 [3.48, 7.57], p < 0.01), PaCO2 (six studies, 460 patients; I2 = 91%, p < 0.01, REM; MD [95%CI] = −7.24 [−10.01, −4.48], p < 0.01), sPAP (three studies, 216 patients; I2 = 88%, p < 0.01, REM; MD [95%CI] = −8.00 [−11.42, −4.58], p < 0.01), dPAP (three studies, 216 patients; I2 = 75%, p < 0.01, REM; MD [95%CI] = −6.06 [−8.19, −3.93], p < 0.01), CAT score (two studies, 114 patients; I2 = 0%, p = 0.83, FEM; MD [95%CI] = −5.96 [−7.05, −4.87], p < 0.01), and efficiency rate (three studies, 205 patients; I2 = 0%, p = 0.47, FEM; RR [95%CI] = 1.25 [1.11, 1.42], p < 0.01).
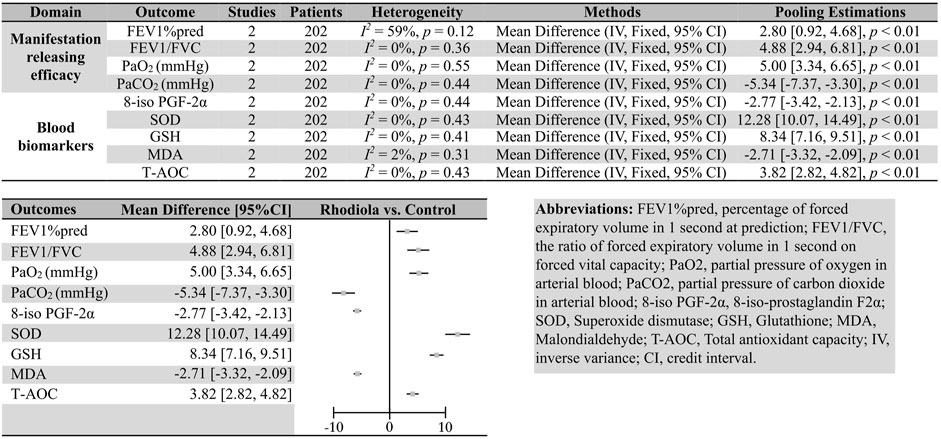
Compared with ambroxol, R. kirilowii also showed superiority in several domains: FEV1%pred (two studies, 202 patients; I2 = 59%, p = 0.12, FEM; MD [95%CI] = 2.80 [0.92, 4.68], p < 0.01), FEV1/FVC (two studies, 202 patients; I2 = 0%, p = 0.36, FEM; MD [95%CI] = 4.88 [2.94, 6.81], p < 0.01), PaO2 (two studies, 202 patients; I2 = 0%, p = 0.55, FEM; MD [95%CI] = 5.00 [3.34, 6.65], p < 0.01), and PaCO2 (two studies, 202 patients; I2 = 0%, p = 0.44, FEM; MD [95%CI] = −5.34 [−7.37, −3.30], p < 0.01).
3.2 Efficacy in patient blood biomarkers
Compared with placebo or ambroxol, patients administered Rhodiola L. showed lower levels of blood biomarkers, CRP (vs. placebo; two studies, 196 patients; I2 = 9%, p = 0.29, FEM; MD [95%CI] = -5.96 [-7.05, -4.87], p < 0.01), NT-pro BNP (vs. placebo; two studies, 196 patients; I2 = 0%, p = 0.66, FEM; MD [95%CI] = −21.09 [−25.32, -16.85], p < 0.01), 8-iso PGF-2α (vs. ambroxol; two studies, 202 patients; I2 = 0%, p = 0.44, FEM; MD [95%CI] = −2.77 [−3.42, −2.13], p < 0.01), SOD (vs. ambroxol; two studies, 202 patients; I2 = 0%, p = 0.43, FEM; MD [95%CI] = 12.28 [10.07, 14.49], p < 0.01), GSH (vs. ambroxol; two studies, 202 patients; I2 = 0%, p = 0.41, FEM; MD [95%CI] = 8.34 [7.16, 9.51], p < 0.01), MDA (vs. ambroxol; two studies, 202 patients; I2 = 2%, p = 0.31, FEM; MD [95%CI] = −2.71 [−3.32, −2.09], p < 0.01), and T-AOC (vs. ambroxol; two studies, 202 patients; I2 = 0%, p = 0.43, FEM; MD [95%CI] = 3.82 [2.82, 4.82], p < 0.01).
3.3 Safety of Rhodiola L. in patients with COPD
Six studies comparing Rhodiola L. and placebo reported that the Rhodiola L. groups did not show more adverse events (373 patients, I2 = 0%, p = 0.65, FEM; RR [95%CI] = 0.90 [0.58, 1.40], p = 0.65).
3.4 Subgroup analysis
Depending on the Rhodiola L. species, the outcomes were divided into four categories: 1) only R. crenulata (vs. placebo: CAT score); 2) only R. wallichiana (vs. placebo: sPAP, dPAP, and SaO2); 3) R. crenulata or R. kirilowii with R. wallichiana (vs. placebo: FEV1%pred, FEV1, FEV1/FVC, PaO2, and PaCO2); and 4) only R. kirilowii (vs. ambroxol: all outcomes). In category three, we conducted subgroup analyses to explore differences between Rhodiola L. species. However, results were inconsistent due to the limited number of included studies (see forest plots in the Supplementary Material).
3.5 Tests of publication bias
The analysis of PaO2, PaCO2, and adverse events included six studies, for which we plotted funnel plots. These plots are all symmetrical, suggesting a lack of significant publication bias (see Supplementary Figure Series).
4 Discussion
This systematic review and meta-analysis included nine studies and 734 patients. The results showed that R. wallichiana, R. crenulata, and R. kirilowii might be safe and promising in relieving the clinical manifestations, pulmonary circulation, inflammation, and oxidative stress of COPD. However, due to the lack of included studies, it might be too early to draw any clinically practical conclusions.
We found that Rhodiola L. may improve FEV1%pred, FEV1, FEV1/FVC, SaO2, PaO2, PaCO2, and CAT score, and efficient rate in COPD, indicating that Rhodiola L. might be beneficial to respiratory function and clinical manifestations in patients with COPD. Previous studies showed that Rhodiola L. might enhance exercise performance and pulmonary ventilation (Kelly, 2001; Zhang et al., 2009). However, the mechanisms of such effects remain unclear. The administration of Rhodiola L. improved adenosine triphosphate (ATP) turnover to increase muscle power (Abidov et al., 2003). The enhancement of the respiratory muscles after using Rhodiola L. might explain this slight recovery of respiratory function. The results of our subgroup analysis were based on limited evidence and are highly inconsistent; therefore, we cannot draw conclusions.
Our results showed that Rhodiola L. may improve sPAP, dPAP, and NT-pro BNP in COPD, indicating that Rhodiola L. might attenuate pulmonary hypertension (PH) in patients with COPD. Previous former studies confirmed the anti-PH effects Rhodiola L. and explored the mechanisms of these effects. Hypoxia is a key point in the occurrence and development of PH; therefore, in patients with COPD patients, hypoxia might also be a core mechanism for the development of PH (Seeger et al., 2013). Rhodiola L. inhibited the thickening of pulmonary vessel walls and maintained the vessel diameter in a PH rat model (Nan et al., 2018). The results of the present study also found that Rhodiola L. also inhibits the expression of several hypoxia-induced proteins, such as proliferating cell nuclear antigen (PCNA), cyclin-dependent kinase 4 (CDK4), and cyclin D1 in pulmonary tissue. Rhodiola L. also inhibited the proliferation of rat pulmonary artery smooth muscle cells (Nan et al., 2022; Zhang et al., 2022).
We found that Rhodiola L. may improve CRP, 8-iso PGF-2α, SOD, GSH, MDA, and T-AOC in COPD, indicating its potential anti-inflammatory and antioxidant effects in COPD. Previous studies demonstrated the anti-inflammatory potency of Rhodiola L. in many diseases including cancer, diabetes, and cardiovascular disease (Pu et al., 2020). This potency might be explained by its multiple metabolites’ inhibition of different inflammatory signaling pathways, including nuclear factor-κ-gene binding (NF- κB), mitogen-activated protein kinase (MAPK), Janus kinase 2/signal transducer and activator of transcription 3 (JAK2/STAT3), and phosphoinositide 3-kinases/Akt (PI3K/Akt) (Chang et al., 2015; Je et al., 2015; Qi et al., 2016). Moreover, Rhodiola L. also demonstrated strong antioxidant activity (Sun et al., 2020), which can suppress the production of reactive oxygen species (ROS) and increase the activity of antioxidant agents such as superoxide dismutase (SOD) and glutathione peroxidase (GSH-Px) (Li et al., 2012; Xing et al., 2014).
This study is the first systematic review and meta-analysis to assess the efficacy and safety of Rhodiola L. in patients with COPD. It included only RCTs and combined mixed evidence to obtain an overall impression of Rhodiola L. in COPD. The study also has some limitations: 1) Although only RCTs were included, the Risk of Bias assessment of the included studies identified some “unclear risks”, which could weaken the strength of the evidence; 2) only nine studies with 734 patients were included; this small number of included trials and sample size limits the strength of the conclusion; 3) although publication bias was tested, the results remain doubtful because of the limited number of included studies. Well-designed, large-scale RCTs are still required; 4) all included studies analyzed the efficacy and safety of Rhodiola L. extract, without considering the specific substances contained in Rhodiola L. such as rosavin or salidroside; 5) this study included only the literature in Chinese and English; however, Rhodiola L. is used in many countries in Asia and Europe. Therefore, the possibility of articles related to this topic being published in other languages cannot be excluded. Therefore, language bias is possible. 6) Rhodiola L. is a large genus with many different species, while this study included only R. wallichiana, R. kirilowii, and R. crenulata; hence, the results can only be applied to these two species and cannot be generalized to other species.
This study analyzed several Rhodiola L. extracts currently available on the market. Limited by the number and quality of included studies, the inferences drawn are weak but are sufficient to raise awareness of these drugs by clinical practitioners. The safety and promising efficacy of Rhodiola L. extracts shown in this study indicated the need for more studies on this topic. As the current RCTs are generally not well designed, conducted, or reported, more effort must be put into these aspects in the future.
5 Conclusion
This study included Rhodiola wallichiana (Hook.) S.H.Fu, Rhodiola kirilowii (Regel) Maxim, and Rhodiola crenulata (Hook. f. and Thomson) H. Ohba, which might be safe and effective in COPD. They might be able to improve respiratory function, clinical manifestation, pulmonary circulation, inflammation, and oxidative stress. Limited by the quantity and quality of included studies, the results of this study are preliminary; therefore, well-designed, large-scale, randomized, controlled trials are required.
Data availability statement
The original contributions presented in the study are included in the article/Supplementary Material; further inquiries can be directed to the corresponding author.
Author contributions
All authors made a significant contribution to the work reported, whether that is in the conception (HY), study design (HY and XS), literature obtaining (LZ, XS, and HY), data extraction (TL, ZF, and HY), analysis and interpretation (HY, MD, and ZH), revising or critically reviewing the article (HG and JL); gave final approval of the version to be published (JL); have agreed on the journal to which the article has been submitted (JL); and agree to be accountable for all aspects of the work (all authors).
Funding
This work was supported by the Science and Technology Projects of Gansu Province (Grant Number 18JR3RA344), and Special Funding for Big Data Research of Analgesic Sedation for Severe Infections. (Grant number Z-2019-1-002). Funders had no role in the design of the study, the collection and analysis of the data, or the preparation of the manuscript.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors, and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fphar.2023.1139239/full#supplementary-material
References
Abidov, M., Crendal, F., Grachev, S., Seifulla, R., and Ziegenfuss, T. (2003). Effect of extracts from Rhodiola rosea and Rhodiola crenulata (Crassulaceae) roots on ATP content in mitochondria of skeletal muscles. Bull. Exp. Biol. Med. 136, 585–587. doi:10.1023/b:bebm.0000020211.24779.15
Boyle, N. B., Billington, J., Lawton, C., Quadt, F., and Dye, L. (2022). A combination of green tea, rhodiola, magnesium and B vitamins modulates brain activity and protects against the effects of induced social stress in healthy volunteers. Nutr. Neurosci. 25, 1845–1859. doi:10.1080/1028415X.2021.1909204
Chang, X., Luo, F., Jiang, W., Zhu, L., Gao, J., He, H., et al. (2015). Protective activity of salidroside against ethanol-induced gastric ulcer via the MAPK/NF-κB pathway in vivo and in vitro. Int. Immunopharmacol. 28, 604–615. doi:10.1016/j.intimp.2015.07.031
Chen, S.-P., Huang Liu, R., Lu, T.-M., Wei, J. C.-C., Wu, T.-C., Tsai, W.-Y., et al. (2015). Complementary usage of rhodiola crenulata (L.) in chronic obstructive pulmonary disease patients: The effects on cytokines and T cells. Phytotherapy Res. 29, 518–525. doi:10.1002/ptr.5259
Chinese Pharmacopoeia Comission (2020). Pharmacopoeia of the people’s Republic of China. Beijing: China Medical Science and Technology Press.
Chuang, M.-L., Wu, T.-C., Wang, Y.-T., Wang, Y.-C., Tsao, T. C.-Y., Wei, J. C.-C., et al. (2015). Adjunctive treatment with rhodiola crenulata in patients with chronic obstructive pulmonary disease--A randomized placebo controlled double blind clinical trial. PLoS One 10, e0128142. doi:10.1371/journal.pone.0128142
Egger, M., Davey Smith, G., Schneider, M., and Minder, C. (1997). Bias in meta-analysis detected by a simple, graphical test. BMJ 315, 629–634. doi:10.1136/bmj.315.7109.629
Gao, L., Wu, C., Liao, Y., and Wang, J. (2020). Antidepressants effects of rhodiola capsule combined with sertraline for major depressive disorder: A randomized double-blind placebo-controlled clinical trial. J. Affect Disord. 265, 99–103. doi:10.1016/j.jad.2020.01.065
GBD Chronic Respiratory Disease Collaborators (2020). Prevalence and attributable health burden of chronic respiratory diseases, 1990-2017: A systematic analysis for the global burden of disease study 2017. Lancet Respir. Med. 8, 585–596. doi:10.1016/S2213-2600(20)30105-3
Global Initiative for Chronic Obstructive Lung Disease (2020). Global initiative for chronic obstructive lung disease - gold. WWW Document. URL Available at: https://goldcopd.org/ (accessed 8 2, 23).
Han, F., Li, Y., Ma, L., Liu, T., Wu, Y., Xu, R., et al. (2016). A rapid and sensitive UHPLC-FT-ICR MS/MS method for identification of chemical constituents in Rhodiola crenulata extract, rat plasma and rat brain after oral administration. Talanta 160, 183–193. doi:10.1016/j.talanta.2016.07.014
Heinrich, M., Jalil, B., Abdel-Tawab, M., Echeverria, J., Kulić, Ž., McGaw, L. J., et al. (2022). Best Practice in the chemical characterisation of extracts used in pharmacological and toxicological research-The ConPhyMP-Guidelines. Front. Pharmacol. 13, 953205. doi:10.3389/fphar.2022.953205
Higgins, J., Thomas, J., Chandler, J., Cumpston, M., Li, T., Page, M., et al. (2022). Cochrane Handbook for systematic reviews of interventions version 6.3. (updated February 2022) [WWW Document]. Cochrane. URL Available at: https://training.cochrane.org/handbook/current (accessed 10 25, 22).
Je, I.-G., Kim, D.-S., Kim, S.-W., Lee, S., Lee, H.-S., Park, E. K., et al. (2015). Tyrosol suppresses allergic inflammation by inhibiting the activation of phosphoinositide 3-kinase in mast cells. PLoS One 10, e0129829. doi:10.1371/journal.pone.0129829
Li, L., Cao, X., Wang, J., Zhang, L., Shi, Y., Sun, S., et al. (2016). [Clinical study on the treatment of chronic obstructive pulmonary disease combined with pulmonary arterial hypertension with Rhodiola rosea injection in combination with simvastatin. Drugs & Clin. 31, 1403–1406.
Li, M.-H., Tang, C.-F., and Ouyang, J.-Q. (2012). Influence of salidroside from Rhodiola Sachalinensis A. Bor on some related indexes of free radical and energy metabolism after exercise in mice. Zhongguo Ying Yong Sheng Li Xue Za Zhi 28, 53–56.
Li, Y., Wu, J., Shi, R., Li, N., Xu, Z., and Sun, M. (2017). Antioxidative effects of rhodiola genus: Phytochemistry and pharmacological mechanisms against the diseases. Curr. Top. Med. Chem. 17, 1692–1708. doi:10.2174/1568026617666161116141334
Mao, J. J., Xie, S. X., Zee, J., Soeller, I., Li, Q. S., Rockwell, K., et al. (2015). Rhodiola rosea versus sertraline for major depressive disorder: A randomized placebo-controlled trial. Phytomedicine 22, 394–399. doi:10.1016/j.phymed.2015.01.010
Mei, H., Pan, H., and Yan, X. (2014). [Clinical efficacy of fasudil combined with Rhodiola rosea in the treatment of pulmonary hypertension in chronic obstructive pulmonary disease. Shenzhen J. Integr. Traditional Chin. West. Med. 24, 35–36. doi:10.16458/j.cnki.1007-0893.2014.11.071
Nan, X., Su, S., Ma, K., Ma, X., Wang, X., Zhaxi, D., et al. (2018). Bioactive fraction of Rhodiola algida against chronic hypoxia-induced pulmonary arterial hypertension and its anti-proliferation mechanism in rats. J. Ethnopharmacol. 216, 175–183. doi:10.1016/j.jep.2018.01.010
Nan, X., Yang, Z., Su, S., Huang, Z., Ma, K., Xu, S., et al. (2022). The mechanism of volatile oil of rhodiola tangutica against hypoxia-induced pulmonary hypertension in rats based on RAS pathway. Biomed. Res. Int. 2022, 9650650. doi:10.1155/2022/9650650
Noah, L., Morel, V., Bertin, C., Pouteau, E., Macian, N., Dualé, C., et al. (2022). Effect of a combination of magnesium, B vitamins, rhodiola, and green tea (L-Theanine) on chronically stressed healthy individuals-A randomized, placebo-controlled study. Nutrients 14, 1863. doi:10.3390/nu14091863
Page, M. J., McKenzie, J. E., Bossuyt, P. M., Boutron, I., Hoffmann, T. C., Mulrow, C. D., et al. (2021). The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ 372, n71. doi:10.1136/bmj.n71
Panossian, A. G., Efferth, T., Shikov, A. N., Pozharitskaya, O. N., Kuchta, K., Mukherjee, P. K., et al. (2021). Evolution of the adaptogenic concept from traditional use to medical systems: Pharmacology of stress- and aging-related diseases. Med. Res. Rev. 41, 630–703. doi:10.1002/med.21743
Pu, W.-L., Zhang, M.-Y., Bai, R.-Y., Sun, L.-K., Li, W.-H., Yu, Y.-L., et al. (2020). Anti-inflammatory effects of rhodiola rosea L.: A review. Biomed. Pharmacother. 121, 109552. doi:10.1016/j.biopha.2019.109552
Qi, Z., Qi, S., Ling, L., Lv, J., and Feng, Z. (2016). Salidroside attenuates inflammatory response via suppressing JAK2-STAT3 pathway activation and preventing STAT3 transfer into nucleus. Int. Immunopharmacol. 35, 265–271. doi:10.1016/j.intimp.2016.04.004
Qv, Q., Xu, J., and Li, S. (2014). [Efficacy of Rhodiola rosea injection in the treatment of acute exacerbation of chronic obstructive pulmonary disease. Chin. Med. Innov. 11, 14–16.
Rhodiola crenulata (Hook.f. & Thomson) H.Ohba. [WWW Document], (2023). Plants of the world online. URL available at: https://mpns.science.kew.org/mpns-portal/plantDetail?plantId=2416737&query=Rhodiola+crenulata&filter=&fuzzy=false&nameType=all&dbs=wcsCmp (accessed 3 14, 23).
Rhodiola crenulata (Hook.f. & Thomson) H.Ohba (2022). World floraonline. WWW Document. URL Available at: http://www.worldfloraonline.org/taxon/wfo-0000399351 (accessed 6 11, 22).
Rhodiola kirilowii (Regel) Maxim. [WWW Document], (2023). Plants of the world online. URL Available at: https://mpns.science.kew.org/mpns-portal/plantDetail?plantId=2416720&query=rhodiola+kirilowii&filter=&fuzzy=false&nameType=all&dbs=wcsCmp (accessed 3 16, 23).
Rhodiola wallichiana (Hook.) S.H.Fu (2023). Plants of the world online. WWW Document. URL Available at: https://mpns.science.kew.org/mpns-portal/plantDetail?plantId=2416913&query=Rhodiola+wallichiana&filter=&fuzzy=false&nameType=all&dbs=wcsCmp (accessed 3 14, 23).
Seeger, W., Adir, Y., Barberà, J. A., Champion, H., Coghlan, J. G., Cottin, V., et al. (2013). Pulmonary hypertension in chronic lung diseases. J. Am. Coll. Cardiol. 62, D109–D116. doi:10.1016/j.jacc.2013.10.036
Sun, S., Tuo, Q., Li, D., Wang, X., Li, X., Zhang, Y., et al. (2020). Antioxidant effects of salidroside in the cardiovascular system. Evid. Based Complement. Altern. Med. 2020, 9568647. doi:10.1155/2020/9568647
Tan, Y. (2014). Analysis of the efficacy of antioxidant treatment for chronic pulmonary heart disease in acute exacerbations of chronic obstructive pulmonary disease in highland areas. J. Beihua Univ. Nat. Sci. 15, 348–351.
Wang, X. (2010). [Effect of Rhodiola rosea on pulmonary artery pressure and its mechanism of action in patients with chronic pulmonary heart disease in highlands. J. High Alt. Med. 20, 36.
Williams, T. D., Langley, H. N., Roberson, C. C., Rogers, R. R., and Ballmann, C. G. (2021). Effects of short-term golden root extract (rhodiola rosea) supplementation on resistance exercise performance. Int. J. Environ. Res. Public Health 18, 6953. doi:10.3390/ijerph18136953
Xing, S., Yang, X., Li, W., Bian, F., Wu, D., Chi, J., et al. (2014). Salidroside stimulates mitochondrial biogenesis and protects against H₂O₂-induced endothelial dysfunction. Oxid. Med. Cell. Longev. 2014, 904834. doi:10.1155/2014/904834
Yang, S., Feng, E., Yan, Z., Yin, H., Zhang, 瑛., Huang, N., et al. (2013). [Study on antioxidant treatment of chronic pulmonary heart disease in combination with acute exacerbation of chronic obstructive pulmonary disease in highland areas. Chin. J. Lung Dis. Electron. Ed. 6, 30–34.
Yu, H. L., Zhang, P. P., Zhang, C., Zhang, X., Li, Z. Z., Li, W. Q., et al. (2019). Effects of rhodiola rosea on oxidative stress and negative emotional states in patients with obstructive sleep apnea. Lin. Chung Er Bi Yan Hou Tou Jing Wai Ke Za Zhi 33, 954–957. doi:10.13201/j.issn.1001-1781.2019.10.013
Yu, H., Su, X., and Lei, T. (2023). Effect of pulmonary hypertension on pulmonary function of patients with COPD: A systematic review and meta-analysis. [WWW Document]. PROSPERO. URL Available at: https://www.crd.york.ac.uk/PROSPERO/display_record.php?RecordID=302881 (accessed 3, 27, 22).
Zhang, R., Li, Z., Liu, C., Yang, Q., Lu, D., Ge, R.-L., et al. (2022). Pretreatment with the active fraction of Rhodiola tangutica (Maxim.) S.H. Fu rescues hypoxia-induced potassium channel inhibition in rat pulmonary artery smooth muscle cells. J. Ethnopharmacol. 283, 114734. doi:10.1016/j.jep.2021.114734
Zhang, Z., Tong, Y., Zou, J., Chen, P., and Yu, D. (2009). Dietary supplement with a combination of Rhodiola crenulata and Ginkgo biloba enhances the endurance performance in healthy volunteers. Chin. J. Integr. Med. 15, 177–183. doi:10.1007/s11655-009-0177-x
Keywords: Rhodiola L., chronic obstructive pulmonary disease, safety, efficacy, systematic review
Citation: Yu H, Lei T, Su X, Zhang L, Feng Z, Dong M, Hou Z, Guo H and Liu J (2023) Efficacy and safety of three species of Rhodiola L. in patients with chronic obstructive pulmonary disease: A systematic review and meta-analysis. Front. Pharmacol. 14:1139239. doi: 10.3389/fphar.2023.1139239
Received: 06 January 2023; Accepted: 20 March 2023;
Published: 06 April 2023.
Edited by:
Alexander N. Shikov, Saint-Petersburg State Chemical Pharmaceutical Academy, RussiaReviewed by:
Claudio Ferrante, University of Studies G. d'Annunzio Chieti and Pescara, ItalyReza Mohebbati, Mashhad University of Medical Sciences, Iran
Copyright © 2023 Yu, Lei, Su, Zhang, Feng, Dong, Hou, Guo and Liu. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Jian Liu, bWVkZWNpbmxpdUBzaW5hLmNvbQ==
 Haichuan Yu
Haichuan Yu Ting Lei
Ting Lei Xiaojie Su
Xiaojie Su Lu Zhang1,2
Lu Zhang1,2 Jian Liu
Jian Liu