- 1Earth and Environment Department, Boston University, Boston, MA, United States
- 2Systems Biology Department, Harvard University, Boston, MA, United States
- 3Sustainability Studio, Massachusetts College of Art and Design, Boston, MA, United States
Integrating cities with the surrounding environment by incorporating green spaces in creative ways would help counter climate change. We propose a rooftop farm system called BIG GRO where air enriched with carbon dioxide (CO2) produced through respiration from indoor spaces is applied through existing ventilation systems to produce a fertilization effect and increased plant growth. CO2 measurements were taken inside 20 classrooms and at two exhaust vents on a rooftop at Boston University in Boston, MA. Exhausted air was directed toward spinach and corn and plant biomass and leaf number were analyzed. High concentrations of CO2 persisted inside classrooms and at rooftop exhaust vents in correlation with expected human occupancy. CO2 levels averaged 1,070 and 830 parts per million (ppm), reaching a maximum of 4,470 and 1,300 ppm CO2 indoors and at exhaust vents, respectively. The biomass of spinach grown next to exhaust air increased fourfold compared to plants grown next to a control fan applying atmospheric air. High wind speed from fans decreased growth by approximately twofold. The biomass of corn, a C4 plant, experienced a two to threefold increase, indicating that alternative environmental factors, such as temperature, likely contribute to growth enhancement. Enhancing growth in rooftop farms using indoor air would help increase yield and help crops survive harsh conditions, which would make their installation in cities more feasible.
Introduction
Rooftops in cities are greatly underutilized areas occupying ~20–50% of urban aerial space (US EPA, 2008; Vaughan and Lenton, 2011; Shafique and Kim, 2017). If vegetated, these spaces could provide considerable environmental and social benefits (Shafique et al., 2018b). Rooftop gardens and farms can mitigate the urban heat island effect by decreasing local temperatures (Ismail et al., 2012; Kleerekoper et al., 2012; Santamouris, 2014; Coutts et al., 2015; ArrowStreet Architecture Design., 2016), increasing storm water retention and precipitation release through evapotranspiration, which decreases flooding (Carter and Rasmussen, 2007; Nagase and Dunnett, 2012; Whittinghill et al., 2015; He et al., 2016; Nitsch Engineering, 2016; Shafique et al., 2018a), providing air pollutant filtration (Rowe, 2011), and decreasing building energy use through increased insulation (Wong et al., 2003; Garrison et al., 2012; Saadatian et al., 2013) and natural cooling (Batchelor et al., 2009; Garrison et al., 2012; Saadatian et al., 2013). Rooftops also offer economic and community building opportunities, aesthetic and mental health benefits (Guite et al., 2006; Johnson et al., 2018), and food security when used for urban agriculture (Oberndorfer et al., 2007; Orsini et al., 2014; Ahmed et al., 2017). Urban food production would decrease dependency on external communities and carbon intensive agricultural systems, increase the redundancy and resiliency of our agricultural system, and address climate change by helping with climate enhanced negative urban environmental impacts and harvesting CO2 (Oberndorfer et al., 2007; Davies et al., 2011; Rowe, 2011; Ismail et al., 2012; Orsini et al., 2014; Whittinghill et al., 2014; Shafique et al., 2020). Decreasing differences between urban and non-urban areas could decrease negative impacts of cities on both urban ecosystems and populations.
A major challenge facing rooftop gardens and farms, hereafter called rooftop farms, on top of increased structural loads is decreased plant growth due to extreme environmental conditions such as higher wind speeds and temperatures, heightened solar radiation, and limited soil moisture content (Ahmed et al., 2017). Enhancing growth in rooftop farms could make them a more viable option and increase the rate of installation in cities. Inducing a CO2 fertilization effect on rooftop vegetation using the CO2 produced in buildings would produce this increased growth effect. When elevated concentrations of CO2 are present, the CO2 fertilization effect increases photosynthetic efficiency mostly linearly at concentrations between 400 and 1,000 ppm (Rogers et al., 1994; Ainsworth and Long, 2005) by decreasing photorespiration. Decreasing photorespiration can increase the amount of carbon taken in by photosynthesis by up to 25% (Sharkey, 1988). The first generation of Free Air Carbon Dioxide Enrichment (FACE) experiments confirmed the existence of the CO2 fertilization effect by administering higher CO2 concentrations to plants within open systems (Kimball et al., 1997; McLeod and Long, 1999; Long et al., 2006; Ainsworth et al., 2008; Leakey et al., 2009; Norby and Zak, 2011). The next generation of FACE experiments further explored this effect (Norby et al., 2016). The effect is strongest between the concentrations of 400 and 1,000 ppm when the increase in growth rate begins to level off as other factors become limiting (Rogers et al., 1994). Most commercially bought CO2 is expensive and produced from fossil fuels. Multiple of the twelve initial FACE experiments are no longer running, in part due to the high cost of purchasing and transporting condensed CO2 (Hendrey et al., 1993; Reece et al., 1995; Miglietta et al., 1997; Ainsworth and Long, 2005; Chakrabarti et al., 2012).
In this paper, we propose a system for Building-Increased Growth in a GreenROof (BIG GRO), that could enhance growth in rooftop farms. To help ameliorate the environmental challenges of rooftops, BIG GRO uses indoor air with CO2 generated from human respiration inside buildings to enhance growth. The average modern-day person spends the majority of their time inside buildings and continuously respires large quantities of CO2 (Lee and Chang, 1999; Apte et al., 2000; Jin et al., 2015). CO2 is typically exhausted from large buildings through Heating, Ventilation, and Air Conditioning (HVAC) systems and exhaust vents on rooftops, where it can directly be applied to rooftop farms. Using waste CO2 from humans makes rooftop farming more feasible than regular rooftop farms due to a higher yield and more cost effective by providing concentrated CO2 in a more sustainable manner (Hendrey et al., 1993; Reece et al., 1995; Chakrabarti et al., 2012). The BIG GRO system leverages the advantages of using waste CO2 and employs the CO2 in facilitating enhanced plant production in rooftop farming.
We hypothesized that when there is a large build-up of CO2 indoors, sufficient CO2 is released from exhaust vents to affect crop growth in rooftop farms, and that plants exposed to exhaust vent air grow larger than plants not exposed to building exhaust. To test this hypothesis, we monitored the “Building Metabolism,” the sources, sinks, and fluxes of CO2 within and out of buildings and atop rooftops (Sanyé-Mengual et al., 2014; Pons et al., 2015). Crops were grown next to exhaust vents to test for a CO2 fertilization effect. Both C3 and C4 crops were grown to attain an initial understanding of whether factors besides CO2 impact growth assuming C4 plants would respond less to CO2 (Ainsworth and Rogers, 2007; Hatfield et al., 2011). We further hypothesized C4 plants would still experience increased growth, indicating the importance of environmental factors besides CO2, such as temperature, in the growth effect. This study provides initial support for a conceptual system that creates a more circular carbon process within buildings, taking high concentrations of CO2 from human respiration (Barrett et al., 2012) and moving it to a rooftop farm where it can be applied for a functional use (Hanson et al., 2000) to produce food that can be eaten by humans and respired anew (Figure 1). Enhancing plant growth would make rooftop farms more productive and potentially capable of surviving harsher conditions, expanding their viability as an urban greening strategy.
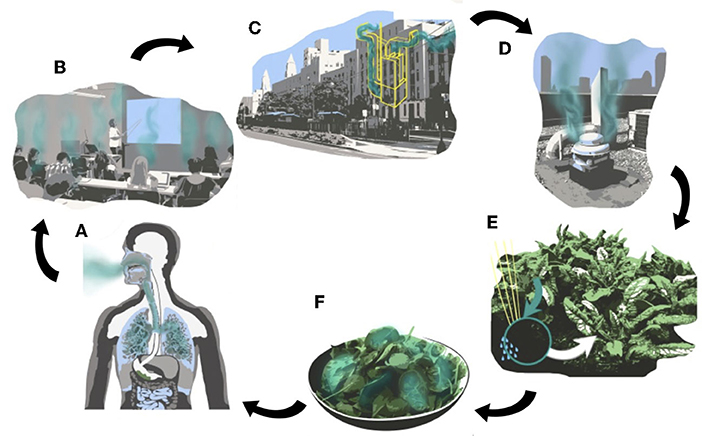
Figure 1. Conceptual diagram showing the carbon cycle within experimental rooftop farms. In the more circular part of this system, CO2 travels (A) from the human body (B) out into the classroom within a building. This CO2 then is brought (C) through the ventilation system to the rooftop and (D) released through an exhaust vent. BIG GRO will apply this CO2 to (E) plants in a rooftop farm after which (F) humans can consume the crops and the carbon can return to the human body.
Materials and methods
Experiments were carried out in Boston, MA in the fall of 2018 and the spring of 2019. CO2 concentrations and other environmental measurements were taken inside of classrooms and at the rooftop exhaust vents. The vent air was directed at the crops, Spinacia oleracea (spinach) and Zea mays (corn), and the effect on growth was measured.
CO2 measurements
Classroom CO2 measurements
From January 2018 through March 2019, CO2 monitors (Onset HOBO Bluetooth Low Energy Carbon Dioxide—Temp—RH Data Logger, #MX1102) were installed in 20 classrooms throughout the College of Arts and Sciences (CAS) Building at Boston University. Sensors were calibrated according to manufacturer's guidelines, to each other, and were found to be within ±50 ppm of a 400 ppm standard compressed air source (AirGas, Rendon, PA). The concentration of 400 ppm is close to the atmospheric background CO2 concentration of 410 ppm at the time of the study (Ng et al., 2019). Sensors were placed within classrooms for a week to capture the dynamics throughout a normal workweek and over the weekend when different usage patterns were expected. 25Live (CollegeNET, Portland OR) scheduling software was used to gather occupancy data along with room size for 20 classrooms for the weeks during which the CO2 sensors were installed.
Rooftop CO2 measurements
The CO2 sensors were also installed on vents on the rooftop of a two story high school on campus, the Boston University Academy (BUA). Two sensors were placed on Direct Drive Centrifugal Roof Exhausters Model PRN (ACME Engineering and Manufacturing Corporation, Muskogee, OK) and two were placed on control fans of the same type installed at separate locations on the BUA rooftop. We chose these vents, commonly referred to as mushroom vents, because of their prevalence on rooftops and the higher than normal CO2 concentrations found in their exhaust air in comparison with other vents (Supplementary Text 1). Through discussions with facilities and inspection of building plans, these vents were also identified as general exhaust from internal human-occupied spaces.
Sensors were first installed at the exhaust vents for 4 weeks from June 25th to July 22nd, 2018 to understand dynamics during periods of time when there were varying amounts of people in the building. Information regarding the schedule of the building was obtained. During the first 2 weeks, only instructional staff (no students) were present during the day, which consisted of 10–15 people. During the second 2 weeks, a camp program was run with around 175 students attending classes regularly throughout the day.
The rooftop sensors were also run throughout the rooftop farm experiments. During this time, a sensor was installed within a bathroom on the second floor of the BUA. Blueprints for the design of the building indicated that this location was most likely the closest point within the building to where the exhaust vents were connected. These vents could be less directly connected to classroom CO2, but given general circulation and diffusion, were assumed to be representative of overall CO2 concentrations within a building.
Rooftop farm experiment
Study species
Spinach (S. oleraceae L.), is an economically and nutritionally important crop (Min et al., 2014; Reddy et al., 2014). It has a high nutritional value (Kuti and Konuru, 2004) and notable quantities of secondary chemicals (Nuutila et al., 2002; Bunea et al., 2008; Shohag et al., 2011; Buckley et al., 2021), which protect against chronic diseases (Howard et al., 2005). It is a cold-season crop from southwestern Asia (Candlish et al., 1987), a property we valued because all plant growth experiments were run in the fall and spring seasons, a time of cold weather in Boston, MA. The fall and spring seasons were selected due to them coinciding with the period when the largest number of people are present in university and high school buildings. The study required people to produce CO2 through respiration to test the impact of CO2-enriched exhaust vent air on plant growth. Spinach also utilizes the C3 photosynthetic metabolic pathway, which is more responsive to elevated CO2 (Kimball et al., 2002; Nowak et al., 2004).
Corn (Z. mays) was grown in the spring to explore the effect of other characteristics of the exhaust vent air such as temperature. Corn is originally from Central America and is the most widely planted crop in the United States with 31.9 million ha planted in 2002 (Kadam and McMillan, 2003). It has less tolerance for cold than spinach (Warrington and Kanemasu, 1983), but is known to use a C4 carbon fixation strategy, which makes it less sensitive to increased CO2 concentrations than C3 species (Ainsworth and Rogers, 2007; Hatfield et al., 2011) and therefore might responds more strongly to other environmental characteristics of the exhaust vent air besides CO2 (Leakey et al., 2004; Long et al., 2006; Ainsworth and Rogers, 2007).
Experimental set up
Plants were grown directly around the two Direct Drive Centrifugal Roof Exhausters that were actively and continuously exhausting air to the rooftop and around the two control fans (Figure 2B). The control fans tested the effect of the fans alone without exhaust vent air from inside of the building. Hereafter these treatments are called “Exhaust Vents” and “Control Fans”. In the spring there was also treatment called “Control” with a fan that was not turned on as an extra control for wind speed. A subset of the environmental data between 8:00 AM and 4:00 PM was selected since this was the time people were expected to be inside the building, as well as when plants undergo photosynthesis and take up CO2. Daytime Exhaust Vent air was similar to Indoor Air and different from Control Fan air as shown by CO2, temperature, and relative humidity measurements (Supplementary Figure 1). CO2 measurements averaged 758 ± 3.7 ppm at the Exhaust Vents, 512 ± 2.8 ppm Inside, and 454 ± 2.5 ppm at the Control Fan. None of the 95% confidence intervals for the Exhaust Vent (765.1–751.0), Indoor measurements (517.6–506.8), and Control Fan (459.4–450.0) overlapped. The Exhaust Vent was most different at 67% higher than the Control Fan compared to Indoor measurements which were 13% higher than the Control Fan. Temperature measurements averaged 22.22 ± 0.02°C at the Exhaust Vent, 21.11 ± 0.018°C Inside, and 14.44 ± 0.14°C at the Control Fan. Since these measurements were taken in the fall, indoor temperatures were higher at the Exhaust Vent and Inside to compensate for cold outdoor temperatures. Confidence intervals for the Exhaust Vent (22.31–22.38°C), Inside (21.03–21.10°C), and Control Fans (14.62–14.09°C) showed a greater difference than with CO2 with Exhaust Vent and Indoor measurements being 25 and 21% larger than Control Fan air. Relative humidity measurements averaged 45% ± 0.38 at the Exhaust Vent, 45% ± 0.40 Inside, and 68% ± 0.45 at the Control Fan. This was also reflected in the confidence intervals for Exhaust Vent (45.8–44.3), Indoor measurements (45.7–44.1), and Control Fan (68.9–67.1), which overlapped for Exhaust Vent and Indoor measurements and were 33.7 and 33.9% over the Control Fan measurements. These differences created the Exhaust Vent treatment experienced by the plants grown next to the fans in the Fall 2018 (Supplementary Figure 1) and Spring 2019 (Supplementary Figures 2,3).
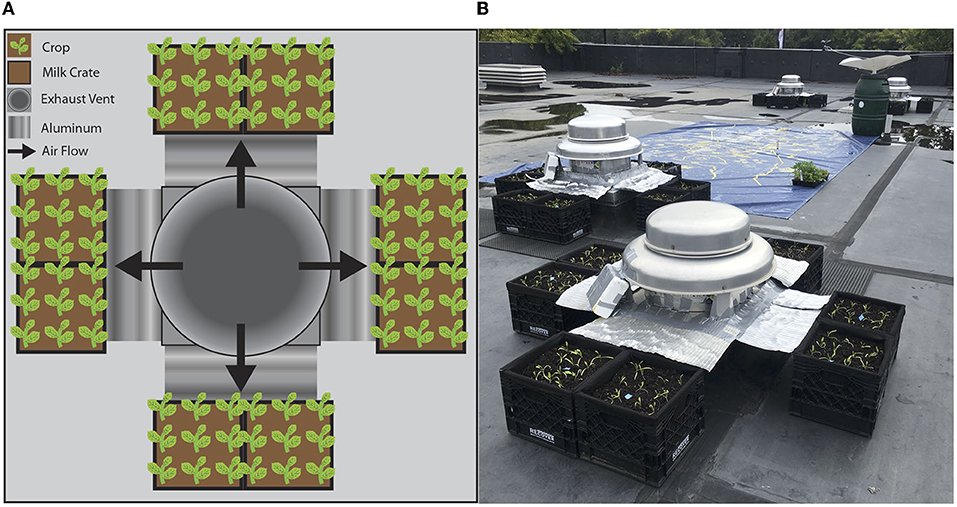
Figure 2. Experimental garden set up around rooftop Exhaust Vents and Control Fans. This shows (A) a diagram of the experimental set up at one vent/fan and (B) a picture of the actual set up. Spinach and corn were planted in milk crates provided by Recover Green Roofs positioned next to various vents for four and a half weeks until harvest after which growth was measured.
Plants were grown in milk cartons, which are large, mobile, and relatively accessible. Milk crates are also the primary container used by current installers of rooftop farms in Boston and across the region, such as Recover Green Roofs (www.recovergreenroofs.com). Recover Green Roofs has developed a RAMM (Recover Aerated Media Module) design with a non-woven Polypropylene Liner with organic compost-based potting mix (Roofs, 2018). Recover Green Roofs graciously lent the milk crate and felt portion of their RAMMs to maintain a system similar to four of the six rooftop farms they have built in Boston. Eight milk crates were placed in pairs around the four sides of each fan for a total of 32 milk crates.
We used an open system to apply the vent air to avoid backpressure. Curved aluminum structures (Figure 2A) were attached around the base of the vent to direct the vent air toward the plants. Aiming the vent air above the media avoided drying the media out, a common issue in rooftop farms (Ahmed et al., 2017). Soil moisture was measured in each milk crate twice a week using a handheld Soil Moisture Meter (Vegetronix, Digital VG-Meter-200). An anemometer (HOLDPEAK 866B Digital Anemometer Handheld) was used to determine the wind speed of the exhaust vents. The wind speeds of the two exhaust vents were ~4.47 and 7.60 m s−1. It was not possible to change the speed of either of these fans. Therefore, the speed of the two control fans were adjusted to reflect these wind speeds.
Plant growth
In the fall of 2018, spinach seeds (Bloomsdale, Long standing, USDA organic) were purchased from Mountain Valley Seed Co. and planted in starter trays for 4 weeks in a classroom in BUA with a large south facing window. They were watered from above approximately every other day until ready to be transplanted on the roof. Plants were hardened for a week before being transplanted by being moved onto the rooftop for increasing amounts of time. The spinach was then moved permanently up to the rooftop and 12 spinach plants, three rows of four, were planted in each of the 32 boxes. The experiment produced a total of 384 plants with 96 in each of four treatments, with each treatment distributed among eight boxes. In spring 2019, the same experiment was done except with corn seeds (Trinity Organic F1) purchased from Johnny's Selected Seeds (Winslow, ME). Nine corn sprouts, three rows of three, were planted in 12 boxes for a total of 81 plants, 27 in each treatment. This represents pseudo replication with multiple plants and boxes surrounding only two exhaust vents and two control fans, each with a different wind speed (Ellsworth et al., 1996) but still provides preliminary insight into an Exhaust Vent effect. In the spring only the low wind speed exhaust vent and control fan was used along with a second control group where the fan was not turned on to further test the effect of the wind speed on the plants. Nature's Care potting soil, and organic, compost-based soil similar to the potting mix in the Recover Green Roof RAMM system was used. After one full day outside, the boxes were moved a foot away from the edge of the vents and fans at the end of the metal sheets directing the air. Plants were watered approximately every other day if it had not rained for three consecutive days.
Crop performance
At each harvest one row of three plants was harvested. Plants were cut at the base just below the soil surface and immediately weighed to measure the wet weight of biomass. All plants were then frozen and kept at −80°C until they were placed within a lyophilizer and dried for 3 days. At the end of this process, plants were again weighed to find the dry weight of biomass. In the fall, three harvests were carried out a week and a half apart and in the spring all plants were harvested at the end of the four and a half weeks. Each week the number of leaves was counted on each plant. A leaf was counted once it had unfolded and the petiole was seen extending from the center of the spinach plant.
Statistical analysis
All statistical analyses and data visualization were performed in R (Version 3.4.4). For a subset of 5 classrooms in the CAS where CO2 concentrations were measured, the scheduling data were merged with the CO2 data. The class times were then highlighted on the CO2 graphs in order to determine overlap of CO2 spikes and class times. Overall effects of the different treatments of air from exhaust vents and control fans on the wet and dry biomass and leaf number of the crops were analyzed with an ANOVA with each plant as the experimental unit. The treatment was the independent variable and the biomass or leaf number the dependent variable. The multiple comparisons using least squares means Tukey's HSD method was used to identify significant differences between treatments. This same analysis was done to determine differences between the effect of different speeds at the four fans. An ANOVA was used for analysis of the environmental characteristics though averaging environmental data over time ignores non-linear impacts on trends. To further characterize these data, 95% confidence intervals and effect size were also calculated.
Results
CO2 measurements
Classroom CO2 measurements
CO2 in classrooms varied highly between day and night with spikes in CO2 occurring during scheduled class times. In general, classes ran between 8:00 am and 4:00 pm. During this time CO2 increased dramatically and maintained high concentrations relative to background CO2 (Figure 3). Throughout all classrooms, CO2 was above 1,000 ppm 37% of the time with an average concentration of 1,060 ppm and maximum of 4,470 ppm. CO2 levels dropped closer to atmospheric concentration around 410 ppm (Ng et al., 2019) by the end of the day (4:00 pm). Weekend CO2 concentrations were more variable (Supplementary Figure 4). Based on the subset of classes that were compared with their schedules, increases and decreases in CO2 concentrations coincided directly with class times (Figure 3). CO2 levels increased during class times though the range of CO2 concentrations appeared to vary based on the classroom (Supplementary Figure 5). This was dependent on number of students and classroom size, which were both provided by the 25Live scheduling data (Supplementary Figure 6).
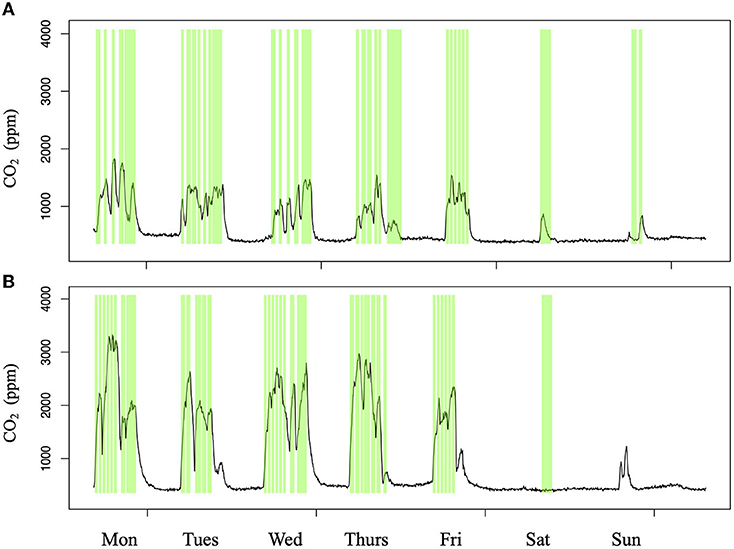
Figure 3. Two examples of classroom CO2 measurements over a week compared with class times, out of 20 classrooms monitored. CO2 sensors (Onset HOBO Bluetooth Low Energy Carbon Dioxide—Temp—RH Data Logger, #MX1102) were installed in classrooms for 1 week. Occupation data was collected for these classrooms from the 25Live scheduling system used by BU. Class times (green boxes) were plotted against CO2 concentrations (black lines) over time to compare CO2 spikes with classroom usage. Spikes occurred primarily during class time in classrooms (A) CAS 201 and (B) CAS 213.
Rooftop CO2 measurements
CO2 from rooftop Exhaust Vents increased when summer camp classes were being held inside the buildings. The measurements taken from the Exhaust Vents over the summer during the first 2 weeks when very few people were in the building were consistently close to atmospheric concentrations, ~410 ppm, and no clear temporal pattern was identified (Figure 4A). Once a camp began during the second 2 weeks of measurements and the number of people in the building increased to ~175 people, a clear pattern became visible. CO2 concentrations approached similar levels as seen within the classrooms (Figure 4B) though they were overall lower most likely due to diffusion and leakage in the system. Over the second 2 weeks of these measurements, Exhaust Vent CO2 stayed above 1,000 ppm 10% of the time with an average daytime concentration of 830 ppm and maximum of 1,300 ppm. Similar to measurements indoors, CO2 concentrations dropped to atmospheric levels around 410 ppm at the end of the day and stayed lower over the weekend. During the rooftop farm experiments, these patterns remained true with CO2 concentrations from the Exhaust Vents during the week being increased to around the same levels as described above (Supplementary Figure 1). These correlated well with the indoor CO2 measurements taken in the second floor bathroom (Supplementary Figure 7A).
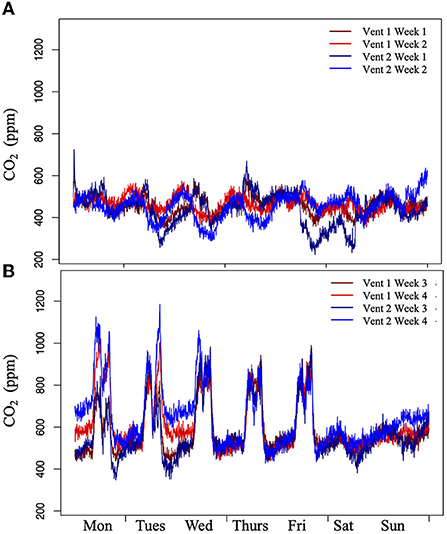
Figure 4. CO2 released from rooftop vents on a building while unoccupied and occupied over 4 weeks. Sensors were attached to two vents on The BUA roof for 4 weeks. (A) During the first 2 weeks, only baseline staff were present and (B) during the second 2 weeks a camp was being run. There were no CO2 peaks in the first 2 weeks and peaks up to 1,300 ppm during the second 2 weeks indicating the connection between Exhaust Vent CO2 and building occupancy.
Rooftop farm crop performance
Wind speed effect
In Fall 2018, there was a significant difference between growth of spinach at the high and low speed fans for both dry (F(1, 1) = 14.96, p < 0.001) and wet (F(1, 1) = 25.98, p < 0.001) biomass, with plants grown next to the higher speed fans (17 mph) significantly smaller than those grown next to the lower speed fan (10 mph) for the Exhaust Vents (Figure 5A; p < 0.001, p < 0.001; Supplementary Figure 8A). The growth differential next to both Control Fans was too low for a difference to be detected (p = 0.96, p = 0.99). Even at the higher wind speed, the growth enhancement effect of the Exhaust Vent still significantly increased dry and wet biomass compared to growth next to both the Control Fans at high (p = 0.0058, p = 0.011) and low speeds (p = 0.0028, p = 0.043). This difference can be observed in representative pictures of the four treatments (Figure 6). To isolate only the exhaust fan effect, going forward, only the data from the plants surrounding the lower speed fans were used.
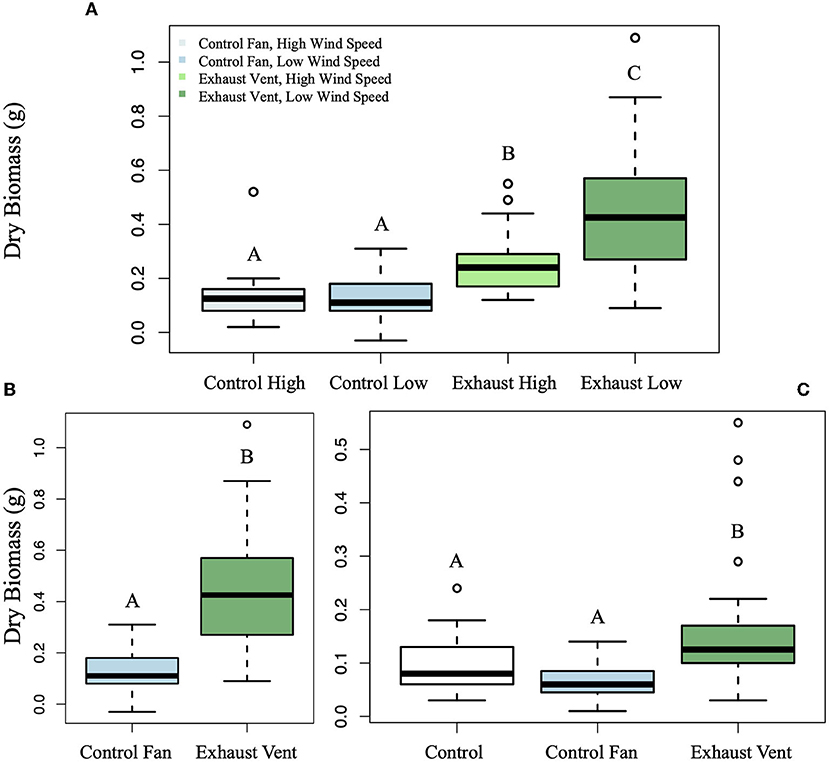
Figure 5. Average dry weight of spinach and corn plants in response to Exhaust Vent air. (A) Spinach was grown in Fall 2018 next to two fans at a higher wind speed (17 mph) and two at a lower speed (10 mph) and one of each of these was a Control Fan and Exhaust Vent. Between the Exhaust Vents, the spinach plants exposed to high wind speeds were smaller (p < 0.001). Even with this, the Exhaust Vent was able to recover growth compared to both the low (p = 0.0058) and high-speed (p = 0.0028) Control Fans. At only low wind speeds there was a significant increase in (B) dry biomass (p < 0.001) with exposure to exhaust air. These data were from harvest 3. (C) Corn grown in Spring 2019 next to the Exhaust Vent was larger than at both the Control Fan (p < 0.001) and control garden (p = 0.0045). Different capital letters indicate statistically significant differences among treatments.

Figure 6. Representative picture of spinach growth next to high and low wind speed exhaust vents and exhaust and control air treatments. (A) Spinach grown next to an exhaust vent and (B) next to a control fan both at low wind speed. (C) Spinach grown next to an exhaust vent and (D) next to a control fan both at high wind speed. The images for each group were ranked by size and the median image is shown here.
The same trend was found for leaf number. Even with wind effects, leaf number was higher for plants surrounding the Exhaust Vent than for plants surrounding the Control Fan (Supplementary Figure 9A; F(1, 1) = 23.64, p < 0.001). The number of leaves grown next to the Exhaust Vent at the higher speed was less than at the Exhaust Vent at the lower speed (p < 0.001), but the difference between leaf number at the high and low speed Control Fans was not significant (p = 0.25). The Exhaust Vent effect did still lead to more leaves being produced at the high speed Exhaust Vent in comparison to the high speed Control Fan (p = 0.0017). However leaf number at the high speed Exhaust Vent was not significantly different than the leaf number next to the low speed Control Fan (p = 0.23).
Biomass and leaf number
In Fall 2018, spinach biomass increased and more leaves grew next to Exhaust Vents (Figure 6). The dry biomass (Figure 5B; F(1, 1) = 56.87, p < 0.001), wet weight (Supplementary Figure 8B; F(1, 1) = 57.69, p < 0.001), and leaf number (Supplementary Figure 9A; F(1, 1) = 41.11, p < 0.001) of the spinach next to Exhaust Vents were significantly greater than those of the spinach grown next to the Control Fan. The average dry weight of plants next to the Exhaust Vent was 0.45 g ± 0.013 (±SE) compared to 0.12 ± 0.041 at the Control Fan. The average leaf number of plants next to the Exhaust Vent was 7 ± 0.08 compared to 5 ± 0.15 at the Control Fan. Dry biomass around the Exhaust Fan was almost four times larger than dry biomass around the Control Fan with means of 0.45 ± 0.04 g and 0.12 ± 0.13 g.
During Spring 2019, there was a significant difference in the corn dry and wet biomass (Figure 5C; F(1, 2) = 10.13, p < 0.001, p < 0.001) with the growth next to the Exhaust Vent being significantly higher than at both the Control Fan and the Control for both dry (p < 0.001, p = 0.0045) and wet (Supplementary Figure 8C; p < 0.001, p = 0.012) biomass. The same trend was found for leaf number (Supplementary Figure 10A). There was also a difference in color between the treatments (Supplementary Figure 11) with corn next to exhaust vents appearing greener than corn next to the control fans which appeared more yellow. The corn was between 2 and 3 times larger at the Exhaust Fan vs. the Control Fan with means of 0.17 ± 0.005 g and 0.065 ± 0.008 g.
Environmental measurements
For soil moisture, data were taken once a week, but only data from the last day of data collection, on the final harvest, were analyzed to be consistent with biomass and leaf number measurements. In both Fall 2018 and Spring 2019 no significant differences were found between the soil moisture measurements at the Control Fans and Exhaust Fans (Supplementary Figures 9B,10B). Frost covering the rooftop in November showed an ~2 m outline around the exhaust vents where the warmer indoor air melted the frost (Supplementary Figure 12A) in contrast to around the control vents (Supplementary Figure 12B) where no outline was seen giving a preliminary indication of the spatial extent of the effect.
Discussion
This study investigated the functionality of the BIG GRO system, which takes advantage of waste CO2 built up from human respiration indoors and applies it to plants in a rooftop farm in order to enhance crop growth. CO2 concentrations inside classrooms were confirmed to be elevated, as well as at exhaust vents on the rooftop, particularly when large numbers of people occupied the building. Plants grown adjacent to these exhaust vents grew larger with the application of indoor air. This supports the potential of BIG GRO as a new way to enhance both growth and survival of crops in rooftop farms.
Building metabolism
In these buildings, humans drove building metabolism by being the largest contributor to CO2 concentrations, leading to high levels both indoors (Figure 3) and in exhaust vent air (Figure 4). CO2 measurements are often used in the context of health and energy efficiency, with governmental agencies recommending CO2 concentrations remains below 1,000 ppm (Apte et al., 2000; ACGIH American Conference of Governmental Industrial Hygienists, 2011; Persily, 2020) or even 800 ppm (MADPH, 2022). High levels of CO2 are associated with increased levels of other indoor air pollutants (Lee and Chang, 1999; Seppanen et al., 1999; Apte et al., 2000) and directly decrease cognitive function (Rice, 2004; Zhang et al., 2018) even when as low as 1,000 ppm CO2 (Satish et al., 2012; Allen et al., 2016; Persily and de Jonge, 2017). Removing CO2 from classrooms is beneficial for humans and this study highlights that the same CO2 can act as a resource for plants in rooftop farms. The human source of waste CO2 can be repurposed and the rooftop farm can both act as a sink for CO2 and recycle that CO2 into crops humans can eat (Figure 1).
Plant growth enhancement
The overall CO2 patterns found for exhaust air (Figure 4) indicate that it could be used opportunistically to induce CO2 fertilization and act as a resource for plant growth enhancement. CO2 in exhaust vents was between 500 and 1,000 ppm, the relevant levels for CO2 fertilization (Rogers et al., 1994). The increased leaf number provides further support for the occurrence of CO2 fertilization since plant physiology has been found to alter at higher CO2 concentrations (Fleisher et al., 2008). BIG GRO does not apply a constant stream of increased CO2. It opportunistically takes advantage of the fact that CO2 is higher during the day in non-residential buildings. The daytime hours coincide with when plants require CO2 for the light phase of photosynthesis and is the most relevant time for CO2 fertilization (Calvert and Slack, 1976; Miglietta et al., 1997; Moore et al., 1999; Edwards et al., 2001; Leakey et al., 2009). BIG GRO is also opportunistic by redirecting air already being expelled through exhaust vents toward plants. It avoids complicated and energy intensive processes such as condensing the CO2 or controlling application to produce a constant CO2 source, which would alter the overall carbon footprint of BIG GRO. This makes the BIG GRO system cheaper and simpler to construct, allowing for wider implementation, especially in lower resource settings.
Factors aside from CO2, such as temperature, precipitation, etc., are also known to affect plant growth (Nijs et al., 1997; Zvereva and Kozlov, 2006; Cai et al., 2016). The inclusion of corn in this study provides an indication that CO2 is one of multiple factors driving the growth enhancement effect. C4 plants should have a weaker response to increased CO2 concentrations (Sharkey, 1988; Kimball et al., 2002; Nowak et al., 2004; Long et al., 2006; Ainsworth and Rogers, 2007; Hatfield et al., 2011). This is because C4 metabolism is a photosynthetic process specifically designed to increase CO2 uptake efficiency, so even at low atmospheric concentrations of CO2 internal CO2 concentrations are already higher (Ainsworth and Rogers, 2007). In this study, both spinach and corn increased in growth (Figure 5), implying other environmental factors such as temperature could be enhancing plant growth. Both spinach and corn are sensitive to temperature and have approximate optimal temperatures of 20°C for spinach and 30°C for corn (Boese and Huner, 1990; Yamori et al., 2006), so the buffering effect of applying heated or cooled internal air would theoretically assist in avoiding extreme temperatures. A full factorial experiment that controls for different variables, such as CO2 and temperature, could separate these effects. While our study was not able to separate these due to its observational design, we developed a proof of concept experiment by looking at the overall effects of ventilation air on crops.
BIG GRO system design and implementation considerations
As found here and in other studies, high windspeed can be detrimental to plant growth (Onoda and Anten, 2011). However, wind speed will also play a role in moving the exhaust vent air across the rooftop to increase the extent of the growth effect. The largest impact of BIG GRO would be realized by applying exhaust air across an entire rooftop area. Therefore, optimizing the physical application of exhaust vent air in regard to appropriate windspeed and spatial extent of application would greatly contribute to the BIG GRO design.
If BIG GRO were optimized, it could also be used as an inexpensive method for conducting future FACE studies that use a free waste source of CO2 on topics not yet fully understood. Open questions remain such as how to optimize production (Ainsworth and Long, 2005; Ainsworth et al., 2008), the effects of multiple environmental factors (Nijs et al., 1997; Cai et al., 2016), and whether or not the CO2 fertilization effect decreases the nutritional content of crops (Idso and Idso, 2001; Loladze, 2002; Myers et al., 2014; Medek et al., 2017). This last question is also relevant in BIG GRO since the response of plant nutrients and secondary chemicals grown in this system might be different than in other FACE studies as there are differences in nutrient cycling in cities when compared to rural areas, undisturbed systems and other agricultural sites (Rao et al., 2014; Templer et al., 2015; Decina et al., 2016). Urban areas have higher CO2 concentrations, temperature, ozone, nitrogen, and other environmental factors (Briber et al., 2015), which could change plant responses (Kangasjärvi et al., 2005; Prajapati, 2012; Rao et al., 2014). The effects of increased CO2 concentrations in urban systems compared to different biomes could be examined. While this study focuses on urban environments, it is important to note that this concept could also be applied to buildings in rural areas if they reach a suitable capacity on a regular basis.
A related project based in Barcelona Spain described an Integrated-Rooftop Greenhouse (i-RTG), rather than a rooftop farm, which similarly drew on indoor air being directed into the greenhouse to assist plant growth (Sanyé-Mengual et al., 2014; Nadal et al., 2017a; Sanjuan-Delmás et al., 2018a,b). Although they did not measure growth in individual plants (Sanjuan-Delmás et al., 2018a,b), indoor air brought to the greenhouse created more hospitable temperatures for growing plants (Nadal et al., 2017b). These two systems further support the relevance of using indoor air as a resource. This study contributes information on increased CO2 since levels of CO2 were not high enough in i-RTG to test for a CO2 fertilization effect, but this was a part of their consideration. BIG GRO highlights the impact on the larger carbon cycle considering the potential carbon sink in a farm. It is also a cheaper system with a relatively small carbon footprint since it does not require larger structures and ideally would use less fertilizer (Vadiee and Martin, 2013; Pons et al., 2015; Sanyé-Mengual et al., 2015a; van Beveren et al., 2015; Sanjuan-Delmás et al., 2018a,b).
Calculating the environmental impact of a scaled up farm
Recycling carbon in the BIG GRO system could help improve a building's overall carbon footprint in multiple ways. Carbon would be directly stored in the plants and soil in the garden. Whittinghill et al. (2014) found that stored carbon varied based on species with herbaceous plants and grasses holding an average of 68.2 kg C m−2 compared to 0.38 kg C m−2 in sedum gardens (Getter et al., 2009). Even considering estimated embodied carbon from construction, rooftop farms are an overall sink over the first few years (Getter et al., 2009; Whittinghill et al., 2014). CO2 emissions from the building could be avoided through decreased energy use (Oberndorfer et al., 2007; Batchelor et al., 2009; Garrison et al., 2012; Saadatian et al., 2013) primarily from 10 to 43% decreases in air conditioning use due to higher albedo and extra cooling from the presence of the garden (Meier, 1990; Garrison et al., 2012), or avoided transport of food from rooftop farms (Pirog et al., 2001; Halweil, 2002; Lower and Restaurant, 2014). Pirog et al. (2001) found produce brought to Chicago travels an average of 1,518 miles and that using local food from the surrounding area reduced the associated carbon emissions from travel by 5–17 times. Studies from the i-RTG project, which contains more carbon costs associated with infrastructure, found the environmental impact of their produce would be lower from decreased packaging and transportation (Sanyé-Mengual et al., 2015a,b).
If implemented widely, a farm integrated into a rooftop farm could make a significant contribution to food requirements for urban communities. Orsini et al. (2014) looked at vegetable production in rooftop farms in a field study conducted to optimize the amount of crops that could be produced on one rooftop. They used their estimate to scale up across the city of Bologna, Italy by identifying all available flat rooftop space for farming and determining that 77% of produce used by the city could be grown on rooftop farms within city limits. Assuming similar increases in yield could be found for different crops across the growing season in different environmental conditions, if BIG GRO were applied in this context, 86–144% of required vegetables could be produced. The results would depend on the number of vents on the buildings and the spatial extent of their effect (Supplementary Table 1). Using the growth rates from Orsini et al. (2014), this concept can be further expanded to Boston M.A. where an estimated 190% of required vegetables could be produced. By adding exhaust vent application systems, 207–290% of Boston's vegetable requirement could be produced (Supplementary Text 4, Supplementary Table 1). This indicates that adding rooftop farms to rooftops within a city could contribute a substantial amount of produce to the overall needs of the city.
Conclusion
As the climate continues to change and the world's population continues to increase, levels of CO2 are reaching new highs and crowded cities are facing extreme weather conditions. Leveraging underutilized rooftop areas to grow crops will help overcome many environmental, economic, and social challenges cities around the world currently face. To our knowledge, this is the first study to take indoor air with elevated CO2 concentrations and apply it to plants grown in a rooftop farm or garden to test for a growth enhancement effect. The enhanced growth found is consistent with a CO2 fertilization effect as characterized by FACE experiments though it also indicates the influence of other environmental characteristics of the exhaust vent air, such as temperature, in increasing growth. BIG GRO is an innovative, sustainable and scalable system for bringing the CO2 fertilization effect in to rooftop gardens. BIG GRO takes advantage of a waste resource, human respiration, a sustainable resource that exists in excess in cities. Implementing this approach on rooftops across cities and increasing overall urban vegetation through farming will help address some environmental challenges facing cities including producing hyper local food more efficiently and sustainably, harvesting carbon, and helping integrate into the surrounding environment.
Data availability statement
The raw data supporting the conclusions of this article will be made available by the author, without undue reservation.
Author contributions
NP and SB conceived the idea and designed the experiment. NP and JM supervised the project and JM contributed to designing the experiment. SB and RS set up the indoor CO2 experiment and collected and analyzed the data. SB set up the rooftop farm experiment with help from FS and EC. SB collected the samples and analyzed the data with help from EC. SB wrote the manuscript with support from FS, RS, EC, and NP. All authors contributed to the article and approved the submitted version.
Funding
This material is based upon work supported by the National Science Foundation Graduate Research Fellowship under Grant No. DGE-1840990 as well as partially funded by NSF Grant #1617053. We would also like to thank the Boston University Pardee Center, Boston University's Initiative on Cities, and the Boston University Urban Biogeosciences and Environmental Health Program for their financial support of this project.
Acknowledgments
We would like to thank Sidney Hare and Tony Pham for their assistance collecting samples. The entire community at the Boston University Academy, especially Paige Brewster, Travis Bain, and Cynthia Taylor, also were essential in carrying out this project. This content also appeared in the PhD thesis titled Enhancing plant growth and carbon harvesting for sustainable agriculture (Buckley, 2020).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fsufs.2022.918027/full#supplementary-material
Abbreviations
FACE, free air carbon dioxide enrichment; CAS, college of arts and sciences; BUA, Boston University Academy.
References
ACGIH American Conference of Governmental Industrial Hygienists (2011). TLVs and BEIs. Cincinnati, OH: American Conference of Governmental Industrial Hygienists.
Ahmed, S., Buckley, S., Stratton, A. E., Asefaha, F., Butler, C., Reynolds, M., et al. (2017). Sedum groundcover variably enhances performance and phenolic concentrations of perennial culinary herbs in an urban edible green roof. Agroecol. Sustain. Food Syst. 41, 1–17. doi: 10.1080/21683565.2017.1279703
Ainsworth, E. A., Leakey, A. D., Ort, D. R., and Long, S. P. (2008). FACE-Ing the facts: inconsistencies and interdependence among field, chamber and modeling studies of elevated [CO2] impacts on crop yield and food supply. New Phytol. 179, 5–9. doi: 10.1111/j.1469-8137.2008.02500.x
Ainsworth, E. A., and Long, S. P. (2005). What have we learned from 15 years of free-air CO2 enrichment (FACE)? A meta-analytic review of the responses of photosynthesis, canopy properties and plant production to rising CO2. New Phytol. 165, 351–372. doi: 10.1111/j.1469-8137.2004.01224.x
Ainsworth, E. A., and Rogers, A. (2007). The response of photosynthesis and stomatal conductance to rising [CO2]: mechanisms and environmental interactions. Plant Cell Environ. 30, 258–270. doi: 10.1111/j.1365-3040.2007.01641.x
Allen, J. G., MacNaughton, P., Satish, U., Santanam, S., Vallarino, J., and Spengler, J. D. (2016). Associations of cognitive function scores with carbon dioxide, ventilation, and volatile organic compound exposures in office workers: a controlled exposure study of green and conventional office environments. Environ. Health Perspect. 124, 805–812. doi: 10.1289/ehp.1510037
Apte, M. G., William, J. F., and Joan, M. D. (2000). Associations between indoor CO2 concentrations and sick building syndrome symptoms in US office buildings: an analysis of the 1994–1996 BASE study data. Indoor Air 10, 246–257. doi: 10.1034/j.1600-0668.2000.010004246.x
ArrowStreet Architecture and Design. (2016). Addressing Campus Sustainability Through MIT' s Roofscapes. Boston, MA: Massachusets Institute Technology.
Barrett, K. E., Barman, S. M., Boitano, S., and Brooks, H. L. (2012). “Respiration physiology,” in Ganong's Review of Medical Physiology, 24th Edn (New York: McGraw-Hill Medical), 619–70.
Batchelor, J., Laurence, S. S., and Tina, S. H. (2009). Green Roof Planning Study. Boston, MA: ArrowStreet.
Boese, S. R., and Huner, N. P. (1990). Effect of growth temperature and temperature shifts on spinach leaf morphology and photosynthesis. Plant Physiol. 94, 1830–1836. doi: 10.1104/pp.94.4.1830
Briber, B. M., Hutyra, L. R., Reinmann, A. B., Raciti, S. M., Dearborn, V. K., Holden, C. E., et al. (2015). Tree productivity enhanced with conversion from forest to urban land covers. PLoS ONE 10, 1–19. doi: 10.1371/journal.pone.0136237
Buckley, S. (2020). Enhancing Plant Growth and Carbon Harvesting for Sustainable Agriculture (Dissertion). Boston University, Boston, MA, United States. Available online at: https://open.bu.edu/ds2/stream/?#/documents/390809/page/1
Buckley, S., Ahmed, S., Griffin, T., and Orians, C. (2021). Extreme precipitation enhances phenolic concentrations of Spinach 2 (S. Oleracea). J. Crop Improv. 34, 618–636. doi: 10.1080/15427528.2020.1750521
Bunea, A., Andjelkovic, M., Socaciu, C., Bobis, O., Neacsu, M., Verhé, R., et al. (2008). Total and individual carotenoids and phenolic acids content in fresh, refrigerated and processed spinach (S. Oleracea L.). Food Chem. 108, 649–656. doi: 10.1016/j.foodchem.2007.11.056
Cai, C., Yin, X., He, S., Jiang, W., Si, C., Struik, P. C., et al. (2016). Responses of wheat and rice to factorial combinations of ambient and elevated CO2 and temperature in FACE experiments. Global Change Biol. 22, 856–874. doi: 10.1111/gcb.13065
Calvert, A., and Slack, G. (1976). Effect of carbon dioxide enrichment on growth, development and yield of glasshouse tomatoes. II. The duration of daily periods of enrichment. J. Hortic. Sci. 51, 401–409.
Candlish, J. K., Gourley, L., and Lee, H. P. (1987). Dietary fiber and starch contents of some southeast Asian vegetables. J. Agric. Food Chem. 35, 319–321. doi: 10.1021/jf00075a008
Carter, T. L., and Rasmussen, T. C. (2007). Hydrologic behavior of vegetated roofs. J. Am. Water Resour. Assoc. 42, 1261–1274. doi: 10.1111/j.1752-1688.2006.tb05611.x
Chakrabarti, B., Singh, S. D., Naresh Kumar, S., Aggarwal, P. K., Pathak, H., and Nagarajan, S. (2012). Low-cost facility for assessing impact of carbon dioxide on crops. Curr. Sci. 102, 1035–1040.
Coutts, E., Ito, K., Nardi, C., and Vuong, T. (2015). Planning Urban Heat Island Mitigation Planning Urban Heat Island Mitigation. Boston, MA: Trust for Public Land. doi: 10.1093/icesjms/fss088
Davies, Z. G., Edmondson, J. L., Heinemeyer, A., Leake, J. R., and Gaston, K. J. (2011). Mapping an urban ecosystem service: quantifying above-ground carbon storage at a city-wide scale. J. Appl. Ecol. 48, 1125–1134. doi: 10.1111/j.1365-2664.2011.02021.x
Decina, S. M., Hutyra, L. R., Gately, C. K., Getson, J. M., Reinmann, A. B., and Gianotti, A. G. S. (2016). Soil respiration contributes substantially to urban carbon fluxes in the greater Boston area. Environ. Pollut. 212, 433–439. doi: 10.1016/j.envpol.2016.01.012
Edwards, G. R., Clark, H., and Newton, P. C. D. (2001). The effects of elevated CO2 on seed production and seedling recruitment in a sheep-grazed pasture. Oecologia 127, 383–394. doi: 10.1007/s004420000602
Ellsworth, D. S., Oren, R., Huang, C., Phillips, N., and Hendrey, G. R. (1996). Erratum: leaf and canopy responses to elevated CO2 in a pine forest under free-air CO2 enrichment (Oecologia (1995) 104 (139–146)). Oecologia 106, 416. doi: 10.1007/BF00328578
Fleisher, D. H., Timlin, D. J., and Reddy, V. R. (2008). Interactive effects of carbon dioxide and water stress on potato canopy growth and development. Agron. J. 100, 711–719. doi: 10.2134/agronj2007.0188
Garrison, N., Horowitz, C., and Lunghino, A. C. (2012). Looking up: how green roofs and cool roofs can reduce energy use, address climate change, and protect water resources in Southern California. NRDC Rep. R 12, 1–33. doi: 10.1109/TDC.2005.1547154
Getter, K. L., Rowe, D. B., Robertson, G. P., Cregg, B. M., and Andresen, J. A. (2009). Carbon sequestration potential of extensive green roofs. Sci. Technol. 43, 7564–7570. doi: 10.1021/es901539x
Guite, H. F., Clark, C., and Ackrill, G. (2006). The impact of the physical and urban environment on mental well-being. Public Health 120, 1117–1126. doi: 10.1016/j.puhe.2006.10.005
Halweil, B. (2002). Home Grown the Case for Local Food in a Global Market. Washington, DC: Worldwatch Institute.
Hanson, P. J., Edwards, N. T., Garten, C. T., and Andrews, J. A. (2000). Separating root and soil microbial contributions to soil respiration: a review of methods and observations. Biogeochemistry 48, 115–146. doi: 10.1023/A:1006244819642
Hatfield, J. L., Boote, K. J., Kimball, B. A., Ziska, L. H., and Izaurralde, R. C. (2011). Climate impacts on agriculture: implications for crop production. Agron. J. 103, 351–370. doi: 10.2134/agronj2010.0303
He, Y., Yu, H., Dong, N., and Ye, H. (2016). Thermal and energy performance assessment of extensive green roof in summer: a case study of a lightweight building in Shanghai. Energy Build. 127, 762–773. doi: 10.1016/j.enbuild.2016.06.016
Hendrey, G. R., Lewin, K. F., and Nagy, J. (1993). Free air carbon dioxide enrichment: development, progress, results. Vegetatio 104, 16–31.
Howard, L. R., Pandjaitan, N., Morelock, T., and Gil, M. I. (2005). Antioxidant capacity and phenolic content of spinach as affected by genetics and maturation. J. Agric. Food Chem. 53, 8618–8623. doi: 10.1021/jf052077i
Idso, S. B., and Idso, K. E. (2001). Effects of atmospheric CO(2) enrichment on plant constituents related to animal and human health. Environ. Exp. Bot. 45, 179–199. doi: 10.1016/S0098-8472(00)00091-5
Ismail, A., Samad, M. H. A., Rahman, A. M. A., and Yeok, F. S. (2012). Cooling potentials and CO2 uptake of ipomoea pes-caprae installed on the flat roof of a single storey residential building in Malaysia. Proc. Soc. Behav. Sci. 35, 361–368. doi: 10.1016/j.sbspro.2012.02.099
Jin, M., Bekiaris-Liberis, N., Weekly, K., Spanos, C., and Bayen, A. (2015). “Sensing by proxy: occupancy detection based on indoor CO2 concentration ming,” in UMBICOMM 2015: The Ninth International Conference on Mobile Ubiquitous Computing, Systems, Services and Technologies, Iaria, Nice, France, 1–10.
Johnson, B. S., Malecki, K. M., Peppard, P. E., and Beyer, K. M. (2018). Exposure to neighborhood green space and sleep: evidence from the survey of the health of Wisconsin. Sleep Health 4, 413–419. doi: 10.1016/j.sleh.2018.08.001
Kadam, K. L., and McMillan, J. D. (2003). Availability of corn stover as a sustainable feedstock for bioethanol production. Bioresour. Technol. 88, 17–25. doi: 10.1016/S0960-8524(02)00269-9
Kangasjärvi, J., Jaspers, P., and Kollist, H. (2005). Signalling and cell death in ozone-exposed plants. Plant Cell Environ. 28, 1021–1036. doi: 10.1111/j.1365-3040.2005.01325.x
Kimball, B. A., Kobayashi, K., and Bindi, M. (2002). Responses of agricultural crops to free-air CO2 enrichment. Adv. Agron. 77, 293–368. doi: 10.1016/S0065-2113(02)77017-X
Kimball, B. A., Pinter, P. J., Wall, G. W., Garcia, R. L., and LaMorte, R. L. (1997). Comparisons of responses of vegetation to elevated carbon dioxide in free air and open-top chamber facilities. Adv. Carbon Dioxide Effects Res. 61, 113–130. doi: 10.2134/asaspecpub61.c5
Kleerekoper, L., Van Esch, M., and Salcedo, T. B. (2012). How to make a city climate-proof, addressing the urban heat Island effect. Resour. Conserv. Recycl. 64, 30–38. doi: 10.1016/j.resconrec.2011.06.004
Kuti, J. O., and Konuru, H. B. (2004). Antioxidant capacity and phenolic content in leaf extracts of tree spinach (Cnidoscolus Spp.). J. Agric. Food Chem. 52, 117–121. doi: 10.1021/jf030246y
Leakey, A. D., Ainsworth, E. A., Bernacchi, C. J., Rogers, A., Long, S. P., and Ort, D. R. (2009). Elevated CO2 effects on plant carbon, nitrogen, and water relations: six important lessons from FACE. J. Exp. Bot. 60, 2859–2876. doi: 10.1093/jxb/erp096
Leakey, A. D., Bernacchi, C. J., Dohleman, F. G., Ort, D. R., and Long, S. P. (2004). Will photosynthesis of maize (Z. Mays) in the US corn belt increase in future [CO2] rich atmospheres? An analysis of diurnal courses of CO2 uptake under free-air concentration enrichment (FACE). Global Change Biol. 10, 951–62. doi: 10.1111/j.1529-8817.2003.00767.x
Lee, S. C., and Chang, M. (1999). Indoor air quality investigations at five classrooms. Indoor Air 9, 134–138. doi: 10.1111/j.1600-0668.1999.t01-2-00008.x
Loladze, I. (2002). Rising atmospheric CO2 and human nutrition: toward globally imbalanced plant stoichiometry? Trends Ecol. Evolut. 17, 457–461. doi: 10.1016/S0169-5347(02)02587-9
Long, S. P., Ainsworth, E. A., Leakey, A. D., Nosberger, J., and Ort, D. R. (2006). Food for thought: lower-than-expected crop yield stimulation with rising CO2 concentrations. Science 312, 1918–1922. doi: 10.1126/science.1114722
Lower, E., and Restaurant, B. (2014). Case Study: economic viability of growing produce organically on-site at restaurants.
MADPH (2022). Appendix A: Carbon Dioxide and its Use in Evaluating Adequacy of Ventilation in Buildings.
McLeod, A. R., and Long, S. P. (1999). Free-air carbon dioxide enrichment (FACE) in global change research: a review. Adv. Ecol. Res. 28, 1–57.
Medek, D. E., Schwartz, J., and Myers, S. S. (2017). Estimated effects of future atmospheric CO2 concentrations on protein intake and the risk of protein deficiency by country and region. Environ. Health Perspect. 125, 1–8. doi: 10.1289/EHP41
Meier, A. K. (1990). Strategic landscaping and air-conditioning savings: a literature review. Energy Build. 15, 479–86. doi: 10.1016/0378-7788(90)90024-D
Miglietta, F., Lanini, M., Bindi, M., and Magliulo, V. (1997). Free air CO2 enrichment of potato (Solanum tuberosum, L.): design and performance of the CO2-fumigation system. Glob. Change Biol. 3, 417–427. doi: 10.1046/j.1365-2486.1997.00076.x
Min, K., Chen, K., and Arora, R. (2014). Effect of short-term versus prolonged freezing on freeze-thaw injury and post-thaw recovery in spinach: Importance in laboratory freeze-thaw protocols. Environ. Exp. Bot. 106, 124–131. doi: 10.1016/j.envexpbot.2014.01.009
Moore, B. D., Cheng, S., Sims, D., and Seemann, J. R. (1999). The biochemical and molecular basis for photosynthetic acclimation to elevated atmospheric CO2. Plant Cell Environ. 22, 567–582.
Myers, S. S., Zanobetti, A., Kloog, I., Huybers, P., Leakey, A. D., Bloom, A. J., et al. (2014). Increasing CO2 threatens human nutrition. Nature 510, 139–42. doi: 10.1038/nature13179
Nadal, A., Alamús, R., Pipia, L., Ruiz, A., Corbera, J., Cuerva, E., et al. (2017a). Urban planning and agriculture. Methodology for assessing rooftop greenhouse potential of non-residential areas using airborne sensors. Sci. Total Environ. 601–602, 493–507. doi: 10.1016/j.scitotenv.2017.03.214
Nadal, A., Llorach-Massana, P., Cuerva, E., López-Capel, E., Montero, J. I., Josa, A., et al. (2017b). Building-integrated rooftop greenhouses: an energy and environmental assessment in the Mediterranean context. Appl. Energy 187, 338–351. doi: 10.1016/j.apenergy.2016.11.051
Nagase, A., and Dunnett, N. (2012). Amount of water runoff from different vegetation types on extensive green roofs: effects of plant species, diversity and plant structure. Landscape Urban Plan. 104, 356–63. doi: 10.1016/j.landurbplan.2011.11.001
Ng, C. W. W., Tasnim, R., and Wong, J. T. F. (2019). Coupled effects of atmospheric CO2 concentration and nutrients on plant-induced soil suction. Plant Soil 439, 393–404. doi: 10.1007/s11104-019-04047-4
Nijs, I., Ferris, R., Blum, H., Hendrey, G., and Impens, I. (1997). Stomatal regulation in a changing climate: a field study using free air temperature increase (FATI) and free air CO2 enrichment (FACE). Plant Cell Environ. 20, 1041–1050. doi: 10.1111/j.1365-3040.1997.tb00680.x
Nitsch Engineering (2016). Harvard Business School Stormwater Plan Update. Boston, MA: Harvard Business School.
Norby, R. J., De Kauwe, M. G., Domingues, T. F., Duursma, R. A., Ellsworth, D. S., Goll, D. S., et al. (2016). Viewpoints model-data synthesis for the next generation of forest free-air CO2 enrichment (FACE) experiments. New Phytol. 209, 17–28. doi: 10.1111/nph.13593
Norby, R. J., and Zak, D. R. (2011). Ecological lessons from free-air CO2 enrichment (FACE) experiments. Annu. Rev. Ecol. Evolut. Syst. 42, 181–203. doi: 10.1146/annurev-ecolsys-102209-144647
Nowak, R. S., Ellsworth, D. S., and Smith, S. D. (2004). Functional responses of plants to elevated atmospheric CO2–do photosynthetic and productivity data from FACE experiments support early predictions? New Phytol. 162, 253–280. doi: 10.1111/j.1469-8137.2004.01033.x
Nuutila, A. M., Kammiovirta, K., and Oksman-Caldentey, K. -M. (2002). Comparison of methods for the hydrolysis of flavonoid and phenolic acids from onion and spinach for HPLC analysis. Food Chem. 76, 519–525. doi: 10.1016/S0308-8146(01)00305-3
Oberndorfer, E., Lundholm, J., Bass, B., Coffman, R. R., Doshi, H., Dunnett, N., et al. (2007). Green roofs as urban ecosystems: ecological structures, functions, and services. BioScience 57, 823–833. doi: 10.1641/B571005
Onoda, Y., and Anten, N. P. (2011). Challenges to understand plant responses to wind. Plant Signal. Behav. 6, 139–141. doi: 10.4161/psb.6.7.15635
Orsini, F., Gasperi, D., Marchetti, L., Piovene, C., Draghetti, S., Ramazzotti, S., et al. (2014). Exploring the production capacity of rooftop gardens (RTGs) in urban agriculture: the potential impact on food and nutrition security, biodiversity and other ecosystem services in the City of Bologna. Food Sec. 6, 781–792. doi: 10.1007/s12571-014-0389-6
Persily, A. (2020). “Quit blaming ASHRAE Standard 62.1 for 1000 ppm CO2,” in The 16th Conference of the International Society of Indoor Air Quality and Climate. p. 1-2.
Persily, A., and de Jonge, L. (2017). Carbon dioxide generation rates for building occupants. Indoor Air 27, 868–879. doi: 10.1111/ina.12383
Pirog, R., Van Pelt, T., Enshayan, K., and Cook, E. (2001). Food, fuel, and freeways: an iowa perspective on how far food travels, fuel usage, and greenhouse gas emissions. Leopold Center Sustain. Agric. 209, 1–37.
Pons, O., Nadal, A., Sanyé-Mengual, E., Llorach-Massana, P., Cuerva, E., Sanjuan-Delmàs, D., et al. (2015). Roofs of the future: rooftop greenhouses to improve buildings metabolism. Proced. Eng. 123, 441–448. doi: 10.1016/j.proeng.2015.10.084
Prajapati, S. K. (2012). Ecological effect of airborne particulate matter on plants. Environ. Skept. Crit. 1, 12–22.
Rao, P., Hutyra, L. R., Raciti, S. M., and Templer, P. H. (2014). Atmospheric nitrogen inputs and losses along an urbanization gradient from Boston to Harvard forest, MA. Biogeochemistry 121, 229–245. doi: 10.1007/s10533-013-9861-1
Reddy, M. T., Begum, H., Sunil, N., Rao, P. S., Sivaraj, N., and Kumar, S. (2014). Preliminary characterization and evaluation of landraces of indian spinach (basella spp. l.) for agro-economic and quality traits. Plant Breed. Biotechnol. 2, 48–63. doi: 10.9787/PBB.2014.2.1.048
Reece, C. F., Krupa, S. V., Jäger, H. J., Roberts, S. W., Hastings, S. J., and Oechel, W. C. (1995). Evaluating the effects of elevated levels of atmospheric trace gases on herbs and shrubs: a prototype dual array field exposure system. Environ. Pollut. 90, 25–31. doi: 10.1016/0269-7491(94)00095-U
Rice, S. A. (2004). “Human health risk assessment of CO2: survirors of accute high-level exposure and populations sensitive to prolonged low-level exposure,” in Third Annual Conference on Carbon Sequestration, Susan A. Rice and Associates Inc., Grass Valley, CA, 1–9.
Rogers, H. H., Runion, G. B., and Krupa, S. V. (1994). Plant reponses to atmospheric CO2 enrichment with emphasis on roots and the rhizosphere. Environ. Pollut. 83, 155–189. doi: 10.1016/0269-7491(94)90034-5
Rowe, D. B. (2011). Green roofs as a means of pollution abatement. Environ. Pollut. 159, 2100–2110. doi: 10.1016/j.envpol.2010.10.029
Saadatian, O., Sopian, K., Salleh, E., Lim, C. H., Riffat, S., and Saadatian, E. (2013). A review of energy aspects of green roofs. Renew. Sustain. Energy Rev. 23, 155–168. doi: 10.1016/j.rser.2013.02.022
Sanjuan-Delmás, D., Llorach-Massana, P., Nadal, A., Ercilla-Montserrat, M., Muñoz, P., Montero, J. I., et al. (2018a). Environmental assessment of an integrated rooftop greenhouse for food production in cities. J. Clean. Prod. 177, 326–337. doi: 10.1016/j.jclepro.2017.12.147
Sanjuan-Delmás, D., Llorach-Massana, P., Nadal, A., Sanyé-Mengual, E., Petit-Boix, A., Ercilla-Montserrat, M., et al. (2018b). “Improving the metabolism and sustainability of buildings and cities through integrated rooftop greenhouses (i-RTG),” in Urban Horticulture: Sustainability for the Future (Cham: Springer International Publishing), 53–72. doi: 10.1007/978-3-319-67017-1_3
Santamouris, M. (2014). Cooling the cities: a review of reflective and green roof mitigation technologies to fight heat Island and improve comfort in urban environments. Solar Energy 103, 682–703. doi: 10.1016/j.solener.2012.07.003
Sanyé-Mengual, E., Cerón-Palma, I., Oliver-Solà, J., Montero, J. I., and Rieradevall, J. (2015a). Integrating horticulture into cities: a guide for assessing the implementation potential of rooftop greenhouses (RTGs) in industrial and logistics parks. J. Urban Technol. 22, 87–111. doi: 10.1080/10630732.2014.942095
Sanyé-Mengual, E., Llorach-Massana, P., Sanjuan-Delmás, D., Oliver-Solà, J., Josa, A., Montero, J. I., et al. (2014). “The ICTA-ICP rooftop greenhouse lab (RTG-Lab): closing metabolic flows (energy, water, CO2) through integrated rooftop greenhouses,” in Finding Places for Productive Cities. Proceedings of 6th International AESOP Sustainable Food Planning Conference, VHL University of Applied Sciences, Barcelona, 693–701.
Sanyé-Mengual, E., Oliver-Solà, J., Montero, J. I., and Rieradevall, J. (2015b). An environmental and economic life cycle assessment of rooftop greenhouse (RTG) implementation in Barcelona, Spain. Assessing new forms of urban agriculture from the greenhouse structure to the final product level. Int. J. Life Cycle Assess. 20, 350–366. doi: 10.1007/s11367-014-0836-9
Satish, U., Mendell, M. J., Shekhar, K., Hotchi, T., Sullivan, D., Streufert, S., et al. (2012). Is CO2 an indoor pollutant? Direct effects of low-to-moderate CO2 concentrations on human decision-making performance. Environ. Health Perspect. 120, 1671–77. doi: 10.1289/ehp.1104789
Seppanen, O. A., Fisk, W. J., and Mendell, M. J. (1999). Association of ventilation rates and CO2 concentrations with health and other responses in commercial and institutional buildings. Indoor Air 9, 226–252. doi: 10.1111/j.1600-0668.1999.00003.x
Shafique, M., and Kim, R. (2017). Retrofitting the low impact development practices into developed urban areas including barriers and potential solution. Open Geosci. 9, 240–254. doi: 10.1515/geo-2017-0020
Shafique, M., Kim, R., and Kyung-Ho, K. (2018a). Green roof for stormwater management in a highly urbanized area: the case of Seoul, Korea. Sustainability 10, 584. doi: 10.3390/su10030584
Shafique, M., Kim, R., and Rafiq, M. (2018b). Green roof benefits, opportunities and challenges: a review. Renew. Sustain. Energy Rev. 90, 757–773. doi: 10.1016/j.rser.2018.04.006
Shafique, M., Xue, X., and Luo, X. (2020). An overview of carbon sequestration of green roofs in urban areas. Urban Forestry Urban Greening 47, 126515. doi: 10.1016/j.ufug.2019.126515
Sharkey, T. D. (1988). Estimating the rate of photorespiration in leaves. Plant Physiol. 73, 147–152.
Shohag, M. J. I., Wei, Y. Y., Yu, N., Zhang, J., Wang, K., Patring, J., et al. (2011). Natural variation of folate content and composition in spinach (S. Oleracea) germplasm. J. Agric. Food Chem. 59, 12520–12526. doi: 10.1021/jf203442h
Templer, P. H., Toll, J. W., Hutyra, L. R., and Raciti, S. M. (2015). Nitrogen and carbon export from urban areas through removal and export of litterfall. Environ. Pollut. 197, 256–261. doi: 10.1016/j.envpol.2014.11.016
Vadiee, A., and Martin, V. (2013). Thermal energy storage strategies for effective closed greenhouse design. Appl. Energy 109, 337–343. doi: 10.1016/j.apenergy.2012.12.065
van Beveren, P. J. M., Bontsema, J., van Straten, G., and van Henten, E. J. (2015). Optimal control of greenhouse climate using minimal energy and grower defined bounds. Appl. Energy 159, 509–519. doi: 10.1016/j.apenergy.2015.09.012
Vaughan, N. E., and Lenton, T. M. (2011). A review of climate geoengineering proposals. Clim. Change 109, 745–790. doi: 10.1007/s10584-011-0027-7
Warrington, I. J., and Kanemasu, E. T. (1983). Corn growth responses to temperature and photoperiod. II. Leaf initiation and leaf appearance rates. Agron. J. 75, 755–761.
Whittinghill, L. J., Rowe, D. B., Andresen, J. A., and Cregg, B. M. (2015). Comparison of stormwater runoff from sedum, native prairie, and vegetable producing green roofs. Urban Ecosyst. 18, 13–29. doi: 10.1007/s11252-014-0386-8
Whittinghill, L. J., Rowe, D. B., Schutzki, R., and Cregg, B. M. (2014). Quantifying carbon sequestration of various green roof and ornamental landscape systems. Landscape Urban Plan. 123, 41–48. doi: 10.1016/j.landurbplan.2013.11.015
Wong, N. H., Cheong, D. K. W., Yan, H., Soh, J., Ong, C. L., and Sia, A. (2003). The effects of rooftop garden on energy consumption of a commercial building in Singapore. Energy Build. 35, 353–364. doi: 10.1016/S0378-7788(02)00108-1
Yamori, W., Suzuki, K., Noguchi, K. O., Nakai, M., and Terashima, I. (2006). Effects of rubisco kinetics and rubisco activation state on the temperature dependence of the photosynthetic rate in spinach leaves from contrasting growth temperatures. Plant Cell Environ. 29, 1659–1670. doi: 10.1111/j.1365-3040.2006.01550.x
Zhang, X., Chen, X., and Zhang, X. (2018). The impact of exposure to air pollution on cognitive performance. Proc. Natl. Acad. Sci. 115, 9193–9197. doi: 10.1073/pnas.1809474115
Keywords: carbon dioxide, rooftop farms, human respiration, enhanced growth, crops, building metabolism, cities
Citation: Buckley S, Sparks R, Cowdery E, Stirling F, Marsching J and Phillips N (2022) Enhancing crop growth in rooftop farms by repurposing CO2 from human respiration inside buildings. Front. Sustain. Food Syst. 6:918027. doi: 10.3389/fsufs.2022.918027
Received: 11 April 2022; Accepted: 05 September 2022;
Published: 24 October 2022.
Edited by:
Amos P. K. Tai, The Chinese University of Hong Kong, ChinaReviewed by:
Emmanuel Amagu Echiegu, University of Nigeria, NigeriaKou Jiancun, Northwest A&F University, China
Copyright © 2022 Buckley, Sparks, Cowdery, Stirling, Marsching and Phillips. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Sarabeth Buckley, c2IyNDgxQGNhbS5hYy51aw==
 Sarabeth Buckley
Sarabeth Buckley Rebecca Sparks1
Rebecca Sparks1 Finn Stirling
Finn Stirling Nathan Phillips
Nathan Phillips