Abstract
ESKAPE (Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumanni, Pseudomonas aeruginosa, and Enterobacter species) pathogens have characteristic multiple-drug resistance and cause an increasing number of nosocomial infections worldwide. Peptide-based therapeutics to treat ESKAPE infections might be an alternative to conventional antibiotics. Histatin 5 (Hst 5) is a salivary cationic histidine-rich peptide produced only in humans and higher primates. It has high antifungal activity against Candida albicans through an energy-dependent, non-lytic process; but its bactericidal effects are less known. We found Hst 5 has bactericidal activity against S. aureus (60–70% killing) and A. baumannii (85–90% killing) in 10 and 100 mM sodium phosphate buffer (NaPB), while killing of >99% of P. aeruginosa, 60–80% E. cloacae and 20–60% of E. faecium was found in 10 mM NaPB. Hst 5 killed 60% of biofilm cells of P. aeruginosa, but had reduced activity against biofilms of S. aureus and A. baumannii. Hst 5 killed 20% of K. pneumonia biofilm cells but not planktonic cells. Binding and uptake studies using FITC-labeled Hst 5 showed E. faecium and E. cloacae killing required Hst 5 internalization and was energy dependent, while bactericidal activity was rapid against P. aeruginosa and A. baumannii suggesting membrane disruption. Hst 5-mediated killing of S. aureus was both non-lytic and energy independent. Additionally, we found that spermidine conjugated Hst 5 (Hst5-Spd) had improved killing activity against E. faecium, E. cloacae, and A. baumannii. Hst 5 or its derivative has antibacterial activity against five out of six ESKAPE pathogens and may be an alternative treatment for these infections.
Introduction
Bacteria causing nosocomial infections are increasingly becoming drug resistant, posing a serious health concern, especially in the Intensive Care Units (ICUs) and in surgical wards. A recent report estimated a total of 722,000 of such drug-resistant infections, with an astonishing 75,000 resulting in deaths (Magill et al., 2014). A group of bacterial pathogens including Enterococcus faecium, Staphylococcus aureus, Klebsiella pneumoniae, Acinetobacter baumanii, Pseudomonas aeruginosa, and Enterobacter species (referred to as ESKAPE pathogens) have been of particular concern in this regard (Rice, 2010). Two-thirds of all healthcare-associated infections are ESKAPE related (Boucher et al., 2009), and many of these bacteria use multiple drug resistance mechanisms to “eskape” killing by both conventional and some newer generation antibiotics (Rice, 2008).
Most nosocomial infections result from an exogenous inoculum including the hospital environment and medical personnel, resulting in colonization of ESKAPE pathogens within various patient niches. E. faecium, K. pneumoniae, P. aeruginosa, and Enterobacter species are common residents of mucosal surfaces such as the oral cavity and the gastrointestinal tract (Keller et al., 1998; Podschun and Ullmann, 1998; Rice, 2010; Vu and Carvalho, 2011). S. aureus is a skin commensal and A. baumanni has been known to have comparatively long survival rates on epithelial surfaces (e.g., skin; Houang et al., 1998; Coates et al., 2014). Also, P. aeruginosa and K. pneumonia were detected in 50 and 31%, respectively, of HIV-positive patients' salivas in hospital settings (Lopes et al., 2015). Thus, colonization of the oral cavity with K. pneumoniae, P. aeruginosa, and Enterobacter species can serve as a potential inoculum source for pneumonia (Sands et al., 2016). Furthermore, Candida albicans, an oral commensal fungus also present in the oral cavity, can contribute to a more robust P. aeruginosa lung infection if the two are present together (Lindsay and Hogan, 2014).
Salivary innate immunity is the first line of defense against transient and pathobionts in the oral cavity (Salvatori et al., 2016). This is illustrated by the example of salivary Histatin 5 (Hst 5), a cationic protein that has strong fungicidal activity against C. albicans (Puri and Edgerton, 2014). Changes in the levels of Hst 5 associated with immunodeficiency may increase susceptibility to oral candidiasis caused by C. albicans (Khan et al., 2013). However, Hst 5 was shown to be largely ineffective against oral commensal bacteria as well as cariogenic or periodontal pathogens (Devine and Hancock, 2002; Groenink et al., 2003; Dale and Fredericks, 2005).
However, antimicrobial activity of Hst 5 against the ESKAPE pathogens has never been examined. Hst 5-mediated candidacidal activity is an energy-dependent, non-lytic process, where multiple intracellular targets are affected after Hst 5 has been transported to the cytosol in an energy dependent manner (Puri and Edgerton, 2014). Given the fact that most ESKAPE pathogens are not present in large numbers in the oral cavity of healthy humans, and do not cause any known oral diseases, we hypothesized that salivary Hst 5 may play a substantial role in keeping at least some of the ESKAPE pathogens in check within the oral environment. Here we show for the first time that Hst 5 demonstrates killing activity against all ESKAPE pathogens except K. pneumoniae. Killing against some ESKAPE pathogens was lytic in nature; however, as for C. albicans, more than one mechanism of killing seems to be involved in Hst 5 activity against ESKAPE bacteria.
Materials and methods
Strains, culture conditions, and peptides
All ESKAPE strains used in this study are clinical isolates and are listed in Table 1. These isolates were collected over the past 20 years directly from the clinical microbiology laboratory at the University at Buffalo, which performed strain identification by approved laboratory methodology. Isolates from de-identified plates were frozen and stored at −80°C. Enterococcus faecium and Staphylococcus aureus were grown in Trypticase soy broth (Sigma-Aldrich; TSB) or TSB agar. All other strains were cultured in Luria-Bertani (LB; BD Biosciences) broth or on LB agar. The incubation temperature for all strains was 37°C. Histatin 5 (Hst 5), FITC-labeled Hst 5 (F-Hst 5), and spermidine conjugated Histatin 5 (Hst 5-Spd) were synthesized by Genemed Synthesis Inc. (San Antonio, Texas). Spermidine was purchased from Sigma-Aldrich.
Table 1
| Species | Strain # | Gram staining | Source* |
|---|---|---|---|
| Enterococcus faecium | 94-346-1139 | + | Buffalo, NY |
| Staphylococcus aureus | 299-0432 | + | Buffalo, NY |
| Staphylococcus aureus (MRSA) | 94-292-1348 | + | Buffalo, NY |
| Klebsiella pneumoniae | Montreal #33 | - | Montreal, Canada |
| Acinetobacter baumannii | AB307-0294 | - | Buffalo, NY (Adams et al., 2008) |
| Acinetobacter baumannii | HUMC1 | - | Luo et al., 2012 |
| Pseudomonas aeruginosa | 94-323-0635 | - | Buffalo, NY |
| Pseudomonas aeruginosa | PAO1 | - | Sutton lab, Buffalo, NY |
| Enterobacter cloacae | 94-293-0624 | - | Buffalo, NY |
ESKAPE strains used in this study.
Isolates from the University at Buffalo were collected over the past 20 years directly from the clinical microbiology laboratory, which performed strain identification by approved laboratory methodology.
Bactericidal assay
Bactericidal assays were performed by microdilution plate method as described for candidacidal assays (Jang et al., 2010), with some modifications. Briefly, a single colony of each strain was inoculated into 10 mL of media and grown for 16 h at 37°C, cultures were diluted to an OD600 = 0.1 in fresh media and incubated with shaking at 37°C to mid-log phase (OD600 ≈ 1.0). Cultures were spun down at 1800 × g for 3 min and washed three times with 10 mM sodium phosphate buffer (pH 7.4; NaPB). Cells were re-suspended (107 cells/mL) in 200 μL NaPB (control) or in NaPB containing peptides (30 μM); then incubated at 37°C (except for S. aureus that was incubated at room temperature for optimal growth). Aliquots were removed after 1 min, 1 h and 5 h incubation, then were serially diluted with phosphate buffered saline (PBS; 137 mM NaCl, 10 mM sodium phosphate, 2.7 mM KCl, pH of 7.4); plated and incubated for 24 h at 37°C to visualize surviving CFUs. Assays were performed in triplicate. Percentage killing was calculated as [1 − number of colonies from peptide-treated cells/mean number of colonies from control cells] × 100%. Bactericidal assays were also performed in 100 mM NaPB and PBS.
Live cell imaging of F-Hst 5 and PI uptake by time-lapse confocal microscopy
Confocal microscopy was performed using live cells of six ESKAPE strains attached to chambered borosilicate coverglass (Lab-Tek #155411, 8 chamber). Cells in mid-log phase were collected and washed three times and re-suspended in 10 mM NaPB. Cells (2 × 107 in 200 μL NaPB) were added to each well, allowed to settle for 15 min, then propidium iodide (PI; 2 μg/mL; Sigma) and F-Hst 5 (30 μM) were added. Time-lapse confocal images were recorded immediately after addition of peptide and PI. Confocal images were acquired with a Zeiss LSM510 Meta Confocal Microscope (Carl Zeiss, Germany) using a Plan Apochromat 63X/1.4 Oil Immersion objective. For E. faecium, S. aureus, K. pneumoniae, and E. cloacae, images were collected every 10 min; and for A. baumannii and P. aeruginosa every 2 min. In order to detect F-Hst 5 and PI simultaneously, the 488 nm line of the argon ion laser and a 561 nm DPSS laser were directed over an HFT UV/488/561 beam splitter, and fluorescence was detected using a Mirror or NFT 565 beam splitter in combination with a BP 500–550 filter for F-Hst 5 and an LP 575 or BP 650–710 filter for PI detection. ImageJ software was used for image acquisition and analysis.
Biofilm killing assays
To evaluate killing of bacterial biofilms by Hst 5, cells were inoculated at 1 × 107 cells/ml into 96 well plates and grown overnight at 37°C and 90 RPM for 12 h to form biofilms. After 12 h, spent media was replaced with fresh media, and biofilms were grown for another 4 h in the same conditions. Media was removed and biofilms were incubated with 30 μM Hst 5 suspended in 10 μM NaPB with 10 μM propidium iodide (PI) for 1 h at 37°C, except for S. aureus which was incubated at RT. After 1 h, biofilms were gently scraped to remove them from each well, diluted, and placed on microscope slides for image acquisition using a Zeiss Axioobserver.Z1 inverted fluorescent microscope, and an Axiocam 503M camera. Percent killing was calculated as the number of PI positive cells divided by the total number of cells from at least 25 fields from two independent wells.
Hst 5 killing activity determination in energy deprived cells
Overnight cultures of E. faecium, S. aureus, A. baumannii, and E. cloacae were diluted and pre-incubated with 10% of media with or without 10 mM NaN3 at 37°C. P. aeruginosa cells were pre-incubated in 10 mM NaPB with 10 mM NaN3 at 37°C due to altered cell viability in the presence of 10% media with NaN3. After 3 h incubation, cells were collected by centrifugation and washed two times with NaPB; then cells were used for Hst 5 bactericidal assay in NaPB as described above.
Susceptibility assay
Minimum inhibitory concentration (MIC) was determined by broth microdilution method based on the guide of Clinical and Laboratory Standards Institute (Clinical Laboratory Standards Institute, 2012) with some modifications. Briefly, all bacterial strains except K. pneumoniae were cultured in respective media and grown overnight at 37°C. Cells were washed with NaPB buffer, diluted in 10% of Mueller-Hinton (MH) broth (Sigma) to obtain a concentration of 5 × 106 CFU/mL that were used as the inoculum. Hst 5 peptide was serially diluted in 10% MH broth (since it is inactive in undiluted broth) in 96-well flat-bottomed plates (Falcon). After adding equal volume of target bacterial suspension to the peptide solution, the 96-well plates were incubated at 37°C for 24 h. The MIC values were determined by visibly recording the lowest concentration of Hst 5 that inhibited growth.
Statistics
Statistical analyses were performed using GraphPad Prism software version 5.0 (GraphPad Software, San Diego, CA, USA) using unpaired Student's t-tests. Differences of P < 0.05 were considered significant. All experiments were performed at least thrice.
Results
Hst 5 has strong bactericidal activity against four ESKAPE pathogens
In order to determine the bactericidal activity of Hst 5 against ESKAPE pathogens, six clinical isolates (Table 1) were tested using bactericidal assays (performed in 10 and 100 mM NaPB buffer) to determine percent bacterial killing following incubation for 1 min, 1, and 5 h with 30 μM Hst 5. E. faecium showed time-dependent killing since only 18.1 ± 8.9 percent of cells were killed by Hst 5 at 1 h, while killing was enhanced to 63.5 ± 3.5 percent after 5 h incubation (Figure 1A). S. aureus cells incubated for only 1 min with 30 μM Hst 5 showed 28.3 ± 2.3 percent killing which increased to 60.3 ± 5.7 and 69.7 ± 0.2 percent after 1 and 5 h, respectively (Figure 1B). A MRSA strain of S. aureus had lower but still significant killing by Hst 5 after 1 h (33.8 ± 3.5%). Interestingly, Hst 5 did not show any killing activity against K. pneumoniae for up to 5 h of incubation (Figure 1C). A. baumannii AB307-0294 had substantial sensitivity to Hst 5 with 31.4 ± 3.7, 90.0 ± 1.1, and 95. ± 2.3 percent killing at 1 min, 1, and 5 h, respectively (Figure 1D), while the A. baumannii HUMC1 strain had even higher sensitivity with 100% killing at 1 h. Similarly, Hst 5 also showed strong killing activity against P. aeruginosa 94-323-0635, with a faster rate of killing. At just 1 min of incubation, 83.2 ± 1.3 percent killing was observed; while over 99.9 percent of cells were killed after 1 h treatment (Figure 1E). P. aeruginosa PAO1 had identical sensitivity to Hst 5 with 100% of cells killed at 1 h. E. cloacae sensitivity was similar to that of A. baumannii showing 20.3 ± 1.5, 66.1 ± 0.6, and 78.1 ± 1.7 percent of cells killed by Hst 5 at 1 min, 1 h, and 5 h, respectively (Figure 1F).
Figure 1
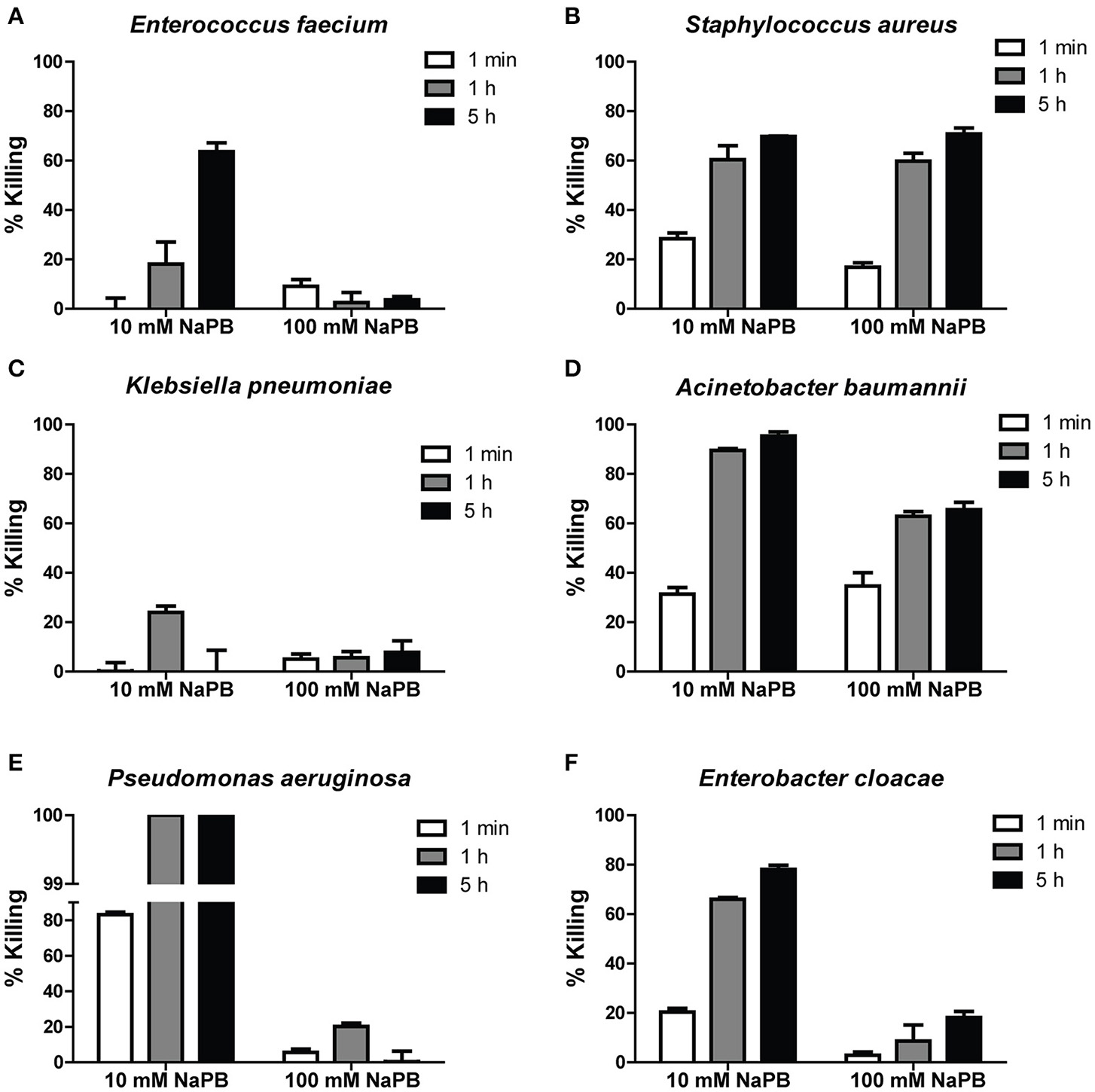
Killing of ESKAPE cells by Hst 5. E. faecium(A), S. aureus(B), K. pneumoniae(C), A. baumannii(D), P. aeruginosa(E), and E. cloacae(F) cells in exponential growth were exposed to 30 μM Hst 5 in 10 mM NaPB or 100 mM NaPB for 1 min, 1, and 5 h. Aliquots taken at different time points were diluted and plated. CFU were determined after 24 h. Error bars represent the standard errors from at least three independent replicates of each strain.
Since the activity of Hst 5 against C. albicans is abolished at higher phosphate buffer concentrations (Helmerhorst et al., 1999), Hst 5 killing activity against ESKAPE strains was also tested in a buffer with higher ionic strength. Killing activities of Hst 5 against E. faecium, P. aeruginosa, and E. cloacae were greatly reduced (Figures 1A,E,F) when carried out in 100 mM NaPB. However, bactericidal activity against S. aureus at 100 mM NaPB was similar to that observed in 10 mM NaPB (Figure 1B), while killing efficiency for A. baumannii were decreased by about 30 percent in 100 mM NaPB after 1 or 5 h incubation (Figure 1D). Effects of PBS on killing were similar to those seen in 100 mM NaPB (data not shown).
Based on these bactericidal results, we next tested growth inhibition by Hst 5 for these ESKAPE pathogens by determining MIC values in 10% MH broth (since Hst 5 is inactive in undiluted broth). Hst 5 MIC values of 38, 47, and 90 μM were observed for A. baumannii, P. aeruginosa, and E. cloacae; while we could not determine MIC values for E. faecium and S. aureus due to media components that inactivate Hst 5 (Table 2).
Table 2
| % Hst 5 killinga | % Hst 5 biofilmKillingb | % Hst 5-spd killingc | Main mechanism of killing | MIC (μM)d | ||
|---|---|---|---|---|---|---|
| Membrane disruption | Energy dependent | |||||
| Enterococcus faecium | 63.6 | ND | 96.6 | - | + | ND |
| Staphylococcus aureus | 69.8 | 3.2 | 69.2 | - | - | ND |
| Klebsiella pneumoniae | 0.0 | 19.5 | 0.0 | N/A | N/A | N/A |
| Acinetobacter baumannii | 95.34 | 15.1 | 96.8 | + | - | 38 |
| Pseudomonas aeruginosa | >99.9 | 59.5 | >99.9 | + | - | 47 |
| Enterobacter cloacae | 78.1 | ND | 90.1 | - | + | 90 |
Hst 5-mediated killing of ESKAPE pathogens.
Hst 5 (30 μM) killing efficiency of planktonic cells in 10 mM NaPB buffer for 5 h.
Hst 5 (30 μM) killing efficiency of biofilm cells in 10 mM NaPB buffer for 1 h.
Hst 5-Spd (30 μM) killing efficiency in 10 mM NaPB buffer for 5 h.
MIC in 10% MH broth.
ND, Not Determined; N/A, Not Applicable.
The activity of Hst 5 against ESKAPE pathogens grown as biofilms was determined after 16 h growth of each bacteria in 96-well plates (Table 2). Biofilms were treated with 30 μM Hst 5 for 1 h, dead cells stained with PI, and percent killing calculated. Neither E. faecium or E. cloacae formed robust biofilms in our hands so that Hst 5 killing could not be reliably determined for these two pathogens. As for planktonic cells, both strains of P. aeruginosa in biofilms were effectively killed by Hst 5 after 1 h (59.5%); however Hst 5 killing of A. baumannii biofilm cells was reduced to 15.1%; while killing of S. aureus biofilm cells (both strains) was negligible (5.1%). Surprisingly, we observed that Hst 5 killed 19.5% of K. pneumoniae when in biofilms, although it had no activity against planktonic cells.
Overall, our results show that Hst 5 has a strong bactericidal activity against P. aeruginosa and E. cloacae (and weaker killing with E. faecium) at lower ionic strength environments; while exerting killing against A. baumannii and S. aureus over a range of ionic strengths. However, biofilm cells of A. baumannii and S. aureus were more resistant to Hst 5 killing. To confirm these findings in real time and to assess whether ionic strength may influence metabolic or membrane lytic mechanisms of killing, we next measured Hst 5 binding, uptake, and time of killing for these ESKAPE pathogens in 10 mM NaPB.
Bactericidal activity of Hst 5 against E. faecium and E. cloacae requires internalization and is energy dependent
Time-lapse confocal microscopy showed that Hst 5 (30 μM) rapidly (< 1 min) bound with the surface of E. faecium cells, although no internalization was evident until after 10 min of incubation (Figure 2A). This slow internalization resulted in only 15% of cells taking up F-Hst 5 by 120 min; but in each instance, Hst 5 internalization was accompanied by PI uptake (indicative of cell death; Figure 2B). This close correlation between intracellular Hst 5 and PI uptake suggested that targets of Hst 5 are intracellular rather than its effects being membrane lytic. However, the percentage of killing from the bactericidal assay was higher than apparent PI staining, suggesting that some portion of killing was delayed. To evaluate this, we pretreated cells with an inhibitor of energy metabolism (NaN3) as a means to determine whether the killing activity of Hst 5-treated E. faecium might be dependent upon cell metabolism. As expected, pretreatment of cells with NaN3 significantly reduced the killing efficiency of Hst 5 at 1 and 5 h (Figure 2C), showing that some portion of Hst 5 killing of E. faecium requires target cell energy, likely for Hst 5 internalization or intracellular organelle localization.
Figure 2
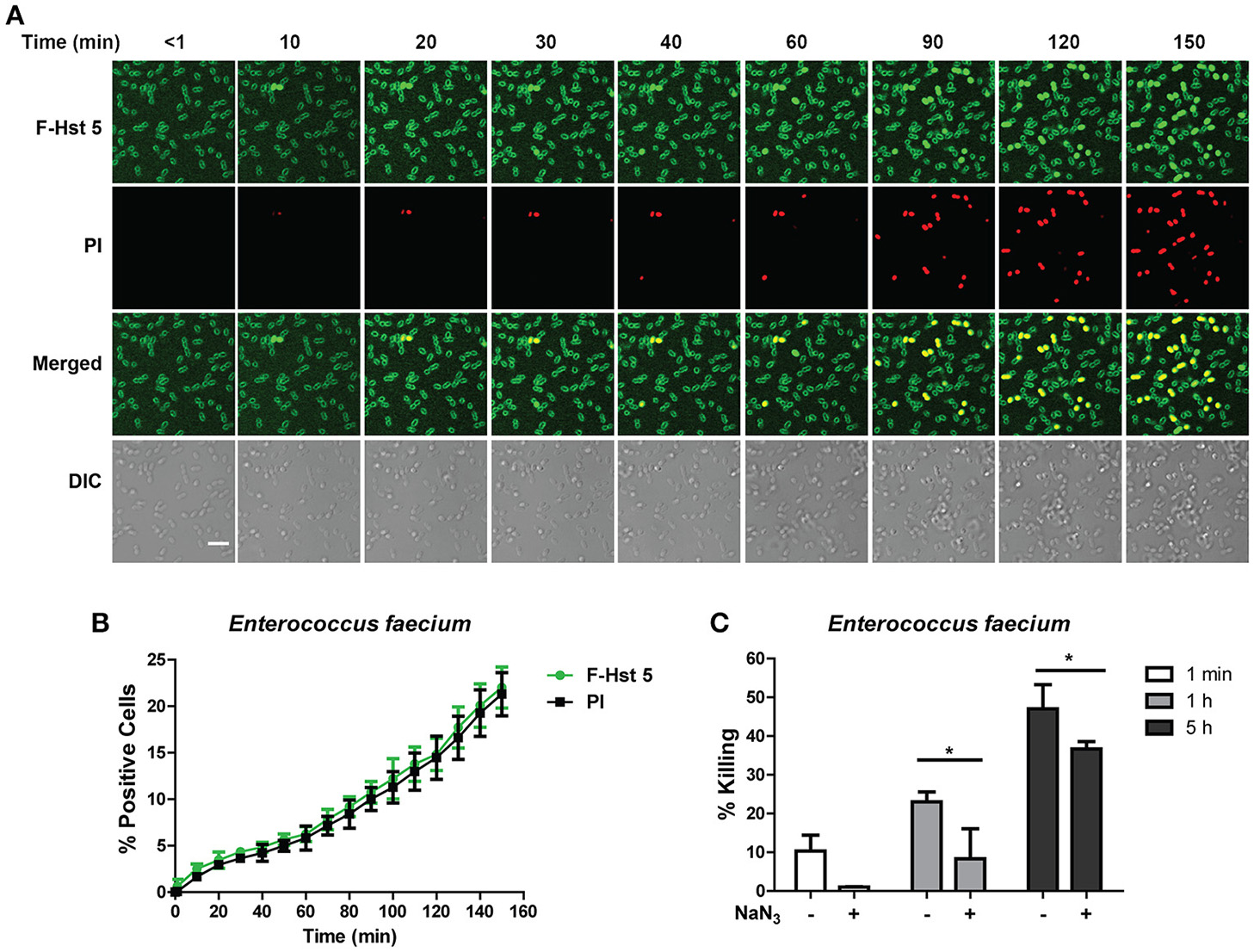
Antibacterial activity of Hst 5 against E. faecium required internalization and is energy dependent. (A) E. faecium cells were exposed to F-Hst 5 (30 μM) and PI (2 μg/mL). F-Hst 5 (green) and PI (red) uptake were measured in parallel by time-lapse confocal microscopy. Images were recorded every 10 min and selected images of indicated time points were shown. (Scale bar: 5 μm) (B) Quantitative analysis of F-Hst 5 uptake (green line) and PI uptake. Error bars represent the standard errors from four different fields of image. (C) Cells pretreated with 10 mM NaN3 at 37°C for 3 h showed decreased susceptibility to Hst 5. (*P < 0.05, Student's t-test).
E. cloacae cells had a very similar response when treated with F-Hst 5, in that the peptide associated with the surface of all cells almost immediately, however entry of F-Hst 5 (and accompanying PI staining) was slow and only occurred in 20% of cells within 30 min (Figure 3A). As for E. faecium, there was a close correlation between intracellular Hst 5 and PI uptake at all time points (Figure 3B), also pointing toward intracellular targets for bactericidal activity. E. cloacae cells pretreated with 10 mM NaN3 prior to exposure with 30μM Hst 5 were more resistant to the killing action of F-Hst 5 (Figure 3C), similar to the energy dependent mechanism found with E. faecium.
Figure 3
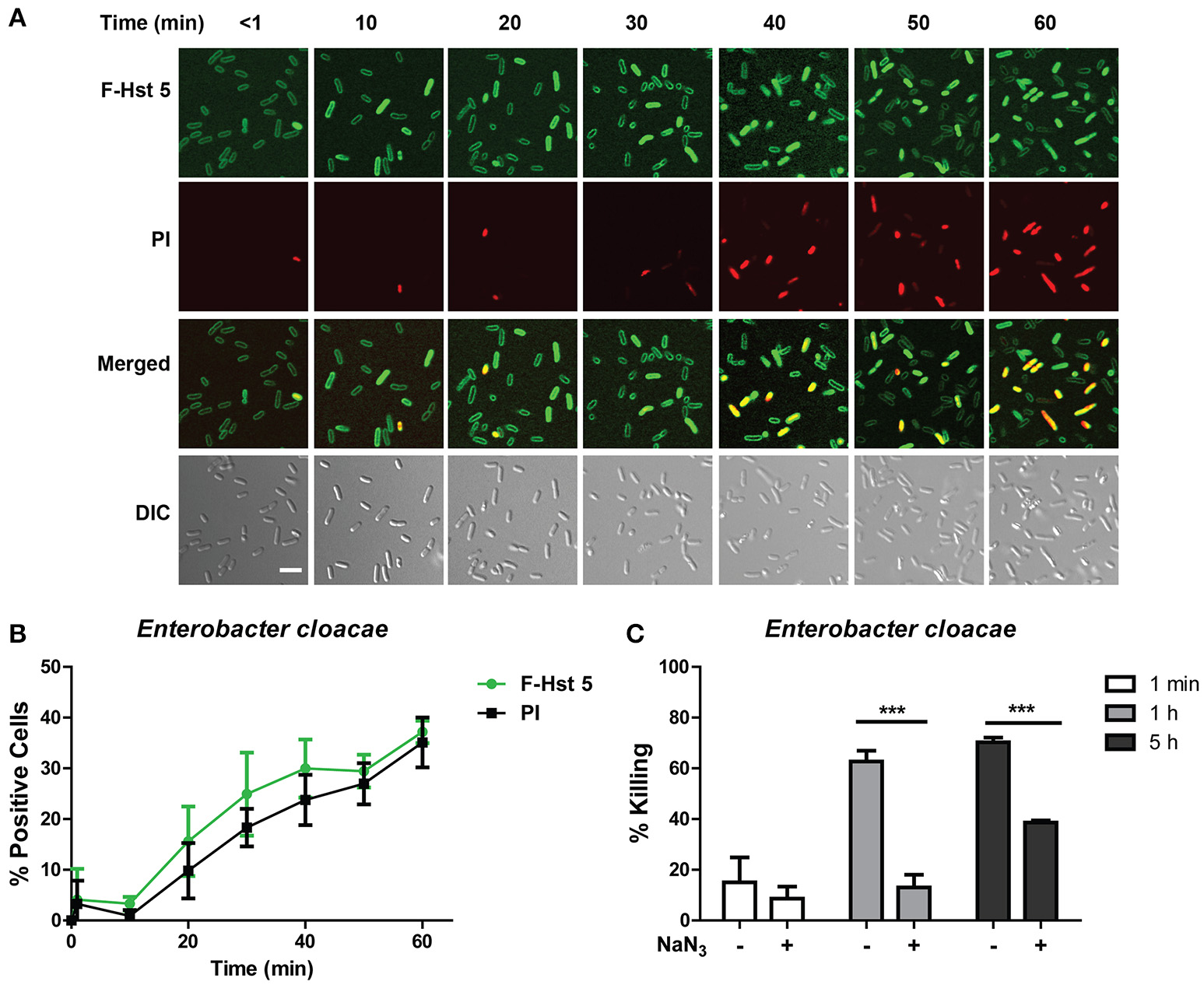
Antibacterial activity of Hst 5 against E. cloacae requires internalization and is energy dependent. (A) E. cloacae cells were exposed to F-Hst 5 (30 μM) and PI (2 μg/mL). The F-Hst 5 (green) and PI (red) uptake were measured in parallel by time-lapse confocal microscopy. Images were recorded every 10 min and selected images as indicated time points were shown. (Scale bar: 5 μm) (B) Quantitative analysis of F-Hst 5 uptake (green line) and PI uptake. Error bars represent the standard errors from four different fields of image. (C) Cells pretreated with 10 mM NaN3 at 37°C for 3 h showed more resistance to Hst 5. (***P < 0.001, Student's t-test).
Membrane disruption is involved in bactericidal activity of Hst 5 against P. aeruginosa and A. baumannii
In agreement with its high bactericidal activity, F-Hst 5 completely covered the cell surface of P. aeruginosa within 1 min, and PI positive cells could be visualized within 2 min (Figure 4A). Quantitative analysis showed that 75% of cells contained F-Hst 5 and were PI positive within 5 min of addition of F-Hst 5 (Figure 4B). Both intracellular localization of Hst 5 and PI staining were rapid and simultaneous, pointing toward membrane lytic activity of Hst 5 mediated killing of P. aeruginosa. Pretreatment of P. aeruginosa cells with 10 mM NaN3 did not reduce the killing activity of Hst 5 (Figure 4C), also suggesting energy-independent membrane disruption as the main pathway for Hst 5 killing activity against P. aeruginosa.
Figure 4
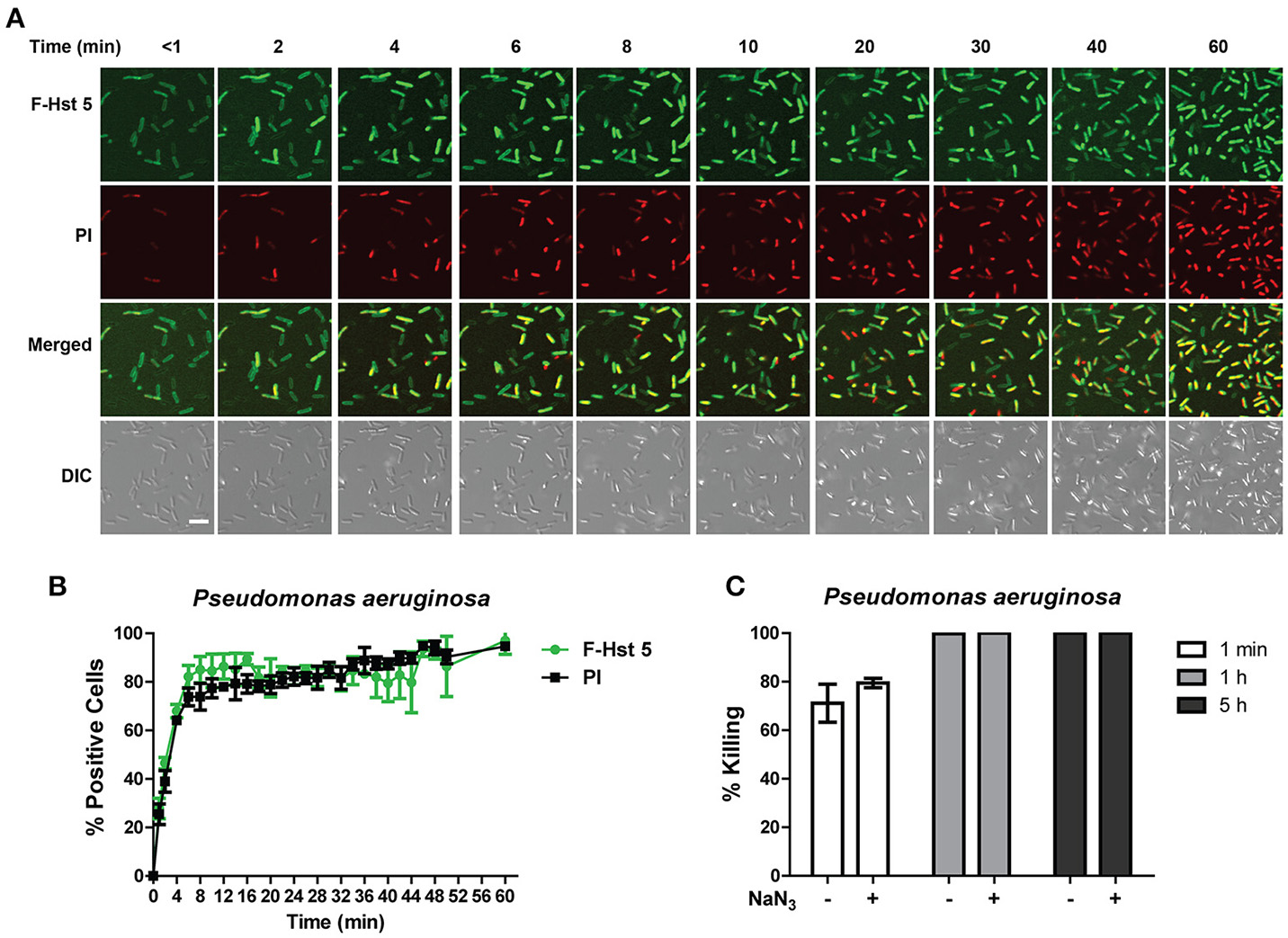
Hst 5 bactericidal activity against P. aeruginosa cells is primarily mediated by membrane disruption. (A) P. aeruginosa cells were exposed to F-Hst 5 (30 μM) and PI (2 μg/mL). F-Hst 5 (green) and PI (red) uptake were measured in parallel by time-lapse confocal microscopy. Images were recorded every 10 min and selected images at indicated time points were shown (Scale bar: 5 μm) (B) Quantitative analysis of F-Hst 5 uptake (green line) and PI uptake. Error bars represent the standard errors from four different fields of image. (C) Cells pretreated with 10 mM NaN3 at 37°C for 3 h did not show significant difference in susceptibility to Hst 5.
A. baumannii showed a very similar profile to P. aeruginosa after exposure to F-Hst 5 in that F-Hst 5 was associated with the surface of all cells within 2 min; and PI positive cells were visualized with 2 min (Figure 5A). Although intracellular uptake of F-Hst 5 was very rapid with 70% of cells containing F-Hst 5 in just 2 min, PI staining did not occur simultaneously so that 70% PI positive cells was seen only after 25 min (Figure 5B). Interestingly, the portion of A. baumannii cells that showed slower PI staining already contained F-Hst 5; so we questioned whether bactericidal activity of these cells was energy-dependent. However, NaN3-pretreated A. baumanni cells had no difference in F-Hst 5 killing (Figure 5C), indicating that Hst 5-mediated killing of A. baumanni is partially due to membrane lysis and is energy independent, although some portion of killing might also be a result of its effect on intracellular targets.
Figure 5
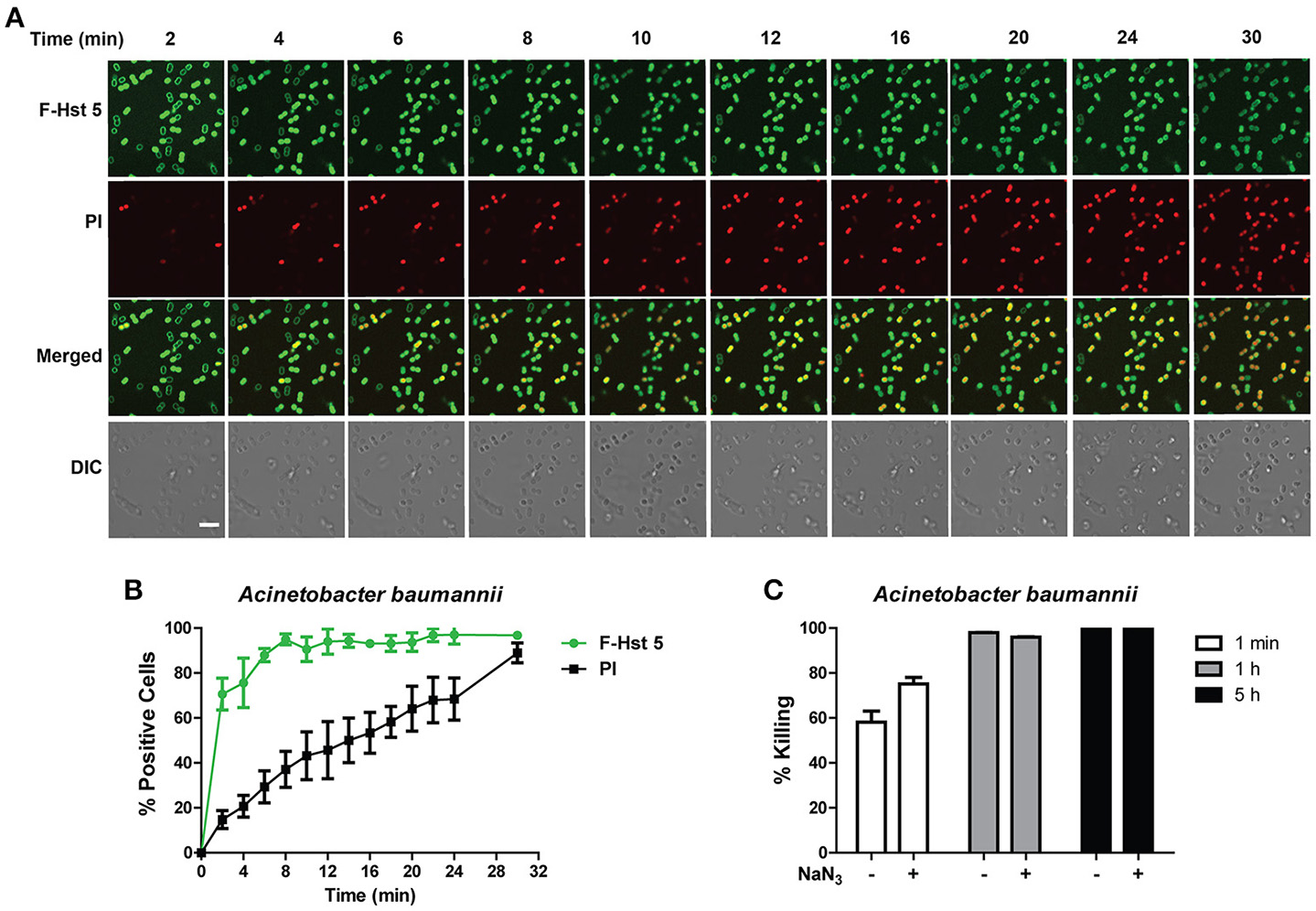
Activity of Hst 5 against A. baumannii is mediated in part by membrane disruption. (A) A. baumannii cells in exponential phase were exposed to F-Hst 5 (30 μM) and PI (2 μg/mL). The F-Hst 5 (green) and PI (red) uptake were measured in parallel by time-lapse confocal microscopy. Images were recorded every 2 min and selected images as indicated time points were shown. (Arrow, cells of F-Hst 5 uptake positive but without PI uptake; Scale bar: 5 μm). (B) Quantitative analysis of F-Hst 5 uptake (green line) and PI uptake. Error bars represent the standard errors from four different fields of image. (C) Cells pretreated with 10 mM NaN3 at 37°C for 3 h did not show significant difference in susceptibility to Hst 5.
Hst 5-mediated killing of S. aureus is delayed and energy independent
F-Hst 5 was visualized binding to the cell surface of all S. aureus cells within 1 min after addition, however intracellular localization and PI uptake occurred slowly (Figure 6A), so that by 120 min only 7% of cells contained F-Hst 5 and were PI positive (Figure 6B). This was surprising since bactericidal activity of Hst 5 after 1 h incubation was found to have 60% killing (Figure 1). Furthermore, bactericidal assay with S. aureus pretreated with 10 mM NaN3 did not decrease the killing efficiency (Figure 6C). These results, combined with the salt-insensitive killing, suggest that there may be multiple targets for Hst 5-mediated killing of S. aureus that are non-lytic and energy independent.
Figure 6
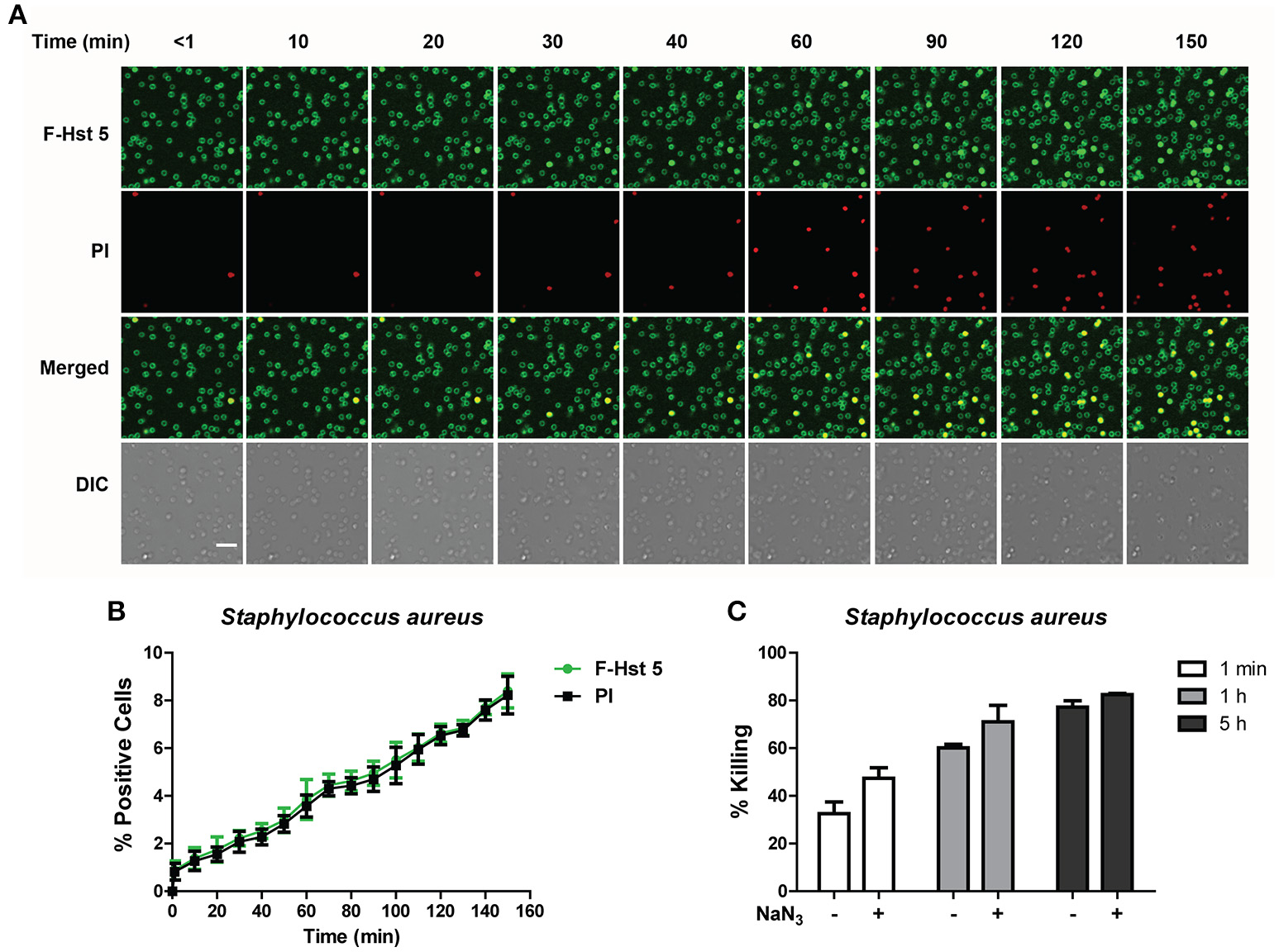
The activity of Hst 5 against S. aureus is mediated by energy-independent mechanisms. (A) S. aureus cells were exposed to F-Hst 5 (30 μM) and PI (2 μg/mL). F-Hst 5 (green) and PI (red) uptake were measured in parallel by time-lapse confocal microscopy. Images were recorded every 10 min and selected images of indicated time points were shown. (Scale bar: 5 μm). (B) Quantitative analysis of F-Hst 5 uptake (green line) and PI uptake. Error bars represent the standard errors from four different fields of image. (C) Cells pretreated with 10 mM NaN3 at 37°C for 3 h did not show significant difference in susceptibility to Hst 5.
Hst 5 is ineffective in killing K. pneumoniae due to lack of sustained binding
Since Hst 5 showed negligible killing against K. pneumoniae (Figure 1C), we examined the reason for this using confocal microscopy. F-Hst bound to most K. pneumoniae cells within 2 min, however by 10 min most binding was lost suggesting detachment of Hst 5 from the surface capsule (Figure 7A). As expected, F-Hst 5 and PI uptake were extremely limited (only 2% of cells), even up to 150 min (Figure 7B). These results suggest that F-Hst 5 is released from the K. pneumoniae after initial binding (perhaps due to lower binding efficacy in the presence of its capsule) and thus Hst 5 is unable to gain entry or lyse the cells and therefore is ineffective against K. pneumoniae.
Figure 7
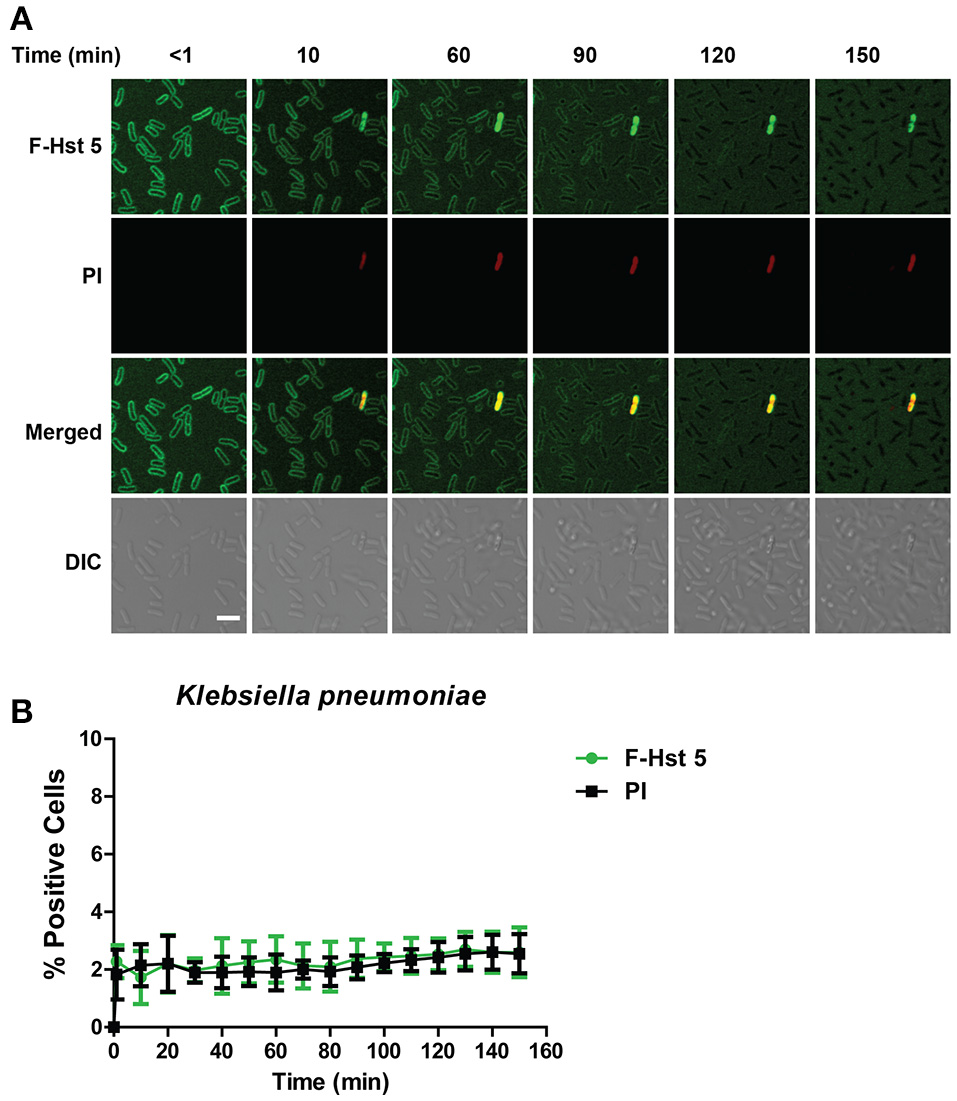
Hst 5 is minimally active against K. pneumoniae. (A) K. pneumoniae cells in exponential phase were exposed to F-Hst 5 (30 μM) and PI (2 μg/mL). F-Hst 5 (green) and PI (red) uptake were measured in parallel by time-lapse confocal microscopy. Images were recorded every 10 min and selected images of indicated time points were shown. (Scale bar: 5 μm). (B) Quantitative analysis of F-Hst 5 uptake (green line) and PI uptake. Error bars represent the standard errors from four different fields of image.
Hst 5-Spd has improved bactericidal efficiency against ESKAPE pathogens that take up Hst 5
We have previously reported that spermidine-conjugated Hst 5 (Hst 5-Spd) has greater activity against C. albicans because the spermidine conjugate translocates more efficiently into the yeast cells (Tati et al., 2014). Therefore, to determine if Hst 5-Spd has improved bactericidal activity against ESKAPE pathogens as well, we tested the killing activity of Hst 5-Spd [30 μM] against all the six strains (Figure 8). Interestingly, Hst 5-Spd had significantly higher killing activity compared with Hst 5 against E. faecium, E. cloacae, and A. baumannii (Figure 8). Hst 5-Spd bactericidal effects were most improved against E. faecium; increased by three-fold after 1 h incubation and by 1.5-fold after 5 h, while Spermidine (Spd) alone had no killing activity even after 5 h treatment (Figure 8A). Hst 5-Spd bactericidal activity was also increased significantly in E. cloacae compared with Hst 5, although by only 15% at 1 h and at 5 h. Hst 5-Spd also showed a small but significant increase in killing activity against A. baumannii compared with Hst 5 (23% at 1 min and 10% at 1 h). However, after 5 h there was no significant difference perhaps due to the high killing that occurred as a result of Spd (30 μM) itself (Figure 8D). These three species of ESKAPE pathogens were also ones that we found to involve Hst 5 uptake and intracellular targets (Figures 2, 3, 5), suggesting that improved Hst 5-Spd activity in these strains is a result of improved uptake. This is supported by our data showing that the killing ability of Hst 5-Spd against P. aeruginosa was not improved, in agreement with a membrane lytic mechanism for this organism. Furthermore, the Hst 5 resistant pathogen K. pneumoniae was not affected by Hst 5-Spd showing that this conjugate does not improve binding to this encapsulated ESKAPE pathogen.
Figure 8
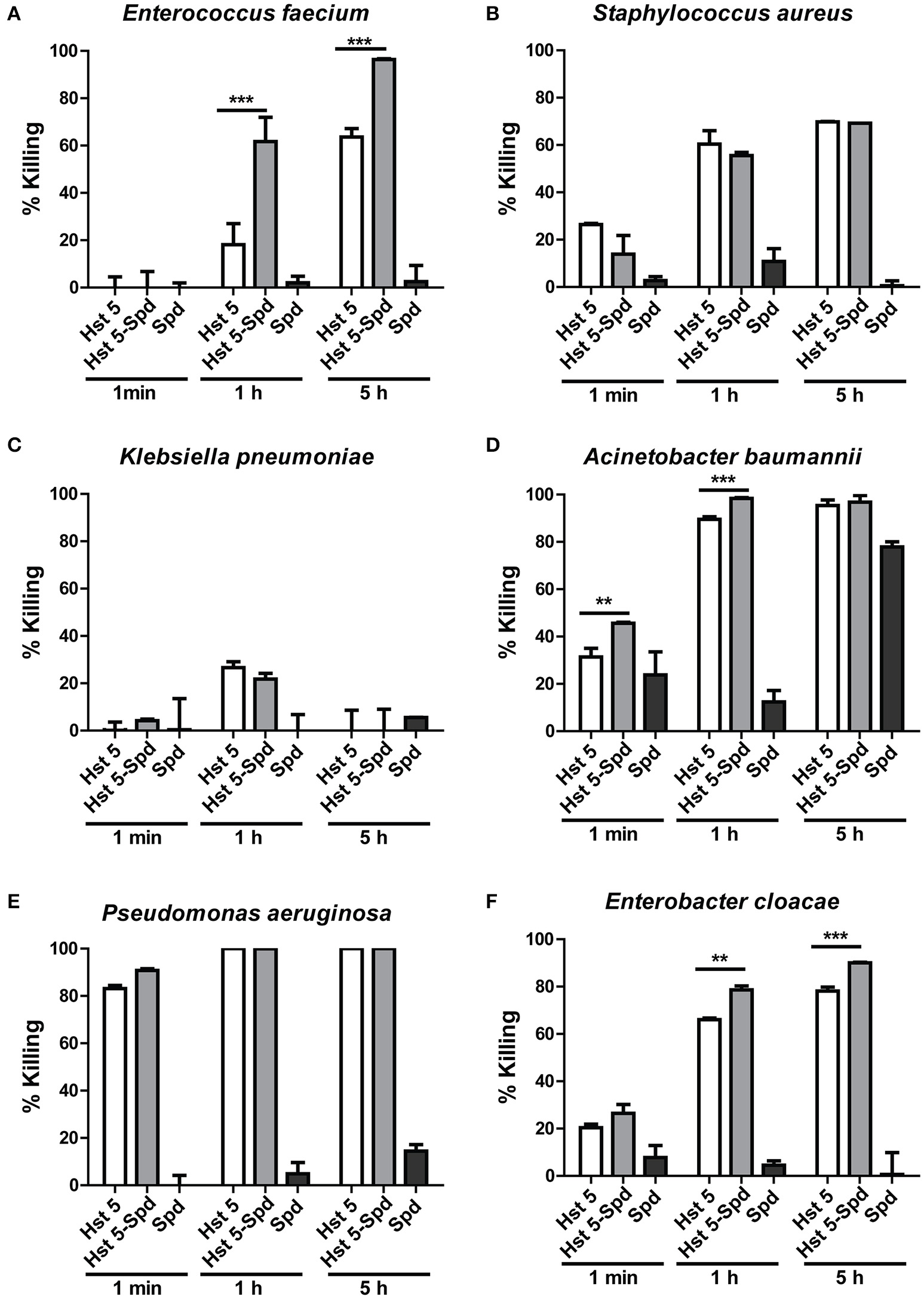
Killing of ESKAPE pathogens by Hst 5-Spd. E. faecium(A), S. aureus(B), K. pneumoniae(C), A. baumannii(D), P. aeruginosa(E), and E. cloacae(F) cells in exponential growth were exposed to 30 μM of Hst 5, F-Hst 5 and spermidine in 10 mM NaPB for 1 min, 1, and 5 h. Aliquots taken at different time points were diluted and plated. CFU were determined after 24 h. Error bars represent the standard errors from at least three independent replicates of each strain. Hst 5-Spd conjugate showed more killing efficiency against E. faecium(A), A. baumannii(D), and E. cloacae(F) (**P < 0.01, ***P < 0.001, Student's t-test).
Discussion
Here we report the remarkable finding that although Hst 5 activity has been believed to be limited to fungi, Hst 5 also demonstrates very high bactericidal activity against several ESKAPE pathogens. Up to now, a few reports have described interactions between Hst 5 and bacteria. Hst 5 was found to have low killing against Streptococcus gordonii (a Gram-positive commensal bacterium of the human oral cavity; Andrian et al., 2012). Zinc-mediated killing of E. faecalis bacteria by histidine–rich histatin analogs was found (Rydengard et al., 2006), and killing activity of another histatin derivative (P113) against P. aeruginosa and S. aureus in vitro was shown (Giacometti et al., 2005). Hst 5 may have other non-bactericidal activities in that it attenuates chemokine responses by binding to Porphyromonas gingivalis hemagglutinin B (Borgwardt et al., 2014). Here we expand the scope of Hst 5 activity by showing that full length Hst 5 and an Hst 5 spermidine conjugate both exert very significant killing of all but one ESKAPE pathogen by multiple mechanisms.
The mechanisms by which Hst 5, a naturally occurring salivary protein, kills C. albicans have been well-studied and suggest the involvement of multiple intracellular targets (Puri and Edgerton, 2014). The effects on fungal targets range from non-lytic leakage of ATP and K+ ions to mitochondrial damage and oxidative stress generation (Puri and Edgerton, 2014); and more recently, potential metal scavenging causing nutritional stress (Puri et al., 2015). However, it has been clearly shown that the killing is non-lytic and energy dependent. Interestingly, the ambiguity in the mechanisms of antimicrobial peptides seems universal. Many antibacterial peptides that were considered to be purely lytic are now believed to involve other killing mechanisms ranging from bacterial cell wall disruption to effects on protein and nucleic acid synthesis (Brogden, 2005). We found that Hst 5 can efficiently kill ESKAPE pathogens killing using a unique blend of lysis and energy dependency.
Antibiotic resistance has been a challenge for treating drug resistant bacterial infections and ESKAPE pathogens are no exception (Rice, 2010). One classic drug resistance mechanism entails efflux of the antimicrobial molecules by bacterial cells (Sun et al., 2014). However, Hst 5 resistance is mediated by efflux through C. albicans Flu1 transporters (Li et al., 2013) and is not likely to occur in bacteria, as there are no similar transporters known in bacteria. In contrast, C. albicans Dur3 transporters (used in polyamine uptake in fungi) are needed for Hst 5 uptake and candidacidal activity (Kumar et al., 2011). Interestingly all ESKAPE pathogens have spermidine/polyamine transporter homologs (Palmer et al., 2010; Ren et al., 2010; Park et al., 2011; Liu et al., 2012; Yao and Lu, 2014; Wang et al., 2016) that might mediate Hst 5 uptake in pathogens that require internalization for killing. Also, the improved bactericidal activity of the Hst 5-spermidine conjugate against E. faecium (whose spermidine transporter has the highest homology to the C. albicans Dur3 transporter, our unpublished data), and to some extent against E. cloacae and A. baumannii may be a result of better uptake due to similarity of polyamine transporters expressed in these cells. Biofilm formation is another resistance mechanism that leads to poor drug penetration and accessibility (del Pozo and Patel, 2007; Hoiby et al., 2010). It has previously been shown that Hst 5 is effective against C. albicans biofilm (Konopka et al., 2010) and thus Hst 5 can potentially make its way through the polysaccharide extracellular biofilm matrix of ESKAPE bacteria as well.
Prophylactic antimicrobial therapy, especially when applied topically, can be of great advantage to prevent surgical, wound, and burn infections. However, depending on the specificity of the agent used, this may also lead to the killing of healthy flora at the site of application that prevents colonization of pathogens. Here we show that salivary Hst 5 that has limited antibacterial activity against most human oral commensal organisms including Streptococcal sp. (found on the human skin; Dale and Fredericks, 2005; Belkaid and Segre, 2014), is extremely effective against P. aeruginosa and A. baumanni. This provides a novel potential therapeutic application for Hst 5 since P. aeruginosa is one of the most important causative agents for burn infections (Tredget et al., 1992). Furthermore, P. aeruginosa is responsible for a majority of bacterial eye infections related to contact lens use (Cope et al., 2016). While Hst 5 is intrinsically absent from human tears, its exogenous application to treat such infections seems plausible, given the high killing activity of Hst 5 against P. aeruginosa. Potential therapeutic use of Hst 5 has some limitations that are inherent to its cationic nature. Since high salt conditions negatively affect Hst 5 microbicidal activity (Helmerhorst et al., 1999; Jang et al., 2010), this may restrict the use of Hst 5 for treatment of systemic disease, although the Hst 5 derivative P113 was found to be effective for systemic use in a rat model of P. aeruginosa sepsis (Cirioni et al., 2004). However Hst 5 has a great potential for application topically, both on human skin and in the eye, especially when carried in hypotonic gels and solutions.
Multiple drugs are sometimes required to completely eradicate drug resistant infections. Although further testing of additional strains of ESKAPE pathogens needs to be done, we show here the killing activity of Hst 5 against ESKAPE pathogens, taken together with how Hst 5 can potentially affects multiple intracellular targets, presents the possibility of using this protein in synergy with other existing antibiotics.
Funding
This work was supported by NIDCR grants DE10641 and DE022720 to ME.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Statements
Author contributions
Conceived and designed the experiments: ME, HD, SP. Performed the experiments: HD, AM, HN. Analyzed the data: HD, AM, HN. Prepared the paper: ME, HD, SP, TR.
Acknowledgments
We thank Dr. Wade J. Sigurdson for assistance in confocal microscopy studies performed in the Confocal Microscopy and Flow Cytometry Facility at University at Buffalo.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
References
1
Adams M. D. Goglin K. Molyneaux N. Hujer K. M. Lavender H. Jamison J. J. et al . (2008). Comparative genome sequence analysis of multidrug-resistant Acinetobacter baumannii. J. Bacteriol.190, 8053–8064. 10.1128/JB.00834-08
2
Andrian E. Qi G. Wang J. Halperin S. A. Lee S. F. (2012). Role of surface proteins SspA and SspB of Streptococcus gordonii in innate immunity. Microbiology158(Pt 8), 2099–2106. 10.1099/mic.0.058073-0
3
Belkaid Y. Segre J. A. (2014). Dialogue between skin microbiota and immunity. Science346, 954–959. 10.1126/science.1260144
4
Borgwardt D. S. Martin A. D. Van Hemert J. R. Yang J. Fischer C. L. Recker E. N. et al . (2014). Histatin 5 binds to Porphyromonas gingivalis hemagglutinin B (HagB) and alters HagB-induced chemokine responses. Sci. Rep.4:3904. 10.1038/srep03904
5
Boucher H. W. Talbot G. H. Bradley J. S. Edwards J. E. Gilbert D. Rice L. B. et al . (2009). Bad bugs, no drugs: no ESKAPE! An update from the Infectious Diseases Society of America. Clin. Infect. Dis.48, 1–12. 10.1086/595011
6
Brogden K. A. (2005). Antimicrobial peptides: pore formers or metabolic inhibitors in bacteria?Nat. Rev. Microbiol.3, 238–250. 10.1038/nrmicro1098
7
Cirioni O. Giacometti A. Ghiselli R. Orlando F. Kamysz W. D'Amato G. et al . (2004). Potential therapeutic role of histatin derivative P-113d in experimental rat models of Pseudomonas aeruginosa sepsis. J. Infect. Dis.190, 356–364. 10.1086/421913
8
Clinical Laboratory Standards Institute (2012). Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria That Grow Aerobically, 9th Edn.Approved standard M07-A9. Wayne, PA: Clinical and Laboratory Standards Institute.
9
Coates R. Moran J. Horsburgh M. J. (2014). Staphylococci: colonizers and pathogens of human skin. Future Microbiol.9, 75–91. 10.2217/fmb.13.145
10
Cope J. R. Collier S. A. Srinivasan K. Abliz E. Myers A. Millin C. J. et al . (2016). Contact lens-related corneal infections - United States, 2005-2015. MMWR Morb. Mortal. Wkly. Rep.65, 817–820. 10.15585/mmwr.mm6532a2
11
Dale B. A. Fredericks L. P. (2005). Antimicrobial peptides in the oral environment: expression and function in health and disease. Curr. Issues Mol. Biol.7, 119–133. 10.1093/jac/dki103
12
del Pozo J. L. Patel R. (2007). The challenge of treating biofilm-associated bacterial infections. Clin. Pharmacol. Ther.82, 204–209. 10.1038/sj.clpt.6100247
13
Devine D. A. Hancock R. E. (2002). Cationic peptides: distribution and mechanisms of resistance. Curr. Pharm. Des.8, 703–714. 10.2174/1381612023395501
14
Giacometti A. Cirioni O. Kamysz W. D'Amato G. Silvestri C. Del Prete M. S. et al . (2005). In vitro activity of the histatin derivative P-113 against multidrug-resistant pathogens responsible for pneumonia in immunocompromised patients. Antimicrob. Agents Chemother.49, 1249–1252. 10.1128/AAC.49.3.1249-1252.2005
15
Groenink J. Ruissen A. L. Lowies D. van't Hof W. Veerman E. C. Nieuw Amerongen A. V. (2003). Degradation of antimicrobial histatin-variant peptides in Staphylococcus aureus and Streptococcus mutans. J. Dent. Res.82, 753–757. 10.1177/154405910308200918
16
Helmerhorst E. J. Breeuwer P. van't Hof W. Walgreen-Weterings E. Oomen L. C. Veerman E. C. et al . (1999). The cellular target of histatin 5 on Candida albicans is the energized mitochondrion. J. Biol. Chem.274, 7286–7291. 10.1074/jbc.274.11.7286
17
Hoiby N. Bjarnsholt T. Givskov M. Molin S. Ciofu O. (2010). Antibiotic resistance of bacterial biofilms. Int. J. Antimicrob. Agents35, 322–332. 10.1016/j.ijantimicag.2009.12.011
18
Houang E. T. Sormunen R. T. Lai L. Chan C. Y. Leong A. S. (1998). Effect of desiccation on the ultrastructural appearances of Acinetobacter baumannii and Acinetobacter lwoffii. J. Clin. Pathol.51, 786–788. 10.1136/jcp.51.10.786
19
Jang W. S. Bajwa J. S. Sun J. N. Edgerton M. (2010). Salivary histatin 5 internalization by translocation, but not endocytosis, is required for fungicidal activity in Candida albicans. Mol. Microbiol.77, 354–370. 10.1111/j.1365-2958.2010.07210.x
20
Keller R. Pedroso M. Z. Ritchmann R. Silva R. M. (1998). Occurrence of virulence-associated properties in Enterobacter cloacae. Infect. Immun.66, 645–649.
21
Khan S. A. Fidel P. L. Jr. Thunayyan A. A. Varlotta S. Meiller T. F. Jabra-Rizk M. A. (2013). Impaired histatin-5 levels and salivary antimicrobial activity against C. albicans in HIV infected individuals. J. AIDS Clin. Res.4:1000193. 10.4172/2155-6113.1000193
22
Konopka K. Dorocka-Bobkowska B. Gebremedhin S. Duzgunes N. (2010). Susceptibility of Candida biofilms to histatin 5 and fluconazole. Antonie Van Leeuwenhoek97, 413–417. 10.1007/s10482-010-9417-5
23
Kumar R. Chadha S. Saraswat D. Bajwa J. S. Li R. A. Conti H. R. et al . (2011). Histatin 5 uptake by Candida albicans utilizes polyamine transporters Dur3 and Dur31 proteins. J. Biol. Chem.286, 43748–43758. 10.1074/jbc.M111.311175
24
Li R. Kumar R. Tati S. Puri S. Edgerton M. (2013). Candida albicans flu1-mediated efflux of salivary histatin 5 reduces its cytosolic concentration and fungicidal activity. Antimicrob. Agents Chemother.57, 1832–1839. 10.1128/AAC.02295-12
25
Lindsay A. K. Hogan D. A. (2014). Candida albicans: molecular interactions with Pseudomonas aeruginosa and Staphylococcus aureus. Fungal Biol. Rev.28, 85–96. 10.1016/j.fbr.2014.10.002
26
Liu P. Li P. Jiang X. Bi D. Xie Y. Tai C. et al . (2012). Complete genome sequence of Klebsiella pneumoniae subsp. pneumoniae HS11286, a multidrug-resistant strain isolated from human sputum. J. Bacteriol.194, 1841–1842. 10.1128/JB.00043-12
27
Lopes A. E. R. Canini S. R. M. D. Reinato L. A. F. Lopes L. P. Gir E. (2015). Prevalence of gram-positive bacteria in patients with HIV in specialized services. Acta Paulista De Enfermagem28, 281–286. 10.1590/1982-0194201500047
28
Luo G. Lin L. Ibrahim A. S. Baquir B. Pantapalangkoor P. Bonomo R. A. et al . (2012). Active and passive immunization protects against lethal, extreme drug resistant-Acinetobacter baumannii infection. PLoS ONE7:e29446. 10.1371/journal.pone.0029446
29
Magill S. S. Edwards J. R. Bamberg W. Beldavs Z. G. Dumyati G. Kainer M. A. et al . (2014). Multistate point-prevalence survey of health care-associated infections. N. Engl. J. Med.370, 1198–1208. 10.1056/NEJMoa1306801
30
Palmer K. L. Carniol K. Manson J. M. Heiman D. Shea T. Young S. et al . (2010). High-quality draft genome sequences of 28 Enterococcus sp. isolates. J. Bacteriol.192, 2469–2470. 10.1128/JB.00153-10
31
Park J. Y. Kim S. Kim S. M. Cha S. H. Lim S. K. Kim J. (2011). Complete genome sequence of multidrug-resistant Acinetobacter baumannii strain 1656-2, which forms sturdy biofilm. J. Bacteriol.193, 6393–6394. 10.1128/JB.06109-11
32
Podschun R. Ullmann U. (1998). Klebsiella spp. as nosocomial pathogens: epidemiology, taxonomy, typing methods, and pathogenicity factors. Clin. Microbiol. Rev.11, 589–603.
33
Puri S. Edgerton M. (2014). How does it kill?: understanding the candidacidal mechanism of salivary histatin 5. Eukaryotic Cell13, 958–964. 10.1128/EC.00095-14
34
Puri S. Li R. Ruszaj D. Tati S. Edgerton M. (2015). Iron binding modulates candidacidal properties of salivary histatin 5. J. Dent. Res.94, 201–208. 10.1177/0022034514556709
35
Ren Y. Ren Y. Zhou Z. Guo X. Li Y. Feng L. et al . (2010). Complete genome sequence of Enterobacter cloacae subsp. cloacae type strain ATCC 13047. J. Bacteriol.192, 2463–2464. 10.1128/JB.00067-10
36
Rice L. B. (2008). Federal funding for the study of antimicrobial resistance in nosocomial pathogens: no ESKAPE. J. Infect. Dis.197, 1079–1081. 10.1086/533452
37
Rice L. B. (2010). Progress and challenges in implementing the research on ESKAPE pathogens. Infect. Control Hosp. Epidemiol.31, S7–S10. 10.1086/655995
38
Rydengard V. Andersson Nordahl E. Schmidtchen A. (2006). Zinc potentiates the antibacterial effects of histidine-rich peptides against Enterococcus faecalis. FEBS J.273, 2399–2406. 10.1111/j.1742-4658.2006.05246.x
39
Salvatori O. Puri S. Tati S. Edgerton M. (2016). Innate immunity and saliva in Candida albicans-mediated oral diseases. J. Dent. Res.95, 365–371. 10.1177/0022034515625222
40
Sands K. M. Wilson M. J. Lewis M. A. Wise M. P. Palmer N. Hayes A. J. et al . (2016). Respiratory pathogen colonization of dental plaque, the lower airways, and endotracheal tube biofilms during mechanical ventilation. J. Crit. Care37, 30–37. 10.1016/j.jcrc.2016.07.019
41
Sun J. Deng Z. Yan A. (2014). Bacterial multidrug efflux pumps: mechanisms, physiology and pharmacological exploitations. Biochem. Biophys. Res. Commun.453, 254–267. 10.1016/j.bbrc.2014.05.090
42
Tati S. Li R. Puri S. Kumar R. Davidow P. Edgerton M. (2014). Histatin 5-spermidine conjugates have enhanced fungicidal activity and efficacy as a topical therapeutic for oral candidiasis. Antimicrob. Agents Chemother.58, 756–766. 10.1128/AAC.01851-13
43
Tredget E. E. Shankowsky H. A. Joffe A. M. Inkson T. I. Volpel K. Paranchych W. et al . (1992). Epidemiology of infections with Pseudomonas aeruginosa in burn patients: the role of hydrotherapy. Clin. Infect. Dis.15, 941–949. 10.1093/clind/15.6.941
44
Vu J. Carvalho J. (2011). Enterococcus: review of its physiology, pathogenesis, diseases and the challenges it poses for clinical microbiology. Front. Biol.6:357. 10.1007/s11515-011-1167-x
45
Wang C. Liu X. Wang J. Zhou J. Cui Z. Zhang L. H. (2016). Design and characterization of a polyamine derivative inhibiting the expression of type III secretion system in Pseudomonas aeruginosa. Sci. Rep.6:30949. 10.1038/srep30949
46
Yao X. Lu C. D. (2014). Functional characterization of the potRABCD operon for spermine and spermidine uptake and regulation in Staphylococcus aureus. Curr. Microbiol.69, 75–81. 10.1007/s00284-014-0556-1
Summary
Keywords
ESKAPE, antimicrobial peptide, Histatin 5, Candida albicans , bactericidal activity
Citation
Du H, Puri S, McCall A, Norris HL, Russo T and Edgerton M (2017) Human Salivary Protein Histatin 5 Has Potent Bactericidal Activity against ESKAPE Pathogens. Front. Cell. Infect. Microbiol. 7:41. doi: 10.3389/fcimb.2017.00041
Received
13 October 2016
Accepted
03 February 2017
Published
15 February 2017
Volume
7 - 2017
Edited by
Ghassan M. Matar, American University of Beirut, Lebanon
Reviewed by
Gill Diamond, University of Florida, USA; Jens Kreth, Oregon Health & Science University, USA
Updates
Copyright
© 2017 Du, Puri, McCall, Norris, Russo and Edgerton.
This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) or licensor are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Mira Edgerton edgerto@buffalo.edu
Disclaimer
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article or claim that may be made by its manufacturer is not guaranteed or endorsed by the publisher.