- 1Department of Medicine, Austin Health, Melbourne Medical School, University of Melbourne, Heidelberg, VIC, Australia
- 2Department of Medicine, Royal Melbourne Hospital, The University of Melbourne, Melbourne, VIC, Australia
- 3Centre for Epidemiology and Biostatistics, Melbourne School of Population and Global Health, The University of Melbourne, Melbourne, VIC, Australia
- 4Department of Endocrinology, Austin Health, Heidelberg, VIC, Australia
Objective: Flash glucose monitoring (FlashGM) is a sensor-based technology that displays glucose readings and trends to people with diabetes. In this meta-analysis, we assessed the effect of FlashGM on glycaemic outcomes including HbA1c, time in range, frequency of hypoglycaemic episodes and time in hypo/hyperglycaemia compared to self-monitoring of blood glucose, using data from randomised controlled trials.
Methods: A systematic search was conducted on MEDLINE, EMBASE and CENTRAL for articles published between 2014 and 2021. We selected randomised controlled trials comparing flash glucose monitoring to self-monitoring of blood glucose that reported change in HbA1c and at least one other glycaemic outcome in adults with type 1 or type 2 diabetes. Two independent reviewers extracted data from each study using a piloted form. Meta-analyses using a random-effects model was conducted to obtain a pooled estimate of the treatment effect. Heterogeneity was assessed using forest plots and the I2 statistic.
Results: We identified 5 randomised controlled trials lasting 10 – 24 weeks and involving 719 participants. Flash glucose monitoring did not lead to a significant reduction in HbA1c. However, it resulted in increased time in range (mean difference 1.16 hr, 95% CI 0.13 to 2.19, I2 = 71.7%) and decreased frequency of hypoglycaemic episodes (mean difference -0.28 episodes per 24 hours, 95% CI -0.53 to -0.04, I2 = 71.4%).
Conclusions: Flash glucose monitoring did not lead to a significant reduction in HbA1c compared to self-monitoring of blood glucose, however, it improved glycaemic management through increased time in range and decreased frequency of hypoglycaemic episodes.
Systematic Review Registration: https://www.crd.york.ac.uk/prospero/, identifier PROSPERO (CRD42020165688).
Introduction
Flash glucose monitoring (FlashGM) technology has revolutionised diabetes management. Its rising popularity has driven a need to assess its impact on key markers of glycaemic management. Hitting glycaemic targets is key to the success of diabetes management and yet, up to 92% of people with diabetes fail to achieve these targets (1). As a result, these individuals are susceptible to micro and macrovascular complications of diabetes, as well as excessive morbidity and increased risk of death (2–4). FlashGM is a sensor-based technology that displays current glucose levels, glucose readings from the past 8 hours and trend arrows (5). Unlike self-monitoring of blood glucose (SMBG), FlashGM produces an ambulatory glucose profile that displays key trends in hypo-, normo- and hyperglycaemia (5). It contributes to clinical care by providing clear, comprehensive glucose data with minimal inconvenience. Such information can be used to guide patient choice, clinical practice and future reimbursement of FlashGM (6).
Previous systematic reviews on FlashGM have reported inconsistent findings (7–9). They have all included observational studies without a control group as a comparator. To minimise bias and confounding factors, we examined randomised controlled trials (RCTs) to assess the effects of FlashGM on glycaemic management. Current literature largely focuses on HbA1c as the main metric of treatment efficacy (8, 9). However, we recognise that glycaemic management has multiple dimensions including time in range, frequency of hypoglycaemic episodes and time in hypo/hyperglycaemia. Less time in range is associated with the increased risk of microvascular complications (10, 11) and hypoglycaemia is linked to life-threatening outcomes including neurocognitive dysfunction and cardiovascular dysfunction (12, 13). To ensure a comprehensive analysis of FlashGM, we assessed these glycaemic outcomes in addition to change in HbA1c.
The aims of this meta-analysis were therefore to assess the effect of FlashGM compared to SMBG on HbA1c, time in range (3.9 – 10.0mmol/L), time in hypoglycaemia (<3.9mmol/L), occurrence of hypoglycaemic episodes and time in hyperglycaemia (>10mmol/L) over a span of 10 to 24 weeks. Our primary hypothesis was that FlashGM leads to reduced HbA1c in individuals with type 1 and 2 diabetes. Our secondary hypotheses were that FlashGM leads to: (i) increase in time in range (3.9 – 10mmol/L), (ii) reduction in time in hypoglycaemia (<3.9mmol/L), (iii) reduction in frequency of hypoglycaemic episodes and (iv) reduction in time in hyperglycaemia (>10mmol/L).
Materials and Methods
This systematic review was registered in PROSPERO (CRD42020165688) and performed in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses statement (14) (Supplementary Figure 1).
Data Sources and Searches
A literature search was conducted on relevant databases including MEDLINE, EMBASE and the Cochrane Central Register of Controlled Trials (CENTRAL). The reference lists of retrieved studies were assessed for further relevant studies. With the help of an expert librarian, we developed a search strategy from Medical Subject Headings (MeSH) and text words related to diabetes, flash glucose monitoring and blood glucose from 1st January 2014 until 13th September 2021 (Supplementary Figure 2). The Cochrane Highly Sensitive Search Strategy was used to restrict the search to randomised controlled trials.
Study Selection
Using Covidence software, two review authors (BL and MH) independently screened titles and abstracts for relevant studies. Covidence was used to exclude the duplicates and the remaining studies were assessed for eligibility by predetermined selection criteria. This was defined as: 1) participants aged ≥ 18 years, 2) type 1 or 2 diabetes, 3) use of FlashGM device as one of the intervention groups, 4) use of SMBG in control group, 5) report of HbA1c (%) and at least one other glycaemic outcome such as time in range (hours or percentage), time in hypo/hyperglycaemia (minutes or percentage) or frequency of hypoglycaemic events per 24 hours, 6) at least 10 weeks duration of intervention and 7) randomised controlled trials. A hypoglycaemic episode was defined by glucose readings below 3.9 mmol/L for at least 15 minutes (15). The end of the episode was defined by readings above 3.9 mmol/L for 15 minutes (15). Studies that blinded participants in the intervention group to sensor glucose data were excluded because it is not reflective of FlashGM use in clinical practice. Access to glucose readings facilitates self-management behaviours as individuals may alter food intake, exercise or medication according to their glucose level (16–18). The influence of FlashGM on behaviour is key to assessing its efficacy in glycaemic management (16). We also excluded studies that used results from other trials and did not have their own original data. Since FlashGM was introduced in 2014, we only included studies published from 2014 onwards. Studies were not excluded on the basis of publication status or on the basis of language.
Data Extraction and Quality Assessment
Studies that met the eligibility criteria were retrieved for full-text assessment. The data from included studies were extracted independently by two review authors (BL and MH) using a piloted form. Results were compared for accuracy and any differences were resolved by consensus. The following data was extracted from each study: 1) author(s) and publication year 2) study population 3) location of study 4) participants’ baseline characteristics such as age, diabetes type, type of treatment regimen and initial HbA1c 5) duration of study 6) glycaemic endpoints including HbA1c (mmol/mol), time in range (hours) and time in hypo/hyperglycaemia (hours) or frequency of hypoglycaemic events depending on availability of data and 7) mean difference and standard error of extracted glycaemic outcomes. The corresponding authors of four studies were contacted via email for missing data but no further information on desired outcomes was provided.
The Cochrane risk-of-bias tool (RoB 2) for randomised trials was used to assess the design, conduction and reporting of included studies. Two review authors (BL and MH) independently conducted the assessment, compared results and resolved differences by consensus. Bias was judged based on five domains: randomisation process, deviations from intended interventions, missing outcome data, measurement of outcome and selection of reported result. Each domain required answers to signalling questions to evaluate the potential risk of bias. The level of bias in each of the five domains was summarised to generate the overall risk of bias and classified as having low bias, high bias, or some concerns regarding bias.
Data Synthesis and Analysis
Changes from baseline in HbA1c, time in range, time in hypo/hyperglycaemia and number of hypoglycaemic episodes per 24 hours were analysed as continuous variables using mean difference between groups and standard error as summary measures. When variability was reported in confidence intervals (CI), standard error (SE) was estimated using the following formula: SE = (upper limit – lower limit)/3.92. A random effects meta-analysis was conducted and effect sizes were reported in the form of mean difference and 95% confidence interval. The I2 statistic was used to quantify the percentage of variability in effect size that was due to heterogeneity. In this meta-analysis, low heterogeneity was defined as 40% or less, moderate heterogeneity was between 30% and 60%, substantial heterogeneity was 50% to 90% (19). For all outcomes, we pre-planned subgroup analyses according to type 1 and type 2 diabetes. However, only one study focused exclusively on type 1 diabetes, thereby precluding subgroup analysis. It should also be noted that time in range and time in hypo/hyperglycaemia were meta-analysed and reported in hours as hours was the unit originally reported in the included as publications. All statistical analyses were performed using STATA, version 16, (STATA, College Station, Texas).
Results
Search Results
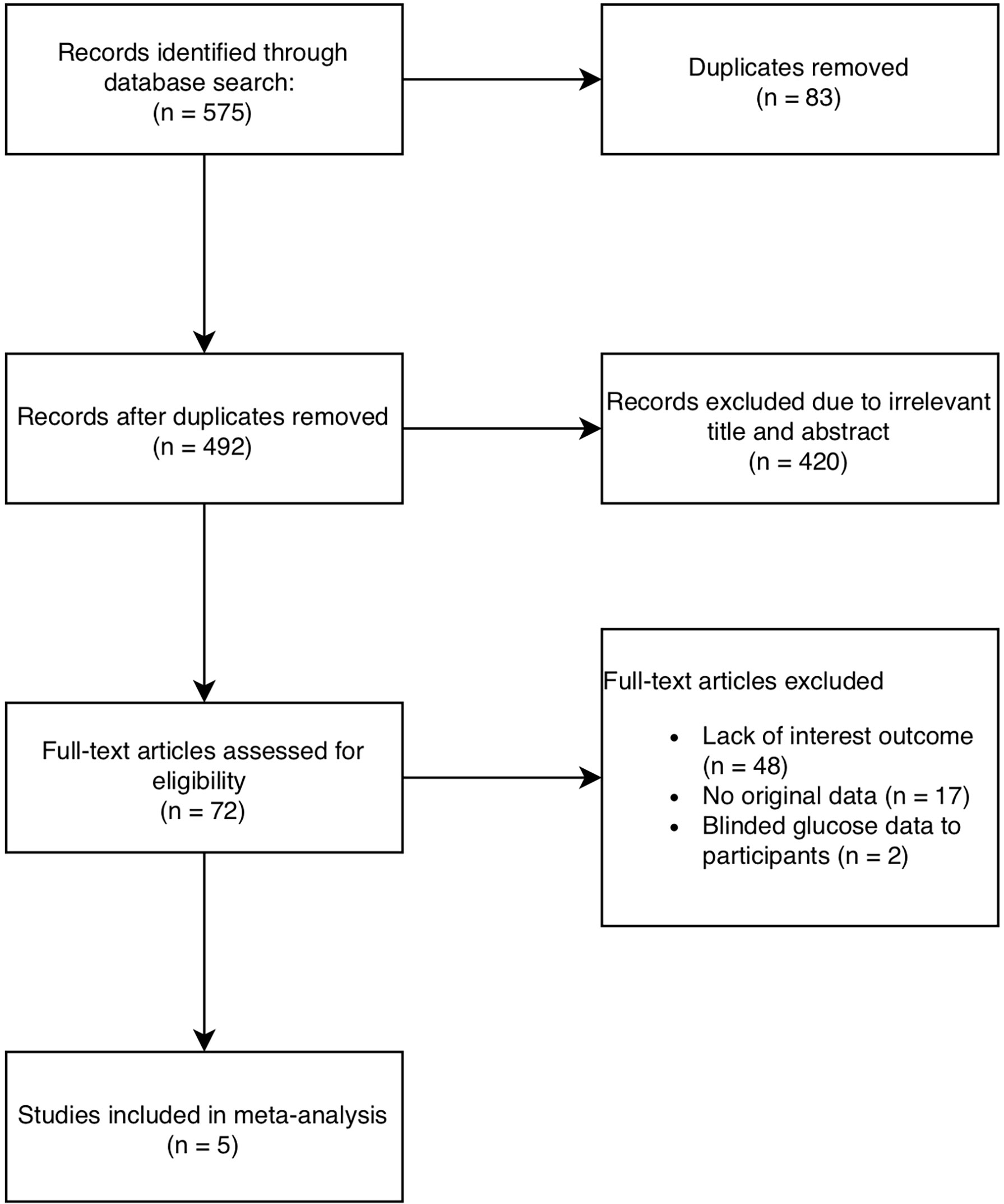
Five hundred and seventy-five articles were identified using MEDLINE, EMBASE and CENTRAL and 492 records remained after duplicates were removed. Five RCTs including a total of 719 participants (400 in the intervention group and 319 in the control group), were included in the meta-analysis (20–24) (Figure 1).
Study and Patient Characteristics
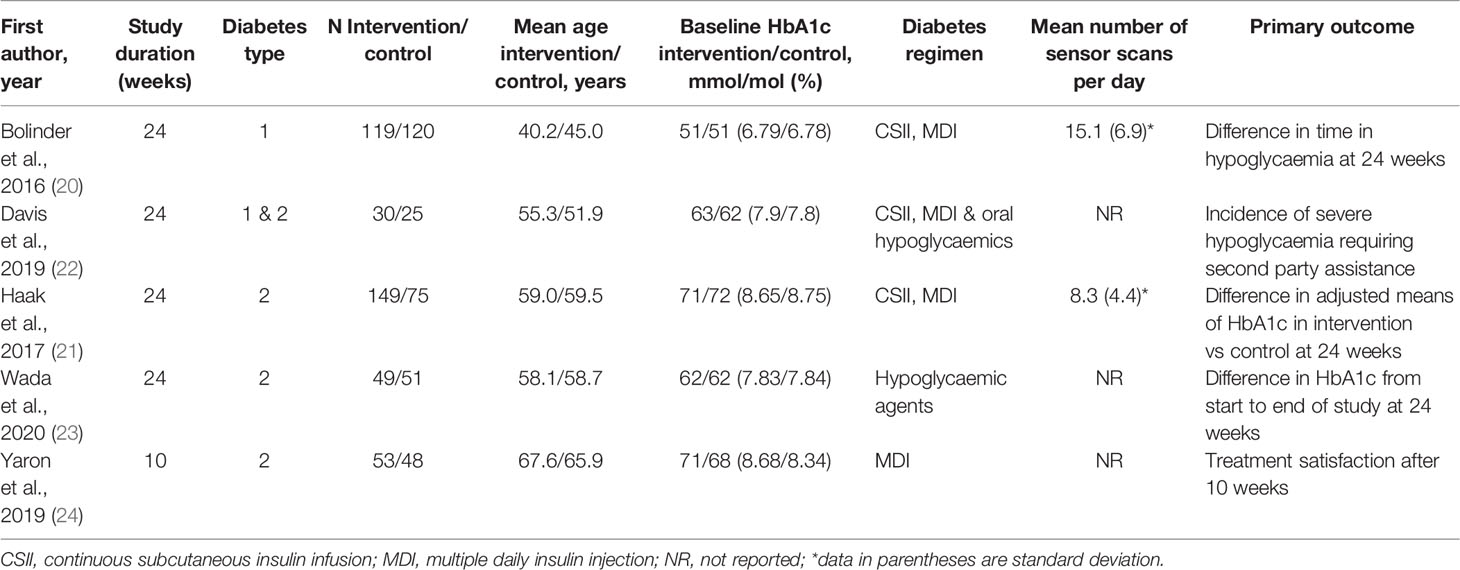
Table 1 shows the characteristics of the included RCTs with baseline characteristics of participants. The trials were published between 2016 and 2020 and recruited participants from Europe, Australia, Japan and Israel. All studies had an open-label design and only included adult participants. All had a duration of 24 weeks except for one, which was 10 weeks long (24). Three studies were conducted on participants on insulin only (20, 21, 24), one study involved participants on both insulin and oral glucose lowering medications (22) and one study was on participants using oral glucose lowering medications only (23). One study was undertaken in people with type 1 diabetes (20), three studies focused on participants with type 2 diabetes (21, 23, 24) and one study assessed both individuals with type 1 and type 2 diabetes (22). Mean patient age ranged from 40.2 to 67.6 years and mean baseline HbA1c levels (%) (SD) ranged from 50.6mmol/mol (6.78%) (0.64) to 72.1mmol/mol (8.75%) (0.98).
Risk of Bias
According to the Cochrane risk-of-bias tool, the risk of bias was evaluated as low in all studies except for one, which had a randomisation process that raised concerns (21) (Supplementary Figure 3). The lack of blinding between participants and personnel was also a potential source of bias. However, it is impractical to blind FlashGM usage as the glucose data informs personal choices regarding self-management (16–18). Blinding FlashGM data would not reflect real life settings and would impinge on the generalisability of our results.
Change in HbA1c
Five studies with 719 participants (400 in the intervention group and 319 in the control group) were pooled for the primary outcome of change in HbA1c. There was no statistically significant decrease in HbA1c at endpoint in the FlashGM group compared to SMBG (mean difference: -0.17%, 95% CI -0.41 to 0.07, p = 0.164). Studies included for analysis had high heterogeneity (I2 = 87.2%, P <0.001) (Figure 2A).
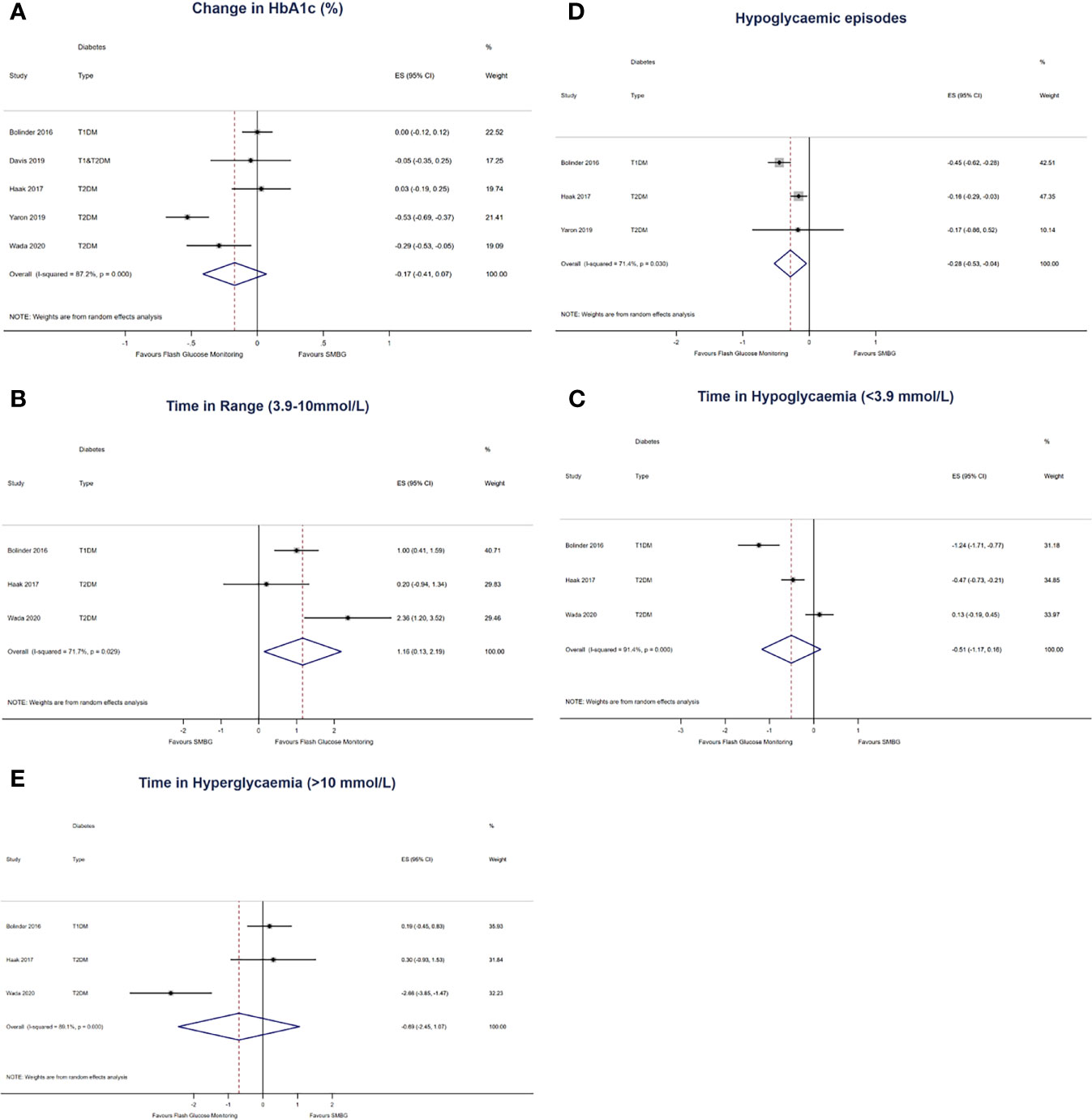
Figure 2 Forest plots of the effect size of (A) first-row left: change in HbA1c from baseline to last follow-up, expressed in % in all 5 comparisons. (B) first-row right: change in time in range (3.9 – 10mmol/L) per 24 hours, expressed in hours (C) second-row left: change in time in hypoglycaemia (<3.9mmol/L) per 24 hours, expressed in hours (D) second-row right: change in number of hypoglycaemic episodes per 24 hours (E) third-row left: change in hyperglycaemia (>10mmol/L), expressed in hours. The results are expressed as mean differences, compared to participants that used SMBG.
Time in Range
Three studies with 563 participants (317 in the intervention group and 246 in the control group) were pooled for the outcome of time in range (20, 21, 23). Compared to SMBG, FlashGM was associated with a statistically significant increase in time spent in target glucose range (mean difference: 1.16 hr, 95% CI 0.13 to 2.19, p = 0.027). There was substantial heterogeneity between comparisons (I2 = 71.7%, p = 0.029) (Figure 2B).
Time in Hypoglycaemia
For this outcome, three studies with 563 participants (317 in the intervention group and 246 in the control group) were included (20, 21, 23). There was no statistically significant decrease in time spent in hypoglycaemia in the FlashGM group compared to SMBG (mean difference: -0.51 hr, 95% CI -1.17 to 0.16, p = 0.137). Overall, there was high heterogeneity between studies that assessed time in hypoglycaemia (I2 = 91.4%, P <0.001) (Figure 2C).
Hypoglycaemic Episodes
The outcome of frequency of hypoglycaemic episodes per 24 hours was based on data from three studies with 564 participants (321 in the intervention group and 243 in the control group) (20, 21, 24). Overall analysis of FlashGM showed a decrease in frequency of hypoglycaemic episodes per 24 hours compared to the control (mean difference: -0.28 episodes per 24 hours, 95% CI -0.53 to -0.04, p = 0.022). Heterogeneity between comparisons was substantial (I2 = 71.4%, p = 0.030) (Figure 2D).
Time in Hyperglycaemia
Three studies with 563 participants (317 in the intervention group and 246 in the control group) were pooled for the outcome of time in hyperglycaemia (20, 21, 23). There was no difference between time in hyperglycaemia in the FlashGM group and the control group (mean difference: -0.69 hr, 95% CI -2.45 to 1.07, p = 0.440). There was high heterogeneity between the comparisons (I2 = 89.1%, P <0.001) (Figure 2E).
Discussion
Main Findings
In this meta-analysis, we found that the use of FlashGM did not result in a significant reduction in HbA1c, however, compared to SMBG, FlashGM resulted in improvements in two key glycaemic markers: time in range and frequency of hypoglycaemic episodes per 24 hours. The use of FlashGM led to a 1.16 hour per day increase in time in range and a 0.28 reduction in number of hypoglycaemic episodes per 24 hours compared to SMBG. FlashGM was not associated with decreased time in hypoglycaemia or hyperglycaemia. Based on this systematic review and meta-analysis, there is insufficient evidence to conclude that FlashGM results in a reduction in HbA1c compared to self-monitoring of blood glucose.
The study findings suggest that FlashGM has a stabilising effect on glucose levels, leading to extended time in range and less frequent hypoglycaemic episodes. HbA1c has been considered the gold standard glycaemic outcome that measures blood glucose in the last three months (19). However, it does not account for the intra- and interday glucose levels like time in range does (25–27). Uncontrolled glucose excursions have short- and long-term repercussions for individuals with diabetes (28, 29). FlashGM enables users to have instant access to glucose data with a painless scan. It promotes self-efficacy and increased self-care behaviours as individuals can make insulin and behavioural adjustments in a timely manner (16–18). Thus, FlashGM aids day-to-day glycaemic management as it improves awareness of glucose levels and gives individuals an immediate opportunity to correct glucose values that are outside of the target range (16–18).
Clinical Implications
FlashGM enables individuals with diabetes to access a comprehensive and personalised glucose profile. Previous studies have demonstrated that an increased frequency of scanning using FlashGM is associated with superior glycaemic management (30–33). Unlike SMBG, individuals do not need to undergo the troublesome procedure of finger-pricking (5). The continuum of glucose data enables people with diabetes to make more informed choices whether it be about food intake, exercise or insulin dosage (16–18). One study showed that participants increased their levels of physical activity after the introduction of FlashGM (16). Easy access to the glucose profile helps to improve self-awareness of exercise levels. Through its data-collecting capacity, FlashGM influences behaviour and encourages individuals to stabilise their glucose levels (16–18, 34).
Comparison to Other Studies
Previous meta-analyses have reported on the HbA1c-reducing effect of FlashGM (7, 9). However, these meta-analyses are susceptible to bias due to their heavy reliance on observational studies (7, 9). Systematic reviews that have included more observational studies tend to report a higher HbA1c reduction. The vast majority of these studies did not have a control group which made them more prone to selection and performance bias. One meta-analysis drew upon the results of 26 observational studies and concluded that the mean change in HbA1c was -0.55% (9). Another systematic review that analysed 10 cohort studies and 3 RCTs reported a -0.51% reduction in HbA1c (7). The meta-analysis on two RCTs and no observational studies reported no difference in HbA1c (8). The inconsistency of findings can be explained by the tendency for observational studies to attribute larger effects than randomised trials (34, 35). In our meta-analysis, we circumvented this limitation by only including RCTs.
Literature on the impact of FlashGM on glycaemic parameters other than HbA1c is lacking. However, there is an increasing body of evidence that supports the use of additional glycaemic metrics (10, 36–38). HbA1c values are skewed in conditions such as pregnancy, anaemia and haemoglobinopathies (26). Unlike HbA1c, time in range provides insight into glucose excursions and acute episodes of hypo/hyperglycaemia (10–12, 25–27). Previous studies have demonstrated an association between decreased time in range and the development of microvascular complications (6, 11). Large-scale observational studies supported our findings that FlashGM increases time in range (32, 39, 40). Hence, FlashGM has the potential capacity to reduce harmful glucose fluctuations and to decrease the risk of developing vascular complications.
Previous publications have reported mixed findings on how FlashGM impacts time in hypoglycaemia (7, 29, 40–42). The difference in results can be due to different definitions of hypoglycaemia as some studies set the threshold at <3.9mmol/L (15, 30, 41) whilst others set it at 3.3mmol/L (43). We set the threshold <3.9mmol/L because that is the glucose concentration at which glucose counterregulatory systems are activated (26). Our meta-analysis did not find a significant decrease amongst FlashGM users and this may be attributed to multiple factors. Firstly, past conclusions have been based on observational studies that use baseline data instead of a control group. A major advantage of RCTs is that they minimise performance bias by having both groups wear a glucose sensor. Furthermore, selection bias is reduced through stratified randomisation in the included RCTs. The avoidance of such biases help to explain the findings of this meta-analysis.
Our results showed a statistically significant decrease in frequency of hypoglycaemic episodes after FlashGM was introduced. Past studies have also found an improvement in number of hypoglycaemic episodes experienced by FlashGM users (40, 44). The device equips people with the data to increase their self-awareness of low glucose levels (17). This is evidenced by an observational study that analysed the glucose data of 10,370 users of the Freestyle Libre Flash Glucose Monitoring system (45). The study showed that there was a significant reduction in the Gold Score for hypoglycaemic unawareness after FlashGM use (45). This finding is key to diabetes treatment because hypoglycaemic episodes are linked to severe vascular complications such as myocardial infarction and cardiac arrhythmias (14, 45–47).
We did not find a significant difference in HbA1c or time in hypo/hyperglycaemia after the introduction of FlashGM. One contributing factor could be the initial HbA1c of participants as those who already have optimal glycaemic management are less likely to further decrease their HbA1c (7, 9, 48). The mean baseline HbA1c in this meta-analysis was 64 mmol/mol (7.97%) which is suboptimal but larger reductions in HbA1c would be expected with higher HbA1c. Another consideration is patient education. Numerous studies have cited the importance of education on FlashGM and its influence on glycaemic management (34, 49, 50). If individuals are unsure about how to adjust insulin, diet and physical activity to their glucose readings, their ability to self-manage is compromised (49, 50).
Past systematic reviews on continuous glucose monitoring (CGM) have reported its efficacy in reducing HbA1c and increasing time in range (8, 48). It should be noted that these systematic reviews included data from other forms of CGM and only drew findings from two RCTs that focused specifically on FlashGM (8, 48). To our knowledge, this is the first meta-analysis to assess glycaemic outcomes of FlashGM based on data from RCTs. Other systematic reviews have assessed different modes of CGM, which precludes a focused assessment of FlashGM and its impact on glycaemic outcomes. Such reviews have concluded that real-time continuous glucose monitoring (RT-CGM) is superior to FlashGM in glycaemic management (8, 48).
Increased treatment satisfaction (24, 34, 42) and improved quality of life (18, 42) are consistently reported outcomes in study participants that use FlashGM. One cohort study measured the responses of 1913 adults on the Diabetes Treatment Satisfaction Questionnaire (DTSQ) and found that treatment satisfaction improved significantly after introduction of FlashGM (42). Four RCTs in our meta-analysis measured DTSQ scores in its participants (20–22). All four studies reported significant improvements in total treatment satisfaction at follow-up. User questionnaire results show a strong preference for the convenience, flexibility and availability of sensor glucose data (20, 21, 23, 24), thereby highlighting the capacity of FlashGM to improve quality of life for people with diabetes.
Strengths
The main strengths of this meta-analysis are the exclusive inclusion of RCTs and the assessment of multiple metrics of glycaemic management. The RCTs provide high quality evidence that help guide the fast-growing usage of FlashGM. Such findings may influence decisions about national reimbursement, insurance funding and integration into clinical practice. In this review, we extended our analysis beyond HbA1c and explored other markers of glycaemic status such as time in range and time in hypo/hyperglycaemia. Such outcomes account for day-to-day glucose fluctuations and are increasingly included in interventional studies (12, 25).
Limitations
This systematic review is limited by the small number of studies which could be included due to its focus on RCTs. The small number of studies precluded subgroup analysis of diabetes type and other variables. Given the inconsistency of different study designs, we found it imperative to prioritise quality of evidence and to only select RCTs. Included studies had an open-label design as participants could access real time glucose readings and accordingly adjust their behaviour. This is characteristic of all continuous glucose monitoring studies as access to sensor-based data is key. Furthermore, this meta-analysis had substantial heterogeneity which could influence the validity of findings. To mitigate this limitation, a random-effects model was used to analyse results.
Future Directions
The widespread uptake of FlashGM drives the need to evaluate and maximise its efficacy in glycaemic management. When there are more available RCTs, they should be included in future meta-analyses. The longest study in this systematic review is 24 weeks so it remains unclear whether such outcomes will be sustained beyond 24 weeks and for years to come and this is an important question which needs to be addressed. Studies have also started to delve into how FlashGM can be optimised for people with diabetes (49, 50). This includes providing structured educational programs to better inform users (49, 50). Recently, the Food and Drug Administration has approved the Freestyle Libre 2 in the United States (51). The new technology has the capacity to alert patients about high and low glucose levels (51). It would be worthwhile to assess the benefits of these additional features for glycaemic management.
Conclusions
The findings of this systematic review and meta-analysis demonstrate that flash glucose monitoring does not result in a difference in HbA1c. However, its usage leads to improvement in key glycaemic markers, including increased time in range and reduced frequency of hypoglycaemic episodes. To our knowledge, this is the first meta-analysis to delineate the effects of FlashGM on glycaemic management using only randomised controlled trial data. Given a relatively small number of RCTs, further research is needed to determine the impact of FlashGM in the long-term. Through this meta-analysis, we found that flash glucose monitoring has the capacity to improve glycaemic variability and overall clinical care of people with type 1 and type 2 diabetes.
Data Availability Statement
The original contributions presented in the study are included in the article/Supplementary Material. Further inquiries can be directed to the corresponding author.
Author Contributions
BL conducted the literature search, data extraction, interpreted study findings and wrote the manuscript. DK and SB conducted data analyses, helped with interpretation of study findings and reviewed the manuscript. MH conducted the literature search, data extraction, reviewed and edited the manuscript. NZ helped conduct the systematic review and reviewed the manuscript. EE conceptualised the study, helped with interpretation of study findings, reviewed and edited the manuscript. All authors approved the final version of the manuscript.
Funding
There was no funding source for this study. EIE’s institutions Austin Health and The University of Melbourne receives research funding from National Health and the Medical Research Council of Australia, Juvenile Diabetes Research Foundation, Sanofi, Novo Nordisk, Eli Lilly, Gilead, Bayer for unrelated research work.
Conflict of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s Note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Acknowledgments
Shanti Nadaraja, senior librarian at Austin Hospital Library, generously assisted with the development and implementation of the electronic search strategy. All authors would like to acknowledge MISCH (Methods and Implementation Support for Clinical and Health research Hub), Faculty of Medicine, Dentistry and Health Sciences at University of Melbourne for its support while conducting the research presented in this work.
Supplementary Material
The Supplementary Material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fcdhc.2022.849725/full#supplementary-material
References
1. Lafeuille MH, Grittner AM, Gravel J, Bailey RA, Martin S, Garber L, et al. Quality Measure Attainment in Patients With Type 2 Diabetes Mellitus. Am J Manag Care (2014) 20:s5–15.
2. Fowler MJ. Microvascular and Macrovascular Complications of Diabetes. Clin Diabetes (2008) 26:77–82. doi: 10.2337/diaclin.26.2.77
3. Landman GW, van Hateren KJ, Kleefstra N, Groenier KH, Gans RO, Bilo HJ. The Relationship Between Glycaemic Control and Mortality in Patients With Type 2 Diabetes in General Practice (ZODIAC-11). Br J Gen Pract (2010) 60:172–5. doi: 10.3399/bjgp10X483517
4. Skyler JS, Bergenstal R, Bonow RO, Buse J, Deedwania P, Gale EA, et al. Intensive Glycemic Control and the Prevention of Cardiovascular Events: Implications of the ACCORD, ADVANCE, and VA Diabetes Trials: A Position Statement of the American Diabetes Association and a Scientific Statement of the American College of Cardiology Foundation and the American Heart Association. Diabetes Care (2009) 32:187–92. doi: 10.2337/dc09-9031
5. Mancini G, Berioli MG, Santi E, Rogari F, Toni G, Tascini G, et al. Flash Glucose Monitoring: A Review of the Literature With a Special Focus on Type 1 Diabetes. Nutrients (2018) 10:992. doi: 10.3390/nu10080992
6. Hirsch IB, Sherr JL, Hood KK. Connecting the Dots: Validation of Time in Range Metrics With Microvascular Outcomes. Diabetes Care (2019) 42:45–348. doi: 10.2337/dci18-0040
7. Castellana M, Parisi C, Di Molfetta S, Di Gioia L, Natalicchio A, Perrini S, et al. Efficacy and Safety of Flash Glucose Monitoring in Patients With Type 1 and Type 2 Diabetes: A Systematic Review and Meta-Analysis. BMJ Open Diabetes Res Care (2020) 8:e001092. doi: 10.1136/bmjdrc-2019-001092
8. Park C, Le QA. The Effectiveness of Continuous Glucose Monitoring in Patients With Type 2 Diabetes: A Systematic Review of Literature and Meta-Analysis. Diabetes Technol Ther (2018) 20:613–21. doi: 10.1089/dia.2018.0177
9. Evans M, Welsh Z, Ells S, Seibold A. The Impact of Flash Glucose Monitoring on Glycaemic Control as Measured by HbA1c: A Meta-Analysis of Clinical Trials and Real-World Observational Studies. Diabetes Ther (2020) 11:83–95. doi: 10.1007/s13300-019-00720-0
10. Beck RW, Bergenstal RM, Riddlesworth TD, Kollman C, Li Z, Brown AS, et al. Validation of Time in Range as an Outcome Measure for Diabetes Clinical Trials. Diabetes Care (2019) 42:400–5. doi: 10.2337/dc18-1444
11. Lu J, Ma X, Zhou J, Zhang L, Mo Y, Ying L, et al. Association of Time in Range, as Assessed by Continuous Glucose Monitoring, With Diabetic Retinopathy in Type 2 Diabetes. Diabetes Care (2018) 41:2370–6. doi: 10.2337/dc18-1131
12. Wright LA, Hirsch IB. Metrics Beyond Hemoglobin A1C in Diabetes Management: Time in Range, Hypoglycemia, and Other Parameters. Diabetes Technol Ther (2017) 19:S16–26. doi: 10.1089/dia.2017.0029
13. Kalra S, Mukherjee JJ, Venkataraman S, Bantwal G, Shaikh S, Saboo B, et al. Hypoglycemia: The Neglected Complication. Indian J Endocrinol Metab (2013) 17:819–34. doi: 10.4103/2230-8210.117219
14. Liberati A, Altman DG, Tetzlaff J, Mulrow C, Gøtzsche PC, Ioannidis JPA, et al. The PRISMA Statement for Reporting Systematic Reviews and Meta-Analyses of Studies That Evaluate Healthcare Interventions: Explanation and Elaboration. Br Med J (2009) 339:b2700. doi: 10.1136/bmj.b2700
15. Danne T, Nimri R, Battelino T, Bergenstal RM, Close KL, DeVries JH, et al. International Consensus on Use of Continuous Glucose Monitoring. Diabetes Care (2017) 40:1631–40. doi: 10.2337/dc17-1600
16. Ida S, Kaneko R, Imataka K, Okubo K, Shirakura Y, Azuma K, et al. Effects of Flash Glucose Monitoring on Dietary Variety, Physical Activity, and Self-Care Behaviors in Patients With Diabetes. J Diabetes Res (2020) 2020:9463648. doi: 10.1155/2020/9463648
17. White ND, Knezevich E. Flash Glucose Monitoring Technology Impact on Diabetes Self-Care Behavior. Am J Lifestyle Med (2020) 14:130–2. doi: 10.1177/1559827619890955
18. Al Hayek AA, Robert AA, Al Dawish MA. Evaluation of FreeStyle Libre Flash Glucose Monitoring System on Glycemic Control, Health-Related Quality of Life, and Fear of Hypoglycemia in Patients With Type 1 Diabetes. Clin Med Insights Endocrinol Diabetes (2017) 10:1179551417746957. doi: 10.1177/1179551417746957
19. Higgins T. HbA1c for Screening and Diagnosis of Diabetes Mellitus. Endocrine (2013) 42:266–73. doi: 10.1007/s12020-012-9768-y
20. Bolinder J, Antuna R, Geelhoed-Duijvestijn P, Kroger J, Weitgasser R. Novel Glucose-Sensing Technology and Hypoglycaemia in Type 1 Diabetes: A Multicentre, non-Masked, Randomised Controlled Trial. Lancet (2016) 388:2254–63. doi: 10.1016/S0140-6736(16)31535-5
21. Haak T, Hanaire H, Ajjan R, Hermanns N, Riveline JP, Rayman G. Flash Glucose-Sensing Technology as a Replacement for Blood Glucose Monitoring for the Management of Insulin-Treated Type 2 Diabetes: A Multicenter, Open-Label Randomized Controlled Trial. Diabetes Ther (2017) 8:55–73. doi: 10.1007/s13300-016-0223-6
22. Davis T, Dwyer P, England M, Fegan G, Davis W. Efficacy of Intermittently Scanned Continuous Glucose Monitoring in the Prevention of Recurrent Severe Hypoglycemia. Diabetes Technol Ther (2019) 2019:14. doi: 10.1089/dia.2019.0331
23. Wada E, Onoue T, Kobayashi T, Handa T, Hayase A, Ito M, et al. Flash Glucose Monitoring Helps Achieve Better Glycemic Control Than Conventional Self-Monitoring of Blood Glucose in non-Insulin-Treated Type 2 Diabetes: A Randomized Controlled Trial. BMJ Open Diabetes Res Care (2020) 8:e001115. doi: 10.1136/bmjdrc-2019-001115
24. Yaron M, Roitman E, Aharon-Hananel G, Landau Z, Ganz T, Yanuv I, et al. Effect of Flash Glucose Monitoring Technology on Glycemic Control and Treatment Satisfaction in Patients With Type 2 Diabetes. Diabetes Care (2019) 42:1178–84. doi: 10.2337/dc18-0166
25. Gabbay MAL, Rodacki M, Calliari LE, Vianna AGD, Krakauer M, Pinto MS, et al. Time in Range: A New Parameter to Evaluate Blood Glucose Control in Patients With Diabetes. Diabetol Metab Syndr (2020) 12:22. doi: 10.1186/s13098-020-00529-z
26. Battelino T, Danne T, Bergenstal RM, Amiel SA, Beck R, Biester T, et al. Clinical Targets for Continuous Glucose Monitoring Data Interpretation: Recommendations From the International Consensus on Time in Range. Diabetes Care (2019) 42:1593–603. doi: 10.2337/dci19-0028
27. Johnson ML, Martens TW, Criego AB, Carlson AL, Simonson GD, Bergenstal RM. Utilizing the Ambulatory Glucose Profile to Standardize and Implement Continuous Glucose Monitoring in Clinical Practice. Diabetes Technol Ther (2019) 21:S217–225. doi: 10.1089/dia.2019.0034
28. Nalysnyk L, Hernandez-Medina M, Krishnarajah G. Glycaemic Variability and Complications in Patients With Diabetes Mellitus: Evidence From a Systematic Review of the Literature. Diabetes Obes Metab (2010) 12:288–98. doi: 10.1111/j.1463-1326.2009.01160.x
29. Hirsch IB. Glycemic Variability and Diabetes Complications: Does It Matter? Of Course It Does! Diabetes Care (2015) 38:1610–4. doi: 10.2337/dc14-2898
30. Gomez-Peralta F, Dunn T, Landuyt K, Xu Y, Merino-Torres JF. Flash Glucose Monitoring Reduces Glycemic Variability and Hypoglycemia: Real-World Data From Spain. BMJ Open Diabetes Res Care (2020) 8:e001052. doi: 10.1136/bmjdrc-2019-001052
31. Jangam S, Dunn T, Xu Y, Hayter G, Ajjan RA. Flash Glucose Monitoring Improves Glycemia in Higher Risk Patients: A Longitudinal, Observational Study Under Real-Life Settings. BMJ Open Diabetes Res Care (2019) 7:e00061. doi: 10.1136/bmjdrc-2018-000611
32. Calliari LEP, Krakauer M, Vianna AGD, Ram Y, Barbieri DE, Xu Y, et al. Real-World Flash Glucose Monitoring in Brazil: Can Sensors Make a Difference in Diabetes Management in Developing Countries? Diabetol Metab Syndr (2020) 12:1. doi: 10.1186/s13098-019-0513-z
33. Laurenzi A, Caretto A, Barrasso M, Andrea Mario B, Molinari C, Zanardini A, et al. Frequency of Flash Glucose Monitoring Scans and Hemoglobin A1c in Real Life. Diabetes (2018) 67(Supplement 1):2. doi: 10.2337/db18-898-P
34. Rouhard S, Preumont V. Evolution of Glycemic Control and Quality of Life of Diabetic Patients After Using Flash Glucose Self-Monitoring: A Prospective Study Involving 248 Patients. Louv Med (2019) 138:143–7.
35. Ioannidis JP, Haidich AB, Pappa M, Pantazis N, Kokori SI, Tektonidou MG, et al. Comparison of Evidence of Treatment Effects in Randomized and Nonrandomized Studies. J Am Med Assoc (2001) 286:821–30. doi: 10.1001/jama.286.7.821
36. Chehregosha H, Khamseh ME, Malek M, Hosseinpanah F, Ismail-Beigi F. A View Beyond HbA1c: Role of Continuous Glucose Monitoring. Diabetes Ther (2019) 10:853–63. doi: 10.1007/s13300-019-0619-1
37. Brown SA, Basu A, Kovatchev BP. Beyond HbA1c: Using Continuous Glucose Monitoring Metrics to Enhance Interpretation of Treatment Effect and Improve Clinical Decision-Making. Diabetic Med (2019) 36:679–87. doi: 10.1111/dme.13944
38. Rama Chandran S, Tay WL, Lye WK, Lim LL, Ratnasingam J, Tan ATB, et al. Beyond HbA1c: Comparing Glycemic Variability and Glycemic Indices in Predicting Hypoglycemia in Type 1 and Type 2 Diabetes. Diabetes Technol Ther (2018) 20:353–62. doi: 10.1089/dia.2017.0388
39. Dunn TC, Xu Y, Hayter G, Ajjan RA. Real-World Flash Glucose Monitoring Patterns and Associations Between Self-Monitoring Frequency and Glycaemic Measures: A European Analysis of Over 60 Million Glucose Tests. Diabetes Res Clin Pract (2018) 137:37–46. doi: 10.1016/j.diabres.2017.12.015
40. Nana M, Moore SL, Ang E, Lee ZX, Bondugulapati LNR. Flash Glucose Monitoring: Impact on Markers of Glycaemic Control and Patient-Reported Outcomes in Individuals With Type 1 Diabetes Mellitus in the Real-World Setting. Diabetes Res Clin Pract (2019) 157:107893. doi: 10.1016/j.diabres.2019.107893
41. Haak T, Hanaire H, Ajjan R, Hermanns N, Riveline JP, Rayman G. Use of Flash Glucose-Sensing Technology for 12 Months as a Replacement for Blood Glucose Monitoring in Insulin-Treated Type 2 Diabetes. Diabetes Ther (2017) 8:573–86. doi: 10.1007/s13300-017-0255-6
42. Charleer S, De Block C, Van Huffel L, Broos B, Fieuws S, Nobels F, et al. Quality of Life and Glucose Control After 1 Year of Nationwide Reimbursement of Intermittently Scanned Continuous Glucose Monitoring in Adults Living With Type 1 Diabetes (FUTURE): A Prospective Observational Real-World Cohort Study. Diabetes Care (2020) 43:389–97. doi: 10.2337/dc19-1610
43. Reddy M, Jugnee N, El Laboudi A, Spanudakis E, Anantharaja S, Oliver N. A Randomized Controlled Pilot Study of Continuous Glucose Monitoring and Flash Glucose Monitoring in People With Type 1 Diabetes and Impaired Awareness of Hypoglycaemia. Diabetic Med (2018) 35:483–90. doi: 10.1111/dme.13561
44. Al Hayek AA, Robert AA, Al Dawish MA. Differences of FreeStyle Libre Flash Glucose Monitoring System and Finger Pricks on Clinical Characteristics and Glucose Monitoring Satisfactions in Type 1 Diabetes Using Insulin Pump. Clin Med Insights (2019) 12:1179551419861102. doi: 10.1177/1179551419861102
45. Deshmukh H, Wilmot EG, Gregory R, Barnes D, Narendran P, Saunders S, et al. Effect of Flash Glucose Monitoring on Glycemic Control, Hypoglycemia, Diabetes-Related Distress, and Resource Utilization in the Association of British Clinical Diabetologists (ABCD) Nationwide Audit. Diabetes Care (2020) 43:2153–60. doi: 10.2337/dc20-0738
46. Desouza CV, Bolli GB, Fonseca V. Hypoglycemia, Diabetes, and Cardiovascular Events. Diabetes Care (2010) 33:1389–94. doi: 10.2337/dc09-2082
47. Clark AL, Best CJ, Fisher SJ. Even Silent Hypoglycemia Induces Cardiac Arrhythmias. Diabetes (2014) 63:1457. doi: 10.2337/db14-0108
48. Maiorino MI, Signoriello S, Maio A, Chiodini P, Bellastella G, Scappaticcio L, et al. Effects of Continuous Glucose Monitoring on Metrics of Glycemic Control in Diabetes: A Systematic Review With Meta-Analysis of Randomized Controlled Trials. Diabetes Care (2020) 43:1146–56. doi: 10.2337/dc19-1459
49. Leelarathna L, Wilmot EG. Flash Forward: A Review of Flash Glucose Monitoring. Diabetic Med (2018) 35:472–82. doi: 10.1111/dme.13584
50. Bruttomesso D, Laviola L, Avogaro A, Bonora E, Del Prato S, Frontoni S, et al. The Use of Real Time Continuous Glucose Monitoring or Flash Glucose Monitoring in the Management of Diabetes: A Consensus View of Italian Diabetes Experts Using the Delphi Method. Nutr Metab Cardiovasc Dis (2019) 29:421–31. doi: 10.1016/j.numecd.2019.01.018
51. DiaTribe. Freestyle Libre 2 Cleared by FDA as iCGM 2020. Available at: https://diatribe.org/freestyle-libre-2-cleared-fda-icgm (Accessed February 2, 2021).
Keywords: flash glucose monitoring, self-monitoring blood glucose, diabetes mellitus, glycated haemoglobin A1c, hyperglycaemia, hypoglycaemia, meta-analysis, systematic review
Citation: Liang B, Koye DN, Hachem M, Zafari N, Braat S and Ekinci EI (2022) Efficacy of Flash Glucose Monitoring in Type 1 and Type 2 Diabetes: A Systematic Review and Meta-Analysis of Randomised Controlled Trials. Front. Clin. Diabetes Healthc. 3:849725. doi: 10.3389/fcdhc.2022.849725
Received: 06 January 2022; Accepted: 17 January 2022;
Published: 22 February 2022.
Edited by:
Aleksandra Jotic, University of Belgrade, SerbiaReviewed by:
Tanja Milicic, University of Belgrade, SerbiaAleksandar Djukic, University of Kragujevac, Serbia
Copyright © 2022 Liang, Koye, Hachem, Zafari, Braat and Ekinci. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Elif I. Ekinci, ZWxpZi5la2luY2lAdW5pbWVsYi5lZHUuYXU=
 Bonnie Liang
Bonnie Liang Digsu N. Koye
Digsu N. Koye Mariam Hachem
Mariam Hachem Neda Zafari1
Neda Zafari1 Sabine Braat
Sabine Braat Elif I. Ekinci
Elif I. Ekinci