- 1Department of Radiation Oncology, National Cancer Center/National Clinical Research Center for Cancer/Cancer Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Beijing, China
- 2Department of Radiation Oncology, National Cancer Center/National Clinical Research Center for Cancer/Cancer Hospital & Shenzhen Hospital, Chinese Academy of Medical Sciences and Peking Union Medical College, Shenzhen, China
by Yang Y, Wang J, Wang W, Zhang T, Zhao J, Wang Y, Li Y, Wang L and Bi N (2022) Front. Oncol. 12:810580. doi: 10.3389/fonc.2022.810580
In the published article, there was an error in the legend for Figure 1 and Figure 3 as published, regarding the light green zone for 95% prediction intervals. The corrected legends appear below.
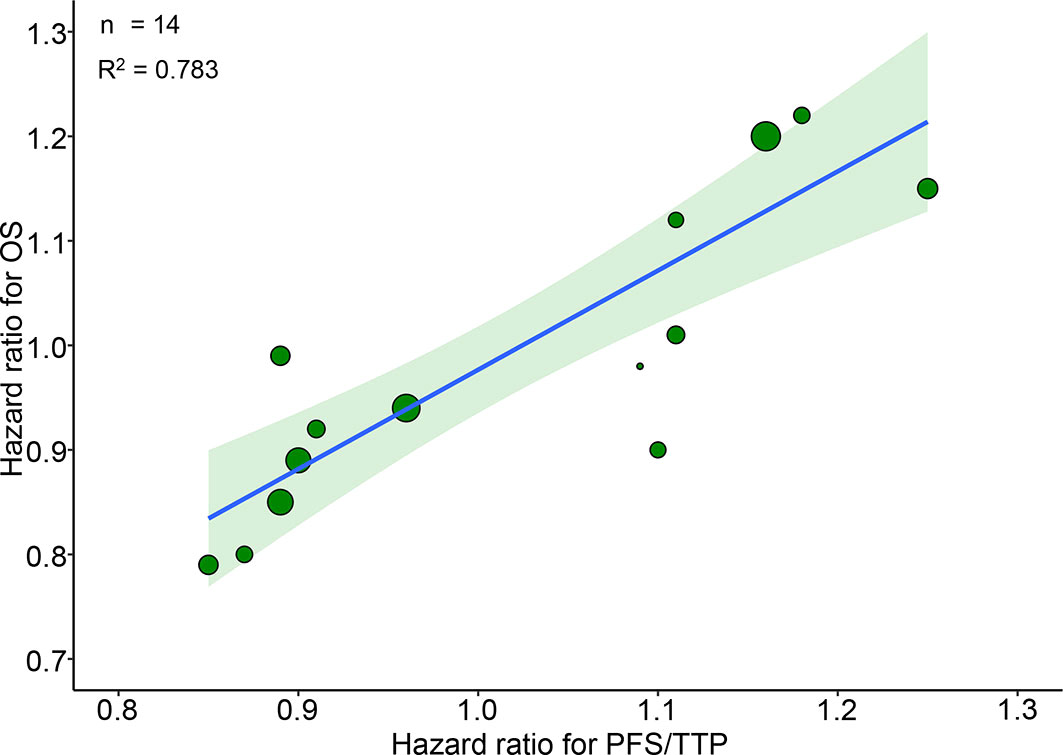
Figure 1 Trial-level correlation between hazard ratios for OS and PFS/TTP in phase III RCTs. Green circles represent trials with a size proportional to the number of patients, blue line for the estimated regression line and the light green zone for 95% confidence intervals. OS, overall survival; PFS/TTP, progression free survival/time to progression; RCTs, randomized controlled trials.
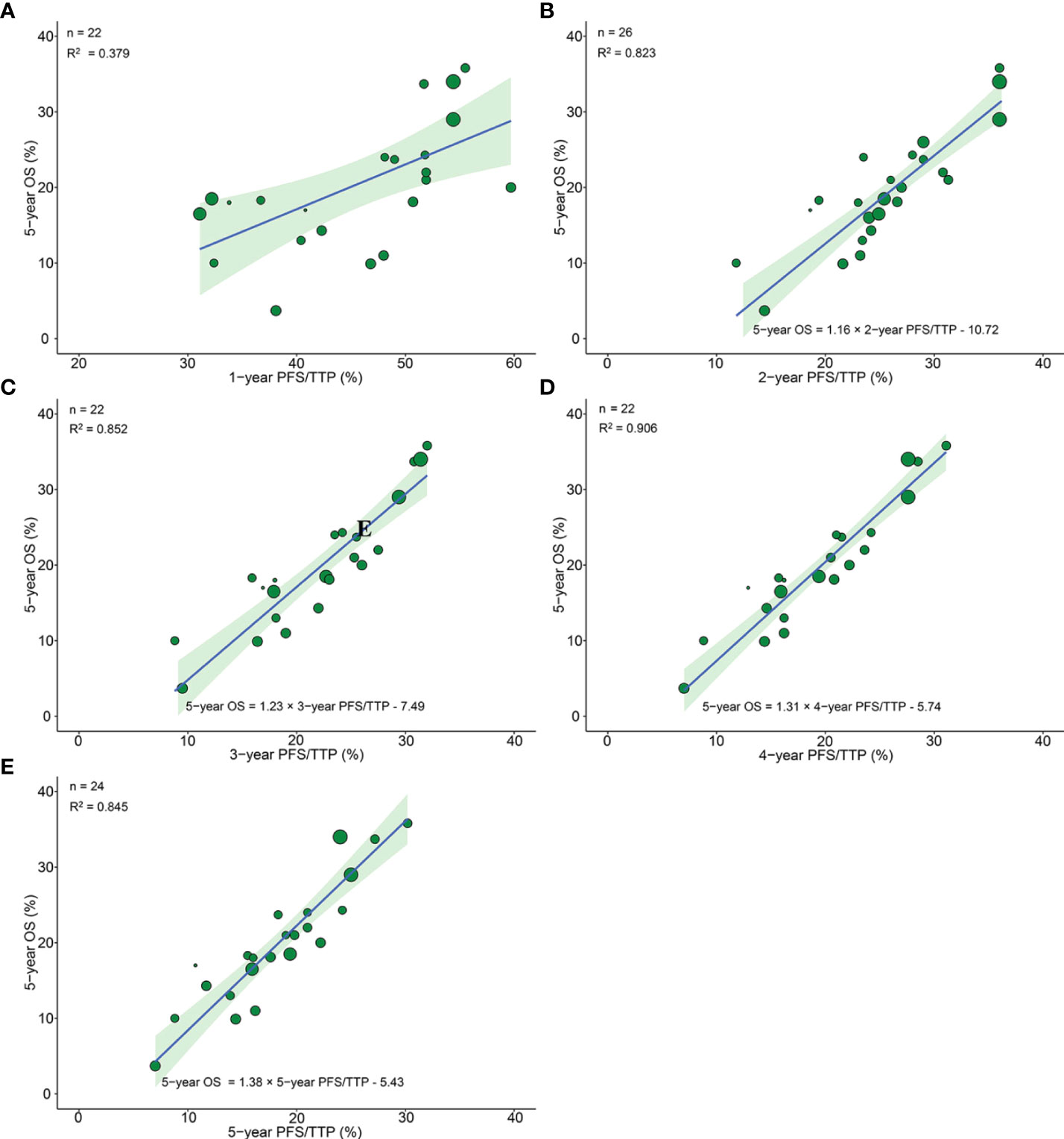
Figure 3 Treatment arm-level correlation between 5-year OS and 1-year PFS/TTP (A), 2-year PFS/TTP (B), 3-year PFS/TTP (C), 4-year PFS/TTP (D), 5-year PFS/TTP (E) in phase III RCTs. Green circles represent treatment arms with a size proportional to the number of patients, blue lines for the estimated regression lines and the light green zones for 95% confidence intervals. OS, overall survival; PFS/TTP, progression free survival/time to progression; RCTs, randomized controlled trials.
The authors apologize for this error and state that this does not change the scientific conclusions of the article in any way. The original article has been updated.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Keywords: Limited-stage small-cell lung cancer, surrogate endpoint, overall survival, progression-free survival, time to progression, chemoradiotherapy
Citation: Yang Y, Wang J, Wang W, Zhang T, Zhao J, Wang Y, Li Y, Wang L and Bi N (2022) Corrigendum: Progression-free survival and time to progression as potential surrogate endpoints for overall survival in chemoradiotherapy trials in limited-stage small-cell lung cancer: A systematic review and meta-analysis. Front. Oncol. 12:1007862. doi: 10.3389/fonc.2022.1007862
Received: 31 July 2022; Accepted: 01 August 2022;
Published: 24 August 2022.
Edited and Reviewed by:
Kristin Higgins, Winship Cancer Institute, Emory University, United StatesCopyright © 2022 Yang, Wang, Wang, Zhang, Zhao, Wang, Li, Wang and Bi. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Nan Bi, YmluYW5fZW1haWxAMTYzLmNvbQ==
†These authors have contributed equally to this work
 Yin Yang1†
Yin Yang1† Tao Zhang
Tao Zhang Jingjing Zhao
Jingjing Zhao Yu Wang
Yu Wang Yexiong Li
Yexiong Li Luhua Wang
Luhua Wang Nan Bi
Nan Bi