- 1Department of Thyroid and Breast Surgery, Southern Central Hospital of Yunnan Province, The First People’s Hospital of Honghe State, Honghe Hospital Affiliated to Kunming Medical University, Gejiu, Yunnan, China
- 2Department of Pathology, Southern Central Hospital of Yunnan Province, The First People’s Hospital of Honghe State, Honghe Hospital Affiliated to Kunming Medical University, Gejiu, Yunnan, China
- 3Department of Hepatobiliary Surgery, Southern Central Hospital of Yunnan Province, The First People’s Hospital of Honghe State, Honghe Hospital Affiliated to Kunming Medical University, Gejiu, Yunnan, China
- 4Department of Rheumatology and Immunology, Southern Central Hospital of Yunnan Province, The First People’s Hospital of Honghe state, Honghe Hospital Affiliated to Kunming Medical University, Gejiu, Yunnan, China
The metastasis of breast cancer to the colon is a rare occurrence, especially in the presence of changes in estrogen and progesterone receptors. To date, literature has only reported two cases of invasive ductal carcinoma and two cases of invasive lobular carcinoma metastasizing to the colon with concurrent changes in hormone receptors. This report describes a 65-year-old woman with a history of left breast cancer, who presented with symptoms of bloody stools and abdominal pain. CT and colonoscopy results revealed a malignant tumor in the ascending colon, and the patient underwent surgery. Pathological results post-surgery indicated changes in hormone receptors, differing from the previous breast cancer pathology, ultimately leading to the diagnosis of breast cancer metastasis to the colon. The patient was found to have liver metastasis 14 months after right hemicolectomy, and systemic metastases in various locations were discovered at the 19-month mark.
Introduction
Breast cancer is the most common malignant tumor in women. Recurrence and distant metastasis often pose challenges in treatment. Common sites of metastasis include bones, liver, and lungs, while gastrointestinal metastases are extremely rare. Previous reports have shown that the pattern of metastasis differs between lobular carcinoma and ductal carcinoma of the breast. Gastrointestinal, gynecologic, and peritoneal metastases are more common in lobular carcinoma (1, 2). ILC (invasive lobular carcinoma) has an increased tendency to metastasize to the GI (gastrointestinal) tract compared with breast carcinomas of NST (non-special type) (40% vs. 2%) (3). We report a case of infiltrating ductal carcinoma that metastasized to the ascending colon, with changes in estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor-2 (HER2) status. To our knowledge, there have been only two similar cases reported previously.
Case presentation
A 65-year-old woman presented to our center with intermittent abdominal pain and hematochezia for several months. She had a previous diagnosis of ER- and PR-positive (Figure 1), HER2 ++ (no further testing was done) invasive ductal carcinoma of the left breast 5 years before. She underwent modified radical mastectomy for breast cancer. The pathological examination did not indicate the presence of axillary lymph node metastasis and received chemotherapy with anthracycline and cyclophosphamide, followed by taxanes. Subsequently, she received 5 years of endocrine therapy with letrozole until she presented to our center. The patient denied having smoked or consumed alcohol in the past.
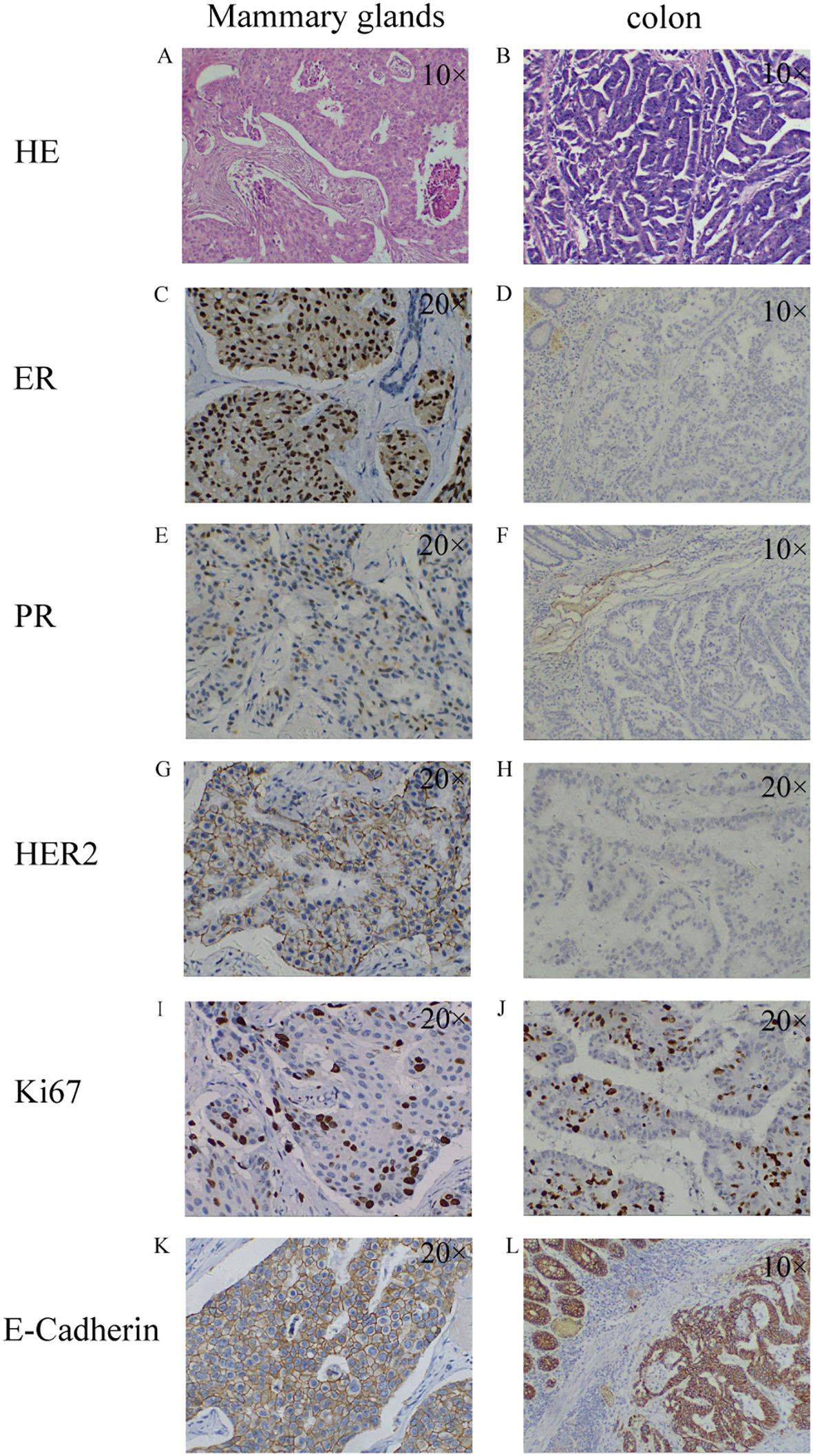
Figure 1. Pathological findings of breast malignancy and colon malignancy. breast malignancy: HE staining (A); ER+ (C); PR+ (E); HER2+ (G); Ki67 15%+ (I); E-Cadherin+ (K); colon malignancy: HE staining (B); ER- (D); PR- (F); HER2- (H); Ki67 50%+ (J); E-Cadherin+ (L). HE staining: Hematoxylin-Eosin staining; ER: estrogen receptor; PR: progesterone receptor; HER2: human epidermal growth factor receptor-2; Ki67: nuclear-associated antigen Ki67.
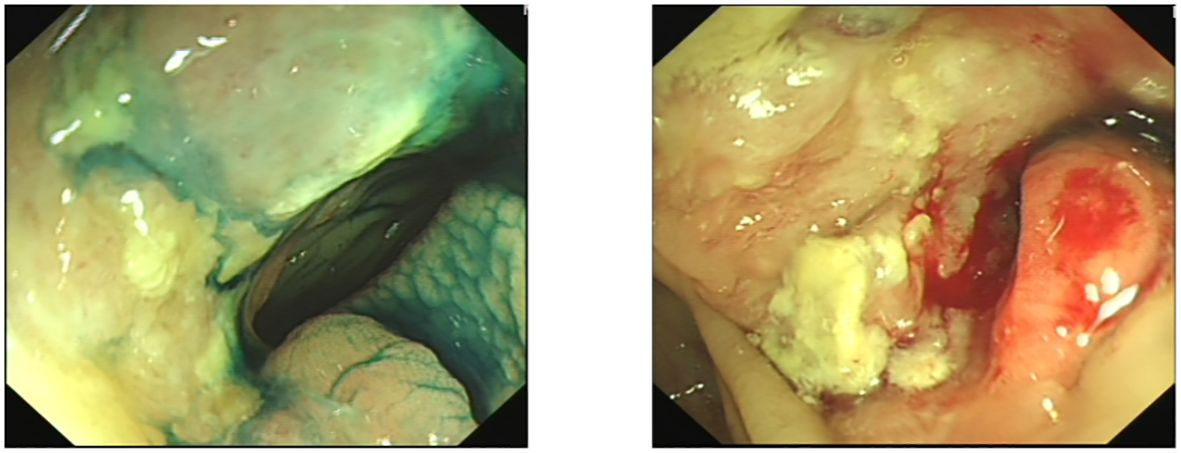
During colonoscopy, a mass was found in the ascending colon (Figure 2). Biopsy results confirmed metastasis of breast cancer to the colon, which was negative for ER, PR, and HER2. However, no local lesions or enlarged lymph nodes were detected in the contralateral breast, chest wall, or axilla through ultrasound, magnetic resonance imaging, and clinical palpation.
After receiving a transfusion of six units of red blood cells to correct anemia, the patient underwent a right hemicolectomy. Intraoperatively, it was observed that the tumor had invaded the right renal fascia. Two lymph node metastases were found on pathological examination after palliative surgery for colonic malignancy. The postoperative pathological examination confirmed the colonoscopy biopsy findings. Further immunohistochemical staining revealed positive results for Cytokeratin 7 (CK7), negative results for Cytokeratin 20 (CK20), and positive results for GATA 3 binding protein (GATA3) and E-Cadherin (Figure 3).
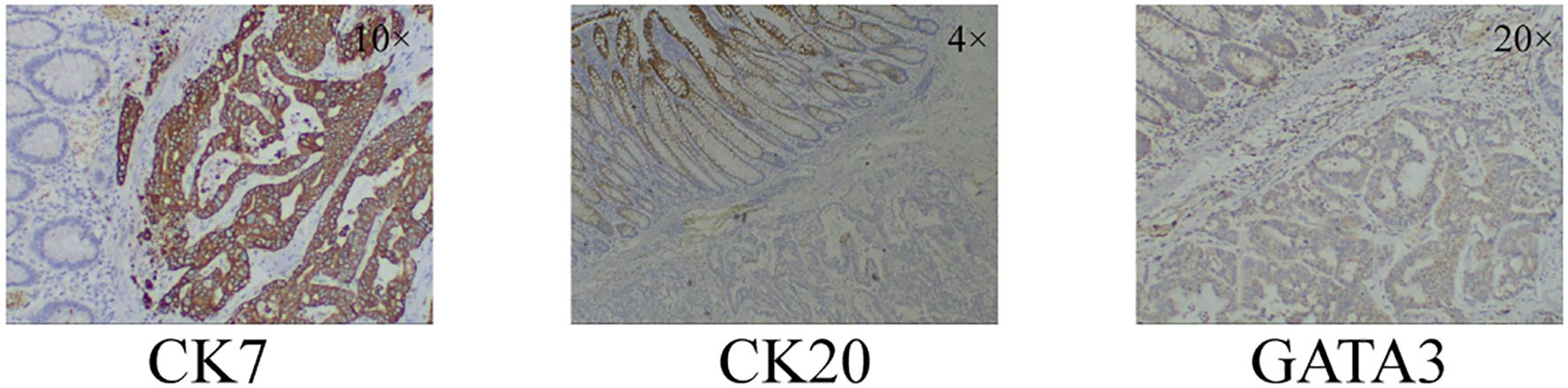
Figure 3. Immunohistochemical staining results of colonic malignancies. CK7+, CK20-, GATA3+. CK7: Cytokeratin 7; CK20: Cytokeratin 20; GATA3: GATA 3 binding protein.
Considering the rarity of this case, we sought opinions from pathologists in different centers to confirm the diagnosis. After considering the majority of expert opinions, it was confirmed as breast cancer metastasis to the colon. The patient was prescribed oral capecitabine at a dosage of 650-mg/m2 twice daily for chemotherapy treatment.
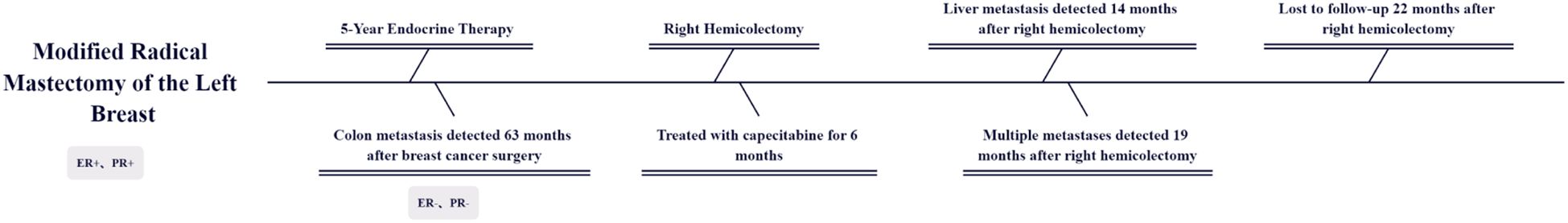
Follow-up imaging studies performed 6 months after the operation did not show any tumor metastasis or recurrence. However, the patient discontinued medication due to severe hand–foot syndrome. In a 14-month postoperative computed tomography (CT) scan, a solitary nodule was detected in the liver, indicating a metastatic lesion (Figure 4). The patient refused intravenous chemotherapy and opted for a second course of oral capecitabine treatment. Nineteen months after surgery, multiple metastases were discovered, including the liver, abdominal lymph nodes, adrenal glands, thoracolumbar spine, lungs, and mediastinal lymph nodes, accompanied by widespread cancer-related pain. The patient declined medication treatment. During the entire follow-up, no local recurrence of breast malignancy or new tumors of the opposite breast were found. The patient was lost to follow-up at 20 months after right hemicolectomy. Treatment and follow-up are organized into a timeline (Figure 5).
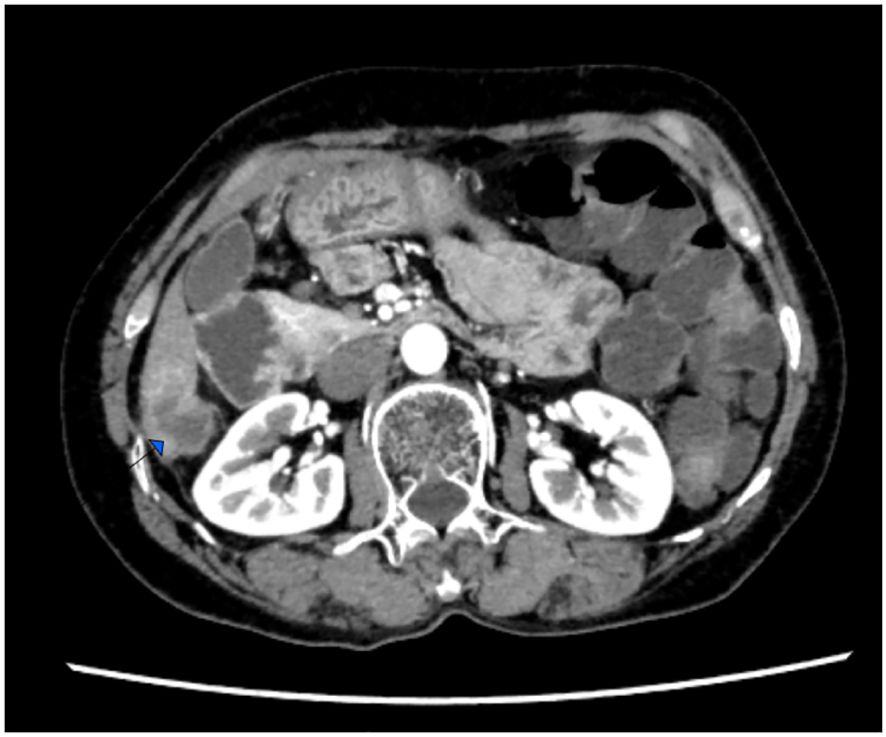
Figure 4. Computedtomography (CT) scan, a solitary nodule was detected in the liver, indicating a metastatic lesion.
Discussion
Breast cancer metastases to the gastrointestinal tract is extremely rare. Previous reports have shown that the most common sites of metastases in breast cancer are the bones, lungs, liver, and brain, while gastrointestinal metastases, especially to the colon, is very rare. The incidence of breast cancer metastases to the gastrointestinal tract, particularly to the colon, is estimated to be around 1%, with a rate of approximately 0.1% specifically for metastases to the colon (4). The patterns of metastases differ between lobular carcinoma and ductal carcinoma of the breast, with gastrointestinal metastases being more common in lobular carcinoma (1, 2). In this case, it was the ductal carcinoma of the breast that had colon metastases.
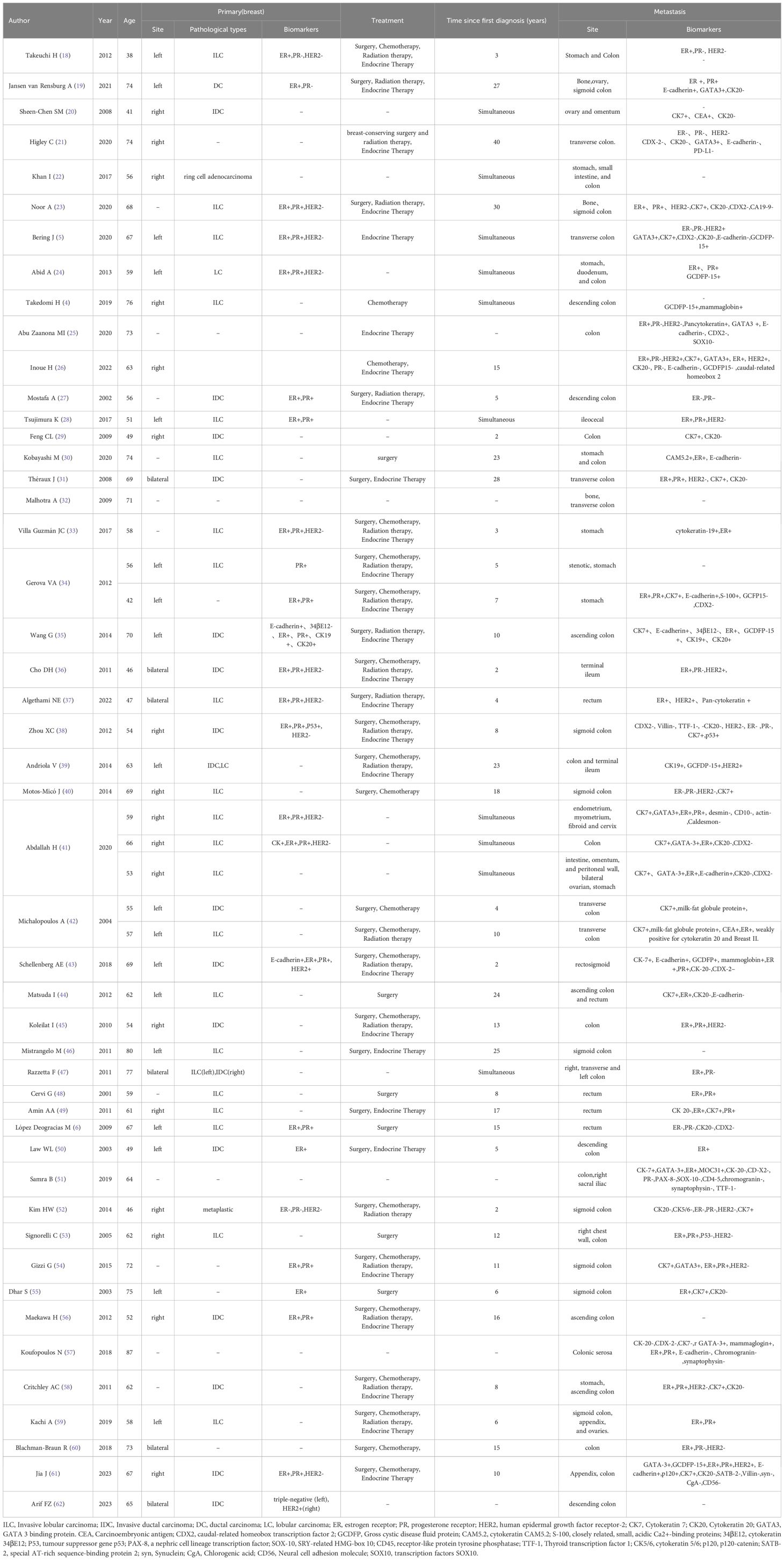
Due to the final diagnosis of breast cancer metastasis to the colon with receptor changes, this case presents significant challenges in terms of diagnosis and treatment. We performed a search in the PubMed database and gathered relevant literature, as detailed in Table 1. Our findings reveal the fourth documented case of ductal carcinoma of breast metastasizing to the colon, accompanied by changes in receptor status. Additionally, there have been two cases where receptor changes were observed when lobular carcinoma metastasized to the colon (5, 6).
In this case, there was a mismatch between the receptors in the primary breast lesion and the colon metastases. Incompatible receptors between the primary and secondary lesions of breast cancer have been reported, with frequencies of alterations in ER, PR, and HER2 being approximately 16.4%–32.4%, 30.9%–37.78%, and 10.2%–14.5%, respectively (7–10). Emilia Montagna reported in 2017 that approximately 82% of breast cancer metastases in the gastrointestinal tract have positive hormone receptor expression (11). In the dozens of case reports we have collected, the majority of patients were found to be hormone receptor positive. Among these patients, most did not exhibit any changes in the hormone receptor status. Previous reports have indicated that the instability of hormone receptors in breast cancer is associated with a poorer prognosis (12, 13), Women whose ER-positive primary tumors transform into ER-negative tumors experience a significant 48% increase in the risk of death (7). In this case report, liver metastases were found at 14 months after surgery, and multiple metastases throughout the body were found at 19 months with subsequent rapid disease progression.
CK7, CK20, and GATA3 are commonly used in pathology for tumor diagnosis and classification. These markers can provide information about the type and origin of the tumor. CK7 was expressed in 89%–98% of non-specified breast cancers (14). Most gastrointestinal, pancreaticobiliary, and ovarian mucinous adenocarcinomas are CK20 positivity, in adenocarcinoma, positivity of CK20 strongly indicates a non-breast origin, and CK7−/CK20+ immunoprofile strongly suggests colorectal origin (15). Based on previous research reports, CK7+/CK20 should be considered indicative of a tumor originating from the breast. GATA3 is also thought to be often positive in primary breast disease (16, 17). It was based on the immunohistochemical staining results of CK7 positive, CK20 negative, and GATA3 positive, combined with the opinions of multicenter pathologists, showing that the case was finally diagnosed as breast cancer with colon metastasis. Even though this case is very rare, we should still pay attention to the occurrence of colon metastasis in the diagnosis and treatment of breast cancer.
Conclusions
This case serves as a reminder to clinicians that they should consider rare sites of metastases and different receptor expression patterns in breast cancer patients in order to make more accurate treatment decisions. However, further research is needed to explore the mechanisms of this metastatic pattern and related treatment strategies.
Data availability statement
The datasets presented in this article are not readily available due to participant privacy. Requests to access the datasets should be directed to the corresponding authors.
Ethics statement
The studies involving humans were approved by Ethics Review Committee of Southern Central Hospital of Yunnan Province. The studies were conducted in accordance with the local legislation and institutional requirements. The participants provided their written informed consent to participate in this study. Written informed consent was obtained from the individual(s) for the publication of any potentially identifiable images or data included in this article.
Author contributions
HL: Investigation, Resources, Writing – original draft, Writing – review & editing. LY: Investigation, Writing – original draft. XS: Investigation, Writing – original draft. GL: Investigation, Writing – original draft. WG: Resources, Writing – original draft. FM: Writing – review & editing. ZW: Writing – review & editing. SQ: Writing – review & editing. CL: Writing – review & editing.
Funding
The author(s) declare financial support was received for the research, authorship, and/or publication of this article. The present study was supported by the Scientific Research Project of Southern Central Hospital of Yunnan Province (grant no.KY202401).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Borst MJ, Ingold JA. Metastatic patterns of invasive lobular versus invasive ductal carcinoma of the breast. Surgery. (1993) 114:637–41; discussion 641-2.
2. Kennecke H, Yerushalmi R, Woods R, Cheang MC, Voduc D, Speers CH, et al. Metastatic behavior of breast cancer subtypes. J Clin Oncol. (2010) 28:3271–7. doi: 10.1200/JCO.2009.25.9820
3. Kioleoglou Z, Georgaki E, Koufopoulos N, Kostek O, Volakakis N, Dimitriadou A, et al. Gastrointestinal metastases from lobular breast carcinoma: A literature review. Cureus. (2024) 16:e65852. doi: 10.7759/cureus.65852
4. Takedomi H, Akutagawa T, Sakata Y. Colon metastasis of breast cancer with a unique endoscopic finding. Clin Gastroenterol Hepatol. (2020) 18:e74. doi: 10.1016/j.cgh.2019.04.014
5. Bering J, Ryan M, Gurudu SR. Breast cancer metastasis presenting as colonic polyps. ACG Case Rep J. (2020) 7:e00411. doi: 10.14309/crj.0000000000000411
6. López Deogracias M, Flores Jaime L, Arias-Camisón I, Zamacola I, Murillo Guibert J, Suescun García R, et al. Rectal metastasis from lobular breast carcinoma 15 years after primary diagnosis. Clin Transl Oncol. (2010) 12:150–3. doi: 10.1007/S12094-010-0481-0
7. Lindström LS, Karlsson E, Wilking UM, Johansson U, Hartman J, Lidbrink EK, et al. Clinically used breast cancer markers such as estrogen receptor, progesterone receptor, and human epidermal growth factor receptor 2 are unstable throughout tumor progression. J Clin Oncol. (2012) 30:2601–8. doi: 10.1200/JCO.2011.37.2482
8. Matsumoto A, Jinno H, Murata T, Seki T, Takahashi M, Hayashida T, et al. Prognostic implications of receptor discordance between primary and recurrent breast cancer. Int J Clin Oncol. (2015) 20:701–8. doi: 10.1007/s10147-014-0759-2
9. Yang Z, Li N, Li X, Lei L, Wang X. The prognostic impact of hormonal receptor and HER-2 expression discordance in metastatic breast cancer patients. Onco Targets Ther. (2020) 13:853–63. doi: 10.2147/OTT.S231493
10. Zhao W, Sun L, Dong G, Wang X, Jia Y, Tong Z. Receptor conversion impacts outcomes of different molecular subtypes of primary breast cancer. Ther Adv Med Oncol. (2021) 13:17588359211012982. doi: 10.1177/17588359211012982
11. Montagna E, Pirola S, Maisonneuve P, De Roberto G, Cancello G, Palazzo A, et al. Lobular metastatic breast cancer patients with gastrointestinal involvement: features and outcomes. Clin Breast Cancer. (2018) 18:e401–401e405. doi: 10.1016/j.clbc.2017.07.003
12. Yang YF, Liao YY, Yang M, Peng NF, Xie SR, Xie YF. Discordances in ER, PR and HER2 receptors between primary and recurrent/metastatic lesions and their impact on survival in breast cancer patients. Med Oncol. (2014) 31:214. doi: 10.1007/s12032-014-0214-2
13. Yamanouchi K, Kuba S, Eguchi S. Hormone receptor, human epidermal growth factor receptor-2, and Ki-67 status in primary breast cancer and corresponding recurrences or synchronous axillary lymph node metastases. Surg Today. (2020) 50:657–63. doi: 10.1007/s00595-019-01831-8
14. Lu S, Yakirevich E, Wang LJ, Resnick MB, Wang Y. Cytokeratin 7-negative and GATA binding protein 3-negative breast cancers: Clinicopathological features and prognostic significance. BMC Cancer. (2019) 19:1085. doi: 10.1186/s12885-019-6295-8
15. Lerwill MF. Current practical applications of diagnostic immunohistochemistry in breast pathology. Am J Surg Pathol. (2004) 28:1076–91. doi: 10.1097/01.pas.0000126780.10029.f0
16. Gown AM, Fulton RS, Kandalaft PL. Markers of metastatic carcinoma of breast origin. Histopathology. (2016) 68:86–95. doi: 10.1111/his.12877
17. Davies T, Chouari T, Ray C, Elgammal S. Appendiceal adenocarcinoma with breast metastases. BMJ Case Rep. (2021) 14. doi: 10.1136/bcr-2020-240808
18. Takeuchi H, Hiroshige S, Yoshikawa Y, Kusumoto T, Muto Y. A case of synchronous metastasis of breast cancer to stomach and colon. Anticancer Res. (2012) 32:4051–5.
19. Jansen van Rensburg A, Riddell A. A case report of ductal carcinoma of the breast metastasizing to the bowel. J Surg Case Rep. (2021) 2021:rjab471. doi: 10.1093/jscr/rjab471
20. Sheen-Chen SM, Liu YW, Sun CK, Lin SE, Eng HL, Huang WT, et al. Abdominal carcinomatosis attributed to metastatic breast carcinoma. Dig Dis Sci. (2008) 53:3043–5. doi: 10.1007/s10620-008-0529-y
21. Higley C, Hsu A, Park BU, Pang M. Back to basics: history and physical examination uncover colonic metastasis in a patient with remote history of breast cancer. ACG Case Rep J. (2020) 7:e00494. doi: 10.14309/crj.0000000000000494
22. Khan I, Malik R, Khan A, Assad S, Zahid M, Sohail MS, et al. Breast cancer metastases to the gastrointestinal tract presenting with anemia and intra-abdominal bleed. Cureus. (2017) 9:e1429. doi: 10.7759/cureus.1429
23. Noor A, Lopetegui-Lia N, Desai A, Mesologites T, Rathmann J. Breast cancer metastasis masquerading as primary colon and gastric cancer: A case report. Am J Case Rep. (2020) 21:e917376. doi: 10.12659/AJCR.917376
24. Abid A, Moffa C, Monga DK. Breast cancer metastasis to the GI tract may mimic primary gastric cancer. J Clin Oncol. (2013) 31:e106–7. doi: 10.1200/JCO.2012.44.6393
25. Abu Zaanona MI, Gulati A, Rowland K. Colon metastases as first clinical manifestation of lobular breast carcinoma with no subsequent evidence of breast disease. BMJ Case Rep. (2020) 13. doi: 10.1136/bcr-2020-236225
26. Inoue H, Arita T, Kuriu Y, Shimizu H, Kiuchi J, Yamamoto Y, et al. Colonic metastasis from breast cancer: A case report and review of the literature. In Vivo. (2022) 36:522–7. doi: 10.21873/invivo.12733
27. Mostafa A, Carpenter R. Colonic metastasis from a breast cancer a case report and a few questions. Eur J Surg Oncol. (2002) 28:462. doi: 10.1053/ejso.2002.1174
28. Tsujimura K, Teruya T, Kiyuna M, Higa K, Higa J, Iha K, et al. Colonic metastasis from breast carcinoma: a case report. World J Surg Oncol. (2017) 15:124. doi: 10.1186/s12957-017-1193-5
29. Feng CL, Chou JW, Huang SF. Colonic metastasis from carcinoma of the breast presenting with colonic erosion. Endoscopy. (2009) 41 Suppl 2:E276–7. doi: 10.1055/s-0029-1215066
30. Kobayashi M, Tashima T, Nagata K, Sakuramoto S, Osaki A, Ryozawa S. Colorectal and gastric metastases from lobular breast cancer that resembled superficial neoplastic lesions. Clin J Gastroenterol. (2021) 14:103–8. doi: 10.1007/s12328-020-01285-3
31. Théraux J, Bretagnol F, Guedj N, Cazals-Hatem D, Panis Y. Colorectal breast carcinoma metastasis diagnosed as an obstructive colonic primary tumor. A case report and review of the literature. Gastroenterol Clin Biol. (2009) 33:1114–7. doi: 10.1016/j.gcb.2009.05.015
32. Malhotra A, Guturu P, Basim MS, Raju GS. A rare case of breast cancer metastasis presenting as linitis plastica of the stomach and colon (with videos). Gastrointest Endosc. (2009) 70:552–3; discussion 553. doi: 10.1016/j.gie.2009.04.029
33. Villa Guzmán JC, Espinosa J, Cervera R, Delgado M, Patón R, Cordero García JM. Gastric and colon metastasis from breast cancer: case report, review of the literature, and possible underlying mechanisms. Breast Cancer (Dove Med Press). (2017) 9:1–7. doi: 10.2147/BCTT.S79506
34. Gerova VA, Tankova LT, Mihova AA, Drandarska IL, Kadian HO. Gastrointestinal metastases from breast cancer: report of two cases. Hepatogastroenterology. (2012) 59:178–81. doi: 10.5754/hge10681
35. Wang G, Wang T, Jiang J, Zhou L, Zhao H. Gastrointestinal tract metastasis from tubulolobular carcinoma of the breast: a case report and review of the literature. Onco Targets Ther. (2014) 7:435–40. doi: 10.2147/OTT.S57831
36. Cho DH, Jeon YS, Choi MY, Lee SK, Kim SM, Hur SM, et al. Ileal metastasis of breast cancer in a patient with a BRCA2 gene mutation: report of a case. Surg Today. (2011) 41:1665–9. doi: 10.1007/s00595-011-4503-5
37. Algethami NE, Althagafi AA, Aloufi RA, Al Thobaiti FA, Abdelaziz HA. Invasive lobular carcinoma of the breast with rectal metastasis: A rare case report. Cureus. (2022) 14:e23666. doi: 10.7759/cureus.23666
38. Zhou XC, Zhou H, Ye YH, Zhang XF, Jiang Y. Invasive ductal breast cancer metastatic to the sigmoid colon. World J Surg Oncol. (2012) 10:256. doi: 10.1186/1477-7819-10-256
39. Andriola V, Piscitelli D, De Fazio M, Altomare DF. Massive colonic metastasis from breast cancer 23 years after mastectomy. Int J Colorectal Dis. (2015) 30:427–8. doi: 10.1007/s00384-014-1992-x
40. Motos-Micó J, Ferrer-Márquez M, Belda-Lozano R, Reina-Duarte Á, Rosado-Cobián R. Metastasis of lobular breast carcinoma in the sigmoid colon. Rev Esp Enferm Dig. (2014) 106:366–7.
41. Abdallah H, Elwy A, Alsayed A, Rabea A, Magdy N. Metastatic breast lobular carcinoma to unusual sites: A report of three cases and review of literature. J Med Cases. (2020) 11:292–5. doi: 10.14740/jmc3538
42. Michalopoulos A, Papadopoulos V, Zatagias A, Fahantidis E, Apostolidis S, Haralabopoulos E, et al. Metastatic breast adenocarcinoma masquerading as colonic primary. Report of two cases. Tech Coloproctol. (2004) 8 Suppl 1:s135–7. doi: 10.1007/s10151-004-0135-8
43. Schellenberg AE, Wood ML, Baniak N, Hayes P. Metastatic ductal carcinoma of the breast to colonic mucosa. BMJ Case Rep. (2018) 2018. doi: 10.1136/bcr-2018-224216
44. Matsuda I, Matsubara N, Aoyama N, Hamanaka M, Yamagishi D, Kuno T, et al. Metastatic lobular carcinoma of the breast masquerading as a primary rectal cancer. World J Surg Oncol. (2012) 10:231. doi: 10.1186/1477-7819-10-231
45. Koleilat I, Syal A, Hena M. Metastatic male ductal breast cancer mimicking obstructing primary colon cancer. Int J BioMed Sci. (2010) 6:66–70. doi: 10.59566/IJBS.2010.6066
46. Mistrangelo M, Cassoni P, Mistrangelo M, Castellano I, Codognotto E, Sapino A, et al. Obstructive colon metastases from lobular breast cancer: report of a case and review of the literature. Tumori. (2011) 97:800–4. doi: 10.1177/030089161109700619
47. Razzetta F, Tassara E, Saro F, Sironi M, D’Ambrosio G. Rare abdominal metastases from occult lobular breast cancer: report of two cases. Updates Surg. (2011) 63:129–33. doi: 10.1007/s13304-011-0047-x
48. Cervi G, Vettoretto N, Vinco A, Cervi E, Villanacci V, Grigolato P, et al. Rectal localization of metastatic lobular breast cancer: report of a case. Dis Colon Rectum. (2001) 44:453–5. doi: 10.1007/BF02234749
49. Amin AA, Reddy A, Jha M, Prasad K. Rectal metastasis from breast cancer: an interval of 17 years. BMJ Case Rep. (2011) 2011. doi: 10.1136/bcr.01.2011.3683
50. Law WL, Chu KW. Scirrhous colonic metastasis from ductal carcinoma of the breast: report of a case. Dis Colon Rectum. (2003) 46:1424–7. doi: 10.1007/s10350-004-6762-3
51. Samra B, Ghanem S, Ilyas G, Taiwo E. Screening colonoscopy unmasking colonic metastasis from an occult breast ductal carcinoma: A case report and review of the literature. Case Rep Oncol Med. (2019) 2019:8432079. doi: 10.1155/2019/8432079
52. Kim HW, Moon DH. Sigmoid colon metastasis from metaplastic breast carcinoma mimicking primary sigmoid colon cancer. Rev Esp Med Nucl Imagen Mol. (2015) 34:211–2. doi: 10.1016/j.remn.2014.09.002
53. Signorelli C, Pomponi-Formiconi D, Nelli F, Pollera CF. Single colon metastasis from breast cancer: a clinical case report. Tumori. (2005) 91:424–7. doi: 10.1177/030089160509100509
54. Gizzi G, Santini D, Guido A, Fuccio L. Single colonic metastasis from breast cancer 11 years after mastectomy. BMJ Case Rep. (2015) 2015:bcr2015211193. doi: 10.1136/bcr-2015-211193
55. Dhar S, Kulaylat MN, Gordon K, Lall P, Doerr RJ. Solitary papillary breast carcinoma metastasis to the large bowel presenting as primary colon carcinoma: case report and review of the literature. Am Surg. (2003) 69:799–803. doi: 10.1177/000313480306900914
56. Maekawa H, Fujikawa T, Tanaka A. Successful laparoscopic investigation and resection of solitary colonic metastasis from breast cancer (with video). BMJ Case Rep. (2012) 2012. doi: 10.1136/bcr-2012-007187
57. Koufopoulos N, Goudeli C, Pigadioti E, Balalis D, Manatakis DK, Antoniadou F, et al. Synchronous colonic adenocarcinoma and metastatic lobular carcinoma in a colectomy specimen: A rare finding. Cureus. (2018) 10:e3207. doi: 10.7759/cureus.3207
58. Critchley AC, Harvey J, Carr M, Iwuchukwu O. Synchronous gastric and colonic metastases of invasive lobular breast carcinoma: case report and review of the literature. Ann R Coll Surg Engl. (2011) 93:e49–50. doi: 10.1308/147870811X582800
59. Kachi A, Nicolas G, Semaan DB, Hashem M, Abou Sleiman C. Unusual pattern of invasive lobular carcinoma metastasis: A case report. Am J Case Rep. (2019) 20:1659–63. doi: 10.12659/AJCR.917237
60. Blachman-Braun R, Felemovicius I, Barker K, Kehrberg E, Khan F. Widespread metastatic breast cancer to the bowel: an unexpected finding during colonoscopy. Oxf Med Case Rep. (2019) 2019:omy133. doi: 10.1093/omcr/omy133
61. Jia J, Huang Y, Li X. Ascending colon metastasis after breast cancer surgery: a case report and literature review. Gland Surg. (2023) 12:309–16. doi: 10.21037/gs-22-642
Keywords: colon metastasis, progesterone receptor, estrogen receptor, non-specific types of breast cancer, breast cancer
Citation: Li H, Yang L, Sun X, Wang Z, Qin S, Li C, Liu G, Xie F and Gao W (2025) Changes in hormone receptor when breast cancer metastasizes to the colon: case report and literature review. Front. Oncol. 14:1391393. doi: 10.3389/fonc.2024.1391393
Received: 22 April 2024; Accepted: 27 December 2024;
Published: 29 January 2025.
Edited by:
Daniel Reis Waisberg, Hospital das Clinicas da Faculdade de Medicina da USP (HC-FMUSP), BrazilReviewed by:
Nektarios I. Koufopoulos, University General Hospital Attikon, GreeceHanen Bouaziz, Salah Azaiez Institute, Tunisia
Copyright © 2025 Li, Yang, Sun, Wang, Qin, Li, Liu, Xie and Gao. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Weiwei Gao, d2Vpd2VpX2dhbzIwMjNAMTYzLmNvbQ==; Fengming Xie, ZmVuZ21pbmdfeGllQDE2My5jb20=
†These authors have contributed equally to this work
 Huimeng Li1†
Huimeng Li1† Weiwei Gao
Weiwei Gao