- 1Department of Thoracic Surgery, The First People’s Hospital of Jiande, Jiande, China
- 2Department of Thoracic Surgery, Affiliated Zhongshan Hospital of Dalian University, Dalian, China
- 3Radiotherapy Department, Second Affiliated Hospital, Zhejiang University School of Medicine, Hangzhou, China
Background: This study examined the effectiveness of preoperative inflammatory markers in predicting the occurrence of postoperative pneumonia (POP) and clinical outcomes based on chest computed tomography (CT) images in patients who underwent surgical resection for non-small cell lung cancer (NSCLC).
Methods: This retrospective study included NSCLC patients who underwent lung cancer surgery at The First People’s Hospital of Jiande between January 2019 and October 2023. Data on demographic characteristics, preoperative inflammatory biomarkers, surgical approach and duration, postoperative outcomes, and CT findings 1 month postoperatively were collected and analyzed. The effectiveness of preoperative inflammatory markers in predicting POP and clinical outcomes 1 month after surgical resection was assessed using propensity score matching.
Results: Among 568 patients, 72 (12.7%) had POP. After matching, 252 patients (POP group: 66; non-POP group: 186) were included in the analysis. The systemic immune-inflammation index (SII) and platelet-to-lymphocyte ratio (PLR) were significantly higher in the POP group than in the non-POP group (433.53 vs. 323.75, P = 0.001; 126.42 vs. 103.64, P < 0.001). The length of hospital stay and the percentage of patients who improved clinically based on chest CT findings 1 month after surgery were significantly higher in the POP group than in the non-POP group (11 days vs. 9 days, P = 0.008; 77.3% vs. 59.7%, P = 0.033). Multivariate analysis showed that PLR and the lymphocyte-to-monocyte ratio (LMR) were independent predictors of POP (AUC of 0.780 and 0.730, both at P < 0.001). However, there were no significant differences in postoperative radiographic outcomes among patients stratified by risk of POP.
Conclusion: PLR and LMR accurately predict POP in surgical patients with NSCLC. Nonetheless, these ratios may not significantly predict radiographic outcomes 1 month after surgical resection.
Introduction
Lung cancer (LC) is the most common malignancy globally, with high morbidity and mortality, imposing a significant health and economic burden (1, 2). Surgery is an effective treatment for patients with LC, especially early-stage non-small cell lung cancer (NSCLC) (3). However, surgical complications, including postoperative pneumonia (POP), may occur, prolonging hospital stay and increasing healthcare costs (4). Despite advancements in antimicrobial therapy and non-invasive respiratory support in the perioperative period, pulmonary infections are a significant cause of mortality in patients with LC (5–8).
The effective prevention and diagnosis of POP are essential components of the surgical treatment of LC. White blood cell (WBC) count, procalcitonin, and C-reactive protein are inflammatory markers of pulmonary infections and have been used to assess the inflammatory status in patients with LC (9–11). The neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), lymphocyte-to-monocyte ratio (LMR), and systemic immune-inflammation index (SII, platelet count × neutrophil count/lymphocyte count) are prognostic factors in LC (12–16). However, the ability of these inflammatory markers to predict the occurrence of POP in surgical patients with LC is unknown.
Our study has important clinical implications. Understanding the role of inflammatory biomarkers in postoperative outcomes can guide personalized perioperative care, potentially reducing the incidence of POP and improving patient recovery. Additionally, the assessment of 1-month pulmonary imaging changes provides a practical and objective measure of postoperative lung function and recovery, which is critical for monitoring patient progress and adjusting treatment plans. This study assessed the clinical value of preoperative inflammatory markers to predict POP and postoperative radiographic outcomes in patients who underwent surgical resection for NSCLC. Elucidating the predictive value of these biomarkers can improve perioperative management and surgical outcomes in patients with NSCLC.
Patients and methods
Study population and eligibility criteria
Patients who underwent surgical resection for LC at The First People’s Hospital of Jiande (Jiande, China) from January 2019 to December 2023 were evaluated retrospectively. The inclusion criteria were patients who underwent lung resection and those with a diagnosis of NSCLC. The exclusion criteria were (1) patients with benign disease (n=56); (2) those who underwent repeat surgery (n=10); (3) patients who underwent more than one surgery within 1 month (n=1); (4) those who received radiotherapy, chemotherapy, immunotherapy, targeted therapy, or other treatments before surgery (n=5); (5) patients with second primary lung tumors (n=4); (6) those with infections or autoimmune diseases requiring antibiotic or hormone therapy, including Crohn’s disease and systemic lupus erythematosus (n=31); (7) patients with stage IV NSCLC or received palliative surgery (n=9); and (8) patients transferred to other hospitals (n=1). Blood samples were collected from all patients within 3 days before surgery, and inflammatory markers were measured.
All patients were re-staged according to the eighth edition of the tumor, node, and metastasis (TNM) classification established by the International Association for the Study of LC (17).
This study complied with the Declaration of Helsinki and was approved by the Ethics Committee of The First People’s Hospital of Jiande. The requirement for informed consent was waived because of the retrospective nature of the study.
Data collection
The following data were collected retrospectively: demographic characteristics (sex, age, body mass index, and smoking history), clinicopathologic features, comorbidities (hypertension, diabetes, coronary heart disease, emphysema, and chronic obstructive pulmonary disease [COPD]), surgical approach, TNM stage, resection site, type of lung resection, number of mediastinal lymph nodes resected and number of nodal stations sampled, surgical duration, intraoperative blood loss volume, drainage time and volume, length of postoperative hospital stay, incidence of POP, histological type of NSCLC, and chest computed tomography (CT) findings 1 month after resection.
The following inflammatory markers were measured to assess inflammatory status preoperatively: neutrophil count, lymphocyte count, macrophage count, platelet count, NLR, PLR, LMR, and SII.
Observation indicators
This study examined three outcomes. The first outcome was the incidence of POP. The diagnosis of POP was based on the presence of at least three of the following features: (1) lung exudation and consolidation on chest radiographs or CT scans, (2) fever (body temperature > 38°C), (3) WBC count > 10000/mm3 or < 3000/mm3, (4) opportunistic pathogens in the sputum or bronchial secretions obtained by bronchoscopy (18). The second outcome was the predictive value of preoperative inflammatory markers in POP. The third outcome was clinical outcomes based on CT images obtained immediately after and 1 month after surgery. Two thoracic surgeons (Yingding Ruan and Jianwei Han) classified the outcomes into three categories based on CT findings: worse, unchanged, and improved. Disagreements were resolved by a third investigator (Ting Zhang).
Statistical analysis
Propensity score matching (PMS) (1:3 ratio) was performed to enhance comparisons and minimize bias. Propensity scores were calculated using a logistic regression model that included gender, age, body mass index, smoking history, surgical approach, TNM stage, resection sites, and type of resection. This approach allowed us to create a cohort that balanced the distribution of these potential confounding factors between the groups.
Additionally, we employed the standardized mean difference (SMD) to assess the balance of these covariates post-matching. Generally, an SMD value less than 0.10 is considered indicative of acceptable balance between groups, with values between 0.10 and 0.34 suggesting minor imbalance, 0.35 to 0.64 indicating moderate imbalance, 0.65 to 1.19 suggesting substantial imbalance, and SMD values of 1.20 or greater indicating a very large imbalance. Our results demonstrated that the SMD values for all covariates were within the acceptable range, confirming good balance between the matched groups.
Normally distributed continuous variables were compared using Student’s t-test and presented as means ± standard deviations. Non-normally distributed continuous variables were compared using the Wilcoxon rank-sum test and expressed as medians and 25th–75th percentiles. Categorical variables were compared using the Chi-square test or Fisher’s exact test and presented as percentages.
Univariate and multivariate analyses were conducted using binary logistic regression models. Variables with a p-value < 0.05 in the univariate analysis were considered significant and were included in the multivariate model to identify independent predictors. The multivariate analysis methods used in the study included backward elimination with a significance level of 0.05 for retention in the model.
Receiver operating characteristic (ROC) curve analysis was performed to determine the optimal cutoff for preoperative inflammatory factors. The area under the curve (AUC) values were calculated, and an AUC ≥ 0.7 was deemed clinically effective. All tests were two-sided, and a p-value of less than 0.05 was considered statistically significant. All statistical analyses were performed using SPSS version 22.0, ensuring the rigorous control of biases and providing detailed insights into the relationships between the study variables.
Results
Demographic and baseline characteristics
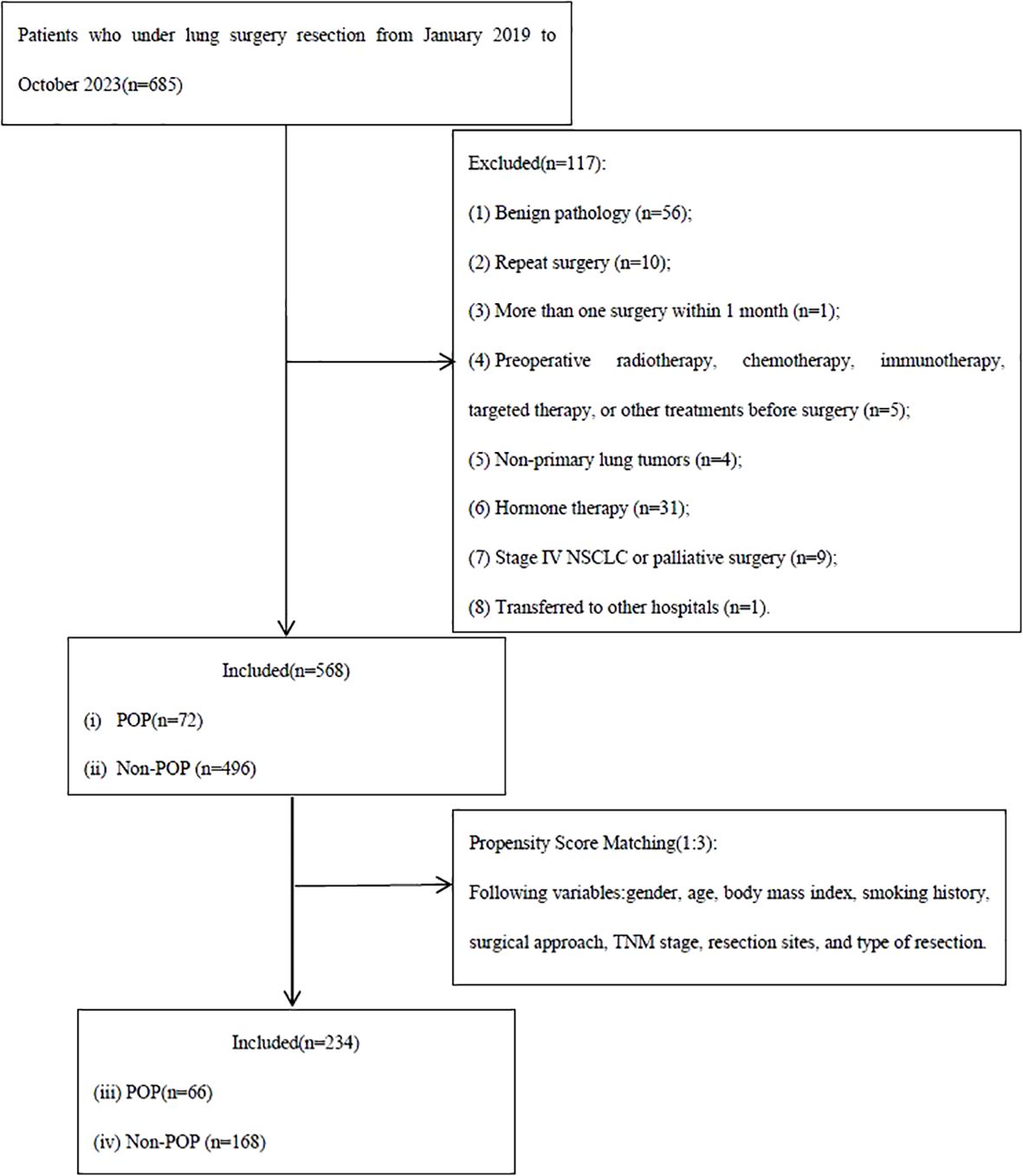
The study enrolled 685 patients treated surgically for LC in our hospital from January 2019 to December 2023. A total of 568 patients were included in the study after applying the eligibility criteria. After PSM, 252 patients (157 males [62.3%] and 95 females [37.7%]; mean age, 63.89 ± 11.43 years; 66 [26.2%] with POP and 186 [73.8%] without POP) were included in the analysis. The flowchart of patient selection is shown in Figure 1. The demographic, clinical, and operative characteristics of the cohort before and after PSM are shown in Table 1.
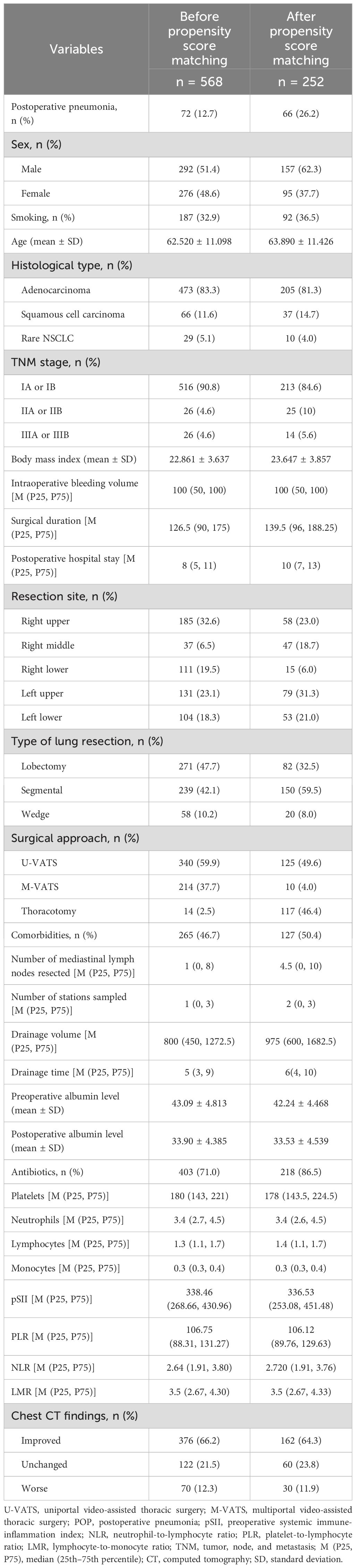
Table 1. Demographic, clinical, and operative characteristics of patients with non-small cell lung cancer before and after propensity score matching.
PSM effectively eliminated confounding factors (Table 2). Compared with controls, the POP group exhibited significantly higher preoperative SII values (433.53 [308.49, 606.14] vs. 323.75 [248.98, 404.31], P = 0.001) and PLR (126.42 [94.11, 167.10] vs. 103.64 [87.17, 121.64], P < 0.001). The LMR was similar between these two groups (3.5 [2.5, 4] vs. 3.5 [2.87, 5], P = 0.051). Postoperative hospital stay was considerably longer in the POP group than in the non-POP group (11 days vs. 9 days; P = 0.008). The analysis of chest CT scans showed that the percentage of patients who improved after surgery was significantly higher in the POP group than in the control group (77.3% vs. 59.7%, P = 0.033).
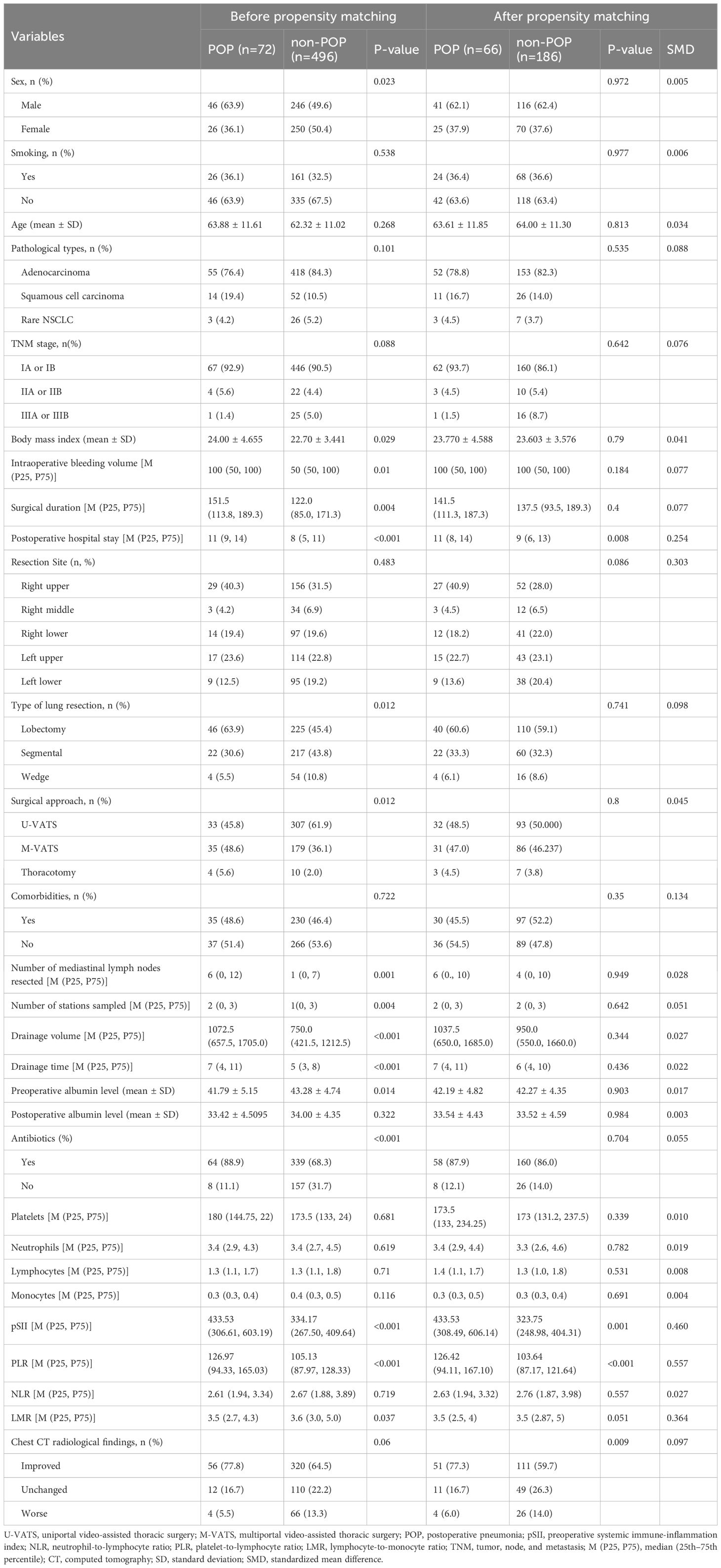
Table 2. Patient characteristics, incidence of POP, and statistical analysis [n(%), mean ± standard deviation, M(P25, P75)].
Risk factors for POP in NSCLC
After PSM, clinical data were analyzed by univariate and multivariate logistic regression analysis. Univariate analysis revealed that SII, PLR, LMR, and preoperative chest CT findings were significant risk factors for POP (P = 0.002, P < 0.001, P = 0.008, and P = 0.013). Multivariate analysis showed that PLR (Exponential(B) [Exp(B)] = 0.988, 95% confidence interval [CI] = 0.980–0.996, P = 0.005) and LMR (Exp(B) = 0.618, 95% CI = 0.494–0.772, P < 0.001) were significant predictors of POP (Tables 3, 4).
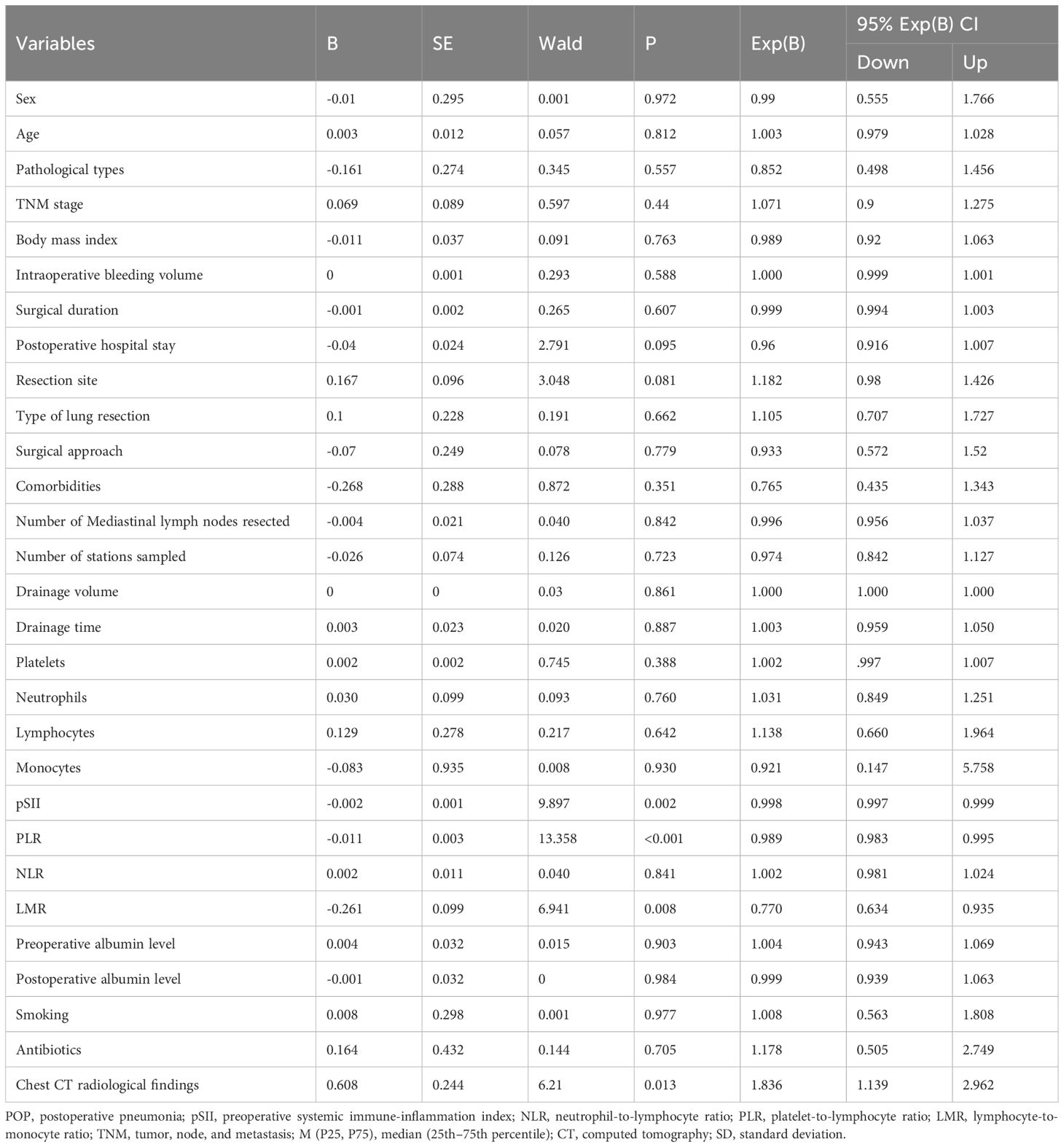
Table 3. Univariate logistic regression analysis of risk factors for postoperative pneumonia in patients with non-small cell lung cancer.
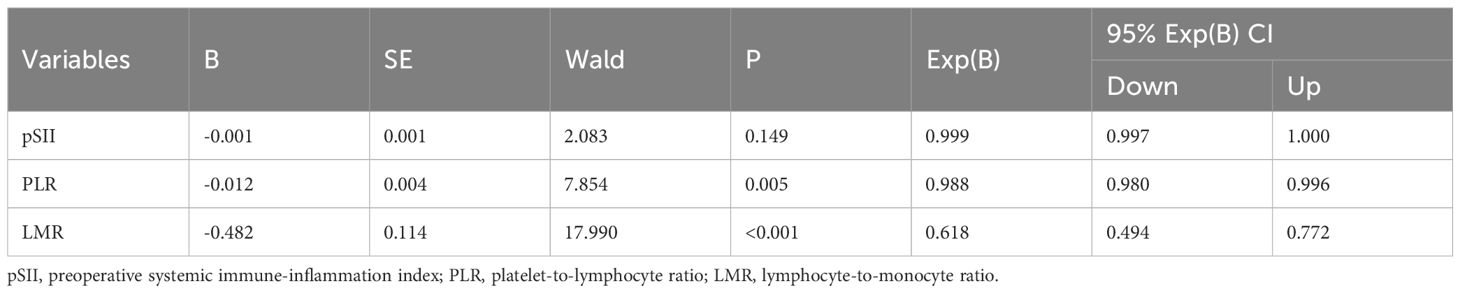
Table 4. Multivariate logistic regression analysis of risk factors for postoperative pneumonia in patients with non-small cell lung cancer.
Effectiveness of PLR and LMR in predicting POP
AUC analysis showed that PLR and LMR had a higher ability to predict POP (AUC of 0.780 and 0.729, both at P < 0.001). The optimal thresholds for PLR and LMR were 140.315 and 4.875. Youden index, sensitivity, and specificity were 0.549, 66.7%, and 88.2% for PLR and 0.362, 56.1%, and 80.1% for LMR. The AUC of preoperative NLR and SII were 0.488 and 0.641 (Figure 2).
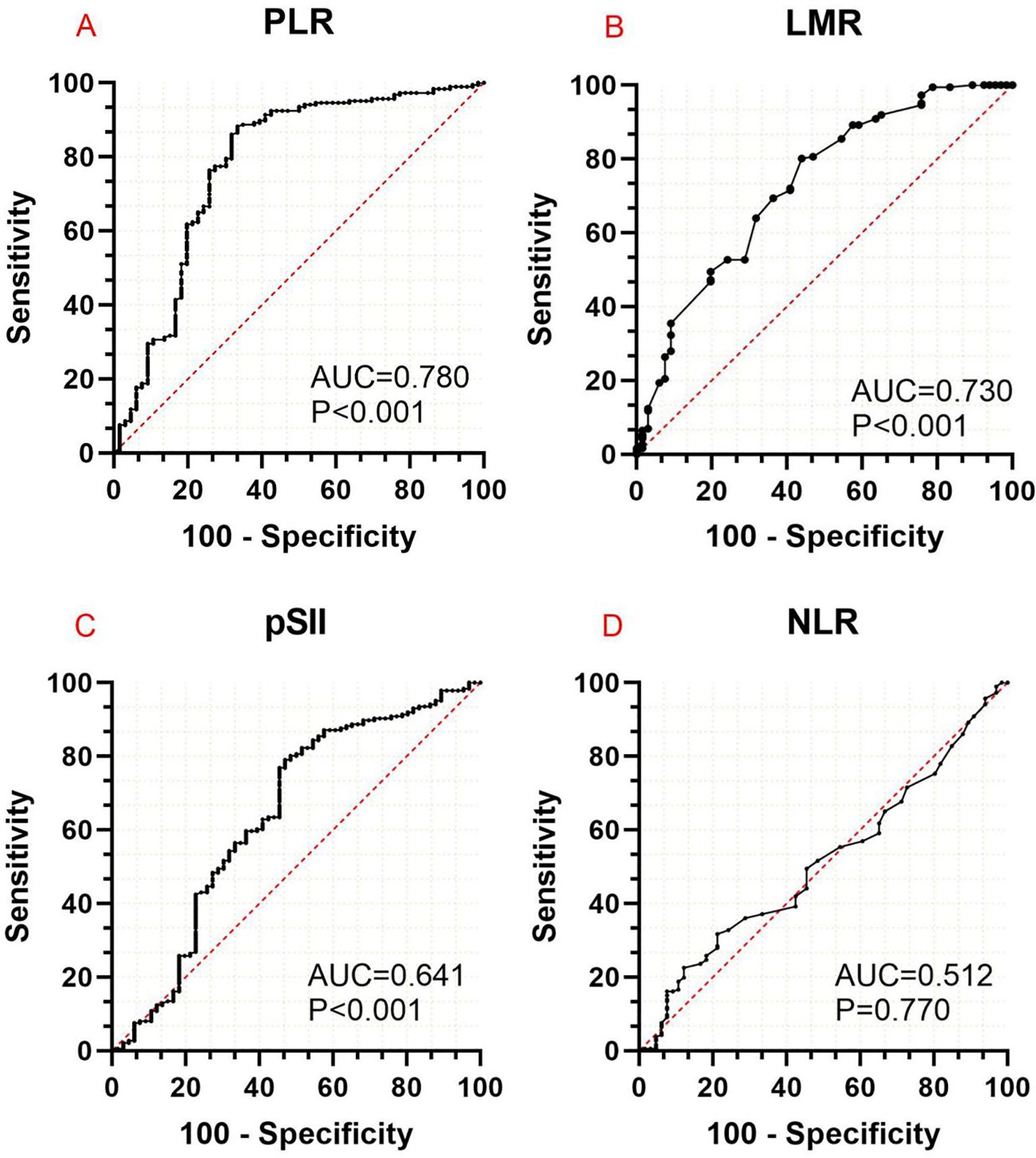
Figure 2. Area under the curve analysis of the effectiveness of PLR (A), LMR (B), SII (C), and NLR (D) in predicting the occurrence of postoperative pneumonia in patients with non-small cell lung cancer. PLR, platelet-to-lymphocyte ratio; LMP, lymphocyte-to-monocyte ratio; pSII, preoperative systemic immune-inflammation index; NLR, neutrophil-to-lymphocyte ratio.
Subgroup analysis of patients with POP
PSM analysis showed that PLR and LMR could accurately predict the occurrence of POP in patients with NSCLC. The optimal cutoff values for PLR and LMR based on AUC analysis were 140.315 and 4.875, respectively. The patients were divided into three risk groups based on threshold values: high (PLR > 140.315 and LMR > 4.875), intermediate (PLR > 140.315 or LMR > 4.875), and low (PLR < 140.315 and LMR < 4.875). Postoperative clinical outcomes in each group were classified into three categories—worse, unchanged, and improved—based on the analysis of CT images obtained immediately after and 1 month after surgery. There were no significant differences in outcomes among these groups (P= 0.976) (Table 5).
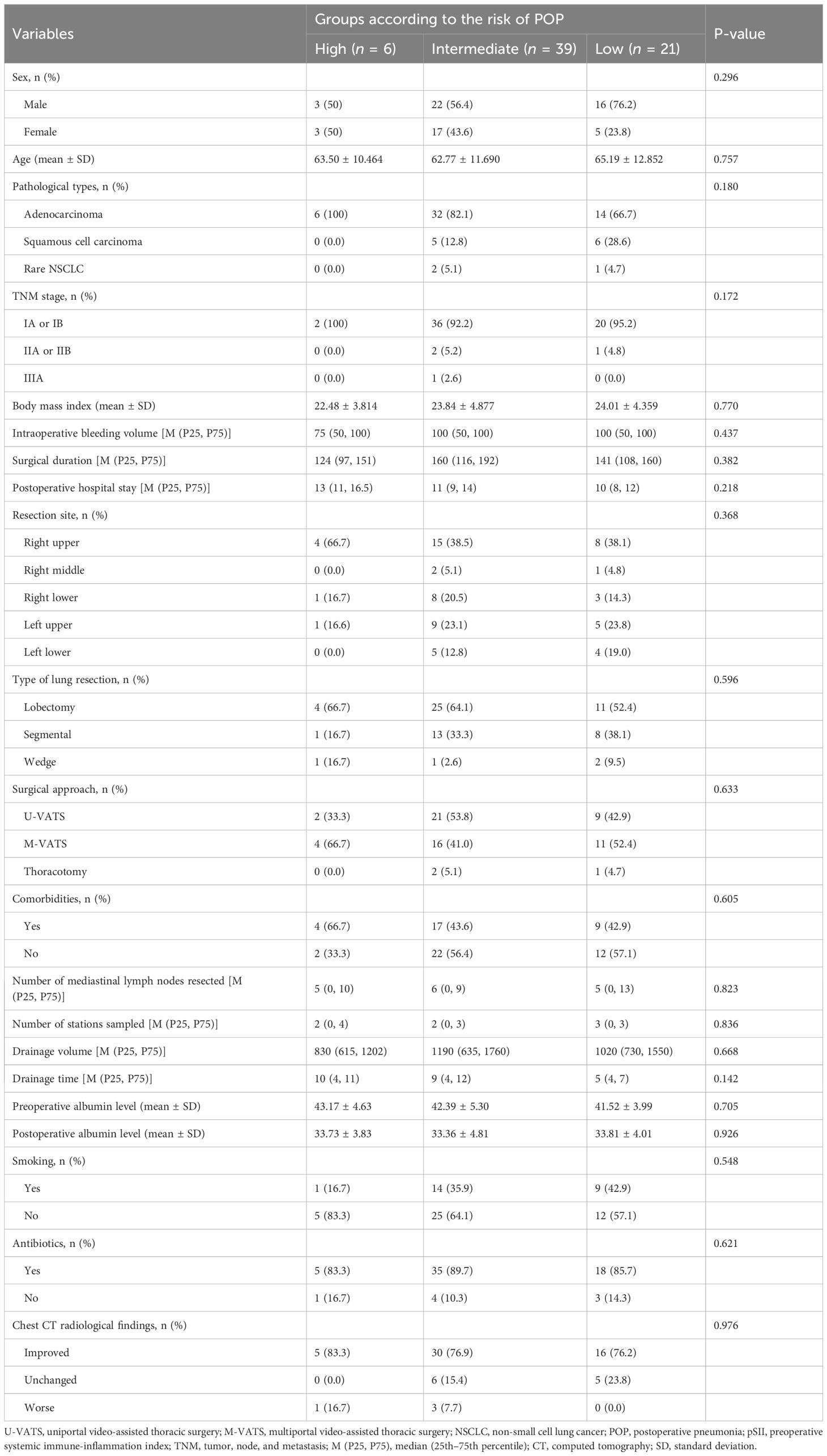
Table 5. Baseline, clinical, and operative characteristics of non-small cell lung cancer patients stratified into three groups according to the risk of POP.
Discussion
Advancements in video-assisted thoracic surgery, surgical equipment, and perioperative management have increased awareness of the benefits of minimally invasive surgery for LC. However, postoperative complications, especially pulmonary infections, can limit clinical recovery. Thus, accurately predicting these complications is crucial to improve prognosis and treatment. This study investigated the relationship between preoperative inflammatory markers (NLR, LMR, SII, and PLR) and POP using PSM. Further, this study is the first to evaluate POP, inflammatory markers, and 1-month radiographic outcomes in patients with NSCLC. The results revealed that PLR and LMR were independent risk factors for POP, with good predictive value, demonstrating the potential clinical utility of these markers in predicting the occurrence of postoperative pulmonary infections. Additionally, clinical outcomes improved 1 month after surgery in patients with POP based on the analysis of chest CT images, independent of PLR and LMR levels. Consequently, calculating PLR and LMR values in surgical patients with LC upon admission can help identify individuals who can benefit from intensive respiratory support and interventions targeting respiratory infections, improving prognosis (19, 20).
Peripheral blood inflammatory markers, including neutrophils, lymphocytes, macrophages, platelets, and natural killer cells, reflect systemic inflammation and have multiple roles in cancer (21, 22). In the early postoperative period, the decreased level and function of lymphocytes and NK cells can impair cellular immunity, increasing the risk of postoperative pneumonia and other inflammatory diseases (23). However, our results showed that preoperative neutrophils, lymphocytes, platelets, and monocytes had no significant impact on POP. In clinical practice, changes in WBC count may alter NLR, LMR, SII, and PLR. These ratios can reflect systemic and local inflammation and predict the occurrence of postoperative complications (12–14).
Our findings suggest that hematological ratios such as PLR and LMR can better predict the risk of POP. PLR, as a marker of platelet activation and systemic inflammation, has good potential to predict the risk of clinical deterioration in patients with pulmonary diseases (24, 25). Platelets play a crucial role in inflammation by releasing inflammatory mediators such as platelet-derived growth factor and thromboxane A2, which promote inflammatory responses (26, 27). Platelets are activated in response to infection and injury and aggregate at the site of damage, forming thrombi and initiating inflammatory reactions. Lymphocytes, as the central regulators of the immune system, are depleted by stress responses. Therefore, elevated PLR may reflect high-grade systemic inflammation, which may promote the occurrence of POP (28–30). Furthermore, platelets interact with immune cells such as lymphocytes and monocytes, influencing their function and activity. Increased PLR may indicate abnormal interactions of platelets with immune cells, potentially disrupting immune responses and increasing the risk of POP (31).
LMR reflects the balance between lymphocytes and monocytes, which have distinct roles in the immune system. Lymphocytes provide antitumor and anti-infectious defenses (32–34). Monocytes are involved in innate immunity and inflammation, killing pathogens by phagocytosis and the release of inflammatory mediators (35, 36). Decreased LMR may indicate lymphocyte depletion or monocyte expansion, compromising immune function and increasing the risk of infections. In POP, lower LMR may indicate weakened immune defense, rendering the lungs more susceptible to infections and inflammatory insults (37, 38). LMR correlates with disease incidence and prognosis (39–42). LMR is an independent predictor of stroke-associated pneumonia in patients with acute ischemic stroke, particularly when LMR was less than 4 (39).
In our study, LMR values were similar in the POP and non-POP groups (3.5 [2.5, 4] vs. 3.5 [2.87, 5], P=0.051), with a cutoff value of 4.875, consistent with a previous study indicating the potential of LMR to predict inflammatory conditions (38). These observations underscore the importance of considering hematological ratios, particularly PLR and LMR, in assessing POP risk and guiding clinical practice.
However, little is known about the ability of NLR, LMR, SII, and PLR to predict the occurrence of POP in surgical patients with LC. A retrospective study involving 1486 patients who underwent LC surgery found that preoperative SII was an independent risk factor for POP and predicted its occurrence (18). SII is also a risk factor for sepsis after intestinal obstruction surgery and for pulmonary complications after LC resection (22, 43). In contrast, NLR had a higher predictive value for POP than PLR and SII in older patients with hip fractures, even after PSM (44). NLR had the highest predictive value for POP in our cohort (AUC = 0.648, 95% CI = 0.594–0.701), maintaining significance even after PSM (Exp(B) = 2.04, 95% CI = 1.31–3.20). These data demonstrate that the impact of preoperative inflammation on POP varies in each study, highlighting the need to clarify this relationship.
In our study, both before and after PSM, PLR and LMR were significantly higher in the POP group than in the non-POP group (before PSM: P < 0.001 for PLR and P = 0.037 for LMR; after PSM: P < 0.001 for PLR and P = 0.051 for LMR). Multivariable analysis after PSM showed that PLR (P = 0.005) and LMR (P < 0.001) were independent risk factors for POP. Further, AUC analysis showed that PLR (ACU = 0.780, P < 0.001) and LMR (ACU = 0.730, P < 0.001) were better predictors of the occurrence of POP than SII and NLR.
Inflammatory burden, PLR, NLR, and other systemic inflammation markers are good prognostic factors in LC (33, 34, 45–47). However, little is known about the ability of these markers to predict postoperative outcomes in patients with pneumonia. To address this gap, we analyzed the effectiveness of PLR and LMR in predicting radiological outcomes 1 month after surgery. In the total cohort, the proportion of patients with improved outcomes before and after PSM was 66.2% (376/568) and 64.3% (162/252), respectively. In the POP group, the percentage of patients with improved outcomes before and after PSM was 77.8% (56/72) and 77.3% (51/66), respectively. The risk of POP was classified into three categories—low, intermediate, and high—based on the optimal cutoff values of PLR and LMR. There were no significant differences in postoperative radiological outcomes among the three risk groups. This result suggests that while PLR and LMR are good predictors of the risk of POP in patients with NSCLC, their predictive value for short-term radiological outcomes is limited.
Although we demonstrated the relationship of preoperative inflammatory biomarkers with POP and changes in lung imaging 1 month postoperatively, this study acknowledges the importance of other potential confounding factors. Moreover, despite employing PSM to minimize the influence of these factors, the roles of confounding variables such as age, gender, body mass index (BMI), smoking history, COPD status, and comorbidities (diabetes, cardiovascular disease, and tuberculosis) must be considered. For instance, the impact of age on the risk of postoperative pneumonia is controversial. Some studies suggest that immune dysfunction in elderly patients increases the risk of POP (48, 49), while others have shown that age is not an independent risk factor (50). BMI may also be a risk factor for POP (49, 51). For instance, BMI ≥ 24.0 kg/m2 was an independent risk factor for POP (51). However, the direct association between BMI and POP has not been demonstrated (48, 50, 52, 53). Smoking and COPD impair lung function and increase the risk of inflammatory responses, while comorbidities may exacerbate inflammatory reactions and affect postoperative recovery (48, 50, 52, 54). Therefore, we considered the effects of these confounding factors to increase the accuracy and reliability of our conclusions.
Surgical approaches, including open surgery and video-assisted thoracoscopic surgery (VATS), may influence the occurrence of POP. For instance, VATS, due to its minimal trauma and rapid recovery, can reduce the risk of POP (55–59). Although this study did not compare these surgical approaches, these findings are important for clinical practice, suggesting that VATS should be considered the preferred surgical option for NSCLC. However, the impacts of different surgical approaches on inflammatory biomarkers and postoperative outcomes require further investigation to improve surgical strategies and patient prognosis.
To prevent POP, we implemented comprehensive management measures, including routine lung rehabilitation training, bronchodilator and steroid therapy for high-risk patients, and anti-inflammatory treatment. In subsequent treatment phases, these measures should be more aggressively implemented for high-risk patients. These approaches have been validated and can significantly reduce the risk of POP (60–62).
This study has limitations. First, the retrospective design may lead to selection bias. Second, data were obtained from hospital records, limiting the effectiveness and reliability of data collection. Notably, we were unable to collect data on patients’ pulmonary function, which could be an important confounder affecting the outcomes of interest. Although biases were reduced using PSM, the small sample size may have limited statistical power. Third, the impact of inflammatory markers on long-term patient survival was not analyzed. Fourth, although PLR and LMR could accurately predict POP, diagnostic sensitivity was low (66.7% and 56.1%, respectively), possibly because of the small sample size, patient specificity, or analytical bias. Thus, large-scale, multicenter, prospective studies are needed to confirm the reliability and clinical value of these markers to predict POP in patients with LC.
Despite these limitations, this study evaluated short-term clinical outcomes in patients who developed pneumonia after lung resection for NSCLC. Moreover, the results provide a basis for using preoperative inflammatory markers to improve treatment outcomes and the postoperative recovery of patients with LC.
In conclusion, preoperative PLR and LMR can predict the occurrence of POP in surgical patients with NSCLC. However, PLR and LMR may not predict radiographic outcomes 1 month after surgical resection.
Data availability statement
The original contributions presented in the study are included in the article/supplementary material. Further inquiries can be directed to the corresponding author.
Ethics statement
The studies involving humans were approved by The First People’s Hospital of Jiande. The studies were conducted in accordance with the local legislation and institutional requirements. The ethics committee/institutional review board waived the requirement of written informed consent for participation from the participants or the participants’ legal guardians/next of kin because this is a retrospective study, and the data collection and analysis are based solely on existing medical records without direct intervention or additional data collection from the research subjects. Therefore, according to the relevant regulations of the ethics review committee, written informed consent has been waived.
Author contributions
YR: Conceptualization, Data curation, Formal analysis, Funding acquisition, Investigation, Methodology, Project administration, Resources, Software, Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing. WC: Data curation, Investigation, Writing – original draft. JH: Data curation, Investigation, Methodology, Writing – original draft. AY: Data curation, Investigation, Methodology, Writing – original draft. JX: Data curation, Investigation, Methodology, Writing – original draft. TZ: Conceptualization, Supervision, Writing – review & editing.
Funding
The author(s) declare that financial support was received for the research and/or publication of this article. This research was supported by grants from the Jiande Municipal Science and Technology Bureau (Grant Nos. 2023YW05 and 2023SJZX22).
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Abbreviations
AUC, area under the curve; CT, computed tomography; LMR, lymphocyte-to-monocyte ratio; NLR, neutrophil-to-lymphocyte ratio; LC, lung cancer; NSCLC, non-small cell lung cancer; PLR, platelet-to-lymphocyte ratio; POP, postoperative pneumonia; ROC, receiver operating characteristic; SII, systemic immune-inflammation index; TNM, tumor, node, and metastasis; WBC, white blood cells; IASLC, The International Association for the Study of Lung Cancer; VATS, video-assisted thoracoscopic surgery.
References
1. Siegel RL, Miller KD, Wagle NS, Jemal A. Cancer statistics, 2023. CA Cancer J Clin. (2023) 73:17–48. doi: 10.3322/caac.21763
2. Zheng RS, Chen R, Han BF, Wang SM, Li L, Sun KX, et al. Cancer incidence and mortality in China, 2022. Zhonghua Zhong Liu Za Zhi. (2024) 46:221–31. doi: 10.3760/cma.j.cn112152-20240119-00035
3. Altorki NK, Wang X, Wigle D, Gu L, Darling G, Ashrafi AS, et al. Perioperative mortality and morbidity after sublobar versus lobar resection for early-stage non-small-cell lung cancer: post-hoc analysis of an international, randomised, phase 3 trial (CALGB/Alliance 140503). Lancet Respir Med. (2018) 6:915–24. doi: 10.1016/S2213-2600(18)30411-9
4. Jones D, Kumar S, Anstee C, Gingrich M, Simone A, Ahmadzai Z, et al. Index hospital cost of adverse events following thoracic surgery: a systematic review of economic literature. BMJ Open. (2023) 13:e069382. doi: 10.1136/bmjopen-2022-069382
5. Schuetz P, Wirz Y, Sager R, Christ-Crain M, Stolz D, Tamm M, et al. Procalcitonin to initiate or discontinue antibiotics in acute respiratory tract infections. Cochrane Database Syst Rev. (2017) 10:CD007498. doi: 10.1002/14651858.CD007498.pub3
6. Kradin RL, Digumarthy S. The pathology of pulmonary bacterial infection. Semin Diagn Pathol. (2017) 34:498–509. doi: 10.1053/j.semdp.2017.06.001
7. Mizgerd JP. Pathogenesis of severe pneumonia: advances and knowledge gaps. Curr Opin Pulm Med. (2017) 23:193–7. doi: 10.1097/MCP.0000000000000365
8. Ruan Y, Han J, Yang A, Ding Q, Zhang T. Impact of preoperative inflammatory indices and postoperative pneumonia on postoperative atrial fibrillation in patients with non-small cell lung cancer: a retrospective study. BMC Pulm Med. (2024) 24:355. doi: 10.1186/s12890-024-03174-8
9. Chen P, Lin C, Jin Q, Ye B, Liu X, Wang K, et al. Investigating mechanisms of Sophora davidii (Franch.) skeels flower extract in treating LPS-induced acute pneumonia based on network pharmacology. J Ethnopharmacol. (2025) 337:118914. doi: 10.1016/j.jep.2024.118914
10. Ma L, Wu H. Clinical characteristics and distinguishing factors of patients with COVID-19 complicated with active pulmonary tuberculosis. J Investig Med. (2025) 73:75–84. doi: 10.1177/10815589241283511
11. Kheirandish E, Mahdizadeh M, Mahdizadeh M, Rezaeitalab F, Yousefi M, Shojaee SSR. Investigating the effect of eChinacea extraction syrup on the outcomes of lower respiratory infections in patients with COVID-19: a randomized clinical trial study. Virol J. (2024) 21:319. doi: 10.1186/s12985-024-02586-6
12. Chen C, Yang H, Cai D, Xiang L, Fang W, Wang R. Preoperative peripheral blood neutrophil-to-lymphocyte ratios (NLR) and platelet-to-lymphocyte ratio (PLR) related nomograms predict the survival of patients with limited-stage small-cell lung cancer. Transl Lung Cancer Res. (2021) 10:866–77. doi: 10.21037/tlcr-20-997
13. Xia L, Huang H, Xiao H, Wang D, Yang Z. Utilization of combined PD-L1 expression and neutrophil-to-lymphocyte ratio prior to surgery as a prognostic factor in non-small cell lung cancer with brain metastasis. Transl Cancer Res. (2019) 8:2864–77. doi: 10.21037/tcr.2019.11.08
14. Wang J, Li H, Xu R, Lu T, Zhao J, Zhang P, et al. NLR, PLR and D-dimer are associated with clinical outcome in lung cancer patients treated with surgery. BMC Pulm Med. (2022) 22:104. doi: 10.1186/s12890-022-01901-7
15. Fu F, Deng C, Wen Z, Gao Z, Zhao Y, Han H, et al. Systemic immune-inflammation index is a stage-dependent prognostic factor in patients with operable non-small cell lung cancer. Transl Lung Cancer Res. (2021) 10:3144–54. doi: 10.21037/tlcr-21-267
16. He H, Guo W, Song P, Liu L, Zhang G, Wang Y, et al. Preoperative systemic immune-inflammation index and prognostic nutritional index predict prognosis of patients with pulmonary neuroendocrine tumors after surgical resection. Ann Transl Med. (2020) 8:630. doi: 10.21037/atm-19-4476
17. Goldstraw P, Chansky K, Crowley J, Rami-Porta R, Asamura H, Eberhardt WE, et al. The IASLC lung cancer staging project: proposals for revision of the TNM stage groupings in the forthcoming (Eighth) edition of the TNM classification for lung cancer. J Thorac Oncol. (2016) 11:39–51. doi: 10.1016/j.jtho.2015.09.009
18. Jiang R, Li P, Shen W, Deng H, Qin C, Qiu X, et al. The predictive value of the preoperative systemic immune-inflammation index in the occurrence of postoperative pneumonia in non-small cell lung cancer: A retrospective study based on 1486 cases. Thorac Cancer. (2023) 14:30–5. doi: 10.1111/1759-7714.14691
19. Buonacera A, Stancanelli B, Colaci M, Malatino L. Neutrophil to lymphocyte ratio: an emerging marker of the relationships between the immune system and diseases. Int J Mol Sci. (2022) 23:3636. doi: 10.3390/ijms23073636
20. Güneylioğlu MM, Güngör A, Göktuğ A, Üner Ç, Bodur İ, Yaradılmış RM, et al. Evaluation of the efficiency of the systemic immune-inflammation index in differentiating parapneumonic effusion from empyema. Pediatr Pulmonol. (2022) 57:1625–30. doi: 10.1002/ppul.25926
21. Cupp MA, Cariolou M, Tzoulaki I, Aune D, Evangelou E, Berlanga-Taylor AJ. Neutrophil to lymphocyte ratio and cancer prognosis: an umbrella review of systematic reviews and meta-analyses of observational studies. BMC Med. (2020) 18:360. doi: 10.1186/s12916-020-01817-1
22. Jaillon S, Ponzetta A, Di Mitri D, Santoni A, Bonecchi R, Mantovani A. Neutrophil diversity and plasticity in tumour progression and therapy. Nat Rev Cancer. (2020) 20:485–503. doi: 10.1038/s41568-020-0281-y
23. Furák J, Németh T, Lantos J, Fabó C, Géczi T, Zombori-Tóth N, et al. Perioperative systemic inflammation in lung cancer surgery. Front Surg. (2022) 9:883322. doi: 10.3389/fsurg.2022.883322
24. Zhao Y, Wang Y, Jiang Y, Yang J, Zhang Y. The prognostic impact of neutrophil-to-lymphocyte ratio and platelet-to-lymphocyte ratio on patients with small cell lung cancer receiving first-line platinum-based chemotherapy: a systematic review and meta-analysis. BMC Pulm Med. (2024) 24:630. doi: 10.1186/s12890-024-03447-2
25. Korkmaz MF, Karacan A, Mehmedali A, Bozdemir ŞE, Arslan D, Oto A, et al. Immature granulocytes as an early clinical predictor in children with respiratory syncytial virus bronchiolitis. Pediatr Int. (2024) 66:e15840. doi: 10.1111/ped.15840
26. Chaplin DD. Overview of the immune response. J Allergy Clin Immunol. (2010) 125:S3–23. doi: 10.1016/j.jaci.2009.12.980
27. Hottz ED, Bozza FA, Bozza PT. Platelets in immune response to virus and immunopathology of viral infections. Front Med (Lausanne). (2018) 5:121. doi: 10.3389/fmed.2018.00121
28. El-Gazzar AG, Kamel MH, Elbahnasy OKM, El-Naggar ME. Prognostic value of platelet and neutrophil to lymphocyte ratio in COPD patients. Expert Rev Respir Med. (2020) 14:111–6. doi: 10.1080/17476348.2019.1675517
29. Zhang S, Sun P. Correlations of platelet-to-lymphocyte ratio, neutrophil-to-lymphocyte ratio and acute physiology, and chronic health evaluation II score with prognosis of elderly patients with chronic obstructive pulmonary disease and respiratory failure. Am J Transl Res. (2024) 16:3751–8. doi: 10.62347/ENAP1762
30. Liao QQ, Mo YJ, Zhu KW, Gao F, Huang B, Chen P, et al. Platelet-to-lymphocyte ratio (PLR), neutrophil-to-lymphocyte ratio (NLR), monocyte-to-lymphocyte ratio (MLR), and eosinophil-to-lymphocyte ratio (ELR) as biomarkers in patients with acute exacerbation of chronic obstructive pulmonary disease (AECOPD). Int J Chron Obstruct Pulmon Dis. (2024) 19:501–18. doi: 10.2147/COPD.S447519
31. Chiba Y, Mizoguchi I, Hasegawa H, Ohashi M, Orii N, Nagai T, et al. Regulation of myelopoiesis by proinflammatory cytokines in infectious diseases. Cell Mol Life Sci. (2018) 75:1363–76. doi: 10.1007/s00018-017-2724-5
32. Moro-García MA, Mayo JC, Sainz RM, Alonso-Arias R. Influence of inflammation in the process of T lymphocyte differentiation: proliferative, metabolic, and oxidative changes. Front Immunol. (2018) 9:339. doi: 10.3389/fimmu.2018.00339
33. Xie H, Ruan G, Wei L, Deng L, Zhang Q, Ge Y, et al. The inflammatory burden index is a superior systemic inflammation biomarker for the prognosis of non-small cell lung cancer. J Cachexia Sarcopenia Muscle. (2023) 14:869–78. doi: 10.1002/jcsm.13199
34. Zhang Y, Yan N, Feng Y, Wu Y, Sun Y, Gao X, et al. Inflammatory markers predict efficacy of immunotherapy in advanced non-small cell lung cancer: a preliminary exploratory study. Discovery Oncol. (2025) 16:8. doi: 10.1007/s12672-025-01753-7
35. Tong X, Cheng A, Yuan X, Zhong X, Wang H, Zhou W, et al. Characteristics of peripheral white blood cells in COVID-19 patients revealed by a retrospective cohort study. BMC Infect Dis. (2021) 21:1236. doi: 10.1186/s12879-021-06899-7
36. Zhao L, Zhang YP, Yang X, Liu X. Eosinopenia is associated with greater severity in patients with coronavirus disease 2019. Allergy. (2021) 76:562–4. doi: 10.1111/all.14455
37. Kosidło JW, Wolszczak-Biedrzycka B, Matowicka-Karna J, Dymicka-Piekarska V, Dorf J. Clinical significance and diagnostic utility of NLR, LMR, PLR and SII in the course of COVID-19: A literature review. J Inflammation Res. (2023) 16:539–62. doi: 10.2147/JIR.S395331
38. Guan J, Wang Q, Zhao Q. Lymphocyte to monocyte ratio is independently associated with futile recanalization in acute ischemic stroke after endovascular therapy. Neuropsychiatr Dis Treat. (2023) 19:2585–96. doi: 10.2147/NDT.S434225
39. Li X, Zhou X, Wang H, Ruan B, Song Z, Zhang G. Association between lymphocyte-to-monocyte ratio and stroke-associated pneumonia: a retrospective cohort study. PeerJ. (2024) 12:e18066. doi: 10.7717/peerj.18066
40. Kose N, Akin F, Yildirim T, Ergun G, Altun I. The association between the lymphocyte-to-monocyte ratio and coronary artery disease severity in patients with stable coronary artery disease. Eur Rev Med Pharmacol Sci. (2019) 23:2570–5. doi: 10.26355/eurrev_201903_17406
41. Mandaliya H, Jones M, Oldmeadow C, Nordman II. Prognostic biomarkers in stage IV non-small cell lung cancer (NSCLC): neutrophil to lymphocyte ratio (NLR), lymphocyte to monocyte ratio (LMR), platelet to lymphocyte ratio (PLR) and advanced lung cancer inflammation index (ALI). Transl Lung Cancer Res. (2019) 8:886–94. doi: 10.21037/tlcr.2019.11.16
42. Wu XB, Huang LX, Huang ZR, Lu LM, Luo B, Cai WQ, et al. The lymphocyte-to-monocyte ratio predicts intracranial atherosclerotic stenosis plaque instability. Front Immunol. (2022) 13:915126. doi: 10.3389/fimmu.2022.915126
43. Yang J, Ran T, Lin X, Xu J, Zhou S, Chen C, et al. Association between preoperative systemic immune inflammation index and postoperative sepsis in patients with intestinal obstruction: A retrospective observational cohort study. Immun Inflammation Dis. (2024) 12:e1187. doi: 10.1002/iid3.1187
44. Yao W, Wang W, Tang W, Lv Q, Ding W. Neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), and systemic immune inflammation index (SII) to predict postoperative pneumonia in elderly hip fracture patients. J Orthop Surg Res. (2023) 18:673. doi: 10.1186/s13018-023-04157-x
45. Xie H, Ruan G, Ge Y, Zhang Q, Zhang H, Lin S, et al. Inflammatory burden as a prognostic biomarker for cancer. Clin Nutr. (2022) 41:1236–43. doi: 10.1016/j.clnu.2022.04.019
46. Wei L, Xie H, Yan P. Prognostic value of the systemic inflammation response index in human Malignancy: A meta-analysis. Med (Baltimore). (2020) 99:e23486. doi: 10.1097/MD.0000000000023486
47. Song M, Zhang Q, Song C, Liu T, Zhang X, Ruan G, et al. The advanced lung cancer inflammation index is the optimal inflammatory biomarker of overall survival in patients with lung cancer. J Cachexia Sarcopenia Muscle. (2022) 13:2504–14. doi: 10.1002/jcsm.13032
48. Song Y, Liu J, Lei M, Wang Y, Fu Q, Wang B, et al. An external-validated algorithm to predict postoperative pneumonia among elderly patients with lung cancer after video-assisted thoracoscopic surgery. Front Oncol. (2021) 11:777564. doi: 10.3389/fonc.2021.777564
49. Sawai S, Nakatani E, Sato S, Hawke P, Mochizuki T, Nishida M, et al. Peak expiratory flow predicts the occurrence of postoperative pneumonia after esophagectomy for esophageal cancer. Dis Esophagus. (2024) 37:doae084. doi: 10.1093/dote/doae084
50. Lugg ST, Agostini PJ, Tikka T, Kerr A, Adams K, Bishay E, et al. Long-term impact of developing a postoperative pulmonary complication after lung surgery. Thorax. (2016) 71:171–6. doi: 10.1136/thoraxjnl-2015-207697
51. Yang R, Du C, Xu J, Yao L, Zhang S, Wu Y. Excessive intravenous crystalloid infusion after video-assisted thoracoscopic surgery lobectomy is associated with postoperative pneumonia. J Cardiothorac Surg. (2019) 14:209. doi: 10.1186/s13019-019-1024-6
52. Zhou J, Wu D, Zheng Q, Wang T, Lin M, Lu T, et al. A clinical prediction model for postoperative pneumonia after lung cancer surgery. J Surg Res. (2023) 284:62–9. doi: 10.1016/j.jss.2022.11.021
53. Bian H, Liu M, Liu J, Dong M, Hong G, Agrafiotis AC, et al. Seven preoperative factors have strong predictive value for postoperative pneumonia in patients undergoing thoracoscopic lung cancer surgery. Transl Lung Cancer Res. (2023) 12:2193–208. doi: 10.21037/tlcr-23-512
54. Liu GW, Sui XZ, Wang SD, Zhao H, Wang J. Identifying patients at higher risk of pneumonia after lung resection. J Thorac Dis. (2017) 9(5):1289–94. doi: 10.21037/jtd.2017.04.42
55. Sihoe ADL. Video-assisted thoracoscopic surgery as the gold standard for lung cancer surgery. Respirology. (2020) 25 Suppl 2:49–60. doi: 10.1111/resp.13920
56. Yun JK, Park I, Kim HR, Choi YS, Lee GD, Choi S, et al. Long-term outcomes of video-assisted thoracoscopic lobectomy for clinical N1 non-small cell lung cancer: A propensity score-weighted comparison with open thoracotomy. Lung Cancer. (2020) 150:201–8. doi: 10.1016/j.lungcan.2020.10.014
57. Matsuura Y, Ichinose J, Nakao M, Ninomiya H, Nishio M, Okumura S, et al. Outcomes of nodal upstaging comparing video-assisted thoracoscopic surgery versus open thoracotomy for lung cancer. Lung Cancer. (2021) 152:78–85. doi: 10.1016/j.lungcan.2020.12.017
58. Yang Y, Mei J, Lin F, Pu Q, Ma L, Liu C, et al. Comparison of the short- and long-term outcomes of video-assisted thoracoscopic surgery versus open thoracotomy bronchial sleeve lobectomy for central lung cancer: A retrospective propensity score matched cohort study. Ann Surg Oncol. (2020) 27:4384–93. doi: 10.1245/s10434-020-08805-y
59. Ye B, Wang M. Video-assisted thoracoscopic surgery versus thoracotomy for non-small cell lung cancer: A meta-analysis. Comb Chem High Throughput Screen. (2019) 22:187–93. doi: 10.2174/1386207322666190415103030
60. Leone M, Bouadma L, Bouhemad B, Brissaud O, Dauger S, Gibot S, et al. Brief summary of French guidelines for the prevention, diagnosis and treatment of hospital-acquired pneumonia in ICU. Ann Intensive Care. (2018) 8:104. doi: 10.1186/s13613-018-0444-0
61. Martin C, Auboyer C, Boisson M, Dupont H, Gauzit R, Kitzis M, et al. Antibioprophylaxis in surgery and interventional medicine (adult patients). Update 2017. Anaesth Crit Care Pain Med. (2019) 38:549–62. doi: 10.1016/j.accpm.2019.02.017
Keywords: preoperative inflammatory biomarkers, postoperative pneumonia, chest computed tomography, surgery, non-small cell lung cancer
Citation: Ruan Y, Cao W, Han J, Yang A, Xu J and Zhang T (2025) Impact of preoperative inflammatory biomarkers on postoperative pneumonia and one-month pulmonary imaging changes after surgery for non-small cell lung cancer. Front. Oncol. 15:1489068. doi: 10.3389/fonc.2025.1489068
Received: 31 August 2024; Accepted: 27 February 2025;
Published: 18 March 2025.
Edited by:
Yu-Chan Chang, National Yang Ming Chiao Tung University, TaiwanReviewed by:
H. Volkan Kara, Istanbul University- Cerrahpasa, TürkiyeGüntuğ Batıhan, Kafkas University, Türkiye
Copyright © 2025 Ruan, Cao, Han, Yang, Xu and Zhang. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Ting Zhang, emV6aHRAemp1LmVkdS5jbg==
†These authors have contributed equally to this work and share first authorship
 Yingding Ruan
Yingding Ruan Wenjun Cao2†
Wenjun Cao2† Ting Zhang
Ting Zhang