- 1Department of Radiology, Sichuan Provincial People’s Hospital, University of Electronic Science and Technology, Chengdu, China
- 2Department of Ultrasound, Ya’ an People’s Hospital, Ya’ an, China
Undifferentiated small round cell sarcoma (USRCS) is an extremely rare and highly aggressive group of malignant neoplasms affecting bones and soft tissues. USRCSs, particularly extraosseous variants pose significant diagnostic challenges due to their rarity, similar clinicoradiological features, nonspecific morphology and the necessity for comprehensive molecular analyses. This paper discusses a rare interesting case of retroperitoneal USRCS in a 12-year-old female with a five-day history of diarrhea and slight right lower abdominal pain. Hematological profile, renal function test, liver function test and tumor markers were in normal limits. Preoperative imaging revealed a well-defined, highly vascularized mass in the lower right retroperitoneal space, featuring patchy calcification and osseous and fatty components that compressed the right ureter, causing obstructive hydroureteronephrosis. Given the clinical and imaging findings. The lesion was initially misdiagnosed as a benign teratoma. However, postoperative pathology and genetic testing confirmed USRCS, remarkably, the Ki-67 index was only 10%. The patient did not undergo any additional postoperative treatment and achieved long-term survival. Despite USRCS in the retroperitoneal space with calcification has been reported, we believe this is the first published case of USRCS with calcification and osseous and fatty components mimicking teratoma. The case is important in that it demonstrates the unusual imaging appearance of retroperitoneal USRCS.
Introduction
Undifferentiated small round cell sarcoma(s) (USRCS) is a group of malignant tumors of uncertain histogenesis that predominantly arise in bones, but rarely in the soft tissues (1). Extraskeletal USRCSs have been found in the head and neck, skin, breast, lung, bone, and deep abdominal wall (2–5); however, USRCS in the retroperitoneal space is extremely rare (6–9). To date, less than 100 cases of retroperitoneal USRCS [including Ewing sarcoma (ES)/primitive neuroectodermal tumor (PNET)] have been published in the medical literature since it was first described in 1981 (10). In addition and, to the best of our knowledge, there have been no reports of retroperitoneal USRCS exhibiting calcification and osseous and fat production. Herein, we present a rare case of a malignant tumor located in the posterior abdominal cavity involving the right ureter. The pathological diagnosis was USRCS with calcification, and osseous and fatty components. This presented a diagnostic challenge because the imaging characteristics and intraoperative findings resembled a benign teratoma. We report imaging findings and discuss the pathogenesis and histogenesis of USRCS.
Case report
A 12-year-old girl presented with a five-day history of diarrhea and slight right lower abdominal pain. Family history did not reveal any malignancies; in particular, there was no history of reproductive system tumors or rare tumors. Physical examination revealed a large, well-defined, palpable mass, measuring approximately 5 cm × 5 cm in the right lower quadrant, and routine laboratory investigations including creatinine, urea nitrogen, glomerular filtration rate, lactate dehydrogenase, and alkaline phosphatase levels were normal. Serum tumor markers, such as CEA, AFP, CA125, CA15-3, CA19-9, CA 72-4, ferritin (FER), CA 50, and the total human chorionic gonadotropin level were within normal limits. Abdominal ultrasonography (USG) (Figure 1) and contrast-enhanced computed tomography (CECT) (Figure 2) revealed a heterogeneous mass with areas of calcification, and osseous and fatty components, measuring 7.3 cm × 6.6 cm × 4.0 cm, in the right posterior abdominal cavity with a rich blood flow. The lesion compressed the right ureter, ileum, cecum, inferior vena cava, abdominal aorta, right common iliac artery, and the internal and external iliac arteries without definitive invasion, resulting in obstructive hydroureteronephrosis. The volume of the right kidney and blood perfusion were reduced. There was no evidence of regional or distant lymphovascular invasion. Based on clinical manifestations and imaging findings, the mass was suspected to be a teratoma. The patient was scheduled for surgery within 1 week of presentation. Upon exploration of the abdomen, a mobile retroperitoneal mass, measuring 7 cm × 6 cm × 5 cm, was identified, which was intimately adhered to the vessels, digestive tract and right ureter without invasion or encasement. The entire mass was removed without any complications or spillage. A ureteral stent was implanted under ureteroscopy, preserving the right ureter, and postoperative renal function and hydronephrosis significantly improved. On the cut surface, there was no hair or bone in the tumor. Microscopic examination revealed that the tumor was composed of small round and spindle-shaped cells with scarce cytoplasm and clear vesiculated nuclei that proliferated in sheets or solid nests with bands of hyalinized fibrous tissue. Areas of osseous matrix production with calcification were also observed; rosettes were not observed. Immunohistochemical examination revealed the following: positive expression of CD99, NKX2.2, and SATB2; negative expression of AFP, CD30, CD56, CK, Desmin, S-100, SALL4, SMA, CgA, Myogenin, p63 and Syn; and a low proliferation rate, as indicated by Ki-67 (10%) (Figure 3). Dual-color, break-apart probe fluorescence in situ hybridization revealed no EWSR1 gene translocation and/or rearrangement. A search for other sites of tumor involvement using whole-body plain CT performed at an outside hospital revealed no abnormalities. The components of undifferentiated small round cell sarcoma in this case could not be further classified due to limited detection capability and economic limitations. These findings supported the diagnosis of localized USRCS arising from the posterior abdominal cavity. However, the patient refused adjuvant standard postoperative chemotherapy and/or radiotherapy. Abdominal USG was performed every 6 months at a local hospital. At the 25-month postoperative follow-up, the patient was alive without any signs of recurrence.
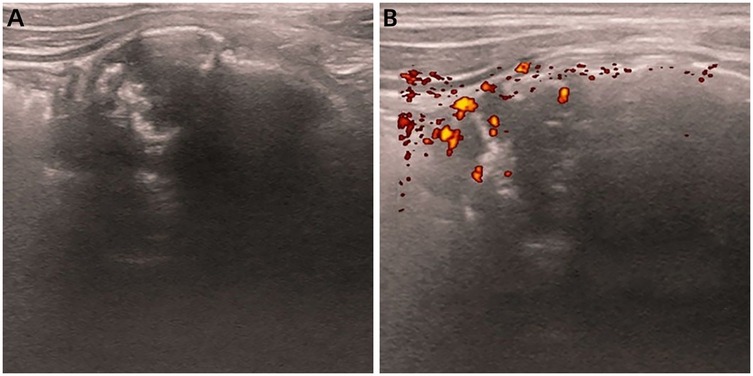
Figure 1. Abdominal ultrasound images of retroperitoneal USRCS in a 12-year-old girl. (A) Plain image shows a heterogeneous echoic mass with posterior acoustic shadowing in the right retroperitoneal region. (B) Transverse color Doppler image shows apparent flow within this structure.
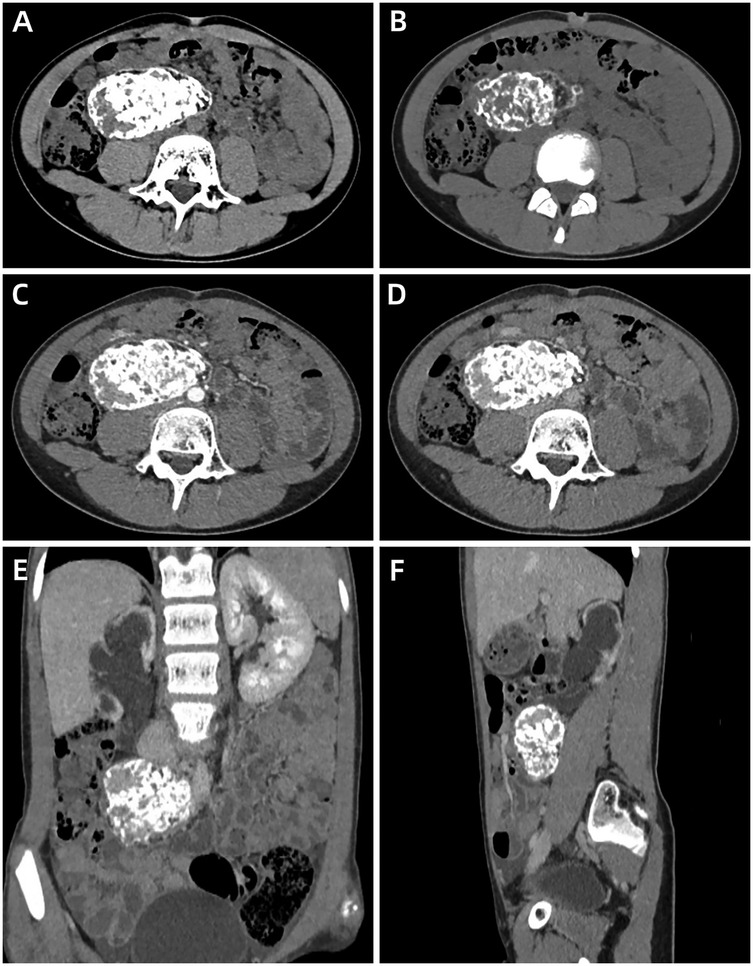
Figure 2. Abdominal contrast-enhanced computed tomography of a retroperitoneal undifferentiated small round cell sarcoma in a 12-year-old girl. (A) (soft window), (B) (bone window) Plain axial images show a mixed mass with areas of patchy calcification, osseous and fat components size in 7.3 cm × 6.6 cm × 4.0 cm, complete capsule. (C,D) Arterial phase (C) and portal venous phase (D) axial images show obvious enhancement of the lesion. (E,F) Coronal (E) and sagittal (F) images demonstrate that the lesion compressed the right ureter and caused obstructive hydroureteronephrosis.
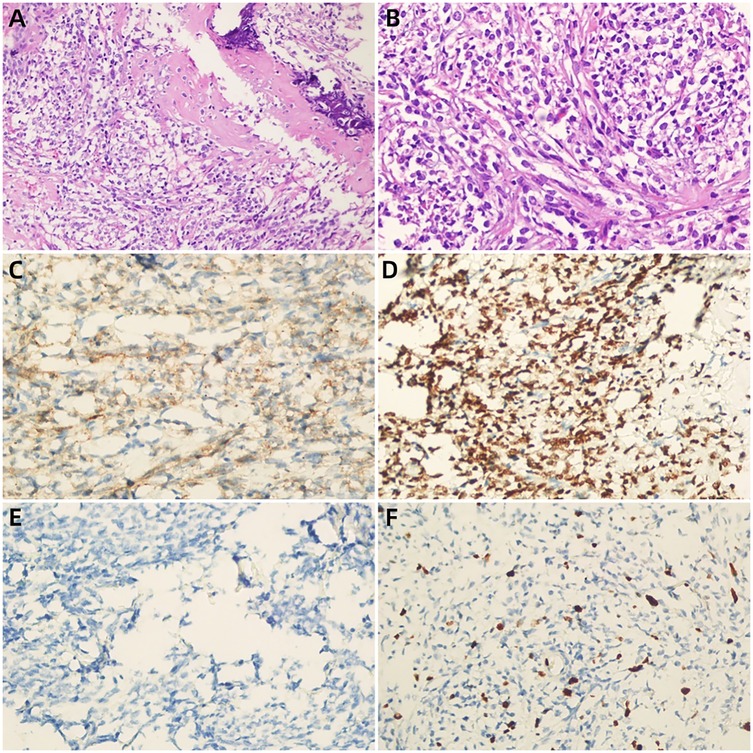
Figure 3. (A,B) (H&E) the tumor cells were small round with sparse cytoplasm, round nuclei and obscure nucleoli, suggesting undifferentiated small round cell sarcoma (a × 200, b × 400),areas of osseous matrix with calcification were observed;immunohistochemical findings showed strongly positivity for CD99 (C), NKX2.2 (D) and negative for AFP (E) the Ki-67 labeling index was approximately 10% (F).
Discussion
We report a rare case of retroperitoneal USRCS mimicking a teratoma, which, to our knowledge, has not been previously reported. This case is unique and interesting due to its unusual location, imaging findings, and biological behavior. Preoperative differential diagnosis of teratoma is difficult, which may cause delays in treatment. Pathological and IHC examinations, along with genetic testing, are crucial for accurate diagnosis and classification.
USRCS represents a heterogeneous group of small round cell tumors in the bone and soft tissues that share histological features but exhibit distinct molecular and clinical behaviors. The latest World Health Organization classification of soft tissue and bone tumors recognizes 4 categories within this group: ES, round cell sarcoma with EWSR1-non-ETS fusions, CIC-rearranged sarcoma, and sarcoma with BCOR alterations (11). ES is driven by fusions of EWSR1 (or much less frequently FUS) and ETS gene family members, most commonly FLI1 (approximately 90%) followed by ERG (approximately 5%) (12). Approximately 80%–90% of ES cases originate in the bones. The ribs, pelvis, or the diaphysis or metadiaphysis of long bones are most frequently involved. Approximately 10%–20% of ES occur in soft tissue, particularly in adults (13). CIC-rearranged sarcoma is defined by the presence of fusion involving CIC gene. Over 90% of tumors have CIC::DUX4 fusion (14), whereas rare alternative fusion partners include NUTM1, FOXO4, LEUTX, and NUTM2A (15). Most CIC-rearranged sarcomas present in the deep soft tissues of the head and neck, retroperitoneum, or pelvis. BCOR alteration sarcoma includes at least three molecular subcategories: BCOR::CCNB3 fusions, BCOR-alternative non-CCNB3 partner fusions and BCOR ITD (16). BCOR-rearranged sarcomas tend to occur more frequently in bone than in soft tissues, accounting for approximately 4% of round cell sarcomas. BCOR–CCNB3 sarcoma involves more frequently the pelvis, the lower limbs, and the paraspinal region. Round cell sarcoma with EWSR1-non-ETS fusions includes two distinct entities, the EWSR1::NFATc2- and the EWSR1::PATZ1-fusion sarcomas. NFATc2 is a transcription factor with an important role in immune regulation and thereby without any relationship to the ETS transcription factor family associated with ES (17). PATZ1 is also located on chromosome 22, only two megabases away from EWSR1, leading to an intrachromosomal rearrangement with pericentric inversion (18). The former occur more frequently in osseous locations rather than in the soft tissues (∼2:1). The latter has a strong predilection for the thoracoabdominal region. However, the aforementioned sites are not absolute; there is an overlap in their occurrence locations. Some cases of USRCS have been reported in the head and neck, lungs, breasts, kidneys, adrenal glands, gastrointestinal tract, and other visceral organs (2–5); however, cases occurring in the retroperitoneal space are extremely rare. Based on a literature review, less than 100 cases of primary USRCSs in the retroperitoneal space have been reported to date (6–9).
The radiological features of USRCS in the retroperitoneal space are nonspecific. On CT/MRI, USRCS usually appear as ill-defined, irregular, or lobular moderately heterogeneous masses with cystic and necrotic areas, and rarely as calcification and hemorrhage, as well as mild-to-moderate heterogeneous enhancement (6–9). Interestingly, in our case, the tumor was a well-defined, regular, mixed-density mass with patchy calcification, osseous components, and fat components, as revealed by CT and USG suggesting that the tumor was a teratoma, but did not raise the suspicion of sarcoma. Although, some researchers (19–21) reported ES/PNET in the retroperitoneal space that presented as lobulated masses with calcification or osseous components, in our case, we identified numerous patchy calcification, and osseous and fatty components accounting more than 80% of the tumor. To our knowledge, such findings have not been previously reported in the medical literature. We speculated that the heterogeneity of the imaging findings may be related to the origin of tumor cells. Although the developmental origins of USRCS have not been precisely determined, accumulating evidence over the past 2 decades suggests that USRCS may arise from mesenchymal stem cells (MSCs) or progenitor cells of specific differentiation stages (22). MSCs are a type of stem cell with multilineage differentiation potential and can differentiate into various human tissues, such as adipogenic, chondrogenic, and osteogenic tissues. In addition, we speculated that USRCS in the retroperitoneal space originated from the transformation of the teratoma along the mesodermal lines. Teratomas are pluripotent tissues capable of transforming into endodermal, ectodermal, or mesodermal lines. Subsequently, it can transform into a PNET, carcinoma, sarcoma, or carcinosarcoma (23). In our case, IHC results revealed negative expression of AFP, CK, S-100, SMA, Myogenin, Syn. As such, this possibility was excluded.
USRCS is a highly invasive and rapidly growing malignant tumor with poor prognosis. ES are highly aggressive with metastases detected at presentation in approximately one in four patients. Despite current aggressive cytotoxic treatment regimens the 5-year OS of patients with metastatic ES ranges from 20% to 35% (24). Even in primary nonmetastatic disease 30%–40% of patients experience recurrence, either local, distant or combined, during follow-up. Survival after recurrence is poor, with 5-year post-relapse survival varying from 15% to 25%, local recurrence doing better than distant recurrence (25). CIC-rearranged sarcomas are molecularly defined, aggressive tumors. With a 5-year overall survival of around 50%, the outcome is poor. There is a high potential for metastasis, often at the time of diagnosis (26).The prognosis of BCOR alteration sarcomas is similar to ES and much better than CIC-rearranged sarcomas (27). The prognosis data about the round cell sarcoma with EWSR1-non-ETS fusions is limited. Initial reports showed a very aggressive behavior (28), with high risk for metastatic spread to lung, pleura, soft tissues and lymph nodes. Although later studies described occasional cases that behaved in a more indolent fashion, however, with the caveat that follow-up of those studies was very short (29). Additional studies with longer follow-up will be essential to better understand their true biological potential.
Interestingly, in our case, the malignant tumor manifested benign behavior. First, on CT, a sarcoma was well-defined with a complete calcified capsule. Yi et al. reported (30) that 53.3% (8/15) of PNET in intra-abdominal and retroperitoneal regions had well-defined margins, which may have two reasons. First, abundant fat surrounding the mass created sharp contrast and a distinct natural boundary between the two different tissues. Second, the tumor's rapid expansion continuously compressed the surrounding connective tissues of the retroperitoneal space, resulting in a complete capsule. In our case, the lesion compressed the right ureter, causing obstructive hydroureteronephrosis. The volume and blood perfusion of the right kidney were reduced, while the volume of the left kidney was increased; however routine urianlysis was normal and there were no symptoms related to urinary tract obstruction. These findings suggest that the growth of the tumor was a chronic process. Second, there were numerous calcification and osseous and fatty components in the lesion mimicking a teratoma, suggesting benign behavior. Third, in our case, the Ki-67 index was only 10%, suggesting mitotic inactivity. In Yi et al. study, they found that the proliferation rate was relatively high (with a median Ki-67 index of 40%, ranging from 20% to 90%) in the 8 cases with clear margins. Given the rapid formation of the capsule, we hypothesized that the capsule did not have sufficient time to be destroyed by tumor invasion, even though its high malignancy and invasiveness.
Currently, there is no standard treatment for USRCS due to its global rarity. Most treatments involve early surgical resection and are supplemented with radiotherapy and chemotherapy. In the present case, the patient did not undergo any adjuvant treatment and is currently alive (25 months postoperatively) without any signs of recurrence. Further studies are needed to determine whether routine chemoradiotherapy is necessary for USRCS with benign radiological features and a low Ki-67 index.
Conclusion
We report the first case of USRCS with calcification and osseous and fatty formation, arising from the right lower retroperitoneal space. Retroperitoneal USRCS can mimic teratoma and exhibit benign behaviors. Our report contributes to the clinical and imaging knowledge on this disease and increases awareness among clinicians and radiologists because early diagnosis of USRCS is imperative for management of the disease and improvement of prognosis.
Data availability statement
The original contributions presented in the study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding author.
Ethics statement
The studies involving humans were approved by The Ethics Committee of Sichuan Provincial People's Hospital. The studies were conducted in accordance with the local legislation and institutional requirements. Written informed consent for participation in this study was provided by the participants' legal guardians/next of kin. Written informed consent was obtained from the individual(s), and minor(s)' legal guardian/next of kin, for the publication of any potentially identifiable images or data included in this article.
Author contributions
DL: Writing – review & editing, Writing – original draft. XLiu: Data curation, Writing – original draft, Investigation. YD: Data curation, Writing – original draft. RW: Data curation, Writing – review & editing. XLi: Data curation, Writing – review & editing, Investigation.
Funding
The author(s) declare that no financial support was received for the research and/or publication of this article.
Acknowledgments
We greatly appreciate the assistance of the staff of the Department of Pediatric Surgery and the Department of Pathology. The authors would like to thank the patient who participated in this study.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Choi JH, Ro JY. The 2020 WHO classification of tumors of soft tissue: selected changes and new entities. Adv Anatomic Pathol. (2021) 28:44–58. doi: 10.1097/PAP.0000000000000284
2. Wright A, Desai M, Bolan CW, Badawy M, Guccione J, Korivi BR, et al. Extraskeletal Ewing sarcoma from head to toe: multimodality imaging review. Radiographics. (2022) 42:1145–60. doi: 10.1148/rg.210226
3. Antonescu CR, Owosho AA, Zhang L, Chen S, Deniz K, Huryn JM, et al. Sarcomas with CIC-rearrangements are a distinct pathologic entity with aggressive outcome: a clinicopathologic and molecular study of 115 cases. Am J Surg Pathol. (2017) 41:941–9. doi: 10.1097/PAS.0000000000000846
4. Kao YC, Owosho AA, Sung YS, Zhang L, Fujisawa Y, Lee JC, et al. BCOR-CCNB3 fusion positive sarcomas: a clinicopathologic and molecular analysis of 36 cases with comparison to morphologic Spectrum and clinical behavior of other round cell sarcomas. Am J Surg Pathol. (2018) 42:604–15. doi: 10.1097/PAS.0000000000000965
5. Liu L, Li L, Ding Y, Kong F, Mo W, Ye H, et al. Report and literature review of four cases of EWSR1::NFATC2 round cell sarcoma. Diagn Pathol. (2024) 19:19. doi: 10.1186/s13000-024-01443-y
6. Qian X, Kai X, Shaodong L, Gaohong C, Hong M, Jingjing L. Radiological and clinicopathological features of pPNET. Eur J Radiol. (2013) 82:e888–93. doi: 10.1016/j.ejrad.2013.08.049
7. Ulusan S, Koc Z, Canpolat ET, Colakoglu T. Radiological findings of primary retroperitoneal Ewing sarcoma. Acta Radiol. (2007) 48:814–18. doi: 10.1080/02841850701408244
8. Somarouthu BS, Shinagare AB, Rosenthal MH, Tirumani H, Hornick JL, Ramaiya NH, et al. Multimodality imaging features, metastatic pattern and clinical outcome in adult extraskeletal Ewing sarcoma: experience in 26 patients. Br J Radiol. (2014) 87:20140123. doi: 10.1259/bjr.20140123
9. Wei Y, Zhang Z, Long C, Huang X, Tang W, Mo X, et al. Case report: colon malignant tumor caused by retroperitoneal small round cell undifferentiated sarcoma. Front Oncol. (2023) 13:1212475. doi: 10.3389/fonc.2023.1212475
10. Blight JEM, Puls JL. Collateral venous flow through uterus caused by retroperitoneal extraosseous Ewing sarcoma. Urology. (1981) 17:386–9. doi: 10.1016/0090-4295(81)90279-x
11. Hwang S, Hameed M, Kransdorf M. The 2020 world health organization classification of bone tumors: what radiologists should know. Skeletal Radiol. (2023) 52:329–48. doi: 10.1007/s00256-022-04093-7
12. Dehner CA, Lazar AJ, Chrisinger JSA. Updates on WHO classification for small round cell tumors: Ewing sarcoma vs. everything else. Hum Pathol. (2024) 147:101–13. doi: 10.1016/j.humpath.2024.01.007
13. Shingde MV, Buckland M, Busam KJ, McCarthy SW, Wilmott J, Thompson JF, et al. Primary cutaneous Ewing sarcoma/primitive neuroectodermal tumour: a clinicopathological analysis of seven cases highlighting diagnostic pitfalls and the role of FISH testing in diagnosis. J Clin Pathol. (2009) 62:915–19. doi: 10.1136/jcp.2008.064014
14. Italiano A, Sung YS, Zhang L, Singer S, Maki RG, Coindre JM, et al. High prevalence of CIC fusion with double-homeobox (DUX4) transcription factors in EWSR1-negative undifferentiated small blue round cell sarcomas. Genes Chromosomes Cancer. (2012) 51:207–18. doi: 10.1002/gcc.20945
15. Linos K, Dermawan JK, Bale T, Rosenblum MK, Singer S, Tap W, et al. Expanding the molecular diversity of CIC-rearranged sarcomas with novel and very rare partners. Mod Pathol. (2023) 36:100103. doi: 10.1016/j.modpat.2023.100103
16. Peters TL, Kumar V, Polikepahad S, Lin FY, Sarabia SF, Liang Y, et al. BCOR-CCNB3 fusions are frequent in undifferentiated sarcomas of male children. Mod Pathol. (2015) 28:575–86. doi: 10.1038/modpathol.2014.139
17. Perret R, Escuriol J, Velasco V, Mayeur L, Soubeyran I, Delfour C, et al. NFATc2-rearranged sarcomas: clinicopathologic, molecular, and cytogenetic study of 7 cases with evidence of AGGRECAN as a novel diagnostic marker. Mod Pathol. (2020) 33:1930–44. doi: 10.1038/s41379-020-0542-z
18. Michal M, Rubin BP, Agaimy A, Kosemehmetoglu K, Rudzinski ER, Linos K, et al. EWSR1-PATZ1-rearranged sarcoma: a report of nine cases of spindle and round cell neoplasms with predilection for thoracoabdominal soft tissues and frequent expression of neural and skeletal muscle markers. Mod Pathol. (2021) 34:770–85. doi: 10.1038/s41379-020-00684-8
19. Patnaik S, Yarlagadda J, Susarla R. Imaging features of Ewing’s sarcoma: special reference to uncommon features and rare sites of presentation. J Cancer Res Ther. (2018) 14:1014–22. doi: 10.4103/jcrt.JCRT_1350_16
20. Kim MS, Kim B, Park CS, Song SY, Lee EJ, Park NH, et al. Radiologic findings of peripheral primitive neuroectodermal tumor arising in the retroperitoneum. Am J Roentgenol. (2006) 186:1125–32. doi: 10.2214/AJR.04.1688
21. Kim JM, Chu YC, Choi CH, Kim L, Choi SJ, Park IS, et al. Peripheral primitive neuroectodermal tumor with osseous component of the small bowel mesentery: a case study. Korean J Pathol. (2013) 47:77–81. doi: 10.4132/KoreanJPathol.2013.47.1.77
22. Kallen ME, Hornick JL. The 2020 WHO classification: what’s new in soft tissue tumor pathology? Am J Surg Pathol. (2021) 45:e1–23. doi: 10.1097/PAS.0000000000001552
23. Matsuo M, Saigo C, Takeuchi T, Onogi A, Watanabe N, Aikyo S, et al. Ovarian clear cell carcinoma and mature cystic teratoma transformed to PNET and carcinosarcoma: a case report with an immunohistochemical investigation. Biomedicines. (2022) 10:547. doi: 10.3390/biomedicines10030547
24. Cotterill SJ, Ahrensn S, Paulussen M, Jurgens HF, Voute PA, Gadner H, et al. Prognostic factors in Ewing’s tumor of bone: analysis of 975 patients from the European intergroup cooperative Ewing’s sarcoma study group. J Clin Oncol. (2000) 18:3108–14. doi: 10.1200/JCO.2000.18.17.3108
25. Stahl M, Ranft A, Paulussen M, Bolling T, Vieth V, Bielack S, et al. Risk of recurrence and survival after relapse in patients with Ewing sarcoma. Pediatr Blood Canc. (2011) 57:549–53. doi: 10.1002/pbc.23040
26. Connolly EA, Bhadri VA, Wake J, Ingley KM, Lewin J, Bae S, et al. Systemic treatments and outcomes in CIC-rearranged sarcoma: a national multi-centre clinicopathological series and literature review. Cancer Med. (2022) 11:1805–16. doi: 10.1002/cam4.4580
27. Palmerini E, Gambarotti M, Italiano A, Nathenson MJ, Ratan R, Dileo P, et al. A global collaboRAtive study of CIC-rearranged, BCOR::CCNB3-rearranged and other ultra-rare unclassified undifferentiated small round cell sarcomas (GRACefUl). Eur J Cancer. (2023) 183:11–23. doi: 10.1016/j.ejca.2023.01.003
28. Watson S, Perrin V, Guillemot D, Reynaud S, Coindre JM, Karanian M, et al. Transcriptomic definition of molecular subgroups of small round cell sarcomas. J Pathol. (2018) 245:29–40. doi: 10.1002/path.5053
29. Dehner CA, Torres-Mora J, Gupta S, Kipp BR, Halling KC, Chen S, et al. Sarcomas harboring EWSR1::PATZ1 fusions: a clinicopathologic study of 17 cases. Mod Pathol. (2024) 37:100400. doi: 10.1016/j.modpat.2023.100400
Keywords: undifferentiated small round cell sarcoma, teratoma, retroperitoneal space, CT, EWSR1 translocation
Citation: Liu D, Liu X, Deng Y, Wu R and Li X (2025) Undifferentiated small round cell sarcomas in the retroperitoneal space in a 12-year-old female: a rare case report. Front. Pediatr. 13:1602157. doi: 10.3389/fped.2025.1602157
Received: 28 March 2025; Accepted: 5 August 2025;
Published: 29 August 2025.
Edited by:
Luca Giacomelli, Polistudium srl, ItalyReviewed by:
Mehmet Sait Ozsoy, Istanbul Medeniyet University, TürkiyeMassimo Della Giustina, University of Udine, Italy
Copyright: © 2025 Liu, Liu, Deng, Wu and Li. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Xin Li, dmljbGl4QDEyNi5jb20=
†These authors have contributed equally to this work and share first authorship
 Dan Liu
Dan Liu Xiaoge Liu2,†
Xiaoge Liu2,†