- 1Department of Pediatric Surgery, Affiliated Hospital of Zunyi Medical University, Zunyi, China
- 2Department of Pediatric Surgery, Guizhou Children’s Hospital, Zunyi, China
Objective: To investigate the clinical features, imaging manifestations, pathological types, and surgical strategies of mediastinal masses in children with this condition, aiming to enhance early diagnosis and perioperative management.
Methods: Clinical data of children diagnosed with mediastinal masses and treated at the Affiliated Hospital of Zunyi Medical University between January 2019 and August 2024 were retrospectively reviewed. Key variables analyzed included demographic characteristics, clinical presentation, imaging findings, surgical procedures, intraoperative management, pathological results, and follow-up outcomes. Risk stratification and intraoperative safety strategies were assessed through representative complex case analyses.
Results: A total of 51 children were enrolled, comprising 29 males and 22 females, with a median age of 5.0 years. Primary clinical presentations included shortness of breath upon exertion (54.9%) and cough (49.0%), while 5 children were asymptomatic. All diagnoses were confirmed by computed tomography (CT), with lesions predominantly located in the middle and posterior mediastinum. Ganglioneuroma, bronchogenic cyst, and schwannoma were the most common types. Benign lesions accounted for 84.3%, whereas 8 cases were malignant. Thoracoscopic surgery was performed in 45 children and open thoracotomy in 6. Intraoperatively, iodine solution was applied to cystic lesions in 16 cases, and sclerosing agent injections were administered to 4 lymphangioma cases. Postoperatively, 10 children with malignant tumors required subsequent oncological treatment. One child encountered mechanical ventilation failure during anesthetic induction, which was resolved by transitioning to a prone position. In bronchogenic cysts, preemptive decompression effectively prevented complications such as bronchial obstruction.
Conclusion: Mediastinal masses in children are predominantly benign, frequently presenting with nonspecific respiratory symptoms, and CT remains the diagnostic modality of choice. Surgical resection was the primary treatment. Individualized intraoperative management based on lesion type, appropriate patient positioning, and decompression procedures can significantly reduce complication risks. Preoperative airway compression assessment and collaboration with anesthesia teams to establish emergency ventilation protocols are essential to ensuring perioperative safety in children.
1 Introduction
Although mediastinal masses are relatively rare in children, their occurrence can rapidly lead to compression of critical structures such as the heart, lungs, great vessels, and airways due to the limited space and complex anatomy of the mediastinum, potentially causing severe and even life-threatening complications. Unlike adults, mediastinal tumors in children are distinct, typically arising from embryonic remnants, neurogenic proliferation, or cystic lesions, resulting in notable differences in clinical presentation, pathology, and therapeutic strategies. Previous studies have demonstrated that neurogenic tumors, cystic lesions, and lymphatic tissue hyperplasia are the most common mediastinal masses in children; however, systematic analyses regarding their precise incidence, anatomical distribution, and surgical risks remain insufficient.
Clinically, mediastinal masses in children often present with nonspecific symptoms, frequently identified during routine physical examination. Larger masses may cause compressive symptoms, including cough, dyspnea, hoarseness, and dysphagia (1, 2). Chest radiography serves as an initial screening method, typically revealing soft tissue masses obscuring normal anatomical borders (1). Computed tomography (CT) remains the preferred diagnostic modality, providing detailed information regarding lesion location, size, internal characteristics, and the relationship to surrounding anatomical structures (2). Magnetic resonance imaging (MRI) offers supplementary value, especially for evaluating neurogenic tumors and complex lesions due to superior soft-tissue resolution. Ultrasound may also assist in lesion localization and cystic characterization, particularly in younger children or when radiation exposure was a concern. Comprehensive analysis integrating imaging characteristics, demographic features, and laboratory findings enhances diagnostic accuracy (2–4).
Surgery continues to be the cornerstone of treatment for children with mediastinal masses (5, 6). In recent years, minimally invasive thoracoscopic surgery has become mainstream due to advancements in pediatric surgical techniques. However, it is critical to acknowledge that surgical risks in children significantly exceed those in adults, particularly in lesions causing tracheobronchial compression, where anesthetic induction may precipitate acute airway obstruction. This scenario necessitates meticulous preoperative evaluation and prompt intraoperative interventions. Airway compression exceeding 50% on CT imaging has been associated with a significantly increased risk of intraoperative ventilation failure (7). Additionally, bronchogenic cysts require special attention since direct intraoperative traction or dissection may cause cyst rupture, resulting in severe airway obstruction. This observation has been reported in previous clinicopathological studies and must be factored into perioperative planning (8). Thus, comprehensive preoperative assessment and intraoperative vigilance are imperative.
Furthermore, current evidence on mediastinal masses in children was predominantly derived from single-center case series with limited long-term follow-up data. This restricts the generalizability of conclusions regarding recurrence risk and functional outcomes. To address these knowledge gaps, we conducted a single-center retrospective study analyzing clinical data from 51 children diagnosed with mediastinal masses and treated at the Affiliated Hospital of Zunyi Medical University between January 2019 and August 2024. We systematically review clinical characteristics, imaging findings, pathological profiles, and surgical strategies, supplemented by detailed analyses of representative high-risk cases. Comparative insights are also offered between benign and malignant tumors and between thoracoscopic and open surgical approaches. Particular emphasis was placed on preoperative airway evaluation, optimization of intraoperative procedures, and emergency perioperative ventilation management. Our aim was to provide clinicians with comprehensive guidance to improve early recognition, diagnostic precision, and therapeutic safety for children with mediastinal masses.
2 Methods
2.1 Study design and ethical approval
This was a single-center, retrospective observational study aiming to systematically analyze the clinical characteristics, imaging features, pathological profiles, and surgical management of children diagnosed with mediastinal masses. Participants were children who underwent surgical treatment for mediastinal masses at the Department of Pediatric Surgery, Affiliated Hospital of Zunyi Medical University, from January 2019 to August 2024.
The study protocol was approved by the Ethics Committee of the Affiliated Hospital of Zunyi Medical University (Approval Number: KLL-2024-706), and strictly adhered to the principles outlined in the Declaration of Helsinki. Written informed consent was obtained from all legal guardians prior to surgery.
2.2 Inclusion and exclusion criteria
Inclusion criteria: 1. Children aged 0–14 years; 2. Confirmed preoperative imaging diagnosis of mediastinal mass; 3. Surgical treatment performed at our hospital with complete pathological diagnosis; 4. Comprehensive clinical records available, including symptoms, physical examination findings, imaging data, surgical records, and follow-up outcomes.
Exclusion criteria: 1. Non-primary mediastinal origin of lesions; 2. Conservative treatment without surgery; 3. Incomplete imaging or pathological data; 4. Lack of definite postoperative pathological diagnosis.
2.3 Data collection
Data were retrospectively extracted from the hospital's electronic medical records and surgical databases. Collected information included basic demographic data (gender, age), initial clinical symptoms and physical signs, imaging modalities used and their findings (x-ray, CT, MRI, ultrasound), anatomical location of masses, surgical techniques employed, special intraoperative interventions (e.g., sclerosing agent injection, iodine-based cyst wall treatment, cyst fluid decompression via aspiration), pathological results, postoperative management (including transfer to oncology departments), and follow-up outcomes. Data were independently collected and cross-verified by two investigators, with challenging cases reviewed by a third-party clinical expert. Selected CT images of children with mediastinal masses are shown below (Figure 1).
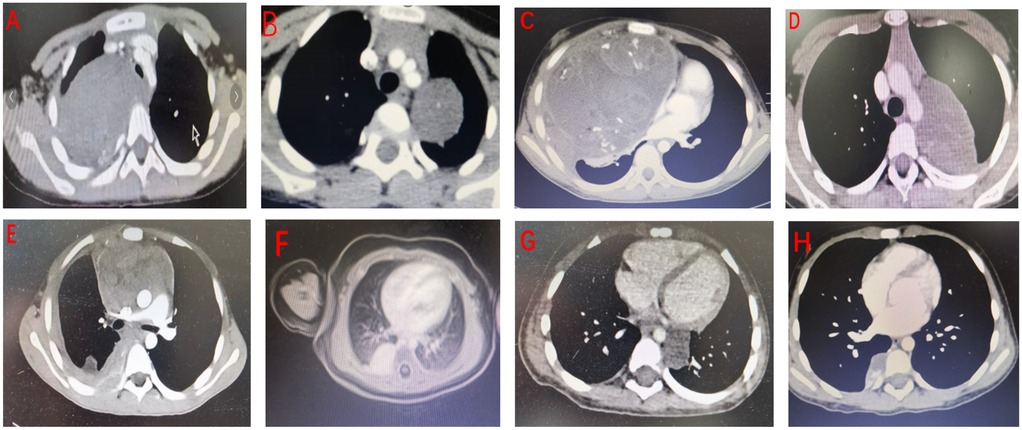
Figure 1. Selected CT images of mediastinal masses from representative patients. Postoperative pathological diagnoses are as follows: (A) ganglioneuroma; (B) ganglioneuroma; (C) mixed germ cell tumor (yolk sac tumor and mature teratoma); (D) schwannoma; (E) lymphoma; (F) hemangioma; (G) esophageal duplication cyst; (H) extralobar pulmonary sequestration.
2.4 Imaging and pathological classification
Mediastinal compartments were categorized into anterior, middle, and posterior regions based on standardized radiological anatomical landmarks. All imaging data were independently reviewed by two senior radiologists (associate chief physician or higher). Disagreements were adjudicated by a third radiologist. Final pathological diagnoses were made by two experienced pathologists according to the latest World Health Organization (WHO) tumor classification system and categorized as benign or malignant.
2.5 Preoperative evaluation and perioperative management
All children underwent preoperative contrast-enhanced CT to assess the location, size, and extent of the mediastinal mass, as well as its relationship to adjacent structures including the trachea, bronchi, heart, and major vessels. In cases where CT imaging revealed airway narrowing, the degree of tracheobronchial compression was semi-quantitatively assessed and categorized as mild (<25%), moderate (25%–50%), or severe (>50%).
For children with moderate to severe airway compression, a multidisciplinary team comprising pediatric surgeons, anesthesiologists, and thoracic specialists jointly developed an individualized perioperative plan. Key elements included:
1. Preserving spontaneous ventilation during anesthetic induction whenever feasible;
2. Availability of advanced airway equipment such as fiberoptic bronchoscopes and video laryngoscopes;
3. Adjustments in intraoperative positioning to relieve airway compression;
4. Implementation of pre-formulated emergency ventilation protocols.
One child experienced acute ventilation failure during induction, attributed to >60% tracheal compression. Emergency oral-to-endotracheal tube insufflation temporarily restored oxygenation, followed by intraoperative repositioning to the prone position, which restored airway patency and enabled safe surgical continuation. This case highlights the importance of incorporating preoperative airway evaluation and individualized intraoperative strategies in high-risk children.
Particular caution was exercised for bronchogenic cysts, given their frequent anatomical proximity to airway structures. Excessive tension on the cyst wall during dissection posed a high risk of rupture and subsequent obstruction (9). As a preventative measure, all bronchogenic cysts underwent controlled aspiration decompression using an 18G needle under direct vision prior to resection (Figure 2).
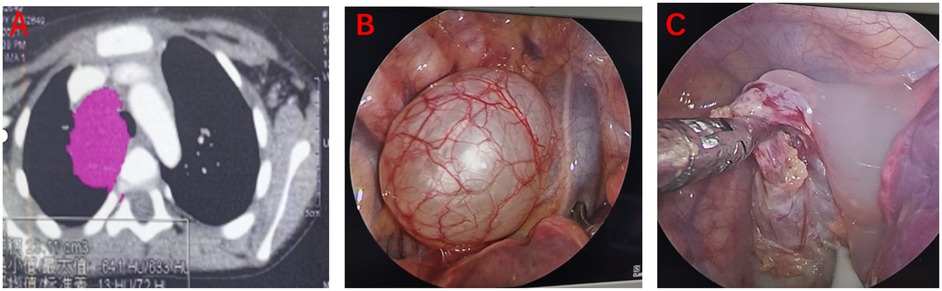
Figure 2. Bronchogenic cyst. (A) Preoperative CT scan; (B) Intraoperative view of the intact cyst; (C) Cystic fluid after decompression.
For selected cystic lesions (n = 16), the residual cyst wall was gently irrigated intraoperatively with 4% tincture of iodine, followed by thorough rinsing with normal saline. This was performed to ablate the secretory function of the cyst epithelium and thereby reduce the risk of recurrence. The technique was adapted from the method reported by Feng et al. (10), in which the inner surface of the cyst was cauterized using 4% iodine tincture, demonstrating favorable efficacy in minimizing postoperative recurrence.
2.6 Statistical analysis
All data were entered into Microsoft Excel 2021 and independently cross-verified. Descriptive statistical analyses were performed using SPSS version 29.0. Categorical variables are presented as frequencies and percentages, while continuous variables are reported as mean ± standard deviation or as medians with interquartile ranges, depending on their distribution.
Given the retrospective and descriptive nature of the study, no inferential statistical tests or multivariable modeling were applied. However, subgroup comparisons—such as between children with benign vs. malignant tumors, or between those who underwent thoracoscopic vs. open surgical approaches—were conducted descriptively to enhance clinical interpretability. All results are summarized using appropriate tables and figures.
3 Results
3.1 Patient demographics and hospitalization characteristics
A total of 51 children diagnosed with mediastinal masses and surgically treated at the Affiliated Hospital of Zunyi Medical University between January 2019 and August 2024 were included in this study. All children had histopathologically confirmed diagnoses. The cohort consisted of 29 males and 22 females (male-to-female ratio, 1.3:1), with a median age of 5.0 years (range: 0.5–12 years). Most cases were managed by the Departments of Pediatric Surgery and Thoracic Surgery, with an average hospitalization duration of 8.3 days (Table 1).
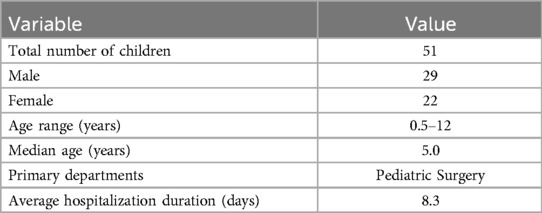
Table 1. Demographic distribution and hospitalization duration among children with mediastinal masses.
3.2 Clinical presentations
The most common presenting symptoms were exertional dyspnea (54.9%) and chronic cough (49.0%), followed by sputum production, fever, and chest pain. Notably, 9.8% of children were asymptomatic, with lesions detected incidentally during routine physical examinations (Table 2).
3.3 Pathological subtypes and anatomical distribution
Histopathologically, benign lesions comprised the majority (84.3%, 43/51). The most common diagnosis was ganglioneuroma (11 cases), predominantly located in the posterior mediastinum. Eight cases (15.7%) were malignant, including neuroblastomas, lymphomas, and germ cell tumors, with a predilection for the anterior mediastinum (Table 3).
3.4 Imaging and surgical management
All children underwent CT imaging, which served as the principal diagnostic tool. Chest x-rays initially identified mediastinal abnormalities in 19 cases, while MRI was employed in 12 children to assess tumor invasiveness, and ultrasound guided localization or aspiration in 8. Minimally invasive thoracoscopic surgery was performed in 88.2% (45/51), while 6 children required open thoracotomy due to tumor size, complex anatomical location, or suspected malignancy (Table 4).
3.5 Intraoperative risk management
Among children with malignant tumors, thoracoscopic resection was feasible in 5 out of 8 cases (62.5%), whereas thoracoscopy was applied in 95.1% (39/41) of those with benign tumors. Malignant tumors were associated with greater intraoperative complexity and longer hospitalization (median stay 10.1 vs. 7.4 days).
Airway compression severity was graded based on CT. Nine children exhibited moderate-to-severe narrowing (compression >25%). One child with 63% tracheal compression experienced acute airway collapse during anesthesia induction. Emergency oral-to-endotracheal tube insufflation, followed by repositioning to the prone position, restored ventilation and permitted completion of surgery without conversion.
3.6 Cyst-specific surgical techniques
Bronchogenic cysts presented a high risk of rupture. All such cases underwent intraoperative decompression via fluid aspiration. Additionally, iodine-based cyst wall irrigation was performed in 16 cases, and sclerotherapy was used in 4 lymphangiomas to reduce recurrence risk.
3.7 Follow-up outcomes
Follow-up data were available for all children with benign lesions. Follow-up duration ranged from 3–36 months (median: 17 months). No recurrences or functional complications were observed. All 10 children with malignant tumors were referred to oncology for postoperative adjuvant therapy.
3.8 Conclusions
This study demonstrates that mediastinal masses in children are predominantly benign and typically present in preschool-aged individuals, with a slight male predominance. Respiratory symptoms, particularly dyspnea on exertion and chronic cough, are the most common presenting features.
CT remains the gold standard for diagnosis and surgical planning, while MRI and ultrasound provide valuable adjunctive information in selected cases. Minimally invasive thoracoscopic surgery was safe and effective for the majority of children, though open thoracotomy remains essential in anatomically complex or malignant lesions.
Airway compression risk stratification using CT was crucial for pre-anesthesia evaluation, and intraoperative airway management, including prone positioning and manual ventilation strategies, should be preemptively prepared. Targeted intraoperative management of cystic lesions, such as decompression and chemical irrigation, can mitigate complication and recurrence risks.
Although long-term outcome data are pending, mid-term follow-up shows excellent prognosis for children with benign lesions, while comprehensive multidisciplinary care remains essential for those with malignant tumors. Prospective multicenter studies are warranted to validate these findings and enhance clinical guidelines (Table 5).
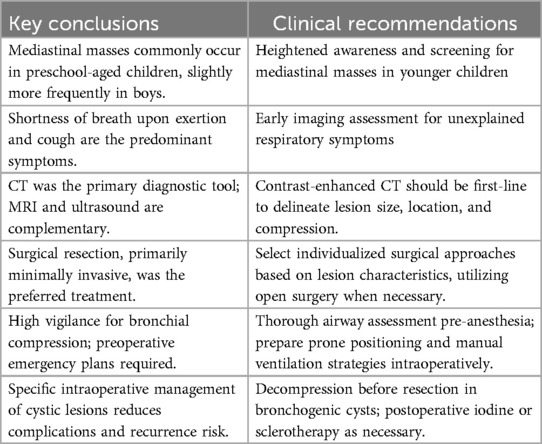
Table 5. Summary of clinical conclusions and practice recommendations for pediatric mediastinal tumors.
4 Discussion
Mediastinal tumors in children, occurring within the central thoracic cavity, present a unique clinical challenge due to their propensity to compress vital mediastinal structures such as the trachea, heart, esophagus, and great vessels (11, 12). While open thoracotomy has historically been the standard approach, the advent of minimally invasive thoracoscopic techniques has significantly broadened surgical options for this population (13). The mediastinum's embryologic complexity, derived from all three germ layers, gives rise to a wide variety of tumor types, most of which are benign (14).
Mediastinal lesions are anatomically classified into anterior, middle, and posterior compartments. Anterior tumors typically include thymic, germ cell, and lymphoid neoplasms; middle compartment lesions are often congenital cysts or vascular anomalies; and posterior tumors predominantly comprise neurogenic neoplasms (15–17). Bronchogenic cysts and esophageal duplication cysts, while relatively uncommon, originate from abnormal foregut budding and account for 13%–15% of congenital thoracic lesions (18).
In children, surgical management must account for narrower airways, limited cardiopulmonary reserve, and anatomical variability. These features necessitate a refined approach to preoperative risk assessment and intraoperative planning. Our findings align with established imaging-based compartmental frameworks (4, 7), emphasizing the importance of vigilant airway evaluation, particularly in anterior and middle mediastinal tumors.
4.1 Tumor distribution and age-dependent histopathology
In our cohort, benign tumors comprised 84.3% of cases, with ganglioneuromas (21.6%) and bronchogenic cysts (13.7%) most prevalent. These lesions were mainly located in the posterior and middle mediastinum. Malignant lesions, including lymphomas and neuroblastomas, represented 15.7% of cases and were predominantly anterior. This compartment-specific distribution reflects developmental origins and was consistent with prior pediatric series (1).
Among posterior mediastinal neurogenic tumors, schwannomas were characterized by slow growth and encapsulation (19), while neurofibromas occurred either as isolated lesions or in association with neurofibromatosis type 1 (20, 21). Ganglioneuromas, derived from sympathetic ganglia, remained the most common and least aggressive.
4.2 Imaging modalities are essential due to nonspecific symptoms
Most children presented with nonspecific symptoms such as exertional dyspnea or chronic cough, which often mimicked upper respiratory conditions. In 9.8% of cases, tumors were detected incidentally during routine evaluations, underscoring the importance of thorough screening and diagnostic imaging.
Computed tomography (CT) achieved 100% diagnostic sensitivity in this study and remains the primary tool for defining tumor location, morphology, and relationship to adjacent structures. Magnetic resonance imaging (MRI) added value in evaluating paraspinal extension or vascular involvement. Ultrasonography was also beneficial, particularly in younger children, offering radiation-free guidance during cyst aspiration or localization.
4.3 Surgical treatment was effective with individualized intraoperative strategies
All children underwent complete surgical resection, with thoracoscopic approaches employed in 88.2% of cases. Minimally invasive surgery was feasible in 95.1% of benign cases, but only 62.5% of malignant cases, likely due to size, invasion, or anatomical constraints. Children undergoing thoracotomy had a longer median hospitalization (10.1 vs. 7.4 days), indicating greater perioperative burden.
Intraoperative strategies were adapted to tumor characteristics. In the management of cystic lesions, we employed an intraoperative strategy involving irrigation of the residual cyst wall with 4% tincture of iodine. This approach aimed to chemically ablate the secretory function of the cyst epithelium and thereby reduce the risk of postoperative recurrence. The irrigation was performed gently, followed by thorough rinsing with normal saline, which enhanced the local therapeutic effect while minimizing chemical irritation to surrounding tissues. Among the 16 children who received this treatment, no recurrence was observed during the follow-up period. These findings suggest that this intraoperative adjunctive technique was both feasible and safe, and may be worthy of further application in similar cases (10). For lymphangiomas, intraoperative sclerotherapy further minimized recurrence risk.
Bronchogenic cysts posed a notable airway hazard. To prevent rupture-induced airway compromise, we performed direct-vision decompression using an 18G needle prior to dissection, a strategy consistent with Limaïem et al. (9). This approach proved effective in avoiding intraoperative airway emergencies.
4.4 High vigilance for airway compression was essential
In one case, tracheal compression >60% led to acute ventilation failure upon anesthetic induction. Emergency manual ventilation via oral endotracheal insufflation, followed by repositioning the child into the prone position, immediately restored airway patency and allowed safe surgical continuation.
CT imaging enabled semi-quantitative grading of airway narrowing: mild (<25%), moderate (25%–50%), or severe (>50%). This grading informed anesthetic strategy. The critical case with 63% compression supports previous findings that compression beyond 50% markedly increases intraoperative respiratory risk (2).
For high-risk children, we coordinated multidisciplinary planning, preserved spontaneous respiration during induction, ensured advanced airway equipment availability, and rehearsed emergency repositioning protocols. These precautions were instrumental in preventing catastrophic outcomes.
4.5 Strengths and limitations
This study was among the few to integrate anatomical location, imaging severity, pathological subtype, and intraoperative technique into a structured perioperative framework for mediastinal tumors in children. The inclusion of real-world high-risk cases reinforced the clinical value of individualized approaches.
Nonetheless, limitations include its retrospective single-center design and limited sample size, which may introduce selection bias. Long-term data remain incomplete; however, mid-term follow-up (3–36 months) of 41 children with benign tumors showed no recurrence or functional sequelae. Five-year outcome tracking is ongoing. Additionally, lack of multivariate analysis limits causal interpretation.
Future prospective, multicenter studies are needed to confirm our findings and further optimize perioperative management protocols.
5 Conclusion
Mediastinal tumors in children are primarily benign but can pose significant perioperative risks, especially when associated with airway compression. CT remains the diagnostic cornerstone, guiding surgical and anesthetic planning. Tailored intraoperative strategies—such as cyst decompression, chemical sclerotherapy, and dynamic positioning—can enhance safety and effectiveness. A multidisciplinary, anatomy-driven approach was essential in achieving optimal outcomes in our pediatric mediastinal surgery cases.
Data availability statement
The original contributions presented in the study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding author.
Ethics statement
The studies involving humans were approved by Ethics Committee of the Affiliated Hospital of Zunyi Medical University. The studies were conducted in accordance with the local legislation and institutional requirements. Written informed consent for participation in this study was provided by the participants' legal guardians/next of kin. Written informed consent was obtained from the individual(s), and minor(s)' legal guardian/next of kin, for the publication of any potentially identifiable images or data included in this article.
Author contributions
XS: Funding acquisition, Resources, Supervision, Writing – review & editing, Methodology, Software, Writing – original draft, Investigation, Formal analysis, Visualization. YuL: Investigation, Visualization, Conceptualization, Formal analysis, Methodology, Data curation, Writing – review & editing. ZL: Writing – original draft, Validation, Funding acquisition, Conceptualization, Methodology. YiL: Writing – original draft, Formal analysis, Supervision, Visualization, Investigation. GZ: Data curation, Writing – original draft, Project administration, Visualization, Software. HT: Methodology, Writing – original draft, Resources, Project administration, Investigation. KM: Supervision, Software, Methodology, Investigation, Writing – original draft.
Funding
The author(s) declare that financial support was received for the research and/or publication of this article. This work was supported by Guizhou Provincial Health Commission [gzwkj2024-431], Guizhou Science and Technology Cooperation Achievement QKH-LC-[2025]-142 and Guizhou Provincial Administration of Traditional Chinese Medicine [QZYY-2024-015]. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Bourgouin PP, Madan R. Imaging of the middle and visceral mediastinum. Radiol Clin North Am. (2021) 59(2):193–204. doi: 10.1016/j.rcl.2020.11.004
2. Zhang Y. Analysis of different CT signs in the differential diagnosis of thymoma and other common malignant mediastinal tumors. Imaging Res Med Appl (2022) 6:50–2.
3. Li J, Xiang G, Zhang J, Jia C, Chang T, Ruan Z, et al. Interpretation of the 2023 version of the NCCN clinical practice guidelines for thymoma and thymic carcinoma. Chin J Clin Thorac Cardiovasc Surg. (2023) 30(04):506–13.
4. Nakazono T, Yamaguchi K, Egashira R, Takase Y, Nojiri J, Mizuguchi M, et al. CT-based mediastinal compartment classifications and differential diagnosis of mediastinal tumors. Jpn J Radiol. (2019) 37(2):117–34. doi: 10.1007/s11604-018-0777-5
5. Ren Z, Ren X, Zhang Y. Observation of short-term clinical efficacy of single-port thoracoscopic surgery for thymoma. Chin J Thorac Surg (Electronic Edition). (2018) 5:230–4.
6. Pan Y, Song Y, Liu L. Clinical analysis of thoracoscopic treatment for anterior mediastinal tumors via the subxiphoid approach in the scissors position versus the lateral thoracic approach in the lateral decubitus position. Chin J Clin Thorac Cardiovasc Surg. (2020) 27:1172–6.
7. Hack HA, Wright NB, Wynn RF. The anaesthetic management of children with anterior mediastinal masses. Anaesthesia. (2008) 63(8):837–46. doi: 10.1111/j.1365-2044.2008.05515.x
8. Nakazono T, Yamaguchi K, Egashira R, Yamasaki F, Fujita M, Hayashi T, et al. Anterior mediastinal lesions: cT and MRI features and differential diagnosis. Jpn J Radiol. (2021) 39:67–81. doi: 10.1007/s11604-020-01031-2
9. Limaïem F, Ayadi-Kaddour A, Djilani H, Kilani T, El Mezni F. Pulmonary and mediastinal bronchogenic cysts: a clinicopathologic study of 33 cases. Lung. (2008) 186:55–61. doi: 10.1007/s00408-007-9056-4
10. Wang X, Chen KZ, Li Y, Liu J, Wang J. Diagnosis and video-assisted thoracoscopic surgical treatment of mediastinal bronchogenic cysts. Chin J Clin Thorac Cardiovasc Surg. (2019) 26(9):848–52. doi: 10.7507/1007-4848.201811026
11. Azizkhan RG, Dudgeon DL, Buck JR, Colombani PM, Yaster M, Nichols D, et al. Life-threatening airway obstruction as a complication to the management of mediastinal masses in children. J Pediatr Surg. (1985) 20(6):816–22. doi: 10.1016/s0022-3468(85)80049-x
12. Brillantino C, Rossi E, Tambaro FP, Minelli R, Bignardi E, Cremone G, et al. Clinical and imaging findings useful in the differential diagnosis of most common childhood mediastinal tumors. Trans Med. (2019) 9:207.
13. Saltsman JA, Danzer E, Hammond WJ, Rhee D, Berhe S, Monteagudo J, et al. Survival and scoliosis following resection of chest wall tumors in children and adolescents: a single-center retrospective analysis. Ann Surg. (2021) 274(2):e167–73. doi: 10.1097/SLA.0000000000003495
14. Yuan Z, Lu Z. Clinical value of CT imaging in the diagnosis of thoracic tumors. Imaging Res Med Appl. (2021) 5:124–5. doi: 10.3969/j.issn.2096-3807.2021.19.059
15. Takeda S, Miyoshi S, Minami M, Ohta M, Masaoka A, Matsuda H. Clinical spectrum of mediastinal cysts. Chest. (2003) 124(1):125–32. doi: 10.1378/chest.124.1.125
16. Sumiyoshi K, Shimizu S, Enjoji M, Iwashita A, Kawakami K. Bronchogenic cyst in the abdomen. Virchows Arch A Pathol Anat Histopathol. (1985) 408(1):93–8. doi: 10.1007/BF00739965
17. Dubrava J, Koren J, Pospisilova V. Mediastinal foregut duplication cyst of enteric type containing a persistent thymus, imitating a pericardial cyst. Bratisl Lek Listy. (2013) 114(8):480–3. doi: 10.4149/bll_2013_100
18. Mampilly T, Kurian R, Shenai A. Bronchogenic cyst -cause of refractory wheezing in infancy. Indian J Pediatr. (2005) 72(4):363–4. doi: 10.1007/BF02724023
19. Ramos AC, Pintado HB, Allende MC. Endoscopic management of tracheal neurofibroma. Arch Bronconeumol (Engl Ed). (2020) 56(2):116. doi: 10.1016/j.arbres.2019.03.003
20. Temelkova I, Tchernev G. Giant pelvic neurofibroma in patient with plexiform sciatic neurofibroma and neurofibromatosis type 1. Open Access Maced J Med Sci. (2019) 7(8):1346–9. doi: 10.3889/oamjms.2019.304
Keywords: children, mediastinal mass, imaging examination, pathological type, surgical treatment, bronchial compression, ventilation management, perioperative safety
Citation: Shang X, Liu Y, Luo Z, Li Y, Zhou G, Tan H and Mao K (2025) Clinical features and surgical treatment of mediastinal masses in children: a retrospective study of 51 cases. Front. Pediatr. 13:1608867. doi: 10.3389/fped.2025.1608867
Received: 9 April 2025; Accepted: 7 August 2025;
Published: 20 August 2025.
Edited by:
Nikolaos Zavras, University General Hospital Attikon, GreeceReviewed by:
Panagiotis Nikolinakos, Chelsea and Westminster Hospital NHS Foundation Trust, United KingdomΕlissavet Anestiadou, G. Papanikolaou General Hospital, Greece
Kimura Daisuke, Hirosaki University, Japan
Copyright: © 2025 Shang, Liu, Luo, Li, Zhou, Tan and Mao. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Yuanmei Liu, eXVhbm1laTExNkBhbGl5dW4uY29t
 Xianhui Shang
Xianhui Shang Yuanmei Liu
Yuanmei Liu Zhen Luo
Zhen Luo Yingbo Li1,2
Yingbo Li1,2 Kaiyi Mao
Kaiyi Mao