- 1Department of Neurosurgery, Beijing Children’s Hospital, National Center for Children’s Health, Capital Medical University, Beijing, China
- 2Department of Neurosurgery, The First Affiliated Hospital of Zhengzhou University, Zhengzhou, China
- 3Department of Neurosurgery, Guangdong Provincial People’s Hospital (Guangdong Academy of Medical Sciences), Southern Medical University, Guangzhou, China
The blood–brain barrier (BBB) plays a vital role in maintaining central nervous system homeostasis but poses a major obstacle to effective drug delivery in pediatric brain tumors. BBB integrity varies significantly in pediatric brain tumors compared to adult ones, and is influenced by the tumor type, molecular subtype, and anatomical location. This review discusses the heterogeneous nature of the BBB across various pediatric brain tumors, including low-grade gliomas, diffuse midline gliomas, medulloblastomas, ependymomas and craniopharyngiomas. We review histological, molecular, and imaging evidence to highlight differences in BBB permeability and their implications for therapeutic delivery and treatment resistance. Special consideration is given to advanced drug delivery strategies, such as focused ultrasound and BBB-disrupting agents, which have been tailored to the unique barrier properties of each tumor subtype. A deeper understanding of tumor-specific BBB architecture is essential for tailoring treatment strategies and improving outcomes in pediatric brain cancer.
Introduction
Pediatric brain tumors are among the most common solid tumors in children and are a leading cause of cancer-related mortality in this population worldwide (1–4). In the United States, central nervous system (CNS) tumors rank the second most common cancer, second only to leukemia, in children aged 0–19 years; however, their mortality rate has surpassed that of leukemia, making CNS tumors the leading cause of cancer-related death in children (3). Approximately 80%–90% of the newly diagnosed pediatric brain tumor cases each year occur in low- and middle-income countries, which together account for approximately 88% of the global pediatric population (5). Studies in China have demonstrated significant regional differences in the annual incidence of pediatric brain tumors (6). Moreover, the peak age for pediatric brain tumors is between 0 and 14 years (7), and some studies have reported a slight male predominance, with a male-to-female ratio of approximately 1.2:1 (8). Common pediatric brain tumor types include low-grade gliomas (accounting for approximately 30% of all gliomas) (9), medulloblastomas (10%–15.2%), ependymomas, craniopharyngiomas, and diffuse midline gliomas, with approximately half of these tumors exhibiting malignant features (10). Anatomically, the most common sites of occurrence are the posterior fossa, ventricular system, and supratentorial regions (10).
The microvasculature of the CNS, collectively termed the blood–brain barrier (BBB), tightly controls the passage of ions, molecules, and cells into the CNS to maintain homeostasis within this evolutionarily conserved region. Drug penetration across the BBB is impacted by factors such as molecular size, lipid solubility, and efflux transporters (11). With respect to the molecular size, most publications report an absolute cutoff of 400–600 Da (12), and the largest substance reported to cross the BBB to date had a molecular weight of 7.8 kDa (13). BBB development occurs synchronously with CNS angiogenesis, and its structure and function support the metabolic demands of neurons and glial cells (14). During development, the brain vasculature acquires a series of specialized molecular and cellular properties, such as tight junction proteins and efflux transporters, which collectively form the BBB and limit passive diffusion between the blood and brain parenchyma (14, 15).
In pediatric brain tumors, the BBB often undergoes structural and functional alterations, forming a blood–brain tumor barrier whose degree of disruption exhibits marked heterogeneity both across different tumor types and among different regions within the same tumor. This heterogeneity likely plays a crucial role in the permeability and therapeutic efficacy of drugs. Moreover, the unique developmental state of the pediatric BBB may further restrict the permeation of therapeutic agents, especially large molecules, such as monoclonal antibodies (mAbs), thereby influencing treatment outcomes (16, 17). To overcome these limitations, recent research efforts have focused on the exploration of strategies such as the use of nanoparticle carriers and BBB-penetrating peptides (e.g., BBB-modulating peptides) to increase drug delivery efficiency; however, these approaches must carefully consider differences in metabolism and sensitivity to toxic substances inherent to the developing pediatric brain (18). Existing BBB models, including stem cell-derived models and microfluidic chip systems, are predominantly based on adult tissue data and lack the capacity to simulate pediatric brain physiology accurately (19). Therefore, there is an urgent need to establish in vitro models that more closely mimic the physiological state of the developing brain, enabling deeper investigations into the dynamic changes in the BBB of pediatric brain tumors (20).
A comprehensive understanding of BBB characteristics in pediatric brain tumors is critical for optimizing therapeutic strategies, improving targeted drug delivery efficiency, and ultimately improving patient outcomes. This review focuses on the features of the BBB in several common pediatric brain tumors (21), with the aim of providing a theoretical foundation and research directions for their precise treatment in the future.
Low-grade gliomas
Pediatric low-grade gliomas (pLGGs) and glioneuronal tumors represent the most common brain tumors, accounting for nearly 30% of pediatric CNS neoplasms. pLGGs are defined as grade 1 or 2 per the recent World Health Organization (WHO) 2021 classification (22). Overall, the BBB function in pLGGs remains relatively intact, with tumor cells causing minimal disruption. This BBB preservation has been observed in magnetic resonance imaging (MRI) examinations. Typically, low-grade gliomas do not exhibit marked contrast enhancement, which distinguishes them from high-grade gliomas. The relatively normal vascular architecture of low-grade gliomas in children contributes to the maintenance of BBB integrity (Figure 1). This integrity protects the brain tissue around the tumor from harmful substances that are secreted by tumor cells, limiting the rapid growth and metastasis of the tumor. According to the report by Hong CS et al. (23), an abundant glial fibrillary acidic protein (GFAP) signal was observed in pediatric pilocytic astrocytoma, which was consistent with the astrocytic lineage of the tumor cells. Aquaporin-4 (AQP4), a marker of astrocytic end-foot processes, has been widely used as a reliable indicator of the structural and functional integrity of astrocyte–endothelial cell interactions (24). AQP4 expression was reduced and exhibited a disorganized pattern within the pilocytic astrocytoma tumor tissue, suggesting that the tumor cells were unable to fulfill their physiological role in supporting the BBB, thereby facilitating increased permeability for therapeutic agents to access the lesion from the vasculature. In low-grade diffuse astrocytoma, immunofluorescence staining revealed an intact BBB, which was characterized by strong GFAP and AQP4 signals surrounding the microvasculature. Notably, AQP4 staining was sufficiently intense to delineate the contours of CD31-positive vessels independently (23). On the basis of data from animal models, Tan J et al. suggested that the use of an I6P7 peptide-mediated MRI probe could be a potential strategy for overcoming the BBB integrity and diagnosing low-grade gliomas (25). The integrity of the BBB remains a major obstacle for conventional chemotherapeutic agents to reach the tumor parenchyma. However, focal BBB disruptions may create opportunities for the use of targeted therapies and advanced drug delivery systems. Recently, focused ultrasound (FUS) combined with microbubble technology has been employed as a noninvasive and reversible method to locally open the BBB, thereby enhancing the penetration of antitumor drugs into pLGGs. Preliminary studies have shown promising results (26, 27). In terms of surgical treatment, sodium fluorescein is among the ideal candidates as an intraoperative marker for actual recognition of tumor extension, since it accumulates in areas with an altered blood–brain barrier, a typical characteristic of pediatric gliomas, and has a low rate of adverse events, which greatly facilitates complete surgical resection (28). To better model and understand the complex BBB status in pLGGs, researchers have utilized human induced pluripotent stem cells (hiPSCs) to develop subtype-specific in vitro models. These models can reflect the interactions between different molecular subtypes of pLGGs and the BBB, providing new technical avenues for personalized drug screening and preclinical platform development (29, 30), However, they are limited by the absence of immune system components and a lack of long-term functional stability.
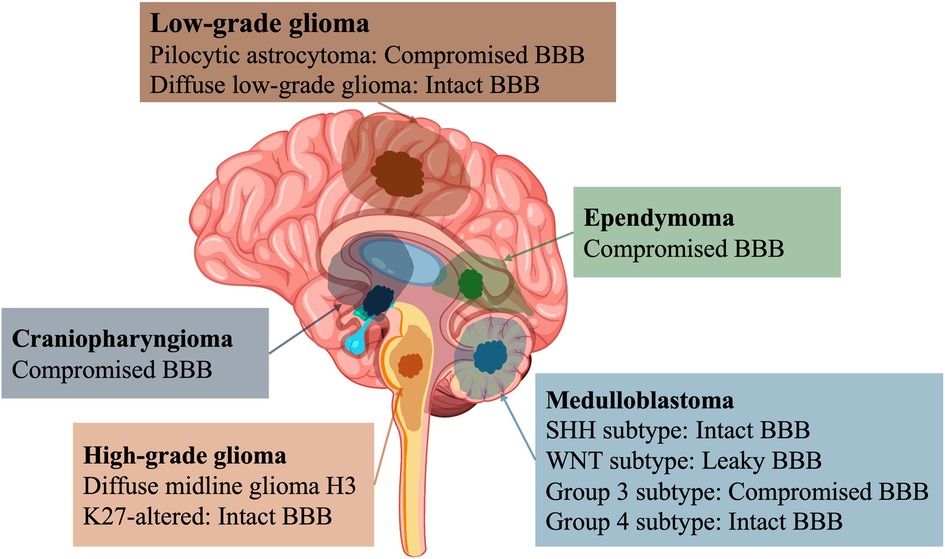
Figure 1. Localization and BBB characteristics in pediatric brain tumors. BBB, blood–brain barrier; SHH, sonic hedgehog; WNT, wingless.
High-grade gliomas
High-grade gliomas account for approximately 10% of CNS tumors in children and are the leading cause of cancer-related death in individuals under 19 years of age. According to the 2021 WHO classification, pediatric diffuse high-grade gliomas are categorized into four distinct subgroups, with diffuse midline glioma (DMG), H3 K27-altered, being the most common subtype (21). DMG is a highly infiltrative and aggressive pediatric brain tumor, with a median survival of approximately nine months following diagnosis. These tumors arise in midline structures of the central nervous system, most commonly in the pons, where they are referred to as diffuse intrinsic pontine gliomas (DIPGs), as well as in the thalamus or spinal cord. More than 80% of DMG cases harbor a specific lysine-to-methionine substitution at position 27 (K27M) in histone H3 variants, occurring either in the canonical H3C1/H3C2 genes (H3.1-K27M) or in the alternative histone H3 variant H3-3A (H3.3-K27M) (31). MRI with a gadolinium-based contrast agent is the most commonly employed method for assessing BBB disruption. Unlike certain CNS tumors that exhibit a compromised BBB, DMG is characterized by a relatively intact BBB (Figure 1) (32–34). However, heterogeneity within the tumor's blood–tumor barrier can result in focal areas of increased permeability. In contrast to many other malignant CNS tumors, the vasculature within DMGs generally retains BBB integrity (35–39). This preservation is largely maintained by tight junction proteins and specialized interendothelial structures that constitute an effective barrier, limiting the penetration of large-molecule therapeutics into the tumor microenvironment. Claudin-3, a key tight junction protein, is known to be overexpressed in ovarian cancer cells, where its upregulation is associated with a reduction in the repressive histone mark H3K27me3 at its promoter region. Similarly, histone mutations that lead to decreased H3K27me3 may contribute to aberrant upregulation of Claudin-3 expression in DMG (40). As a result, the intact BBB presents a major challenge to the delivery and efficacy of molecularly targeted therapies and chemotherapeutic agents (36, 37, 41). Notably, a study by McCully et al. demonstrated that the BBB is not uniformly impermeable; rather, it is heterogeneous, as evidenced by the differential penetration of temozolomide between the brainstem and cortical regions (42). To date, more than 250 DMG-targeting clinical trials have failed to overcome the BBB limitation, resulting in persistently high treatment failure rates (38). At the molecular level, the hallmark genetic alteration in DMG, the H3 K27M mutation, causes global hypomethylation of histone H3, potentially regulating the expression of BBB-related genes through epigenetic mechanisms and thus influencing the dynamic functionality of the barrier (43–45). In an in vitro study that investigated the DIPG-specific BBB, Deligne et al. reported that over the course of one week of DIPG cell development, there was no significant change in either the permeability of the barrier to chemotherapeutic agents or the expression levels of efflux transporters (34), which underscores the complex and multifactorial nature of chemoresistance in DMGs (34). Imaging studies using diffusion-weighted imaging (DWI) and apparent diffusion coefficient (ADC) analysis have revealed significant differences in ADC values between H3 K27M-mutant DMGs and other midline gliomas, with these parameters potentially serving as indirect indicators of the BBB functional status and tumor microenvironment characteristics (46, 47). With respect to treatment, small molecules such as ONC201 (dordaviprone) or epigenetic agents demonstrate relatively good BBB penetration and have shown preliminary efficacy in some patients with the H3 K27M mutation, making these agents a current focus of clinical research (48). Several studies have reported the expression of efflux transporters, such as P-glycoprotein/MDR1 (P-gp) and breast cancer resistance protein 1 (BCRP1), not only on the surface of DIPG cells but also within the surrounding tumor microenvironment (49). P-gp, in particular, contributes to reduced intracellular drug accumulation by actively exporting therapeutic agents in an ATP-dependent manner (50). Although CAR-T-cell therapy has shown some potential to penetrate the BBB, many immunotherapeutic agents, including immune checkpoint inhibitors, are poorly delivered to the tumor site (51, 52). Moreover, the tumor immune microenvironment in the DMG is generally immunosuppressive and, in combination with the intact BBB, further diminishes the efficacy of systemic therapies (53). Radiotherapy, as the current standard of care, can temporarily alleviate symptoms but does not effectively overcome BBB-related drug delivery barriers. However, its long-term efficacy and BBB modulation effects remain controversial (54). Various techniques that are aimed at bypassing or disrupting the BBB, such as convection-enhanced delivery (CED) and focused ultrasound, have demonstrated promising results. Clinical trials investigating techniques such as CED and FUS for targeted drug delivery to the brain are increasing in number (55, 56) In addition to enhancing drug penetration, FUS can transiently disrupt the BBB, enabling the release of tumor-derived biomarkers into the bloodstream, which may facilitate noninvasive monitoring of tumor progression and treatment response through liquid biopsy. Furthermore, FUS holds promise for delivering larger therapeutic molecules, such as monoclonal antibodies, to otherwise inaccessible brain regions. These approaches hold significant potential for overcoming the formidable challenge that the BBB presents to drug delivery to the central nervous system (35, 44). Future breakthroughs in DMG treatment may depend on effective modulation of the BBB combined with molecular-targeted and immunotherapeutic strategies.
Medulloblastoma
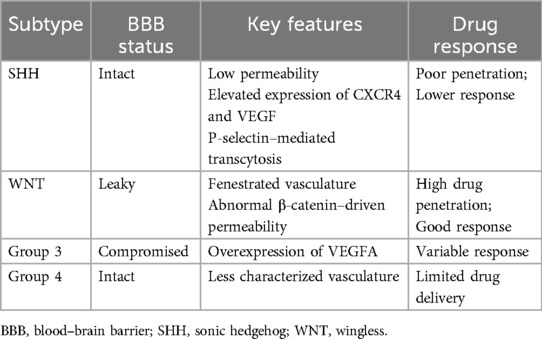
Medulloblastoma is one of the most common malignant CNS tumors in children. During tumor progression, the BBB undergoes pathological changes, resulting in a structurally and functionally compromised BBB. The extent of BBB disruption is significantly heterogeneous across different tumor regions and among molecular subtypes (57). Medulloblastomas are classified into Wingless (WNT), Sonic Hedgehog (SHH), Group 3, and Group 4 molecular subtypes, each with a distinct tumor microenvironment and vascular architecture. Notably, the SHH subtype displays specific vascular biological features and differs in the BBB morphology and permeability from the other subtypes (Table 1) (58). The SHH and Group 4 subtypes of medulloblastoma exhibit an intact BBB, whereas the Group 3 subtype shows mild BBB disruption. In contrast, in WNT-activated medulloblastoma, the BBB is notably leaky (Figure 1) (33, 59). Phoenix et al. demonstrated in genetically engineered mouse models that medulloblastomas driven by alterations in the WNT signaling pathway exhibited a compromised BBB, resulting in greater exposure to systemically administered chemotherapeutic agents and an enhanced tumor response (60). The WNT subgroup has an innately porous BBB, which is driven by abnormal β-catenin signaling and causes a fenestrated vasculature (60, 61). This intrinsic property likely enables a chemotherapy drug to pass into the tumor more easily. In contrast, medulloblastomas that are associated with alterations in the SHH pathway tend to maintain a more intact BBB, which renders them less responsive to chemotherapy (60). The SHH subtype also has upregulated angiogenesis pathway molecules, including chemokine receptor type 4 (CXCR4) and vascular endothelial growth factor (VEGF) (62, 63). Research conducted by Tylawsky DE et al. revealed that P-selectin-targeted nanocarriers induced active crossing of the blood‒brain barrier via caveolin-1-dependent transcytosis in the SHH subtype (64), demonstrating that even tumors with intact BBBs (e.g., SHH subtype) may be susceptible to targeted transcytosis-based drug delivery strategies. These preclinical findings align with clinical observations, as patients with WNT-driven medulloblastoma typically exhibit superior survival outcomes compared to those with SHH-driven tumors under similar treatment regimens. While enhanced BBB permeability in the WNT subtype may contribute to improved drug delivery, other contributing factors such as a favorable immune microenvironment and inherent chemosensitivity are also believed to influence the improved prognosis observed in patients (65). Compared with the other subgroups, the Group 3 subtype has significantly greater vascular endothelial growth factor A (VEGFA) mRNA expression (66), which may be directly driven by MYC activation (67). Although some degree of BBB disruption is observed in medulloblastomas, most tumors retain partial barrier function, which poses a major obstacle for systemic therapies to effectively penetrate into the tumor core (57). Group 3 subtype is associated with a poor prognosis, characterized by a survival rate below 40% and frequent metastases (68). Medulloblastoma frequently metastasizes through cerebrospinal fluid (CSF) dissemination, with the therapeutic challenge being especially pronounced in patients presenting with leptomeningeal spread or distant metastases. In these cases, the BBB remains a significant obstacle to effective drug delivery. Intrathecal administration offers considerable potential as a therapeutic strategy to bypass the BBB and directly target tumor cells within the CSF compartment (69, 70). The interaction between tumor cells and the vascular microenvironment also plays a crucial role in maintaining BBB stability. Some studies have shown that SOX2-positive medulloblastoma stem-like cells tightly envelop capillaries via a mechanosensitive ion channel Piezo2-dependent mechanism, thereby influencing local tissue mechanical properties and vascular permeability (71, 72). Targeting the Piezo2 channel (e.g., via knockout) not only significantly enhances drug delivery efficiency but also has the potential to prolong survival, indicating that Piezo2 is a novel target with clinical translational potential (72). Furthermore, radiotherapy has been demonstrated to improve the efficiency of fucoidan nanocarrier-mediated transcytosis via P-selectin, thus increasing drug delivery (64). However, radiation also impacts BBB-associated structures, such as the perivascular space (PVS), potentially causing side effects, including cerebral edema and alterations in the tumor microenvironment (73). Owing to the barrier effect of the BBB, liquid biopsy markers such as circulating microRNAs in peripheral blood have shown limited sensitivity for detecting medulloblastoma (74). Therefore, the clinical focus has increasingly shifted toward the use of imaging parameters to evaluate the BBB status. MRI-based biomarkers, such as the ADC, hold promise for noninvasively monitoring BBB integrity within tumors and assessing treatment responses (75, 76).
Ependymoma
Ependymoma is a neuroepithelial tumor that arises from the ependymal layer bordering the cerebral ventricles and spinal canal. Intracranial ependymoma represents a major encephalic tumor in children, whereas spinal ependymoma develops more frequently in adults (77). In pediatric ependymomas, the structural integrity of the BBB remains preserved (Figure 1) (78). However, ependymal cells constitute a key component of both the cerebrospinal fluid–brain barrier and the blood–CSF barrier, playing a vital role in maintaining CNS homeostasis. During ependymoma development, these barrier cells can be directly disrupted by the tumor, leading to abnormal infiltration of cerebrospinal fluid components into the tumor parenchyma, which alters the local microenvironment and may promote tumor progression (79). Animal model studies further demonstrated that intracranial tumor growth could directly damage the ependymal epithelial layer, disturbing its barrier function and thereby enhancing interactions between cerebrospinal fluid and tumor tissue (80). Significant regional heterogeneity of the BBB has been observed in ependymomas, particularly in supratentorial tumors. In 9 out of 20 cases, tumor cells in supratentorial ependymomas highly expressed the tight junction protein claudin-5, a marker of BBB integrity, a feature not observed in infratentorial tumors (81). The heterogeneity of the BBB in ependymomas is a major contributor to treatment failure and poor drug delivery efficiency. Targeted therapies face clinical challenges, partly because of the variable drug permeability across different tumor regions, which is governed mainly by differences in BBB permeability and molecular tumor characteristics (82). Some studies have shown that the regulation of BBB functionality is closely linked to the glycosylation of certain transmembrane proteins, such as basigin, which is upregulated in ependymomas. Basigin may influence barrier permeability by modulating endothelial tight junctions and transport mechanisms (83). Ginguené C et al. reported that a biochemical, transporter-dependent blood‒tumor barrier might exist in ependymomas, which may reduce the tumoral bioavailability of lipophilic and amphiphilic anticancer drugs (77). It is noteworthy that two pediatric cases of relapsing demyelination, occurring after and in conjunction with radiation therapy for ependymoma, have been reported with features consistent with a multiple sclerosis phenotype, suggesting radiation-induced disruption of the BBB (84). Such BBB impairment may enhance the permeability of therapeutic agents, potentially improving drug delivery to the central nervous system. In parallel, Ependymal cells regulate CSF dynamics and contribute to CSF production via the choroid plexus. Their strategic ventricular location enables broad distribution of therapeutic proteins through CSF pathways, potentially bypassing the BBB and offering a route for targeted brain tumor treatment while minimizing neuronal toxicity (85). Therefore, a deeper understanding of BBB functional states and underlying molecular mechanisms across ependymoma is critical for improving drug delivery strategies and enhancing therapeutic responses.
Craniopharyngioma
Craniopharyngioma is a benign tumor originating from embryonic remnants of Rathke's pouch or the residual craniopharyngeal duct epithelium. In pediatric patients, the peak incidence of craniopharyngioma occurs between 5 and 15 years of age, with the adamantinomatous subtype being the most predominant form observed in this population. Pediatric adamantinomatous craniopharyngiomas (ACPs) predominantly occur in the sellar and suprasellar regions, adjacent to critical neurovascular structures such as the hypothalamic‒pituitary axis and optic pathways (86). The integrity of the BBB in this area is crucial for maintaining neuroendocrine system homeostasis. Craniopharyngiomas, though histologically benign, can exert significant mass effects that compress or infiltrate adjacent brain structures, leading to focal BBB disruption (Figure 1) (57, 87). For instance, hypothalamic damage frequently results in multiple pituitary hormone deficiencies and may induce metabolic disorders such as nonalcoholic fatty liver disease, suggesting that BBB dysfunction may play a significant role in these pathological changes (88, 89). Moreover, the tumors' clinically aggressive behavior may further impair BBB integrity, causing neuroendocrine abnormalities (e.g., growth hormone deficiency or diabetes insipidus) as well as visual impairment (90, 91). Studies indicate that pediatric patients face a significantly greater risk of postoperative obesity, cognitive decline, and cranial nerve damage than adults do, which is potentially related to inflammation or oxidative stress triggered by BBB disruption (92, 93). A small phase 0 study, which involved three child patients, provided compelling evidence that the humanized monoclonal antibody tocilizumab effectively reached ACP tumors and cyst fluid after systemic delivery (94). These findings support existing data that indicate that tocilizumab may play a role in systemic therapy for ACP. Furthermore, these findings imply that the ACP-associated BBB is compromised relative to other regions of the CNS (94). Moreover, abnormal expression of tight junction proteins such as claudin-5 within the gut–brain axis is thought to exacerbate BBB dysfunction, suggesting that the peripheral system may influence central nervous system function through the modulation of barrier integrity (95). Currently, surgical resection and radiotherapy remain the primary treatment modalities in patients with craniopharyngiomas but may exacerbate BBB damage to some extent. For example, perioperative BBB disruption is closely associated with cognitive impairment and increased postoperative mortality (93). Oxytocin treatment of ACP patients can restore the integrity of the BBB and reduce the inflammatory response that is activated by infiltrating peripheral immune cells, thus blocking the lipotoxicity of hypothalamic neural stem cells induced by the diffusion of peripheral low-density lipoprotein cholesterol into the third ventricle (96). To minimize hypothalamic injury, some studies recommend conservative treatment strategies, such as limited resection combined with moderate radiotherapy (97, 98). Additionally, the permeability of the BBB in ACP can vary considerably, resulting in inconsistent therapeutic delivery (99). To address this challenge, localized treatment strategies such as intracystic drug administration and nanotechnology-based delivery systems are being investigated as potential approaches to bypass the BBB and improve treatment efficacy (100). However, there is a lack of sufficient evidence regarding the long-term efficacy of these approaches in protecting or restoring BBB function, highlighting the need for further systematic investigations.
Conclusion
The review underscores the heterogeneous integrity of the BBB across pediatric brain tumors and its significant implications for drug delivery and therapeutic resistance. By integrating histopathological, molecular, and imaging data, it highlights the subtype-specific characteristics of BBB disruption and preservation. Furthermore, the review evaluates emerging therapeutic strategies, such as FUS and CED, that aim to overcome BBB-associated barriers, thereby enhancing drug delivery and potentially improving clinical outcomes in affected children.
Author contributions
QG: Writing – review & editing, Data curation, Writing – original draft. DS: Data curation, Writing – original draft. DX: Writing – original draft. XC: Writing – original draft, Data curation. MG: Writing – review & editing.
Funding
The author(s) declare that no financial support was received for the research and/or publication of this article.
Acknowledgments
The authors thank Yafang Deng for her assistance in image archiving and processing.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Nyeko R, Kambugu JB, Angom R, Senyonjo H, Kibudde S, Geriga F, et al. The clinicopathological profile and value of multidisciplinary management of pediatric brain tumors in a low-income setting. Pediatr Hematol Oncol. (2023) 403:267–80. doi: 10.1080/08880018.2022.2140861
2. Nyalundja AD, Kanmounye US, Karekezi C, Laeke T, Thango N, Balogun JA. Pediatric brain tumors in sub-Saharan Africa: a systematic review and meta-analysis. J Neurosurg Pediatr. (2024) 336:524–35. doi: 10.3171/2024.1.PEDS23282
3. Grigore FN, Yang SJ, Chen CC, Koga T. Pioneering models of pediatric brain tumors. Neoplasia. (2023) 36:100859. doi: 10.1016/j.neo.2022.100859
4. Estevez-Ordonez D, Gary SE, Atchley TJ, Maleknia PD, George JA, Laskay NMB, et al. Immunotherapy for pediatric brain and spine tumors: current state and future directions. Pediatr Neurosurg. (2023) 585:313–36. doi: 10.1159/000528792
5. Das AK, Mani SK, Singh SK, Kumar S. Management and outcome of unusual pediatric brain tumors: challenges experienced at a tertiary care center of a developing country. Childs Nerv Syst. (2023) 391:169–83. doi: 10.1007/s00381-022-05694-2
6. Yao B, Wang H, Wu X, Wang C, Tang T, An W, et al. A system review of central nervous system tumors on children in China: epidemiology and clinical characteristics. BMC Cancer. (2024) 241:138. doi: 10.1186/s12885-024-11883-w
7. Tripathy A, John V, Wadden J, Kong S, Sharba S, Koschmann C. Liquid biopsy in pediatric brain tumors. Front Genet. (2023) 13:1114762. doi: 10.3389/fgene.2022.1114762
8. Abuhamed J, Nikkilä A, Raitanen J, Alimam W, Lohi O, Pitkäniemi J, et al. Incidence trends of childhood central nervous system tumors in Finland 1990–2017. BMC Cancer. (2022) 221:784. doi: 10.1186/s12885-022-09862-0
9. Roth C. Pediatric supratentorial brain tumors. Radiologie (Heidelb). (2023) 63(8):592–7. doi: 10.1007/s00117-023-01158-z
10. Rjoub A, Abu Zahra W, Issa N, Dumaidi Y, Abuawad M, Daqour A, et al. Epidemiology and anatomical distribution of primary brain tumors among children in Palestine: a 6-year national referral institution study. World Neurosurg. (2024) 186:e470–80. doi: 10.1016/j.wneu.2024.03.160
11. Daneman R, Prat A. The blood-brain barrier. Cold Spring Harb Perspect Biol. (2015) 7(1):a020412. doi: 10.1101/cshperspect.a020412
12. Banks WA. Characteristics of compounds that cross the blood-brain barrier. BMC Neurol. (2009) 9 Suppl 1(Suppl 1):S3. doi: 10.1186/1471-2377-9-S1-S3
13. Pan W, Kastin AJ. Changing the chemokine gradient: CINC1 crosses the blood-brain barrier. J Neuroimmunol. (2001) 115(1-2):64–70. doi: 10.1016/S0165-5728(01)00256-9
14. Rattner A, Wang Y, Nathans J. Signaling pathways in neurovascular development. Annu Rev Neurosci. (2022) 45:87–108. doi: 10.1146/annurev-neuro-111020-102127
15. Brown SM, Sinha BK, Cannon RE. A role for iNOS in Erastin mediated reduction of P-glycoprotein transport activity. Cancers Basel. (2024) 169:1733. doi: 10.3390/cancers16091733
16. Schwinghamer K, Kopec BM, Ayewoh E, Tao X, Sadekar S, Sreedhara A, et al. Exploring how antibody format drives clearance from the brain. Mol Pharm. (2024) 219:4416–29. doi: 10.1021/acs.molpharmaceut.4c00354
17. Gao X, Xu J, Yao T, Liu X, Zhang H, Zhan C. Peptide-decorated nanocarriers penetrating the blood-brain barrier for imaging and therapy of brain diseases. Adv Drug Deliv Rev. (2022) 187:114362. doi: 10.1016/j.addr.2022.114362
18. Zhao Z, Chen Y, Sun T, Jiang C. Nanomaterials for brain metastasis. J Control Release. (2024) 365:833–47. doi: 10.1016/j.jconrel.2023.12.001
19. Matsuo K, Nshihara H. Rebuilding insight into the pathophysiology of Alzheimer’s disease through new blood-brain barrier models. Neural Regen Res. (2024) 19(9):1954–60. doi: 10.4103/1673-5374.390978
20. Chavarria D, Abbaspour A, Celestino N, Shah N, Sankar S, Baker AB. A high throughput blood-brain barrier model incorporating shear stress with improved predictive power for drug discovery. Biomicrofluidics. (2023) 174:044105. doi: 10.1063/5.0150887
21. Malbari F. Pediatric neuro-oncology. Continuum (Minneap Minn). (2023) 29(6):1680–709. doi: 10.1212/CON.0000000000001360
22. Louis DN, Perry A, Wesseling P, Brat DJ, Cree IA, Figarella-Branger D, et al. The 2021 WHO classification of tumors of the central nervous system: a summary. Neuro Oncol. (2021) 238:1231–51. doi: 10.1093/neuonc/noab106
23. Hong CS, Ho W, Piazza MG, Ray-Chaudhury A, Zhuang Z, Heiss JD. Characterization of the blood brain barrier in pediatric central nervous system neoplasms. J Interdiscip Histopathol. (2016) 42:29–33. doi: 10.5455/jihp.20160623053540
24. Roales-Buján R, Páez P, Guerra M, Rodríguez S, Vío K, Ho-Plagaro A, et al. Astrocytes acquire morphological and functional characteristics of ependymal cells following disruption of ependyma in hydrocephalus. Acta Neuropathol. (2012) 1244:531–46. doi: 10.1007/s00401-012-0992-6
25. Tan J, Sun W, Lu L, Xiao Z, Wei H, Shi W, et al. I6p7 peptide modified superparamagnetic iron oxide nanoparticles for magnetic resonance imaging detection of low-grade brain gliomas. J Mater Chem B. (2019) 740:6139–47. doi: 10.1039/c9tb01563a
26. Chesney KM, Keating GF, Patel N, Kilburn L, Fonseca A, Wu CC, et al. The role of focused ultrasound for pediatric brain tumors: current insights and future implications on treatment strategies. Childs Nerv Syst. (2024) 408:2333–44. doi: 10.1007/s00381-024-06413-9
27. Seas AA, Malla AP, Sharifai N, Winkles JA, Woodworth GF, Anastasiadis P. Microbubble-enhanced focused ultrasound for infiltrating gliomas. Biomedicines. (2024) 126:1230. doi: 10.3390/biomedicines12061230
28. de Laurentis C, Pasquali C, Beuriat PA, Mottolese C, Szathmari A, Vinchon M, et al. Role of sodium fluorescein in pediatric low-grade glioma surgery: an update. Childs Nerv Syst. (2024) 4010:3143–54. doi: 10.1007/s00381-024-06402-y
29. Anastasaki C, Chatterjee J, Cobb O, Sanapala S, Scheaffer SM, De Andrade Costa A, et al. Human induced pluripotent stem cell engineering establishes a humanized mouse platform for pediatric low-grade glioma modeling. Acta Neuropathol Commun. (2022) 101:120. doi: 10.1186/s40478-022-01428-2
30. Yvone GM, Breunig JJ. Pediatric low-grade glioma models: advances and ongoing challenges. Front Oncol. (2023) 13:1346949. doi: 10.3389/fonc.2023.1346949
31. Bruschi M, Midjek L, Ajlil Y, Vairy S, Lancien M, Ghermaoui S, et al. Diffuse midline glioma invasion and metastasis rely on cell-autonomous signaling. Neuro Oncol. (2024) 263:553–68. doi: 10.1093/neuonc/noad161
32. Chaves C, Declèves X, Taghi M, Menet MC, Lacombe J, Varlet P, et al. Characterization of the blood-brain barrier integrity and the brain transport of SN-38 in an orthotopic xenograft rat model of diffuse intrinsic pontine glioma. Pharmaceutics. (2020) 125:399. doi: 10.3390/pharmaceutics12050399
33. Gomez-Zepeda D, Taghi M, Scherrmann JM, Decleves X, Menet MC. ABC transporters at the blood-brain interfaces, their study models, and drug delivery implications in gliomas. Pharmaceutics. (2019) 121:20. doi: 10.3390/pharmaceutics12010020
34. Deligne C, Hachani J, Duban-Deweer S, Meignan S, Leblond P, Carcaboso AM, et al. Development of a human in vitro blood-brain tumor barrier model of diffuse intrinsic pontine glioma to better understand the chemoresistance. Fluids Barriers CNS. (2020) 171:37. doi: 10.1186/s12987-020-00198-0
35. Martinez PJ, Green AL, Borden MA. Targeting diffuse midline gliomas: the promise of focused ultrasound-mediated blood-brain barrier opening. J Control Release. (2024) 365:412–21. doi: 10.1016/j.jconrel.2023.11.037
36. Jovanovich N, Habib A, Head J, Hameed F, Agnihotri S, Zinn PO. Pediatric diffuse midline glioma: understanding the mechanisms and assessing the next generation of personalized therapeutics. Neurooncol Adv. (2023) 51:vdad040. doi: 10.1093/noajnl/vdad040
37. Tazhibi M, McQuillan N, Wei HJ, Gallitto M, Bendau E, Webster Carrion A, et al. Focused ultrasound-mediated blood-brain barrier opening is safe and feasible with moderately hypofractionated radiotherapy for brainstem diffuse midline glioma. J Transl Med. (2024) 221:320. doi: 10.1186/s12967-024-05096-9
38. Parekh K, LeBlang S, Nazarian J, Mueller S, Zacharoulis S, Hynynen K, et al. Past, present and future of focused ultrasound as an adjunct or complement to DIPG/DMG therapy: a consensus of the 2021 FUSF DIPG meeting. Neoplasia. (2023) 37:100876. doi: 10.1016/j.neo.2023.100876
39. Feng Y, Xu Q, Fang M, Hu C. Anlotinib combined with temozolomide for the treatment of patients with diffuse midline glioma: a case report and literature review. Transl Cancer Res. (2022) 1110:3876–82. doi: 10.21037/tcr-22-1073
40. Kwon MJ, Kim SS, Choi YL, Jung HS, Balch C, Kim SH, et al. Derepression of CLDN3 and CLDN4 during ovarian tumorigenesis is associated with loss of repressive histone modifications. Carcinogenesis. (2010) 316:974–83. doi: 10.1093/carcin/bgp336
41. Sasaki T, Watanabe J, He X, Katagi H, Suri A, Ishi Y, et al. Intranasal delivery of nanoliposomal SN-38 for treatment of diffuse midline glioma. J Neurosurg. (2022) 1386:1570–9. doi: 10.3171/2022.9.JNS22715
42. McCully CM, Pastakia D, Bacher J, Thomas ML 3rd, Steffen-Smith EA, Saleem K, et al. Model for concomitant microdialysis sampling of the pons and cerebral cortex in rhesus macaques Macaca mulatta. Comp Med. (2013) 63(4):355–60.24209972
43. Jackson ER, Persson ML, Fish CJ, Findlay IJ, Mueller S, Nazarian J, et al. A review of current therapeutics targeting the mitochondrial protease ClpP in diffuse midline glioma, H3 K27-altered. Neuro Oncol. (2024) 26(Supplement_2):S136–54. doi: 10.1093/neuonc/noad144
44. Nonnenbroich LF, Bouchal SM, Millesi E, Rechberger JS, Khatua S, Daniels DJ. H3K27-altered diffuse midline glioma of the brainstem: from molecular mechanisms to targeted interventions. Cells. (2024) 13(13):1122. doi: 10.3390/cells13131122
45. Donev K, Sundararajan V, Johnson D, Balan J, Chambers M, Paulson VA, et al. Diffuse hemispheric glioma with H3 p.K28M K27M mutation: unusual non-midline presentation of diffuse midline glioma, H3 K27M-altered? J Neuropathol Exp Neurol. (2024) 835:357–64. doi: 10.1093/jnen/nlae018
46. Zhao JP, Liu XJ, Lin HZ, Cui CX, Yue YJ, Gao S, et al. MRI comparative study of diffuse midline glioma, H3 K27-altered and glioma in the midline without H3 K27-altered. BMC Neurol. (2022) 221:498. doi: 10.1186/s12883-022-03026-0
47. Li J, Zhang P, Qu L, Sun T, Duan Y, Wu M, et al. Deep learning for noninvasive assessment of H3 K27M mutation status in diffuse midline gliomas using MR imaging. J Magn Reson Imaging. (2023) 58(3):850–61. doi: 10.1002/jmri.28606
48. Odia Y, Hall MD, Cloughesy TF, Wen PY, Arrillaga-Romany I, Daghistani D, et al. Selective DRD2 antagonist and ClpP agonist ONC201 in a recurrent non-midline H3 K27M-mutant glioma cohort. Neuro Oncol. (2024) 26(Supplement_2):S165–72. doi: 10.1093/neuonc/noae021
49. Veringa SJ, Biesmans D, van Vuurden DG, Jansen MH, Wedekind LE, Horsman I, et al. In vitro drug response and efflux transporters associated with drug resistance in pediatric high grade glioma and diffuse intrinsic pontine glioma. PLoS One. (2013) 8(4):e61512. doi: 10.1371/journal.pone.0061512
50. Bredel M, Zentner J. Brain-tumour drug resistance: the bare essentials. Lancet Oncol. (2002) 3(7):397–406. doi: 10.1016/S1470-2045(02)00786-6
51. Lertsumitkul L, Iliopoulos M, Wang SS, McArthur SJ, Ebert LM, Davenport AJ, et al. EphA3-targeted chimeric antigen receptor T cells are effective in glioma and generate curative memory T cell responses. J Immunother Cancer. (2024) 12(8):e009486. doi: 10.1136/jitc-2024-009486
52. Filieri S, Miciaccia M, Armenise D, Baldelli OM, Liturri A, Ferorelli S, et al. Can focused ultrasound overcome the failure of chemotherapy in treating pediatric diffuse intrinsic pontine glioma due to a blood-brain barrier obstacle? Pharmaceuticals Basel. (2025) 18(4):525. doi: 10.3390/ph18040525
53. Thomas BC, Staudt DE, Douglas AM, Monje M, Vitanza NA, Dun MD. CAR T cell therapies for diffuse midline glioma. Trends Cancer. (2023) 9(10):791–804. doi: 10.1016/j.trecan.2023.07.007
54. Liu C, Kuang S, Wu L, Cheng Q, Gong X, Wu J, et al. Radiotherapy and radio-sensitization in H3K27M -mutated diffuse midline gliomas. CNS Neurosci Ther. (2023) 29(7):1721–37. doi: 10.1111/cns.14225
55. Zhu H, Allwin C, Bassous MG, Pouliopoulos AN. Focused ultrasound-mediated enhancement of blood-brain barrier permeability for brain tumor treatment: a systematic review of clinical trials. J Neurooncol. (2024) 170(2):235–52. doi: 10.1007/s11060-024-04795-z
56. Spinazzi EF, Argenziano MG, Upadhyayula PS, Banu MA, Neira JA, Higgins DMO, et al. Chronic convection-enhanced delivery of topotecan for patients with recurrent glioblastoma: a first-in-patient, single-centre, single-arm, phase 1b trial. Lancet Oncol. (2022) 23(11):1409–18. doi: 10.1016/S1470-20452200599-X
57. Morris EK, Daignault-Mill S, Stehbens SJ, Genovesi LA, Lagendijk AK. Addressing blood-brain-tumor-barrier heterogeneity in pediatric brain tumors with innovative preclinical models. Front Oncol. (2023) 13:1101522. doi: 10.3389/fonc.2023.1101522
58. Kunhiraman H, McSwain L, Shahab SW, Gershon TR, MacDonald TJ, Kenney AM. IGFBP2 promotes proliferation and cell migration through STAT3 signaling in sonic hedgehog medulloblastoma. Acta Neuropathol Commun. (2023) 11(1):62. doi: 10.1186/s40478-023-01557-2
59. Engelhardt B, Sorokin L. The blood-brain and the blood-cerebrospinal fluid barriers: function and dysfunction. Semin Immunopathol. (2009) 31(4):497–511. doi: 10.1007/s00281-009-0177-0
60. Phoenix TN, Patmore DM, Boop S, Boulos N, Jacus MO, Patel YT, et al. Medulloblastoma genotype dictates blood brain barrier phenotype. Cancer Cell. (2016) 29(4):508–22. doi: 10.1016/j.ccell.2016.03.002
61. Benz F, Wichitnaowarat V, Lehmann M, Germano RF, Mihova D, Macas J, et al. Low wnt/β-catenin signaling determines leaky vessels in the subfornical organ and affects water homeostasis in mice. Elife. (2019) 8:e43818. doi: 10.7554/eLife.43818
62. Ward SA, Warrington NM, Taylor S, Kfoury N, Luo J, Rubin JB. Reprogramming medulloblastoma-propagating cells by a combined antagonism of sonic hedgehog and CXCR4. Cancer Res. (2017) 77(6):1416–26. doi: 10.1158/0008-5472.CAN-16-0847
63. Yuan L, Zhang H, Liu J, Rubin JB, Cho YJ, Shu HK, et al. Growth factor receptor-src-mediated suppression of GRK6 dysregulates CXCR4 signaling and promotes medulloblastoma migration. Mol Cancer. (2013) 12:18. doi: 10.1186/1476-4598-12-18
64. Tylawsky DE, Kiguchi H, Vaynshteyn J, Gerwin J, Shah J, Islam T, et al. P-selectin-targeted nanocarriers induce active crossing of the blood-brain barrier via caveolin-1-dependent transcytosis. Nat Mater. (2023) 22(3):391–9. doi: 10.1038/s41563-023-01481-9
65. Gajjar A, Robinson GW, Smith KS, Lin T, Merchant TE, Chintagumpala M, et al. Outcomes by clinical and molecular features in children with medulloblastoma treated with risk-adapted therapy: results of an international phase III trial SJMB03. J Clin Oncol. (2021) 39(7):822–35. doi: 10.1200/JCO.20.01372
66. Thompson EM, Keir ST, Venkatraman T, Lascola C, Yeom KW, Nixon AB, et al. The role of angiogenesis in group 3 medulloblastoma pathogenesis and survival. Neuro Oncol. (2017) 19(9):1217–27. doi: 10.1093/neuonc/nox033
67. Baudino TA, McKay C, Pendeville-Samain H, Nilsson JA, Maclean KH, White EL, et al. c-Myc is essential for vasculogenesis and angiogenesis during development and tumor progression. Genes Dev. (2002) 16(19):2530–43. doi: 10.1101/gad.1024602
68. Łastowska M, Trubicka J, Niemira M, Paczkowska-Abdulsalam M, Karkucińska-Więckowska A, Kaleta M, et al. Medulloblastoma with transitional features between group 3 and group 4 is associated with good prognosis. J Neurooncol. (2018) 138(2):231–40. doi: 10.1007/s11060-018-2797-5
69. Khang M, Lee JH, Lee T, Suh HW, Lee S, Cavaliere A, et al. Intrathecal delivery of nanoparticle PARP inhibitor to the cerebrospinal fluid for the treatment of metastatic medulloblastoma. Sci Transl Med. (2023) 15720:eadi1617. doi: 10.1126/scitranslmed.adi1617
70. Chen Y, Zhao H, Zhang H, Wang B, Ma J. Cytokine profile of cerebrospinal fluid in pediatric patients with metastatic medulloblastoma. Heliyon. (2024) 10(21):e38504. doi: 10.1016/j.heliyon.2024.e38504
71. Chen X, Momin A, Wanggou S, Wang X, Min HK, Dou W, et al. Mechanosensitive brain tumor cells construct blood-tumor barrier to mask chemosensitivity. Neuron. (2023) 111(1):30–48.e14. doi: 10.1016/j.neuron.2022.10.007
72. Sprowls SA, Lathia JD. Breaking down the barrier to medulloblastoma treatment: piezo2 knockout disrupts the BTB and increases vascular permeability. Neuron. (2023) 111(1):3–5. doi: 10.1016/j.neuron.2022.12.008
73. Song R, Glass JO, Wu S, Li Y, Robinson GW, Gajjar A, et al. Perivascular space imaging during therapy for medulloblastoma. PLoS One. (2025) 20(2):e0318278. doi: 10.1371/journal.pone.0318278
74. Liu Y, Peng Y, Zhang C, Chen R, Zhang K. Single-molecule detection of serum MicroRNAs for medulloblastoma with biphasic sandwich hybridization-assisted plasmonic resonant scattering imaging. Anal Chem. (2024) 96(47):18655–63. doi: 10.1021/acs.analchem.4c02665
75. Aljaafary M, Alali AA. Association of imaging biomarkers with molecular subtypes of medulloblastoma. Neuroradiol J. (2025) 38(3):304–11. doi: 10.1177/19714009241303065
76. Gonçalves FG, Tierradentro-Garcia LO, Kim JDU, Zandifar A, Ghosh A, Viaene AN, et al. The role of apparent diffusion coefficient histogram metrics for differentiating pediatric medulloblastoma histological variants and molecular groups. Pediatr Radiol. (2022) 52(13):2595–609. doi: 10.1007/s00247-022-05411-w
77. Ginguené C, Champier J, Maallem S, Strazielle N, Jouvet A, Fèvre-Montange M, et al. P-glycoprotein ABCB1 and breast cancer resistance protein ABCG2 localize in the microvessels forming the blood-tumor barrier in ependymomas. Brain Pathol. (2010) 20(5):926–35. doi: 10.1111/j.1750-3639.2010.00389.x
78. Malik JR, Podany AT, Khan P, Shaffer CL, Siddiqui JA, Baranowska-Kortylewicz J, et al. Chemotherapy in pediatric brain tumor and the challenge of the blood-brain barrier. Cancer Med. (2023) 12(23):21075–96. doi: 10.1002/cam4.6647
79. Nelles DG, Hazrati LN. Ependymal cells and neurodegenerative disease: outcomes of compromised ependymal barrier function. Brain Commun. (2022) 4(6):fcac288. doi: 10.1093/braincomms/fcac288
80. Norton ES, Whaley LA, Ulloa-Navas MJ, García-Tárraga P, Meneses KM, Lara-Velazquez M, et al. Glioblastoma disrupts the ependymal wall and extracellular matrix structures of the subventricular zone. Fluids Barriers CNS. (2022) 19(1):58. doi: 10.1186/s12987-022-00354-8
81. Virág J, Haberler C, Baksa G, Piurkó V, Hegedüs Z, Reiniger L, et al. Region specific differences of claudin-5 expression in pediatric intracranial ependymomas: potential prognostic role in supratentorial cases. Pathol Oncol Res. (2017) 23(2):245–52. doi: 10.1007/s12253-016-0084-3
82. Upton DH, Ung C, George SM, Tsoli M, Kavallaris M, Ziegler DS. Challenges and opportunities to penetrate the blood-brain barrier for brain cancer therapy. Theranostics. (2022) 12(10):4734–52. doi: 10.7150/thno.69682
83. Cui D, Yamamoto K, Ikeda E. High-mannose-type glycan of basigin in endothelial cells is essential for the opening of the blood-brain barrier induced by hypoxia, cyclophilin A, or tumor necrosis factor-alpha. Am J Pathol. (2024) 194(4):612–25. doi: 10.1016/j.ajpath.2023.11.007
84. Smith JD, Mandel G, Niazi T, Bradley JA, Indelicato DJ, Khatib Z. Multifocal and multiphasic demyelinating lesions after radiation for ependymoma in a pediatric population. J Child Neurol. (2022) 37(7):609–16. doi: 10.1177/08830738221079476
85. Carrell EM, Chen YH, Ranum PT, Coffin SL, Singh LN, Tecedor L, et al. VWA3A-derived ependyma promoter drives increased therapeutic protein secretion into the CSF. Mol Ther Nucleic Acids. (2023) 33:296–304. doi: 10.1016/j.omtn.2023.07.016
86. Memon F, Humayun KN, Riaz Q, Arif M, Huda NU, Laghari AA, et al. Pediatric craniopharyngioma: a 20-year study on epidemiological features, clinical presentation, and survival outcomes in a tertiary care center from LMIC. Childs Nerv Syst. (2024) 40(2):427–34. doi: 10.1007/s00381-023-06177-8
87. Shen K, Shi Y, Wang X, Leung SW. Cellular components of the blood-brain barrier and their involvement in aging-associated cognitive impairment. Aging Dis. (2024) 16(3):1513–34. doi: 10.14336/AD.202.0424
88. Zhang XY, Yuan K, Fang YL, Wang CL. Growth hormone ameliorates hepatopulmonary syndrome and nonalcoholic steatohepatitis secondary to hypopituitarism in a child: a case report. World J Clin Cases. (2022) 10(18):6211–7. doi: 10.12998/wjcc.v10.i18.6211
89. Zhu R, Wang L, Zhao L, Liu X. Central precocious puberty secondary to postoperative craniopharyngioma: two case reports and a literature review. Childs Nerv Syst. (2024) 40(7):2245–50. doi: 10.1007/s00381-024-06406-8
90. Stec NE, Barker FG 2nd, Brastianos PK. Targeted treatment for craniopharyngioma. J Neurooncol. (2025) 172(3):503–13. doi: 10.1007/s11060-025-04942-0
91. Colliander R, Sharma S, Shlobin NA, Fernandez LG, LoPresti MA, Lam S, et al. Visual outcomes after treatment of craniopharyngioma in children: a systematic review. Childs Nerv Syst. (2024) 40(6):1641–59. doi: 10.1007/s00381-024-06328-5
92. Pang JC, Chung DD, Wang J, Abiri A, Lien BV, Himstead AS, et al. Characteristics and outcomes in pediatric versus adult craniopharyngiomas: a systematic review and meta-analysis. Neurosurgery. (2023) 92(6):1112–29. doi: 10.1227/neu.0000000000002346
93. Qiu Y, Mo C, Xu S, Chen L, Ye W, Kang Y, et al. Research progress on perioperative blood-brain barrier damage and its potential mechanism. Front Cell Dev Biol. (2023) 11:1174043. doi: 10.3389/fcell.2023.1174043
94. Prince EW, Hoffman LM, Vijmasi T, Dorris K, McWilliams JA, Jordan KR, et al. Adamantinomatous craniopharyngioma associated with a compromised blood-brain barrier: patient series. J Neurosurg Case Lessons. (2021) 119:CASE2150. doi: 10.3171/CASE2150
95. Tanır Y, Cahid Örengül A, Esad Özdemir Y, Karayağmurlu A, Bilbay Kaynar T, Merve Baki A, et al. Serum zonulin and claudin-5 but not interferon-gamma and interleukin-17A levels increased in children with specific learning disorder: a case-control study. Psychiatry Clin Psychopharmacol. (2023) 33(3):211–7. doi: 10.5152/pcp.2023.23660
96. Wang C, Zhang H, Fan J, Li Q, Guo R, Pan J, et al. Inhibition of integrated stress response protects against lipid-induced senescence in hypothalamic neural stem cells in adamantinomatous craniopharyngioma. Neuro Oncol. (2023) 25(4):720–32. doi: 10.1093/neuonc/noac261
97. Aldave G, Okcu MF, Chintagumpala M, Ruggieri L, Minard CG, Malbari F, et al. Comparison of neurocognitive and quality-of-life outcomes in pediatric craniopharyngioma patients treated with partial resection and radiotherapy versus gross-total resection only. J Neurosurg Pediatr. (2023) 31(5):453–62. doi: 10.3171/2022.12.PEDS22367
98. Agresta G, Campione A, Veiceschi P, Gallo D, Agosti E, Massimi L, et al. Clinical and oncological outcomes in single-stage versus staged surgery for pediatric craniopharyngiomas: a multicenter retrospective study. J Endocrinol Invest. (2023) 46(6):1219–32. doi: 10.1007/s40618-022-01993-2
99. Muller HL. The diagnosis and treatment of craniopharyngioma. Neuroendocrinology. (2020) 110(9–10):753–66. doi: 10.1159/000504512
Keywords: pediatric brain tumors, blood-brain barrier, tumor heterogeneity, permeability, drug delivery
Citation: Gao Q, Song D, Xu D, Chai X and Ge M (2025) Characteristics of the blood–brain barrier in pediatric brain tumors. Front. Pediatr. 13:1646641. doi: 10.3389/fped.2025.1646641
Received: 13 June 2025; Accepted: 7 August 2025;
Published: 29 August 2025.
Edited by:
Eugene Hwang, Children's National Hospital, United StatesReviewed by:
Enzi Jiang, CSL, United StatesJohn Bianco, Princess Maxima Center for Pediatric Oncology, Netherlands
Copyright: © 2025 Gao, Song, Xu, Chai and Ge. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Ming Ge, bWluZ19nZUAxMjYuY29t; Xinyi Chai, MjI5MDg5NzIyN0BxcS5jb20=
†These authors have contributed equally to this work
 Qiang Gao
Qiang Gao Dengpan Song
Dengpan Song Dingkang Xu3
Dingkang Xu3 Xinyi Chai
Xinyi Chai