- 1Department of Urology and Laboratory of Pelvic Floor Muscle Function, Shenzhen Children’s Hospital, Shenzhen, Guangdong, China
- 2Department of Urology, Shenzhen Children's Hospital of Shantou University Medical College, Shenzhen, Guangdong, China
Purpose: To resolve conflicting evidence on androgen receptor (AR) expression in hypospadias, we compared preputial AR levels between affected children and controls.
Methods: Forty patients with isolated hypospadias (age 2.9 ± 0.79 years, range 1.6–5.2 years) and 40 normal boys (age 3.2 ± 0.79 years, range 1.8–4.7 years) who underwent circumcision for phimosis were included in our study. Inner plates of preputial tissues were analyzed by immunohistochemistry for AR detection.
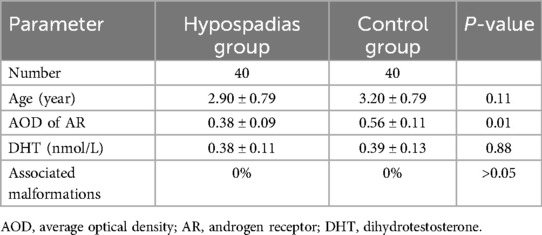
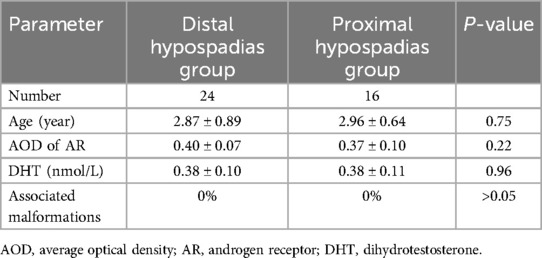
Results: AR immunoreactivity was predominantly localized to the basal epithelial layer. Significantly reduced AR expression was observed in hypospadias patients vs. controls [average optical density (AOD), 0.38 ± 0.09 vs. 0.56 ± 0.11]. However, there was no difference in AR expression between the proximal and distal hypospadias (AOD, 0.40 ± 0.07 vs. 0.37 ± 0.10, P = 0.22).
Conclusions: Preputial AR downregulation in hypospadias implicates AR deficiency in pathogenesis, providing evidence to resolve a key controversy in the field.
Introduction
Hypospadias is a common congenital malformation of the urinary system in male children, with a prevalence of 18.6 per 10,000 births (1). It is defined as incomplete development of the male urethra leading to aberrant localization of the urinary meatus on the ventral side of the penis in a variable position from the glans to the perineum. The etiology of hypospadias remains unknown. Although some studies have implicated androgen receptors (ARs) in hypospadias development, recent studies report conflicting results regarding AR expression.
Vottero et al. demonstrated that AR expression in the preputial tissue of patients with hypospadias was lower than that of normal children (2). Chao et al. examined the AR in the prepuce of patients with hypospadias and controls using immunohistochemistry (IHC) and observed that AR expression in 68 hypospadias tissues was significantly lower than that in 68 normal Preputial tissues (3). Silva T et al. studied 17 hypospadias and 41 control patients and observed significantly less AR mRNA levels in the urethral mucosa of patients with hypospadias than in controls (4). Recent epigenetic studies further highlight transcriptional dysregulation of AR in preputial mucosa (5, 6). In contrast, Pichler et al. discovered that AR protein was higher in boys with hypospadias than in boys without hypospadias (133.25 ± 6.17 vs. 100 ± 4.45, P = 0.014) (7). Similarly, Kocaturk et al. documented higher AR expression in hypospadias-affected preputial tissue than in normal tissue (8). Hence, the expression of AR in patients with hypospadias remains controversial.
We conducted a large prospective study to clarify discrepancies in AR expression between hypospadias patients and normal children, and to assess the relationship between AR dysregulation and hypospadias development.
Materials and methods
Patients and study design
From January 2022 to December 2024, 40 patients with isolated hypospadias and 40 healthy boys who underwent circumcision for phimosis were included in our study. All subjects were ethnic Han people of Chinese origin. Distal and proximal hypospadias were present in 24 and 16 patients, respectively. Patients with DSD, cryptorchidism, or other anomalies and those with a history of preoperative hormonal treatment were excluded. Preoperative blood samples were collected for hormone analysis; preputial tissues were harvested intraoperatively for histopathological examination.
Hormone measurements
Serum dihydrotestosterone (DHT) levels were tested in all subjects before surgery. Blood samples were centrifuged immediately after collection. Then, the plasma was stored at −20 ℃ until assay. Serum testosterone and DHT levels were measured using commercial kits (ELISA, Mlbio Company, Shanghai, China). The sensitivity of the hormone assay was 99%.
Immunohistochemistry (IHC)
The inner plates (mucosal surface) of prepuce tissues were procured intraoperatively during circumcision or hypospadias repair, fixed in 4% paraformaldehyde for ≥6 h, rinsed three times, and dehydrated through a graded series of isopropanol and xylene. Subsequently, tissues were embedded in paraffin, and 4 μm sections cut at 200 μm intervals were prepared for staining.
For AR detection, sections underwent standardized immunohistochemistry:
• Deparaffinization in xylene;
• Heat-induced antigen retrieval (100°C, 20 min in citrate buffer, pH 6.0);
• Primary antibody incubation with monoclonal mouse anti-human AR antibody (AR441, MXB Biotechnologies; 1:100 dilution, 8 h at 4°C);
• Secondary antibody incubation with biotinylated goat anti-mouse IgG (37°C, 2 h);
• Signal amplification with streptavidin-HRP (30 min);
• DAB chromogenic development (monitored in real-time under microscopy);
• Counterstaining with hematoxylin (1 min).
All procedures were conducted by a single blinded pathologist.
Human prostate carcinoma tissue served as the positive control. AR immunoreactivity was quantified as average optical density (AOD) using ImageJ (NIH v1.53k). For each specimen, five random fields per section (totaling 25 fields/sample) were analyzed via superimposing a 100-point grid. Mean AOD values were compared among hypospadias patients, controls, and distal/proximal subtypes.
Institutional review board approval
This study was approved by the Institutional Review Board of Shenzhen Children's Hospital (IRB No. 202210802), and informed consent was obtained from the parents of the children.
Statistical analyses
Continuous data were compared using independent t-test. Categorical variables were analyzed with the chi-squared test or logistic regression analysis. All statistical analyses were performed with a two-tailed significance level of α = 0.05 using SPSS 26.0 (IBM Corp., Armonk, NY).
Results
The hypospadias group included 40 patients; the mean age was 2.90 ± 0.79 (range, 1.6–5.2) years. Forty patients who underwent circumcision were in the control group, and the mean age was 3.2 ± 0.79 (range: 1.8–4.7) years. There was no difference in age between the hypospadias and control groups (Table 1). In addition, there was no difference in serum DHT concentration between the hypospadias and control groups (0.38 ± 0.11 vs. 0.39 ± 0.13 nmol/L, P = 0.88).
IHC of inner plate preputial tissues revealed that the AR protein was majorly expressed on the basal cell layer, while some were expressed in the fibroblasts (Figure 1). Furthermore, AR expression was lower in the hypospadias group than in the control group (AOD, 0.38 ± 0.09 vs. 0.56 ± 0.11, P = 0.01) (Table 1) (Figure 1). However, there was no difference in AR expression between the proximal and distal hypospadias (AOD, 0.40 ± 0.07 vs. 0.37 ± 0.10, P = 0.22) (Table 2).
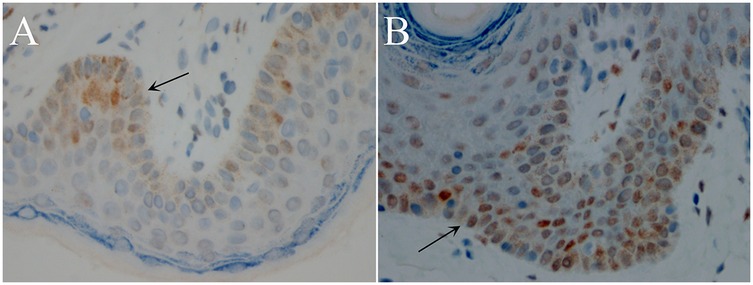
Figure 1. Androgen receptor (AR) immunostaining in inner plate preputial tissues (400×, transverse sections). Strongest nuclear staining in basal epithelial layer (arrows). (A) Hypospadias group: Sparse immunopositive cells with weak staining intensity. (B) Control group: Numerous strongly stained immunopositive cells. DAB chromogen, hematoxylin counterstain.
Discussion
Our study demonstrated significantly reduced AR expression in hypospadias patients compared to healthy controls, with no differential expression between proximal and distal subtypes.
Although extensive studies have characterized the histology of hypospadias, its etiology remains elusive. Multiple genes—including ZEB1, Hsa_circ_0000417, SRY, 5α-reductase, and AR—have been implicated as contributors, though none are established as causative (2, 3, 9).
The second trimester is a critical period for human fetal urethral development. During this period, androgens are critical for the differentiation and development of sexual genitalia in male fetuses (11). DHT-AR signaling is essential for urethral closure, with AR functioning as a ligand-dependent nuclear receptor transcription factor (12). AR deficiency disrupts androgen signaling, impairing male sex differentiation and potentially contributing to hypospadias pathogenesis.
The predominant localization of AR to the basal epithelial layer is biologically significant, as this compartment contains progenitor cells critical for urethral morphogenesis. Basal AR signaling likely orchestrates epithelial-mesenchymal interactions (EMT) during urethral closure by regulating downstream targets (e.g., ZEB1, FGF10). Reduced AR in basal cells may disrupt EMT, impairing urethral tubularization, resulting in failed urethral fusion and contributing to hypospadias pathogenesis (9, 10).
Numerous studies have investigated the AR-hypospadias relationship, yet yielded conflicting results. Vottero et al. reported reduced AR expression in preputial tissue from hypospadias patients vs. healthy controls (2). Conversely, Pichler et al. documented elevated AR protein in affected boys (133.25 ± 6.17 vs. 100 ± 4.45, P = 0.014) (7). These discrepancies likely reflect methodological variations and limited sample sizes in prior studies. To resolve this controversy, we employed a large cohort to definitively establish the AR-hypospadias association.
Our study demonstrated that AR expression is significantly decreased in patients with hypospadias than in healthy boys. This may be associated with the etiology of hypospadias. Lower AR expression causes abnormal combination and dysfunction of androgens; thus, urethral development is affected, and urethral closure cannot be achieved, contributing to various hypospadias. Vottero et al. documented reduced AR in preputial tissue from affected patients vs. controls (2), a result corroborated by Chao et al. using immunohistochemistry (3). Silva et al. further reported diminished AR mRNA in urethral mucosa (4), while An et al. noted AR reduction in penile skin, particularly within the urethral plate (10). Consistent with our protein-level findings, Yıldız et al. demonstrated downregulated AR expression and associated DNA methylation changes in foreskin tissue of hypospadias patients (6). Similarly, İnanç et al. reported dysregulation of AR and related genes (ESR1, FGFR2) in preputial tissues, reinforcing end-organ resistance in hypospadias pathogenesis (5). This convergence of protein and gene evidence reinforces AR deficiency as a hallmark of hypospadias. Conversely, several groups report contradictory data. Kocaturk H. described increased AR expression in affected preputial tissue (8). Balaji et al. similarly noted heightened AR levels, with proximal subtypes exhibiting greater expression than distal forms, potentially indicating end-organ overexpression (13).
We observed no difference in AR expression between the proximal and distal hypospadias. Although AR mediates external genitalia development, its levels did not correlate with phenotypic severity—a finding consistent with Celayir et al. who reported no AR-severity association (14). In contrast, Qiao et al. described elevated AR in severe vs. mild hypospadias (P < 0.05), though small sample sizes preclude definitive conclusions (15). Our null finding regarding AR expression across proximal/distal subtypes suggests that baseline AR deficiency may be a universal feature in hypospadias irrespective of severity. We speculate that compensatory mechanisms (e.g., local androgen synthesis) or alternative signaling pathways may modulate phenotypic expression beyond AR abundance alone.
Impaired AR transcriptional activity in hypospadias may be ameliorated by androgen supplementation. Kaya et al. demonstrated that preoperative DHT gel reduces postoperative complications (e.g., fistula rates) and improves cosmetic outcomes, potentially via enhanced angiogenesis and signaling rescue (16). Our data support DHT trials in AR-deficient hypospadias, reinforcing this therapeutic approach.
Our findings contrast with prior reports of AR overexpression in hypospadias (7, 8, 13). Specifically, we observed reduced AR expression in patients vs. healthy controls. These discrepancies likely reflect methodological differences. Pichler et al. quantified AR in whole-tissue lysates via Western blotting, whereas our IHC precisely localized AR to basal epithelial cells—the primary site for androgen signaling during urethrogenesis (7).
While our study identifies AR downregulation, future work should explore upstream mechanisms (e.g., AR promoter methylation, miRNA regulation) and functional consequences via in vitro models of urethral development.
In conclusion, the inner plate preputial tissues of patients with hypospadias showed decreased AR expression, suggesting that AR may be involved in hypospadias development and contributes to resolving a key controversy. However, further studies are necessary to assess whether epigenetic abnormalities or gene mutations are responsible for the decreased AR expression.
Data availability statement
The original contributions presented in the study are included in the article/Supplementary Material, further inquiries can be directed to the corresponding author.
Ethics statement
The studies involving humans were approved by Medical Ethics Committee of Shenzhen Children's Hospital. The studies were conducted in accordance with the local legislation and institutional requirements. Written informed consent for participation in this study was provided by the participants’ legal guardians/next of kin. Written informed consent was obtained from the minor(s)' legal guardian/next of kin for the publication of any potentially identifiable images or data included in this article.
Author contributions
ZY: Visualization, Formal analysis, Project administration, Data curation, Funding acquisition, Resources, Validation, Writing – review & editing, Software, Methodology, Supervision, Writing – original draft, Investigation, Conceptualization. PZ: Data curation, Methodology, Project administration, Conceptualization, Software, Formal analysis, Writing – original draft. TZ: Methodology, Data curation, Writing – original draft, Conceptualization, Funding acquisition, Resources. JY: Investigation, Writing – review & editing, Data curation, Methodology, Visualization, Resources, Validation. GZ: Software, Data curation, Formal analysis, Writing – original draft, Conceptualization. WZ: Project administration, Investigation, Writing – original draft, Funding acquisition, Data curation, Formal analysis, Resources. PC: Formal analysis, Data curation, Resources, Software, Writing – original draft. JL: Investigation, Methodology, Conceptualization, Writing – review & editing, Visualization. SL: Investigation, Resources, Conceptualization, Funding acquisition, Writing – review & editing, Data curation, Supervision, Writing – original draft, Formal analysis, Methodology, Visualization.
Funding
The author(s) declare that financial support was received for the research and/or publication of this article. This work was supported by the Guangdong High-level Hospital Construction Fund and the Shenzhen Fund for Guangdong Provincial High-level Clinical Key Specialties (SZXK035).
Acknowledgments
We would like to acknowledge Pan Zhao and Wanhua Xu for refining this manuscript.
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The author(s) declare that no Generative AI was used in the creation of this manuscript.
Any alternative text (alt text) provided alongside figures in this article has been generated by Frontiers with the support of artificial intelligence and reasonable efforts have been made to ensure accuracy, including review by the authors wherever possible. If you identify any issues, please contact us.
Publisher's note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
References
1. Bergman JE, Loane M, Vrijheid M, Pierini A, Nijman RJ, Addor MC, et al. Epidemiology of hypospadias in Europe: a registry-based study. World J Urol. (2015) 33(12):2159–67. doi: 10.1007/s00345-015-1507-6
2. Vottero A, Minari R, Viani I, Tassi F, Bonatti F, Neri TM, et al. Evidence for epigenetic abnormalities of the androgen receptor gene in foreskin from children with hypospadias. J Clin Endocrinol Metab. (2011) 96(12):E1953–62. doi: 10.1210/jc.2011-0511
3. Chao C, Jian YY, Zhao XY, Liu YL, Xie QK. The involvement of hsa_circ_0000417 in the development of hypospadias by regulating AR. Differentiation. (2020) 116:9–15. doi: 10.1016/j.diff.2020.09.003
4. Silva TS, Richeti F, Cunha DP, Amarante AC, de Souza Leão JQ, Longui CA. Androgen receptor mRNA measured by quantitative real time PCR is decreased in the urethral mucosa of patients with middle idiopathic hypospadias. Horm Metab Res. (2013) 45(7):495–500. doi: 10.1055/s-0033-1333717
5. Inanc I, Avlan D, Eker D, Gurkan H. A genetics study in the foreskin of boys with hypospadias. Mol Syndromol. (2023) 14(3):185–90. doi: 10.1159/000527405
6. Yıldız S, Inanç I, Zhuri D, Atlı E, Avlan D. The possible role of epigenetics in the etiology of hypospadias. J Pediatr Urol. (2024) 20(5):877–83. doi: 10.1016/j.jpurol.2024.07.001
7. Pichler R, Djedovic G, Klocker H, Heidegger I, Strasak A, Loidl W, et al. Quantitative measurement of the androgen receptor in prepuces of boys with and without hypospadias. BJU Int. (2013) 112(2):265–70. doi: 10.1111/j.1464-410X.2012.11731.x
8. Kocaturk H, Özkaya F, Adanur S, Yapanoglu T, Aksoy Y. Expression of androgen receptor and estrogen receptor-beta in foreskin tissue of hypospadiac and healthy boys. Arch Esp Urol. (2020) 73(2):119–25.32124842
9. Qiao L, Tasian GE, Zhang H, Cunha GR, Baskin L. ZEB1 Is estrogen responsive in vitro in human foreskin cells and is over expressed in penile skin in patients with severe hypospadias. J Urol. (2011) 185(5):1888–93. doi: 10.1016/j.juro.2010.12.066
10. An Y, Liu X, Tang Y, Liu J, Li J, Luo Y, et al. [Expression of androgen receptor in genital tissue of patients with congenital hypospadias and simple chordee]. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. (2012) 26(4):461–5.22568330
11. Bouty A, Ayers KL, Pask A, Heloury Y, Sinclair AH. The genetic and environmental factors underlying hypospadias. Sex Dev. (2015) 9(5):239–59. doi: 10.1159/000441988
12. Lu NZ, Wardell SE, Burnstein KL, Defranco D, Fuller PJ, Giguere V, et al. International union of pharmacology. LXV. The pharmacology and classification of the nuclear receptor superfamily: glucocorticoid, mineralocorticoid, progesterone, and androgen receptors. Pharmacol Rev. (2006) 58(4):782–97. doi: 10.1124/pr.58.4.9
13. Balaji DR, Reddy G, Babu R, Paramaswamy B, Ramasundaram M, Agarwal P, et al. Androgen receptor expression in hypospadias. J Indian Assoc Pediatr Surg. (2020) 25(1):6–9. doi: 10.4103/jiaps.JIAPS_166_18
14. Celayir A, Moralioglu S, Cetiner H, Kir G, Celayir S. Expression of androgen, estrogen, and progesterone hormone receptors in the penile tissues of children with different types of hypospadias. North Clin Istanb. (2019) 6(2):110–6. doi: 10.14744/nci.2018.47108
15. Qiao L, Tasian GE, Zhang H, Cao M, Ferretti M, Cunha GR, et al. Androgen receptor is overexpressed in boys with severe hypospadias, and ZEB1 regulates androgen receptor expression in human foreskin cells. Pediatr Res. (2012) 71(4 Pt 1):393–8. doi: 10.1038/pr.2011.49
Keywords: hypospadias, androgen receptor, prepuce, children, expression
Citation: Yang Z, Zhang P, Zhang T, Yin J, Zhou G, Zhao W, Chen P, Li J and Li S (2025) Downregulated androgen receptor expression in preputial tissues of children with hypospadias. Front. Pediatr. 13:1660777. doi: 10.3389/fped.2025.1660777
Received: 6 July 2025; Accepted: 8 August 2025;
Published: 29 August 2025.
Edited by:
Christian Schöfer, Medical University of Vienna, AustriaReviewed by:
Irem Inanc, Trakya University Faculty of Medicine, TürkiyeSadettin Yildiz, Trakya University, Türkiye
Copyright: © 2025 Yang, Zhang, Zhang, Yin, Zhou, Zhao, Chen, Li and Li. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Shoulin Li, bGlzaG91bGluODMwMDgwODhAMTYzLmNvbQ==
 Zhilin Yang
Zhilin Yang Peiliang Zhang2
Peiliang Zhang2 Weiguang Zhao
Weiguang Zhao Pengyu Chen
Pengyu Chen Jiaqiang Li
Jiaqiang Li