- 1Discipline of Clinical Pharmacy, School of Pharmaceutical Sciences, Universiti Sains Malaysia, Georgetown, Malaysia
- 2Pharmacy Department, Hospital Pulau Pinang, Georgetown, Malaysia
- 3Clinical Research Center, Hospital Pulau Pinang, Georgetown, Malaysia
Adverse drug reaction (ADR) primarily caused by many drugs including antibiotics. At present, the incidence and pattern of ADR caused by antibiotics have remained as neglected area in Malaysia. This study was conducted to determine the incidence and analyze the pattern of ADR caused by antibiotics among patients in a tertiary care hospital. It is a 2-year retrospective observational study conducted at Hospital Pulau Pinang, Malaysia. All eligible patients who had antibiotic prescribed belonging to any age group either from outpatient or inpatient that had experienced ADR was included in this study. The outcomes were measured with the aid of Naranjo’s and Hartwig’s scales. The incidence of the ADRs among patients prescribed with antibiotics in Hospital Pulau Pinang is about 1.1%. Vancomycin and Trimethoprim/Sulfamethoxazole both are considered to be the major contributors to ADR incidences. The skin was the most affected organ by ADRs followed by gastrointestinal system. Most of the severe ADRs were caused by Penicillin. The causality relationship of all the severe reactions was mostly probable. General Medicine unit had reported the highest number of ADRs caused by antibiotics. The common manifestations of ADRs are acute kidney injury and exanthem. In addition, majority of the ADRs caused by antibiotics were reversible. A large multicenter study is suggested to confirm the present findings.
Introduction
One of the major threats in the healthcare system is the adverse drug reaction (ADR), which profusely affects the health, transience, and the quality of life especially among the hospitalized patients (Lazarou et al., 1998; Pirmohamed et al., 1998, 2004; Davies et al., 2009). In many countries, ADRs rank among the top 10 leading causes of mortality. Therefore, there is a need to study ADRs seriously to create awareness among patients and to motivate health care professionals in the hospital to report ADRs as in to minimize the risk (Hurwitz, 1969; Hepler, 2003; Morris and Cantrill, 2003; Schnipper et al., 2006; Knudsen et al., 2007).
A study conducted in Malaysia among the pediatric population revealed that anti-infective for systemic use (61.9%) was the most common therapeutic group reported for ADRs in children below 2-years-old (Rosli et al., 2016). Another 8 years retrospective study conducted in Malaysia revealed that anti-infective agents were among the drugs caused various ADRs (35%) (Ibrahim et al., 2013). Pharmacovigilance activities are regulated by the National Center for Adverse Drug Monitoring, a division within the National Pharmaceutical Regulatory Agency (NPRA), which was established under Drug Control Authority (DCA) in Malaysia. Malaysian Adverse Drug Reactions Advisory Committee (MADRAC) provides DCA with important information pertaining to local and international drug safety issues and plays a role as advisor to DCA on risk management and risk communication following effective assessment of the benefit-risk profile of drugs (Malaysian Guidelines for the Reporting and Monitoring of Adverse drug Reactions, 2017). MADRAC revealed that 50% of the top 10 drugs most commonly reported for cutaneous ADR in the year 2008 belong to the antibiotic group (Ding et al., 2010).
Although there are various studies underlying the ADR effects among patients prescribed with antibiotics, but the data on the ADR incidence and pattern have remained as neglected area. Thus, this study was conducted with the aim to determine the incidence and analyze the pattern of the ADRs among patients prescribed with antibiotics in Hospital Pulau Pinang, Malaysia. This study will help to gain insight on the number of patients that are affected with ADR as a consequence from antibiotic use and provide the much needed data to prevent reoccurrence of the ADR associated with those specific antibiotics especially in different patients and further reduces the impact on the patients’ quality of life.
Materials and Methods
Study Settings and Design
This was a 2-year retrospective observational study using patients’ medical records conducted at Hospital Pulau Pinang, Malaysia. A total of 648 patients’ medical records from January 2015 till December 2016 were retrieved via the MADRAC registry. All eligible patients’ records belonging to any age group prescribed with an antibiotic by the prescriber either from outpatient or inpatient whom had experienced ADR were included in this study. Validated data collection form was used in obtaining patients’ clinical and the socio-demographics data. The total population of patients whom visited Hospital Pulau Pinang was 15,712 for the duration of 2 years. Written approval was obtained from the Medical Research and Ethics Committee (NMRR-16-1740-32642), Ministry of Health Malaysia prior to the embankment of the study. No personal identifiers of the patients were collected during the study duration. The data were accessed anonymously.
Verification of Scoring System
Naranjo’s scale was used for the causality assessment in this study (Naranjo et al., 1981). The first scoring of the scale was given by the reporter whom could be any of the healthcare professionals. Subsequently, the pharmacist whom handled the ADR submissions to the MADRAC re-scored the scale for further confirmation and verified the details being reported. Additionally, Hartwig’s scale was used for the severity assessment scale in this study (Hartwig et al., 1992). The first scoring of the scale was given by the reporter whom could be any of the healthcare professionals. However, the researcher verified the scores given by the reporter with the information written in the bed head ticket (BHT). Any queries or doubts arises during the process were clarified immediately with the reporter.
Statistical Analysis
The data were entered and analyzed using the Statistical Package for Social Sciences (SPSS) base version 22.0; SPSS, Inc., Chicago, IL, United States. Descriptive data analysis was used for the analysis of this study. The normally distributed continuous variables are summarized in mean and standard deviation while the non-normalized are expressed as median and inter quartile range. For categorical variables, frequencies and percentage were tabulated. Two-sided p-values of less than 0.05 were considered statistically significant.
Results
During the study period, a total of 648 patients’ records that had documented the event of ADR were screened at the study site. Out of it, only 175 reactions were reported caused by antibiotics. And all 175 reports met the eligibility criteria and subjected for further analysis. None was excluded. Baseline clinical and socio-demographic characteristics of the patients enrolled in the study are depicted in Table 1.
Table 2 describes the various analyses of the ADRs caused by antibiotics. Skin manifestations deemed to be the highest organ to be affected by ADRs followed by gastrointestinal system. Penicillins are proven to be the main culprit in causing the most of the adverse reactions followed by Cephalosporins. General Medicine unit had reported the highest number of ADRs. This study revealed that all the ADRs were moderate reactions in terms of severity and believed to have probable causality the most. Precisely, majority of the ADRs caused by antibiotics were reversible.
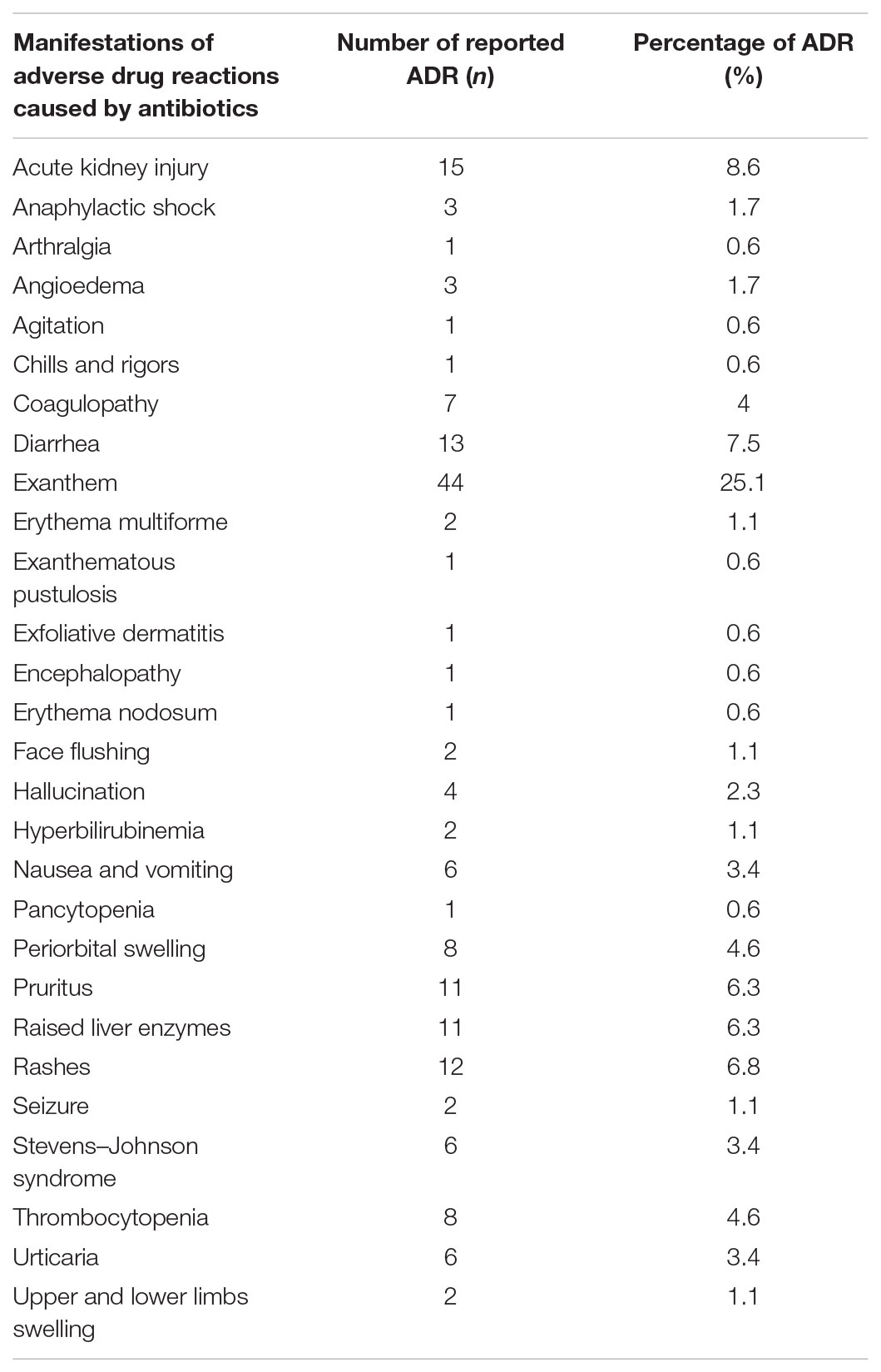
The manifestations of the ADRs were further illustrated in Table 3. Exanthem has accounted as the common manifestation in quite a number of patients. And the subsequent common manifestation seen in this study was acute kidney injury.
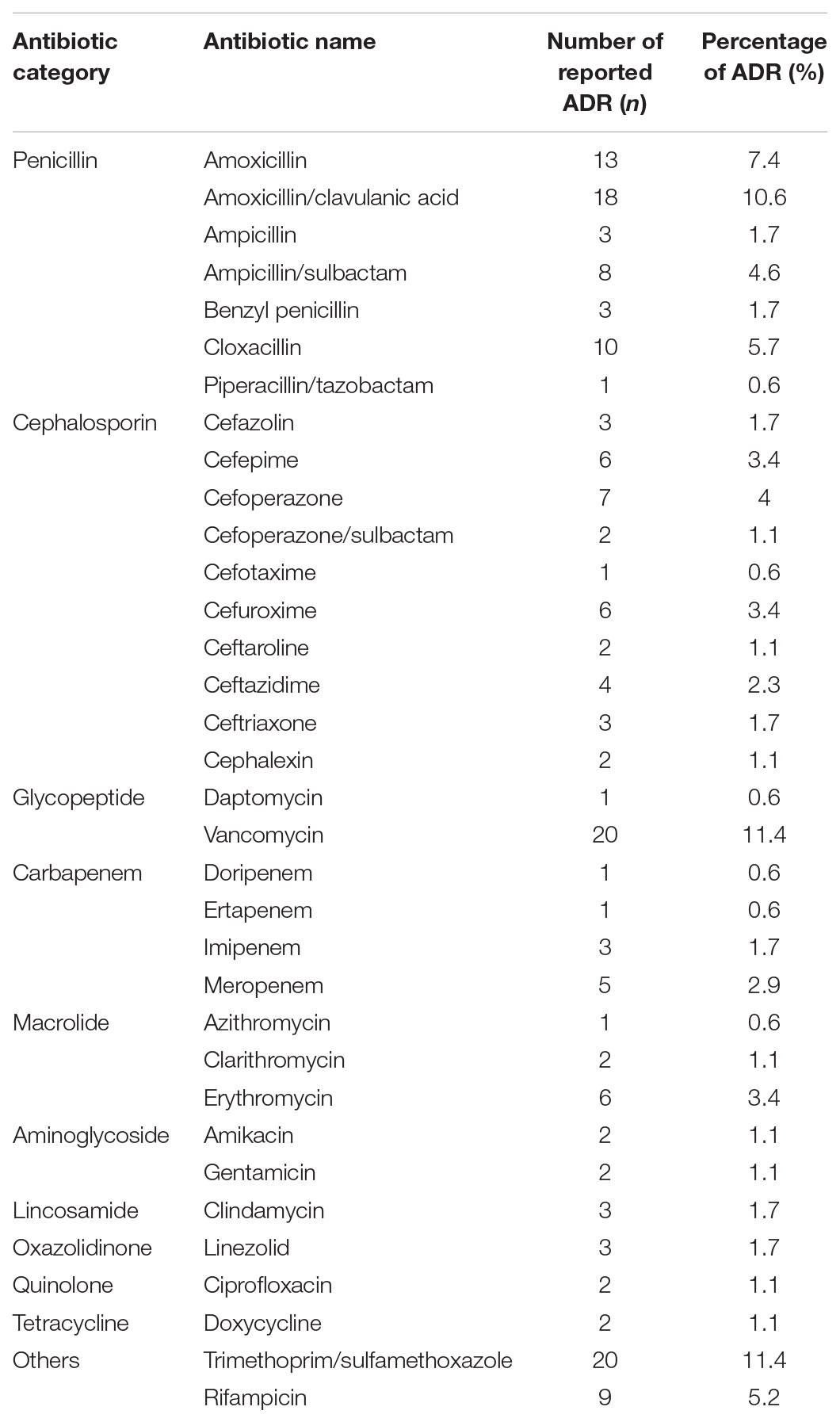
Further analysis of the antibiotics causing ADRs based on the various pharmacological categories is depicted in Table 4. Vancomycin and Trimethoprim/Sulfamethoxazole are known to cause the highest number of ADRs equally. The manifestations of ADRs are acute kidney injury and exanthem respectively.
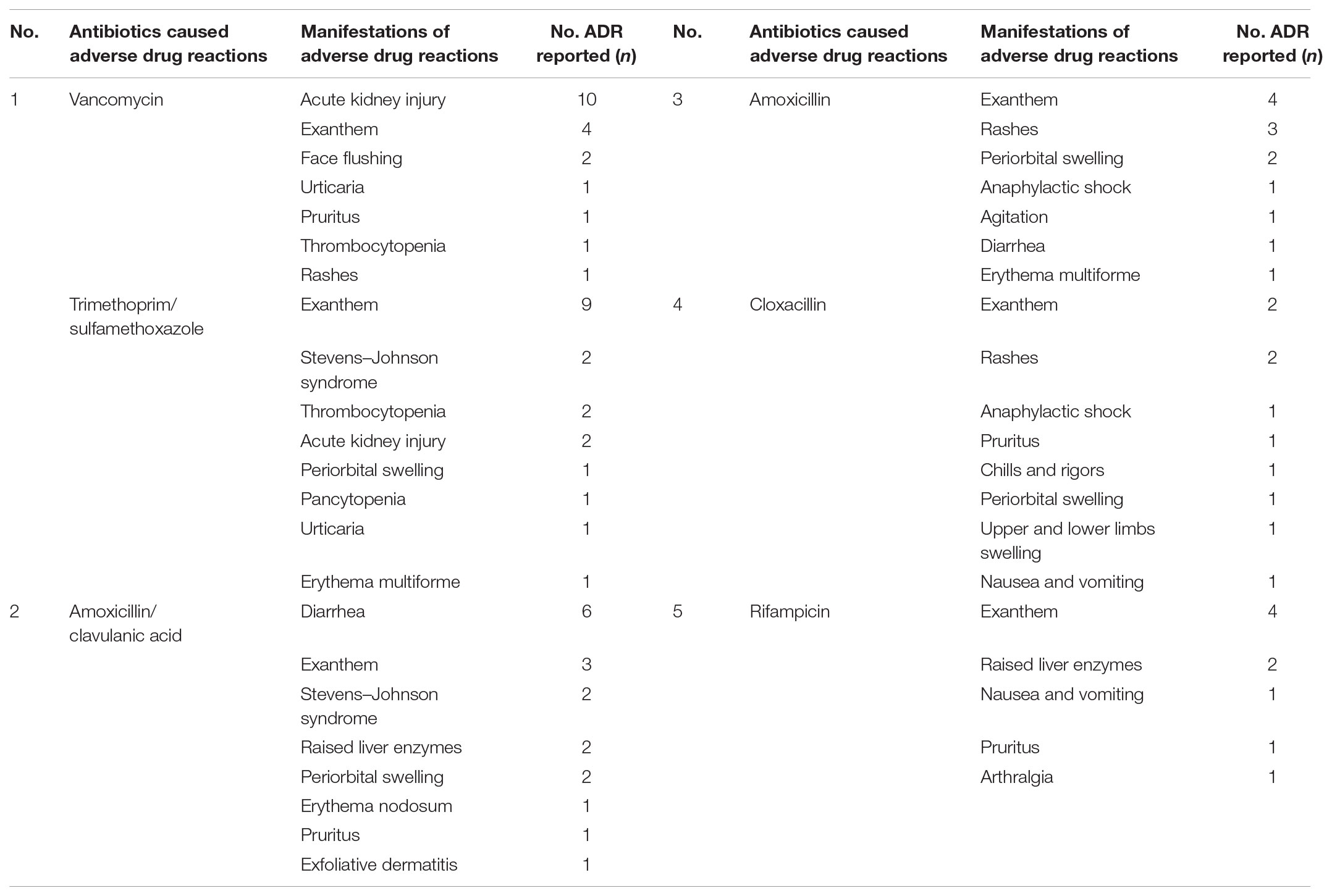
Table 5 portrayed the top six antibiotics causing the ADRs and their manifestations respectively.
Discussion
To the best of our knowledge, this is the first study which analyzed the pattethe pattern of ADRs caused by antibiotics in Malaysia. This study provides the much needed data about the incidence rate of antibiotics causing ADRs especially in a tertiary care hospital. The incidence rate of antibiotics causing ADRs in this study was found to be comparatively low when compared to other studies (Ding et al., 2010; Priyadharsini et al., 2011; Shamna et al., 2014; Dhar et al., 2015). It accounted about 1.1% (n = 175) out of the total population which had been reported past 2 years. The incidence rate was lower probably due to the effective intervention of respective clinical pharmacists at the study site. Also, we were informed that the health care professionals in this hospital receive constant updates from the Medication Safety team formed merely within Hospital Pulau Pinang, thus, keeping them updated of the latest events related to drug safety via their routine publication of bulletins.
In this study, the predominance of male sex for ADRs which is similar to those studies conducted elsewhere (Jose et al., 2008; Dhar et al., 2015). According to the data obtained from the record office, majority of the admitted patients was male throughout the study period. Analysis of the age wise distribution showed the predominance of adult patients followed by pediatric patients and lastly geriatrics. Probably adult patients were more prone to antibiotic ADRs due to age related pharmacokinetics and pharmacodynamics changes and the presence of co-morbid illnesses and multiple drugs along with infectious diseases.
On the other hand, the most affected organ system by ADRs due to antibiotics is skin which is parallel with another study (Ibrahim et al., 2013) followed by gastrointestinal, hematologic, renal, hepatic, neurologic, ophthalmic, musculoskeletal, and others. Contrary to our study, other studies conducted elsewhere (Shamna et al., 2014; Dhar et al., 2015) claimed that the most affected organ system was the gastrointestinal system. Out of the many types of skin reactions, exanthem was the most commonly accounted type of adverse reaction in this study. Another two studies (Ding et al., 2010; Priyadharsini et al., 2011) concluded that the most common manifestation observed were maculopapular eruption and urticaria, respectively.
Another noteworthy finding of the current study was the distribution of antibiotic classes that caused ADRs. Penicillins was the most accounted antibiotic category causing ADRs which is similar to other studies conducted elsewhere (Priyadharsini et al., 2011; Rosli et al., 2016), followed by Cephalosporins, others precisely Trimethoprim/Sulfamethoxazole and Rifampicin, Glycopeptides, Carbapenems, Macrolides, Aminoglycosides, both Lincosamides and Oxazolidinones and lastly Quinolones and Tetracyclines. Penicillins was the most accounted antibiotics category causing ADRs compared to the rest of the pharmacological categories is because the usage of Penicillins was the highest throughout the study period compared to Cephalosporins. This study also revealed Vancomycin and Trimethoprim/Sulfamethoxazole are both antibiotics with highest number of incidences of ADRs. Vancomycin as it is known to be a nephrotoxic drug, deemed to cause most of the ADRs as in acute kidney injury and the usage was high consistent with the high occurrence of Methicillin-resistant Staphylococcus aureus (MRSA) infection. Trimethoprim/Sulfamethoxazole usage was also high due to high prevalence of Stenotrophomonas maltophilia isolates which resulted from heavy usage of Carbapenems. Trimethoprim/Sulfamethoxazole were reported to have most of skin reactions, especially exanthem. We suppose this exanthem reaction was due to presence of sulfonamides in Trimethoprim/Sulfamethoxazole.
The highest quantity of ADR reports were received from General Medicine followed by Nephrology, Pediatrics, Respiratory, Skin, Surgical, both Cardiology and Cardiothoracic, Hematology, Gastroenterology, Obstetrics and Gynecology and Oncology, and last of all were from Orthopedics. In this study, most of the reported ADRs were from General Medicine is because the department used ranges of antibiotics tremendously right from the low end to the high end throughout the study period for both prophylaxis and treatment purposes. Furthermore, the Infectious Unit is grouped under General Medicine Department in this hospital.
Of all the reported ADRs, moderate reactions accounted the most and followed by mild and severe reactions as per the Hartwig’s scale. Other studies (Jose et al., 2008; Stavreva et al., 2008; Priyadharsini et al., 2011; Shamna et al., 2014; Dhar et al., 2015) also disclosed comparable findings as ours. This study revealed that none of the severe ADRs were fatal provided all the cases were given intensive medical care accordingly. Most of the severe ADRs were caused by Penicillins and the most common manifestation of severe ADR is Stevens–Johnson syndrome (SJS). The one death reported in severe category was purely due to severe head injury post-trauma. The adults were accounted the most with severe ADRs compared to the rest of the age groups. Male gender was identified to be more prominent than female in this sub-analysis.
Moreover, the causality assessment of reported ADRs as per the Naranjo’s scale and revealed that mostly were probable, then possible and limited reactions were definite. There were no ADR reports grouped as unlikely. These findings are analogous as Stavreva et al. (2008); Priyadharsini et al. (2011), Shamna et al. (2014), and Dhar et al. (2015). The cases were categorized as definite after “rechallenging” with the respective drugs. All the definite cases were given medical care accordingly and none of them is fatal. Most of the definite ADRs were caused by Rifampicin and the most common manifestation of definite ADR is exanthem. Perhaps, the adults were accounted the most with definite ADRs compared to the rest of the age groups. Female gender was identified to be more prominent than male in this sub-analysis.
In this study, it is noted that majority of the total reported ADRs were reversible. Limited numbers of reactions were irreversible. All the irreversible reactions were reported from General Medicine. The reason behind the irreversibility is because all the patients involved died even before the ADRs were treated. Indeed, those patients died because of severe head injury post-MVA. The utmost common manifestation of the irreversibility reaction is acute kidney injury and caused by vancomycin and gentamicin. Another antibiotic namely, Trimethoprim/Sulfamethoxazole had caused irreversible thrombocytopenia. None of the reported irreversible ADRs is due to inappropriate doses or frequencies. All the irreversible ADRs were experienced by adults and mostly were male patients.
Conclusion
The incidence rate of antibiotics causing ADRs in this study was found to be as low as 1.1%. Thus, educational interventions on the main causes of ADRs, patient monitoring, and appropriate prescribing policy should be deployed to both prevent ADRs and minimize their impact, where they do occur. Dissemination of information related to ADRs to both health care professionals and patients has the potential to improve awareness and subsequently heightens the patients’ quality of life.
Strength and Limitations
The strength of the study is that the information collected was directly from the experienced health care professionals itself. This group of professionals receive constant updates from the Medication Safety team formed merely within Hospital Pulau Pinang, thus, keeping them updated of the latest events or news related to drug safety via their routine publication of bulletins. Being a study from a single center in a high burden country with ADRs caused by antibiotics, the findings of the present study should be interpreted with the major limitation of small number of patients enrolled. However, Hospital Pulau Pinang is a center for referral especially for the northern region population and also being a second largest hospital in Malaysia. Thus, the usage of antibiotics is tremendously high and involves huge variation of antibiotics. Nevertheless, a multicenter study with a large sample size is needed to confirm the findings of the current study. On the other hand, there is possibility of under reporting especially by the newly joined health care professionals such as house physicians and provisionally registered pharmacists.
Author Contributions
AA and MD conception and design, analysis interpretation of the data, and statistical expertise. AA collection and assembly of data and drafting of the article. SS critical revision of the article for important intellectual content and final approval of the article.
Conflict of Interest Statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
The authors would like to thank the Director General of Health Malaysia for his permission to publish this article.
References
Davies, E. C., Green, C. F., Taylor, S., Williamson, P. R., Mottram, D. R., and Pirmohamed, M. (2009). Adverse drug reactions in hospital in-patients: a prospective analysis of 3695 patient-episodes. PLoS One 4:e4439. doi: 10.1371/journal.pone.0004439
Dhar, K., Sinha, A., Gaur, P., Goel, R., Chopra, V., and Bajaj, U. (2015). Pattern of adverse drug reactions to antibiotics commonly prescribed in department of medicine and pediatrics in a tertiary care teaching hospital, Ghaziabad. J. Appl. Pharm. Sci. 5, 78–82. doi: 10.7324/JAPS.2015.50413
Ding, W. Y., Lee, C. K., and Choon, S. E. (2010). Cutaneous adverse drug reactions seen in a tertiary hospital in Johor, Malaysia. Int. J. Dermatol. 49, 834–841. doi: 10.1111/j.1365-4632.2010.04481.x
Hartwig, S. C., Siegel, J., and Schneider, P. J. (1992). Preventability and severity assessment in reporting adverse drug reactions. Am. J. Hosp. Pharm. 49, 2229–2232.
Hepler, C. (2003). Understanding Adverse Drug Therapy Outcomes. Preventing Medication Errors and Improving Drug Therapy Outcomes: A Management Systems Approach. Boca Raton, FL: CRC Press LLC. doi: 10.1201/9780203010730
Hurwitz, N. (1969). Admissions to hospital due to drugs. Br. Med. J. 1, 539–540. doi: 10.1136/bmj.1.5643.539
Ibrahim, N. S., Ramlee, N., Lah, N. A. A., and Rosemi, N. A. (2013). Adverse drug reaction reported in hospital universiti sains Malaysia (HUSM). Age 14, 15–24.
Jose, J., Rao, P. G., and Jimmy, B. (2008). Adverse drug reactions to fluoroquinolone antibiotics–Analysis of reports received in a tertiary care hospital. Int. J. Risk Saf. Med. 20, 169–180.
Knudsen, P., Herborg, H., Mortensen, A., Knudsen, M., and Hellebek, A. (2007). Preventing medication errors in community pharmacy: frequency and seriousness of medication errors. Qual. Saf. Health Care 16, 291–296. doi: 10.1136/qshc.2006.018770
Lazarou, J., Pomeranz, B. H., and Corey, P. N. (1998). Incidence of adverse drug reactions in hospitalized patients: a meta-analysis of prospective studies. J. Am. Med. Assoc. 279, 1200–1205. doi: 10.1001/jama.279.15.1200
Malaysian Guidelines for the Reporting and Monitoring of Adverse drug Reactions (2017). Available at: http://npra.moh.gov.my/index.php/about-npcb/malaysian-adverse-drug-reactions-advisory-committee-madrac/introduction
Morris, C., and Cantrill, J. (2003). Preventing drug-related morbidity–the development of quality indicators. J. Clin. Pharm. Ther. 28, 295–305. doi: 10.1046/j.1365-2710.2003.00496.x
Naranjo, C., Busto, U., Sellers, E., Sandor, P., Ruiz, I., and Roberts, E. (1981). A method for estimating the probability of adverse drug reactions. Clin. Pharmacol. Ther. 30, 239–245. doi: 10.1038/clpt.1981.154
Pirmohamed, M., Breckenridge, A. M., Kitteringham, N. R., and Park, B. K. (1998). Adverse drug reactions. Br. Med. J. 316, 1295–1298. doi: 10.1136/bmj.316.7140.1295
Pirmohamed, M., James, S., Meakin, S., Green, C., Scott, A. K., Walley, T. J., et al. (2004). Adverse drug reactions as cause of admission to hospital: prospective analysis of 18 820 patients. Br. Med. J. 329, 15–19. doi: 10.1136/bmj.329.7456.15
Priyadharsini, R., Surendiran, A., Adithan, C., Sreenivasan, S., and Sahoo, F. (2011). A study of adverse drug reactions in pediatric patients. J. Pharmacol. Pharmacother. 2, 277–280. doi: 10.4103/0976-500X.85957
Rosli, R., Ming, L., Abd Aziz, N., and Manan, M. (2016). A retrospective analysis of spontaneous adverse drug reactions reports relating to paediatric patients. PLoS One 11:e0155385. doi: 10.1371/journal.pone.0155385
Schnipper, J. L., Kirwin, J. L., Cotugno, M. C., Wahlstrom, S. A., Brown, B. A., Tarvin, E., et al. (2006). Role of pharmacist counseling in preventing adverse drug events after hospitalization. Arch. Intern. Med. 166, 565–571. doi: 10.1001/archinte.166.5.565
Shamna, M., Dilip, C., Ajmal, M., Mohan, P. L., Shinu, C., Jafer, C., et al. (2014). A prospective study on adverse drug reactions of antibiotics in a tertiary care hospital. Saudi Pharm. J. 22, 303–308. doi: 10.1016/j.jsps.2013.06.004
Keywords: adverse drug reactions, vancomycin, trimethoprim/sulfamethoxazole, penicillin, skin
Citation: Arulappen AL, Danial M and Sulaiman SAS (2018) Evaluation of Reported Adverse Drug Reactions in Antibiotic Usage: A Retrospective Study From a Tertiary Care Hospital, Malaysia. Front. Pharmacol. 9:809. doi: 10.3389/fphar.2018.00809
Received: 03 April 2018; Accepted: 05 July 2018;
Published: 20 August 2018.
Edited by:
Brian Godman, Karolinska Institutet (KI), SwedenReviewed by:
Gaurav Deshpande, HealthCore, Inc., United StatesMaria Margarita Salazar-Bookaman, Central University of Venezuela, Venezuela
Joseph O. Fadare, Ekiti State University, Nigeria
Copyright © 2018 Arulappen, Danial and Sulaiman. This is an open-access article distributed under the terms of the Creative Commons Attribution License (CC BY). The use, distribution or reproduction in other forums is permitted, provided the original author(s) and the copyright owner(s) are credited and that the original publication in this journal is cited, in accordance with accepted academic practice. No use, distribution or reproduction is permitted which does not comply with these terms.
*Correspondence: Ann L. Arulappen, YW5ubGlzYWFydWxAeWFob28uY29t
 Ann L. Arulappen1,2*
Ann L. Arulappen1,2* Monica Danial
Monica Danial